Introduction
Major depressive disorder (MDD) belongs to severe mood disorders. Global prevalence of MDD is about 4.7% with regional differences (Ferrari et al. 2013; Steel et al. 2014). The lifetime prevalence varies from 3.3% to 16% across countries (Gu et al. 2013; Otte et al. 2016). It has been estimated that MDD will be a leading cause of disability by 2030 (Mathers and Loncar 2006). MDD is associated with great individual and family suffering, disability, poor daily functioning, insomnia, and negative prognosis for comorbid medical conditions (Fröjd et al. 2008; Buysse et al. 2008; Lynch and Clarke 2006; Kupfer et al. 2012). MDD is also associated with an elevated risk of suicide, psychotic presentations (Penninx 2017; Kendler et al. 2009), and other psychiatric disorders (Cassano et al. 2004). Treating MDD usually involves pharmacological agents and/or psychotherapy. Treatment coverage of MDD has clear regional differences: the coverage of treatment of psychiatric disorders including MDD varies between 7.0% and 20.3% across countries (Bijl et al. 2003; Demyttenaere et al. 2004). In the United States, less than 30% of adults who are positively screened for depression received any treatment (Olfson et al. 2016). In China, the discordant treatment ranges from 4% to 60% of MDD patients who ever received psychosocial therapy or pharmacotherapy (Yu et al. 2015; Liu et al. 2017; Chin et al. 2015). Overall, variations in epidemiology, clinical presentation, and treatment of MDD are significantly influenced by sociocultural and economic factors (Kleinman 2004; Compton et al. 2006; Zhong et al. 2018).
In the 1950s, imipramine and iproniazid were used, for the first time, for the pharmacotherapy of MDD, thus opening the history of antidepressant treatment (López-Muñoz and Alamo 2009). Consequently, these two agents contributed to the formulation of the first mechanistic hypothesis of depressive disorders, the monoaminergic theory. In the 1960s, the catecholamine hypothesis was further advanced, with the role of noradrenergic imbalance suggested to be the key pathological factor in the occurrence of MDD (Schildkraut 1965). Serotonin was at the focus of these hypotheses, which resulted in the invention of fluoxetine, the first selective serotonin reuptake inhibitor (SSRI). In recent decades, pathophysiological research of depression allowed pharmacological treatments to target specific neurotransmitters as well as the specific brain regions and types of MDD (Kutzer et al. 2020). For example, ketamine and esketamine are proposed to treat resistant depression (Abdallah et al. 2015; Stenovec et al. 2020), while brexanolone, a natural steroid, is the first Food and Drug Administration (FDA)-approved therapeutic medication of postpartum depression (Meltzer-Brody et al. 2018).
Despite considerable advances in studying MDD pathophysiology in recent decades, and an abundance of pharmacological agents approved for MDD treatment, a substantial number of MDD patients remains resistant to available antidepressants. A real-life sequential treatment study demonstrates that nearly 30% of MDD patients fail to achieve remission, even after multiple treatment attempts (Rush et al. 2004, 2006). This may reflect the fact that existing theories cannot fully explain pathological mechanisms of MDD; similarly, pharmacological mechanisms of action of available antidepressants are in need of further research. Overreliance on the monoaminergic hypothesis may stipulate limited success of antidepressants (Ceskova 2016; Ceskova and Silhan 2018).
Growing evidence supports the key role of astrocytes in the pathophysiology and pharmacology of MDD. Astrocytes are homeostatic cells of the central nervous system (CNS). They control multiple aspects of brain physiology at all levels of organization from molecular to organ and systemic (Verkhratsky and Butt 2013; Verkhratsky and Parpura 2015; Verkhratsky and Nedergaard 2018). An individual protoplasmic astrocyte interacts with as many as 100,000 synapses in mice and possibly up to 2,000,000 synapses in humans (Bushong et al. 2002; Oberheim et al. 2009); these astrocytic perisynaptic structures, known as synaptic cradle (Verkhratsky and Nedergaard 2014), are fundamental for maintaining neurotransmission in the CNS. Pathological changes in astrocytes, classified into astrogliosis, astrodegeneration, or atrophy with loss of function, are salient features of all neurological disorders (Pekny et al. 2016; Verkhratsky et al. 2017) including neurodevelopmental (Zeidán-Chuliá et al. 2014) and neuropsychiatric (Verkhratsky et al. 2014; Peng et al. 2015, 2016) disorders. Based on reports of decreased astroglial numbers in postmortem histopathological studies of MDD patients, the dysfunction of astrocytes was postulated to contribute to the pathophysiology of depression (Cotter et al. 2001; Rajkowska et al. 2007; Hercher et al. 2009; Verkhratsky et al. 2014).
Expression of multiple receptors and transporters makes astrocytes potential targets for specific therapy, and indeed, astrocytes respond to many antidepressants (Hertz et al. 2014; Saura et al. 1992; Zhang et al. 2010). Astrocytes possess serotonin receptor subtypes associated with the pharmacology and pathophysiology of depression (Hertz et al. 2010, 2015; Ding et al. 2013; Kirischuk et al. 1996). In this chapter, we focus on astrocytic serotonin receptors and discuss their relation to MDD.
Serotonin Receptors in the Treatments of MDD
A multitude of neurotransmitter receptors is expressed in astrocytes, including ionotropic and metabotropic glutamate receptors, purinoceptors, α/β-adrenoceptors, and serotonin receptors (Hertz et al. 2015), all associated with the intracellular excitability (Verkhratsky and Parpura 2010; Verkhratsky et al. 2020, 2021). Astrocytic serotonin receptors are of particular importance for MDD because of the widespread application of SSRIs in clinical practice. Stimulation of serotonin receptors in astrocytes activates cytosolic and nuclear signaling pathways, altering cellular functions as well as gene expression (Peng et al. 2015).
There are seven subtypes of serotonin receptors designated as 5-HT1A-1F, 5-HT2A-2C, 5-HT3, 5-HT4, 5-HT5A, 5-HT6, and 5-HT7. Among these subtypes, 5-HT1A-1F, 5-HT2A-2C, 5-HT6, and 5-HT7 receptors were discovered in cerebral or spinal astrocytes in humans and rodents (Hirst et al. 1997, 1998; Meuser et al. 2002; Merzak et al. 1996; Li et al. 2012). These serotonin receptors can be involved in the pathophysiology of MDD through numerous cellular signaling pathways. All astrocytic serotonin receptors are G-coupled protein receptors being linked to phospholipase C and inositol-1,4,5-trisphosphate (InsP3)-dependent Ca2+ signaling (Verkhratsky et al. 2012).
5-HT1 Receptors
The 5-HT1A and 5-HT1B receptors attract particular attention because of their involvement in the pathophysiology of MDD; these receptors are also pharmacological targets in the clinical therapeutics of depression (Moret and Briley 2000; Murrough et al. 2011; Murrough and Neumeister 2011; Ruf and Bhagwagar 2009; Sari 2004; Savitz et al. 2009; Tiger et al. 2014). The 5-HT1A and 5-HT1B receptors display 43% amino acid sequence homology, and both are the Gi/o protein-coupled receptors (Hoyer et al. 2002). These receptor subtypes have different cellular localizations, with 5-HT1A receptors mainly expressed in somata and dendrites (Sotelo et al. 1990) and 5-HT1B receptors being primarily localized in axon terminals (Boschert et al. 1994). Furthermore, 5-HT1A receptor is one of the most abundant in cortical regions implicated in mood and emotion (Albert and François 2010; Albert and Lemonde 2004). The 5-HT1B receptors are widely distributed throughout the brain regions, with particularly dense expression in the substantia nigra and globus pallidus (Bonaventure et al. 1997; Varnäs et al. 2011).
The 5-HT1A receptors act as autoreceptors and heteroreceptors. The presynaptic 5-HT1A receptors are mainly the autoreceptors localized in the cell bodies and dendrites of serotonergic neurones in dorsal raphe nuclei. These 5-HT1A receptors modulate the release of serotonin and cell discharge rates. Stimulation of 5-HT1A autoreceptors decreases the firing activity of central serotonergic neurones and synthesis and release of serotonin by a negative feedback mechanism while promoting the depressive-like behaviors and the resistance responses to antidepressants, thus inhibiting their activity and having opposite results (Richardson-Jones et al. 2010, 2011; Milak et al. 2018). The postsynaptic 5-HT1A receptors are heteroreceptors, localized in glutamatergic and γ-aminobutyric acid (GABA)-ergic neurones; these receptors show substantial expression in the hippocampus, medial septum, prefrontal cortex, and midbrain ventral tegmental regions where they regulate the release of acetylcholine, glutamate, and dopamine (Comley et al. 2015; Borroto-Escuela et al. 2018; López-Gil et al. 2008; Jeltsch-David et al. 2010; Di Matteo et al. 2008). The 5-HT1A heteroreceptors play key roles in desensitization of postsynaptic 5-HT1A receptors, especially in prefrontal cortex regions (Taciak et al. 2018; Naumenko et al. 2014). Inhibition of 5-HT1A presynaptic autoreceptors or stimulation of 5-HT1A postsynaptic heteroreceptors may produce antidepressant effects, and hence 5-HT1A receptor antagonists or agonists may act as antidepressants or antidepressants potentiators, respectively (Richardson-Jones et al. 2011; Philippe et al. 2018).
There are indeed novel antidepressants based on 5-HT1A receptor agonism; these include YL-0919, brexpiprazole, vilazodone, and vortioxetine hydrobromide. Some of these 5-HT1A receptor agonists also act as SSRIs and as agonists or antagonists of other serotonin subunits. For example, vortioxetine hydrobromide is the agonist of 5-HT1A receptor and SSRI; it is a partial agonist of 5-HT1B receptors and antagonist of 5-HT3 and 5-HT7 receptors (Wang et al. 2019). Compound MIN-117, an antagonist of 5-HT1A autoreceptors, is considered for adjunctive therapy to improve acute antidepressant effects of SSRIs (Artigas et al. 2001; Sahli et al. 2016).
Similar to 5-HT1A receptors, 5-HT1B receptors are classified into autoreceptors and heteroreceptors. In postmortem tissue from the suicide MDD subjects, the level of 5-HT1B receptors is decreased in the hippocampus; the mRNA of 5-HT1B receptors is also lower in the frontal cortex. Conversely, mRNA of 5-HT1B receptors is higher in the hypothalamus, compared with a healthy group (Anisman et al. 2008). Serotonin binding to the 5-HT1B receptors in serotonergic neurones decreases the formation of cAMP and the downstream signaling responses, which results in the reduction of transmitter release (Barnes and Sharp 1999; Leenders and Sheng 2005; Middlemiss and Hutson 1990). Furthermore, 5-HT1B receptors could modulate serotonin transporters (SERT), and thus 5-HT1B autoreceptors can regulate the serotonin release from serotonergic neurones (Hagan et al. 2012; Montañez et al. 2013). An agonist of 5-HT1B receptor CP-93129 decreases 5-HT release in the hippocampus in rats (Hjorth and Tao 1991). In the hippocampus and cortex of wild-type mice, an agonist of 5-HT1B receptor decreases the level of serotonin, while an antagonist of this receptor has the opposite effect on the serotonin level. Both effects are diminished in the transgenic mice with genetically deleted 5-HT1B receptors (Rutz et al. 2006). In addition, 5-HT1B receptors localized in non-serotonergic neurones can modulate the release of glutamate, GABA, acetylcholine, and dopamine (Ruf and Bhagwagar 2009).
Serotonin release is suppressed by 5-HT1B receptor antagonists SB-616234-A (Dawson et al. 2006), AZD3783 (Zhang et al. 2011), and AR-A000002 (Hudzik et al. 2003), which makes them candidates for antidepressive therapeutics. Stimulation of 5-HT1 receptors may neutralize the positive effects of SSRIs on serotonin levels to produce the therapeutic latency (Blier and de Montigny 1994; Nutt 2002). Therefore, the antagonists of 5-HT1B receptors may act as rapid antidepressants. Available 5-HT1B receptor antagonists are not sufficiently effective as antidepressants but can be used as potential adjuvants (Ruf and Bhagwagar 2009). As a result, the combination of SSRIs with 5-HT1B receptor antagonists represents a new therapeutic scheme for MDD (Matzen et al. 2000).
5-HT2 Receptors
The 5-HT2A, 5-HT2B, and 5-HT2C receptors are Gq protein-coupled receptors, which activate phospholipase C (PLC) or protein kinase C and trigger rapid InsP3-dependent Ca2+ signaling or stimulate phospholipase A2 (PLA2) thus increasing arachidonic acid release (Masson et al. 2012; Li et al. 2008). The link between 5-HT2A receptors and astrocytes emerged in 1999 when 5-HT2A receptors were found to be significantly upregulated in reactive astrocytes (Wu et al. 1999). Trazodone, a triazolopyridine derivative, could play a neuroprotective role by inhibiting 5-HT2A/2C receptors in human reactive astrocytes, while fluoxetine also has a similar neuroprotective effects by blocking the same receptors (Daniele et al. 2015). Antagonists of 5-HT2A receptors such as EMD281014, FG5893a, and M100907 suppress depressive-like behavior as reported by forced swim test, the classical rodent behavioral test for depressive phenotype (Patel et al. 2004; Albinsson et al. 1994; Marek et al. 2005). Antagonist of 5-HT2A receptors promotes noradrenaline release under SSRI treatment (Dremencov et al. 2007), suggesting a feedback loop between 5-HT1A receptors in glutamatergic neurones and 5-HT2A receptors in GABAergic neurones. Such interaction may enhance the antidepressant activity of SSRIs and noradrenaline reuptake inhibitor (SNRIs) YM992, which is a potent 5-HT2A antagonist (Szabo and Blier 2002). Some recent studies show that functional disruption of the 5-HT2A receptors may be related to postpartum depression disorder or psychotic disorders (Gao et al. 2018).
Even though 5-HT2A receptors are widely researched with regard to the pharmacological mechanism of antidepressant action, most research focusses on neurones (Szabo and Blier 2001a, b). However, fluoxetine does not affect the mRNA expression of 5-HT2A receptors in neurones or astrocytes, isolated and sorted from transgenic mice treated with chronic unpredictable mild stress (Li et al. 2012). In contrast, astrocytic 5-HT2B receptors have shown significant changes.
Fluoxetine increases mRNA expression of 5-HT2B receptors decreased by the treatment with chronic stress solely in astrocytes, and not in neurones (Dong et al. 2015; Li et al. 2012). Similarly, in the Parkinson’s disease model, 5-HT2B receptors are downregulated in astrocytes, but not in neurones; this downregulation develops in parallel with depressive-like behaviors (Diaz et al. 2016). The 5-HT2B receptor is widely distributed in mammalian brains, including the frontal cortex, septal nuclei, dorsal hypothalamus, and medial amygdala (Duxon et al. 1997; Kursar et al. 1994; Li et al. 2012; Dong et al. 2015). In the primary cultured astrocytes or in the fluorescence-activated cell sorted astrocytes from hgfap::EGFP transgenic mice (expressing enhanced green fluorescence protein driven by a fragment of the human glial fibrillary acidic protein promoter), 5-HT2B receptors are co-expressed with 5-HT2A and 5-HT2C receptors (Li et al. 2012; Hertz et al. 2010). However, astrocytic 5-HT2B receptor has a much higher affinity for serotonin than the other two 5-HT2 receptors subtypes (Li et al. 2010). Stimulation of 5-HT2B receptors with 1 nM serotonin triggers phosphorylation of extracellular signal-regulated kinase 1/2 (ERK1/2), while 5-HT2C receptor-mediated ERK1/2 phosphorylation occurs at 100 nM, and 5-HT2A receptors are activated by serotonin in concentrations above 10μM (Li et al. 2010). All SSRIs including fluoxetine act as agonists of astrocytic 5-HT2B receptors suggesting the role of these receptors in antidepressive action (Zhang et al. 2010; Hertz et al. 2012; Li et al. 2009a, 2012, 2017).
