Abstract
With the current worldwide pandemic of COVID-19, there is an urgent need to develop effective treatment and prevention methods against SARS-CoV-2 infection. We have previously reported that the proanthocyanidin (PAC) fraction in blueberry (BB) leaves has strong antiviral activity against hepatitis C virus (HCV) and human T-lymphocytic leukemia virus type 1 (HTLV-1). In this study, we used Kunisato 35 Gou (K35) derived from the rabbit eye blueberry (Vaccinium virgatum Aiton), which has a high PAC content in the leaves and stems. The mean of polymerization (mDP) of PAC in K35 was the highest of 7.88 in Fraction 8 (Fr8) from the stems and 12.28 of Fraction 7 (Fr7) in the leaves. The composition of BB-PAC in K35 is that most are B-type bonds with a small number of A-type bonds and cinchonain I as extension units. A strong antiviral effect was observed in Fr7, with a high polymerized PAC content in both the leaves and stems. Furthermore, when we examined the difference in the action of BB-PAC before and after SARS-CoV-2 infection, we found a stronger inhibitory effect in the pre-infection period. Moreover, BB-PAC Fr7 inhibited the activity of angiotensin II converting enzyme (ACE2), although no effect was observed in a neutralization test of pseudotyped SARS-CoV-2. The viral chymotrypsin-like cysteine protease (3CLpro) of SARS-CoV-2 was also inhibited by BB-PAC Fr7 in leaves and stems. These results indicate that BB-PAC has at least two different inhibitory effects, and that it is effective in suppressing SARS-CoV-2 infection regardless of the time of infection.
Keywords: SARS-CoV-2, Proanthocyanidins, Blueberry, Angiotensin II converting Enzyme, Viral chymotrypsin-like cysteine protease (3CLpro)
1. Introduction
The outbreak of coronavirus disease 2019 (COVID-19) is ongoing, with more than 230 million confirmed infections and 4.7 million coronavirus-related deaths. In Japan, on the other hand, the number of infected people is 1.7 million and the number of deaths is only 17,000; however, the number of COVID-19 patients is constantly increasing as a major domestic problem. Considered one of the most devastating global crises in recent years, infection with COVID-19 causes symptoms similar to those of acute respiratory distress syndrome [1]. Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), the causative virus of COVID-19, is a novel coronavirus strain [2]. To date, remdecivir has been licensed for emergency use as a therapeutic agent [3], and the efficacy of an urgently developed mRNA vaccine has been evaluated [4]. However, it will take some time to achieve herd immunity through vaccination, and although some new drugs for COVID-19 have been marketed, alternative measures are still needed to prevent infection.
We have reported that proanthocyanidins (PACs) contained in rabbit blueberry (BB) leaves have various functions, such as inhibiting hepatitis C virus replication [5], suppressing fatty liver development [6], inhibiting the progression of hepatocarcinogenesis [7], suppressing the development of adult T-cell leukemia [8], and inhibiting the proliferation of HTLV-1 virus-infected cells [9]. In recent years, we have developed Kunisato 35 gou (K35), which is suitable for use in tea leaves with high PAC content, and we are conducting functional analysis of PAC in K35 [10]. In this manuscript, we report a new function of the PAC fraction from K35 against a new type of coronavirus disease (COVID-19). The leaves and stems of K35 were extracted using hot water, and the extracts inhibited the RNA copy number of SARS-CoV-2 or the plaque-forming assay of SARS-CoV-2. After the extracts were fractionated using organic solvents, the fraction with high polymerization PAC content (BB-Fr7) was found to have a stronger suppressive effect against SARS-CoV-2. At the same time, BB-Fr7 with high PAC content showed potential as a prophylactic administration because it suppressed viral replication after infection by pretreatment. In addition, BB-Fr7 was found to inhibit angiotensin II converting enzyme (ACE2) and viral chymotrypsin-like cysteine protease (3CLpro), suggesting that it inhibits cell surface receptors necessary for infection and inhibits viral replication after infection. Therefore, administration of BB extracts containing highly polymerized PACs is expected to prevent new coronavirus infections.
2. Materials and methods
2.1. Fractionation of blueberry leaf and stem extraction from K35
The fractionation of blueberry leaf and stem extracts has been previously described [8]. Lyophilized blueberry leaves and stem powder (300 g) were extracted for 30 min with boiling water (15 l) and then cooled in an ice bath. After centrifugation (10 min at 1700×g), the supernatant was lyophilized and stored at −20 °C until use. The lyophilized powder (20.4 g) of blueberry leaf hot water extracts (BBL-HWE) was suspended in distilled water (1.2 l) and sonicated at 50 °C for 30 min. The insoluble material was then removed via filtration. The filtrate was directly subjected to Sephadex LH-20 column chromatography (3.5 cm i.d. × 23 cm) (Sigma Aldrich, St. Louis, MO, USA), eluted with water containing increasing proportions of methanol (100% water, 20%, 40%, 60%, 80%, and 100% methanol), 60% acetone, and finally eluted with 100% acetone. The eluate was collected in an Erlenmeyer flask (1000 ml in each) to yield ten fractions (BBFr0–BBFr9), concentrated with a vacuum evaporator, and completely freeze-dried. The lyophilized powder (100 g) of blueberry stem hot water extracts (BBS-HWE) was fractionated in the same manner as above.
