Abstract
The present study aimed to investigate the cardioprotective role of berberine in sepsis-induced cardiac dysfunction and consider the underlying mechanisms. C57BL/6J mice were randomized into four groups, namely, Control, lipopolysaccharide (LPS), LPS + berberine and LPS + Nω-nitro-L-arginine methyl ester (L-NAME) + berberine. A single dose (10 mg/kg body weight) of LPS was intraperitoneally administered to mice to induce cardiac dysfunction, whereas the Control group was administered with an equivalent volume of saline. In the LPS + berberine and LPS + L-NAME + berberine group, berberine (10 mg/kg body weight) dissolved in hot water was intraperitoneally administered 30 min after the LPS treatment. In the LPS + L-NAME + berberine group, L-NAME (100 mg/kg body weight) dissolved in saline was intraperitoneally administered 30 min before the LPS treatment. Then, ~6 h after the LPS treatment, a significant decrease was observed in the left ventricular ejection fraction (LVEF) and left ventricular fractional shortening (LVFS). Meanwhile, the plasma myocardial injury markers, inflammatory factors and oxidative stress levels were significantly increased in the LPS group compared with the Control group. The administration of berberine improved the ventricular function and decreased the plasma myocardial injury markers, inflammatory factors and oxidative stress levels. In addition, it increased the heart total nitric oxide synthase (NOS) activity and upregulated the protein expressions of p-Akt and phosphorylated endothelial (e)NOS, which indicated that the Akt/eNOS pathway was activated by berberine. However, the cardioprotective effects of berberine were counteracted by L-NAME, an NOS inhibitor, which inhibited the eNOS activity. In conclusion, berberine attenuated sepsis-induced cardiac dysfunction by upregulating the Akt/eNOS pathway in mice.
Keywords: berberine, cardiac dysfunction, sepsis, Akt, endothelial nitric oxide synthase
Introduction
Sepsis is a life-threatening organ dysfunction caused by a dysregulated host response to an infection. It is currently the major cause of morbidity and mortality in intensive care units worldwide (1). The heart is one of the important target organs in sepsis and a large number of studies have demonstrated that ~50% of patients with sepsis exhibited cardiac dysfunction (2,3). It has been reported that septic patients with cardiac dysfunction had significantly higher mortality rates compared with those without cardiac dysfunction (4). The degree of myocardial structural damage and functional impairment is associated with the clinical adverse outcomes of the illness. Although much remains unknown, over the past decades, substantial research has improved the understanding of its underlying pathophysiologic mechanisms, including an excessive inflammatory response, autonomic nervous system dysregulation, endothelial dysfunction, cardiac autophagy and apoptosis, calcium regulation disorders and metabolic reprogramming (5-10). Among these factors, nitric oxide (NO) synthesis impairment serves a crucial role in sepsis-induced cardiac dysfunction (11). NO is endogenously produced from L-arginine by nitric oxide synthase (NOS), an enzyme with three isoforms; ineuronal, endothelial and inducible NOS (nNOS, eNOS and iNOS, respectively). In addition, it serves a wide range of physiological and pathophysiological roles in the cardiovascular system (12). Some studies have demonstrated that sepsis-induced cardiac dysfunction is caused by enhanced iNOS activity and resultant NO (13,14). Conversely, another study revealed that the brain-derived neurotrophic factor attenuated cardiac dysfunction by increasing myocardial eNOS expression in sepsis models (15). In view of this, it is particularly important to understand the role that NOS serves in sepsis-induced cardiac dysfunction. Thus, despite improvements in antibiotic therapies and supportive care, but due to the lack of specific treatment, the expected results in clinical applications have not been achieved and new therapeutic interventions need to be explored.
Berberine is a natural pentacyclic isoquinoline alkaloid that is the principal bioactive ingredient of Rhizoma coptidis (also named Huang Lian in Chinese). It is also the principal component of many other medicinal herbs. Berberine exhibits a wide spectrum of pharmacological effects and is widely used in clinical conditions for the treatment of different diseases, including hypertension, stroke, diabetes, cancers, atherosclerosis and viral infections (16-18). It has been demonstrated that berberine functions as a negative regulator in lipopolysaccharide (LPS)-induced sepsis and reduces sepsis-related organ damage by suppressing inflammatory responses (19-21). However, the cardioprotective effect of berberine on sepsis-induced cardiac dysfunction has not been fully understood and the underlying mechanisms remain unclear.
Thus, the present study aimed to investigate the cardioprotective role of berberine in sepsis-induced cardiac dysfunction. It aimed to determine whether the cardioprotective effects of berberine were mediated by upregulating the Akt/eNOS pathway.
