Abstract
Neutrophils play significant roles in immune homeostasis and as neutralizers of microbial infections. Recent evidence further suggests heterogeneity of neutrophil developmental and activation states that exert specialized effector functions during inflammatory disease conditions. Neutrophils can play multiple roles during viral infections, secreting inflammatory mediators and cytokines that contribute significantly to host defense and pathogenicity. However, their roles in viral immunity are not well understood. In this review, we present an overview of neutrophil heterogeneity and its impact on the course and severity of viral respiratory infectious diseases. We focus on the evidence demonstrating the crucial roles neutrophils play in the immune response toward respiratory infections, using influenza as a model. We further extend the understanding of neutrophil function with the studies pertaining to COVID‐19 disease and its neutrophil‐associated pathologies. Finally, we discuss the relevance of these results for future therapeutic options through targeting and regulating neutrophil‐specific responses.
Keywords: anti‐viral functions, COVID‐19, heterogeneity, Influenza A, neutrophils, tissue damage, Viral Respiratory Diseases
Graphical Abstract
Review on the role neutrophils play during viral respiratory diseases, evaluating their various pro‐ and anti‐ viral functions leading to disease resolution or pathology.
1. INTRODUCTION
Neutrophils are known as early responders against bacterial or fungal infections, releasing their powerful antimicrobial arsenal to neutralize and contain the infection. Neutrophils were first reported by Elie Metchnikoff in 1883 as professional phagocytes. 1 They are capable of engulfing foreign microbes and neutralizing them with various secretory granules 2 and potent reactive oxygen species (ROS). 3 Neutrophils also have the unique ability to release neutrophil extracellular traps (NETs) to limit the spread of infections. 4 These NETs were shown to help neutralize large fungal hypha together with the secretion of cytotoxic granules. 5 , 6 The effects of neutrophil‐mediated immunity are further amplified as neutrophils work in large numbers, forming a concerted effort to eliminate foreign threats. Although beneficial, these effects can often be detrimental to the host tissues and the immune response, 7 , 8 including further exacerbation of organ damage, resulting in critical and life‐threatening conditions. 9 In viral diseases, however, the roles neutrophils play are not well understood. Furthermore, the recent evidence of a strong neutrophil presence in severe acute respiratory syndrome coronavirus (SARS‐CoV2)‐infected patients has garnered interest in the function these neutrophils elicit in the progression of COVID‐19 disease. 10 , 11
Neutrophils are innate immune cells that are frequently overlooked in discussions of viral immunity, in part because adaptive immune cells such as T and B cells are recognized to play essential roles in initiating cytotoxic killing and antibody generation in response to viral infection, both of which are major steps toward viral clearance. While the role of neutrophils in viral infection is still unclear, growing evidence suggests that neutrophils play a role in resolving viral infection. For instance, data from preclinical mouse models of influenza A virus (IAV) infection show depletion of neutrophils by anti‐Ly6G or anti‐Gr1 antibody treatment led to severe disease outcomes in infected mice. 12 , 13 Similar findings were also made in encephalitis 14 and HSV‐1 15 infection models, supporting the need for neutrophils to achieve optimal viral immunity. Studies have also demonstrated that mice with deficient inflammasome signaling through the NLPR3 pathway have poor neutrophil recruitment due to decreased production of neutrophil chemokines such as KC, TNF‐α, and IL‐1b, which results in severe pathology and mortality following an IAV infection. 16 , 17 Neutrophils in respiratory syncytial virus (RSV) infections, on the other hand, has been proven in several studies to have no influence on viral load or pathogenicity. 18 , 19 , 20 Although some studies suggest that neutrophils play a beneficial or redundant role in the body, clinical observation indicated that a high neutrophil count is associated with the severity of many diseases. For instance, hematologic analysis and clinical studies of COVID‐19 patients’ neutrophil counts showed strong association with disease severity. 21 , 22 , 23 It has been proposed that the neutrophil‐to‐lymphocyte ratio (NLR), which has been used to stratify cancer patients, 24 , 25 may be utilized as a predictive and prognostic marker for COVID‐19 patients. 10 , 11 , 26 These results suggest that the presence of neutrophils in severe types of inflammation is linked to disease severity in COVID‐19 patients. Here, we will review the current evidence of neutrophil heterogeneity and consolidate information on neutrophil function in viral respiratory infection. We expect to better understand the role of neutrophils in the immune response to viral respiratory infections and their participation in the pathology of the most severe cases.
2. DEVELOPMENT AND FUNCTIONAL HETEROGENEITY OF NEUTROPHILS
The daily production of billions of neutrophils takes place within the bone marrow, where committed progenitors reside in specialized niches providing growth signals and developmental cues. Due to the short lifespan of neutrophils, this developmental process is critical in producing a steady supply to the circulation. Granulopoiesis is therefore well studied, and is described by the characterization of the various maturation stages through their granule content and morphologic attributes. 27 According to their granule content and nuclear shape, neutrophils are produced in a step‐wise order of early myoblasts → promyelocytes →> myelocytes → metamyelocytes → band cells → segmented neutrophils. 28 , 29 , 30 , 31 Under homeostatic conditions, only the mature segmented neutrophils exit the bone marrow, performing their various roles. 32 However, during inflammatory conditions, hematologists typically observe immature phenotypes of neutrophils in the circulation. This is commonly known as a left shift and is used as an indicator of inflammation. 33
These immature phenotypes have been widely reported in various inflammatory conditions, ranging from cancer, pregnancy, stress, cardiovascular diseases, and notably viral infections. 34 , 35 , 36 , 37 , 38 The way these immature neutrophils are characterized differs between research groups. There is no standardized approach for phenotyping neutrophil subsets, making it difficult to corroborate functional data from multiple laboratories. Some groups have proposed markers to identify these subsets, such as CD10, 39 CD177, 40 Olfactomedin‐4 (OLFM4), 41 and CD49d. 42 Separating these subtypes can be difficult since they might emerge from various distinct neutrophil precursor stages with different degrees of maturity. A more profound knowledge of the ontogeny of the neutrophils will be required to overcome this issue. In recent years, several research groups have characterized neutrophil development at each step of differentiation and maturation. The advancements in single‐cell technologies and sequencing techniques have led to the identification and characterization of various neutrophil progenitors and precursors in both mice and humans. 32 , 43 , 44 , 45 , 46 This includes the early progenitors, such as the proNeus 46 and eNePs, 45 as well as late precursors preNeus, 32 NePs, 44 and NeuPs. 47 In‐depth single‐cell analyses further reveal finer transcriptomic distinct developmental stages 43 , 48 (Figure 1). These studies demonstrate that immature neutrophils can exist in the circulation during disease states, creating a heterogeneity of developmental states that can potentially elicit various unique functions in response to the inflammatory stimulus. However, it is still unclear if these immature forms go on to become functionally distinct mature neutrophils.
FIGURE 1.
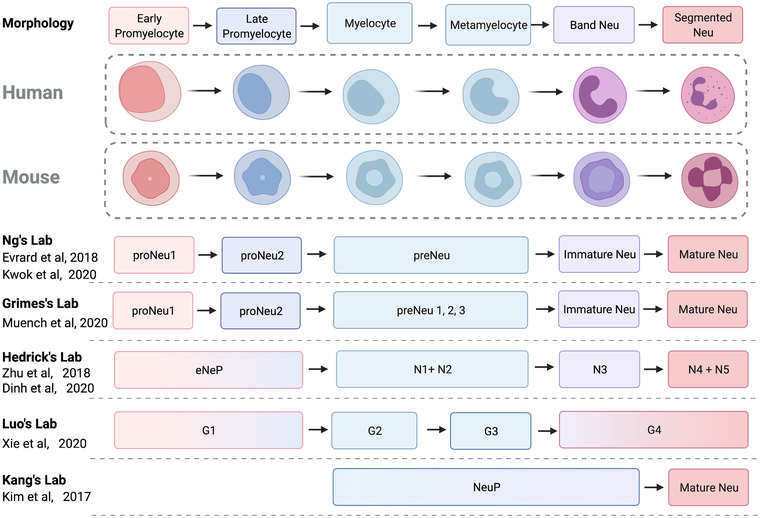
Characterization of neutrophil subsets in mice and humans. Neutrophil development is historically characterized by various morphologic structures and granules using bone marrow smears. Undifferentiated myeloblasts differentiate into promyelocytes, myelocytes, nonproliferating meta‐myelocytes, band cells, and finally mature as segmented neutrophils. These stages are accompanied by stage‐specific granules. The advent of high dimensional single‐cell phenotyping technologies enabled both the transcriptome and protein expression characterization of the developmental continuum, giving rise to the identification of discrete subsets proposed by various groups. These enable the study of neutrophils in viral inflammatory conditions and discover subset‐specific functions leading to disease pathology and resolution. Created with BioRender.com
Mature neutrophils undergo an ageing process when released into the circulation, decreasing their expression of L selectin (CD62L) while increasing chemokine receptor CXCR4 expression. 49 , 50 Aged neutrophils are then cleared from the circulation by specialized efferocytic macrophages present in the lung, liver, and spleen. 27 This physiologic process regulates granulopoiesis, providing feedback signaling to the bone marrow through an IL‐23 and IL‐17‐mediated manner. 51 Clearance of aged neutrophils also prevents unwanted necrosis or spillage of their store of cytotoxic granules. 52 Interfering with this clearance process results in an accumulation of circulating aged neutrophils. This build‐up of exhausted phagocytes has been shown to play significant roles in disease. Aged neutrophils have a much higher phagocytic activity as compared with the non‐aged neutrophils. 53 Additionally, they respond faster toward inflammatory signals, migrating to sites of infection to neutralize threats. A transgenic mouse model affecting the ageing process in neutrophils showed these distinctions in functionality between aged and fresh neutrophils. In this model, the group showed that mice with mostly aged neutrophils were able to survive better against fungal infections as compared with mice with mostly fresh neutrophils. However, aged neutrophils confer a worse disease pathology in a vascular inflammation model, resulting in larger infarction sizes and poorer survival. 54 Therefore, it is important to consider the heterogeneity of neutrophil age states in addition to the developmental state of the circulating neutrophils during inflammatory states such as viral infections.
Neutrophils in the circulation also comprise a subset of myeloid‐derived suppressor cells that possess immune‐modulating properties observed in various conditions such as cancer, pregnancy, and sepsis. 35 , 55 , 56 These neutrophils possess a general identity of Ly6G+CD11b+ and can have a morphologic resemblance of both immature and mature neutrophil phenotypes as described by various groups. 57 These suppressor cells are reported to inhibit T cell proliferation and activation, dampening the immune response. They may also be akin to low‐density neutrophils (LDN) found in the mononuclear fraction after density gradient separation. 58 LDNs are also reported as a combination of immature and mature phenotypes observed first in SLE 58 and rheumatoid arthritis. 59 These cells have perhaps degranulated and accumulated in the bloodstream of these patients, as reported by some groups. 60 , 61 This indicates that neutrophils can exist in various states, eliciting both beneficial and detrimental functions to the immune response.
In the lung specifically, a major subset of neutrophils exists as a marginated, intravascular pool, adhering to the endothelium of capillaries and postcapillary venules. These neutrophils have been shown to be activated once in the lung, acquiring higher expression of adhesion receptors such as CD11b but lower expression of CD62L. 62 Notably, the authors argued that this phenomenon occurs regardless of the inflammatory condition. Recent evidence in mice further proposes that neutrophils change at the transcriptome level when they infiltrate various tissues. 63 In the study, the group showed a lung‐specific signature in the neutrophils, possessing proangiogenic genes involved in vascular growth and repair. Intravital imaging of lung neutrophils showed that they interact closely with B cells, allowing for their clearance by macrophages. A disruption of neutrophil clearance by B cell depletion was shown to cause pathologic consequences. 64 This opens new questions on how tissue neutrophils, such as those in the lungs, are activated during viral respiratory diseases (VRDs) and how this might impact their function against the infection.
