Abstract
Background
Specialist medical practitioners have conducted clinics in primary care and rural hospital settings for a variety of reasons in many different countries. Such clinics have been regarded as an important policy option for increasing the accessibility and effectiveness of specialist services and their integration with primary care services.
Objectives
To undertake a descriptive overview of studies of specialist outreach clinics and to assess the effectiveness of specialist outreach clinics on access, quality, health outcomes, patient satisfaction, use of services, and costs.
Search methods
We searched the Cochrane Effective Practice and Organisation of Care (EPOC) specialised register (March 2002), the Cochrane Controlled Trials Register (CCTR) (Cochrane Library Issue 1, 2002), MEDLINE (including HealthStar) (1966 to May 2002), EMBASE (1988 to March 2002), CINAHL (1982 to March 2002), the Primary‐Secondary Care Database previously maintained by the Centre for Primary Care Research in the Department of General Practice at the University of Manchester, a collection of studies from the UK collated in "Specialist Outreach Clinics in General Practice" (Roland 1998), and the reference lists of all retrieved articles.
Selection criteria
Randomised trials, controlled before and after studies and interrupted time series analyses of visiting specialist outreach clinics in primary care or rural hospital settings, either providing simple consultations or as part of complex multifaceted interventions. The participants were patients, specialists, and primary care providers. The outcomes included objective measures of access, quality, health outcomes, satisfaction, service use, and cost.
Data collection and analysis
Four reviewers working in pairs independently extracted data and assessed study quality.
Main results
73 outreach interventions were identified covering many specialties, countries and settings. Nine studies met the inclusion criteria. Most comparative studies came from urban non‐disadvantaged populations in developed countries. Simple 'shifted outpatients' styles of specialist outreach were shown to improve access, but there was no evidence of impact on health outcomes. Specialist outreach as part of more complex multifaceted interventions involving collaboration with primary care, education or other services wasassociated with improved health outcomes, more efficient and guideline‐consistent care, and less use of inpatient services. The additional costs of outreach may be balanced by improved health outcomes.
Authors' conclusions
This review supports the hypothesis that specialist outreach can improve access, outcomes and service use, especially when delivered as part of a multifaceted intervention. The benefits of simple outreach models in urban non‐disadvantaged settings seem small. There is a need for good comparative studies of outreach in rural and disadvantaged settings where outreach may confer most benefit to access and health outcomes.
Plain language summary
Specialist outreach clinics in primary care and rural hospital settings may improve access to care, quality of care, health outcomes, patient satisfaction and use of hospital services. They may also be more costly.
This review examines the benefits and costs of outreach in a range of specialties and in a variety of settings. Simple 'shifted outpatients' styles of specialist outreach were shown to improve access, but there was no evidence of their impact on health outcomes. Outreach as part of more complex multifaceted interventions involving primary care collaborations, education and other services was associated with improved health outcomes, more efficient and guideline‐consistent care, and less use of inpatient services. There is a need for better quality evidence evaluating specialist outreach in all settings, but especially in rural and disadvantaged populations.
Background
Specialist medical practitioners usually consult in outpatient areas of major hospital facilities or large metropolitan clinics. In some places, visiting specialist services, otherwise known as 'specialist outreach', have been established to improve access to specialist care, enhance primary‐specialist care relationships, reduce pressures on hospitals, shift the balance of care to community‐based services, or reduce health service costs. Specialist outreach has emerged as specific policy initiatives, as initiatives of individual practitioners or organisations, and as a secondary effect of other policies. General practice fundholding in the UK during the 1990s is an example of the latter, whereby many general practitioners (GPs) "purchased" visiting specialist services from hospital trusts.
Three main types of settings are apparent: (1) specialist clinics in urban primary care settings instead of hospital outpatient departments; (2) specialist clinics in rural community primary health centres or hospitals where there is no resident specialist; and (3) sub‐specialist clinics in major regional centres where there is only a resident 'general' specialist service (e.g. general physician, general surgeon). Accordingly outreach serves a range of urban, rural and remote populations of varying degrees of health, healthcare and socioeconomic disdadvantage.
It is not surprising, given the variety of objectives and settings, that "specialist outreach" is therefore a blanket term that covers a heterogenous group of activities. British psychiatrists were the first to identify four main models of working with primary care doctors and clinics: the "shifted outpatients" model, where outreach provides similar consultations, investigations and procedures to those provided in hospital clinics; the "replacement" model, where specialists replace the general practitioner as the doctor of first contact; the "consultation" model, where the relationship between specialist and primary care is enhanced, but most patient care is mediated through the primary carer; and the "liaison attachment" model, where the specialist is part of a team of visiting services.(Williams 1981;Strathdee 1984;Mitchell 1985;Tyrer 1990) To a greater or lesser degree these styles are employed in other specialties, and they are often combined.
The aims and potential benefits of outreach services over hospital clinics relate to improved access to specialists and hospital‐based services, improved liaison between specialists and primary carers, and the benefits of consultations in primary care settings, such as familiarity and less stigma for patients and fewer distractions for providers. The potential costs relate to additional costs of service provision, the inconvenience for traveling specialists, and opportunity costs associated with taking specialists out of their main practice and with further investment in specialist medical services as opposed to other sectors.
Studies of outreach have been conducted in the UK where the 'shifted outpatients' model has predominated. A systematic review of these studies, which included only one RCT and one other study that controlled for case‐mix, concluded that outreach leads to improved communication between GPs and specialists, improved patients' experiences and better access, but at greater cost and with less efficient use of specialists' time (Powell). In the UK the decision to provide specialist outreach services has been summarised as one of whether the added benefits of convenience to patients are worth the extra cost. There has been little analysis of the quality of care compared with hospital clinics, nor of the implications for equity of access.
In countries where specialists are less accessible and where outreach may be difficult to establish and maintain, costs and benefits may be different from those in the UK. Compared to mainstream urban populations, people living in rural and remote areas, and some city‐dwelling groups such as the homeless or mentally‐ill, tend to have worse health and worse access to medical care. Planning specialist outreach services requires detailed knowledge of the targeted population, the gaps in existing resources and the potential contribution of specialist medical practitioners. In order to facilitate such planning, a systematic review is needed to provide information about the relative benefits and costs of specialist outreach delivered through different modes and in the full range of settings in which it has been employed, and to identify what further research is necessary.
Objectives
1. To undertake a descriptive overview of all studies of specialist outreach clinics. 2. To undertake a systematic review of rigorous evaluations of specialist outreach clinics in primary care and rural hospital settings to estimate their effects on: i. Access to specialist care ii. Quality and appropriateness of care iii. Health outcomes iv. Patient and provider satisfaction v. Use of services vi. Costs.
3. To assess the influence of different contexts and styles of service delivery on these outcomes.
Methods
Criteria for considering studies for this review
Types of studies
Descriptive overview of all studies of specialist outreach clinics: Any comparative or descriptive study.
Systematic review: Randomised controlled trials (RCTs), controlled clinical trials (CCTs), controlled before and after studies (CBAs), and interrupted time series analyses (ITSs).
Types of participants
1. Patients who are eligible for specialist care (in some countries a referral from primary care practitioners, such as general practitioners, nurses or health workers, is required); 2. Primary health care practitioners; and 3. Specialists.
Types of interventions
Specialist outreach clinics: defined as planned and regular visits by specialist‐trained medical practitioners from a usual practice location (hospital or specialist center) to primary care or rural hospital settings. Studies of domiciliary (home) visits, and studies where the specialist's main practice location is in a primary care setting were excluded. Only outreach visits that involved patient consultations were included. The intervention was characterised as "simple" if specialist consultations only were provided in a primary care or remote hospital setting, and "multifaceted" if outreach involved joint consultations, education sessions or seminars for primary caregivers, or enhanced patient care of which outreach was a component.
Types of outcome measures
A wide variety of outcome measures have been studied and reported. Only objective and standardised measures have been included in this review. Outcome categories include: 1. Access (perceived by patients; measured access determinants; realised access (total attendance and attendance rates)); 2. Quality of care (guideline‐consistent referral and treatment; adherence to treatment); 3. Health outcomes; 4. Patient and provider satisfaction; 5. Use of hospital and primary care services (investigations; consultations; inpatient and outpatient treatment); and 6. Costs (per patient costs, cost‐effectiveness, opportunity costs and harms).
Measures of health outcomes, and in their absence, measures of access to care and quality of care, are regarded as primary outcomes for the purpose of this review. Patient satisfaction is very important but will, at least in part, be determined by access, quality and clinical outcome factors, and is often measured using non‐standardised scales. Independent of clinical outcomes, the use of health services and costs vary considerably with local factors and are of relatively little assistance to policy‐makers. Cost‐effectiveness, however, is regarded as an important outcome.
Search methods for identification of studies
A. The following electronic databases were searched:
Cochrane Effective Practice and Organisation of Care (EPOC) specialised register (March 2002), the Cochrane Controlled Trials Register (CCTR) (Cochrane Library Issue 1, 2002), MEDLINE (including HealthStar) (1966 to May 2002), EMBASE (1988 to March 2002) and CINAHL (1982 to March 2002).
The search strategy included a combination of textword and index terms relating to community care, primary care, rural health services and outreach clinics. The MEDLINE search strategy is shown below, and was adapted for the other databases:
1. Health services accessibility/ 2. exp Ambulatory care facilities/ 3. Community health services/ 4. Primary health care/ 5. Family practice/ 6. Rural population/ 7. Hospitals, rural/ 8. Rural health services/ 9. remote.tw 10. rural.tw 11. "referral and consultation"/ 12. or/1‐11 13. "Allergy and Immunology"/ 14. Anesthesiology/ 15. Dermatology/ 16. exp Internal Medicine/ 17. Neurology/ 18. exp Pediatrics/ 19. exp Physical Medicine/ 20. exp Psychiatry/ 21. exp Radiology/ 22. Reproductive Medicine/ 23. Venereology/ 24. exp Specialties, Surgical/ 25. or/13‐24 26. 12 and 25 27. 26 and (outreach or visit$).tw 28. ((special$ or consultant?) adj3 (outreach or visit$)).tw 29. (outreach adj3 (clinic? or service? or consultation?)).tw 30. (visiting adj3 (service? or clinic? or consultation?)).tw 31. or/27‐30
B. The Primary‐Secondary Care Database ‐ an inactive specialist bibliographic database previously maintained by the Centre for Primary Care Research in the Department of General Practice, University of Manchester, containing published research relating to the interface between primary and secondary care up to the early 1990s.
C. "Specialist Outreach Clinics in General Practice", a collection of studies from the UK (Roland 1998) D. The reference list of each retrieved article was scanned to identify further studies.
Data collection and analysis
This review was conducted in accordance with EPOC guidelines.
Methods of screening and obtaining Studies
An initial search was based on titles and abstracts, and relevant articles obtained. The titles cited in the reference lists of obtained articles were examined to identify further studies for retrieval. Editorials, commentaries, other reviews, feasibility studies and theoretical analyses were excluded. Descriptive studies, with no comparison group, were included in the desriptive overview of interventions, but were excluded from further analyses. Studies reporting a comparison group (concurrent or otherwise) were entered for review. Reviewers worked in three pairs (the contact reviewer was common to each pair) each evaluating one third of comparative studies. In each pair, both reviewers independently assessed the studies for inclusion in the EPOC component of the review, as well as the quality and findings of the study. Any discrepancies between reviewers were resolved through discussion.
Data Abstraction
In each study, the setting was characterized by: 1. the country; and 2. the population served, characterized as one of four possibilities: urban (outreach to a major population centre); urban disadvantaged (outreach to an explicitly‐defined disadvantaged population in a major population centre); rural (outreach to rural centres without resident specialists, where patients must otherwise travel to another population centre to obtain specialist care); or rural disadvantaged (outreach to rural centres where it is clearly stated that geographic and other types of access barriers are likely to prevent many patients from otherwise obtaining specialist care).
In each study, the intervention was characterized by: 1. the specialty(such as medicine, surgery, otolaryngology, ophthalmology, paediatrics, psychiatry) and any specific disease being targeted, 2. the nature of the intervention. For the purposes of this review we have regarded the shifted outpatients model as "simple outreach", and outreach that is enhanced by increased collaboration with primary care practitioners, joint consultations, case‐conferences, seminars or other education sessions, or is part of a broader intervention involving other personnel and services, we refer to as "multifaceted outreach'".
Each reported outcome was allocated to one specific category of outcome measure as described above. Each outcome measure was characterized as showing improvement, worsening, a mixed picture, or no evidence of improvement when compared with hospital‐based services.
Data abstraction and quality assessment from studies in the systematic review of rigorous evaluations was facilitated by a modified version of the EPOC Data Extraction Checklist. EPOC quality criteria for RCTs involved consideration of unit of allocation and analysis, concealment of allocation, follow up rates, blinding, comparability of groups at baseline, reliability of outcome assessment and protection against contamination. For inclusion in the review, CBAs had to have contemporaneous data collection and use appropriate control groups. Quality criteria included comparability of the characteristics of intervention and control groups, similarity of baseline measurements in intervention and control groups, blind assessment of primary outcome, protection against contamination, follow up rates and reliability of outcome assessment. For inclusion in the review, ITSs had to include an intervention delivered at a defined point in time and report three or more data points before and after the intervention. Quality criteria for ITSs was based on the EPOC guidelines (Bero 2000) and recently proposed enhanced criteria (Ramsay) which together considered protection against secular changes, an explicit rationale for the number and spacing of data points, pre‐specification of the shape of intervention effect, formal test for trend preferably using appropriate time‐series techniques, data collection uncontaminated by the intervention, data identical before and after the intervention, blind assessment of outcome, reliable outcomes, and completeness of the data set.
Follow‐up rates varied at different times and for different outcomes in some studies. Where an intention to treat analysis was not done, the denominators at the relevant time periods were used to calculate results. The denominators in some of the tables may therefore vary within individual studies.
In accordance with EPOC guidelines, where possible results were presented in terms of: (1) Absolute difference (mean or proportion of clinical behaviour in intervention/experimental group minus control); (2) Relative percentage difference (absolute difference divided by post‐intervention score in the control group); (3) Absolute change from baseline (pre to post changes in both groups); and (4) Difference in absolute change from baseline. In studies without baseline data, only absolute difference and relative percentage difference were calculated.
In order to give a visual representation of the results, forest plots of the outcome categories were presented. This required conversion of data to follow the convention that positive or desirable effects of the intervention are indicated to the left of the null‐effect line. Studies with unit of analysis error were not included in forest plots. Only studies that were similar in terms of setting, intervention and outcome assessment were subjected to statistical meta‐analysis. In such cases, summary statistics, using a fixed‐effects model, are presented in the text. Extreme heterogeneity, however, was expected to exist among the results of most included studies due in part to their different populations, problems addressed and services delivered, and in such cases meta‐analysis was not performed.
Data abstraction from comparative and descriptive studies not meeting EPOC inclusion criteria was limited to characteristics of the setting, intervention and results, without further assessment of the design or methods. They are summarised in the "Description of Studies", and comment on the excluded comparative studies is made separately in each section.
Results
Description of studies
1. Descriptive overview of all studies of specialist outreach
137 publications relating to specialist outreach were found, as shown in Figure 1. In the order the search was conducted, none were identified from the Cochrane Controlled Trials Register, two came from the EPOC register, 61 from Medline, ten from EMBASE and CINAHL, 50 from article references and 14 from other sources known to the reviewers.
1.
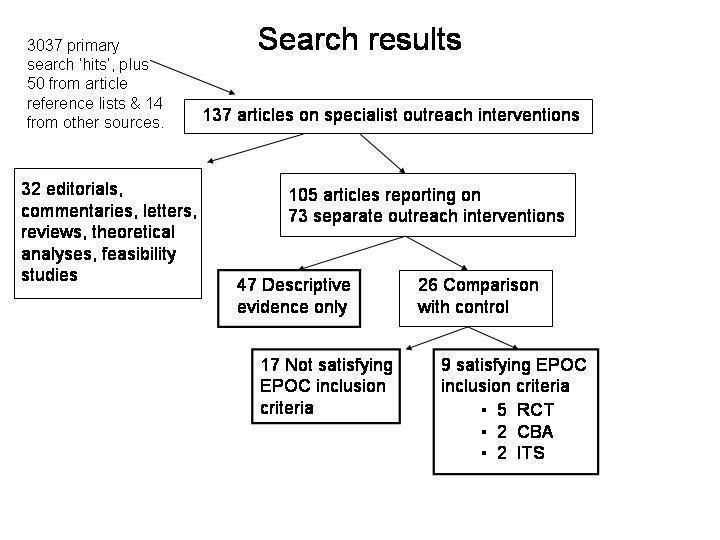
Search results
Twenty‐five were editorials, commentaries or letters (Creed 1989; Denham 1995; Dunbar 1994; Gibson 1966; Gillam 2001; Gish; Golele 1994; Hanlon 1989; Harris 1994; Jurkiewicz 1989; London 1995; McCormick 1923; Mitchell 1983; Mitchell 1985; Mitchell 1990; Ollivere 2001; Petchey 1998; Raasen 1998; Robb 1995; Robinson 2001; Sprod 2001; Swash 1993; Tod 1993; Tracy 1996; White 2001), three were feasibility studies/surveys for specialist outreach (Brown;Musk; Rees), two were theoretical models (Bergmo, Shah) and two were collective reviews (Balestrieri;Stolee), all of which were excluded from the review and which are listed in the Additional References. The reviewers were aware of several Australian government reports (e.g. AMWAC; Taylor) that provided overviews of some outreach activities, but these were not included because they were not primary studies, did not contain the results of evaluative studies using comparative methods, and the search strategy did not encompass government reports that were not otherwise referenced in the medical and health services literature. An additional systematic review limited to UK studies that was published after the search was conducted is also included in Additional References (Powell).
The remaining 105 articles, when grouped, reported findings from 73 outreach interventions summarised in Table 1 and represented in Figure 2. Twenty‐eight were from the UK, twelve from Australia, eleven from the USA, seven from Canada, four from South Africa, three from East Africa (Kenya, Tanzania and Uganda), three from Israel, and one each from Zimbabwe, Holland, Norway, Ecuador and Hong Kong.
