Abstract
Background
To examine whether change in neutrophil–lymphocyte ratio, a marker of systemic inflammation, differs by childhood adversity and prenatal depression.
Methods
Prenatal complete blood count data were used to calculate neutrophil–lymphocyte ratio in first and third trimesters. The Adverse Childhood Experiences scale measured childhood adversity, and the Patient Health Questionnaire-9 measured depression. This is a secondary analysis of a study of predictors of risk for sleep-disordered breathing.
Results
Participants were 98 pregnant women, mean age 30 years (SD = 5), mean body mass index of 35 kg/m2 (SD = 7), 61% identified as white, and 28% identified as Hispanic. Women who reported childhood sexual abuse history displayed greater increase in neutrophil–lymphocyte ratio over pregnancy relative to women without childhood sexual abuse. Change in neutrophil–lymphocyte ratio across pregnancy did not differ by prenatal depression.
Conclusion
Experiences of sexual abuse in childhood may impact markers of systemic inflammation in pregnancy.
Keywords: Pregnancy, prenatal depression, childhood adversity, inflammation, neutrophil, lymphocyte
Introduction
According to the biological embedding model of early adversity, 1 experiences of childhood adversity program immune cells to have an exaggerated inflammatory response to stimuli and a decreased sensitivity to feedback from glucocorticoids, resulting in non-resolving inflammation and a “pro-inflammatory phenotype.” Nonpregnant victims of childhood adversity display elevations in inflammatory cytokines (IL-6, IL-1β, TNF-α),2–4 and there is some evidence, including work by our group, 5 that elevations in inflammation among victims of childhood adversity persist into pregnancy.6–8 Prenatal depression is also highly prevalent among women who experienced childhood adversity 9 and is associated with elevated inflammation; higher depressive symptoms in pregnancy have been associated with higher C-reactive protein (CRP), 10 IL-6, 11 and IL-1β 12 in past research.
While childhood adversity is a risk factor for prenatal depression, 9 the relative contribution of childhood adversity and prenatal depression on inflammation is not currently understood. Several previous studies conducted with nonpregnant adults have evaluated the relative contribution of childhood and adult adversity on adult biology. In a study of four life-course trajectories of childhood and adulthood socioeconomic status (i.e., low–low, low–high, high–low, and high–high socioeconomic conditions), Austin et al. 13 found that early-life socioeconomic disadvantage, but not current, predicted advanced cellular aging. Conversely, Levine et al. 14 found that trauma in childhood and low socioeconomic status in adulthood interacted to affect inflammatory gene expression. Similarly, adults with histories of child sexual and/or physical abuse with comorbid depression displayed exacerbated biological responses to a laboratory challenge relative to adults with histories of child abuse but without depression.15,16
In this study, we will examine the neutrophils-to-lymphocytes ratio (NLR) from peripheral blood via complete blood counts (CBCs), a measure of systemic inflammation and one that is frequently reported as a prognostic factor for diseases including cancer 17 and cardiovascular diseases. 18 Neutrophils increase due to nonspecific inflammation, and lymphocytes decrease in response to inflammation and glucocorticoid release, resulting in elevations in NLR. In nonpregnant adults, Penz et al. 19 reported that experiences of stressful life events were associated with an increase in NLR across one year. Aydin Sunbul et al. 20 reported that higher NLR levels were associated with greater depressive symptoms. In obstetric populations, research indicates that NLR values are higher in women with gestational diabetes, 21 preeclampsia,22,23 premature rupture of membranes, 24 and early preterm delivery. 25 NLR is routinely collected across pregnancy during prenatal care. Over pregnancy, NLR reach peak values in second trimester, and normative ranges of NLR in pregnancy have been published. 26
The aims of this study are to examine the contribution of both childhood adversity and prenatal depression on maternal NLR values across gestation. We expected that women with history of childhood adversity and high prenatal depression would demonstrate greater increases in NLR across pregnancy relative to women without childhood adversity or prenatal depression. Results from this study will add to a growing body of literature on the biological consequences of childhood adversity, and the relative contribution of childhood adversity and prenatal depression, on maternal inflammation across pregnancy.
