Abstract
B cells secreting IL-10 functionally are recognized as functional regulatory B (Breg) cells; however, direct evidence concerning the phenotype, regulation, and functional and clinical relevance of IL-10-secreting Breg cells in humans is still lacking. Here, we demonstrate that, although IL-10 itself is anti-inflammatory, IL-10+ functional Breg cells in patients with systemic lupus erythematosus (SLE) display aggressive inflammatory features; these features shift their functions away from inducing CD8+ T cell tolerance and cause them to induce a pathogenic CD4+ T cell response. Functional Breg cells polarized by environmental factors (e.g., CPG-DNA) or directly isolated from patients with SLE mainly exhibit a CD24intCD27−CD38−CD69+/hi phenotype that is different from that of their precursors. Mechanistically, MAPK/ERK/P38-elicited sequential oncogenic c-Myc upregulation and enhanced glycolysis are necessary for the generation and functional maintenance of functional Breg cells. Consistently, strategies that abrogate the activity of ERK, P38, c-Myc, and/or cell glycolysis can efficiently eliminate the pathogenic effects triggered by functional Breg cells.
Subject terms: Inflammation, Lymphocytes
Introduction
Regulatory B (Breg) cells are now recognized as suppressive cells that support immunological tolerance1. A shared signature of all described Breg subsets is their ability to release IL-10, and in parallel, release of IL-10 is extensively used to define functional Breg cells2–4. Nevertheless, in addition to producing IL-10, Breg cells also express other molecules (e.g., PD-L1, FasL, and granzyme B), which induce additional pathological effects5–7. Thus, IL-10 is necessary, but not sufficient, for Breg cell activities. Studies have established that immature transitional CD24hiCD38hi B cells8, together with mature CD24hiCD27+ B cells9, represent important cellular sources of peripheral Breg cells, both in healthy individuals and in patients. Of note, the occurrence of a CD24hiCD38hi or CD24hiCD27+ phenotype for B cells does not necessarily reflect their functionality. In fact, in patients with autoimmune diseases, CD24hiCD38hi and CD24hiCD27+ B cells hardly process immunosuppressive functions directly8,9. Direct evidence supporting a role for functional Breg cells in the immunopathogenesis of human diseases is still lacking. Furthermore, a related issue that must be addressed in this context is whether functional Breg cells exhibit unique functions that are different from those functions that are already known and, if so, how B cells exert these effects.
Metabolic reprogramming is emerging as a crucial process for reshaping immune cell differentiation and function10,11. Oxidative phosphorylation (OXPHOS) is employed by naïve and memory B cells to supply the basal energy needs of metabolic quiescence12. In contrast, high-strength glycolysis together with aggressive OXPHOS controls the antibody secretion of effector B cells (also termed plasma cells), and this process depends on active glucose and amino acid uptake machinery13,14. Notably, although not directly related to B cells, glycolysis is also considered an efficient activator of the inflammatory response15,16, whereas OXPHOS has been implicated in the anti-inflammatory response and immune tolerance17,18. To date, the precise metabolic programs of IL-10-secreting functional Breg cells under pathological conditions are not known. In other words, evaluating the metabolic programs of functional Breg cells in pathological conditions is crucial for understanding the polarization and functional status of these cells in immunopathogenesis.
Systemic lupus erythematosus (SLE) is a potentially fatal autoimmune disease characterized by extensive inflammation and tissue damage that can affect any part of the body. In patients with SLE, defects in suppressive cell functions are necessary for enhanced inflammation and the active stage of disease19,20. It has been assumed that Breg cells are a protective factor that impedes the inflammatory response based on their ability to produce IL-101–4. However, by probing the phenotypic features and functions of IL-10-secreting B cells in patients with SLE, we herein report that functional Breg cells mainly serve as a pathogenic factor but do not serve as a suppressor of inflammation. Functional Breg cells triggered by SLE pathological factors (e.g., CPG-DNA) display a previously unrecognized CD24intCD27−CD38−CD69+/hi phenotype and simultaneously produce high amounts of inflammatory mediators, and this process is dominated by MAPK/ERK/P38 signal-elicited sequential oncogenic c-Myc upregulation and enhanced glycolysis. More importantly, we demonstrate that these inflammatory features repurpose Breg cell functions away from those inducing CD8+ T cell tolerance and toward those inducing an inflammatory TH cell response. Accordingly, inhibiting the activity of ERK, P38, c-Myc, or the glycolytic enzyme PFKFB3 in functional Breg cells from patients with SLE effectively impairs the polarization of pathogenic inflammatory TH cell subsets.
Results
Polarization of unrecognized functional Breg cells in human SLE correlates with disease progression
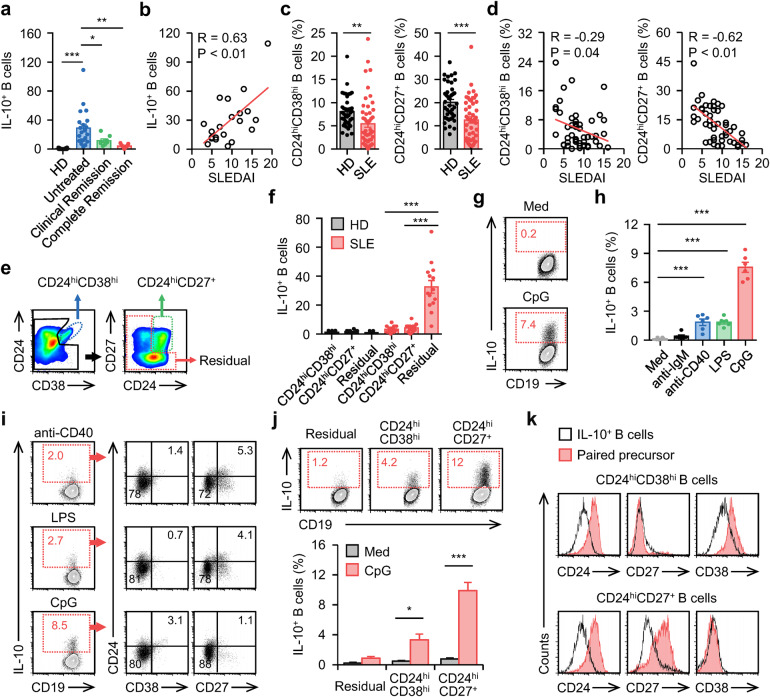
We used an ELISpot detection system to analyze IL-10 production by B cells purified from blood samples of 20 healthy donors, 21 untreated SLE patients, 8 SLE patients with clinical remission, and 7 SLE patients with complete remission (Supplementary Table 1). IL-10 was hardly secreted by B cells from the blood of healthy donors (Fig. 1a). Unexpectedly, this cytokine was spontaneously produced by a fraction of B cells from the blood of untreated SLE patients (Fig. 1a), and the density of these IL-10+ B cells positively correlated with disease progression (Fig. 1b and Supplementary Fig. 1a) but declined in the blood of SLE patients with clinical remission or complete remission (Fig. 1a). Thus, IL-10+ functional Breg cells accumulated in the blood of SLE patients with active disease. CD24hiCD38hi and CD24hiCD27+ B cells are considered the major cellular sources of peripheral Breg cells in humans8,9. However, inconsistent with the above observations, the proportions of CD24hiCD38hi and CD24hiCD27+ B cells in the blood of SLE patients were profoundly decreased (Fig. 1c and Supplementary Fig. 1b), and these decreases were also positively correlated with active disease (Fig. 1d and Supplementary Fig. 1c), suggesting that functional Breg cells exhibit a phenotype unrelated to the CD24hiCD38hi or CD24hiCD27+ phenotype. To test this hypothesis, we sorted peripheral CD24hiCD38hi and CD24hiCD27+ B cells from healthy donors and SLE patients (Fig. 1e and Supplementary Fig. 1d) and probed their ability to spontaneously secrete IL-10. In all FACS-sorted CD24hiCD38hi and CD24hiCD27+ B cells, IL-10 signals were rarely detected (Fig. 1f). By comparison, significantly higher frequencies of IL-10 signals were observed in residual B cells from SLE patients (Fig. 1f). This prompted us to further investigate the environmental factors and cellular sources that contribute to the polarization of functional Breg cells in SLE patients.
