Abstract
Alzheimer's disease (AD) is a common neurodegenerative brain disorder that causes cellular response alterations, such as impaired cholinergic mechanism, amyloid-beta (Aβ) AD aggregation, neuroinflammation, and several other pathways. AD is still the most prevalent form of dementia and affects many individuals across the globe. The exact cause of the disorder is obscure. There are yet no effective medications for halting, preventing, or curing AD's progress. Plenty of natural products are isolated from several sources and analyzed in preclinical and clinical settings for neuroprotective effects in preventing and treating AD. In addition, natural products and their derivatives have been promising in treating and preventing AD. Natural bioactive compounds play an active modulatory role in the pathological molecular mechanisms of AD development. This review focuses on natural products from plant sources and their derivatives that have demonstrated neuroprotective activities and maybe promising to treat and prevent AD. In addition, this article summarizes the literature pertaining to natural products as agents in the treatment of AD. Rapid metabolism, nonspecific targeting, low solubility, lack of BBB permeability, and limited bioavailability are shortcomings of most bioactive molecules in treating AD. We can use nanotechnology and nanocarriers based on different types of approaches.
1. Introduction
Alzheimer's disease (AD) is a progressive neurological disorder that affects memory and cognitive function as individuals age [1–3]. It accounts for 60-80% of all dementia cases, ranking it the fifth leading cause of mortality [4]. As a neurodegenerative disorder (NDD), AD steadily and permanently diminishes memory, cognition, and ultimately the capability of regular activities, necessitating full-time care. The disease is most prevalent in those over 65 but can also harm younger people. One of the most significant risk factors for AD is age. Research findings showed that the percentage of people with Alzheimer's dementia increases dramatically with age, where 3% of people aged 65-74, 17% of people aged 75-84, and 32% of people aged 85 or older have AD dementia [5, 6]. The pathogenesis of AD is affected by both environmental and genetic factors [7]. In this respect, extracellular depositing in amyloid and intracellular neurofibrillary tangles (NFTs) are two prominent pathologic hallmarks of AD [8]. Accumulation of amyloid causes cognitive decline, leading to clinical dementia [9]. Both amyloid precursor protein (APP) and presenilin mutations end up with different production of amyloid-beta (Aβ) peptides and neuronal death, which is involved in AD development [10, 11]. Surprisingly, new data indicated that neuroinflammation is a critical pathological constituent of Alzheimer's [12]. During AD pathogenesis, the extracellular protein Aβ aggregates, whereas intracellular neurofibrillary tangles (NFTs) reflect hyperphosphorylation of tau proteins in neurons. These neurons generate neuronal cell death and are primarily found in the brain's cerebral cortex and hippocampus [13]. Deposition of aggregated Aβ protein in the synapses of Alzheimer's patients causes inflammation and oxidative stress (OS). In addition, AD consists of excessive glutamatergic neurotransmission and cholinergic neurotransmission depletion [14].
The most frequent and early symptom of AD dementia is the inability to recall memories [15]. Immediate memory impairment frequently occurs, but distant memory is also impacted later, and short-term memory impairment is generally the first clinical sign. On the other hand, memory processing that does not require hippocampal structures is not impaired in AD [16, 17]. In late-stage severe dementia due to AD, people generally lose the ability to move and communicate coherently, leading to substantial memory loss and the loss of their sense of time and place. In this case, patients require additional care. Over the next 50 years, it is anticipated that therapeutic intervention that can delay the onset or progression of AD would dramatically reduce the number of cases [18]. Natural products and their bioactive molecules have been widely shown as successful and promising sources of potential drug leads in the development of medications for the treatment of AD [19, 20]. In this respect, mixtures or extracts of natural products contain natural bioactive molecules that could be a potential therapeutic strategy for treating or preventing AD [21–23]. Furthermore, several extracts and natural sources are broadly used in animal models and clinical trials to cure AD [24, 25]. Based on the previous discussion, and owing to the wide range of preventive and therapeutic options of natural products of plant origin, this review highlights the therapeutic ability of plant-based natural products that may have neuroprotective properties for the control and treatment of AD through various mechanisms.
2. Methodology
We performed a literature search, and recent relevant references have been obtained from different databases such as Scopus, Science Direct, Elsevier, PubMed, and Web of Science. In our search, we used the following terms: medicinal plant, neuroprotection, Alzheimer's disease, antioxidant, and inflammation. Research reports, review articles, and original research articles in English published up to October 2021 were selected and evaluated. In addition, we examined the citations therein and included them when appropriate. According to the recommendations of Page et al. [26, 27], an algorithm was used that inserted all of the steps involved for selecting the relevant information in the study, as indicated in the flow chart in Figure 1. We reviewed 390 references and included 254 of them in the present review.
Figure 1.
Stages involved in selecting published data for inclusion in the present study are depicted in a flow chart; n: number of literature reports.
3. Pathology of AD
Amyloid plaques and NFTs are the pathological hallmarks of AD. In addition, neuropil threads, dystrophic neurites, related astrogliosis, microglial activation, and cerebral amyloid angiopathy are also seen in AD [28]. Neurodegeneration, including synaptic and neuronal loss, leads to macroscopic atrophy due to these pathologic processes. Mixed pathology, which comprises vascular disease and Lewy bodies, is also common neurodegenerative dementia in older people [29]. Indeed, Lewy body pathology frequently coexists with familial AD, the mechanism for which is unknown [30]. Moreover, TDP-43 pathology is becoming more acknowledged as a pathology [31].
Amyloid plaques are extracellular accumulations primarily made up of improperly folded Aβ proteins with 40 or 42 amino acids (Aβ40 and Aβ42), two by-products of APP metabolism. Because of its increased rate of insolubility and fibrillation, Aβ42 is more common in plaques than Aβ40. Amyloid deposition may not necessarily comply with a predictable development sequence, but it generally begins in the isocortex and only affects subcortical regions later. Unlike NFTs, amyloid plaques have a minor impact on the entorhinal cortex and hippocampal formations. On the other hand, hyperphosphorylated tau paired helical filaments (PHFs) make up the majority of NFTs. Tau disease frequently starts in the medial temporal lobes (entorhinal cortex and hippocampus) and then spreads to the associative isocortex; the primary sensory, motor, and visual domains are mostly freed. Because neuronal and synapse loss often coincides with tangle development, pathology of NFT is better connected with clinical characteristics and severity of AD [32]. Aβ pathology, on the other hand, achieves a plateau early in the disease's clinical phase [33].
4. Etiology and Pathophysiology of AD
Although Aloise Alzheimer, a German doctor, initially described AD over a century ago, the fundamental mechanisms responsible for its development are still unknown [34]. Dementia, memory loss, mobility dysfunction, sadness, delusion, spatial awareness impairment, and hallucination are common AD signs, and anomic aphasia, acalculia, and apathy are common symptoms. Furthermore, patients in the terminal stages of the disease cannot speak vocally, have lost their independence, and cannot conduct basic daily tasks [35, 36]. These behavioral abnormalities observed in AD are symptoms of the underlying CNS processes. Despite ongoing research, the etiopathogenesis of this condition has yet to be fully understood. However, some distinct pathways have been found at the cellular and tissue levels. In this context, the buildup of Aβ is a common symptom of AD. Aβ is a short peptide synthesized from naturally existing APP and produces senile plaque APP. Furthermore, Aβ regulates synaptic plasticity, has a role in axonal growth, and modulates axonal growth under physiological settings [36, 37].
Other pathological alterations that disrupt the structure of pyramidal neurons often accompany development. Increased phosphorylation of tau proteins causes these processes, resulting in tau tangles's formation (Figure 2). In addition, tau proteins are involved in the microtubule's stabilization and, therefore, the cytoskeleton structure in physiological settings. In this respect, microtubules provide a proper neuron function and synaptic signaling by acting as an enzyme transporter and cellular protein. Microtubules are delicate structures that rely on interactions between their fundamental ingredients, tubulins, and tau proteins, for stability. Increased tau phosphorylation induces microtubule disassembly and increases the production of tau tangles, which is characteristic of AD. Similarly, the increased concentration of Ca2+ ions caused directly through the aggregation of Aβ inside nerve cells can trigger cyclin-dependent kinase five as one of the agents implicated in this process. Thus, microtubules depolymerize, the cytoskeleton deforms, intracellular transport is disrupted, and the neuron's overall function is reduced. Toxic aggregates represented in tau tangles activate microglia, causing inflammation, neuronal damage, and cell death to combine into toxic aggregates.
Figure 2.
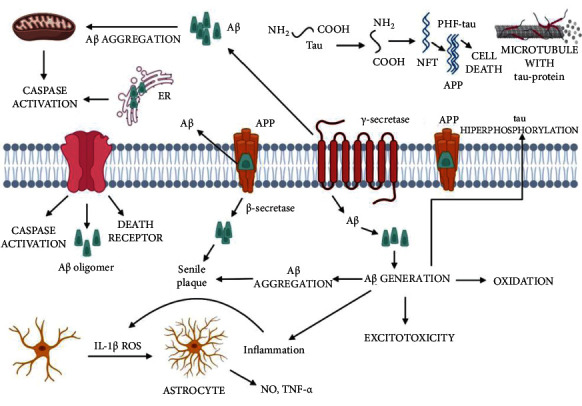
Amyloid cascade in Alzheimer's disease (AD). ER: endoplasmic reticulum; Aβ: amyloid-beta; NFT: neurofibrillary tangle; APP: amyloid precursor protein, ROS: reactive oxygen species; NO: nitric oxide; TNF-α: tumor necrosis factor-alpha [38].
Although several hypotheses have been proposed to explain the pathology of AD, the exact mechanism remains unknown and complex [39]. The following are a few of the hypotheses that have been proposed:
4.1. Cholinergic Hypothesis
Mental state, brain adaptation, sleep-wake cycle regulation, cerebral blood flow control, and neuronal function are affected by cholinergic neurotransmission. According to investigations, the cholinergic system is also essential in cognitive performance. Therefore, impairment could result in memory loss [40, 41]. During cholinergic neurotransmission, acetylcholine (ACh) is released within synapses. Hydrolysis of ACh, which results in signal termination, is carried out by acetylcholinesterase (AChE) and butyrylcholinesterase (BuChE). In this context, certain AD patients showed unaltered or enhanced BuChE activity. AChE is also linked to the development of neurotoxic Aβ fibrils, implying that AChE-induced Aβ aggregation contributes to the advancement of AD, which leads to AChE and BuChE inhibition as a hopeful technique for treating AD [42].
4.2. Hypothesis of Amyloid
Aβ precursor protein (APP) is encoded by a single gene on chromosome 21's 19 exons and considers a type I transmembrane sialoglycoprotein. There are three isoforms of APP: APP751, APP770, and APP695. Neurite outgrowth, cell adhesion, intracellular calcium level stabilization, and synaptic plasticity regulation are APP functions. APP in its soluble form exhibits neurotrophic and neuroprotective effects [43]. There are two types of APP processing pathways: nonamyloidogenic and amyloidogenic. The significant process involves cleavage of Lys16 of the APP by α-secretase enzyme, which results in an Aβ soluble peptide and C-terminal fragment. On the other hand, the nonamyloidogenic peptide p3 is produced by dividing the C-terminal portion by γ-secretase. In contrast, β-secretase cleaves APP to create soluble Aβ peptides and a C-terminal segment. Furthermore, γ-secretase cleaves APP at numerous locations resulting in an Aβ monomer containing amino acids in the 38–43 range. Then, Aβ monomers self-assemble into neurotoxic oligomers; subsequently, the creation of fibrillary aggregates causes neuronal malfunction, which eventually leads to dementia [44]. Aggregated oligomers also cause the production of senile plaques, which are a hallmark of AD. In Alzheimer's patients, levels of the Aβ42 peptide are relatively higher than usual. Furthermore, genetic studies show that APOE, PSEN1, and PSEN2 affect Aβ pathogenesis [45].
4.3. Hypothesis of Tau
Tau protein is a phosphoprotein with 38 phosphorylation sites and six isoforms which vary in length from 352 to 441 amino acids. In this respect, the domains of tau are defined based on their microtubule interactions and their amino acid character. In this case, the amino-terminal fragment does not bind to microtubules but instead projects away from the microtubule surface is termed “projection domain.” Furthermore, the projection domain is separated into amino-terminal and proline-rich regions rich in acidic residues. Likewise, there is division of the tubulin-binding part and the acidic carboxy-terminal portion of the microtubule-binding domain [39]. Phosphorylated tau protein helps stabilize axonal microtubule assembly and participates in intracellular trafficking by interacting with tubulin [46]. Due to aberrant tau phosphorylation, normal tau is transferred to NFTs and paired helical filament tau (PHF-tau). Hyperphosphorylated tau destabilizes microtubules, resulting in nerve cell death. Findings indicated that hyperphosphorylated tau in an AD patient's brain is three to four times higher than in the normal brain [47].
4.4. Neuroinflammation
Elevated levels of microglia and astrocytes cause chronic neuroinflammation by releasing proinflammatory cytokines such as interferon, interleukin-1, and tumor necrosis factor (TNF), which have been found in AD patients and affect the brain. In this regard, reactive oxygen species (ROS) enhance the effect of β-secretase, which cleaves APP to generate an Aβ peptide [46]. Consequently, anti-inflammatory techniques have been employed to produce new compounds that can be used to treat and prevent AD [48].
4.5. Biometal Dyshomeostasis
Metals such as copper, iron, and zinc are essential in biologically essential activities such as protein structure stability, metabolism, catalyst activity, and cellular signal communication [49]. Increased DNA, proteins, and lipids may be produced by free radical generation by the Fenton reaction, primarily powered by redox-active Fe2+ and Cu2+. Therefore, in neurodegenerative disorders such as AD, dysregulation of biometals leads to an increase in oxidative stress, which is why metal chelators could have a role in breaking AD development [50, 51].
4.6. Oxidative Stress (OS)
Consumption of oxygen and cellular signaling produce ROS such as hydroxyl radical, superoxide anion radical, peroxide, and hydrogen peroxide. The intrinsic antioxidant system regulates ROS balance in normal conditions [52]. However, there is a discrepancy between ROS generation and clearance in pathological situations, leading to high ROS levels [53]. Research findings indicated that brain OS may be an early event in AD and might affect disease development [54, 55]. The brain is the most energy-demanding organ, consumes more oxygen than other organs, and conducts mitochondrial respiration, thus increasing the risk of exposure to ROS. On the other hand, protein oxidation and lipid peroxidation play a role in forming and deposition of Aβ in AD [56, 57].
4.7. Insulin-Degrading Enzyme
AD is linked to type 2 diabetes and insulin resistance in the brain. Studies have associated the insulin-degrading enzyme (IDE) to the deposition of Aβ and tau hyperphosphorylation. Insulin and Aβ consider IDE as a competing substrate that plays a role in the pathophysiology of AD. In addition, clearing of Aβ in the brain is linked to IDE. Consequently, IDE activators can be used to treat AD.
4.8. Homocysteine
After demethylation of methionine, homocysteine (HCy) is a nonproteinogenic homolog of cysteine. HCy binds to glutamate NMDA receptors, causing glutamate excitotoxicity, which leads to neurotoxicity and, eventually, neuronal death. Oxidative damage, apoptosis, Aβ aggregation, and tau protein hyperphosphorylation are linked to high levels of HCy [58, 59].
4.9. Phosphodiesterase
Phosphodiesterases (PDEs) are enzymes that help to break down cyclic guanosine monophosphate (cGMP) and cyclic adenosine monophosphate (cAMP). Moreover, they play a role in synaptic plasticity and intracellular signaling cascade control. Changes in PDE4, PDE7, and PDE8 expression, in particular, have been linked to AD [60].
5. Natural Products
Research findings indicated that some dietary components lessen the incidence of AD, prompting scientists to investigate the activity of plant bioactive molecules [61]. Natural bioactive molecules are regarded as “secondary metabolites” of plants. In this respect, numerous chemicals extracted from several plants, including roots, rhizomes, leaves, and seeds, have been shown to inhibit harmful plaque development and increase cholinergic signaling [62, 63] (Figure 3).
Figure 3.