Methods
Recruitment and participation
Participants were pregnant women enrolled in an ongoing study of predictors of risk for obstructive sleep apnea. Women were recruited for the parent study through medical chart review. Pregnant women with a singleton pregnancy, body mass index (BMI) of 27kg/m2 or more, and gestational age less than 18 weeks were approached at prenatal care appointments and asked if they snored regularly in pregnancy. Inclusion criteria were: self-reported habitual snoring, BMI 27kg/m2 or more at recruitment, under 18 weeks’ gestation at enrollment, over 18 years of age, pregnant with a singleton pregnancy, and able to provide informed consent. Women were excluded if they had a serious physical or mental illness that would affect participation.
Participants completed a study visit at approximately 11 weeks’ gestation (SD = 2) in which they completed self-report measures of childhood adversity and prenatal depression. NLRs were calculated from complete blood counts (CBC) collected across prenatal care and extracted from the medical record. As continuous positive airway pressure therapy can affect NLR levels, 27 women were excluded from the current analyses if they received treatment for obstructive sleep apnea. All participants provided written informed consent prior to their participation. The Rhode Island Hospital Institutional Review Board approved the study.
Measures
Adverse Childhood Experience (ACE) Scale
The ACE is a widely used 10-item self-report instrument that asks adults to indicate whether or not they experienced a number of adverse events prior to the age of 18 years old. In this study, we categorized women based on having four or more versus three or less adverse childhood experiences, based on evidence from a recent systematic review and meta-analysis reporting that individuals who experienced at least four ACEs were at highest risk for physical and mental illnesses. 28 We also examined if specific experiences of childhood physical, sexual, or emotional abuse predicted change in NLR over pregnancy. Specifically, women responded yes/no to three questions: (1) “Did a parent or other adult in the household often or very often… Swear at you, insult you, put you down, or humiliate you? or Act in a way that made you afraid that you might be physically hurt?” (i.e., emotional abuse); (2) “Push, grab, slap, or throw something at you? or Ever hit you so hard that you had marks or were injured?” (i.e., physical abuse); and (3) “Touch or fondle you or have you touch their body in a sexual way? or Attempt or actually have oral, anal, or vaginal intercourse with you?” (i.e., sexual abuse). Internal reliability of the ACE was α = .79 reflecting good internal reliability.
Prenatal depression
Symptoms of prenatal stress were measured using the Patient Health Questionnaire (PHQ)-9. 29 The PHQ-9 is a self-report measure of depressive symptoms in the previous two weeks. Items correspond to each of the nine diagnostic symptoms of major depressive disorders as defined by the Diagnostic and Statistical Manual of Mental Disorders (DSM). Responses range from 0 (not at all) to 3 (nearly every day). Scores of 10 or greater reflect clinically significant levels of depressive symptoms. Internal reliability of the PHQ9 was α = .82 reflecting good internal reliability. The PHQ9 is considered to be a reliable measure of depression in pregnancy.30,31
Neutrophil-to-lymphocyte ratio (NLR)
Prenatal CBC data (absolute neutrophil and lymphocyte counts) that were collected as part of standard prenatal care were extracted from medical records across gestation. Electrical impedance, also known as Coulter’s Principle, was used to produce cell counts. The NLR at each time point was calculated by dividing the number of neutrophils by the number of lymphocytes. CBCs from emergency room visits or during active acute or chronic inflammatory conditions or infections were excluded.
Adverse maternal and neonatal pregnancy outcomes
While this study was not designed to detect differences in pregnancy outcomes by NLR, we explored if change in NLR was associated with adverse maternal or neonatal pregnancy outcomes. We created a composite variable to indicate if women had any of the following adverse maternal pregnancy outcomes: gestational diabetes, gestational hypertension, and/or preeclampsia. We created a composite variable to indicate if neonates experienced any of the following adverse outcomes: preterm birth and/or low birth weight. We defined births prior to 37 weeks’ gestation as preterm birth, and birthweight less than 2500 g as low birth weight. Pregnancy outcomes were collected by chart review following delivery by a member of the research team with a medical degree.