1. List of all specialist outreach interventions.
| Label | Year | Country | Specialty | Setting (U,UD,R,RD) | Outreach type (S,M) | Evidence (E,C,D) | Setting description | Outreach description |
| Almehdi | 2000 | Australia | general surgery | RD | S | D | 3 Indigenous communities in far north‐west Queensland | 6‐8 weekly clinic by specialists from Mt Isa Hospital for consultations and minor procedures. |
| Ayshford | 1995‐1998 | UK | ENT | U | S | D | 2 GP surgeries ‐ one in a large city, one in a small city. | consultant clinics |
| Bailey | 1993 | UK | psychiatry, general medicine, general surgery | U | S | D | 96 consultants and 88 GPs in England & Wales | survey of outreach activity |
| Baker | 1998‐2000 | Australia | obstetrics & gynaecology | RD | S | D | 24 towns in 1 million square kilometres of central Queensland. | regular visits by aircraft with consultant, anaesthetist and pilot, with laparoscopy, ultrasound and anaesthetic and surgical equipment, for operating and consulting. |
| Baskett* | 1971‐1977 | Canada | obstetrics & gynaecology | RD | M | D | Health center in Churchill on west side of Hudson Bay, 600 miles north of Winnipeg | 3 consultants from Winnipeg sharing monthly visits for operating, consulting and in‐service education for local medical & nursing staff. |
| Baumann | 1978 | USA | neurology | R | S | D | 16 Appalachian counties in eastern Kentucky 60‐175 miles from regional center Lexington. | team of neurologist, technician and students spend 3 days per month traveling, visiting each clinic 5 times per year. |
| Black | 1994‐1995 | UK | orthopaedics, dermatology | U | S | C | 3 districts of England | weekly clinics in general practice |
| Blair | 1994‐1995 | UK | paediatrics | U | S | D | Various settings in Nottingham, with population of 125,000 children under 15 years of age. | community pediatrician clinics in schools, social service family centers, teenage clinics, residential children's homes and child development center. |
| Bowling | 1995‐1997 | UK | cardiology, ENT, internal medicine, general surgery, gynaecology, paediatrics, rheumatology | U | S | C | 14 hospital trust areas across England | average 3 hours every 4 weeks in any particular general practice |
| Bruusgaard | 1975 | Norway | paediatrics, internal medicine, gynaecology, ophthalmology, orthodontics | R | S | D | 4 communities with total population 13,000 in southern Norway 65 miles from nearest hospital. | 60 specialist visits over 2.5 years for examinations and consulting. |
| Buck | 1984‐1985 | USA | ophthalmology | R | S | D | rural areas of Arizona | visiting specialists to consult and provide day surgery. |
| Buhrich | 1988‐1992 | Australia | psychiatry (schizophrenia) | UD | M | C | 4 inner city refuges for the homeless in Sydney, Australia. | Weekly evening clinics by consultant & registrar with 2 other MHWs. Assertive case management intervention included medication supervision, counselling, regular review and access to social services. |
| Carr | 1989‐1991 | Australia | psychiatry | U | M | D | 4 primary care group practices in Newcastle. | half day clinic per week in each practice by a psychiatric registrar working in collaboration with general practitioner. |
| Chelala | 1994‐1996 | Ecuador | general surgery | R | S | D | small rural hospitals and health centres. | mobile surgical unit including surgeons, anaesthetists, nurses and support staff from Quito in a truck that visits health centres for 2‐3 days. |
| Coetzee | 1994‐1996 | South Africa | haematology | RD | S | D | Free State and Northern Cape, covering 40% area of South Africa but only 10% population. Travel by train up to 300km. | Fortnightly to monthly outreach visits from tertiary centre to 3 regional hospitals to consult with patients |
| Cotton | 1990‐1995 | Zimbabwe | general surgery | RD | S | D | 10 hospitals up to 316 km from Bulawayo | 117 fortnightly visits by aircraft over 5 years 1990‐5, for operating first day and consulting the second. |
| Crosier | 1992 | South Africa | orthopaedics | RD | S | D | Western Cape | Monthly orthopaedic clinics in 11 centres involving consultant, registrar & allied health. |
| Cybulska | 1997 | UK | genitourinary medicine | U | M | D | variety of urban street settings in London | variety of clinics and contact tracing in a variety of street and inner‐city settings, usually in conjunction with other social services. |
| Dart | 1984 | UK | ophthalmology | U | S | D | A community health centre in London | Consultant clinics on three half days per week. |
| Desch | 1987‐1992 | USA | oncology | R | M | C | Two 80‐110 bed non‐profit community hospitals each serving 50,000 people in rural Virginia, 80‐90 miles from the closest tertiary hospital. | Weekly visits by oncologist and nurse specialist to develop & implement care plan with local physicians. |
| Feild | 1981‐1988 | USA | paediatrics | R | S | D | 9 Arkansas communities outside Little Rock. | 19 faculty paediatricians alternate, traveling with child psychologist and speech therapist. |
| Gillam | 1992‐1993 | UK | ophthalmology | U | S | C | 17 general practices in London in 1992‐1993. | monthly clinics by consultant and nurse. |
| Gruen | 1997‐1999 | Australia | general surgery, obstetrics & gynaecology, ophthalmology, ENT | RD | S | E | Remote Aboriginal community clinics in Australia's Northern Territory | Regular specialist visits by air/road for consulting and procedures |
| Hansom | 1979 | Canada | paediatrics | R | S | D | clinics and hospitals in Nova Scotia | Traveling clinics in vans and specialist as consultant to smaller hospitals |
| Haynes | 2000 | UK | ophthalmology (cataract) | R | S | C | community hospital with no inpatient facilities in Norfolk, 40km from main district hospital. | consultant visits lasting a day to operate and consult. |
| Helliwell | 1993‐1994 | UK | rheumatology | U | S | C | general practice clinic | weekly clinic at one of 3 general practices or a cottage hospital |
| Hess | 1997‐1998 | Canada | palliative care | U | M | D | acute care and hospice settings in Ottawa | palliative care physician & multidisciplinary team (clinical, support, allied health, social services) sees patients when referred 24 hours per day in the clinic or home. |
| Hindler | 1995 | UK | psychiatry | UD | S | D | 2 community psychiatric outpatient clinics in a deprived inner city area of London | consultant clinic in community health center |
| Ho | 1994‐1995 | Hong Kong | child psychiatry | U | S | C | a community child mental health clinic | 3 sessions every two weeks by visiting psychiatrists from the main hospital. |
| Hodges | 2000 | Uganda | plastics & reconstructive surgery (cleft lip/palate) | RD | S | D | 20 rural and remote hospitals | Team of surgeon and anaesthetist bringing all surgical equipment by road. |
| Howe | 1988‐1992 | USA | oncology (breast cancer) | R | S | E | 9 rural hospitals with resident general physicians and surgeons. | regular visits to consult and administer treatment protocols. |
| Humphrey | 1991‐1992 | Australia | obstetrics & gynaecology | RD | S | D | 1759 patients in 14 small towns and Aboriginal communities in far‐north Queensland | 141 visit days by specialist from Cairns Base Hospital via car or aircraft for consultations, including mobile ultrasonography and colposcopy. |
| Hunter | 1978‐1980 | UK | psychiatry | U | S | D | 3 general practices in the Aberdeen area. | fortnightly consultant clinics |
| Kates | 1995‐1996? | Canada | psychiatry | U | M | C | family physicians' offices in Hamilton, Ontario. | nine psychiatrists sharing visits to each practice for half a day every 1 to 4 weeks (equivalent of 2 full time consultants) for joint specialist‐GP inititial patient consultations and others as required. |
| Katon1 | 1992? | USA | psychiatry (depression) | U | M | E | large urban HMO primary clinic with 22 family physicians | 2 or more spec consultations in primary care clinic during weeks 2 & 4 as part of collaborative care program including physician education, regular case conferences, patient education booklets and videotapes, and individualised treatment algorithms. |
| Katon2 | 1997? | USA | psychiatry (persistent major depression) | U | M | E | 4 large HMOs | As above, for major depression meeting diagnosis of major depression 6 to 8 weeks after therapy |
| Kerrison | 1995 | UK | general surgery, gynaecology, orthopaedics, ENT, dermatology, urology, psychiatry, general medicine, ophthalmology, rheumatology, diabetology, cardiology, paediatrics, vasectomy | U | S | D | South East Thames region 1995 | presence of any outreach |
| Kierath | 1995‐1997 | Australia | general surgery | RD | S | D | 7 major rural towns in Western Australia, each with population over 20,000, and up to 1000 miles from Perth. | a surgeon, a medical student and a coordinator spend 2 days per month on visits for consultations and operations. |
| Klein | 1994‐1995 | South Africa | cardiothoracic surgery | RD | S | D | 2 rural 400‐bed hospitals with no ICU and basic perioperative facilities, general nursing and medical staff. | Monthly visits by coardiothoracic team (consultant, 3 registrars, 2 medical officers, 6 nurses, perfusionist & all equipment) for operating and consulting. |
| Leaming | 1965‐1968 | Australia | general surgery | RD | S | D | 20 rural hospitals in inland Queensland, Australia covering 70,000 people over area of 200,000 sq miles. | Flying surgeon team of surgeon, anaesthetist & pilot visiting each hospital once per month for consulting and operating. |
| Little | 1992 | UK | ophthalmology (screening) | UD | S | D | Inner city London | Clinics by consultant and nurse in 3 general practices. |
| Loefler | 1975‐1985 | Kenya | general surgery | RD | S | D | 9 hospitals in rural Kenya and Uganda | 266 visits by aircraft over a 10‐year period for operating, consulting and teaching. |
| Marsh | 1967‐1968 | UK | paediatrics | U | M | D | one group practice centre in England. | joint consultations between consultant and GP |
| Martin* | 1993‐1994 | Canada | gynaecology (cervical dysplasia) | RD | S | D | Regional center (Churchill) serving 7 Canadian Central Arctic communities on the west side of Hudson Bay with total population 5292, 89% Inuit. | 2‐monthly visits by consultant from Winnipeg 350km south. |
| McKechnie | 1975‐1977 | UK | psychiatry | U | M | C | 2 GP practices in separate towns in West Lothian district | fortnightly consultant clinic, with weekly psychologist clinic visits and social work home visits, weekly discussion of cases, and 1 hour for liaison with GPs. |
| Nakar | 1997? | Israel | cardiology, nephrology, internal medicine, psychiatry | U | S | D | 38 primary care practices (180,000 patients) in one district. | outreach clinics with or without family physician present. |
| Oboler | 1980‐1981 | USA | Internal medicine | R | S | C | Area of 56,000 sq miles in eastern Colorado where patients live average 105 miles from Denver. | 30‐foot van equipped as an internist's office making weekly visits to 4 cities with internist, technician & coordinator. |
| O'Brien | 1999? | UK | orthodontics | U | S | E | 3 urban community health centres 0.5‐15km from main base in Manchester | Orthodontic triage consultation in community clinic. |
| Owen | 1997? | Australia | psychiatry | R | M | C | 6 Towns in rural New South Wales. | 10 psychiatrist and mental health worker visits by aircraft from Sydney. Direct care, peer support for local staff, and education to health staff. |
| Parish | 1982‐1998 | Australia | ophthalmology | RD | S | D | Remote Indigenous communities in Australia's Northern Territory. Representative of several reports of similar ophthalmology outreach in other Australian states. | infrequent community visits for consulting and arranging procedures |
| Payne | 1984‐1990 | South Africa | anaesthetics | R | S | D | 13 provincial hospitals in the Western Cape | visits by surgical team. |
| Perrett | 1994‐1995 | UK | gynaecology, orthopaedics, general surgery | U | S | C | 37 primary care clinics in Sheffield in 1994‐5. | outreach clinics in primary care. |
| Pullen | 1956‐1988 | UK | psychiatry | U | S | D | primary care and rural areas of Scotland | clinics in primary care settings |
| Raasen | 1994‐1995 | Kenya, Tanzania | general surgery | RD | S | D | mission and government hospitals in rural areas of Kenya & Tanzania. | Flying surgical service (AMREF) from Nairobi for 16.5 to 31 days of surgical work over 5 years. |
| Riley | 1995? | UK | gynaecology, orthopaedics, urology | U | S | C | one GP practice in Worcestorshire | outreach clinics in primary care |
| Roy‐Byrne | 1999? | USA | psychiatry (panic disorder) | U | M | E | 3 urban primary care clinics in Seattle – 2 university‐associated with 30 physicians & 6000‐8000 patients and 1 community family clinic with 8 physicians & 10000 patients | 2 consultations with psychiatrist in 1st 8 weeks, as part of collaborative care involving physician education, patient education with videotapes, and structured telephone liaison for medication adjustment and provider feedback. |
| Seidel | 1991? | Australia | geriatric psychiatry | U | M | D | Residential facilities in one half of the city of Adelaide. | Visits by psychiatrist and psychiatric nurse prompted by referral from GP. |
| Sewell | 1983 | Australia | paediatrics | UD | M | D | 11 community agencies (health centres, relief centres, schools) up to 5 km from Royal Childrens' Hospital | Weekly, fortnightly and monthly half‐day visits by consultants, registrars and resident medical officers to provide direct care, clinical & educational support, & community‐oriented training for specialists. |
| Shanon | 1992‐1993 | Israel | paediatrics | U | S | D | medical community centre which does not provide primary care services in a suburb of Tel Aviv, | daily clinics staffed by paediatricians from Sheba Medical Centre |
| Shulman | 1996‐1999? | USA | paediatrics (children of parents with substance abuse) | UD | M | D | Methadone maintenance treatment center in New York | weekly half‐day visits by consultant, psychologist, speech pathologist, nutritionist and administrative assistant. |
| Spencer | 1979‐1989 | UK | paediatrics | U | M | C | 18 GP surgeries in Sheffield 1979‐1989. | monthly consultant visits for joint consultations with GPs, direct care, regular discussions and lunchtime education sessions. |
| Steele | 1997 | Canada | child psychiatry | R | S | D | Rural communities | survey of practice patterns |
| Strathdee1 | 1987? | UK | psychiatry | U | S | D | survey of UK psychiatrists | assessing level of outreach activity |
| Strathdee2 | 1988? | UK | psychiatry | UD | S | D | 3 primary care clinics in deprived areas of urban London | outpatient clinics in primary care clinics |
| Toupin | 1951‐1990 | Canada | rheumatology (arthritis) | R | M | D | 21 community clinics, 4 regional centres in British Columbia and Yukon provinces. | visits from Vancouver by rheumatologist for direct care and education sessions, in conjunction with allied health and social service outreach. |
| Tyrer | 1982‐1985 | UK | psychiatry | U | S | E | 5 clinics in general practices in Nottingham. | weekly to monthly clinics by consultant. |
| Viehrhout | 1993? | Holland | orthopaedics | U | M | E | Urban & rural primary care clinics | Joint consultation between orthopaedic surgeon, 3 general practitioners & patient in primary care setting. |
| Wagstaff | 1991‐1996 | UK | paediatric cardiology | U | S | D | Referring hospitals to Royal Brompton Hospital London | 3‐monthly visits by specialist paediatric cardiologist at the hospital with local paediatricians. |
| Wakefield | 1989‐1994 | USA | all specialties | R | S | D | rural communities in Iowa | regular visiting consultant clinics to rural hospitals |
| Walshe | 1995 | UK | dermatology, paediatrics, orthopaedics, ENT | U | S | C | GP clinics in Wolverhampton | consultant visits in primary care |
| Weingarten | 1982‐1996 | Israel | psychiatry | UD | M | D | Primary care clinic in a town of 15,000 jewish immigrants from Yemen | visits from psychiatrist, psychologist and social worker out of Psychiatric hospital. Provided direct care, case discussions, education sessions, Balint sessions & video analysis of consultations. |
| Williams | 1972‐1981 | UK | psychiatry | U | S | E | Ecological study of UK by region. | Psychiatric consultations in primary care settings. |
| Zegelman | 1984‐1985 | UK | psychiatry | U | S | C | Health centre in southwest sector of Edinburgh. | outreach clinics in primary care |
2.
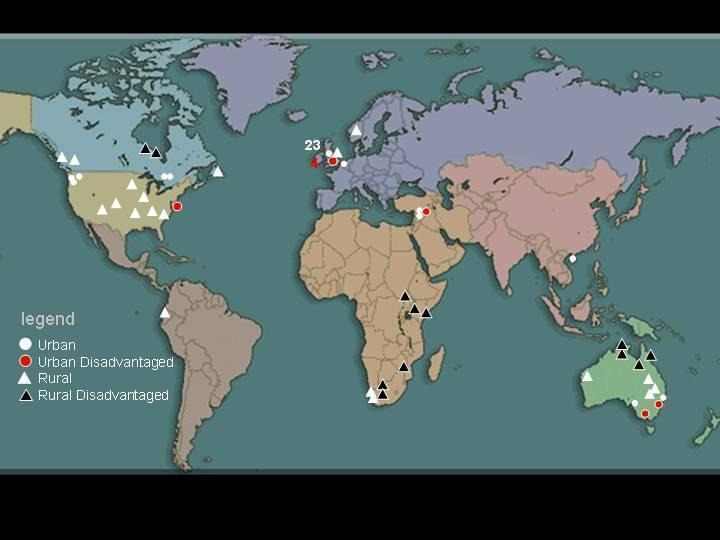
Reports of all identified specialist outreach interventions
A wide range of settings was represented. At one extreme specialists visited urban general practice clinics near major hospitals. At the other extreme, they used small aircraft or four‐wheel‐drive vehicles to visit sparsely populated rural and remote areas of Africa, Australia and Canada. Thirty‐five studies described outreach to "urban" populations, 7 to "urban disadvantaged" populations, 19 to "rural" populations, and 12 to "rural disadvantaged" populations.
A wide range of specialties were represented. Eleven studies reported outreach in multiple specialties ‐ of these studies seven were from the UK and one each from Norway, Israel, Australia and the USA. Each of the remainder was a study of a single specialty ‐ twenty of psychiatry, eight of paediatrics, seven of general surgery, six of ophthalmology, four of obstetrics and gynaecology, two of each of orthopaedics, oncology and rheumatology, and one of each of haematology, neurology, otolaryngology (ENT), palliative care, genitourinary medicine, plastic and reconstructive surgery, cardiothoracic surgery, internal medicine, orthodontics, anaesthetics and paediatric cardiology.
Fifty‐three interventions were simple outreach with minimal interaction with local staff, and no additional services. Twenty outreach interventions were multi‐faceted, most commonly involving joint consultations or education sessions for local primary‐care staff, and some involved the specialist as part of a visiting team that included other health professionals. The proportion of studies in which outreach was multifaceted was as follows: psychiatry (10/20), paediatrics (4/8), oncology (1/2), palliative care (1/1), genitourinary medicine (1/1), rheumatology (1/1), orthopaedics (1/2), and obstetrics and gynaecology (1/4).
Nine studies met the EPOC Inclusion Criteria, 17 were comparative studies that did not meet EPOC criteria, and 47 were descriptive only. Of the 26 studies with a comparison group, 14 were from the UK (50% of UK studies), six from the USA (55% of USA studies), 3 from Australia (25% of Australian studies), and one each from Canada (14% of Canadian studies), Holland (100%) and Hong Kong (100%). Eighteen out of 26 related to urban non‐disadvantaged populations, and two‐thirds of these were from the UK. The distribution across settings is shown in Table 2. We combined the 'urban‐disadvantaged', 'rural' and 'rural‐disadvantaged' categories and compared the distribution of evidence 'quality' against the 'urban non‐disadvantaged' category, and found that the overall quality of studies was significantly higher in evaluations of outreach to urban non‐disadvantaged populations (df=2, Chi‐square=10.34, p=0.006).
2. Quality of evidence by population type.
| Population type | Satisfies EPOC | Comparative_not EPOC | Descriptive only | Total |
| Urban | 7 | 12 | 16 | 35 |
| Urban Disadvantaged | 0 | 1 | 6 | 7 |
| Rural | 1 | 4 | 14 | 19 |
| Rural Disadvantaged | 1 | 0 | 11 | 12 |
| Total | 9 | 17 | 47 | 73 |
2. Systematic review of rigourous evaluations of specialist outreach clinics
Studies that met our Inclusion Criteria are listed in the Table of Included Studies, and the reason for exclusion of others is given in the Table of Excluded Studies. Two controlled before‐after studies were excluded due to inappropriate control groups (Buhrich, Owen).