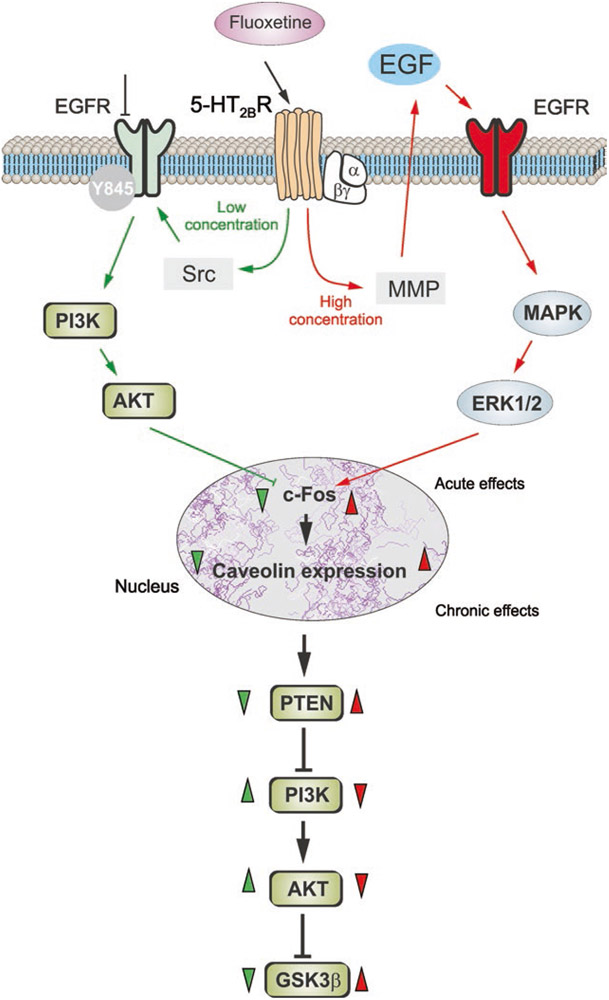
Astrocytic 5-HT2B receptors treated with various concentrations of fluoxetine trigger different signaling pathways to regulate gene expressions. Acute treatment with fluoxetine at concentrations lower than 1μM decreases mRNA expression of c-Fos through phosphoinositide 3 kinase(PI3K)-protein kinase B (also known as AKT) signaling pathway; at higher concentrations (more than 5μM), however, fluoxetine elevates this gene expression by mitogen-activated protein kinase (MAPK)-ERK signaling pathway (Fig. 1; Li et al. 2017). Stimulation of 5-HT2B receptors by fluoxetine in astrocytes leads to the transactivation of epidermal growth factor receptors (EGFR); this in turn activates downstream MAPK/ERK or PI3K/AKT signaling cascades thus regulating expressions of genes possibly related to mood disorders. These genes include Ca2+-dependent phospholipase A2 (cPLA2), subtype 2 of adenosine deaminases acting on RNA’s (ADAR2), or subtype 2 of kainate receptors (GluK2) (Li et al. 2009b, 2011, 2012; Rao et al. 2006). Chronic treatment with fluoxetine also induces RNA editing of ADAR2, resulting in the loss of function of 5-HT2B receptors, although expression of these receptors is increased (Peng et al. 2014). Rapid increase in [Ca2+]i triggered by an agonist of 5-HT2B receptors was suppressed by chronic treatment with fluoxetine (Li et al. 2011; Peng et al. 2014). Malfunction of 5-HT2B receptors has been discovered in chronic stress animal models of depression and in animals with depressive-like behaviors associated with Parkinson’s disease or hyperammonemia (Li et al. 2012; Zhang et al. 2015; Yue et al. 2019). This evidence indicates that 5-HT2B receptors are a target for SSRIs, and it is indicative of the potential contribution of astrocytic 5-HT2B receptors to the pathophysiology of MDD (Kennett et al. 1996, 1998). Agonist of 5-HT2B receptors BW723C86 displays anxiolytic-like effects, whereas 5-HT2C/2B receptor antagonist SB200646A improves depressive-like behaviors (Kennett et al. 1995).
Fig. 1.
Schematic illustration of biphasic concentration-dependent regulation of Cav-1 gene expression and GSK-3β activity by fluoxetine in astrocytes. Fluoxetine is an agonist to serotonin 2B receptors (5-HT2BR). Acute treatment with fluoxetine at low concentrations (green arrows) stimulates Src which phosphorylates epidermal growth factor (EGF) receptors (EGFR) at Y845 residue and in turn activates phosphoinositide 3 kinase (PI3K)-AKT signaling pathway. The AKT phosphorylation by fluoxetine at low concentrations inhibits c-Fos gene expression and subsequently decreases caveolin-1 (Cav-1) gene expression (chronic effects) that in turn decreases membrane content of PTEN (phosphatase and tensin homolog), induces phosphorylation and stimulation of PI3K, and elevates glycogen synthase kinase 3 (GSK-3β) phosphorylation thus suppressing its activity. At higher concentrations, fluoxetine (red arrows) stimulates metalloproteinase (MMP) and induces shedding of growth factor which stimulates EGFR and activates mitogen-activated protein kinase (MAPK) extracellular signal-regulated kinases 1/2 (ERK1/2) signal pathway. The ERK1/2 phosphorylation by fluoxetine at high concentrations stimulates cFos gene expression and subsequently increases Cav-1 gene expression (chronic effects), which acts on PTEN/PI3K/AKT/GSK-3β in an inverse fashion. (From Li et al. 2017)
The 5-HT2C receptors are upregulated by fluoxetine specifically in neurones sorted from Thy1::YFP transgenic mice, expressing yellow fluorescence protein driven by Thy1 promoter (Li et al. 2012). Antagonist of 5-HT2C receptor S32006 demonstrates antidepressive effects and increases the level of dopamine and noradrenaline in the frontal cortex in rats (Dekeyne et al. 2008). An inverse agonist of 5-HT2C receptor, S32212, also has antidepressant activity in behavioral tests (Dekeyne et al. 2012). Fluoxetine is reported to act as an antagonist of 5-HT2C receptors (Ni and Miledi 1997), while chronic treatment with SSRIs may promote receptor desensitization (Yamauchi et al. 2004). Mianserin and several classical tricyclic antidepressants (TCAs), such as amitriptyline, imipramine, and clomipramine, all work as antagonists with moderate to high affinity at 5-HT2C receptors (Jenck et al. 1993; Millan 2005). SNRIs desipramine and maprotiline as well as atypical antidepressants mirtazapine, trazodone, and nefazodone similarly act as 5-HT2C antagonists (Ni and Miledi 1997).
Increased density of 5-HT2C receptors was found in the frontal cortex of subjects who committed suicide (Pandey et al. 2006). This increase may reflect changes in mRNA alternative splicing and/or RNA editing (Martin et al. 2013), which has been proposed as a risk factor for suicidal attempts (Dracheva et al. 2008). Clinical studies similarly indicated increases in mRNA editing of 5-HT2C receptors in the frontal cortex of depressed suicide individuals (Niswender et al. 2001; Gurevich et al. 2002; Iwamoto and Kato 2003; Schmauss 2003). A recent study demonstrated that astrocytic 5-HT2C receptors are involved in the depressive- and anxious-like behaviors induced by alcohol addiction. Fluoxetine at low concentration (1μM) can alleviate these anxiety-like behaviors and motor activity induced by alcohol by suppressing the expression of ADAR2 and mRNA editing of 5-HT2C receptors (Li et al. 2020).
The study in chronic social defeat animal model of depression reported increased sensitivity of 5-HT2C receptors that can be reversed by chronic antidepressants (Berton et al. 2006; Rygula et al. 2005, 2006). Pretreatment with 5-HT2C receptor agonist m-chlorophenylpiperazine increases behavioral presentations of defeat in hamsters (Harvey et al. 2012). In rats, however, chronic stress increases depressive-like behavior, this effect being arguably mediated through 5-HT2C receptors (Kimura et al. 2008; Moreau et al. 1993). Suppression of dopamine release mediated by 5-HT2C receptors has been observed in rat models of depression (Dremencov et al. 2005; Oba et al. 2013). Antagonists of 5-HT2C receptors can alleviate stress-induced depressive-like behaviors in mice, while their antidepressive effects may relate to dopaminergic neurones in the ventral tegmental area (Opal et al. 2013). Chronic treatment with 5-HT2C receptor agonist RO60-0175 alleviates anhedonia in stressed mice by desensitization of the receptor (Moreau et al. 1993). Depressive-like behaviors in the learned helplessness model can be improved by selective antagonists of 5-HT2C receptors (Strong et al. 2009). Likewise, clomipramine, a potential antagonist of 5-HT2C receptors, decreases elevated mRNA expression of 5-HT2C receptors in chronic stress treated rats (Pitychoutis et al. 2012).
All these observations suggest that the dysfunctions of 5-HT2 receptors may be a consequence of stress and can be linked to anxiety, depression, and suicide. Antidepressive effects of agonists and antagonists of 5-HT2 receptors remain controversial and poorly understood, thus warranting more research.
Other Serotonin Receptors
The 5-HT3 is the only ionotropic member of the serotonin receptors family. This 5HT-gated channel is assembled from five subunits, 5HT3A, 5HT3B, 5HT3D, 5HT3E, and 5HT3F, expression of which was confirmed in humans (Niesler et al. 2007). In rodents, however, only 5-HT3A and 5HT3B subunits have been identified (Karnovsky et al. 2003). Ondansetron and zacopride, antagonists of 5HT3 receptors, have been used in clinical therapeutics of depression, and both reversed the depressive-like behaviors in the rat learned helplessness model (Martin et al. 1992; Thiebot and Martin 1991). Antagonists of 5HT3 receptors potentiate the pharmacological efficiency of clinical antidepressants (Nakagawa et al. 1998; Redrobe and Bourin 1997). Vortioxetine is a multimodal drug which binds to 5-HT1A, 5-HT1B, 5-HT1D, and 5-HT7 receptors and to SERT; it also acts as a potent antagonist of 5HT3 receptors. Vortioxetine demonstrated antidepressive effects in rodent models of depression (Guilloux et al. 2013; Mork et al. 2012; Wallace et al. 2014). Furthermore, preclinical data also show that vortioxetine as an antagonist of 5HT3 receptors produced antidepressant response faster than classical antidepressants (Bétry et al. 2013). In addition, vortioxetine demonstrates antidepressive performance in patients resistant to SSRIs or SNRIs (Alvarez et al. 2014). 5HT3 receptor antagonists may be promising therapeutic agents, although certain unresolved issues remain, including those of heterogeneity of the species used and of the inconsistent data on effective doses as antidepressants (Adrien et al. 1992; Costall et al. 1987; Johnson et al. 2002, 2006; Martin et al. 1992; Ramamoorthy et al. 2008).
5-HT4 receptors are widely distributed in limbic regions, such as the amygdala and hippocampus (Hannon and Hoyer 2008; Tanaka et al. 2012). Being Gs-coupled, 5-HT4 receptors increase intracellular cAMP by stimulating adenylyl cyclase (Dumuis et al. 1988; Fagni et al. 1992). Decrease in 5-HT4 receptors was observed in the striatum of depressed patients and in mice models (Amigó et al. 2016; Madsen et al. 2014). In postmortem studies of depressed suicide victims, various binding forms of 5-HT4 receptors and the different levels of cAMP in distinct brain areas have been reported (Rosel et al. 2004). A selective partial agonist of 5-HT4 receptors, RS67333, has a rapid antidepressant effect in chronic depression models of rodents (Lucas et al. 2007).
In MDD context, studies of 5-HT5, 5-HT6, and 5-HT7 receptors are scarce. Administration of N-methyl-D-aspartate receptor antagonist ketamine, at a dose that leads to drug abuse, can trigger schizophrenic-like symptoms in animals (de la Salle et al. 2016). In these ketamine pretreated mice, an agonist of 5-HT6 receptors E-6837 as well as an antagonist of these receptors SB-271046 can both produce depressive-like behaviors (Suárez-Santiago et al. 2017). Antagonists of 5-HT7 receptors have faster antidepressant effects when compared to fluoxetine in the rat model of depression (Mnie-Filali et al. 2011).
To summarize, there are many uncertainties and controversies concerning MDD-related pharmacology of serotonin receptors. In general, agonists of 5-HT1A, 5-HT2B, and 5-HT4 receptors and antagonists of 5-HT1B, 5-HT2A, 5-HT2C, 5-HT3, 5-HT6, and 5-HT7 act as antidepressants. None of these drugs, however, can substantially affect clinical manifestations of depression.
Astrocytes as a Target for Classic Antidepressants
Classic antidepressants are SSRIs, SNRIs, TCAs, inhibitors of monoamine oxidase (MAO), and lithium. Major theories of MDD pathophysiology focus on neurones, with particular emphasis on monoamines (Marathe et al. 2018), aberrant signaling of brain-derived neurotrophic factor (BDNF), leptin dysregulation (Milaneschi et al. 2017), myo-inositol abnormalities (Yu and Greenberg 2016), and glutamate homeostasis failure (Shirayama et al. 2017). Here, we discuss the MDD from the astrocytic angle and portray astrocytes as targets for therapy.
SSRIs
SSRIs, which include fluoxetine, fluvoxamine, sertraline, paroxetine, and citalopram, are the most popular antidepressants and anxiolytics. An increase in extracellular serotonin availability following SERT inhibition has been regarded as the main and the only pharmacological mechanism of SSRIs, despite the well-known delay in clinical effect contrasting to the immediate inhibition of SERT. Recently, the contribution of astrocytes to MDD has gained increased attention. Postmortem histological analysis of the frontal cortex reported a reduced number of glia in patients suffering from MDD (Cotter et al. 2001, 2002). Decreased density of neuroglia and decrease in glia-to-neurone ratio have also been observed in the amygdala (Bowley et al. 2002; Hamidi et al. 2004). Experimental ablation of astrocytes (but not neurones) in the frontal cortex is sufficient to initiate depressive-like behavior in rats (Banasr and Duman 2008). Similarly, loss of astrocyte-derived ATP induces depressive phenotypes in rodents (Cao et al. 2013). Fluoxetine reverses the loss of hippocampal astrocytes in an animal model of depression (Czéh et al. 2006), while increasing the release of ATP from astrocytes by regulating vesicular nucleotide transporter (Kinoshita et al. 2018).
Fluoxetine acts as an agonist of astrocytic 5-HT2B receptors. However, acute and chronic treatments of astrocytes with fluoxetine produce different outcomes. Acute treatment with 10μM fluoxetine triggers Ca2+ signals, transactivates the EGFR, and phosphorylates downstream ERK1/2 signaling cascade; ERK1/2 enters nuclei and regulates the mRNA and protein expression of c-Fos and FosB, consequently modulating the expression of several key proteins (Li et al. 2008). For instance, fluoxetine, by stimulating 5-HT2B receptors, increases protein expression of cPLA2, GluK2, ADAR2, transient receptor potential canonical 1 channels, L-type Cav1.2 Ca2+ channels, caveolin-1, and BDNF (Li et al. 2009a, 2011, 2017, 2019; Hertz et al. 2015; Peng et al. 2018). Increased expression of ADAR2 in astrocytes following chronic treatment with fluoxetine catalyzes RNA editing, promoting loss of function of 5-HT2B receptors and GluK2, which affects Ca2+ signaling and phosphorylation of ERK1/2 (Li et al. 2011; Hertz et al. 2012). Decreased expression of astrocytic 5-HT2B receptors was also observed in chronic stress models and Parkinson’s disease (Dong et al. 2015; Zhang et al. 2015). Fluoxetine can selectively upregulate the expression of 5-HT2B receptors in astrocytes (Li et al. 2012). However, PI3K/AKT and MAPK/ERK signaling pathways induced by low and high concentrations of fluoxetine result in the opposite regulation of caveolin-1 and glycogen synthase kinase-3β (GSK-3β) (Fig. 1) (Li et al. 2017). Some effects of fluoxetine mediated by 5-HT2B receptors may contribute to both therapeutic and adverse effects of this drug; hence, selective stimulation or inhibition of various signaling pathways triggered by fluoxetine may promote the efficacy or lessen the side effects of antidepressants.
