Abstract
The COVID‐19 pandemic is a highly dramatic concern for mankind. In Italy, the pandemic exerted its major impact throughout the period of February to June 2020. To date, the awkward amount of more than 134,000 deaths has been reported. Yet, post‐mortem autopsy was performed on a very modest number of patients who died from COVID‐19 infection, leading to a first confirmation of an immune‐thrombosis of the lungs as the major COVID‐19 pathogenesis, likewise for SARS. Since then (June–August 2020), no targeted early therapy considering this pathogenetic issue was approached. The patients treated with early anti‐inflammatory, anti‐platelet, anticoagulant and antibiotic therapy confirmed that COVID‐19 was an endothelial inflammation with immuno‐thrombosis. Patients not treated or scarcely treated with the most proper and appropriate therapy and in the earliest, increased the hospitalization rate in the intensive care units and also mortality, due to immune‐thrombosis from the pulmonary capillary district and alveoli. The disease causes widespread endothelial inflammation, which can induce damage to various organs and systems. Therapy must be targeted in this consideration, and in this review, we demonstrate how early anti‐inflammatory therapy may treat endothelia inflammation and immune‐thrombosis caused by COVID‐19, by using drugs we are going to recommend in this paper.
Keywords: immunopharmacology, immunotoxicology, infection, inflammation, pharmacoepidemiology
1. INTRODUCTION ON THE IMMUNO‐THROMBOTIC ETIOPATHOGENESIS OF COVID‐19
The coronavirus disease 2019 (COVID‐19) had a huge impact on health management worldwide. It generated controversial issues and great concerns even on authorized and agreed therapeutic protocols for SARS‐CoV2 pandemic, particularly in people staying at home with early symptoms of COVID‐19. 1 , 2 , 3 , 4 From a pathogenetic point of view, COVID‐19 is a clinical syndrome caused by an infection associated with a severe acute respiratory syndrome (ARDS) from SARS‐CoV‐2, essentially due to damage to the lung functionality. ARDS is most probably caused by a progressive disruption of the barrier integrity in endothelia, therefore causing a diffuse lung damage. Impaired cross talk between coagulation cascade and immunity is a major hallmark of ARDS, which rapidly leads to an exacerbated inflammatory response and a diffuse fibrin deposition both in vascular micro‐circulation and in alveoli. Concurrent factors such as activated platelets, innate immune cells, neutrophils' NETs and proteases from coagulation lead altogether to the immune‐thrombosis, which can be reported as the leading cause of the lung functional failure. 5 , 6 , 7 Patients with a severe clinical stage exhibit an exaggerated activation of the immune system which can cause a range of damage to various organs, as well as pneumonia and acute respiratory distress syndrome (ARDS). 8 The infection process of SARS‐CoV2 increases the cyclo‐oxygenase 2 (COX‐2) expression, and anti‐inflammatory drugs enable to inhibit COX‐2 may exert a formidable action in preventing the onset of COVID‐19. 9
SARS‐CoV2 infection induces, therefore, a pathogenetic process known as immuno‐thrombosis. 5
Although many individuals develop asymptomatic or pauci‐symptomatic infection, those ones with severe and critical disease exhibit a dysregulated over‐activation of the immune system which can cause an abnormal immune response with impaired production of cytokines, which is likely to be responsible for most of the organ damage that occurs in the final stage of the disease. 10 , 11 , 12
To date, recent investigation has outlined that SARS‐CoV‐2 infection induces a pro‐thrombotic state expressed mainly as immuno‐thrombosis, that is, thrombotic events associated with inflammation in microcirculation, 5 an evidence even reported for SARS and in the very few post‐mortem investigations (autoptic data), which we missed as a major evidence to shed a light on the real COVID‐19 pathogenesis. 13 , 14 , 15 , 16 Autopsy, which was widely neglected to highlight and elucidate COVID‐19 pathogenesis, is the most awkward ‘missing link’ to address COVID‐19 pathogenesis, and hence a therapy protocol with the highest successful outcome.
Moreover, restrictions due to the several lockdown policies worldwide have exacerbated the difficulty in having a plain consultancy with practitioners, family doctors and other physicians, particularly due to COVID‐19 emergency in hospitals. This engulfed a huge need of medical expertise and caregivers, often subtracting people from having an available consultancy at home or at their disposal. In this perspective, suggestions and recommendations were even reported elsewhere in the literature, 1 , 4 despite some critical comment. 17 , 18 , 19 , 20 The fundamental role of a home protocol to address mild COVID‐19 or the early symptoms of this pathology is to ensure a rapid pharmacological intervention in order to absolutely prevent hospitalization, often due to exacerbation more than excessive caution. In this sense, therapeutic drugs must be promptly available, easy to be provided and able to target the first steps leading to the etiopathogenesis of COVID‐19. 9
However, the first line of choice is quite never addressing this recommendation, but simply reducing and dampening any early painful symptom, such as fever.
In this respect, antipyretics such as acetaminophen (paracetamol) underwent a huge increase in their use during COVID‐19 pandemic, though its use may be hazardous for COVID‐19 patients. 17 , 21 , 22 , 23 In Italy, the government's recommendation to use paracetamol (acetaminophen) against the early symptoms of COVID‐19 (pauci‐symptomatic subjects) was officially forwarded. 17 It is difficult to state if the need for an official recommendation would like to supply the paucity of available doctors, most of whom engaged in COVID‐19 units, yet recent data have outlined that reducing fever with paracetamol may lead patients to a severe form of COVID‐19, compelling physicians to recommend for their immediate hospitalization. 19 Furthermore, a certain frenzy in retrieving novel and straightforward therapeutic strategies against SARS‐CoV2, such as the very expensive monoclonal antibodies, overshadowed optimal and frequently used anti‐inflammatory drugs, probably due to the up‐growing conviction that SARS‐CoV2 was an unusual virus, poorly addressable by commonly used therapy. 9
In this review, an attempt in elucidating the high potentiality of an easy to perform and priceless therapy against COVID‐19 at home, upon physician counselling, is reported. Our firm opinion is that a proper therapeutic protocol in the early symptoms of COVID‐19 at home should have rescued many patients, reporting them to their own healthy lives. The present review aims at demonstrating how early anti‐inflammatory therapy is able to treat endothelia‐vascular disorder caused by immuno‐thrombosis in COVID‐19, for which recommendations about the drugs that can be used, are herein recommended.
2. FEVER AS AN EARLY SYMPTOM OF COVID‐19. ACETAMINOPHEN SHOULD NOT BE RECOMMENDED AT ALL
During the early stages of the SARS‐CoV2 infection, fever is the most frequent and common symptom, affecting about 42% of children and 45–89% of adults. 24 , 25 , 26 Both patients and physicians very frequently use and prescribe, respectively, antipyretics as a commonly available and over‐the‐counter medication to dampen fever‐related discomfort and pain. 21 , 23 , 27 , 28 Yet, in this context, the use of acetaminophen (N‐[4‐hydroxyphenyl]‐ethanamide), known also as paracetamol, to treat early pre‐hospitalization COVID‐19 symptoms, such as fever, has been recently debated by papers dealing with fever in COVID‐19. 29 , 30 , 31 , 32 Peluso et al., 32 who described fever as a complex phenomenology during SARS‐CoV2 infection, suggested to properly managing fever during severe COVID‐19, reporting that targeting temperature management should help patients with severe symptoms to increase their survival possibility, although the complexity of body temperature maintenance during COVID‐19 cannot allow physicians to recommend an aggressive antipyretic therapy. In his paper, Peluso et al. 32 reported that, because of the high incidence in vascular thrombosis during COVID‐19, an increase in damage‐associated molecular patterns (DAMPs) should occur, so triggering an immune response and release of pyrogenic factors. However, they admit also that there are no data about the optimal timing, duration and severity of fever to promote a correct intervention on the patient's body temperature. Peluso et al. 32 suggest to use indifferently either anti‐pyretics, such as paracetamol or anti‐inflammatory pharmaceuticals.
Despite their recommendation in reducing fever in more critical stages of COVID‐19, suggesting some consideration regarding the use of antipyretics in severe COVID‐19, that is, mainly during hospitalization, a more rigorous debate about the role of fever in COVID‐19 should be undertaken. Certainly, the role of selective inhibitors of COX‐2, such as celecoxib, might give fundamental insights even in this occurrence, as the increase in COX‐2 in the lung enhances the damage to this tissue particularly during the mechanical ventilation. 33
The major concern of acetaminophen upon SARS‐CoV2 infection is associated with its use in early symptomatology, during a mild COVID‐19, while staying at home.
Taking pills without a direct physician's assistance may lead to severe adverse effects, particularly metabolic acidosis. 34 , 35 , 36 Some authors are persuaded that fever during COVID‐19, even with mild or moderate symptomatology, has a formidable role in strengthening the host's immune response. 37 Pain and discomfort overwhelm any good intention to fit this consideration, however. Keaveney et al. 30 reported a paper where they assessed the reduced risk perception on one's own health in taking and even abusing with acetaminophen, despite its acknowledged danger. 30 , 38 However, the more general consideration about acetaminophen is that the antipyretic drug is harmless, probably because people consider that fever is very common.
Actually, the COVID‐19 pandemic registered a dramatic increase in the use of this drug. 39 A survey on subjects suffering from mild COVID‐19 and possibly requiring further acute and follow‐up care reported that acetaminophen held the 87.1% of responses gathered from providers recruited in the study. 40 The use of acetaminophen was even preferred respect to non‐steroidal anti‐inflammatory drugs (NSAIDs), to relieve pain, discomfort and fever due to early COVID‐19 symptoms. 41 Furthermore, the association between early pain in COVID‐19 and mortality was recently addressed by Knox et al., 42 who reported that acute pain during COVID‐19 was less associated with intensive care request and mortality risk than mild or absent pain.
While acetaminophen is widely and quite indiscriminately used (or abused) during the early COVID‐19 symptomatology, a more complex debate should be expanded about early stages in SARS‐CoV2 infection, because particularly crucial to prevent or not, further exacerbation and hospitalization in intensive care units (ICUs). During the ACE2 receptor invasion, SARS‐CoV2 begins to induce a marked dysregulation in the reactive oxygen species (ROS) level as signalling molecules, rapidly creating a marked oxidative stress. Native immune cells are involved in addressing this stress and counteracting the initial damage towards the fine cross‐talk endothelia platelets. A drug potentiating this damage may have a dramatic impact on exacerbating COVID‐19.
