Abstract
There is a clear clinical need for efficient cartilage healing strategies for treating cartilage defects which burdens millions of patients physically and financially. Different strategies including microfracture technique, osteochondral transfer, and scaffold-based treatments have been suggested for curing cartilage injuries. Although some improvements have been achieved in several facets, current treatments are still less than satisfactory. Recently, different hydrogel-based biomaterials have been suggested as a therapeutic candidate for cartilage tissue regeneration due to their biocompatibility, high water content, and tunability. Specifically, magnetic hydrogels are becoming more attractive due to their smart response to magnetic fields remotely. We seek to outline the context-specific regenerative potential of magnetic hydrogels for cartilage tissue repair. In this review, first, we explained conventional techniques for cartilage repair and then compared them with new scaffold-based approaches. We illustrated various hydrogels used for cartilage regeneration by highlighting the magnetic hydrogels. Also, we gathered in vitro and in vivo studies of how magnetic hydrogels promote chondrogenesis as well as studied the biological mechanism which is responsible for cartilage repair due to the application of magnetic hydrogel.
1. Introduction
Cartilage injuries occur due to degenerative disease and surgical and traumatic injuries [1, 2]. The hyaline cartilage legions account for the highest rate of world disability [3]; only in the USA, more than 250000 patients require knee arthroplasty for articular cartilage each year [4]. International Cartilage Repair Society assessed that more than 60% of the patients undergoing knee arthroscopy demonstrated cartilage damage [4, 5]. Furthermore, more than 50% of the world population older than 65 years old suffer from osteoarthritis (OA) which is traditionally characterized as cartilage damage [6, 7]. Articular cartilage disorder occurs in conjunction between the bones and worsens over time by constant mechanical degeneration and loss of cartilage tissue, resulting in osteoarthritis [3]. Cartilage has a limited ability for self-repair due to its low vascularity which constraints the replicative ability of the chondrocyte [8, 9]. In contrast with the bone tissue, the cartilage has a few cells and metabolic activity, and the limited number of these cells, if any, is specialized in cartilage remodeling [9]. Without cells and mediators or the lack of accessibility to abundant nutrients even small cartilage defect does not have the innate ability to achieve sufficient healing [10]. Furthermore, the lack of specific diagnostic biomarkers is another reason that makes regeneration of the cartilage a clinical issue for decades [11].
Traditional techniques for curing articular cartilage defects include microfracture, osteochondral autographs, and allographs [12]. Shortcoming reported for current treatment includes the requirement of secondary surgery for osteochondral autographs or immune rejection and transmission of donor pathogen after allographs [13, 14]. Lack of efficient regeneration technique for articular cartilage necessitates tissue engineering approaches combining scaffolds, cells, and growth factors [13]. Cartilage formation is a complex cascade that is influenced by a cocktail of cytokines and growth factors that guide inflammatory cells to the injury site [15]. Hydrogels inducing chondrogenesis via chemical agents can be good therapeutic agents; however, they are not still ideal for clinical application. The limitations associated with the use of growth factors and chemical agents for cartilage tissue regeneration include protein denaturation, side effects, and off-target effects. New studies revealed that the fabrication of novel magnetic hydrogels that ensure cell function and sufficient biomechanical properties to the regenerative environment can be useful for cartilage tissue regeneration [16]. The electromagnetic field (EMF) was previously shown to promote osteogenic differentiation of mesenchymal stem cells (MSC) [17]. It is hypothesized that EMF can also trigger chondrogenic differentiation of stem cells which can be used for the preparation of novel cartilage tissue scaffolds [18–21].
The application of the magnetic hydrogel for cartilage tissue regeneration is still at the beginning of the road and much has not been done in this area. There is a need for a detailed study of magnetically induced chondrogenesis and magnetic hydrogels. In this review, a study was done on the fabrication of magnetic hydrogels that induce chondrogenesis. Furthermore, the influence of magnetically labeled stem cells in enhancing the cartilage matrix synthesis was investigated. In addition, the results of in vitro and in vivo studies of magnetic hydrogel for articular cartilage defects were discussed.
A variety of natural and synthetic polymers have been studied for the fabrication of hydrogels for cartilage regeneration. Commonly used polymers for cartilage tissue regeneration are summarized in Table 1. Collagen, hyaluronic acid (HA), and chondroitin sulfate (CS) are widely used for cartilage regeneration since they can mimic natural cartilage ECM and induce chondrogenesis [22, 23]. Lack of mechanical properties and quick degradation are the shortcomings of most hydrogel scaffolds [24, 25]. Insufficient mechanical or chemical properties can be rectified by crosslinking the polymer with other materials in order to obtain a hybrid polymer [20]. However, a hybrid hydrogel-based scaffold should be designed and manufactured in a way to overcome different shortcomings other than mechanical properties. For instance, the low number of chondrocytes in the cartilage is a limitation that should be addressed by a scaffold that can enhance chondrogenic cell differentiation [26]. Lack of chondrogenesis in the conventional hydrogel-based scaffolds necessitates the fabrication of smart hydrogels that can respond to certain cues. The desire to remotely regulate the cartilage physical and chemical microenvironment has attracted attention to the fabrication of magnetically responsive hydrogels [27]. Therefore, fabricating magnetic hydrogels that can create a microenvironment that ensures chondrocyte cell viability and proliferation and enhances chondrogenesis can be a good cartilage tissue engineering approach.
Table 1.
Most common polymers for cartilage hydrogel scaffolds and their biological activities.
| Polymer | Properties | References |
|---|---|---|
| Collagen | It has chondro-inductive properties which lead to a suitable 3-D microenvironment for enhancing MSC chondrogenesis. Collagen can also provide immunomodulatory properties by reducing certain immunogenic effects. | [28–30] |
| Chondroitin sulfate | It provides chondro-protective and anti-inflammatory properties to enhance cartilage tissue regeneration. Furthermore, it increased the production of collagen type II. | [23] |
| Hyaluronic acid (HA) | It improves early-stage chondrogenesis and was proved to repair osteochondral defects in vivo studies. HA interacts with receptors such as CD44 to adjust signal transduction and stem cell differentiation. | [11, 31, 32] |
| Alginate | It enhances the proliferation of chondrocytes and maintains the chondrocyte phenotype. Its fast and simple gelation makes it suitable for injection. | [33, 34] |
| Chitosan | The similarity of its structure with GAG leads to chondrocyte proliferation and chondrogenesis. It also improves chondrocyte homeostasis. | [35, 36] |
2. Cartilage Structure
Hyaline cartilage is a nonvascularized connective tissue and the most abundant cartilage type of the body which is found in the costal cartilage, trachea, and articular cartilage [16, 37]. The articular cartilage has a highly organized but heterogeneous structure that is composed of small numbers of chondrocytes and a multicomponent matrix [37, 38]. Between 70 and 85% of the articular cartilage is water and 30% of the dry tissue is proteoglycan [38]. As demonstrated in Figure 1, the articular structure of the cartilage is categorized into four zones the superficial, middle, deep, and calcified zone [39]. The superficial zone is the thinnest layer which is composed of ellipsoid chondrocyte and collagen fibrils [40]. Collagen fibers are packed in a superficial layer and are oriented parallel to the articular surface [41]. The superficial zone has the highest water content and provides a gliding surface to the cartilage as well as protection from the synovial fluid immune cells [40, 41]. The middle or transitional zone has a relative proportion between 40 and 60%, while the superficial layer is 10-20% of the articular cartilage [42, 43]. This zone is composed of spheroid-shaped cells and thick collagen fibers that are oriented obliquely [40]. The middle zone induces compatibility between shear forces of the superficial layer and compressive forces of the deeper zone [40]. Deep or radial zone is composed of spheroidal-shaped chondrocytes as well as the highest concentration of proteoglycans [44]. Chondrocytes are larger and collagens are thicker in the deep zone as compared with the other zones [45]. Collagens are oriented perpendicularly to facilitate load distribution and resist compression [40]. The glycosaminoglycan (GAG) amount is usually lower in the superficial zone, enhances in the middle layer, and drops in the deep layer towards the tidemark [42]. Extension of collagen from deep zone to calcified zone preserves cartilage and bone integrity [40]. The calcified zone is the boundary between cartilage and subchondral bone and provides a barrier to diffusion from blood vessels supplying the bone [40].
Figure 1.