Materials and methods
Animals and treatment
All animal experimental procedures were conducted in accordance with the Guidelines for the Care and Use of Laboratory Animals (8th edition; pubmed.ncbi.nlm.nih.gov/21595115/) and were approved by the ethics committee of Bengbu Medical College [approval no. (2020) 211]. Male C57BL/6J 8-12-week-old mice were fed a standard laboratory diet and tap water ad libitum and were housed in plastic cages in a room with a 12-h light/dark cycle, temperature of 22-24˚C and humidity of 60%. After 1 week of acclimation, the mice were randomly divided into the following four groups (n=6 in each group): Control, LPS, LPS + berberine and LPS + Nω-nitro-L-arginine methyl ester (L-NAME) + berberine. To develop a mouse septic cardiac dysfunction model, a single dose (10-mg/kg body weight) of LPS was administered intraperitoneally (22) and the Control group was intraperitoneally administered with the equivalent volume of saline. In the LPS + berberine and LPS + L-NAME + berberine groups, berberine (10-mg/kg body weight) (23) dissolved in hot water was administered intraperitoneally 30 min after the LPS treatment. In the LPS + L-NAME + berberine group, L-NAME (100-mg/kg body weight) (24) dissolved in saline was administered intraperitoneally 30 min before the LPS treatment. Berberine and LPS were purchased from Beijing Solarbio Science & Technology Co., Ltd. L-NAME was purchased from MilliporeSigma.
Echocardiographic study
After 6 h of LPS treatment, the mice were anesthetized with 1.5-2% of isoflurane and echocardiography was performed to assess cardiac function using a Vevo 2100 ultrasound device (FUJIFILM VisualSonics Inc.). In total, three consecutive cardiac cycle measurements were averaged and the left ventricular ejection fraction (LVEF) and left ventricular fractional shortening (LVFS) were measured to evaluate heart function. Subsequently, mice were euthanized by an overdose of sodium pentobarbital (250 mg/kg; intraperitoneal injection). Blood was collected from the eyelids of the mice and placed into tubes containing 4% sodium citrate. The plasma was then centrifuged for 10 min at 1,000 x g at 4˚C and the heart tissues were obtained and frozen at -80˚C.
Plasma biochemical and inflammatory cytokine analysis
The plasma levels of lactate dehydrogenase (LDH), creatine kinase (CK) and creatine kinase-MB (CK-MB) were determined using corresponding assay kits (cat. nos. A020-2-2, A032-1-1 and H197-1-2 for CK-MB, Nanjing Jiancheng Bioengineering Institute) according to the manufacturer's protocols. The plasma inflammatory cytokines, tumor necrosis factor (TNF)-α and interleukin (IL)-1β were measured using ELISA kits (cat. nos. MTA00B for TNF-α, MLB00C for IL-1β, R&D Systems, Inc.) according to the manufacturer's protocols.
Measurement of oxidative stress
The heart tissues were homogenized with 50-mmol/l potassium phosphate buffer. Following centrifugation for 5 min at 12,000 g at 4˚C, the supernatant was used to measure the oxidative stress levels. The levels of cardiac hydrogen peroxide (H2O2), malondialdehyde (MDA) and glutathione (GSH) were determined using the corresponding assay kits (S0038 for H2O2, S0131S for MDA, S0053 for GSH, Beyotime Institute of Biotechnology) and the activity of cardiac superoxide dismutase (SOD) was measured using a total SOD assay kit with WST-8 (S0101S, Beyotime Institute of Biotechnology) according to the manufacturer's protocols. The above values were standardized by protein content, determined using a bicinchoninic acid (BCA) protein assay kit (P0010S, Beyotime Institute of Biotechnology).
Measurement of the heart NOS activity
The heart NOS activity was measured using the Griess method with the corresponding assay kits (Nanjing Jiancheng Bioengineering Institute) according to the manufacturer's protocols. The heart NOS activity was standardized by protein content that was determined using the BCA protein assay kit.
Western blot analysis
Protein used for western blot was extracted from the myocardial tissue using RIPA lysis buffer containing phosphatase and a protease inhibitor cocktail (Beyotime Institute of Biotechnology) and then quantified using a BCA protein assay kit. Equal amounts of protein (80 µg/lane) were separated with 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) and then transferred onto polyvinylidene fluoride membranes (MilliporeSigma). After blocking with 5% non-fat milk at 4˚C for 1 h, the membranes were incubated with primary antibodies, namely, phosphorylated (p)-Akt (Ser473, 1:1,000, ProteinTech Group, Inc.), Akt (1:1,000, ProteinTech Group, Inc.), p-eNOS (Ser1177, 1:1,000, Abcam) and eNOS (1:1,000, Abcam) at 4˚C overnight. GAPDH (1:1,000, ProteinTech Group, Inc.) was used as the loading control. After washing with TBST three times, the membranes were incubated with horseradish peroxidase-conjugated secondary antibodies (cat. no. SA00001-2;1:2,000; ProteinTech Group, Inc.) at room temperature for 1 h and the target signal was visualized using the ECL exposure system. The intensity of the bands was quantified using an image analysis system (ImageJ V1.8.0; National Institutes of Health).
Statistical analysis
All statistical analyses were conducted using SPSS software, version 21.0 (IBM Corp.). The data were expressed as mean ± SEM and the differences between ≥ groups were assessed using one-way analysis of variance followed by Tukey post hoc tests. P<0.05 was considered to indicate a statistically significant difference.
