Abstract
People with HIV (PWH) have a high burden of medical comorbidities, potentially putting them at increased risk for severe COVID-19. Additionally, during the COVID-19 pandemic, HIV care delivery has been restructured and the impact on HIV outcomes is unknown. The objectives of this study were first, to examine the risk of severe COVID-19 among PWH, using a definition incorporating clinical risk factors, and second, to examine the pandemic’s impact on HIV care. We used data from the DC Cohort, a large cohort of people receiving HIV care in Washington, DC. We found that a high proportion of participants across all age groups qualified as increased (58%) or high risk (34%) for severe COVID-19. Between 2019 and 2020, encounters increased (17.7%, increasing to 23.5% of active DC Cohort participants had an encounter) while laboratory utilization decreased (14.4%, decreasing to 11.4% of active DC Cohort participants had an HIV RNA test performed). Implications of our work include the importance of protecting vulnerable people with HIV from acquiring COVID-19 and potentially manifesting severe complications through strategies including vaccination. Additionally, acknowledging that HIV service delivery will likely be changed long-term by the pandemic, adaptation is required to ensure continued progress towards 90-90-90 goals.
Supplementary Information
The online version contains supplementary material available at 10.1007/s10461-022-03662-0.
Keywords: HIV, COVID-19, Comorbidities, Utilization, HIV RNA
Introduction
COVID-19 has had a severe global and national impact with over 464 million infections and 6 million deaths as of March 17, 2022 [1]. In the United States, the pandemic has highlighted the fragility of our public health and medical infrastructures and has unveiled many longstanding health disparities as reflected by the excessively high rates of COVID-19 among persons of color and those of lower socioeconomic status [2, 3]. COVID-19 also disproportionately impacts those who are over 65 years old and have underlying medical conditions. Our understanding of the impact of COVID-19 on people with HIV (PWH) continues to evolve [4]. Studies examining the impact of HIV on COVID-19 outcomes have been mixed, with some showing similar outcomes for people with and without HIV [5–8] and others showing worse outcomes for PWH [9, 10] or more specifically for PWH with low CD4 cell counts [11].
It remains unclear whether HIV itself or the comorbidities in PWH contribute more to risk of severe outcomes for COVID-19 [12]. Certainly, in the general population, presence of certain medical comorbidities portends worse outcomes from COVID-19. These medical comorbidities include cardiovascular disease, diabetes mellitus, hypertension, and chronic lung disease, among others [13–15]. Racial and income level disparities in the distribution of comorbidities that are most likely to be associated with severe COVID-19 have also been noted [16]. Medical comorbidities are even more common among PWH, especially older PWH [17–20], when compared with the general population. Many PWH exhibit at least one other high risk condition, and this comorbidity, or multimorbidity if they exhibit multiple other risk factors, increases their chance of having severe COVID-19 [21].
In Washington, DC, the first COVID-19 case was reported on March 7, 2020 [22]; since then there have been over 134,000 cases and 1319 deaths as of February 28, 2022 [23]. In a densely populated city where 57.5% of the population are racial/ethnic minorities, 54.7% of the population has at least one chronic medical condition [24] and there are an estimated 12,300 PWH [25], the city has become one of the national hotspots for COVID-19 infection [26]. At the same time that PWH living in this area are grappling with the impacts of COVID-19 on daily life and their potential increased health risk from COVID-19, they must continue to maintain their HIV-related care, medication-taking behavior, and viral suppression. Given this background, we used data from the DC Cohort to assess the impact of COVID-19 among HIV outpatients in Washington, DC during the first year of the pandemic. Specifically, we studied the proportion at high risk of severe COVID-19 disease and assessed the impact of the COVID-19 pandemic on HIV service utilization.
Methods
Study Setting
This was a secondary data analysis of DC Cohort data. The DC Cohort is a clinical cohort of people receiving HIV care at 15 outpatient clinical sites in Washington, DC. Following informed consent at the 14 out of 15 DC Cohort sites where separate consent to participate in the DC Cohort is required, participants in the DC Cohort have their demographic and clinical data electronically and manually abstracted from the electronic health record (EHR) and entered into a centralized database on a monthly basis. Data are periodically linked to DC Department of Health HIV/AIDS, Hepatitis, STD, TB Administration (HAHSTA) data [27, 28]. All study procedures are approved by the George Washington University Institutional Review Board (IRB).
Inclusion Criteria
Included in this analysis were participants who were DC residents greater than 18 years old and with “active” study status at select DC Cohort sites as of June 30, 2019 and as of June 30, 2020. Active study status in each of those time intervals was defined as having an encounter in the 18 months prior to June 30, 2019 or in the 18 months prior to June 30, 2020. Sites that were included had utilization data (i.e. HIV encounter date or HIV viral load test date) available from both March–June 2019 and March–June 2020 (9 out of 15 clinic sites). Patients did not have to be active at both time points; these were separate samples used to compare 2019 to 2020. Descriptive statistics were calculated using the sample active on June 30, 2019. The sample in 2019 was used to characterize risk of severe COVID-19.
Outcome: Risk of Severe COVID-19
We classified participants into three categories to indicate risk of severe COVID-19 disease, adapted from Banerjee et al. [21], which was a general population study and therefore did not include CD4 count as a risk factor. The high-risk category included individuals with BMI > 40 m/kg2, cardiovascular disease, diabetes and renal disease. Increased risk included BMI > 30 m/kg2, hypertension, respiratory disease, history of transplant, liver disease, autoimmune/rheumatologic disease, cancer, any smoking history, substance abuse, asthma, hemoglobin disorder and HIV with immunosuppression (CD4 < 200 cells/mm3 or not on ART). Low risk had none of the conditions. Additional details of this categorization, including ICD 9/10 codes, are displayed in Supplemental Table 1. Low risk participants had none of the conditions displayed in Supplemental Table 1, while increased risk and high-risk participants had at least one of the increased risk or high-risk conditions, respectively. We described the proportion of participants meeting the definition of increased risk and high risk, and the proportion in each risk category overall and by age group.