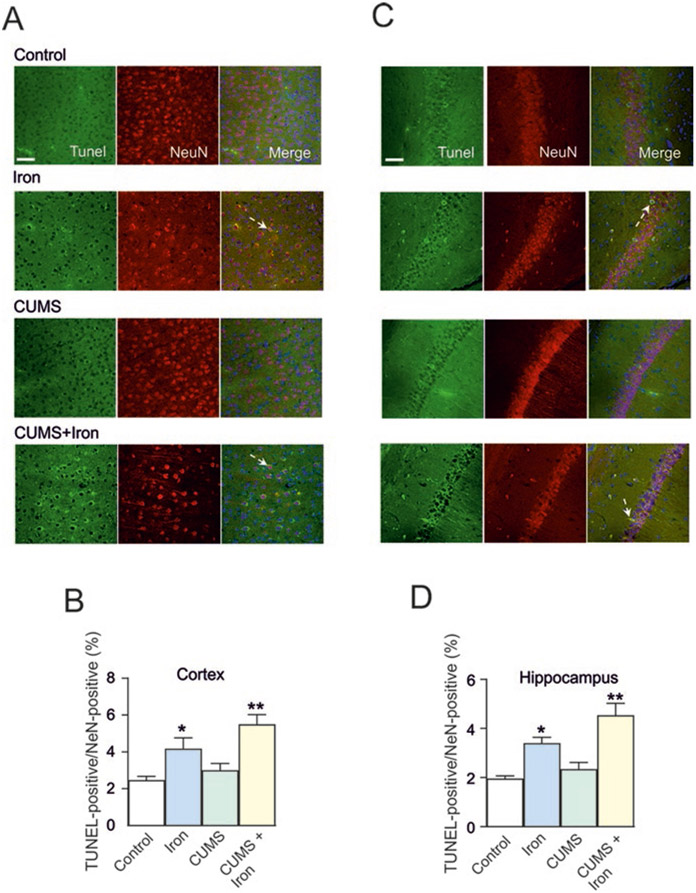
Depressive disorders are associated with many neurological conditions including sleep disturbances, Alzheimer’s, and Parkinson’s diseases (Xia et al. 2018; Orgeta et al. 2017; Huang et al. 2019). Aging of astrocytes (Verkhratsky et al. 2020, 2021) may contribute to age-dependent diseases as well as to depressive phenotypes. Sleep disturbance in particular is linked to both depression and neurodegeneration. Insomnia and depression can easily combine into a vicious cycle, as decreased sleep time increases the risk for MDD, which further exacerbates sleep disorders (Roberts and Duong 2014). The glymphatic system (Iliff et al. 2012) is responsible for the clearance of brain metabolite waste via paravascular pathways (Xie et al. 2013). Astrocytic endfeet forming glia limitans vascularis and endfeet-polarized expression of aquaporin 4 (AQP4) support the glymphatic system. In aging, astroglial atrophy decreases glymphatic clearance facilitating extracellular accumulation of disease-related proteins, such as β-amyloid (Kress et al. 2014). Increased accumulation of β-amyloid has been reported by positron emission tomography in the right hippocampus and thalamus of human subjects having their moods affected by sleep deprivation (Shokri-Kojori et al. 2018). In animals, chronic stress increases β-amyloid42 in the frontal cortex and hippocampus. Fluoxetine upregulates the expression of AQP4 and promotes clearance of β-amyloid42 by improving glymphatic clearance; this also rescues depressive-like behaviors (Xia et al. 2017). Toxic load with iron also impairs the glymphatic system, thus exacerbating depressive-like phenotypes and increasing neuronal apoptosis (Fig. 2; Liang et al. 2020).
Fig. 2.
Treatment with chronic stress enhances neuronal apoptosis triggered by overload with iron. Mice were pretreated without or with chronic unpredictable mild stress (CUMS) for 6 weeks; in week 6, animals were randomly separated to be injected with dextran (CUMS group) or iron dextran (CUMS+Iron group) for 6 days. Apoptosis was detected by TUNEL assay in the cortex (a) and hippocampus (c), where neuronal nuclei were stained with NeuN (red), while all the cell nuclei were labeled with DAPI (blue in merge). Percentage of cell death was determined by the ratio of TUNEL+ and NeuN+ cells, with the cortical and hippocampal regions analyzed in (b, d), respectively. Data are presented as mean ± SEM, n = 6. *p < 0.05, statistically significant difference compared with control, untreated group; **p < 0.05, statistically significant difference compared with any other group. (From Liang et al. 2020)
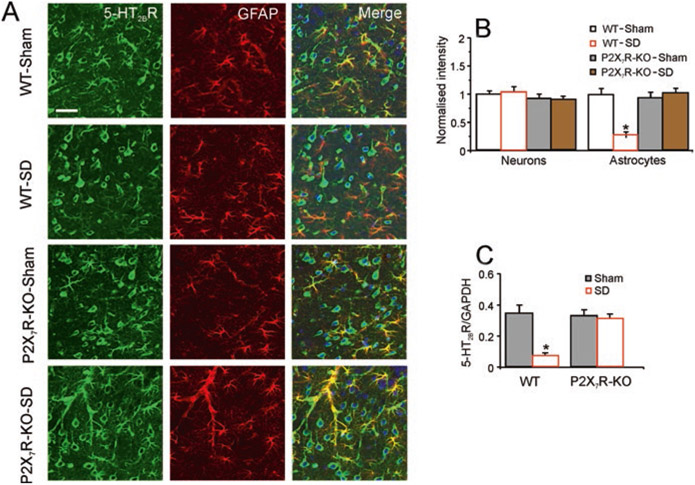
Growing evidence indicates a strong link between sleep deprivation and activation of nucleotide-binding domain and leucine-rich repeat protein-3 (NLRP3) inflammasome; this activation is mediated by astrocytic P2X7 receptors in astrocytes, and it facilitates neuronal apoptosis (Xia et al. 2018; Zielinski et al. 2017). Chronic sleep deprivation selectively downregulates expression of astrocytic 5-HT2B receptors, the effect of which is similarly mediated by P2X7 receptors (Fig. 3; Xia et al. 2020). These changes result in depressive-like behaviors associated with the gradual elevation of extracellular ATP (Xia et al. 2020). Fluoxetine increases the phosphorylation of signal transducer and activator of transcription 3, thus suppressing astrocytic NLRP3 activation and neuronal apoptosis (Xia et al. 2018; Zielinski et al. 2017). Inhibition of astrocytic NLRP3 inflammasome by fluoxetine is also mediated by 5-HT2B receptors (Li et al. 2019). In addition, fluoxetine, again acting through 5-HT2B receptors, recovers BDNF levels suppressed by sleep deprivation (Li et al. 2019). Pre-treatment with the 16 kDa hormone leptin increases expression of astrocytic 5-HT2B receptors, thus enhancing positive effects of fluoxetine on BDNF and depressive-like behaviors (Li et al. 2019). Of note, long-term, chronic stress may induce sleep disorders and depression associated with decreased levels of BDNF (Schmitt et al. 2016).
Fig. 3.
P2X7 receptors mediate the selective decrease of 5-HT2B receptors in astrocytes treated with chronic sleep deprivation. (a) Immunolabelled 5-HT2B receptors (green) were co-stained with glial fibrillary acidic protein (GFAP, red) and NeuN (blue) in the mice frontal cortex treated with sham (Control) or exposed to sleep deprivation for 3 weeks of wild-type (WT) and P2X7 receptor knockout (P2X7R-KO) mice. Scale bar, 20μm. (b) 5-HT2B receptors immunolabeling intensity of neurones and astrocytes, respectively, relative to the cell-free parenchyma in the cortex, normalized to the intensity of a matching sham group. (c) RT-PCR analysis of 5-HT2B receptors mRNA expression in WT or P2X7R-KO mice treated with sleep deprivation for 3 weeks, expressed as the relative expression ratio of 5-HT2B receptor/GAPDH, the latter used as loading control. Data represent mean ± SEM. *Indicates statistically significant (p < 0.05) difference from matching sham groups, n = 6. (From Xia et al. 2020)
Pathological features and pathogenesis of sleep disturbances, depressive disorders, and neurodegenerative pathology, such as Alzheimer’s disease, usually overlap, and all these may be linked to astrocytes. Morphological and functional abnormalities of astrocytes can impair BDNF signaling and the glymphatic system, with failed glymphatic clearance being a key contributor to diseases of cognition (Nedergaard and Goldman 2020), which could further worsen the deposition of brain waste and impair neuronal functions. Fluoxetine and other SSRIs may provide neuroprotection by arresting astrogliopathology through direct stimulation of 5-HT2B receptors.
SNRIs
Serotonin/noradrenaline reuptake inhibitors exert their effects through increasing availability of serotonin and noradrenaline in the nervous tissue, and they are commonly used to treat patients with refractory depression (Wang et al. 2014). Venlafaxine is one of the most commonly prescribed SNRI with a broad range of antidepressant effects. Accumulating evidence shows that venlafaxine may target astrocytes by affecting the metabolism of lysine, tyrosine, glutamate, methionine, ethanolamine, fructose-6-phosphate, and phosphorylethanolamine (Sun et al. 2017). Duloxetine, another SNRI widely used in the treatment of MDD (Nemeroff et al. 2002), was reported to suppress ischemia-induced astrocytic reactivity and oxidative stress (Lee et al. 2016).
TCAs and MAOIs
Astrocytic-derived neurotrophic growth factors receive attention because of their links to the mechanism of action of antidepressants (Tsybko et al. 2017). In primary cultured astrocytes, amitriptyline increases expression of BDNF, vascular endothelial growth factor (VEGF), fibroblast growth factor 2 (FGF2), and glial cell line-derived neurotrophic factor (GDNF) (Kajitani et al. 2012). Furthermore, clomipramine (TCA), fluvoxamine (SSRI), and duloxetine (SNRI) all elevate the expression of FGF2 and BDNF in astrocytes (Hisaoka-Nakashima et al. 2016; Kajitani et al. 2012). Fluoxetine and paroxetine both enhance the mRNA expression of VEGF in primary astrocytes (Allaman et al. 2011).
Mirtazapine, a tetracyclic piperazinoazepine antidepressant, has a distinct pharmacological mechanism associated with inhibition of α2-adrenoceptors and 5-HT2 and 5-HT3 receptors (Benjamin and Doraiswamy 2011; Croom et al. 2009). Unlike amitriptyline and other antidepressants, mirtazapine does not affect noradrenaline or serotonin reuptake (Al-Majed et al. 2018). Moreover, mirtazapine has a faster onset time as compared with SNRIs in the treatment of acute-phase MDD (Nagao et al. 2013; Watanabe et al. 2011). In primary culture of astrocytes, mirtazapine increases the expression of GDNF and BDNF by stimulating phosphorylation of ERK1/2 (Hisaoka-Nakashima et al. 2016).
Astrocytes in Treatments of Bipolar Disorders
Therapeutic strategy for bipolar disorders (BD) is distinct from treating MDD, due to reflecting different pathophysiology. Lithium ion (Li+) in various salts, valproic acid (VPA), and carbamazepine (CBZ) are three classical anti-bipolar drugs. Lithium salt was first to be used for treating mania and depressive episodes of bipolar depression; importantly, lithium may also prevent relapses (Brown and Tracy 2013). Both CBZ and VPA are used for treating manic episodes and epileptic seizures (Nevitt et al. 2019; Xu et al. 2019; Post et al. 2007). These three classical anti-BD drugs have nothing in common in their chemical structures, and only a few metabolic parameters in the brain are affected by them in a similar manner (Li et al. 2007). Revealing shared effects of these drugs is important because common mechanisms of action may provide information about the pathophysiology of BP and facilitate drug development.
Astroglial cell numbers in the cortex from BD patients are reduced (Ongür et al. 1998, 2008; Rajkowska 2000). This coincides with exaggerated activity of the glutamatergic system, as demonstrated by MRI, in the brains of BD patients (Ongür et al. 2008; Michael et al. 2003; Chen et al. 2010; Eastwood and Harrison 2010), which could be due to suppressed capacity of astrocytes to maintain glutamate homeostasis. In astrocytes, riluzole can increase the expression and activity of plasmalemmal glutamate transporters in order to increase the glutamate clearance from the extracellular space (Carbone et al. 2012). Riluzole can also improve the glutamine-glutamate cycle and the glutamatergic transmission in brains of BD patients (Brennan et al. 2010) and even alleviate related disease symptoms (Zarate and Manji 2008), which is also indicative of potential astrocytic glutamatergic dysfunction.
We outline some common targets of these three classical anti-BD drugs on astrocytes. Firstly, the decreased expression of GluK2 was reported in the entorhinal cortex and the hippocampus of BD patients, most of whom had received drug therapy (Beneyto et al. 2007; Benes et al. 2001). In primary cultures of astrocytes treated with Li+, VPA, or CBZ at clinically relevant doses the mRNA expression of GluK2 receptors decreased, whereas the expression of other subunits of GluK receptors remained unchanged (Li et al. 2009b). In vivo, after 2 weeks of intraperitoneal injection, CBZ reduced the gene expression of GluK2 which is encoded by GRIK2 (Li et al. 2009b); of note, this gene in a genetic linkage region (6q21) is associated with BD (Schulze et al. 2004). In other studies, CBZ, VPA, or Li+ suppresses GluK2-mediated phosphorylation of ERK1/2 in primary cultured astrocytes, while anticonvulsant topiramate is ineffective (Li et al. 2009b). Release of glutamate from astrocytes induced by ATP is inhibited by all three anti-BD drugs; this may suppress hyperactivity of the glutamatergic transmission in brains of patients with BD (Liu et al. 2015).
In addition to classical targets, Li+ can play a key role in the metabolism of inositol. The intracellular myoinositol needs continuous re-supply from the diet (Spector and Lorenzo 1975) and de novo synthesis, which occurs in the vasculature (Wong et al. 1987). In primary culture of mouse astrocytes, chronic treatment with Li+ at 1 mM inhibits the uptake of inositol, although this effect is absent during acute Li+ treatment (Wolfson et al. 1998). Changes in myoinositol levels may have opposite effects depending on its concentration. For example, myoinositol at low concentration promotes the uptake of the extracellular inositol, whereas at high concentrations the uptake is inhibited. This may be related to the existence of two inositol transporters in astrocytes, the high-affinity Na+-dependent myoinositol transporter (SMIT/SLC5A3), which accounts for most of the uptake at low (25μM) inositol concentration, and the lower-affinity H+-dependent myoinositol transporter (HMIT/SLC2A13), which dominates the uptake at higher (50μM) inositol concentrations (Wolfson et al. 2000). Because the elevated intracellular pH (pHi) stimulates SMIT but inhibits HMIT, the effects of anti-BD drugs on the inhibition of inositol may be caused by the intracellular alkalinization. This was further confirmed by studying the uptake of [3H]myoinositol in astrocytes at various myoinositol doses or at various pHi and corroborated by analysis of different myoinositol transporter operation (Fu et al. 2012). Chronic treatment by any of the three anti-BD drugs increases astrocytic pHi (Song et al. 2008, 2013). The Na+–H+ exchanger (NHE) exchanger (NHE) is one of the acid-controlling transporters, widely distributed in almost all cell types, including neurones and astrocytes (McAlear and Bevensee 2006). The NHE/SLC9 family has seven subtypes, NHE1-NHE7. The NHE1/SLC9A1 is expressed in cultured astrocytes from the rat hippocampus (Pizzonia et al. 1996) and from the mouse cerebral cortex (Song et al. 2008). Chronic treatment with Li+ at 1 mM alkalinize pHi by about 0.10, but 0.5 mM is ineffective (Song et al. 2008).