Five RCTs (O'Brien,Vierhout,Katon1,Katon2; Roy‐Byrne), two CBAs (Howe; Tyrer) and two ITSs (Gruen, Williams) met the EPOC criteria for inclusion in the review. The stated reasons that outreach was initiated in these studies included improving access to specialist care (Gruen; O'Brien), improving quality of care (Howe; Williams; Katon2), improving outcomes (Katon2), reducing unnecessary use of services (Vierhout; O'Brien; Williams), improving collaboration between specialists and primary care (Tyrer) and making care more efficient (Vierhout; Williams).
02.01 Randomised Controlled Trials
The only RCT examining a 'simple' outreach intervention was O'Brien's study of orthodontic visits to clinics at three community health centres 500m to 15km from the university hospital base in Manchester, UK. The trial aimed to test the effect of outreach on access, satisfaction, outcome of the consultation, and cost. 324 referred adult patients were randomised to receive a consultation in the main base hospital or in an outreach clinic. Visiting specialists and specialists in the outpatient clinic provided triage consultations, and all treatment was carried out at a later date in the main hospital. Outcomes measured included attendance at the appointment, total time spent in the clinic, acceptance for treatment, patients' perceptions of travel arrangements and costs, and costs based on capital, overheads, equipment and consultant travel. Appropriate referrals were assessed according to guidelines that had been developed by the researchers as part of a previous study and had been disseminated to local dentists by post along with an invitation from a local consultant to attend a seminar.
Vierhout's Dutch study examined whether monthly joint consultations between a visiting specialist orthopaedic surgeon and three general practitioners simultaneously in their primary care practice reduced unnecessary interventions and referrals, when compared to referral to hospital clinics. Four separate practices were involved. A patient was entered into the study if he or she was 10 to 75 years old and had a locomotor system disorder, and the primary practitioner was uncertain about the management or if a referral was considered. 333 patients were randomised using numbered envelopes in a 'randomised consent' design (patients were not informed that they were participating in a trial until after randomisation, and then given information only about their arm of the study). 25 intervention and 36 control patients withdrew after randomisation, the majority of whom did not complete the survey forms. The primary outcome measures were patient investigation, treatment and referral rates over the following year. Other patient outcomes included self assessed health status of patients and clinical examination by a 'blinded' specialist at one year (to assess for an effect on health outcomes). Primary care providers' knowledge and skills were also assessed by a written examination.
Three related RCTs by one psychiatry group in Seattle, USA, were included. All three studies employed a similar collaborative care intervention involving weekly consultations alternating between the primary care physician and the psychiatrist on consecutive weeks for 4 to 6 weeks, as well as primary care physician education, case conferences, patient education using information booklets and video‐tapes, and individualised treatment algorithms. Control patients received usual care from their primary care physician. All three studies were conducted in clinics of large health maintenance organizations (HMOs) in urban Seattle, and enrolled only adults. Katon1 randomised 217 patients with depression (91 major and 126 minor depression). Primary outcome measures included adherence to medication (both self‐reported and dispensed from pharmacies at doses consistent with national published treatment guidelines), satisfaction with care, and level of depressive symptoms, assessed by blinded telephone interviews. This trial found that the intervention was most effective for patients with major depression who required a change in medication. Therefore the Katon2 trial was designed to assess 'stepped collaborative care' in which patients with major depression were enrolled if they met certain criteria of failure of treatment in primary care. 228 patients were randomised to intervention or control treatments similar to the previous trial. Similar outcome measures were assessed and, in addition, a cost‐effectiveness study was performed taking into account costs to the HMO but not patient‐related costs. The third trial (Roy‐Byrne) followed a similar design to that in Katon1, except that it addressed panic disorder rather than depression. 115 patients were randomised to collaborative care or usual care. Adherence, satisfaction and symptom outcomes were again assessed. In all three studies outreach formed only part of the multifaceted intervention and, while the specialist visits were necessary for many aspects of the intervention, outcomes could not be attributed to the outreach component alone.
02.02 Controlled Before‐After studies
Howe retrospectively studied visiting oncology clinics in five rural hospitals in Illinois, USA between 1986 and 1991. Only female Caucasian patients diagnosed with breast cancer were included. The intervention involved patient consultation and administration of treatment protocols, with the aim of improving on previously identified inadequate rates of 'state‐of‐the‐art' breast cancer care. The control group included patients managed in four rural hospitals that did not receive specialist visits, who may or may not have been referred to centralised specialist clinics. The control hospitals and their primary care and general specialist providers (ie general surgeons), however, did receive an additional program of audit and feedback, without visits from a specialist oncologist. The observed effect of outreach may therefore be less than it would have been if the control had received no intervention. The five rural hospitals in the intervention group contributed 96 patients before the intervention began (1986‐1987) and 105 patients after (1990‐1991). The control hospitals had records of 47 patients in 1986‐7 and 67 patients in 1990‐1. Primary outcomes were oncology consultations and 'state‐of‐the‐art' care (defined as being consistent with the current "Physician's Data Query" of the National Cancer Institute, where annually‐updated standards for breast cancer management are posted, and which included bilateral mammography, tumour staging and type‐specific treatment during the period of study). Patients records were retrospectively reviewed.
Tyrer's study examined psychiatric hospital admissions among 15‐64 year olds in Nottingham, UK during the period 1978‐1985, spanning 'sectorization' of the psychiatric service in 1981, a policy which led to rapid increase in the number of psychiatric outreach clinics. This was particularly pronounced in the East Nottingham sector, with a population of 85,000, and the analysis included in this review was limited to the comparison of admission rates prior to and after sectorization in East Nottingham, with the control group being the remainder of Nottingham (population approximately 300,000) where outreach clinics were less pronounced. While psychiatrists provided a 'shifted outpatient' model of care, there were parallel changes that involved expansion of nursing and social work clinics in general practices for care of psychiatric patients. Multiple analyses were performed, however the most relevant was a comparison of the change in mean rate of admissions before and after 1981, compared with the control. From the graphical data, it appears that an important change in trend of admission rates in the intervention group (but not the control group) occurred at the same time, which was not analysed further in the original study, and the data was unobtainable.
02.03 Interrupted Time Series analyses
Both ITS studies are ecological studies over long time periods that assessed changes in hospital‐based service use that may be due to outreach activities. Williams study is similar to Tyrer in that it examines the effect of psychiatrist outreach clinics on psychiatric admissions in the UK. Williams, however, used national hospital psychiatric admission statistics from 1964‐1981, divided geographically into ten 'super‐regions,' and data they had previously collected by survey indicating the amount of outreach activity (number of consultants and clinics). Data is presented showing that the amount of outreach increased considerably after 1973, and they chose this as the 'beginning' of the intervention for the purpose of analysis. The change in trend in admission rates in each region after 1973 compared with before was calculated. The change in trend of amount of outreach in each region post‐1973 compared with before was also calculated. The two changes in trend were then correlated and the strength of association assessed. Gruen's study of outreach in four surgical disciplines to rural Indigenous communities in northern Australia was the only included study to examine a rural disadvantaged population. Specialists in general surgery, gynaecology, ophthalmology and ENT participated in a coordinated government‐funded program of three to six‐monthly outreach visits to many small communities up to 900 km distant. 5184 patient consultations were conducted and described, the most common being follow‐up appointments and new referrals for colposcopy, retinopathy screening, cataract, abdominal/gastrointestinal conditions, cutaneous surgical conditions, and chronic otitis media with tympanic perforation and hearing loss. The outreach service began in 1997. Comparison was made between the trends in annual number of consultations with remote community patients (hospital and outreach) before and after 1997, and the trends in the annual number of consultations held in hospital outpatients (requiring patients to travel) before and after 1997. Analysis was limited to the specialty of gynaecology in which outreach had been most consistent. The trends before and after the introduction of outreach were estimated using Poisson regression, and the change in trend was assessed.
Risk of bias in included studies
1. Randomised Controlled Trials
Randomisation and allocation concealment was satisfactory in four of five studies, and not described in one (Katon1). Vierhout's randomised consent design, where patients were first randomised and then received information exclusively about the procedure within their own group, was constructed because the study could not be blinded, and it obtained institutional ethics committee approval. Twenty‐seven patients 'did not wish to participate', but 17 of them were excluded for failing to complete the study forms after randomisation. Patient allocated to intervention and control groups who then withdrew did not differ in important respects, however. All RCTs except O'Brien reported baseline characteristics proving comparability of intervention and control groups. Follow‐up rates were over 80% in all but O'Brien. None of the RCTs reported a sensitivity analysis allocating the non‐attenders to intervention or control groups. Sample sizes ranged from 115 to 324, with a mean of 231.
There was potential for detection bias in O'Brien's study, where the researchers provided the intervention and assessed the outcome of intervention, need for treatment, and reason for treatment decisions, and classified the referral as appropriate or inappropriate, albeit using predefined criteria. Most outcome measures were automated or collected by individuals blinded to the allocation. Standardised measures, validated in other settings, were used by O'Brien for satisfaction, and Vierhout for self‐reported health. However, several non‐standardised self‐reported measures of satisfaction, perceived health status and impression of the quality of care remained.
Denominators at follow‐up intervals were obtained from the authors in Katon1; Katon2, and Roy‐Byrne. All five RCTs undertook an "Intention to treat" analysis. There was potential for contamination in Roy‐Byrne, (and possibly in Katon1 and Katon2) where some primary care physicians had both intervention and control group patients, making it likely that management of control patients would have been affected by the physician education aspects of the intervention.
2. Controlled Before‐After studies
Both CBA studies were vulnerable to selection bias. Neither study reported baseline characteristics of intervention and control groups, except that Howe presented mean ages. It is likely that Howe's 4 hospitals that did not receive outreach differed in some ways from the 5 receiving outreach. Tyrer stated that the region served by outreach was more socio‐economically depressed than the rest of Nottingham, and may therefore have had a greater potential to benefit. In both studies it is possible that, for these reasons, the control group is inappropriate. Howe claimed follow‐up of 99%, but used a process of record review that may not have been blinded. Tyrer used data from automated records. The validity of Tyrer's allocated year in which outreach began is unclear. Both studies were subject to unit of analysis error, because the intervention was delivered at practice and hospital level but outcomes were measured in individual patients. Neither study addressed this in their analysis. Because of these issues, and because these were generally poor quality studies, they are discussed in the text and presented in the tables, but not included in the forest plots nor considered for meta‐analysis.
3. Interrupted Time Series analyses
The ITS studies spanned long time periods (Williams 1964‐1981 and Gruen 1992‐1999) during which other health policy and health service changes may have influenced the outcomes of interest (admission rates and referral rates, respectively). Like Tyrer, the validity of Williams' allocated year in which outreach began is unclear. Both studies used automatically‐collected data. Neither study used ARIMA methods of analysis or time‐series regression models with tests for serial correlation, and neither prespecified the expected effect nor gave a rationale for the number and spacing of data‐points. These studies are also represented in tables and the text, but not in forest plots.
4. A comment on the quality of comparative studies excluded from the main analysis
Many studies were excluded from the main analysis on the basis of study design, most being comparative studies without baseline outcome measures prior to the intervention. Some appeared to be methodologically stronger in some respects than all four CBA and ITS studies included in the review (e.g. Black, Bowling, Gillam, Haynes, Helliwell, Walshe). Their strengths were in description of the characteristics as well as the outcomes of outreach and control groups, involvement of large numbers of patients from multiple centres, and corroboration of survey data with other health service data. Due to their design, however, they are also subject to residual confounding. Details of their methods and results are shown in Table 3.
3. Results of all comparative studies.
| Label | Pop Type (U,UD,R,RD) | Country | Specialty | Comparative method | Effect of outreach |
| Black | U | UK | orthopaedics & dermatology | Comparison of 6 outreach clinics in orthopaedics and dermatology with 6 hospital clinics by the same consultants regarding process, costs, health status and patients' satisfaction | Dermatology ‐ shorter waiting time for appointment (69 v 97 days), less travel time (20 v 40 mins), longer waiting time at clinic (30 v 15 mins), less total cost to patient (4.49 v 5.51), lower non‐attendance rate (11% v 20%), no difference in satisfaction except waiting time at clinic, fewer total follow‐up appointments made (20 v 45), fewer items prescribed per patient (1.41 v 1.78), fewer total excisions/cautery (7 v 15), more longstanding conditions, less improvement in health status at 3 months on HSQ‐12 questionnaire, lower health service costs per pt (43.78 v 63.92) but higher marginal costs (7.79 v 3.62). Orthopaedics ‐ longer waiting time for appointment (61.5 v 48.5 days), less travel time (30 v 40 mins), shorter waiting time at clinic (10 v 25 mins), less total cost to patient (4.98 v 8.86), higher non‐attendance rates (9% v 3%), higher satisfaction with location of clinic, length of consultation, time in clinic and specialist's explanation, more surgical procedures in clinic (15 v 3), no difference in health status at 3 months on HSQ‐12, fewer patients booked for surgery, fewer total follow‐up appointments (12 v 31), more items prescribed per patient (1.43 v 1.17), more tests initiated in total (19 v 9), higher health service costs (252.71 v 160.71), higher marginal costs (15.68 v 6.09). |
| Bowling | U | UK | cardiology, ENT, general medicine, general surgery, gynaecology, paediatrics, rheumatology | Comparison of 38 outreach clinics and 38 hospital clinics on measures of process, costs, health status and patient satisfaction. | less wait (5.7 v 7.9 wks), lower non‐attendance (10% v 16%), shorter time at clinic (15.9 v 32.8 mins), improved satisfaction (on non‐validated scale) of patient & provider, more discharged from clinic (36% v 27%), perceived health improvement on HSQ12 (+2.58) but NB different baseline (slightly healthier) and no difference on mental and physical function questions. Cheaper cost to patient (3.96 v 8.40) but more expensive to NHS (149.59 v 106.79). More new patients (65% v 31%) and fewer patients per clinic (range 5‐28 v 8‐50). More likely to be referred for surgery or investigation (43% v 31%). |
| Gillam | U | UK | ophthalmology | Comparison of referral patterns, costs and patient satisfaction with 17 matched control general practices. | Less travel time (73% <10 mins v 18.7%), distance (1.3% >5 mi v 22%), time in clinic (94.9% < 30 mins v 86%), more costly (48.09 v 15.71). Fewer referrals to hospital outpatients than from control practices (3.8 v 9.5 per 10000 registered patients). |
| Helliwell | U | UK | rheumatology | Comparison of community clinic with main hospital clinic regarding patients' satisfaction, case‐mix and cost. | shorter travel distance (1.6 v 4.9 mi), shorter wait for appointment (47% <1 mo v 27%), longer consultation times (49% twenty mins or longer v 31%), higher patient satisfaction (82% said "all their questions were answered" v 52%), fewer patients per clinic (8.6 v 14.1 per doctor), fewer new patients (old/new ratio 6.04 v 3.96), cost per patient higher (15.93 v 10.35), casemix similar. |
| McKechnie | U | UK | psychiatry | Comparison of 72 referrals from one practice to outreach clinic with 54 patients from another practice to hospital clinic in terms of patients referred, case‐mix and admissions. | more new referrals (67% v 50%), fewer total admissions (10 v 24) and shorter total days in hospital (339 v 587). |
| O'Brien | U | UK | orthodontics | See Table of Included Studies | See outcomes of Included studies |
| Perrett | U | UK | gynaecology, orthopaedics, general surgery. | Comparison of waiting times between 37 outreach clinics and hospital clinics. Description of extent and use of clinics. | shorter waiting time for appointment (97.0% <3 mo v 88.1%), more booked for elective surgery (17.4% v 9.4%). |
| Riley | U | UK | gynaecology, orthopaedics, urology | Comparison of outreach clinics and hospital outpatient clinics with respect to patient satisfaction, process & outcomes. | Shorter waiting times (4.8 wks v 8.6 wks), shorter time in clinic (75% <10mins v 35%), more referred for investigation (76% v 57%), more booked for surgery (67% v 54%), fewer patients seen per clinic (9 v 28). |
| Spencer | U | UK | paediatrics | Comparison of patients seen in 18 different outreach clinics by one consultant versus that consultant's hospital clinics over 1year. | Fewer new patients per session (2.5 v 4), fewer patients seen, less non‐attendance rate (20% v 25%), less 'organic' disease (38% v 44%), more referrals (5.5 v 0), fewer admissions (3% v 7%), fewer children investigated (6.5 v 47), fewer investigations per child (1.2 v 2.1), fewer children prescribed treatment (7% v 23%). |
| Walshe | U | UK | dermatology, ophthalmology, gynaecology & ENT | Comparisons of 138 patients at 4 outreach clinics and 4 hospital clinics (different consultants) on measures of process, costs and patient satisfaction, using routine data, patient survey & patient interviews. | Non‐attendance rates lower overall but variation between specialties (range 3.3‐16.9% v 8.0‐16.6%), higher proportion of new referrals (range 48.6‐81.8% v 23.2 v 34.6%), fewer number of appointments per patient episode (range 1.2‐2.1 v 2.9‐4.3), similar mean waiting time from referral to first appointment (range 7.1‐11.3 weeks v 8.6‐11.0 weeks), less travel time (23.2 mins v 51.7 mins), less time at clinic (24.3 mins v 64 mins), lower cost per clinic session (range 130‐196 v 191‐306), higher cost per clinic appointment (range 13‐20 v 5‐13), lower cost per outpatient episode (range 20‐28 v 17‐85), greater percent making positive comments about clinic (93% v 42%) and fewer making negative comments (7% v 58%). |
| Williams | U | UK | psychiatry | See Table of Included Studies | See outcomes of Included studies |
| Zegelman | U | UK | psychiatry | Comparison of non‐attendance rates at outreach clinic compared with hospital outpatient clinic. | No difference observed. |
| Katon1 | U | USA | psychiatry (depression) | See Table of Included Studies | See outcomes of included studies |
| Katon2 | U | USA | psychiatry (persistent major depression) | See Table of Included Studies | See outcomes of Included studies |
| Roy‐Byrne | U | USA | psychiatry (panic disorder) | See Table of Included Studies | See outcomes of Included studies |
| Kates | U | Canada | psychiatry | Comparison of 80 patients seen in family physicians' offices with 145 patients seen in psychiatric clinic for patient and problem characteristics, and a study of 1515 patients in 88 family physicians offices in 36 practices in urban Hamilton, Ontario. | fewer anxiety cases (7% v 20%), fewer number of visits per patient illness episode (2.5 v 4.8), |
| Viehrhout | U | Holland | orthopaedics | See Table of Included Studies | See outcomes of Included studies |
| Ho | U | Hong Kong | child psychiatry | Comparison of 56 children seen in community clinic and 42 seen in hospital clinic regarding clinical presentation, treatment and inpatient care. | Higher hyperactivity scores (4.61 v 3.26), shorter duration of treatment (4.29 months v 9.20) & less inpatient care (3.6% v 14.3%). |
| Tyrer | U | UK | psychiatry | See Table of Included Studies | See outcomes of Included studies |
| Buhrich | UD | Australia | psychiatry (schizophrenia) | Comparison of patients' rate & duration of hospital admissions in the year after attending or failing to attend outreach clinics. | Lower rate and duration of admission (nb problems) |
| Owen | R | Australia | psychiatry | Comparison of changes in prescribing patterns pre/post in the region with those for the state overall. (NB this is not a valid comparison) | mixed picture of prescribing anxiolytics, dexamphetamine, antipsychotics and anti‐depressants. |
| Haynes | R | UK | ophthalmology (cataract) | Comparison by interview and questionairre of 198 patients attending outreach clinic for surgery with 201 patients attending district hospital clinic on measures of acess, satisfaction and costs. | Outreach patients older (77.5 v 75.6), less affluent and poorer general health (SF12), shorter travel time (24 min v 27), shorter time in hospital (2.9 hr v 3.3), fewer required companion to take time off work (10% v 17%), shorter time lost off work (10hr v 17 hr), less out of pocket cost to patient (0.57 v 0.80), less cost to health service per patient (72.98 v 110.50), more satisfied with arrangements & facilities and with medical care at 3 months. No measurable differences in outcomes. |
| Desch | R | USA | oncology | Financial analysis of total costs of 1745 patients pre and post introduction of the outreach program. | Increased use of specialist level care (452%). Less cost per patient ($4392 v $10,233) |
| Howe | R | USA | oncology (breast cancer) | See Table of Included Studies | See outcomes of included studies |
| Oboler | R | USA | Internal medicine | Simple cost comparison. | No significant cost difference per patient ($67 v $68) |
| Gruen | RD | Australia | general surgery, gynaecology, ophthalmology, ENT | See Table of Included Studies | See outcomes of included studies |
Effects of interventions
Detailed results of Included Studies are presented in outcome tables (Table 4; Table 5; Table 6; Table 7; Table 8; Table 9) and in the forest plots in the Metaview Analysis Graphs.