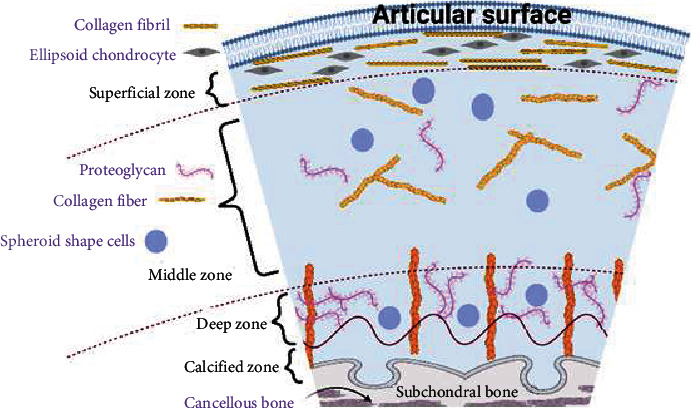
Schematic of the articular cartilage.
3. Cartilage Injuries and Repair
Several pathological conditions such as infectious disease, osteoarthritis, cancer, or traumatic injuries can lead to cartilage defects. Articular cartilage has a low intrinsic capacity for repair so treatment of defects of articular cartilage whether from trauma or degenerative disease is a significant challenge for orthopedic surgeons [46, 47]. Although many techniques have been introduced for the treatment of cartilage defects, not enough is known about the suitable treatment modality for a particular lesion [48]. Due to the fact that no universally accepted system is established to define a lesion, it may be hard to decide which lesion requires treatment and discuss the results of treatment of focal cartilage impairments [48]. Two major issues when repairing the articular cartilage are filling the defect void with the material of similar mechanical properties with cartilage and inducing integration between repair tissue and native cartilage [49]. Current most common strategies for healing cartilage defects include microfracture, osteochondral transfer, autologous chondrocyte implantation (ACI), and scaffolds.
3.1. Microfracture Technique for Cartilage Repair
Microfracture is a safe, effective, minimally invasive, and marrow-stimulating technique that aims at triggering vascular response to injury [50]. This technique includes abrading the tidemark and creating small holes perpendicular to the subchondral bone plate to allow bleeding into the defect. In this technique, the stem cells extravasate from the subchondral area to the chondral defect and they differentiate to the fibrocartilage tissue [50]. At the same time, the body's own tissue healing process maintains the integrity of marrow clot and subchondral plate, inducing durable cartilage repair [51]. Microfractures usually result in a fibrous-fibro hyaline unstructured repair tissue that lacks the biomechanical and viscoelastic features of hyaline cartilage [52–54]. Of note, collagen scaffolds are commonly inserted postmicrofracture to augment marrow stimulation [55]. Different factors are influential in determining the success of the microfracture technique such as the dimension and location of the defect as well as the age and gender of the patient [56].
3.2. Osteochondral Transfer
When the articular defect size is bigger than 15 mm, the transplantation of autogenic and allogenic tissue is preferred over marrow-stimulating techniques [57]. This technique introduces a new hyaline cartilage surface, while microfracture yields a fibrocartilage repair [58]. The source of tissue for the osteochondral transfer may be either autogenic or allogenic. Allogenic tissue is obtained from cadaveric tissues, but the autogenic tissue can provide better mechanical stability and biocompatibility. Although articular cartilage is located in an immunologically privileged position, the immune response is still a major concern with the osteochondral transfer technique [49]. Another disadvantage of this technique is donor-site morbidity after osteochondral transfer due to the cell death at the wound margin which results in tissue degeneration over time [27].
3.3. Autologous Chondrocyte Implantation (ACI)
ACI is the first cell-based biological approach to the treatment of grade III and grade IV cartilage lesions [52, 59] that introduces end-differentiated chondrocytes into the prepared defect, resulting in the formation of a hyaline-like repair tissue [60, 61]. ACI was initially used for the treatment of focal chondral injury and later this technique utilized various 3D polymeric scaffolds [62]. The ACI technique includes creating small biopsy of the hyaline cartilage, extracting chondrocytes from a less-weight-bearing area of articular surface, and culturing the cells in vitro [63]. Then, chondrocyte cells are expanded in vitro to enhance the number of cells to provide enough number to fill a focal articular defect [64]. Once the chondrocyte cell population achieved a certain level in vitro, they are implanted into the cartilage defect. After chondrocytes are implanted into the articular defect, they start to produce cartilage matrix that gradually fills out the cartilage defect. This is similar to the mesenchymal condensation that happens during the limb formation [63, 65]. The periosteal graft may cause complications after ACI which can be addressed to some extent by using collagen scaffolds instead of periosteal patches [55].
3.4. Scaffold
Three-dimensional scaffolds are becoming popular because they are cost-effective, time-efficient, and require a single-stage procedure. Furthermore, they provide a high capacity for cell attachment as well as adjustability for having appropriate mechanical properties [66, 67]. Furthermore, scaffolds have been promising in improving the conventional cartilage repair techniques such as microfracture and ACI by promoting chondrocyte transfer and graft incorporation to enhance hyaline cartilage [55].
Since the beforementioned treatments (microfracture, osteochondral transfer, and ACI) have limitations, the hopes are in scaffold tissue engineering. However, because the chemical and mechanical properties of the cartilage tissue are not consistent through the entire tissue, it is complicated to create a scaffold that can fully mimic the natural cartilage [68]. Therefore, multiphasic scaffolds are more preferred than monophasic scaffolds since they have several layers with controlled properties to imitate the local microenvironment of cartilage tissue [69]. For example, Nguyen et al. [70] fabricated three-layer polyethylene glycol-based scaffold with the chemical gradient of chondroitin sulfate, matrix metalloproteinase-sensitive peptides, and hyaluronic acid by being inspired by the depth-dependent morphology of the cartilage. They showed that MSCs within the scaffold have undergone chondrogenic differentiation and also matrix production profile was compatible with the specific zone of the articular cartilage [70]. Similarly, Liu et al. [71] developed BMDS-laden 3D-bioprinted multilayer scaffold and studied its effect on the animal model of osteochondral defect repair. They observed enhanced collagen type II production as well as decreased inflammatory cytokines in the injury site which has resulted in increased chondrogenesis [71].
Different materials have been suggested for cartilage regeneration purposes (synthetic or natural) and they can be used in various physical forms of fibers, meshes, and hydrogels. Hyaluronan and collagen-based scaffolds are polymers widely used for the fabrication of cartilage scaffolds due to their similarity to the natural cartilage tissue [49, 64]. Scaffolds are designed to be chondro-conductive and they can be used with or without cells. Although chondrocytes are the most commonly used cells for tissue engineering purposes, the potential of MSCs for cartilage regeneration is being investigated [72].
Recently, multidisciplinary effort has been put into the fabrication of scaffolds that can specifically and molecularly interact with the cartilage microenvironment. Biological signals such as growth factors and signals are often incorporated into the hydrogels to enhance chondrogenesis in large cartilage defects [73]. For example, Zhou et al. [74] synthesized gelatin methacrylate (GelMA) and poly(ethylene glycol) diacrylate (PEGDA) and added DNA-based analogs to improve chondrogenesis of adipose-derived stem cells (ADSC). As shown in Figure 2, they printed a three-gradient scaffold, cultured ADSCs on it, and observed chondrogenic differentiation of ADSCs as well as ECM formation [74]. As another example, Kisiday et al. [75] devised a self-assemble peptide hydrogel scaffold to encapsulate chondrocytes. The molecularly engineered peptide used in their study provides tailored degradation rate and induced accumulation of cartilage-like ECM. Moreover, the manufactured hydrogel peptide provided the potential for tethering of growth factors and targeted delivery of growth factors to the chondrocytes [75].
Figure 2.
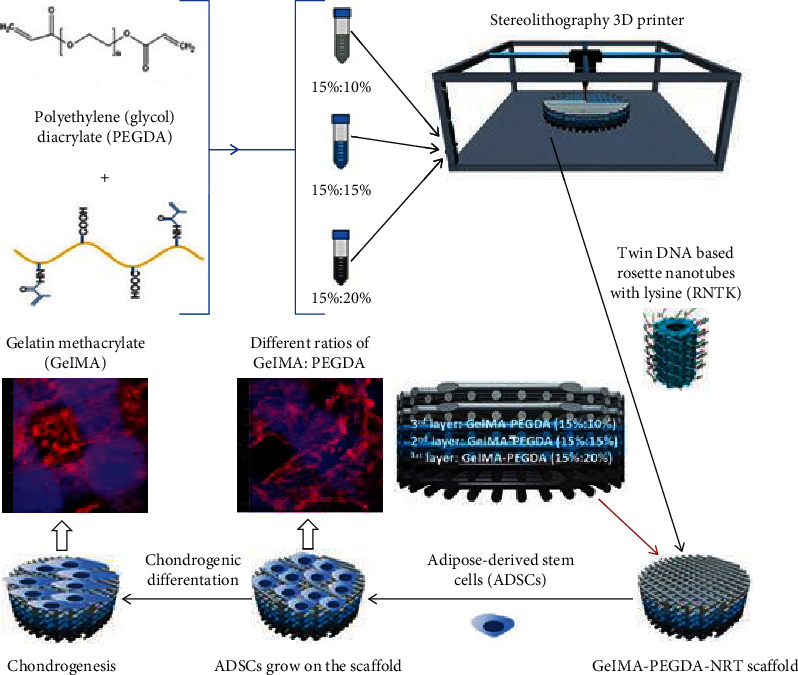
3D-printing hydrogel coated by DNA-based nanotubes [74].
