Abstract
The vasa vasorum are a vital microvascular network supporting the outer wall of larger blood vessels. Although these dynamic microvessels have been studied for centuries, the importance and impact of their functions in vascular health and disease are not yet fully realized. There is now rich knowledge regarding what local progenitor cell populations comprise and cohabitate with the vasa vasorum and how they might contribute to physiological and pathological changes in the network or its expansion via angiogenesis or vasculogenesis. Evidence of whether vasa vasorum remodeling incites or governs disease progression or is a consequence of cardiovascular pathologies remains limited. Recent advances in vasa vasorum imaging for understanding cardiovascular disease severity and pathophysiology open the door for theranostic opportunities. Approaches that strive to control angiogenesis and vasculogenesis potentiate mitigation of vasa vasorum–mediated contributions to cardiovascular diseases and emerging diseases involving the microcirculation.
Advances over the centuries highlight the importance of vasa vasorum in cardiovascular disease.
INTRODUCTION
“All kinds of vessels” was written in English by Willis (1) to describe the outermost layer of larger blood vessels when he authored Pharmaceutice rationalis in 1678. Willis also noted that microvessels penetrated deeper layers of the wall. This description by Willis is thought to be the first documented observation of what the Dutch botanist and anatomist Frederic Ruysch of the Netherlands later called “vasa arteriosa” when he made an important illustration in 1696 that showed what we now know to be vasa vasorum (2). Christian Ludwig was the first to use the term “vasa vasorum” in 1739, and this became the adopted terminology despite incidences of misnomers and grammatical errors over past centuries. For an excellent historical review of the terminological and grammatical use and misuse of terms describing these fascinating microvessels, readers are encouraged to visit the insightful review of Musil et al. (3). The organization and structure of these nourishing microvessels within larger parent vessels reflect other patterns in nature as in tree branches, river tributaries, and orchid petal pigmentation.
Investigation of the vasa vasorum spans nearly three- and one-half centuries and progressing over a series of fits and starts since the mid-1920s. An overt curiosity and a current research focus on the vasa vasorum in the author’s laboratory motivated this review of the literature to bring forth several yet unanswered questions. These include the following: (i) Are changes in vasa vasorum density, structure, and function a cause or consequence of cardiovascular pathologies? (ii) What cells contribute to vasa vasorum expansion? (iii) What imaging modalities accurately monitor vasa vasorum integrity and function? (iv) What properties of the local tissue niche support vasa vasorum function? and (v) What therapeutic opportunities do the vasa vasorum offer?
Foundational studies in human and animal tissues laid important observational groundwork that informed learners of the basic vasa vasorum structure and function (4–8). These landmark studies paved the way for hypothesis-generating data regarding the role of the vasa vasorum in vessel health. This review summarizes pivotal studies that begin to unravel causative and consequential changes of vasa vasorum within different cardiovascular pathologies. Furthermore, this article turns the spotlight on recent advances expected to inspire creative approaches to answering the important yet unanswered questions about the role of vasa vasorum in healthy and diseased blood vessels.
Anatomical origins of vasa vasorum
Blood vessels are composed of three distinct layers: the intima (inner, abluminal layer consisting of endothelial cells), the media (middle, smooth muscle, and elastic layer), and the adventitia (outer, fibrous layer). The vasa vasorum are a network of microvessels within larger arteries and veins with importance to the overall health of the larger “host” vessel. There exist arterial vasa vasorum to deliver oxygen and nutrients to the parent vessel and venous and lymphatic vasa vasorum, which remove waste and extracellular fluid to adjacent veins and lymphatic vessels, respectively.
Two types of vasa vasorum (e.g., externa and interna) are described in the literature, characterized on the basis of their anatomical origin. The vasa vasorum externa reside mainly within the adventitia and originate from myriad anatomical locations. Figure 1 depicts the originating vessels of the vasa vasorum as well as their presence and relative densities for exemplar parent vessels. Vasa vasorum externa in the ascending aorta arise from the brachiocephalic and coronary arteries (8, 9). The intercostal arteries give rise to the vasa vasorum externa of the descending thoracic aorta, and the mesenteric and lumbar arteries are continuous with the vasa vasorum of the abdominal aorta (9, 10). Vasa vasorum penetrate the medial layer in larger arteries (e.g., thoracic aorta, carotid, femoral, and coronary arteries) (9). Other studies of porcine vessels describe vasa vasorum originating from the parent vessel’s lumen as in normal coronary arteries (11) and are thus designated as vasa vasorum interna. The earliest reports describe an avascular zone of 0.5 mm (12) and then, later, as dependent not on the thickness but rather the number (>29) of elastic lamellae in the vessel wall at birth (7). Approximately 50 years later, another report quantified vasa vasorum density among five equal transverse segments of the porcine aorta and demonstrated differing densities (13). Detection and quantification of vasa vasorum density in hematoxylin and eosin–stained full thickness sections of porcine (13) and human (14) aorta recently provided new evidence that normal vasa vasorum may exist closer to the intima of human thoracic aorta than previously thought. Thus, vasa vasorum remodeling under normal and pathological settings may be occur via multiple mechanisms and are likely influenced by hemodynamics, vessel architecture, and developmental origin of local cells.
Fig. 1. Density and origins of vasa vasorum in different vessels.
In the ascending thoracic aorta, vasa vasorum originate from the brachiocephalic and coronary arteries. Intercostal arteries feed the vasa vasorum of the descending thoracic aorta, which are as similarly dense as the ascending thoracic aorta. Vasa vasorum are least dense in the infrarenal abdominal aorta than in the thoracic aorta, do not penetrate the medial layer as in the thoracic aorta, and stem from the lumber and mesenteric arteries. Only the basilar and vertebral intracranial arteries contain vasa vasorum.
Density of vasa vasorum also varies by anatomic location within different vascular beds. Examining aortic autopsy specimens of patients without a history of aortic disease, Sano et al. (15) confirmed that arterial vasa vasorum density varied across different regions of human aorta, with maximum density in the aortic arch decreasing distally to reach a minimum in the infrarenal abdominal aorta (Fig. 1). The authors also noted denser lymphatic vasa vasorum within the infrarenal abdominal aorta and identified a positive correlation between the density of lymphatic vasa vasorum and intimal thickness in agreement with observations in human carotid and iliac arteries and aorta. However, the density of adventitial vasa vasorum did not correlate with intimal thickness in the infrarenal abdominal aorta. Thus, vasa vasorum density may differentially influence aortic diseases in a region- and etiology-specific manner by influencing inflammatory status. Moreover, vasa vasorum penetrate the medial layer in human thoracic aorta but not the abdominal aorta (7). In autopsy-collected vessels, coronary arteries were found to have a higher density of vasa vasorum when compared to femoral and renal arteries (16). Study of porcine vessels revealed higher density in the coronary arteries, followed by carotid, femoral, and renal arteries, with random intravessel distribution in all vessels examined (17).
Vasa vasorum exist in the extracranial vessels but are rare among intracranial vessels, with most animal studies describing a lack of intracranial vasa vasorum. However, a summary of data from human autopsy specimens reveals differing observations, particularly with age and cause of death (18). Vasa vasorum are not present within human intracranial arteries at birth but rather seem to develop during adulthood (19) and increase with age (18). It is thought that the absence of vasa vasorum within intracranial arteries is due to structural differences from extracranial arteries such as thinner medial and adventitial layers, a thicker and fenestrated internal elastic lamina, and lack of external elastic lamina, together allowing for greater diffusion of blood. In addition to blood from the lumen, the intracranial vessel wall is nourished with oxygen and nutrients via cerebrospinal fluid (20). The anatomy of the parent vessel dictates the need for vasa vasorum, and, likewise, vasa vasorum prevalence and organization govern vessel health and homeostasis.
Organization and physiology of vasa vasorum
The 1960s was the first decade to see an exponential increase in the number of publications on vasa vasorum. Clarke (4, 9, 21–38) published 20 articles alone between 1964 and 1966, describing the distribution of vasa vasorum in several human blood vessels including the location of medial vasa vasorum. The aortic vasa vasorum was observed at the border between the middle and inner third of the aortic media and reported as completely mature medial vasa vasorum by age 13 in the human (9).
Vasa vasorum are endarteries (10), meaning that they are vessels that do not anastomose (i.e., connect) with neighboring arteries and are solely responsible for oxygenating the local tissue. A range of lumen diameters have been reported for various normal arteries and veins of animal models and human specimens. Microbeads less than 50 μm in diameter were suitable for image-based measurement of vasa vasorum flow in the canine aorta and pulmonary artery (39). Vasa vasorum lumen diameters in human great saphenous vein ranged from 11 to ~36 μm in human and animal specimens (40). A range of sizes was previously described for vasa vasorum within specimens of human femoral, abdominal aorta, iliac, and renal arteries (41) and human carotid artery (42). A study by the author’s laboratory reported vasa vasorum density calculated separately for different ranges of lumen diameters as the mean of all vessels measured for a given specimen: small (<50 μm), mid-size (50 to 100 μm), and large (>100 μm) (43). Looking anew at the raw dataset, the 462 vessels of adventitial vasa vasorum measured exhibited a broad spectrum of lumen diameters (2 to 392 μm) among the cohort of 23 individual patient specimens of normal human ascending aorta. The mean ± SD for all patient specimens examined was found to be 40.0 ± 15.5 μm with approximately three quarters of all vessels measuring <50 μm (72.2 ± 17.5%). Vessel size and vessel orientation are both deliberately important for adequate nourishment of the parent vessel.
The vasa vasorum network within parent arteries and veins follows a similar hierarchy as artery-arteriole-capillary where vessels oriented along the longitudinal axis of the host vessel are known as “first-order” and are similar in size to arterioles (Fig. 2). “Second-order” vessels in capillary size range branch from first-order vessels and extend circumferentially around the host vessel. First- and second-order vasa vasorum contain smooth muscle cell (SMC) layer(s), while smaller second-order vessels (<25 μm) vasa vasorum exhibit pericyte coverage with α–smooth muscle expression in subsets (44). In hearts of hypercholesterolemic pigs, the ratio of the density for first-order to second-order coronary artery vasa vasorum was increased (45). Balloon-injured porcine coronary arteries also exhibited an overall reduction in vasa vasorum density despite a shift to more second-order than first-order vessels and formation of new first- and second-order vessels, both with smaller diameters than vessels in noninjured arteries (46). Thus, the ratio of first-order to second-order vasa vasorum seems highly important for proper health of the host vessel, and identifying mechanisms of microvascular remodeling and factors inciting dysfunction should yield important insights in parent vessel pathophysiology.
Fig. 2. Organization and hierarchy of arterial vasa vasorum.
Image of arterial vasa vasorum of the human ascending thoracic aorta (procured during aortic replacement surgery with approval of the University of Pittsburgh Institutional Review Board (protocol no. STUDY20040179). First-order vasa vasorum are oriented longitudinally along the ascending aorta (arrowheads). Second-order vasa vasorum branch from first-order vessels and extend circumferentially around the vessel (white arrows). The proximal end of the vessel is at the bottom. Image was captured using a Nikon SMZ25 Stereoscope with a SHR Plan Apo 1× objective. Images were captured using NIS Elements software (version 4.60.00 and 3× digital zoom). Scale bar, 500 μm.
In the late 1970s, the microsphere method emerged to measure blood flow through the vasa vasorum. The first report of flow measurements through aortic vasa vasorum is thought to be by Heistad et al. (47). In porcine models, studies applying micro–computed tomography (CT) imaging methods revealed the branching patterns of vasa vasorum and determined the diameter gradient in microsphere-injected vessels. Similar methods proved that vasa vasorum are endarteries rather than a plexus because occlusion of proximal arteries led to decreased perfusion of vasa vasorum and widespread areas of diminished vasa vasorum density.
Vasa vasorum are also able to regulate their own tone (48), as they are vasoresponsive to physiologic and neural stimuli (49). Adenosine-treated vasa vasorum exhibited increased blood flow to the outer but not the inner media (47). Porcine vasa vasorum exhibited concentration-dependent constriction in response to endothelin-1 (ET-1) and, unlike other arteries, were found to be insensitive to other known vasoconstrictors. Porcine vessels serve as useful models; however, what differences may exist among species and how can this information be extrapolated for translational directions?
Cross species similarities and differences
Wolinsky and Glagov (7) were the first to identify a diversity among species based on the presence of medial vasa vasorum in the aorta. Studying 12 different mammalian species varying in size from mouse to horse, they determined that the presence or absence of a vascularized zone within the media was dependent on the number of medial lamellar units at birth, not the total wall thickness at maturity. Species with vessels composed of less than 29 layers of medial elastic lamellar units at birth (e.g., rats and rabbits) exhibited an entirely avascular media, whereas species with 29 or greater lamellar units showed medial vasa vasorum. Across all species studied, the avascular zone was found to be uniformly 29 lamellar units, irrespective of age. Total wall thickness, including that of the avascular zone, increased with age and body size. More recently, evidence of cells expressing endothelial markers (i.e., CD31/platelet, endothelial cell adhesion molecule 1, and endothelial nitric oxide synthase) was found, on average, less than 150 μm from the intima of normal vessels (13, 14), further substantiating an emerging theory that aortic vasa vasorum may extend deeper into the media than were previously understood, which may have additional implications for gaining a new understanding of interlayer cell–matrix relationships and of pathological remodeling and dysfunction.
The adventitia and cellular composition of vasa vasorum
The adventitial layer has long been recognized for its biomechanical importance to overall aortic integrity. The adventitia represents the final barrier to rupture during the highly lethal aortic dissection. Free rupture occurs when the stress of dissection tear propagation exceeds the biomechanical strength of the adventitia. We now know more about other biological influences, including paracrine signaling and the importance of the vasa vasorum as the adventitia’s resident microvasculature in both healthy vessels and in vascular and cardiovascular pathologies. Evidence emerging over the past decade has also shed light on multiple cell subpopulations with demonstrated plasticity including several cell types associated with the vasa vasorum.
The cellular origins of vasa vasorum during development are not entirely understood. Moreover, postnatal vasculogenesis for normal vascular health or pathological microvascular remodeling may involve ancestral cells with endothelial lineage potential or cells with endothelial plasticity. Recent advances in the classification and functional characterization of adventitial cells provide new insight as to which cells may contribute to vasa vasorum development, expansion, and regeneration.
The adventitia plays home to numerous cell types including immune cells, dendritic cells, and fibroblasts. This microenvironment also serves as a repository of progenitor cells, many of which have shown vasculogenic potential including endothelial plasticity. In recent years, evidence came forth that a perivascular source of mesenchymal stromal cells resides within the small blood vessels of adipose (50) and other vascularized tissues (51–54). Several groups purport that multipotent progenitor cells also reside within the tunica adventitia of larger blood vessels including human pulmonary artery (55), vena saphena (56), adult thoracic aorta (57, 58), and fetal aorta (59). Using human white adipose tissue and fetal muscle, lung, and bone marrow as a paradigm, Corselli and colleagues (60) demonstrated that the adventitia houses a repository of progenitor cells from which SMCs, pericytes, and endothelial cells originate in a so-called “vasculogenesis zone” intermediate to the adventitial and medial layers of larger blood vessels.