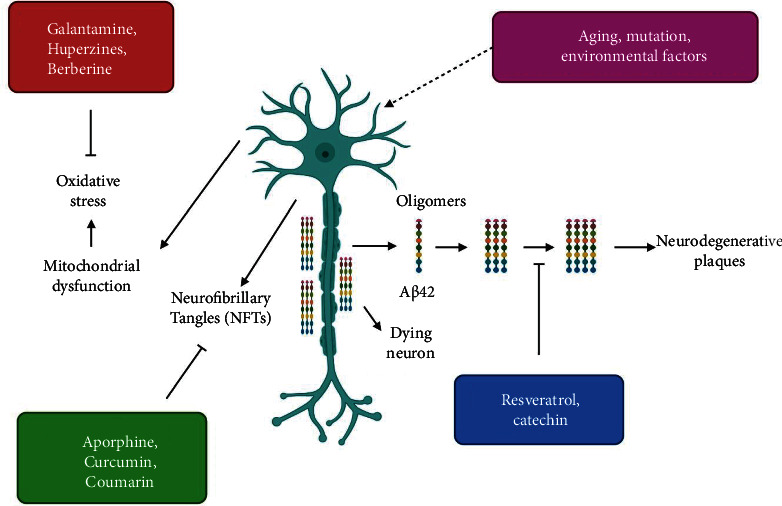
Illustration of the mode of actions by which natural products block Alzheimer's disease (AD).
Antioxidant-rich foods lower oxidative stress in the brain. Accordingly, plant-derived products exhibit a wide range of pharmacological effects and attract the attention of scientists who want to use them to develop molecules to cure various ailments [64, 65]. Findings showed that some natural bioactive compounds are adequate for AD management. Below are details about these compounds (Figure 4).
Figure 4.
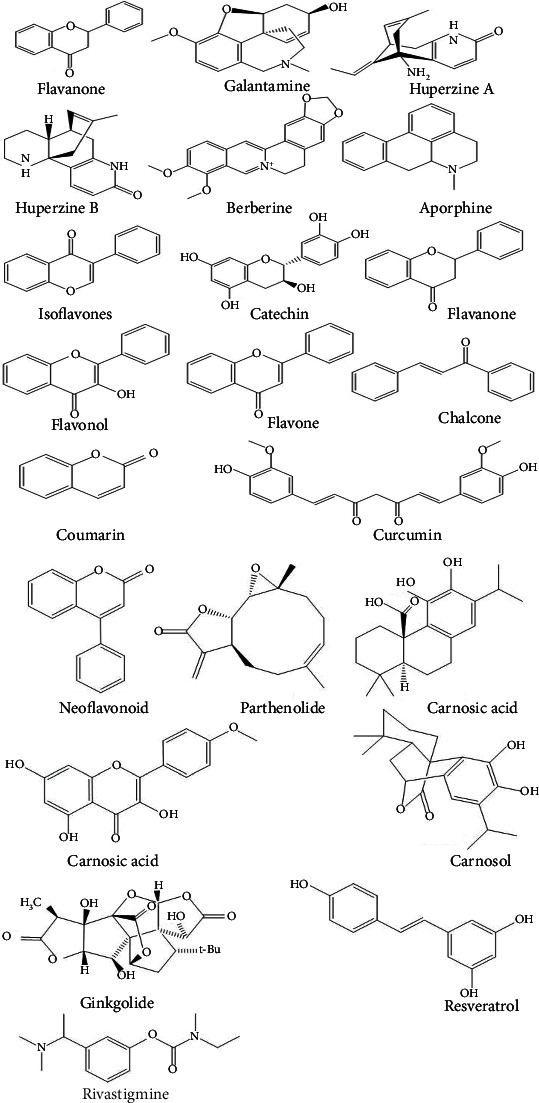
Chemical structures of some chemical compounds that are effective against Alzheimer's disease (AD).
5.1. Alkaloids
Alkaloids, a family of nitrogenous chemical substances, are abundant in confirmed flowering plant families. Many species contain a limited number of alkaloids, but others, such as the Solanaceae, Papaveraceae, Amaryllidaceae, and Ranunculaceae, have many alkaloids [62, 66]. In addition, amphibians include the poison dart frog, rodents like the new world beaver, and fungi like ergot produce alkaloids. Interestingly, rivastigmine and galantamine, two AChEIs approved by the Food and Drug Administration (FDA) in the United States, are alkaloids [64, 66].
5.1.1. Galantamine
Galantamine is an isoquinoline alkaloid found in the flowers and bulbs of Galanthus caucasicus, Galanthus sworonowii, and Leucojum aestivum, all of which are members of the Amaryllidaceae family [67]. Galantamine functions as an allosteric modulator of nicotinic acetylcholine receptors (nAChRs) [68, 69]. Several galantamine derivatives were synthesized by connecting them to memantine with linkers of various lengths and compositions. These compounds were tested as AChE inhibitors and for selectivity toward NMDAR binders and NMDAR subunit 2B (NR2B). Some synthesized compounds showed outstanding inhibitory activity against AChE where IC50 was nanomolar and micromolar affinities for NMDAR. When tested for selectivity toward NMDAR containing the 2B subunit (NR2B), some derivatives exhibited a micromolar affinity for NR2B. Finally, selected compounds were tested using a cell-based assay to measure their neuroprotective activity. In addition, three of the prepared compounds exhibited showed remarkable neuroprotective activities, inhibiting the NMDA-induced neurotoxicity at subnanomolar concentrations, and the IC50 value of one of the prepared compounds was 0.28 nM [70].
In addition, a new dual-site binding hybrid comprising galantamine and indole was synthesized and docked on rhAChE. Results revealed AChE inhibitory activity with IC50 values of 0.011 μM, 0.012 μM, and 0.015 μM for three of the synthesized compounds. The galantamine moiety interacts with the CAS, while the indole portion interacts with the aromatic residues in the peripheral anionic site, resulting in galantamine-indole derivatives that act as dual site binders to the rhAChE enzyme [71].
5.1.2. Huperzines
Huperzines A and B, two lycopodium alkaloids, are isolated from Huperzia serrata (club moss), a Chinese medicinal herb, which can treat illnesses such as swelling, confusion, schizophrenia, fever, and strain. Huperzine A inhibits AChE and BuChE in a specific, effective, and reversible manner, with IC50 values of 0.82 and 74.43 nM, respectively [72, 73]. Similarly, huperzine B is a reversible inhibitor of AChE with an IC50 of 14.3 μM [74]. Consequently, huperzines A and B are frequently employed as natural moieties in developing more powerful AChEIs. In addition, novel AChE inhibitors based on huperzine A's carbobicyclo and tacrine's 4-aminoquinoline substructures with several substituents have been presented as possible AChE inhibitors [75]. Moreover, various heterodimers containing donepezil dimethoxyindanone and huperzine A pyridone connected via a different methylene linker have been considered AChE inhibitors with possible importance in treating AD [76]. Interestingly, novel huperzine A and imine derivatives containing an extra tiny substituted aromatic ring demonstrate efficiency in the nanomolar range, as hAChE inhibitors, where the huperzine derivatives' aromatic rings reveal a π-π stacking with AChE amino acid residues [77].
Furthermore, novel huperzine B compounds were wisely developed. The huperzine B moiety is attached to the terminal aromatic ring by a tether chain, favoring interaction with the peripheral anionic site (PAS) [78]. The hydroxyanthraquinone system of rhein has been connected to a unit of heparin Y using a variety of linkers to create novel multitarget rhein-huprin hybrids. Huprin Y interacts with an active catalytic site (CAS), whereas rhein's aromatic rings form π-π stacking contacts with AChE's PAS, resulting in a dual-site inhibitor [79].
5.1.3. Berberine
Berberine is a benzylisoquinoline alkaloid isolated from rhizomes, stems, roots, and bark of Berberis spp. and Phellodendron amurense. It exhibits potent antioxidant, anti-inflammatory, anticancer, antibacterial, cardioprotective, and neuroprotective activities [80–82]. In addition, berberine inhibits both AChE and BuChE; however, AChE inhibition is more selective. Berberine also inhibits the voltage-dependent potassium current and exhibits an antagonistic effect against the NMDA receptor (especially NR1), resulting in neuroprotection. As a result, berberine improves cognitive impairment in AD via increasing cholinergic stimulation [83, 84]. Furthermore, Torpedo californica acetylcholinesterase (TcAChE) was docked with novel dual-site binding derivatives of triazole and berberine moieties [85]. Berberine-thiophenyl hybrids were synthesized by replacing the oxygen or NH group of the berberine derivatives with a sulfur atom, which caused increasing antioxidant properties. These hybrids also showed antioxidant effects and prevented Aβ aggregation [86].
5.1.4. Aporphine
Aporphine alkaloids are part of the alkaloid isoquinoline class and feature a tetrahydroisoquinoline substructure and are isolated from Menispermum dauricum [87, 88]. Aporphine alkaloids such as oxoisoaporphine and oxoaporphine exert numerous biological activities, including the ability to inhibit telomerase cholinesterase and Aβ aggregation and antioxidant activity [87, 89]. In this regard, synthetic oxoaporphine derivatives are two to three times less potent than their oxoisoaporphine analogs as AChE inhibitors. According to molecular modeling studies, the azabenzanthrone moiety of the oxoisoaporphine alkaloids could attach to Trp279 residue of PAS of AChE via π-π stacking interaction. Using amines or ammonium groups as spacers considerably improved the water solubility and selectivity of the oxoisoaporphine alkaloid toward AChE [87]. An aminoalkyl tether connected a new set of oxoisoaporphine-tacrine hybrids. These novel compounds exhibited antiaggregating activity; they were potent inhibitors of self-induced Aβ aggregation at 10 μM concentrations (35.5–85.8%) [89]. Moreover, dealkylation and ring aromatization processes created eight nuciferine derivatives. AChE inhibitors with IC50 values of 28 and 25 μg/mL were discovered in 1,2-dihydroxyaporphine and dehydronuciferine products [90].
5.2. Flavonoids and Other Polyphenols
Flavonoids are polyphenols found in fruits and vegetables. These are abundant in Polygonaceae, Rutaceae, and Leguminosae plant families [47, 64]. Flavonoids exhibit neuroprotective activities because of their polyphenolic composition; they scavenge free radicals such as superoxide radicals and hydrogen peroxide. The frequency and position of hydroxyl groups in polyphenols influence their capacity to scavenge free radicals. Because of their antioxidant characteristics, a new line of flavonoid derivatives was synthesized [65, 66]. The position of the B ring, the degree of unsaturation, and oxidation of the C ring split flavonoids into various subgroups. Isoflavones are compounds with the B ring connected to position 3 of the C ring, resulting in the 3-phenylchromen-4-one structure. In contrast, in neoflavonoids, the B ring is attached to position 4 of the C ring, resulting in a 4-phenylcoumarine structure. The B ring is connected to position 2 of the C ring in the following subgroups: flavones, flavonols, flavanones, flavan-3-ol or flavanols or catechins, and chalcones; only the structural properties of the C ring differ [91].
Furthermore, flavonoids have become widely employed phytochemicals with different medicinal effects. Due to their core repressive activities against proinflammatory transcription factors, flavonoids play a central role in reducing neuroinflammation in AD [92]. In addition, this category of substances activates transcription factors of antioxidant and anti-inflammatory. Though flavonoids have the potential to be a natural treatment in preclinical AD models, it is, however, worth mentioning that the parent flavonoids' average bioavailability is frequently low. Flavonoids also pass the blood-brain barrier (BBB) because of their intense polarization [12].
5.2.1. Flavones
Flavones are frequently found in numerous medicinal plants, providing several health benefits. Flavones and derivatives inhibit advanced glycation products (AGEs) and exhibit biological activities such as antioxidant, anti-inflammatory, and neuroprotective. These compounds could also be promising agents for treating and preventing AD [92–100].
5.2.2. Isoflavones
Isoflavonoids can be obtained in leguminous plants, such as soybeans, and extracted from microorganisms. During plant-microbe interactions, they function as precursors for the formation of phytoalexin. These compounds exert an inhibitory action on AChE and MAO-B [101].
5.2.3. Flavanones
Another significant subgroup of flavonoids is flavanones such as hesperetin. Flavanones are abundant in citrus fruits like oranges, grapefruit, tangerines, lemons, and limes. In this respect, citrus fruits exhibit free radical scavenging characteristics and act as anti-inflammatory and blood lipid-lowering agents. Thus, the utilization of flavanones in the synthesis of multitarget-directed ligands (MTDL) has increased [97, 98].
5.2.4. Chalcones
Chalcones are another significant subgroup of open chain flavonoids since the fundamental flavonoid skeleton structure lacks ring C. Numerous vegetables such as ladies' fingers and tomatoes contain a certain level of chalcones. Chalcones and their derivatives have attracted researchers' interest as anti-Alzheimer's agents due to their wide range of biological effects [97, 98, 102–105].
5.2.5. Neoflavonoids
Neoflavonoids are natural products that belong to polyphenolic compounds. While flavonoids have the 2-phenylchromen-4-one backbone, neoflavonoids have the 4-phenylchromen backbone with no hydroxyl group substitution at position 2. In this respect, coumarin is a neoflavonoid found in various plants and has a wide range of therapeutic functions. According to molecular modeling studies, it interacts with AChE's peripheral anionic site (PAS) and parts as a potent inhibitor of AChE and inhibits Aβ aggregation [97, 98]. A derivative where the piperazine-based alkyl spacer connected both scaffolds to a new tacrine-coumarin hybrid was prepared. Due to its amide linkage, the product exhibited substantial inhibitory action against EeAChE (0.092 μM) and moderate activity against EqBuChE (0.234 μM), in addition to showing antiaggregation capabilities [106]. The 6 and 7 positions of coumarin are associated with alkyl spacers of various lengths with a terminal diethylamino group in coumarin-based MTDL derivatives, resulting in human AChE inhibition at nanomolar concentrations. In addition, these compounds exhibit remarkable inhibitory activity against Aβ42 self-aggregation (around 60%), thus providing a neuroprotective effect and making them a possible disease modifier [107].
5.3. Curcumin
Curcumin is a natural compound that has been used for centuries to treat a variety of diseases [108]. Curcumin's antioxidant and anti-inflammatory properties made it an effective neuroprotective drug to treat various neurological illnesses. Endoproteolytic breakdown of APP yields the Aβ peptide, which is 40-42 amino acids long. After combining Aβ with curcumin, research findings indicated that rats treated with Aβ show reduced oxidative stress, inflammation, and cognitive deficits [109, 110]. Curcumin's potential to prevent Aβ aggregation and fibril formation has been demonstrated in vivo and in vitro experiments. Along this line, amyloid plaques forming factors are metal chelation, low cholesterol levels, lipid peroxidation, facilitated transcription, and reduced production of β-secretase enzyme; curcumin modulated extracellular amyloid accumulation through these signaling pathways [111, 112]. In addition, curcumin suppresses protein aggregation by affecting the production of heat shock protein (HSP), which is another pathway. In this respect, HSP is molecular chaperones that prevent the formation of protein aggregation. Curcumin increased the formation of HSP in both in vivo and in vitro experimental settings. It also inhibited the formation of harmful amyloid aggregates and proinflammatory cytokines in the brain [111].
On the other hand, the intraneuronal tau protein's accumulations are another vital cause of AD. The β-sheet in tau protein, which drugs can inhibit like curcumin, causes aggregation. Moreover, curcumin's numerous systemic actions have made it a pleiotropic and cost-effective treatment for neuronal dysfunction [3]. Curcumin also suppressed the accumulation of Aβ in PC12 cells and human umbilical vein endothelial cells. In addition, curcumin exhibited antioxidant and anti-inflammatory properties in a Tg2576 mouse model of AD [113]. Similarly, curcumin exerted neuroprotective effects in primary neuronal cell culture against quinolinic acid-induced neurotoxicity. Findings showed that pretreatment with curcumin resulted in a substantial decrease in neuronal nitric oxide synthase [3].
5.4. Terpenes
Terpenoids are a collection of substances chemically known as 2-methyl-1 or 3-butadiene, generated biosynthetically from a mixture of two or more isoprene units [108]. Tanacetum parthenium contains parthenolide, a physiologically active sesquiterpene lactone that increases cognitive performance and lowers TNF-α and IL-6 levels in the cortex and rat hippocampus regions [114]. TLR4/NF-κB-mediated reduction in the ranks of TNF-α, IL-6, and IL-17 in the brain ipsilateral hemispheres was recently shown to control neuroinflammation in intracerebral hemorrhage, which stimulates brain damage in rats [12]. Parthenolide's diverse inhibitory effects in multiple neuropathologies involving inflammation appear to be due to its NF-κB inhibitory action. However, the neuroprotective effects of this sesquiterpene lactone must be validated in AD clinical studies.