Actually, some reports showed the negative impact of paracetamol on endothelia, though referring fundamentally to paracetamol overdosage and drug‐caused hepatotoxicity. 43 , 44 Moreover, in the oxidative stress micro‐environment, glutathione (GSH) availability is particularly crucial when COVID‐19 is characterized by a significant reduction in GSH availability. 45 Glutathione biosynthesis is critical for the correct endothelia function and in anti‐fibrotic responses. 46 A close relationship between GSH homeostasis and fever has been reported recently, assessing that the role of GSH in the febrile response to inflammation and the role of fever on GSH availability and biosynthesis are closely intertwined. 47
In this context, questions arise about whether and how to use drugs to relieve fever during early COVID‐19 without consulting a physician.
How to check early COVID‐19 symptoms and promptly treat them?
An awkward terminology used by the Italian major Health Authorities, for example, is ‘watchful waiting’, usually alongside acetaminophen (paracetamol) recommendation. What does the patient have to wait for?
Recent data showed that acetaminophen induces a strong depletion of GSH in type II pneumocytes and alveolar macrophages. 48
As suggested before, some authors wondered if the use of antipyretics is really useful in managing COVID‐19 with respect to letting fever run. 37 , 47 , 49 To deepen this consideration and expand the debate about the usefulness in adopting the antipyretic strategy in early COVID‐19, a thorough understanding of COVID‐19 pathogenesis should be considered. Bonaventura et al. 5 highlighted the role of endothelia damage due to the SARS‐CoV2 infection, then causing coagulation disorders and an immuno‐thrombosis. Hence, COVID‐19 is not merely a simple airway inflammation sickness, like the flu and only severely targeting the lungs. Physicians are persuaded to reduce fever during early COVID‐19. Actually, past reports described an increase in fibrinogen due to fever, 50 and during SARS‐CoV2 infection, an increase in fibrinogen was actually reported. 5 The pro‐thrombotic mechanisms elicited by SARS‐CoV2 may be driven, at least initially, by platelets, which is thought to express ACE2 receptors on their membranes, 51 , 52 although an ACE2‐independent mechanism was also described. 53 A recent paper showed that a pre‐hospitalization anti‐platelet therapy greatly reduces the incidence of acute respiratory distress syndrome (ARDS), usually present in severe COVID‐19. 54
The potential noxious role of acetaminophen in this context might be particularly crucial.
Despite the past observation that paracetamol dose dependently inhibits platelet function, this action was exclusively reported for healthy subjects 55 and particularly in association with NSAIDs. 56 , 57 , 58 Probably, in the presence of a worsening endothelia dysfunction, as occurring during COVID‐19, the complex cross‐talk between endothelia and platelets is dramatically impaired and even the very modest COX‐2 inhibitory effect of acetaminophen may be insufficient to act as a friend, rather than a foe, against platelet targeting by SARS‐CoV2. 59 , 60 The SARS‐CoV2 spike (S) protein is the major cause of the endothelia damage in microcirculation. 5 This damage should prevent endothelia to counteract platelet aggregation via the production of the endothelial ecto‐ADP‐ase CD39, which by metabolizing the platelet activator ADP, prevents the ADP‐mediated platelet activation, besides the nitric oxide (NO) and prostacyclin release. 59
Past reports have elucidated that acetaminophen inhibits prostacyclin synthesis by endothelia, 61 an evidence that would emphasize the possible deleterious role of acetaminophen in activating circulating platelets. 61 Noticeably, the impairment in the endothelia function may reduce the actual potential of anti‐aggregating drugs such acetylsalicylic acid. 60 Therefore, endothelial impairment due to SARS‐CoV2 infection, once recruited the non‐classical endothelia‐associated monocytes, may promote the induction of an immuno‐thrombosis, where the inflammatory nature of the latter prolongs the febrile status and the subsequent abuse of acetaminophen. Interestingly, past reports, then confirmed, showed that acetaminophen at its usual oral dosage may inhibit prostacyclin release from endothelia even in pregnant women. 61 , 62 , 63 Acetaminophen inhibits prostacyclin synthesis in endothelia, via the scavenging of the activator of the prostaglandin endoperoxide H2 synthase, the peroxynitrite. 64
Briefly speaking, paracetamol should not be recommended even in association with NSAIDs. We cannot be ensured about the suggestion that acetaminophen to relieve pain and dampen fever in early COVID‐19 may exacerbate the inflammatory status of endothelia caused by SARS‐CoV2 infection, but evidence supports the idea that acetaminophen (i.e., paracetamol) may not be properly suited in the early COVID‐19 symptomatology. Moreover, even the prolonged use of acetaminophen may exacerbate this context, at least as observed in laboratory animals. During the induced acute liver failure by acetaminophen in mice, platelet‐microparticles (PMPs) expressing CD39 were generated. 65
PMPs were observed also during COVID‐19; they bear a CD39 marker than can disrupt the endothelia CD39‐mediated dampening of platelet‐induced thrombosis. 66 Despite COX‐1 inhibitors may paradoxically exacerbate the production of prostacyclin from endothelia, the COX‐2 inhibitor ibuprofen resulted very efficaciously in preventing the inhibition of endothelial prostacyclin, an evidence known for many years. 67 , 68 , 69 , 70 In this sense, recent clinical and observational studies have highlighted how the prevalent use of COX‐2 inhibitors, in the first line of therapy in early COVID‐19, with respect to acetaminophen, reduced significantly the rate of people undergoing hospitalization. 19
From a pharmacological point of view, some authors reported recently the ability of acetaminophen to diminish the availability of reduced glutathione (GSH), a circumstance that may exacerbate the impact of the drug on platelet functions. 9 , 70 , 71 , 72 , 73 , 74 , 75 GSH depletion is critical in the elderly, particularly if affected by age‐associated comorbidities, such as hypertension and even deep venous thrombosis, which may occur in severe COVID‐19, worsens GSH levels by enhancing glutathione peroxidase. 75 , 76 A recent in vitro study on VeroE6 cells reported that SARS‐CoV2 impairs the cellular redox balance by lowering cellular thiols, particularly glutathione (GSH). 77 Polonikov 78 reported that GSH deficiency is associated with severity and even death in COVID‐19. A possible mechanism is that SARS‐CoV2 hijacks serine metabolism in the mono‐carbon pathway to build up purines, so contributing in the reduction of a fundamental GSH precursor, the cystathionine. 79 Considering that acetaminophen inhibits glutathione reductase, the concurrence of SARS‐CoV2 infection, comorbidity in elderly and the prolonged use of acetaminophen in mild COVID‐19 may be particularly concerning for patients' health. 80 This leads us to conclude that prescribing paracetamol in the early stages of COVID‐19 should not be recommended.
3. NSAIDS AS THE FIRST CHOICE RECOMMENDED DRUGS IN THE COVID‐19 HOME THERAPY
3.1. Ibuprofen
Non‐steroidal anti‐inflammatory drugs (NSAIDs), usually known as cyclooxygenase 2 (COX‐2) inhibitors, have recently come to the spotlight as the first choice in addressing the early symptoms in COVID‐19. 19 Past research, yet confirmed to date, reported that ibuprofen, the propionic acid derivative known as RS)‐2‐(4‐[2‐methyl‐propyl]‐phenyl)‐propanoic acid, targets platelet function, 80 , 81 , 82 , 83 likewise aspirin. 84 In this context, ibuprofen, altogether with other NSAIDs, encompasses both an anti‐inflammatory and anti‐aggregating potential, particularly suited to address the early stage of COVID‐19 following a symptomatic SARS‐CoV2 infection, and also preventing the onset of a cytokine storm. 85 , 86 A recent paper by Valenzuela et al. 86 reported that ibuprofen upregulates ACE2 receptor expression and concomitantly the anti‐inflammatory renin‐angiotensin system (RAS) in the lungs. Criticism was raised in the past on NSAID use in COVID‐19, due to their role in up‐regulating ACE2 receptors, 87 , 88 , 89 although further data support the beneficial role of NSAIDs in COVID‐19. 90 , 91 , 92 Recent data disproved the prejudicial consideration about ibuprofen as a harmful therapeutic. 93
Ibuprofen promotes the RAS‐dependent anti‐inflammatory pathway, that is, the angiotensin 1–7 with the G‐coupled receptor Mas, and the pathway Ang 1–7/Mas/AT2, 94 , 95 whereas it inhibits the pro‐inflammatory axis made by the pathway AngII/AT1/NADPH oxidase), thus increasing ACE2 levels and reducing the activity of ADAM17. 96 , 97 , 98 Interestingly, it is well known that SARS‐CoV2 increases the activity of ADAM17, leading to the augmentation in ACE2 shedding, expression and membrane recycling, whereas ibuprofen blocks this activity and moreover lowers the activity of TMPRSS2, a well‐known trans‐membrane serine protease which activates viral Spike protein. 99
Ibuprofen activity is not limited to the inhibition of COX‐2. But this narrative review about the pharmacological action of NSAIDs such as ibuprofen on SARS‐CoV2 infection and ACE2 expression shows some apparent controversial issues. The impact of NSAIDs on ACE2 expression seems not so easy to foresee, 100 but probably, ACE2 is not the major target by NSAIDs action. The relationship between COX‐2 and ACE2, for example, is particularly complex during SARS‐CoV2 infection. As for instance, SARS‐CoV2 induces COX‐2 activity and increases COX‐2 expression, without affecting viral entry or viral replication. 9 , 101
Chen et al. 101 have recently demonstrated that the use of well‐known COX‐1 and COX‐2 inhibitors, that is, NSAIDs, such as ibuprofen and meloxicam, do not affect SARS‐CoV2 entry, therefore rejecting the previously forwarded criticism that, as NSAIDs enhance ACE2, they may increase susceptibility to viral infection. The activity of NSAIDs on COX‐2 may have further intriguing roles; for example, it is well known that an inverse relationship exists between ACE2 and COX‐2 in endothelia. 102 , 103 Moreover, it is well known that ibuprofen inhibits platelets aggregation. 104
At the same time, ibuprofen, as recently observed in the arginate form, can rescue the eNOS activity in the damaged endothelia and revert the tissue to its normal function, that is, saving the NO pathway while it is inhibiting COX‐2. 105 It is tempting to speculate that the intake of ibuprofen, while SARS‐CoV2 is attacking the endothelial ability in counteracting thrombosis via NO and prostacyclin, can arrest and dampen the damaging process. Also, the previously reported belief that COX‐2 inhibitors may promote thromboembolic events by their inhibitory action on vascular prostacyclin synthesis and poor effect on platelet thromboxane A2 production was completely refuted by more recent data, assessing that COX‐2 inhibitors, such as celecoxib, ibuprofen or diclofenac, particularly ibuprofen, cannot cause any thromboembolic event. 105 , 106 , 107 , 108
3.2. Other affordable NSAIDs
COX‐2‐inhibiting NSAIDs are able to target different mechanisms in COVID‐19.