Fig. 1. Polarization of IL-10-producing Breg cells in human SLE.
a Enzyme-linked immunospot (ELISpot) detection of IL-10 production by circulating B cells from 20 healthy donors (HD), 21 untreated SLE patients, 8 clinical remission SLE patients, and 7 complete remission SLE patients. b Associations of circulating IL-10+ B cells with patients’ SLE disease activity index (SLEDAI) (n = 21). c FACS analysis of CD24hiCD38hi and CD24hiCD27+ B cells from healthy donors (n = 40) and untreated SLE patients. d Associations of circulating CD24hiCD38hi and CD24hiCD27+ B cells with patients’ SLEDAI (n = 49). e CD24hiCD38hi, CD24hiCD27+, and residual B cells from HD and untreated SLE patients were sorted by FACS. f IL-10 production by these cells were detected using ELISpot (n = 14). g–i Total B cells from HD were cultured in medium or treated with an anti-IgM antibody, anti-CD40 antibody, LPS (h, i), or CPG-DNA ODN (CPG) (g–i) for 3 d. IL-10+ (g, h), CD24hiCD38hi (i), and CD24hiCD27+ (i) B cells were detected by FACS (n = 6). j, k CD24hiCD38hi, CD24hiCD27+, and residual B cells from HD were cultured in medium or were treated with CPG-DNA for 3 d. IL-10+ (j, k), CD24hiCD38hi (k), and CD24hiCD27+ (k) B cells were detected by FACS (n = 5). Data were acquired from more than four independent experiments and shown as means ± SEM. Pearson’s correlation analysis for b and d. Significance was examined with one-way ANOVA test (a, f, h) or Student’s t test (c, j), *p < 0.05, **p < 0.01, ***p < 0.001
It is noteworthy that elevated levels of plasma DNA enriched in hypomethylated CPGs are a biomarker of active SLE21. Therefore, we determined whether CPG-DNA promotes the polarization of functional Breg cells. As shown in Fig. 1g, exposing total B cells from healthy donors to CPG-DNA for 3 days led to marked polarization of functional Breg cells. Other environmental factors, including anti-IgM [B cell receptor (BCR)-triggering] antibodies, anti-CD40 [a B cell costimulatory signal], and LPS (a TLR4 agonist)22–24, also triggered polarization of IL-10+ functional Breg cells, although to a lesser extent (Fig. 1h). In support of in vivo observations (Fig. 1f), the functional Breg cells generated in vitro mainly exhibited a unique CD38−CD24intCD27− phenotype (Fig. 1i). Furthermore, we also assessed the influences of CPG-DNA on FACS-sorted CD24hiCD38hi and CD24hiCD27+ B cells, as well as residual B cells. Unexpectedly, CD24hiCD38hi and CD24hiCD27+ B cells, but not the residual B cells, represented major cell populations that could be differentiated into functional Breg cells (Fig. 1j). Notably, the functional Breg cells polarized from CD24hiCD38hi or CD24hiCD27+ B cells displayed a phenotype unrelated to CD24hi, CD27+, and/or CD38hi (Fig. 1k), which is consistent with the finding that, in the blood of SLE patients, increasing frequencies of functional Breg cells were accompanied by decreasing ratios of CD24hiCD38hi and CD24hiCD27+ B cells (Fig. 1a, c).
Functional Breg cells exhibit a unique CD25+CD69+/hiCD80+CD86+/hi phenotype that differs from that of their precursors
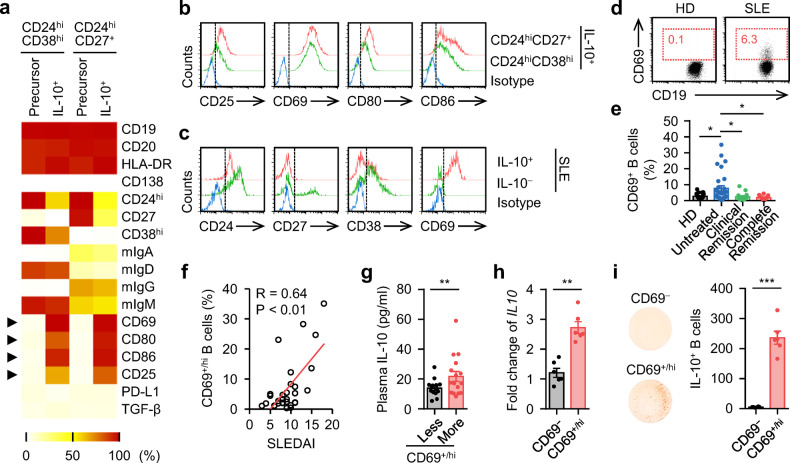
We next investigated the phenotypic characteristics of functional Breg cells. Generally, CPG-DNA-induced Breg cells, as well as their precursors CD24hiCD38hi and CD24hiCD27+ B cells, displayed a non-plasma cell phenotype with substantial CD19, CD20, and HLA-DR but rare CD138 expression (Fig. 2a). Compared with CD24hiCD38hi B cells, CD24hiCD27+ B cells displaying a memory phenotype and expressed higher frequencies of membrane immunoglobulin A (mIgA) and mIgG but lower levels of mIgD and mIgM. Interestingly, unlike the expression patterns of CD24, CD27, and CD38, the expression profiles of mIg in CD24hiCD38hi and CD24hiCD27+ B cells remained virtually unchanged after their differentiation into functional Breg cells (Fig. 2a), indicating that the generation of functional Breg cells is a process unrelated to Ig class-switch recombination. Notably, although CPG-DNA-induced Breg cells lost the conventional phenotypic features related to their precursors, these cells developed a unique CD25+CD69+/hiCD80+CD86+ phenotype (Fig. 2a, b). These data suggest that functional B cells purified from SLE patients directly may exhibit a phenotype that is related to CD24intCD27−CD38−CD69+/hi. In support, IL-10+ B cells from blood of SLE patients did displayed a CD24dim/−CD27lo/−CD38lo/−CD69+/hi phenotype (Fig. 2c and Supplementary Fig. 2a).
Fig. 2. Phenotypic characteristics of IL-10-secreting Breg cells in SLE.
a, b FACS analysis of the phenotypic characteristics of the untreated CD24hiCD38hi and CD24hiCD27+ B cells (precursor), as well as IL-10+ cells derived from CD24hiCD38hi and CD24hiCD27+ B cells by CPG-DNA induction (a). Representative histogram of CD25, CD69, CD80, and CD86 expression were shown in b (n = 5). c Expression of CD24, CD27, CD38, and CD69 in IL-10+ B cells purified from untreated SLE patients was detected by FACS (n = 6). d, e FACS analysis of CD69+/hi B cells from healthy donors and untreated SLE patients. Representative FACS plots were shown in d. The portions of CD69+/hi B cells relative to total B cells from 21 healthy donors, 30 untreated SLE patents, 14 clinical remission SLE patients, and 12 complete remission SLE patients were shown in e. f, g Associations of circulating CD69+/hi B cells with disease progression (f) and plasma level of IL-10 from untreated SLE patients (g) (n = 30). h, i Expression of IL-10 by CD69− and CD69+/hi B cells isolated from untreated SLE patients were detected by PCR (h) and ELISpot (i), respectively (each n = 6). Data were acquired from more than four independent experiments and shown as means ± SEM. Pearson’s correlation analysis for f. Significance was determined with one-way ANOVA test (e), Student’s t test (g–i). *p < 0.05, **p < 0.01, ***p < 0.001
Based on the above-mentioned findings, we proposed that CD69+/hi B cells were more potent in producing IL-10 functionally. In fact, significantly higher frequencies of CD69+/hi B cells were detected in the blood of untreated SLE patients than in blood from healthy donors (Fig. 2d, e), and the proportion of these cells positively correlated with disease progression in patients (Fig. 2f and Supplementary Fig. 2b). Of note, increased levels of plasma IL-10 in patients with increased frequencies of peripheral CD69+/hi B cells were detected (Fig. 2g) and most CD69+/hi B cells in blood of SLE patients were CD24lo/−CD27lo/−CD38lo/− (Supplementary Fig. 2c). We subsequently sorted the CD69+/hi B cells from the blood of SLE patients. In agreement with our hypothesis, CD69+/hi B cells had more potential to express IL-10 mRNA (Fig. 2h). Indeed, the CD69+/hi B cells displayed a 50-fold increase in IL-10 production compared with the CD69− population (Fig. 2i).