Other groups have also centered on this region near the adventitia-media border as a progenitor cell niche (61–64). A domain of sonic hedgehog–expressing cells was shown to support a population of Sca1+ progenitor cells at the border between the adventitia and media in the mouse aortic root where the extracellular matrix (ECM) is particularly rich (62). Sca1+ adventitial cells were detected between E15.5 (embryonic day 15.5) and E18.5 in the mouse embryo, and the niche was maintained in the adult mouse. Sca1+ cells isolated from the adventitia express transcription factors governing the SMC lineage and can be differentiated toward mature SMCs and contribute to intimal hyperplasia in atherosclerosis (65). Work from Toledo-Flores and colleagues (66) discerned populations of murine adventitial cells based on their expression of Sca1 and CD45 and deemed Sca1+/CD45+ subsets as vasculogenic with endothelial plasticity, thus implicating an involvement of these cells in vasa vasorum expansion in the setting of atherosclerosis, separate from their role as macrophage progenitors. Other populations in mice coexpressing the markers Gli, Sca1, and CD34 contributed to intimal lesions in atherosclerotic vessels of apolipoprotein E (ApoE)−/− mice (65), could differentiate toward the SMC lineage, and localize to areas of vascular calcification (67). An exciting report from the Psaltis group (63) expanded on their discovery of Sca1+/CD45+ cells in mouse arteries that give rise to macrophages. Sca1+/CD45+ cells lost CD45 expression and acquired endothelial cell markers during ex vivo culture. Their data demonstrate a contribution (albeit of unknown relative abundance) of Sca1+/CD45+ cells to neovessels and adventitial vasa vasorum in atherosclerotic carotid arteries, indicating that these adventitial cells exhibit potential for endothelial lineage progression and/or endothelial plasticity and contribute to postnatal vasculogenesis of vasa vasorum. A CD45− cultured subset of Sca1+ cells exhibited a similar proclivity toward the endothelial lineage as the CD45+ subset. Cultured CD45−/CD146+/CD31− cells isolated from human adventitia and shown to reside in an in situ perivascular location associated with vasa vasorum exhibited similar behavior as CD45+ and CD45− subsets of Sca1+ cells from mouse adventitia. These cells demonstrated formation of branching structures from colonies on Matrigel culture substrates, although human vasa vasorum–associated pericytes did so at an accelerated rate [6 hours (44) versus 5 days (66)]. Embryologic origins of Sca1+ cells are not known although are unlikely to be derived from bone marrow because they lack hematopoietic lineage potential (63) and lineage-tracing experiments failed to demonstrate a contribution of the bone marrow compartment to Sca1+ cells (68). Nearly all (99%) SMC-derived Sca1+ cells were found to lack CD45 expression. These data support the theory proposed by Wolinsky and Glagov (7) in 1967 that additional lamellar units are derived from adventitial cells as the organism matures. A fate mapping study revealed that differentiated medial SMCs can also dedifferentiate to become adventitial Sca1+ progenitor cells capable of giving rise to SMCs, endothelial cells, and macrophages and contribute prominently to adventitial remodeling during vascular injury (69). One could also speculate that Sca1+ cells are analogous to other perivascular cells in human arteries with vasculogenic potential (44, 51). Future studies could be designed to uncover a human cell counterpart to Sca1 that remains yet unidentified, thus precluding its use to reliably label and track the fate of human progenitor cells.
Larger blood vessels containing vasa vasorum are themselves vascularized tissues. Motivated by a curiosity as to whether the vasa vasorum itself also serves as a repository of progenitor cells, work by the author’s team characterized vasa vasorum–associated cells from human aorta (44). Using a combination of analytical flow cytometry and immunolabeling approaches, nonhematopoietic cells (CD45−/CD34+/CD31+) were identified in situ and, in freshly isolated, noncultured, shown to localize to the vasa vasorum intima and may constitute tissue-resident endothelial colony-forming cells (ECFCs), with a surface marker profile consistent with cells described in other reports with the moniker endothelial progenitor cells (EPCs) (70–72). Cells previously termed circulating EPCs have been shown to include two phenotypically and functionally distinct populations with one “early EPC” population showing transcriptomic and proteomic similarities to monocytes and the second “outgrowth endothelial cells” exhibiting high resemblance to endothelial cells (73). Outgrowth endothelial cells are also known as myeloid endothelial cells. While some studies report incorporation of circulating EPC-like cells into the vasculature in vivo, others show evidence to the contrary (74, 75). However, despite ambiguity in marker profile characterization and a lack of consistency in isolation methodology within various tissues and species amidst heterogeneity and hierarchy within vessels (76), tissue-resident ECFCs and circulating myeloid angiogenic cells have been shown to adopt marker expression and functional features of cells of endothelial lineages in vitro and were identified within preexisting vessels (77). ECFCs also participate in physiological and pathophysiological neovessel formation in vivo, demonstrated using robust methodology (e.g., parasymbiosis models, bone marrow transplant, and clonal analysis) (70–72, 78, 79), thus elevating their candidacy as direct contributors of arterial vasa vasorum development and expansion.
Lymphatic endothelial cells express unique markers distinct from blood vessel endothelial cells such as lymphatic endothelial cell receptor 1 (LYVE-1) (80) and podoplanin (81). LYVE-1 functions as a gatekeeper of leukocyte trafficking and immune activation as a receptor for hyaluronan transport into the lumen of lymphatic vessels. LYVE-1 binds to hyaluronan in the glycocalyx of dendritic cells (82), thus permitting their entry and migration to lymph nodes. LYVE-1’s ability to bind growth factors and associate with growth factor receptors to engage downstream kinase activity potentiates myriad physiological and pathophysiological roles (83). Podoplanin plays a key role in lymphatic function by preventing blood from entering lymphatic vessels and facilitating migration of dendritic cells to lymph nodes (84). Both LYVE-1 and podoplanin have been reported to distinguish lymphatic vasa vasorum from arterial vasa vasorum by selectively labeling lymphatic endothelial cells, whereas von Willebrand factor expression defined arterial vasa vasorum (15). While the markers LYVE-1 and podoplanin distinguish endothelial cells in lymphatic vessels from arterial and venous vasa vasorum, it remains unknown whether endothelial cells of vasa vasorum are transcriptionally distinct from intimal endothelial cells of larger arteries and veins. Whether vasa vasorum among multiple parent vessels arise from the same ancestral cells as blood vessel endothelial cells is also unknown and relatively unexplored. The advent of single-cell RNA sequencing and advances in fluorescent in situ hybridization multiplexing approaches in addition to genetic fat mapping strategies are likely to cast a spotlight on similarities and putative phenotype distinctions among endothelial cells and other local cell populations. Nonendothelial cells such as bone marrow–derived cells and macrophages have also been shown to express the lymphatic endothelial cell marker LYVE-1. Notably, these LYVE-1+ cells are distributed as single cells within tissues and are distinct from cells comprising lymphatic vessels (85) that coexpress other blood vessel endothelial-specific surface markers such as CD31. LYVE-1+ macrophages located with the adventitia can help to maintain arterial homeostasis through regulation of collagen expression via direct interaction of LYVE-1 with hyaluronan receptors on SMCs (86). Of additional interest would be pericytes associated with vasa vasorum and other capillaries that are intimately associated with endothelial cells and play key roles in microvascular angiogenesis and function. Figure 3 displays several discussed examples of the various cell subpopulations in the adventitia, associated with the vasa vasorum or potentially involved in vasa vasorum postnatal angiogenesis. Table 1 further summarizes the phenotype and proposed role of perivascular progenitor cells in physiological and/or pathophysiological postnatal neovascularization discussed in this article.
Fig. 3. Schematic of large elastic artery.
Image depicts a large artery (i.e., aorta) with basal lamina and intimal layer (endothelium), medial smooth muscle layer with elastic fibers, and the outer, highly heterogeneous adventitial layer. Arterial and venous vasa vasorum are shown in the adventitial and medial layers. In addition, lymphatic vasa vasorum and nervi vasorum (nerves) are shown. Numerous cells with postnatal vasculogenic potential are denoted in the adventitia. Note that components are not drawn to scale.
Table 1. Examples of potential vasculogenic cells contributing to postnatal angiogenesis of vasa vasorum.
| Cell type | Markers and subsets | Location | Function | References |
| Adventitia-laden progenitor- like cells |
CD34+/CD146−/CD45−/ CD31−/CD90+/VEGR2+/TIE2+ |
Adventitia/media border; perivascular to adventitial vasa vasorum |
Macrophage precursors and formation of endothelial sprouts; mural cells, endothelium support; multipotent, precursors of pericyte-like cells |
Zengin et al. (64), Corselli et al. (60), Billaud et al. (44) |
| Pericytes | CD146±/CD90±/CD34±/ CD31− Other subsets: NG2+/ PDGFRβ+/αSMA+/ CD105+/CD73+ |
Abluminal, under the basement membrane; around capillaries and vasa vasorum |
Stabilization of new vessels; sprouting behavior in vitro |
Crisan et al. (51), Corselli et al.
(60), Billaud et al. (44) |
| Tissue-resident ECFCs | CD45−/CD31+/CD146−/CD34+ | Endothelium; liver, intestine; vasa vasorum intima |
Differentiate into endothelial cells; form neovessels in ischemic and nonischemic tissues |
Ingram et al. (76), Aicher et al.
(78), Naito et al. (77), Billaud et al. (44) |
| Circulating bone marrow–derived myeloid angiogenic cells |
CD45+/CD31+/CD34+/Flk+ | Peripheral blood, bone marrow |
Comprise neovessels in murine ischemic hindlimb model; increased capillary density in mouse heart infarct model; formation of subcutaneous neovessels |
Asahara et al. (70), Kawamoto (71), Yoder et al. (72), Medina et al. (73) |
| Bone marrow–derived cells | c-kit+ | Peripheral blood, bone marrow |
Neovessels in atherosclerotic vessels and pulmonary hypertension |
Davie et al. (182), Sata et al.
(123), Montani et al. (195) |
| Sca1+ | CD45+/Gli1+/CD34+ Other subsets: CD45−; CD31+/CD105+; CD144−/CD31− |
Adventitia/media border; adventitia |
Vasa vasorum expansion in atherosclerosis; smooth muscle cell progenitors; macrophage progenitors, endothelial precursors |
Toledo-Flores et al. (66), Kramann et al. (67), Passman et al. (62), Psaltis et al. (63), Majesky et al. (69) |
A variety of adventitial cells with potentially overlapping marker profiles, similar if not identical phenotypes, and compensatory functions and potency for multilineage progression have been identified and characterized in varying breadth and depth and using several different ex vivo, in vitro, and in vivo approaches. The relationships among cell populations known to be involved in postnatal angiogenesis and putatively in vasa vasorum expansion are depicted in Fig. 4. Characterization of these many perivascular cell populations in humans has been a daunting challenge to traverse from in vivo native location to ex vivo propagation (87). The work of Traktuev and colleagues (50) was among the first reports of adipose stromal cells that shared surface markers with pericytes and mesenchymal stem cells. These cells were shown to express CD34 and, despite their perivascular location adjacent to CD31+ cells, are distinct from CD34+/CD31+/CD144+ endothelial cells as well as the pericytes described by Crisan and colleagues (51) as CD146+/CD34− within adult adipose, skeletal muscle, and pancreas and fetal muscle. However, it remains undetermined whether adventitial cells that exhibit a progenitor cell–like marker profile also demonstrate differential multipotency and adopt progenitor cell–like function upon ex vivo culture and whether vasa vasorum–associated cells exhibit different features of progenitor cells in both normal and diseased large blood vessels.
Fig. 4. Lineage hierarchy of vascluogenic cells participating in postnatal angiogenesis.
Probable cellular origins of postnatal vasa vasorum angiogenesis in normal and pathological states include differentiation of adventitia-laden progenitor cells toward a pericyte phenotype, and subsequent migration toward PDGF-BB producing branching endothelial tip cells and/or differentiation toward SMCs. Tissue-resident ECFCs give rise to smooth muscle and mature endothelial cells. Pericytes can adopt a more mature SMC phenotype. Bone marrow–derived cells are precursors of myeloid angiogenic cells, macrophages, SMCs, pericytes, and endothelial cells. Endothelial cells can transdifferentiate toward smooth muscle and pericytes through endothelial-to-mesenchymal transition (EndoMT). Tissue-resident Sca1+ cells that are non–bone marrow–derived may originate from medial SMCs and give rise to mature SMCs, endothelial cells, and macrophages.
It is also nonmutually exclusive that multiple cell types residing within this important vascular layer have vasculogenic potential and can directly and/or indirectly support formation of neovessels and angiogenesis during expansion of existing vasa vasorum. For example, it was recently shown that pericyte-specific deletion of Ninjurin1 led to formation of immature vasa vasorum in the adventitia of wire-injured mouse vessels, increased inflammatory cells, and exacerbated intimal hyperplasia (88). Pericytes are among the most important cells for vasa vasorum function and expansion, in part, due to their known direct interactions with endothelial cells and platelet-derived growth factor receptor β (PDGFRβ)–dependent functions for proper vasculogenesis and angiogenesis (89, 90). While tissue-resident and circulating progenitor cells have been shown to directly contribute to neovessel formation in vivo (72), human pericytes delivered in vivo also directly home to host microvasculature and improved function in the mouse (54) and porcine infarcted hearts (91), their mechanism(s) of action remains unknown. Pericyte delivery also improved regeneration of injured or diseased skeletal muscle, likely as producers of proregenerative cytokines (92). Notably, bone marrow–derived cells were shown to give rise to perivascular cells of fibroblast growth factor 2 (FGF2)–induced neovessels in the mouse cornea (93). Nearly all macrophages and more than half of the pericytes within neovessels of subcutaneous FGF2-impregnated hydrogel implants in mice were of bone marrow origin versus just 10% of endothelial cells (94). Similarly, human adipose-derived stromal cells injected into the mesentery arteries of adult nude rats were identified in a perivascular location of microvessels and expressed the pericyte markers α–smooth muscle actin and neuron glial antigen factor 2 (95). These studies demonstrate a prominent contribution of bone marrow–derived cells to perivascular cells during postnatal angiogenesis. Populations of pericytes (CD146+/CD34±/CD31−) associate with the adventitial vasa vasorum (44); however, a direct contribution of pericytes (or their precursors) to (patho)physiological neovascularization of vasa vasorum has not been examined. Studies that use labeling and/or genetic fate mapping strategies represent a priority for further rigorous studies to determine the cellular origins of vasa vasorum.
The ability of mesenchymal stromal–like cells to directly participate in vasculogenesis remains controversial, where it is more commonly accepted that mesenchymal stromal cell secreted factors stimulate paracrine angiogenic signaling and mesenchymal stromal cells directly interact with endothelial cells to participate in angiogenesis instead of undergoing differentiation toward the endothelial lineage. Results of fate mapping studies in mice failed to show an appreciable contribution of cells in the pericyte/smooth muscle lineage to adipogenic and myogenic lineages during aging or induced disease states (96), thus arguing against a previous theory of in vivo plasticity of perivascular cells. More recent lineage tracing studies support a role for pericytes in postnatal vascular remodeling by revealing that neuron-glial antigen 2 (NG2)–expressing cells accumulated around lung arterioles in a mouse model of hypoxia-induced pulmonary hypertension and pericytes isolated from lungs of patient with pulmonary hypertension exhibited an increased propensity to differentiate toward SMCs (97). Ex vivo cultured vasa vasorum–associated pericytes from human aorta could be differentiated toward mature SMCs in vitro as shown by their responsiveness to cytokine-induced up-regulation of contractile SMC markers α–smooth muscle actin, smooth muscle myosin heavy chain, and calponin. Another interesting cell population within human aortic adventitia was classified as nonhematopoietic (CD45−), nonpericyte (CD146−), and nonendothelial (CD31−) cells described as CD90+/CD34+ cells (44). These cells were deemed “supra-vasa” because they were noted to reside in a perivascular location around vasa vasorum within the adventitia. CD90+ cells, in particular, have also been described as a rare population of adventitial cells coexpressing mesenchymal stromal cell markers (61). Ex vivo cultured CD90+ cells isolated from human atherosclerotic and/or aneurysmal aortas secreted less proangiogenic factors when compared with cells isolated from health aortas (five to six subjects per group) (61). These CD90+/CD34+ supra-vasa cells express a similar surface proteome to nonhematopoietic progenitor cells in the adventitia and adipose-derived cells from the stromal vascular fraction described by others (60, 61, 98, 99). Pericytes and other local cells in the perivascular niche of vasa vasorum represent candidate populations for additional lineage specification and potential for transdifferentiation, lineage progression during vasculogenesis, and angiogenesis.
Endothelial cells can also give rise to pericytes and SMCs through a process of endothelial-to-mesenchymal transition, as was demonstrated using lineage tracing in the mouse embryonic heart (100), adding further complexity to the potential relationships among vasculogenic cells potentially contributing to vasa vasorum expansion. An important identified gap in knowledge is whether tissue-resident or circulating precursor endothelial cells and local pericytes (or their precursors) directly participate in physiological or pathological expansion of vasa vasorum. It will be interesting to find whether and how these many adventitial cells directly and/or indirectly participate in vasa vasorum postnatal angiogenic processes within larger normal and diseased vessels.
Despite several lines of evidence regarding cell hierarchy and contributions to angiogenesis in physiological and pathological models and specimens, questions about cellular origins of vasa vasorum development and postnatal remodeling remain. Since the vasa vasorum can be considered analogous to small arteries in other vascularized tissues, what cells in the adventitia contribute to vascularization of the adventitia and expansion of the vasa vasorum into the media? An important area of new investigation will be in uncovering what factors incite microvascular remodeling and pruning and may influence or exhibit antiangiogenic signaling in different pathological states. Moreover, because of the responsiveness of adventitial cells to paracrine factors produced by medial cells and evidence of their cellular origins in the media, these perivascular cells likely sense and respond accordingly to signals from the media and intimal layers through interlayer cross-talk. The vasa vasorum nourishes, reflects overall health, and stands poised to adapt to the needs of the parent vessel.