4. Outcomes ‐ Access.
| Study | Study type | Outcome | Results | Notes |
| Howe | CBA | Proportion getting oncology consult | Int Pre 25/96(26%), Con Pre 17/47(15%), Int 57/105(54%), Con 23/67(34%), Absolute difference=20%, Relative % difference=59%, Absolute change from baseline Int 28% Con 19% Difference in absolute change from baseline=9% | |
| O'Brien | RCT | Cost for consumer | Int u=0.95 (sd=1.43) Con u=1.17 (sd=1.22) Absolute difference= ‐0.22 Relative % difference= ‐19% | |
| O'Brien | RCT | Hours to clinic | Int u=0.37 (sd=0.28) Con u=0.63 (sd=0.42) Absolute difference= ‐0.26 Relative % difference= ‐41% | |
| O'Brien | RCT | Miles to clinic | Int u=4.18 (sd=4.36) Con u=5.85 (sd=4.48) Absolute difference= ‐1.67 Relative % difference= ‐29% | |
| O'Brien | RCT | Minutes in clinic | Int u=28.1 (sd=13.3) Con u=42.1 (sd=26.6) Absolute difference= ‐14 Relative % difference= ‐33% | |
| O'Brien | RCT | Attendance rate | Int=134/169(83%) Con=125/155(81%) Absolute difference=2% Relative % difference=2% |
5. Outcomes ‐ Quality.
| Study | Study type | Outcome | Results | Notes |
| Howe | CBA | Proportion getting 'state of the art' care | Int Pre 56/96(58%), Con Pre 27/47(57%), Int 66/105(63%), Con 37/67(55%), Absolute difference=8%, Relative % difference=15%, Absolute change from baseline Int=5% Con=‐2%, Difference in absolute change from baseline=7% | |
| Katon1 | RCT | Proportion adhering to treatment at 30days | Int=43/49(88%) Con=24/42(57%) Absolute difference=29% Relative % difference=51% | |
| Katon1 | RCT | Proportion adhering to treatment at 90days | Int=37/49(76%) Con=21/42(50%) Absolute difference=26% Relative % difference=52% | |
| Katon2 | RCT | Adherence to treatment (dispensed at minimum guidelines dose) | Int=69% Con=44% Absolute difference=25% Relative % difference=57% | |
| Katon2 | RCT | Adherence to treatment at 6 mo (interview) | Int=73 % Con=51% Absolute difference=22% Relative % difference=43% | |
| Roy‐Byrne | RCT | Adherence to treatment 25/30 days at 3 mo. | Int=27/51(53%) Con=13/48(27%) Absolute difference=26% Relative % difference=96% | |
| Roy‐Byrne | RCT | Proportion receiving appropriate dose/duration of medications | Int=31/51(61%) Con=16/48(33%) Absolute difference=28% Relative % difference=85% | |
| Roy‐Byrne | RCT | Proportion receiving appropriate type of medications | Int=39/51(76%) Con=23/48(48%) Absolute difference=28% Relative % difference=58% | |
| O'Brien | RCT | Appropriate referrals | Int=80/115(70%) Con=67/108(62%) Absolute difference=8% Relative % difference=13% | More appropriately referred (the intervention was to triage for treatment) |
6. Outcomes ‐ Health outcomes.
| Study | Study type | Outcome | Results | Notes |
| Katon1 | RCT | Improved >=50% on SCL score at 7 months | Int=31/41(76%), Con=15/35(43%) Absolute difference=33% Relative % difference=77% | More improved on severity score at 7 mo. |
| Katon2 | RCT | Depression‐free days in 6 mo | Int u=87.7(sd=4.5) Con u= 70.9(sd=2.9) Absolute difference=16.8 Relative % difference=24% | More depression free days |
| Katon2 | RCT | Resolution at 6 mo. | Int=48/97(50%) Con=40/95(42%) Absolute difference=8% Relative % difference=19% | More resolved by 6 mo. |
| Roy‐Byrne | RCT | Recovery at 6 mo (ANSI criteria) | Int=22/45(49%) Con=8/47(17%) Absolute difference=32% Relative % difference=188% | |
| Roy‐Byrne | RCT | Improved at 6 mo (PDSS criteria) | Int=34/45(76%) Con=18/47(38%) Absolute difference=38% Relative % difference=100% | |
| Vierhout | RCT | Disorder‐free at 1 yr | Int=35% Con=23% Absolute difference=12% Relative % difference=52% |
7. Outcomes ‐ Patient satisfaction.
| Study | Study type | Outcome | Results | Notes |
| Katon1 | RCT | Rated quality of care at least 'Good' at 4 months | Int 40/43(93%) Con 25/33(76%) Absolute difference=17% Relative % difference=22% | |
| Katon1 | RCT | Medication helping at least 'somewhat' at 4 months | Int=38/43(88%), Con=21/33(64%), Absolute difference= 24% Relative % difference=38% | |
| Katon2 | RCT | Satisfaction with treatment rated 'good' to 'excellent' | Int=79/97(81%) Con=66/95(69%) Absolute difference=12% Relative % difference=17% | |
| Roy‐Byrne | RCT | Satisfaction with quality of care in first 6 mo. | Int=37/45(82%) Con=20/47(43%) Absolute difference=39% Relative % difference=91% |
8. Outcomes ‐ Service use.
| Study | Study type | Outcome | Results | Notes |
| Katon1 | RCT | Additional primary care visits in 1 yr | Int u=4.5(sd=3.7) Con= u=3.7(sd=2.4) Absolute difference=0.8 Relative % difference=22% | |
| Katon1 | RCT | Had outpatient clinic psychiatric consult in 1 yr | Int=3/108(3%) Con=11/109(10%) Absolute difference= ‐7% Relative % difference= ‐70% | |
| Katon1 | RCT | Seen mental health worker outside PC clinic in 1 yr | Int=30/108(28%) Con=34/109(31%) Absolute difference= ‐3% Relative % difference= ‐10% | |
| Katon2 | RCT | PC visits 1 yr | Int u=3.4 (sd=4.3) Con u=3.3 (sd=3.1) Absolute difference=0.1 Relative % difference=3% | |
| O'Brien | RCT | Referred for hospital treatment | Int=42/115(37%) Con=37/108(34%) Absolute difference= ‐3% Relative % difference= ‐9% | |
| O'Brien | RCT | Use of other hospital outpatient services | Int=18/115(16%) Con=13/108(12%) Absolute difference=4% Relative % difference=33% | |
| O'Brien | RCT | Referral to general dental practitioner | Int=6/115(5%) Con=11/108(10%) Absolute difference= ‐5% Relative % difference= ‐50% | |
| O'Brien | RCT | Referral to Xray | Int=3/115(3%) Con=0/108(0%) Absolute difference=3% Relative % difference=incalculable | |
| Vierhout | RCT | Total Injection treatments | Int=49/144(34%) Con=54/128(42%) Absolute difference= ‐8% Relative % difference= ‐19% | '%' = number per 100 patients |
| Vierhout | RCT | Total Lab tests | Int=23/144(16%) Con=43/128(34%) Absolute difference= ‐18% Relative % difference= ‐53% | '%' = number per 100 patients |
| Vierhout | RCT | Total Medication scripts | Int=35/144(24%) Con=38/128(30%) Absolute difference= ‐6% Relative % difference= ‐20% | '%' = number per 100 patients |
| Vierhout | RCT | Total Operations | Int=25/144(17%) Con=23/128(18%) Absolute difference= ‐1% Relative % difference= ‐5% | '%' = number per 100 patients |
| Vierhout | RCT | Total Radiology requests | Int=79/144(55%) Con=120/128(94%) Absolute difference= ‐39% Relative % difference= ‐41% | '%' = number per 100 patients |
| Vierhout | RCT | Total referrals to physiotherapy | Int=72/144(50%) Con=93/128(73%) Absolute difference= ‐23% Relative % difference= ‐32% | '%' = number per 100 patients |
| Tyrer | CBA | Admission rate | Int Pre=39/10,000 Con Pre=28/10,000 Int=33/10,000 Con=25/10,000 Absolute difference=8/10,000 Relative % difference=32% Absolute change from baseline ‐6/10,000 vs ‐3/10,000 Difference in absolute change from baseline= ‐3/10,000 | Data estimated from graph (Fig 3a in Tyrer et al 1990) |
| Gruen | ITS | Trend in hospital outpatient consultations in gynaecology | Int= ‐26% p.a. Con= +89% p.a. Relative % difference= ‐115% |
9. Outcomes ‐ Cost.
| Study | Study type | Outcome | Results | Notes |
| Katon1 | RCT | Cost per patient | Int=$1337 Con=$850 Absolute difference=$487 Relative % difference=57% | |
| Katon1 | RCT | Cost effectiveness (avge treatment cost divided by measure of treatment effectiveness) | Int=1797 Con=1941 Absolute difference= ‐144 Relative % difference= ‐7.4% | |
| Katon1 | RCT | Incremental cost effectiveness (=((cost intervention)‐(cost control)) / ((effectiveness of intervention)‐(effectiveness of control))) | 1592 | |
| Katon2 | RCT | Cost per patient | Int=2406 Con=2110 Absolute difference=296 Relative % difference=14% | |
| O'Brien | RCT | Cost of treatment | Int=8.40 Con=9.11 Absolute difference= ‐0.71 Relative % difference= ‐8% | |
| Gruen | ITS | Avge cost per patient | Int=277 Con=450 Absolute difference= ‐173 Relative % difference= ‐38% |
1. Access to specialist care
01.01 Perceived access
No measures of perceived access used standardised scales. O'Brien found outreach led to 7.5% of patients reporting "cost being a problem" compared to 23.2% of controls, and 15.3% "having difficulty parking" compared with 73.1% of controls.
01.02 Measures of access
O'Brien was the only included study to report objective measures of access, and found that outreach reduced the cost for the consumer by 19%, reduced the distance to the clinic by 29% and the time taken getting to the clinic by 41%, although the absoluted differences were small (22 pence, 1.67 miles and 16 minutes, respectively). Patients also spent on average 14 minutes (33%) less at the outreach clinic.
01.03 Realised access (attendance) The improved access measures in O'Brien's study were associated with an increased in the attendance at booked appointments from 81% to only 83%. In the studies in rural populations, where access changes may be more significant, Howe found outreach led to 9% more breast cancer patients receiving an oncology consult, and Gruen found a large increase in numbers of specialist consultations involving remote community patients (390%).
2. Quality of care
02.01 Guideline‐consistent care and referrals
Howe reported 7% more breast cancer patients received guideline‐consistent care. Roy‐Byrne reported that almost twice as many patients in the intervention group than in the control group received the appropriate type, dose and duration of medication. O'Brien reported 8% more patients were appropriately referred to the specialist, although only 2.2% more were offered treatment by the specialist.
02.02 Adherence to treatment
Self‐reported adherence to treatment was greater for outreach in all three Seattle studies, and was sustained at one and three months in Katon1. When pooled these three studies showed outreach had a combined relative risk for not adhering to treatment of 0.62 (95% CI 0.49‐0.78, Z=4.10, p<0.0001) and were statistically homogenous (Chi2=0.82, df=2, P=0.67).
3. Health outcomes
Vierhout reported no improvement in objective clinical assessment or subjective measures of symptoms, except on the variable "disorder free at one year," which was a self‐reported assessment of psychological and physical health, reported favourably by 35% of intervention patients and 23% of controls. Katon1, Katon2 and Roy‐Byrne, on the other hand, all reported substantial improvements in objective measures of symptom improvement and disease resolution. Pooling of the 3 Seattle studies led to combined relative risk of persistent symptoms for the outreach group of 0.63 (95% CI=0.52‐0.77, Z=4.44, p<0.00001), although they were statistically heterogeneous (Chi2=8.6, df=2, P=0.01).
4. Patient and provider satisfaction
No measures of satisfaction used standardised scales. All three Seattle studies (Katon1; Katon2; Roy‐Byrne) reported greater patient satisfaction with quality of care, the effect of medication, and overall treatment, with a combined relative risk of being unsatisfied of 0.43 (95% CI 0.29‐0.62, Z=4.38, P<0.0001), which was statistically homogenous (Chi2=2.53, df=2, P=0.28).
No studies reported measures of provider satisfaction 5. Use of services
05.01 Primary care services
Both studies (Katon1; Katon2) that examined numbers of primary care physician visits in 1 year found that outreach led to an increase that was not statistically significant (combined standardised mean difference in number of visits of 0.14 (95% CI ‐0.05‐0.32, Z=1.45, P=0.15). There was a reduction in use of other non‐hospital services reported by Katon1 (3% fewer consuted a mental health worker), and Vierhout (23% fewer were referred to physiotherapy).
05.02 Hospital outpatient clinics
Use of relevant hospital outpatient clinics was examined by Katon1, who found outreach reduced hospital attendances from 10% to 3%, and Gruen who demonstrated a significant trend reversal (positive to negative) in annual hospital outpatient consulations for members of rural disadvantaged communities. O'Brien found a small increases in the use of other hospital outpatient clinics, including referrals to other specialties.
05.03 Investigations
Vierhout found outreach almost halved the numbers of laboratory tests and radiology requests. O'Brien, on the other hand, found a slight increase in the numbers of radiology requests.
05.04 Treatment
Outpatient treatment modalities, including medication scripts and injections, were all reduced in Vierhout's study. Admissions for inpatient treatment were reduced by outreach in all studies examining this outcome (O'Brien, Vierhout, Tyrer and Williams).
6. Costs
Katon1and Katon2 found outreach to be more expensive to provide per patient ($487 and $296 more per patient, respectively), whereas O'Brien (urban) and Gruen (rural disadvantaged) found it less expensive per patient (71 pence and AUD$173 less respectively). Katon1 also demonstrated that, despite being more costly to deliver, their multifaceted outreach intervention was 7.4% more cost‐effective than usual care when health outcomes were considered.
7. Comments on the results of comparative studies excluded from the main analysis.
The methods and findings of all/excluded comparative studies are summarised in Table 3. The findings are synthesized and visually represented in Figure 3. In urban non‐disadvantaged populations, the most consistently‐reported findings were that outreach clinics were more expensive and provided for fewer patient consultations per clinic, but were associated with improved patient satisfaction and improved access (perceived by patients, measured in terms of distance, waiting times, cost and convenience, and realised in attendance rates at booked appointments). Sometimes the reported benefits were considerable, and in other cases they were small. Five out of seven studies that reported use of hospital outpatient services showed a reduction associated with outreach. Outreach clinics were associated with more appropriate use of treatment services, which included reductions in psychiatric hospital admissions, and a mixed picture of use of elective treatment and investigations. A broader casemix of patients were seen in outreach and more patients were discharged from outreach clinics than hospital clinics in two out of two studies. Among the seven studies of outreach to rural and disadvantaged populations reports of improved measures of access, quality of care, patient satisfaction and less cost per patient while overall costs were greater, although these were mostly results from single studies.
3.
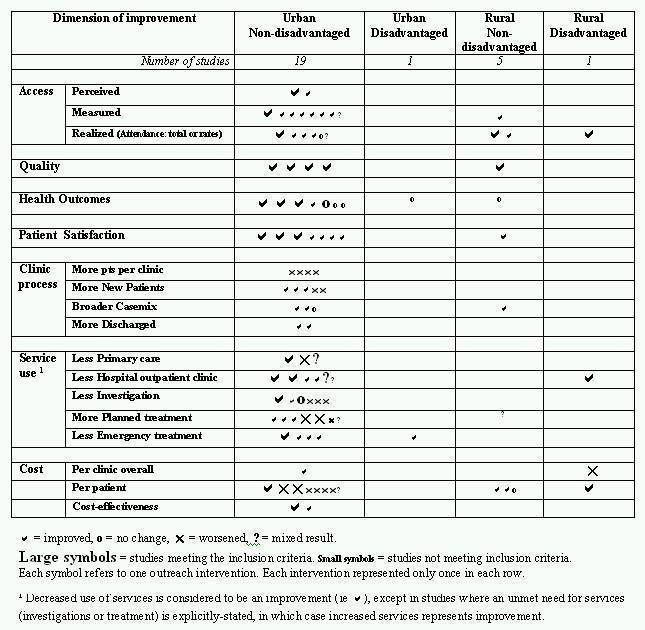
Results of all comparative studies, by population type
Discussion
1. Overview of studies
This review identified 73 specialist outreach interventions in 14 countries on five continents. There are likely to be many others throughout the world that have not been described in the medical literature. Outreach has been established to improve access to specialists and specialist services, foster collaboration between specialist and primary care providers, and improve efficiency and the appropriateness health care service use. Virtually all clinical disciplines are represented. Outreach has taken many different forms, ranging from simple consulting services to complex multifaceted interventions that have included joint consultations, case‐conferences and education seminars, multi‐disciplinary teams, and other care enhancements.
Many articles provided rich descriptions of the outreach initiatives, the context in which they developed, the rationale for their establishment, the services they provided, and the difficulties they faced. Studies comparing the effectiveness of outreach to the more standard model of hospital or office‐based specialist care have measured access, health outcomes, patient satisfaction, costs, use of services and effects on health service and clinical processes. Pooling of three methodogically and statistically homogenous studies was performed, but the heterogeneity of contexts and outcomes of other studies precluded further meta‐analysis. Nonetheless, we have been able to draw some conclusions about the quality and distribution of the available evidence, and about the effectiveness of outreach in different settings and in different forms.
2. Quality and distribution of evidence
02.01 Quality of studies
The majority of comparative studies did not meet EPOC inclusion criteria for study design. Only five RCTS were identified. Most of the other comparative studies failed to make pre‐intervention (baseline) observations of the intervention and control groups, and few made explicit the characteristics of the intervention and control groups. In those that did, differences between the types of patients seen in outreach and hospital clinics often existed, with outreach clinics usually seeing a greater proportion of new referrals, and a broader range of problems. As such, most of the excluded studies could not rule out residual confounding of their results.