Results
Berberine improved cardiac function in LPS-induced septic mice, but it was inhibited by L-NAME
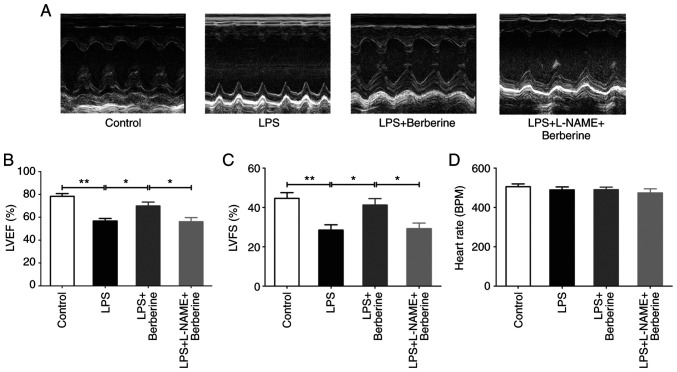
As presented in Fig. 1A-C, echocardiography revealed that there was a significant decrease in the LVEF and LVFS after LPS injection compared with the Control group. Berberine treatment increased LVEF and LVFS as compared with the LPS group. However, LVEF and LVFS were significantly decreased in LPS + L-NAME + berberine group as compared with LPS + berberine group. There were no significant differences in heart rate among the four groups (Fig. 1D).
Figure 1.
Berberine improved cardiac function in LPS-induced septic mice, but it was inhibited by L-NAME. (A) Representative M-mode images. Changes of (B) LVEF, (C) LVFS and (D) HR. Results are means ± SEM. *P<0.05 and **P<0.01 were considered significant. LPS, lipopolysaccharide; L-NAME, Nω-nitro-L-arginine methyl ester; LVEF, left ventricular ejection fraction; LVFS, left ventricular fractional shortening; HR, heart rate.
Berberine alleviated cardiac injury in LPS-induced septic mice, but it was inhibited by L-NAME
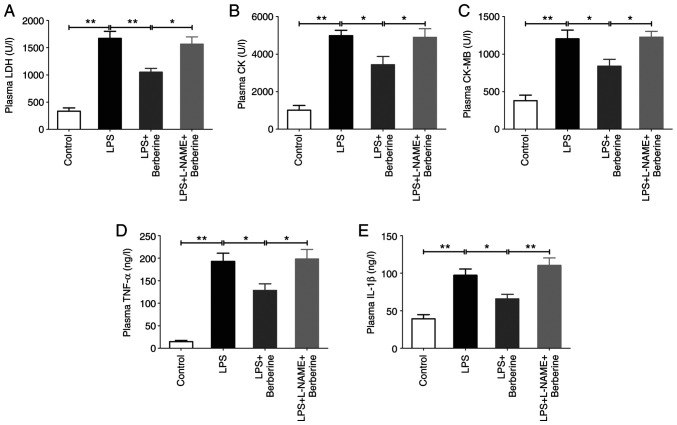
The levels of plasma LDH, CK and CK-MB, which are myocardial injury markers, were significantly increased in the LPS group compared with the Control group (Fig. 2A-C). The inflammatory factors TNF-α and IL-1β in plasma were also significantly increased in the LPS group(Fig. 2D and E). However, berberine treatment decreased plasma LDH, CK, CK-MB, TNF-α and IL-1β levels as compared with the LPS group, which was inhibited by L-NAME pre-treatment.
Figure 2.
Berberine alleviated cardiac injury in LPS-induced septic mice, but it was inhibited by L-NAME. (A) LDH, (B) CK, (C) CK-MB, (D) TNF-α and (E) IL-1β levels in the plasma. Results are means ± SEM. *P<0.05 and **P<0.01 were considered significant. LPS, lipopolysaccharide; L-NAME, Nω-nitro-L-arginine methyl ester; LDH, lactate dehydrogenase; CK, Creatine kinase; CK-MB, Creatine kinase-MB.
Berberine reduced oxidative stress in LPS-induced cardiac dysfunction, but it was inhibited by L-NAME
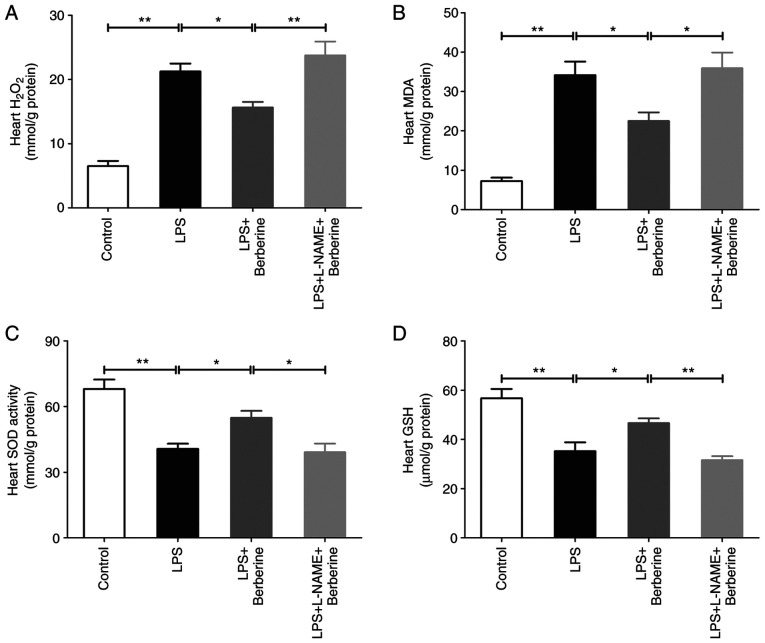
Sepsis-induced cardiac dysfunction was associated with oxidative stress injury. As presented in Fig. 3A and B, the heart H2O2 and MDA levels significantly increased after the LPS injection compared with the Control group, whereas the berberine treatment markedly attenuated the elevation of H2O2 and MDA in the hearts of the LPS-induced septic mice. There was also a significant decrease in the activity of SOD and levels of GSH after the LPS injection as compared with the Control group and berberine treatment significantly enhanced the activity of SOD and level of GSH in the hearts of the LPS-induced septic mice (Fig. 3C and D). L-NAME pre-treatment increased heart H2O2 and MDA levels in the LPS + berberine group, but decreased SOD activity and GSH levels.