Involvement of vasa vasorum in cardiovascular diseases
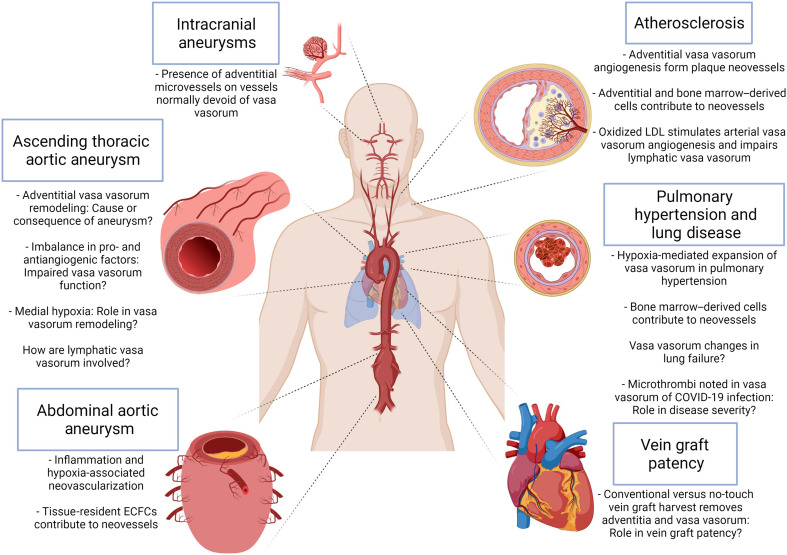
For decades, the vasa vasorum have been implicated, associated, and targeted in the setting of various cardiovascular diseases. The location of vasa vasorum within larger vessels and the vital function of these vessels demand a better understanding of how they contribute to and/or are influenced by cardiovascular pathologies. Nearly all of what we know regarding vasa vasorum in cardiovascular disease pertains to the arterial vasa vasorum with relatively little understood about the role of venous vasa vasorum and lymphatic vasa vasorum. Delineating whether vasa vasorum alterations are a cause and/or a consequence of disease pathophysiology in humans is not only difficult to test experimentally but also challenging to interpret. Undoubtedly, the pursuit to answer such questions is worthwhile.
Atherosclerosis
Involvement of vasa vasorum in atherosclerotic disease is irrefutable. Several lines of evidence point to microvascular dysfunction not only as an initiator but also as an exacerbating mechanism of progressive atherosclerosis. Numerous lines of inquiry point to a role for vasa vasorum in the initiation, progression, and susceptibility to atherosclerotic disease. Notably, the anatomic specificity of larger vessel decoration with vasa vasorum may influence that vessel’s propensity for atherosclerotic lesion development. The seminal work of Koester (5) in 1876 showed that occlusion of vasa vasorum led to medial necrosis and speculated this to be an inciting event of atherosclerotic lesion development. Nearly 200 years later, Nakashima and Tashiro (101) explained early proatherosclerotic lipid deposition in the outer coronary artery as independent of intimal endothelium damage and due instead to neovascularization of vasa vasorum within the outer layers of a thickened intima, where biglycan, a proteoglycan with selective binding for lipoproteins, is present in normal and diseased arteries. During atherogenesis, an increase in transport of low-density lipoprotein (LDL), oxidized products, and inflammatory cytokines across the endothelium that precedes cellular infiltration has been described, purportedly because of endothelial dysfunction and a decreased capacity for clearance by the venous vasa vasorum (102). Furthermore, vasa vasorum dysfunction may contribute to atherogenesis via hypoperfusion-induced low oxygen tension, which could further impair effective LDL transport. Moreover, oxidized LDL can contribute to neovascularization of vasa vasorum in atherosclerosis through epigenetic modifications in endothelial cells (103).
Neovascularization from adventitial vasa vasorum occurs after expansion of the diffuse intimal thickening and precedes formation of histopathologic features of atherosclerosis, giving rise to an alternative hypothesis for the mechanism causing coronary atherosclerosis in the setting of normal LDLs (104). Other work in a porcine model of hypercholesterolemia showed evidence of vasa vasorum neovascularization before onset of endothelial dysfunction in coronary arteries (102). Observations that stenotic and calcified human coronary arteries exhibited higher vasa vasorum density when compared with nonstenotic vessels (105) and extend from the adventitia, branching into the intima (106), demonstrate that neovascularization of arterial vasa vasorum occurs in atherosclerosis-prone vessels.
Adventitial lymphangiogenesis has also been linked to atherosclerosis. Although the role of lymphangiogenesis in atherogenesis remains unclear, disruption of lymph flow in the mouse aorta increased adventitial inflammation and plaque development and limited plaque regression in ezetimibe-treated mice (107). Lymphatic vessels expand in atherosclerotic vessels; however, angiogenesis seems to play more of a role in neovascularization within atherosclerotic plaques than lymphangiogenesis. In a study of 23 autopsy cases, LYVE-1–positive lymphatic vessels were rarely detected in atherosclerotic intima of human carotid arteries (108), and increased levels of oxidized LDL noted in the adventitia near lymphatic vessels are thought to inhibit lymphangiogenesis. Lymphatic vessels mediate the removal of oxidized LDL, thus reducing atherosclerotic lesion formation. Human lymphatic-derived endothelial cells exposed to oxidized LDL exhibit reduced angiogenic activity in vitro and neovessel formation in vivo via CD36 receptor–mediated signaling (109). Whether this signaling axis may be exploited for therapeutic benefit in atherosclerotic patients would be an advantageous line of inquiry. Understanding how vasa vasorum function affects atherosclerosis development and progression, and therapeutic effectiveness is of the utmost importance.
In atherosclerotic monkeys, blood flow through the vasa vasorum of the coronary arteries (more prominently in the outer layer) was increased, presumably because of neovessel growth rather than dilation of existing vessels and atherosclerotic vessels maintained adenosine-induced dilation (110). ET-1 is up-regulated in atherosclerosis and, therefore, may cause increased vasa vasorum constriction, limiting blood flow and nutrient delivery (111, 112). Furthermore, occlusion of rabbit vasa vasorum was found to be associated with increased activity of cholesterol acyltransferase (113), suggesting that the resulting hypoperfusion leads to tissue hypoxia. Adventitial delivery of FGF2 in ApoE−/− mice caused vasa vasorum expansion and aggravated plaque development (114). Endothelial-to-mesenchymal transition has been linked to loss of FGF receptor 1 expression in endothelial cells and accelerated atherosclerotic disease in ApoE-deficient mice and was associated with disease severity in human coronary arteries (115). When vasa vasorum neovascularization extends to the intima, the plaque may experience increased nourishment through delivery of hormones, oxygen, and other nutrients. However, there also appears to be a relationship between the vasa vasorum and plaque instability. Plaque microvessels were denser in ruptured versus nonruptured plaques and less numerous in fibrocalcific plaques versus lipid-rich plaques (116). Since inflammation was associated with neovessel density where severe inflammation was associated with the highest density of plaque microvessels, the vasa vasorum may, therefore, serve as a dynamic conduit for influx of macrophages and other proinflammatory factors that contribute to increased production of matrix metalloproteinases and further aggravate atherosclerotic lesions. It is well accepted that inflammatory cell infiltration in cardiovascular disease occurs via the vasa vasorum, although a comparison of leukocyte adhesion molecules between vasa vasorum–associated endothelial cells within normal and atherosclerotic or aneurysmal human vessels or with neighboring cell types is lacking and relationships are not well understood, representing an additional important area to be explored. A single study of human abdominal aorta and iliac artery revealed detection of endothelial leukocyte adhesion molecule 1 with increasing severity of atherosclerosis (117). More vasa vasorum–associated endothelial cell functions have been gleaned from animal model investigations. For example, a porcine coronary artery balloon angioplasty model revealed up-regulation of P-selectin, E-selectin, and vascular cell adhesion molecule 1 in vasa vasorum–associated endothelial cells to facilitate infiltration of inflammatory cells when compared with noninjured vessels absent of inflammatory cells (118). Consistent with the known involvement of macrophages in hypoxia-mediated angiogenesis, positron emission tomographic imaging of 18F-floromisonidazole–treated rabbits revealed colocalization with areas of tissue hypoxia and macrophages near intraplaque neovessels of advanced atherosclerosis (119). Similar noninvasive imaging studies in humans identified comparable overlapping signals for hypoxia and macrophages in vascularized human carotid plaques (120) to further support inflammatory cell involvement in hypoxia-mediated angiogenesis.
The presence of vasa vasorum is also associated with atherosclerotic intracranial arteries. A study examining 157 autopsy specimens found adventitial vasa vasorum in approximately half of all samples and in normal and atherosclerotic middle cerebral, vertebral, and basilar arteries, with the majority of vasa vasorum observed in the vertebral artery (121). Vasa vasorum in the vertebral arteries were found to be associated with preatherosclerotic lesions and progressive atherosclerosis and more numerous in vessels with a heavier plaque load increased density of intraplaque calcification (121) and in more severe atherosclerosis (122). Thus far, there have been no fate mapping studies that delineate origins of vasa vasorum expansion in atherosclerotic intracranial arteries, but they likely arise from local vasculogenic cells.
Vasa vasorum expansion in atherosclerosis appears to arise by vasculogenesis, whereby circulating and local tissue progenitor cells such as those residing in the adventitia give rise to endothelial cells. Bone marrow–derived cells have exhibited vasculogenic potential to give rise to smooth muscle actin–positive cells within plaques of hyperlipidemia-induced atherosclerotic mice (123). Conversely, it has also been shown that bone marrow–derived cells rarely participate in plaque endothelium in the ApoE-deficient mouse model of atherosclerosis (124), consistent with prior studies of neovascularization (94). Within the previous 2 years, Sca1+/CD45+ cells were identified within the murine aortic adventitia that coexpress CD31+ in atherosclerotic aorta of ApoE−/− mice and frequently colocalized with von Willebrand factor and vasa vasorum (66). Moreover, ex vivo cultured Sca1+ cells contributed to de novo perfused vasa vasorum in the carotid artery of atherosclerotic mice and neovessels with increased perfusion in an ischemic hindlimb (66). Additional growth factors may be secreted by infiltrating inflammatory cells from the vasculature, other resident adventitial cells, and from periadventitial fat to influence vasa vasorum expansion and altered flow in atherosclerotic vessels in a paracrine fashion. In hypoglycemic ApoE−/− mice, the expected neovascularization of vasa vasorum is absent in the larger atherosclerotic plaques that form relative to normoglycemic ApoE−/− mice (125). In contrast to such observations in diabetic animal models, patients with diabetes mellitus exhibit increased microvascular density in atherosclerotic plaques when compared to similar plaques in nondiabetic patients (126). The noted discrepancy from observations in the mouse model may be due to unknown ApoE genotype, which was not determined for human subjects examined.
Developing strategies to inhibit plaque angiogenesis as a treatment for atherosclerosis has been a longstanding focal area of research. In ApoE−/− mice, subcutaneous injection of the antiangiogenic factors endostatin or TPN-470 (a fumagillin analog) or angiostatin led to reduced lesion formation, thus raising these factors as potential therapeutic candidates to prevent atherogenesis (127, 128). However, despite this initial enthusiasm, there are currently no clinical trials focusing on endostatin or TPN-470 in atherosclerosis (clinicaltrials.gov). More recently, local blockade of vascular endothelial growth factor receptor 2 (VEGFR2) in hypercholesterolemic mice resulted in less intraplaque hemorrhage and a decrease in the number of extravasated red blood cells when compared with sham-operated controls (129). Thus, prevention of neovessel instability may prove therapeutic by markedly reducing the impact of vasa vasorum expansion. A small-molecule inhibitor of FGF2 represents another candidate for plaque stabilization due to its ability to reduce macrophage infiltration and intraplaque angiogenesis (130). A gene transfer strategy to deliver decoy VEGFRs to the adventitia of rabbit vessels implanted with bare metal stents prevented vasa vasorum neovascularization in stent restenosis (131). However, when intravenous bevacizumab was administered to pigs after intercostal artery ligation with the premise of combating hypoxia-induced angiogenesis, deleterious outcomes were noted (132). An inflammatory response persisted and was accompanied by other complications such as altered hemodynamics, blood dyscrasia, and severe gastric lesions. Future efficacious treatments will likely require local, controlled delivery of agents that appropriately prevent overgrowth of vasa vasorum.
While some interpret the effects of vasa vasorum removal, occlusion, or, otherwise, hypoperfused microvessels as proatherosclerotic events, others view these data of consequent vascular wall necrosis and disruption as indicative of aneurysm or dissection. One useful example is the recent work of Colman et al. (133), who, while studying the role of deleted in breast cancer gene in angiotensin II (Ang-II)–infused hypertensive mice, made the somewhat unexpected observation of aortic delamination associated with extravasated red blood cells from adventitial vasa vasorum and intramural hematoma (IMH) formation. Current experimental models have not enabled sufficiently rigorous experimentation to determine whether vasa vasorum dysfunction is an aggravator as opposed to a consequence of lesion development in atherosclerotic disease. Addressing this important pathophysiological question will require more study and novel sophisticated animal models.
Vein graft patency
Vasa vasorum in veins also lie within the adventitia and penetrate the media in close proximity to the lumen (134). The presence and integrity of the vasa vasorum are thought to be important for vein graft patency (135, 136). Saphenous vein graft harvest via a “no-touch” technique presumably protects the adventitia, preserves vasa vasorum endothelial function, and is considered to be superior to the conventional technique, which is less invasive and therefore imposes reduced donor site morbidity, but is more damaging to the harvested vessel itself in that it strips away the adventitia, thus removing most of the vasa vasorum (134). The no-touch technique includes a pedicle of stromal tissue, adventitia, vasa vasorum, and perivascular adipose tissue. This local tissue niche in close proximity to the vasa vasorum is a source of contractile and relaxant factors with potential to influence vasa vasorum function and contribute to vein graft patency (137). A recent multicenter randomized controlled trial comparing outcomes in no-touch versus conventional saphenous vein graft harvest revealed no differences between harvest techniques in total graft occlusion, stenosis, or cardiovascular death, although inclusion in a meta-analysis uncovered a reduction in graft occlusion associated with no-touch harvest (138). In one study, comparing saphenous vein graft explants harvested either by conventional technique or no-touch technique revealed a higher density of CD34+ immunoreactive cells in vessels harvested with a no-touch technique despite similar density and size of CD34+ vessels in both the adventitia and media when compared with conventionally harvested vessels (139). Meanwhile, progenitor-like cells that comprise atherosclerotic lesions of vein grafts have been shown to be derived from tissue-resident stem cells in the adventitia (65). Inclusion of the adventitia, perivascular adipose, and vasa vasorum together is thought to serve as key biological and mechanical support components for vein graft patency in coronary artery bypass grafting. Although these clinically relevant examples highlight the importance of perivascular tissues and vasa vasorum in the health and function of parent vessels, what remains unknown and mostly untested is whether microvascular dysfunction and local cells directly incite pathologies of larger vessels.
Aortic aneurysm and dissection
The aorta carries a higher blood volume than any other vessel, and its high number of elastic lamellae dictates a clear need for a robust vasa vasorum network to nourish the outer layers of aortic media. Several lines of evidence collected using animal surgical models reveal the impact that loss of vasa vasorum imparts on aortic function. In the mid-1960s, an investigation by Wilens et al. (6) demonstrated in a canine model that stoppage of blood flow through the vasa vasorum via ligation at the origin of four pairs of intercostal arteries resulted in elastin degeneration and medial necrosis, mainly in the middle third of the aorta media of the descending thoracic aorta with no sign of aortic dissection. Mitotic adventitial fibroblasts, neovascularization, and thickening of the adventitia were noted within 1 week after ligature. Endothelial proliferation and leukocyte infiltration around adventitial vessels in addition to neovascularization of the outer third of the media were observed within 2 weeks. Notably, Wilens et al. (6) do not report development of aneurysm in any of the five dogs that underwent intercostal artery ligation when compared with a single sham–operated dog. In 1981, a similar report of a larger canine study (n = 22 dogs) revealed that intercostal artery ligation resulted in dilated vasa vasorum within the middle third of the aortic media, which were unresponsive to the vasodilator adenosine as measured by tracking injected microspheres (140). Medial necrosis and loss of elastin were reproducible findings noted, absent of gross changes in aortic size. Vasa vasorum underwent modest recovery of vasodilator capacity at later time points. The authors’ observations of an increased prominence of vasa vasorum led them to suggest that regenerative changes occurred following intercostal ligation. Aggregated medial cells of unknown phenotype were also observed, which opens questions of cell dedifferentiation, transdifferentiation, or infiltration. The same group later demonstrated that an aortic interposition graft disrupted vasa vasorum blood flow and led to medial necrosis in the ascending aorta but not in the abdominal aorta and concluded that adventitial vasa vasorum is not required for nourishment of the medial layer (141). Adventitial vasa vasorum penetrate the outer two-thirds of the medial layer in the ascending aorta; therefore, their essential role in nourishment of the medial layer in this aortic region remains critical.