The CBA studies, however, were also potentially subject to residual confounding. Two were excluded from the analysis because the control group differed in important characteristics and was clearly inappropriate (Buhrich,Owen). Neither of the remaining two provided a detailed comparison of intervention and control groups (Howe, Tyrer). Both Howe and Tyrer also failed to account for unit of analysis error, making estimates of confidence intervals impossible without obtaining the data. Furthermore, neither of the included ITS studies commented in detail on factors other than outreach that could have been responsible for the observed effects on outpatient throughput (Gruen) nor psychiatric admission rates (Williams).
On the other hand, some large studies from the UK that were excluded on the EPOC design criteria (eg Black, Bowling, Gillam, Haynes, Helliwell, Walshe) appeared to be methodologically stronger in some respects than all four included CBA and ITS studies. As observational studies, they provide insights into the 'real‐world' implementation of outreach. The trade‐off is that they are less powerful than well‐designed intervention studies for attributing benefits or harms to the outreach intervention. We felt that these studies, in particular, added valuable supplementary information in this review. Essentially their findings both supported and added to the insights gained from the Included Studies. On very few accounts did the findings reported in the excluded studies contradict in important ways those of the studies meeting the EPOC design criteria.
02.02 Comparison with a previous systematic review of UK Studies
It is useful to examine these findings in light of the recent systematic review by Powell. Powell restricted the review to UK studies and used a search based on keywords, hand searches, internet searches, reference lists and contact with experts. He obtained 15 studies. We searched the international literature, including the Cochrane Library, with a combination of keyword and MeSH search terms, used article reference lists and referred to other known sources and obtained 73 reports of outreach, 28 of which were from the UK. Powell's review detected two of the three UK studies included in this review (Tyrer, O'Brien), but did not include Williams. Powell reviewed, in considerable detail, the evidence from the UK studies mentioned above. Our review of these studies concurs with his findings. Adding Williams and giving extra weight to O'Brien and Tyrer does not change the conclusions significantly, as Tyrer and Williams can only, at best, provide indirect evidence of improved outcomes and use of hospital services.
However Powell's review was largely a review of the 'simple' shifted outpatients model that has predominated in the UK. In our review, studies from outside the UK provided valuable insights about clinical outcomes and service utilisation when outreach formed part of more complex multifaceted interventions. Furthermore, our review highlights the important differences in outreach in different settings, and we caution against applying Powell 's conclusions regarding the UK experience, to other settings where they may not be relevant. 02.03 Distribution of evidence
We have clearly shown the distribution of research evidence is inequitable. The available high quality evidence is skewed to urban non‐disadvantaged populations, despite the fact that outreach is an intervention with relevance to both resource‐rich and resource‐poor settings. Many studies from the UK, USA, Europe and urban Australia were comparative, but the vast majority from rural Australia, rural Canada, Africa, South America, and the Middle East were descriptive studies. The latter populations may depend on visiting services for access to specialists, without whom care depends on local primary care staff. While these communities may have significant potential to benefit, the quality of studies seriously limits the ability to draw conclusions about whether specialist care leads to improved health outcomes and, if so, at what cost. Most of the high quality research is done in populations where the potential to benefit is marginal, and there is little evidence about the effectiveness of outreach in settings that have the most to gain. It is invalid to use the UK experience, for example, to predict the benefits and costs of outreach in rural or disadvantaged settings.
A variety of reasons could be responsible for the maldistribution of evidence. First, it may be a reflection of the under‐representation of journal content about the health needs of resource‐poor peoples and countries (Horton). Second, high quality comparative studies require good data, skills and other resources to conduct research, all of which are more available in affluent countries. Third, there may be less demand for rigorous evaluation in resource‐poor settings, where specialist outreach is usually an initiative of non‐government organizations, individuals or groups, and might not be a major policy consideration. Nonetheless, providers, patients, funders and other stakeholders in any setting have legitimate needs for evidence of effectiveness on which to justify establishment, continuation, modification or discontinuation of resource‐intensive activities such as specialist outreach.
3. Effectiveness of outreach
Evidence of clinical improvement is the outcome measure to which we assigned greatest importance. The three Seattle studies estimated that their interventions reduced by 37% the risk of the psychiatric disorder persisting for 6 months (RR 0.63, 95% CI 0.52‐0.77). Their interventions were complex, involving collaborative care, patient and physician education, and protocol driven management that, in effect, provided 'intensive outpatient care' for patients with depression or panic disorder. They also all used a similar urban Seattle‐based HMO population. The generalisability of their findings to different populations is unclear from this review, although it seems reasonable to predict that a similar intervention would have benefit in other urban populations for patients with psychiatric disorders.
Vierhout was the only other study to directly measure clinical outcomes, but did so to detect poorer clinical outcomes that could potentially result from reduced use of services. No worsening was found, and on one measure, a combined self‐reported measure of well‐being, the intervention led to clinical improvement. The significance of this in relation to outreach is uncertain. Both Tyrer and Williams used admission rates as an outcome. Given they were both studies of psychiatry outreach, admission can be interpreted as a poor clinical outcome. These were not high quality studies, but both showed lowering of psychiatric admission rates in association with outreach.
In the absence of measures of clinical outcome, we regarded access to specialist care, and quality of care determined by accepted guidelines, to be proxies for health outcomes. This is particularly relevant for populations without ready access to specialist services. O'Brien provided the sort of access data that is useful for this purpose, including patient‐centred measures of cost, time and distance, and of realised access using attendance rates. Although statistically significant, the effect size in O'Brien's study was small because the potential to benefit in an urban setting was relatively low. Unfortunately no such detailed examination of access exists in rural and disadvantaged contexts. Howe and Gruen provide indirect evidence of improved access in terms of increased relative (Howe) and absolute (Gruen) numbers of patients consulting with a specialist.
Howe and O'Brien both found specialist involvement was associated with approximately 8% more patients receiving guideline‐based standard of care in breast cancer treatment and orthodontic referral, respectively. In the former improved care may have made an unmeasured clinical difference, but in the latter it did not seem to have much effect on the number of patients ultimately booked for treatment. The Seattle studies of psychiatry found their intervention had greater benefit on adherence rates for medication (combined effect of relative risk of non‐adherence 0.62, 95% CI 0.49‐0.78), which may have contributed to improved clinical outcomes. Undertreated patients with depression may have considerably greater potential to benefit in terms of adherence to treatment and in short term measurable clinical outcomes than do patients with breast cancer in rural communities. Features of the context that influence the potential roles, benefits and costs of specialist outreach include factors relating to health needs and the potential to benefit, local health resources and access to hospital services, the structure of the health system and the role of specialists within it, and health care financing arrangements. We have found the distinction between urban, urban‐disadvantaged, rural and rural‐disadvantaged populations to be a useful one. Across these categories specialist outreach has been established for different reasons, the quality of evidence varies, and the methodology and outcomes differ.
03.01 Urban non‐disadvantaged populations
Urban non‐disadvantaged populations, when compared with rural or disadvantaged populations, have relatively little to gain from specialist outreach in terms of improving access to specialists and hospital services. This has been borne out in studies from the UK. O'Brien's measures of improved access were mostly issues of convenience (patients' travel savings averaged 22 pence, 16 minutes and 1.67 miles, and the time in the clinic was shortened by an average of 14 minutes). Patients reported valuing the convenience. These findings were also quite consistent across studies excluded from the review. There were some excluded studies, however, (eg Black) in which outreach performed worse on access indicators such as waiting time at the clinic. Furthermore, neither O'Brien nor the excluded studies suggested that the combination of improved access and patient preference for outreach clinics translated into consistently higher attendance rates or lasting health benefits. The only evidence of clinical outcomes from UK studies in the review came in the form of population‐based admission rates for psychiatric disorders (Tyrer; Williams) and may have been influenced by other health service changes. Outreach clinics in the UK have generally cost the health service more (due to less throughput and higher marginal overheads) and are therefore potentially less cost‐effective than hospital outpatient clinics. In an excluded study, Bowling modelled clinic characteristics and outcomes and concluded that the fact that outreach clinics were smaller led to most of their advantages. Consequently, increasing clinic size would reduce costs but would also reduce the advantages.
In summary, neither included nor excluded UK studies suggested that outreach in urban non‐disadvantaged settings provides any significant benefit in health improvements or the effectiveness of healthcare delivery. Outreach clinics may have benefits in terms of patient experience while hospital clinics have advantages in terms of efficiency and, in the end, specialist outreach in the UK has essentially been a trade off between patient convenience and health service costs. In fact, we found few examples where specialist outreach has been a planned policy initiative to improve access in urban non‐disadvantaged populations. Most outreach in the UK was fuelled by general practice fundholding, and was driven primarily by health system interests, rather than patient care interests. As fundholding declined in the late‐1990s, so did the amount of outreach activity (Martin Roland, personal communication, 24/10/2001). Furthermore, some of the excluded studies reported that specialists found outreach clinics inconvenient, and GPs had not had the interaction with specialists that was anticipated.
The UK experience helps to illustrate that moving specialist services to primary care settings does not necessarily change the way that consultants and GPs work and relate to each other (Black). The potential for simple outreach interventions to have any effect beyond repositioning the consultation was limited by practical constraints at the local level. What the UK picture does not portray, except in a few excluded reports written before fundholding (Marsh, McKechnie, Spencer), are any complex outreach interventions that explicitly aim to improve the processes of care and health outcomes.
In this sense, the multifaceted interventions in urban non‐disadvantaged populations in North America (Katon1, Katon2, Roy‐Byrne, Hess, Kates), Europe (Vierhout) and Australia (Carr, Seidel) are of particular interest. Many would regard them to be 'collaborative care' initiatives rather than 'outreach' per se, although visiting specialists were essential components of them. They represent outreach as a vehicle for integrating specialist services and community services in order to improve the processes of care, reduce inappropriate referrals and unnecessary tests, and ultimately improve health outcomes. Among the included studies, the RCTs of Katon1, Katon2, Roy‐Byrne and Vierhout provide good evidence that such end‐points are achievable. In the only detailed cost‐effectiveness analysis, a multifaceted intervention addressing major depression (Katon1) was more cost‐effective than hospital outpatient clinics. In urban non‐disadvantaged populations, therefore, the main potential benefits of specialist outreach activities appear to relate not to access, but to improving the processes of care, and this can only be achieved through complex multifaceted interventions. These findings are consistent with those of other systematic reviews of complex interventions that address a range of factors and barriers to best practice (Renders, Bower,Bero 1998).
In rural and disadvantaged populations, however, access issues are more salient, and outreach may be established with access, clinical process and/or health outcome goals in mind. There is, however, less evidence on which to base conclusions about its effectiveness in meeting such goals.
03.02 Urban disadvantaged populations
Urban disadvantaged populations, such as the homeless and mentally ill, are often marginalised and face a variety of social, cultural, language and health service barriers. Specialist outreach potentially has a very important role to play in overcoming these barriers. There were no Included studies in the systematic review fom urban disadvantaged populations. The overview identified examples of relevant interventions from inner‐city London (Hindler, Little, Strathdee2), Australia (Buhrich, Sewell) and a community of Yemenese immigrants in Israel (Weingarten) that involved regular consulting clinics in community locations such as primary care clinics, schools, refuges and shelters. Only the Australian studies were complex interventions, and none undertook a reliable comparison with other patterns of care. There is a clear need for more rigorous studies in these populations.
03.03 Rural populations
Rural communities possibly have the most to gain from outreach in terms of client outcomes. The fact that specialist services are usually disproportionately concentrated in major urban centres results in access inequities and can compound the fact that, in resource‐rich and resource‐poor countries alike, rural populations are usually less healthy. We defined rural non‐disadvantaged populations as those where patients must travel to another population centre for specialist or hospital care, but where access barriers are not so great that many patients would forego that care. Howe was the only Included study in the systematic review from this category. By finding that 8% more breast cancer patients received an oncology consultation and 7% more received guideline‐consistent care, Howe provided an indication that access to specialist care was improved. While the advantages of outreach in terms of cost and convenience could be considerable, no studies in this group examined specific dimensions of access.
The overview identified examples of specialists travelling alone by car or aeroplane to communities, and with large vehicles fitted with or carrying specialist equipment (Chelala, Hansom, Oboler). Clinics lasted from half a day to one week. Five comparative studies were identified: two in oncology (Howe, Desch), one in internal medicine (Oboler), one in ophthalmology (cataract surgery) (Haynes), and one in psychiatry (Owen). The excluded studies complement Howe in as much as they suggest that access to guideline‐consistent specialist care may be improved (Howe, Desch, Haynes), that it may be delivered safely in rural locations without compromising outcomes (Haynes, Howe, Desch), and that overall costs including costs borne by the patient may be similar (Oboler) or lower (Haynes, Desch) for the outreach group. While the quality of this evidence is mixed, it does make a reasonable argument that specialist care, including elective surgery, can be performed efficiently and effectively in rural centres. There is a need for more rigorous evaluation in rural populations, too.
03.04 Rural disadvantaged populations
We defined rural disadvantaged populations as those for whom the barriers faced in accessing specialist services are so great that many patients would forego specialist care if visiting services were not available. The only Included Study in the systematic review was Gruen's which demonstrated increased numbers of consultations (up to 390%) with specialists in conjunction with change in trend of annual hospital outpatient clinic consultations from positive to negative, leading to reduced use of hospital outpatient clinics. Otherwise questions about access, quality, health outcomes, service use and costs, relative to usual care processes, are unanswered.
The overview found other reports of specialist outreach serving rural populations in Africa (Coetzee, Cotton, Crosier, Hodges, Klein, Loefler, Raasen), and remote and Indigenous populations in Australia (Almehdi, Baker, Gruen, Humphrey, Kierath, Leaming, Parish) and Canada (Baskett, Martin). Many utilised aircraft and brought their own specialised equipment. Some performed major operative procedures, including cardiothoracic surgery (Klein) and others primarily provided a consulting service to integrate with treatment in the major hospital. Only one intervention (Baskett) reported local education and joint consultations as major components of the service. The remainder were simple outreach interventions.
The policy environment in these rural disadvantaged settings was diverse. Many outreach activities were initiatives of individuals and non‐government groups. In Australia, and possibly in Canada and South Africa, however, there were examples that were an integrated part of the health care system delivered through regional centres or academic institutions (Gruen, Almehdi, Humphrey, Kierath, Parish, Baskett, Martin, Coetzee, Crosier). Policy decisions confronted in these populations are often not just about whether to provide specialist outreach services, but also how much to invest in specialist services at all, as opposed to other services, including primary health care. Although there is little supporting or refuting evidence, multifaceted specialist outreach may in fact be a means for both improving access to specialists and bolstering local primary health care services. Like in the preceding two categories, there is a need to build a more rigorous evidence base for outreach interventions to rural‐disadvantaged areas.
4. Costs and harms associated with outreach
There are potential harms associated with specialist outreach. These include extra costs and additional resources required to provide the clinics, inefficient use of specialists' time when they could be consulting in their primary practice, and the opportunity costs associated with additional investment in the specialist sector. In urban settings, at least, the additional costs seem particularly related to fewer patients per clinic and the higher overhead or marginal costs in primary care settings than in hospital outpatient departments.
The fact that outreach delivered more cost effective care in Katon1 was an important finding, although relatively small additional expense was required and the population was urban psychiatric patients who seemed to have potential for considerable improvement. For rural and disadvantaged populations, where costs are greater but the potential to benefit is also greater, the question remains unanswered. The increased patient throughput in Gruen's study in a disadvantaged rural population was an indication that an unmet need or demand existed. An ongoing study is seeking to determine the nature of this increased demand and whether there is evidence of improved access on the basis of individualised needs (Gruen2).
Several UK commentaries and surveys of providers found specialists to be critical of outreach as an inefficient use of scarce specialist resources. On the other hand some showed specialists to be praising the effectiveness of outreach. No study formally compared providers' attitudes, and no study examined in detail the opportunity costs associated with specialists being absent from their primary practice. Additionally, no study particularly examined the opportunity costs associated with outreach when compared with investment in other parts of the health care sector.
5. Generalisability across specialties
Five of the nine included studies in the systematic review and 20 of the 73 studies in the overview related to psychiatry. The benefits associated with specialist psychiatrist visits, particularly when delivered as part of a multifaceted intervention, are consistent with the findings of an EPOC review of the effect on professional practice of visiting mental health workers in primary care (Bower). Given that one of the criticisms of outreach has been that many patients need to go to hospital for investigations or procedures anyway, non‐psychiatric and procedure‐based disciplines deserve special attention. The non‐psychiatric studies in the review included three surgical specialties and oncology. O'Brien found that simple orthodontic outreach visits reduced the total number and increased the appropriateness of referrals for hospital‐based treatment. Similarly, Vierhout demonstrated that joint consultations between orthopaedic surgeons and general practitioners reduced the the number of inpatient treatments, and also reduced the number of laboratory and radiology tests, without affecting clinical outcomes. Several comparative studies that were excluded from the review, however, did find that outreach clinics resulted in higher numbers of patients booked for inpatient treatment (Black, Bowling, Perrett and Riley). In rural settings, Howe found that outreach improved the proportion of patients receiving guideline‐consistent breast cancer care and, while Gruen did not examine hospital‐based investigation or treatment, this study showed that outreach in four surgical specialties increased numbers of consultations overall and reduced hospital outpatient visits. These effects correspond to reduced numbers of 'unnecessarily' duplicated tests, more on‐site procedures, coordination of complex care, and upskilling of primary care staff. The only non‐psychiatric studies to evaluate the outcomes of outreach‐based treatment were two comparative studies that were excluded from the review. Haynes found that cataract outcomes were no worse when the operation was done in a rural hospital during outreach visits. Bowling found perceived health improvement by patients, although the outreach population was generally healthier. In summary, therefore, there is little evidence suggesting that the overall benefits and costs of outreach are different for surgical or other high‐tech specialties than they are in psychiatry.
Authors' conclusions
Implications for practice.
The evidence presented provides support for the hypothesis that specialist outreach can improve access to specialist care on a range of patient‐based measures, health outcomes to a clinically important degee, and efficiency in the use of hospital‐based services by reducing duplication and unnecessary referrals and investigations. The pooled effect size in three similar RCTs is large (RR of 0.63 and 0.62 for poor clinical outcomes and non‐adherence to treatment, respectively), however given the relatively small size of these studies (560 patients in total), it may only take a single large study showing no effect to change this conclusion (although no such study has been reported). Outreach seems also to facilitate engagement between specialists and primary care practitioners, although such engagement cannot be presumed. Interaction is greatest when outreach is part of a complex multifaceted intervention which involves case‐conferences, joint consultations, seminars and education sessions, other health professionals or other care enhancements. On the basis of four RCTs, it appears that this can lead to improved health outcomes and more efficient use of services. Simple models in urban non‐disadvantaged settings, where outreach involves little more than a shift in the location of the consultation, confer relatively little benefit, limited to measures of convenience and patient satisfaction.
Outreach usually requires additional investment on the part of providers and health care systems when compared with hospital‐based care, although these extra costs may be at least partly offset by reduced costs for the patient and greater cost‐effectiveness of multifaceted interventions. Additional investment is most required when providing outreach to rural populations, and an increase in demand for hospital services may result from uncovering unmet needs.