3. NEUTROPHILS AND VRDs
Respiratory viruses are one of the greatest contributors to endemics and pandemics in the history of humankind, with a significant morbidity and mortality rate. 65 , 66 The most common ones are influenza virus, RSV, parainfluenza virus, metapneumovirus, rhinovirus (RV), coronavirus, and adenovirus. They can affect the upper respiratory tract, which presents mild to moderate symptoms such as fever, cough, sore throat, and/or running nose. 67 , 68 This infection can progress to the lower respiratory tract, which causes damage to the lungs and results in severe symptoms such as pneumonia. 65 , 68 , 69 , 70 , 71 , 72 , 73 , 74 , 75 , 76 , 77 , 78 In pediatric hospitalizations for lower respiratory tract illnesses (LRTIs), 40% are caused by seasonal parainfluenza virus epidemics. 72 RV, typically associated with upper respiratory tract illnesses (URTIs) such as the common cold, has been reported to result in severe LRTIs and is the second leading cause of pneumonia and bronchiolitis in infants and young children. 74 In IAV infections, clinical studies have reported pneumonia as the most frequent severe clinical manifestation, affecting an estimated one‐third of the IAV‐infected and hospitalized patients. 79 , 80 , 81 Severe pneumonia can progressively develop into acute respiratory distress syndrome (ARDS), the most severe form of acute respiratory failure. 82 A life‐threatening respiratory condition with a pooled mortality rate of 43% across various evaluated studies, ARDS is characterized by pulmonary edema with large infiltration of neutrophils into the interstitial and bronchioalveolar space. 83
Infiltration of neutrophils is a common trait in VRDs, reported in IAV, RSV, metapneumovirus, RV, adenovirus, and coronaviruses. 79 , 84 , 85 , 86 , 87 , 88 In RSV infection, an increased number of neutrophils in the lung is a hallmark for disease severity in both humans and mice. 89 , 90 Severe RSV‐infected infants have neutrophils as the predominant cell type in the bronchoalveolar lavage (BAL). 89 An increase in lung neutrophils, markers, and genes of neutrophil function and activation has been shown in severe IAV and SARS‐CoV‐2 infections. 91 , 92 , 93 , 94 , 95 , 96 , 97 Additionally, neutrophil infiltration is also observed in URTIs. RV and adenovirus are typical causes of URTIs, such as the common cold. In the early course of cold from symptomatic RV‐infected patients, neutrophils have been shown to infiltrate the nasal mucosa and secretion. 98 During the common cold of adenovirus‐infected children, a high level of neutrophils, HNP‐1, ‐3, and ‐4 was observed in their upper respiratory tracts. 99 This increased level of neutrophils observed across respiratory viral infections emphasizes the importance of studying the role of neutrophils in VRDs.
4. TAKING EVIDENCE FROM IAV INFECTIONS
Before delving into the biologic significance of neutrophil subsets for viral immunity, we first integrate the current evidence of neutrophils in viral respiratory infections to ascertain their functions, both beneficial and detrimental to the host. Among the numerous research involving neutrophils and viral infections, IAV infection investigations have made significant contributions to our understanding of the several functions neutrophils can play during a VRD. IAV is the cause of the yearly seasonal flu and the global human flu pandemics. 100 Since 1900, 5 influenza pandemics has hit the world, with the most recent 2009 IAV H1N1 causing over half a million deaths globally. 101 Moreover, a global estimate of 5 million severely diseased and 650 thousand respiratory deaths has been associated with seasonal influenza each year. 102 Due to its seasonal emergence and multiple subtypes, IAV is a persistent global public health concern that results in a spectrum of pathologic severity. 100 Due to the diversity of individual patients and the difficulties of conducting mechanistic research on human individuals, IAV infection studies are widely carried out on mice, which provides clues on the pathology and responses that neutrophils participate in. 103 It is important to consider murine models of viral infections as well as human studies as they, together, form a cohesive understanding of the mechanisms and underlying pathologies. When possible, we also incorporate relevant studies of other respiratory viruses to complement our comprehension of neutrophil function.
5. ANTIVIRAL NEUTROPHIL FUNCTIONS IN VRDs
Neutrophils are phagocytes, and their ability to engulf viral particles suggests a possible antiviral function. Indeed, Mullarkey et al. 104 demonstrated that neutrophils perform antibody‐dependent cellular phagocytosis (ADCP) on IAV through Hemagglutinin stalk protein‐specific IgG antibodies. The opsonized viral particles are phagocytosed by neutrophils resulting in the generation of ROS and perhaps the elimination of virus. In a study using a flank model of Modified Vaccinia Ankara (MVA) infection, it was demonstrated that neutrophils harboring viruses were shown to be APCs. Duffy et al. 105 discovered the infected neutrophils that homed to the bone marrow activated residing CD8+ memory T cells specific to MVA. This effect was later seen to be abrogated by a disruption in neutrophil migration through CCR1 signaling. Additionally, Hufford et al. 106 reported de novo synthesis of viral RNA and protein of IAV in neutrophils, suggesting how they can be infected and serve as antigen‐presenter cells to CD8+ T cells for antiviral immunity function. Additionally, Hufford's group showed an increase in IFNγ production in activated CD8+ T cells, but not CD4+ T cells, and the depletion of neutrophils through anti‐Ly6G antibodies showed a significant decrease of these activated CD8+ T cells in the lungs. 106 Notably, while being infected and act as transporters/presenters, neutrophils were incapable of supporting active IAV growth. 107 In an elegant study by Lim et al., 108 neutrophils were shown to secrete the chemokine CXCL12, which induced the migration of influenza‐specific CD8+ T cells to the infected lung. This function of T cell recruitment was lost with either neutrophil‐specific knock‐out of CXCL12 or blocking its receptor, CXCR4, through the inhibitor AMD3100.
Degranulation of antimicrobial peptides and mediators has been shown to greatly damage lung epithelial cells and tight junction integrity. 109 , 110 These powerful proteinases and granules serve to eliminate microbial threats and contain infections. Though it seems unnecessary in viral infections, these mediators are key players in viral clearance. In VRDs like RSV, matrix metalloproteinase 9 (MMP‐9) is important for viral clearance. 111 Dabo et al. 111 demonstrated that MMP‐9 decreases RSV infectivity and modulates neutrophil recruitment and cytokine generation in the lung using an MMP‐9 knock‐out model of RSV infection. Another study reported similar findings, showing that secretion of MMP‐9 through TLR signaling was required for neutrophil migration to the lung on IAV‐infected mice. 112 It is still unclear how MMP‐9 reduces infectivity of RSV, possibly disrupting viral attachment or indirectly triggering signaling cascades of receptors MMP‐9 is known to bind to. 113 , 114 Apart from MMP‐9, it was demonstrated that the neutrophil‐derived cathelicidin LL‐37 has direct antiviral effects, interacting with the virus and decreasing its virulence, suppressing both IAV and RSV infections. Separately, antimicrobial peptides called human neutrophil peptides can neutralize IAV through a process of viral aggregation. This process was shown to promote uptake of IAV by neutrophils, preventing further infections. 115 , 116 Along with antimicrobial peptides, neutrophils produce considerable amounts of ROS via oxidative burst, which can be used to remove phagocytosed material or cause tissue damage. Oxidative burst has been detected in mice during IAV and RSV infection. 117 , 118 , 119 Excessive amounts of ROS, when inflammation is prolonged, can often lead to severe disease pathology in IAV infection. 117 , 120 Additionally, mediators like myeloperoxidase (MPO) and neutrophil elastase (NE) can either proteolyse or catalyze ROS production. 121 MPO was shown to be important for viral clearance but also a contributor of lung tissue damage. 122 Similarly, NE has been found in the respiratory tract and serum of severe RSV‐infected infants, potentially contributing to disease pathology. 123 , 124 Interestingly, individuals with chronic granulomatous disease exhibit ROS‐deficient neutrophils but no increased vulnerability to VRDs, indicating that ROS may be primarily engaged in disease pathogenesis as the disease progresses. 125
ROS is also linked to another important neutrophil function: NET formation. NETosis has garnered much contention as an antiviral role in VRDs. Persistent NET formation in IAV‐infected models was associated with lung damage and increased pathogenesis, as NETs are highly toxic. 126 , 127 ARD‐related histopathologic manifestations such as diffuse alveolar damage (DAD) caused by alveolar injury were entangled with NETs. NET formation and endothelial damage were detected when alveolar epithelial cells (AECs) were infected, suggesting the potential link of NET formation to alveolar damage in IAV‐infected patients. Additionally, high extracellular histones levels were found in the lungs of IAV‐infected mice and were shown to exacerbate lung pathology. 128 RSV‐infected epithelial cells were shown to be recognized by neutrophils, triggering NETosis, 129 , 130 , 131 , 132 , 133 which can capture RSV particles in vitro. 133 Additionally, histones have been shown to neutralize H3N2 and H1N1 IAV. 134 These findings highlight NETosis as potential antiviral capabilities against VRDs. On the other hand, close examinations of patients’ sputum, lung biopsies, or mouse models with severe VRDs frequently reveal elevated NETs and neutrophil levels, suggesting neutrophils as major drivers of disease pathology and mortality in severe disease. 128 , 135 , 136 Most of the airway obstructions during severe RSV infections were found to have NETs plugs. 133 As a result of this, airway obstruction and poor prognosis may be linked to an increased neutrophil response by NETs. However, these observations are likely a consequence of uncontrolled disease progression, led by dysregulation of cellular signaling and unresolved inflammation. 137 , 138
After executing their functions, neutrophils can perform an additional function in the lung by secreting epidermal growth factor (EGF), which has been demonstrated to be required for monocyte development into competent APCs. A recent study showed that the absence of EGF significantly reduced CD8+ T cell cytokine expression and activation, and showed a marked decrease in dendritic cell maturation markers like MHCII and CD86. 139 Collectively, the data suggest that neutrophils may carry out their antiviral activities through indirect processes.
6. RECRUITMENT OF NEUTROPHILS TO THE INFECTED RESPIRATORY TRACT
The innate immune players in the lung include the residential lung leukocytes and the epithelial cells lining the alveolar surface and the conducting airways. 140 The immune cells in healthy lung tissues are ∼95% alveolar macrophages, 1–4% lymphocytes, and ∼1% neutrophils. Pattern recognition receptors (PRRs) such as TLRs are found on alveolar walls and epithelial cells lining the respiratory tract to sense pathogen‐associated molecular patterns and damage‐associated molecular patterns. 141 , 142 , 143 , 144 These epithelial cells in the airway are the first to be infected, 145 secreting proinflammatory cytokines and chemokines to activate and recruit leukocytes into the infected respiratory tract. 146 , 147 These mediators include MIP‐1α, TNF‐α, IL‐1, IL‐6, CXCL1, CXCL2, CXCL10, and IL‐17. 148 , 149 , 150 , 151 Enhanced levels of proinflammatory cytokines and Type I IFNs are also contributed by infected alveolar macrophages. 152 , 153 , 154 , 155 , 156 Depending on the inflammatory stimuli, massive infiltration can occur, leading to lung tissue damage and loss of lung function 147 , 157 , 158 (Figure 2).
FIGURE 2.
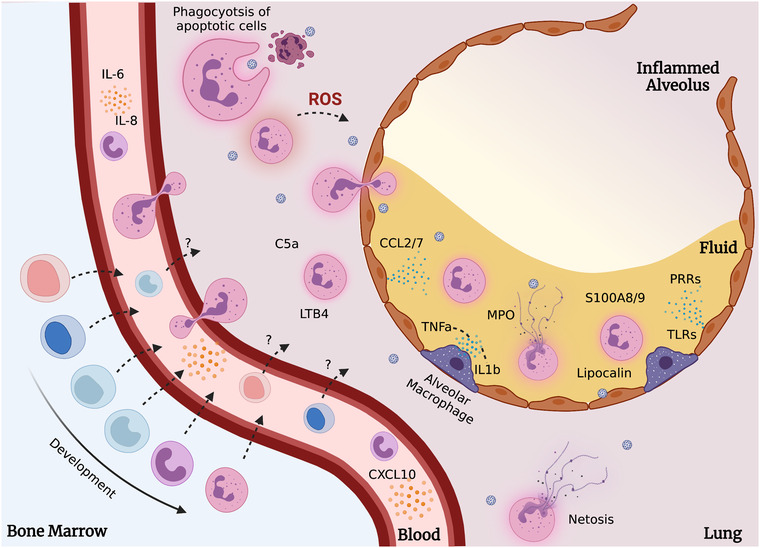
Neutrophil recruitment and action in the infected airway tract. Evidence now shows the presence of developing neutrophil precursors in the circulation during viral infections. These subsets may provide immunosuppressive or effector functions that are not fully understood. In the infected lungs, through recruitment by cytokines (such as IL‐1b, LTB4, C5a, and TNF‐α), TLRs, and pattern recognition receptors (PRRs), neutrophils perform various functions that help control the infection. Left unchecked, these actions lead toward increased disease severity and tissue damage. Created with BioRender.com
Following IAV infection and activation of tissue‐resident macrophages and epithelial cells, neutrophils are the first wave of immune cells recruited to the site of infection, which is approximately 2 days after the IAV infection. 13 , 159 , 160 , 161 During IAV, RSV, and SARS‐CoV infection, MyD88/TRIF signaling is crucial for recruitment of neutrophil to the lung. 84 , 162 , 163 , 164 , 165 Following IAV infection, an increase in both local and systemic levels of neutrophils was commonly observed in clinical patients, mice, and ferrets. This neutrophilia correlates with the increased IL‐6, CCL2, CXCL8, and CXCL10 levels and disease severity of IAV infections. 153 , 166 , 167 , 168 The release of proinflammatory cytokines (e.g., TNF‐α , IL‐1, and IL‐6), chemokines (CCL2, CCL7, CXCR8), complement component C5a, and leukotriene B4 attracts neutrophils to the infected respiratory airway. 83 , 169 , 170 , 171 , 172 Similarly, these molecules were highly associated with increased disease severity.