Other canine studies in the mid-1990s revealed that the removal of the perivascular fat from the left subclavian artery to the coronary arteries containing the vasa vasorum similarly led to medial necrosis, elastin degeneration, and a decrease in aortic distensibility, although aneurysm formation was not evident (142, 143). When blood flow through the vasa vasorum of the porcine descending aorta was disrupted, straightening of elastin and collagen fibers and increased tissue stiffness was noted without changes in collagen or elastin protein content (144). The authors also described a detachment of layers near the adventitia-media border. These observations led to the notion that a mechanical defect due to tissue remodeling predisposes the aorta to dissection, a theory supported by vasa vasorum alterations observed in human aneurysmal specimens (43) and dissected specimens (145) and by the typical location of dissection near the adventitia-media border. Occlusion of the porcine vasa vasorum of the descending thoracic aorta led to “ischemic necrosis” in the outer aortic media evidenced by localized SMC loss and areas of elastin interspersed with fragmented elastin fibers (144). These phenomena are reminiscent of the histopathological hallmark of human thoracic aortic aneurysm (TAA) known as cystic medial degeneration. This work in animal models, albeit generally limited in sample size, supports a vital role for the vasa vasorum in aortic wall integrity, although it is important to emphasize that these microvascular perturbations did not lead to overt aortic dilatation, dissection, or rupture, thus limiting the usefulness of the models and interpretation of the findings. Further studies should determine the impact of disruption of the vasa vasorum on biomechanical strength of the aorta to better understand the influence of these microvessels on tissue biomechanics and a resultant influence on the propensity for dissection or rupture.
Aneurysms of the abdominal aorta can arise idiopathically or because of lifestyle conditions. Aneurysms in this locale involve extensive inflammation-mediated wall degeneration, and intraluminal thrombi (ILT) commonly develop. Macrophage accumulation and increased production of reactive oxygen species are thought to provoke increased production and related activity of matrix metalloproteinases, the drivers of elastin and collagen degeneration and a hallmark of abdominal aortic aneurysm (AAA) pathophysiology. Unlike the thoracic aorta, human abdominal aortic media are typically avascular and devoid of vasa vasorum (7), although increased microvascular density has been reported for human AAA (124, 146–149). Histopathological studies of human AAAs revealed increased presence of medial microvasculature as shown by lectin- and laminin-positive cells (150), as well as CD31+ (146) and CD34+ (151) cells when compared with normal abdominal aorta, indicative of vasa vasorum expansion into the media. Another study compared small (<5 cm), mid-sized (5 to 7 cm), and large (>7 cm) AAAs and reported a modest reduction in CD31+ cells in the aortic media of AAA specimens (147). When all diameters were considered and compared with a smaller set of normal aortic specimens (n = 6), a higher area fraction of CD31+ cells were found in large aneurysms when compared with small AAA. Cells participating in AAA-associated vasa vasorum neovascularization and intimal hyperplasia are believed to have originated in the adventitia. In addition to the aortic lumen, medial vasa vasorum expansion is a possible route of administration for inflammatory cells to the media (149) and production of mitogenic cytokines that can induce SMC proliferation and intimal hyperplasia. In aneurysmal sac specimens from human AAA, luminal stenosis was noted in adventitial vasa vasorum explained by intimal hyperplasia and proliferative smooth muscle within the wall of the vasa vasorum evidenced by Ki67-positive cells (148). This study also reported hypoxia inducible factor 1α (HIF-1α)–positive cells within the adventitial vasa vasorum of aneurysm sac specimens near the intima in addition to HIF-1α immunoreactivity with partial overlap with the macrophage marker calprotectin within medial specimens of the aneurysm sac and neck. Vasa vasorum expansion in human AAA was found in abundance within the adventitia and outer media, although this was not observed in the associated ILTs (152). Further support for the theory that hypoperfusion of vasa vasorum invokes and exacerbates progression of AAA comes from work in a rat model (153–156). Removal of perivascular tissue, insertion of a polyurethane catheter in the infrarenal abdominal aorta, and suture ligation of the aorta over the catheter together resulted in hypoperfusion of the adventitial vasa vasorum without disrupting blood flow in the aortic lumen and led to a twofold expansion of the aortic diameter within 14 days (155). A follow-up study using the same hypoperfusion technique in rats noted diffuse and hyperlocal HIF-1α expression in the aortic wall within 24 hours of hypoperfusion onset with elastin fragmentation, increased gelatinase activity, decreased collagen content, and apoptotic SMCs observed after 28 days (153). Most recently, the same group reported rapid detection of HIF-1α in medial SMCs and macrophages (within 3 hours after ligation) and, despite an increased number of vasa vasorum, inferred that their hypoperfusion exerts time-dependent effects on aortic wall biology including potential hypoxia-mediated changes in contractile SMCs before onset of AAA (156). Pretreatment with a phosphodiesterase inhibitor (K-134) was shown to prevent aortic rupture in the hypoperfusion rat model, with noted decreases in vasa vasorum stenosis and medial hypoxia as well as prevention of rupture in elastase- and Ang-II–induced models of AAA (157). These studies in rat models definitively demonstrate that when vasa vasorum perfusion is physically compromised, wall hypoxia and degeneration ensue, resulting in histological phenomena reminiscent of human aneurysmal pathology. That hypoxic cells in the wall of vasa vasorum incite inflammatory cell infiltration and transition of SMCs from a contractile to synthetic phenotype is plausible, although further study using new experimental techniques is needed to determine whether vasa vasorum alterations precede or result from aneurysmal dilatation in the human abdominal aorta. Nonetheless, involvement of the arterial vasa vasorum in AAA offers an opportunity for therapeutic intervention.
There may be no vessel more life-sustaining than the aorta, and when support from its resident microvessels is compromised or restricted, the parent vessel consequently exhibits histopathologic features consistent with wall weakening and vulnerability such as medial degeneration. Involvement of vasa vasorum in both AAA and TAA has long been hypothesized. The likely compromised function of the parent vessel requires further interrogation to determine putative biomechanical changes due to loss of or reduced function of vasa vasorum. A prior review summarized evidence supporting a role for the vasa vasorum in aortic dissection, a theory built on a clinical and experimental knowledge base that ECM deformations and alterations in contractile forces reduce microvascular tone and lead to an influx of interstitial fluid and an increase in transmural pressure (158). Others have reviewed the literature and suggest that faulty mechanosensing is involved in aortic disease, especially because of genetic defects in key genes that regulate cell contractility and the elastin-contractile unit and are associated with aneurysm and dissection (159). Thus, it has been proposed that loss of microvascular tone coupled with faulty mechanosensing cause aortic dissection. Additional studies that incorporate multidisciplinary approaches to comprehensively evaluating manipulation of vascular tone are needed to definitively test this theory.
Other summaries postulate that SMC origins may contribute to dissection tear location. This developmentally minded theory is based on observations that dissection tears were histologically identified in the outer third of the aortic media in the vast majority (95%) of a 21-patient cohort and emerging genetic fate mapping studies, revealing clear demarcation of cellular origins in the ascending aorta with neural crest–derived cells noted throughout the entire media and second heart field–derived cells confined to the outer media (160). Several other factors likely influence tear location and the clinical manifestations of dissection (161) including material properties of the aortic wall (162), hemodynamics, wall tensile stress (163), wall shear stress (WSS) (164), and wall stiffness unrelated to WSS (165).
Studies centered on human ascending aortic specimens support the theory of vasa vasorum involvement in aortic disease including aneurysm and dissection. Osada et al. (145) considered specimens from 21 patients undergoing aortic replacement operation due to either Stanford type A (n = 20) or Stanford type B (n = 1) dissection. The researchers located dissection tears in the outer third of the aortic media and adventitial inflammation and thickening. Examples of histological phenomena consistent with remodeling of the vasa vasorum including tortuosity, elastosis, intimal fibrosis, and muscular hyperplasia were noted in two to four patient specimens per feature. Adventitial inflammation was found to be associated with sclerosis of the vasa vasorum evidenced by lymphoid aortitis. In their report, the authors allude to red blood cell extravasation from the vasa vasorum as a “probable initial feature of dissection.” IMH absent of an intimal tear occurs in a minority of dissection autopsies (166) yet has been frequently speculated to stem from rupture of medial vasa vasorum aneurysm (167), yet data to support this notion remain sparse (168, 169). More rarely, an associated penetrating atherosclerotic ulcer is thought to cause vasa vasorum rupture, leading to formation of an IMH, a concept that is also empirically unsubstantiated as of yet. This hemorrhage from vasa vasorum could, in theory, progress outwardly, causing rupture, or inwardly, as an intimal tear and dissection initiation. Histological artifacts notwithstanding these provocative findings encourage additional studies to evaluate causation, and contemporary surgical expert opinion continues to mount a logical and convincing argument for a role for vasa vasorum in aortic dissection (170). Hemorrhage of the vasa vasorum inciting IMH and leading to aortic dissection reveals an additional facet of aortic pathophysiology and, with the advent of novel imaging modalities and multiparameter tissue level studies, may offer new methods for preventative therapies through microvascular protection.
Reminiscent of the work of Heistad in canine models (140, 171), Kessler et al. (172) noted increased medial vasa vasorum density in specimens of human TAA as evidenced by a higher number of von Willebrand factor–positive vessels. This study found increased expression of angiogenic factors, angiopoietin 1 and 2, in the media of human TAA specimens, which were unchanged in adventitial specimens collected from just four patients when compared with healthy specimens from nine patients. Conditioned medium from TAA specimens reduced impedance of cultured endothelial cells and prevented their migration/proliferation when compared with conditioned medium from healthy aortic specimens. A supposition of microvessel leakage was inferred from increased expression of plasma proteins albumin and plasminogen in the media of human aneurysm specimens and qualitative observations of poor endothelial junctional associations and mural cell coverage from electron micrographs. The authors interpret these data as evidence that any angiogenic process in the aortic media likely spawned from within the adventitial layer. Although perhaps proangiogenic factors in the media also exert chemotactic influence on adventitial cells. Furthermore, additional in-depth studies that interrogate endothelial integrity, cell-cell junctions, and communication are needed to confirm endothelial dysfunction in vasa vasorum of human TAA.
Research initiatives from the author’s laboratory enthusiastically support a fresh perspective of the adventitia in aortic biology and agree with writings by Kessler et al. (172), Osada et al. (145), and others (15, 173) that this important blood vessel layer directly affects medial biology and deserves our attention and inquiry to delineate previously unknown aortic disease mechanisms. Work by the author’s team in human aneurysmal specimens partially agree with the findings of Kessler et al. (172), with respect to decreased Vegf and elevated thrombospondin 1 (TSP1), although this was found in the adventitia rather than the aortic media (43). In contrast to Kessler et al., who observed increased vasa vasorum in the media of human TAA specimens, the study of Billaud et al. (43) identified decreased adventitial vasa vasorum density. Only adventitial vasa vasorum morphometrics were quantified as medial vessels are more difficult to discern amidst smooth muscle and elastic lamellae. Decreased density of adventitial vasa vasorum in TAA specimens was associated with increased lumen area and thickened walls. Moreover, increase in lumen size may be due to poor pericyte-endothelial cell interactions. These features of microvascular remodeling were associated with locally increased expression of glucose transporter 1 (Glut1), a marker of chronic hypoxia, in the aortic media of aneurysmal specimens. Elevated Glut1 was most prominent in specimens of degenerative TAA and was found near regions of intense elastin fragmentation. There were fewer areas of focally elevated Glut1 in specimens of bicuspid aortic valve–associated human aneurysm, perhaps due to notably less elastin fragmentation in these specimens. This evidence of vasa vasorum remodeling and medial hypoxia is strongly suggestive of poor microvascular function in TAA. Likewise, inflammatory infiltrates have been commonly observed near vasa vasorum (43, 174–177) particularly in the adventitia and have been proposed as an influence of putative neovessel formation in the aortic media. However, medial vasa vasorum are challenging to delineate by histological methods, and evidence of neovessels is unconvincing—absent of definitive and quantitative markers of neovessel formation. Additional studies are needed to determine the underlying cause of hypoxia and vasa vasorum remodeling and how one phenomenon may influence the other. Increased lumen area may also be a response to hypoxia as was shown in a prior canine study examining effects of acute systemic hypoxia (39). Conversely, vasa vasorum dysfunction due to vessel occlusion as in atherosclerotic vessels or constriction (178, 179) could cause the aortic media to become hypoxic. Elevated expression of the antiangiogenic factor TSP1 and down-regulation of hypoxic gene targets and proangiogenic factors Vegf, Hif1α, and Mt-1a were also noted in the adventitia of human TAA specimens (43). Hypoxia evokes a cascade of proangiogenic signaling, causing neovessel formation as demonstrated in the hypertensive rat aorta (180). It is well established that hypoxia triggers angiogenic activity including the vasa vasorum in the rat aorta (180), human carotid artery (181), and bovine pulmonary artery (182). Paradoxically, multiple lines of evidence demonstrate that chronic hypoxia can also inhibit an angiogenic response (183, 184). Work focusing on the ECM of the adventitia in human aorta revealed that a biomaterial composed of decellularized aneurysmal tissue contained less proangiogenic factors (e.g., angiopoietin 2, VEGF-A, and FGF2) than nonaneurysmal aortic tissue (185). These studies suggest that vasa vasorum rarefaction and/or lack of angiogenesis in human TAA may be influenced by an imbalance of pro- and antiangiogenic signals in the adventitia, owing to, contributing to, and/or perpetuating a state of chronic hypoxia in human TAA. Current avenues of inquiry are focused on identifying what factors regulate vasa vasorum function in the setting of TAA.
The reduction in vasa vasorum density that is observed in human TAA differs from what is seen in human AAA, where vasa vasorum expand and contribute to neovascularization of an associated atherosclerotic lesion (150). The law of LaPlace applies to aortic dilatation where wall weakening, biomechanical deformation, and increased tangential stress together can likely create an ischemic environment in the media and adventitia. Together with the presence of an ILT additionally contributing to local tissue hypoxia, the prompted accumulation of inflammatory cytokines can enable local vasa vasorum expansion. When canine aortic segments were subjected to dilatation via balloon catheters, the vasa vasorum became damaged such that extravasation occurred (186). Tissue remodeling after balloon injury included proliferation of vasa vasorum, formation of a neointima, and adventitial hyperplasia (186), revealing that vasa vasorum can reestablish after disruption due to parent vessel dilation, thus protecting from further aneurysm progression. Additional studies including new animal models are needed to better delineate whether and how vasa vasorum remodeling causes, exacerbates, or alleviates aneurysmal pathophysiology in the thoracic and abdominal aorta.