The mechanical properties of hydrogels are considered their main limitation for load-bearing tissues [76]. Different methods have been used to address the poor mechanical properties of hydrogels. For example, Mohabatpour et al. [77] produced electrospun fiber polylactic acid (PLA) and grafted hyaluronic acid and alginate to enhance weight ratio nanofibers in composite to promote mechanical properties [77]. Similarly, Fenbo et al. [78] synthesized strontium alginate/chondroitin sulfate (Alg/CS-Sr) hydrogel with tunable stiffness by changing the concentration of strontium chloride. They inserted the hydrogel in the rabbit cartilage defect model and confirmed cartilage regeneration [78].
4. Hydrogel for Cartilage Tissue Regeneration
Hydrogels are good candidates for the construction of three-dimensional structures due to their tunable biomechanical properties and biocompatibility. Hydrogel scaffolds can regulate different materials to promote cartilage repair [11]. The hydrogel can be modified with cell adhesion ligands and its internal aqueous environment protects the cell and allows nutrient transportation [13].
Hydrogels can be used as filling agents, a delivery vehicle for bioactive molecules, and a three-dimensional structure for organizing cells [13, 88]. Different synthetic and natural polymers have been used for the fabrication of scaffold hydrogel for bone tissue engineering [89]. Although cartilage tissue is not successful in self-repair, recent hydrogel fabrication techniques are promising. For example, PLGA-gelatin/chondroitin/hyaluronate hydrogel scaffold was seeded with MSC, enhancing the MSC proliferation and GAG synthesis [90, 91]. Then, autologous differentiated MSC/PLGA-GCH was implanted in rabbit contralateral cartilage defect and it demonstrated better chondrocyte morphology and integration of continuous subchondral bone [13].
In order to generate MSC-based functional hyaline cartilage, MSCs should be directed to chondrogenic lineage and initiate the formation of cartilage matrix including collagen type II and glycosaminoglycan (GAGs) [90]. Although chondrogenic chemicals or growth factors have been used for chondrogenic differentiation of MSCs, protein denaturation, probability of carrying a pathogen, and undesired side effects limit their application [67, 92].
There have been several types of research to expand and improve the hydrogel that induces chondrogenesis; however, researchers are looking for physical cues that can promote cartilage regeneration after hydrogel is added [64]. The electromagnetic hydrogels are the new generation of hydrogel for cartilage tissue generation that is shown to be successful in in vitro and in vivo studies. These hydrogels have not still reached clinical practice and their long-term results have not been investigated yet. This review is focused on the hydrogels that induce cartilage regeneration through electromagnetic properties.
5. Magnetic Hydrogels for Cartilage Tissue Regeneration
The electromagnetic field (EMF) and pulsed electromagnetic field (PEMF) are FDA-approved techniques that are previously shown to promote osteogenic differentiation of MSC [17]. Human mesenchymal stem cells are widely accepted seeding cells and control of them by physical cues is of great interest in regenerative medicine [93]. Jiang et al. studied the uptake of magnetic nanoparticles by MSCs under the magnetic field. Iron-oxide nanoparticles-loaded bovine serum albumin (BSA) internalized by MSCs increased osteogenic differentiation and expression of collagen type I and osteocalcin significantly [94]. Several groups have reported that electromagnetic field promotes cartilage formation in vitro and in vivo. For example, adipose-derived stem cells were cultured in a hyaluronan microenvironment and treated with a pulsed electromagnetic field (PEMF) [95]. It was shown that under chondrogenic induction, PEMF stimulation promoted the expression of main chondrogenic genes such as SOX-9, collagen II, and aggrecan [92]. Similarly, Hou et al. [11] incorporated SPIONs into hyaluronic acid-graft-amphiphilic gelatin hydrogel and injected it into the rabbit's knee to study chondrogenic commitment. "They observed magnetic field upregulated Col II and SOX9 gene expression"" and magnetic derived" should be omitted. [11].
Therapeutic applications of the magnetic field keep expanding because magnetic nanoparticles can apply remote magnetic-induced physical stimulation which enables targeting of the specific site as well [11]. Mesenchymal stem cells labeled with magnetic nanoparticles were shown to induce higher differentiation [96]. In one study, human bone marrow-derived MSC was under static magnetic field and magnetic-derived shear stress via magnetic nanoparticles. It was demonstrated that biophysical stimulation resulted in higher chondrogenic differentiation efficiency [11].
It is shown that an early essential step for initiating the chondrogenic differentiation of the stem cell is its condensation [97]. For example, a decrease in intercellular spaces in the area of the cartilage and bone formation precedes cartilage differentiation during limb skeletogenesis [98]. In one study, magnetic labeling of the stem cell with the maghemite citrate-coated iron oxide resulted in cell condensation into aggregates. The magnetically cellularized scaffolds were exposed to transduction and shear stress stimuli produced high collagen type II [97]. The combination of magnetic cell seeding with dynamic differentiation induces chondrogenic differentiation as well as the creation of a millimeter-sized cartilage cellular construct [88]. Similarly, in another study [14, 99], Maghemite nanoparticles were used for labeling MSCs, and magnetic cells were suspended in chondrogenesis culture medium. Gene expression and histological studies proved that major cartilage matrix proteins (collagen II and aggrecan) were elevated [67, 88].
Magnetic nanoparticles can induce chondrogenic differentiation under the magnetic field when they bind to the cell surface. For example, Pulse electromagnetic fields (PEMFs) was applied to bone marrow mesenchymal stem cell (BMSC) cultured on the electromagnetic hydrogel. The hydrogel composed of gelatin, beta-cyclodextrin (beta-CD), and magnetic iron oxide (Fe3O4) induced the expression of late chondrogenic differentiation markers including COL2 and aggrecan [9]. In addition, the hydrogel was implanted in rabbit knee cartilage defect revealing regenerative tissue that has completely filled the gap, and the histology staining was similar to that of natural cartilage.
Superparamagnetic iron oxide nanoparticles (SPIONs) can guide cells and serve as physical stimulation. In one study, SPIONs were encapsulated in a hydrophobic shell of hyaluronic acid-graft-amphiphilic gelatin (HA-AGMCs) microsphere. Hyaluronic acid is one of the major components of the chondrocyte extracellular matrix (ECM) which provides a backbone for aggrecan aggregation [11]. Furthermore, it interacts with the CD44 receptor to regulate signal transduction, cell migration, and differentiation [11]. Applying a magnetic field to the SPIONs incorporated into the (HA-AGMC) microsphere led to the expression of CoI II and SOX9 which are cartilage tissue-specific genes. In addition, this novel platform initiated chondrogenesis and sGAG synthesis [11].
In another study, magnetic nanoparticle-vesicle (MNVP) was assembled by cross-linking of phospholipid vesicles and magnetite nanoparticles. Chondrocytes and nanoparticle-vesicle assemblies were coimmobilized within a calcium alginate hydrogel. This smart biomaterial responds to the alternating magnetic field by translating noninvasive magnetic signals into cellular responses [13]. Their research proved that the chondrocyte in the gel responded to the magnetic release of ascorbic acid-2-phosphate (AAP) which was applied as an additive by producing a high level of collagen [13]. Similarly, in another study, dextran-coated magnetic nanoparticles were integrated into the distinct layer of agarose construct to create trilayered ferrogel [100]. An external magnetic field of the 0.5 T was applied to the bovine chondrocytes seeded in ferrogels demonstrating that sGAG content increased over time [100].
The other advantage provided by the incorporation of magnetic nanoparticles into the hydrogel is they can serve as a contrast agent for imaging [101]. Magnetic resonance imaging (MRI) is a noninvasive technique that enables longitudinal imaging at successive time points and can be used to visualize the molecular changes and remodeling of the repairing tissue [101, 102]. Yang et al. [101] incorporated SPIONs-Kartogenin into cellulose nanocrystal/dextran hydrogel. The SPION-labeled hydrogel not only induced chondrogenesis both in vitro and in vivo but also demonstrated magnetic resonance contrast enhancement [101]. Similarly, Chen et al. [103] fabricated USPIO-labeled cellulose nanocrystal (CNC)/silk fibroin (SF) composite hydrogel which enhanced chondrogenic gene upregulation in vitro as well as provided a mean for measuring hydrogel degradation through MRI imaging [103].