After migration to the infected airway, neutrophils are found to confer a protective function to the host in both the early and late stages of the infection. 67 Cell depletion studies in mice revealed that recuperation from severe cases of IAV depended heavily on neutrophils presence and contribution. 13 Interestingly, mild IAV infection did not exhibit a similar dependency on neutrophils. Moreover, studies in mice using IAV strains with varying levels of virulence suggest that neutrophil response is dependent on the pathogenicity of the viral infection. 173
In the lungs, the release of proinflammatory cytokine IL‐6 is essential to stimulate neutrophil survival and promote viral clearance, as it can reinstate antiapoptotic factors levels (Mcl‐1 and Bcl‐XL) suppressed by IAV. 168 Low levels of IL‐6, coupled with low neutrophil numbers, strongly correlated with increased disease severity. However, retention of neutrophils might not be necessarily beneficial. Retainment of lung neutrophils has been associated with IAV disease severity, and observed especially in highly pathogenic IAV strains. 112 , 155 , 173 , 174 CXCL8 (a neutrophil chemoattractant) increase in lung airspaces was correlated to elevated neutrophil numbers from recovered patients with ARDS. 175 , 176 Notably, this increase was correlated to higher disease severity and symptoms.
As mentioned, neutrophils are needed in the early stage of IAV infection for viral clearance. They are effectors in viral clearance, but the accumulation of neutrophils are signs of dysregulated inflammatory signaling and life‐threatening tissue damage. In IAV, mild lung pathology can be observed in neutrophil‐depleted mice, while ARDS‐like pathology and excessive neutrophil infiltration were found in macrophage‐depleted mice. 110 Hence, neutrophil responses need to be well balanced during IAV to have an adequate but not excessive inflammation response. It might also be more beneficial for neutrophils to have early apoptosis later into the IAV infection to prevent excessive neutrophil accumulation in the lung.
7. SEX, AGE, AND THE ACCUMULATION OF NEUTROPHILS IN INFECTED LUNGS
IAV infection severity has been associated with multiple demographic factors such as age and sex. 177 For both seasonal and pandemic strains of IAV, children below the age of 10 and adults above age 65 have an increased risk of disease severity. Their immunocompromised nature leaves them more vulnerable to infections. Interestingly, young adults (15–49 years of age) have severe outcomes during IAV pandemics as compared with seasonal outbreaks, and this increased risk is seen more in females than in males. This observation was attributed to the male reproductive hormone, testosterone, that has been shown to confer protection in male mice. 178 Interestingly, treatment of female mice with high doses of exogenous estradiol showed higher survival rates. 179 In estradiol‐treated female mice, increased neutrophil recruitment was observed, and the depletion of neutrophils reverses the protective effects of the treatment. 180 This suggests that the protective effect of estradiol is influenced by its amount produced, which might account for the higher risk in females. Future studies are required to study the role of sex differences and the response toward IAV and other VRDs.
While other respiratory viruses such as the RSV, parainfluenza virus, metapneumovirus, RV, and adenovirus affect mainly children, 68 the influenza virus and coronavirus have more disease complications in elderly of 65 years old and above. 65 , 181 , 182 Young children can excrete the viruses earlier and over a longer period of time than adults. 65 A recent study conducted by Kulkarni et al. 174 between lung neutrophil infiltration and age demonstrated an increased level of neutrophils in aged compared with young mice. This observation might have been contributed by the elevated secretion of neutrophil‐recruiting chemokines, CXCL1 and CXCL2, by AECs in infected, aged mice. The receptor of these neutrophil chemoattractants, CXCR2, is highly expressed on circulating neutrophils for chemotaxis to the site of inflammation. 183 CXCR2 has been shown to play a major role in neutrophil migration to the lungs during influenza infection, but the neutrophils recruited were reported as dispensable for influenza viral clearance. 86 , 184 , 185 , 186 This suggests that the increase in disease severity and mortality rate concerning age is related to the increased secretion of CXCL1 and CXCL2, which attract excessive numbers of neutrophils to the lungs and confer a pathogenic effect. 174 This corresponds to ageing studies in humans, where individuals aged above 60 have deficiencies in circulating neutrophils and reductions in neutrophil effector functions such as phagocytosis of bacteria, production of NETs, ROS, and migration. 187
However, a study conducted by Lu et al. 188 demonstrated increased resistance to IAV infection in aged as compared with young mice. While younger mice showed a faster viral clearance, they also had a higher mortality rate and tissue damage. Due to their weakened immune system, aged mice cleared viruses more slowly without causing tissue damage. Studies by Kulkarni et al. 174 and Lu et al. 188 employed the same viral strain (H1N1; PR8) and mouse strain (female C57BL/6), however their findings were inconsistent. Notably, in the second study by Lu et al., 188 the age range of the mice is significantly older. Thus, this age difference in mice may account for the observed disparity in experimental results. Hence, this suggests that stronger immune responses might not result in better survival rates, and therapies targeted at reducing excessive neutrophil levels may have to take into consideration the age group and sex of the patient.
With age, neutrophil functions such as phagocytosis and ROS production decline. 189 , 190 Numerous retrospective studies have demonstrated, severe COVID‐19 disease is frequently observed in elderly patients. 22 , 191 , 192 , 193 , 194 Transcriptomic and cytokine analyses of aged COVID‐19 patients revealed higher degranulation signatures and IFN‐γ signaling. This was also similarly seen using a non‐human primate model of SARS‐CoV2 infection, including a higher level of VEGF in the lungs of old macaques. 195 Interestingly, the airway epithelial cells of children with SARS‐CoV2 possessed augmented antiviral sensing and immune cell activation. 196 This, along with the higher viral sensing and IFN production in myeloid cells, prevents children from acquiring severe disease pathology.
8. CORONAVIRUS (SARS, MERS, SARS‐COV2): HIGH NEUTROPHIL COUNTS OBSERVED IN CORONAVIRUS‐INFECTED PATIENTS
Coronaviruses are enveloped, positive‐sense single‐stranded RNA viruses with spike‐like structures on their viral surfaces when observed under the electron microscope. 197 , 198 Similar to IAV, they can cause mild to severe respiratory infections in humans that progress toward fatal outcomes. 197 Since 2000, the emergence of highly pathogenic coronavirus as pandemics has been an unsolved global public health concern. SARS‐CoV in 2002 and Middle East respiratory syndrome coronavirus (MERS‐CoV) in 2012 have caused a high mortality rate of 9.6% and 35.5%, respectively. 199
Only a few investigations have been conducted to determine the involvement of neutrophils in SARS‐CoV and MERS‐CoV infection. Animal models that are ideal for studying these coronaviruses are currently being investigated. 200 , 201 , 202 , 203 In the acute phase of SARS‐CoV infection, a high neutrophil count and a low number of CD4+ and CD8+ T cells were observed in patients' blood, which was correlated with unfavorable consequences. 204 , 205 , 206 The excessive neutrophils may have contributed to lesion formation in SARS‐CoV‐infected patients via secretion of granules and ROS that give rise to necrosis in neighboring cells and recruitment of other inflammatory cells. For MERS‐CoV infection, high levels of IL‐8 (CXCL8), IL‐1α, and IL‐1β were expressed in the lower respiratory tract of patients. 207 These cytokines positively correlated with case fatality rates that were reported. Notably, the high expression level of IL‐8 and IL‐1β in MER‐CoV patients can be drawn parallel to the studies conducted with IAV. 175 , 176 This suggests a similar inflammatory response observed in both VRDs, allowing us to understand deeper the mechanisms that underlie coronavirus infections through IAV infection studies.
Since December 2019, the world was hit by SARS‐CoV‐2. It is a novel, highly contagious and pathogenic coronavirus that has resulted in more than a million deaths worldwide. Transmitted between humans via respiratory droplets and close contact, SARS‐CoV‐2 causes COVID‐19, which primarily presents as respiratory symptoms. 208 , 209 , 210 To this date, SARS‐CoV‐2 has overtaken both SARS‐CoV and MERS in the total number of infected individuals and death toll. 210 , 211 , 212 Viral pneumonia caused by SARS‐CoV‐2 can be grouped into 2 phenotypes: a milder pneumonia phenotype and an ARDS‐like phenotype. COVID‐19 patients can recover from mild pneumonia or progressively develop to the more ARDS‐like severe symptoms. 213 During this progression, conditions such as atelectasis (blockage of the airway) and lung derecruitment (collapse of the lungs) are developed. Progression into ARDS occurs 20−30% of the time. Primary histologic manifestation of severe COVID‐19 includes bilateral DAD, pulmonary edema, and hyaline membranes. 214 DAD is also associated with high mortality in patients. Postmortem histopathologic analysis of COVID‐19 patients reveals endothelialitis, which is the inflammation of the endothelium and alveolar wall injury with the presence of neutrophils and lymphocytes. 215 , 216 This suggests that neutrophils and lymphocytes contribute to the lung tissue damage and pathogenesis of COVID‐19.
Hence, the association between the NLR and COVID‐19 disease severity has been studied and identified as an early indicator for severe COVID‐19 disease. 10 , 11 , 217 , 218 , 219 , 220 , 221 A retrospective cohort study in Wuhan consisting of 210 COVID‐19 patients (87 of them are severe cases) showed a higher NLR of 6‐fold difference in severe cases compared with mild cases, with an increased level of neutrophils and a significant reduction in T cell level. 218 Neutrophil markers including resistin, lipocalin‐2, hepatocyte growth factor, IL‐8, G‐CSF, and dual endothelin‐1 and VEGF signal peptide‐activated Receptor (DEspR) have also been identified as predictors of severe COVID‐19 disease. 97 , 222 In critical cases, high NLR and neutrophilia have been associated with excessive ROS, which may contribute to COVID‐19 disease severity through induction of disease manifestation such as tissue damage, atherosclerosis, thrombosis, and endothelialitis. 223 Additionally, excessive ROS may cause an imbalance of iron homeostasis and contribute to inefficient oxygen transport. 224 Furthermore, the elevation of circulating NETs was observed. 225 Studies in patients revealed a pathogenic role of NETs in disease progression, 93 , 226 , 227 , 228 , 229 , 230 , 231 , 232 , 233 , 234 , 235 , 236 , 237 , 238 , 239 contributing to immunothrombosis. In vitro experiments found SARS‐CoV‐2 capable of activating healthy neutrophils and inducing the release of NETs, which promotes lung epithelial apoptosis. 226 These suggest that both excessive ROS and NET formation from dysregulated levels of neutrophils in the lung contribute to disease pathology. Hence, NETosis is a potential therapeutic target against severe COVID‐19 disease progression. 240 R406, an ATP‐competitive SYK inhibitor and active metabolite of fostamatinib, inhibited NETosis of healthy donor neutrophils in COVID‐19 patient plasma, demonstrating its potential to inhibit NETosis in COVID‐19 patients. 241 Interestingly, these neutrophil‐related pathologies closely resemble IAV infections, reinforcing how neutrophil functions can be closely studied using IAV as a model.
To better study COVID‐19, animal models are actively being developed. 242 , 243 While angiotensin‐converting enzyme 2 (ACE2) is the functional receptor in humans for SARS‐CoV‐2, ACE2 in mice does not bind to the virus effectively. 244 Strategies to overcome this problem are being explored. Currently, no mouse model replicates all aspects of COVID‐19 displayed in humans. 245 However, continued refinement may result in models even for these aspects of the human disease. Recent studies using a transgenic mouse model with human ACE2 demonstrated that the neutrophil mediator S100A8/9 is highly expressed in SARS‐CoV2 infection and inhibition through the drug paquinimod reduced pathogenesis. 246 These models will pave the way for new insights into the mechanisms that neutrophils partake in during SARS‐CoV2 and other VRDs to elucidate possible treatment regimens that can target neutrophil subsets at various stages of the disease.