Cerebral aneurysm
Clarke (28) first stated in 1965 that cerebral arteries lack vasa vasorum; however, intracranial arteries were since found to contain vasa vasorum that do not extend to the media or intima. Later studies described adventitial microvessels of the canine internal carotid and vertebral arteries (20). Despite multiple studies in the late 1990s that sought to settle the debate on the existence of vasa vasorum in intracranial arteries, the matter remains unresolved at present with some studies, failing to find evidence of vasa vasorum (19), while others report the presence of adventitial vessels (122) described in some reports as being confined to the proximal ends of the internal carotid and vertebral arteries (19, 187). An area of agreement is the existence of vasa vasorum in the media of intracranial arteries in a pathological setting (e.g., atherosclerosis, thrombosis, or aneurysm). It has been suggested that the same structural features that could explain a lack of vasa vasorum in cerebral arteries may also contribute to the formation of aneurysm in these vessels; these include a fenestrated internal elastic lamina, a thinner vessel wall, a lack of external elastic lamina, and a dearth of stroma in the perivascular subarachnoid space (18). Vasa vasorum are not thought to cause aneurysm formation in the intracranial arteries, an interpretation based on reproducible findings that vasa vasorum are seen in saccular aneurysms of >4 mm (188–190). Specimens from a cohort of unruptured intracranial specimens revealed via histological methods not only the presence of vasa vasorum mainly in the adventitia but also the ILT in 61% of specimens (14 of 23) (191). Rather, the presence of vasa vasorum seems to play a role in aneurysm enlargement. A recent article noted vasa vasorum in 25 specimens of intracranial aneurysms in a range of sizes from 3 to 27 mm associated with increased inflammatory infiltrate and intimal hyperplasia (192). Since vasa vasorum are relatively fragile and under pathological conditions such as aneurysm, which disrupt flow and vessel homeostasis, occluded vessels could lead to intralaminar hematoma, triggering an inflammatory response, and release of mitogenic growth factors as reported for AAA (193). Leakiness, bleeding, and hyperplasia of vasa vasorum likely contribute to aneurysm growth, wall weakening, and an increased propensity for rupture.
More recently, a newly established model of intracranial aneurysm was developed in ovariectomized rats (194). This was achieved by several manipulations of the vasculature including ligation of the left common carotid artery, the right external carotid artery, and the right pterygopalatine artery. Systemic hypertension was induced via high-salt diet and peritoneal injection of Ang-II and inhibitors of nitric oxide synthase, and the copper-dependent matrix cross-linking enzyme lysyl oxidase (194). Histological analysis of 12 ruptured aneurysms revealed the presence of vasa vasorum in approximately one-half of the specimens. The number of vasa vasorum in the ruptured lesions was relatively low (~1 to 2 vessels); however, the diameters of smooth muscle actin–positive vasa vasorum appear to be on the order of 25 to 100 μm, and the area occupied was reported to be ~600 μm2. The authors also observed higher Fgf2 expression in rupture-prone intracranial aneurysms when compared with lesions near bifurcations without rupture. Therefore, the presence of local proangiogenic factors could favor development of vasa vasorum and contribute to a propensity for rupture of the host vessel. Similar to the pursuit of preventing rupture of atherosclerotic plaques through VEGFR blockade, administration of agents that limit VEGF or FGF2 signaling may lessen risk for intracranial rupture through modulation of vasa vasorum expansion.
Vasa vasorum in pulmonary hypertension
A role for vasa vasorum remodeling in pulmonary hypertension has been demonstrated over the past two decades via extensive work in sophisticated animal models and observations of human specimens. A calf model of hypoxia-induced pulmonary hypertension revealed vasa vasorum expansion (182). Consistent with studies reporting a contribution of bone marrow–derived cells to atherosclerosis-related arterial remodeling and plaque development (123), an increased proportion of c-kit+ cells in peripheral blood versus bone marrow and the presence of c-kit+ cells in pulmonary artery vasa vasorum were interpreted as bone marrow–derived progenitor cells, migrating to areas of neovascularization in hypoxic calves. In pulmonary artery specimens from patients with idiopathic pulmonary arterial hypertension, similar progenitor-like cells coexpressing c-kit and CD34 were noted in the vascular wall along with vasa vasorum expansion and increased numbers of circulating bone marrow–derived cells (195). Extravasation from vasa vasorum was noted in hypoxic calves as well, indicating that neovessels exhibit leakiness, similar to vasa vasorum noted in TAA (43) or dissection (145). Later work identified that the highly proliferative c-kit+ cells isolated from hypoxic calves included more ECFC-like cells among vasa vasorum–associated endothelial cells than control (normoxic) calves (196). Most recently, transcriptional control of angiogenic activity of vasa vasorum–associated endothelial cells isolated from hypoxia-exposed calves was defined (197). It was also learned that adenosine receptors mediate vasa vasorum endothelial barrier function (198), thus identifying a putative therapeutic target to improve vasa vasorum function in pulmonary hypertension. These elegant studies broadly advanced our knowledge of pathological vasa vasorum remodeling not only by uncovering involvement of hypoxia-mediated vasa vasorum expansion in pulmonary hypertension but also by identifying the contributing cell populations, elucidating the underlying signaling pathways leading to neovessel formation and uncovering the putative molecular mediators to therapeutically improve endothelial function in disease. It will be important to apply similarly rigorous and elegant experimental designs to learn more about vasa vasorum remodeling in aneurysmal and atherosclerotic disease and cardiovascular manifestations of other diseases, including those involving emerging pathogens.
Vasa vasorum and severe acute respiratory syndrome coronavirus 2 infection
Given the extensively reported involvement of the microvasculature and angiotensin-converting enzyme 2–positive cells including pericytes and the frequent occurrence of microthrombi in fatalities from cardiovascular complication of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection (199), the vasa vasorum have become part of the conversation as a center point of SARS-CoV-2–induced microthrombi in lung microvasculature and potentially other large arteries (200, 201). Autopsy case reports of patients with coronavirus disease 2019 (COVID-19) describe histological evidence of thrombotic microangiopathy and vasculitis in thrombosed mid-sized vessels and perivascular lymphocytic infiltration (202). Fibrin microthrombi near pericytes were noted in the lungs. Another study reported the development of an occlusive arterial disease requiring lower left limb amputation and, upon histological examination, identified lymphocytic infiltrate and edema of adventitial vasa vasorum (203). More recently, multiple occlusive microthrombi were noted in the vasa vasorum amidst numerous atherosclerotic plaques in the aorta of a patient with a fatal COVID-19 infection (204). Evidence of focal inflammation of the endothelium and endothelial detachment in the vasa vasorum was also noted. These studies and emerging theories on the role of vasa vasorum and associated pericytes represent just the beginning of what we have yet to study, understand, and conquer in the ongoing battle against COVID-19 infection.
Advances in vasa vasorum imaging
Clinical imaging of vasa vasorum formation, remodeling, and function could serve as powerful diagnostic and prognostic tools in the evaluation of the many cardiovascular diseases where vasa vasorum are involved whether as a cause or consequence of the pathophysiology. Contrast-enhanced clinical imaging modalities (e.g., magnetic resonance and CT) that identify changes in vasa vasorum would be helpful in both diagnosis and prognosis of cardiovascular disease. Micro-CT, optical coherence tomography (OCT), and magnetic resonance imaging (MRI) are high-resolution imaging modalities that have successfully detected microvasculature in preclinical models. A disadvantage of micro-CT for broad clinical adoption is the exposure to radioisotopes and contrast agents. OCT is a high-resolution intravascular imaging technique with demonstrated success in detection of vasa vasorum in humans and in animal models, yet it suffers from inadequate depth of tissue penetration. Furthermore, OCT availability in cardiac catheterization centers remains sparse, thus limiting translational feasibility and potential. Likewise, MRI systems are impractical for repeated imaging due to device size and cost. Conventional noninvasive ultrasound such as Doppler and contrast-enhanced ultrasound (CEU) is compatible for vascular imaging but lacks the spatial resolution necessary for imaging microvessels the size of vasa vasorum. High-frequency intravascular ultrasound results in sufficient resolution but has the drawback of being invasive (205).
Super-resolution ultrasound (SRU) is an approach inspired by other super-resolution imaging techniques such as photoactivated localization microscopy and stochastic optical reconstruction microscopy. In SRU, contrast agents perfuse vascular networks to supplant the role of fluorophores in the optical super-resolution imaging techniques. The desirable larger imaging depth achieved through ultrasound is unfortunately at the expense of spatial resolution. Intravascular ultrasound using microbubbles can adequately quantify vasa vasorum density but is susceptible to motion artifact and is not applicable to larger vessels such as the aorta or carotid arteries. A simulated approach was applied for detection of coronary artery vasa vasorum using microbubble perfused cellulose tubing, thus demonstrating additional feasibility of using vasa vasorum remodeling as a metric for plaque rupture risk (206). CEU has been postured as an attractive approach for risk stratification of atherosclerotic plaques. Using microbubbles, transcutaneous CEU allows for visualization of adventitial vasa vasorum expansion in human atherosclerotic carotid arteries (207) and femoral arteries in pig and rabbit models of atherosclerotic plaque development (208–211). CEU in rabbit femoral arteries revealed increasing vasa vasorum density associated with atherosclerotic plaques, findings that were validated histologically (210). Despite a perceived promise of the CEU approach to enabling derivation of a diagnostic index for identifying unstable plaques, this strategy has also faced criticism that enhancement may lead to inaccurate detection of vasa vasorum. Recent advances in SRU imaging demonstrated detection of vasa vasorum in atherosclerotic plaques within rabbit femoral arteries, validated with ex vivo micro-CT and histological methods (208). Despite these promising modalities, implementation of a clinically impactful noninvasive imaging tool with sufficient spatial resolution to quantify metrics of vasa vasorum is hindered by long scan times, control of microbubble concentration, and acoustic attenuation in deeper tissues. Eventual high-resolution bioimaging technologies that balance spatial resolution with depth of penetration and enable detection of regional vasa vasorum remodeling will offer additional advantages. The numerous associated cell populations and how they dynamically interact within the adventitia and influence cells of the medial and intima layer provide additional biologic imaging targets of interest to better understand vasa vasorum presence and function in normal and diseased vessels using emerging technical innovations in noninvasive imaging.
Therapeutic opportunities in the vasa vasorum
Therapeutic strategies should be tailored to what pathological changes the vasa vasorum undergo during different disease processes and the pertinent consequences of these changes. For example, in aneurysmal disease of the ascending aorta where adventitial vasa vasorum have been shown to be less dense and the media is hypoxic (43), a strategy that restores the balance in pro- and antiangiogenic signals might be a candidate strategy for treatment. The author’s laboratory demonstrated that human adventitia contains numerous angiogenesis-related factors and that adventitial ECM derived from aneurysmal aortic specimens exhibited less proangiogenic and high amounts of TSP1 (185). ECM hydrogel bioscaffolds prepared from decellularized porcine aortic adventitia exhibited proangiogenic activity in vitro and in vivo predominantly via FGF receptor 2– and VEGFR2-dependent mechanism (185). FGF2 was previously inferred as the main proangiogenic factor in the adventitia, governing pathologic expansion of the vasa vasorum due to an observation of elevated Fgf2 mRNA and unchanged levels of another well-known proangiogenic factor, Vegf, in a hypercholesterolemic mouse model (212). Conversely, in atherosclerotic disease and aneurysms of the abdominal aorta, prevention of vasa vasorum–derived neovessels that facilitate neointimal formation and nurture its persistence could be abrogated through inhibition of proangiogenic signals. There are numerous studies that seek to limit plaque development through inhibition of angiogenesis including a first-in-man clinical trial with bevacizumab-eluting stent (213). However, in part, because preclinical models failed to show efficacy of intravenous bevacizumab where intercostal artery ligation led to hypoxia-induced neovascularization in the descending aorta of pigs (132), any therapy addressing this vital microvascular network should be carefully tailored. A successful strategy will take into consideration not only the vasculogenic potential of local cell populations and their abundance but also the delicate balance of pro- and antiangiogenic factors in a temporal and spatially appropriate manner, as well as the contribution of pro- and anti-inflammatory signals. A balance among these many facets must be the goal.
Final perspectives
Vasa vasorum remodeling is likely both a cause and consequence of parent vessel disease and reaching a deep understanding of both perspectives could have tremendous impact on how cardiovascular pathologies are diagnosed and clinically managed, in addition to how they are studied toward the development of novel therapeutic interventions. The establishment of these microvascular network patterns during development likely stem from spatial cues of soluble and immobilized morphogens that signal to local vasculogenic cells. Postnatal thickening of the parent vessel via addition of elastic lamellae directs vasa vasorum expansion from the adventitia to the media in some vessels. ECM biochemical and biophysical cues and overall health of the host vessel, especially the presence of atherosclerotic or aneurysmal disease, likely guide postnatal vasa vasorum remodeling. Advances in imaging modalities such as contrast enhanced ultrasound offer new opportunities to differentiate vasa vasorum changes related to disease onset and severity; these, together with basic science discovery, will pave the way toward theranostic interventions for diseases involving vasa vasorum. New animal models could help discern which changes in the vasa vasorum incite disease of the parent vessel or exacerbate disease progression while serving as test beds for the development of novel imaging modalities and therapeutics. Several perivascular populations of vasculogenic progenitor cells identified within the adventitia may contribute to vasa vasorum expansion and remodeling in normal and pathological vessels, offering additional insight into vessel pathophysiology and opportunities for constructive tissue remodeling, regeneration, and repair.
Several questions regarding the role of vasa vasorum in cardiovascular disease persist. Involvement of arterial vasa vasorum expansion in atherosclerosis, AAA, and pulmonary hypertension is clear from both animal studies and observations in human specimens with hypoxia-driven neovascularization a main pathway of disease progression. However, what is the cadence for adventitial cell migration and differentiation toward vascular lineages to form plaque neovessels? What are the spatial and temporal matrix cues that drive pathological neovascularization? Evidence for a role of vasa vasorum dysfunction in ascending aortic disease is less clear, perhaps because of potential for artifactual observations and differences in human specimens by ethnicity and/or disease etiology and severity, as well as other patient-specific and sample differences among study cohorts, thus limiting the ability for study reproduction. However, observations of red blood cell extravasation in diseased human ascending aortas have been noted by multiple groups, implicating reduced vasa vasorum integrity as a putative driver of IMH and aortic dissection. Limitations of human vessel studies aside, animal models support a role for vasa vasorum as both an inciting and exacerbating component of aneurysm and/or dissection. Is arterial vasa vasorum remodeling a cause or consequence of medial hypoxia in human ascending aortic disease, or both? A definitive answer will require further study via novel animal models complemented by in vitro disease models of human tissue and cells. That bone marrow–derived cells contribute to neovessel formation in atherosclerotic vessels and vasa vasorum expansion in pulmonary hypertension, it is likely that circulating progenitor cells also contribute to neovascularization in AAA. Could these and other tissue-resident precursors of the endothelial, pericyte, and SMC lineage be harnessed for therapeutic vasa vasorum regeneration in thoracic aortic disease or improvement of vein/arterial graft patency for coronary artery bypass graft? A summary of the evidence for vasa vasorum involvement in several cardiovascular pathologies and the persisting unanswered questions are depicted in Fig. 5 and remain the areas of intense research interest for the author and other researchers.
Fig. 5. Vasa vasorum in diseased vessels.
The schematic depicts the known involvement of vasa vasorum involvement in several cardiovascular and pulmonary pathologies including intracranial aneurysm, atherosclerosis, pulmonary hypertension, COVID-19, and aneurysms of the abdominal and ascending thoracic aorta. The unanswered questions that persist and deserve future indepth study are outlined.
The vasa vasorum have captivated relatively few adoring anatomists/scientists and artists for centuries and anew amidst the ongoing global threat of the COVID-19 pandemic. This vital microvascular network is expected to provide additional clues of disease origins and opportunities for intervention by medicinal, cellular, or biomaterial-based therapies and surgical and image-guided therapies. The vasa vasorum remain worthy of our continued curiosity and deserving of increased attention to advance our understanding of great vessel disease and organ and tissue failure.
Acknowledgments
I acknowledge T. Richards for exceptional editorial assistance on this manuscript. Figure 1 was drafted in collaboration with Ella Maru Studio Inc. with the final version created by the author using BioRender (BioRender.com). Figures 3 to 5 were created using BioRender.
Funding: This work was supported by National Institutes of Health (grant HL131632), Commonwealth of PA CURE Program grant, UPMC Pellegrini Chair in Cardiothoracic Surgery, University of Pittsburgh Physicians, and Root Family Fund for Lung Transplantation and Lung Failure Research.
Competing interests: The author declares that she has no competing interests.
Data and materials availability: All data needed to evaluate the conclusions in the paper are present in the paper.
REFERENCES AND NOTES
- 1.T. Willis, Pharmaceutice rationalis (1678).