Data analysis
NLR values were log transformed due to significant skewness (>2). Demographic characteristics, one-way analysis of variance (ANOVA) and Chi-squared analyses were performed to examine differences in NLR according to ACE measures and prenatal depression using IBM SPSS version 25. Repeated measures ANOVA were used to examine change in NLR from first to third trimester according to history of (1) childhood adversity, (2) prenatal depression, (3) both childhood adversity and prenatal depression, and (4) adverse maternal and neonatal pregnancy outcomes. All analyses adjusted for covariates including maternal age, BMI assessed in pregnancy, race, cigarette smoking in pregnancy, and diagnosis of sleep-disordered breathing (defined as an apnea–hypopnea index of 5 or more as measured by at home sleep study).
Results
Participant characteristics
Data from 98 women were included in this study. Women were, on average, 30 years old (SD = 5, range: 18–42), BMI measured at the study visit was 35kg/m2 (SD = 7, range: 27–58), 8% reported cigarette smoking in pregnancy, and 4% had obstructive sleep apnea. The distribution of ethnicity and race was as follows: 28% Hispanic, 61% White, 18% Black, 4% Asian, 4% American Indian/Alaskan Native, 10% multiracial, and 10% unreported.
ACE scores ranged from 0 to 10, with average scores of 2 (SD = 2). Twenty-six percent of the sample reported four or more ACEs; 25% of the sample reported childhood emotional abuse, 16% reported childhood physical abuse, and 20% of participants reported childhood sexual abuse. PHQ-9 scores ranged from 0 to 23, with average scores of 6 (SD = 5). Nineteen percent of the sample reported clinically elevated prenatal depression scores (scores of 10 or greater on the PHQ9). Women with four or more experiences of childhood adversity were significantly more likely to experience prenatal depression (χ2 = 7.43, p = .006). Average NLR values in the first trimester were 3.13 (SD = 1.13), and in third trimester 4.18 (SD = 1.55). Of note, these values fall within the published ranges for normal pregnancy. 26 In terms of adverse pregnancy outcomes, 36% of participants had an adverse maternal outcome and 14% had an adverse neonatal pregnancy outcome. Participant characteristics for the total sample and by ACE group are displayed in Table 1.
Table 1.
Demographic characteristics.
| TotalN = 98 | High childhood adversity (4+)N = 25 | Low childhood adversity (4−)N = 73 | ||
|---|---|---|---|---|
| M (SD) | M (SD) | M (SD) | p value | |
| Maternal age | 30 (5) | 30 (6) | 31 (5) | .52 |
| Race (%) | ||||
| White | 61% | 70% | 64% | .54 |
| Black | 18% | 19% | 13% | .50 |
| Asian | 4% | 0% | 2% | .42 |
| American Indian | 4% | 7% | 1% | .086 |
| Multiracial | 10% | 15% | 6% | .148 |
| Ethnicity (% Hispanic) | 28% | 28% | 27% | .096 |
| Prenatal body mass index (kg/m2) | 35 (7) | 35 (6) | 35 (6) | .59 |
| OSA diagnosis (% Yes) | 4% | 10% | 0% | .009 |
| Cigarette smoking (% Yes) | 8% | 20% | 5% | .004 |
| Depressive symptoms | 6 (5) | 8 (5) | 5 (4) | .003 |
| Prenatal depression (% Yes) | 19% | 36% | 12% | .006 |
| Childhood sexual abuse (% Yes) | 20% | 46% | 10% | <.001 |
| Childhood physical abuse (% Yes) | 16% | 51% | 3% | <.001 |
| Childhood emotional abuse (% Yes) | 25% | 76% | 6% | <.001 |
OSA: obstructive sleep apnea.
Note: Demographic characteristics were compared between high and low childhood adversity groups using independent samples t-tests or chi-squared analyses.