Tailoring specialist services, including specialist outreach, will depend on an intimate understanding of local contexts. Allocation to one of urban, urban disadvantaged, rural, or rural disadvantaged populations is a helpful first step that allows the weight of the evidence to be considered. Finally, outreach appears to offer potential benefits to both procedural and non‐procedural specialties.
Implications for research.
The evidence located by the review is especially deficient for important subpopulations, namely rural and disadvantaged communities, who may benefit most from outreach interventions. Most studies addressing these populations were descriptive studies. The process of conducting an overview of all studies, and then stratifying the systematic review by population type, enabled the inequitable distribution of evidence about effectiveness to be identified and highlighted.
Health systems seeking to address inequalities in health and access to specialist care faced by rural and disadvantaged populations will need to build an evidence base on the effectiveness of outreach on important outcomes. Of particular relevance will be whether improved access to specialists results in improved health outcomes, and whether there is a difference between simple and multifaceted styles of service delivery in these environments. Cost‐effectiveness and equity considerations will require thoughtful evaluation particularly in places where specialist outreach may form an integral part of specialist medical services.
Many comparative studies of specialist outreach are post‐hoc analyses of services already established and for which evaluation had not been planned prior to implementing the intervention. As a consequence many studies have been unable to rule out potentially important confounders, and have been excluded from this review. This seems to be a greater problem than in systematic reviews of other primary care interventions (Renders; Bower). The conclusions from this review are therefore drawn on the basis of few studies and particular interventions. They provide insight into the sorts of outcomes that are achievable with specialist outreach. Confirmatory or contradictory evidence in similar interventions, and new evidence in other interventions and settings are needed to make more generalizable the conclusions about the merits of specialist outreach. Such studies will need to be designed with an eye to appropriate methods and control groups that minimise the risk of confounding and take into account clustering in design.
Finally some other important questions emerging from this review remain unanswered by the current research. First, the label 'multifaceted' has been useful for this review, but in fact it represents a heterogenous group of activities and forms of interaction between specialists and primary care practitioners. It will be important to tease out the types of interaction that are most beneficial for patient outcomes if the goals of improving care coordination and interaction between specialist and primary carers are to be achieved. Future studies could test the effectiveness of specialist outreach interventions against usual care, and against other forms of case management in primary care settings that do not involve visiting specialists. Second, opportunity costs, particularly the effect of specialists' absence from their main practice while doing outreach, have been alluded to but not examined in any detail. Third, it appeared that many of these outreach interventions lasted a finite period of time. If indeed there is benefit in delivering services in this fashion, as we conclude there can be, then the requirements for sustainable outreach are important issues. And fourth, the right balance of outreach and hospital‐based care must be tailored to each situation. It will be interesting to consider which of the benefits of multifaceted interventions can be achieved through other pathways and which are dependent for their implementation on visiting specialist services.
What's new
| Date | Event | Description |
|---|---|---|
| 31 July 2008 | Amended | Converted to new review format. |
History
Protocol first published: Issue 3, 2003 Review first published: Issue 1, 2004
| Date | Event | Description |
|---|---|---|
| 15 August 2003 | New citation required and conclusions have changed | Substantive amendment |
Acknowledgements
The reviewers would like to thank Jeremy Grimshaw, Merrick Zwarenstein and the Australasian Cochrane Centre for advice and support throughout, Cynthia Fraser for assistance with the search strategy and performing some of the searches, Sally Bailey for retrieval of articles, and Peter Morris for critical review of a previous draft.
Data and analyses
Comparison 1. Outreach v no outreach.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Measures of Access (continuous) | 1 | Std. Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 1.1 Distance travelled to appointment | 1 | Std. Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 1.2 Travel time to appointment | 1 | Std. Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 1.3 Cost for patient to attend appointment | 1 | Std. Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 1.4 Time spent in clinic at appointment | 1 | Std. Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 2 Measures of Access (categorical) | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 2.1 Difficulty parking | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 2.2 Cost of attendance is a problem | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 3 Realized Access (Attendance) | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 3.1 Not attending specialist appointment when made | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 4 Quality of care | 4 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 4.1 Inappropriate non‐referral when indicated | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 4.2 Inappropriate referrals when not indicated | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 4.3 Not prescribed appropriate type of care | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 4.4 Not prescribed appropriate dose/duration of treatment | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 4.5 Not adhering to treatment | 3 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 5 Health Outcomes (categorical) | 4 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 5.1 Persistent symptoms | 4 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 6 Health outcomes (continuous) | 1 | Std. Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 6.1 Persistent symptoms (days/6mo.) | 1 | Std. Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 7 Patient satisfaction | 4 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 7.1 Unsatisfied with overall care | 3 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 7.2 Unsatisfied with medication/treatment | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 7.3 Unsatisfied with setting | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 7.4 Prefer hospital clinic | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 8 Service utilisation (continuous) | 2 | Std. Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 8.1 Primary care consultations | 2 | Std. Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 9 Service utilisation (categorical) | 3 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 9.1 Other non‐hospital outpatient services | 2 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 9.2 Hospital‐based outpatient services | 2 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 9.3 Laboratory tests | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 9.4 Radiological tests | 2 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 9.5 Medication | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 9.6 Minor procedures (e.g. joint injections) | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 9.7 Planned (elective) inpatient services | 2 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 9.8 Primary care consultations | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
1.1. Analysis.
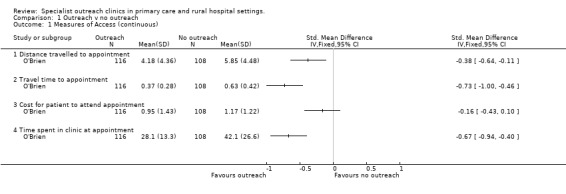
Comparison 1 Outreach v no outreach, Outcome 1 Measures of Access (continuous).
1.2. Analysis.
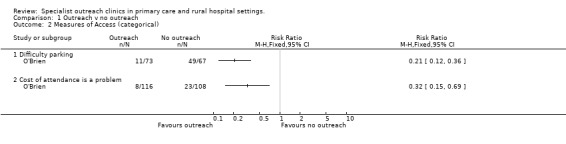
Comparison 1 Outreach v no outreach, Outcome 2 Measures of Access (categorical).
1.3. Analysis.
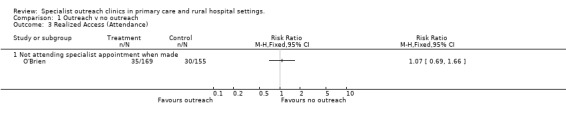
Comparison 1 Outreach v no outreach, Outcome 3 Realized Access (Attendance).
1.4. Analysis.
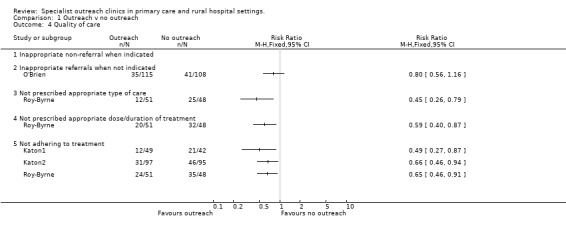
Comparison 1 Outreach v no outreach, Outcome 4 Quality of care.
1.5. Analysis.
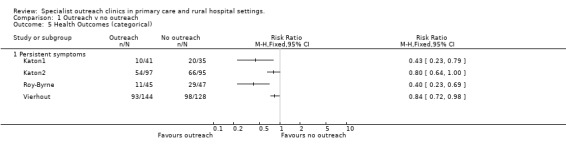
Comparison 1 Outreach v no outreach, Outcome 5 Health Outcomes (categorical).
1.6. Analysis.

Comparison 1 Outreach v no outreach, Outcome 6 Health outcomes (continuous).
1.7. Analysis.
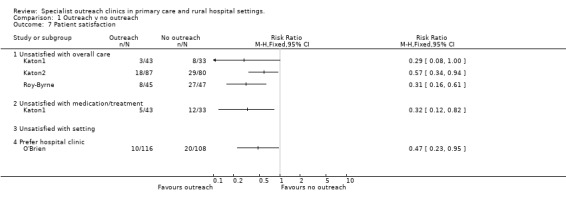
Comparison 1 Outreach v no outreach, Outcome 7 Patient satisfaction.
1.8. Analysis.
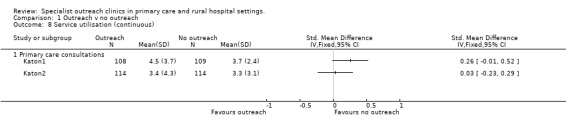
Comparison 1 Outreach v no outreach, Outcome 8 Service utilisation (continuous).
1.9. Analysis.
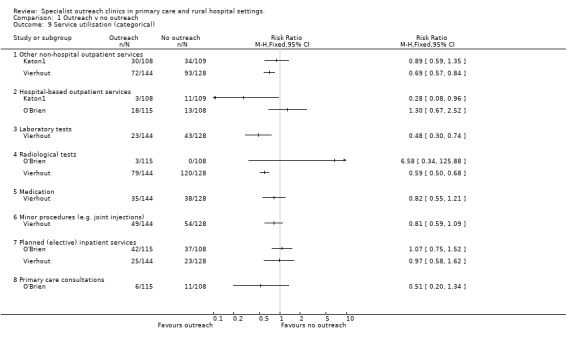
Comparison 1 Outreach v no outreach, Outcome 9 Service utilisation (categorical).
Characteristics of studies
Characteristics of included studies [ordered by study ID]
Gruen.
| Methods | Interrupted Time Series Analysis. Independent of other changes: NOT CLEAR Sufficient data points: DONE Test for trend: DONE, ARIMA/time‐series regression: NOT DONE Intervention independent of data collection: NOT DONE Blinded assessment: DONE Completeness of data: NOT CLEAR Reliable outcomes: DONE Shape of intervention effect prespecified: NOT CLEAR Rationale for number/spacing of data points: NOT DONE | |
| Participants | Australia, 33 remote Indigenous communities 50 ‐ 900 km from main hospital base, all patients with surgical, ophthalmological, obstetric/gynaecological, disorders consulting a specialist 1993‐1999. | |
| Interventions | Regular outpatient clinics in remote community primary care clinics, for consultation and minor procedures commencing 1997. Purpose: To improve access to appropriate specialist care. Comparison: Hospital outpatient clinics. | |
| Outcomes | Access: Total annual number of consultations with specialist, annual count of consultations at hospital outpatients. | |
| Notes | This publication has been independently assessed against EPOC inclusion and quality criteria by a reviewer external to this review group. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment? | Unclear risk | D ‐ Not used |
Howe.
| Methods | Controlled Before‐After Study (communities). Baseline: DONE Comparable control: NOT DONE Blinded assessment: NOT CLEAR Protection against contamination: DONE Reliable outcomes: NOT CLEAR Follow up: providers: DONE patients: DONE | |
| Participants | USA, 9 rural hospitals & clinics, all female Caucasian patients diagnosed with breast cancer 1986‐1991. | |
| Interventions | Regular oncologist visits to rural hospitals to consult and administer treatment protocols commencing 1988. Prior to this it had been identified that too few patients were receiving bilateral mammography or adequate tumour staging. Purpose: To improve rates of bilateral mammography, tumour staging, and 'state of the art' stage and type‐specific cancer care. Comparison: A program of audit and intervention feedback to family doctors and rural‐based specialists, without outreach. | |
| Outcomes | Quality of care: Guideline‐consistent care, including diagnostic and prognostic assessment, oncology consultation and appropriate treatment. Access: Proportion receiving oncology consult. | |
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment? | Unclear risk | D ‐ Not used |
Katon1.
| Methods | Randomised Controlled Trial (patients). Allocation concealment: NOT CLEAR Follow‐up: providers: DONE; patients: DONE Blinded assessment: DONE Baseline: DONE Reliable outcomes:DONE Protection against contamination: NOT CLEAR | |
| Participants | USA, 1 large urban HMO primary care clinic with 22 family physicians serving 22,000 adults; English‐speaking patients aged 18‐80 yrs with minor or major depression but without substance abuse, psychosis, suicidal ideation, dementia or terminal illness 1992‐1993. | |
| Interventions | Each patient has 2 or more consultations with psychiatrist in primary care clinic during week 2 (50 minutes) & week 4 (20 minutes) as part of collaborative care program which also included: physician education and regular case conferences, patient education utilising information booklets and videotapes, and individualised treatment algorithms. Purpose: To improved treatment of depression to the level recommended by practice guidelines in a way that is acceptable to patients and primary care providers, and improve short‐term clinical outcomes. Comparison: Usual care by family physician, of whom 31% were seen by other mental health worker, and 10% by a psychiatrist. | |
| Outcomes | Realised Access: Adherence to treatment at 30 days and 90 days; Patient satisfaction: Satisfaction with quality of care and with medication; Service utilisation: Additional PC physician visits in 1 yr; Additional mental health worker visits in 1 yr; Outpatient clinic psychiatric visits in 1 yr; Health outcomes: Improvement >=50% on SCL score at 7 months; Somatic distress; self rated overall health; time off work; restricted activity days; Cost: Cost per patient treated; Cost effectiveness; incremental cost effectiveness. | |
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment? | Unclear risk | B ‐ Unclear |
Katon2.
| Methods | Randomised Controlled Trial (patients). Allocation concealment: DONE Follow‐up: providers: DONE; patients: DONE Blinded assessment: DONE Baseline: DONE Reliable outcomes: NOT CLEAR Protection against contamination: DONE | |
| Participants | USA, 4 large urban primary care clinics, Adults with persistent major depression after 6‐8 weeks of treatment by primary care | |
| Interventions | 2 or more consultations by psychiatrist in primary care clinic as part of stepped collaborative care involving an educational book/videotape, tailored antidepressant therapy, other community‐based resources and ongoing monitoring of adherence to medication; comparison with usual care. Purpose: Test if pts with persistent depressions 6‐8 wks after routine primary care Rx who then got collaborative care had more adequate guideline‐based Rx, were more satisfied, & had better outcomes over 6mo. Comparison: Usual care by primary care physician, which included referral of 27.2% to a nonstudy mental health specialist (compared with 24.6% of intervention patients, p=0.76). | |
| Outcomes | Health outcomes: Depression free days. Patient satisfaction: Adherence to treatment; satisfaction score. Costs: overall; per patient. | |
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment? | Low risk | A ‐ Adequate |
O'Brien.
| Methods | Randomised Controlled Trial (patients). Allocation concealment: DONE Follow‐up: providers: DONE; patients: NOT DONE Blinded assessment: DONE Baseline: NOT CLEAR Reliable outcomes: DONE Protection against contamination: DONE | |
| Participants | Manchester, UK, 3 urban community health centres 0.5‐15km from main base, patients referred for orthodontic consultation | |
| Interventions | Orthodontic triage consultation in community clinic. Purpose: To test null hypothesis: No difference between the main base and outreach consultation service in: consumer's access to care, views of their care, outcome of consults & costs to consumers. Comparison: Hospital outpatient clinics | |
| Outcomes | Access: Waiting time and total time in clinic; attendance rate; patient costs. Satisfaction: Satisfaction measure; preference for site; Cost: Treatment cost Quality: proportion accepted for orthodontic treatment. | |
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment? | Low risk | A ‐ Adequate |
Roy‐Byrne.
| Methods | Randomised Controlled Trial (patients). Allocation concealment: DONE Follow‐up: providers: DONE; patients: DONE (up to 6 mo, NOT DONE >6mo) Blinded assessment: DONE Baseline: DONE Reliable outcomes: NOT CLEAR Protection against contamination: NOT DONE | |
| Participants | USA, 3 urban primary care clinics in Seattle ‐ 2 university‐associated internal medicine clinics with 30 physicians serving 8000 & 6000 people 50‐60% privately insured; and 1 community family clinic with 8 physicians serving 10000 people 80‐90% with private insurance. English‐speaking patients 18‐65 y.o. who had panic disorder, but who did not have substance abuse, suicidal ideation, psychosis, terminal illness, dementia, or pregnancy. 37% were minority groups, 36% unemployed. | |
| Interventions | 2 consultations with psychiatrist in 1st 8 weeks, as part of collaborative care involving physician education, patient education with videotapes, and improved care which also included structured program of 2 or more telephone calls in 1st 8 weeks, then regular calls over 12 months, leading to medication adjustment and feedback to providers. Purpose: To improve proportion of patients receiving adequate pharmacotherapy, improve clinical and functional outcomes and improve patients' satisfaction with their care compared with usual care by primary care providers. Comparison: Usual care by primary care providers, of whom 25% were referred to a psychiatrist. | |
| Outcomes | Realised access / Appropriate care: Proportion of patients receiving appropriate type, dose & duration of medication; Proportion adherent for at least 25 days/month. Patient satisfaction: Satisfaction with care. Health outcomes: Severity of panic disorder (measured on Panic Disorder Severity Scale and the Anxiety Sensitivity Inventory). | |
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment? | Low risk | A ‐ Adequate |
Tyrer.
| Methods | Controlled Before‐After Study (regions). Baseline: DONE Comparable control: NOT DONE Blinded assessment: DONE Protection against contamination: DONE Reliable outcomes: DONE Follow up: providers: DONE patients: DONE | |
| Participants | East Nottingham, UK, general practice clinics in urban area of 130 km2, patients 15‐64 years old contacting specialist psychiatric services 1978‐1985. | |
| Interventions | Specialist psychiatric clinics based in general practice clinics. Increased in number after sectorization 1981. Purpose: To improve liaison and collaboration between general practice and psychiatry. Comparison: Rest of Nottingham where outreach clinics less prevalent. | |
| Outcomes | Service utilisation: Admissions to hospital, proportion receiving inpatient care. | |
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment? | Unclear risk | D ‐ Not used |
Vierhout.
| Methods | Randomised Controlled Trial (patients). Allocation concealment: DONE Follow‐up: providers: DONE patients: DONE Blinded assessment: DONE Baseline: DONE Reliable outcomes: DONE Protection against contamination: DONE | |
| Participants | Holland, urban & rural primary care clinics, patients 10‐75 y.o. with orthopaedic problems | |
| Interventions | Joint consultation between orthopaedic surgeon, 3 general practitioners & patient in primary care setting. Purpose: To determine the extent to which unnecessary interventions and referrals can be prevented and whether more efficient care can be provided. Comparison: Usual care, which may or may not involve referral to hospital outpatients. | |
| Outcomes | Health outcomes: symptom free at one year; Service utilisation: referrals to hospital outpatients; diagnostic tests in 1 year; PC Integration: Knowledge of primary care practitioners | |
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment? | Low risk | A ‐ Adequate |
Williams.