Figure 3.
Berberine reduced oxidative stress in LPS-induced cardiac dysfunction, but it was inhibited by L-NAME. (A) H2O2 and (B) MDA levels in heart tissues. (C) SOD activities in heart tissues. (D) GSH levels in heart tissues. Results are means ± SEM. *P<0.05 and **P<0.01 were considered significant. LPS, lipopolysaccharide; L-NAME, Nω-nitro-L-arginine methyl ester; MDA, malondialdehyde; SOD, superoxide dismutase; GSH, glutathione.
Berberine upregulated the Akt/eNOS pathway in LPS-induced cardiac dysfunction
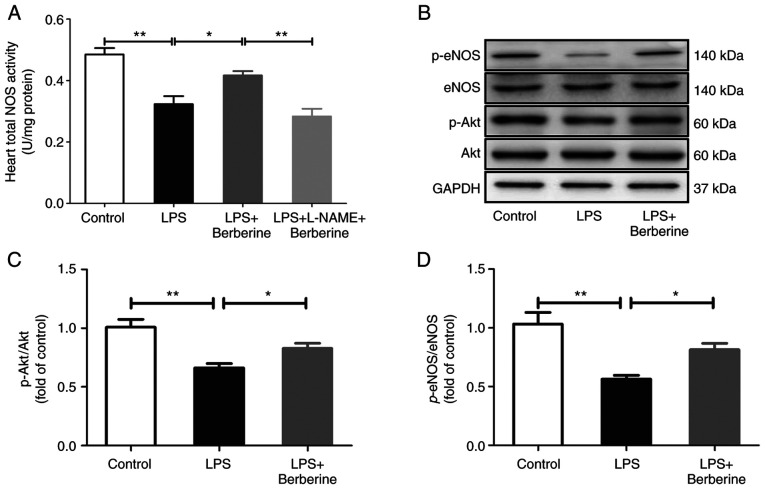
As presented in Fig. 4A, the heart total NOS activity was decreased after the LPS injection as compared with the Control group. There was also a significant decrease in the protein levels of p-Akt at Ser473 (Fig. 4C) and p-eNOS at Ser1177 (Fig. 4D). The berberine treatment increased the heart total NOS activity which was inhibited by L-NAME pre-treatment and berberine treatment also upregulated the protein expressions of p-Akt and p-eNOS.
Figure 4.
Berberine upregulated Akt/eNOS pathway in LPS-induced cardiac dysfunction. (A) Total NOS activities in heart tissues. (B-D) Representative western blots and quantitative analysis for p-Akt/Akt and p-eNOS/eNOS expression. GAPDH was used as the internal control. Results are means ± SEM. *P<0.05 and **P<0.01 were considered significant. eNOS, endothelial nitric oxide synthase; p-, phosphorylated.
Discussion
The present study found that berberine had a protective effect on sepsis-induced cardiac dysfunction and this was accompanied by a decrease in myocyte injury marker enzymes, which reduced cardiac inflammation and oxidative stress. Mechanistically, it provided evidence showing that berberine upregulated the Akt/eNOS pathway by which it ameliorated sepsis-induced cardiac dysfunction.
Sepsis-induced cardiac dysfunction, which is defined as a global but reversible dysfunction (systolic and diastolic) of the heart induced by sepsis, is extremely common and contributes to the adverse clinical outcomes of patients with sepsis (25). As a bioactive alkaloid in traditional Chinese medicine, berberine has been used in critical care medicine to treat sepsis and sepsis-related organ failure according to its various pharmacological properties (26). Although it is shown to have beneficial effects (27,28), the therapeutic effects of berberine for sepsis-induced cardiac dysfunction have not been studied in detail to date. In the present study, mice were intraperitoneally injected with 10-mg/kg body LPS to induce cardiac dysfunction, which is a widely accepted noninvasive reliable model (29,30). After 6 h of LPS treatment, cardiac function was severely impaired, as evidenced by the decreases in LVEF and LVFS and the increase in myocardial injury markers. Berberine administration could significantly increase LVEF and LVFS and decrease myocardial injury markers. The above results were in line with a recent report that found that berberine attenuated septic cardiomyopathy by inhibiting TLR4/NF-κB signaling in rats (31).