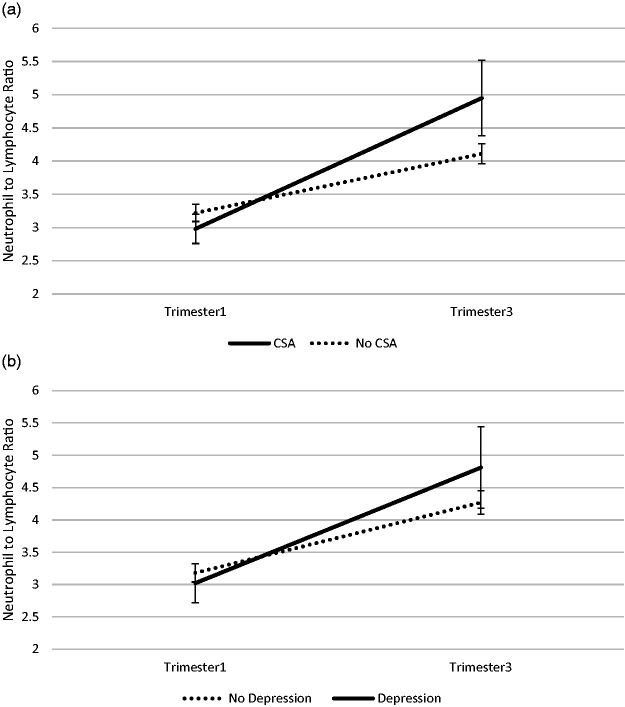
We examined if ACE group, and specific types of childhood adversity, predicted change in NLR across gestation. Repeated measures ANOVA revealed that ACE group marginally predicted change in NLR across gestation (F = 3.90, p = .053) such that women who experienced four or more ACEs displayed greater increases in NLR over time than women with 3 or fewer ACEs. History of childhood sexual abuse significantly predicted change in NLR over pregnancy (F = 6.39, p = .014) such that women with sexual abuse histories displayed greater increases in NLR over pregnancy compared to women without histories of childhood sexual abuse. See Table 2 and Figure 1(a). However, history of childhood physical abuse (F = 1.17, p = .28) or emotional abuse (F = 0.61, p = .44) did not significantly predict change in NLR over pregnancy.
Table 2.
Childhood sexual abuse history predicts increases in neutrophil-to-lymphocyte ratio over pregnancy.
| Model 1 (CSA) | Model 2 (Depression) | Model 3 (CSA + Depression) | |
|---|---|---|---|
| F (p value) | F (p value) | F (p value) | |
| Maternal age | 0.04 (.84) | 0.07 (.79) | 0.00 (.99) |
| Race | 0.51 (.48) | 0.03 (.86) | 0.23 (.63) |
| Body mass index | 0.73 (.40) | 0.32 (.58) | 0.67 (.42) |
| OSA diagnosis | 0.14 (.71) | 0.02 (.89) | 0.09 (.77) |
| Cigarette smoking | 0.03 (.87) | 0.04 (.85) | 0.10 (.75) |
| Childhood sexual abuse | 6.39 (.01) | – | 4.97 (.03) |
| Prenatal depression | – | 1.46 (.23) | 0.56 (.46) |
CSA: childhood sexual abuse; OSA: obstructive sleep apnea.
Note: Repeated measures analysis of variance was performed to examine effects of childhood adversity and prenatal depression on change in neutrophils-to-lymphocytes ratio (NLR) over gestation. Childhood sexual abuse history was measured using the Adverse Childhood Experiences (ACE) scale. Prenatal depression was measured using the Patient Health Questionnaire-9 (PHQ9).
Figure 1.
Pregnant women who experienced childhood sexual abuse, but not prenatal depression, exhibited greater increases in systemic inflammation over pregnancy. (a) Maternal history of childhood sexual abuse and neutrophil-to-lymphocyte ratio. (b) Prenatal depression and neutrophil-to-lymphocyte ratio. Note: Childhood adversity was measured using the Adverse Childhood Experiences Scale. Prenatal depression was measured using the Patient Health Questionnaire 9 (PHQ9). Neutrophils and lymphocytes were collected during routine prenatal care. The neutrophil-to-lymphocyte ratios were log transformed for analyses. We categorized women based on those with and without histories of childhood sexual abuse (a), and those with and without prenatal depression (b). Models adjusted for age, BMI, race, cigarette smoking in pregnancy, and obstructive sleep apnea diagnosis. Error bars reflect standard errors. CSA: childhood sexual abuse.
Prenatal depression did not significantly predict change in NLR across gestation (F = 1.46, p = .23). See Table 2 and Figure 1(b). NLR displayed greater increase over pregnancy among women with prenatal depression; however this did not reach statistical significance. When both prenatal depression and history of childhood sexual abuse were included as independent variables in the repeated measures ANOVA, history of childhood sexual abuse remained a significant predictor of change in NLR over gestation (F = 4.97, p = .030). See Table 2.