Naproxen seems to exhibit a direct anti‐viral activity, as this NSAID suppresses viral replication and N‐oligomerization in VeroA6 in vitro cells. 109 Interestingly, the association of low doses acetyl‐salicylic acid (ASA) with naproxen leads to a significant reduction in the production of thromboxane B2, a direct metabolite of platelets' thromboxane A2. 110 Nimesulide is a powerful inhibitor of the BOAT1 subunit in SARS‐CoV2 receptor, namely, the plasma membrane transporter of aminoacids, known as SLC6A19, which is linked with ACE2 in a dimer and stabilizes the link with SARS‐CoV2 Spike protein: nimesulide, therefore, hampers viral entry into the cell. 111
Selective inhibitors of COX‐2, which are currently used in clinics, such as the sulphonamide celecoxib and the methylsulphone etoricoxib, have been reported as capable of reducing significantly COVID‐19 patients' hospitalization rate. 18 Many of these pharmaceuticals are endowed with an anti‐viral activity. 112
Indomethacin is another NSAID, which was effective against the first SARS coronavirus in the outbreak of more than 15 years ago. 113 Actually, Amici et al. 113 reported that indomethacin significantly reduced in vitro both SARS‐COV‐1 on monkey VERO6 cells and human lung epithelial A549 cells, and canine coronavirus, both in A72 canine cells and in vivo dogs. This drug is a powerful anti‐inflammatory agent that not selectively inhibits cyclooxygenase (COX)‐1 and ‐2 enzymes and has been mostly used to treat inflammatory conditions of the musculoskeletal system. Since indomethacin inhibits COX‐2 and viral protein synthesis, anti‐viral activities of indomethacin against different viruses have been previously explored, including Herpes virus 6, Cytomegalovirus, Hepatitis B virus and so forth. 114 , 115 , 116 More recently, Napolitano et al. 117 have reported that indomethacin downregulates ACE2 receptors by suppressing the gene in the ACE2 pathway, while previously other authors showed that indomethacin inhibits cathepsin L activity for fusion with host cells, and that no other anti‐inflammatory drug aside from indomethacin exhibited a role in the cathepsin activity. 118 Furthermore, a direct evidence of effectiveness of indomethacin against SARS‐CoV‐2 has been provided by Xu et al. 119 They have shown the anti‐viral effect of indomethacin in vitro and in vivo in an infected canine model.
Indomethacin also reduces the levels of interleukine‐6 (IL‐6) and tumour necrosis factor (TNF), cytokines that greatly increase during COVID‐19, and are responsible for some of its detrimental effects. 120 It has been also hypothesized that indomethacin can counteract bradykinin effects in COVID‐19 patients, thus possibly reducing COVID‐19‐induced dry cough or other bradykinin‐induced symptoms. 121 The use of indomethacin to relieve pain and discomfort has been compared to other NSAIDs even in past reports; for example, indomethacin has no anaesthetic properties with respect to diclofenac, 122 yet recent evidence recommends to better target COX‐2 and use panels of drugs against COVID‐19 including indomethacin, diclofenac and celecoxib. 123 A comparison between indomethacin and ibuprofen showed comparable efficacy and safety. 124
Alongside with indomethacin, diclofenac potassium also is a NSAID included in the silver line of anti‐inflammatory drugs against SARS‐CoV2 infection. 123 Recent reports showed that diclofenac potassium increases lymphocyte recruitment for the immune response against SARS‐CoV2. 125
The correct selection of NSAIDs for treating patients with early COVID‐19 symptoms may be particularly burdensome, as some very common NSAIDs, such as ibuprofen and meloxicam, seem to have no effect on ACE2 expression, viral entry and replication. 126 The correct choice of therapeutic drugs to be used in the early COVID‐19 symptomatology needs to be refreshed with new clinical studies.
Ravichandran et al. 127 have recently reported a clinical study, comparing 72 COVID‐19 patients treated with indomethacin with 109 COVID‐19 patients treated with paracetamol (indomethacin and paracetamol were both given along with standard therapy). The authors concluded that the symptomatic relief in the cohort of indomethacin patients was remarkably higher than the paracetamol group, and that only one of 72 patients in the indomethacin group required supplementary oxygen as compared to 28 patients in the paracetamol patient cohort group. 127
Moreover, an author is absolutely persuaded that the early use of NSAIDs, particularly regarding ibuprofen, might revert COVID‐19 pathogenesis, prevent exacerbation and improve clinical outcomes. 125 The route in which these drugs should operate encompasses the early phases of COVID‐19 pathogenesis, therefore inflammation and immuno‐thrombosis. In this first stage, inflammation is occurring in the patient, as fever is one of its major symptoms.
Commonly widespread anti‐inflammatory drugs were not widely considered in COVID‐19 pharmacotherapy, probably because of misleading communication about medical expertise and practice. 128 Dampening inflammation, which most probably is affecting the endothelia‐platelet cross talk in microcirculation, is a leading target of early therapy against COVID‐19. Therefore, more than dampening fever is of the utmost importance to reducing the patient's inflammatory response to SARS‐CoV2, in order to lower the possible damage on endothelia. In this context, for example, ibuprofen reduces the levels of IL‐6 and IL‐8. 129 The anti‐inflammatory activity of NSAIDs reduces also fever, so a patient in the early stages of COVID‐19 can simply take up NSAIDs rather than acetaminophen, to relieve pain and discomfort due to fever.
The activity of NSAIDs on inflammation an endothelial function should be the leading cause of NSAIDs effectiveness in COVID‐19. 130
Past reports showed that nimesulide is able to block neutrophils diapedesis through TNF‐α‐activated endothelia, thus switching inflammation off and dampening neutrophils‐mediated pro‐oxidative injury. 131 Celecoxib is able to directly target SARS‐CoV2 and block its activity, 112 an evidence that can explain why patients using celecoxib and other NSAIDs more than acetaminophen in the early symptoms of COVID‐19, had the lowest risk of being hospitalized. 18 , 132 The use of NSAIDs in home therapy against COVID‐19 was not so widespread and counselled as due, in order to prevent disease worsening. The panel of NSAIDs is particularly enriched of novel therapeutic drugs, which could successfully address COVID‐19 in its early phases. If endothelia are rapidly targeted by SARS‐CoV2, generating an inflammatory response in microcirculation, a therapy able to modulate inflammation, endothelial dysfunction and pro‐oxidant stress, is particularly suitable to address COVID‐19 at the earliest.
3.3. Acetyl‐salicylic acid (ASA)
The recent evidence about the endothelial involvement in COVID‐19 has promoted the medical counselling towards anti‐aggregating drugs in the early stages of COVID‐19. 133
Some authors have also suggested dypiridamole, as an anti‐platelet drug, but its use is highly concerning and never entred the routinely and conventional therapy protocols in early COVID‐19. 134 So far, the consideration that COVID‐19 is prevalently an endothelial dysfunction with inflammation, enabled in promoting an immuno‐thrombosis, ASA is not so thoroughly included in the therapy panels recommended for early or mild COVID‐19, as physicians are particularly cautious in addressing drugs targeting the coagulation/thrombotic mechanisms. The overall agreed consideration is that COVID‐19 is an inflammatory illness, for which anti‐inflammatory drugs are properly suited.
ASA is probably the most common anti‐aggregating pharmaceutical drug. A recent paper by Liu et al. 135 reported that when treating COVID‐19 patients with acetyl‐salicylate, mortality dropped down significantly and that patients with metabolic comorbidities (cardiovascular disease and hypertension) infected with SARS‐CoV2, when treated with 100 mg/day aspirin reduced their risk of mortality. In this circumstance, the authors are talking about a preventive use of aspirin against the thrombus‐embolic risk associated with COVID‐19. 135 A decrease in mortality was also observed by Osborne et al., 136 in a study from the US Veterans Health Administration. The authors performed a retrospective study on a great number of veterans from across the country, having resulted positive to SARS‐CoV2 and included 35,370 patients in the period of 2 March 2020 to 13 September 2020 in the 14‐day mortality cohort, and 32,836 patients were included from 2 March 2020 to 28 August 2020, in the 30‐day mortality cohort. Their results showed that previous aspirin prescription reduced significantly the overall mortality at 14 days (OR 0.38, 95% CI 0.32–0.46) and at 30 days (OR 0.38, 95% CI 0.33–0.45). 136
However, an interesting meta‐analysis performed by Martha et al. 137 reported much more cautious results. The authors, following the inclusion of only six eligible studies, encompassing about 13 993 patients, concluded that the use of low‐doses aspirin was only independently associated with a reduced mortality (RR 0.46 [95% confidence interval (CI) 0.35–0.61], P < 0.001; I 2 = 36.2%). No practitioner or physician would like to prescribe the sole aspirin to address the early symptoms of COVID‐19, yet aspirin might be useful in reducing the pro‐thrombotic impact of SARS‐CoV2 infection. The recent meta‐analysis performed by the Department of Pharmacology and Clinical Research, Delhi Institute of Pharmaceutical Sciences and Research (DIPSAR), New Delhi, India, reported an odd ratio (OR) = 0.70 (0.68–0.77), that is, a lesser probability to die for COVID‐19 in patients having taken aspirin respect to the control, 138 although this evidence may be criticized by the existence of outliers. The reliability of papers selected for the meta‐analysis was assessed by the Newcastle‐Ottawa scale. In COVID‐19 development, platelets should play a major role. 139 However, a paper reported that previous treatment with anti‐platelet drugs did not reduce COVID‐19 severity. 140 Actually, COVID‐19 pathology is not a direct cause of platelet dysfunction but of the endothelia‐platelets cross talk strongly impaired by immuno‐inflammatory causes. Therefore, in our opinion, a simple therapeutic drug with both anti‐inflammatory and anti‐aggregating properties may be suitable to address COVID‐10 in its early symptoms.