| Methods | Interrupted Time Series Analysis. Independent of other changes: NOT DONE Sufficient data points: DONE Test for trend: DONE, ARIMA/time‐series regression: NOT DONE Intervention independent of data collection: DONE Blinded assessment: DONE Completeness of data: DONE Reliable outcomes: DONE Shape of intervention effect prespecified: NOT CLEAR Rationale for number/spacing of data points: NOT DONE | |
| Participants | UK, ecological study of all urban & rural admissions for psychiatric disorders. | |
| Interventions | Psychiatric consultations in primary care settings, assessed by relative prevalence of outreach in large administrative regions. Purpose: To improve the standard of clinical care and prevent hospital admissions. | |
| Outcomes | Service utilisation: Admissions for psychotic and non‐psychotic conditions.Service utilisation: Admissions for psychotic and non‐psychotic conditions. | |
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment? | Unclear risk | D ‐ Not used |
Characteristics of excluded studies [ordered by study ID]
| Study | Reason for exclusion |
|---|---|
| Almehdi | Descriptive study |
| Ayshford | Descriptive study |
| Bailey | Descriptive study |
| Baker | Descriptive study, before‐after comparison of procedures with no control |
| Baskett | Descriptive study |
| Baumann | Descriptive study |
| Black | Comparative study without pre‐intervention measures |
| Blair | Descriptive study. Community‐based paediatrician without hospital base. |
| Bowling | Comparative study with matched controls of treating clinician,, but without pre‐intervention measures. RCT design rejected on basis of cost. |
| Bruusgaard | Descriptive study |
| Buck | Descriptive study |
| Buhrich | CBA with inappropriate control group (non‐attenders at the clinic) |
| Carr | Descriptive study |
| Chelala | Descriptive study |
| Coetzee | Descriptive study |
| Cotton | Descriptive study |
| Crosier | Descriptive study |
| Cybulska | Descriptive study |
| Dart | Descriptive study |
| Desch | Before‐after study with no control group |
| Feild | Descriptive study |
| Gillam | Comparative study with no preintervention measures |
| Hansom | Descriptive study |
| Haynes | Comparative study with inappropriate control group (and no pre‐intervention measures) |
| Helliwell | Comparative study with inappropriate control group (and no pre‐intervention measures) |
| Hess | Descriptive study |
| Hindler | Descriptive study |
| Ho | Comparative study with inadequate control of confounders and no pre‐intervention measures |
| Hodges | Descriptive study |
| Humphrey | Descriptive study |
| Hunter | Descriptive study |
| Kates | Comparative study of presenting complaints without attempting to control for confounders. |
| Kerrison | Descriptive study |
| Kierath | Descriptive study |
| Klein | Descriptive study |
| Leaming | Descriptive study |
| Little | Descriptive study |
| Loefler | Descriptive study |
| Marsh | Descriptive study |
| Martin | Descriptive study |
| McKechnie | Comparative study of presentations without attempting to control for casemix. |
| Nakar | Descriptive study |
| Oboler | Simple cost comparison without controlling for casemix |
| Owen | CBA with inappropriate control group (urban population with regular specialist care versus rural with intermittent specialist care). |
| Parish | Descriptive study |
| Payne | Descriptive study |
| Perrett | Comparative study with inadequate control of confounders and no pre‐intervention measures |
| Pullen | Descriptive study |
| Raasen | Descriptive study |
| Riley | Comparative study with inadequate control of confounders |
| Seidel | Descriptive study |
| Sewell | Descriptive study |
| Shanon | Descriptive study |
| Shulman | Descriptive study |
| Spencer | Comparative study of presentations with no attempt to control for confounders or casemix |
| Steele | Descriptive study |
| Strathdee1 | Descriptive study |
| Strathdee2 | Descriptive study |
| Toupin | Descriptive study |
| Wagstaff | Descriptive study |
| Wakefield | Descriptive study |
| Walshe | Comparative study with no pre‐intervention measures |
| Weingarten | Descriptive study |
| Zegleman | Comparative study with no controlling for confounders and casemix |
Characteristics of ongoing studies [ordered by study ID]
Gruen2.
| Trial name or title | Specialist outreach to Indigenous communities of Australia's Northern Territory: effects on utilisation, access and health outcomes. |
| Methods | |
| Participants | Remote Indigenous communities in Australia's Northern Territory (as described in Gruen 2001) |
| Interventions | Outreach as described in Gruen 2001 |
| Outcomes | Rate of completion of referrals, rate of definitive treatment. |
| Starting date | January 2000. |
| Contact information | russg@menzies.edu.au |
| Notes | further study of the outreach intervention described in Gruen 2000. |
Contributions of authors
All authors were responsible for the planning of the review and writing the protocol. Russell Gruen conducted the searches, extracted and recorded trial data and wrote the main draft of the review. Tarun Weeramanthri, Ross Bailie and Stephen Knight extracted data and assisted in the writing of the review.
Sources of support
Internal sources
Menzies School of Health Research, Australia.
Flinders University of South Australia, Australia.
Northern Territory Department of Health and Community Services, Australia.
KwaZulu‐Natal Health Department, South Africa.
University of Natal, South Africa.
External sources
National Health and Medical Research Council, Australia.
Royal Australasian College of Surgeons, Australia.
Australasian Cochrane Centre, Australia.
Declarations of interest
The contact reviewer (RG) and two co‐reviewers (RB & TW) are authors of an included study (Gruen). This study was identified in the Medline section of the search, and has been independently assessed against the EPOC inclusion and quality criteria by a reviewer external to the review group.
Edited (no change to conclusions)
References
References to studies included in this review
Gruen {published and unpublished data}
- Gruen RL, Bailie R. Evaluation of the Specialist Outreach Service in the Top End of the Northern Territory. (Menzies Occasional Papers). Darwin, Australia: Menzies School of Health Research, February 2000. [ISBN 0 9587722 3 1] [Google Scholar]
- Gruen RL, Bailie RS, d'Abbs PH, O'Rourke IC, O'Brien MM, Verma N. Improving access to specialist care for remote Aboriginal communities: evaluation of a specialist outreach service. Med J Aust 2001;174(10):507‐511. [DOI] [PubMed] [Google Scholar]
Howe {published data only}
- Howe HL, Katterhagen JG, Yates J, Lehnherr M. Urban‐rural differences in the management of breast cancer. Cancer Causes Control 1992;3(6):533‐539. [DOI] [PubMed] [Google Scholar]
- Howe HL, Lehnherr M, Katterhagen JG. Education of rural physicians about breast cancer through an oncology outreach program. Public Health Rep 1994;109(6):804‐808. [PMC free article] [PubMed] [Google Scholar]
- Howe HL, Lehnherr M, Katterhagen JG. Effects of physician outreach programs on rural‐urban differences in breast cancer management. J Rural Health 1997;13(2):109‐117. [DOI] [PubMed] [Google Scholar]
- Katterhagen JG, Howe HL, Lehnherr M. Effectiveness of an oncology outreach program. Journal of Oncology Management 1993;March/April:32‐37. [Google Scholar]
Katon1 {published and unpublished data}
- Katon W, Korff M, Lin E, Simon G, Walker E, Bush T, et al. Collaborative management to achieve depression treatment guidelines. J Clin Psychiatry 1997;58(Suppl 1):20‐23. [PubMed] [Google Scholar]
- Katon W, Korff M, Lin E, Walker E, Simon GE, Bush T, et al. Collaborative management to achieve treatment guidelines. Impact on depression in primary care. JAMA 1995;273(13):1026‐1031. [PubMed] [Google Scholar]
- Simon GE, Katon W, Rutter C, VonKorff M, Lin E, Robinson P, et al. Impact of improved depression treatment in primary care on daily functioning and disability. Psychol Med 1998;28(3):693‐701. [DOI] [PubMed] [Google Scholar]
- Korff M, Katon W, Bush T, Lin EH, Simon GE, Saunders K, et al. Treatment costs, cost offset, and cost‐effectiveness of collaborative management of depression. Psychosom Med 1998;60(2):143‐149. [DOI] [PubMed] [Google Scholar]
Katon2 {published data only}
- Katon W, Korff M, Lin E, Simon G, Walker E, Unutzer J, et al. Stepped collaborative care for primary care patients with persistent symptoms of depression: a randomized trial. Arch Gen Psychiatry 1999;56(12):1109‐1115. [DOI] [PubMed] [Google Scholar]
- Simon GE, Katon WJ, VonKorff M, Unutzer J, Lin EH, Walker EA, et al. Cost‐effectiveness of a collaborative care program for primary care patients with persistent depression. Am J Psychiatry 2001;158(10):1638‐1644. [DOI] [PubMed] [Google Scholar]
O'Brien {published data only}
- O'Brien K, Mattick R, Mandall N, Wright J, Conboy F, Gosden T. Are specialist outreach clinics for orthodontic consultation effective? A randomised controlled trial. Br Dent J 2001;191(4):203‐207. [DOI] [PubMed] [Google Scholar]
Roy‐Byrne {published and unpublished data}
- Roy‐Byrne PP, Katon W, Cowley DS, Russo J. A randomized effectiveness trial of collaborative care for patients with panic disorder in primary care. Arch Gen Psychiatry 2001;58(9):869‐876. [DOI] [PubMed] [Google Scholar]
Tyrer {published data only}
- Ferguson B. Psychiatric clinics in general practice: an asset for primary care. Health Trends 1987;19:22. [PubMed] [Google Scholar]
- Ferguson B, Cooper S, Brothwell J, Markantonakis A, Tyrer P. The clinical evaluation of a new community psychiatric service based on general practice psychiatric clinics. Br J Psychiatry 1992;160:493‐497. [DOI] [PubMed] [Google Scholar]
- Tyrer P. Psychiatric clinics in general practice. An extension of community care. Br J Psychiatry 1984;145:9‐14. [DOI] [PubMed] [Google Scholar]
- Tyrer P, Ferguson B, Wadsworth J. Liaison psychiatry in general practice: the comprehensive collaborative model. Acta Psychiatr Scand 1990;81:359‐363. [DOI] [PubMed] [Google Scholar]
- Tyrer P, Seivewright N, Wollerton S. General practice psychiatric clinics. Impact on psychiatric services. Br J Psychiatry 1984;145:15‐19. [DOI] [PubMed] [Google Scholar]
Vierhout {published data only}
- Vierhout WP, Knottnerus JA, Ooij A, Crebolder HF, Pop P, Wesselingh‐Megens AM, et al. Effectiveness of joint consultation sessions of general practitioners and orthopaedic surgeons for locomotor‐system disorders. Lancet 1995;346(8981):990‐994. [DOI] [PubMed] [Google Scholar]
Williams {published data only}
- Strathdee G, Williams P. A survey of psychiatrists in primary care: the silent growth of a new service. J R Coll Gen Pract 1984;34(268):615‐618. [PMC free article] [PubMed] [Google Scholar]
- Williams P, Balestrieri M. Psychiatric clinics in general practice: do they reduce admissions?. Br J Psychiatry 1989;154:67‐71. [DOI] [PubMed] [Google Scholar]
- Williams P, Strathdee G. The expanding concept of community psychiatric care: the British experience. New Trends in Experimental & Clinical Psychiatry 1985;2:217‐228. [Google Scholar]
References to studies excluded from this review
Almehdi {published data only}
- Almehdi R, Wisnewski A, Gallery R, Donnelly P. Surgical outreach in Queensland's far northwest: preliminary analysis of a new surgical service. Aust NZ J Surg 2001;71(5Suppl):RS20. [Google Scholar]
Ayshford {published data only}
- Ayshford CA, Johnson AP, Chitnis JG. What is the value of ENT specialist outreach clinics?. Journal of Laryngology & Otology 2001;115(6):441‐443. [DOI] [PubMed] [Google Scholar]
Bailey {published data only}
- Bailey J, Black M, Wilkin D. Outreach clinics. The special branch. Health Service Journal 1994;104(5413):30‐31. [PubMed] [Google Scholar]
- Bailey JJ, Black ME, Wilkin D. Specialist outreach clinics in general practice. BMJ 1994;308(6936):1083‐1086. [DOI] [PMC free article] [PubMed] [Google Scholar]
Baker {published data only}
- Baker JW, Buttini MJ. The Flying Obstetric and Gynaecology Service in rural Queensland: its first two years. Med J Aust 1991;154(9):578‐582. [DOI] [PubMed] [Google Scholar]
- Cyna AM. Anaesthesia in rural Queensland: clinical experience with the Flying Obstetric and Gynaecology Service. Anaesthesia & Intensive Care 1993;21(6):831‐836. [DOI] [PubMed] [Google Scholar]
Baskett {published data only}
- Baskett TF. A university department's involvement with medical care in the Canadian North. CMAJ: Canadian Medical Association Journal 1979;120(3):298‐300. [PMC free article] [PubMed] [Google Scholar]
- Baskett TF. Obstetric care in the central Canadian Arctic. BMJ 1978;2(6143):1001‐1004. [DOI] [PMC free article] [PubMed] [Google Scholar]
Baumann {published data only}
- Baumann RJ, Leonidakis MG. Extending neurologic services to rural children. Neurology 1979;29(9 Pt 1):1298‐30. [DOI] [PubMed] [Google Scholar]
Black {published data only}
- Black M, Gosden T, Leese B, Mead N. Outreach clinics in dermatology and orthopaedics.. In: Roland M, Shapiro J editor(s). Specialist outreach clinics in general practice. Abingdon: Radcliffe Medical Press, 1998:49‐57. [Google Scholar]
- Black M, Leese B, Gosden T, Mead N. Specialist outreach clinics in general practice: what do they offer?. Br J Gen Pract 1997;47(422):558‐561. [PMC free article] [PubMed] [Google Scholar]
- Gosden T, Black M, Mead N, Leese B. The efficiency of specialist outreach clinics in general practice: is further evaluation needed?. J Health Serv Res Policy 1997;2(3):174‐179. [DOI] [PubMed] [Google Scholar]
- Leese B, Mead N, Black M, Gosden T. Outreach clinics. Dr Livingstone, I presume?. Health Serv J 1996;106(5532):24‐26. [PubMed] [Google Scholar]
Blair {published data only}
- Blair M, Horn N, Polnay L. General practitioners use of hospital and community based paediatric out‐patient services in Nottingham. Public Health 1997;111(2):97‐100. [DOI] [PubMed] [Google Scholar]
- Blair ME, Pullan CR, Rands CE, Crown N. Community paediatrics moves on‐an analysis of changing work patterns 1994‐97. Public Health 2000;114(1):61‐64. [PubMed] [Google Scholar]
Bowling {published data only}
- Bond M, Bowling A, Abery A, McClay M, Dickinson E. Evaluation of outreach clinics held by specialists in general practice in England. J Epidemiol Community Health 2000;54(2):149‐156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bowling A, Bond M. A national evaluation of specialists' clinics in primary care settings. Br J Gen Pract 2001;51(465):264‐269. [PMC free article] [PubMed] [Google Scholar]
- Bowling A, Stramer K, Dickinson E, Windsor J, Bond M. Evaluation of specialists' outreach clinics in general practice in England: process and acceptability to patients, specialists, and general practitioners. J Epidemiol Community Health 1997;51(1):52‐61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bowling A, Stramer K, Dickinson E, Windsor J, Bond M, Abery A. Outreach clinics in rheumatology, ENT and gynaecology.. In: Roland M, Shapiro J editor(s). Specialist outreach clinics in general practice. Abingdon: Radcliffe Medical Press, 1998:37‐48. [Google Scholar]
Bruusgaard {published data only}
- Bruusgaard D. Specialists visiting rural general practices in Norway. Journal of the Royal College of General Practitioners ‐ Occasional Paper 1980;10:1‐2. [Google Scholar]
Buck {published data only}
- Buck K. Issues in establishing rural ophthalmic surgery centers. College Review 1987;4(2):43‐52. [PubMed] [Google Scholar]
Buhrich {published data only}
- Buhrich N. Success of a psychiatric outreach service based in refuges for the homeless.. Med J Aust 1991;154(3):218. [DOI] [PubMed] [Google Scholar]
- Buhrich N, Teesson M. Impact of a psychiatric outreach service for homeless persons with schizophrenia. Psychiatr Serv 1996;47(6):644‐646. [DOI] [PubMed] [Google Scholar]
Carr {published data only}
- Carr VJ, Donovan P. Psychiatry in general practice. A pilot scheme using the liaison‐ attachment model. Med J Aust 1992;156(6):379‐382. [PubMed] [Google Scholar]
Chelala {published data only}
- Chelala C. Bringing surgery to the rural areas of Ecuador. Lancet 1998;352(9129):715. [DOI] [PubMed] [Google Scholar]
Coetzee {published data only}
- Coetzee MJ, Badenhorst PN, Loux EP, Doman MJ. Haematology outreach clinics in the Free State and northern Cape. S Afr Med J 1998;88(6):702‐706. [PubMed] [Google Scholar]
Cotton {published data only}
- Cotton MH. Five years as a flying surgeon in Zimbabwe. World J Surg 1996;20(8):1127‐1130. [DOI] [PubMed] [Google Scholar]
Crosier {published data only}
- Crosier J, Stoops N, Goldschmidt RB. Rural orthopaedic services in the Western Cape. South African Bone and Joint Surgery 1994;4(1):8‐10. [Google Scholar]
Cybulska {published data only}
- Cybulska BA. AGUM meeting on Outreach Services in Genitourinary Medicine. Association of Genito‐Urinary Medicine. International Journal of STD & AIDS 1997;8(5):347‐350. [DOI] [PubMed] [Google Scholar]
Dart {published data only}
- Dart JK. Eye disease at a community health centre. Br Med J (Clin Res Ed) 1986;293(6560):1477‐1480. [DOI] [PMC free article] [PubMed] [Google Scholar]
Desch {published data only}
- Desch CE, Grasso MA, McCue MJ, Buonaiuto D, Grasso K, Johantgen MK, et al. A rural cancer outreach program lowers patient care costs and benefits both the rural hospitals and sponsoring academic medical center. J Rural Health 1999;15(2):157‐167. [DOI] [PubMed] [Google Scholar]
- Smith TJ, Desch CE, Grasso MA, McCue MJ, Buonaiuto D, Grasso K, et al. The rural cancer outreach program: clinical and financial analysis of palliative and curative care for an underserved population. Cancer Treat Rev 1996;22(Suppl A):97‐101. [DOI] [PubMed] [Google Scholar]
Feild {published data only}
- Feild CR, Dungan WT, Marsh C, Patterson P, Fiser RH, Jr. Pediatric outreach clinics in Arkansas. J Ark Med Soc 1989;86(7):270‐271. [PubMed] [Google Scholar]
Gillam {published data only}