There was no statistically significant difference in change in NLR over pregnancy by adverse maternal pregnancy outcomes (F = 3.75, p = .058). We did not observe differences in change in NLR across gestation according to adverse neonatal outcomes (F = 0.17, p = .68).
Discussion
Results from this study indicate that NLR, a measure of systemic inflammation that is routinely collected during prenatal care, displayed greater increases over gestation among women who experienced histories of childhood sexual abuse relative to women who did not experience sexual abuse in childhood. Change in NLR over gestation did not vary by prenatal depression. Study findings are consistent with the biological embedding model of early adversity, indicating that childhood adversity results in a “pro-inflammatory phenotype” in adulthood. 1 Findings are also in line with results from our previous cross-sectional study in which pregnant women with histories of childhood adversity displayed elevated pro-inflammatory cytokine levels. 5
Rates of childhood sexual abuse in this study (20%) were aligned with national averages,32,33 and consistent with past evidence that childhood sexual abuse is uniquely dysregulating to stress-related biology in pregnancy. Specifically, studies have found that women with histories of childhood sexual abuse, with or without childhood physical abuse, displayed greater inflammation, 5 greater salivary cortisol 34 and greater hair cortisol 35 in pregnancy, particularly in the third trimester. Together, these findings suggest that childhood sexual abuse may lead to dysregulation in maternal hypothalamic–pituitary–adrenal (HPA) and immune activity over pregnancy. More research is needed to understand the psychological and biological underpinnings of these associations.
Neutrophils are the most abundant type of white blood cells and are the first to respond to bacterial infection or environmental insult. 36 Neutrophils also lead to secretion of several pro-inflammatory cytokines including IL-1β and IL-6. 37 The greater increase in NLR among women with histories of childhood sexual abuse, as found in this study, may suggest an increase in secretion of pro-inflammatory cytokines, as has been reported in past studies.5–8 In a population-based study, NLR values were observed to be highest in the second trimester. 26 In the current study, NLR levels were lower in first trimester and showed a greater rise in third trimester. This aberrant pattern in women with histories of childhood sexual abuse is consistent with findings from a previous study of the cortisol awakening response among pregnant women with childhood sexual abuse histories. 34 Specifically, cortisol awakening responses were lower in early pregnancy, and then rose precipitously in third trimester among women with histories of childhood sexual abuse. Taken together, these results suggest comparable patterns of dysregulated HPA and immune functioning across gestation in women with histories of childhood sexual abuse. Future research using larger sample sizes is needed to evaluate if greater increases in NLR over pregnancy have implications for adverse pregnancy outcomes and neonatal development.
There was no evidence that prenatal depression predicted changes in NLR across gestation. This finding is in contrast to prior evidence that prenatal depression is associated with elevated inflammation.10–12,38,39 Study results are consistent with past research that found that childhood adversity exerted a greater effect on biological dysregulation than adult stress. 13 Depressive symptoms among women in this study were also comparable to scores in samples of women with chronic health conditions such as migraine, diabetes, and hypertension.40–44 Patterns of results, while not statistically significant, indicated that women with prenatal depression displayed greater increases in NLR over gestation relative to women without prenatal depression. Future studies are needed using larger sample sizes to further evaluate the association between prenatal depression and NLR over gestation.
Increases in NLR over gestation were higher among women with adverse maternal outcomes of pregnancy, including gestational diabetes, gestational hypertension, or preeclampsia, which is consistent with past research, 21 however this did not reach statistical significance. No differences in NLR over gestation were found according to adverse neonatal outcomes. Given the small sample size and lack of statistical power to test differences in NLR according to adverse pregnancy outcomes, results should be interpreted with caution. More research is needed to evaluate the clinical utility of NLR in predicting adverse pregnancy outcomes.
Results from this study should be interpreted in light of several limitations. Most notably, participants in these analyses were from a larger study of pregnant women at risk for sleep disordered breathing based on obesity status and habitual snoring. Therefore, the sample is homogenous, and results may not generalize to a general obstetric population who are not overweight or obese and do not have other risk factors for sleep-disordered breathing. The sample size was also small, and more research is needed within larger samples to further evaluate trajectories of NLR across gestation among women who experienced childhood sexual abuse and prenatal depression. Information on socioeconomic factors was not available, and this is a limitation given the established association between low socioeconomic status and inflammation. 45 Finally, information on childhood adversity was collected via self-report and could be subject to recall bias or underreporting.
