Introduction
Nonhealing wounds of the scalp have a broad range of differential diagnoses. Clinicians must rely on patient history, physical diagnosis, and pathologic findings to develop a differential diagnosis that may include keratinocyte carcinomas, primary blistering diseases, picker’s ulcerations, pyoderma, and traumatic wounds. An additional consideration is erosive pustular dermatosis of the scalp (EPDS), an uncommon idiopathic disease that often occurs in elderly patients.1 First described in 1977,2 it is commonly associated with actinic damage and baldness, although it has been linked to drug reactions, surgical closures, and trauma.3,4 Patients with EPDS present with recurrent pustules and nonhealing crusted ulcerations, producing scarring alopecia with skin atrophy. The diagnosis is often delayed because it is a diagnosis of exclusion. Histopathologic findings demonstrate nonspecific inflammatory changes, epidermal atrophy, and ulceration. Neutrophils, plasma cell proliferation, and foreign body–type giant cells may be noted.1 Paradoxically, high-potency topical steroids usually avoided in ulcerations are the most effective treatment. There have been variable responses to antimicrobials, tacrolimus, and photodynamic therapy.5 Chronic ulceration in untreated EPDS may take years to heal.6
Case report
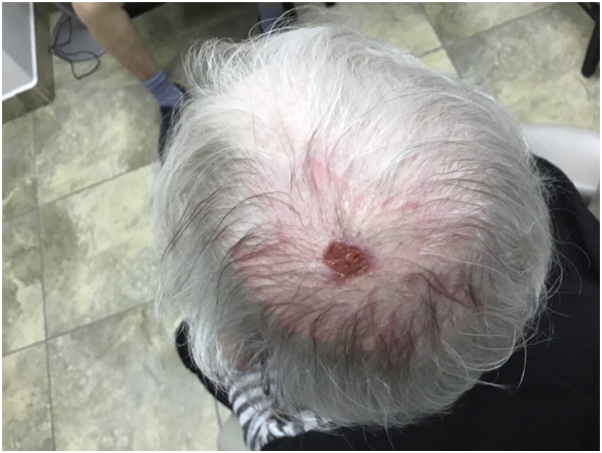
An 84-year–old woman presented with a 1-year history of a chronic, crusted ulceration on the parietal aspect of scalp. As per patient history, it had failed to respond to a twice-daily regimen of clobetasol 0.05% ointment used for 2 months before presentation. The patient also reported using petroleum jelly and mupirocin ointment before topical steroids with no response. There was no history of trauma. Past medical history was significant for androgenic alopecia. A painless 3.5-cm ulcerated plaque with yellow crusting, erythema, and oozing was present on the parietal aspect of the scalp (Fig 1). A punch biopsy specimen demonstrated a nonspecific ulcer with a thick overlying scale crust and an underlying scar with acute and chronic inflammation (Fig 2). Solar elastosis was present, but no keratinocyte atypia or evidence of invasive cancer was observed. The pathology report stated that EPDS was the likely diagnosis to be considered. Bacterial culture demonstrated moderate growth of Staphylococcus epidermidis.
Fig 1.

A 3.5-cm × 2.5-cm ulcer with crusting was noted on the apex of the scalp. The patient did not respond to mupirocin 2% ointment and petroleum jelly over 1 year.
Fig 2.
The shave specimen showed a broad crusted ulceration with underlying dermal edema, acute and chronic inflammation, and telangiectatic vessels. Re-epithelialization and an underlying scar were noted at 1 pole of the specimen. There was no evidence of leukocytoclastic vasculitis, vasculopathy, or an invasive tumor. (Hematoxylin-eosin stain; original magnification: ×100.)
Because of the persistence of this ulceration on the scalp in an elderly patient, lack of significant bacterial pathogens on culture, absence of clinical features of pyoderma gangrenosum, absence of malignancy on biopsy, as well as the dermatopathology report, the favored diagnosis was erosive pustular dermatosis. Treatment was initiated with sulfamethoxazole/trimethoprim 160 mg/800 mg twice daily for 2 weeks to suppress secondary bacterial infection. After no improvement was noted, powdered umbilical remnant allograft (URA) was packed into the wound, topped with a pea-sized amount of mupirocin 2% ointment, and covered with a saline-soaked gauze secured with a nonstick pad and paper tape. The dressing was changed weekly in the clinic for 8 weeks after gentle cleansing and modest debridement with a 12-ply gauze soaked in chlorhexidine and rinsing prior to the reapplication of URA, mupirocin, and saline gauze at each visit. At week 4, the wound was 45% smaller (Fig 3). Near-complete resolution occurred at week 8 (Fig 4), and treatment was discontinued without recurrence over 9 months.