The anti-bipolar effects of lithium are often attributed to the classical hypothesis of the depletion of myoinositol induced by Li+ (Berridge et al. 1989). According to this hypothesis, Li+ causes the non-competitive inhibition of inositol-phosphate hydrolysis; InsP3 is produced by PLC and degraded to myoinositol by inositol phosphatases, whereas diacylglycerol is converted to phosphatidate and condensed with cytidine triphosphate to form cytidine mono-phosphoryl-phosphatidate, which together with myoinositol regenerates phosphatidylinositol 4,5-bisphosphate (PIP2) (Hallcher and Sherman 1980; Inhorn and Majerus 1987). Because Li+ inhibits the formation of inositol from InsP3, it decreases the regeneration of PIP2 and blocks PLC pathway. The [Ca2+]i response to noradrenaline in cultured astrocytes is suppressed by chronic treatments with Li+ (Chen and Hertz 1996).
As astrocytes are the direct targets of Li+, identification of the unique astroglial transcriptional networks is of importance. Recently, it was found that Li+ can induce a specific astrocytic phenotype (Rivera and Butt 2019). By pharmacogenomic analyses, the roles of the extracellular matrix (ECM) regulatory enzyme lysyl oxidase (LOX) and peroxisome proliferator-activated receptor γ (PPAR-γ) were shown to modulate astrocyte morphogenesis. According to genomic analyses, LOX is the most highly regulated Li+-responsive astroglial gene. Irreversible LOX inhibitor mimics effects of Li+ suggesting that LOX is a major regulator of astrocyte morphology and proliferation. Treatment with Li+ also actives PPAR-γ (Liu et al. 2011), while PPAR-γ has been reported to inhibit LOX (Segond et al. 2013). These effects may involve GSK3β-Wnt signaling, which is implicated in neuropsychiatric disorders and interacts with ECM remodeling. In addition, PPAR-γ agonists decrease TGF-β1 signaling leading to decreased fibrosis (Vallee et al. 2017), providing a further link with LOX and the actions of Li+.
Neuroprotective effects of anti-bipolar drugs have been discovered in other neuronal diseases. Pretreatment with VPA increases expression of plasmalemmal glutamate transporter 1 (GLT-1) in astrocytes and promotes the recovery of locomotor activities in a rodent model (Johnson Jr et al. 2018). Lithium can mediate neuroprotection by inhibition of GSK3β after spinal cord injury (Li et al. 2018). Chronic treatment with Li+ activates α1 subunit of neuronal Na+-K+-ATPase (Li et al. 2018). In contrast, chronic treatment with CBZ increases astrocyte specific α2 subunit containing Na+/K+ ATPase (Li et al. 2013).
Conclusion
In this chapter, we introduced the contribution of astrocytic serotonin receptors to the pathophysiology of MDD and to pharmacological mechanisms of classic antidepressants. Direct stimulation of astroglial 5-HT2B receptors may be the common target of SSRIs. Activation of astrocytic serotonin receptors associates with several signaling pathways and regulates expression of numerous genes that are related to depressive-like behaviors. Recent discoveries overviewed in this chapter highlight the key roles of astrocytes in the pathogenesis and treatment of mood disorders.
Acknowledgments
BL’s work is supported by the National Natural Science Foundation of China (grant number 8187185), LiaoNing Revitalization Talents Program (grant number XLYC1807137), the Scientific Research Foundation for Returned Scholars of Education Ministry of China (grant number 20151098), LiaoNing Thousands Talents Program (grant number 202078), and “ChunHui” Program of Education Ministry of China (grant number 2020703). CS’s work is supported by a grant from the Italian Ministry of Education, University and Research (2015KP7T2Y_002) and a grant from the Sapienza University of Rome (RM11916B7A8D0225). VP’s work is supported by a grant from the National Institute of General Medical Sciences of the National Institutes of Health (R01GM123971). VP is an Honorary Professor at the University of Rijeka, Croatia.
Contributor Information
Alexei Verkhratsky, Faculty of Biology, Medicine and Health, The University of Manchester, Manchester, UK; Achucarro Center for Neuroscience, IKERBASQUE, Basque Foundation for Science, Bilbao, Spain.
Vladimir Parpura, Department of Neurobiology, The University of Alabama at Birmingham, Birmingham, AL, USA.
Caterina Scuderi, Department of Physiology and Pharmacology “Vittorio Erspamer”, SAPIENZA University of Rome, Rome, Italy.
Baoman Li, Department of Forensic Analytical Toxicology, School of Forensic Medicine, China Medical University, Shenyang, People’s Republic of China.
References
- Abdallah CG, Sanacora G, Duman RS, Krystal JH (2015) Ketamine and rapid-acting antidepressants: a window into a new neurobiology for mood disorder therapeutics. Annu Rev Med 66:509–523 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Adrien J, Tissier MH, Lanfumey L, Haj-Dahmane S, Jolas T, Franc B, Hamon M (1992) Central action of 5-HT3 receptor ligands in the regulation of sleep-wakefulness and raphe neuronal activity in the rat. Neuropharmacology 31:519–529 [DOI] [PubMed] [Google Scholar]
- Albert PR, François BL (2010) Modifying 5-HT1A receptor gene expression as a new target for antidepressant therapy. Front Neurosci 4:35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Albert PR, Lemonde S (2004) 5-HT1A receptors, gene repression, and depression: guilt by association. Neuroscientist 10:575–593 [DOI] [PubMed] [Google Scholar]
- Albinsson A, Björk A, Svartengren J, Klint T, Andersson G (1994) Preclinical pharmacology of FG5893: a potential anxiolytic drug with high affinity for both 5-HT1A and 5-HT2A receptors. Eur J Pharmacol 261:285–294 [DOI] [PubMed] [Google Scholar]
- Allaman I, Fiumelli H, Magistretti PJ, Martin JL (2011) Fluoxetine regulates the expression of neurotrophic/growth factors and glucose metabolism in astrocytes. Psychopharmacology 216:75–84 [DOI] [PubMed] [Google Scholar]
- Al-Majed A, Bakheit AH, Alharbi RM, Abdel Aziz HA (2018) Mirtazapine. Profiles Drug Subst Excip Relat Methodol 43:209–254 [DOI] [PubMed] [Google Scholar]
- Alvarez E, Perez V, Artigas F (2014) Pharmacology and clinical potential of vortioxetine in the treatment of major depressive disorder. Neuropsychiatr Dis Treat 10:1297–1307 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Amigó J, Díaz A, Pilar-Cuéllar F, Vidal R, Martín A, Compan V, Pazos A, Castro E (2016) The absence of 5-HT4 receptors modulates depression- and anxiety-like responses and influences the response of fluoxetine in olfactory bulbectomised mice: adaptive changes in hippocampal neuroplasticity markers and 5-HT1A autoreceptor. Neuropharmacology 111:47–58 [DOI] [PubMed] [Google Scholar]
- Anisman H, Du L, Palkovits M, Faludi G, Kovacs GG, Szontagh-Kishazi P, Merali Z, Poulter MO (2008) Serotonin receptor subtype and p11 mRNA expression in stress-relevant brain regions of suicide and control subjects. J Psychiatry Neurosci 33:131–141 [PMC free article] [PubMed] [Google Scholar]
- Artigas F, Celada P, Laruelle M, Adell A (2001) How does pindolol improve antidepressant action? Trends Pharmacol Sci 22:224–228 [DOI] [PubMed] [Google Scholar]
- Banasr M, Duman RS (2008) Glial loss in the prefrontal cortex is sufficient to induce depressive-like behaviors. Biol Psychiatry 64:863–870 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barnes NM, Sharp T (1999) A review of central 5-HT receptors and their function. Neuropharmacology 38:1083–1152 [DOI] [PubMed] [Google Scholar]
- Benes FM, Todtenkopf MS, Kostoulakos P (2001) GluR5,6,7 subunit immunoreactivity on apical pyramidal cell dendrites in hippocampus of schizophrenics and manic depressives. Hippocampus 11:482–491 [DOI] [PubMed] [Google Scholar]
- Beneyto M, Kristiansen LV, Oni-Orisan A, McCullumsmith RE, Meador-Woodruff JH (2007) Abnormal glutamate receptor expression in the medial temporal lobe in schizophrenia and mood disorders. Neuropsychopharmacology 32:1888–1902 [DOI] [PubMed] [Google Scholar]
- Benjamin S, Doraiswamy PM (2011) Review of the use of mirtazapine in the treatment of depression. Expert Opin Pharmacother 12:1623–1632 [DOI] [PubMed] [Google Scholar]
- Berridge MJ, Downes CP, Hanley MR (1989) Neural and developmental actions of lithium: a unifying hypothesis. Cell 59:411–419 [DOI] [PubMed] [Google Scholar]
- Berton O, McClung CA, Dileone RJ, Krishnan V, Renthal W, Russo SJ, Graham D, Tsankova NM, Bolanos CA, Rios M, Monteggia LM, Self DW, Nestler EJ (2006) Essential role of BDNF in the mesolimbic dopamine pathway in social defeat stress. Science 311:864–868 [DOI] [PubMed] [Google Scholar]
- Bétry C, Pehrson AL, Etiévant A, Ebert B, Sánchez C, Haddjeri N (2013) The rapid recovery of 5-HT cell firing induced by the antidepressant vortioxetine involves 5-HT3 receptor antagonism. Int J Neuropsychopharmacol 16:1115–1127 [DOI] [PubMed] [Google Scholar]
- Bijl RV, de Graaf R, Hiripi E, Kessler RC, Kohn R, Offord DR, Ustun TB, Vicente B, Vollebergh WA, Walters EE, Wittchen HU (2003) The prevalence of treated and untreated mental disorders in five countries. Health Aff (Millwood) 22:122–133 [DOI] [PubMed] [Google Scholar]
- Blier P, de Montigny C (1994) Current advances and trends in the treatment of depression. Trends Pharmacol Sci 15:220–226 [DOI] [PubMed] [Google Scholar]
- Bonaventure P, Schotte A, Cras P, Leysen JE (1997) Autoradiographic mapping of 5-HT1B- and 5-HT1D receptors in human brain using [3H]alniditan, a new radioligand. Recept Channels 5:225–230 [PubMed] [Google Scholar]
- Borroto-Escuela DO, Narváez M, Ambrogini P, Ferraro L, Brito I, Romero-Fernandez W, Andrade-Talavera Y, Flores-Burgess A, Millon C, Gago B, Narvaez JA, Odagaki Y, Palkovits M, Diaz-Cabiale Z, Fuxe K (2018) Receptor−receptor interactions in multiple 5-HT1A heteroreceptor complexes in raphe-hippocampal 5-HT transmission and their relevance for depression and its treatment. Molecules 23:1341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boschert U, Amara DA, Segu L, Hen R (1994) The mouse 5-hydroxytryptamine1B receptor is localized predominantly on axon terminals. Neuroscience 58:167–182 [DOI] [PubMed] [Google Scholar]
- Bowley MP, Drevets WC, Ongür D, Price JL (2002) Low glial numbers in the amygdala in major depressive disorder. Biol Psychiatry 52:404–412 [DOI] [PubMed] [Google Scholar]
- Brennan BP, Hudson JI, Jensen JE, McCarthy J, Roberts JL, Prescot AP, Cohen BM, Pope HG Jr, Renshaw PF, Ongür D (2010) Rapid enhancement of glutamatergic neurotransmission in bipolar depression following treatment with riluzole. Neuropsychopharmacology 35:834–846 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown KM, Tracy DK (2013) Lithium: the pharmacodynamic actions of the amazing ion. Ther Adv Psychopharmacol 3:163–176 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bushong EA, Martone ME, Jones YZ, Ellisman MH (2002) Protoplasmic astrocytes in CA1 stratum radiatum occupy separate anatomical domains. J Neurosci 22:183–192 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buysse DJ, Angst J, Gamma A, Ajdacic V, Eich D, Rössler W (2008) Prevalence, course, and comorbidity of insomnia and depression in young adults. Sleep 31:473–480 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cao X, Li LP, Wang Q, Wu Q, Hu HH, Zhang M, Fang YY, Zhang J, Li SJ, Xiong WC, Yan HC, Gao YB, Liu JH, Li XW, Sun LR, Zeng YN, Zhu XH, Gao TM (2013) Astrocyte-derived ATP modulates depressive-like behaviors. Nat Med 19:773–777 [DOI] [PubMed] [Google Scholar]
- Carbone M, Duty S, Rattray M (2012) Riluzole elevates GLT-1 activity and levels in striatal astrocytes. Neurochem Int 60:31–38 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cassano GB, Rucci P, Frank E, Fagiolini A, Dell’Osso L, Shear MK, Kupfer DJ (2004) The mood spectrum in unipolar and bipolar disorder: arguments for a unitary approach. Am J Psychiatry 161:1264–1269 [DOI] [PubMed] [Google Scholar]
- Ceskova E (2016) Current pharmacotherapy of depression – focused on multimodal/multifunctional antidepressants. Expert Opin Pharmacother 17:1835–1837 [DOI] [PubMed] [Google Scholar]
- Ceskova E, Silhan P (2018) Novel treatment options in depression and psychosis. Neuropsychiatr Dis Treat 14:741–747 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen Y, Hertz L (1996) Inhibition of noradrenaline stimulated increase in [Ca2+]iin cultured astrocytes by chronic treatment with a therapeutically relevant lithium concentration. Brain Res 711:245–248 [DOI] [PubMed] [Google Scholar]
- Chen CH, Suckling J, Ooi C, Jacob R, Lupson V, Bullmore ET, Lennox BR (2010) A longitudinal fMRI study of the manic and euthymic states of bipolar disorder. Bipolar Disord 12:344–347 [DOI] [PubMed] [Google Scholar]
- Chin WY, Chan KT, Lam CL, Lam TP, Wan EY (2015) Help-seeking intentions and subsequent 12-month mental health service use in Chinese primary care patients with depressive symptoms. BMJ Open 5:e006730. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Comley RA, van der Aart J, Gulyás B, Garnier M, Iavarone L, Halldin C, Rabiner EA (2015) In vivo occupancy of the 5-HT1A receptor by a novel pan 5-HT1(A/B/D) receptor antagonist, GSK588045, using positron emission tomography. Neuropharmacology 92:44–48 [DOI] [PubMed] [Google Scholar]
- Compton WM, Conway KP, Stinson FS, Grant BF (2006) Changes in the prevalence of major depression and comorbid substance use disorders in the United States between 1991–1992 and 2001–2002. Am J Psychiatry 163:2141–2147 [DOI] [PubMed] [Google Scholar]
- Costall B. Domeney AM. Gerrard PA. Kelly ME. Naylor RJ. Tyers MB (1987) Effects of the 5-HT3 receptor antagonists GR38032F, CS 215-930 and BRL 43694 in tests for anxiolytic activity. Br J Pharmacol 91:195P [Google Scholar]
- Cotter D. Mackay D. Landau S. Kerwin R. Everall I (2001) Reduced glial cell density and neuronal size in the anterior cingulate cortex in major depressive disorder. Arch Gen Psychiatry 58:545–553 [DOI] [PubMed] [Google Scholar]