To quantify PAC, the fractions were measured using the butanol/HCl method [5]. Each fraction was quantitated from 0.5 mg/ml (for Fr0–Fr4) or 0.2 mg/ml (for Fr5–Fr7) in methanol. Briefly, 400 μl of the solution was mixed with 3.5 ml of n-butanol/HCl (95:5, v/v) and 100 μl of 2% (w/v) NH4Fe(SO4)2·12H2O in 2 M HCl and heated in an oil bath at 105 °C for 30 min. After heating, the reaction mixture was cooled in water for 15 min and the absorbance was measured at 550 nm. PAC content was expressed in milligrams of cyanidin chloride equivalent/g dry weight.
The mean polymerization (mDP) and composition of PAC were estimated by thiolysis followed by reverse-phase high-performance liquid chromatography (HPLC). Fr4 to Fr8 (2.5 mg) were dissolved in 500 μl ethanol solution consisting of 5% (v/v) 2-mercaotoethanol, 4% (v/v) 0.5 M HCl, and 32% (v/v) H2O, and the mixture was heated at 70 °C for 7 h. The resulting thiolytic products were filtered and the filtrate was injected into the HPLC system. HPLC was carried out on a Q-Exactive mass spectrometry system (Thermo Fisher Scientific, Waltham, MA, USA) equipped with a UV–visible detector and an ODS Hypersil C18 column (4.6 mm·250 mm, 5 μm, Thermo Fisher Scientific). The separation conditions were as follows: flow rate, 0.8 ml/min; elution solvent, A (0.1% formic acid in water) and B (acetonitrile); and gradient program, 10%–28% B from 0 to 30 min, 28%–82% B from 30 to 40 min. UV detection was performed at 280 nm wavelength. The mean degree of polymerization (mDP) of PAC was calculated as follows: mDP = [sum of (2-hydroxyethylthio adducts · n) + sum of (free flavan-3-ol · n)]/[total free flavan-3-ol], where n represents the degree of polymerization of the detected flavan-3-ol by thiolysis. Average percentage of A-type bonds = [sum of (thiolysis compounds containing A-type bonds · n)/[total free flavan-3-ol]/mDP × 100. Average percentage of cinchonain I units = [sum of (thiolysis compounds containing cinchonain I unit · n)]/[total free flavan-3-ol]/mDP × 100.
2.2. Cells and virus preparation
VeroE6/TMPRSS2 cells were obtained from the Japanese Collection of Research Bioresources (JCRB) Cell Bank in Japan (https://cellbank.nibiohn.go.jp/english/) (JCRB no. JCRB1819) [12]. VeroE6/TMPRSS2 cells were cultured in Dulbecco's modified Eagle's medium (DMEM) containing 10% fetal bovine serum (FBS), penicillin (100 U/mL), streptomycin (100 μg/mL), and 1 mg/ml G418 (Gibco), and were maintained in a humidified atmosphere at 37 °C and 5% CO2. A strain of SARS-CoV-2 isolated from a patient who developed COVID-19 on the cruise ship Diamond Princess in Japan in February 2020 was obtained from the Kanagawa Prefectural Institute of Public Health (SARS-CoV-2/Hu/DP/Kng/19–027, LC528233) [13]. The virus was propagated in VeroE6/TMPRSS2 cells cultured in DMEM supplemented with 2% FBS. Viruses in the culture medium were diluted with DMEM and adjusted to 2.0 × 104 plaque-forming units (PFU)/ml.
2.3. Viral load quantitation of SARS-CoV-2
VeroE6/TMPRSS2 cells were seeded in a 96-well plate at a density of 1 × 105 cells/ml. A viral mixture containing DMSO (final conc. 0.2%), epigallocatechin (EGC, 50 μg/ml), or each fraction of BB-Leaf or BB-Stem (50 μg/ml) were inoculated into VeroE6/TMPRSS2 cells. After 2 h, the inoculum was removed, washed with PBS, and replaced with fresh culture medium without BB fraction. After incubation of 24 h, the copy number of viral RNA in the culture supernatant was quantified by real-time RT-PCR, as previously described [14], with some modifications. In brief, 5 μl of culture supernatant was mixed with 5 μl of 2 × RNA lysis buffer [2% Triton X-100, 50 mM KCl, 100 mM Tris-HCl (pH 7.4), 40% glycerol, 0.8 U/μl recombinant RNase inhibitor (Takara, cat# 2313B)] and incubated at room temperature for 10 min. The samples were diluted with RNase-free water (90 μl), and the diluted sample (2.5 μl) was used as the template for real-time RT-PCR performed according to the manufacturer's protocol using the One Step TB Green PrimeScript PLUS RT-PCR kit (Takara, cat# RR096A) and the following primers: 026N–F, 5′-AGCCTCTTCTCGTTCCTCATCAC-3'; and 026N-R, 5′-CCGCCATTGCCAGCCATTC-3'. Fluorescent signals were acquired using a QuantStudio 3 Real-Time PCR system (Thermo Fisher Scientific).