Strengths of this study include the novel investigation of NLR over pregnancy. NLR is an inexpensive and readily available marker of systemic inflammation that is collected routinely during prenatal care and has been investigated as a prognostic indicator for obstetric complications. Future research should be conducted to determine if NLR is useful in evaluating effectiveness of behavioral health interventions in the prenatal period, or if NLR could be used to inform the development of new interventions to improve perinatal health among women with histories of childhood adversity.
To conclude, maternal history of childhood sexual abuse was associated with increased systemic inflammation across gestation. These findings support recent guidelines which emphasize the importance of screening for history of sexual violence in prenatal women46,47 as trauma-informed care and psychological treatments may help to reduce risk for elevated inflammation among women with histories of childhood sexual abuse.
Acknowledgements
We would like to thank the women who participated in the study and Beth Hott for her assistance with this submission.
Declaration of conflicting interests: The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding: The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by National Heart Lung and Blood Institute, National Institutes of Health (R01HL130702 to GB); National Institute for Child Health and Human Development (R01HD 078515 to GB); National Institute of General Medical Sciences, National Institutes of Health (P20 GM103652 to MB); and Brown Medical School Summer Research Assistantship (to TF). Funding sources had no involvement in the design, collection, analysis and interpretation of the data, writing of the report, and in the decision to submit the article for publication.
Ethical approval: This study was approved by the Rhode Island Hospital Institutional Review Board (#781944 approved 23 September 2015).
Informed consent: Not applicable.
Guarantor: MB.
Contributorship: MB was responsible for data analysis and draft of the manuscript. TF was responsible for data collection of NLR and contributed to the first draft of the manuscript. MS contributed to interpretation of study findings and manuscript writing. MS contributed to data collection and manuscript writing. GB was responsible for study design and writing of the manuscript.
ORCID iDs: Margaret H Bublitz https://orcid.org/0000-0001-8765-7846
Ghada Bourjeily https://orcid.org/0000-0002-0543-4719
References
- 1.Miller GE, Chen E, Parker KJ. Psychological stress in childhood and susceptibility to the chronic diseases of aging: moving toward a model of behavioral and biological mechanisms. Psychol Bull 2011; 137: 959–997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Danese A, Pariante CM, Caspi A, et al. Childhood maltreatment predicts adult inflammation in a life-course study. Proc Natl Acad Sci U S A 2007; 104: 1319–1324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Loucks EB, Pilote L, Lynch JW, et al. Life course socioeconomic position is associated with inflammatory markers: the Framingham Offspring Study. Soc Sci Med 2010; 71: 187–195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Matthews KA, Chang YF, Thurston RC, et al. Child abuse is related to inflammation in mid-life women: role of obesity. Brain Behav Immun 2014; 36: 29–34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Bublitz M, De La Monte S, Martin S, et al. Childhood maltreatment and inflammation among pregnant women with gestational diabetes mellitus: a pilot study. Obstet Med 2017; 10: 120–124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Walsh K, Basu A, Werner E, et al. Associations among child abuse, depression, and interleukin-6 in pregnant adolescents: paradoxical findings. Psychosom Med 2016; 78: 920–930. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Blackmore ER, Moynihan JA, Rubinow DR, et al. Psychiatric symptoms and proinflammatory cytokines in pregnancy. Psychosom Med 2011; 73: 656–663. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Mitchell AM, Porter K, Christian LM. Examination of the role of obesity in the association between childhood trauma and inflammation during pregnancy. Health Psychol 2018; 37: 114–124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Wajid A, van Zanten SV, Mughal MK, et al. Adversity in childhood and depression in pregnancy. Arch Womens Ment Health 2020; 23: 169–180. [DOI] [PubMed] [Google Scholar]