It is well known that inflammation and oxidative stress serve a key role in cardiac dysfunction induced by sepsis. After activation by LPS, proinflammatory cytokines, such as TNF-α and IL-1β, are released to promote inflammatory reaction and then, they induce oxidative stress injury. However, the inhibition of inflammation and oxidative stress injury can ameliorate sepsis-induced cardiac dysfunction (32,33). The present study found that berberine suppressed inflammatory responses and ameliorated oxidative stress injury, as evidenced by the significant decrease in the levels of H2O2 and MDA and enhancement of the activity of SOD and levels of GSH in the heart. A growing body of evidence has suggested that NO is an antioxidant and serves an anti-inflammatory role and that the downregulation and uncoupling of the NOS protein leads to inflammation and increased oxidative stress (34,35). In addition, NO serves a crucial role in sepsis-induced cardiac dysfunction, although this is still controversial. It has been reported that cardiac dysfunction in sepsis is caused by the induction of NO production via iNOS hyperactivity (36) and that the inhibition of iNOS reversed cardiac dysfunction (10). On the other hand, other studies demonstrate that high doses of L-N-monomethyl arginine, a nonselective NOS inhibitor, increases mortality in patients with septic shock and cardiomyocyte-specific overexpression of eNOS decreases systemic inflammation and prevents cardiac dysfunction in the murine models of septic shock (37,38). In the present study, the heart total NOS activity and the protein levels of p-eNOS at Ser1177 were decreased following the LPS injection, whereas the berberine treatment increased the total NOS activity in the heart and upregulated the protein expressions of p-eNOS, which indicated that the eNOS activity was increased by berberine. The results were in agreement with the study by Wang et al (39), who found that berberine prevents hyperglycemia-induced endothelial injury and enhances vasodilatation by enhancing phosphorylation of eNOS at Ser1177. It is well known that phosphorylation of the amino acids Ser1177 in eNOS is facilitated by the Akt pathway activation (40). The results of the present study indicated that there was a significant decrease in the protein levels of p-Akt at Ser473 following LPS treatment, whereas berberine upregulated the protein expressions of p-Akt. Berberine is reported to exert an anti-apoptotic effect and improve cardiac functional recovery following myocardial ischemia/reperfusion by activating PI3K-Akt-eNOS signaling in diabetic rats (41). It is also reported that berberine protects endothelial progenitor cells from TNF-α damage via the PI3K/AKT/eNOS signaling pathway (42). L-NAME is used to inhibit the activity of eNOS (43). The results of the present study indicated that cardiac function was significantly decreased in the LPS + L-NAME +berberine group compared with the LPS + berberine group, which was accompanied by a significant decrease in the heart total NOS activity. The myocardial injury markers, inflammatory factors and heart oxidative stress levels were significantly increased after the L-NAME pre-treatment. These data were in line with the report of Bougaki et al (44), who found that eNOS knockout mice exhibit shorter survival times in murine polymicrobial sepsis and that eNOS deficiency worsens systemic inflammation and exaggerated myocardial dysfunction.
There are several limitations to the present study. One limitation is that L-NAME is just a non-specific NOS inhibitor. So eNOS knockout mice should be used for the further studies and p-eNOS and p-Akt expression changes should be detected after L-NAME treatment or eNOS knockout. Another limitation is that the present study was designed as pre-clinical animal experiment. Whether it can be applied direct to clinical practice, requires further work.
In conclusion, the present study suggested that berberine could attenuate sepsis-induced cardiac dysfunction by upregulating the Akt/eNOS pathway. Therefore, berberine has the potential to become a therapeutic medicine for septic patients with cardiac dysfunction.
Acknowledgements
Not applicable.
Funding Statement
Funding: The present study was supported by fund of Bengbu Medical College (grant no. BYKY2019053ZD).
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Authors' contributions
HZ and GYL designed the experiments and wrote the manuscript. HZ and YYT performed the experiments. HZ and XFW analyzed and interpreted the data. XFW and GYL confirm the authenticity of all the raw data. All authors read and approved the final manuscript.
Ethics approval and consent to participate
All animal experimental procedures were conducted in accordance with the Guidelines for the Care and Use of Laboratory Animals (8th edition, pubmed.ncbi.nlm.nih.gov/21595115/) and were approved by the ethics committee of Bengbu Medical College [approval no. (2020) 211].