2.4. Cytotoxicity assay
To test biological activity, each BB fraction was dissolved in dimethyl sulfoxide (DMSO; Nacalai Tesque, Japan) at a concentration of 100 mg/ml (stock solution). Serial 4-fold dilutions of BB, ranging from 1000 μg/ml to 0.25 μg/ml, were prepared in DMEM containing 10% FBS, penicillin (100 units/mL), streptomycin (100 μg/mL), and 1 mg/ml G418 (Gibco). Diluted DMSO was used as the negative control. The cytotoxic effect of each extract was tested using the Cell Counting Kit-8 (CCK-8; Dojindo, Japan) assay following the manufacturer's instructions. The absorbance was measured at 450 nm using a microplate reader (Bio-Rad, Japan). The concentration of the BB fraction that produced a 50% reduction in cell viability (cytotoxic concentration, CC50) and the concentration that maintained >95% cell viability, the safe concentration, were calculated by regression analysis as described previously [15]. A solution below the calculated safe concentration of each BB was used in the antiviral assays.
(The remaining materials and methods are in the supplementary file.)
3. Results
3.1. Purified fractionation of hot water extracts from leaves and stems of blueberry Kunisato 35 gou (K35)
We previously reported the inhibitory effects of PACs from rabbit-eye blueberry leaves on HTLV-1-infected T lymphocytes [8], ATLL, and HCV virus [5]. Recently, we developed Kunisato 35 gou (K35) from rabbit eye blueberries (Vaccinium virgatum Aiton) for leaf tea production [10] and examined its effect on the SARS virus.
To investigate the chemical structure of PAC, hot water extraction (BB-HWE) was carried out according to a previously published method (reference), and the extracts were fractionated using eluting solvents according to standard methods (Tables S1 and S2). To investigate the chemical structure of PAC, the DMACA and butanol hydrochloric acid methods were used to determine the PAC content in each fraction (Tables S3 and S4). The mean polymerization (mDP) and composition of PAC were estimated by thiolysis followed by HPLC. The results showed that Fr4–Fr8 contained higher amounts of PAC. The mDP of PAC was the highest of 7.88 Fr8 from the stem and 12.28 Fr7 in the leaves, resulting in much higher degree of polymerization of the leaves than that of the stems. Regarding the structure of PAC, the content of cinchonain I as an extension unit and A-type bond was quite small in one unit of PAC, and most of them were B-type bonds (Table S5). Interestingly, the structure of PACs from the stem showed mostly B-type bonds, whereas PACs from leaves were characterized by slightly more A-type bonds and cinchonain I units. Based on these results, we next investigated the antiviral activity of Fr4 to Fr7, which had higher PAC contents in the same fractions from the leaves and stems.
3.2. Purified fractions containing highly polymerized PAC from blueberry hot water extracts significantly inhibit SARS-CoV-2 replication
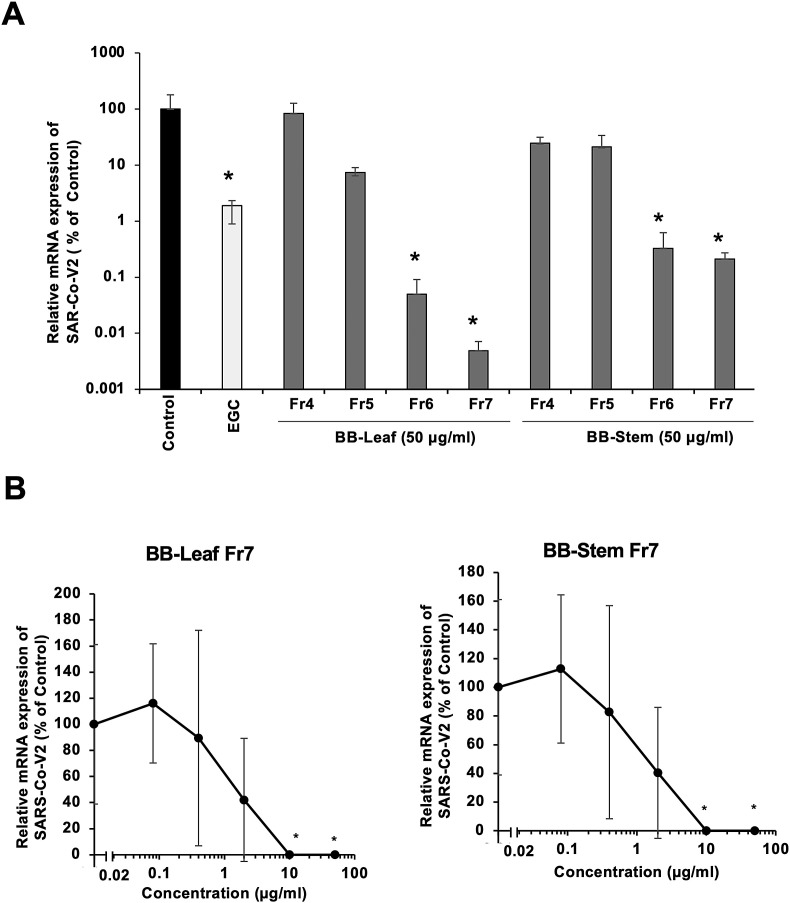
To investigate the inhibitory effect of HWE and Fr4–Fr7, the viral RNA expression rate in SARS-CoV-2 infected VeroE6/TMPRSS2 cells was determined. The results showed that Fr7 from BB-Stem or BB-Leaf, containing highly polymerized PAC, showed the strongest inhibition of viral RNA expression (Fig. 1 A). In particular, the inhibitory activity of BB-Leaf Fr7 was over 100 times higher than that of EGC as a control, and that of the stem component (Fr6 and Fr7) was more than 10 times higher. Therefore, the fraction with the highly polymerized PAC was found to have a stronger inhibitory effect against SARS-CoV-2.