Functional Breg cells co-polarize with inflammatory B cell populations
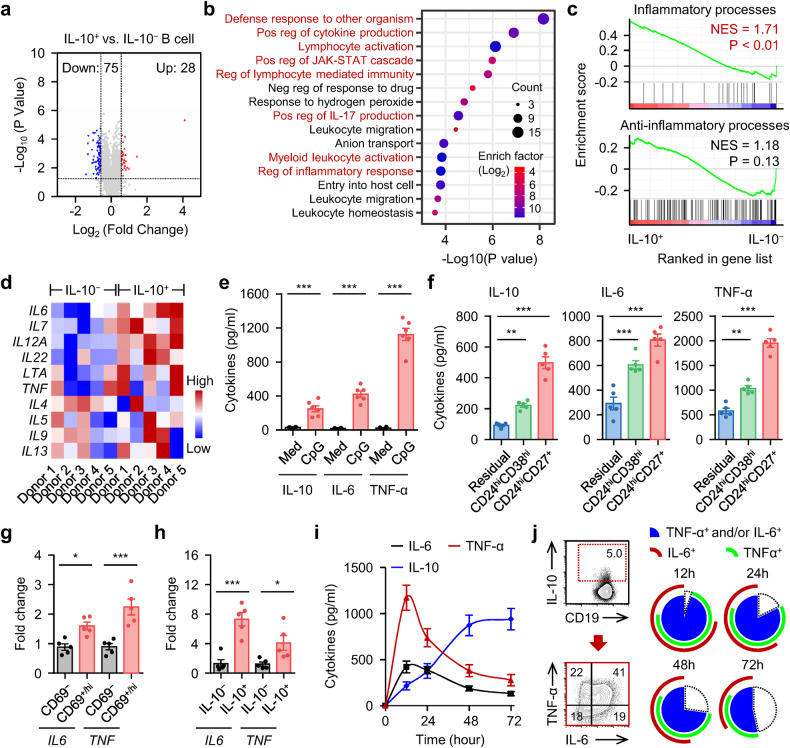
To further probe the characteristics of functional Breg cells, we analyzed the transcriptional profiles of those cells with RNA sequencing (GSE50895). We identified 103 genes upregulated or downregulated at least 1.5-fold in functional Breg cells (IL-10+ B cells) and annotated these genes using Gene Ontology (GO) (Fig. 3a, b). Surprisingly, although IL-10 itself is classically considered anti-inflammatory, activation of pathways related to the anti-inflammatory response was completely lacking (Fig. 3b). Instead, pathways related to inflammatory processes, including cytokine production, the defense response against organisms, and the activation of lymphocytes or myeloid cells, were intensively enriched (Fig. 3b), which was further confirmed by Gene Set Enrichment Analysis (GSEA) (Fig. 3c and Supplementary Fig. 3a, b). In support of these findings, significantly higher IL6, IL7, IL12A, IL22, LTA, and TNF levels were detected in IL-10+ B cells than in their IL-10− counterparts (Fig. 3d). In contrast, irregular variations in the levels of the anti-inflammatory molecules IL4, IL5, IL9, or IL13 were detected (Fig. 3d).
Fig. 3. Inflammatory features of IL-10-secreting Breg cells in SLE.
a The changes of genes in IL-10+ B cells versus IL-10− B cells (GSE50895) were shown in volcano plot. The P value of each gene was calculated using DESeq2 and adjusted with Benjamini-Hochberg false discovery rate correction (n = 5). b Functional annotation of 103 differential gene expression listed in a were analyzed by DAVID tool. The top 15 enrichment GO terms are listed. c GSEA of inflammatory processes (WP530) and anti-inflammatory processes (GO: 0050728) in IL-10+ B cells versus IL-10− B cells. NES, normalized enrichment score. d Heat map displaying inflammatory and anti-inflammatory cytokine expression in IL-10+ B cells versus IL-10− B cells (n = 5). e, f Purified total B cells (e), CD24hiCD38hi, CD24hiCD27+, and residual B cells (f) from HD were cultured in medium or were treated with CPG-DNA for 12 h. IL-10, IL-6, and TNF-α production were detected by ELISA (n = 6). g, h Expression of IL6 and TNF in CD69− and CD69+/hi B cells (g), or in IL-10− and IL-10+ B cells (h) isolated from untreated SLE patients were detected by real-time PCR (n = 5). i, j Total B cells purified from HD were cultured in medium or treated with CPG-DNA. Dynamics of IL-10, IL-6, and TNF-α production was measured over the indicated times by ELISA (i). Frequencies of TNF-α+ and/or IL-6+ and TNF-α−IL-6− B cells relative to total B-cell were examined by FACS (j) (n = 6 for i; n = 5 for j). Data were acquired from more than four independent experiments and shown as means ± SEM. Significance was determined with Student’s t-test (e, g, h) or one-way ANOVA test (f). *p < 0.05, **p < 0.01, ***p < 0.001
To ascertain whether functional Breg cells and inflamed B cells are interrelated, we tested the inflammatory profiles of functional Breg cells generated in vitro. Reliably, exposure to CPG-DNA, a factor contributing to functional Breg cell generation, also resulted in substantial production of TNF-α and IL-6 by these cells (Fig. 3e). Analogously, CD24hiCD38hi and CD24hiCD27+ B cells, two major groups of functional Breg cell precursors, secreted significantly more IL-6 and TNF-α than other B cell populations (Fig. 3f). In parallel, CD69+/hi B cells and IL-10+ B cells, both directly isolated from the blood of SLE patients, expressed higher levels of IL6 and TNF than other B cell populations (Fig. 3g, h). Furthermore, measurement of the cytokines produced by B cells exposed to CPG-DNA over time revealed rapid accumulation of TNF-α and IL-6 in culture supernatants, with a maximum or a plateau reached within 24 h, followed by a gradual decline (Fig. 3i). In contrast, accumulation of IL-10 was delayed, but a sustained elevation did last for at least 3 days after stimulation (Fig. 3i). Given these results, we concluded that IL-10+ B cells were IL-6+ and/or TNF-α+ at the early measurement time point, and these cells, at the subsequent measurement time points, gradually and partially lost the ability to secrete these cytokines (Fig. 3j).
Metabolic program that acts in differentiation of functional Breg cells in SLE patients
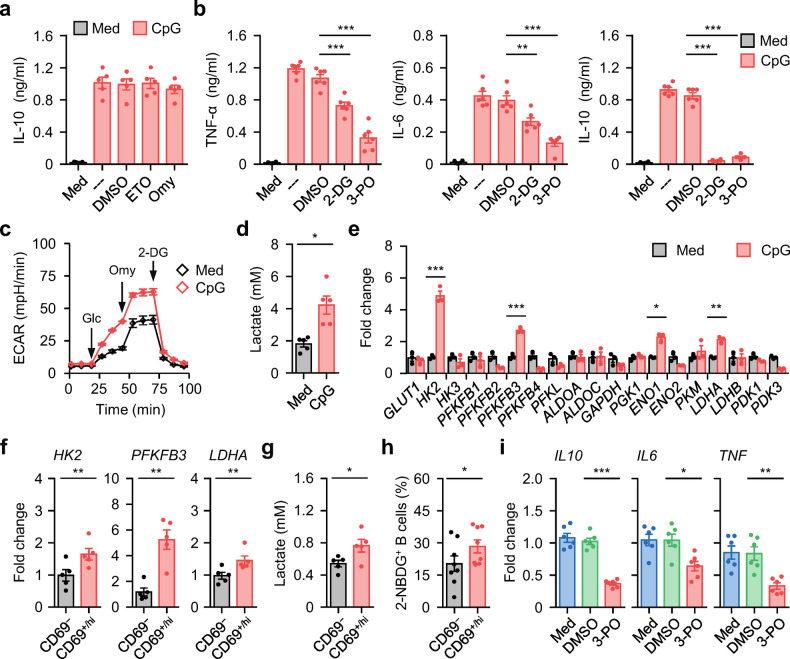
Metabolic reprogramming is essential for both lineage differentiation and the function of immune cells10,11. Notably, regulatory T (Treg) cells depend on OXPHOS and fatty acid metabolism11,17. To investigate whether such a metabolic program is also employed by functional Breg cells, we exposed CPG-DNA-treated B cells to etomoxir (ETO), a widely used small-molecule inhibitor of fatty acid oxidation25, or to oligomycin (Omy), an inhibitor of ATP synthase;26 however, we detected negligible changes in CPG-DNA-elicited functional Breg cell polarization by measuring the IL-10 concentration (Fig. 4a and Supplementary Fig. 4a, b), suggesting that OXPHOS and fatty acid metabolism are not involved.