Similarly, artemisinin, a sesquiterpene lactone present in the Artemisia annua of the Asteraceae family, was first used to treat multidrug-resistant malaria. This molecule and some of its synthetic counterparts have recently been found to exhibit promising neuroprotective action in AD, owing to their anti-inflammatory properties [115]. Because of their lipophilicity, artemisinin and its synthetic derivatives have been proven to pass the BBB [116]. Carnosic acid and carnosol, both present in Rosmarinus officinalis, are brain-permeable natural diterpenes with considerable neuroprotective activity [117]. No data demonstrate that ginkgolides are beneficial in AD treatment in clinical trials. Clinical trials involving Ginkgo biloba extracts resulted in mixed outcomes. They enhanced cognitive performance, neuropsychiatric symptoms, and functional capacities in patients with mild to moderate AD or vascular dementia, according to results of a randomized controlled trial (RCT) [12]. However, compared to placebo, another trial evaluated the efficacy of long-term use of Ginkgo biloba extract (120 mg) for reducing the incidence of AD in older persons with memory complaints; results showed that the extract does not diminish the progression of AD [118, 119].
5.5. Resveratrol
Resveratrol is an essential nonflavonoid found in red wine, almonds, and grapes [120]. Resveratrol exhibits numerous pharmacological activities, such as anti-inflammatory, antioxidant, anticarcinogenic, and antimutagenic [121]. It additionally demonstrated neuroprotective activity in vitro and in vivo models of AD. Aside from its antioxidant and anti-inflammatory properties, findings suggest that resveratrol enhances nonamyloidogenic APP division and aids in eliminating neurotoxic Aβ peptides, which is crucial in preventing and slowing down AD pathology [122]. Resveratrol also reduces neuronal cell loss through various mechanisms, the most important of which is the activity of NAD+-dependent histone deacetylase enzymes known as sirtuins [123]. Furthermore, resveratrol acts as an antioxidant by inhibiting ROS generation, increasing the amount of GSH and intracellular Ca21 in neurons, and altering the second messengers' calcium-dependent AMP-activated protein kinase (cAMP) and nitric oxide [124]. It also binds to Aβ plaques, which leads to the elimination of the Aβ peptide and inhibits AChE activity in in vitro cells [125].
6. Neuroprotective Mechanisms of Natural Products for AD
6.1. Antioxidative Neuroprotective Activity of Natural Products for AD
Reactive nitrogen species (RNS) and ROS are highly reactive molecules containing radical and nonradical oxidants. Processes such as cell cycle modulation, enzyme, and receptor stimulation, inflammation monitoring, phagocytosis, gene expression, and signal transduction depend on establishing a regulated proportion of these oxidizing agents in the human body [126–129]. In this respect, the nuclear factor E2-related factor 2 (Nrf2) route is one of the most used ways to regulate these reactive species. In response to OS, the transcription factor Nrf2 increases the production of antioxidant genes. The antioxidant response element (ARE) is intensely engaged in lowering OS, inflammation, and an assortment of a few toxic residues [130]. Since elevated metabolic activity and brain-limited cellular regeneration, OS has a more significant impact on the brain than other body regions [131]. Thus, OS is extensively regarded as a critical factor in gradually destroying neuron structure and decreasing neuron activity, one of the leading causes of NDDs, such as AD [127]. Therefore, much work has gone into developing antioxidant treatments as neuroprotective drugs for AD [132]. Published work showed that OS plays a vital role in advancing AD, and antioxidants act against OS damaging effects [133].
Natural and synthetic antioxidants are divided into two categories based on their natural prevalence, with the predominance of antioxidant compounds derived from natural sources. Vitamins A, C, and E, carotenoids, and flavonoids are the most well-known natural antioxidants, and they all contribute to protecting an organism from ROS-induced degradation. Numerous natural antioxidants, such as carotenoids and antioxidant vitamins, are derived from plants and correspond to phenolic and polyphenolic chemical structures, including hydroxyl groups on their aromatic ring(s). These natural compounds demonstrated substantial antioxidant activity as free radical scavengers and hydrogen atom donors [134]. Additionally, these phenolic and polyphenolics exhibit antioxidative functions due to their structures, notably the hydroxyl groups [135]. Flavonoids are the most abundant polyphenolic chemicals, and they possess a broad spectrum of antioxidative properties [136]. Furthermore, dietary polyphenols reduce neuron damage and apoptosis by lowering ROS levels [137]. Because phenolic compounds are naturally occurring substances, they reduce chronic NDDs, particularly AD. Polyphenolic compounds and their derivatives have recently received attention owing to their characteristics as neuroprotective agents for better management of AD [132].
6.2. Antineuroinflammatory and Neuroprotective Activity of Natural Products for AD
Although the specific pathophysiological process of AD has yet to be discovered, several assumptions such as the Aβ, the tau, the cholinergic, and the inflammatory hypotheses exist to interpret this complex disorder [138]. Along this line, the accumulation of Aβ in the brain, a prominent feature in the pathogenesis of AD, has been linked to neuroinflammation [139]. Elevated levels of ROS characterize the neuroinflammatory process in AD, enhance microglial activation, activate nuclear factor kappa B (NF-κB), and increase cytokine production [140]. Additionally, immune cell activation leads to the secretion of IFN-γ, IL-1β, and TNF-α as proinflammatory cytokines [141] that drive neighboring astrocytes to produce Aβ42 oligomers [139]. These proinflammatory cytokines were discovered in high concentrations in AD patients' brains, serum, and cerebrospinal fluid [142]. In this respect, researchers have linked memory impairment in AD to elevated cytokine levels at all stages of the disease [143].
Moreover, it is associated with brain function, particularly in neurological diseases like AD. [144, 145]. Its activation has been associated with Aβ-initiated neurotoxicity [146], and it has been found in the brains of Alzheimer's patients [147]. Natural substances can hinder AD neurodegeneration with fewer adverse effects than synthetic medications. Additionally, natural compounds with anti-inflammatory effects may act as a pharmacological intervention to mitigate AD signs in the early stages [148]. The anti-inflammatory actions of these natural compounds are associated with several targets and several signaling pathways. In addition, natural products can stimulate the effects of antiamyloid as a sound output in AD management due to their ability to reduce neuroinflammation [149]. Consequently, natural substances or combinations of natural products should be evaluated for multitarget anti-inflammatory action as prospective therapeutic alternatives for managing AD [150]. Listed in Table 1 are neuroprotective of some plant-based natural products, extracts, and mixtures.
Table 1.
Neuroprotective status of some plant-based natural products, extracts, and mixtures.
| Plant | Extract | Neuroprotective outcomes | Study model | Reference |
|---|---|---|---|---|
| Panax ginseng | Root extracts | Reduced Aβ formation and aggregation, inhibited AChE, restored synaptophysin and ChAT activity, and decreased Aβ formation and aggregation | In vitro, in vivo | [151–157] |
| Ginkgo biloba | Leaf extract | Scavenged free radicals, averted the mitochondrial malfunction, activated the JNK and ERK pathways, and blocked neuronal death | In vivo | [158–160] |
| Pistacia vera | Kernel | Improved cognitive and motor deficits caused by inhibited cisplatin or vincristine | In vivo | [161] |
| Phyllanthus emblica | Ethanol extract | Improved learning, memory, and antioxidant potential; inhibited AChE activity | In vivo | [162] |
| Hibiscus sabdariffa | Anthocyanin-enriched extracts | Reduced memory impairment by decreasing STZ-induced neuroinflammation and amyloidogenesis | In vitro, In vivo | [163] |
| Spirulina maxima | Ethanol extract | Reduced hippocampus Aβ1–42, APP, and BACE1 expression levels, which reduced AChE activity; lowered hippocampal OS, and elevated BDNF levels | In vivo | [164–166] |
| Ishige foliacea | Phlorotannin-rich fraction | Lowered brain AChE activity, reduced OS, and activated the ERK-BDNF-CREB signaling pathway | In vivo | [167] |
| Juglans regia | Defatted protein | Lowered proinflammatory cytokine expression and AChE levels, extensively restored antioxidant enzyme levels and reduced NF-κB expression | In vivo | [168–172] |
| Almond (Prunus dulcis) | Paste | Reduced AChE activity, lowered cholesterol and triglyceride levels, increased brain tryptophan monoamine levels and serotonergic turnover, and improved learning and memory. | In vivo | [173–176] |
| Hazelnut (Corylus avellana) | Kernel | Improved memory, reduced anxiety, and lowered neuroinflammation and apoptosis | In vivo | [173, 177, 178] |
| Vitis vinifera | Juice, polyphenolic extract | Exhibited antioxidant, antineuroinflammatory, and antiamnesic properties and inhibited Aβ aggregation | In vivo | [179–184] |
| Oryza sativa | Dietary supplement | Reduced hippocampal AChE activity and lipid peroxidation products | In vivo | [185] |
| Zingiber officinale | Root extract | Acted as AChE inhibitor, suppressed lipid peroxidation, caused NMDA receptor overstimulation, and inhibited the generation of free radicals | In vivo | [186, 187] |
| Benincasa hispida | Aqueous extract | Prevented substance P (SP) formation, as were antioxidant scavenging effects. | In vivo | [188] |
| Fuzhisan | Herbal complex | Exhibited antiapoptosis and anti-Aβ buildup activity, increased ACh concentrations, and provided neurotrophic benefits | In vivo | [189–192] |
| Bojungikgi-tang | Herbal formula | Prevented the accumulation and Aβ peptide expression, NeuN, and BDNF in the hippocampus by inhibiting the aggregation of Aβ, enhanced BACE activity in vivo, and increased antioxidant action | In vitro, in vivo | [193] |
| Pistacia integerrima | Gall extracts | Exhibited cholinesterase inhibitory and free radical scavenging activity | In vitro | [194] |
| Phyllanthus acidus | Methanol extract | Increased brain antioxidant enzymes, improved cognitive functioning, and reduced OS | In vitro | [195] |
| Hedera nepalensis | Crude extract | Increased catalase (CAT) and superoxide dismutase (SOD) levels and decreased glutathione (GSH) levels | In vivo | [196] |
| Thalassospira profundimaris | Crude extract | Preserved the synaptic structure and prevented cell cycle-related neuron death | In vitro, in vivo | [197] |
| Eisenia bicycles | Methanol extract | Attenuated OS and reduced neuronal cell death | In vitro | [198] |
| Curcuma longa | Ethanol extract | Reduced CeCl3-induced OS, increased antioxidant enzyme activity, and inhibited AChE activity | In vitro, in vivo | [108, 199–205] |
| Allium sativum | Aged garlic extract | Reduced microglial activation and IL-1 levels and the inflammatory response and reduced psychological stress via modulating stress hormones and the OS response in the brain | In vivo | [206–209] |
| Momordica charanti | Dried and ground fruit | Reduced gliosis, oligomeric Aβ levels, tau hyperphosphorylation, and neuronal death; increased synaptic-related protein and pS9-GSK3b expression levels | In vitro, in vivo | [188, 210] |
| Bacopa monnieri | Extract | Reduced cholinergic degeneration, improved cognition, and suppressed AChE activity | In vivo | [211–216] |
| Viscum album | Extract | Significantly raised serum BDNF levels and reduced AlCl3-induced neurotoxicity | In vitro, in vivo | [217] |
| Pistacia atlantica | Ethyl acetate and aqueous extracts | Inhibitory action of AChE | In vitro | [218] |
| Nardostachys jatamansi | Ethanol extract | Inhibited cell death caused by Aβ | In vitro, in vivo | [219, 220] |
| Phyllanthus amarus, Cynodon dactylon | Methanol extract | Increased superoxide dismutase, catalase, and NADH dehydrogenase levels | In vivo | [221] |
| Salvia miltiorrhiza | Root extract | Inhibited OS and the mitochondria-dependent apoptotic pathway, inhibited production of NO and iNOS expression, induced neuron cell development in rat mesenchymal stem cells, and enhanced the differentiation ability of iPSCs and the survival and neuronal maturation of iPSC-derived neurons transplanted | In vitro, in vivo | [222–225] |
7. Therapeutic Targets for AD
7.1. Amyloid-β (Aβ) and Its Related Enzymes
Amyloid beta-peptide (Aβ) is obtained via the proteolytic processing of APP by β- and γ-secretases [226]. Published research demonstrated that immunization against Aβ has a neuroprotective effect and lessens memory deficits in transgenic mice without lowering the load of Aβ plaques [227, 228]. These findings show that Aβ in plaques may not cause synapse degradation, and that other types of Aβ play a significant role in neurotoxicity in AD brains [229]. Along this line, and based on evidence that Aβ plays a significant role in the etiology of AD, several therapeutic trials using passive and active vaccinations against Aβ were conducted [230]. However, neither technique was effective in clinical studies owing to side effects such as encephalitis and limited treatment efficiency. The action of γ-secretase and β-site APP-cleaving enzyme 1 (BACE1) on APP is required for the accumulation of Aβ peptide, which contributes to the pathophysiology of AD. Before its discovery, the BACE1 activity had been identified in cells and tissues [231].
On the other hand, overexpression of BACE1 enhances the synthesis of Aβ and BACE1-cleaved APP fragments [232]. In this respect, BACE1 splits APP carrying the Swedish familial AD causing mutation around ten to one hundred times more effectively than wild-type APP [231]. Moreover, some BACE1 inhibitors have been tested as antidrugs [233]. Alzheimer's early-onset AD is caused by mutations in presenilin genes [234]. Presenilins are multipass membrane proteins that were later discovered as γ-secretase catalytic components; the membrane implanted aspartyl protease complexes that generate the carboxyl terminus of Aβ from APP [235]. The γ-secretase activity is a therapeutic target for drugs that ease the amyloid plaque, whose accumulation is thought to cause AD.
Meanwhile, the notch signaling is associated with γ-secretase, a primary target for developing an AD therapy. In this regard, compounds that modulate Aβ synthesis by β-secretase without affecting Notch proteolysis and signaling are now in the therapeutic development pipeline at various levels. It is worth mentioning that flurbiprofen, a γ-secretase modulator, was the first drug to go through a clinical study. However, it was unsuccessful because of lack of therapeutic efficacy. However, numerous potent Notch-sparing inhibitors have recently been identified and tested in different clinical trial stages [236]; GSI-953 from Wyeth is an example [237].
7.2. Glycogen Synthase Kinase 3 (GSK3) and Tau Protein
Tau is a mature neuron's major microtubule-associated protein (MAP). It plays a vital role in the pathogenesis of AD and other related disorders, which are known as tauopathies, where tau is deposited in brain regions affected by the disease [238]. In the AD brain, tau is approximately three to fourfold more hyperphosphorylated than the average adult brain tau. Pathologic tau in AD is hyperphosphorylated and abnormally cleaved [239]. Accordingly, inhibition of abnormal hyperphosphorylation of tau offers a promising therapeutic target for AD and related tauopathies [228]. Recent studies have linked early modifications in the structures of soluble tau proteins, particularly their phosphorylation, to neurodegeneration [240, 241]. In this context, GSK3 is a potential kinase that regulates tau binding to microtubules, tau breakdown, and tau aggregation by phosphorylating tau protein [242]. Moreover, findings showed that Aβ enhances GSK3 activation and tau phosphorylation in AD [243]. Some GSK3 inhibitors are currently being studied for their therapeutic potential in AD. Furthermore, research findings revealed that AZD1080, a strong and selective GSK3 inhibitor, can inhibit tau phosphorylation in cells expressing human tau and in intact rat brain [244].
7.3. Acetylcholine-Related Molecules
Cholinergic deficiency owing to basal forebrain atrophy was detected in AD, in addition to the complicated clinical and metabolic abnormalities implicated in the neuronal symptoms of AD, such as the development of NFT and Aβ aggregation [17]. Furthermore, because of the favored loss of neurons expressing nAChRs, there is a significant drop in cerebral nAChR levels in AD [245]. Additionally, the interaction of acetylcholine and nicotinic ligands with nAChRs is essential in mental activities, and nAChR activation by nicotinic ligands can also protect neurons [246]. As a result, new chemicals have been developed to increase acetylcholine levels and directly stimulate nAChRs to reverse mental insufficiencies and shield neurons from Aβ neurotoxicity [247]. Thus, acetylcholinesterase (AChE) inhibitors that limit hydrolysis of acetylcholine and particular agonists for nAChRs have been discovered [248]. Since cholinergic deficiency is a persistent and early result of disease development, AChE has proven to be a critical treatment target for attaining clinical development in AD [249]. In this respect, galantamine, rivastigmine, and donepezil are three of the four medications now available for AD treatment. Galantamine, produced from the bulbs of the common snowdrop and various Amaryllidaceae, has been accepted for the symptomatic treatment of AD-associated senile dementia in several countries [250].
8. Discussion
AD is the most prevalent NDD, and it is one of society's major social and economic challenges. Due to the lack of specific diagnosis and treatment practices, AD is facing barriers in curative strategies. Several bioactive substances and natural extracts are mentioned herein to cure and prevent AD. Until today, most natural chemicals investigated have been sourced mainly from plant sources, with only a few molecules recovered from animals and marine sources. Because AD is a complex condition, these natural substances were linked to various treatment strategies. However, natural substances' neuroprotective effects are dependent on their capacity to penetrate the BBB.