- Gillam SJ. Ophthalmology outreach clinics in Barnet.. In: Roland M, Shapiro J editor(s). Specialist outreach clinics in general practice. Abingdon: Radcliffe Medical Press, 1998:59‐68. [Google Scholar]
- Gillam SJ, Ball M, Prasad M, Dunne H, Cohen S, Vafidis G. Investigation of benefits and costs of an ophthalmic outreach clinic in general practice. Br J Gen Pract 1995;45(401):649‐652. [PMC free article] [PubMed] [Google Scholar]
Hansom {published data only}
- Hansom T. Outreach program in paediatrics. Dimensions in Health Service 1979;56(5):54‐58. [PubMed] [Google Scholar]
Haynes {published data only}
- Haynes R, Gale S, Mugford M, Davies P. Cataract surgery in a community hospital outreach clinic: patients' costs and satisfaction. Soc Sci Med 2001;53(12):1631‐1640. [DOI] [PubMed] [Google Scholar]
Helliwell {published data only}
- Helliwell PS. Comparison of a community clinic with a hospital out‐patient clinic in rheumatology. Br J Rheumatol 1996;35(4):385‐388. [DOI] [PubMed] [Google Scholar]
Hess {published data only}
- Hess M. The new Palliative Care Outreach Program‐‐a resounding success. Supportive Care in Cancer 1999;7(5):298‐301. [DOI] [PubMed] [Google Scholar]
Hindler {published data only}
- Hindler C. Psychiatric outreach clinics held in a general practice setting and community mental health care. Psychiatric Bulletin 2002;19(8):470‐474. [Google Scholar]
Ho {published data only}
- Ho TP, Luk C. Comparison of child psychiatric patients in hospital and community clinics in Hong Kong. Gen Hosp Psychiatry 1997;19(5):362‐369. [DOI] [PubMed] [Google Scholar]
Hodges {published data only}
- Hodges AM, Hodges SC. A rural cleft project in Uganda. Br J Plast Surg 2000;53(1):7‐11. [DOI] [PubMed] [Google Scholar]
Humphrey {published data only}
- Humphrey MD, Dudgeon GI, Smith WJ. FROGS (Far North Regional Obstetric and Gynaecological Service) ‐ the development of a specialist outreach service. Australian Health Review 1993;16(3):268‐272. [PubMed] [Google Scholar]
Hunter {published data only}
- Hunter D, McCance C. Referral to the psychiatric services by general practitioners in relation to the introduction of sessions by psychiatrists in health centres. Health Bulletin (Scotland) 1983;41:78‐83. [PubMed] [Google Scholar]
Kates {published data only}
- Kates N. Psychiatric consultation in the family physician's office: advantages and hidden benefits. Gen Hosp Psychiatry 1988;10:431‐437. [DOI] [PubMed] [Google Scholar]
- Kates N, Craven MA, Crustolo AM, Nikolaou L, Allen C, Farrar S. Sharing care: the psychiatrist in the family physician's office. Canadian Journal of Psychiatry ‐ Revue Canadienne de Psychiatrie 1997;42(9):960‐965. [DOI] [PubMed] [Google Scholar]
Kerrison {published data only}
- Kerrison S, Corney R. Private provision of 'outreach' clinics to fundholding general practices in England. J Health Serv Res Policy 1998;3(1):20‐22. [DOI] [PubMed] [Google Scholar]
Kierath {published data only}
- Kierath A, Hamdorf JM, House AK, House J. Developing visiting surgical services for rural and remote Australian communities. Med J Aust 1998;168(9):454‐457. [DOI] [PubMed] [Google Scholar]
Klein {published data only}
- Klein M, Ramoroko SP, Jacobs AG, Bomela MD, Mokhobo KP, Mohlala ML. The Cardiothoracic Outreach Programme‐‐a pilot project. S Afr Med J 1996;86(12):1533‐1535. [PubMed] [Google Scholar]
Leaming {published data only}
- Leaming DB, Smith AG. The Flying Surgeon Service of Queensland. Med J Aust 1970;1(8):387‐390. [PubMed] [Google Scholar]
Little {published data only}
- Little BC, Aylward GW, Gregson R, Wormald R, Courtney P. Community ophthalmology pilot study. Eye 1993;7(Pt 1):180‐183. [DOI] [PubMed] [Google Scholar]
Loefler {published data only}
- Loefler IJP. Visits to out‐lying hospitals. Proc Assoc Surg East Aftrica 1986;9:32‐36. [Google Scholar]
Marsh {published data only}
- Marsh GN, Tompkins AB. Paediatric care in general practice: an experiment in collaboration. Br Med J 1969;2(649):106‐108. [DOI] [PMC free article] [PubMed] [Google Scholar]
Martin {published data only}
- Martin B, Smith W, Orr P, Guijon F. Investigation and management of cervical intraepithelial neoplasia in Canadian Inuit: enhancing access to care. Arctic Med Res 1995;54 Suppl 1:117‐121. [PubMed] [Google Scholar]
McKechnie {published data only}
- McKechnie AA, Philip AE, Ramage JG. Psychiatric services in primary care: specialized or not?. J R Coll Gen Pract 1981;31(231):611‐614. [PMC free article] [PubMed] [Google Scholar]
Nakar {published data only}
- Nakar S, Vinker S, Lustman A, Hyam E. Outreach clinics in Israel: a common but unregulated phenomenon. Fam Pract 1999;16(2):149‐151. [DOI] [PubMed] [Google Scholar]
Oboler {published data only}
- Oboler SK, Blieden MA, Carter SA, Jahnigen DW, Luck TC, Mathew M, et al. A mobile internal medicine clinic. Arch Intern Med 1983;143(1):97‐99. [PubMed] [Google Scholar]
Owen {published data only}
- Owen C, Tennant C, Jessie D, Jones M, Rutherford V. A model for clinical and educational psychiatric service delivery in remote communities. Aust N Z J Psychiatry 1999;33(3):372‐378. [DOI] [PubMed] [Google Scholar]
Parish {published data only}
- Parish D. 'The eye mob comin': Eye health services to Aboriginal people in the Northern Territory 1982‐1998. Darwin: NT Aboriginal Eye Health Committee (NTAEHC), 1999. [Google Scholar]
Payne {published data only}
- Payne KA, Muller CJ, Coetzee AR. Rural anaesthetic practices in the western Cape. S Afr Med J 1991;80(1):21‐22. [PubMed] [Google Scholar]
Perrett {published data only}
- Perrett K. Sheffield.. In: Roland M, Shapiro J editor(s). Specialist outreach clinics in general practice. Abingdon: Radcliffe Medical Press, 1998:9‐15. [Google Scholar]
- Perrett K. Specialist outreach clinics in Sheffield: a faster tier of out‐patient provision for the patients of fundholding GPs?. J Public Health Med 1997;19(3):347‐353. [DOI] [PubMed] [Google Scholar]
Pullen {published data only}
- Pullen IM, Yellowlees P. Scottish psychiatrists in primary care: a silent majority. Br J Psychiatry 1988;153:663‐666. [DOI] [PubMed] [Google Scholar]
Raasen {published data only}
- Furnas DW, Gilchrist D, Rees TD, Wood AM. Surgical safaris with the East Africa Flying Doctor Service. Arch Surg 1979;114(10):1143‐1148. [DOI] [PubMed] [Google Scholar]
- Rees TD, Raassen T, Wachira J, Mustafa O, Adams‐Ray B. The flying doctors of east Africa. Bull Am Coll Surg 1994;79(10):12‐20. [PubMed] [Google Scholar]
- Wachira J, Nordberg E. Airborne surgical outreach services in eastern Africa [see comments]. East Afr Med J 1998;75(10):563‐566. [PubMed] [Google Scholar]
- Wood AM. Surgery in East Africa: Communications. Proc Assoc Surg East Aftrica 1981;4:115‐120. [Google Scholar]
Riley {published data only}
- Riley K, Kirby R. Outreach clinics. Practice does not make perfect. Health Service Journal 1996;106(5502):29. [PubMed] [Google Scholar]
Seidel {published data only}
- Seidel G, Smith C, Hafner RJ, Holme G. A psychogeriatric community outreach service: description and evaluation. International Journal of Geriatric Psychiatry 1992;7(5):347‐350. [Google Scholar]
Sewell {published data only}
- Sewell J, Oberklaid F, Debelle G, Cullen J, Carmichael A, Finnocchiaro C. Community outreach: an expanding role for hospitals. Aust Paediatr J 1986;22(4):295‐298. [DOI] [PubMed] [Google Scholar]
Shanon {published data only}
- Shanon A, Reisner S, Paswell J, Friedman N, Koren N. The role of a secondary pediatric ambulatory care service within the community. Pediatr Emerg Care 1997;13(1):50‐53. [DOI] [PubMed] [Google Scholar]
Shulman {published data only}
- Shulman LH, Shapira SR, Hirshfield S. Outreach developmental services to children of patients in treatment for substance abuse. Am J Public Health 2000;90(12):1930‐1933. [DOI] [PMC free article] [PubMed] [Google Scholar]
Spencer {published data only}
- Spencer NJ. Consultant paediatric outreach clinics‐‐a practical step in integration. Arch Dis Child 1993;68(4):496‐500. [DOI] [PMC free article] [PubMed] [Google Scholar]
Steele {published data only}
- Steele MM, Wolfe VV. Child psychiatry practice patterns in Ontario. Can J Psychiatry 1999;44(8):788‐792. [DOI] [PubMed] [Google Scholar]
Strathdee1 {published data only}
- Strathdee G. Psychiatrists in primary care: the general practitioner viewpoint. Fam Pract 1988;5(2):111‐115. [DOI] [PubMed] [Google Scholar]
Strathdee2 {published data only}
- Strathdee G, Brown RM, Doig. R.J. Psychiatric clinics in primary care: the effect on general practitioner referral patterns. Soc Psychiatry Psychiatr Epidemiol 1990;25:95‐100. [DOI] [PubMed] [Google Scholar]
Toupin {published data only}
- Toupin A, Denford‐Nelson B. Arthritis Society outreach program: British Columbia and Yukon Division. Canadian Journal of Rehabilitation 1993;6(4):238‐243. [Google Scholar]
Wagstaff {published data only}
- Wagstaff MH, Rigby ML, Redington AN. Increasing workload and changing referral patterns in paediatric cardiology outreach clinics: implications for consultant staffing. Heart 1998;79(3):223‐224. [DOI] [PMC free article] [PubMed] [Google Scholar]
Wakefield {published data only}
- Wakefield DS, Tracy R, Einhellig J. Trends and implications of visiting medical consultant outpatient clinics in rural hospital communities. Hospital & Health Services Administration 1997;42(1):49‐66. [PubMed] [Google Scholar]
Walshe {published data only}
- Walshe K, Shapiro J. Wolverhampton.. In: Roland M, Shapiro J editor(s). Specialist outreach clinics in general practice. Abingdon: Radcliffe Medical Press, 1998:18‐35. [Google Scholar]
Weingarten {published data only}
- Weingarten M, Granek M. Psychiatric liaison with a primary care clinic‐‐14 years' experience. Isr J Psychiatry Relat Sci 1998;35(2):81‐88. [PubMed] [Google Scholar]
Zegleman {published data only}
- Zegleman FE. Psychiatric clinics in different settings ‐ default rates. Health Bull 1988;46:286‐291. [PubMed] [Google Scholar]
References to ongoing studies
Gruen2 {published and unpublished data}
- Specialist outreach to Indigenous communities of Australia's Northern Territory: effects on utilisation, access and health outcomes.. Ongoing study January 2000..
Additional references
AMWAC
- Australian Medical Workforce Advisory Committee (AMWAC). Sustainable specialist services: a compendium of requirements. Report No. 1998.7.. Sydney: AMWAC, 1998. [Google Scholar]
Balestrieri
- Balestrieri M, Williams P, Wilkinson G. Specialist mental health treatment in general practice: a meta‐analysis. Psychol Med 1988;18(3):711‐717. [DOI] [PubMed] [Google Scholar]
Bergmo
- Bergmo TS. An economic analysis of teleradiology versus a visiting radiologist service. Journal of Telemedicine and Telecare 1996;2(3):136‐142. [DOI] [PubMed] [Google Scholar]
Bero 1998
- Bero LA, Grilli R, Grimshaw JM, Harvey E, Oxman AD, Thomson MA. Getting research findings into practice: Closing the gap between research and practice: an overview of systematic reviews of interventions to promote the implementation of research findings. BMJ 1998;317(7156):465‐468. [DOI] [PMC free article] [PubMed] [Google Scholar]
Bero 2000
- Bero LA, Grilli R, Grimshaw JM, Mowatt G, Oxman AD, Zwarenstein M. The Cochrane Effective Practice and Organization of Care Group (EPOC). The Cochrane Library. Oxford: Update Software, 2000. [Google Scholar]
Bower
- Bower P, Sibbald B. On‐site mental health workers in primary care: effects on professional practice. Cochrane Database of Systematic Reviews 2003, Issue 3. [DOI: 10.1002/14651858.CD000532] [DOI] [PubMed] [Google Scholar]
Brown
- Brown LM, Tower JE. Psychiatrists in primary care: would general practitioners welcome them? [see comments.]. Br J Gen Pract 1990;40(338):369‐371. [PMC free article] [PubMed] [Google Scholar]
Creed 1989
- Creed F, Marks B. Liaison psychiatry in general practice: a comparison of the liaison‐attachment scheme and shifted outpatient clinic models. J R Coll Gen Pract 1989;39(329):514‐517. [PMC free article] [PubMed] [Google Scholar]
Denham 1995
- Denham JW. How do we bring an acceptable level of radiotherapy services to a dispersed population?. Australas Radiol 1995;39(2):171‐173. [DOI] [PubMed] [Google Scholar]
Dunbar 1994
- Dunbar JA, Vincent DS, Meikle JN, Dunbar AP, Jones PA. Outreach clinics in general practice. BMJ 1994;308(6945):1714. [DOI] [PMC free article] [PubMed] [Google Scholar]
Gibson 1966
- Gibson R, Forbes JM, Stoddart IW, Cooke JJ, Jenkins W, McKeith SA. Psychiatric care in general practice: an experiment in collaboration. BMJ 1966;1:1287‐1289. [DOI] [PMC free article] [PubMed] [Google Scholar]
Gillam 2001
- Gillam S. Outreach clinics in the new NHS: not yet the end of outpatients. Br J Gen Pract 2001;51(465):261‐262. [PMC free article] [PubMed] [Google Scholar]
Gish
- Gish O, Walker G. Mobile health services. London: Tri Medical Books, 1977. [DOI] [PubMed] [Google Scholar]
Golele 1994
- Golele R. Orthopaedic services in peripheral hospitals. South African Bone and Joint Surgery 1994;4(1):24‐26. [Google Scholar]
Hanlon 1989
- Hanlon CR. Itinerant surgery: outreach or outrage?. Bulletin of the American College of Surgeons 1989;74(10):6‐8. [PubMed] [Google Scholar]
Harris 1994
- Harris A. Specialist outreach clinics. BMJ 1994;308(6936):1053. [DOI] [PMC free article] [PubMed] [Google Scholar]
Horton
- Horton R. Medical journals: evidence of bias against the diseases of poverty. The Lancet 2003;361(9359):712‐713. [DOI] [PubMed] [Google Scholar]
Jurkiewicz 1989
- Jurkiewicz MJ. Itinerant surgery in the guise of outreach surgery. J Med Assoc Ga 1989;78(12):805‐806. [PubMed] [Google Scholar]
Katon W, Von Ko1
- Katon W, Korff M, Lin E, Simon G. Rethinking practitioner roles in chronic illness: the specialist, primary care physician, and the practice nurse. Gen Hosp Psychiatry 2001;23(3):138‐144. [DOI] [PubMed] [Google Scholar]
London 1995
- London D. Reaching out is hard to do. Health Serv J 1995;11 May:15. [Google Scholar]
McCormick 1923
- McCormick B. Hospital policy on outreach surgery. Trustee 1923;42(1):17. [PubMed] [Google Scholar]
Mitchell 1983
- Mitchell AR. Liaison psychiatry in general practice. Br J Hosp Med 1983;30:100‐106. [PubMed] [Google Scholar]
Mitchell 1985
- Mitchell AR. Psychiatrists in primary health care settings. Br J Psychiatry 1985;147:371‐379. [DOI] [PubMed] [Google Scholar]
Mitchell 1990
- Mitchell R. Psychiatric liaison in general practice. Update 1990;34:834‐841. [Google Scholar]
Musk
- Musk AW, Rhys‐Hearn C. Review of specialist medical services to rural and remote Aboriginal communities in Western Australia. Perth: University of Western Australia, 1996. [Google Scholar]
Ollivere 2001
- Ollivere PT. Outreach clinics. Br Dent J 2001;191(11):594. [DOI] [PubMed] [Google Scholar]
Petchey 1998
- Petchey R. Specialist Outreach Clinics in General Practice. BMJ 1998;316(7136):1028. [PubMed] [Google Scholar]
Powell
- Powell J. Systematic review of outreach clinics in primary care in the UK. J Health Serv Res Policy 2002;7(3):177‐183. [DOI] [PubMed] [Google Scholar]
Raasen 1998
- Raasen TJ. Airborne surgical outreach services. East Afr Med J 1998;75(10):561‐562. [PubMed] [Google Scholar]
Ramsay
Rees
- Rees GJ, Deutsch GP, Dunlop PR, Priestman TJ. Clinical oncology services to district general hospitals: report of a working party of the Royal College of Radiologists. Clin Oncol (R Coll Radiol ) 1991;3(1):41‐45. [DOI] [PubMed] [Google Scholar]
Renders
- Renders CM, Valk GD, Griffin S, Wagner EH, Eijk JThM van, Assendelft WJJ. Interventions to improve the management of diabetes in primary care, outpatient and community settings. Cochrane Database of Systematic Reviews 2003, Issue 3. [DOI: 10.1002/14651858.CD001481] [DOI] [PMC free article] [PubMed] [Google Scholar]
Robb 1995
- Robb PJ. Otolaryngology in the GP fundholder practice. J Laryngol Otol 1995;109:95. [PubMed] [Google Scholar]
Robinson 2001
- Robinson PG. Outreach clinics. Br Dent J 2001;191(11):595. [PubMed] [Google Scholar]
Roland 1998
- Roland MO, Shapiro J (eds). Specialist outreach clinics in general practice. Abingdon: Radcliffe Medical Press, 1998. [Google Scholar]
Shah
- Shah A. Cost comparison of psychiatric outpatient clinics based in hospitals and in primary care (general practice health center). Acta Psychiatr Scand 1995;92(1):30‐34. [DOI] [PubMed] [Google Scholar]
Sprod 2001
- Sprod A. Outreach clinics. Br Dent J 2001;191(11):594. [DOI] [PubMed] [Google Scholar]
Stolee
- Stolee P, Clair JK, Kessler L. Geriatric psychiatry consultation in the community. Can J Psychiatry 1994;39(8 Suppl 1):S27‐S33. [DOI] [PubMed] [Google Scholar]
Strathdee 1984
- Strathdee G, Williams P. A survey of psychiatrists in primary care: the silent growth of a new service. J R Coll Gen Pract 1984;34(268):615‐618. [PMC free article] [PubMed] [Google Scholar]
Swash 1993
- Swash M. Should consultants do sessions in GP fundholders' practices? A medical director's view. Br J Hosp Med 1993;50(11):634‐636. [PubMed] [Google Scholar]
Taylor
- Taylor HR. Eye health in Aboriginal and Torres Strait Islander Communities. Canberra: Commonwealth of Australia, 1997. [Google Scholar]
Tod 1993
- Tod ED. Should consultants do sessions in GP fundholders' practices? A GP's view. Br J Hosp Med 1993;50(11):636‐637. [PubMed] [Google Scholar]
Tracy 1996
- Tracy R, Saltzman KL, Wakefield DS. Considerations in establishing visiting consultant clinics in rural hospital communities. Hospital & Health Services Administration 1996;41(2):255‐265. [PubMed] [Google Scholar]
Tyrer 1990
- Tyrer P, Ferguson B, Wadsworth J. Liaison psychiatry in general practice: the comprehensive collaborative model. Acta Psychiatr Scand 1990;81:359‐363. [DOI] [PubMed] [Google Scholar]
White 2001
- White P. Specialist outreach in the NHS. Br J Gen Pract 2001;51(468):581. [PMC free article] [PubMed] [Google Scholar]
Williams 1981
- Williams P, Clare A. Changing patterns of psychiatric care. Br Med J (Clin Res Ed) 1981;282(6261):375‐377. [DOI] [PMC free article] [PubMed] [Google Scholar]