9. NEUTROPHIL HETEROGENEITY IN VRDS
The current literature on neutrophils in VRDs, covered in this review, provides the foundation for understanding the role of neutrophil subsets in VRDs. There are many layers of heterogeneity to consider, resulting from environmental, developmental, and activation states. 27 , 59 , 247 , 248 The first layer of heterogeneity is the neutrophil's infection status. IAV‐infected neutrophils were shown to produce less ROS, cathelicidin LL‐37, and lipocalin. 249 , 250 , 251 This implicates their functional capacity for viral clearance and may explain higher viral loads that are observed with high infiltrating neutrophil numbers. As discussed, these infected neutrophils can also act as antigen‐presenters for CD8+ T cell immunity. 106
The second layer of heterogeneity is the neutrophil's developmental status. During inflammation, the appearance of immature band cells has been observed, marked as a “left shift” of precursor neutrophils. 33 , 39 Retrospective studies of infants with various VRDs noted high frequencies of immature neutrophils, and this observation was not influenced by bacterial coinfections. 252 Cortjens et al. 253 further identified 3 blood neutrophil subsets during viral infections in infants, namely immature CD16loCD62Lhi neutrophils, mature CD16hiCD62Lhi neutrophils, and a suppressive CD16hiCD62Llo subset. The group previously identified the suppressive CD16hiCD62Llo subset in LPS‐treated individuals. 254 It is unclear if this LPS‐induced CD16hiCD62Llo subset is analogous to the VRD‐induced subset identified. Recent evidence with single‐cell sequencing technologies has shown that neutrophils in SARS‐CoV2‐infected patients are frequently developmentally immature, with lower expression levels of S100a8, S100a9, CD10, and CD101. 255 These neutrophils were also reported to consist of proneutrophils, preneutrophils, and immature neutrophils that associate highly with severe COVID‐19 disease. 256 Immature neutrophils, in particular, showed a strong correlation with disease severity and is suggested to perform better than the NLR. 257 Martinelli et al. 258 used microarray profiling and compared immature bone marrow neutrophils with mature blood neutrophils, showing that immature neutrophils lacked type‐1 IFN signaling receptors and associated genes. They further showed the lack of STAT‐1 phosphorylation during IFNα or IFNγ stimulation in immature neutrophils. 258 These results suggest a differential ability to respond toward VRDs, leading to a lower propensity for NET formation and cytokine release. Single‐cell analysis of SARS‐CoV2‐infected patient leukocytes confirms this, showing proNeus and preNeus in the blood have much lower expression of IFN signaling genes. 256 Moreover, both proNeus and preNeus have low or no expression of CD16 required for ADCP as mentioned earlier. This further differentiates the antiviral potential between mature and immature neutrophil subsets.
The role of immature neutrophils during VRDs is still unclear. It is proposed that their appearance and accumulation is a consequence of the high inflammation present in the patients that stimulates the premature mobilization of immature neutrophils from the bone marrow to the circulation and sites of inflammation. These banded nuclei cells are perhaps less efficient in providing viral clearance and forming NETs and may trigger the recruitment of more immature granulocytes to compensate for the loss of efficiency in viral clearance. Studies on immature neutrophils during inflammatory conditions suggest a high ROS function and migration capacity, but low NETs and phagocytosis function. 259 , 260 This was also shown with in vitro human studies, demonstrating an increased immature neutrophil migration through CXCL8 signaling, but a higher propensity for NET formation correlating with severe COVID‐19 disease. 261
The third layer of heterogeneity is the neutrophil's activation status. Reports of LDNs in VRDs have suggested a degranulated form of neutrophils capable of immunosuppression. 262 , 263 This subset of neutrophils consisted of a mixture of immature and mature phenotypes, unlike what is commonly thought. 262 , 263 , 264 Morrissey et al. 262 identified a specific CD16int LDN subset that was shown to possess enhanced cytokine production upon stimulation. RNA sequencing analysis further suggests increased phagocytosis and degranulation function when compared with CD16hi subset. 262 However, recent evidence of LDNs questions this difference with normal density neutrophils (NDNs). Hardisty et al. 265 were able to generate LDNs from activating NDNs with TNF‐α, fMLP, or LPS. These LDNs were shown to have no significant difference in ROS production or surface marker expression. Moreover, no differential effect on T cell proliferation or IFNγ production was observed. 265 One key difference was the lowered ability for NET formation, possibly linked to the decrease of granules such as NE and MPO required for NETosis. Similarly, LDNs of patients with severe fever with thrombocytopenia syndrome (SFTS) was shown to be derived from NDNs after culture with SFTS media. However, Li's group 266 showed that LDNs could secrete higher amounts of IL‐8, IL‐6 and IL‐17, suggesting a contributor of higher cytotoxicity to endothelial cells. In their study, Li showed that LDNs had higher viral loads, signifying possible differences in antigen‐presentation potential.
10. FUTURE OPPORTUNITIES AND CHALLENGES
Neutrophils can play various roles during VRDs. However, some of these activities that aid in viral clearance can become harmful to the host when dysregulated. Complicated by a spectrum of heterogeneity in neutrophil subtypes and states, identifying pathologic neutrophils remains a significant hurdle in devising biomarkers and novel treatment strategies. Current evidence of neutrophils in VRDs lack a consensus of identifying neutrophil subsets. Lung neutrophils are, at physiologic conditions, different from blood neutrophils phenotypically. 63 Surface markers such as CD62L and CD11b, which are used to identify suppressive and activated neutrophils, were shown to be lower in expression in lung neutrophils. 63 , 253 , 267 Moreover, many surface markers, used in differentiate neutrophil subsets might alter drastically depending on severity and disease progression. It is obvious that emergency granulopoiesis occurs during VRDs, mobilizing immature precursors into the periphery. 268 Therefore, an inflammation‐stable maturation marker, such as CD10 (human) and CD101 (mouse), could be incorporated into future studies to dissect development‐specific changes in neutrophil activity during VRDs. 32 , 39 Future work also should focus on lung neutrophils from BAL samples as the role of the local environment is increasingly appreciated. We anticipate that these future findings will lead to novel interventions and therapies capable of modulating neutrophil activity to improve clinical outcomes.
AUTHORSHIP
Y.Z., I.K., C.R.M, Q.W. and L.G.N. wrote and edited the manuscript. L.G.N reviewed the manuscript. Y.Z. and I.K. designed the graphics. I.K. and L.G.N. supervised the work and conceptualised the review focus.
DISCLOSURE
The authors declare no conflict of interest.
ACKNOWLEDGMENTS
We thank all members of L.G.N. laboratory for their kind support. L.G.N. and I.K. are supported by Singapore Immunology Network (SIgN) core funding, A*STAR, Singapore. C. R. M. is funded under the University and Institute Innovation Team Project of Jinan (Grant No. 2020GXRC036) and the Innovation Pilot Project of Integration of Science, Education and Industry of Shandong Province, China (Grant No. 2020KJC‐ZD011). We apologize to all colleagues whose work was omitted from this article. All figures were created with Biorender.com.
Zhang Y, Wang Q, Mackay CR, Ng LG, Kwok I. Neutrophil subsets and their differential roles in viral respiratory diseases. J Leukoc Biol. 2022;111:1159–1173. 10.1002/JLB.1MR1221-345R
Lai Guan Ng and Immanuel Kwok are the co‐corresponding authors.
REFERENCES
- 1. Segal AW. How neutrophils kill microbes. Annu Rev Immunol. 2005;23:197‐223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Borregaard N. Neutrophils, from marrow to microbes. Immunity. 2010;33:657‐670. [DOI] [PubMed] [Google Scholar]
- 3. Winterbourn CC, Kettle AJ, Hampton MB. Reactive oxygen species and neutrophil function. Annu Rev Biochem. 2016;85:765‐792. [DOI] [PubMed] [Google Scholar]
- 4. Papayannopoulos V. Neutrophil extracellular traps in immunity and disease. Nat Rev Immunol. 2018;18:134‐147. [DOI] [PubMed] [Google Scholar]
- 5. Brinkmann V. Neutrophil extracellular traps kill bacteria. Science. 2004;303:1532‐1535. [DOI] [PubMed] [Google Scholar]
- 6. Branzk N, Lubojemska A, Hardison SE, et al. Neutrophils sense microbe size and selectively release neutrophil extracellular traps in response to large pathogens. Nat Immunol. 2014;15:1017‐1025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Kruger P, Saffarzadeh M, Weber ANR, et al. Neutrophils: between host defence, immune modulation, and tissue injury. PLoS Pathog. 2015;11:e1004651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Peiseler M, Kubes P. More friend than foe: the emerging role of neutrophils in tissue repair. J Clin Invest. 2019;129:2629‐2639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Mortaz E, Alipoor SD, Adcock IM, et al. Update on neutrophil function in severe inflammation. Front Immunol. 2018;9. 10.3389/fimmu.2018.02171. Epub ahead of print. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Liu Y, Du X, Chen J, et al. Neutrophil‐to‐lymphocyte ratio as an independent risk factor for mortality in hospitalized patients with COVID‐19. J Infect. 2020:S0163445320302085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Yang A‐P, Liu J, Tao W, et al. The diagnostic and predictive role of NLR, d‐NLR and PLR in COVID‐19 patients. Int Immunopharmacol. 2020. 10.1016/j.intimp.2020.106504. Epub ahead of print April 13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Egan CE, Sukhumavasi W, Bierly AL, et al. Understanding the multiple functions of Gr‐1(+) cell subpopulations during microbial infection. Immunol Res. 2008;40:35‐48. [DOI] [PubMed] [Google Scholar]
- 13. Tate MD, Deng Y‐M, Jones JE, et al. Neutrophils ameliorate lung injury and the development of severe disease during influenza infection. J Immunol. 2009;183:7441‐7450. [DOI] [PubMed] [Google Scholar]
- 14. Zhou J, Stohlman SA, Hinton DR, et al. Neutrophils promote mononuclear cell infiltration during viral‐induced encephalitis. J Immunol. 2003;170:3331‐3336. [DOI] [PubMed] [Google Scholar]
- 15. Tumpey TM, Chen SH, Oakes JE, et al. Neutrophil‐mediated suppression of virus replication after herpes simplex virus type 1 infection of the murine cornea. J Virol. 1996;70:898‐904. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Thomas PG, Dash P, Aldridge JR, et al. The intracellular sensor NLRP3 mediates key innate and healing responses to influenza a virus via the regulation of Caspase‐1. Immunity. 2009;30:566‐575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Allen IC, Scull MA, Moore CB, et al. The NLRP3 inflammasome mediates in vivo innate immunity to influenza a virus through recognition of viral RNA. Immunity. 2009;30:556‐565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Kirsebom F, Michalaki C, Agueda‐Oyarzabal M, et al. Neutrophils do not impact viral load or the peak of disease severity during RSV infection. Sci Rep. 2020;10:1110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Cortjens B, Lutter R, Boon L, et al. Pneumovirus‐induced lung disease in mice is independent of neutrophil‐driven inflammation. PLoS One. 2016;11:e0168779. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Stokes KL, Currier MG, Sakamoto K, et al. The respiratory syncytial virus fusion protein and neutrophils mediate the airway mucin response to pathogenic respiratory syncytial virus infection. J Virol. 2013;87:10070‐10082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Huang C, Wang Y, Li X, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. The Lancet. 2020;395:497‐506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Wang D, Hu B, Hu C, et al. Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus–Infected pneumonia in Wuhan, China. JAMA. 2020;323:1061‐1069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Terpos E, Ntanasis‐Stathopoulos I, Elalamy I, et al. Hematological findings and complications of COVID‐19. Am J Hematol. 10.1002/ajh.25829. n/a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Howard R, Kanetsky PA, Egan KM. Exploring the prognostic value of the neutrophil‐to‐lymphocyte ratio in cancer. Sci Rep. 2019;9:19673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Faria SS, Jr PCF, Silva MJB, et al. The neutrophil‐to‐lymphocyte ratio: a narrative review. Epub ahead of print December 12, 2016. DOI: 10.3332/ecancer.2016.702. [DOI] [PMC free article] [PubMed]
- 26. Kuri‐Cervantes L, Pampena MB, Meng W, et al. Comprehensive mapping of immune perturbations associated with severe COVID‐19. Sci Immunol. 2020;5. 10.1126/sciimmunol.abd7114. Epub ahead of print July 15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Yvan‐Charvet L, Ng LG. Granulopoiesis and neutrophil homeostasis: a metabolic, daily balancing act. Trends Immunol. 2019;40:598‐612. [DOI] [PubMed] [Google Scholar]
- 28. Gaillard CM, Tokuyasu TA, Passegué E, et al. PML‐RARα deregulates an unexpectedly small number of genes in pre‐leukemic promyelocytes. Blood. 2012;120:3526. [Google Scholar]
- 29. Gaillard C, Tokuyasu TA, Rosen G, et al. Transcription and methylation analyses of preleukemic promyelocytes indicate a dual role for PML/RARA in leukemia initiation. Haematologica. 2015;100:1064‐1075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Hu N, Mora‐Jensen H, Theilgaard‐Mönch K, et al. Differential expression of granulopoiesis related genes in neutrophil subsets distinguished by membrane expression of CD177. PLoS One. 2014;9:e99671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Bjerregaard MD, Jurlander J, Klausen P, et al. The in vivo profile of transcription factors during neutrophil differentiation in human bone marrow. Blood. 2003;101:4322‐4332. [DOI] [PubMed] [Google Scholar]
- 32. Evrard M, Kwok IWH, Chong SZ, et al. Developmental analysis of bone marrow neutrophils reveals populations specialized in expansion, trafficking, and effector functions. Immunity. 2018;48:364‐379.e8. [DOI] [PubMed] [Google Scholar]