Outcome: Utilization
We also examined the impact of the pandemic on encounters and HIV-related laboratory testing. An encounter could be either an in-person or remote encounter (audio or video). The proportion of active participants with an encounter during a given month in 2019 was compared with the proportion of active participants with an encounter during the same month in 2020, after the start of the pandemic. Additionally, the proportion of active patients with an HIV RNA test during a given month in 2019 was compared with the proportion of active patients with an HIV RNA test during the same month in 2020. HIV RNA results are results available in the EHR for that patient, regardless of location (e.g., clinic vs outside lab provider) performed. Finally, we examined the proportion of participants who were virally suppressed (HIV RNA < 50 copies/mL) on the last assessment from 2019 compared with the proportion virally suppressed on the last assessment from 2020. Chi-square testing was used to compare proportions.
Predictor Variables
At enrollment into the DC cohort, and during the annual update process, the following variables used in this analysis were collected by manual abstraction from the medical record: Age, race, gender, housing, employment, smoking, and substance use disorder. The following are electronically abstracted from the medical record: antiretroviral (ART) prescription, HIV RNA, and CD4 cell count.
Results
As shown in Table 1, of the 3584 participants, the median age was 53 years and 1242 (34.7%) were aged 51–60. Most were male (64.2%) and Non-Hispanic Black (81.0%). A significant proportion were unemployed or disabled (38.5%). The participants had a high median recent CD4 count (643 cells/mm3). A small proportion (8.6%) had last HIV RNA > 200 copies/ml. A high proportion (58.8%) had ever smoked. Comorbidities and elevated BMI were common. Many participants were either at increased (58.3%) or high (33.8%) risk for severe COVID-19 if they were to become infected.
Table 1.
Characteristics of actively enrolled DC residents as of June 30, 2019, DC Cohort, (n = 3584)
| Variable | N | % |
|---|---|---|
| Age (median, IQR) | 53 (43–60) | |
| 19–30 | 160 | 4.5 |
| 31–40 | 568 | 15.8 |
| 41–50 | 782 | 21.8 |
| 51–60 | 1242 | 34.7 |
| 61–70 | 704 | 19.6 |
| 71–80 | 118 | 3.3 |
| 81 + | 10 | 0.2 |
| Gender | ||
| Cisgender male | 2300 | 64.2 |
| Cisgender female | 1189 | 33.2 |
| Transgender female (assigned male at birth) | 88 | 2.5 |
| Transgender male (assigned female at birth) | 7 | 0.2 |
| Unknown | 0 | 0.0 |
| Race/ethnicity | ||
| Non-Hispanic black | 2904 | 81.0 |
| Non-Hispanic white | 348 | 9.7 |
| Hispanic | 203 | 5.7 |
| Other/unknown | 129 | 2.6 |
| Unstably housed/homeless | 400 | 11.2 |
| Unemployed/disabled | 1380 | 38.5 |
| Recent CD4 count (last year only)a (median, IQR) | 643 (445–883) | |
| Recent HIV viral load (last year only)a, copies/mL (median, IQR) | UD (UD-30) | |
| Recent HIV viral load > 200 copies/mL (last year only)a | 308 | 8.6 |
| Not on ART | 159 | 4.44 |
| Ever smoker | 2108 | 58.8 |
| Substance use disorder | 520 | 14.5 |
| Chronic kidney disease/ESRD/hemodialysis | 428 | 11.9 |
| Liver disease | 237 | 6.6 |
| Cancer | 318 | 8.9 |
| Hypertensiona | 2115 | 59.0 |
| Diabetesa | 642 | 17.9 |
| BMI 30 + (weight last year only)a kg/m2 | 995 | 27.8 |
| BMI 40 + (weight last year only)a kg/m2 | 228 | 6.4 |
| Cardiovascular disease | 337 | 9.4 |
| Respiratory disease | 686 | 19.1 |
| Asthma | 599 | 16.7 |
| History of transplant | 0 | 0.0 |
| Autoimmune/rheumatologic disease | 97 | 2.7 |
| Hemoglobin disorder | 7 | 0.2 |
| Dementia | 26 | 0.7 |
| Risk for severe COVID-19 | ||
| Low risk | 283 | 7.9 |
| Increased risk | 2090 | 58.3 |
| High risk | 1211 | 33.8 |
ESRD end stage renal disease, ART antiretroviral therapy, BMI body mass index, UD undetectable
aDue to missing values for clinical/laboratory results in the last year (Jul 2018–Jun 2019), not all participants could be evaluated for all conditions: BMI (n = 3133 with weight in last year and height ever); blood pressure (n = 3,198); glucose (n = 3158); HbA1c (n = 800), CD4 count (n = 2848), HIV viral load (n = 2822)
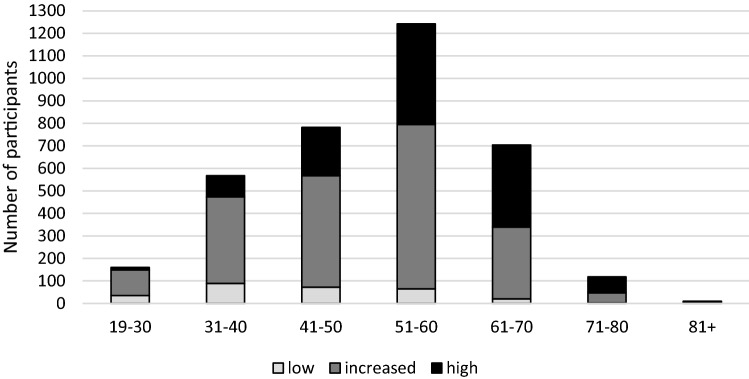
Table 2 and Fig. 1 show the distribution of increased/high risk of severe COVID-19 disease stratified by age group. The vast majority of DC Cohort participants at all ages fell into the increased or high risk of severe COVID-19 groups. The highest number of high-risk participants was among the 51–60 year olds (N = 447) and the highest proportion of high risk participants was among those older than 80 years of age (80%).