Drug bioavailability and the difficulty of crossing the BBB are critical barriers to developing novel therapeutics. Both observational and experimental research show that bioactive substances enhance cognitive functioning in AD patients. Their modes of action vary, but the critical beneficial effects include the following: reduction of Aβ levels and tau phosphorylation rate, prevention of Aβ and tau aggregation, defense against OS, anti-inflammatory activity, and protection of cellular structures and inhibition of neuronal apoptosis.
Finally, despite recent significant discoveries, the development of potent and selective bioactive plant derivative natural compounds endowed with favorable ADMET (absorption, distribution, metabolism, excretion, and toxicity) properties, particularly higher metabolic stability and lower toxicity, remains a challenging goal for medicinal chemists. Thus, bioactive compounds will likely stay on stage for some time, even if no new natural compound has been advanced to the clinical trial stage in several decades. Compounds that have been shown to have neuroprotective properties in vivo should be investigated further in clinical studies.
9. Future Perspectives
In summary, AD is a terrible neurological condition that has affected humans for decades. Although few medications are accessible today for the treatment of AD, and several plants and their extracts have been widely used in animal studies and AD patients, no drug or plant extract could adequately reverse the symptoms of the disease [251–254]. AD is a multifactorial disease with various causes. Based on AChE inhibition or NMDA receptor antagonism, the present treatments provide some symptomatic relief but do not affect the disease morbidity or mortality. Because of these shortcomings, more serious work is required to comprehend the hallmarks of NDDs, their origins, and claimed therapeutic interventions. It is believed that combining natural product chemistry, medicinal chemistry, pharmacology, biology, and other related fields could be the most promising path to drug discovery and ensuring that natural products and natural-based products are more likely to become therapeutically relevant pharmaceuticals in the treatment and prevention of AD. The implementation of nanotechnology and nanocarrier-based techniques in the delivery of natural products and their separated constituents may aid in the improvement and enhancement of therapeutic responses and efficacy. The use of nanoparticles in the delivery system can improve the bioavailability of natural goods and their constituents.
10. Conclusions
Given the variety and complexity of genetic and epigenetic factors underlying AD, numerous abnormal physiological aspects such as exposure to environmental toxins and risk factors linked to cardiovascular abnormalities decreased quality of life diet, and aging should be considered in managing AD cases. In addition, more pragmatic and extensive quality control regulations protect the safety and potency of these neuroprotective agents. Furthermore, innovative techniques and methods to enhance CNS direct exposure of these neuroprotective agents, including implementation of nanoscience in the transfer of natural products, could occupy an essential role in stopping the progress of dementia. In short, this review has highlighted the ability of nature to prevent and treat AD. In this respect, fruits, spices, nuts, and herbs contain vital bioactive chemicals that can help prevent and treat various ailments, including AD, without serious adverse side effects.
Contributor Information
Talha Bin Emran, Email: talhabmb@bgctub.ac.bd.
Polrat Wilairatana, Email: polrat.wil@mahidol.ac.th.
Mohammad S. Mubarak, Email: mmubarak@ju.edu.jo.
Conflicts of Interest
The authors declare no conflict of interest.
Authors' Contributions
Conceptualization was done by F.I., T.B.E., and P.W.; data curation was contributed by F.I. and J.F.K.; writing—original draft preparation was performed by F.I., J.F.K., and M.H.O.R.; validation was done by F.I., M.S.R., M.H.N, M.R.I., A.A., and T.B.E.; formal analysis was performed by F.I., M.H.O.R., M.M.I., and M.S.R.; investigation was contributed by F.I., M.H.O.R., M.S.R, M.H.N., M.R.I, A.A., and M.S.R.; resources were contributed by T.B.E. and M.S.M.; writing—reviewing and editing was contributed by F.I., T.B.E., P.W., and M.S.M; visualization was contributed by F.I., J.F.K., M.H.O.R., M.S.R, M.R.I, A.A., T.B.E., P.W., and M.S.M.; supervision was done by T.B.E.; project administration was done by T.B.E., P.W., and M.S.M. All authors have read and agreed to the submitted version of the manuscript.
References
- 1.Rai S. N., Singh C., Singh A., Singh M., Singh B. K. Mitochondrial dysfunction: a potential therapeutic target to treat Alzheimer’s disease. Molecular Neurobiology . 2020;57(7):3075–3088. doi: 10.1007/s12035-020-01945-y. [DOI] [PubMed] [Google Scholar]
- 2.Meilandt W. J., Ngu H., Gogineni A., et al. Trem2 deletion reduces late-stage amyloid plaque accumulation, elevates the Aβ42: Aβ40 ratio, and exacerbates axonal dystrophy and dendritic spine loss in the PS2APP Alzheimer's mouse model. Journal of Neuroscience . 2020;40(9):1956–1974. doi: 10.1523/JNEUROSCI.1871-19.2019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Essa M. M., Vijayan R. K., Castellano-Gonzalez G., Memon M. A., Braidy N., Guillemin G. J. Neuroprotective effect of natural products against Alzheimer’s disease. Neurochemical Research . 2012;37(9):1829–1842. doi: 10.1007/s11064-012-0799-9. [DOI] [PubMed] [Google Scholar]
- 4.Grodzicki W., Dziendzikowska K. The role of selected bioactive compounds in the prevention of Alzheimer’s disease. Antioxidants . 2020;9(3):p. 229. doi: 10.3390/antiox9030229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Chen X., Drew J., Berney W., Lei W. Neuroprotective natural products for Alzheimer’s disease. Cells . 2021;10(6, article 1309) doi: 10.3390/cells10061309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Singh A. K., Rai S. N., Maurya A., et al. Therapeutic potential of phytoconstituents in management of Alzheimer’s disease. Evidence-based Complementary and Alternative Medicine . 2021;2021:19. doi: 10.1155/2021/5578574.5578574 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Chauhan V., Chauhan A. Oxidative stress in Alzheimer's disease. Pathophysiology . 2000;13(3):195–208. doi: 10.1016/j.pathophys.2006.05.004. [DOI] [PubMed] [Google Scholar]
- 8.Rai S. N., Tiwari N., Singh P., et al. Therapeutic potential of vital transcription factors in alzheimer’s and parkinson’s disease with particular emphasis on transcription factor eb mediated autophagy. Frontiers in Neuroscience . 2021;15:p. 1703. doi: 10.3389/fnins.2021.777347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Javaid N., Shah M. A., Rasul A., et al. Neuroprotective effects of Ellagic acid in Alzheimer's disease: focus on underlying molecular mechanisms of therapeutic potential. Current Pharmaceutical Design . 2021;27(34):3591–3601. doi: 10.2174/1381612826666201112144006. [DOI] [PubMed] [Google Scholar]
- 10.Brown J. Mutations in amyloid precursor protein gene and disease. The Lancet . 1991;337(8746):923–924. doi: 10.1016/0140-6736(91)90262-N. [DOI] [PubMed] [Google Scholar]
- 11.Srivastava P., Tripathi P. N., Sharma P., et al. Design and development of some phenyl benzoxazole derivatives as a potent acetylcholinesterase inhibitor with antioxidant property to enhance learning and memory. European Journal of Medicinal Chemistry . 2019;163:116–135. doi: 10.1016/j.ejmech.2018.11.049. [DOI] [PubMed] [Google Scholar]
- 12.Olajide O. A., Sarker S. D. Anti-inflammatory studies on Adenanthera pavonina seed extract. Inflammopharmacology . 2004;12(2):196–202. doi: 10.1163/1568560041352310. [DOI] [PubMed] [Google Scholar]
- 13.Butterfield D. A., Reed T., Newman S. F., Sultana R. Roles of amyloid β-peptide-associated oxidative stress and brain protein modifications in the pathogenesis of Alzheimer's disease and mild cognitive impairment. Free Radical Biology and Medicine . 2007;43(5):658–677. doi: 10.1016/j.freeradbiomed.2007.05.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Mariani E., Polidori M., Cherubini A., Mecocci P. Oxidative stress in brain aging, neurodegenerative and vascular diseases: an overview. Journal of Chromatography B . 2005;827(1):65–75. doi: 10.1016/j.jchromb.2005.04.023. [DOI] [PubMed] [Google Scholar]
- 15.Uddin M. S., Mamun A. A., Takeda S., Sarwar M. S., Begum M. M. Analyzing the chance of developing dementia among geriatric people: a cross-sectional pilot study in Bangladesh. Psychogeriatrics . 2019;19(2):87–94. doi: 10.1111/psyg.12368. [DOI] [PubMed] [Google Scholar]
- 16.Markowitsch H. J., Staniloiu A. Memory, autonoetic consciousness, and the self. Consciousness and cognition . 2011;20(1):16–39. doi: 10.1016/j.concog.2010.09.005. [DOI] [PubMed] [Google Scholar]
- 17.Tripathi P. N., Srivastava P., Sharma P., Seth A., Saraf P., Shrivastava S. K. Design, synthesis, and biological evaluation of ferulic acid based 1,3,4-oxadiazole hybrids as multifunctional therapeutics for the treatment of Alzheimer's disease. Bioorganic Chemistry . 2020;95:p. 103506. doi: 10.1016/j.bioorg.2019.103506. [DOI] [PubMed] [Google Scholar]
- 18.Gregory J., Vengalasetti Y. V., Bredesen D. E., Rao R. V. Neuroprotective herbs for the management of Alzheimer’s disease. Biomolecules . 2021;11(4) doi: 10.3390/biom11040543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Akter A., Islam F., Bepary S., et al. CNS depressant activities of Averrhoa carambola leaves extract in thiopental-sodium model of Swiss albino mice: implication for neuro-modulatory properties. Biologia . 2022;77 doi: 10.1007/s11756-022-01057-z. [DOI] [Google Scholar]
- 20.Kennedy D. O., Wightman E. L. Polyphenols and the human brain: plant “secondary metabolite” ecologic roles and endogenous signaling functions drive benefits. Advances in Nutrition . 2014;5(5):515–533. doi: 10.3945/an.114.006320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Lee W.-H., Loo C.-Y., Bebawy M., Luk F., Mason R. S., Rohanizadeh R. Neuron-to-microglia Crosstalk in Psychiatric Disorders. Current Neuropharmacology . 2020;18(2):84–86. doi: 10.2174/1570159X1802200109163818. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Rahman M. M., Islam F., Anwar Parvez M. A., Ashraf G. M., Ullah M. F., Ahmed M. Citrus limonL. (lemon) seed extract shows neuro-modulatory activity in an in vivo thiopental-sodium sleep model by reducing the sleep onset and enhancing the sleep duration. Journal of Integrative Neuroscience . 2022;21(1) doi: 10.31083/j.jin2101042. [DOI] [PubMed] [Google Scholar]
- 23.Rahman M., Rahaman M., Islam M., et al. Role of phenolic compounds in human disease: current knowledge and future prospects. Molecules . 2021;27:p. 233. doi: 10.3390/molecules27051738. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Akram M., Nawaz A. Effects of medicinal plants on Alzheimer's disease and memory deficits. Neural Regeneration Research . 2017;12(4):660–670. doi: 10.4103/1673-5374.205108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Andrade S., Ramalho M. J., Loureiro J. A., Pereira M. D. C. Production and Characterization of Chitooligosaccharides: Evaluation of Acute Toxicity, Healing, and Anti-Inflammatory Actions. International Journal of Molecular Sciences . 2021;22(19) doi: 10.3390/ijms221910631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Page M. J., McKenzie J. E., Bossuyt P. M., et al. Updating guidance for reporting systematic reviews: development of the PRISMA 2020 statement. Journal of Clinical Epidemiology . 2022;134:103–112. doi: 10.1016/j.jclinepi.2022.03.003. [DOI] [PubMed] [Google Scholar]
- 27.Page M. J., McKenzie J. E., Bossuyt P. M., et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. International Journal of Surgery . 2021;88, article 105906 doi: 10.1016/j.ijsu.2021.105906. [DOI] [PubMed] [Google Scholar]
- 28.Serrano-Pozo A., Frosch M. P., Masliah E., Hyman B. T. Neuropathological alterations in Alzheimer disease. Cold Spring Harbor Perspectives in Medicine . 2011;1(1) doi: 10.1101/cshperspect.a006189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Schneider J. A., Arvanitakis Z., Leurgans S. E., Bennett D. A. Three faces of the same gene: FA2H links neurodegeneration with brain iron accumulation, leukodystrophies, and hereditary spastic paraplegias. Annals of Neurology . 2010;68(5):575–577. doi: 10.1002/ana.22211. [DOI] [PubMed] [Google Scholar]
- 30.Revesz T., McLaughlin J., Rossor M., Lantos P. Dementia in Parkinsonism . Springer; 1997. Pathology of Familial Alzheimer’s Disease with Lewy Bodies; pp. 121–135. [DOI] [PubMed] [Google Scholar]
- 31.James B. D., Wilson R. S., Boyle P. A., Trojanowski J. Q., Bennett D. A., Schneider J. A. TDP-43 stage, mixed pathologies, and clinical Alzheimer’s-type dementia. Brain . 2016;139(11):2983–2993. doi: 10.1093/brain/aww224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Fan Z. J., Aditya I. Obesity and Alzheimer’s disease: a bridge between two worlds. The Meducator . 2014;1(25) [Google Scholar]
- 33.Ingelsson M., Fukumoto H., Newell K., et al. Early A accumulation and progressive synaptic loss, gliosis, and tangle formation in AD brain. Neurology . 2004;62(6):925–931. doi: 10.1212/01.wnl.0000115115.98960.37. [DOI] [PubMed] [Google Scholar]
- 34.Hodes R. J., Buckholtz N., Cahan V., Morrison-Bogorad M. Eyes on the prize: Federal Alzheimer’s research effort aims to facilitate interventions. Alzheimer's & Dementia . 2008;4(1S1):S37–S47. doi: 10.1016/j.jalz.2007.11.002. [DOI] [PubMed] [Google Scholar]
- 35.Du X., Wang X., Geng M. Alzheimer’s disease hypothesis and related therapies. Translational Neurodegeneration . 2018;7(1) doi: 10.1186/s40035-018-0107-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Sharma P., Srivastava P., Seth A., Tripathi P. N., Banerjee A. G., Shrivastava S. K. Comprehensive review of mechanisms of pathogenesis involved in Alzheimer's disease and potential therapeutic strategies. Progress in Neurobiology . 2019;174:53–89. doi: 10.1016/j.pneurobio.2018.12.006. [DOI] [PubMed] [Google Scholar]
- 37.Kinney J. W., Bemiller S. M., Murtishaw A. S., Leisgang A. M., Salazar A. M., Lamb B. T. Inflammation as a central mechanism in Alzheimer's disease. Alzheimer's & Dementia: Translational Research & Clinical Interventions . 2018;4:575–590. doi: 10.1016/j.trci.2018.06.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.dos Santos P., Leide C., Ozela P. F., et al. Alzheimer's disease: a review from the pathophysiology to diagnosis, new perspectives for pharmacological treatment. Current medicinal chemistry . 2018;25(26):3141–3159. doi: 10.2174/0929867323666161213101126. [DOI] [PubMed] [Google Scholar]