- 33. Honda T, Uehara T, Matsumoto G, et al. Neutrophil left shift and white blood cell count as markers of bacterial infection. Clin Chim Acta Int J Clin Chem. 2016;457:46‐53. [DOI] [PubMed] [Google Scholar]
- 34. Lipiński M, Rydzewska G. Immature granulocytes predict severe acute pancreatitis independently of systemic inflammatory response syndrome. Przegla̜d Gastroenterol. 2017;12:140‐144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Roehrl MHA, Wang JY. Immature granulocytes in pregnancy: a story of Virchow, anxious fathers, and expectant mothers. Am J Hematol. 2011;86:307‐308. [DOI] [PubMed] [Google Scholar]
- 36. Reader BF, Nicole PD, Andrew TJ, et al. 72. Social stress enhances immature neutrophil release from bone marrow in murine Aspergillus fumigatus‐induced allergic airway inflammation. Brain Behav Immun. 2014;40:e21. [Google Scholar]
- 37. Katahira Y, Higuchi H, Matsushita H, et al. Increased granulopoiesis in the bone marrow following epstein‐barr virus infection. Sci Rep. 2019;9:13445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. de Monteiro Júnior JG M, de Oliveira Cipriano Torres D, Filho DCS. Hematological parameters as prognostic biomarkers in patients with cardiovascular diseases. Curr Cardiol Rev. 2019;15:274‐282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Marini O, Costa S, Bevilacqua D, et al. Mature CD10+ and immature CD10− neutrophils present in G‐CSF‐–treated donors display opposite effects on T cells. Blood. 2017;129:1343‐1356. [DOI] [PubMed] [Google Scholar]
- 40. Zhou G, Yu L, Fang L, et al. CD177+ neutrophils as functionally activated neutrophils negatively regulate IBD. Gut. 2018;67:1052‐1063. [DOI] [PubMed] [Google Scholar]
- 41. Alder MN, Mallela J, Opoka AM, et al. Olfactomedin 4 marks a subset of neutrophils in mice. Innate Immun. 2019;25:22‐33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Pliyev BK, Shmidt EI, Ivanova AV, et al. Circulating CD35(‐)/CD49d(+) neutrophils in influenza virus infection patients. Hum Immunol. 2012;73:1087‐1090. [DOI] [PubMed] [Google Scholar]
- 43. Xie X, Shi Q, Wu P, et al. Single‐cell transcriptome profiling reveals neutrophil heterogeneity in homeostasis and infection. Nat Immunol. 2020;21:1119‐1133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Zhu YP, Padgett L, Dinh HQ, et al. Identification of an early unipotent neutrophil progenitor with pro‐tumoral activity in mouse and human bone marrow. Cell Rep. 2018;24:2329‐2341.e8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Dinh HQ, Eggert T, Meyer MA, et al. Coexpression of CD71 and CD117 identifies an early unipotent neutrophil progenitor population in human bone marrow. Immunity. 2020;53:319‐334.e6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Kwok I, Becht E, Xia Y, et al. Combinatorial single‐cell analyses of granulocyte‐monocyte progenitor heterogeneity reveals an early uni‐potent neutrophil progenitor. Immunity. 2020;53:303‐318.e5. [DOI] [PubMed] [Google Scholar]
- 47. Kim M‐H, Yang D, Kim M, et al. A late‐lineage murine neutrophil precursor population exhibits dynamic changes during demand‐adapted granulopoiesis. Sci Rep. 2017;7:39804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Muench DE, Olsson A, Ferchen K, et al. Mouse models of neutropenia reveal progenitor‐stage‐specific defects. Nature. 2020;582:109‐114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Adrover JM, Nicolás‐Ávila JA, Hidalgo A. Aging: a temporal dimension for neutrophils. Trends Immunol. 2016;37:334‐345. [DOI] [PubMed] [Google Scholar]
- 50. Kolaczkowska E. The older the faster: aged neutrophils in inflammation. Blood. 2016;128:2280‐2282. [DOI] [PubMed] [Google Scholar]
- 51. Stark MA, Huo Y, Burcin TL, et al. Phagocytosis of apoptotic neutrophils regulates granulopoiesis via IL‐23 and IL‐17. Immunity. 2005;22:285‐294. [DOI] [PubMed] [Google Scholar]
- 52. Nathan C. Points of control in inflammation. Nature. 2002;420:846‐852. [DOI] [PubMed] [Google Scholar]
- 53. Uhl B, Vadlau Y, Zuchtriegel G, et al. Aged neutrophils contribute to the first line of defense in the acute inflammatory response. Blood. 2016;128:2327‐2337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Adrover JM, del Fresno C, Crainiciuc G, et al. A neutrophil timer coordinates immune defense and vascular protection. Immunity. 2019;50:390‐402.e10. [DOI] [PubMed] [Google Scholar]
- 55. Gabrilovich DI. Myeloid‐derived suppressor cells. Cancer Immunol Res. 2017;5:3‐8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Schrijver IT, Théroude C, Roger T. Myeloid‐derived suppressor cells in sepsis. Front Immunol. 2019;10. 10.3389/fimmu.2019.00327. Epub ahead of print February 27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57. Veglia F, Perego M, Gabrilovich D. Myeloid‐derived suppressor cells coming of age. Nat Immunol. 2018;19:108‐119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. Hacbarth E, Kajdacsy‐Balla A. Low density neutrophils in patients with systemic lupus erythematosus, rheumatoid arthritis, and acute rheumatic fever. Arthritis Rheum. 1986;29:1334‐1342. [DOI] [PubMed] [Google Scholar]
- 59. Grieshaber‐Bouyer R, Nigrovic PA. Neutrophil heterogeneity as therapeutic opportunity in immune‐mediated disease. Front Immunol. 2019;10. 10.3389/fimmu.2019.00346. Epub ahead of print. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60. Sagiv JY, Michaeli J, Assi S, et al. Phenotypic diversity and plasticity in circulating neutrophil subpopulations in cancer. Cell Rep. 2015;10:562‐573. [DOI] [PubMed] [Google Scholar]
- 61. Hassani M, Hellebrekers P, Chen N, et al. On the origin of low‐density neutrophils. J Leukoc Biol. 2020;107:809‐818. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62. Fortunati E, Kazemier KM, Grutters JC, et al. Human neutrophils switch to an activated phenotype after homing to the lung irrespective of inflammatory disease. Clin Exp Immunol. 2009;155:559‐566. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63. Ballesteros I, Rubio‐Ponce A, Genua M, et al. Co‐option of neutrophil fates by tissue environments. Cell. 2020. 10.1016/j.cell.2020.10.003. Epub ahead of print October 23. [DOI] [PubMed] [Google Scholar]
- 64. Kim JH, Podstawka J, Lou Y, et al. Aged polymorphonuclear leukocytes cause fibrotic interstitial lung disease in the absence of regulation by B cells. Nat Immunol. 2018;19:192‐201. [DOI] [PubMed] [Google Scholar]
- 65. Seitz R, Heiden M, Offergeld R, et al. Influenza virus. Transfus Med Hemotherapy. 2009;36:32‐39. [Google Scholar]
- 66. Nichols WG, Peck Campbell AJ, Boeckh M. Respiratory viruses other than influenza virus: impact and therapeutic advances. Clin Microbiol Rev. 2008;21:274‐290. table of contents. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67. Tate MD, Brooks AG, Reading PC. The role of neutrophils in the upper and lower respiratory tract during influenza virus infection of mice. Respir Res. 2008;9:57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68. Boncristiani HF, Criado MF, Arruda E. Respiratory viruses. Encyclopedia of Microbiology. Elsevier:500‐518. [Google Scholar]
- 69. Schuster JE, Williams JV. Human Metapneumovirus. Pediatr Rev. 2013;34:558‐565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70. Cheemarla NR, Guerrero‐Plata A. How does the human metapneumovirus regulate neutrophil infiltration into the airways?. Future Virol. 2018;13:233‐235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71. Williams JV, Harris PA, Tollefson SJ, et al. Human metapneumovirus and lower respiratory tract disease in otherwise healthy infants and children. N Engl J Med. 2004;350:443‐450. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72. Branche A, Falsey A. Parainfluenza virus infection. Semin Respir Crit Care Med. 2016;37:538‐554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73. Scott MK, Chommanard C, Lu X, et al. Human adenovirus associated with severe respiratory infection, Oregon, USA, 2013–2014. Emerg Infect Dis. 2016;22:1044‐1051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74. Hayden FG. Rhinovirus and the lower respiratory tract. Rev Med Virol. 2004;14:17‐31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75. Ljubin‐Sternak S, Meštrović T, Ivković‐Jureković I, et al. The emerging role of rhinoviruses in lower respiratory tract infections in children – clinical and molecular epidemiological study from croatia, 2017–2019. Front Microbiol. 2019;10:2737. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76. Aponte FE, Taboada B, Espinoza MA, et al. Rhinovirus is an important pathogen in upper and lower respiratory tract infections in Mexican children. Virol J. 2015;12:31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77. Louie JK, Roy‐Burman A, Guardia‐LaBar L, et al. Rhinovirus associated with severe lower respiratory tract infections in children. Pediatr Infect Dis J. 2009;28:337‐339. [DOI] [PubMed] [Google Scholar]
- 78. Liu P, Xu M, Cao L, et al. Impact of COVID‐19 pandemic on the prevalence of respiratory viruses in children with lower respiratory tract infections in China. Virol J. 2021;18:159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79. Garg S, Jain S, Dawood FS, et al. Pneumonia among adults hospitalized with laboratory‐confirmed seasonal influenza virus infection—United States, 2005–2008. BMC Infect Dis. 2015;15:369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80. Maruyama T, Fujisawa T, Suga S, et al. Outcomes and prognostic features of patients with influenza requiring hospitalization and receiving early antiviral therapy. Chest. 2016;149:526‐534. [DOI] [PubMed] [Google Scholar]
- 81. Casalino E, Antoniol S, Fidouh N, et al. Influenza virus infections among patients attending emergency department according to main reason to presenting to ED: a 3‐year prospective observational study during seasonal epidemic periods. PLoS One. 2017;12:e0182191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82. Lee K‐Y. Pneumonia, acute respiratory distress syndrome, and early immune‐modulator therapy. Int J Mol Sci. 2017:15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83. Williams AE, José RJ, Mercer PF, et al. Evidence for chemokine synergy during neutrophil migration in ARDS. Thorax. 2017;72:66‐73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84. Kirsebom FCM, Kausar F, Nuriev R, et al. Neutrophil recruitment and activation are differentially dependent on MyD88/TRIF and MAVS signaling during RSV infection. Mucosal Immunol. 2019;12:1244‐1255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85. Totura AL, Whitmore A, Agnihothram S, et al. Toll‐Like Receptor 3 signaling via TRIF contributes to a protective innate immune response to severe acute respiratory syndrome coronavirus infection. mBio. 2015;6:e00638‐15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86. Wareing MD, Shea AL, Inglis CA, et al. CXCR2 Is required for neutrophil recruitment to the lung during influenza virus infection, but is not essential for viral clearance. Viral Immunol. 2007;20:369‐378. [DOI] [PubMed] [Google Scholar]
- 87. McCarthy MK, Zhu L, Procario MC, et al. IL‐17 contributes to neutrophil recruitment but not to control of viral replication during acute mouse adenovirus type 1 respiratory infection. Virology. 2014;456–457:259‐267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88. Camp JV, Jonsson CB. A role for neutrophils in viral respiratory disease. Front Immunol. 2017;8:550. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89. McNamara PS. Bronchoalveolar lavage cellularity in infants with severe respiratory syncytial virus bronchiolitis. Arch Dis Child. 2003;88:922‐926. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90. Goritzka M, Makris S, Kausar F, et al. Alveolar macrophage–derived type I interferons orchestrate innate immunity to RSV through recruitment of antiviral monocytes. J Exp Med. 2015;212:699‐714. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91. Cavallaro EC, Liang K‐K, Lawrence MD, et al. Neutrophil infiltration and activation in bronchiolitic airways are independent of viral etiology: neutrophil activity in viral bronchiolitis. Pediatr Pulmonol. 2017;52:238‐246. [DOI] [PubMed] [Google Scholar]
- 92. Tang BM, Shojaei M, Teoh S, et al. Neutrophils‐related host factors associated with severe disease and fatality in patients with influenza infection. Nat Commun. 2019;10:3422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93. Zuo Y, Yalavarthi S, Shi H, et al. Neutrophil extracellular traps in COVID‐19. JCI Insight. 2020. 10.1172/jci.insight.138999. Epub ahead of print April 24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94. Xiong Y, Liu Y, Cao L, et al. Transcriptomic characteristics of bronchoalveolar lavage fluid and peripheral blood mononuclear cells in COVID‐19 patients. Emerg Microbes Infect. 2020;9:761‐770. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95. Lucas C, Wong P, Klein J, et al. Longitudinal analyses reveal immunological misfiring in severe COVID‐19. Nature. 2020;584:463‐469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96. Investigators MOSAIC, Dunning J, Blankley S, et al. Progression of whole‐blood transcriptional signatures from interferon‐induced to neutrophil‐associated patterns in severe influenza. Nat Immunol. 2018;19:625‐635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97. Meizlish ML, Pine AB, Bishai JD, et al. A neutrophil activation signature predicts critical illness and mortality in COVID‐19. Blood Adv. 2021;5:1164‐1177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98. Turner RB. The role of neutrophils in the pathogenesis of rhinovirus infections. Pediatr Infect Dis J. 1990;9:832‐835. [DOI] [PubMed] [Google Scholar]
- 99. Priyadharshini VS, Ramírez‐Jiménez F, Molina‐Macip M, et al. Human neutrophil defensin‐1, ‐3, and ‐4 are elevated in nasal aspirates from children with naturally occurring adenovirus infection. Can Respir J. 2018;2018:1‐6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100. Neumann G, Noda T, Kawaoka Y. Emergence and pandemic potential of swine‐origin H1N1 influenza virus. Nature. 2009;459:931‐939. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101. Cavallazzi R, Ramirez JA. Influenza and Viral Pneumonia. Clin Chest Med. 2018;39:703‐721. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102. WHO . Influenza (Seasonal). World Health Organisation Available from: https://www.who.int/news‐room/fact‐sheets/detail/influenza‐(seasonal). 2018.