Table 2.
Distribution of increased/high risk of severe COVID-19 disease stratified by age group, DC Cohort, 6/3/2019 (n = 3584)
| Age | Total in age group | Low risk | Row % | Increased risk | High risk | Row % | |
|---|---|---|---|---|---|---|---|
| N | n | n | Row % | n | |||
| 19–30 | 160 | 35 | 21.9 | 114 | 71.3 | 11 | 6.9 |
| 31–40 | 568 | 89 | 15.7 | 384 | 67.6 | 95 | 16.7 |
| 41–50 | 782 | 72 | 9.2 | 496 | 63.4 | 214 | 27.4 |
| 51–60 | 1242 | 65 | 5.2 | 730 | 58.8 | 447 | 35.9 |
| 61–70 | 704 | 20 | 2.8 | 319 | 45.3 | 365 | 51.8 |
| 71–80 | 118 | 2 | 1.7 | 45 | 38.1 | 71 | 60.2 |
| 81+ | 10 | 0 | 0 | 2 | 20.0 | 8 | 80.0 |
| Overall | 3584 | 283 | 7.9 | 2090 | 58.3 | 1211 | 33.8 |
Fig. 1.
Risk group of DC Cohort participants by age, N = 3584
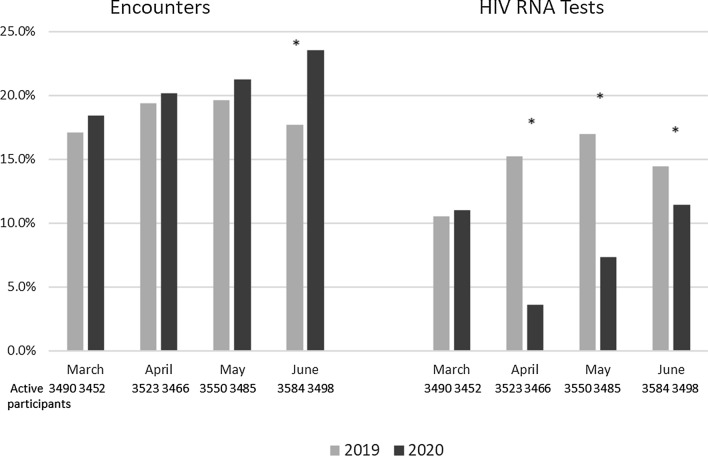
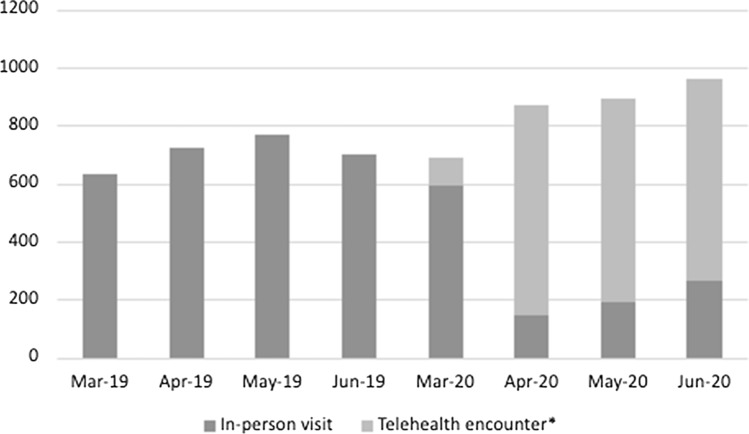
Figure 2 and Table 3 show utilization (encounters) and lab tests in 2019 and 2020. Comparing monthly encounters between 2019 and 2020, the proportion of participants with at least 1 encounter was similar in March and April but was significantly higher in June (23.5% vs. 17.7%, p < 0.0001). Figure 3 shows the proportion of encounters that were remote by month. Two-thirds (65.1%) of the encounters in March to June 2020 were remote encounters. Additionally, the proportion of individuals with at least one HIV RNA test was significantly lower in April, May and June 2020 compared to April, May and June 2019 (as an example, 15.2% of the cohort had an HIV RNA test in 2019 while 3.6% of the cohort had an HIV RNA test in 2020, p < 0.0001).The proportion of participants with an undetectable viral load (< 50 copies/ml) declined from 79.4% in 2019 to 76.2% in 2020 (p = 0.04).
Fig. 2.
Proportion of active patients who had an HIV-related encounter or HIV RNA tests performed, March–June 2019 and March–June 2020, DC Cohort
Table 3.
Utilization and labs, 3/1/19–6/30/19 and 3/1/20–6/30/20
| Utilization/labs | March 2019 | April 2019 | May 2019 | June 2019 |
|---|---|---|---|---|
| Number of active participants at end of month | 3490 | 3523 | 3550 | 3584 |
| Number of unique participants with > 1 encounter | 596 | 682 | 697 | 635 |
| % with > 1 encounter | 17.1% | 19.4% | 19.6% | 17.7% |
| Number of encounters, all participants | 634 | 722 | 769 | 702 |
| Number of unique participants with HIV RNA test | 368 | 537 | 603 | 517 |
| % with > 1 HIV RNA test | 10.5% | 15.2% | 17.0% | 14.4% |
| Number of HIV RNA tests, all participants | 570 | 751 | 816 | 702 |
| Number of unique participants with CD4 test | 350 | 537 | 609 | 505 |
| Number of CD4 tests, all participants | 552 | 736 | 810 | 682 |
| Utilization/labs | March 2020 | April 2020 | May 2020 | June 2020 |
|---|---|---|---|---|
| Number of active participants at end of month | 3452 | 3466 | 3485 | 3498 |
| Number of unique participants with > 1 encounter | 636 | 698 | 740 | 823 |
| % with > 1 encounter | 19.8% | 20.1% | 212.2% | 23.5% |
| Number of encounters, all participants | 686 | 872 | 893 | 963 |
| Number of unique participants with HIV RNA test | 381 | 124 | 255 | 399 |
| % with > 1 HIV RNA test | 11.0% | 3.5% | 7.3% | 11.4% |
| Number of HIV RNA tests, all participants | 392 | 131 | 259 | 411 |
| Number of unique participants with CD4 test | 370 | 114 | 253 | 380 |
| Number of CD4 tests, all participants | 401 | 131 | 280 | 419 |
| Utilization/labs | 3/1/19–6/30/19 | 3/1/20–6/30/20 |
|---|---|---|
| Number of active participants at end of interval | 3584 | 3498 |
| Number of unique participants with > 1 encounter | 2610 | 1997 |
| Number of encounters, all participants | 2834 | 3414 |
| Number of participants with HIV RNA test | 2025 | 1082 |
| Number of HIV RNA tests, all participants | 2839 | 1352 |
| Number of unique participants with CD4 test | 2001 | 1063 |
| Number of CD4 tests, all participants | 2780 | 1193 |
| Proportion with last viral load in interval suppressed (< 50 copies/mL) | 79.4% | 76.2% |
The two proportions with last viral load in interval suppressed between 2019 and 2020 are not equal. The p value is 0.04 and statistically significant
Fig. 3.