Fig 3.
The ulcer contracted, and excellent granulation tissue was noted at the base after 4 weekly treatments with a powdered formulation of dehydrated umbilical remnant allograft.
Fig 4.
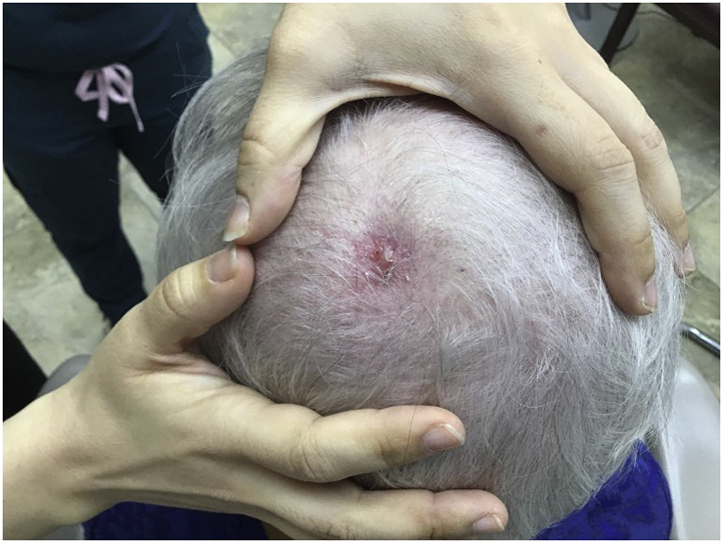
The ulcer had almost completely healed after the eighth weekly application of umbilical remnant allograft.
Discussion
EPDS is a diagnosis of exclusion with nonspecific clinical and histopathologic findings. Any delay in diagnosis is a missed opportunity for the initiation of an effective treatment. Although a diagnosis of EPDS can be inferred from this patient’s presentation, nonhealing scalp wounds have a broad range of differential diagnoses. It can be mistaken for a chronic bacterial infection because of the presence of sterile pustules early in its course, eroded basal or squamous cell carcinoma, or picker’s ulceration.1 It is important to set proper expectations with the patient before treatment. Even after the inflammatory cascade is reduced using potent topical steroids, wound healing is slow and a permanent scar will be present.7 This case demonstrated that a wound refractory to mupirocin ointment, petroleum jelly, and ultrapotent steroids over the course of a year could heal within months of using a weekly treatment with powdered URA.
Mechanisms of healing
There has been an increased interest in the use of stem cells for wound healing over the past 10 years. Options include platelet-rich plasma, bone marrow aspirate, adipose tissue aspirate, amniotic fluid, amniotic membrane, umbilical cord–derived Wharton jelly, and cord blood.8 Most notably, amniotic/chorionic allographs have been used in the treatment of EPDS.7 The healing capabilities of these products are attributed to the presence of stem cells, growth factors, cytokines, hyaluronic acid, and/or extracellular vesicles, including exosomes.9
Dehydrated URA (Corplex P, Stimlabs LLC; www.stimlabs.com) is a new product used for acute and chronic wounds. It contains large amounts of extracellular matrix components, including collagen, hyaluronic acid, and various growth factors that are implicated in the advancement of the wound-healing process.8,10 Recent sterilization techniques permit the retention of intact extracellular matrix structures and regulatory proteins from Wharton jelly in a powdered form.10,11 However, stem cells would not survive this processing.10 The primary benefit of this powdered form is its stable shelf life of 3 years as well as its ability to conform to irregular wound shapes.
Although the exact mechanism of action impacting accelerated wound healing with URA made from Wharton jelly is unknown, the introduction of extracellular matrix proteins (collagen 1, chondroitin/dermatan sulfate proteoglycans, hyaluronic acid, and glycosaminoglycans) to a depleted erosion may provide scaffolding for granulation and epithelialization.9 Additionally, cell signaling cascades have been implicated. Insulin-like growth factor 1, transforming growth factor β, and fibroblast growth factor are all linked with cellular proliferation and are found within the extracellular matrix (a component of URA).10 Cytokines associated with wound healing and tissue regeneration have also been found (intracellular adhesion molecule 1, granulocyte colony-stimulating factor, growth differentiation factor 15, and growth hormone).8
Conclusion
To our knowledge, this is the first report of URA used effectively in the treatment of a chronic wound resistant to traditional therapy for EPDS (ultrapotent topical steroids). Further studies comparing several therapies would be required to determine the relative value of this treatment.