Fig. 4. Metabolic reprogramming involved in inflammatory Breg cell induction.
a, b Total B cells from healthy donors were cultured in medium or pretreated with DMSO, inhibitors against fatty acid oxidation (Etomoxir, ETO) (a), oxidative phosphorylation (Oligomycin, Omy) (a), or glycolysis (2-DG; 3-PO) (b). Thereafter, the cells were incubated in the presence or absence of CPG-DNA. Cytokine production were detected by ELISA (n = 5 for a; n = 6 for b). c–e Total B cells purified from healthy donors were cultured in medium or treated with CPG-DNA for 3 d. Extracellular acidification (ECAR) (c), accumulated lactate production (d), and expression of glycolytic enzymes (e) were measured by seahorse analyzer, lactate assay kit, and real-time PCR, respectively (n = 3 for c and e; n = 5 for d). f–h Expression of key rate-limiting glycolytic enzymes (f), accumulated lactate production (g), and capabilities of glucose incorporation (2-NBDG+) (h) by CD69− and CD69+/hi B cells purified from untreated SLE patients were examined by real-time PCR (f), lactate assay kit (g), and FACS (h), respectively (n = 5 for f and g; n = 8 for h). i Purified CD69+/hi B cells from blood of untreated SLE patients were cultured in medium or in the presence of DMSO, or 3-PO for 12 h. Expression of IL10, IL6, and TNF were examined by real-time PCR. Data were acquired from more than four independent experiments and shown as means ± SEM. Significance was determined with one-way ANOVA test (a, b, i) or Student’s t test (d–h). *p < 0.05, **p < 0.01, ***p < 0.001
Considering the requirement of glycolysis during the inflammatory response, CPG-DNA-treated B cells were incubated with 2-deoxyglucose (2-DG), a glucose analog that inhibits glucose incorporation27, or with 3-(3-pyridinyl)−1-(4-pyridinyl)−2-propen-1-one (3-PO), an inhibitor of the glycolytic enzyme PFKFB328. Interestingly, cells that received either treatment not only exhibited decreased production of the inflammatory molecules TNF-α and IL-6 but also completely lost their ability to polarize into functional Breg cells (Fig. 4b and Supplementary Fig. 4a, b). Thus, increased glycolysis may represent a cardinal feature during functional Breg cell polarization. Consistent with this finding, after B cells were exposed to CPG-DNA, there was a rapid increase in the extracellular acidification rate (ECAR) (Fig. 4c and Supplementary Fig. 4c) and the extracellular lactate concentration (Fig. 4d). We further established that B cells undergoing CPG-DNA stimulation expressed significantly increased expression of the glycolytic enzymes HK2, PFKFB3, and LDHA (Fig. 4e). In line with this, CD69+/hi B cells, as well as IL-10+ B cells, purified from SLE patients showed a similar expression pattern of glycolytic enzymes (Fig. 4f and Supplementary Fig. 4d), and these cells cultured ex vivo produced significantly more lactate (Fig. 4g) and displayed a greater capacity to incorporate the fluorescent glucose analog 2-(N-[7-nitrobenz-2-oxa-1,3-diazol-4-yl] amino)−2-deoxyglucose (2-NBDG) (Fig. 4h and Supplementary Fig. 4e). Supporting our hypothesis, suppression of glycolysis in CD69+/hi B cells ex vivo by 3-PO successfully impeded their Breg cell signature and inflammatory features (Fig. 4i).
MAPK-mediated c-Myc signaling is essential for glycolysis-triggered functional Breg cells
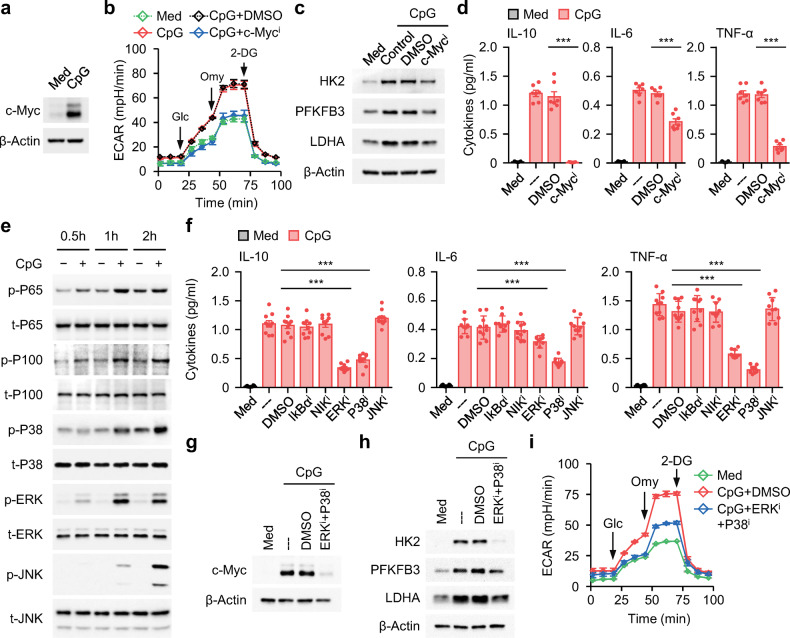
It is generally thought that increased AKT/mTOR signaling induces HIF-1α activity and subsequent glycolysis29. Indeed, exposing B cells to CPG-DNA triggered marked AKT/mTOR activation and HIF-1α upregulation (Supplementary Fig. 5a), which suggested the involvement of the AKT-mTOR-HIF-1α pathway during glycolysis-elicited functional Breg cell polarization. However, this assumption was rapidly refuted by further observation that inhibiting the activity of either mTOR or HIF-1α in CPG-DNA-treated B cells did not affect the increase in the ECAR (Supplementary Fig. 5b), the upregulation of the glycolytic enzymes HK2, PFKFB3, and LDHA (Supplementary Fig. 5c, d), the production of the inflammatory mediators IL-6 and TNF-α, or the polarization of functional Breg cells (Supplementary Fig. 5e). Notably, it is also reported that c-myc can regulate energy metabolism by directly activating genes involved in glycolysis, glutamine metabolism and mitochondrial biogenesis30. Interestingly, via GSEA, we noted that the c-Myc signaling pathway was intensively enriched in IL-10+ B cells (Supplementary Fig. 5f, g). Additionally, c-Myc was robustly upregulated in B cells exposed to CPG-DNA (Fig. 5a). In agreement with these findings, suppressing c-Myc signaling in either CPG-DNA-treated B cells or IL-10+ B cells purified from blood of SLE patients efficiently decreased glycolysis (Fig. 5b, c and Supplementary Fig. 5h), as well as the subsequent inflammatory response and functional Breg cell generation (Fig. 5d and Supplementary Fig. 5i).
Fig. 5. Signals and transcription factors that triggered B-cell glycolysis and inflammatory Breg cell differentiation.
a Total B cells purified from healthy donors were cultured in medium or treated with CPG-DNA. c-Myc expression was examined by immunoblotting (n = 3). b–d Total B cells purified from healthy donors were cultured in medium or pretreated with DMSO, or inhibitor against c-Myc signals. Thereafter, the cells were cultured in the presence or absence of CPG-DNA. Basal extracellular acidification (b), expression of glycolytic enzymes (c), and cytokine secretion (d) were detected by seahorse analyzer, immunoblotting, and ELISA, respectively (n = 3 for b and c; n = 7 for d). e Purified total B cells from healthy donors were cultured in medium or stimulated with CPG-DNA. Activation of MAP kinase and NF-κB signals were determined by immunoblotting (n = 3). f–i Total B cells purified from healthy donors were cultured in medium or pretreated with DMSO, or inhibitor against MAP kinase (f–i) or NF-κB signals (f). Thereafter, the cells were cultured in the presence or absence of CPG-DNA. Cytokine production (f), expression of c-Myc (g) and glycolytic enzymes (h), and extracellular acidification (i) were detected by immunoblotting, seahorse analyzer, and ELISA, respectively (n = 10 for f; n = 3 for g–i). Data were acquired from more than four independent experiments and shown as means ± SEM. Significance was determined with one-way ANOVA test (d, f). ***p < 0.001
To further explore the mechanisms involved in triggering functional Breg cells, we analyzed the activation kinetics of the MAPK and canonical and noncanonical NFκB pathways. After B cells were exposed to CPG-DNA, all the signaling pathways analyzed were rapidly activated, although to varying extents (Fig. 5e). Notably, using inhibitors to block the signal transduction of ERK and P38 in B cells effectively impaired the CPG-DNA-triggered inflammatory response and functional Breg cell generation, whereas abolishing the phosphorylation of JNK or the NF-κB subunits P65 and P100 had only a marginal effect (Fig. 5f). In support of the above-mentioned findings that c-Myc-mediated glycolysis was a key upstream event for functional Breg cell generation, we established that combined inhibition of ERK and P38 in B cells effectively abrogated CPG-DNA-mediated c-Myc upregulation (Fig. 5g) and subsequent glycolysis (Fig. 5h, i).