- 103. Thangavel RR, Bouvier NM. Animal models for influenza virus pathogenesis, transmission, and immunology. J Immunol Methods. 2014;410:60‐79. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104. Mullarkey CE, Bailey MJ, Golubeva DA, et al. Broadly neutralizing hemagglutinin stalk‐specific antibodies induce potent phagocytosis of immune complexes by neutrophils in an Fc‐dependent manner. mBio;7:e01624‐16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105. Duffy D, Perrin H, Abadie V, et al. Neutrophils transport antigen from the dermis to the bone marrow, initiating a source of memory CD8+ T Cells. Immunity. 2012;37:917‐929. [DOI] [PubMed] [Google Scholar]
- 106. Hufford MM, Richardson G, Zhou H, et al. Influenza‐infected neutrophils within the infected lungs act as antigen presenting cells for anti‐viral CD8+ T cells. PLoS One. 2012;7:e46581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107. Ivan FX, Tan KS, Phoon MC, et al. Neutrophils infected with highly virulent influenza H3N2 virus exhibit augmented early cell death and rapid induction of type I interferon signaling pathways. Genomics. 2013;101:101‐112. [DOI] [PubMed] [Google Scholar]
- 108. Lim K, Hyun Y‐M, Lambert‐Emo K, et al. Neutrophil trails guide influenza‐specific CD8+ T cells in the airways. Science. 2015;349:aaa4352‐aaa4352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109. Moraes TJ, Zurawska JH, Downey GP. Neutrophil granule contents in the pathogenesis of lung injury. Curr Opin Hematol. 2006;13:21‐27. [DOI] [PubMed] [Google Scholar]
- 110. Narasaraju T, Yang E, Samy RP, et al. Excessive neutrophils and neutrophil extracellular traps contribute to acute lung injury of influenza pneumonitis. Am J Pathol. 2011;179:199‐210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111. Dabo AJ, Cummins N, Eden E, et al. Matrix metalloproteinase 9 exerts antiviral activity against respiratory syncytial virus. PloS One. 2015;10:e0135970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112. Bradley LM, Douglass MF, Chatterjee D, et al. Matrix metalloprotease 9 mediates neutrophil migration into the airways in response to influenza virus‐induced toll‐like receptor signaling. PLoS Pathog. 2012;8:e1002641. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113. Fridman R, Toth M, Chvyrkova I, et al. Cell surface association of matrix metalloproteinase‐9 (gelatinase B). Cancer Metastasis Rev. 2003;22:153‐166. [DOI] [PubMed] [Google Scholar]
- 114. Ellerbroek SM, Halbleib JM, Benavidez M, et al. Phosphatidylinositol 3‐kinase activity in epidermal growth factor‐stimulated matrix metalloproteinase‐9 production and cell surface association. Cancer Res. 2001;61:1855‐1861. [PubMed] [Google Scholar]
- 115. Hartshorn KL, White MR, Tecle T, et al. Innate defense against influenza A virus: activity of human neutrophil defensins and interactions of defensins with surfactant protein D. J Immunol Baltim Md 1950. 2006;176:6962‐6972. [DOI] [PubMed] [Google Scholar]
- 116. Doss M, White MR, Tecle T, et al. Interactions of alpha‐, beta‐, and theta‐defensins with influenza A virus and surfactant protein D. J Immunol Baltim Md 1950. 2009;182:7878‐7887. [DOI] [PubMed] [Google Scholar]
- 117. Akaike T, Noguchi Y, Ijiri S, et al. Pathogenesis of influenza virus‐induced pneumonia: involvement of both nitric oxide and oxygen radicals. Proc Natl Acad Sci USA. 1996;93:2448‐2453. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118. Bataki EL, Evans GS, Everard ML. Respiratory syncytial virus and neutrophil activation. Clin Exp Immunol. 2005;140:470‐477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119. Johansson C, Kirsebom FCM. Neutrophils in respiratory viral infections. Mucosal Immunol. 2021;14:815‐827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120. Vlahos R, Stambas J, Bozinovski S, et al. Inhibition of Nox2 oxidase activity ameliorates influenza a virus‐induced lung inflammation. PLoS Pathog. 2011;7:e1001271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121. Borregaard N. Development of neutrophil granule diversity. Ann N Y Acad Sci. 1997;832:62‐68. [DOI] [PubMed] [Google Scholar]
- 122. Sugamata R, Dobashi H, Nagao T, et al. Contribution of neutrophil‐derived myeloperoxidase in the early phase of fulminant acute respiratory distress syndrome induced by influenza virus infection: r. Sugamata et al. Microbiol Immunol. 2012;56:171‐182. [DOI] [PubMed] [Google Scholar]
- 123. Emboriadou M, Hatzistilianou M, Magnisali C, et al. Human neutrophil elastase in RSV bronchiolitis. Ann Clin Lab Sci. 2007;37:79‐84. [PubMed] [Google Scholar]
- 124. Abu‐Harb M, Bell F, Finn A, et al. IL‐8 and neutrophil elastase levels in the respiratory tract of infants with RSV bronchiolitis. Eur Respir J. 1999;14:139. [DOI] [PubMed] [Google Scholar]
- 125. Anjani G, Vignesh P, Joshi V, et al. Recent advances in chronic granulomatous disease. Genes Dis. 2020;7:84‐92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126. N T. Neutrophils as possible therapeutic targets in severe influenza pneumonia. J Infect Pulm Dis. 2016;2. 10.16966/2470-3176.115. Epub ahead of print. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127. Zhu L, Liu L, Zhang Y, et al. High level of neutrophil extracellular traps correlates with poor prognosis of severe influenza a infection. J Infect Dis. 2018;217:428‐437. [DOI] [PubMed] [Google Scholar]
- 128. Ashar HK, Mueller NC, Rudd JM, et al. The role of extracellular histones in influenza virus pathogenesis. Am J Pathol. 2018;188:135‐148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129. Funchal GA, Jaeger N, Czepielewski RS, et al. Respiratory syncytial virus fusion protein promotes TLR‐4–dependent neutrophil extracellular trap formation by human neutrophils. PLoS One. 2015;10:e0124082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130. Geerdink RJ, Hennus MP, Westerlaken GHA, et al. LAIR‐1 limits neutrophil extracellular trap formation in viral bronchiolitis. J Allergy Clin Immunol. 2018;141:811‐814. [DOI] [PubMed] [Google Scholar]
- 131. Muraro SP, De Souza GF, Gallo SW, et al. Respiratory syncytial virus induces the classical ROS‐dependent NETosis through PAD‐4 and necroptosis pathways activation. Sci Rep. 2018;8:14166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132. Hägglund S, Blodörn K, Näslund K, et al. Proteome analysis of bronchoalveolar lavage from calves infected with bovine respiratory syncytial virus—Insights in pathogenesis and perspectives for new treatments. PLoS One. 2017;12:e0186594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133. Cortjens B, de Boer OJ, de Jong R, et al. Neutrophil extracellular traps cause airway obstruction during respiratory syncytial virus disease: nETs in RSV disease. J Pathol. 2016;238:401‐411. [DOI] [PubMed] [Google Scholar]
- 134. Hoeksema M, Tripathi S, White M, et al. Arginine‐rich histones have strong antiviral activity for influenza A viruses. Innate Immun. 2015;21:736‐745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135. Radermecker C, Detrembleur N, Guiot J, et al. Neutrophil extracellular traps infiltrate the lung airway, interstitial, and vascular compartments in severe COVID‐19. J Exp Med. 2020;217. 10.1084/jem.20201012. Epub ahead of print September 14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136. Bendib I, de Chaisemartin L, Granger V, et al. Neutrophil extracellular traps are elevated in patients with pneumonia‐related acute respiratory distress syndrome. Anesthesiology. 2019;130:581‐591. [DOI] [PubMed] [Google Scholar]
- 137. Heltzer ML, Coffin SE, Maurer K, et al. Immune dysregulation in severe influenza. J Leukoc Biol. 2009;85:1036‐1043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138. Tahaghoghi‐Hajghorbani S, Zafari P, Masoumi E, et al. The role of dysregulated immune responses in COVID‐19 pathogenesis. Virus Res. 2020;290:198197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139. Lim K, Kim T, Trzeciak A, et al. In situ neutrophil efferocytosis shapes T cell immunity to influenza infection. Nat Immunol. 2020;21:1046‐1057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140. Martin TR. Innate immunity in the lungs. Proc Am Thorac Soc. 2005;2:403‐411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141. Janeway CA, Medzhitov R. Innate immune recognition. Annu Rev Immunol. 2002;20:197‐216. [DOI] [PubMed] [Google Scholar]
- 142. van de Sandt CE, Kreijtz JHCM, Rimmelzwaan GF. Evasion of influenza a viruses from innate and adaptive immune responses. Viruses. 2012;4:1438‐1476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143. Williams MR, Azcutia V, Newton G, et al. Emerging mechanisms of neutrophil recruitment across endothelium. Trends Immunol. 2011;32:461‐469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144. Pittman K, Kubes P. Damage‐associated molecular patterns control neutrophil recruitment. J Innate Immun. 2013;5:315‐323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145. Chen X, Liu S, Goraya MU, et al. Host immune response to influenza a virus infection. Front Immunol. 2018;9:320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146. Waugh T, Ching JCH, Zhou Y, et al. Influenza A virus (H1N1) increases airway epithelial cell secretion by up‐regulation of potassium channel KCNN4. Biochem Biophys Res Commun. 2013;438:581‐587. [DOI] [PubMed] [Google Scholar]
- 147. Tavares LP, Teixeira MM, Garcia CC. The inflammatory response triggered by Influenza virus: a two edged sword. Inflamm Res. 2017;66:283‐302. [DOI] [PubMed] [Google Scholar]
- 148. Schmitz N, Kurrer M, Bachmann MF, et al. Interleukin‐1 is responsible for acute lung immunopathology but increases survival of respiratory influenza virus infection. J Virol. 2005;79:6441‐6448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149. Short KR, Kroeze EJBV, Fouchier RAM, et al. Pathogenesis of influenza‐induced acute respiratory distress syndrome. Lancet Infect Dis. 2014;14:57‐69. [DOI] [PubMed] [Google Scholar]
- 150. Ma W‐T, Yao X‐T, Peng Q, et al. The protective and pathogenic roles of IL‐17 in viral infections: friend or foe?. Open Biol. 2019;9:190109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151. Grommes J, Soehnlein O. Contribution of neutrophils to acute lung injury. Mol Med. 2011;17:293‐307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152. Högner K, Wolff T, Pleschka S, et al. Macrophage‐expressed IFN‐β contributes to apoptotic alveolar epithelial cell injury in severe influenza virus pneumonia. PLoS Pathog. 2013;9:e1003188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153. Kobasa D, Jones SM, Shinya K, et al. Aberrant innate immune response in lethal infection of macaques with the 1918 influenza virus. Nature. 2007;445:319‐323. [DOI] [PubMed] [Google Scholar]
- 154. Lee SMY, Gardy JL, Cheung CY, et al. Systems‐level comparison of host‐responses elicited by avian H5N1 and seasonal H1N1 influenza viruses in primary human macrophages. PLoS One. 2009;4:e8072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155. Perrone LA, Plowden JK, García‐Sastre A, et al. H5N1 and 1918 pandemic influenza virus infection results in early and excessive infiltration of macrophages and neutrophils in the lungs of mice. PLoS Pathog. 2008;4:e1000115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156. Cline TD, Beck D, Bianchini E. Influenza virus replication in macrophages: balancing protection and pathogenesis. J Gen Virol. 2017;98:2401‐2412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157. Griffith JW, Sokol CL, Luster AD. Chemokines and chemokine receptors: positioning cells for host defense and immunity. Annu Rev Immunol. 2014;32:659‐702. [DOI] [PubMed] [Google Scholar]