- 10.Lahti-Pulkkinen M, Girchenko P, Robinson R, et al. Maternal depression and inflammation during pregnancy. Psychol Med 2019; 50: 1–13. [DOI] [PubMed] [Google Scholar]
- 11.Christian LM. Effects of stress and depression on inflammatory immune parameters in pregnancy. Am J Obstet Gynecol 2014; 211: 275–277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Cassidy-Bushrow AE, Peters RM, Johnson DA, et al. Association of depressive symptoms with inflammatory biomarkers among pregnant African-American women. J Reprod Immunol 2012; 94: 202–209. [DOI] [PubMed] [Google Scholar]
- 13.Austin MK, Chen E, Ross KM, et al. Early-life socioeconomic disadvantage, not current, predicts accelerated epigenetic aging of monocytes. Psychoneuroendocrinology 2018; 97: 131–134. [DOI] [PubMed] [Google Scholar]
- 14.Levine ME, Cole SW, Weir DR, et al. Childhood and later life stressors and increased inflammatory gene expression at older ages. Soc Sci Med 2015; 130: 16–22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Heim C, Newport DJ, Bonsall R, et al. Altered pituitary-adrenal axis responses to provocative challenge tests in adult survivors of childhood abuse. Am J Psych 2001; 158: 575–581. [DOI] [PubMed] [Google Scholar]
- 16.Pace TW, Mletzko TC, Alagbe O, et al. Increased stress-induced inflammatory responses in male patients with major depression and increased early life stress. Am J Psych 2006; 163: 1630–1633. [DOI] [PubMed] [Google Scholar]
- 17.Templeton AJ, McNamara MG, Seruga B, et al. Prognostic role of neutrophil-to-lymphocyte ratio in solid tumors: a systematic review and meta-analysis. J Natl Cancer Inst 2014; 106: dju124. [DOI] [PubMed] [Google Scholar]
- 18.Wang X, Zhang G, Jiang X, et al. Neutrophil to lymphocyte ratio in relation to risk of all-cause mortality and cardiovascular events among patients undergoing angiography or cardiac revascularization: a meta-analysis of observational studies. Atherosclerosis 2014; 234: 206–213. [DOI] [PubMed] [Google Scholar]
- 19.Penz M, Kirschbaum C, Buske-Kirschbaum A, et al. Stressful life events predict one-year change of leukocyte composition in peripheral blood. Psychoneuroendocrinology 2018; 94: 17–24. [DOI] [PubMed] [Google Scholar]
- 20.Aydin Sunbul E, Sunbul M, Yanartas O, et al. Increased neutrophil/lymphocyte ratio in patients with depression is correlated with the severity of depression and cardiovascular risk factors. Psychiatry Investig 2016; 13: 121–126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Sargin MA, Yassa M, Taymur BD, et al. Neutrophil-to-lymphocyte and platelet-to-lymphocyte ratios: are they useful for predicting gestational diabetes mellitus during pregnancy? Ther Clin Risk Manag 2016; 12: 657–665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Serin S, Avci F, Ercan O, et al. Is neutrophil/lymphocyte ratio a useful marker to predict the severity of pre-eclampsia? Pregnancy Hypertens 2016; 6: 22–25. [DOI] [PubMed] [Google Scholar]
- 23.Kang Q, Li W, Yu N, et al. Predictive role of neutrophil-to-lymphocyte ratio in preeclampsia: a meta-analysis including 3982 patients. Pregnancy Hypertens 2020; 20: 111–118. [DOI] [PubMed] [Google Scholar]
- 24.Christoforaki V, Zafeiriou Z, Daskalakis G, et al. First trimester neutrophil to lymphocyte ratio (NLR) and pregnancy outcome. J Obstet Gynaecol 2020; 40: 59–64. [DOI] [PubMed] [Google Scholar]
- 25.Kurban Y, Uyar AY, İ, Atak Z, et al. Investigation of neutrophil/lymphocyte ratio and mean platelet volume in patients diagnosed with preterm labor. Paediatr Respir Rev. Epub ahead of print 2020. [DOI] [PubMed] [Google Scholar]
- 26.Hershko Klement A, Hadi E, Asali A, et al. Neutrophils to lymphocytes ratio and platelets to lymphocytes ratio in pregnancy: a population study. PLoS One 2018; 13: e0196706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Al-Halawani M, Kyung C, Liang F, et al. Treatment of obstructive sleep apnea with CPAP improves chronic inflammation measured by neutrophil-to-lymphocyte ratio. J Clin Sleep Med 2020; 16: 251–257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Hughes K, Bellis MA, Hardcastle KA, et al. The effect of multiple adverse childhood experiences on health: a systematic review and meta-analysis. Lancet Public Health 2017; 2: e356–e66. [DOI] [PubMed] [Google Scholar]