5.1. Methods for Synthesizing and Characterizing Magnetic Hydrogels
The cartilage microenvironment can be triggered by magnetic stimuli through being exposed to the external magnetic field or magnetic scaffolding [104]. Approaches used in order to synthesize the magnetic scaffolds include both conventional and modern techniques [105]. The conventional method of fabrication of magnetic hydrogel includes mixing nanoparticles with hydrogel, precipitation, blending, and grafting method [105], while the recent research has focused on additive manufacturing. Farzaneh et al. [104] mixed cobalt ferrite nanoparticles (CFNs) with hydrogel precursors to produce magnetic hydrogel. The magnetic properties of a synthesized hydrogel such as hysteresis curve were studied by vibrating sample magnetometer (VSM) instrument [104]. Furthermore, Huang et al. [106] added the iron oxide magnetic nanoparticles as embedment to the gelatin and β-cyclodextrin and inserted them into the rabbit cartilage defect. MSCs seeded in hydrogel exposed to the PEMF promoted the differentiation of the stem cells by promoting COL2 and aggrecan [106]. Similarly, Zhang et al. [107] first coprecipitated the iron oxide nanoparticles with polyvinyl alcohol and then synthesized it with the combination of collagen II, polyethylene glycol, and hyaluronic acid to fabricate magnetic hybrid gel which mimics the natural cartilage ECM [107].
The additive manufacturing technique is promising for the fabrication of magnetic hydrogels due to their potential to be customized and be reproduced [108]. De Santis et al. [108] synthesized polycaprolactone-polyethylene glycol-based magnetic scaffold by stereolithography approaches for articular cartilage tissue regeneration [108]. Also, Choi et al. [109] fabricated a scaffold composed of self-healing hydrogel combined with ferrogel via extrusion 3D printing technique as demonstrated in Figure 3. The ferrogel demonstrated superparamagnetic properties due to containing SPIONs. They conducted in vitro study by exposing the cells encapsulated in hydrogel to the external magnetic field. They showed that the magnetic stimulation upregulates the SOX-9 and COL-2 which indicates the potential of fabricated magnetic hydrogel for cartilage tissue regeneration [109].
Figure 3.
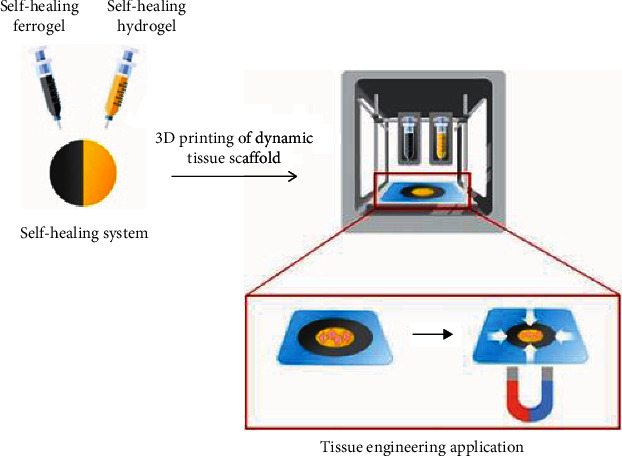
3D printing of scaffold including self-healing hydrogel and ferrogel [109].
5.2. Underlying Biological Mechanism of Enhanced Chondrogenesis Dependent on Magnetic Stimuli
Magnetic hydrogels can be used to influence cellular mechanotransduction and translational and paracrine responses of the cells to encourage cartilage repair through different cellular and molecular mechanisms [19]. The mechanisms by which magnetic signals promote cartilage include enhancing cell-cell interaction, MSC adherence to the defect area, and activating mechanosensitive channels [110]. For instance, Kamei et al. [111] proved that cell adhesion molecules such as integrin α2 (ITG α2), integrin α6 (ITG α6), integrin β3-binding protein (ITG β3BP), intercellular adhesion molecule–2 (ICAM-2), and platelet/endothelial cell adhesion molecule–1 (PECAM-1) have been upregulated in magnetically labeled MSCs. Increased adhesion rate of MSCs can make them engraft to the defect area biologically [111].
Also, the electromagnetic field can induce cartilage tissue regeneration by stimulating chondrocyte maturation [112–115]. For instance, Yi et al. reported that magnetic field upregulates the chondrogenic genes in stem cells and also enhances the synthesis of proteoglycans. They suggested that this might be due to increased expression of transforming growth factor beta in presence of magnetic field. Yan et al. [117] showed that increased cartilage regeneration under EMF is associated with enhanced (TGF-β) and it results from the activation of the Wnt1/LRP6/β-catenin signaling pathway [117]. Moreover, magnetic signals have been shown to contribute to chondrogenesis by activating calcium-permeable transient receptor potential (TRP) channels [19]. Parate et al. [19] have demonstrated that paracrine activity of MSC secretome provides the potential for chondrogenic differentiation of MSCs when they are exposed to the magnetic signal [19]. Table 2 summarizes the advantages and disadvantages of different cartilage repair strategies.
Table 2.
Advantages and disadvantages of different cartilage repair strategies.
| Technique | Advantages | Disadvantages | References |
|---|---|---|---|
| Microfracture technique | It is minimally invasive and there is no need for a tissue graft. | It does not restore normal hyaline cartilage and leads to losing undamaged cartilage. This technique is just applied for the lesion size less than 2.5 cm2. |
[53, 55, 79] |
| Osteochondral transfer | Autograft provides a fresh viable cartilage tissue from the patient. Allograft is a useful technique for different defect sizes and locations. Also, it decreases the operation time compared with autograft. |
Autograft may lead to donor-site morbidity. Also, some patients do not have proper donor tissue. Moreover, the autograft cannot be normally used for repairing large defects. Allograft may lead to graft host reaction. |
[80–84] |
| ACI | Can repair large cartilage defects with minimum donor-site morbidity | It leads to periosteal hypertrophy and graft delamination | [55, 85] |
| Scaffold | High biocompatibility, incorporating growth factors and tunable properties | [86, 87] |
6. Future Directions and Limitations
Magnetic hydrogels have been shown to be promising for the treatment of cartilage lesions due to their influence on the stem cell fate as well as chondrocyte behavior [14]. Magnetic hydrogels can provide other advantages such as being used for delivery and timely release of the growth factors. In addition, magnetic strength in tissue formation controls the processes influencing interface regeneration and the homeostasis [118]. Recent research proved the application of electromagnetic field promotes progress of the fascinating line in the field of regenerative medicine for cartilage tissue regeneration. Moreover, magnetic nanoparticles can manipulate the microstructure by noncontact magnetic forces or inducing mechanical stresses at the microscopic level through applied magnetic field which may result in cell proliferation and differentiation [16]. However, these hydrogels still struggle with cell differentiation [11] and they need improvements to generate a more biomimetic and functional cartilage substitute for future preclinical applications [16].