- Cotter D. Mackay D. Chana G. Beasley C. Landau S. Everall IP (2002) Reduced neuronal size and glial cell density in area 9 of the dorsolateral prefrontal cortex in subjects with major depressive disorder. Cereb Cortex 12:386–394 [DOI] [PubMed] [Google Scholar]
- Croom KF, Perry CM. Plosker GL (2009) Mirtazapine: a review of its use in major depression and other psychiatric disorders. CNS Drugs 23:427–452 [DOI] [PubMed] [Google Scholar]
- Czéh B. Simon M. Schmelting B. Hiemke C. Fuchs E (2006) Astroglial plasticity in the hippocampus is affected by chronic psychosocial stress and concomitant fluoxetine treatment. Neuropsychopharmacology 31:1616–1626 [DOI] [PubMed] [Google Scholar]
- Daniele S. Zappelli E. Martini C (2015) Trazodone regulates neurotrophic/growth factors, mitogen-activated protein kinases and lactate release in human primary astrocytes. J Neuroinflammation 12:225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dawson LA. Hughes ZA. Starr KR, Storey JD. Bettelini L. Bacchi F. Dawson LA. Hughes ZA. Starr KR. Storey JD. Bettelini L. Bacchi F. Arban R. Poffe A. Melotto S. Hagan JJ, Price GW (2006) Characterisation of the selective 5-HT1B receptor antagonist SB-616234-A (1-[6-(cis-3,5-dimethylpiperazin-1-yl)-2,3-dihydro-5-methoxyindol-1-yl]-1-[2′-methyl-4′-(5-methyl-1,2,4-oxadiazol-3-yl)biphenyl-4-yl]methanone hydrochloride): in vivo neurochemical and behavioural evidence of anxiolytic/antidepressant activity. Neuropharmacology 50:975–983 [DOI] [PubMed] [Google Scholar]
- de la Salle S. Choueiry J. Shah D. Bowers H. McIntosh J. Ilivitsky V. Knott V (2016) Effects of ketamine on resting-state EEG activity and their relationship to perceptual/dissociative symptoms in healthy humans. Front Pharmacol 7:348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dekeyne A. Mannoury la Cour C. Gobert A. Brocco M. Lejeune F. Serres F. Sharp T, Daszuta A, Soumier A, Papp M. Rivet JM, Flik G, Cremers TI, Muller O, Lavielle G, Millan MJ (2008) S32006, a novel 5-HT2C receptor antagonist displaying broad-based antidepressant and anxiolytic properties in rodent models. Psychopharmacology 199:549–568 [DOI] [PubMed] [Google Scholar]
- Dekeyne A, Brocco M, Loiseau F, Gobert A, Rivet JM, Di Cara B, Cremers TI. Flik G. Fone KC. Watson DJ, Papp M, Sharp T, Serres F, Cespugho R, Olivier B, Chan JS, Lavielle G, Millan MJ (2012) S32212, a novel serotonin type 2C receptor inverse agonist/α2-adrenoceptor antagonist and potential antidepressant: II. A behavioral, neurochemical, and electrophysiological characterization. J Pharmacol Exp Ther 340:765–780 [DOI] [PubMed] [Google Scholar]
- Demyttenaere K, Bruffaerts R, Posada-Villa J, Gasquet I, Kovess V, Lepine JP, Angermeyer MC, Bernert S, de Girolamo G, Morosini P, Polidori G, Kikkawa T, Kawakami N, Ono Y, Takeshima T, Uda H, Karam EG, Fayyad JA, Karam AN, Mneimneh ZN, Medina-Mora ME, Borges G, Lara C, de Graaf R, Ormel J, Gureje O, Shen Y, Huang Y, Zhang M, Alonso J, Haro JM, Vilagut G, Bromet EJ, Gluzman S, Webb C. Kessler RC, Merikangas KR, Anthony JC, Von Korff MR, Wang PS, Brugha TS, Aguilar-Gaxiola S, Lee S, Heeringa S, Pennell BE, Zaslavsky AM, Ustun TB, Chatterji S, WHO World Mental Health Survey Consortium (2004) Prevalence, severity, and unmet need for treatment of mental disorders in the World Health Organization World Mental Health Surveys. JAMA 291:2581–2590 [DOI] [PubMed] [Google Scholar]
- Diaz SL, Narboux-Nême N, Boutourlinsky K, Doly S, Maroteaux L (2016) Mice lacking the serotonin 5-HT2B receptor as an animal model of resistance to selective serotonin reuptake inhibitors antidepressants. Eur Neuropsychopharmacol 26:265–279 [DOI] [PubMed] [Google Scholar]
- Di Matteo V, Di Giovanni G, Pierucci M, Esposito E (2008) Serotonin control of central dopaminergic function: focus on in vivo microdialysis studies. Prog Brain Res 172:7–44 [DOI] [PubMed] [Google Scholar]
- Ding F, O’Donnell J, Thrane AS, Zeppenfeld D, Kang H, Xie L, Wang F, Nedergaard M (2013) α1-adrenergic receptors mediate coordinated Ca2+ signaling of cortical astrocytes in awake, behaving mice. Cell Calcium 54:387–394 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dong L, Li B, Verkhratsky A, Peng L (2015) Cell type-specific in vivo expression of genes encoding signalling molecules in the brain in response to chronic mild stress and chronic treatment with fluoxetine. Psychopharmacology 232:2827–2835 [DOI] [PubMed] [Google Scholar]
- Dracheva S, Chin B, Haroutunian V (2008) Altered serotonin2C receptor RNA splicing in suicide: association with editing. Neuroreport 19:379–382 [DOI] [PubMed] [Google Scholar]
- Dremencov E, Newman ME, Kinor N, Blatman-Jan G, Schindler CJ, Overstreet DH, Yadid G (2005) Hyperfunctionality of serotonin-2C receptor-mediated inhibition of accumbal dopamine release in an animal model of depression is reversed by antidepressant treatment. Neuropharmacology 48:34–42 [DOI] [PubMed] [Google Scholar]
- Dremencov E, El Mansari M, Blier P (2007) Noradrenergic augmentation of escitalopram response by risperidone: electrophysiologic studies in the rat brain. Biol Psychiatry 61:671–678 [DOI] [PubMed] [Google Scholar]
- Dumuis A, Bouhelal R, Sebben M, Cory R, Bockaert J (1988) A nonclassical 5-hydroxytryptamine receptor positively coupled with adenylate cyclase in the central nervous system. Mol Pharmacol 34:880–887 [PubMed] [Google Scholar]
- Duxon MS, Flanigan TP, Reavley AC, Baxter GS, Blackburn TP, Fone KC (1997) Evidence for expression of the 5-hydroxytryptamine-2B receptor protein in the rat central nervous system. Neuroscience 76:323–329. Editing. Neuroscientist. 9:237–42. [DOI] [PubMed] [Google Scholar]
- Eastwood SL, Harrison PJ (2010) Markers of glutamate synaptic transmission and plasticity are increased in the anterior cingulate cortex in bipolar disorder. Biol Psychiatry 67:1010–1016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fagni L, Dumuis A, Sebben M, Bockaert J (1992) The 5-HT4 receptor subtype inhibits K+ current in colliculi neurones via activation of a cyclic AMP-dependent protein kinase. Br J Pharmacol 105:973–979 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ferrari AJ, Somerville AJ, Baxter AJ, Norman R, Patten SB, Vos T, Whiteford HA (2013) Global variation in the prevalence and incidence of major depressive disorder: a systematic review of the epidemiological literature. Psychol Med 43:471–481 [DOI] [PubMed] [Google Scholar]
- Fröjd SA, Nissinen ES, Pelkonen MU, Marttunen MJ, Koivisto AM, Kaltiala-Heino R (2008) Depression and school performance in middle adolescent boys and girls. J Adolesc 31:485–498 [DOI] [PubMed] [Google Scholar]
- Fu H, Li B, Hertz L, Peng L (2012) Contributions in astrocytes of SMIT1/2 and HMIT to myoinositol uptake at different concentrations and pH. Neurochem Int 61:187–194 [DOI] [PubMed] [Google Scholar]
- Gao J, Wu R, Davis C, Li M (2018) Activation of 5-HT2A receptor disrupts rat maternal behavior. Neuropharmacology 128:96–105 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gu L, Xie J, Long J, Chen Q, Chen Q, Pan R, Yan Y, Wu G, Liang B, Tan J, Xie X, Wei B, Su L (2013) Epidemiology of major depressive disorder in mainland China: a systematic review. PLoS One 8:e65356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guilloux JP, Mendez-David I, Pehrson A, Guiard BP, Repérant C, Orvoën S, Gardier AM, Hen R, Ebert B, Miller S, Sanchez C, David DJ (2013) Antidepressant and anxiolytic potential of the multimodal antidepressant vortioxetine (Lu AA21004) assessed by behavioural and neurogenesis outcomes in mice. Neuropharmacology 73:147–159 [DOI] [PubMed] [Google Scholar]
- Gurevich I, Tamir H, Arango V, Dwork AJ, Mann JJ, Schmauss C (2002) Altered editing of serotonin 2C receptor pre-mRNA in the prefrontal cortex of depressed suicide victims. Neuron 34:349–356 [DOI] [PubMed] [Google Scholar]
- Hagan CE, McDevitt RA, Liu Y, Furay AR, Neumaier JF (2012) 5-HT(1B) autoreceptor regulation of serotonin transporter activity in synaptosomes. Synapse 66:1024–1034 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hallcher LM, Sherman WR (1980) The effects of lithium ion and other agents on the activity of myo-inositol-1-phosphatase from bovine brain. J Biol Chem 255:10896–10901 [PubMed] [Google Scholar]
- Hamidi M, Drevets WC, Price JL (2004) Glial reduction in amygdala in major depressive disorder is due to oligodendrocytes. Biol Psychiatry 55:563–569 [DOI] [PubMed] [Google Scholar]
- Hannon J, Hoyer D (2008) Molecular biology of 5-HT receptors. Behav Brain Res 195:198–213 [DOI] [PubMed] [Google Scholar]
- Harvey ML, Swallows CL, Cooper MA (2012) A double dissociation in the effects of 5-HT2A and 5-HT2C receptors on the acquisition and expression of conditioned defeat in Syrian hamsters. Behav Neurosci 126:530–537 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hercher C, Turecki G, Mechawar N (2009) Through the looking glass: examining neuroanatomical evidence for cellular alterations in major depression. J Psychiatr Res 43:947–961 [DOI] [PubMed] [Google Scholar]
- Hertz L, Lovatt D, Goldman SA, Nedergaard M (2010) Adrenoceptors in brain: cellular gene expression and effects on astrocytic metabolism and [Ca(2+)]i. Neurochem Int 57:411–420 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hertz L, Li B, Song D, Ren J, Dong L, Chen Y, Peng L (2012) Astrocytes as a 5-HT2B-mediated SERT-independent SSRI target, slowly altering depression-associated genes and function. Curr Signal Transduction Ther 7:65–80 [Google Scholar]
- Hertz L, Song D, Li B, Du T, Xu J, Gu L, Chen Y, Peng L (2014) Signal transduction in astrocytes during chronic or acute treatment with drugs (SSRIs, antibipolar drugs, GABA-ergic drugs, and benzodiazepines) ameliorating mood disorders. J Signal Transduct 2014:593934. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hertz L, Rothman DL, Li B, Peng L (2015) Chronic SSRI stimulation of astrocytic 5-HT2B receptors change multiple gene expressions/editings and metabolism of glutamate, glucose and glycogen: a potential paradigm shift. Front Behav Neurosci 9:25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hirst WD, Price GW, Rattray M, Wilkin GP (1997) Identification of 5-hydroxytryptamine receptors positively coupled to adenylyl cyclase in rat cultured astrocytes. Br J Pharmacol 120:509–515 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hirst WD, Cheung NY, Rattray M, Price GW, Wilkin GP (1998) Cultured astrocytes express messenger RNA for multiple serotonin receptor subtypes, without functional coupling of 5-HT1 receptor subtypes to adenylyl cyclase. Brain Res Mol Brain Res 61:90–99 [DOI] [PubMed] [Google Scholar]
- Hisaoka-Nakashima K, Kajitani N, Kaneko M, Shigetou T, Kasai M, Matsumoto C, Yokoe T, Azuma H, Takebayashi M, Morioka N, Nakata Y (2016) Amitriptyline induces brain-derived neurotrophic factor (BDNF) mRNA expression through ERK-dependent modulation of multiple BDNF mRNA variants in primary cultured rat cortical astrocytes and microglia. Brain Res 1634:57–1667 [DOI] [PubMed] [Google Scholar]
- Hjorth S, Tao R (1991) The putative 5-HT1B receptor agonist CP-93,129 suppresses rat hippocampal 5-HT release in vivo: comparison with RU 24969. Eur J Pharmacol 209:249–252 [DOI] [PubMed] [Google Scholar]
- Hoyer D, Hannon JP, Martin GR (2002) Molecular, pharmacological and functional diversity of 5-HT receptors. Pharmacol Biochem Behav 71:533–554 [DOI] [PubMed] [Google Scholar]
- Huang MH, Cheng CM, Huang KL, Hsu JW, Bai YM, Su TP, Li CT, Tsai SJ, Lin WC, Chen TJ, Chen MH (2019) Bipolar disorder and risk of Parkinson disease: a nationwide longitudinal study. Neurology 92:e2735–e2742 [DOI] [PubMed] [Google Scholar]
- Hudzik TJ, Yanek M, Porrey T, Evenden J, Paronis C, Mastrangelo M, Ryan C, Ross S, Stenfors C (2003) Behavioral pharmacology of AR-A000002, a novel, selective 5-hydroxytryptamine(1B) antagonist. J Pharmacol Exp Ther 304:1072–1084 [DOI] [PubMed] [Google Scholar]
- Iliff JJ, Wang M, Liao Y, Plogg BA, Peng W, Gundersen GA, Benveniste H, Vates GE, Deane R, Goldman SA, Nagelhus EA, Nedergaard M (2012) A paravascular pathway facilitates CSF flow through the brain parenchyma and the clearance of interstitial solutes, including amyloid β. Sci Transl Med 4:147ra111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Inhorn RC, Majerus PW (1987) Inositol polyphosphate 1-phosphatase from calf brain. Purification and inhibition by Li+, Ca2+, and Mn2+. J Biol Chem 262:15946–15952 [PubMed] [Google Scholar]
- Iwamoto K, Kato T (2003) RNA editing of serotonin2C receptor in human postmortem brains of major mental disorders. Neurosci Lett 346:169–172 [DOI] [PubMed] [Google Scholar]
- Jeltsch-David H, Koenig J, Cassel JC (2010) Modulation of cholinergic functions by serotonin and possible implications in memory: general data and focus on 5-HT(1A) receptors of the medial septum. Behav Brain Res 195:86–97 [DOI] [PubMed] [Google Scholar]
- Jenck F, Moreau JL, Mutel V, Martin JR, Haefely WE (1993) Evidence for a role of 5-HT1C receptors in the antiserotonergic properties of some antidepressant drugs. Eur J Pharmacol 231:223–229 [DOI] [PubMed] [Google Scholar]
- Johnson J Jr, Pajarillo EAB, Taka E, Reams R, Son DS, Aschner M, Lee E (2018) Valproate and sodium butyrate attenuate manganese-decreased locomotor activity and astrocytic glutamate transporters expression in mice. Neurotoxicology 64:230–239 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnson BA, Roache JD, Ait-Daoud N, Zanca NA, Velazquez M (2002) Ondansetron reduces the craving of biologically predisposed alcoholics. Psychopharmacology 160:408–413 [DOI] [PubMed] [Google Scholar]