- 29.Kroenke K, Spitzer RL, Williams JB. The PHQ-9: validity of a brief depression severity measure. J Gen Intern Med 2001; 16: 606–613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Flynn HA, Sexton M, Ratliff S, et al. Comparative performance of the Edinburgh Postnatal Depression Scale and the Patient Health Questionnaire-9 in pregnant and postpartum women seeking psychiatric services. Psych Res 2011; 187: 130–134. [DOI] [PubMed] [Google Scholar]
- 31.Yawn BP, Pace W, Wollan PC, et al. Concordance of Edinburgh Postnatal Depression Scale (EPDS) and Patient Health Questionnaire (PHQ-9) to assess increased risk of depression among postpartum women. J Am Board Fam Med 2009; 22: 483–491. [DOI] [PubMed] [Google Scholar]
- 32.Frankenberger DJ, Clements-Nolle K, Yang W. The association between adverse childhood experiences and alcohol use during pregnancy in a representative sample of adult women. Womens Health Issues 2015; 25: 688–695. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Smith MV, Gotman N, Yonkers KA. Early childhood adversity and pregnancy outcomes. Matern Child Health J 2016; 20: 790–798. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Bublitz MH, Stroud LR. Childhood sexual abuse is associated with cortisol awakening response over pregnancy: preliminary findings. Psychoneuroendocrinology 2012; 37: 1425–1430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Schreier HM, Enlow MB, Ritz T, et al. Childhood abuse is associated with increased hair cortisol levels among urban pregnant women. J Epidemiol Commun Health 2015; 69: 1169–1174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Jacobs L, Nawrot TS, de Geus B, et al. Subclinical responses in healthy cyclists briefly exposed to traffic-related air pollution: an intervention study. Environ Health 2010; 9: 64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Tecchio C, Micheletti A, Cassatella MA. Neutrophil-derived cytokines: facts beyond expression. Front Immunol 2014; 5: 508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Coussons-Read ME, Okun ML, Nettles CD. Psychosocial stress increases inflammatory markers and alters cytokine production across pregnancy. Brain Behav Immun 2007; 21: 343–350. [DOI] [PubMed] [Google Scholar]
- 39.Coussons-Read ME, Okun ML, Schmitt MP, et al. Prenatal stress alters cytokine levels in a manner that may endanger human pregnancy. Psychosom Med 2005; 67: 625–631. [DOI] [PubMed] [Google Scholar]
- 40.Katon JG, Russo J, Gavin AR, et al. Diabetes and depression in pregnancy: is there an association? J Womens Health 2011; 20: 983–989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Katon WJ, Russo JE, Melville JL, et al. Depression in pregnancy is associated with preexisting but not pregnancy-induced hypertension. Gen Hosp Psych 2012; 34: 9–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Orta OR, Gelaye B, Qiu C, et al. Depression, anxiety and stress among pregnant migraineurs in a pacific-northwest cohort. J Affect Disord 2015; 172: 390–396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Huang JY, Arnold D, Qiu CF, et al. Association of serum vitamin D with symptoms of depression and anxiety in early pregnancy. J Womens Health 2014; 23: 588–595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Zhong Q, Gelaye B, Rondon M, et al. Comparative performance of Patient Health Questionnaire-9 and Edinburgh Postnatal Depression Scale for screening antepartum depression. J Affect Disord 2014; 162: 1–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Milaniak I, Jaffee SR. Childhood socioeconomic status and inflammation: a systematic review and meta-analysis. Brain Behav Immun 2019; 78: 161–176. [DOI] [PubMed] [Google Scholar]
- 46.ACOG Committee Opinion No. 757. Screening for perinatal depression. Obstet Gynecol 2018; 132: e208–e12. [DOI] [PubMed] [Google Scholar]
- 47.ACOG Committee Opinion No. 777. Sexual assault. Obstet Gynecol 2019; 133: e296–e302. [DOI] [PubMed] [Google Scholar]