Fig. 1.
Inhibition of viral mRNA copy number using PAC-rich fractions from blueberry extracts (K35). A. After SARS-CoV-2-infected VeroE6/TMPRSS2 cells were treated with fractions 4 to 7 with high PAC content (50 μg/ml each), mRNA expression of SARS-CoV-2 was determined, which is shown as % of control. 0.2% of DMSO solution used as a solvent for the BB fractions was used as a control. EGC; Epigallocatechin. ∗; <0.05. B. Determination of effective concentration (IC50) for inhibition of viral mRNA expression using hot water extracts or BB-Fr7of K35 leaves and stems.
Therefore, IC50 was determined by adding various concentrations of BB-Leaf Fr7 and BB-Stem Fr7 (Fig. 1B). The IC50 of BB-Leaf Fr7 was 1.52 μg/ml and 1.39 μg/ml for BB-Stem Fr7. Based on these results, subsequent experiments were conducted using BB-HWE and BB-Fr7 media.
3.3. Inhibitory effect of BB-Fr7 solution on viral replication of SARS-CoV-2 by plaque-forming assay
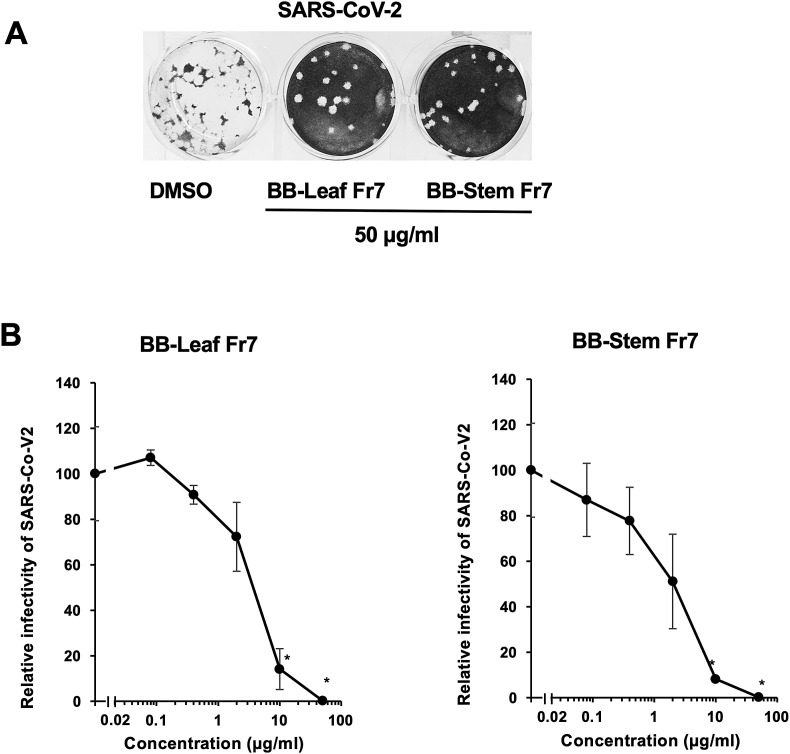
Because the addition of high concentrations of BB-Fr7 solution to VeroE6/TMPRSS2 cells caused cytotoxicity, we decided to use a BB-Fr7 solution of less than 50 μg/ml, which does not cause cytotoxicity in the experimental system (Fig. S1). To determine the suppressive activity of viral replication on SARS-CoV-2, we conducted a viral plaque assay at a concentration of 50 μg/ml of BB-Leaf or Stem Fr7. The results showed that the number of plaques was significantly reduced in BB-Leaf Fr7-and Stem Fr7 treated cells (Fig. 2 A). We also examined the concentration-dependent inhibition of plaque formation. The results showed that the IC50 values were 1.08 μg/ml for leaves and 1.92 μg/ml for stems (Fig. 2B). Therefore, BB-Fr7 was found to inhibit SARS-CoV-2 replication directly or indirectly.
Fig. 2.
Plaque formation assay in SARS-CoV-2-infected VeroE6/TMPRSS2 cells. A. Plaque formation assay in SARS-CoV-2-infected VeroE6/TMPRSS2 cells after the treatment of BB-Leaf or -Stem Fr7. BB-Fr7 (50 μg/ml) solution was diluted in multiples and cultured for 3 days after treatment with the diluted BB-Leaf or -Stem Fr7 solution against VeroE6/TMPRSS2 cells infected with SARS-CoV-2. The same experiment (N = 3) was repeated three times. B. The 50% inhibitory concentration (IC50) of BB-Leaf or -Stem Fr7 in the plaque formation assay described above was determined. ∗; <0.05.