- 2.F. Ruysch, Frederici Ruyschii responsio ad eruditum Dr. Johannem Gaubium, in Gaubius J Epistola Anatomica, Problem-Atica Tertia, J. Wolters, Ed. (Ab Amstelodami, 1696). [Google Scholar]
- 3.Musil V., Sach J., Kachlik D., Patzelt M., Stingl J., Vasa vasorum: An old term with new problems. Surg. Radiol. Anat. 40, 1159–1164 (2018). [DOI] [PubMed] [Google Scholar]
- 4.Clarke J. A., An x-ray microscopic study of the vasa vasorum of the normal human thoracic aorta. Z. Anat. Entwicklungsgesch. 124, 261–267 (1964). [DOI] [PubMed] [Google Scholar]
- 5.Koester W., Enarteritis and arteritis. Berl. Klin. Wochenschr. 13, 454–455 (1876). [Google Scholar]
- 6.Wilens S. L., Malcolm J. A., Vazquez J. M., Experimental infarction (medial necrosis) of the dog’s aorta. Am. J. Pathol. 47, 695–711 (1965). [PMC free article] [PubMed] [Google Scholar]
- 7.Wolinsky H., Glagov S., Nature of species differences in the medial distribution of aortic vasa vasorum in mammals. Circ. Res. 20, 409–421 (1967). [DOI] [PubMed] [Google Scholar]
- 8.Woodruff C. E., Studies on the vasa vasorum. Am. J. Pathol. 2, 567–570 565 (1926). [PMC free article] [PubMed] [Google Scholar]
- 9.Clarke J. A., An x-ray microscopic study of the postnatal development of the vasa vasorum in the human aorta. J. Anat. 99, 877–889 (1965). [PMC free article] [PubMed] [Google Scholar]
- 10.Gössl M., Rosol M., Malyar N. M., Fitzpatrick L. A., Beighley P. E., Zamir M., Ritman E. L., Functional anatomy and hemodynamic characteristics of vasa vasorum in the walls of porcine coronary arteries. Anat Rec A Discov Mol Cell Evol Biol 272, 526–537 (2003). [DOI] [PubMed] [Google Scholar]
- 11.Kwon H. M., Sangiorgi G., Ritman E. L., Lerman A., McKenna C., Virmani R., Edwards W. D., Holmes D. R., Schwartz R. S., Adventitial vasa vasorum in balloon-injured coronary arteries: Visualization and quantitation by a microscopic three-dimensional computed tomography technique. J. Am. Coll. Cardiol. 32, 2072–2079 (1998). [DOI] [PubMed] [Google Scholar]
- 12.Geiringer E., Intimal vascularization and atherosclerosis. J. Pathol. Bacteriol. 63, 201–211 (1951). [DOI] [PubMed] [Google Scholar]
- 13.Tonar Z., Tomasek P., Loskot P., Janácek J., Králicková M., Witter K., Vasa vasorum in the tunica media and tunica adventitia of the porcine aorta. Ann. Anat. 205, 22–36 (2016). [DOI] [PubMed] [Google Scholar]
- 14.Federspiel J. M., Tschernig T., Laschke M. W., Wagenpfeil S., Schnabel P., Schafers H. J., The vasa vasorum reach deep into the human thoracic aorta. Ann. Anat. 225, 54–56 (2019). [DOI] [PubMed] [Google Scholar]
- 15.Sano M., Unno N., Sasaki T., Baba S., Sugisawa R., Tanaka H., Inuzuka K., Yamamoto N., Sato K., Konno H., Topologic distributions of vasa vasorum and lymphatic vasa vasorum in the aortic adventitia—Implications for the prevalence of aortic diseases. Atherosclerosis 247, 127–134 (2016). [DOI] [PubMed] [Google Scholar]
- 16.Hildebrandt H. A., Gossl M., Mannheim D., Versari D., Herrmann J., Spendlove D., Bajanowski T., Malyar N. M., Erbel R., Lerman L. O., Lerman A., Differential distribution of vasa vasorum in different vascular beds in humans. Atherosclerosis 199, 47–54 (2008). [DOI] [PubMed] [Google Scholar]
- 17.Galili O., Herrmann J., Woodrum J., Sattler K. J., Lerman L. O., Lerman A., Adventitial vasa vasorum heterogeneity among different vascular beds. J. Vasc. Surg. 40, 529–535 (2004). [DOI] [PubMed] [Google Scholar]
- 18.Portanova A., Hakakian N., Mikulis D. J., Virmani R., Abdalla W. M., Wasserman B. A., Intracranial vasa vasorum: Insights and implications for imaging. Radiology 267, 667–679 (2013). [DOI] [PubMed] [Google Scholar]
- 19.Aydin F., Do human intracranial arteries lack vasa vasorum? A comparative immunohistochemical study of intracranial and systemic arteries. Acta Neuropathol. 96, 22–28 (1998). [DOI] [PubMed] [Google Scholar]
- 20.Zervas N. T., Liszczak T. M., Mayberg M. R., Black P. M., Cerebrospinal fluid may nourish cerebral vessels through pathways in the adventitia that may be analogous to systemic vasa vasorum. J. Neurosurg. 56, 475–481 (1982). [DOI] [PubMed] [Google Scholar]
- 21.Clarke J. A., An x-ray microscopic study of the vasa vasorum of normal human pulmonary arteries. Thorax 19, 561–567 (1964). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Clarke J. A., An x-ray microscopic study of the vasa vasorum of normal human coronary arteries. J. Anat. 98, 539–543 (1964). [PMC free article] [PubMed] [Google Scholar]
- 23.Clarke J. A., An x-ray microscopic study of the postnatal development of the vasa vasorum in the root of neck arteries. Anat. Anz. 117, 280–290 (1965). [PubMed] [Google Scholar]
- 24.Clarke J. A., An x-ray microscopic study of the vasa vasorum of the human ductus arteriosus. J. Anat. 99, 527–537 (1965). [PMC free article] [PubMed] [Google Scholar]
- 25.Clarke J. A., An x-ray microscopic study of the postnatal development of the vasa vasorum in the pulmonary trunk and arteries. Thorax 20, 348–356 (1965). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Clarke J. A., An x-ray microscopic study of the vasa vasorum of the normal human internal carotid and vertebral arteries. J. Neurol. Sci. 2, 301–306 (1965). [DOI] [PubMed] [Google Scholar]
- 27.Clarke J. A., An x-ray microscopic study of the postnatal development of the vasa vasorum in human upper limb arteries. Anat. Anz. 116, 345–351 (1965). [PubMed] [Google Scholar]
- 28.Clarke J. A., An x-ray microscopic study of the vasa vasorum of the intracranial arteries. Z. Anat. Entwicklungsgesch. 124, 396–400 (1965). [DOI] [PubMed] [Google Scholar]
- 29.Clarke J. A., An x-ray microscopic study of the vasa vasorum of the human umbilical arteries. Z. Zellforsch. Mikrosk. Anat. 66, 293–299 (1965). [DOI] [PubMed] [Google Scholar]
- 30.Clarke J. A., An x-ray microscopic study of the vasa vasorum of the normal human abdominal aorta. Z. Zellforsch. Mikrosk. Anat. 66, 212–218 (1965). [DOI] [PubMed] [Google Scholar]
- 31.Clarke J. A., An x-ray microscopic study of the postnatal development of the vasa vasorum in human lower limb arteries. Z. Zellforsch. Mikrosk. Anat. 65, 897–903 (1965). [DOI] [PubMed] [Google Scholar]
- 32.Clarke J. A., The vasa vasorum of normal human upper limb arteries. Anat. Anz. 116, 1–5 (1965). [PubMed] [Google Scholar]
- 33.Clarke J. A., An x-ray microscopic study of the vasa vasorum of the normal human aortic arch. Thorax 20, 76–81 (1965). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Clarke J. A., An x-ray microscopic study of the vasa vasorum of the normal human ascending aorta. Br. Heart J. 27, 99–104 (1965). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Clarke J. A., The vasa vasorum of normal human lower limb arteries. Acta Anat. 61, 481–487 (1965). [DOI] [PubMed] [Google Scholar]
- 36.Clarke J. A., An x-ray microscopic study of the vasa vasorum of the normal human pulmonary trunk. Acta Anat. 61, 6–14 (1965). [DOI] [PubMed] [Google Scholar]
- 37.Clarke J. A., An x-ray microscopic study of the postnatal development of the vasa vasorum of normal human coronary arteries. Acta Anat. 64, 506–516 (1966). [DOI] [PubMed] [Google Scholar]
- 38.Clarke J. A., An x-ray microscopic study of the development of the vasa vasorum in the human foetal aorta and pulmonary trunk. Acta Anat. 63, 55–70 (1966). [DOI] [PubMed] [Google Scholar]
- 39.Heistad D. D., Armstrong M. L., Amundsen S., Blood flow through vasa vasorum in arteries and veins: Effects of luminal PO2. Am. J. Phys. 250, H434–H442 (1986). [DOI] [PubMed] [Google Scholar]
- 40.Kachlik D., Baca V., Stingl J., Sosna B., Lametschwandtner A., Minnich B., Setina M., Architectonic arrangement of the vasa vasorum of the human great saphenous vein. J. Vasc. Res. 44, 157–166 (2007). [DOI] [PubMed] [Google Scholar]
- 41.Vasuri F., Fittipaldi S., Buzzi M., Degiovanni A., Stella A., D’Errico-Grigioni A., Pasquinelli G., Nestin and WT1 expression in small-sized vasa vasorum from human normal arteries. Histol. Histopathol. 27, 1195–1202 (2012). [DOI] [PubMed] [Google Scholar]
- 42.Giannoni M. F., Vicenzini E., Citone M., Ricciardi M. C., Irace L., Laurito A., Scucchi L. F., Di Piero V., Gossetti B., Mauriello A., Spagnoli L. G., Lenzi G. L., Valentini F. B., Contrast carotid ultrasound for the detection of unstable plaques with neoangiogenesis: A pilot study. Eur. J. Vasc. Endovasc. Surg. 37, 722–727 (2009). [DOI] [PubMed] [Google Scholar]
- 43.Billaud M., Hill J. C., Richards T. D., Gleason T. G., Phillippi J. A., Medial hypoxia and adventitial vasa vasorum remodeling in human ascending aortic aneurysm. Front. Cardiovas. Med. 5, 124 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Billaud M., Donnenberg V. S., Ellis B. W., Meyer E. M., Donnenberg A. D., Hill J. C., Richards T. D., Gleason T. G., Phillippi J. A., Classification and functional characterization of vasa vasorum-associated perivascular progenitor cells in human aorta. Stem Cell Rep. 9, 292–303 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Kwon H. M., Sangiorgi G., Ritman E. L., McKenna C., Holmes D. R. Jr., Schwartz R. S., Lerman A., Enhanced coronary vasa vasorum neovascularization in experimental hypercholesterolemia. J. Clin. Invest. 101, 1551–1556 (1998). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Kwon T. G., Gulati R., Matsuzawa Y., Aoki T., Guddeti R. R., Herrmann J., Lennon R. J., Ritman E. L., Lerman L. O., Lerman A., Proliferation of coronary adventitial vasa vasorum in patients with spontaneous coronary artery dissection. J. Am. Coll. Cardiol. Img. 9, 891–892 (2016). [DOI] [PubMed] [Google Scholar]
- 47.Heistad D. D., Marcus M. L., Law E. G., Armstrong M. L., Ehrhardt J. C., Abboud F. M., Regulation of blood flow to the aortic media in dogs. J. Clin. Invest. 62, 133–140 (1978). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Scotland R. S., Vallance P. J., Ahluwalia A., Endogenous factors involved in regulation of tone of arterial vasa vasorum: Implications for conduit vessel physiology. Cardiovasc. Res. 46, 403–411 (2000). [DOI] [PubMed] [Google Scholar]
- 49.Heistad D. D., Marcus M. L., Martins J. B., Effects of neural stimuli on blood flow through vasa vasorum in dogs. Circ. Res. 45, 615–620 (1979). [DOI] [PubMed] [Google Scholar]
- 50.Traktuev D. O., Merfeld-Clauss S., Li J., Kolonin M., Arap W., Pasqualini R., Johnstone B. H., March K. L., A population of multipotent CD34-positive adipose stromal cells share pericyte and mesenchymal surface markers, reside in a periendothelial location, and stabilize endothelial networks. Circ. Res. 102, 77–85 (2008). [DOI] [PubMed] [Google Scholar]
- 51.Crisan M., Yap S., Casteilla L., Chen C. W., Corselli M., Park T. S., Andriolo G., Sun B., Zheng B., Zhang L., Norotte C., Teng P. N., Traas J., Schugar R., Deasy B. M., Badylak S., Buhring H. J., Giacobino J. P., Lazzari L., Huard J., Péault B., A perivascular origin for mesenchymal stem cells in multiple human organs. Cell Stem Cell 3, 301–313 (2008). [DOI] [PubMed] [Google Scholar]
- 52.Okada M., Payne T. R., Zheng B., Oshima H., Momoi N., Tobita K., Keller B. B., Phillippi J. A., Peault B., Huard J., Myogenic endothelial cells purified from human skeletal muscle improve cardiac function after transplantation into infarcted myocardium. J. Am. Coll. Cardiol. 52, 1869–1880 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Park T. S., Gavina M., Chen C. W., Sun B., Teng P. N., Huard J., Deasy B. M., Zimmerlin L., Peault B., Placental perivascular cells for human muscle regeneration. Stem Cells Dev. 20, 451–463 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Chen C. W., Okada M., Proto J. D., Gao X., Sekiya N., Beckman S. A., Corselli M., Crisan M., Saparov A., Tobita K., Peault B., Huard J., Human pericytes for ischemic heart repair. Stem Cells 31, 305–316 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Hoshino A., Chiba H., Nagai K., Ishii G., Ochiai A., Human vascular adventitial fibroblasts contain mesenchymal stem/progenitor cells. Biochem. Biophys. Res. Commun. 368, 305–310 (2008). [DOI] [PubMed] [Google Scholar]
- 56.Campagnolo P., Cesselli D., Al Haj Zen A., Beltrami A. P., Krankel N., Katare R., Angelini G., Emanueli C., Madeddu P., Human adult vena saphena contains perivascular progenitor cells endowed with clonogenic and proangiogenic potential. Circulation 121, 1735–1745 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Pasquinelli G., Tazzari P. L., Vaselli C., Foroni L., Buzzi M., Storci G., Alviano F., Ricci F., Bonafe M., Orrico C., Bagnara G. P., Stella A., Conte R., Thoracic aortas from multiorgan donors are suitable for obtaining resident angiogenic mesenchymal stromal cells. Stem Cells 25, 1627–1634 (2007). [DOI] [PubMed] [Google Scholar]
- 58.Pasquinelli G., Pacilli A., Alviano F., Foroni L., Ricci F., Valente S., Orrico C., Lanzoni G., Buzzi M., Luigi Tazzari P., Pagliaro P., Stella A., Paolo Bagnara G., Multidistrict human mesenchymal vascular cells: Pluripotency and stemness characteristics. Cytotherapy 12, 275–287 (2010). [DOI] [PubMed] [Google Scholar]
- 59.Fang B., Li Y., Song Y., Li N., Isolation and characterization of multipotent progenitor cells from the human fetal aorta wall. Exp. Biol. Med. 235, 130–138 (2010). [DOI] [PubMed] [Google Scholar]
- 60.Corselli M., Chen C. W., Sun B., Yap S., Rubin J. P., Peault B., The tunica adventitia of human arteries and veins as a source of mesenchymal stem cells. Stem Cells Dev. 21, 1299–1308 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Michelis K. C., Nomura-Kitabayashi A., Lecce L., Franzen O., Koplev S., Xu Y., Santini M. P., D’Escamard V., Lee J. T. L., Fuster V., Hajjar R., Reddy R. C., Chikwe J., Stelzer P., Filsoufi F., Stewart A., Anyanwu A., Bjorkegren J. L. M., Kovacic J. C., CD90 identifies adventitial mesenchymal progenitor cells in adult human medium- and large-sized arteries. Stem Cell Rep. 11, 242–257 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Passman J. N., Dong X. R., Wu S. P., Maguire C. T., Hogan K. A., Bautch V. L., Majesky M. W., A sonic hedgehog signaling domain in the arterial adventitia supports resident Sca1+ smooth muscle progenitor cells. Proc. Natl. Acad. Sci. U.S.A. 105, 9349–9354 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Psaltis P. J., Puranik A. S., Spoon D. B., Chue C. D., Hoffman S. J., Witt T. A., Delacroix S., Kleppe L. S., Mueske C. S., Pan S., Gulati R., Simari R. D., Characterization of a resident population of adventitial macrophage progenitor cells in postnatal vasculature. Circ. Res. 115, 364–375 (2014). [DOI] [PubMed] [Google Scholar]
- 64.Zengin E., Chalajour F., Gehling U. M., Ito W. D., Treede H., Lauke H., Weil J., Reichenspurner H., Kilic N., Ergun S., Vascular wall resident progenitor cells: A source for postnatal vasculogenesis. Development 133, 1543–1551 (2006). [DOI] [PubMed] [Google Scholar]