4. ANCILLARY AND SUPPORTING THERAPY IN THE EARLY COVID‐19 HOME TREATMENT
Attempts in improving home therapy against COVID‐19 have evaluated also the introduction of micronutrients and plant‐derived polyphenolic substances, usually as nutraceuticals, to enhance the anti‐oxidant and anti‐inflammatory activity of conventional drugs. In this perspective, anti‐oxidant molecules, most of which coming from natural vitamins or flavonoids, may help NSAIDs in acting much more effectively against SARS‐CoV2‐caused damage. Actually, flavonoids and nutraceuticals cannot be used as pharmaceuticals, yet. 141
The reduction in glutathione (GSH), caused by SARS‐CoV2 infection, 77 should be addressed. Liver usually produces glutathione but a well‐ruled dietary regimen may give its fundamental contribution. 142 Several reports have also outlined the possible role of L‐ascorbate. 14 , 143 Obviously, vitamin C is not a therapy; molecules able to scavenge reactive oxygen species (ROS), such as L‐ascorbate or other agents with anti‐oxidant properties, 144 , 145 might even contribute in notably reducing the impact of COVID‐19 on endothelia and immuno‐thrombotic mechanisms.
Anyway, anti‐oxidants can be particularly helpful in the first 3 days upon early COVID‐19 symptoms along with anti‐inflammatory drugs such as NSAIDs, as they can act as cofactors in the ability of NSAIDs to counteract pro‐thrombotic mechanisms. 146 , 147 , 148 , 149 , 150
In the first stage (see further on), NSAIDs can be associated with vitamins and anti‐oxidants, such as ascorbate (500 mg/day), vitamin D3 (10.000 UI/day), vitamin K2 (200 μg/day), quercetin (300 mg/day) or other flavonoids, such as hesperidin (500 mg/day), lactoferrin (300–400 mg/day), resveratrol (500–1,000 mg/day) and even supplementation with Zn (30 mg/day).
Furthermore, some pharmaceuticals, used to relieve COVID‐19‐associated symptoms such as cough, may even contribute in ameliorating the performance of NSAIDs, despite some controversial results.
A pilot study of intravenous N‐acetyl‐cysteine (NAC) reported negative results in severe COVID‐19 patients, as intravenous NAC did not reach a statistical difference with respect to not treated subjects. 143
Despite some encouraging claim about NAC, 151 , 152 , 153 the novelty should prevalently focus on the use of NAC during the early stages of COVID‐19 or lately as a preventive therapy, rather than a curative approach. NAC reduces the oxidative stress causing endothelial injury via the complement activation and apoptosis. 154 In this sense, this compound should be particularly useful in a therapeutic formula uptaken during early symptoms in COVID‐19. In this perspective, cough syrups might have an intriguing role in addressing SARS‐CoV2 infection and its exacerbation. 125 , 155 Anyway, NAC remains as a coadjutant factor in the conventional therapeutic protocol for treating COVID‐19 in its early symptomatology.
According to the most recent evidence, SARS‐CoV2 infection should rapidly affect endothelia and platelets, sequestering from circulating blood non‐classical endothelia‐associated monocytes, which should scavenge pro‐oxidant stressors from promoting pro‐thrombotic events. However, NAC is not an elective therapy against COVID‐19, but a possible adjuvant.
Further on, we will address the role of anti‐oxidant compounds as adjuvants alongside therapeutic anti‐inflammatory drugs and anti‐microbials, such as azithromycin. To date, acetyl salicylic acid seems to work better as a preventive agent, against COVID‐19 exacerbation.
5. TOWARDS A PHARMACOLOGICAL PROPOSAL FOR EARLY COVID‐19 SYMPTOMS
To date, no official proposal reached the attention of leading authorities, as COVID‐19 home therapy would be based on affordable and widely available anti‐inflammatory and anti‐thrombotic drugs to reduce the impact on any socio‐economic burden and reduce hospitalization overload. Recent clinical data, despite someone yet published as a pre‐print, assessed that COX‐2 inhibitors, either commonly purchasable NSAIDs and selective inhibitors, are a better choice to treat COVID‐19 in its early symptomatology. 19 , 156 Pharmaceuticals such as nimesulide and celecoxib are strongly suggested to greatly reduce the risk of being hospitalized for the patient with early COVID‐19 symptoms. 19 , 156 The proposal should be exerted usually at home, as in the first stages of COVID‐19 symptomatology the patient feels pain, discomfort, even cough or dyspnoea and fever while staying at her/his own home. Major recommended NSAIDs may be therefore celecoxib, nimesulide, ibuprofen and indomethacin, 156 taking into account that any protocol considering one of these NSAIDs might include ASA (>44%) and antibiotics such as azithromycin (>48%). 156 However, using ASA alongside with NSAIDs might cause adverse effects, already described in the literature. 157
The suggestion comes from observing the different ability of these. Therefore, the therapeutic proposal herein forwarded is a ‘home therapy proposal’ to treat SARS‐CoV2 infection in its earliest onset (within 72 h) and therefore prevent hospitalization. Noteworthy, besides vaccination, early home therapy against COVID‐19 should represent the second major pillar of the political and institutional response to COVID‐19 emergency.
Figure 1 shows how the current available therapy drugs, even in home COVID‐19 treatment, can hamper and inhibit SARS‐CoV2 spreading in the organism and COVID‐19 pathogenesis.
FIGURE 1.
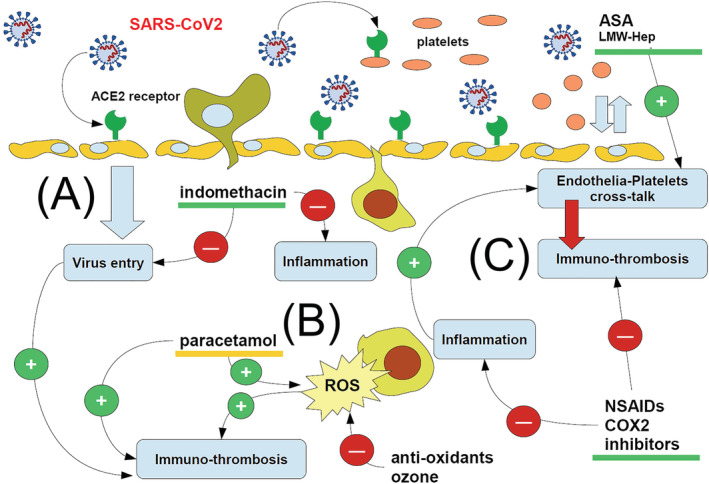
Cartoon showing the major steps through which available therapeutic drugs may address SARS‐CoV2 infection and hence COVID‐19. A first line, which is rapidly replaced by anti‐COX‐2 NSAIDs, is the use of anti‐aggregation drugs, such as acetyl salicylic acid (ASA) and low‐molecular weight heparin (LMW‐Hep), to rescue the correct endothelia‐platelets cross talk impaired by early SARS‐CoV2 infection. This might even be associated with drugs particularly targeting viral entry (such as indomethacin) (A) and inhibiting inflammation. The onset of a huge oxidative stress, with production of reactive oxygen species (ROS), is the major hallmark of innate immune cell activity and of inflammation (B); therefore, the early inhibition of inflammation leads to the reduction of those mechanisms leading to immuno‐thrombosis, where NSAIDs exert their major activity (C). While most of drugs in the cartoon (green underlined) are useful to hamper immuno‐thrombosis, paracetamol (orange underlined) increases the oxidative stress, so allowing inflammation an immuno‐thrombosis to go ahead and exacerbating COVID‐19 progress. Red circles: inhibition; green circles: promotion
Fundamentally, we can distinguish between three major phases in the COVID‐19 pathogenetic development: (A) Phase 1 (early infection): Symptoms are flu‐like with a prevalence of low‐grade fever or a frank fever, headache, arthralgia and myalgia. (B) Phase 2 (pulmonary phase): The symptoms mainly refer to the respiratory tract with dyspnoea, polypnoea, chest pain and mild desaturation; (C) Phase 3 (systemic inflammation with thromboembolic and immune‐thrombotic events): The symptoms refer to multi‐organ inflammatory involvement with thrombo‐embolism from vasculitis to deep venous thrombosis and possible disseminated thrombosis. Although this classification serves only as a simplistic way to show how COVID‐19 usually develops in clinics, representing an easy synopsis, therapy must be undertaken at the earliest, at least within the first 3 days of symptom appearance, even prior of any swabbing delay, if the patient has a history of previous contact. The viral attack on the endothelial‐platelet‐coagulation system is immediate, and the symptomatology in phase 3 is only the exacerbation of a progressing damage in the microcirculation. 5
During Phase 1, NSAIDs such as ibuprofen, nimesulide and celecoxib 19 but also indomethacin and acetyl salicylic acid may be easily considered elective therapeutic drugs. These drugs must be easy to be provided and should be administered within 72 h from early symptoms and never in associated forms.
Much more caution should be paid towards the use of low molecular weight heparin (LMW‐Hep). 158 Usually, LMW‐Hep, as well as other major anti‐coagulant therapy, is prescribed during hospitalization 158 ; the use of LMW‐Hep is not currently recommended in addressing early COVID‐19 symptoms, that is, mild and pauci‐symptomatic subjects, due to the many critical issues raised when the platelet‐coagulation system is targeted. 159
Such a protocol, which completely replaces acetaminophen with NSAIDs, has proven to reduce dramatically the hospitalization rate with respect to paracetamol only. Considering that hospitalization may exacerbate the clinical status even in hospital‐acquired infections, 160 this should lead to a reduced mortality particularly in elderly patients with early COVID‐19 symptoms.