Conflicts of interest
Dr Brodell reports honoraria for presentations from Allergan, Galderma, 3M/Graceway Pharmaceuticals, GlaxoSmithKline/Stiefel, Dermik/BenzaClin Speaker Bureau, Novartis Pharmaceuticals Corporation, Sanofi-Aventis, Medicis Speaker’s Bureau, PharmaDerm (a division of Nycomed US Inc), and Veregen Speakers Bureau; reports consultant fees from Galderma Laboratories, L.P., Medicis, Dow Pharmaceuticals Sciences, and Promius; serves on the advisory boards of Dow Pharmaceuticals Sciences and Nycomed US Inc; and has performed clinical trials for Genentech, Galderma, Abbott Laboratories, and Dow Pharmaceuticals Science. Authors Buttars, Rashid, and Al-Rubaie have no conflicts of interest to declare.
Footnotes
Funding sources: None.
IRB approval status: Not applicable.
References
- 1.Piccolo V., Russo T., Bianco S., Ronchi A., Alfano R., Argenziano G. Erosive pustular dermatosis of the scalp: why do we miss it? Dermatology. 2019;235(5):390–395. doi: 10.1159/000500909. [DOI] [PubMed] [Google Scholar]
- 2.Burton J.L. Case for diagnosis. Pustular dermatosis of scalp. Br J Dermatol. 1977;97(suppl 15):67–69. doi: 10.1111/j.1365-2133.1977.tb14339.x. [DOI] [PubMed] [Google Scholar]
- 3.Saridakis S., Giesey R.L., Ezaldein H.H., Scott J.F. Erosive pustular dermatosis of the scalp following surgical procedures: a systematic review. Dermatol Online J. 2020;26(4) 13030/qt9d80k39g. [PubMed] [Google Scholar]
- 4.Grattan C.E., Peachey R.D., Boon A. Evidence for a role of local trauma in the pathogenesis of erosive pustular dermatosis of the scalp. Clin Exp Dermatol. 1988;13(1):7–10. doi: 10.1111/j.1365-2230.1988.tb00639.x. [DOI] [PubMed] [Google Scholar]
- 5.Lee K.H., Carley S.K., Kraus C.N., Mesinkovska N.A. Treatment of erosive pustular dermatosis: a systematic review of the literature. Int J Dermatol. 2020;59(7):770–786. doi: 10.1111/ijd.14744. [DOI] [PubMed] [Google Scholar]
- 6.Levakov O., Gajić B. Erosive pustular dermatosis of the scalp-is it really a rare condition? Vojnosanit Pregl. 2014;71(3):307–310. doi: 10.2298/vsp120216053l. [DOI] [PubMed] [Google Scholar]
- 7.Kempton D.M., Maarouf M., Hendricks A.J., Shi V.Y. Erosive pustular dermatosis of the scalp associated with lamellar ichthyosis successfully treated with dehydrated human amnion/chorion membrane allograft. JAAD Case Rep. 2018;4(10):1059–1061. doi: 10.1016/j.jdcr.2018.09.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Gupta A., El-Amin S.F., III, Levy H.J., Sze-Tu R., Ibim S.E., Maffulli N. Umbilical cord-derived Wharton’s jelly for regenerative medicine applications. J Orthop Surg Res. 2020;15(1):49. doi: 10.1186/s13018-020-1553-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.La Rocca G., Anzalone R. Perinatal stem cells revisited: directions and indications at the crossroads between tissue regeneration and repair. Curr Stem Cell Res Ther. 2013;8(1):2–5. doi: 10.2174/1574888x11308010002. [DOI] [PubMed] [Google Scholar]
- 10.Corrao S., La Rocca G., Lo Iacono M., Corsello T., Farina F., Anzalone R. Umbilical cord revisited: from Wharton's jelly myofibroblasts to mesenchymal stem cells. Histol Histopathol. 2013;28(10):1235–1244. doi: 10.14670/HH-28.1235. [DOI] [PubMed] [Google Scholar]
- 11.Roy A., Griffiths S. Intermediate layer contribution in placental membrane allografts. J Tissue Eng Regen Med. 2020;14(8):1126–1135. doi: 10.1002/term.3086. [DOI] [PubMed] [Google Scholar]