- 39.Cummings J., Lee G., Ritter A., Zhong K. Alzheimer's disease drug development pipeline: 2018. Alzheimer's & Dementia: Translational Research & Clinical Interventions . 2018;4(1):195–214. doi: 10.1016/j.trci.2018.03.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Birks J. S., Harvey R. J. Donepezil for dementia due to Alzheimer's disease. Cochrane Database of systematic reviews . 2006;25 doi: 10.1002/14651858.cd001190.pub2. [DOI] [PubMed] [Google Scholar]
- 41.Bekdash R. A. The cholinergic system, the adrenergic system and the neuropathology of alzheimer’s disease. International Journal of Molecular Sciences . 2021;22(3) doi: 10.3390/ijms22031273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Jiang Y., Gao H., Turdu G. Azaphilone derivatives with anti-inflammatory activity from the mangrove endophytic fungus Penicillium sclerotiorum ZJHJJ-18. Bioorganic Chemistry . 2022;122:p. 105721. doi: 10.1016/j.bioorg.2022.105721. [DOI] [PubMed] [Google Scholar]
- 43.Villaflores O. B., Chen Y.-J., Chen C.-P., Yeh J.-M., Wu T.-Y. Curcuminoids and resveratrol as anti-Alzheimer agents. Taiwanese Journal of Obstetrics and Gynecology . 2012;51(4):515–525. doi: 10.1016/j.tjog.2012.09.005. [DOI] [PubMed] [Google Scholar]
- 44.Lin A. J., Koike M. A., Green K. N., et al. Spatial frequency domain imaging of intrinsic optical property contrast in a mouse model of Alzheimer’s disease. Annals of biomedical engineering . 2011;39(4):1349–1357. doi: 10.1007/s10439-011-0269-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Tanzi R. E., Bertram L. Twenty Years of the Alzheimer's Disease Amyloid Hypothesis: A Genetic Perspective. Cell . 2005;120(4):545–555. doi: 10.1016/j.cell.2005.02.008. [DOI] [PubMed] [Google Scholar]
- 46.Jakob-Roetne R., Jacobsen H. Alzheimer's disease: from pathology to therapeutic approaches. Angewandte Chemie International Edition . 2009;48(17):3030–3059. doi: 10.1002/anie.200802808. [DOI] [PubMed] [Google Scholar]
- 47.Savelieff M. G., Nam G., Kang J., Lee H. J., Lee M., Lim M. H. Development of multifunctional molecules as potential therapeutic candidates for Alzheimer’s disease, Parkinson’s disease, and amyotrophic lateral sclerosis in the last decade. Chemical Reviews . 2019;119(2):1221–1322. doi: 10.1021/acs.chemrev.8b00138. [DOI] [PubMed] [Google Scholar]
- 48.Qu Z., Mossine V. V., Cui J., Sun G. Y., Gu Z. Protective effects of AGE and its components on neuroinflammation and neurodegeneration. Neuromolecular medicine . 2016;18(3):474–482. doi: 10.1007/s12017-016-8410-1. [DOI] [PubMed] [Google Scholar]
- 49.Min J.-y., Min K.-b. Association between exposure to traffic-related air pollution and pediatric allergic diseases based on modeled air pollution concentrations and traffic measures in Seoul, Korea: a comparative analysis. Environmental Health . 2020;19(1) doi: 10.1186/s12940-020-0563-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Sharma C., Kim S. R. Relationship between Vitamin C Deficiency and Cognitive Impairment in Older Hospitalised Patients: A Cross-Sectional Study. Antioxidants . 2022;11(3) doi: 10.3390/antiox11030463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Faller P., Hureau C., Berthoumieu O. Role of metal ions in the self-assembly of the Alzheimer’s amyloid-β peptide. Inorganic Chemistry . 2013;52(21):12193–12206. doi: 10.1021/ic4003059. [DOI] [PubMed] [Google Scholar]
- 52.Binjhade N., Supare V., Ghaywat S., Trivedi S., Wadher K., Umekar M. Agmatine: a potential Neurotherapeutic Agent. Journal of Drug Delivery and Therapeutics . 2021;11(4):88–92. doi: 10.22270/jddt.v11i4.4855. [DOI] [Google Scholar]
- 53.Lajmanovich R. C., Attademo A. M., Lener G., et al. Glyphosate and glufosinate ammonium, herbicides commonly used on genetically modified crops, and their interaction with microplastics: ecotoxicity in anuran tadpoles. Science of The Total Environment . 2022;804, article 150177 doi: 10.1016/j.scitotenv.2021.150177. [DOI] [PubMed] [Google Scholar]
- 54.Bonda D. J., Lee H.-g., Blair J. A., Zhu X., Perry G., Smith M. A. Role of metal dyshomeostasis in Alzheimer's disease. Metallomics . 2011;3(3):267–270. doi: 10.1039/c0mt00074d. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Greenough M. A., Camakaris J., Bush A. I. Metal dyshomeostasis and oxidative stress in Alzheimer's disease. Neurochemistry International . 2013;62(5):540–555. doi: 10.1016/j.neuint.2012.08.014. [DOI] [PubMed] [Google Scholar]
- 56.Cheignon C. M., Tomas M., Bonnefont-Rousselot D., Faller P., Hureau C., Collin F. Oxidative stress and the amyloid beta peptide in Alzheimer's disease. Redox Biology . 2018;14:450–464. doi: 10.1016/j.redox.2017.10.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Sultana R., Butterfield D. A. Redox proteomics studies of in vivo amyloid beta-peptide animal models of Alzheimer's disease: insight into the role of oxidative stress. Proteomics Clinical Applications . 2008;2(5):685–696. doi: 10.1002/prca.200780024. [DOI] [PubMed] [Google Scholar]
- 58.Zhuo J.-M., Portugal G. S., Kruger W. D., Wang H., Gould T. J., Praticò D. Diet-induced hyperhomocysteinemia increases Amyloid-β formation and deposition in a mouse model of Alzheimers disease. Current Alzheimer Research . 2010;7(2):140–149. doi: 10.2174/156720510790691326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Zhuo J.-M., Wang H., Praticò D. Is hyperhomocysteinemia an Alzheimer's disease (AD) risk factor, an AD marker, or neither? Trends in Pharmacological Sciences . 2011;32(9):562–571. doi: 10.1016/j.tips.2011.05.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Rabal O., Sánchez-Arias J. A., Cuadrado-Tejedor M., et al. Design, synthesis, and biological evaluation of first-in-class dual acting histone deacetylases (HDACs) and phosphodiesterase 5 (PDE5) inhibitors for the treatment of Alzheimer’s disease. Journal of Medicinal Chemistry . 2016;59(19):8967–9004. doi: 10.1021/acs.jmedchem.6b00908. [DOI] [PubMed] [Google Scholar]
- 61.Grant W. B. Using multicountry ecological and observational studies to determine dietary risk factors for Alzheimer's disease. Journal of the American College of Nutrition . 2016;35(5):476–489. doi: 10.1080/07315724.2016.1161566. [DOI] [PubMed] [Google Scholar]
- 62.Ansari N., Khodagholi F. Natural products as promising drug candidates for the treatment of Alzheimer’s disease: molecular mechanism aspect. Current Neuropharmacology . 2013;11(4):414–429. doi: 10.2174/1570159X11311040005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Chitre N. M., Moniri N. H., Murnane K. S. Omega-3 fatty acids as druggable therapeutics for neurodegenerative disorders. CNS & Neurological Disorders-Drug Targets (Formerly Current Drug Targets-CNS & Neurological Disorders) . 2019;18(10):735–749. doi: 10.2174/1871527318666191114093749. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Silva T., Reis J., Teixeira J., Borges F. Alzheimer's disease, enzyme targets and drug discovery struggles: from natural products to drug prototypes. Ageing Research Reviews . 2014;15:116–145. doi: 10.1016/j.arr.2014.03.008. [DOI] [PubMed] [Google Scholar]
- 65.Williams P., Sorribas A., Howes M.-J. R. Employing chemical synthesis to study the structure and function of colibactin, a dark matter metabolite. Natural Product Reports . 2020;37(11):1532–1548. doi: 10.1039/d0np00072h. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Bhattacharjee A., Ramakrishna A., Obulesu M., Bhattacharjee A., Ramakrishna A. Phytomedicine and Alzheimer’s Disease . CRC Press; 2020. Plant Alkaloids and Their Derivatives Relevant to Alzheimer’s Disease; pp. 271–284. [DOI] [Google Scholar]
- 67.Lazarova M. I., Tsekova D. S., Tancheva L. P., et al. New galantamine derivatives with inhibitory effect on acetylcholinesterase activity. Journal of Alzheimer's Disease . 2021;83(3):1211–1220. doi: 10.3233/jad-210577. [DOI] [PubMed] [Google Scholar]
- 68.Heinrich M., Teoh H. L. Best practice in research - Overcoming common challenges in phytopharmacological research. Journal of Ethnopharmacology . 2020;246:p. 112230. doi: 10.1016/j.jep.2019.112230. [DOI] [PubMed] [Google Scholar]
- 69.Choueiry J., Blais C. M., Shah D., et al. CDP-choline and galantamine, a personalized α7 nicotinic acetylcholine receptor targeted treatment for the modulation of speech MMN indexed deviance detection in healthy volunteers: a pilot study. Psychopharmacology . 2020;237(12):3665–3687. doi: 10.1007/s00213-020-05646-1. [DOI] [PubMed] [Google Scholar]
- 70.Simoni E., Daniele S., Bottegoni G., et al. Combining galantamine and memantine in multitargeted, new chemical entities potentially useful in Alzheimer’s disease. Journal of Medicinal Chemistry . 2012;55(22):9708–9721. doi: 10.1021/jm3009458. [DOI] [PubMed] [Google Scholar]
- 71.Atanasova M., Stavrakov G., Philipova I., Zheleva D., Yordanov N., Doytchinova I. Galantamine derivatives with indole moiety: docking, design, synthesis and acetylcholinesterase inhibitory activity. Bioorganic & Medicinal Chemistry . 2015;23(17):5382–5389. doi: 10.1016/j.bmc.2015.07.058. [DOI] [PubMed] [Google Scholar]
- 72.Upadhyay S. D., Ahmad Y., Sharma R. K., Kohli S. Huperzinea: pharmacological and therapeutic potential. Pharmacognosy Communications . 2020;10(4):146–149. doi: 10.5530/pc.2020.4.31. [DOI] [Google Scholar]
- 73.Wu X.-D., Li X.-N., Peng L.-Y., Zhao Q.-S. Huperserratines A and B, two MacrocyclicLycopodiumAlkaloids with an unusual skeleton fromHuperzia serrata. The Journal of Organic Chemistry . 2020;85(10):6803–6807. doi: 10.1021/acs.joc.0c00623. [DOI] [PubMed] [Google Scholar]
- 74.Shi Y.-f., Zhang H.-y., Wang W., et al. Novel 16-substituted bifunctional derivatives of huperzine B: multifunctional cholinesterase inhibitors. Acta Pharmacologica Sinica . 2009;30(8):1195–1203. doi: 10.1038/aps.2009.91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Mak S., Li W., Fu H., et al. Promising tacrine/huperzine A-based dimeric acetylcholinesterase inhibitors for neurodegenerative disorders: From relieving symptoms to modifying diseases through multitarget. Journal of Neurochemistry . 2021;158(6):1381–1393. doi: 10.1111/jnc.15379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Dietrich S., Hüllein J., Hundemer M., et al. Continued response off treatment after BRAF inhibition in refractory hairy cell leukemia. Journal of Clinical Oncology . 2013;31(19):e300–e303. doi: 10.1200/JCO.2012.45.9495. [DOI] [PubMed] [Google Scholar]
- 77.Yan J., Sun L., Wu G., et al. Rational design and synthesis of highly potent anti-acetylcholinesterase activity huperzine A derivatives. Bioorganic & Medicinal Chemistry . 2009;17(19):6937–6941. doi: 10.1016/j.bmc.2009.08.017. [DOI] [PubMed] [Google Scholar]
- 78.Viayna E., Sola I., Bartolini M., et al. Synthesis and multitarget biological profiling of a novel family of rhein derivatives as disease-modifying anti-Alzheimer agents. Journal of Medicinal Chemistry . 2014;57(6):2549–2567. doi: 10.1021/jm401824w. [DOI] [PubMed] [Google Scholar]
- 79.Imanshahidi M., Hosseinzadeh H. Pharmacological and therapeutic effects of Berberis vulgaris and its active constituent, berberine. Phytotherapy Research . 2008;22(8):999–1012. doi: 10.1002/ptr.2399. [DOI] [PubMed] [Google Scholar]
- 80.Jung H. A., Min B.-S., Yokozawa T., Lee J.-H., Kim Y. S., Choi J. S. Anti-Alzheimer and antioxidant activities of Coptidis Rhizoma alkaloids. Biological and Pharmaceutical Bulletin . 2009;32(8):1433–1438. doi: 10.1248/bpb.32.1433. [DOI] [PubMed] [Google Scholar]
- 81.Küpeli E., Koşar M., Yeşilada E., Başer K. H. C. A comparative study on the anti-inflammatory, antinociceptive and antipyretic effects of isoquinoline alkaloids from the roots of Turkish _Berberis_ species. Life Sciences . 2002;72(6):645–657. doi: 10.1016/s0024-3205(02)02200-2. [DOI] [PubMed] [Google Scholar]
- 82.Durairajan S. S. K., Liu L.-F., Lu J.-H., et al. Berberine ameliorates β-amyloid pathology, gliosis, and cognitive impairment in an Alzheimer's disease transgenic mouse model. Neurobiology of Aging . 2012;33(12):2903–2919. doi: 10.1016/j.neurobiolaging.2012.02.016. [DOI] [PubMed] [Google Scholar]
- 83.Kulkarni S., Dhir A. Berberine: a plant alkaloid with therapeutic potential for central nervous system disorders. Phytotherapy Research . 2010;24(3):317–324. doi: 10.1002/ptr.2968. [DOI] [PubMed] [Google Scholar]
- 84.Shi A., Huang L., Lu C., He F., Li X. Synthesis, biological evaluation and molecular modeling of novel triazole containing berberine derivatives as acetylcholinesterase and β-amyloid aggregation inhibitors. Bioorganic & Medicinal Chemistry . 2011;19(7):2298–2305. doi: 10.1016/j.bmc.2011.02.025. [DOI] [PubMed] [Google Scholar]
- 85.Su T., Xie S., Wei H., Yan J., Huang L., Li X. Synthesis and biological evaluation of berberine-thiophenyl hybrids as multi- functional agents: Inhibition of acetylcholinesterase, butyrylcholinesterase, and Aβ aggregation and antioxidant activity. Bioorganic & Medicinal Chemistry . 2013;21(18):5830–5840. doi: 10.1016/j.bmc.2013.07.011. [DOI] [PubMed] [Google Scholar]
- 86.Tang H., Ning F.-X., Wei Y.-B., et al. Derivatives of oxoisoaporphine alkaloids: a novel class of selective acetylcholinesterase inhibitors. Bioorganic & Medicinal Chemistry Letters . 2007;17(13):3765–3768. doi: 10.1016/j.bmcl.2007.04.015. [DOI] [PubMed] [Google Scholar]
- 87.Tang H., Wei Y.-B., Zhang C., et al. Synthesis, biological evaluation and molecular modeling of oxoisoaporphine and oxoaporphine derivatives as new dual inhibitors of acetylcholinesterase/butyrylcholinesterase. European Journal of Medicinal Chemistry . 2009;44(6):2523–2532. doi: 10.1016/j.ejmech.2009.01.021. [DOI] [PubMed] [Google Scholar]
- 88.Himeno E., Ohyagi Y., Ma L., et al. Apomorphine treatment in Alzheimer mice promoting amyloid-β degradation. Annals of Neurology . 2011;69(2):248–256. doi: 10.1002/ana.22319. [DOI] [PubMed] [Google Scholar]
- 89.Tang H., Zhao L.-Z., Zhao H.-T., et al. Hybrids of oxoisoaporphine-tacrine congeners: Novel acetylcholinesterase and acetylcholinesterase-induced β -amyloid aggregation inhibitors. European Journal of Medicinal Chemistry . 2011;46(10):4970–4979. doi: 10.1016/j.ejmech.2011.08.002. [DOI] [PubMed] [Google Scholar]
- 90.Yang Z., Song Z., Xue W., et al. Synthesis and structure–activity relationship of nuciferine derivatives as potential acetylcholinesterase inhibitors. Medicinal Chemistry Research . 2014;23(6):3178–3186. doi: 10.1007/s00044-013-0905-9. [DOI] [Google Scholar]
- 91.Panche A., Diwan A., Chandra S. Flavonoids: an overview. Journal of Nutritional Science . 2016;5:p. e47. doi: 10.1017/jns.2016.41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Rengasamy K. R., Khan H., Gowrishankar S., et al. The role of flavonoids in autoimmune diseases: therapeutic updates. Pharmacology & Therapeutics . 2019;194:107–131. doi: 10.1016/j.pharmthera.2018.09.009. [DOI] [PubMed] [Google Scholar]
- 93.Cruz I., Puthongking P., Cravo S., et al. Xanthone and flavone derivatives as dual agents with acetylcholinesterase inhibition and antioxidant activity as potential anti-alzheimer agents. Journal of Chemistry . 2017;2017:16. doi: 10.1155/2017/8587260.8587260 [DOI] [Google Scholar]