- 158. Stifter SA, Bhattacharyya N, Pillay R, et al. Functional interplay between Type I and II interferons is essential to limit influenza a virus‐induced tissue inflammation. PLoS Pathog. 2016;12:e1005378. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159. Tumpey TM, García‐Sastre A, Taubenberger JK, et al. Pathogenicity of influenza viruses with genes from the 1918 pandemic virus: functional roles of alveolar macrophages and neutrophils in limiting virus replication and mortality in mice. J Virol. 2005;79:14933‐14944. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160. Atkin‐Smith GK, Duan M, Chen W, et al. The induction and consequences of Influenza A virus‐induced cell death. Cell Death Dis. 2018;9:1002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161. Brandes M, Klauschen F, Kuchen S, et al. A systems analysis identifies a feedforward inflammatory circuit leading to lethal influenza infection. Cell. 2013;154:197‐212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162. Rudd BD, Schaller MA, Smit JJ, et al. MyD88‐mediated instructive signals in dendritic cells regulate pulmonary immune responses during respiratory virus infection. J Immunol. 2007;178:5820‐5827. [DOI] [PubMed] [Google Scholar]
- 163. Seo S‐U, Kwon H‐J, Song J‐H, et al. MyD88 signaling is indispensable for primary influenza a virus infection but dispensable for secondary infection. J Virol. 2010;84:12713‐12722. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164. Sheahan T, Morrison TE, Funkhouser W, et al. MyD88 is required for protection from lethal infection with a mouse‐adapted SARS‐CoV. PLoS Pathog. 2008;4:e1000240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165. Goritzka M, Pereira C, Makris S, et al. T cell responses are elicited against respiratory syncytial virus in the absence of signalling through TLRs, RLRs and IL‐1R/IL‐18R. Sci Rep. 2016;5:18533. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166. van den Brand JMA, Stittelaar KJ, van Amerongen G, et al. Comparison of temporal and spatial dynamics of seasonal H3N2, PANDEMIC H1N1 and highly pathogenic avian influenza H5N1 virus infections in ferrets. PLoS One. 2012;7:e42343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167. Shinya K, Gao Y, Cilloniz C, et al. Integrated clinical, pathologic, virologic, and transcriptomic analysis of H5N1 influenza virus‐induced viral pneumonia in the rhesus macaque. J Virol. 2012;86:6055‐6066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168. Dienz O, Rud JG, Eaton SM, et al. Essential role of IL‐6 in protection against H1N1 influenza virus by promoting neutrophil survival in the lung. Mucosal Immunol. 2012;5:258‐266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169. Prince LR, Whyte MK, Sabroe I, et al. The role of TLRs in neutrophil activation. Curr Opin Pharmacol. 2011;11:397‐403. [DOI] [PubMed] [Google Scholar]
- 170. Thomas CJ, Schroder K. Pattern recognition receptor function in neutrophils. Trends Immunol. 2013;34:317‐328. [DOI] [PubMed] [Google Scholar]
- 171. Bjornson AB, Mellencamp MA, Schiff GM, Complement is activated in the upper respiratory tract during influenza virus infection. Am Rev Respir Dis. 143:1062–1066. [DOI] [PubMed]
- 172. Borregaard N, Sørensen OE, Theilgaard‐Mönch K. Neutrophil granules: a library of innate immunity proteins. Trends Immunol. 2007;28:340‐345. [DOI] [PubMed] [Google Scholar]
- 173. Tate MD, Ioannidis LJ, Croker B, et al. The role of neutrophils during mild and severe influenza virus infections of mice. PLoS One. 2011;6:e17618. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174. Kulkarni U, Zemans RL, Smith CA, et al. Excessive neutrophil levels in the lung underlie the age‐associated increase in influenza mortality. Mucosal Immunol. 2019;12:545‐554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175. Groeneveld JAB, Raijmakers PGHM, Hack EC, et al. Interleukin 8‐related neutrophil elastase and the severity of the adult respiratory distress syndrome. Cytokine. 1995;7:746‐752. [DOI] [PubMed] [Google Scholar]
- 176. Miller EJ, Cohen AB, Nagao S, et al. Elevated levels of NAP‐1/Interleukin‐8 are present in the airspaces of patients with the adult respiratory distress syndrome and are associated with increased mortality. Am Rev Respir Dis. 1992;146:427‐432. [DOI] [PubMed] [Google Scholar]
- 177. vom Steeg LG, Klein SL. Sex and sex steroids impact influenza pathogenesis across the life course. Semin Immunopathol. 2019;41:189‐194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178. vom Steeg LG, Vermillion MS, Hall OJ, et al. Age and testosterone mediate influenza pathogenesis in male mice. Am J Physiol‐Lung Cell Mol Physiol. 2016;311:L1234‐L1244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179. Robinson DP, Lorenzo ME, Jian W, et al. Elevated 17β‐estradiol protects females from influenza a virus pathogenesis by suppressing inflammatory responses. PLoS Pathog. 2011;7. 10.1371/journal.ppat.1002149. Epub ahead of print July 28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180. Robinson DP, Hall OJ, Nilles TL, et al. 17β‐estradiol protects females against influenza by recruiting neutrophils and increasing virus‐specific CD8 T cell responses in the lungs. J Virol. 2014;88:4711‐4720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181. Kang S‐J, Jung SI. Age‐related morbidity and mortality among patients with COVID‐19. Infect Chemother. 2020;52:154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182. Flerlage T, Boyd DF, Meliopoulos V, et al. Influenza virus and SARS‐CoV‐2: pathogenesis and host responses in the respiratory tract. Nat Rev Microbiol. 2021;19:425‐441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183. Eash KJ, Greenbaum AM, Gopalan PK, et al. CXCR2 and CXCR4 antagonistically regulate neutrophil trafficking from murine bone marrow. J Clin Invest. 2010;120:2423‐2431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184. Belperio JA, Keane MP, Burdick MD, et al. Critical role for CXCR2 and CXCR2 ligands during the pathogenesis of ventilator‐induced lung injury. J Clin Invest. 2002;110:1703‐1716. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 185. Sawant KV, Xu R, Cox R, et al. Chemokine CXCL1‐mediated neutrophil trafficking in the lung: role of CXCR2 activation. J Innate Immun. 2015;7:647‐658. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186. Tavares LP, Garcia CC, Machado MG, et al. CXCR1/2 antagonism is protective during influenza and post‐influenza pneumococcal infection. Front Immunol. 2017;8:1799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187. Oh S‐J, Lee JK, Shin OS. Aging and the immune system: the impact of immunosenescence on viral infection, immunity and vaccine immunogenicity. Immune Netw. 2019;19. 10.4110/in.2019.19.e37. Epub ahead of print November 14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188. Lu J, Duan X, Zhao W, et al. Aged mice are more resistant to influenza virus infection due to reduced inflammation and lung pathology. Aging Dis. 2018;9:358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 189. Sauce D, Dong Y, Campillo‐Gimenez L, et al. Reduced oxidative burst by primed neutrophils in the elderly individuals is associated with increased levels of the CD16 bright /CD62L dim Immunosuppressive subset. J Gerontol A Biol Sci Med Sci. 2017;72:163‐172. [DOI] [PubMed] [Google Scholar]
- 190. Wenisch C, Patruta S, Daxböck F, et al. Effect of age on human neutrophil function. J Leukoc Biol. 2000;67:40‐45. [DOI] [PubMed] [Google Scholar]
- 191. Guo Y, Liu X, Deng M, et al. Epidemiology of COVID‐19 in older persons, Wuhan, China. Age Ageing. 2020;49:706‐712. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192. Wu JT, Leung K, Bushman M, et al. Estimating clinical severity of COVID‐19 from the transmission dynamics in Wuhan, China. Nat Med. 2020;26:506‐510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193. O'Driscoll M, Ribeiro Dos Santos G, Wang L, et al. Age‐specific mortality and immunity patterns of SARS‐CoV‐2. Nature. 2021;590:140‐145. [DOI] [PubMed] [Google Scholar]
- 194. Pastor‐Barriuso R, Pérez‐Gómez B, Hernán MA, et al. Infection fatality risk for SARS‐CoV‐2 in community dwelling population of Spain: nationwide seroepidemiological study. BMJ. 2020;371:m4509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195. Rosa BA, Ahmed M, Singh DK, et al. IFN signaling and neutrophil degranulation transcriptional signatures are induced during SARS‐CoV‐2 infection. Commun Biol. 2021;4:1‐14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 196. Loske J, Röhmel J, Lukassen S, et al. Pre‐activated antiviral innate immunity in the upper airways controls early SARS‐CoV‐2 infection in children. Nat Biotechnol. 2021. 10.1038/s41587-021-01037-9. Epub ahead of print August 18. [DOI] [PubMed] [Google Scholar]
- 197. Cui J, Li F, Shi Z‐L. Origin and evolution of pathogenic coronaviruses. Nat Rev Microbiol. 2019;17:181‐192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 198. Gui M, Liu X, Guo D, et al. Electron microscopy studies of the coronavirus ribonucleoprotein complex. Protein Cell. 2017;8:219‐224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199. Lu L, Zhong W, Bian Z, et al. A comparison of mortality‐related risk factors of COVID‐19, SARS, and MERS: a systematic review and meta‐analysis. J Infect. 2020;81:e18‐e25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 200. Gretebeck LM, Subbarao K. Animal models for SARS and MERS coronaviruses. Curr Opin Virol. 2015;13:123‐129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201. Roberts A, Lamirande EW, Vogel L, et al. Animal models and vaccines for SARS‐CoV infection. Virus Res. 2008;133:20‐32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 202. Subbarao K, Roberts A. Is there an ideal animal model for SARS?. Trends Microbiol. 2006;14:299‐303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203. Yuan L, Tang Q, Cheng T, et al. Animal models for emerging coronavirus: progress and new insights. Emerg Microbes Infect. 2020;9:949‐961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 204. Chen J, Lau YF, Lamirande EW, et al. Cellular immune responses to severe acute respiratory syndrome coronavirus (SARS‐CoV) infection in senescent BALB/c mice: cD4+ T cells are important in control of SARS‐CoV infection. J Virol. 2010;84:1289‐1301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 205. Franks TJ, Chong PY, Chui P, et al. Lung pathology of severe acute respiratory syndrome (SARS): a study of 8 autopsy cases from Singapore. Hum Pathol. 2003;34:743‐748. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 206. Graham RL, Donaldson EF, Baric RS. A decade after SARS: strategies for controlling emerging coronaviruses. Nat Rev Microbiol. 2013;11:836‐848. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 207. Alosaimi B, Hamed ME, Naeem A, et al. MERS‐CoV infection is associated with downregulation of genes encoding Th1 and Th2 cytokines/chemokines and elevated inflammatory innate immune response in the lower respiratory tract. Cytokine. 2020;126:154895. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 208. Liu J, Liao X, Qian S, et al. Community transmission of severe acute respiratory syndrome coronavirus 2, Shenzhen, China, 2020. Emerg Infect Dis. 2020;26. 10.3201/eid2606.200239. Epub ahead of print June. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 209. Li Q, Guan X, Wu P, et al. Early transmission dynamics in Wuhan, China, of novel coronavirus–infected pneumonia. N Engl J Med. 2020;382:1199‐1207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 210. Singhal T. A review of coronavirus disease‐2019 (COVID‐19). Indian J Pediatr. 2020;87:281‐286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 211. Deng S‐Q, Peng H‐J. Characteristics of and public health responses to the coronavirus disease 2019 Outbreak in China. J Clin Med. 2020;9:575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 212. Han Q, Lin Q, Jin S, et al. Coronavirus 2019‐nCoV: a brief perspective from the front line. J Infect. 2020;80:373‐377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 213. Gattinoni L, Chiumello D, Caironi P, et al. COVID‐19 pneumonia: different respiratory treatments for different phenotypes?. Intensive Care Med. 2020;46:1099‐1102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 214. Chau VQ, Oliveros E, Mahmood K, et al. The imperfect cytokine storm. JACC Case Rep. 2020;2:1315‐1320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 215. Aguiar D, Lobrinus JA, Schibler M, et al. Inside the lungs of COVID‐19 disease. Int J Legal Med. 2020;134:1271‐1274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 216. Borczuk AC, Salvatore SP, Seshan SV, et al. COVID‐19 pulmonary pathology: a multi‐institutional autopsy cohort from Italy and New York City. Mod Pathol. 2020;33:2156‐2168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 217. Chan AS, Rout A. Use of neutrophil‐to‐lymphocyte and Platelet‐to‐Lymphocyte ratios in COVID‐19. J Clin Med Res. 2020;12:448‐453. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 218. Kong M, Zhang H, Cao X, et al. Higher level of neutrophil‐to‐lymphocyte is associated with severe COVID‐19. Epidemiol Infect. 2020;148:e139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 219. Pimentel GD, Dela Vega MCM, Laviano A. High neutrophil to lymphocyte ratio as a prognostic marker in COVID‐19 patients. Clin Nutr ESPEN. 2020:S240545772030173X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 220. Jimeno S, Ventura PS, Castellano JM, et al. Prognostic implications of neutrophil‐lymphocyte ratio in COVID‐19. Eur J Clin Invest. 2021;51 . 10.1111/eci.13404. Epub ahead of print January. [DOI] [PubMed] [Google Scholar]