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Singer M, Deutschman CS, Seymour CW, Shankar-Hari M, Annane D, Bauer M, Bellomo R, Bernard GR, Chiche JD, Coopersmith CM, et al. The third international consensus definitions for sepsis and septic shock (Sepsis-3) JAMA. 2016;315:801–810. doi: 10.1001/jama.2016.0287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.L'Heureux M, Sternberg M, Brath L, Turlington J, Kashiouris MG. Sepsis-induced cardiomyopathy: A comprehensive review. Curr Cardiol Rep. 2020;22(35) doi: 10.1007/s11886-020-01277-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Wang J, Wang XT, Liu DW, Zhang HM, Su LX. Induction and deduction in sepsis-induced cardiomyopathy: Five typical categories. Chin Med J (Engl) 2020;133:2205–2211. doi: 10.1097/CM9.0000000000000929. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Landesberg G, Gilon D, Meroz Y, Georgieva M, Levin PD, Goodman S, Avidan A, Beeri R, Weissman C, Jaffe AS, Sprung CL. Diastolic dysfunction and mortality in severe sepsis and septic shock. Eur Heart J. 2012;33:895–903. doi: 10.1093/eurheartj/ehr351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Li N, Zhou H, Wu H, Wu Q, Duan M, Deng W, Tang Q. STING-IRF3 contributes to lipopolysaccharide-induced cardiac dysfunction, inflammation, apoptosis and pyroptosis by activating NLRP3. Redox Biol. 2019;24(101215) doi: 10.1016/j.redox.2019.101215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Carrara M, Ferrario M, Bollen Pinto B, Herpain A. The autonomic nervous system in septic shock and its role as a future therapeutic target: A narrative review. Ann Intensive Care. 2021;11(80) doi: 10.1186/s13613-021-00869-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Winkler MS, Nierhaus A, Poppe A, Greiwe G, Gräler MH, Daum G. Sphingosine-1-Phosphate: A potential biomarker and therapeutic target for endothelial dysfunction and sepsis? Shock. 2017;47:666–672. doi: 10.1097/SHK.0000000000000814. [DOI] [PubMed] [Google Scholar]
- 8.Sun Y, Yao X, Zhang QJ, Zhu M, Liu ZP, Ci B, Xie Y, Carlson D, Rothermel BA, Sun Y, et al. Beclin-1-dependent autophagy protects the heart during sepsis. Circulation. 2018;138:2247–2262. doi: 10.1161/CIRCULATIONAHA.117.032821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Sepúlveda M, Gonano LA, Viotti M, Morell M, Blanco P, López Alarcón M, Peroba Ramos I, Bastos Carvalho A, Medei E, Vila Petroff M. Calcium/calmodulin protein kinase ii-dependent ryanodine receptor phosphorylation mediates cardiac contractile dysfunction associated with sepsis. Crit Care Med. 2017;45:e399–e408. doi: 10.1097/CCM.0000000000002101. [DOI] [PubMed] [Google Scholar]
- 10.Hollenberg SM, Singer M. Pathophysiology of sepsis-induced cardiomyopathy. Nat Rev Cardiol. 2021;18:424–434. doi: 10.1038/s41569-020-00492-2. [DOI] [PubMed] [Google Scholar]
- 11.Kawaguchi S, Okada M, Ijiri E, Koga D, Watanabe T, Hayashi K, Kashiwagi Y, Fujita S, Hasebe N. β3-Adrenergic receptor blockade reduces mortality in endotoxin-induced heart failure by suppressing induced nitric oxide synthase and saving cardiac metabolism. Am J Physiol Heart Circ Physiol. 2020;318:H283–H294. doi: 10.1152/ajpheart.00108.2019. [DOI] [PubMed] [Google Scholar]
- 12.Farah C, Michel LYM, Balligand JL. Nitric oxide signalling in cardiovascular health and disease. Nat Rev Cardiol. 2018;15:292–316. doi: 10.1038/nrcardio.2017.224. [DOI] [PubMed] [Google Scholar]
- 13.Ndongson-Dongmo B, Lang GP, Mece O, Hechaichi N, Lajqi T, Hoyer D, Brodhun M, Heller R, Wetzker R, Franz M, et al. Reduced ambient temperature exacerbates SIRS-induced cardiac autonomic dysregulation and myocardial dysfunction in mice. Basic Res Cardiol. 2019;114(26) doi: 10.1007/s00395-019-0734-1. [DOI] [PubMed] [Google Scholar]
- 14.Ndongson-Dongmo B, Heller R, Hoyer D, Brodhun M, Bauer M, Winning J, Hirsch E, Wetzker R, Schlattmann P, Bauer R. Phosphoinositide 3-kinase gamma controls inflammation-induced myocardial depression via sequential cAMP and iNOS signalling. Cardiovasc Res. 2015;108:243–253. doi: 10.1093/cvr/cvv217. [DOI] [PubMed] [Google Scholar]