Data Availability
SPSS data of the participant can be requested from the authors. Please write to the corresponding author if you are interested in such data.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
References
- 1.Armiento A., Stoddart M. J., Alini M., Eglin D. Biomaterials for articular cartilage tissue engineering: learning from biology. Journal of cell science . 2018;65:1–20. doi: 10.1016/j.actbio.2017.11.021. [DOI] [PubMed] [Google Scholar]
- 2.Guilak F., Nims R. J., Dicks A., Wu C. L., Meulenbelt I. Osteoarthritis as a disease of the cartilage pericellular matrix. Matrix Biology . 2018;71-72:40–50. doi: 10.1016/j.matbio.2018.05.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Babaniamansour P., Ebrahimian-Hosseinabadi M., Zargar-Kharazi A. Designing an optimized novel femoral stem. Journal of medical signals and sensors . 2017;7(3):p. 170. doi: 10.4103/jmss.JMSS_1_17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Fini M., Pagani S., Giavaresi G., et al. Functional tissue engineering in articular cartilage repair: is there a role for electromagnetic biophysical stimulation? Tissue Engineering Part B: Reviews . 2013;19(4):353–367. doi: 10.1089/ten.teb.2012.0501. [DOI] [PubMed] [Google Scholar]
- 5.Ershadinia N., Mortazavinia N., Babaniamansour S., Najafi-Nesheli M., Babaniamansour P., Aliniagerdroudbari E. The prevalence of autoimmune diseases in patients with multiple sclerosis: A cross-sectional study in Qom, Iran, in 2018. Current Journal of Neurology . 2020;19(3):98–102. doi: 10.18502/cjn.v19i3.5421. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Li Y., Wei X. C., Zhou J. M., Wei L. The age-related changes in cartilage and osteoarthritis. BioMed Research International . 2013;2013:12. doi: 10.1155/2013/916530.916530 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Zhao Z., Tan Q., Jiang A., et al. Evidence of subchondral bone's effects on articular cartilage damage in OVX-OA rat. Engineering Fracture Mechanics . 2020;233:p. 107081. doi: 10.1016/j.engfracmech.2020.107081. [DOI] [Google Scholar]
- 8.Tuan R. S., Chen A. F., Klatt B. A. Cartilage regeneration. The Journal of the American Academy of Orthopaedic Surgeons . 2013;21(5):303–311. doi: 10.5435/JAAOS-21-05-303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Huey D. J., Hu J. C., Athanasiou K. A. J. S. Unlike bone, cartilage regeneration remains elusive. Science . 2012;338(6109):917–921. doi: 10.1126/science.1222454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Edalatfar M., Piri S. M., Mehrabinejad M. M., et al. Biofluid biomarkers in traumatic brain injury: a systematic scoping review. Neurocritical care . 2021;35(2):559–572. doi: 10.1007/s12028-020-01173-1. [DOI] [PubMed] [Google Scholar]
- 11.Hou K.-T., Liu T. Y., Chiang M. Y., Chen C. Y., Chang S. J., Chen S. Y. Cartilage tissue-mimetic pellets with multifunctional magnetic hyaluronic acid-graft-amphiphilic gelatin microcapsules for chondrogenic stimulation. 2020;12(4):p. 785. doi: 10.3390/polym12040785. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Rahmani S., Ahmadzadeh A., Hajihassani O., Mirhosseini S., Gorgin S. An efficient multi-core and many-core implementation of k-means clustering. ACM-IEEE International Conference on Formal Methods and Models for System Design (MEMOCODE); 2016. [Google Scholar]
- 13.Liao J., Shi K., Ding Q., Qu Y., Luo F., Qian Z. Recent developments in scaffold-guided cartilage tissue regeneration. Journal of biomedical nanotechnology . 2014;10(10):3085–3104. doi: 10.1166/jbn.2014.1934. [DOI] [PubMed] [Google Scholar]
- 14.Farahmand S., Abdolhoseini A., Aliniagerdroudbari E., Babaniamansour S., Baratloo A., Bagheri-Hariri S. Point-of-care ultrasound modalities in terms of diagnosing acute decompensated heart failure in emergency department; a diagnostic accuracy study. Internal and Emergency Medicine . 2020;15(3):491–499. doi: 10.1007/s11739-019-02233-x. [DOI] [PubMed] [Google Scholar]
- 15.Roshandel M., Sotudeh-Gharebagh R., Mirzakhanlouei S., Hajiaghaee R., Ghaffarzadegan R. Statistical optimization of production conditions of polycaprolactone-chitosan-curcumin Particles. Journal of Chemical and Petroleum Engineering . 2018;52(2):181–191. [Google Scholar]
- 16.Bonhome-Espinosa A. B., Campos F., Durand-Herrera D., et al. In vitro characterization of a novel magnetic fibrin-agarose hydrogel for cartilage tissue engineering. Journal of the mechanical behavior of biomedical materials . 2020;104:p. 103619. doi: 10.1016/j.jmbbm.2020.103619. [DOI] [PubMed] [Google Scholar]
- 17.Mohtasebi M., Huang C., Mazdeyasna S., et al. optical techniques in neurosurgery, Neurophotonics, and Optogenetics . International Society for Optics and Photonics; 2021. Noncontact optical assessment of spontaneous low-frequency fluctuations of cerebral blood flow in neonatal intraventricular hemorrhage. [Google Scholar]
- 18.Ross C. L., Ang D. C. Targeting mesenchymal stromal cells/pericytes (MSCs) with pulsed electromagnetic field (pemf) has the potential to treat rheumatoid arthritis. Frontiers in immunology . 2019;10:p. 266. doi: 10.3389/fimmu.2019.00266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Parate D., Kadir N. D., Celik C., et al. Pulsed electromagnetic fields potentiate the paracrine function of mesenchymal stem cells for cartilage regeneration. Stem cell research & therapy . 2020;11(1):1–16. doi: 10.1186/s13287-020-1566-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Hajivandi S., Dachek A., Salimi A., et al. Comparison of the separate and combined effects of physiotherapy treatment and corticosteroid injection on the range of motion and pain in nontraumatic rotator cuff tear: a randomized controlled trial. Advances in Orthopedics . 2021;2021:7. doi: 10.1155/2021/6789453.6789453 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Uzieliene I., Bernotas P., Mobasheri A., Bernotiene E. The role of physical stimuli on calcium channels in chondrogenic differentiation of mesenchymal stem cells. International journal of molecular sciences . 2018;19(10):p. 2998. doi: 10.3390/ijms19102998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Setayeshmehr M., Esfandiari E., Rafieinia M., et al. Hybrid and composite scaffolds based on extracellular matrices for cartilage tissue engineering. Tissue Engineering Part B: Reviews . 2019;25(3):202–224. doi: 10.1089/ten.teb.2018.0245. [DOI] [PubMed] [Google Scholar]
- 23.Balakrishnan B., Banerjee R. Biopolymer-based hydrogels for cartilage tissue engineering. Chemical Reviews . 2011;111(8):4453–4474. doi: 10.1021/cr100123h. [DOI] [PubMed] [Google Scholar]
- 24.Cao Y., Xiong D., Wang K., Niu Y. Semi-degradable porous poly (vinyl alcohol) hydrogel scaffold for cartilage repair: evaluation of the initial and cell-cultured tribological properties. Journal of the mechanical behavior of biomedical materials . 2017;68:163–172. doi: 10.1016/j.jmbbm.2017.02.001. [DOI] [PubMed] [Google Scholar]
- 25.Shekouhi N., Dick D., Baechle M. W., et al. Clinically relevant finite element technique based protocol to evaluate growing rods for early onset scoliosis correction. Jor Spine . 2020;3(3):p. e1119. doi: 10.1002/jsp2.1119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Koo Y., Choi E. J., Lee J. Y., Kim H. J., Kim G. H., Do S. H. 3D printed cell-laden collagen and hybrid scaffolds for _in vivo_ articular cartilage tissue regeneration. Journal of Industrial and Engineering Chemistry . 2018;66:343–355. doi: 10.1016/j.jiec.2018.05.049. [DOI] [Google Scholar]