- 221. King AH, Mehkri O, Rajendram P, et al. A high neutrophil‐lymphocyte ratio is associated with increased morbidity and mortality in patients with coronavirus disease 2019. Crit Care Explor. 2021;3:e0444. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 222. deKay JT, Emery IF, Rud J, et al. DEspRhigh neutrophils are associated with critical illness in COVID‐19. Sci Rep. 2021;11:22463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 223. Darbousset R, Thomas GM, Mezouar S, et al. Tissue factor–positive neutrophils bind to injured endothelial wall and initiate thrombus formation. Blood. 2012;120:2133‐2143. [DOI] [PubMed] [Google Scholar]
- 224. Laforge M, Elbim C, Frère C, et al. Tissue damage from neutrophil‐induced oxidative stress in COVID‐19. Nat Rev Immunol. 2020;20:515‐516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 225. Golonka RM, Saha P, Yeoh BS, et al. Harnessing innate immunity to eliminate SARS‐CoV‐2 and ameliorate COVID‐19 disease. Physiol Genomics. 2020;52:217‐221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 226. Veras FP, Pontelli MC, Silva CM, et al. SARS‐CoV‐2–triggered neutrophil extracellular traps mediate COVID‐19 pathology. J Exp Med. 2020;217:e20201129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 227. Wang J, Li Q, Yin Y, et al. Excessive neutrophils and neutrophil extracellular traps in COVID‐19. Front Immunol. 2020;11:2063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 228. Narasaraju T, Tang BM, Herrmann M, et al. Neutrophilia and NETopathy as key pathologic drivers of progressive lung impairment in patients with COVID‐19. Front Pharmacol. 2020;11:870. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 229. Blasco A, Coronado M‐J, Hernández‐Terciado F, et al. Assessment of neutrophil extracellular traps in coronary thrombus of a case series of patients with COVID‐19 and myocardial infarction. JAMA Cardiol. 2021;6:469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 230. Zhang Y, Han K, Du C, et al. Carboxypeptidase B blocks ex vivo activation of the anaphylatoxin‐neutrophil extracellular trap axis in neutrophils from COVID‐19 patients. Crit Care. 2021;25:51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 231. Ng H, Havervall S, Rosell A, et al. Circulating markers of neutrophil extracellular traps are of prognostic value in patients with COVID‐19. Arterioscler Thromb Vasc Biol. 2021;41:988‐994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 232. Thierry AR, Roch B. Neutrophil extracellular traps and by‐products play a key role in COVID‐19: pathogenesis, risk factors, and therapy. J Clin Med. 2020;9:2942. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 233. Ondracek AS, Lang IM. Neutrophil extracellular traps as prognostic markers in COVID‐19: a welcome piece to the puzzle. Arterioscler Thromb Vasc Biol. 2021;41:995‐998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 234. Middleton EA, He X‐Y, Denorme F, et al. Neutrophil extracellular traps contribute to immunothrombosis in COVID‐19 acute respiratory distress syndrome. Blood. 2020;136:1169‐1179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 235. Obermayer A, Jakob L‐M, Haslbauer JD, et al. Neutrophil extracellular traps in fatal COVID‐19‐associated lung injury. Dis Markers. 2021;2021:1‐10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 236. Radermecker C, Detrembleur N, Guiot J, et al. Neutrophil extracellular traps infiltrate the lung airway, interstitial, and vascular compartments in severe COVID‐19. J Exp Med. 2020;217:e20201012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 237. Ackermann M, Anders H‐J, Bilyy R, et al. Patients with COVID‐19: in the dark‐NETs of neutrophils. Cell Death Differ. 2021;28:3125‐3139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 238. Leppkes M, Knopf J, Naschberger E, et al. Vascular occlusion by neutrophil extracellular traps in COVID‐19. EBioMedicine. 2020;58:102925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 239. Arcanjo A, Logullo J, Menezes CCB, et al. The emerging role of neutrophil extracellular traps in severe acute respiratory syndrome coronavirus 2 (COVID‐19). Sci Rep. 2020;10:19630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 240. Barnes BJ, Adrover JM, Baxter‐Stoltzfus A, et al. Targeting potential drivers of COVID‐19: neutrophil extracellular traps. J Exp Med. 2020;217:e20200652. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 241. Strich JR, Ramos‐Benitez MJ, Randazzo D, et al. Fostamatinib inhibits neutrophils extracellular traps induced by COVID‐19 patient plasma: a potential therapeutic. J Infect Dis. 2021;223:981‐984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 242. Dinnon KH, Leist SR, Schäfer A, et al. A mouse‐adapted model of SARS‐CoV‐2 to test COVID‐19 countermeasures. Nature. 2020;586:560‐566. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 243. Johansen MD, Irving A, Montagutelli X, et al. Animal and translational models of SARS‐CoV‐2 infection and COVID‐19. Mucosal Immunol. 2020;13:877‐891. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 244. Wan Y, Shang J, Graham R, et al. Receptor recognition by the novel coronavirus from wuhan: an analysis based on decade‐long structural studies of SARS coronavirus. J Virol. 2020;94:e00127‐20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 245. Muñoz‐Fontela C, Dowling WE, Funnell SGP, et al. Animal models for COVID‐19. Nature. 2020;586:509‐515. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 246. Guo Q, Zhao Y, Li J, et al. Induction of alarmin S100A8/A9 mediates activation of aberrant neutrophils in the pathogenesis of COVID‐19. Cell Host Microbe. 2021;29:222‐235.e4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 247. Deniset JF, Kubes P. Neutrophil heterogeneity: bona fide subsets or polarization states?. J Leukoc Biol. 2018;103:829‐838. [DOI] [PubMed] [Google Scholar]
- 248. Ng LG, Ostuni R, Hidalgo A. Heterogeneity of neutrophils. Nat Rev Immunol. 2019;19:255‐265. [DOI] [PubMed] [Google Scholar]
- 249. Navarini AA, Recher M, Lang KS, et al. Increased susceptibility to bacterial superinfection as a consequence of innate antiviral responses. Proc Natl Acad Sci USA. 2006;103:15535‐15539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 250. Tripathi S, Verma A, Kim E‐J, et al. LL‐37 modulates human neutrophil responses to influenza A virus. J Leukoc Biol. 2014;96:931‐938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 251. Robinson KM, McHugh KJ, Mandalapu S, et al. Influenza A virus exacerbates Staphylococcus aureus pneumonia in mice by attenuating antimicrobial peptide production. J Infect Dis. 2014;209:865‐875. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 252. Noyola E, Noor A, Sweeney N, et al. Prevalence of bandemia in respiratory viral infections: a pediatric emergency room experience. Front Pediatr. 2020;8:845. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 253. Cortjens B, Ingelse SA, Calis JC, et al. Neutrophil subset responses in infants with severe viral respiratory infection. Clin Immunol. 2017;176:100‐106. [DOI] [PubMed] [Google Scholar]
- 254. Pillay J, Tak T, Kamp VM, et al. Immune suppression by neutrophils and granulocytic myeloid‐derived suppressor cells: similarities and differences. Cell Mol Life Sci. 2013;70:3813‐3827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 255. Silvin A, Chapuis N, Dunsmore G, et al. Elevated calprotectin and abnormal myeloid cell subsets discriminate severe from mild COVID‐19. Cell. 2020;0. 10.1016/j.cell.2020.08.002. Epub ahead of print August 5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 256. Schulte‐Schrepping J, Reusch N, Paclik D, et al. Severe COVID‐19 is marked by a dysregulated myeloid cell compartment. Cell. 2020;0. 10.1016/j.cell.2020.08.001. Epub ahead of print August 5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 257. Carissimo G, Xu W, Kwok I, et al. Whole blood immunophenotyping uncovers immature neutrophil‐to‐VD2 T‐cell ratio as an early marker for severe COVID‐19. Nat Commun. 2020;11:5243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 258. Martinelli S, Urosevic M, Daryadel A, et al. Induction of genes mediating interferon‐dependent extracellular trap formation during neutrophil differentiation. J Biol Chem. 2004;279:44123‐44132. [DOI] [PubMed] [Google Scholar]
- 259. Wang L, Ai Z, Khoyratty T, et al. ROS‐producing immature neutrophils in giant cell arteritis are linked to vascular pathologies. JCI Insight. 2020;5. 10.1172/jci.insight.139163. Epub ahead of print October 15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 260. Drifte G, Dunn‐Siegrist I, Tissières P, et al. Innate Immune functions of immature neutrophils in patients with sepsis and severe systemic inflammatory response syndrome *. Crit Care Med. 2013;41:820‐832. [DOI] [PubMed] [Google Scholar]
- 261. Belchamber K, Thein O & Hazeldine J et al. Altered neutrophil phenotype and function in non‐ICU hospitalised COVID‐19 patients correlated with disease severity. Preprint, Intensive Care and Critical Care Medicine. Epub ahead of print June 8, 2021. DOI: 10.1101/2021.06.08.21258535.
- 262. Morrissey SM, Geller AE, Hu X, et al. A specific low‐density neutrophil population correlates with hypercoagulation and disease severity in hospitalized COVID‐19 patients. JCI Insight. 2021;6. 10.1172/jci.insight.148435. Epub ahead of print May 10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 263. Cabrera LE, Pekkarinen PT, Alander M, et al. Characterization of low‐density granulocytes in COVID‐19. PLoS Pathog. 2021;17:e1009721. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 264. Tay SH, Celhar T, Fairhurst A‐M. Low‐density neutrophils in systemic lupus erythematosus. Arthritis Rheumatol. 2020;72:1587‐1595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 265. Hardisty GR, Llanwarne F, Minns D, et al. High purity isolation of low density neutrophils casts doubt on their exceptionality in health and disease. Front Immunol. 2021;12:2057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 266. Li Y, Li H, Wang H, et al. The proportion, origin and pro‐inflammation roles of low density neutrophils in SFTS disease. BMC Infect Dis. 2019;19:109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 267. Tak T, Rygiel TP, Karnam G, et al. Neutrophil‐mediated suppression of influenza‐induced pathology requires CD11b/CD18 (MAC‐1). Am J Respir Cell Mol Biol. 2018;58:492‐499. [DOI] [PubMed] [Google Scholar]
- 268. Manz MG, Boettcher S. Emergency granulopoiesis. Nat Rev Immunol. 2014;14:302‐314. [DOI] [PubMed] [Google Scholar]