3.4. Blueberry-derived highly polymerized PAC components can significantly inhibit viral replication by prophylactic administration
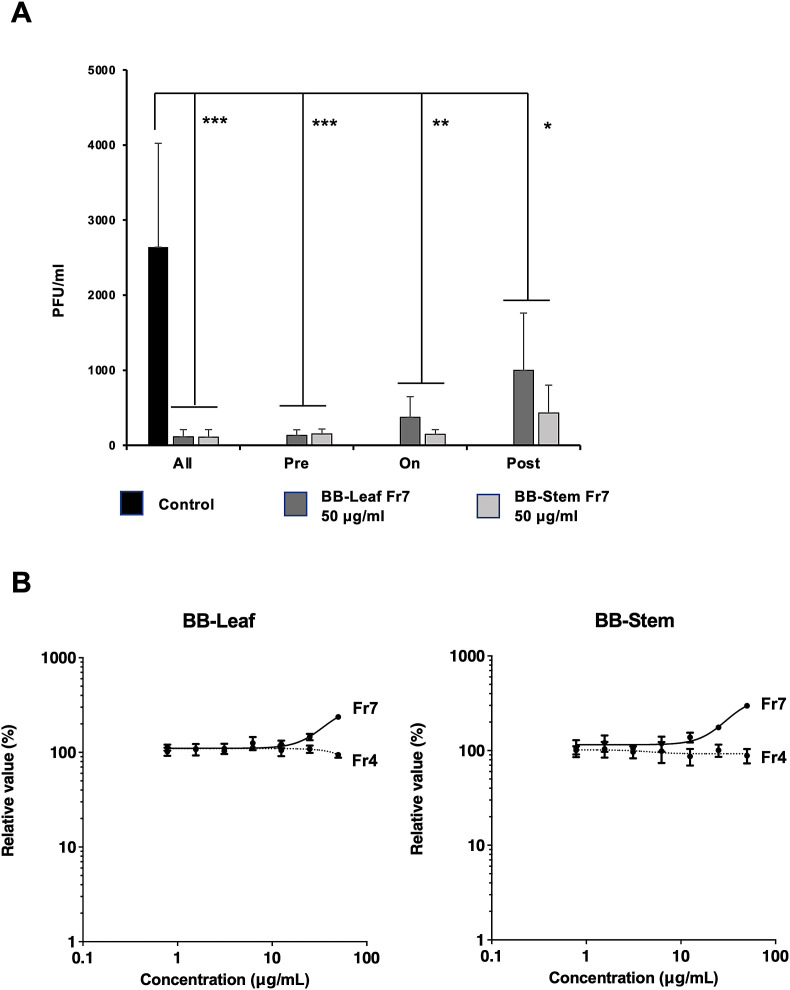
To determine the mechanism by which BB-Fr7 inhibits SARS-CoV-2 replication, we applied BB-Fr7 to virus-infected cells at different time points. As shown in Fig. S2, we applied BB-Fr7 as a pre-infection treatment (Pre), for 2 h during infection (On), and 72 h after infection (Post). The results showed that pre-infection treatment (Pre) was the strongest inhibitor of viral replication. Furthermore, a significant suppression of viral replication was observed during and after infection (Fig. 3 A), suggesting that highly polymerized PAC, the main component of BB-Fr7, has multiple inhibitory functions against SARS-CoV-2 infection before and after infection. In particular, pre-infection treatment showed stronger suppression of SARS-CoV-2 replication, indicating that prophylactic administration of BB-Fr7 was the most efficient method of administration.
Fig. 3.
An experiment to examine the ability of BB-Fr7 to inhibit virus replication at different times of treatment. A. As in Fig. S2, the effect of BB-Fr7 treatment on VeroE6/TMPRSS2 cells was investigated using three different treatment methods: before (Pre), during (On), and after (Post) the virus infection period as indicated in Materials and Methods. As a control, we included those not treated with BB-Fr7 (No) and those treated with BB-Fr7 (All) at all experimental times. The same experiment (N = 3) was repeated three times. ∗; p < 0.05, ∗∗; p < 0.005, ∗∗∗; p < 0.001. B. To examine the inhibition of infection by BB-PAC, infection of HeLa-ACE2-TMPRSS2 cells with SARS pseudovirus was measured by luciferase activity. The left figure shows the results for Fr4 (dotted line) and Fr7 (solid line) in blueberry leaves, and the right figure shows the results for the fractions of Fr4 and Fr7 from blueberry stems. Luciferase activity is shown relative to the condition without BB-PAC fraction as 100%.
To understand the mode of inhibitory activity against infection, we tested whether BB-PAC could directly target SARS-CoV-2 virus particles. To this end, we mixed BB-Fr7 with the SARS-CoV-2 spike (S) protein-pseudotyped lentiviral luciferase reporter virus for 1 h and inoculated them into HeLa-ACE2-TMPRSS2 cells. All BB-PAC Fr7 from leaves, stems, and Fr4 did not suppress pseudovirus infection, even at higher concentrations (100 μg/ml) (Fig. 3B). Thus, these results suggest that BB-PAC does not directly target the viral spike protein in defense against infection.