Encounter type by month, 2019–2020
Tables 4 and 5 examine the association between demographic and clinical characteristics and having an encounter in the interval between 1 March 2020 and 30 June 2020 (Table 4) or having an HIV RNA test performed in the interval between 1 March 2020 and 30 June 2020 (Table 5). As shown in Table 4, cisgender females were more likely to have an encounter [aOR 1.36 (1.17, 1.58)], as were all other races compared to non-Hispanic white patients (aOR ranging from 1.67 to 2.45, all statistically significant). Individuals who were at high risk for COVID-19 were most likely to have an encounter [aOR 1.39 (1.05, 1.84)].As shown in Table 5, there was no significant difference by gender in having an HIV RNA test. Similarly to the results shown for having an encounter in Table 4, all races other than non Hispanic white were more likely to have an HIV RNA test. Those who were unstably housed were less likely to have an HIV RNA test, while those who were unemployed or disabled were more likely. There was a borderline p-value for an inverse association between being at increased risk for severe COVID-19 and having an HIV RNA test (p = 0.048).
Table 4.
Factors associated with having an encounter, 3/1/2020–6/30/2020, DC Cohort
| Variable | OR (95% CI) (univariate analysis) | aOR (95% CI) (multivariate analysis) |
|---|---|---|
| Age (per 5 year increase) | 1.01 (0.98, 1.04) | 1 (0.97, 1.04) |
| Gender | ||
| Cisgender male (Ref) | 1 | 1 |
| Cisgender female | 1.51 (1.31, 1.75)*** | 1.36 (1.17, 1.58)*** |
| Transgender female (assigned male at birth) | 1.20 (0.79, 1.86) | 1.13 (0.74, 1.76) |
| Transgender male (assigned female at birth) | 2.29 (0.49, 16.0) | 2.08 (0.44, 14.7) |
| Race/ethnicity | ||
| Non-Hispanic Black | 2.29 (1.83, 2.89)*** | 1.93 (1.52, 2.46)*** |
| Non-Hispanic white (Ref) | 1 | 1 |
| Hispanic | 2.53 (1.77, 3.61)*** | 2.45 (1.71, 3.51)*** |
| Other/unknown | 1.87 (1.24, 2.82)* | 1.67 (1.10, 2.53)* |
| Unstably housed/homeless [Yes vs. No (Ref)] | 1.05 (0.85, 1.29) | 1.01 (0.81, 1.25) |
| Unemployed/disabled [(Yes vs. No (Ref)] | 1.27 (1.11, 1.46)*** | 1.10 (0.96, 1.27) |
| Risk for severe COVID-19 | ||
| Low risk (ref) | 1 | 1 |
| Increased risk | 1.25 (0.98, 1.61) | 1.15 (0.89, 1.48) |
| High risk | 1.62 (1.25, 2.10)*** | 1.39 (1.05, 1.84)* |
*p < 0.05; **p < 0.01; ***p < 0.001
Table 5.
Factors associated with having an HIV RNA test performed, 3/1/2020–6/30/2020, DC Cohort
| Variable | OR (95% CI) (univariate analysis) | aOR (95% CI) (multivariate analysis) |
|---|---|---|
| Age (per 5 year increase) | 1.01 (0.98, 1.05) | 1.01 (0.98, 1.05) |
| Gender | ||
| Cisgender male (Ref) | 1 | 1 |
| Cisgender female | 1.18 (1.01, 1.37)* | 1.12 (0.96, 1.31) |
| Transgender female (assigned male at birth) | 0.86 (0.52, 1.38) | 0.89 (0.53, 1.43) |
| Transgender male (assigned female at birth) | 0.41 (0.02, 2.38) | 0.42 (0.02, 2.54) |
| Race/ethnicity | ||
| Non-Hispanic Black | 1.46 (1.13, 1.91)* | 1.34 (1.02, 1.78)* |
| Non-Hispanic white (Ref) | 1 | 1 |
| Hispanic | 2.09 (1.43, 3.05)*** | 2.10 (1.43, 3.07)*** |
| Other/unknown | 1.56 (0.99, 2.43)* | 1.48 (0.93, 2.31) |
| Unstably housed/homeless [Yes vs. No (Ref)] | 0.73 (0.57, 0.92)* | 0.73 (0.57, 0.93)* |
| Unemployed/disabled [Yes vs. No (Ref)] | 1.33 (1.15, 1.54)*** | 1.29 (1.11, 1.51)*** |
| Risk for severe COVID-19 | ||
| Low risk (ref) | 1 | 1 |
| Increased risk | 0.80 (0.61,1.04) | 0.76 (0.58,1.00)* |
| High risk | 0.93 (0.71, 1.23) | 0.84 (0.63, 1.13) |
*p < 0.05; **p < 0.01; ***p < 0.001
Discussion
We demonstrated that a large proportion of PWH receiving HIV care in Washington, DC are at high risk for severe COVID-19 if they acquire SARS-CoV-2 infection. Additionally, we found that while HIV-related encounters were higher in March–June 2020 compared with the same months in the previous year, HIV RNA test utilization was lower. Among those who did receive an HIV RNA test, the proportion who were undetectable was lower.