- 27.Liu Z., Liu J., Cui X., Wang X., Zhang L., Tang P. Recent advances on magnetic sensitive hydrogels in tissue engineering. Frontiers in chemistry . 2020;8:p. 124. doi: 10.3389/fchem.2020.00124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Chen X., Zhang F., He X., et al. Chondrogenic differentiation of umbilical cord-derived mesenchymal stem cells in type I collagen-hydrogel for cartilage engineering. Injury . 2013;44(4):540–549. doi: 10.1016/j.injury.2012.09.024. [DOI] [PubMed] [Google Scholar]
- 29.Zhang L., Yuan T., Guo L., Zhang X. An in vitro study of collagen hydrogel to induce the chondrogenic differentiation of mesenchymal stem cells. Journal of biomedical materials research Part A . 2012;100(10):2717–2725. doi: 10.1002/jbm.a.34194. [DOI] [PubMed] [Google Scholar]
- 30.Yuan T., Zhang L., Li K., et al. Collagen hydrogel as an immunomodulatory scaffold in cartilage tissue engineering. Journal of Biomedical Materials Research Part B: Applied Biomaterials . 2014;102(2):337–344. doi: 10.1002/jbm.b.33011. [DOI] [PubMed] [Google Scholar]
- 31.Matsiko A., Levingstone T. J., O’Brien F. J., Gleeson J. P. Addition of hyaluronic acid improves cellular infiltration and promotes early- stage chondrogenesis in a collagen-based scaffold for cartilage tissue engineering. Journal of the Mechanical Behavior of Biomedical Materials . 2012;11:41–52. doi: 10.1016/j.jmbbm.2011.11.012. [DOI] [PubMed] [Google Scholar]
- 32.Unterman S. A., Gibson M., Lee J. H., et al. Hyaluronic acid-binding scaffold for articular cartilage repair. Tissue Engineering Part A . 2012;18(23-24):2497–2506. doi: 10.1089/ten.tea.2011.0711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Zeng L., Yao Y., Wang D. A., Chen X. Effect of microcavitary alginate hydrogel with different pore sizes on chondrocyte culture for cartilage tissue engineering. Materials Science and Engineering: C . 2014;34:168–175. doi: 10.1016/j.msec.2013.09.003. [DOI] [PubMed] [Google Scholar]
- 34.Wei W., Ma Y., Yao X., et al. Advanced hydrogels for the repair of cartilage defects and regeneration. Bioactive Materials . 2021;6(4):998–1011. doi: 10.1016/j.bioactmat.2020.09.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Meng Q., Man Z., Dai L., et al. A composite scaffold of MSC affinity peptide-modified demineralized bone matrix particles and chitosan hydrogel for cartilage regeneration. Scientific Reports . 2015;5(1):p. 17802. doi: 10.1038/srep17802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Comblain F., Rocasalbas G., Gauthier S., Henrotin Y. Chitosan: a promising polymer for cartilage repair and viscosupplementation. Bio-medical Materials and Engineering . 2017;28(s1):S209–S215. doi: 10.3233/BME-171643. [DOI] [PubMed] [Google Scholar]
- 37.Carballo C. B., Nakagawa Y., Sekiya I., Rodeo S. A. Basic science of articular cartilage. Clinics in sports medicine . 2017;36(3):413–425. doi: 10.1016/j.csm.2017.02.001. [DOI] [PubMed] [Google Scholar]
- 38.Mansour J. M. Biomechanics of cartilage. Kinesiology: the mechanics and pathomechanics of human movement . 2003;2:69–83. [Google Scholar]
- 39.Mansfield J. C., Bell J. S., Winlove C. P. The micromechanics of the superficial zone of articular cartilage. Osteoarthritis and cartilage . 2015;23(10):1806–1816. doi: 10.1016/j.joca.2015.05.030. [DOI] [PubMed] [Google Scholar]
- 40.Killen M.-C., Charalambous C. P. Advances in Medical and Surgical Engineering . Elsevier; 2020. Advances in cartilage restoration techniques; pp. 71–83. [DOI] [Google Scholar]
- 41.Gannon A. R., Nagel T., Kelly D. J. The role of the superficial region in determining the dynamic properties of articular cartilage. Osteoarthritis and cartilage . 2012;20(11):1417–1425. doi: 10.1016/j.joca.2012.08.005. [DOI] [PubMed] [Google Scholar]
- 42.Antons J., Marascio M. G. M., Nohava J., et al. Zone-dependent mechanical properties of human articular cartilage obtained by indentation measurements. Journal of Materials Science: Materials in Medicine . 2018;29(5):p. 57. doi: 10.1007/s10856-018-6066-0. [DOI] [PubMed] [Google Scholar]
- 43.Sharafat Vaziri A., Aghaghazvini L., Jahangiri S., et al. Determination of normal reference values for meniscal extrusion using ultrasonography during the different range of motion. Journal of Ultrasound in Medicine . 2022 doi: 10.1002/jum.15955. [DOI] [PubMed] [Google Scholar]
- 44.Bagheri-Hariri S., Bahreini M., Farshidmehr P., et al. The effect of extended-focused assessment with sonography in trauma results on clinical judgment accuracy of the physicians managing patients with blunt thoracoabdominal trauma. Archives of Trauma Research . 2019;8(4):207–213. doi: 10.4103/atr.atr_57_19. [DOI] [Google Scholar]
- 45.Dowthwaite G. P., Bishop J. C., Redman S. N., et al. The surface of articular cartilage contains a progenitor cell population. Journal of Cell Science . 2004;117(6):889–897. doi: 10.1242/jcs.00912. [DOI] [PubMed] [Google Scholar]
- 46.Dhinsa S., Adesida B. Current clinical therapies for cartilage repair, their limitation and the role of stem cells. Current stem cell research & therapy . 2012;7(2):143–148. doi: 10.2174/157488812799219009. [DOI] [PubMed] [Google Scholar]
- 47.Sheikhbahaei E., Mirghaderi S. P., Moharrami A., Habibi D., Motififard M., Mortazavi S. M. J. Incidence of symptomatic COVID-19 in unvaccinated patients within one month after elective total joint arthroplasty: a multicenter study. Arthroplasty Today . 2022;14:110–115. doi: 10.1016/j.artd.2022.01.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Brittberg M., Winalski C. S. Evaluation of cartilage injuries and repair. JBJS . 2003;85(supplement_2):58–69. doi: 10.2106/00004623-200300002-00008. [DOI] [PubMed] [Google Scholar]
- 49.Redman S., Oldfield S., Archer C. J. E. C. M. Current strategies for articular cartilage repair. European Cells & Materials . 2005;9(23-32):23–32. doi: 10.22203/eCM.v009a04. [DOI] [PubMed] [Google Scholar]
- 50.Yen Y.-M., Kocher M. S. Chondral lesions of the hip. Sports Medicine and Arthroscopy Review . 2010;18(2):83–89. doi: 10.1097/JSA.0b013e3181de1189. [DOI] [PubMed] [Google Scholar]
- 51.Gobbi A., Nunag P., Malinowski K. J. K. S. Treatment of full thickness chondral lesions of the knee with microfracture in a group of athletes. Knee Surgery, Sports Traumatology, Arthroscopy . 2005;13(3):213–221. doi: 10.1007/s00167-004-0499-3. [DOI] [PubMed] [Google Scholar]
- 52.Peterson L., Vasiliadis H. S., Brittberg M., Lindahl A. Autologous chondrocyte implantation. The American journal of sports medicine . 2010;38(6):1117–1124. doi: 10.1177/0363546509357915. [DOI] [PubMed] [Google Scholar]
- 53.Mithoefer K., McAdams T., Williams R. J., Kreuz P. C., Mandelbaum B. R. Clinical efficacy of the microfracture technique for articular cartilage repair in the knee. The American journal of sports medicine . 2009;37(10):2053–2063. doi: 10.1177/0363546508328414. [DOI] [PubMed] [Google Scholar]
- 54.Basad E., Ishaque B., Bachmann G., Stürz H., Steinmeyer J. Matrix-induced autologous chondrocyte implantation versus microfracture in the treatment of cartilage defects of the knee: a 2-year randomised study. Knee surgery, sports traumatology, arthroscopy . 2010;18(4):519–527. doi: 10.1007/s00167-009-1028-1. [DOI] [PubMed] [Google Scholar]
- 55.Mollon B., Kandel R., Chahal J., Theodoropoulos J. The clinical status of cartilage tissue regeneration in humans. Osteoarthritis and cartilage . 2013;21(12):1824–1833. doi: 10.1016/j.joca.2013.08.024. [DOI] [PubMed] [Google Scholar]