6. CONCLUSION
Addressing home therapy against COVID‐19 is a crucial and very urgent topic in this pandemic emergency, which may be prolonged by the SARS‐CoV2 delta and South Africa variants. Hospitalization represents a huge burden for the community of citizens, both from economic and social perspectives. Therefore, a proper policy of physicians visiting patients in their home in order to take care of them is preferable.
In this review, we collected major evidence regarding the use of very common therapeutic drugs against COVID‐19, promptly available and fully enabled in addressing a mild COVID‐19 or its early symptomatic manifestations. Hospitalization may even exacerbate the progress of the coronavirus sickness, due to hospital‐acquired infections, crowding and presence of people with severe forms of COVID‐19. In Italy, a civil association of citizens and professionals joined the proposal to sensitize politicians and healthcare workers on this great concern and thoroughly revise the Italian Health Authorities' protocols, currently counselling a home therapy highly debatable and controversial, due to the paracetamol issue. Therapy against COVID‐19 does exist, is promptly available and is easy to manage even at home, and the knowledge of it must only be widespread among professionals to make them fully aware of this issue.
CONFLICT OF INTEREST
The Authors state they have no conflict of interest.
Pandolfi S, Chirumbolo S, Ricevuti G, et al. Home pharmacological therapy in early COVID‐19 to prevent hospitalization and reduce mortality: Time for a suitable proposal. Basic Clin Pharmacol Toxicol. 2022;130(2):225-239. doi: 10.1111/bcpt.13690
REFERENCES
- 1. D'Amato G, Acanfora L, Delli Paoli L, D'Amato M. Preventive home therapy for symptomatic patients affected by COVID‐19 and followed by teleconsultations. Multidiscip Respir Med. 2021;16(1):748. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Adiletta G, Baglioni S, Bettoncelli G, et al. Comments on “Preventive home therapy for symptomatic patients affected by COVID‐19 and followed by teleconsultations” by D'Amato et al. Multidiscip Respir Med. 2021;16(1):757. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Cazzola M, de Novellis V, Bianco A, Rogliani P, Matera MG. Disputes over the production and dissemination of misinformation in the time of COVID‐19. Respir Med. 2021;182:106380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Donno DR, Grattagliano I, Rossi A, et al. How to treat COVID‐19 patients at home in the Italian context: an expert opinion. Infect Dis Rep. 2021;13(1):251‐258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Bonaventura A, Vecchié A, Dagna L, et al. Endothelial dysfunction and immunothrombosis as key pathogenic mechanisms in COVID‐19. Nat Rev Immunol. 2021;21(5):319‐329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Albasheer OB, Almutairi HA, Almalki FM, Malaka HM. Massive arterial cerebral thrombosis in a 59‐year old female patient with severe COVID‐19. Cureus. 2021;13(6):e15553. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Ignacio‐Ibarra G, García‐Lee MT, González‐Ávila AI, et al. A proposal for the management of COVID‐19‐induced coagulopathy in adults. Gac Med Mex. 2021;157(2):201‐206. [DOI] [PubMed] [Google Scholar]
- 8. Semeraro N, Colucci M. The prothrombotic state associated with SARS‐CoV‐2 infection: pathophysiological aspects. Mediterr J Hematol Infect Dis. 2021;13(1):e2021045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Pandolfi S, Chirumbolo S. Home therapy of COVID‐19 at the earliest may greatly prevent hospitalization. Basic Clin Pharmacol Toxicol. 2021;129(6):395‐396. 10.1111/bcpt.13650 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Galati D, Zanotta S, Capitelli L, Bocchino M. A bird's eye view on the role of dendritic cells in SARS‐CoV‐2 infection: perspectives for immune‐based vaccines. Allergy. 2021. 10.1111/all.15004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Sepehrinezhad A, Gorji A, Sahab Negah S. SARS‐CoV‐2 may trigger inflammasome and pyroptosis in the central nervous system: a mechanistic view of neurotropism. Inflammopharmacology. 2021;29(4):1049‐1059. 10.1007/s10787-021-00845-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Yang L, Xie X, Tu Z, Fu J, Xu D, Zhou Y. The signal pathways and treatment of cytokine storm in COVID‐19. Signal Transduct Target Ther. 2021;6(1):255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Cipolloni L, Sessa F, Bertozzi G, et al. Preliminary post‐mortem COVID‐19 evidence of endothelial injury and factor VIII hyperexpression. Diagnostics (Basel). 2020;10(8):575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Huang L, Wang L, Tan J, Liu H, Ni Y. High‐dose vitamin C intravenous infusion in the treatment of patients with COVID‐19: A protocol for systematic review and meta‐analysis. Medicine (Baltimore). 2021;100(19):e25876. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Dwiputra Hernugrahanto K, Novembri Utomo D, Hariman H, et al. Thromboembolic involvement and its possible pathogenesis in COVID‐19 mortality: lesson from post‐mortem reports. Eur Rev Med Pharmacol Sci. 2021;25(3):1670‐1679. [DOI] [PubMed] [Google Scholar]
- 16. Elezkurtaj S, Greuel S, Ihlow J, et al. Causes of death and comorbidities in hospitalized patients with COVID‐19. Sci Rep. 2021;11(1):4263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Pandolfi S, Simonetti V, Ricevuti G, Chirumbolo S. Paracetamol in the home treatment of early COVID‐19 symptoms: a possible foe rather than a friend for elderly patients? J Med Virol. 2021;93(10):5704‐5706. 10.1002/jmv.27158 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Hashemi SA, Kyani A, Bathaie SZ. The in silico mechanism of hVKOR interaction with acetaminophen and its metabolite, as well as N‐acetyl cysteine: caution on application in COVID‐19 patients. J Biomol Struct Dyn Apr. 2021;20:1‐12. 10.1080/07391102.2021.1910570 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Suter F, Consolaro E, Pedroni S, et al. A simple, home‐therapy algorithm to prevent hospitalisation for COVID‐19 patients: A retrospective observational matched‐cohort study. EClinicalMedicine. 2021;37:100941. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Reese JT, Coleman B, Chan L, Blau H, Callahan TJ, Cappelletti L, Fontana T, Bradwell KR, Harris NL, Casiraghi E, Valentini G, Karlebach G, Deer R, McMurry JA, Haendel MA, Chute CG, Pfaff E, Moffitt R, Spratt H, Singh J, Mungall CJ, Williams AE, Robinson PN. Cyclooxygenase inhibitor use is associated with increased COVID‐19 severity. 2021. medRxiv [Preprint]. Apr 20:2021.04.13.21255438.
- 21. Wastesson JW, Martikainen JE, Zoëga H, Schmidt M, Karlstad Ø, Pottegård A. Trends in use of paracetamol in the Nordic countries. Basic Clin Pharmacol Toxicol. 2018;123(3):301‐307. [DOI] [PubMed] [Google Scholar]
- 22. Morthorst BR, Erlangsen A, Nordentoft M, Hawton K, Hoegberg LCG, Dalhoff KP. Availability of paracetamol sold over the counter in Europe: a descriptive cross‐sectional international survey of pack size restriction. Basic Clin Pharmacol Toxicol. 2018;122(6):643‐649. [DOI] [PubMed] [Google Scholar]
- 23. Romano S, Galante H, Figueira D, Mendes Z, Rodrigues AT. Time‐trend analysis of medicine sales and shortages during COVID‐19 outbreak: data from community pharmacies. Res Social Adm Pharm. 2021;17(1):1876‐1881. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Lu X, Zhang L, Du H, et al. SARS‐CoV‐2 infection in children. N Engl J Med. 2020;382(17):1663‐1665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Guan WJ, Ni ZY, Hu Y, et al. Clinical characteristics of coronavirus disease 2019 in China. N Engl J Med. 2020;382(18):1708‐1720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Lechien JR, Chiesa‐Estomba CM, Place S, et al. Clinical and epidemiological characteristics of 1420 European patients with mild‐to‐moderate coronavirus disease 2019. J Intern Med. 2020;288(3):335‐344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Duong M, Gulmez SE, Salvo F, et al. Usage patterns of paracetamol in France. Br J Clin Pharmacol. 2016;82(2):498‐503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Diener HC, Schneider R, Aicher B. Per‐capita consumption of analgesics: a nine‐country survey over 20 years. J Headache Pain. 2008;9(4):225‐231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Shader RI. Acetaminophen (paracetamol), COVID‐19, and misleading conclusions: a commentary. J Clin Psychopharmacol. 2021;41(2):98‐99. [DOI] [PubMed] [Google Scholar]
- 30. Keaveney A, Peters E, Way B. Effects of acetaminophen on risk taking. Soc Cogn Affect Neurosci. 2020;15(7):725‐732. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Rodríguez‐Morales AJ, Cardona‐Ospina JA, Murillo‐Muñoz MM. Gastroenterologists, hepatologists, COVID‐19 and the use of acetaminophen. Clin Gastroenterol Hepatol. 2020;18(9):2142‐2143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Peluso L, Abella BS, Ferrer R, Kucher N, Sunde K, Taccone FS. Fever management in COVID‐19 patients. Minerva Anestesiol. 2021;87(1):1‐3. [DOI] [PubMed] [Google Scholar]
- 33. Robertson JA, Sauer D, Gold JA, Nonas SA. The role of cyclooxygenase‐2 in mechanical ventilation‐induced lung injury. Am J Respir Cell Mol Biol. 2012;47(3):387‐394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Zand Irani A, Borchert G, Craven B, Gibbons H. Flucloxacillin and paracetamol induced pyroglutamic acidosis. BMJ Case Rep. 2021;14(1):e237536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Hitzing S, Böttcher A, Laube M. Metabolische azidose unter paracetamol—eine ungewöhnliche Nebenwirkung [Metabolic acidosis under acetaminophen intake—an unordinary side effect]. Anasthesiol Intensivmed Notfallmed Schmerzther. 2018;53(10):718‐722. [DOI] [PubMed] [Google Scholar]