Our study participants have a mean age of 53 years, and most are on antiretroviral therapy (ART), are virally suppressed, and have CD4 cell counts over 500 cells/mm3. While they may be at lower risk for complications from COVID-19 based on these parameters and having relatively stable HIV disease, most are persons of color and are living with at least one condition known to increase the risk of complications and mortality from COVID-19 [29]. Our findings were similar to an analysis in the general U.S. population showing that a high proportion of Americans (75.4%) are at increased risk for severe COVID-19 given the high prevalence of comorbidities such as obesity and hypertension [30]. These findings highlight that regardless of whether a person is living with HIV, we need to protect those most vulnerable. This involves providing resources including access to medical care for non-COVID-19—related issues, providing insurance to those in need, and providing locations to properly isolate and/or quarantine if needed.
In evaluating clinical utilization among our sample, encounters were stable or higher, which is encouraging news. At the height of the COVID-19 pandemic, the ability to receive in-person services was extremely difficult and impractical [31]. With social distancing measures and other COVID-19-related restrictions, in-person clinic encounters were greatly restricted, and the transition to telehealth and a virtual environment posed some new obstacles and opportunities. Despite those challenges, there has been a significant increase in phone and/or video consultations with care providers, and clinic websites and social media have been updated to reflect current protocols and offer assistance when needed [31]. In general, many HIV clinics had historically high no-show rates pre-pandemic [32] and the increasing use of phone/telehealth encounters may have actually been beneficial in increasing visit adherence. This may be particularly true for those patients with challenges in obtaining transportation, child care, or time off from work [33] or individuals for whom the experience of coming to clinic is stigmatizing and reminds them of their HIV status [34]. However, individuals with less reliable access to phone and/or data may not reap the same benefits and telehealth potentially could worsen disparities [35]. In addition, the clinical impact of not having face-to-face encounters is unknown. Also, the use of preventive services has declined during COVID-19 [36] which may be particularly detrimental to people with HIV who are at higher risk of cardiovascular disease, cancer, and cancer death [37–39].
An analysis of data from the HIV Outpatient Study (HOPS) found an increase in telemedicine visits in 2020, however, determined that total encounters had not rebounded to pre-pandemic levels by September 2020 [40]. However, we found that a higher proportion of active patients had visits in May and June 2020 compared to May and June 2020. The explanation for this difference is not clear. Clinics in DC may have made extra effort to increase encounters in May/June 2020. As clinics envision the ways they will provide care in the future, engagement in care may be enhanced by ensuring the ongoing availability of both in-person and telehealth visits.
Our results showed that lab monitoring, which is an important part of HIV care, was lower during the pandemic. People who are adherent and in good general health may not need as frequent lab monitoring. However, those who are potentially most vulnerable to poor health outcomes may do worse with less frequent lab monitoring. Our finding of a lower proportion of undetectable results in individuals receiving an HIV RNA test in 2020 compared with 2019 has several potential explanations. It may represent challenges obtaining ART due to pandemic-related service interruptions, resulting in less suppression. Or, it may represent that clinicians advised clinically stable patients to delay laboratory testing.
In examining factors associated with using telehealth or having an HIV RNA test, we found that women were more likely to have an encounter, as were individuals who were not of white race. We also showed that the individuals at highest risk for severe COVID-19 were more likely to have an encounter in March–June 2020 and less likely to have an HIV RNA. The significance of these findings is unclear. Generally, women are more likely to seek medical care [41]. Perhaps women in our sample desired the support of their clinic provider or were more worried about COVID and wanted to engage in medical care to discuss their concerns with their provider. With individuals of non-white race being more vulnerable to COVID infection, hospitalization, and death [42, 43], perhaps clinics or providers were making additional effort to engage those patients. Finally, it is interesting that those at increased risk for severe COVID-19 were more likely to have an encounter and less likely to have a HIV RNA test. This may indicate that physicians were advising their most medically fragile patients to stay home if their HIV was stable or medically fragile patients choosing to stay home because of their concern for being exposed to COVID.
Our findings represent a preliminary investigation of how HIV care delivery has been impacted by the COVID pandemic and what those changes may mean for HIV care outcomes. Additional studies that have examined these issues have shown that the pandemic may impact maintenance of viral suppression due to decreased access to clinical services through in-person visits [44]. Our findings were similar to those of Mayer et al. in Boston area HIV clinic [45] with a surge in telehealth and similar viral suppression and differed somewhat from a Midwestern HIV clinic [46] where viral suppression was stable and retention in care decreased. These findings were presented in a review of the impact of COVID 19 [47] which concluded that telehealth offers many opportunities and simultaneously presents many challenges, particularly for the most vulnerable patients. It remains to be seen if people who always did well in HIV care (i.e., were always retained and suppressed) will continue to do well in HIV care, whatever the delivery modality, and people whose needs were not well met by our medical care delivery system prior to the pandemic will continue to struggle to remain engaged and suppressed. We can use DC Cohort data to examine these outcomes over time in future analyses.
The major limitation of our analysis is that we have not yet linked risk of severe COVID-19 to occurrence of severe COVID-19 and/or change in behavior to mitigate COVID-19 acquisition risk. However, we have a major survey underway that will address COVID-19 incidence (self-reported, and electronic health record confirmed) as well as the adoption of risk mitigation behaviors, the social and mental health impacts on PWH, and the use of telehealth. Another limitation is that this analysis included 9 out of 15 DC Cohort clinics based on data availability. Although all DC Cohort clinics were not included, the clinics with data available represented a mix of clinics located in academic centers and community-based clinics. However, these results may not be fully generalizable to all people receiving HIV care in Washington, DC. Another limitation is that we did not collect information on whether HIV RNA test data came from the primary clinical site or an outside lab site. If the primary clinical site was the only place that a patient could get labs, and it was closed, that could explain lack of viral load testing. We are conducting a site survey to capture information about COVID-related service changes and this issue could potentially be addressed in future research.