- 65.Hu Y., Zhang Z., Torsney E., Afzal A. R., Davison F., Metzler B., Xu Q., Abundant progenitor cells in the adventitia contribute to atherosclerosis of vein grafts in ApoE-deficient mice. J. Clin. Invest. 113, 1258–1265 (2004). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Toledo-Flores D., Williamson A., Schwarz N., Fernando S., Dimasi C., Witt T. A., Nguyen T. M., Puranik A. S., Chue C. D., Delacroix S., Spoon D. B., Bonder C. S., Bursill C. A., Di Bartolo B. A., Nicholls S. J., Simari R. D., Psaltis P. J., Vasculogenic properties of adventitial Sca-1+CD45+ progenitor cells in mice: A potential source of vasa vasorum in atherosclerosis. Sci. Rep. 9, 7286 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Kramann R., Goettsch C., Wongboonsin J., Iwata H., Schneider R. K., Kuppe C., Kaesler N., Chang-Panesso M., Machado F. G., Gratwohl S., Madhurima K., Hutcheson J. D., Jain S., Aikawa E., Humphreys B. D., Adventitial MSC-like cells are progenitors of vascular smooth muscle cells and drive vascular calcification in chronic kidney disease. Cell Stem Cell 19, 628–642 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Patel J., Seppanen E. J., Rodero M. P., Wong H. Y., Donovan P., Neufeld Z., Fisk N. M., Francois M., Khosrotehrani K., Functional definition of progenitors versus mature endothelial cells reveals key SoxF-dependent differentiation process. Circulation 135, 786–805 (2017). [DOI] [PubMed] [Google Scholar]
- 69.Majesky M. W., Horita H., Ostriker A., Lu S., Regan J. N., Bagchi A., Dong X. R., Poczobutt J., Nemenoff R. A., Weiser-Evans M. C., Differentiated smooth muscle cells generate a subpopulation of resident vascular progenitor cells in the adventitia regulated by Klf4. Circ. Res. 120, 296–311 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Asahara T., Murohara T., Sullivan A., Silver M., van der Zee R., Li T., Witzenbichler B., Schatteman G., Isner J. M., Isolation of putative progenitor endothelial cells for angiogenesis. Science 275, 964–967 (1997). [DOI] [PubMed] [Google Scholar]
- 71.Kawamoto A., Tkebuchava T., Yamaguchi J., Nishimura H., Yoon Y. S., Milliken C., Uchida S., Masuo O., Iwaguro H., Ma H., Hanley A., Silver M., Kearney M., Losordo D. W., Isner J. M., Asahara T., Intramyocardial transplantation of autologous endothelial progenitor cells for therapeutic neovascularization of myocardial ischemia. Circulation 107, 461–468 (2003). [DOI] [PubMed] [Google Scholar]
- 72.Yoder M. C., Mead L. E., Prater D., Krier T. R., Mroueh K. N., Li F., Krasich R., Temm C. J., Prchal J. T., Ingram D. A., Redefining endothelial progenitor cells via clonal analysis and hematopoietic stem/progenitor cell principals. Blood 109, 1801–1809 (2007). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Medina R. J., O’Neill C. L., Sweeney M., Guduric-Fuchs J., Gardiner T. A., Simpson D. A., Stitt A. W., Molecular analysis of endothelial progenitor cell (EPC) subtypes reveals two distinct cell populations with different identities. BMC Med. Genet. 3, 18 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Ziegelhoeffer T., Fernandez B., Kostin S., Heil M., Voswinckel R., Helisch A., Schaper W., Bone marrow-derived cells do not incorporate into the adult growing vasculature. Circ. Res. 94, 230–238 (2004). [DOI] [PubMed] [Google Scholar]
- 75.Purhonen S., Palm J., Rossi D., Kaskenpaa N., Rajantie I., Yla-Herttuala S., Alitalo K., Weissman I. L., Salven P., Bone marrow-derived circulating endothelial precursors do not contribute to vascular endothelium and are not needed for tumor growth. Proc. Natl. Acad. Sci. U.S.A. 105, 6620–6625 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Ingram D. A., Mead L. E., Moore D. B., Woodard W., Fenoglio A., Yoder M. C., Vessel wall-derived endothelial cells rapidly proliferate because they contain a complete hierarchy of endothelial progenitor cells. Blood 105, 2783–2786 (2005). [DOI] [PubMed] [Google Scholar]
- 77.Naito H., Kidoya H., Sakimoto S., Wakabayashi T., Takakura N., Identification and characterization of a resident vascular stem/progenitor cell population in preexisting blood vessels. EMBO J. 31, 842–855 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Aicher A., Rentsch M., Sasaki K., Ellwart J. W., Fandrich F., Siebert R., Cooke J. P., Dimmeler S., Heeschen C., Nonbone marrow-derived circulating progenitor cells contribute to postnatal neovascularization following tissue ischemia. Circ. Res. 100, 581–589 (2007). [DOI] [PubMed] [Google Scholar]
- 79.Isner J. M., Asahara T., Angiogenesis and vasculogenesis as therapeutic strategies for postnatal neovascularization. J. Clin. Invest. 103, 1231–1236 (1999). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Banerji S., Ni J., Wang S. X., Clasper S., Su J., Tammi R., Jones M., Jackson D. G., LYVE-1, a new homologue of the CD44 glycoprotein, is a lymph-specific receptor for hyaluronan. J. Cell Biol. 144, 789–801 (1999). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Breiteneder-Geleff S., Soleiman A., Kowalski H., Horvat R., Amann G., Kriehuber E., Diem K., Weninger W., Tschachler E., Alitalo K., Kerjaschki D., Angiosarcomas express mixed endothelial phenotypes of blood and lymphatic capillaries: Podoplanin as a specific marker for lymphatic endothelium. Am. J. Pathol. 154, 385–394 (1999). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Johnson L. A., Banerji S., Lawrance W., Gileadi U., Prota G., Holder K. A., Roshorm Y. M., Hanke T., Cerundolo V., Gale N. W., Jackson D. G., Dendritic cells enter lymph vessels by hyaluronan-mediated docking to the endothelial receptor LYVE-1. Nat. Immunol. 18, 762–770 (2017). [DOI] [PubMed] [Google Scholar]
- 83.Jackson D. G., Hyaluronan in the lymphatics: The key role of the hyaluronan receptor LYVE-1 in leucocyte trafficking. Matrix Biol. 78-79, 219–235 (2019). [DOI] [PubMed] [Google Scholar]
- 84.Bianchi R., Russo E., Bachmann S. B., Proulx S. T., Sesartic M., Smaadahl N., Watson S. P., Buckley C. D., Halin C., Detmar M., Postnatal deletion of podoplanin in lymphatic endothelium results in blood filling of the lymphatic system and impairs dendritic cell migration to lymph nodes. Arterioscler. Thromb. Vasc. Biol. 37, 108–117 (2017). [DOI] [PubMed] [Google Scholar]
- 85.Xu H., Chen M., Reid D. M., Forrester J. V., LYVE-1–positive macrophages are present in normal murine eyes. Invest. Ophthalmol. Vis. Sci. 48, 2162–2171 (2007). [DOI] [PubMed] [Google Scholar]
- 86.Lim H. Y., Lim S. Y., Tan C. K., Thiam C. H., Goh C. C., Carbajo D., Chew S. H. S., See P., Chakarov S., Wang X. N., Lim L. H., Johnson L. A., Lum J., Fong C. Y., Bongso A., Biswas A., Goh C., Evrard M., Yeo K. P., Basu R., Wang J. K., Tan Y., Jain R., Tikoo S., Choong C., Weninger W., Poidinger M., Stanley E. R., Collin M., Tan N. S., Ng L. G., Jackson D. G., Ginhoux F., Angeli V., Hyaluronan receptor LYVE-1-expressing macrophages maintain arterial tone through hyaluronan-mediated regulation of smooth muscle cell collagen. Immunity 49, 1191 (2018). [DOI] [PubMed] [Google Scholar]
- 87.Murray I. R., West C. C., Hardy W. R., James A. W., Park T. S., Nguyen A., Tawonsawatruk T., Lazzari L., Soo C., Peault B., Natural history of mesenchymal stem cells, from vessel walls to culture vessels. Cell. Mol. Life Sci. 71, 1353–1374 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Horiuchi K., Kano K., Minoshima A., Hayasaka T., Yamauchi A., Tatsukawa T., Matsuo R., Yoshida Y., Tomita Y., Kabara M., Nakagawa N., Takehara N., Hasebe N., Kawabe J. I., Pericyte-specific deletion of ninjurin-1 induces fragile vasa vasorum formation and enhances intimal hyperplasia of injured vasculature. Am. J. Physiol. Heart Circ. Physiol. , (2021). [DOI] [PubMed] [Google Scholar]
- 89.Levéen P., Pekny M., Gebre-Medhin S., Swolin B., Larsson E., Betsholtz C., Mice deficient for PDGF B show renal, cardiovascular, and hematological abnormalities. Genes Dev. 8, 1875–1887 (1994). [DOI] [PubMed] [Google Scholar]
- 90.Soriano P., Abnormal kidney development and hematological disorders in PDGF beta-receptor mutant mice. Genes Dev. 8, 1888–1896 (1994). [DOI] [PubMed] [Google Scholar]
- 91.Alvino V. V., Fernandez-Jimenez R., Rodriguez-Arabaolaza I., Slater S., Mangialardi G., Avolio E., Spencer H., Culliford L., Hassan S., Ballesteros L. S., Herman A., Ayaon-Albarran A., Galan-Arriola C., Sanchez-Gonzalez J., Hennessey H., Delmege C., Ascione R., Emanueli C., Angelini G. D., Ibanez B., Madeddu P., Transplantation of allogeneic pericytes improves myocardial vascularization and reduces interstitial fibrosis in a swine model of reperfused acute myocardial infarction. J. Am. Heart Assoc. 7, e006727 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Chen C. W., Corselli M., Peault B., Huard J., Human blood-vessel-derived stem cells for tissue repair and regeneration. J. Biomed. Biotechnol. 2012, 597439 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Ozerdem U., Alitalo K., Salven P., Li A., Contribution of bone marrow-derived pericyte precursor cells to corneal vasculogenesis. Invest. Ophthalmol. Vis. Sci. 46, 3502–3506 (2005). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Tigges U., Hyer E. G., Scharf J., Stallcup W. B., FGF2-dependent neovascularization of subcutaneous Matrigel plugs is initiated by bone marrow-derived pericytes and macrophages. Development 135, 523–532 (2008). [DOI] [PubMed] [Google Scholar]
- 95.Amos P. J., Shang H., Bailey A. M., Taylor A., Katz A. J., Peirce S. M., IFATS collection: The role of human adipose-derived stromal cells in inflammatory microvascular remodeling and evidence of a perivascular phenotype. Stem Cells 26, 2682–2690 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Guimarães-Camboa N., Cattaneo P., Sun Y., Moore-Morris T., Gu Y., Dalton N. D., Rockenstein E., Masliah E., Peterson K. L., Stallcup W. B., Chen J., Evans S. M., Pericytes of multiple organs do not behave as mesenchymal stem cells in vivo. Cell Stem Cell 20, 345–359.e5 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Bordenave J., Tu L., Berrebeh N., Thuillet R., Cumont A., Le Vely B., Fadel E., Nadaud S., Savale L., Humbert M., Huertas A., Guignabert C., Lineage tracing reveals the dynamic contribution of pericytes to the blood vessel remodeling in pulmonary hypertension. Arterioscler. Thromb. Vasc. Biol. 40, 766–782 (2020). [DOI] [PubMed] [Google Scholar]
- 98.West C. C., Hardy W. R., Murray I. R., James A. W., Corselli M., Pang S., Black C., Lobo S. E., Sukhija K., Liang P., Lagishetty V., Hay D. C., March K. L., Ting K., Soo C., Péault B., Prospective purification of perivascular presumptive mesenchymal stem cells from human adipose tissue: Process optimization and cell population metrics across a large cohort of diverse demographics. Stem Cell Res Ther 7, 47 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Zimmerlin L., Donnenberg V. S., Pfeifer M. E., Meyer E. M., Peault B., Rubin J. P., Donnenberg A. D., Stromal vascular progenitors in adult human adipose tissue. Cytometry. A 77, 22–30 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Chen Q., Zhang H., Liu Y., Adams S., Eilken H., Stehling M., Corada M., Dejana E., Zhou B., Adams R. H., Endothelial cells are progenitors of cardiac pericytes and vascular smooth muscle cells. Nat. Commun. 7, 12422 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Nakashima T., Tashiro T., Early morphologic stage of human coronary atherosclerosis. Kurume Med. J. 15, 235–242 (1968). [DOI] [PubMed] [Google Scholar]
- 102.Herrmann J., Lerman L. O., Rodriguez-Porcel M., Holmes D. R. Jr., Richardson D. M., Ritman E. L., Lerman A., Coronary vasa vasorum neovascularization precedes epicardial endothelial dysfunction in experimental hypercholesterolemia. Cardiovasc. Res. 51, 762–766 (2001). [DOI] [PubMed] [Google Scholar]
- 103.Kumar A., Kumar S., Vikram A., Hoffman T. A., Naqvi A., Lewarchik C. M., Kim Y. R., Irani K., Histone and DNA methylation-mediated epigenetic downregulation of endothelial Kruppel-like factor 2 by low-density lipoprotein cholesterol. Arterioscler. Thromb. Vasc. Biol. 33, 1936–1942 (2013). [DOI] [PubMed] [Google Scholar]
- 104.Subbotin V. M., Neovascularization of coronary tunica intima (DIT) is the cause of coronary atherosclerosis. Lipoproteins invade coronary intima via neovascularization from adventitial vasa vasorum, but not from the arterial lumen: A hypothesis. Theor. Biol. Med. Model. 9, 11 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Gossl M., Versari D., Hildebrandt H. A., Bajanowski T., Sangiorgi G., Erbel R., Ritman E. L., Lerman L. O., Lerman A., Segmental heterogeneity of vasa vasorum neovascularization in human coronary atherosclerosis. J. Am. Coll. Cardiol. Img. 3, 32–40 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Virmani R., Kolodgie F. D., Burke A. P., Finn A. V., Gold H. K., Tulenko T. N., Wrenn S. P., Narula J., Atherosclerotic plaque progression and vulnerability to rupture: Angiogenesis as a source of intraplaque hemorrhage. Arterioscler. Thromb. Vasc. Biol. 25, 2054–2061 (2005). [DOI] [PubMed] [Google Scholar]
- 107.Yeo K. P., Lim H. Y., Thiam C. H., Azhar S. H., Tan C., Tang Y., See W. Q., Koh X. H., Zhao M. H., Phua M. L., Balachander A., Tan Y., Lim S. Y., Chew H. S., Ng L. G., Angeli V., Efficient aortic lymphatic drainage is necessary for atherosclerosis regression induced by ezetimibe. Sci. Adv. 6, eabc2697 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Nakano T., Nakashima Y., Yonemitsu Y., Sumiyoshi S., Chen Y. X., Akishima Y., Ishii T., Iida M., Sueishi K., Angiogenesis and lymphangiogenesis and expression of lymphangiogenic factors in the atherosclerotic intima of human coronary arteries. Hum. Pathol. 36, 330–340 (2005). [DOI] [PubMed] [Google Scholar]
- 109.Singla B., Lin H. P., Ahn W., White J., Csanyi G., Oxidatively modified LDL suppresses lymphangiogenesis via CD36 signaling. Antioxidants 10, 331 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Heistad D. D., Armstrong M. L., Blood flow through vasa vasorum of coronary arteries in atherosclerotic monkeys. Arteriosclerosis 6, 326–331 (1986). [DOI] [PubMed] [Google Scholar]
- 111.Bacon C. R., Cary N. R., Davenport A. P., Endothelin peptide and receptors in human atherosclerotic coronary artery and aorta. Circ. Res. 79, 794–801 (1996). [DOI] [PubMed] [Google Scholar]
- 112.Scotland R., Vallance P., Ahluwalia A., Endothelin alters the reactivity of vasa vasorum: Mechanisms and implications for conduit vessel physiology and pathophysiology. Br. J. Pharmacol. 128, 1229–1234 (1999). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Martin J. F., Booth R. F., Moncada S., Arterial wall hypoxia following thrombosis of the vasa vasorum is an initial lesion in atherosclerosis. Eur. J. Clin. Investig. 21, 355–359 (1991). [DOI] [PubMed] [Google Scholar]