- Johnson BA, Roache JD, Ait-Daoud N, Javors MA, Harrison JM, Elkashef A, Mojsiak J, Li SH, Bloch DA (2006) A preliminary randomized, double-blind, placebo-controlled study of the safety and efficacy of ondansetron in the treatment of cocaine dependence. Drug Alcohol Depend 84:256–263 [DOI] [PubMed] [Google Scholar]
- Kajitani N, Hisaoka-Nakashima K, Morioka N, Okada-Tsuchioka M, Kaneko M, Kasai M, Shibasaki C, Nakata Y, Takebayashi M (2012) Antidepressant acts on astrocytes leading to an increase in the expression of neurotrophic/growth factors: differential regulation of FGF-2 by noradrenaline. PLoS One 7:e51197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karnovsky AM, Gotow LF, McKinley DD, Piechan JL, Ruble CL, Mills CJ, Schellin KA, Slightom JL, Fitzgerald LR, Benjamin CW, Roberds SL (2003) A cluster of novel serotonin receptor 3-like genes on human chromosome 3. Gene 319:137–148 [DOI] [PubMed] [Google Scholar]
- Kendler KS, Gardner CO, Fiske A, Gatz M (2009) Major depression and coronary artery disease in the Swedish twin registry: phenotypic, genetic, and environmental sources of comorbidity. Arch Gen Psychiatry 66:857–863 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kennett GA, Bailey F, Piper DC, Blackburn TP (1995) Effect of SB 200646A, a 5-HT2C/5-HT2B receptor antagonist, in two conflict models of anxiety. Psychopharmacology 118:178–182 [DOI] [PubMed] [Google Scholar]
- Kennett GA, Bright F, Trail B, Baxter GS, Blackburn TP (1996) Effects of the 5-HT2B receptor agonist, BW 723C86, on three rat models of anxiety. Br J Pharmacol 117:1443–1448 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kennett GA, Trail B, Bright F (1998) Anxiolytic-like actions of BW 723C86 in the rat Vogel conflict test are 5-HT2B receptor mediated. Neuropharmacology 37:1603–1610 [DOI] [PubMed] [Google Scholar]
- Kimura Y, Naitou Y, Wanibuchi F, Yamaguchi T (2008) 5-HT(2C) receptor activation is a common mechanism on proerectile effects of apomorphine, oxytocin and melanotan-II in rats. Eur J Pharmacol 589:157–162 [DOI] [PubMed] [Google Scholar]
- Kinoshita M, Hirayama Y, Fujishita K, Shibata K, Shinozaki Y, Shigetomi E, Takeda A, Le HPN, Hayashi H, Hiasa M, Moriyama Y, Ikenaka K, Tanaka KF, Koizumi S (2018) Anti-depressant fluoxetine reveals its therapeutic effect via astrocytes. EBioMedicine 32:72–83 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kirischuk S, Tuschick S, Verkhratsky A, Kettenmann H (1996) Calcium signalling in mouse Bergmann glial cells mediated by alpha1-adrenoreceptors and H1 histamine receptors. Eur J Neurosci 8:1198–1208 [DOI] [PubMed] [Google Scholar]
- Kleinman A (2004) Culture and depression. N Engl J Med 351:951–953 [DOI] [PubMed] [Google Scholar]
- Kress BT, Iliff JJ, Xia M, Wang M, Wei HS, Zeppenfeld D, Xie L, Kang H, Xu Q, Liew JA, Plog BA, Ding F, Deane R, Nedergaard M (2014) Impairment of paravascular clearance pathways in the aging brain. Ann Neurol 76:845–861 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kupfer DJ, Frank E, Phillips ML (2012) Major depressive disorder: new clinical, neurobiological, and treatment perspectives. Lancet 379:1045–1055 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kursar JD, Nelson DL, Wainscott DB, Baez M (1994) Molecular cloning, functional expression, and mRNA tissue distribution of the human 5-hydroxytryptamine2B receptor. Mol Pharmacol 46:227–234 [PubMed] [Google Scholar]
- Kutzer T, Dick M, Scudamore T, Wiener M, Schwartz T (2020) Antidepressant efficacy and side effect burden: an updated guide for clinicians. Drugs Context 9:2020-2-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee TK, Park JH, Ahn JH, Shin MC, Cho JH, Bae EJ, Kim YM, Won MH, Lee CH (2016) Pretreated duloxetine protects hippocampal CA1 pyramidal neurons from ischemia-reperfusion injury through decreases of glial activation and oxidative stress. J Neurol Sci 370:229–236 [DOI] [PubMed] [Google Scholar]
- Leenders AG, Sheng ZH (2005) Modulation of neurotransmitter release by the second messenger-activated protein kinases: implications for presynaptic plasticity. Pharmacol Ther 105:69–84 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li B, Gu L, Zhang H, Huang J, Chen Y, Hertz L, Peng L (2007) Up-regulation of cPLA(2) gene expression in astrocytes by all three conventional anti-bipolar drugs is drug-specific and enzyme-specific. Psychopharmacology 194:333–345 [DOI] [PubMed] [Google Scholar]
- Li B, Zhang S, Zhang H, Nu W, Cai L, Hertz L, Peng L (2008) Fluoxetine-mediated 5-HT2B receptor stimulation in astrocytes causes EGF receptor transactivation and ERK phosphorylation. Psychopharmacology 201:443–458 [DOI] [PubMed] [Google Scholar]
- Li B, Zhang S, Li M, Hertz L, Peng L (2009a) Chronic treatment of astrocytes with therapeutically relevant fluoxetine concentrations enhances cPLA2 expression secondary to 5-HT2B-induced, transactivation-mediated ERK1/2 phosphorylation. Psychopharmacology (Berlin) 207:1–12 [DOI] [PubMed] [Google Scholar]
- Li B, Zhang S, Li M, Zhang H, Hertz L, Peng L (2009b) Down-regulation of GluK2 kainate receptor expression by chronic treatment with mood-stabilizing anti-convulsants or lithium in cultured astrocytes and brain, but not in neurons. Neuropharmacology 57:375–385 [DOI] [PubMed] [Google Scholar]
- Li B, Zhang S, Li M, Hertz L, Peng L (2010) Serotonin increases ERK1/2 phosphorylation in astrocytes by stimulation of 5-HT2B and 5-HT2C receptors. Neurochem Int 57:432–439 [DOI] [PubMed] [Google Scholar]
- Li B, Zhang S, Zhang H, Hertz L, Peng L (2011) Fluoxetine affects GluK2 editing, glutamate-evoked Ca(2+) influx and extracellular signal-regulated kinase phosphorylation in mouse astrocytes. J Psychiatry Neurosci 36:322–338 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li B, Dong L, Wang B, Cai L, Jiang N, Peng L (2012) Cell type-specific gene expression and editing responses to chronic fluoxetine treatment in the in vivo mouse brain and their relevance for stress-induced anhedonia. Neurochem Res 37:2480–2495 [DOI] [PubMed] [Google Scholar]
- Li B, Hertz L, Peng L (2013) Cell-specific mRNA alterations in Na+, K+-ATPase α and β isoforms and FXYD in mice treated chronically with carbamazepine, an anti-bipolar drug. Neurochem Res 38:834–841 [DOI] [PubMed] [Google Scholar]
- Li B, Jia S, Yue T, Yang L, Huang C, Verkhratsky A, Peng L (2017) Biphasic regulation of caveolin-1 gene expression by fluoxetine in astrocytes: opposite effects of PI3K/AKT and MAPK/ERK signaling pathways on c-fos. Front Cell Neurosci 11:335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li B, Ren J, Yang L, Li X, Sun G, Xia M (2018) Lithium inhibits GSK3β activity via two different signaling pathways in neurons after spinal cord injury. Neurochem Res 43:848–856 [DOI] [PubMed] [Google Scholar]
- Li X, Liang S, Li Z, Li S, Xia M, Verkhratsky A, Li B (2019) Leptin increases expression of 5-HT2B receptors in astrocytes thus enhancing action of fluoxetine on the depressive behavior induced by sleep deprivation. Front Psych 9:734. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li Z, Lu Y, Liang S, Li S, Chen B, Zhang M, Xia M, Guan D, Verkhratsky A, Li B (2020) Fluoxetine improves behavioural deficits induced by chronic alcohol treatment by alleviating RNA editing of 5-HT2C receptors. Neurochem Int 134:104689. [DOI] [PubMed] [Google Scholar]
- Liang S, Lu Y, Li Z, Li S, Chen B, Zhang M, Chen B, Ji M, Gong W, Xia M, Verkhratsky A, Wu X, Li B (2020) Iron aggravates the depressive phenotype of stressed mice by compromising the glymphatic system. Neurosci Bull 36:1542–1546 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu KJ, Lee YL, Yang YY, Shih NY, Ho CC, Wu YC, Huang TS, Huang MC, Liu HC, Shen WW, Leu SJ (2011) Modulation of the development of human monocyte-derived dendritic cells by lithium chloride. J Cell Physiol 226:424–433 [DOI] [PubMed] [Google Scholar]
- Liu Z, Song D, Yan E, Verkhratsky A, Peng L (2015) Chronic treatment with anti-bipolar drugs suppresses glutamate release from astroglial cultures. Amino Acids 47:1045–1051 [DOI] [PubMed] [Google Scholar]
- Liu XJ, Zhou Y, Dong L, Guo Q, Chen WC, Psychiatryet DO (2017) A survey of major depression among elderly population in Wuhan (in Chinese). Chin Ment Health J 11:851–856 [Google Scholar]
- López-Gil X, Artigas F, Adell A (2008) Unraveling monoamine receptors involved in the action of typical and atypical antipsychotics on glutamatergic and serotonergic transmission in prefrontal cortex. Curr Pharm Des 16:502–515 [DOI] [PubMed] [Google Scholar]
- López-Muñoz F, Alamo C (2009) Monoaminergic neurotransmission: the history of the discovery of antidepressants from 1950s until today. Curr Pharm Des 15:1563–1586 [DOI] [PubMed] [Google Scholar]
- Lucas G, Rymar VV, Du J, Mnie-Filali O, Bisgaard C, Manta S, Lambas-Senas L, Wiborg O, Haddjeri N, Piñeyro G, Sadikot AF, Debonnel G (2007) Serotonin(4) (5-HT(4)) receptor agonists are putative antidepressants with a rapid onset of action. Neuron 55:712–725 [DOI] [PubMed] [Google Scholar]
- Lynch FL, Clarke GN (2006) Estimating the economic burden of depression in children and adolescents. Am J Prev Med 31:S143–S151 [DOI] [PubMed] [Google Scholar]
- Madsen K, Torstensen E, Holst KK, Haahr ME, Knorr U, Frokjaer VG, Brandt-Larsen M, Iversen P, Fisher PM, Knudsen GM (2014) Familial risk for major depression is associated with lower striatal 5-HT4 receptor binding. Int J Neuropsychopharmacol 18:pyu034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marathe SV, D’almeida PL, Virmani G, Bathini P, Alberi L (2018) Effects of monoamines and antidepressants on astrocyte physiology: implications for monoamine hypothesis of depression. J Exp Neurosci 12:1179069518789149 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marek GJ, Martin-Ruiz R, Abo A, Artigas F (2005) The selective 5-HT2A receptor antagonist M100907 enhances antidepressant-like behavioral effects of the SSRI fluoxetine. Neuropsychopharmacology 30:2205–2215 [DOI] [PubMed] [Google Scholar]
- Martin KF, Hannon S, Phillips I, Heal DJ (1992) Opposing roles for 5-HT1B and 5-HT3 receptors in the control of 5-HT release in rat hippocampus in vivo. Br J Pharmacol 106:139–142 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martin CB, Ramond F, Farrington DT, Aguiar AS Jr, Chevarin C, Berthiau AS, Caussanel S, Lanfumey L, Herrick-Davis K, Hamon M, Madjar JJ, Mongeau R (2013) RNA splicing and editing modulation of 5-HT(2C) receptor function: relevance to anxiety and aggression in VGV mice. Mol Psychiatry 18:656–665 [DOI] [PubMed] [Google Scholar]
- Masson J, Emerit MB, Hamon M, Darmon M (2012) Serotonergic signaling: multiple effectors and pleiotropic effects. WIREs Membr Transport Signaling 1:685–713 [Google Scholar]
- Mathers CD, Loncar D (2006) Projections of global mortality and burden of disease from 2002 to 2030. PLoS Med 3:e442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matzen L, van Amsterdam C, Rautenberg W, Greiner HE, Harting J, Seyfried CA, Böttcher H (2000) 5-HT reuptake inhibitors with 5-HT(1B/1D) antagonistic activity: a new approach toward efficient antidepressants. J Med Chem 43:1149–1157 [DOI] [PubMed] [Google Scholar]
- McAlear SD, Bevensee MO (2006) A cysteine-scanning mutagenesis study of transmembrane domain 8 of the electrogenic sodium/bicarbonate cotransporter NBCe1. J Biol Chem 281:32417–32427 [DOI] [PubMed] [Google Scholar]
- Meltzer-Brody S, Colquhoun H, Riesenberg R, Epperson CN, Deligiannidis KM, Rubinow DR, Li H, Sankoh AJ, Clemson C, Schacterle A, Jonas J, Kanes S (2018) Brexanolone injection in post-partum depression: two multicentre, double-blind, randomised, placebo-controlled, phase 3 trials. Lancet 392:1058–1070 [DOI] [PubMed] [Google Scholar]
- Merzak A, Koochekpour S, Fillion MP, Fillion G, Pilkington GJ (1996) Expression of serotonin receptors in human fetal astrocytes and glioma cell lines: a possible role in glioma cell proliferation and migration. Brain Res Mol Brain Res 41:1–7 [DOI] [PubMed] [Google Scholar]
- Meuser T, Pietruck C, Gabriel A, Xie GX, Lim KJ, Pierce Palmer P (2002) 5-HT7 receptors are involved in mediating 5-HT-induced activation of rat primary afferent neurons. Life Sci 71:2279–2289 [DOI] [PubMed] [Google Scholar]
- Michael N, Erfurth A, Ohrmann P, Gössling M, Arolt V, Heindel W, Pfleiderer B (2003) Acute mania is accompanied by elevated glutamate/glutamine levels within the left dorsolateral prefrontal cortex. Psychopharmacology 168:344–346 [DOI] [PubMed] [Google Scholar]
- Middlemiss DN, Hutson PH (1990) The 5-HT1B receptors. Ann N Y Acad Sci 600:132–147; discussion 347-48. [DOI] [PubMed] [Google Scholar]
- Milak MS, Pantazatos S, Rashid R, Zanderigo F, DeLorenzo C, Hesselgrave N, Ogden RT, Oquendo MA, Mulhern ST, Miller JM, Burke AK, Parsey RV, Mann JJ (2018) Higher 5-HT1A autoreceptor binding as an endophenotype for major depressive disorder identified in high risk offspring – a pilot study. Psychiatry Res Neuroimaging 276:15–23 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Milaneschi Y, Lamers F, Bot M, Drent ML, Penninx BW (2017) Leptin dysregulation is specifically associated with major depression with atypical features: evidence for a mechanism connecting obesity and depression. Biol Psychiatry 81(9):807–814 [DOI] [PubMed] [Google Scholar]
- Millan MJ (2005) Serotonin 5-HT2C receptors as a target for the treatment of depressive and anxious states: focus on novel therapeutic strategies. Therapie 60:441–460 [DOI] [PubMed] [Google Scholar]
- Mnie-Filali O, Faure C, Lambás-Señas L, El Mansari M, Belblidia H, Gondard E, Etiévant A, Scarna H, Didier A, Berod A, Blier P, Haddjeri N (2011) Pharmacological blockade of 5-HT7 receptors as a putative fast acting antidepressant strategy. Neuropsychopharmacology 36:1275–1288 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Montañez S, Munn JL, Owens WA, Horton RE, Daws LC (2013) 5-HT1B receptor modulation of the serotonin transporter in vivo: studies using KO mice. Neurochem Int 73:127–131 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moreau JL, Jenck F, Martin JR, Perrin S, Haefely WE (1993) Effects of repeated mild stress and two antidepressant treatments on the behavioral response to 5HT1C receptor activation in rats. Psychopharmacology 110:140–144 [DOI] [PubMed] [Google Scholar]