A major benefit of this survey is that we will be able to link diagnosis and other clinical data to patient-reported survey data. A strength of our study is the large sample and the availability of utilization data with a relatively short turnaround, giving us the ability to examine the impact of COVID-19 on clinical utilization in the short-term. In future analyses, we will use subsequent data to evaluate changes beyond the immediate start of the pandemic (March–June 2020). This will allow us to determine whether these patterns hold throughout 2020 and into 2021, or if they were temporary.
In summary, we found high risk for severe disease among DC Cohort participants and declines in laboratory utilization despite increases in HIV-related contacts. There are several important implications of these findings. First, because many people with HIV have a high risk of severe disease they should be a high priority group for the SARS-CoV-2 vaccine. All those providing health care and wraparound services to PWH should emphasize the importance of vaccination. Our findings additionally reinforce the importance of ensuring continuity of care and consistent access to ARTs and HIV care so that we do not lose momentum and the gains we have made to date in curbing the HIV epidemic in DC and nationally in achieving the 90-90-90 goals. The conduct of systematic studies to further understand interactions between COVID-19 and HIV are needed. These will be facilitated by using existing longitudinal cohorts such as the DC Cohort and enable researchers to assess the incidence of COVID-19 among PWH, as well as its longer- term impacts on the care continuum. Finally, given that we found an impact of the pandemic on service provision for PWH, future studies could assess the impact of the pandemic on services such as STI prevention as well as look at whether the rates of new HIV diagnoses increased and whether those newly diagnosed were able to get started on ART in a timely manner.
Supplementary Information
Below is the link to the electronic supplementary material.
Acknowledgements
Data in this manuscript were collected by the DC Cohort Study Group with investigators and research staff located at: Children's National Medical Center Adolescent (Lawrence D’Angelo) and Pediatric (Natella Rakhmanina) clinics; The Senior Deputy Director of the DC Department of Health HAHSTA (Michael Kharfen); Family and Medical Counseling Service (Michael Serlin); Georgetown University (Princy Kumar); The George Washington University Biostatistics Center (Vinay Bhandaru, Tsedenia Bezabeh, Nisha Grover-Fairchild, Lisa Mele, Susan Reamer, Alla Sapozhnikova, Greg Strylewicz, Marinella Temprosa, and Kevin Xiao); The George Washington University Department of Epidemiology (Morgan Byrne, Amanda Castel, Alan Greenberg, Maria Jaurretche, Paige Kulie, Anne Monroe, James Peterson, Bianca Stewart, and Brittany Wilbourn) and Department of Biostatistics and Bioinformatics (Yan Ma); The George Washington University Medical Faculty Associates (Hana Akselrod); Howard University Adult Infectious Disease Clinic (Jhansi L. Gajjala) and Pediatric Clinic (Sohail Rana); Kaiser Permanente Mid-Atlantic States (Michael Horberg); La Clinica Del Pueblo (Ricardo Fernandez); MetroHealth (Annick Hebou); National Institutes of Health (Carl Dieffenbach, Henry Masur); Washington Health Institute, formerly Providence Hospital (Jose Bordon); Unity Health Care (Gebeyehu Teferi); Veterans Affairs Medical Center (Debra Benator); Washington Hospital Center (Maria Elena Ruiz); and Whitman-Walker Institute (Stephen Abbott).
Author Contributions
ADC, MEL, and AKM designed the study, MEL, JX, MT, AKM processed the data, performed the analysis, and designed the tables and figures. AKM, JX, AEG, MEL, MT, ADC aided in interpreting the results. AKM took the lead in writing the manuscript with assistance from JBR. All authors provided critical feedback and helped shape the research, analysis and manuscript.
Funding
National Institute of Allergy and Infectious Diseases, 1R24AI152598-01.
Data Availability
Deidentified data available upon written request.
Code Availability
Analytic code available upon written request.
Declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Ethical Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. The study was approved by the George Washington University Institutional Review Board (IRB 071029).
Consent to Participate
Informed consent was obtained from all individual participants included in the study.
Consent for Publication
N/A.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Anne K. Monroe, Email: amonroe@gwu.edu
The DC Cohort Executive Committee:
Lawrence D’Angelo, Natella Rakhmanina, Michael Kharfen, Michael Serlin, Princy Kumar, Vinay Bhandaru, Tsedenia Bezabeh, Nisha Grover-Fairchild, Lisa Mele, Susan Reamer, Alla Sapozhnikova, Greg Strylewicz, Marinella Temprosa, Kevin Xiao, Morgan Byrne, Amanda Castel, Alan Greenberg, Maria Jaurretche, Paige Kulie, Anne Monroe, James Peterson, Bianca Stewart, Brittany Wilbourn, Yan Ma, Hana Akselrod, Jhansi L. Gajjala, Sohail Rana, Michael Horberg, Ricardo Fernandez, Annick Hebou, Carl Dieffenbach, Henry Masur, Jose Bordon, Gebeyehu Teferi, Debra Benator, Maria Elena Ruiz, and Stephen Abbott
References
- 1.Johns Hopkins Coronavirus Resource Center. COVID-19 dashboard by the Center for Systems Science and Engineering (CSSE) at Johns Hopkins University (JHU). https://coronavirus.jhu.edu/map.html. Accessed 17 Mar 2022.