- 15.Zeng N, Xu J, Yao W, Li S, Ruan W, Xiao F. Brain-derived neurotrophic factor attenuates septic myocardial dysfunction via eNOS/NO pathway in rats. Oxid Med Cell Longev. 2017;2017(1721434) doi: 10.1155/2017/1721434. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Cai Y, Xin Q, Lu J, Miao Y, Lin Q, Cong W, Chen K. A new therapeutic candidate for cardiovascular diseases: Berberine. Front Pharmacol. 2021;12(631100) doi: 10.3389/fphar.2021.631100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Yang S, Li D, Yu Z, Li Y, Wu M. Multi-pharmacology of berberine in atherosclerosis and metabolic diseases: Potential contribution of gut microbiota. Front Pharmacol. 2021;12(709629) doi: 10.3389/fphar.2021.709629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Šudomová M, Berchová-Bímová K, Marzocco S, Liskova A, Kubatka P, Hassan STS. Berberine in human oncogenic herpesvirus infections and their linked cancers. Viruses. 2021;13(1014) doi: 10.3390/v13061014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Yuan C, Wu M, Xiao Q, Zhao W, Li H, Zhong Y, Zhao M, Li C, Li Y, Yang X. Blocking Msr1 by berberine alkaloids inhibits caspase-11-dependent coagulation in bacterial sepsis. Signal Transduct Target Ther. 2021;6(92) doi: 10.1038/s41392-021-00483-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Wang Y, Du P, Jiang D. Berberine functions as a negative regulator in lipopolysaccharide-induced sepsis by suppressing NF-κB and IL-6 mediated STAT3 activation. Pathog Dis. 2020;78(ftaa047) doi: 10.1093/femspd/ftaa047. [DOI] [PubMed] [Google Scholar]
- 21.He Y, Yuan X, Zuo H, Li X, Sun Y, Feng A. Berberine induces ZIP14 expression and modulates zinc redistribution to protect intestinal mucosal barrier during polymicrobial sepsis. Life Sci. 2019;233(116697) doi: 10.1016/j.lfs.2019.116697. [DOI] [PubMed] [Google Scholar]
- 22.Chen YH, Teng X, Hu ZJ, Tian DY, Jin S, Wu YM. Hydrogen sulfide attenuated sepsis-induced myocardial dysfunction through TLR4 pathway and endoplasmic reticulum stress. Front Physiol. 2021;12(653601) doi: 10.3389/fphys.2021.653601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Xu L, Zheng X, Wang Y, Fan Q, Zhang M, Li R, Ye J, Wu X, Zhao W, Zhang Y. Berberine protects acute liver failure in mice through inhibiting inflammation and mitochondria-dependent apoptosis. Eur J Pharmacol. 2018;819:161–168. doi: 10.1016/j.ejphar.2017.11.013. [DOI] [PubMed] [Google Scholar]
- 24.Bülbül M, Sinen O. Dual autonomic inhibitory action of central Apelin on gastric motor functions in rats. Auton Neurosci. 2018;212:17–22. doi: 10.1016/j.autneu.2018.03.005. [DOI] [PubMed] [Google Scholar]
- 25.Antonucci E, Fiaccadori E, Donadello K, Taccone FS, Franchi F, Scolletta S. Myocardial depression in sepsis: From pathogenesis to clinical manifestations and treatment. J Crit Care. 2014;29:500–511. doi: 10.1016/j.jcrc.2014.03.028. [DOI] [PubMed] [Google Scholar]
- 26.Fan TT, Cheng BL, Fang XM, Chen YC, Su F. Application of Chinese medicine in the management of critical conditions: A review on sepsis. Am J Chin Med. 2020;48:1315–1330. doi: 10.1142/S0192415X20500640. [DOI] [PubMed] [Google Scholar]
- 27.Li Y, Zhou J, Qiu J, Huang Z, Wang W, Wu P, Feng A. Berberine reduces gut-vascular barrier permeability via modulation of ApoM/S1P pathway in a model of polymicrobial sepsis. Life Sci. 2020;261(118460) doi: 10.1016/j.lfs.2020.118460. [DOI] [PubMed] [Google Scholar]
- 28.He Y, Yuan X, Zuo H, Sun Y, Feng A. Berberine exerts a protective effect on Gut-vascular barrier via the modulation of the Wnt/beta-catenin signaling pathway during sepsis. Cell Physiol Biochem. 2018;49:1342–1351. doi: 10.1159/000493412. [DOI] [PubMed] [Google Scholar]
- 29.Mou SQ, Zhou ZY, Feng H, Zhang N, Lin Z, Aiyasiding X, Li WJ, Ding W, Liao HH, Bian ZY, Tang QZ. Liquiritin attenuates lipopolysaccharides-induced cardiomyocyte injury via an AMP-Activated protein kinase-dependent signaling pathway. Front Pharmacol. 2021;12(648688) doi: 10.3389/fphar.2021.648688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Dai S, Ye B, Zhong L, Chen Y, Hong G, Zhao G, Su L, Lu Z. GSDMD Mediates LPS-induced septic myocardial dysfunction by regulating ROS-dependent NLRP3 inflammasome activation. Front Cell Dev Biol. 2021;9(779432) doi: 10.3389/fcell.2021.779432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Chen H, Liu Q, Liu X, Jin J. Berberine attenuates septic cardiomyopathy by inhibiting TLR4/NF-κB signalling in rats. Pharm Biol. 2021;59:121–128. doi: 10.1080/13880209.2021.1877736. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Liu Y, Yang W, Sun X, Xie L, Yang Y, Sang M, Jiao R. SS31 ameliorates sepsis-induced heart injury by inhibiting oxidative stress and inflammation. Inflammation. 2019;42:2170–2180. doi: 10.1007/s10753-019-01081-3. [DOI] [PubMed] [Google Scholar]