Targeting c-Myc-elicited glycolysis abolishes the pathogenic response induced by functional Breg cells
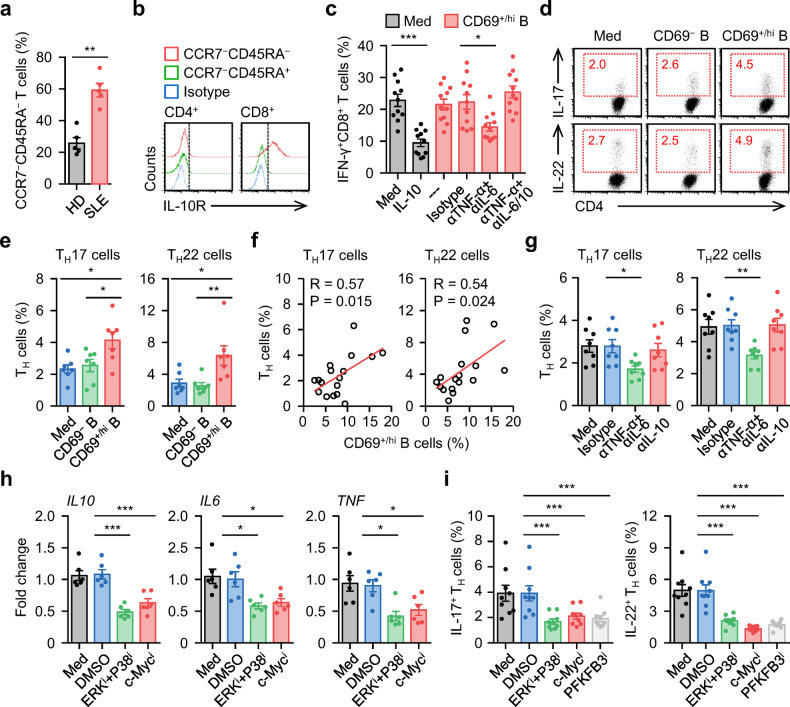
Given that functional Breg cells (IL-10+ B cells) were co-polarized with inflammatory B cells and potently correlated with the disease progression of SLE patients (Figs. 1–3), we determined whether inflammatory features repurposed Breg cell function away from immune tolerance and toward a pathogenic response. In the blood of SLE patients, most T cells mainly exhibited a CCR7−CD45RA− effector memory phenotype (Fig. 6a). Analyzing the IL-10 receptor (IL-10R) in T cells from SLE blood revealed that this receptor was primarily expressed by CD8+ but not CD4+ T cells (Fig. 6b), implying that IL-10 mainly affects CD8+ T cell function. Accordingly, exposure of CD8+ T cells to recombinant human (rh) IL-10 led to a marked reduction in IFN-γ in cells (Fig. 6c). However, coculture of CD8+ T cells and autologous CD69+/hi B cells, both of which were purified from the blood of SLE patients, showed marginal effects (Fig. 6c). Interestingly, in such a coculture system, adding antibodies against IL-6 plus TNF-α effectively restored CD69+/hi B cell-mediated CD8+ T cell tolerance, and this process could be reversed by additional neutralization of IL-10 (Fig. 6c).
Fig. 6. Targeting c-Myc-elicited glycolysis abrogates inflammatory Breg cell mediated pathogenic response.
a, b FACS analysis of CCR7−CD45RA− effector memory cells T cells in circulating T cells (a) and IL-10R expression (b) on effector memory T cells in blood samples from 5 healthy donors and 5 untreated SLE patients. c Purified T cells from SLE patients were cultured in medium, treated with recombinant human IL-10, or cocultured with CD69+/hi B cells in the presence of isotype control, anti-TNF-α antibody plus anti-IL-6 antibody, or anti-TNF-α antibody plus anti-IL-6 antibody plus anti-IL-10 antibody for 7 d. IFN-γ expression in CD8+ T cells was examined using FACS (n = 11). d, e Purified T cells from SLE patients were left untreated or cultured with CD69− B cells or CD69+/hi B cells for 7 d. Differentiation of TH17 and TH22 was determined by FACS (n = 7). Representative dotplots and statistical data were shown in d and e, respectively. f Associations of circulating CD69+/hi B cells with circulating TH17 and TH22 cells in untreated SLE patients (n = 17). g Purified T cells from untreated SLE patients were cultured with autologous CD69+/hi B cells in the presence of isotype control, anti-TNF-α antibody plus anti-IL-6 antibody, or anti-IL-10 antibody for 7 d. Differentiation of TH17 and TH22 was determined by FACS (n = 8). h, i Purified CD69+/hi B cells from SLE patients were cultured in medium or pretreated with DMSO, inhibitor against c-Myc, PFKFB3, or ERK plus P38. Thereafter, the cells were incubated with autologous T cells for 7 d. Cytokine expression in B cells (h) and differentiation of TH17 and TH22 (i) was measured by real-time PCR and FACS (n = 6 for h; n = 9 for i). Data were acquired from more than four independent experiments and shown as means ± SEM. Pearson’s correlation analysis for f, significance was determined with Student’s t test (a) or one-way ANOVA test (c, e, g–i). *p < 0.05, **p < 0.01, ***p < 0.001
Next, we further considered the effects of functional Breg cells on CD4+ T cell function. We found that exposure of CD4+ T cells to autologous CD69+/hi B cells resulted in marked differentiation of TH17 and TH22 cells ex vivo (Fig. 6d, e). In the blood of SLE patients, the proportion of CD69+/hi B cells positively correlated with that of TH17 or TH22 cells (Fig. 6f). Consistent with our hypothesis, blocking the effect of IL-6 plus TNF-α on CD4+ T cells successfully suppressed the polarization of TH17 and TH22 cells, but neutralizing IL-10 had minimal effects (Fig. 6g). Because MAPK-mediated c-Myc upregulation is also required for glycolysis-triggered B cell inflammatory effects (Fig. 5), we pretreated CD69+/hi B cells with inhibitors of ERK, P38, c-Myc, or the glycolytic enzyme PFKFB3 before culturing them with autologous CD4+ T cells. Consistently, all of the pretreatment groups showed an attenuated inflammatory response of CD69+/hi B cells and impaired subsequent differentiation of TH17 or TH22 cells (Fig. 4i, and Fig. 6h, i). These data indicate that these inflammatory features repurposed Breg cell function away from inducing CD8+ T cell tolerance and toward inducing a pathogenic CD4+ T cell response.
Discussion
Previous studies have implied a suppressive role of Breg cells solely based on the anti-inflammatory signature of IL-101. In the present investigation, by using SLE as a model system, we identified a disruptive role of IL-10-secreting functional Breg cells in maintaining the pathogenic disease response. We substantially demonstrated that functional Breg cells generated in pathological environments of SLE exhibit inflammatory features rather than anti-inflammatory features, suggesting an unrecognized immune-editing mechanism by which B cells regulate the progression of inflammatory diseases.
Despite recent success in identifying several subpopulations with Breg cell potential, little is known about the phenotype and regulation of functional Breg cells in humans1. At present, the conclusion that Breg cells hamper the progression of autoimmune diseases remains controversial31,32. It has been suggested that CD24hiCD38hi and CD24hiCD27+ B cells comprise the major cellular groups of peripheral Breg cells in humans8,9. In fact, neither of these subpopulations isolated from healthy individuals or patients with SLE functionally produce IL-10. Hence, the ratio of CD24hiCD38hi or CD24hiCD27+ B cells cannot reflect the contribution of functional Breg cells to the progression of SLE. More strikingly, although IL-10 itself is anti-inflammatory, IL-10+ functional Breg cells increase significantly with the progression of SLE, suggesting a pathogenic role. Of note, the environmental factors (e.g., CPG-DNA) that trigger functional Breg cells in SLE also effectively contribute to the progression of disease21. It is plausible that IL-10 is merely a marker for functional Breg cells and cannot represent all the functions of those cells. This notion is supported by our observation that functional Breg cells display a previously unrecognized CD24intCD27−CD38−CD69+/hi phenotype different from that of their precursors, CD24hiCD38hi and CD24hiCD27+ B cells, and these cells produce high amounts of inflammatory mediators in their early differentiation stage. Thus, Breg cells are versatile in that they create inflammation and/or suppression according to their differentiation stage and exogenous signals. Consistently, several groups also reported that Breg cells can serve as killer cells, as described for CD8+ T or NK cells7.