- 2.Mackey K, Ayers CK, Kondo KK, Saha S, Advani SM, Young S, et al. Racial and ethnic disparities in COVID-19–related infections, hospitalizations, and deaths. Ann Intern Med. 2020 doi: 10.7326/m20-6306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Seligman B, Ferranna M, Bloom DE. Social determinants of mortality from COVID-19: a simulation study using NHANES. PLoS Med. 2021;18:1–13. doi: 10.1371/journal.pmed.1003490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Lesko C, Bengtson A. HIV and COVID-19: intersecting epidemics with many unknowns. Am J Epidemiol. 2021;190:10–16. doi: 10.1093/aje/kwaa158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Blanco J, Ambrosioni J, Garcia F, Martinez E, Soriano A, Mallolas J, et al. COVID-19 in patients with HIV: clinical case series. Lancet HIV. 2020 doi: 10.1016/S2352-3018(20)30111-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Gervasoni C, Meraviglia P, Riva A, Giacomelli A, Oreni L, Minisci D, et al. Clinical features and outcomes of patients with human immunodeficiency virus with COVID-19. Clin Infect Dis. 2020 doi: 10.1093/cid/ciaa579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Sigel K, Swartz T, Golden E. Covid-19 and people with HIV infection: outcomes for hospitalized patients in New York city. Clin Infect Dis. 2020 doi: 10.1093/cid/ciaa880. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Karmen-Tuohy S, Carlucci P, Zervou F, Zacharioudakis I, Rebick G, Klein E, et al. Outcomes among HIV-positive patients hospitalized with COVID-19. J Acquir Immune Defic Syndr. 2020;395:10229. doi: 10.1097/QAI.0000000000002423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Del Amo J, Polo R, Moreno S. Incidence and severity of COVID-19 in HIV-positive persons receiving antiretroviral therapy. Ann Intern Med. 2020 doi: 10.7326/M20-3689. [DOI] [PubMed] [Google Scholar]
- 10.Bhaskaran K, Rentsch CT, MacKenna B, Schultze A, Mehrkar A, Bates CJ, et al. HIV infection and COVID-19 death: a population-based cohort analysis of UK primary care data and linked national death registrations within the OpenSAFELY platform. Lancet HIV. 2021;8:e24–e32. doi: 10.1016/S2352-3018(20)30305-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Dandachi D, Geiger G, Montgomery MW, Karmen-Tuohy S, Golzy M, Antar AAR, et al. Characteristics, comorbidities, and outcomes in a multicenter registry of patients with human immunodeficiency virus and coronavirus disease 2019. Clin Infect Dis. 2020;65212:1–9. doi: 10.1093/cid/ciaa1339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Collins LF. Clinical characteristics, comorbidities and outcomes among persons with HIV hospitalized with coronavirus disease 2019 in Atlanta Georgia. AIDS. 2020;34:1789. doi: 10.1097/QAD.0000000000002632. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Cunningham JW, Vaduganathan M, Claggett BL, Jering KS, Bhatt AS, Rosenthal N, et al. Clinical outcomes in young US adults hospitalized with COVID-19. JAMA Intern Med. 2020 doi: 10.1001/jamainternmed.2020.5313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Petrilli CM, Jones SA, Yang J, Rajagopalan H, O’Donnell L, Chernyak Y, et al. Factors associated with hospital admission and critical illness among 5279 people with coronavirus disease 2019 in New York City: prospective cohort study. BMJ. 2020 doi: 10.1136/bmj.m1966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Williamson EJ, Walker AJ, Bhaskaran K, Bacon S, Bates C, Morton CE, et al. Factors associated with COVID-19-related death using OpenSAFELY. Nature. 2020;584:430–436. doi: 10.1038/s41586-020-2521-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Weiser JK, Tie Y, Beer L, Neblett Fanfair R, Shouse RL. Racial/ethnic and income disparities in the prevalence of comorbidities that are associated with risk for severe COVID-19 among adults receiving HIV care, United States, 2014–2019. J Acquir Immune Defic Syndr. 2021;86(3):297–304. doi: 10.1097/QAI.0000000000002592. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Levy ME, Greenberg AE, Hart R, Powers Happ L, Hadigan C, Castel A, et al. High burden of metabolic comorbidities in a citywide cohort of HIV outpatients: evolving health care needs of people aging with HIV in Washington, DC. HIV Med. 2017;18:724–735. doi: 10.1111/hiv.12516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Collins LF, Armstrong WS. What it means to age with HIV infection: years gained are not comorbidity free. JAMA Netw Open. 2020;3:e208023. doi: 10.1001/jamanetworkopen.2020.8023. [DOI] [PubMed] [Google Scholar]
- 19.Gallant J, Hsue PY, Shreay S, Meyer N. Comorbidities among US patients with prevalent HIV infection—a trend analysis. J Infect Dis. 2017;216:1525–1533. doi: 10.1093/infdis/jix238. [DOI] [PubMed] [Google Scholar]
- 20.Mayer KH, Loo S, Crawford PM, Crane HM, Leo M, Denouden P, et al. Excess clinical comorbidity among HIV-infected patients accessing primary care in US community health centers. Public Health Rep. 2018;133:109–118. doi: 10.1177/0033354917748670. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Banerjee A, Pasea L, Harris S, et al. Estimating excess 1-year mortality associated with the COVID-19 pandemic according to underlying conditions and age: a population-based cohort study. Lancet. 2020;395:1715. doi: 10.1016/S0140-6736(20)30854-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.The New York Times. Tracking Coronavirus in Washington, D.C.: latest map and case count. https://www.nytimes.com/interactive/2020/us/washington-dc-coronavirus-cases.html. Accessed 17 Mar 2022.
- 23.DC Health. COVID-19 Surveillance. https://coronavirus.dc.gov/data. Accessed 17 Mar 2022.
- 24.Partnership to fight chronic disease. What is the impact of chronic disease on Washington, D.C.? https://www.fightchronicdisease.org/sites/default/files/download/PFCD_DC_Factsheet_FINAL1.pdf. Accessed 17 Mar 2022.
- 25.DC Health. Annual Epidemiology & Surveillance Report. https://dchealth.dc.gov/sites/default/files/dc/sites/doh/publication/attachments/2021%20Annual%20Surveillance%20Report_final_3.29.pdf. Accessed 17 Mar 2022.
- 26.Washington Post. White House says D.C. region among worst in country, as summer closures continue. https://www.washingtonpost.com/local/white-house-says-dc-region-among-worst-in-country-as-summer-closures-continue/2020/05/22/31e4cc8c-9c3a-11ea-ac72-3841fcc9b35f_story.html. Accessed 22 May 2020.