- Moret C, Briley M (2000) The possible role of 5-HT(1B/D) receptors in psychiatric disorders and their potential as a target for therapy. Eur J Pharmacol 404:1–12 [DOI] [PubMed] [Google Scholar]
- Mork A, Pehrson A, Brennum LT, Nielsen SM, Zhong H, Lassen AB, Miller S, Westrich L, Boyle NJ, Sánchez C, Fischer CW, Liebenberg N, Wegener G, Bundgaard C, Hogg S, Bang-Andersen B, Stensbøl TB (2012) Pharmacological effects of Lu AA21004: a novel multimodal compound for the treatment of major depressive disorder. J Pharmacol Exp Ther 340:666–675 [DOI] [PubMed] [Google Scholar]
- Murrough JW, Neumeister A (2011) The serotonin 1B receptor: a new target for depression therapeutics? Biol Psychiatry 69:714–715 [DOI] [PubMed] [Google Scholar]
- Murrough JW, Henry S, Hu J, Gallezot JD, Planeta-Wilson B, Neumaier JF, Neumeister A (2011) Reduced ventral striatal/ventral pallidal serotonin1B receptor binding potential in major depressive disorder. Psychopharmacology 213:547–553 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nagao K, Kishi T, Moriwaki M, Fujita K, Hirano S, Yamanouchi Y, Funahashi T, Iwata N (2013) Comparative clinical profile of mirtazapine and duloxetine in practical clinical settings in Japan: a 4-week open-label, parallel-group study of major depressive disorder. Neuropsychiatr Dis Treat 9:781–786 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakagawa Y, Ishima T, Takashima T (1998) The 5-HT3 receptor agonist attenuates the action of antidepressants in the forced swim test in rats. Brain Res 786:189–193 [DOI] [PubMed] [Google Scholar]
- Naumenko VS, Popova NK, Lacivita E, Leopoldo M, Ponimaskin EG (2014) Interplay between serotonin 5-HT1A and 5-HT7 receptors in depressive disorders. CNS Neurosci Ther 20:582–590 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nedergaard M, Goldman SA (2020) Glymphatic failure as a final common pathway to dementia. Science 370:50–56 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nemeroff CB, Schatzberg AF, Goldstein DJ, Detke MJ, Mallinckrodt C, Lu Y, Tran PV (2002) Duloxetine for the treatment of major depressive disorder. Psychopharmacol Bull 36:106–132 [PubMed] [Google Scholar]
- Nevitt SJ, Sudell M, Tudur Smith C, Marson AG (2019) Topiramate versus carbamazepine monotherapy for epilepsy: an individual participant data review. Cochrane Database Syst Rev 6:CD012065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ni YG, Miledi R (1997) Blockage of 5HT2C serotonin receptors by fluoxetine (Prozac). Proc Natl Acad Sci U S A 94:2036–2040 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Niesler B, Walstab J, Combrink S, Möller D, Kapeller J, Rietdorf J, Bönisch H, Göthert M, Rappold G, Brüss M (2007) Characterization of the novel human serotonin receptor subunits 5-HT3C,5-HT3D, and 5-HT3E. Mol Pharmacol 72:8–17 [DOI] [PubMed] [Google Scholar]
- Niswender CM, Herrick-Davis K, Dilley GE, Meltzer HY, Overholser JC, Stockmeier CA, Emeson RB, Sanders-Bush E (2001) RNA editing of the human serotonin 5-HT2C receptor. Alterations in suicide and implications for serotonergic pharmacotherapy. Neuropsychopharmacology 24:478–491 [DOI] [PubMed] [Google Scholar]
- Nutt DJ (2002) The neuropharmacology of serotonin and noradrenaline in depression. Int Clin Psychopharmacol 17(Suppl 1):S1–S12 [DOI] [PubMed] [Google Scholar]
- Oba A, Nakagawasai O, Onogi H, Nemoto W, Yaoita F, Arai Y, Tan-No K, Tadano T (2013) Chronic fluvoxamine treatment changes 5-HT(2A/2C) receptor-mediated behavior in olfactory bulbectomized mice. Life Sci 92:119–124 [DOI] [PubMed] [Google Scholar]
- Oberheim NA, Takano T, Han X, He W, Lin JH, Wang F, Xu Q, Wyatt JD, Pilcher W, Ojemann JG, Ransom BR, Goldman SA, Nedergaard M (2009) Uniquely hominid features of adult human astrocytes. J Neurosci 29:3276–3287 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Olfson M, Blanco C, Marcus SC (2016) Treatment of adult depression in the United States. JAMA Intern Med 176:1482–1491 [DOI] [PubMed] [Google Scholar]
- Ongür D, Drevets WC, Price JL (1998) Glial reduction in the subgenual prefrontal cortex in mood disorders. Proc Natl Acad Sci U S A 95:13290–13295 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ongür D, Jensen JE, Prescot AP, Stork C, Lundy M, Cohen BM, Renshaw PF (2008) Abnormal glutamatergic neurotransmission and neuronal-glial interactions in acute mania. Biol Psychiatry 64:718–726 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Opal MD, Klenotich SC, Morais M, Bessa J, Winkle J, Doukas D, Kay LJ, Sousa N, Dulawa SM (2013) Serotonin 2C receptor antagonists induce fast-onset antidepressant effects. Mol Psychiatry 19:1106–1114 [DOI] [PubMed] [Google Scholar]
- Orgeta V, Tabet N, Nilforooshan R, Howard R (2017) Efficacy of antidepressants for depression in Alzheimer’s disease: systematic review and meta-analysis. J Alzheimers Dis 58:725–733 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Otte C, Gold SM, Penninx BW, Pariante CM, Etkin A, Fava M, Mohr DC, Schatzberg AF (2016) Major depressive disorder. Nat Rev Dis Primers 2:16065. [DOI] [PubMed] [Google Scholar]
- Pandey GN, Dwivedi Y, Ren X, Rizavi HS, Faludi G, Sarosi A, Palkovits M (2006) Regional distribution and relative abundance of serotonin(2c) receptors in human brain: effect of suicide. Neurochem Res 31:167–176 [DOI] [PubMed] [Google Scholar]
- Patel JG, Bartoszyk GD, Edwards E, Ashby CR Jr (2004) The highly selective 5-hydroxytryptamine (5-HT)2A receptor antagonist, EMD 281014, significantly increases swimming and decreases immobility in male congenital learned helpless rats in the forced swim test. Synapse 52:73–75 [DOI] [PubMed] [Google Scholar]
- Pekny M, Pekna M, Messing A, Steinhäuser C, Lee JM, Parpura V, Hol EM, Sofroniew MV, Verkhratsky A (2016) Astrocytes: a central element in neurological diseases. Acta Neuropathol 131:323–345 [DOI] [PubMed] [Google Scholar]
- Peng L, Gu L, Li B, Hertz L (2014) Fluoxetine and all other SSRIs are 5-HT2B agonists – importance for their therapeutic effects. Curr Neuropharmacol 12:365–379 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peng L, Verkhratsky A, Gu L, Li B (2015) Targeting astrocytes in major depression. Expert Rev Neurother 15:1299–1306 [DOI] [PubMed] [Google Scholar]
- Peng L, Li B, Verkhratsky A (2016) Targeting astrocytes in bipolar disorder. Expert Rev Neurother 16:649–657 [DOI] [PubMed] [Google Scholar]
- Peng L, Song D, Li B, Verkhratsky A (2018) Astroglial 5-HT2B receptor in mood disorders. Expert Rev Neurother 18:435–442 [DOI] [PubMed] [Google Scholar]
- Penninx BW (2017) Depression and cardiovascular disease: epidemiological evidence on their linking mechanisms. Neurosci Biobehav Rev 74:277–286 [DOI] [PubMed] [Google Scholar]
- Philippe TJ, Vahid-Ansari F, Donaldson ZR, Le François B, Zahrai A, Turcotte-Cardin V, Daigle M, James J, Hen R, Merali Z, Albert PR (2018) Loss of MeCP2 in adult 5-HT neurons induces 5-HT1A autoreceptors, with opposite sex-dependent anxiety and depression phenotypes. Sci Rep 8:5788. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pitychoutis PM, Dalla C, Sideris AC, Tsonis PA, Papadopoulou-Daifoti Z (2012) 5-HT(1A), 5-HT(2A), and 5-HT(2C) receptor mRNA modulation by antidepressant treatment in the chronic mild stress model of depression: sex differences exposed. Neuroscience 210:152–167 [DOI] [PubMed] [Google Scholar]
- Pizzonia JH, Ransom BR, Pappas CA (1996) Characterization of Na+/H+ exchange activity in cultured rat hippocampal astrocytes. J Neurosci Res 44:191–198 [DOI] [PubMed] [Google Scholar]
- Post RM, Ketter TA, Uhde T, Ballenger JC (2007) Thirty years of clinical experience with carbamazepine in the treatment of bipolar illness: principles and practice. CNS Drugs 21:47–71 [DOI] [PubMed] [Google Scholar]
- Rajkowska G (2000) Postmortem studies in mood disorders indicate altered numbers of neurons and glial cells. Biol Psychiatry 48:766–777 [DOI] [PubMed] [Google Scholar]
- Rajkowska G, O’Dwyer G, Teleki Z, Stockmeier CA, Miguel-Hidalgo JJ (2007) GABAergic neurons immunoreactive for calcium binding proteins are reduced in the prefrontal cortex in major depression. Neuropsychopharmacology 32:471–482 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ramamoorthy R, Radhakrishnan M, Borah M (2008) Antidepressant-like effects of serotonin type-3 antagonist, ondansetron: an investigation in behaviour-based rodent models. Behav Pharmacol 19:29–40 [DOI] [PubMed] [Google Scholar]
- Rao JS, Ertley RN, Lee HJ, Rapoport SI, Bazinet RP (2006) Chronic fluoxetine upregulates activity, protein and mRNA levels of cytosolic phospholipase A2 in rat frontal cortex. Pharmacogenomics J 6:413–420 [DOI] [PubMed] [Google Scholar]
- Redrobe JP, Bourin M (1997) Partial role of 5-HT2 and 5-HT3 receptors in the activity of antidepressants in the mouse forced swimming test. Eur J Pharmacol 325:129–135 [DOI] [PubMed] [Google Scholar]
- Richardson-Jones JW, Craige CP, Guiard BP, Stephen A, Metzger KL, Kung HF, Gardier AM, Dranovsky A, David DJ, Beck SG, Hen R, Leonardo ED (2010) 5-HT1A autoreceptor levels determine vulnerability to stress and response to antidepressants. Neuron 65:40–52 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Richardson-Jones JW, Craige CP, Nguyen TH, Kung HF, Gardier AM, Dranovsky A, David DJ, Guiard BP, Beck SG, Hen R, Leonardo ED (2011) Serotonin-1A autoreceptors are necessary and sufficient for the normal formation of circuits underlying innate anxiety. J Neurosci 31:6008–6018 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rivera AD, Butt AM (2019) Astrocytes are direct cellular targets of lithium treatment: novel roles for lysyl oxidase and peroxisome-proliferator activated receptor-γ as astroglial targets of lithium. Transl Psychiatry 9:211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roberts RE, Duong HT (2014) The prospective association between sleep deprivation and depression among adolescents. Sleep 37:239–244 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rosel P, Arranz B, Urretavizcaya M, Oros M, San L, Navarro MA (2004) Altered 5-HT2A and 5-HT4 postsynaptic receptors and their intracellular signalling systems IP3 and cAMP in brains from depressed violent suicide victims. Neuropsychobiology 49:189–195 [DOI] [PubMed] [Google Scholar]
- Ruf BM, Bhagwagar Z (2009) The 5-HT1B receptor: a novel target for the pathophysiology of depression. Curr Drug Targets 10:1118–1138 [DOI] [PubMed] [Google Scholar]
- Rush AJ, Fava M, Wisniewski SR, Lavori PW, Trivedi MH, Sackeim HA, Thase ME, Nierenberg AA, Quitkin FM, Kashner TM, Kupfer DJ, Rosenbaum JF, Alpert J, Stewart JW, McGrath PJ, Biggs MM, Shores-Wilson K, Lebowitz BD, Ritz L, Niederehe G, STAR*D Investigators Group (2004) Sequenced treatment alternatives to relieve depression (STAR*D): rationale and design. Control Clin Trials 25:119–142 [DOI] [PubMed] [Google Scholar]
- Rush AJ, Trivedi MH, Wisniewski SR, Nierenberg AA, Stewart JW, Warden D, Niederehe G, Thase ME, Lavori PW, Lebowitz BD, McGrath PJ, Rosenbaum JF, Sackeim HA, Kupfer DJ, Luther J, Fava M (2006) Acute and longer-term outcomes in depressed outpatients requiring one or several treatment steps: a STAR*D report. Am J Psychiatry 163:1905–1917 [DOI] [PubMed] [Google Scholar]
- Rutz S, Riegert C, Rothmaier AK, Buhot MC, Cassel JC, Jackisch R (2006) Presynaptic serotonergic modulation of 5-HT and acetylcholine release in the hippocampus and the cortex of 5-HT1B-receptor knockout mice. Brain Res Bull 70:81–93 [DOI] [PubMed] [Google Scholar]
- Rygula R, Abumaria N, Flügge G, Fuchs E, Rüther E, Havemann-Reinecke U (2005) Anhedonia and motivational defificits in rats: impact of chronic social stress. Behav Brain Res 162:127–134 [DOI] [PubMed] [Google Scholar]
- Rygula R, Abumaria N, Domenici E, Hiemke C, Fuchs E (2006) Effects of fluoxetine on behavioral deficits evoked by chronic social stress in rats. Behav Brain Res 174:188–192 [DOI] [PubMed] [Google Scholar]
- Sahli ZT, Banerjee P, Tarazi FI (2016) The preclinical and clinical effects of vilazodone for the treatment of major depressive disorder. Expert Opin Drug Discov 11:515–523 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sari Y (2004) Serotonin1B receptors: from protein to physiological function and behavior. Neurosci Biobehav Rev 28:565–582 [DOI] [PubMed] [Google Scholar]
- Saura J, Kettler R, Da Prada M, Richards JG (1992) Quantitative enzyme radioautography with 3H-Ro 41-1049 and 3H-Ro 19-6327 in vitro: localization and abundance of MAO-A and MAO-B in rat CNS, peripheral organs, and human brain. J Neurosci 12:1977–1999 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Savitz J, Lucki I, Drevets WC (2009) 5-HT(1A) receptor function in major depressive disorder. Prog Neurobiol 88:17–31 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schildkraut JJ (1965) The catecholamine hypothesis of affective disorders: a review of supporting evidence. Am J Psychiatry 122:509–522 [DOI] [PubMed] [Google Scholar]
- Schmauss C (2003) Serotonin2C receptors: suicide, serotonin, and runaway RNA. Neuroscientist 9:237–242 [DOI] [PubMed] [Google Scholar]
- Schmitt K, Holsboer-Trachsler E, Eckert A (2016) BDNF in sleep, insomnia, and sleep deprivation. Ann Med 48:42–51 [DOI] [PubMed] [Google Scholar]
- Schulze TG, Buervenich S, Badner JA, Steele CJ, Detera-Wadleigh SD, Dick D, Foroud T, Cox NJ, MacKinnon DF, Potash JB, Berrettini WH, Byerley W, Coryell W, DePaulo JR Jr, Gershon ES, Kelsoe JR, McInnis MG, Murphy DL, Reich T, Scheftner W, Nurnberger JI Jr, McMahon FJ (2004) Loci on chromosomes 6q and 6p interact to increase susceptibility to bipolar affective disorder in the national institute of mental health genetics initiative pedigrees. Biol Psychiatry 56:18–23 [DOI] [PubMed] [Google Scholar]