In both humans and mice, glycolysis participated in M1 macrophage polarization15, whereas OXPHOS is restricted to M2 macrophages18. Analogously, increased OXPHOS maintains the suppressive activity of Treg cells;17 whereas potent glycolysis is responsible for the effector activity of cytotoxic T cells33. Unexpectedly, the metabolic program employed by functional Breg cells differed from that used by Treg cells. We demonstrated that glycolysis, but not OXPHOS, is vital for the polarization and function of Breg cells in SLE patients, and this was proved by four sets of experiments. First, the environmental factors (e.g., CPG-DNA) contributing to the induction of functional Breg cells also result in increased expression of the glycolytic enzymes HK2, PFKFB3, and LDHA, as well as marked production of extracellular lactate. Second, blocking glucose incorporation or suppressing the activity of the glycolytic enzyme PFKFB3 in CPG-DNA-treated B cells successfully impairs the differentiation of functional Breg cells in vitro, whereas incubating B cells with inhibitors of OXPHOS has only marginal effects. Third, functional Breg cells isolated from SLE patients express higher levels of glycolytic enzymes, and when cultured ex vivo, these cells produce significantly increased levels of lactate and display a greater capacity to incorporate glucose. Fourth, suppressing glycolysis in CD69+/hi functional Breg cells ex vivo with 3-PO successfully impedes their Breg cell signature and inflammatory features. Therefore, activation of glycolysis in cells with Breg cell potential may represent a novel route for triggering a pathogenic response in SLE patients. This hypothesis is compatible with several previous studies showing that the induction of glycolysis in macrophages and dendritic cells also enhances the pathogenic response in inflammatory diseases15,34.
AKT/mTOR signal-triggered HIF-1α activity is essential for myeloid cell glycolysis and subsequent inflammatory responses29. However, although this pathway is also active in B cells treated with CPG-DNA, inhibiting mTOR and HIF-1α activity cannot attenuate glycolysis and the subsequent differentiation of functional Breg cells, suggesting the involvement of other signaling pathways. Notably, Myc proteins regulate cell growth and are oncogenic in many cancers, particularly B cell lymphoma30. In fact, c-Myc is also crucial for CPG-DNA-mediated glycolysis and the subsequent differentiation of functional Breg cells. More importantly, suppressing c-Myc signaling in functional Breg cells ex vivo efficiently reverses the pathogenic effects of functional Breg cells. Additionally, abolishing the phosphorylation of ERK and P38 in B cells effectively impairs c-Myc-triggered glycolysis, the inflammatory response and functional Breg cell generation. These findings agree with data reported by Wei and colleagues showing that evolutionarily conserved MAPK/ERK pathway triggers T cell response via c-Myc-elicited glycolysis35. Thus, a comprehensive study of the signaling network of functional Breg cells will be beneficial for designing rational therapeutic strategies for autoimmune diseases36,37.
Binding of IL-10R by IL-10 should trigger dysfunction of effector T cells38,39. The elevated expression of the proinflammatory factors IL-6 and TNF-α in functional Breg cells in patients with SLE abrogates the likelihood of engagement of this pathway. Coincidentally, inflammatory TNF-α has also been proven to reverse the suppressive function of Treg cells by phosphorylating Foxp340,41. Thus, it is not the suppressive cells per se but rather the inflammatory “context” that determines the ability of the suppressive cells42,43. Moreover, during the progression of autoimmune diseases, TH cells regularly display pathogenic function by triggering and sustaining inflammation44,45. The current study provides important new insights into the previously unrecognized helper role of functional Breg cells. After exposure to inflammatory mediators released by functional Breg cells, TH cells obtain the ability to polarize into proinflammatory TH subsets and that contribute to chronic inflammation.
In our current study, all data are obtained based on clinical observations and ex vivo (or in vitro) experiments using human primary immune cells. It should be emphasized that some of our findings may not fully reflect the in vivo conditions of SLE patients. However, in mouse lupus model, the progenitors and their phenotypic characteristics of functional Breg cells are different from those of human functional Breg cells46. Therefore, establishing a suitable animal model which can reproduce the immune environment of human SLE may help us to better understand the potential mechanism in the development of the disease.
Materials And methods
Patients
A total of 317 peripheral blood samples from SLE patients who met the Systemic Lupus International Collaborating Clinics classification criteria were collected at the First Affiliated Hospital of Nanjing Medical University and the Third Affiliated Hospital of Sun Yat-sen University from May 2018 to January 2022. 71 untreated patients were used for analyzing the clinical relevance of peripheral B cell subsets (Supplementary Table 1, cohort 1 and Supplementary Table 2); Another 219 untreated patients were enrolled for the isolation of leukocytes for subsequent ex vivo experiments (Supplementary Table 1, cohort 2); 15 asymptomatic clinical remission patients who may get treatment with hydroxychloroquine, glucocorticoids, or tacrolimus and another 12 complete remission patients who achieved a serologically and clinically quiescent period lasting for at least 2 years were also used for the isolation of leukocytes (Supplementary Table 1, cohort 3 and 4). Pregnant women and patients with severe infection, other systemic diseases were excluded in the study. The control samples were obtained from age-matched healthy volunteers registered in the physical examination center of the Third Affiliated Hospital of Sun Yat-sen University, who would be excluded once with other diagnosed systemic diseases, oral ulcer, skin rash, a complaint of joint symptoms, fever, and positive family history of SLE or other possible symptoms of SLE. All samples were anonymously coded in accordance with local ethical guidelines (as stipulated by the Declaration of Helsinki). Written informed consent was obtained from the patients, and the protocol was approved by the Review Board of Sun Yat-sen University.
Peripheral blood mononuclear cells (PBMC) isolation
PBMC were purified by density gradient centrifugation in Ficoll. Thereafter, the PBMC were washed and resuspended in culture medium (RPMI 1640 + 10% FBS). B cells and T cells were isolated from the PBMC by magnetic separation with MACS column (Miltenyi Biotec). Thereafter, CD24hiCD38hi and CD24hiCD27+ B cells, as well as CD69+/hi and CD69− B cells were sorted by FACS (MoFlo, Beckman Coulter). IL-10− and IL-10+ B cells were further purified using the IL-10 secretion assay-detection kits. These cells were used in subsequent experiments.
Immunoblotting
B cells from in vitro culture system were harvested and washed with PBS. Protein lysis and immunoblotting were conducted as previously described16. The antibodies used are shown in Supplementary Table 3.
Flow cytometry (FACS)
B cells and T cells purified from blood samples were incubated with fluorochrome-conjugated antibodies and subsequently examined by FACS. Under certain circumstances, B cells and T cells from blood samples, in vitro or ex vivo coculture experiments were treated with Leukocyte Activation Cocktail (BD Bioscience) for 5 h at 37 °C. Afterward, the cells stained with surface markers were fixed and permeabilized using IntraPrep reagent (Beckman Coulter), and finally stained with intracellular markers. Data were obtained using Gallios flow cytometer (Beckman Coulter). Antibodies used are shown in Supplementary Table 4.
Enzyme-linked immunospot assay (ELISpot)
ELISpot assays were conducted with kits (BD Bioscience). For IL-10 detection, 1 × 105 B cells were cultured for 24 h. The images were obtained using an ELISpot reader (CTL). Thereafter, the spot numbers were calculated.
Enzyme-linked immunosorbent assay (ELISA)
Concentrations of TNF-α, IL-6 and IL-10 in the culture supernatants or in the plasma from SLE patients were detected using ELISA kits purchased from eBioscience. Antibodies used are listed in Supplementary Table 5.