- 33.Giustina AD, Bonfante S, Zarbato GF, Danielski LG, Mathias K, de Oliveira AN Jr, Garbossa L, Cardoso T, Fileti ME, De Carli RJ, et al. Dimethyl fumarate modulates oxidative stress and inflammation in organs after sepsis in rats. Inflammation. 2018;41:315–327. doi: 10.1007/s10753-017-0689-z. [DOI] [PubMed] [Google Scholar]
- 34.Daiber A, Kröller-Schön S, Oelze M, Hahad O, Li H, Schulz R, Steven S, Münzel T. Oxidative stress and inflammation contribute to traffic noise-induced vascular and cerebral dysfunction via uncoupling of nitric oxide synthases. Redox Biol. 2020;34(101506) doi: 10.1016/j.redox.2020.101506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Ilari S, Dagostino C, Malafoglia V, Lauro F, Giancotti LA, Spila A, Proietti S, Ventrice D, Rizzo M, Gliozzi M, et al. protective effect of antioxidants in nitric Oxide/COX-2 interaction during inflammatory pain: The role of nitration. Antioxidants (Basel) 2020;9(1284) doi: 10.3390/antiox9121284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Dal-Secco D, DalBó S, Lautherbach NES, Gava FN, Celes MRN, Benedet PO, Souza AH, Akinaga J, Lima V, Silva KP, et al. Cardiac hyporesponsiveness in severe sepsis is associated with nitric oxide-dependent activation of G protein receptor kinase. Am J Physiol Heart Circ Physiol. 2017;313:H149–H163. doi: 10.1152/ajpheart.00052.2016. [DOI] [PubMed] [Google Scholar]
- 37.López A, Lorente JA, Steingrub J, Bakker J, McLuckie A, Willatts S, Brockway M, Anzueto A, Holzapfel L, Breen D, et al. Multiple-center, randomized, placebo-controlled, double-blind study of the nitric oxide synthase inhibitor 546C88: Effect on survival in patients with septic shock. Crit Care Med. 2004;32:21–30. doi: 10.1097/01.CCM.0000105581.01815.C6. [DOI] [PubMed] [Google Scholar]
- 38.Ichinose F, Buys ES, Neilan TG, Furutani EM, Morgan JG, Jassal DS, Graveline AR, Searles RJ, Lim CC, Kaneki M, et al. Cardiomyocyte-specific overexpression of nitric oxide synthase 3 prevents myocardial dysfunction in murine models of septic shock. Circ Res. 2007;100:130–139. doi: 10.1161/01.RES.0000253888.09574.7a. [DOI] [PubMed] [Google Scholar]
- 39.Wang Y, Huang Y, Lam KS, Li Y, Wong WT, Ye H, Lau CW, Vanhoutte PM, Xu A. Berberine prevents hyperglycemia-induced endothelial injury and enhances vasodilatation via adenosine monophosphate-activated protein kinase and endothelial nitric oxide synthase. Cardiovasc Res. 2009;82:484–492. doi: 10.1093/cvr/cvp078. [DOI] [PubMed] [Google Scholar]
- 40.Kim HJ, Yoo HY, Jang JH, Lin HY, Seo EY, Zhang YH, Kim SJ. Wall stretch and thromboxane A2 activate NO synthase (eNOS) in pulmonary arterial smooth muscle cells via H2O2 and Akt-dependent phosphorylation. Pflugers Arch. 2016;468:705–716. doi: 10.1007/s00424-015-1778-1. [DOI] [PubMed] [Google Scholar]
- 41.Chen K, Li G, Geng F, Zhang Z, Li J, Yang M, Dong L, Gao F. Berberine reduces ischemia/reperfusion-induced myocardial apoptosis via activating AMPK and PI3K-Akt signaling in diabetic rats. Apoptosis. 2014;19:946–957. doi: 10.1007/s10495-014-0977-0. [DOI] [PubMed] [Google Scholar]
- 42.Xiao M, Men LN, Xu MG, Wang GB, Lv HT, Liu C. Berberine protects endothelial progenitor cell from damage of TNF-α via the PI3K/AKT/eNOS signaling pathway. Eur J Pharmacol. 2014;743:11–16. doi: 10.1016/j.ejphar.2014.09.024. [DOI] [PubMed] [Google Scholar]
- 43.Aremu OO, Oyedeji AO, Oyedeji OO, Nkeh-Chungag BN, Rusike CRS. In vitro and in vivo antioxidant properties of taraxacum officinale in Nω-Nitro-l-Arginine Methyl ester (L-NAME)-induced hypertensive rats. Antioxidants (Basel) 2019;8(309) doi: 10.3390/antiox8080309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Bougaki M, Searles RJ, Kida K, Yu J, Buys ES, Ichinose F. Nos3 protects against systemic inflammation and myocardial dysfunction in murine polymicrobial sepsis. Shock. 2010;34:281–290. doi: 10.1097/SHK.0b013e3181cdc327. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.