- 94.Li F., Wu J.-J., Wang J., et al. Synthesis and pharmacological evaluation of novel chromone derivatives as balanced multifunctional agents against Alzheimer's disease. Bioorganic & Medicinal Chemistry . 2017;25(14):3815–3826. doi: 10.1016/j.bmc.2017.05.027. [DOI] [PubMed] [Google Scholar]
- 95.Li R.-S., Wang X.-B., Hu X.-J., Kong L.-Y. Design, synthesis and evaluation of flavonoid derivatives as potential multifunctional acetylcholinesterase inhibitors against Alzheimer’s disease. Bioorganic & Medicinal Chemistry Letters . 2013;23:2636–2641. doi: 10.1016/j.bmcl.2013.02.095. [DOI] [PubMed] [Google Scholar]
- 96.Luo W., Su Y.-B., Hong C., et al. Design, synthesis and evaluation of novel 4-dimethylamine flavonoid derivatives as potential multi-functional anti-Alzheimer agents. Bioorganic & Medicinal Chemistry . 2013;21(23):7275–7282. doi: 10.1016/j.bmc.2013.09.061. [DOI] [PubMed] [Google Scholar]
- 97.Shen Y., Zhang J., Sheng R., et al. Synthesis and biological evaluation of novel flavonoid derivatives as dual binding acetylcholinesterase inhibitors. Journal of Enzyme Inhibition and Medicinal Chemistry . 2009;24(2):372–380. doi: 10.1080/14756360802187885. [DOI] [PubMed] [Google Scholar]
- 98.Sheng R., Lin X., Zhang J., et al. Design, synthesis and evaluation of flavonoid derivatives as potent AChE inhibitors. Bioorganic & Medicinal Chemistry . 2009;17(18):6692–6698. doi: 10.1016/j.bmc.2009.07.072. [DOI] [PubMed] [Google Scholar]
- 99.Singh M., Kaur M., Singh N., Silakari O. Corrigendum to Exploration of multi-target potential of chromen-4-one based compounds in Alzheimer's disease: Design, synthesis and biological evaluations. Bioorganic & Medicinal Chemistry . 2018;26(14):4360–4361. doi: 10.1016/j.bmc.2018.06.009. [DOI] [PubMed] [Google Scholar]
- 100.Singh M., Silakari O. Design, synthesis and biological evaluation of novel 2-phenyl-1-benzopyran-4-one derivatives as potential poly-functional anti-Alzheimer's agents. RSC Advances . 2016;6(110):108411–108422. doi: 10.1039/C6RA17678J. [DOI] [Google Scholar]
- 101.Feng B., Li X., Xia J., Wu S. Discovery of novel isoflavone derivatives as AChE/BuChE dual-targeted inhibitors: synthesis, biological evaluation and molecular modelling. Journal of Enzyme Inhibition and Medicinal Chemistry . 2017;32(1):968–977. doi: 10.1080/14756366.2017.1347163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Duan K., Liu H., Fan H., Zhang J., Wang Q. Synthesis and anticholinesterase inhibitory activity of Mannich base derivatives of flavonoids. Journal of Chemical Research . 2014;38(7):443–446. doi: 10.3184/174751914X14031988231263. [DOI] [Google Scholar]
- 103.Fosso M. Y., LeVine H., 3rd, Green K. D., Tsodikov O. V., Garneau-Tsodikova S. Effects of structural modifications on the metal binding, anti-amyloid activity, and cholinesterase inhibitory activity of chalcones. Organic & Biomolecular Chemistry . 2015;13(36):9418–9426. doi: 10.1039/c5ob01478f. [DOI] [PubMed] [Google Scholar]
- 104.Liu H.-r., Liu X.-j., Fan H.-q., Tang J.-j., Gao X.-h., Liu W.-K. Design, synthesis and pharmacological evaluation of chalcone derivatives as acetylcholinesterase inhibitors. Bioorganic & Medicinal Chemistry . 2014;22(21):6124–6133. doi: 10.1016/j.bmc.2014.08.033. [DOI] [PubMed] [Google Scholar]
- 105.Xiao G., Li Y., Qiang X., et al. Design, synthesis and biological evaluation of 4'-aminochalcone-rivastigmine hybrids as multifunctional agents for the treatment of Alzheimer's disease. Bioorganic & Medicinal Chemistry . 2017;25(3):1030–1041. doi: 10.1016/j.bmc.2016.12.013. [DOI] [PubMed] [Google Scholar]
- 106.Xie S.-S., Wang X.-B., Li J.-Y., Yang L., Kong L.-Y. Design, synthesis and evaluation of novel tacrine-coumarin hybrids as multifunctional cholinesterase inhibitors against Alzheimer's disease. European Journal of Medicinal Chemistry . 2013;64:540–553. doi: 10.1016/j.ejmech.2013.03.051. [DOI] [PubMed] [Google Scholar]
- 107.Saeed A., Zaib S., Ashraf S., et al. Synthesis, cholinesterase inhibition and molecular modelling studies of coumarin linked thiourea derivatives. Bioorganic Chemistry . 2015;63:58–63. doi: 10.1016/j.bioorg.2015.09.009. [DOI] [PubMed] [Google Scholar]
- 108.Ganguli M., Chandra V., Kamboh M. I., et al. Apolipoprotein E polymorphism and Alzheimer disease: The Indo-US cross-national dementia study. Archives of Neurology . 2000;57(6):824–830. doi: 10.1001/archneur.57.6.824. [DOI] [PubMed] [Google Scholar]
- 109.Lobine D., Sadeer N., Jugreet S., et al. Potential of medicinal plants as neuroprotective and therapeutic properties against amyloid-β-related toxicity, and glutamate-induced excitotoxicity in human neural cells. Current Neuropharmacology . 2021;19(9):1416–1441. doi: 10.2174/1570159X19666210412095251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Tagde P., Tagde P., Islam F., et al. The multifaceted role of curcumin in advanced nanocurcumin form in the treatment and management of chronic disorders. Molecules . 2021;26(23):p. 7109. doi: 10.3390/molecules26237109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Chen M., Du Z.-Y., Zheng X., Li D.-L., Zhou R.-P., Zhang K. Use of curcumin in diagnosis, prevention, and treatment of Alzheimer's disease. Neural Regeneration Research . 2018;13(4):742–752. doi: 10.4103/1673-5374.230303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Rahman M., Majumder S., Akter F., Islam F., Shahriar M., Alam J. Pre-clinical investigation of analgesic, anti-diarrheal and CNS depressant effect of Pterocarpus indicus in Swiss albino mice. Jordan Journal of Pharmaceutical Sciences . 2021;14(1) [Google Scholar]
- 113.Alattiya R. H., Tarish F. K., Hashim L. L., Hussain S. A. The role of polyphenols in the treatment of Alzheimer's disease: curcumin as a prototype: polyphenols in Alzheimer's disease. Al-Rafidain Journal of Medical Sciences . 2021;1:53–61. doi: 10.54133/ajms.v1i.31. [DOI] [Google Scholar]
- 114.Khare P., Datusalia A. K., Sharma S. S. Parthenolide, an NF-κB inhibitor ameliorates diabetes-induced behavioural deficit, neurotransmitter imbalance and neuroinflammation in type 2 diabetes rat model. Neuromolecular Medicine . 2017;19(1):101–112. doi: 10.1007/s12017-016-8434-6. [DOI] [PubMed] [Google Scholar]
- 115.Zhu C., Xiong Z., Chen X., et al. Artemisinin attenuates lipopolysaccharide-stimulated proinflammatory responses by inhibiting NF-κB pathway in microglia cells. PLoS One . 2012;7(4) doi: 10.1371/journal.pone.0035125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Navaratnam V., Mansor S. M., Sit N.-W., Grace J., Li Q., Olliaro P. Pharmacokinetics of artemisinin-type compounds. Clinical Pharmacokinetics . 2000;39(4):255–270. doi: 10.2165/00003088-200039040-00002. [DOI] [PubMed] [Google Scholar]
- 117.de Oliveira M. R. The dietary components carnosic acid and carnosol as neuroprotective agents: a mechanistic view. Molecular Neurobiology . 2016;53(9):6155–6168. doi: 10.1007/s12035-015-9519-1. [DOI] [PubMed] [Google Scholar]
- 118.Vellas B., Coley N., Ousset P.-J., et al. Long-term use of standardised Ginkgo biloba extract for the prevention of Alzheimer's disease (GuidAge): a randomised placebo-controlled trial. The Lancet Neurology . 2012;11(10):851–859. doi: 10.1016/S1474-4422(12)70206-5. [DOI] [PubMed] [Google Scholar]
- 119.Zhang Y., Huang Q., Wang S., et al. The Food Additive β-Caryophyllene Exerts Its Neuroprotective Effects Through the JAK2-STAT3-BACE1 Pathway. Frontiers in Aging Neuroscience . 2022;14 doi: 10.3389/fnagi.2022.814432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Liang Z., Owens C. L., Zhong G.-Y., Cheng L. Polyphenolic profiles detected in the ripe berries of Vitis vinifera germplasm. Food Chemistry . 2011;129(3):940–950. doi: 10.1016/j.foodchem.2011.05.050. [DOI] [PubMed] [Google Scholar]
- 121.Alarcon De La Lastra C., Villegas I. Resveratrol as an anti-inflammatory and anti-aging agent: mechanisms and clinical implications. Molecular Nutrition & Food Research . 2005;49(5):405–430. doi: 10.1002/mnfr.200500022. [DOI] [PubMed] [Google Scholar]
- 122.Braidy N., Jugder B.-E., Poljak A., et al. Resveratrol as a potential therapeutic candidate for the treatment and management of Alzheimer’s disease. Current Topics in Medicinal Chemistry . 2016;16(17):1951–1960. doi: 10.2174/1568026616666160204121431. [DOI] [PubMed] [Google Scholar]
- 123.Schemies J., Uciechowska U., Sippl W., Jung M. NAD+dependent histone deacetylases (sirtuins) as novel therapeutic targets. Medicinal Research Reviews . 2010;30(6):861–889. doi: 10.1002/med.20178. [DOI] [PubMed] [Google Scholar]
- 124.Li F., Gong Q., Dong H., Shi J. Resveratrol, a neuroprotective supplement for Alzheimer's disease. Current Pharmaceutical Design . 2012;18(1):27–33. doi: 10.2174/138161212798919075. [DOI] [PubMed] [Google Scholar]
- 125.Lee E. O., Park H. J., Kang J. L., Kim H. S., Chong Y. H. Resveratrol reduces glutamate-mediated monocyte chemotactic protein-1 expression via inhibition of extracellular signal-regulated kinase 1/2 pathway in rat hippocampal slice cultures. Journal of Neurochemistry . 2010;112(6):1477–1487. doi: 10.1111/j.1471-4159.2009.06564.x. [DOI] [PubMed] [Google Scholar]
- 126.Hussain T., Tan B., Yin Y., Blachier F., Tossou M. C., Rahu N. Oxidative stress and inflammation: what polyphenols can do for us? Oxidative Medicine and Cellular Longevity . 2016;2016:9. doi: 10.1155/2016/7432797.7432797 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Islam M. T. Oxidative stress and mitochondrial dysfunction-linked neurodegenerative disorders. Neurological Research . 2017;39(1):73–82. doi: 10.1080/01616412.2016.1251711. [DOI] [PubMed] [Google Scholar]
- 128.Manoharan S., Guillemin G. J., Abiramasundari R. S., Essa M. M., Akbar M., Akbar M. D. The role of reactive oxygen species in the pathogenesis of Alzheimer’s disease, Parkinson’s disease, and Huntington’s disease: a mini review. Oxidative Medicine and Cellular Longevity . 2016;2016:15. doi: 10.1155/2016/8590578.8590578 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Liu Z., Zhou T., Ziegler A. C., Dimitrion P., Zuo L. Oxidative stress in neurodegenerative diseases: from molecular mechanisms to clinical applications. Oxidative Medicine and Cellular Longevity . 2017;2017:11. doi: 10.1155/2017/2525967.428010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Otsuki A., Yamamoto M. Cis-element architecture of Nrf2–sMaf heterodimer binding sites and its relation to diseases. Archives of Pharmacal Research . 2020;43(3):275–285. doi: 10.1007/s12272-019-01193-2. [DOI] [PubMed] [Google Scholar]
- 131.Liguori I., Russo G., Curcio F., et al. Oxidative stress, aging, and diseases. Clinical Interventions in Aging . 2018;13:p. 757. doi: 10.2147/CIA.S158513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Teleanu R. I., Chircov C., Grumezescu A. M., Volceanov A., Teleanu D. M. Antioxidant therapies for neuroprotection a review. Journal of Clinical Medicine . 2019;8(10):p. 1659. doi: 10.3390/jcm8101659. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Juszczyk G., Mikulska J., Kasperek K., Pietrzak D., Mrozek W., Herbet M. Chronic stress and oxidative stress as common factors of the pathogenesis of depression and Alzheimer’s disease: the role of antioxidants in prevention and treatment. Antioxidants . 2021;10(9):p. 1439. doi: 10.3390/antiox10091439. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Chandran R., Sajeesh T., Parimelazhagan T. Total phenolic content, anti-radical property and HPLC profiles ofCaralluma diffusa(Wight) N.E. Br. Journal of Biologically Active Products from Nature . 2014;4(3):188–195. doi: 10.1080/22311866.2014.933082. [DOI] [Google Scholar]
- 135.Cosme P., Rodríguez A. B., Espino J., Garrido M. Plant phenolics: Bioavailability as a key determinant of their potential health-promoting applications. Antioxidants . 2020;9(12) doi: 10.3390/antiox9121263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Kumar S., Pandey A. K. Chemistry and biological activities of flavonoids: an overview. The Scientific World Journal . 2013;2013:16. doi: 10.1155/2013/162750.162750 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Calabrese V., Butterfield D. A., Stella A. Nutritional antioxidants and the heme oxygenase pathway of stress tolerance: novel targets for neuroprotection in Alzheimer's disease. The Italian journal of biochemistry . 2003;52(4):177–181. [PubMed] [Google Scholar]
- 138.Yiannopoulou K. G., Papageorgiou S. G. Current and future treatments for Alzheimer’s disease. Therapeutic Advances in Neurological Disorders . 2013;6(1):19–33. doi: 10.1177/1756285612461679. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Prà I. D., Chiarini A., Gui L., et al. Do astrocytes collaborate with neurons in spreading the “infectious” Aβ and Tau drivers of Alzheimer’s disease? The Neuroscientist . 2015;21(1):9–29. doi: 10.1177/1073858414529828. [DOI] [PubMed] [Google Scholar]
- 140.Von Bernhardi R., Cornejo F., Parada G., Eugenín J. Role of TGFβ signaling in the pathogenesis of Alzheimer’s disease. Frontiers in Cellular Neuroscience . 2015;9:p. 426. doi: 10.3389/fncel.2015.00426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Tan M.-S., Yu J.-T., Jiang T., Zhu X.-C., Tan L. The NLRP3 inflammasome in Alzheimer’s disease. Molecular Neurobiology . 2013;48(3):875–882. doi: 10.1007/s12035-013-8475-x. [DOI] [PubMed] [Google Scholar]
- 142.Simpson D. S., Oliver P. L. ROS generation in microglia: understanding oxidative stress and inflammation in neurodegenerative disease. Antioxidants . 2020;9(8):p. 743. doi: 10.3390/antiox9080743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Garcez M. L., Mina F., Bellettini-Santos T., et al. Minocycline reduces inflammatory parameters in the brain structures and serum and reverses memory impairment caused by the administration of amyloid β (1-42) in mice. Progress in Neuro-Psychopharmacology and Biological Psychiatry . 2017;77:23–31. doi: 10.1016/j.pnpbp.2017.03.010. [DOI] [PubMed] [Google Scholar]
- 144.Sandberg M., Patil J., D'Angelo B., Weber S. G., Mallard C. NRF2-regulation in brain health and disease: implication of cerebral inflammation. Neuropharmacology . 2014;79:298–306. doi: 10.1016/j.neuropharm.2013.11.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Yu M., Li H., Liu Q., et al. Nuclear factor p65 interacts with Keap1 to repress the Nrf2-ARE pathway. Cellular Signalling . 2011;23(5):883–892. doi: 10.1016/j.cellsig.2011.01.014. [DOI] [PubMed] [Google Scholar]
- 146.Longpré F., Garneau P., Ramassamy C. Protection by EGb 761 against β-amyloid-induced neurotoxicity: involvement of NF-κB, SIRT1, and MAPKs pathways and inhibition of amyloid fibril formation. Free Radical Biology and Medicine . 2006;41(12):1781–1794. doi: 10.1016/j.freeradbiomed.2006.08.015. [DOI] [PubMed] [Google Scholar]
- 147.Uddin M. S., Hasana S., Ahmad J., et al. Anti-neuroinflammatory potential of polyphenols by inhibiting NF-κB to halt Alzheimer's disease. Current Pharmaceutical Design . 2021;27(3):402–414. doi: 10.2174/1381612826666201118092422. [DOI] [PubMed] [Google Scholar]