- 27.Greenberg AE, Hays H, Castel AD, Subramanian T, Happ LP, Jaurretche M, et al. Development of a large urban longitudinal HIV clinical cohort using a web-based platform to merge electronically and manually abstracted data from disparate medical record systems: technical challenges and innovative solutions. J Am Med Inform Assoc. 2016;23:635–643. doi: 10.1093/jamia/ocv176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Castel AD, Terzian A, Opoku J, Happ LP, Younes N, Kharfen M, et al. Defining care patterns and outcomes among persons living with HIV in Washington, DC: linkage of clinical cohort and surveillance data. JMIR Public Health Surveill. 2018;4:e23. doi: 10.2196/publichealth.9221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Centers for Disease Control. People at increased risk and other people who need to take extra precautions. 2021. https://www.cdc.gov/coronavirus/2019-ncov/need-extra-precautions/people-at-higher-risk.html. Accessed 3 Jan 2021
- 30.Ajufo E, Rao S, Navar AM, Pandey A, Ayers CR, Khera A. U.S. population at increased risk of severe illness from COVID-19. Am J Prev Cardiol. 2021;6:100156. doi: 10.1016/j.ajpc.2021.100156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Pinto R, Park S. COVID-19 pandemic disrupts HIV continuum of care and prevention: implications for research and practice concerning community-based organizations and frontline providers. AIDS Behav. 2020;24:1–4. doi: 10.1007/s10461-020-02893-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Adams JA, Whiteman K, McGraw S. Reducing missed appointments for patients with HIV: an evidence-based approach. J Nurs Care Qual. 2020;35:165–170. doi: 10.1097/NCQ.0000000000000434. [DOI] [PubMed] [Google Scholar]
- 33.Yehia BR, Stewart L, Momplaisir F, Mody A, Holtzman CW, Jacobs LM, et al. Barriers and facilitators to patient retention in HIV care. BMC Infect Dis. 2015;15:1–10. doi: 10.1186/s12879-015-0990-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Vanable PA, Carey MP, Blair DC, Littlewood RA. Impact of HIV-related stigma on health behaviors and psychological adjustment among HIV-positive men and women. AIDS Behav. 2006;10:473–482. doi: 10.1007/s10461-006-9099-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Velasquez D, Mehrotra A. Ensuring the growth of telehealth during COVID-19 does not exacerbate disparities in care. Health Aff Blog. 2020 doi: 10.1377/hblog20200505.591306. [DOI] [Google Scholar]
- 36.Martin K, Kurowski D, Given P, Kennedy K, Clayton E. The impact of COVID-19 on the use of preventive healthcare. 2021. https://healthcostinstitute.org/hcci-research/the-impact-of-covid-19-on-the-use-of-preventive-health-care. Accessed 1 Mar 2021
- 37.Coghill AE, Pfeiffer RM, Shiels MS, Engels EA. Excess mortality among HIV-infected individuals with cancer in the United States. Cancer Epidemiol Biomarkers Prev. 2017;26:1027–1033. doi: 10.1158/1055-9965.EPI-16-0964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Alonso A, Barnes AE, Guest JL, Shah A, Shao IY, Marconi V. HIV infection and incidence of cardiovascular diseases: an analysis of a large healthcare database. J Am Heart Assoc. 2019 doi: 10.1161/JAHA.119.012241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Engels EA, Biggar RJ, Hall HI, Cross H, Crutchfield A, Finch JL, et al. Cancer risk in people infected with human immunodeficiency virus in the United States. Int J Cancer. 2008;123:187–194. doi: 10.1002/ijc.23487. [DOI] [PubMed] [Google Scholar]
- 40.Tedaldi EM, Hou Q, Armon C, Palella FJ, Li J, Simoncini G, et al. HIV Ambulatory care during COVID-19 pandemic in US: visits and viral load testing. Topics Antivir Med. 2021;2021:294. [Google Scholar]
- 41.Brett KM and Burt CW. Utilization of ambulatory medical care by women; United States, 1997-1998. (2001). Accessed from https://www.cdc.gov/nchs/data/series/sr_13/sr13_149.pdf [DOI] [PubMed]
- 42.Price-Haywood EG, Burton J, Fort D, Seoane L. Hospitalization and mortality among black patients and white patients with Covid-19. N Engl J Med. 2020;382(26):2534–2543. doi: 10.1056/NEJMsa2011686. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.CDC. Risk for COVID-19 Infection, Hospitalization, and death by race/ethnicity https://www.cdc.gov/coronavirus/2019-ncov/covid-data/investigations-discovery/hospitalization-death-by-race-ethnicity.html. Accessed 31 Oct 2021
- 44.Spinelli MA, Hickey MD, Glidden DV, Nguyen JQ, Oskarsson JJ, Havlir D, Gandhi M. Viral suppression rates in a safety-net HIV clinic in San Francisco destabilized during COVID-19. AIDS. 2020;34(15):2328–2331. doi: 10.1097/QAD.0000000000002677. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Mayer KH, Levine K, Grasso C, Multani A, Gonzalez A, Biello K. 541. Rapid migration to telemedicine in a Boston Community Health Center is associated with maintenance of effective engagement in HIV care. Open Forum Infect Dis. 2020;7(Suppl 1):S337–S338. doi: 10.1093/ofid/ofaa439.735. [DOI] [Google Scholar]
- 46.Fadul N. 112. A quality management project of a midwestern academic HIV clinic operation during COVID-19: implementation strategy and preliminary outcomes. Open Forum Infect Dis. 2020;7:S184–S185. doi: 10.1093/ofid/ofaa439.422. [DOI] [Google Scholar]
- 47.Budak JZ, Scott JD, Dhanireddy S, Wood BR. The impact of COVID-19 on HIV care provided via telemedicine-past, present, and future. Curr HIV/AIDS Rep. 2021;18(2):98–104. doi: 10.1007/s11904-021-00543-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Deidentified data available upon written request.
Analytic code available upon written request.