- 56.Bark S., Piontek T., Behrens P., Mkalaluh S., Varoga D., Gille J. Enhanced microfracture techniques in cartilage knee surgery: fact or fiction? World Journal of Orthopedics . 2014;5(4):444–449. doi: 10.5312/wjo.v5.i4.444. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.VanTienderen R. J., Dunn J. C., Kusnezov N., Orr J. D. Osteochondral allograft transfer for treatment of osteochondral lesions of the talus: a systematic review. Arthroscopy: The Journal of Arthroscopic & Related Surgery . 2017;33(1):217–222. doi: 10.1016/j.arthro.2016.06.011. [DOI] [PubMed] [Google Scholar]
- 58.Werner B. C., Cosgrove C. T., Gilmore C. J., et al. Accelerated return to sport after osteochondral autograft plug transfer. Orthopaedic Journal of Sports Medicine . 2017;5(4) doi: 10.1177/2325967117702418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Jones D. G., Peterson L. Autologous chondrocyte implantation. JBJS . 2006;88(11):2501–2520. doi: 10.2106/00004623-200611000-00025. [DOI] [PubMed] [Google Scholar]
- 60.Minas T., Ogura T., Bryant T. Autologous chondrocyte implantation. JBJS Essential Surgical Techniques . 2016;6(2):p. e24. doi: 10.2106/JBJS.ST.16.00018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Harris J. D., Siston R. A., Pan X., Flanigan D. C. Autologous Chondrocyte Implantation. The Journal of bone and joint surgery. American volume . 2010;92(12):2220–2233. doi: 10.2106/JBJS.J.00049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Harris J. D., Siston R. A., Pan X., Flanigan D. C. Autologous chondrocyte implantation: a systematic review. The Journal of bone and joint surgery. American volume . 2010;92(12):2220–2233. doi: 10.2106/JBJS.J.00049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Vasiliadis H. S., Wasiak J., Salanti G. Autologous chondrocyte implantation for the treatment of cartilage lesions of the knee: a systematic review of randomized studies. Knee surgery, sports traumatology, arthroscopy . 2010;18(12):1645–1655. doi: 10.1007/s00167-010-1050-3. [DOI] [PubMed] [Google Scholar]
- 64.Faraji M., Behnam H., Norizadeh Cherloo M., Shojaeifard M. Novel approach for automatic mid‐diastole frame detection in 2D echocardiography sequences for performing planimetry of the mitral valve orifice. IET Image Processing . 2020;14(12):2890–2900. doi: 10.1049/iet-ipr.2019.1757. [DOI] [Google Scholar]
- 65.Marlovits S., Zeller P., Singer P., Resinger C., Vécsei V. Cartilage repair: generations of autologous chondrocyte transplantation. European journal of radiology . 2006;57(1):24–31. doi: 10.1016/j.ejrad.2005.08.009. [DOI] [PubMed] [Google Scholar]
- 66.McNickle A. G., Provencher M. T., Cole B. J. Overview of existing cartilage repair technology. Sports Medicine and Arthroscopy Review . 2008;16(4):196–201. doi: 10.1097/JSA.0b013e31818cdb82. [DOI] [PubMed] [Google Scholar]
- 67.Eivazzadeh-Keihan R., Asgharnasl S., Bani M. S., et al. Magnetic copper ferrite nanoparticles functionalized by aromatic polyamide chains for hyperthermia applications. Langmuir . 2021;37(29):8847–8854. doi: 10.1021/acs.langmuir.1c01251. [DOI] [PubMed] [Google Scholar]
- 68.Krishnan R., Park S., Eckstein F., Ateshian G. A. Inhomogeneous cartilage properties enhance superficial interstitial fluid support and frictional properties, but do not provide a homogeneous state of stress. Journal of Biomechanical Engineering . 2003;125(5):569–577. doi: 10.1115/1.1610018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Yousefi A.-M., Hoque M. E., Prasad R. G. S. V., Uth N. Current strategies in multiphasic scaffold design for osteochondral tissue engineering: a review. Journal of Biomedical Materials Research Part A . 2015;103(7):2460–2481. doi: 10.1002/jbm.a.35356. [DOI] [PubMed] [Google Scholar]
- 70.Correia C. R., Reis R. L., Mano J. F. Engineering Mineralized and Load Bearing Tissues . Springer; 2015. Multiphasic, multistructured and hierarchical strategies for cartilage regeneration; pp. 143–160. [DOI] [PubMed] [Google Scholar]
- 71.Liu Y., Peng L., Li L., et al. 3D-bioprinted BMSC-laden biomimetic multiphasic scaffolds for efficient repair of osteochondral defects in an osteoarthritic rat model. Biomaterials . 2021;279:p. 121216. doi: 10.1016/j.biomaterials.2021.121216. [DOI] [PubMed] [Google Scholar]
- 72.Kon E., Roffi A., Filardo G., Tesei G., Marcacci M. Scaffold-based cartilage treatments: with or without cells? A systematic review of preclinical and clinical evidence. Arthroscopy: The Journal of Arthroscopic & Related Surgery . 2015;31(4):767–775. doi: 10.1016/j.arthro.2014.11.017. [DOI] [PubMed] [Google Scholar]
- 73.Keeney M., Lai J. H., Yang F. Recent progress in cartilage tissue engineering. Current Opinion in Biotechnology . 2011;22(5):734–740. doi: 10.1016/j.copbio.2011.04.003. [DOI] [PubMed] [Google Scholar]
- 74.Zhou X., Tenaglio S., Esworthy T., et al. Three-dimensional printing biologically inspired DNA-based gradient scaffolds for cartilage tissue regeneration. ACS Applied Materials & Interfaces . 2020;12(29):33219–33228. doi: 10.1021/acsami.0c07918. [DOI] [PubMed] [Google Scholar]
- 75.Kisiday J., Jin M., Kurz B., et al. Self-assembling peptide hydrogel fosters chondrocyte extracellular matrix production and cell division: implications for cartilage tissue repair. Proceedings of the National Academy of Sciences . 2002;99(15):9996–10001. doi: 10.1073/pnas.142309999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Ahearne M., Yang Y., Liu K. K. Mechanical characterisation of hydrogels for tissue engineering applications. Topics in tissue Engineering . 2008;4(12):1–16. [Google Scholar]
- 77.Mohabatpour F., Karkhaneh A., Sharifi A. M. A hydrogel/fiber composite scaffold for chondrocyte encapsulation in cartilage tissue regeneration. RSC advances . 2016;6(86):83135–83145. doi: 10.1039/C6RA15592H. [DOI] [Google Scholar]
- 78.Ma F., Ge Y., Liu N., Pang X., Shen X., Tang B. In situ fabrication of a composite hydrogel with tunable mechanical properties for cartilage tissue engineering. Journal of Materials Chemistry B . 2019;7(15):2463–2473. doi: 10.1039/C8TB01331D. [DOI] [PubMed] [Google Scholar]
- 79.Lee K. B., Wang V. T., Chan Y. H., Hui J. H. A novel, minimally-invasive technique of cartilage repair in the human knee using arthroscopic microfracture and injections of mesenchymal stem cells and hyaluronic acid—a prospective comparative study on safety and short-term efficacy. Annals, Academy of Medicine, Singapore . 2012;41(11):511–517. [PubMed] [Google Scholar]
- 80.de Girolamo L., Ragni E., Cucchiarini M., van Bergen C. J. A., Hunziker E. B., Chubinskaya S. Cells, soluble factors and matrix harmonically play the concert of allograft integration. Knee surgery, sports traumatology, arthroscopy . 2019;27(6):1717–1725. doi: 10.1007/s00167-018-5182-1. [DOI] [PubMed] [Google Scholar]
- 81.Hurley E. T., Murawski C. D., Paul J., et al. Osteochondral autograft: proceedings of the international consensus meeting on cartilage repair of the ankle. Foot & Ankle International . 2018;39(1_supplement):28S–34S. doi: 10.1177/1071100718781098. [DOI] [PubMed] [Google Scholar]
- 82.Euler S., Kralinger F. S., Hengg C., Wambacher M., Blauth M. Allograft augmentation in proximal humerus fractures. Operative Orthopädie und Traumatologie . 2016;28(3):153–163. doi: 10.1007/s00064-016-0446-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Shekouhi N., DD B. M., Kaeley D. K., Goel V. K. Finite element based test protocol to evaluate the effect of distraction on growth rods spanning over multiple spinal segments for pediatric scoliosis patients. ORS Annual Meeting . 2020.