- 148.Cooper E. L., Ma M. J. Alzheimer disease: clues from traditional and complementary medicine. Journal of Traditional and Complementary Medicine . 2017;7(4):380–385. doi: 10.1016/j.jtcme.2016.12.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Zhao L., Wang J.-L., Liu R., Li X.-X., Li J.-F., Zhang L. Neuroprotective, anti-amyloidogenic and neurotrophic effects of apigenin in an Alzheimer’s disease mouse model. Molecules . 2013;18(8):9949–9965. doi: 10.3390/molecules18089949. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Wollen K. A. Alzheimer’s disease: the pros and cons of pharmaceutical, nutritional, botanical, and stimulatory therapies, with a discussion of treatment strategies from the perspective of patients and practitioners. Altern Med Rev . 2010;15(3):223–244. [PubMed] [Google Scholar]
- 151.Seo J.-S., Yun J.-H., Baek I.-S., et al. Oriental medicine Jangwonhwan reduces Aβ (1–42) level and β-amyloid deposition in the brain of Tg-APPswe/PS1dE9 mouse model of Alzheimer disease. Journal of Ethnopharmacology . 2010;128(1):206–212. doi: 10.1016/j.jep.2016.07.022. [DOI] [PubMed] [Google Scholar]
- 152.Kim J., Kim S. H., Lee D.-S., Kim S. H., Chung S., Yang H. O. Effects of fermented ginseng on memory impairment and β-amyloid reduction in Alzheimer’s disease experimental models. Journal of Ginseng Research . 2013;37(1):p. 100. doi: 10.1016/j.jgr.2021.04.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Choi J. G., Kim N., Huh E., et al. White ginseng protects mouse hippocampal cells against amyloid-beta oligomer toxicity. Phytotherapy Research . 2017;31(3):497–506. doi: 10.1002/ptr.7336. [DOI] [PubMed] [Google Scholar]
- 154.Heo J.-H., Lee S.-T., Oh M. J., et al. Improvement of cognitive deficit in Alzheimer’s disease patients by long term treatment with Korean red ginseng. Journal of Ginseng Research . 2011;35(4):p. 457. doi: 10.5142/jgr.2011.35.4.457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Heo J.-H., Park M.-H., Lee J.-H. Effect of Korean red ginseng on cognitive function and quantitative EEG in patients with Alzheimer's disease: a preliminary study. The Journal of Alternative and Complementary Medicine . 2016;22(4):280–285. doi: 10.1089/acm.2015.0265. [DOI] [PubMed] [Google Scholar]
- 156.Kim H.-J., Jung S.-W., Kim S.-Y., et al. Panax ginseng as an adjuvant treatment for Alzheimer's disease. Journal of Ginseng Research . 2018;42(4):401–411. doi: 10.1016/j.jgr.2021.04.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Lee M. R., Yun B. S., In O. H., Sung C. K. Comparative study of Korean white, red, and black ginseng extract on cholinesterase inhibitory activity and cholinergic function. Journal of Ginseng Research . 2011;35(4):p. 421. doi: 10.1016/j.jgr.2022.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Singh S. K., Srivastav S., Castellani R. J., Plascencia-Villa G., Perry G. Neuroprotective and antioxidant effect of Ginkgo biloba extract against AD and other neurological disorders. Neurotherapeutics . 2019;16(3):666–674. doi: 10.1007/s13311-019-00767-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Zhao J., Li K., Wang Y., et al. Enhanced anti-amnestic effect of donepezil by Ginkgo biloba extract (EGb 761) via further improvement in pro-cholinergic and antioxidative activities. Journal of Ethnopharmacology . 2021;269:p. 113711. doi: 10.1016/j.jep.2020.113711. [DOI] [PubMed] [Google Scholar]
- 160.Liu H., Ye M., Guo H. An updated review of randomized clinical trials testing the improvement of cognitive function of Ginkgo biloba extract in healthy people and Alzheimer’s patients. Frontiers in Pharmacology . 2020;10:p. 1688. doi: 10.3389/fphar.2019.01688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Golchin L., Shabani M., Harandi S., Razavinasab M. Pistachio supplementation attenuates motor and cognition impairments induced by cisplatin or vincristine in rats. Advanced Biomedical Research . 2015;4 doi: 10.4103/2277-9175.156656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Uddin M. S., Al Mamun A., Hossain M. S., Akter F., Iqbal M. A., Asaduzzaman M. Exploring the effect of Phyllanthus emblica L. on cognitive performance, brain antioxidant markers and acetylcholinesterase activity in rats: promising natural gift for the mitigation of Alzheimer's disease. Annals of Neurosciences . 2016;23(4):218–229. doi: 10.1159/000481812. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.El-Shiekh R. A., Ashour R. M., Abd El-Haleim E. A., Ahmed K. A., Abdel-Sattar E. Hibiscus sabdariffa L.: a potent natural neuroprotective agent for the prevention of streptozotocin-induced Alzheimer’s disease in mice. Biomedicine & Pharmacotherapy . 2020;128:p. 110303. doi: 10.1016/j.biopha.2020.110303. [DOI] [PubMed] [Google Scholar]
- 164.Lee J.-C., Hou M.-F., Huang H.-W., et al. Marine algal natural products with anti-oxidative, anti-inflammatory, and anti-cancer properties. Cancer Cell International . 2013;13(1):1–7. doi: 10.1186/1475-2867-13-55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Koh E.-J., Kim K.-J., Song J.-H., et al. Spirulina maxima extract ameliorates learning and memory impairments via inhibiting GSK-3β phosphorylation induced by intracerebroventricular injection of amyloid-β 1–42 in mice. International Journal of Molecular Sciences . 2017;18(11):p. 2401. doi: 10.3390/ijms23020596. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Koh E.-J., Kim K.-J., Choi J., Kang D.-H., Lee B.-Y. Spirulina maxima extract prevents cell death through BDNF activation against amyloid beta 1-42 (Aβ1-42) induced neurotoxicity in PC12 cells. Neuroscience Letters . 2018;673:33–38. doi: 10.1097/APO.0000000000000503. [DOI] [PubMed] [Google Scholar]
- 167.Um M. Y., Lim D. W., Son H. J., Cho S., Lee C. Phlorotannin-rich fraction from Ishige foliacea brown seaweed prevents the scopolamine-induced memory impairment via regulation of ERK-CREB-BDNF pathway. Journal of Functional Foods . 2018;40:110–116. doi: 10.1016/j.jff.2017.10.014. [DOI] [Google Scholar]
- 168.Pribis P., Bailey R. N., Russell A. A., et al. Effects of walnut consumption on cognitive performance in young adults. British Journal of Nutrition . 2012;107(9):1393–1401. doi: 10.1017/S0007114511004302. [DOI] [PubMed] [Google Scholar]
- 169.Zou J., Cai P.-s., Xiong C.-m., Ruan J.-l. Neuroprotective effect of peptides extracted from walnut (Juglans Sigilata Dode) proteins on Aβ25-35-induced memory impairment in mice. Journal of Huazhong University of Science and Technology [Medical Sciences] . 2016;36(1):21–30. doi: 10.1007/s11596-016-1536-4. [DOI] [PubMed] [Google Scholar]
- 170.Li W., Zhao T., Zhang J., et al. Effect of walnut protein hydrolysate on scopolamine-induced learning and memory deficits in mice. Journal of Food Science and Technology . 2017;54(10):3102–3110. doi: 10.1007/s13197-021-05080-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Chauhan A., Chauhan V. Beneficial effects of walnuts on cognition and brain health. Nutrients . 2020;12(2):p. 550. doi: 10.3390/nu13051635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Muthaiyah B., Essa M. M., Chauhan V., Chauhan A. Protective effects of walnut extract against amyloid beta peptide-induced cell death and oxidative stress in PC12 cells. Neurochemical Research . 2011;36(11):2096–2103. doi: 10.1007/s11064-011-0533-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.Kulkarni K. S., Kasture S., Mengi S. Efficacy study of Prunus amygdalus (almond) nuts in scopolamine-induced amnesia in rats. Indian Journal of Pharmacology . 2010;42(3):p. 168. doi: 10.4103/ijp.ijp_508_21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Haider S., Batool Z., Haleem D. Nootropic and hypophagic effects following long term intake of almonds (Prunus amygdalus) in rats. Nutricion Hospitalaria . 2012;27(6):2109–2115. doi: 10.3305/nh.2012.27.6.6140. [DOI] [PubMed] [Google Scholar]
- 175.Batool Z., Sadir S., Liaquat L., et al. Repeated administration of almonds increases brain acetylcholine levels and enhances memory function in healthy rats while attenuates memory deficits in animal model of amnesia. Brain Research Bulletin . 2016;120:63–74. doi: 10.1016/j.brainresbull.2015.11.001. [DOI] [PubMed] [Google Scholar]
- 176.Batool Z., Agha F., Ahmad S., et al. Attenuation of cadmium-induced decline in spatial, habituation and recognition memory by long-term administration of almond and walnut supplementation: role of cholinergic function. Pak J Pharm Sci . 2017;30(1):273–279. [PubMed] [Google Scholar]
- 177.Gorji N., Moeini R., Memariani Z. Almond, hazelnut and walnut, three nuts for neuroprotection in Alzheimer’s disease: a neuropharmacological review of their bioactive constituents. Pharmacological Research . 2018;129:115–127. doi: 10.1016/j.phrs.2017.12.003. [DOI] [PubMed] [Google Scholar]
- 178.Bahaeddin Z., Yans A., Khodagholi F., Hajimehdipoor H., Sahranavard S. Hazelnut and neuroprotection: improved memory and hindered anxiety in response to intra-hippocampal Aβ injection. Nutritional Neuroscience . 2017;20(6):317–326. doi: 10.1080/1028415X.2015.1126954. [DOI] [PubMed] [Google Scholar]
- 179.Peters R., Peters J., Warner J., Beckett N., Bulpitt C. Alcohol, dementia and cognitive decline in the elderly: a systematic review. Age and Ageing . 2008;37(5):505–512. doi: 10.1093/ageing/afy193. [DOI] [PubMed] [Google Scholar]
- 180.Loureiro J. A., Andrade S., Duarte A., et al. Resveratrol and grape extract-loaded solid lipid nanoparticles for the treatment of Alzheimer’s disease. Molecules . 2017;22(2):p. 277. doi: 10.3390/molecules24020243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181.Siahmard Z., Alaei H., Reisi P., Pilehvarian A. A. The effect of red grape juice on Alzheimer's disease in rats. Advanced Biomedical Research . 2012;1:p. 63. doi: 10.4103/2277-9175.100188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182.Sun Q., Jia N., Li X., Yang J., Chen G. Grape seed proanthocyanidins ameliorate neuronal oxidative damage by inhibiting GSK-3β-dependent mitochondrial permeability transition pore opening in an experimental model of sporadic Alzheimer’s disease. Aging . 2019;11(12):p. 4107. doi: 10.18632/aging.203839. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Lian Q., Nie Y., Zhang X., et al. Effects of grape seed proanthocyanidin on Alzheimer's disease in vitro and in vivo. Experimental and Therapeutic Medicine . 2016;12(3):1681–1692. doi: 10.3892/etm.2016.3530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.Borai I. H., Ezz M. K., Rizk M. Z., et al. Therapeutic impact of grape leaves polyphenols on certain biochemical and neurological markers in AlCl3-induced Alzheimer’s disease. Biomedicine & Pharmacotherapy . 2017;93:837–851. doi: 10.1016/j.biopha.2017.07.038. [DOI] [PubMed] [Google Scholar]
- 185.Pannangrong W., Wattanathorn J., Muchimapura S., Tiamkao S., Tong-Un T. Purple rice berry is neuroprotective and enhances cognition in a rat model of Alzheimer's disease. Journal of Medicinal Food . 2011;14(7-8):688–694. doi: 10.1089/jmf.2010.1312. [DOI] [PubMed] [Google Scholar]
- 186.Oboh G., Ademiluyi A. O., Akinyemi A. J. Inhibition of acetylcholinesterase activities and some pro-oxidant induced lipid peroxidation in rat brain by two varieties of ginger (Zingiber officinale) Experimental and Toxicologic Pathology . 2012;64(4):315–319. doi: 10.1016/j.etp.2010.11.018. [DOI] [PubMed] [Google Scholar]
- 187.Ali B. H., Blunden G., Tanira M. O., Nemmar A. Some phytochemical, pharmacological and toxicological properties of ginger (Zingiber officinale Roscoe): a review of recent research. Food and Chemical Toxicology . 2008;46(2):409–420. doi: 10.1016/j.fct.2021.111980. [DOI] [PubMed] [Google Scholar]
- 188.Huang H. Y., Huang J. J., Tso T. K., Tsai Y. C., Chang C. K. Antioxidant and angiotension converting enzyme inhibition capacities of various parts of Benincasa hispida (wax gourd) Food/Nahrung . 2004;48(3):230–233. doi: 10.1002/food.200300428. [DOI] [PubMed] [Google Scholar]
- 189.Li X. L., Zhao B. Q., Li Q., et al. Effects of Chinese herbal medicine fuzhisan on aged rats. Experimental Gerontology . 2008;43(9):853–858. doi: 10.1016/j.exger.2008.05.018. [DOI] [PubMed] [Google Scholar]
- 190.Zhao J., Wang D., Duan S., Wang J., Bai J., Li W. Analysis of Fuzhisan and quantitation of baicalin and ginsenoside Rb 1 by HPLC-DAD-ELSD. Archives of Pharmacal Research . 2009;32(7):989–996. doi: 10.1007/s12272-018-1049-8. [DOI] [PubMed] [Google Scholar]
- 191.Sanders O., Rajagopal L. Phosphodiesterase inhibitors for Alzheimer’s disease: a systematic review of clinical trials and epidemiology with a mechanistic rationale. Journal of Alzheimer's Disease Reports . 2020;4(1):185–215. doi: 10.3233/ADR-200191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Guo P., Wang D., Wang X., et al. Effect and mechanism of fuzhisan and donepezil on the sirtuin 1 pathway and amyloid precursor protein metabolism in PC12 cells. Molecular Medicine Reports . 2016;13(4):3539–3546. doi: 10.3892/mmr.2021.12481. [DOI] [PubMed] [Google Scholar]
- 193.Lim H.-S., Kim Y. J., Sohn E., Yoon J., Kim B.-Y., Jeong S.-J. Bojungikgi-Tang, a traditional herbal formula, exerts neuroprotective effects and ameliorates memory impairments in Alzheimer’s disease-like experimental models. Nutrients . 2018;10(12):p. 1952. doi: 10.3390/nu10121952. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Zahoor M., Zafar R., Rahman N. U. Isolation and identification of phenolic antioxidants from Pistacia integerrima gall and their anticholine esterase activities. Heliyon . 2018;4(12):p. e01007. doi: 10.1016/j.heliyon.2021.e06367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195.Uddin M. S., Al Mamun A., Hossain M. S., et al. Neuroprotective effect of Phyllanthus acidus L. on learning and memory impairment in scopolamine-induced animal model of dementia and oxidative stress: natural wonder for regulating the development and progression of Alzheimer’s disease. Advances in Alzheimer's Disease . 2016;5(2):53–72. [Google Scholar]
- 196.Hashmi W. J., Ismail H., Mehmood F., Mirza B. Neuroprotective, antidiabetic and antioxidant effect of Hedera nepalensis and lupeol against STZ+ AlCl 3 induced rats model. DARU Journal of Pharmaceutical Sciences . 2018;26(2):179–190. doi: 10.1007/s40199-018-0223-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197.Zhu B., Li Z., Qian P. Y., Herrup K. Marine bacterial extracts as a new rich source of drugs against Alzheimer’s disease. Journal of Neurochemistry . 2020;152(4):493–508. doi: 10.1111/jnc.15307. [DOI] [PubMed] [Google Scholar]
- 198.Ahn B. R., Moon H. E., Kim H. R., Jung H. A., Choi J. S. Neuroprotective effect of edible brown alga Eisenia bicyclis on amyloid beta peptide-induced toxicity in PC12 cells. Archives of Pharmacal Research . 2012;35(11):1989–1998. doi: 10.1007/s12272-012-1116-5. [DOI] [PubMed] [Google Scholar]