- 36. Mendoza CD, Heard K, Dart RC. Coma, metabolic acidosis and normal liver function in a child with a large serum acetaminophen level. Ann Emerg Med. 2006;48(5):637. [DOI] [PubMed] [Google Scholar]
- 37. Steiner AA. Should we let fever run its course in the early stages of COVID‐19? J R Soc Med. 2020;113(10):407‐409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Hodgman MJ, Garrard AR. A review of acetaminophen poisoning. Crit Care Clin. 2012;28(4):499‐516. [DOI] [PubMed] [Google Scholar]
- 39. Ortiz‐Prado E, Fernandez‐Naranjo R, Torres‐Berru Y, Lowe R, Torres I. Exceptional prices of medical and other supplies during the COVID‐19 pandemic in Ecuador. Am J Trop Med Hyg. 2021;1(aop):81‐87. 10.4269/ajtmh.21-0221 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. O'Keefe JB, Newsom LC, Taylor TH Jr. A survey of provider‐reported use and perceived effectiveness of medications for symptom management in telemedicine and outpatient visits for mild COVID‐19. Infect Dis Ther. 2021;10(2):839‐851. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Yousefifard M, Zali A, Zarghi A, Madani Neishaboori A, Hosseini M, Safari S. Non‐steroidal anti‐inflammatory drugs in management of COVID‐19; a systematic review on current evidence. Int J Clin Pract. 2020;74(9):e13557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Knox N, Lee CS, Moon JY, Cohen SP. Pain manifestations of COVID‐19 and their association with mortality: a multicenter prospective observational study. Mayo Clin Proc. 2021;96(4):943‐951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Williams AM, Langley PG, Osei‐Hwediah J, Wendon JA, Hughes RD. Hyaluronic acid and endothelial damage due to paracetamol‐induced hepatotoxicity. Liver Int. 2003;23(2):110‐115. [DOI] [PubMed] [Google Scholar]
- 44. Badmann A, Langsch S, Keogh A, Brunner T, Kaufmann T, Corazza N. TRAIL enhances paracetamol‐induced liver sinusoidal endothelial cell death in a Bim‐ and Bid‐dependent manner. Cell Death Dis. 2012;3(12):e447. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Khanfar A, Al Qaroot B. Could glutathione depletion be the Trojan horse of COVID‐19 mortality? Eur Rev Med Pharmacol Sci. 2020;24(23):12500‐12509. [DOI] [PubMed] [Google Scholar]
- 46. Espinosa‐Díez C, Miguel V, Vallejo S, et al. Role of glutathione biosynthesis in endothelial dysfunction and fibrosis. Redox Biol. 2018;14:88‐99. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Wrotek S, Sobocińska J, Kozłowski HM, Pawlikowska M, Jędrzejewski T, Dzialuk A. New insights into the role of glutathione in the mechanism of fever. Int J Mol Sci. 2020;21(4):1393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Dimova S, Hoet PH, Dinsdale D, Nemery B. Acetaminophen decreases intracellular glutathione levels and modulates cytokine production in human alveolar macrophages and type II pneumocytes in vitro. Int J Biochem Cell Biol. 2005;37(8):1727‐1737. [DOI] [PubMed] [Google Scholar]
- 49. Geddes L. The fever paradox. New Sci. 2020;246(3277):39‐41. 10.1016/S0262-4079(20)30731-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Egeberg O. The effect of unspecific fever induction on the blood clotting system. Scand J Clin Lab Invest. 1962;14(5):471‐474. [Google Scholar]
- 51. Zhang J, Huang X, Ding D, Tao Z. Platelet‐driven coagulopathy in COVID‐19 patients: in comparison to seasonal influenza cases. Exp Hematol Oncol. 2021a;10(1):34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Campbell RA, Boilard E, Rondina MT. Is there a role for the ACE2 receptor in SARS‐CoV‐2 interactions with platelets? J Thromb Haemost. 2021;19(1):46‐50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Shen S, Zhang J, Fang Y, et al. SARS‐CoV‐2 interacts with platelets and megakaryocytes via ACE2‐independent mechanism. J Hematol Oncol. 2021;14(1):72. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Erlich JM, Talmor DS, Cartin‐Ceba R, Gajic O, Kor DJ. Prehospitalization antiplatelet therapy is associated with a reduced incidence of acute lung injury: a population‐based cohort study. Chest. 2011;139(2):289‐295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Munsterhjelm E, Munsterhjelm NM, Niemi TT, Ylikorkala O, Neuvonen PJ, Rosenberg PH. Dose‐dependent inhibition of platelet function by acetaminophen in healthy volunteers. Anesthesiology. 2005;103(4):712‐717. [DOI] [PubMed] [Google Scholar]
- 56. Munsterhjelm E, Niemi TT, Ylikorkala O, Silvanto M, Rosenberg PH. Characterization of inhibition of platelet function by paracetamol and its interaction with diclofenac in vitro. Acta Anaesthesiol Scand. 2005b;49(6):840‐846. [DOI] [PubMed] [Google Scholar]
- 57. Galliard‐Grigioni KS, Fehr M, Reinhart WH. Influence of combinations of acetylsalicylic acid, acetaminophen, and diclofenac on platelet aggregation. Eur J Pharmacol. 2008;595(1–3):65‐68. [DOI] [PubMed] [Google Scholar]
- 58. Hinz B, Cheremina O, Brune K. Acetaminophen (paracetamol) is a selective cyclooxygenase‐2 inhibitor in man. FASEB j. 2008;22(2):383‐390. [DOI] [PubMed] [Google Scholar]
- 59. Hamilos M, Petousis S, Parthenakis F. Interaction between platelets and endothelium: from pathophysiology to new therapeutic options. Cardiovasc Diagn Ther. 2018;8(5):568‐580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60. Sahai A, Bhandari R, Koupenova M, Freedman J, Godwin M, McIntyre T, Chung M, Iskandar JP, Kamran H, Aggarwal A, Kalra A, Bartholomew J, McCrae K, Elbadawi A, Svensson L, Kapadia S, Hariri E, Cameron S. SARS‐CoV‐2 receptors are expressed on human platelets and the effect of aspirin on clinical outcomes in COVID‐19 patients. 2020. Res Sq [Preprint]. Dec 23:rs.3.rs‐119031
- 61. Grèen K, Drvota V, Vesterqvist O. Pronounced reduction of in vivo prostacyclin synthesis in humans by acetaminophen (paracetamol). Prostaglandins. 1989;37(3):311‐315. [DOI] [PubMed] [Google Scholar]
- 62. O'Brien WF, Krammer J, O'Leary TD, Mastrogiannis DS. The effect of acetaminophen on prostacyclin production in pregnant women. Am J Obstet Gynecol. 1993;168(4):1164‐1169. [DOI] [PubMed] [Google Scholar]
- 63. Schwartz JI, Musser BJ, Tanaka WK, et al. Inhibition of prostacyclin and thromboxane biosynthesis in healthy volunteers by single and multiple doses of acetaminophen and indomethacin. Clin Pharmacol Drug Dev. 2015;4(5):337‐345. [DOI] [PubMed] [Google Scholar]
- 64. Schildknecht S, Daiber A, Ghisla S, Cohen RA, Bachschmid MM. Acetaminophen inhibits prostanoid synthesis by scavenging the PGHS‐activator peroxynitrite. FASEB J. 2008;22(1):215‐224. [DOI] [PubMed] [Google Scholar]
- 65. Schmelzle M, Splith K, Andersen LW, et al. Increased plasma levels of microparticles expressing CD39 and CD133 in acute liver injury. Transplantation. 2013;95(1):63‐69. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66. Zahran AM, El‐Badawy O, Ali WA, Mahran ZG, Mahran EEMO, Rayan A. Circulating microparticles and activated platelets as novel prognostic biomarkers in COVID‐19; relation to cancer. PLoS ONE. 2021;16(2):e0246806. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67. Czervionke RL, Smith JB, Fry GL, Hoak JC, Haycraft DL. Inhibition of prostacyclin by treatment of endothelium with aspirin. Correlation with platelet adherence. J Clin Invest. 1979;63(5):1089‐1092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68. Parks WM, Hoak JC, Czervionke RL. Comparative effect of ibuprofen on endothelial and platelet prostaglandin synthesis. J Pharmacol Exp Ther. 1981;219(2):415‐419. [PubMed] [Google Scholar]
- 69. Wessels DA, Hempel SL. Ibuprofen protects human endothelial cell prostaglandin H synthase from hydrogen peroxide. Am J Physiol. 1996;271(6 Pt 1):C1879‐C1886. [DOI] [PubMed] [Google Scholar]
- 70. Gorudko IV, Shamova EV, Shishlo LM, et al. Glutathione‐dependent regulation of platelet aggregation with neutrophils and tumor cells. Biofizika. 2012;57(1):93‐98. [PubMed] [Google Scholar]
- 71. Essex DW, Li M. Redox control of platelet aggregation. Biochemistry. 2003;42(1):129‐136. [DOI] [PubMed] [Google Scholar]
- 72. Essex DW, Li M, Feinman RD, Miller A. Platelet surface glutathione reductase‐like activity. Blood. 2004;104(5):1383‐1385. [DOI] [PubMed] [Google Scholar]
- 73. Ball C, Vijayan KV, Nguyen T, et al. Glutathione regulates integrin alpha (IIb)beta(3)‐mediated cell adhesion under flow conditions. Thromb Haemost. 2008;100(5):857‐863. [PubMed] [Google Scholar]
- 74. Kozer E, Evans S, Barr J, et al. Glutathione, glutathione‐dependent enzymes and antioxidant status in erythrocytes from children treated with high‐dose paracetamol. Br J Clin Pharmacol. 2003;55(3):234‐240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75. Ekim M, Sekeroglu MR, Balahoroglu R, Ozkol H, Ekim H. Roles of the oxidative stress and ADMA in the development of deep venous thrombosis. Biochem Res Int. 2014;2014:703128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76. Rybka J, Kupczyk D, Kędziora‐Kornatowska K, et al. Glutathione‐related antioxidant defense system in elderly patients treated for hypertension. Cardiovasc Toxicol. 2011;11(1):1‐9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77. Bartolini D, Stabile AM, Bastianelli S, et al. SARS‐CoV2 infection impairs the metabolism and redox function of cellular glutathione. Redox Biol. 2021;45:102041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78. Polonikov A. Endogenous deficiency of glutathione as the most likely cause of serious manifestations and death in COVID‐19 patients. ACS Infect Dis. 2020;6(7):1558‐1562. [DOI] [PubMed] [Google Scholar]