Extracellular acidification (ECAR) analyses
The ECAR of B cells was measured via XF-24 Extracellular Flux Analyzer (Seahorse Bioscience). In some experiments, B cells were left untreated or were pretreated with DMSO, or inhibitor against mTOR, HIF-1α, c-Myc, ERK or P38 signals. Thereafter, the cells were incubated in the presence or absence of CPG-DNA for 12 h. B cells were suspended in XF Base Medium Minimal DMEM supplemented with L-Glutamine (2 mM) and then placed on a cell culture microplate (5 × 105 cells/well; XF-24, Seahorse Bioscience). During real-time measurement of the ECAR, Glucose (10 mM), oligomycin A (1 μM), and 2DG (50 mM) were added to the cells. The related reagents used are shown in Supplementary Table 5.
Glucose uptake assay
B cells were purified from SLE patients. Thereafter, these cells were cultured for 1 h in PBS and then incubated with fluorescence-labeled 2-NBDG for 0.5 h at 37 °C. Finally, the cells were harvested and examined by FACS (Gallios).
Lactate assay
Purified B cells from healthy blood were cultured in medium or treated with CPG-DNA for 3 d. Thereafter, the culture supernatants were collected. In some cases, CD69+/hi and CD69− B cells purified from SLE were culture for 3 days. Concentrations of the lactate were determined using L-lactate assay Kit (eton bioscience). L-lactate assay Kit used are shown in Supplementary Table 5.
In vitro or ex vivo experiments for B cells
In vitro experiments, purified B cells (Miltenyi Biotec) from healthy donor were left untreated or were treated with an anti-IgM antibody (5 μg/ml), an anti-CD40 antibody (2 μg/ml), LPS (5 μg/ml), or CpG-DNA (2.5 μg/ml). In some in vitro experiments, before treatment with CPG-DNA, B cells were incubated with DMSO, 2-DG (10 mM), or a specific inhibitor against glycolytic enzyme PFKFB3 (3-PO, 10 μM), ATP synthase (Oligomycin, 0.2 µM), fatty acid oxidation (etomoxir, 50 µM), mTOR (Rapamycin, 10 µM), HIF-1α (Echinomycin, 1 ng/ml), c-Myc ((JQ1, 20 μM), JNK (SP 600125, 5 μM), ERK (U0126, 10 μM), P38 (SB 203580, 10 μM), canonical NFκB (BAY 11-7082, 5 μM) or non-canonical NF-κB (Amgen16, 1 μM) signals. Thereafter, IL-10 production was detected by indicated time. In some ex vivo experiments, CD69+/hi and CD69−, or IL-10+ and IL-10− B cells purified from untreated SLE patients’ blood were left untreated or treated with DMSO, or a specific inhibitor against c-Myc (JQ1), PFKFB3 (3-PO) for 12 h, then the expression of cytokine and glycolytic enzyme were measured. The inhibitors are shown in Supplementary Table 5.
Ex vivo coculture system of T cells
Purified T cells were cultured in medium, or treated with human IL-10 (10 ng/ml), or were cultured with autologous CD69− or CD69+/hi B cells in the presence of anti-CD3 (2.5 µg/ml) as well as anti-CD28 (2.5 µg/ml) antibodies (eBioscience) for 2 days. Thereafter, the cells were incubated for 5 d with IL-2 (20 IU/ml) (eBioscience). In some cases, CD69+/hi B cells were cultured in medium or pretreated with a specific inhibitor against PFKFB3 (3-PO), c-Myc (JQ1), ERK (U0126) or P38 (SB 203580). Thereafter, the B cells were washed and cocultured with autologous T cells. In other experiments, T cells were incubated with blocking antibodies against IL-10 (10 µg/ml), IL-6 (25 µg/ml), or TNF-α (25 µg/ml) (R&D Systems) and afterward cultured with autologous B cells. The neutralizing antibodies are shown in Supplementary Table 5.
Real-time polymerase chain reaction (PCR)
Total RNA of B cells from blood of SLE patients or from ex vivo or in vitro culture system were extracted using Trizol reagent (Invitrogen). PCR was conducted as previously described16. The primers are shown in Supplementary Table 6. All data are shown in arbitrary units compared with the expression of 18 S rRNA.
Statistical analysis
The gene expression profiling of human IL-10− and IL-10+ B cells were downloaded from GEO database (GSE50895). Data are presented as means ± SEM. All statistical tests were performed as two-sided. We applied the Student t test to compare normally distributed data; and the non-parametric exact Wilcoxon signed-rank test was used to compare data not normally distributed. For multiple comparisons, an analysis of variance followed by Bonferroni’s correction was applied. R values were calculated based on the analysis of Pearson’s correlation. All statistical tests were analyzed using GraphPad Prism (v.6) software. The p values < 0.05 were considered statistically significant.
Supplementary information
Acknowledgements
The study was supported by project grants from the National Natural Science Foundation of China (82071767, 82025016, 31830025, 81901585, and 81802403), and the Natural Science Foundation of Guangdong Province, China (2018B030308010 and 2019A1515011770).
Author contributions
X.Y.W., Y.W. and X.W. conducted most of the experiments and analyzed data. B.H. and Y.L. collected clinical samples. W.H.W., C.X.H. and M.M. contributed to FACS and analyzed the related data. Z.Y.L and D.P.C contributed to seahorse experiments. J.R.Q. and X.D.C guided computational analysis. Y.C., X.H.W. and D.M.K. contributed to study design, supervised the study, and/or contributed to writing the manuscript. All authors have read and approved the article.
Data availability
All data are available in the main text or the supplementary materials.
Competing interests
The authors declare no competing interests.
Footnotes
These authors contributed equally: Xu-Yan Wang, Yuan Wei, Bo Hu, Yuan Liao, Xiaodong Wang.
Change history
3/22/2024
A Correction to this paper has been published: 10.1038/s41392-024-01783-7
Contributor Information
Dong-Ming Kuang, Email: kdming@mail.sysu.edu.cn.
Xue-Hao Wang, Email: wangxh@njmu.edu.cn.
Yun Chen, Email: chenyun@njmu.edu.cn.
Supplementary information
The online version contains supplementary material available at 10.1038/s41392-022-00948-6.