- 199.Khazdair M. R., Anaeigoudari A., Hashemzehi M., Mohebbati R. Neuroprotective potency of some spice herbs, a literature review. Journal of Traditional and Complementary Medicine . 2019;9(2):98–105. doi: 10.1016/j.jtcme.2018.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 200.Pulido-Moran M., Moreno-Fernandez J., Ramirez-Tortosa C., Ramirez-Tortosa M. Curcumin and health. Molecules . 2016;21(3):p. 264. doi: 10.3390/molecules21030264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201.Geun Kim H., Sook Oh M. Herbal medicines for the prevention and treatment of Alzheimer's disease. Current Pharmaceutical Design . 2012;18(1):57–75. doi: 10.2174/138161212798919002. [DOI] [PubMed] [Google Scholar]
- 202.Yuliani S., Mustofa, Partadiredja G. The neuroprotective effects of an ethanolic turmeric (Curcuma longa L.) extract against trimethyltin-induced oxidative stress in rats. Nutritional Neuroscience . 2019;22(11):797–804. doi: 10.1080/1028415X.2018.1447267. [DOI] [PubMed] [Google Scholar]
- 203.Kadri Y., Nciri R., Brahmi N., et al. Protective effects of Curcuma longa against neurobehavioral and neurochemical damage caused by cerium chloride in mice. Environmental Science and Pollution Research . 2018;25(20):19555–19565. doi: 10.1007/s11356-018-2151-7. [DOI] [PubMed] [Google Scholar]
- 204.Hishikawa N., Takahashi Y., Amakusa Y., et al. Effects of turmeric on Alzheimer's disease with behavioral and psychological symptoms of dementia. Ayu . 2012;33(4):499–504. doi: 10.4103/0974-8520.110524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 205.Ng T.-P., Chiam P.-C., Lee T., Chua H.-C., Lim L., Kua E.-H. Curry consumption and cognitive function in the elderly. American Journal of Epidemiology . 2006;164(9):898–906. doi: 10.1093/aje/kwz220. [DOI] [PubMed] [Google Scholar]
- 206.Mathew B., Biju R. Neuroprotective effects of garlic a review. Libyan Journal of Medicine . 2006;3(1):23–33. doi: 10.4176/071110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 207.Sripanidkulchai B. Benefits of aged garlic extract on Alzheimer's disease: possible mechanisms of action. Experimental and Therapeutic Medicine . 2020;19(2):1560–1564. doi: 10.3892/etm.2019.8390. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 208.Nillert N., Pannangrong W., Welbat J. U., Chaijaroonkhanarak W., Sripanidkulchai K., Sripanidkulchai B. Neuroprotective effects of aged garlic extract on cognitive dysfunction and neuroinflammation induced by β-amyloid in rats. Nutrients . 2017;9(1):p. 24. doi: 10.3390/nu9010024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 209.Hwang K.-A., Hwang Y.-J., Hwang I.-G., Song J., Kim Y. J. Low temperature-aged garlic extract suppresses psychological stress by modulation of stress hormones and oxidative stress response in brain. Journal of the Chinese Medical Association . 2019;82(3):191–195. doi: 10.1097/JCMA.0000000000000655. [DOI] [PubMed] [Google Scholar]
- 210.Huang H.-J., Chen S.-L., Chang Y.-T., Chyuan J.-H., Hsieh-Li H. M. Administration of Momordica charantia enhances the neuroprotection and reduces the side effects of LiCl in the treatment of Alzheimer’s disease. Nutrients . 2018;10(12):p. 1888. doi: 10.3390/nu10121888. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 211.Russo A., Borrelli F. Bacopa monniera, a reputed nootropic plant: an overview. Phytomedicine . 2005;12(4):305–317. doi: 10.1016/j.phymed.2006.09.010. [DOI] [PubMed] [Google Scholar]
- 212.Dhanasekaran M., Tharakan B., Holcomb L. A., Hitt A. R., Young K. A., Manyam B. V. Neuroprotective mechanisms of ayurvedic antidementia botanical Bacopa monniera. Phytotherapy Research . 2007;21(10):965–969. doi: 10.1002/ptr.2405. [DOI] [PubMed] [Google Scholar]
- 213.Uabundit N., Wattanathorn J., Mucimapura S., Ingkaninan K. Cognitive enhancement and neuroprotective effects of Bacopa monnieri in Alzheimer's disease model. Journal of Ethnopharmacology . 2010;127(1):26–31. doi: 10.1016/j.jep.2009.09.056. [DOI] [PubMed] [Google Scholar]
- 214.Bhattacharya S., Bhattacharya A., Kumar A., Ghosal S. Antioxidant activity ofBacopa monniera in rat frontal cortex, striatum and hippocampus. Phytotherapy Research . 2000;14(3):174–179. doi: 10.1002/ptr.7218. [DOI] [PubMed] [Google Scholar]
- 215.Limpeanchob N., Jaipan S., Rattanakaruna S., Phrompittayarat W., Ingkaninan K. Neuroprotective effect of _Bacopa monnieri_ on beta-amyloid-induced cell death in primary cortical culture. Journal of Ethnopharmacology . 2008;120(1):112–117. doi: 10.1016/j.jep.2008.07.039. [DOI] [PubMed] [Google Scholar]
- 216.Sadhu A., Upadhyay P., Agrawal A., et al. Management of cognitive determinants in senile dementia of Alzheimer’s type: therapeutic potential of a novel polyherbal drug product. Clinical Drug Investigation . 2014;34(12):857–869. doi: 10.1007/s40261-014-0235-9. [DOI] [PubMed] [Google Scholar]
- 217.Szurpnicka A., Kowalczuk A., Szterk A. Biological activity of mistletoe: in vitro and in vivo studies and mechanisms of action. Archives of Pharmacal Research . 2008;43(6):1–37. doi: 10.1007/s12272-020-01247-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 218.Moeini R., Memariani Z., Asadi F., Bozorgi M., Gorji N. Pistacia genus as a potential source of neuroprotective natural products. Planta Medica . 2019;85(17):1326–1350. doi: 10.1055/a-1014-1075. [DOI] [PubMed] [Google Scholar]
- 219.Liu Q. F., Jeon Y., Sung Y.-w., et al. Nardostachys jatamansi ethanol extract ameliorates Aβ42 cytotoxicity. Biological and Pharmaceutical Bulletin . 2018;41(4):470–477. doi: 10.1248/bpb.b17-00750. [DOI] [PubMed] [Google Scholar]
- 220.Liu Q. F., Lee J. H., Kim Y.-M., et al. In vivo screening of traditional medicinal plants for neuroprotective activity against Aβ42 cytotoxicity by using Drosophila models of Alzheimer’s disease. Biological and Pharmaceutical Bulletin . 2015;38:b15–00459. doi: 10.1248/bpb.b21-00691. [DOI] [PubMed] [Google Scholar]
- 221.Alagan A., Jantan I., Kumolosasi E., Ogawa S., Abdullah M. A., Azmi N. Protective effects of Phyllanthus amarus against lipopolysaccharide-induced neuroinflammation and cognitive impairment in rats. Frontiers in Pharmacology . 2019;10:p. 632. doi: 10.3389/fphar.2019.00632. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 222.Yu H., Yao L., Zhou H., et al. Neuroprotection against Aβ25-35-induced apoptosis by Salvia miltiorrhiza extract in SH-SY5Y cells. Neurochemistry International . 2014;75:89–95. doi: 10.1016/j.neuint.2014.06.001. [DOI] [PubMed] [Google Scholar]
- 223.Jiang W.-Y., Jeon B.-H., Kim Y.-C., Lee S. H., Sohn D. H., Seo G. S. PF2401-SF, standardized fraction of Salvia miltiorrhiza shows anti-inflammatory activity in macrophages and acute arthritis in vivo. International Immunopharmacology . 2013;16(2):160–164. doi: 10.1016/j.intimp.2021.108345. [DOI] [PubMed] [Google Scholar]
- 224.Hu L., Yu J., Li F., Chen B., Li L., Liu G. Effects of Salvia miltorrhiza in neural differentiation of rat mesenchymal stem cells with optimized protocol. Journal of Ethnopharmacology . 2011;136(2):334–340. doi: 10.1016/j.jep.2022.115049. [DOI] [PubMed] [Google Scholar]
- 225.Shu T., Pang M., Rong L., et al. Effects of Salvia miltiorrhiza on neural differentiation of induced pluripotent stem cells. Journal of Ethnopharmacology . 2014;153(1):233–241. doi: 10.1016/j.jep.2014.02.028. [DOI] [PubMed] [Google Scholar]
- 226.Dar N. J., Glazner G. W. Deciphering the neuroprotective and neurogenic potential of soluble amyloid precursor protein alpha (sAPPα) Cellular and Molecular Life Sciences . 2020;77(12):2315–2330. doi: 10.1007/s00018-019-03404-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 227.He P., Schulz P., Sierks M. R. A conformation-specific antibody against oligomeric β-amyloid restores neuronal integrity in a mouse model of Alzheimer's disease. Journal of Biological Chemistry . 2021;296:p. 100241. doi: 10.1074/jbc.RA120.015327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 228.Kabir M. T., Uddin M., Mathew B., Das P. K., Perveen A., Ashraf G. M. Emerging promise of immunotherapy for Alzheimer’s disease: a new hope for the development of Alzheimer’s vaccine. Current Topics in Medicinal Chemistry . 2020;20(13):1214–1234. doi: 10.2174/1568026620666200618114924. [DOI] [PubMed] [Google Scholar]
- 229.Lackie R. E., Marques-Lopes J., Ostapchenko V. G., et al. Increased levels of Stress-inducible phosphoprotein-1 accelerates amyloid-β deposition in a mouse model of Alzheimer’s disease. Acta Neuropathologica Communications . 2020;8(1):119–143. doi: 10.1186/s40478-020-01013-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 230.Lobello K., Ryan J. M., Liu E., Rippon G., Black R. Targeting beta amyloid: a clinical review of immunotherapeutic approaches in Alzheimer's disease. International Journal of Alzheimer’s Disease . 2012;2012:14. doi: 10.1155/2012/628070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 231.Vassar R., Kandalepas P. C. The β-secretase enzyme BACE1 as a therapeutic target for Alzheimer's disease. Alzheimer's Research & Therapy . 2011;3(3):1–6. doi: 10.1186/alzrt82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 232.Li Y., Zhou W., Tong Y., He G., Song W. Control of APP processing and Aβ generation level by BACE1 enzymatic activity and transcription. The FASEB Journal . 2006;20(2):285–292. doi: 10.1096/fj.202101307R. [DOI] [PubMed] [Google Scholar]
- 233.Zhang C. Natural compounds that modulate BACE1-processing of amyloid-beta precursor protein in Alzheimer’s disease. Discovery Medicine . 2012;14(76):189–197. [PubMed] [Google Scholar]
- 234.De Strooper B., Iwatsubo T., Wolfe M. S. Presenilins and Secretase: Structure, Function, and role in Alzheimer disease. Cold Spring Harbor Perspectives in Medicine . 1998;2(1):p. a006304. doi: 10.1101/cshperspect.a006304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 235.Escamilla-Ayala A., Wouters R., Sannerud R., Annaert W. Contribution of the presenilins in the cell biology, structure and function of γ-secretase. In Seminars in Cell & Developmental Biology . 2020;105:12–26. doi: 10.1016/j.semcdb.2020.02.005. [DOI] [PubMed] [Google Scholar]
- 236.Kreft A. F., Martone R., Porte A. Recent advances in the identification of γ-secretase inhibitors to clinically test the Aβ oligomer hypothesis of Alzheimer’s disease. Journal of Medicinal Chemistry . 2009;52(20):6169–6188. doi: 10.1021/jm900188z. [DOI] [PubMed] [Google Scholar]
- 237.Mayer S. C., Kreft A. F., Harrison B., et al. Discovery of begacestat, a Notch-1-sparing γ-secretase inhibitor for the treatment of Alzheimer’s disease. Journal of Medicinal Chemistry . 2020;51(23):7348–7351. doi: 10.1021/jm801252w. [DOI] [PubMed] [Google Scholar]
- 238.Hanger D., Anderton B., Noble W. Phosphorylation: the therapeutic challenge for neurodegenerative disease. Trends Mol. Med . 2009;15:112–119. doi: 10.1016/j.molmed.2009.01.003. [DOI] [PubMed] [Google Scholar]
- 239.Kolarova M., García-Sierra F., Bartos A., Ricny J., Ripova D. Structure and pathology of tau protein in Alzheimer disease. International Journal of Alzheimer’s Disease . 2012;2012:13. doi: 10.1155/2012/731526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 240.Santacruz K., Lewis J., Spires T., et al. Tau suppression in a neurodegenerative mouse model improves memory function. Science . 2005;309(5733):476–481. doi: 10.1126/science.1113694. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 241.Andorfer C., Acker C. M., Kress Y., Hof P. R., Duff K., Davies P. Cell-cycle reentry and cell death in transgenic mice expressing nonmutant human tau isoforms. Journal of Neuroscience . 2005;25(22):5446–5454. doi: 10.1523/JNEUROSCI.4637-04.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 242.Hernández F., de Barreda E. G., Fuster-Matanzo A., Lucas J. J., Avila J. GSK3: a possible link between beta amyloid peptide and tau protein. Experimental Neurology . 2010;223(2):322–325. doi: 10.1016/j.expneurol.2021.113837. [DOI] [PubMed] [Google Scholar]
- 243.Lee H.-K., Kumar P., Fu Q., Rosen K. M., Querfurth H. W. The insulin/Akt signaling pathway is targeted by intracellular β-amyloid. Molecular Biology of the Cell . 2009;20(5):1533–1544. doi: 10.1091/mbc.E20-10-0631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 244.Georgievska B., Sandin J., Doherty J., et al. AZD 1080, a novel GSK 3 inhibitor, rescues synaptic plasticity deficits in rodent brain and exhibits peripheral target engagement in humans. Journal of Neurochemistry . 2013;125(3):446–456. doi: 10.1111/jnc.12203. [DOI] [PubMed] [Google Scholar]
- 245.Sugaya K., Giacobini E., Chiappinelli V. Nicotinic acetylcholine receptor subtypes in human frontal cortex: changes in Alzheimer's disease. Journal of Neuroscience Research . 1990;27(3):349–359. doi: 10.1002/jnr.490270314. [DOI] [PubMed] [Google Scholar]
- 246.Kihara T., Shimohama S., Urushitani M., et al. Stimulation of α 4 β 2 nicotinic acetylcholine receptors inhibits β -amyloid toxicity. Brain Research . 1998;792(2):331–334. doi: 10.1016/S0006-8993(98)00138-3. [DOI] [PubMed] [Google Scholar]
- 247.Buckingham S. D., Jones A. K., Brown L. A., Sattelle D. B. Nicotinic acetylcholine receptor signalling: roles in Alzheimer's disease and amyloid neuroprotection. Pharmacological Reviews . 2009;61(1):39–61. doi: 10.1124/pr.108.000562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 248.Alonso E., Vale C., Vieytes M. R., Laferla F. M., Giménez-Llort L., Botana L. M. The cholinergic antagonist gymnodimine improves Aβ and tau neuropathology in an in vitro model of Alzheimer disease. Cellular Physiology and Biochemistry . 2011;27(6):783–794. doi: 10.1159/000330086. [DOI] [PubMed] [Google Scholar]
- 249.Mehta M., Adem A., Sabbagh M. New acetylcholinesterase inhibitors for Alzheimer's disease. International Journal of Alzheimer’s Disease . 2012;2012:8. doi: 10.1155/2012/728983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 250.Sramek J. J., Frackiewicz E. J., Cutler N. R. Review of the acetylcholinesterase inhibitor galanthamine. Expert Opinion on Investigational Drugs . 2000;9(10):2393–2402. doi: 10.1517/13543784.10.4.741. [DOI] [PubMed] [Google Scholar]
- 251.Akhondzadeh S., Noroozian M., Mohammadi M., Ohadinia S., Jamshidi A., Khani M. Melissa officinalis extract in the treatment of patients with mild to moderate Alzheimer’s disease: a double blind, randomised, placebo controlled trial. Journal of Neurology, Neurosurgery & Psychiatry . 2003;74(7):863–866. doi: 10.1136/jnnp.74.7.863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 252.Akhondzadeh S., Noroozian M. Alzheimer's disease: pathophysiology and pharmacotherapy. IDrugs: The Investigational Drugs Journal . 2002;5(11):1062–1069. [PubMed] [Google Scholar]
- 253.Mok V. C., Pendlebury S., Wong A., et al. Tackling challenges in care of Alzheimer's disease and other dementias amid the COVID-19 pandemic, now and in the future. Alzheimer's & Dementia . 2020;16(11):1571–1581. doi: 10.1002/alz.12143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 254.Ning H., Li R., Ye X., Zhang Y., Liu L. A review on serious games for dementia care in ageing societies. IEEE Journal of Translational Engineering in Health and Medicine . 2020;8:1–11. doi: 10.1109/JTEHM.2020.2998055. [DOI] [PMC free article] [PubMed] [Google Scholar]