- 79. Zhang Y, Guo R, Kim SH, et al. SARS‐CoV‐2 hijacks folate and one‐carbon metabolism for viral replication. Nat Commun. 2021b;12(1):1676. 10.1038/s41467-021-21903-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80. Rousar T, Pařík P, Kucera O, Bartos M, Červinková Z. Glutathione reductase is inhibited by acetaminophen‐glutathione conjugate in vitro. Physiol Res. 2010;59(2):225‐232. [DOI] [PubMed] [Google Scholar]
- 81. McIntyre BA, Philp RB, Inwood MJ. Effect of ibuprofen on platelet function in normal subjects and hemophiliac patients. Clin Pharmacol Ther. 1978;24(5):616‐621. [DOI] [PubMed] [Google Scholar]
- 82. Ikeda Y. The effect of ibuprofen on platelet function in vivo. Keio J Med. 1977;26(4):213‐222. [DOI] [PubMed] [Google Scholar]
- 83. De la Cruz JP, Reyes JJ, Ruiz‐Moreno MI, Lopez‐Villodres JA, Jebrouni N, Gonzalez‐Correa JA. Differences in the in vitro antiplatelet effect of dexibuprofen, ibuprofen, and flurbiprofen in human blood. Anesth Analg. 2010;111(6):1341‐1346. [DOI] [PubMed] [Google Scholar]
- 84. Chakraborty K, Khan GA, Banerjee P, Ray U, Sinha AK. Inhibition of human blood platelet aggregation and the stimulation of nitric oxide synthesis by aspirin. Platelets. 2003;14(7–8):421‐427. [DOI] [PubMed] [Google Scholar]
- 85. Kelleni MT. NSAIDs/nitazoxanide/azithromycin repurposed for COVID‐19: potential mitigation of the cytokine storm interleukin‐6 amplifier via immunomodulatory effects. Expert Rev Anti Infect Ther Jun. 2021a;15:1‐5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86. Valenzuela R, Pedrosa MA, Garrido‐Gil P, et al. Interactions between ibuprofen, ACE2, renin‐angiotensin system, and spike protein in the lung. Implications for COVID‐19. Clin Transl Med. 2021;11(4):e371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87. Moore N, Carleton B, Blin P, Bosco‐Levy P, Droz C. Does ibuprofen worsen COVID‐19? Drug Saf. 2020a;43(7):611‐614. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88. Jamerson BD, Haryadi TH. The use of ibuprofen to treat fever in COVID‐19: a possible indirect association with worse outcome? Med Hypotheses. 2020;144:109880. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89. Mitchell JA, Lucas R, Vojnovic I, Hasan K, Pepper JR, Warner TD. Stronger inhibition by nonsteroid anti‐inflammatory drugs of cyclooxygenase‐1 in endothelial cells than platelets offers an explanation for increased risk of thrombotic events. FASEB J. 2006;20(14):2468‐2475. [DOI] [PubMed] [Google Scholar]
- 90. Kutti Sridharan G, Kotagiri R, Chandiramani VH, et al. COVID‐19 and avoiding ibuprofen. How good is the evidence? Am J Ther. 2020;27(4):e400‐e402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91. Pergolizzi JV Jr, Varrassi G, Magnusson P, et al. COVID‐19 and NSAIDS: a narrative review of knowns and unknowns. Pain Ther. 2020;9(2):353‐358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92. Abu Esba LC, Alqahtani RA, Thomas A, Shamas N, Alswaidan L, Mardawi G. Ibuprofen and NSAID use in COVID‐19 infected patients is not associated with worse outcomes: a prospective cohort study. Infect Dis Ther. 2021;10(1):253‐268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93. Poutoglidou F, Saitis A, Kouvelas D. Ibuprofen and COVID‐19 disease: separating the myths from facts. Expert Rev Respir Med. 2021;15(8):979‐983. [DOI] [PubMed] [Google Scholar]
- 94. Simões e Silva AC, Silveira KD, Ferreira AJ, Teixeira MM. ACE2, angiotensin‐(1‐7) and Mas receptor axis in inflammation and fibrosis. Br J Pharmacol. 2013;169(3):477‐492. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95. Santos RAS, Sampaio WO, Alzamora AC, et al. The ACE2/angiotensin‐(1‐7)/MAS axis of the renin‐angiotensin system: focus on angiotensin‐(1‐7). Physiol Rev. 2018;98(1):505‐553. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96. Franco R, Lillo A, Rivas‐Santisteban R, et al. Functional complexes of angiotensin‐converting enzyme 2 and renin‐angiotensin system receptors: expression in adult but not fetal lung tissue. Int J Mol Sci. 2020;21(24):9602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97. Deshotels MR, Xia H, Sriramula S, Lazartigues E, Filipeanu CM. Angiotensin II mediates angiotensin converting enzyme type 2 internalization and degradation through an angiotensin II type I receptor‐dependent mechanism. Hypertension. 2014;64(6):1368‐1375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98. Xu J, Sriramula S, Xia H, et al. Clinical relevance and role of neuronal AT1 receptors in ADAM17‐mediated ACE2 shedding in neurogenic hypertension. Circ Res. 2017;121(1):43‐55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99. Kashiwagi E, Shiota M, Yokomizo A, Inokuchi J, Uchiumi T, Naito S. EP2 signaling mediates suppressive effects of celecoxib on androgen receptor expression and cell proliferation in prostate cancer. Prostate Cancer Prostatic Dis. 2014;17(1):10‐17. [DOI] [PubMed] [Google Scholar]
- 100. Saheb Sharif‐Askari N, Saheb Sharif‐Askari F, Mdkhana B, et al. Effect of common medications on the expression of SARS‐CoV‐2 entry receptors in liver tissue. Arch Toxicol. 2020;94(12):4037‐4041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101. Chen JS, Alfajaro MM, Wei J, Chow RD, Filler RB, Eisenbarth SC, Wilen CB. Cyclooxgenase‐2 is induced by SARS‐CoV‐2 infection but does not affect viral entry or replication. 2020. bioRxiv [Preprint]. Sep 25:2020.09.24.312769. 10.1101/2020.09.24.312769 [DOI]
- 102. Kohlstedt K, Brandes RP, Müller‐Esterl W, Busse R, Fleming I. Angiotensin‐converting enzyme is involved in outside‐in signaling in endothelial cells. Circ Res. 2004;94(1):60‐67. [DOI] [PubMed] [Google Scholar]
- 103. Kohlstedt K, Busse R, Fleming I. Signaling via the angiotensin‐converting enzyme enhances the expression of cyclooxygenase‐2 in endothelial cells. Hypertension. 2005;45(1):126‐132. [DOI] [PubMed] [Google Scholar]
- 104. Martini WZ, Rodriguez CM, Deguzman R, et al. Dose responses of ibuprofen in vitro on platelet aggregation and coagulation in human and pig blood samples. Mil Med. 2016;181(5 Suppl):111‐116. [DOI] [PubMed] [Google Scholar]
- 105. White WB, Faich G, Whelton A, et al. Comparison of thromboembolic events in patients treated with celecoxib, a cyclooxygenase‐2 specific inhibitor, versus ibuprofen or diclofenac. Am J Cardiol. 2002;89(4):425‐430. [DOI] [PubMed] [Google Scholar]
- 106. Arjomandi Rad A, Vardanyan R, Tas NR. Ibuprofen and thromboembolism in SARS‐COV2. J Thromb Haemost. 2020;18(9):2425‐2427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107. Cui S, Chen S, Ke L. Reply to “Ibuprofen and thromboembolism in SARS‐COV2”. J Thromb Haemost. 2020;18(9):2427‐2428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108. Moore N. No arguments for extra risk from ibuprofen in SARS‐COV2 infection. Therapie. 2020b;75(5):513‐514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109. Terrier O, Dilly S, Pizzorno A, et al. Antiviral properties of the NSAID drug naproxen targeting the nucleoprotein of SARS‐CoV‐2 coronavirus. Molecules. 2021;26(9):2593. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110. Gurbel PA, Bliden KP, Zhu J, et al. Thromboxane inhibition during concurrent therapy with low‐dose aspirin and over‐the‐counter naproxen sodium. J Thromb Thrombolysis. 2018;45(1):18‐26. [DOI] [PubMed] [Google Scholar]
- 111. Scalise M, Indiveri C. Repurposing nimesulide, a potent inhibitor of the B0AT1 subunit of the SARS‐CoV‐2 receptor, as a therapeutic adjuvant of COVID‐19. SLAS Discov. 2020;25(10):1171‐1173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112. Kumar S, Singh B, Kumari P, et al. Identification of multipotent drugs for COVID‐19 therapeutics with the evaluation of their SARS‐CoV2 inhibitory activity. Comput Struct Biotechnol J. 2021;19:1998‐2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113. Amici C, Di Caro A, Ciucci A, et al. Indomethacin has a potent antiviral activity against SARS coronavirus. Antivir Ther. 2006;11(8):1021‐1030. [PubMed] [Google Scholar]
- 114. Reynolds AE, Enquist LW. Biological interactions between herpesviruses and cyclooxygenase enzymes. Rev Med Virol. 2006;16(6):393‐403. [DOI] [PubMed] [Google Scholar]
- 115. Schröer J, Shenk T. Inhibition of cyclooxygenase activity blocks cell‐to‐cell spread of human cytomegalovirus. Proc Natl Acad Sci U S A. 2008;105(49):19468‐19473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116. Bahrami H, Daryani NE, Haghpanah B, et al. Effects of indomethacin on viral replication markers in asymptomatic carriers of hepatitis B: a randomized, placebo‐controlled trial. Am J Gastroenterol. 2005;100(4):856‐861. [DOI] [PubMed] [Google Scholar]
- 117. Napolitano F, Gambardella G, Carrella D, Gao X, di Bernardo D. Computational drug repositioning and elucidation of mechanism of action of compounds against SARS‐COV‐2. 2020. https://arxiv.org/abs/2004.07697v2
- 118. Raghav N, Kamboj RC, Singh H. Effect of some steroidal & non‐steroidal anti‐inflammatory drugs on purified goat brain cathepsin L. Indian J Med Res. 1993;98:188‐192. [PubMed] [Google Scholar]
- 119. Xu T, Gao X, Wu Z, Selinger DW, Zhou Z. Indomethacin has a potent antiviral activity against SARS‐COV‐2 in vitro and canine coronavirus in vivo. BioRXiv (preprint) April 2020. 10.1101/2020.04.01.017624 [DOI]