References
- 1.Rosser EC, Mauri C. Regulatory B cells: Origin, phenotype, and function. Immunity. 2015;42:607–612. doi: 10.1016/j.immuni.2015.04.005. [DOI] [PubMed] [Google Scholar]
- 2.Candando KM, Lykken JM, Tedder TF. B10 cell regulation of health and disease. Immunol. Rev. 2014;259:259–272. doi: 10.1111/imr.12176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Lin X, et al. IL-10-producing regulatory B cells restrain the T follicular helper cell response in primary Sjögren’s syndrome. Cell. Mol. Immunol. 2019;16:921–931. doi: 10.1038/s41423-019-0227-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Boldison J, Da Rosa LC, Davies J, Wen L, Wong FS. Dendritic cells license regulatory B cells to produce IL-10 and mediate suppression of antigen-specific CD8 T cells. Cell. Mol. Immunol. 2020;17:843–855. doi: 10.1038/s41423-019-0324-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Lundy SK, Fox DA. Reduced Fas ligand-expressing splenic CD5+ B lymphocytes in severe collagen-induced arthritis. Arthritis Res. Ther. 2009;11:R128. doi: 10.1186/ar2795. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Khan AR, et al. PD-L1hi B cells are critical regulators of humoral immunity. Nat. Commun. 2015;6:5997. doi: 10.1038/ncomms6997. [DOI] [PubMed] [Google Scholar]
- 7.Lindner S, et al. Interleukin 21-induced granzyme B-expressing B cells infiltrate tumors and regulate T cells. Cancer Res. 2013;73:2468–2479. doi: 10.1158/0008-5472.CAN-12-3450. [DOI] [PubMed] [Google Scholar]
- 8.Blair PA, et al. CD19(+)CD24(hi)CD38(hi) B cells exhibit regulatory capacity in healthy individuals but are functionally impaired in systemic Lupus Erythematosus patients. Immunity. 2010;32:129–140. doi: 10.1016/j.immuni.2009.11.009. [DOI] [PubMed] [Google Scholar]
- 9.Iwata Y, et al. Characterization of a rare IL-10-competent B-cell subset in humans that parallels mouse regulatory B10 cells. Blood. 2011;117:530–541. doi: 10.1182/blood-2010-07-294249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Loftus RM, Finlay DK. Immunometabolism: Cellular metabolism turns immune regulator. J. Biol. Chem. 2016;291:1–10. doi: 10.1074/jbc.R115.693903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Saravia J, Chapman NM, Chi H. Helper T cell differentiation. Cell. Mol. Immunol. 2019;16:634–664. doi: 10.1038/s41423-019-0220-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Khalsa JK, et al. Functionally significant metabolic differences between B and T lymphocyte lineages. Immunology. 2019;158:104–120. doi: 10.1111/imm.13098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Boothby M, Rickert RC. Metabolic regulation of the immune humoral response. Immunity. 2017;46:743–755. doi: 10.1016/j.immuni.2017.04.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Lam WY, et al. Mitochondrial pyruvate import promotes long-term survival of antibody-secreting plasma cells. Immunity. 2016;45:60–73. doi: 10.1016/j.immuni.2016.06.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Shirai T, et al. The glycolytic enzyme PKM2 bridges metabolic and inflammatory dysfunction in coronary artery disease. J. Exp. Med. 2016;213:337–354. doi: 10.1084/jem.20150900. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Zeng QH, et al. B cells polarize pathogenic inflammatory T helper subsets through ICOSL-dependent glycolysis. Sci. Adv. 2020;6:eabb6296. doi: 10.1126/sciadv.abb6296. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Michalek RD, et al. Cutting edge: Distinct glycolytic and lipid oxidative metabolic programs are essential for effector and regulatory CD4+ T cell subsets. J. Immunol. 2011;186:3299–3303. doi: 10.4049/jimmunol.1003613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Wang F, et al. Glycolytic stimulation is not a requirement for M2 macrophage differentiation. Cell Metab. 2018;28:463–475. doi: 10.1016/j.cmet.2018.08.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Gao N, et al. Impaired suppressive capacity of activation-induced regulatory B cells in systemic lupus erythematosus. Arthritis Rheumatol. 2014;66:2849–2861. doi: 10.1002/art.38742. [DOI] [PubMed] [Google Scholar]
- 20.Ohl K, Tenbrock K. Regulatory T cells in systemic lupus erythematosus. Eur. J. Immunol. 2015;45:344–355. doi: 10.1002/eji.201344280. [DOI] [PubMed] [Google Scholar]
- 21.Pogribny LP, Beland FA. DNA hypomethylation in the origin and pathogenesis of human diseases. Cell. Mol. Life Sci. 2009;66:2249–2261. doi: 10.1007/s00018-009-0015-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Lee TP, et al. Transgenic overexpression of anti-double-stranded DNA autoantibody and activation of toll-like receptor 4 in mice induce severe systemic lupus erythematosus syndromes. J. Autoimmun. 2010;35:358–367. doi: 10.1016/j.jaut.2010.07.007. [DOI] [PubMed] [Google Scholar]
- 23.Hamilton JA, Hsu HC, Mountz JD. Autoreactive B cells in SLE, villains or innocent bystanders? Immunol. Rev. 2019;292:120–138. doi: 10.1111/imr.12815. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Ramanujam M, et al. Phoenix from the flames: Rediscovering the role of the CD40-CD40L pathway in systemic lupus erythematosus and lupus nephritis. Autoimmun. Rev. 2020;19:102668. doi: 10.1016/j.autrev.2020.102668. [DOI] [PubMed] [Google Scholar]
- 25.Singh SR, et al. The lipolysis pathway sustains normal and transformed stem cells in adult Drosophila. Nature. 2016;538:109–113. doi: 10.1038/nature19788. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Symersky J, Osowski D, Walters DE, Mueller DM. Oligomycin frames a common drug-binding site in the ATP synthase. Proc. Natl Acad. Sci. USA. 2012;109:13961–13965. doi: 10.1073/pnas.1207912109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Rowe I, et al. Defective glucose metabolism in polycystic kidney disease identifies a new therapeutic strategy. Nat. Med. 2013;19:488–493. doi: 10.1038/nm.3092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Clem B, et al. Small-molecule inhibition of 6-phosphofructo-2-kinase activity suppresses glycolytic flux and tumor growth. Mol. Cancer Ther. 2008;7:110–120. doi: 10.1158/1535-7163.MCT-07-0482. [DOI] [PubMed] [Google Scholar]
- 29.Yecies JL, Manning BD. Transcriptional control of cellular metabolism by mTOR signaling. Cancer Res. 2011;71:2815–2820. doi: 10.1158/0008-5472.CAN-10-4158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Dang CV. MYC on the path to cancer. Cell. 2012;149:22–35. doi: 10.1016/j.cell.2012.03.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Hu F, et al. Pathogenic conversion of regulatory B10 cells into osteoclast-priming cells in rheumatoid arthritis. J. Autoimmun. 2017;76:53–62. doi: 10.1016/j.jaut.2016.09.002. [DOI] [PubMed] [Google Scholar]
- 32.Xu L, et al. Impairment of Granzyme B-producing regulatory B cells correlates with exacerbated rheumatoid arthritis. Front. Immunol. 2017;8:768. doi: 10.3389/fimmu.2017.00768. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Phan AT, et al. Constitutive glycolytic metabolism supports CD8+ T cell effector memory differentiation during viral Infection. Immunity. 2016;45:1024–1037. doi: 10.1016/j.immuni.2016.10.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Krawczyk CM, et al. Toll-like receptor-induced changes in glycolytic metabolism regulate dendritic cell activation. Blood. 2010;115:4742–4749. doi: 10.1182/blood-2009-10-249540. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Wei X, et al. The evolutionarily conserved MAPK/Erk signaling promotes ancestral T-cell immunity in fish via c-Myc-mediated glycolysis. J. Biol. Chem. 2020;295:3000–3016. doi: 10.1074/jbc.RA119.012231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Kodama S, Davis M, Faustman DL. The therapeutic potential of tumor necrosis factor for autoimmune disease: a mechanistically based hypothesis. Cell. Mol. Life Sci. 2005;62:1850–1862. doi: 10.1007/s00018-005-5022-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.O’Neill LA, Kishton RJ, Rathmell J. A guide to immunometabolism for immunologists. Nat. Rev. Immunol. 2016;16:553–565. doi: 10.1038/nri.2016.70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Ruffell B, et al. Macrophage IL-10 blocks CD8+ T cell-dependent responses to chemotherapy by suppressing IL-12 expression in intratumoral dendritic cells. Cancer Cell. 2014;26:623–637. doi: 10.1016/j.ccell.2014.09.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Xiao X, et al. PD-1hi identifies a novel regulatory B-cell population in human hepatoma that promotes disease progression. Cancer Disco. 2016;6:546–559. doi: 10.1158/2159-8290.CD-15-1408. [DOI] [PubMed] [Google Scholar]
- 40.Kryczek I, et al. FOXP3 defines regulatory T cells in human tumor and autoimmune disease. Cancer Res. 2009;69:3995–4000. doi: 10.1158/0008-5472.CAN-08-3804. [DOI] [PubMed] [Google Scholar]
- 41.Nie H, et al. Phosphorylation of FOXP3 controls regulatory T cell function and is inhibited by TNF-α in rheumatoid arthritis. Nat. Med. 2013;19:322–328. doi: 10.1038/nm.3085. [DOI] [PubMed] [Google Scholar]
- 42.Kryczek I, et al. IL-17+ regulatory T cells in the microenvironments of chronic inflammation and cancer. J. Immunol. 2011;186:4388–4395. doi: 10.4049/jimmunol.1003251. [DOI] [PubMed] [Google Scholar]
- 43.Maj T, et al. Oxidative stress controls regulatory T cell apoptosis and suppressor activity and PD-L1-blockade resistance in tumor. Nat. Immunol. 2017;18:1332–1341. doi: 10.1038/ni.3868. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Zenewicz LA, Antov A, Flavell RA. CD4 T-cell differentiation and inflammatory bowel disease. Trends Mol. Med. 2009;15:199–207. doi: 10.1016/j.molmed.2009.03.002. [DOI] [PubMed] [Google Scholar]
- 45.Wilke CM, Bishop K, Fox D, Zou W. Deciphering the role of Th17 cells in human disease. Trends Immunol. 2011;32:603–611. doi: 10.1016/j.it.2011.08.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Yanaba K, et al. A regulatory B cell subset with a unique CD1dhiCD5+ phenotype controls T cell-dependent inflammatory responses. Immunity. 2008;28:639–650. doi: 10.1016/j.immuni.2008.03.017. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
All data are available in the main text or the supplementary materials.