3.5. Blueberry-derived highly polymerized PAC components protects against SARS-CoV-2 infection by inhibiting ACE2 and viral protease
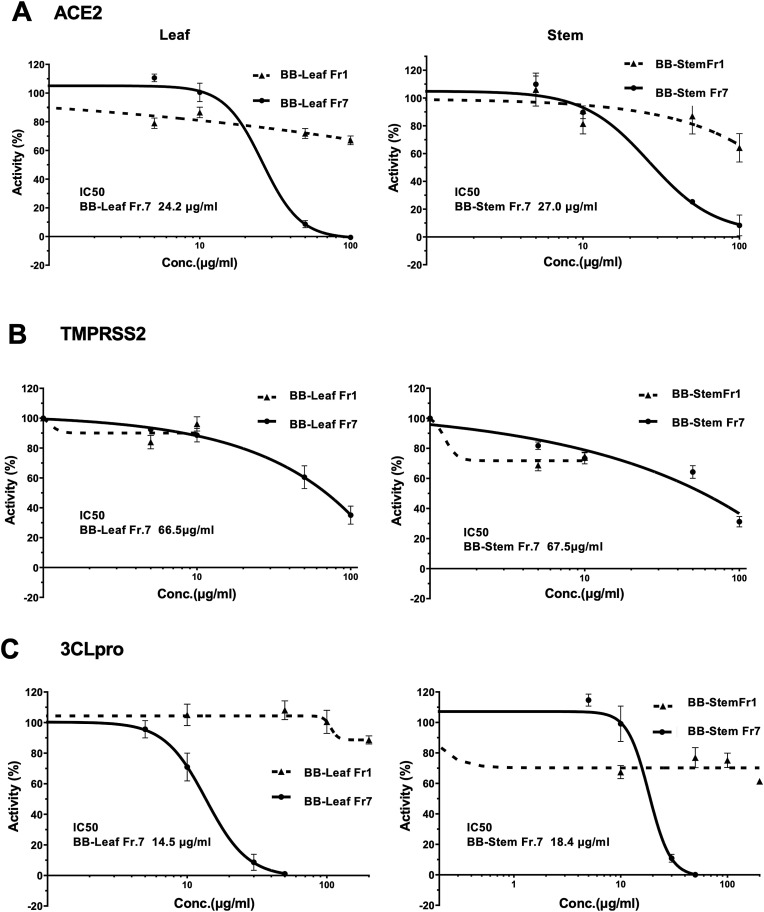
Next, we examined the inhibitory effects of the BB-PAC fraction on the angiotensin-converting enzyme 2 (ACE2) receptor and transmembrane protease, serine 2 (TMPRSS2), which are essential for SARS-CoV-2 infection on the plasma membrane. The enzymatic activities of ACE2 and TMPRSS2 were examined using BB-PAC Fr7 from the leaves or stems and Fr1 as a control Fr1 had no inhibitory effect on ACE2, while Fr7 had an inhibitory effect with 24.2 μg/ml as IC50 for leaves and 27.0 μg/ml for stems, respectively (Fig. 4 A). On the other hand, Fr1 showed no inhibition for TMPRSS2, while Fr7 showed 66.5 μg/ml for leaves and 67.5 μg/ml for stems as IC50 (Fig. 4B). The inhibitory activity of TMPRSS2 was observed at higher concentrations, resulting in weaker inhibitory activity of TMPRSS2 compared to the IC50 of ACE2.
Fig. 4.
Inhibitory activity of ACE2, BSS, and virus-derived 3CL protease enzymes by BB-PAC. A. ACE2 enzyme activity by BB-PAC fractions of Fr1(dotted line) as a control and Fr7 (solid line) at several different concentrations was measured by the ACE2 activity assay kit (BPS Bioscience). The right figure shows the results using the blueberry leaf fraction and the left figure shows the results using the blueberry stem fraction. B. TMPRSS2 enzyme activity by BB-PAC fractions of Fr1(dotted line) as a control and Fr7 (solid line) at several different concentrations was measured by the TMPRSS2 activity assay kit (BPS Bioscience). C. 3CLpro enzyme activity by BB-PAC fractions of Fr1(dotted line) as a control and Fr7 (solid line) at several different concentrations was measured by the 3CLpro activity assay kit (BPS Bioscience).
In addition, the effect of BB-PAC on the protease activity of SARS-CoV-2 was examined, as viral replication was suppressed by BB-PAC even after infection. In addition, since several natural compounds have been investigated using computational methods to inhibit proteases, proanthocyanidins have been identified as promising drug targets for the main protease (Mpro)/chymotrypsin-like cysteine protease (3CLpro) [16]. Therefore, we investigated the enzyme inhibition ability of our purified BB-PAC fractions (Fr1 as a control and Fr7) using a 3PLpro assay kit (Fig. 4C). Fr1, the control from leaves and stems, showed no inhibitory effect, while Fr7 showed an inhibitory effect at both, 14.5 μg/ml for leaves and 18.4 μg/ml for stems. These results indicate that BB-PAC inhibits viral replication by blocking the ACE2 receptor as a defense against viral infection and inhibiting the main viral protease in the cell.