- Segond N, Degrelle SA, Berndt S, Clouqueur E, Rouault C, Saubamea B, Dessen P, Fong KS, Csiszar K, Badet J, Evain-Brion D, Fournier T (2013) Transcriptome analysis of PPARγ target genes reveals the involvement of lysyl oxidase in human placental cytotrophoblast invasion. PLoS One 8:e79413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shirayama Y, Takahashi M, Osone F, Hara A, Okubo T (2017) Myo-inositol, glutamate, and glutamine in the prefrontal cortex, hippocampus, and amygdala in major depression. Biol Psychiatry Cogn Neurosci Neuroimaging 2:196–204 [DOI] [PubMed] [Google Scholar]
- Shokri-Kojori E, Wang GJ, Wiers CE, Demiral SB, Guo M, Kim SW, Lindgren E, Ramirez V, Zehra A, Freeman C, Miller G, Manza P, Srivastava T, De Santi S, Tomasi D, Benveniste H, Volkow ND (2018) β-Amyloid accumulation in the human brain after one night of sleep deprivation. Proc Natl Acad Sci U S A 115:4483–4488 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Song D, Du T, Li B, Cai L, Gu L, Li H, Chen Y, Hertz L, Peng L (2008) Astrocytic alkalinization by therapeutically relevant lithium concentrations: implications for myo-inositol depletion. Psychopharmacology 200:187–195 [DOI] [PubMed] [Google Scholar]
- Song D, Man Y, Li B, Xu J, Hertz L, Peng L (2013) Comparison between drug-induced and K+-induced changes in molar acid extrusion fluxes (JH+) and in energy consumption rates in astrocytes. Neurochem Res 38:2364–2374 [DOI] [PubMed] [Google Scholar]
- Sotelo C, Cholley B, El Mestikawy S, Gozlan H, Hamon M (1990) Direct immunohistochemical evidence of the existence of 5-HT1A autoreceptors on serotoninergic neurons in the midbrain raphe nuclei. Eur J Neurosci 2:1144–1154 [DOI] [PubMed] [Google Scholar]
- Spector R, Lorenzo AV (1975) The origin of myo-inositol in brain, cerebrospinal fluid and choroid plexus. J Neurochem 25:353–354 [DOI] [PubMed] [Google Scholar]
- Steel Z, Marnane C, Iranpour C, Chey T, Jackson JW, Patel V, Silove D (2014) The global prevalence of common mental disorders: a systematic review and meta-analysis 1980–2013. Int J Epidemiol 43:476–493 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stenovec M, Li B, Verkhratsky A, Zorec R (2020) Astrocytes in rapid ketamine antidepressant action. Neuropharmacology 173:108158. [DOI] [PubMed] [Google Scholar]
- Strong PV, Greenwood BN, Fleshner M (2009) The effects of the selective 5-HT(2C) receptor antagonist SB 242084 on learned helplessness in male Fischer 344 rats. Psychopharmacology 203:665–675 [DOI] [PubMed] [Google Scholar]
- Suárez-Santiago JE, Briones-Aranda A, Espinosa-Raya J, Picazo O (2017) Agonist E-6837 and antagonist SB-271046 of 5-HT6 receptors both reverse the depressive-like effect induced in mice by subchronic ketamine administration. Behav Pharmacol 28:582–585 [DOI] [PubMed] [Google Scholar]
- Sun L, Fang L, Lian B, Xia JJ, Zhou CJ, Wang L, Mao Q, Wang XF, Gong X, Liang ZH, Bai SJ, Liao L, Wu Y, Xie P (2017) Biochemical effects of venlafaxine on astrocytes as revealed by 1H NMR-based metabolic profiling. Mol BioSyst 13:338–349 [DOI] [PubMed] [Google Scholar]
- Szabo ST, Blier P (2001a) Response of the norepinephrine system to antidepressant drugs. CNS Spectr 6:679–684 [DOI] [PubMed] [Google Scholar]
- Szabo ST, Blier P (2001b) Serotonin (1A) receptor ligands act on norepinephrine neuron firing through excitatory amino acid and GABA(A) receptors: a microiontophoretic study in the rat locus coeruleus. Synapse 42:203–212 [DOI] [PubMed] [Google Scholar]
- Szabo ST, Blier P (2002) Effects of serotonin (5-hydroxytryptamine, 5-HT) reuptake inhibition plus 5-HT2A receptor antagonism on the fifiring activity of norepinephrine neurons. J Pharmacol Exp Ther 302:983–991 [DOI] [PubMed] [Google Scholar]
- Taciak PP, Lysenko N, Mazurek AP (2018) Drugs which influence serotonin transporter and serotonergic receptors: pharmacological and clinical properties in the treatment of depression. Pharmacol Rep 70:37–46 [DOI] [PubMed] [Google Scholar]
- Tanaka KF, Samuels BA, Hen R (2012) Serotonin receptor expression along the dorsal-ventral axis of mouse hippocampus. Philos Trans R Soc Lond Ser B Biol Sci 367:2395–2401 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thiebot MH, Martin P (1991) Effects of benzodiazepines, 5-HT1A agonists and 5-HT3 antagonists in animal models sensitive to antidepressant drugs. In: Rodgers RJ, Cooper SJ (eds) 5-HT1A agonists, 5-HT3 antagonists and benzodiazepines: their comparative behavioural pharmacology. Wiley, Chichester, pp 159–194 [Google Scholar]
- Tiger M, Rück C, Forsberg A, Varrone A, Lindefors N, Halldin C, Farde L, Lundberg J (2014) Reduced 5-HT(1B) receptor binding in the dorsal brain stem after cognitive behavioural therapy of major depressive disorder. Psychiatry Res 223:164–170 [DOI] [PubMed] [Google Scholar]
- Tsybko AS, Ilchibaeva TV, Popova NK (2017) Role of glial cell line-derived neurotrophic factor in the pathogenesis and treatment of mood disorders. Rev Neurosci 28:219–233 [DOI] [PubMed] [Google Scholar]
- Vallee A, Lecarpentier Y, Guillevin R, Vallee JN (2017) Interactions between TGFbeta1, canonical WNT/beta-catenin pathway and PPAR gamma in radiationinduced fibrosis. Oncotarget 8:90579–90604 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Varnäs K, Nyberg S, Halldin C, Varrone A, Takano A, Karlsson P, Andersson J, McCarthy D, Smith M, Pierson ME, Söderström J, Farde L (2011) Quantitative analysis of [11C]AZ10419369 binding to 5-HT1B receptors in human brain. J Cereb Blood Flow Metab 31:113–123 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Verkhratsky A, Butt AM (2013) Glial physiology and pathophysiology. Wiley-Blackwell, Chichester [Google Scholar]
- Verkhratsky A, Nedergaard M (2014) Astroglial cradle in the life of the synapse. Philos Trans R Soc Lond Ser B Biol Sci 369:20130595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Verkhratsky A, Nedergaard M (2018) Physiology of astroglia. Physiol Rev 98:239–389 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Verkhratsky A, Parpura V (2010) Recent advances in (patho)physiology of astroglia. Acta Pharmacol Sin 31:1044–1054 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Verkhratsky A, Parpura V (2015) Physiology of astroglia: channels, receptors, transporters, ion signaling and gliotransmission. In: Verkhratsky A, Parpura V (eds) Colloquium series on neuroglia in biology and medicine: from physiology to disease. Morgan & Claypool Publishers, San Rafael, p 172 [Google Scholar]
- Verkhratsky A, Rodríguez JJ, Parpura V (2012) Calcium signalling in astroglia. Mol Cell Endocrinol 353:45–56 [DOI] [PubMed] [Google Scholar]
- Verkhratsky A, Rodriguez JJ, Steardo L (2014) Astrogliopathology: a central element of neuropsychiatric diseases? Neuroscientist 20:576–588 [DOI] [PubMed] [Google Scholar]
- Verkhratsky A, Zorec R, Parpura V (2017) Stratification of astrocytes in healthy and diseased brain. Brain Pathol 27:629–644 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Verkhratsky A, Semyanov A, Zorec R (2020) Physiology of astroglial excitability. Function 1:zqaa016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Verkhratsky A, Augusto-Oliveira M, Pivoriunas A, Popov A, Brazhe A, Semyanov A (2021) Astroglial asthenia and loss of function, rather than reactivity, contribute to the ageing of the brain. Pflugers Arch 473:753–774 [DOI] [PubMed] [Google Scholar]
- Wallace A, Pehrson AL, Sánchez C, Morilak DA (2014) Vortioxetine restores reversal learning impaired by 5-HT depletion or chronic intermittent cold stress in rats. Int J Neuropsychopharmacol 17:1695–1706 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang J, Qiao J, Zhang Y, Wang H, Zhu S, Zhang H, Hartle K, Guo H, Guo W, He J, Kong J, Huang Q, Li XM (2014) Desvenlafaxine prevents white matter injury and improves the decreased phosphorylation of the rate-limiting enzyme of cholesterol synthesis in a chronic mouse model of depression. J Neurochem 131:229–238 [DOI] [PubMed] [Google Scholar]
- Wang L, Zhang Y, Du X, Ding T, Gong W, Liu F (2019) Review of antidepressants in clinic and active ingredients of traditional Chinese medicine targeting 5-HT1A receptors. Biomed Pharmacother 120:109408. [DOI] [PubMed] [Google Scholar]
- Watanabe N, Omori IM, Nakagawa A, Cipriani A, Barbui C, Churchill R, Furukawa TA (2011) Mirtazapine versus other antidepressive agents for depression. Cochrane Database Syst Rev 7:CD006528. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wolfson M, Hertz E, Belmaker RH, Hertz L (1998) Chronic treatment with lithium and pretreatment with excess inositol reduce inositol pool size in astrocytes by different mechanisms. Brain Res 787:34–40 [DOI] [PubMed] [Google Scholar]
- Wolfson M, Bersudsky Y, Zinger E, Simkin M, Belmaker RH, Hertz L (2000) Chronic treatment of human astrocytoma cells with lithium, carbamazepine or valproic acid decreases inositol uptake at high inositol concentrations but increases it at low inositol concentrations. Brain Res 855:158–161 [DOI] [PubMed] [Google Scholar]
- Wong YH, Kalmbach SJ, Hartman BK, Sherman WR (1987) Immunohistochemical staining and enzyme activity measurements show myo-inositol-1-phosphate synthase to be localized in the vasculature of brain. J Neurochem 48:1434–1442 [DOI] [PubMed] [Google Scholar]
- Wu C, Singh SK, Dias P et al. (1999) Activated astrocytes display increased 5-HT2a receptor expression in pathological states. Exp Neurol 158:529–533 [DOI] [PubMed] [Google Scholar]
- Xia M, Yang L, Sun G, Qi S, Li B (2017) Mechanism of depression as a risk factor in the development of Alzheimer’s disease: the function of AQP4 and the glymphatic system. Psychopharmacology (Berlin) 234:365–379 [DOI] [PubMed] [Google Scholar]
- Xia M, Li X, Yang L, Ren J, Sun G, Qi S, Verkhratsky A, Li B (2018) The ameliorative effect of fluoxetine on neuroinflammation induced by sleep deprivation. J Neurochem 146:63–75 [DOI] [PubMed] [Google Scholar]
- Xia M, Li Z, Li S, Liang S, Li X, Chen B, Zhang M, Dong C, Verkhratsky A, Guan D, Li B (2020) Sleep deprivation selectively Down-regulates astrocytic 5-HT2B receptors and triggers depressive-like behaviors via stimulating P2X7 receptors in mice. Neurosci Bull 36:1259–1270 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xie L, Kang H, Xu Q, Chen MJ, Liao Y, Thiyagarajan M, O’Donnell J, Christensen DJ, Nicholson C, Iliff JJ, Takano T, Deane R, Nedergaard M (2013) Sleep drives metabolite clearance from the adult brain. Science 342:373–377 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu Z, Jing X, Li G, Sun J, Guo H, Hu Y, Sun F, Wen X, Chen F, Wang T, Lu XP (2019) Valproate decreases vitamin D levels in pediatric patients with epilepsy. Seizure 71:60–65 [DOI] [PubMed] [Google Scholar]
- Yamauchi M, Tatebayashi T, Nagase K, Kojima M, Imanishi T (2004) Chronic treatment with fluvoxamine desensitizes 5-HT2C receptor-mediated hypolocomotion in rats. Pharmacol Biochem Behav 78:683–689 [DOI] [PubMed] [Google Scholar]
- Yu W, Greenberg ML (2016) Inositol depletion, GSK3 inhibition and bipolar disorder. Future Neurol 11:135–148 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu C, Lyu J, Chen Y, Guo Y, Paul S, Bian Z, Zhou H, Tan Y, Chen J, Chen Z, Li L (2015) Epidemiology of major depressive episodes among Chinese adults aged 30-79 years: data from the China Kadoorie Biobank. Zhonghua Liu Xing Bing Xue Za Zhi 36:52–56 [PubMed] [Google Scholar]
- Yue T, Li B, Gu L, Huang J, Verkhratsky A, Peng L (2019) Ammonium induced dysfunction of 5-HT2B receptor in astrocytes. Neurochem Int 129:104479. [DOI] [PubMed] [Google Scholar]
- Zarate CA, Manji HK (2008) Riluzole in psychiatry: a systematic review of the literature. Expert Opin Drug Metab Toxicol 4:1223–1234 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zeidán-Chuliá F, Salmina AB, Malinovskaya NA, Noda M, Verkhratsky A, Moreira JC (2014) The glial perspective of autism spectrum disorders. Neurosci Biobehav Rev 38:160–172 [DOI] [PubMed] [Google Scholar]
- Zhang S, Li B, Lovatt D, Xu J, Song D, Goldman SA, Nedergaard M, Hertz L, Peng L (2010) 5-HT2B receptors are expressed on astrocytes from brain and in culture and are a chronic target for all five conventional ‘serotonin-specific reuptake inhibitors’. Neuron Glia Biol 6:113–125 [DOI] [PubMed] [Google Scholar]
- Zhang M, Zhou D, Wang Y, Maier DL, Widzowski DV, Sobotka-Briner CD, Brockel BJ, Potts WM, Shenvi AB, Bernstein PR, Pierson ME (2011) Preclinical pharmacology and pharmacokinetics of AZD3783, a selective 5-hydroxytryptamine 1B receptor antagonist. J Pharmacol Exp Ther 339:567–578 [DOI] [PubMed] [Google Scholar]
- Zhang X, Song D, Gu L, Ren Y, Verkhratsky A, Peng L (2015) Decrease of gene expression of astrocytic 5-HT2B receptors parallels development of depressive phenotype in a mouse model of Parkinson’s disease. Front Cell Neurosci 9:388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhong XM, Dong M, Wang F, Zhang Q, Ungvari GS, Ng CH, Chiu HFK, Si TM, Sim K, Avasthi A, Grover S, Chong MY, Chee KY, Kanba S, Lee MS, Yang SY, Udomratn P, Kallivayalil RA, Tanra AJ, Maramis MM, Shen WW, Sartorius N, Mahendran R, Tan CH, Shinfuku N, Xiang YT (2018) Physical comorbidities in older adults receiving antidepressants in Asia. Psychogeriatrics 18:351–356 [DOI] [PubMed] [Google Scholar]
- Zielinski MR, Gerashchenko D, Karpova SA, Konanki V, McCarley RW, Sutterwala FS, Strecker RE, Basheer R (2017) The NLRP3 inflammasome modulates sleep and NREM sleep delta power induced by spontaneous wakefulness, sleep deprivation and lipopolysaccharide. Brain Behav Immun 62:137–150 [DOI] [PMC free article] [PubMed] [Google Scholar]