- 120. Russell B, Moss C, George G, et al. Associations between immune‐suppressive and stimulating drugs and novel COVID‐19‐a systematic review of current evidence. Ecancermedicalscience. 2020;14:1022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121. Alkotaji M, Al‐Zidan RN. Indomethacin: can it counteract bradykinin effects in COVID‐19 patients? Curr Pharmacol Rep. 2021;22(3):1‐5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122. Cochener B, Kling F, Savary‐Le Floch G, Colin J. Indométhacine (Indocollyre) versus Diclofénac (Voltarène) sur la douleur post‐photoablation excimer de surface [Indomethacin (indocollyre) versus diclofenac (voltarene) for the control of pain following excimer photoablation]. J Fr Ophtalmol. 2000;23(6):555‐562. French. [PubMed] [Google Scholar]
- 123. Prasher P, Sharma M, Gunupuru R. Targeting cyclooxygenase enzyme for the adjuvant COVID‐19 therapy. Drug Dev Res. 2021;82(4):469‐473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124. Malikiwi A, Roufaeil C, Tan K, Sehgal A. Indomethacin vs ibuprofen: comparison of efficacy in the setting of conservative therapeutic approach. Eur J Pediatr. 2015;174(5):615‐620. [DOI] [PubMed] [Google Scholar]
- 125. Kelleni MT. Early use of non‐steroidal anti‐inflammatory drugs in COVID‐19 might reverse pathogenesis, prevent complications and improve clinical outcomes. Biomed Pharmacother. 2021b;133:110982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126. Chen JS, Alfajaro MM, Chow RD, et al. Non‐steroidal anti‐inflammatory drugs dampen the cytokine and antibody response to SARS‐CoV‐2 infection. J Virol. 2021;95(7). e00014–e00021 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127. Ravichandran R, Purna P, Vijayaragavan S, Kalavakollu RT, Gaidhane S, Kumar RK. Efficacy and safety of indomethacin in COVID‐19 patients. 2020. medRxiv 12.14.20245266. 10.1101/2020.12.14.20245266 [DOI]
- 128. Mehta N, Mazer‐Amirshahi M, Alkindi N, Pourmand A. Pharmacotherapy in COVID‐19; a narrative review for emergency providers. Am J Emerg Med. 2020;38(7):1488‐1493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129. Sun F, Zhang Y, Li Q. Therapeutic mechanisms of ibuprofen, prednisone and betamethasone in osteoarthritis. Mol Med Rep. 2017;15(2):981‐987. [DOI] [PubMed] [Google Scholar]
- 130. Grosser T. The pharmacology of selective inhibition of COX‐2. Thromb Haemost. 2006;96(4):393‐400. [PubMed] [Google Scholar]
- 131. Dapino P, Ottonello L, Dallegri F. The anti‐inflammatory drug nimesulide inhibits neutrophil adherence to and migration across monolayers of cytokine‐activated endothelial cells. Respiration. 1994;61(6):336‐341. [DOI] [PubMed] [Google Scholar]
- 132. Baghaki S, Yalcin CE, Baghaki HS, Aydin SY, Daghan B, Yavuz E. COX2 inhibition in the treatment of COVID‐19: review of literature to propose repositioning of celecoxib for randomized controlled studies. Int J Infect Dis. 2020;101:29‐32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133. García‐Álvarez JL, García‐Vigil JL. Guidelines for clinical management of SARS‐CoV‐2 infection. Gac Med Mex. 2020;156(6):576‐583. [DOI] [PubMed] [Google Scholar]
- 134. Akhter J, Quéromès G, Pillai K, et al. The combination of bromelain and acetylcysteine (BromAc) synergistically inactivates SARS‐CoV‐2. Viruses. 2021;13(3):425. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135. Liu Q, Huang N, Li A, et al. Effect of low‐dose aspirin on mortality and viral duration of the hospitalized adults with COVID‐19. Medicine (Baltimore). 2021;100(6):e24544. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136. Osborne TF, Veigulis ZP, Arreola DM, Mahajan SM, Röösli E, Curtin CM. Association of mortality and aspirin prescription for COVID‐19 patients at the Veterans Health Administration. PLoS ONE. 2021;16(2):e0246825. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137. Martha JW, Pranata R, Lim MA, Wibowo A, Akbar MR. Active prescription of low‐dose aspirin during or prior to hospitalization and mortality in COVID‐19: A systematic review and meta‐analysis of adjusted effect estimates. Int J Infect Dis. 2021;108:6‐12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138. Srivastava R, Kumar A. Use of aspirin in reduction of mortality of COVID‐19 patients: a metanalysis. Int J Clin Pract. 2021;e14515. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139. Violi F, Cammisotto V, Pignatelli P. Thrombosis in Covid‐19 and non‐Covid‐19 pneumonia: role of platelets. Platelets. 2021;7:1‐9. 10.1080/09537104.2021.1936478 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140. Pan D, Ip A, Zhan S, et al. Pre‐hospital antiplatelet medication use on COVID‐19 disease severity. Heart Lung. 2021;50(5):618‐621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141. Chirumbolo S. Nutraceuticals and dietary supplements should not be used to treat COVID‐19 as pharmaceuticals. Nutrition. 2021, in press;111494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142. Minich DM, Brown BI. A review of dietary (phyto)nutrients for glutathione support. Nutrients. 2019;11(9):2073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143. Hall K, Mfone F, Shallcross M, Pathak V. Review of pharmacotherapy trialed for management of the coronavirus disease‐19. Eur J Med. 2021;53(2):137‐143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144. Chirumbolo S, Valdenassi L, Simonetti V, et al. Insights on the mechanisms of action of ozone in the medical therapy against COVID‐19. Int Immunopharmacol. 2021;96:107777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145. Franzini M, Valdenassi L, Ricevuti G, et al. Oxygen‐ozone (O2‐O3) immunoceutical therapy for patients with COVID‐19. Preliminary evidence reported. Int Immunopharmacol. 2020;88:106879. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146. Di Pierro F, Iqtadar S, Khan A, et al. Potential clinical benefits of quercetin in the early stage of covid‐19: results of a second, pilot, randomized, controlled and open‐label clinical trial. Int J Gen Med. 2021;14:2807‐2816. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147. Kandeil A, Mostafa A, Kutkat O, et al. Bioactive polyphenolic compounds showing strong antiviral activities against severe acute respiratory syndrome coronavirus 2. Pathogens. 2021;10(6):758. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148. Alesci A, Aragona M, Cicero N, Lauriano ER. Can nutraceuticals assist treatment and improve covid‐19 symptoms? Nat Prod Res. 2021;5:1‐20. [DOI] [PubMed] [Google Scholar]
- 149. Ho P, Zheng JQ, Wu CC, et al. Perspective adjunctive therapies for COVID‐19: beyond antiviral therapy. Int J Med Sci. 2021;18(2):314‐324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150. Taher A, Lashgari M, Sedighi L, Rahimi‐Bashar F, Poorolajal J, Mehrpooya M. A pilot study on intravenous N‐Acetylcysteine treatment in patients with mild‐to‐moderate COVID19‐associated acute respiratory distress syndrome. Pharmacol Rep. 2021;10(6):1‐10. 10.1007/s43440-021-00296-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151. Shi Z, Puyo CA. N‐acetylcysteine to combat COVID‐19: an evidence review. Ther Clin Risk Manag. 2020;16:1047‐1055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152. De Flora S, Balansky R, La Maestra S. Rationale for the use of N‐acetylcysteine in both prevention and adjuvant therapy of COVID‐19. FASEB J. 2020;34(10):13185‐13193. 10.1096/fj.202001807 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153. Poe FL, Corn J. N‐Acetylcysteine: a potential therapeutic agent for SARS‐CoV‐2. Med Hypotheses. 2020;143:109862. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154. Zhang R, Qi J, Zhou M, et al. Upregulation of Nrf‐2 attenuates oxidative stress‐induced complement activation‐associated endothelial injury and apoptosis in transplant‐associated thrombotic microangiopathy. Transplant Cell Ther. 2021c;S2666‐6367(21):01002. [DOI] [PubMed] [Google Scholar]
- 155. Atefi N, Behrangi E, Mozafarpoor S, Seirafianpour F, Peighambari S, Goodarzi A. N‐acetylcysteine and coronavirus disease 2019: may it work as a beneficial preventive and adjuvant therapy? A comprehensive review study. J Res Med Sci. 2020;25:109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156. Consolaro E, Suter F, Rubis N, Nurse R, Pedroni S, Moroni C, Pastò E, Paganini MV, Pravettoni G, Cantarelli U, Perico N, Perna A, Peracchi TS, Ruggenenti P, Remuzzi G. A home‐treatment algorithmbased on anti‐inflammatory drugs to prevcent hospitalization of patients with early COVID‐19. A matched‐cohort study (COVER 2). MedRXiv Oct 1 2021. 10.1101/2021.09.29.21264298 [DOI] [PMC free article] [PubMed]
- 157. Kelleni MT. NSAIDs/nitazoxanide/azithromycin immunomodulatory protocol used in adult, geriatric, pediatric, pregnant, and immunocompromised COVID‐19 patients: a real‐world experience. Canad J Medicine. 2021;3(3):121‐143. [Google Scholar]
- 158. Kow CS, Hasan SS. Use of low‐molecular‐weight heparin in COVID‐19 patients. J Vasc Surg Venous Lymphat Disord. 2020;8(5):900‐901. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159. di Micco P, Tufano A, Cardillo G, et al. The impact of risk‐adjusted heparin regimens on the outcome of patients with COVID‐19 Infection. A prospective cohort study. Viruses. 2021;13(9):1720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160. Chirumbolo S, Simonetti V, Franzini M, Valdenassi L, Bertossi D, Pandolfi S. Estimating coronavirus disease 2019 (COVID‐19)‐caused deaths in hospitals and healthcare units: do hospital‐acquired infections play a role? Comments with a proposal. Infect Control Hosp Epidemiol Mar. 2021b;19:1‐2. [DOI] [PMC free article] [PubMed] [Google Scholar]