4. Discussion
In this study, we report that extracts containing high levels of highly polymerized PACs from blueberry leaves or stems are effective in protecting against SARS-CoV-2 infection. We created Kunisato 35 gou (K35) from rabbit eye blueberries that contained a large amount of highly polymerized PAC in their leaves and stems for use in these experiments. The PAC structure of rabbit-eye blueberry leaves (Vaccinium virgatum Aiton) has been reported that the structure of polymerized PAC is mainly B-type bonds but there is also type A bonds and cinchonain I units [11]. In this study, we analyzed the structure of PAC from K35, and the degree of polymerization in leaves and stems was 5–12, and the structure also contained mostly B-type bonds with type A bonds and cinchonain 1 units. Moreover, PAC from leaves or stems was also found to have a strong inhibitory effect on SARS-CoV-2 replication and was found to be maximally effective when treated prior to infection, indicating that it should be used prophylactically.
As a result of our investigations using BB-PAC, we have found a wide range of antiviral effects, including those against HTLV-1 and HCV, as well as antioxidant effects, anti-inflammatory effects, and anti-obesity effects due to its effects on fat metabolism [[5], [6], [7], [8], [9], [10]]. In the present study, we showed that BB-PAC inhibits the ACE2 enzyme as a receptor for the SARS virus [17], suggesting that BB-PAC may function in defense against infection by blocking viral entry into the cell. In addition, the BB-PAC fraction inhibited 3CL protease, which encodes an essential 3CL protease (3CLpro or Mpro) that processes polyproteins, making it an attractive target for viral inhibitors [18]. Therefore, BB-PAC shows inhibition with respect to these two major therapeutic targets, ACE2 receptor on the cell membrane and viral 3CL protease and has the potential to act as an effective inhibitor of SARS-associated coronavirus infection. Therefore, BB-PAC can be considered a safe and regular protective agent against the spread of COVID-19 as a food ingredient [19]. Of course, further validation is needed, but future development should be aimed at using it not only in COVID-19 but also in general coronavirus infections, including the common cold.
Declaration of conflicts of interest
The authors declare no conflicts of interest.
Acknowledgments
This work was funded in part by the Takeda Science Foundation (KM).
Footnotes
Supplementary data to this article can be found online at https://doi.org/10.1016/j.bbrc.2022.04.072.
Appendix A. Supplementary data
The following is the Supplementary data to this article:
References
- 1.Carson Welker C., Jeffrey Huang J., Nunez Gil I.J., Ramakrishna H. 2021 acute respiratory distress syndrome update, with Coronavirus Disease 2019. Focus J. Cardiothoracic Vascular Anesthesia. 2021:1–8. doi: 10.1053/j.jvca.2021.02.053. 00. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Hu Ben, Guo Hua, Zhou Peng, Shi Zheng-Li. Characteristics of SARS-CoV-2 and COVID-1. Nat. Rev. Microbiol. 2021;19:141–154. doi: 10.1038/s41579-020-00459-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Eastman T.R., Roth Jacob S., Brimacombe Kyle R., Simeonov Anton, Shen Min, Patnaik Samarjit, Matthew D. Hall remdesivir: a review of its discovery and development leading to emergency use authorization for treatment of COVID-19. ACS Cent. Sci. 2020;6:672–683. doi: 10.1021/acscentsci.0c00489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Garcia-Montero Ci, Fraile-Martinez O., Bravo C., Torres-Carranza D., Sanchez-Trujillo L., Gomez-Lahoz A.M., Guijarro L.G., Garcia-Honduvilla N., Asnsolo A., Bujan J., Monserrat J., Serrano E., MAlvarez-Mon J.A., De Leon-Luis, Alvarez-Mon M., Ortega M.A. An updated review of SARS-CoV-2 vaccines and the importance of effective vaccination programs in pandemic times. Vaccines. 2021;9:433–465. doi: 10.3390/vaccines9050433. 9, 433-465 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Takeshita M., Ishida Y., Akamatsu E., Ohmori Y., Sudoh M., Uto H., Tsubouchi H., Kataoka H. Proanthocyanidin from blueberry leaves suppresses expression of subgenomic hepatitis C virus RNA∗. J. Biol. Chem. 2009;284:21165–21176. doi: 10.1074/jbc.M109.004945. https://pubmed.ncbi.nlm.nih.gov/19531480/ [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Fujii K., Ota Yuma, Nishiyama K., Kunitake H., Yamasaki Y., Tari H., Araki K., Arakawa T., Yamasaki M. Blueberry leaf polyphenols prevent body fat accumulation in mice fed high-fat. High-Sucrose Diet J. Oleo Sci. 2019;68:471–479. doi: 10.5650/jos.ess18226. https://www.jstage.jst.go.jp/article/jos/68/5/68_ess18226/_article [DOI] [PubMed] [Google Scholar]