- 84.Snow M. The Illustrative Book of Cartilage Repair . Springer; 2021. The illustrative osteochondral allograft-based cartilage repair; pp. 219–240. [DOI] [Google Scholar]
- 85.Lavender C., Sina Adil S. A., Singh V., Berdis G. Autograft cartilage transfer augmented with bone marrow concentrate and allograft cartilage extracellular matrix. Arthroscopy techniques . 2020;9(2):e199–e203. doi: 10.1016/j.eats.2019.09.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Li L., Yu F., Zheng L., et al. Natural hydrogels for cartilage regeneration: modification, preparation and application. Journal of orthopaedic translation . 2019;17:26–41. doi: 10.1016/j.jot.2018.09.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Levato R., Webb W. R., Otto I. A., et al. The bio in the ink: cartilage regeneration with bioprintable hydrogels and articular cartilage-derived progenitor cells. Acta biomaterialia . 2017;61:41–53. doi: 10.1016/j.actbio.2017.08.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Shelat R., Bhatt L. K., Paunipagar B., Kurian T., Khanna A., Chandra S. Regeneration of hyaline cartilage in osteochondral lesion model using L‐lysine magnetic nanoparticles labeled mesenchymal stem cells and their in vivo imaging. Journal of Tissue Engineering and Regenerative Medicine . 2020;14(11):1604–1617. doi: 10.1002/term.3120. [DOI] [PubMed] [Google Scholar]
- 89.Roshandel M., Dorkoosh F. Cardiac tissue engineering, biomaterial scaffolds, and their fabrication techniques. Polymers for Advanced Technologies . 2021;32(6):2290–2305. doi: 10.1002/pat.5273. [DOI] [Google Scholar]
- 90.Abasian P., Ghanavati S., Rahebi S., Nouri Khorasani S., Khalili S. Polymeric nanocarriers in targeted drug delivery systems: a review. Polymers for Advanced Technologies . 2020;31(12):2939–2954. doi: 10.1002/pat.5031. [DOI] [Google Scholar]
- 91.Shepard J. A., Huang A., Shikanov A., Shea L. D. Balancing cell migration with matrix degradation enhances gene delivery to cells cultured three-dimensionally within hydrogels. Journal of controlled release . 2010;146(1):128–135. doi: 10.1016/j.jconrel.2010.04.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Chen C.-H., Lin Y. S., Fu Y. C., et al. Electromagnetic fields enhance chondrogenesis of human adipose-derived stem cells in a chondrogenic microenvironment in vitro. Journal of Applied Physiology . 2013;114(5):647–655. doi: 10.1152/japplphysiol.01216.2012. [DOI] [PubMed] [Google Scholar]
- 93.Xu H.-Y., Gu N. Magnetic responsive scaffolds and magnetic fields in bone repair and regeneration. Frontiers of Materials Science . 2014;8(1):20–31. doi: 10.1007/s11706-014-0232-1. [DOI] [Google Scholar]
- 94.Jiang P., Zhang Y., Zhu C., Zhang W., Mao Z., Gao C. Fe3O4/BSA particles induce osteogenic differentiation of mesenchymal stem cells under static magnetic field. Acta Biomaterialia . 2016;46:141–150. doi: 10.1016/j.actbio.2016.09.020. [DOI] [PubMed] [Google Scholar]
- 95.Abasian P., Radmansouri M., Habibi Jouybari M., et al. Incorporation of magnetic NaX zeolite/DOX into the PLA/chitosan nanofibers for sustained release of doxorubicin against carcinoma cells death in vitro. International journal of biological macromolecules . 2019;121:398–406. doi: 10.1016/j.ijbiomac.2018.09.215. [DOI] [PubMed] [Google Scholar]
- 96.Przekora A. Current trends in fabrication of biomaterials for bone and cartilage regeneration: materials modifications and biophysical stimulations. International journal of molecular sciences . 2019;20(2):p. 435. doi: 10.3390/ijms20020435. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Van de Walle A., Wilhelm C., Luciani N. 3D magnetic stem cell aggregation and bioreactor maturation for cartilage regeneration. JoVE . 2017;(122) doi: 10.3791/55221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Tacchetti C., Tavella S., Dozin B., Quarto R., Robino G., Cancedda R. Cell condensation in chondrogenic differentiation. Experimental Cell Research . 1992;200(1):26–33. doi: 10.1016/S0014-4827(05)80067-9. [DOI] [PubMed] [Google Scholar]
- 99.Singh P., Schwarzbauer J. E. Fibronectin matrix assembly is essential for cell condensation during chondrogenesis. Journal of cell science . 2014;127(20):4420–4428. doi: 10.1242/jcs.150276. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Brady M. A., Talvard L., Vella A., Ethier C. R. Bio-inspired design of a magnetically active trilayered scaffold for cartilage tissue engineering. Journal of Tissue Engineering and Regenerative Medicine . 2017;11(4):1298–1302. doi: 10.1002/term.2106. [DOI] [PubMed] [Google Scholar]
- 101.Yang W., Zhu P., Huang H., et al. Functionalization of novel theranostic hydrogels with kartogenin-grafted USPIO nanoparticles to enhance cartilage regeneration. ACS Applied Materials & Interfaces . 2019;11(38):34744–34754. doi: 10.1021/acsami.9b12288. [DOI] [PubMed] [Google Scholar]
- 102.Saldanha K. J., Doan R. P., Ainslie K. M., Desai T. A., Majumdar S. Micrometer-sized iron oxide particle labeling of mesenchymal stem cells for magnetic resonance imaging-based monitoring of cartilage tissue engineering. Magnetic Resonance Imaging . 2011;29(1):40–49. doi: 10.1016/j.mri.2010.07.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Chen Z., Yan C., Yan S., et al. Non-invasive monitoring of in viv ohydrogel degradation and cartilage regeneration by multiparametric MR imaging. Theranostics . 2018;8(4):1146–1158. doi: 10.7150/thno.22514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Farzaneh S., Hosseinzadeh S., Samanipour R., Hatamie S., Ranjbari J., Khojasteh A. Fabrication and characterization of cobalt ferrite magnetic hydrogel combined with static magnetic field as a potential bio-composite for bone tissue engineering. Journal of Drug Delivery Science and Technology . 2021;64:p. 102525. doi: 10.1016/j.jddst.2021.102525. [DOI] [Google Scholar]
- 105.Satarkar N. S., Zhang W., Eitel R. E., Hilt J. Z. Magnetic hydrogel nanocomposites as remote controlled microfluidic valves. Lab on a Chip . 2009;9(12):1773–1779. doi: 10.1039/b822694f. [DOI] [PubMed] [Google Scholar]
- 106.Huang J., Jia Z., Liang Y., et al. Pulse electromagnetic fields enhance the repair of rabbit articular cartilage defects with magnetic nano-hydrogel. RSC Advances . 2020;10(1):541–550. doi: 10.1039/C9RA07874F. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Zhang N., Lock J., Sallee A., Liu H. Magnetic nanocomposite hydrogel for potential cartilage tissue engineering: synthesis, characterization, and cytocompatibility with bone marrow derived mesenchymal stem cells. ACS Applied Materials & Interfaces . 2015;7(37):20987–20998. doi: 10.1021/acsami.5b06939. [DOI] [PubMed] [Google Scholar]
- 108.De Santis R., Gloria A., Russo T., et al. Viscoelastic properties of rapid prototyped magnetic nanocomposite scaffolds for osteochondral tissue regeneration. Procedia CIRP . 2016;49:76–82. doi: 10.1016/j.procir.2015.07.037. [DOI] [Google Scholar]
- 109.Choi Y., Kim C., Kim H. S., Moon C., Lee K. Y. 3D printing of dynamic tissue scaffold by combining self-healing hydrogel and self-healing ferrogel. Colloids and Surfaces B: Biointerfaces . 2021;208:p. 112108. doi: 10.1016/j.colsurfb.2021.112108. [DOI] [PubMed] [Google Scholar]
- 110.Dobson J., Cartmell S. H., Keramane A., el Haj A. J. Principles and design of a novel magnetic force mechanical conditioning bioreactor for tissue engineering, stem cell conditioning, and dynamic in vitro screening. IEEE transactions on nanobioscience . 2006;5(3):173–177. doi: 10.1109/TNB.2006.880823. [DOI] [PubMed] [Google Scholar]
- 111.Kamei G., Kobayashi T., Ohkawa S., et al. Articular cartilage repair with magnetic mesenchymal stem cells. The American journal of sports medicine . 2013;41(6):1255–1264. doi: 10.1177/0363546513483270. [DOI] [PubMed] [Google Scholar]
- 112.Rodriguez-Merchan E. C. Regeneration of articular cartilage of the knee. Rheumatology international . 2013;33(4):837–845. doi: 10.1007/s00296-012-2601-3. [DOI] [PubMed] [Google Scholar]
- 113.Boopalan P., Arumugam S., Livingston A., Mohanty M., Chittaranjan S. Pulsed electromagnetic field therapy results in healing of full thickness articular cartilage defect. International orthopaedics . 2011;35(1):143–148. doi: 10.1007/s00264-010-0994-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Hoghoughi M. A., Kazemi T., Khojasteh A., et al. The effect of intervelar veloplasty under magnification (Sommerlad’s technique) without tympanostomy on middle ear effusion in cleft palate patients. BMC Pediatrics . 2021;21(1):1–6. doi: 10.1186/s12887-021-02856-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Salimi M. Late-onset hip dysplasia and multiple hip dislocation in osteogenesis imperfecta: a case report. Journal of Sports Medicine and Orthopedic Advances . 2021;1(1):23–25. [Google Scholar]
- 116.Yi H. G., Kang K. S., Hong J. M., et al. Effects of electromagnetic field frequencies on chondrocytes in 3D cell-printed composite constructs. Journal of Biomedical Materials Research Part A . 2016;104(7):1797–1804. doi: 10.1002/jbm.a.35714. [DOI] [PubMed] [Google Scholar]
- 117.Yan J., Liu C., Tu C., et al. Hydrogel-hydroxyapatite-monomeric collagen type-I scaffold with low-frequency electromagnetic field treatment enhances osteochondral repair in rabbits. Stem Cell Research & Therapy . 2021;12(1):p. 572. doi: 10.1186/s13287-021-02638-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Gil S., Mano J. F. Magnetic composite biomaterials for tissue engineering. Biomaterials science . 2014;2(6):812–818. doi: 10.1039/C4BM00041B. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
SPSS data of the participant can be requested from the authors. Please write to the corresponding author if you are interested in such data.