- 7.Matsuura Y., Sakakibara H., Kawaguchi M., Murayama E., Yokoyama D., Yukizaki C., Kunitake H., Sakono M. Effects of blueberry leaf and stem extracts on hepatic lipid levels in rats consuming a high sucrose diet. Functional Foods Health Dis. 2018;8:447–461. https://ffhdj.com/index.php/ffhd/article/view/538 [Google Scholar]
- 8.Nagahama K., Eto N., Sakakibar Y., Matsusita Y., Sugamoto K., Morishita K. M Suiko Oligomeric proanthocyanidins from rabbiteye blueberry leaves inhibits the proliferation of human T-cell lymphotropic virus type 1-associated cell lines via apoptosis and cell cycle arrest. J of Func. Foods. 2014;6:356–366. https://www.sciencedirect.com/science/article/pii/S1756464613002557 [Google Scholar]
- 9.Kai H., Fuse T., Kunitake H., Morishita H.,K., Matsuno K K.,K. Medicines; Basel: 2014. Comparison of Cultivars and Seasonal Variation in Blueberry (Vaccinium Species) Leaf Extract on Adult T_Cell Leukemia Cell Line Growth Suppression; pp. 3–11.https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5532976/ 1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Tetsumura T., Kawjiwara Y., Honsho C., Sato-Yamaguchi M., Sugamoto K., Kunitake H. Effective micropropagation of rabbiteye blueberries for leaf tea production environmental control in biology. Environ. Contr. Biol. 2012;50:289–296. https://www.jstage.jst.go.jp/article/ecb/50/3/50_289/_article/-char/ja [Google Scholar]
- 11.Matsuo Y., Fujita Y., Ohnishi S., Tanaka T., Hirabaru H., Kai T., Sakaida H., Nishizono S., Kouno I. Chemical constituents of leaves or rabbiteye blueberry (Vaccinium ashei) and characterization of proanthocyanidins containing phenylpropanoid units and A-type linkages. Food Chem. 2010;121(2020):1073–1079. doi: 10.1016/j.foodchem.2020.02.052. [DOI] [Google Scholar]
- 12.Matsuyama S., Nao N., Shirato K., Kawase M., Saito S., Takayama I., Nagata N., Sekizuka T., Katoh H., Kato F., Sakata M., Tahara M., Kutsuna S., Ohmagari N., Kuroda M., Suzuki T., Kageyama T., Takeda M. Enhanced isolation of SARS-CoV-2 by TMPRSS2-expressing cells. Proc. Natl. Acad. Sci. U. S. A. 2020;117:7001–7003. doi: 10.1073/pnas.2002589117. https://www.pnas.org/content/117/13/7001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Mallapaty S. What the cruise-ship outbreaks reveal about COVID-19. Nature. 2020;580 doi: 10.1038/d41586-020-00885-w. https://www.nature.com/articles/d41586-020-00885-w 18. [DOI] [PubMed] [Google Scholar]
- 14.Mugisha C.S., R Vuong H., Puray-Chavez M., L Bailey A., M Fox J., E Chen R., W Wessel A., M Scott J., H Harastani H., Boon A.C.M., Shin H., B Kutluay S. A simplified quantitative real-time pcr assay for monitoring SARS-CoV-2 growth in cell culture. mSphere. 2020 Sep 2;5(5) doi: 10.1128/mSphere.00658-20. https://pubmed.ncbi.nlm.nih.gov/32878932/ [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Kwon Y.-C., Jewett M.C. High-throughput preparation methods of crude extract for robust cell-free protein synthesis. Sci. Rep. 2015;5:8663. doi: 10.1038/srep08663. https://www.nature.com/articles/srep08663 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.MF AlAjmi, A Azhar, S Hasan, AZ Alshabr, A Hussain, M T Rehman Identification of natural compounds (proanthocyanidin and rhapontin) as high-affinity inhibitor of SARS-CoV-2 Mpro and PLpro using computational strategies. Arch. Med. Sci. Preprint DOI: 10.5114/aoms/133706 https://doi.org/10.1152/ajpendo.00298.2020. [DOI] [PMC free article] [PubMed]
- 17.Hoffmann M., Kleine-Weber H., Schroeder S., Krüger N., Herrler T., Erichsen S., Schiergens T.S., Herrler G., Wu N.H., Nitsche A., Müller M.A., Drosten C., Pöhlmann S. SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell. 2020;181:271–280. doi: 10.1016/j.cell.2020.02.052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Jin Z., Du X., Xu Y., Deng Y., Liu M., Zhao Y., Zhang B., Zhang Li X., Peng L., Duan C., Yu Y., Wang J., Yang L., Liu K., Jiang F., Yang R., You X., Liu T., Yang X., Bai X., Liu F., Liu H., Guddat X., Xu L.W., Xiao W., Qin G., Shi C., Jiang Z., Rao Z H., Yang H. Structure of M(pro) from SARS-CoV-2 and discovery of its inhibitors. Nature. 2020;582:289–293. doi: 10.1038/s41586-020-2223-y. [DOI] [PubMed] [Google Scholar]
- 19.Levy E., Delvin E., Marcil V. S Spahis Can phytotherapy with polyphenols serve as a powerful approach for the prevention and therapy tool of novel coronavirus disease 2019 (COVID-19)? Am. J. Physiol. Endocrinol. Metab. 2020;319:E689–E708. doi: 10.1152/ajpendo.00298.2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.