- 114.Tanaka K., Nagata D., Hirata Y., Tabata Y., Nagai R., Sata M., Augmented angiogenesis in adventitia promotes growth of atherosclerotic plaque in apolipoprotein E-deficient mice. Atherosclerosis 215, 366–373 (2011). [DOI] [PubMed] [Google Scholar]
- 115.Chen P. Y., Qin L., Baeyens N., Li G., Afolabi T., Budatha M., Tellides G., Schwartz M. A., Simons M., Endothelial-to-mesenchymal transition drives atherosclerosis progression. J. Clin. Invest. 125, 4514–4528 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Moreno P. R., Purushothaman K. R., Fuster V., Echeverri D., Truszczynska H., Sharma S. K., Badimon J. J., O’Connor W. N., Plaque neovascularization is increased in ruptured atherosclerotic lesions of human aorta: Implications for plaque vulnerability. Circulation 110, 2032–2038 (2004). [DOI] [PubMed] [Google Scholar]
- 117.Kasper H. U., Schmidt A., Roessner A., Expression of the adhesion molecules ICAM, VCAM, and ELAM in the arteriosclerotic plaque. Gen. Diagn. Pathol. 141, 289–294 (1996). [PubMed] [Google Scholar]
- 118.Okamoto E., Couse T., De Leon H., Vinten-Johansen J., Goodman R. B., Scott N. A., Wilcox J. N., Perivascular inflammation after balloon angioplasty of porcine coronary arteries. Circulation 104, 2228–2235 (2001). [DOI] [PubMed] [Google Scholar]
- 119.Mateo J., Izquierdo-Garcia D., Badimon J. J., Fayad Z. A., Fuster V., Noninvasive assessment of hypoxia in rabbit advanced atherosclerosis using (1)(8)F-fluoromisonidazole positron emission tomographic imaging. Circ. Cardiovasc. Imaging 7, 312–320 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Nie X., Elvington A., Laforest R., Zheng J., Voller T. F., Zayed M. A., Abendschein D. R., Bandara N., Xu J., Li R., Randolph G. J., Gropler R. J., Lapi S. E., Woodard P. K., (64)Cu-ATSM positron emission tomography/magnetic resonance imaging of hypoxia in human atherosclerosis. Circ. Cardiovasc. Imaging 13, e009791 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Zheng L., Yang W. J., Niu C. B., Zhao H. L., Wong K. S., Leung T. W. H., Chen X. Y., Correlation of adventitial vasa vasorum with intracranial atherosclerosis: A postmortem study. J. Stroke 20, 342–349 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Takaba M., Endo S., Kurimoto M., Kuwayama N., Nishijima M., Takaku A., Vasa vasorum of the intracranial arteries. Acta Neurochir. 140, 411–416 (1998). [DOI] [PubMed] [Google Scholar]
- 123.Sata M., Saiura A., Kunisato A., Tojo A., Okada S., Tokuhisa T., Hirai H., Makuuchi M., Hirata Y., Nagai R., Hematopoietic stem cells differentiate into vascular cells that participate in the pathogenesis of atherosclerosis. Nat. Med. 8, 403–409 (2002). [DOI] [PubMed] [Google Scholar]
- 124.Hagensen M. K., Shim J., Thim T., Falk E., Bentzon J. F., Circulating endothelial progenitor cells do not contribute to plaque endothelium in murine atherosclerosis. Circulation 121, 898–905 (2010). [DOI] [PubMed] [Google Scholar]
- 125.Veerman K. J., Venegas-Pino D. E., Shi Y., Khan M. I., Gerstein H. C., Werstuck G. H., Hyperglycaemia is associated with impaired vasa vasorum neovascularization and accelerated atherosclerosis in apolipoprotein-E deficient mice. Atherosclerosis 227, 250–258 (2013). [DOI] [PubMed] [Google Scholar]
- 126.Purushothaman K.-R., Purushothaman M., Muntner P., Lento P. A., O’Connor W. N., Sharma S. K., Fuster V., Moreno P. R., Inflammation, neovascularization and intra-plaque hemorrhage are associated with increased reparative collagen content: Implication for plaque progression in diabetic atherosclerosis. Vasc. Med. 16, 103–108 (2011). [DOI] [PubMed] [Google Scholar]
- 127.Moulton K. S., Heller E., Konerding M. A., Flynn E., Palinski W., Folkman J., Angiogenesis inhibitors endostatin or TNP-470 reduce intimal neovascularization and plaque growth in apolipoprotein E–deficient mice. Circulation 99, 1726–1732 (1999). [DOI] [PubMed] [Google Scholar]
- 128.Moulton K. S., Vakili K., Zurakowski D., Soliman M., Butterfield C., Sylvin E., Lo K. M., Gillies S., Javaherian K., Folkman J., Inhibition of plaque neovascularization reduces macrophage accumulation and progression of advanced atherosclerosis. Proc. Natl. Acad. Sci. U.S.A. 100, 4736–4741 (2003). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.de Vries M. R., Parma L., Peters H. A. B., Schepers A., Hamming J. F., Jukema J. W., Goumans M., Guo L., Finn A. V., Virmani R., Ozaki C. K., Quax P. H. A., Blockade of vascular endothelial growth factor receptor 2 inhibits intraplaque haemorrhage by normalization of plaque neovessels. J. Intern. Med. 285, 59–74 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Parma L., Peters H. A. B., Sluiter T. J., Simons K. H., Lazzari P., de Vries M. R., Quax P. H. A., bFGF blockade reduces intraplaque angiogenesis and macrophage infiltration in atherosclerotic vein graft lesions in ApoE3*Leiden mice. Sci. Rep. 10, 15968 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Hytonen J. P., Taavitsainen J., Laitinen J. T. T., Partanen A., Alitalo K., Leppanen O., Yla-Herttuala S., Local adventitial anti-angiogenic gene therapy reduces growth of vasa-vasorum and in-stent restenosis in WHHL rabbits. J. Mol. Cell. Cardiol. 121, 145–154 (2018). [DOI] [PubMed] [Google Scholar]
- 132.Castro C., Pereira A. H., Histological changes secondary to use of anti-angiogenic therapy after interruption of vasa vasorum flow in the descending aorta: Results in a porcine model. J. Vasc. Bras. 18, e20180095 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Colman L., Caggiani M., Leyva A., Bresque M., Liechocki S., Maya-Monteiro C. M., Mazal D., Batthyany C., Calliari A., Contreras P., Escande C., The protein Deleted in Breast Cancer-1 (DBC1) regulates vascular response and formation of aortic dissection during Angiotensin II infusion. Sci. Rep. 10, 6772 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Loesch A., Dashwood M. R., Vasa vasorum inside out/outside in communication: A potential role in the patency of saphenous vein coronary artery bypass grafts. J. Cell Commun. Signal. 12, 631–643 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Dashwood M. R., Anand R., Loesch A., Souza D. S., Hypothesis: A potential role for the vasa vasorum in the maintenance of vein graft patency. Angiology 55, 385–395 (2004). [DOI] [PubMed] [Google Scholar]
- 136.Loesch A., Dashwood M. R., A brief comment on vasa vasorum of human saphenous vein: Relevance for coronary artery bypass surgery. Braz. J. Cardiovasc. Surg. 36, 106–111 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Loesch A., Dashwood M. R., Saphenous vein vasa vasorum as a potential target for perivascular fat-derived factors. Braz. J. Cardiovasc. Surg. 35, 964–969 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Deb S., Singh S. K., de Souza D., Chu M. W. A., Whitlock R., Meyer S. R., Verma S., Jeppsson A., Al-Saleh A., Brady K., Rao-Melacini P., Belley-Cote E. P., Tam D. Y., Devereaux P. J., Novick R. J., Fremes S. E., Investigators S. S. S., SUPERIOR SVG: No touch saphenous harvesting to improve patency following coronary bypass grafting (a multi-Centre randomized control trial, NCT01047449). J. Cardiothorac. Surg. 14, 85 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Dreifaldt M., Souza D. S., Loesch A., Muddle J. R., Karlsson M. G., Filbey D., Bodin L., Norgren L., Dashwood M. R., The “no-touch” harvesting technique for vein grafts in coronary artery bypass surgery preserves an intact vasa vasorum. J. Thorac. Cardiovasc. Surg. 141, 145–150 (2011). [DOI] [PubMed] [Google Scholar]
- 140.Heistad D. D., Marcus M. L., Larsen G. E., Armstrong M. L., Role of vasa vasorum in nourishment of the aortic wall. Am. J. Phys. 240, H781–H787 (1981). [DOI] [PubMed] [Google Scholar]
- 141.Williams J. K., Armstrong M. L., Heistad D. D., Blood flow through new microvessels: Factors that affect regrowth of vasa vasorum. Am. J. Phys. 254, H126–H132 (1988). [DOI] [PubMed] [Google Scholar]
- 142.Stefanadis C., Vlachopoulos C., Karayannacos P., Boudoulas H., Stratos C., Filippides T., Agapitos M., Toutouzas P., Effect of vasa vasorum flow on structure and function of the aorta in experimental animals. Circulation 91, 2669–2678 (1995). [DOI] [PubMed] [Google Scholar]
- 143.Stefanadis C. I., Karayannacos P. E., Boudoulas H. K., Stratos C. G., Vlachopoulos C. V., Dontas I. A., Toutouzas P. K., Medial necrosis and acute alterations in aortic distensibility following removal of the vasa vasorum of canine ascending aorta. Cardiovasc. Res. 27, 951–956 (1993). [DOI] [PubMed] [Google Scholar]
- 144.Angouras D., Sokolis D. P., Dosios T., Kostomitsopoulos N., Boudoulas H., Skalkeas G., Karayannacos P. E., Effect of impaired vasa vasorum flow on the structure and mechanics of the thoracic aorta: Implications for the pathogenesis of aortic dissection. Eur. J. Cardio Thorac. Surg. 17, 468–473 (2000). [DOI] [PubMed] [Google Scholar]
- 145.Osada H., Kyogoku M., Ishidou M., Morishima M., Nakajima H., Aortic dissection in the outer third of the media: What is the role of the vasa vasorum in the triggering process? Eur. J. Cardiothorac. Surg. 43, e82–e88 (2013). [DOI] [PubMed] [Google Scholar]
- 146.Blassova T., Tonar Z., Tomasek P., Hosek P., Hollan I., Treska V., Molacek J., Inflammatory cell infiltrates, hypoxia, vascularization, pentraxin 3 and osteoprotegerin in abdominal aortic aneurysms—A quantitative histological study. PLOS ONE 14, e0224818 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Eberlova L., Tonar Z., Witter K., Krizkova V., Nedorost L., Korabecna M., Tolinger P., Kocova J., Boudova L., Treska V., Houdek K., Molacek J., Vrzalova J., Pesta M., Topolcan O., Valenta J., Asymptomatic abdominal aortic aneurysms show histological signs of progression: A quantitative histochemical analysis. Pathobiology 80, 11–23 (2013). [DOI] [PubMed] [Google Scholar]
- 148.Tanaka H., Zaima N., Sasaki T., Hayasaka T., Goto-Inoue N., Onoue K., Ikegami K., Morita Y., Yamamoto N., Mano Y., Sano M., Saito T., Sato K., Konno H., Setou M., Unno N., Adventitial vasa vasorum arteriosclerosis in abdominal aortic aneurysm. PLOS ONE 8, e57398 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Thompson M. M., Jones L., Nasim A., Sayers R. D., Bell P. R., Angiogenesis in abdominal aortic aneurysms. Eur. J. Vasc. Endovasc. Surg. 11, 464–469 (1996). [DOI] [PubMed] [Google Scholar]
- 150.Holmes D. R., Liao S., Parks W. C., Thompson R. W., Medial neovascularization in abdominal aortic aneurysms: A histopathologic marker of aneurysmal degeneration with pathophysiologic implications. J. Vasc. Surg. 21, 761–771; discussion 771-762 (1995). [DOI] [PubMed] [Google Scholar]
- 151.Rodella L. F., Rezzani R., Bonomini F., Peroni M., Cocchi M. A., Hirtler L., Bonardelli S., Abdominal aortic aneurysm and histological, clinical, radiological correlation. Acta Histochem. 118, 256–262 (2016). [DOI] [PubMed] [Google Scholar]
- 152.Fontaine V., Touat Z., Mtairag el M., Vranckx R., Louedec L., Houard X., Andreassian B., Sebbag U., Palombi T., Jacob M. P., Meilhac O., Michel J. B., Role of leukocyte elastase in preventing cellular re-colonization of the mural thrombus. Am. J. Pathol. 164, 2077–2087 (2004). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Tanaka H., Zaima N., Sasaki T., Sano M., Yamamoto N., Saito T., Inuzuka K., Hayasaka T., Goto-Inoue N., Sugiura Y., Sato K., Kugo H., Moriyama T., Konno H., Setou M., Unno N., Hypoperfusion of the adventitial vasa vasorum develops an abdominal aortic aneurysm. PLOS ONE 10, e0134386 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Kugo H., Zaima N., Tanaka H., Hashimoto K., Miyamoto C., Sawaragi A., Urano T., Unno N., Moriyama T., Pathological analysis of the ruptured vascular wall of hypoperfusion-induced abdominal aortic aneurysm animal model. J. Oleo Sci. 66, 499–506 (2017). [DOI] [PubMed] [Google Scholar]
- 155.Tanaka H., Unno N., Yata T., Kugo H., Zaima N., Sasaki T., Urano T., Creation of a rodent model of abdominal aortic aneurysm by blocking adventitial vasa vasorum perfusion. J. Vis. Exp. 2017, 55763 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Kugo H., Sukketsiri W., Tanaka H., Fujishima R., Moriyama T., Zaima N., Time-dependent pathological changes in hypoperfusion-induced abdominal aortic aneurysm. Biology 10, 149 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Unno N., Tanaka H., Yata T., Kayama T., Yamanaka Y., Tsuyuki H., Sano M., Inuzuka K., Naruse E., Takeuchi H., K-134, a phosphodiesterase 3 inhibitor, reduces vascular inflammation and hypoxia, and prevents rupture of experimental abdominal aortic aneurysms. JVS Vasc. Sci. 1, 219–232 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Mallat Z., Tedgui A., Henrion D., Role of microvascular tone and extracellular matrix contraction in the regulation of interstitial fluid: Implications for aortic dissection. Arterioscler. Thromb. Vasc. Biol. 36, 1742–1747 (2016). [DOI] [PubMed] [Google Scholar]
- 159.Milewicz D. M., Trybus K. M., Guo D. C., Sweeney H. L., Regalado E., Kamm K., Stull J. T., Altered smooth muscle cell force generation as a driver of thoracic aortic aneurysms and dissections. Arterioscler. Thromb. Vasc. Biol. 37, 26–34 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Sawada H., Rateri D. L., Moorleghen J. J., Majesky M. W., Daugherty A., Smooth muscle cells derived from second heart field and cardiac neural crest reside in spatially distinct domains in the media of the ascending aorta-brief report. Arterioscler. Thromb. Vasc. Biol. 37, 1722–1726 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Sharif M., Yap Z. J., Ghazal A., Bashir M., Harky A., Tear size and location influence the pressure of false lumen following type a aortic dissection: Perspective of current evidence. Heart Lung Circ. 29, 178–187 (2020). [DOI] [PubMed] [Google Scholar]
- 162.Pasta S., Phillippi J. A., Gleason T. G., Vorp D. A., Effect of aneurysm on the mechanical dissection properties of the human ascending thoracic aorta. J. Thorac. Cardiovasc. Surg. 143, 460–467 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Emerel L., Thunes J., Kickliter T., Billaud M., Phillippi J. A., Vorp D. A., Maiti S., Gleason T. G., Predissection-derived geometric and distensibility indices reveal increased peak longitudinal stress and stiffness in patients sustaining acute type A aortic dissection: Implications for predicting dissection. J. Thorac. Cardiovasc. Surg. 158, 355–363 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Pasta S., Rinaudo A., Luca A., Pilato M., Scardulla C., Gleason T. G., Vorp D. A., Difference in hemodynamic and wall stress of ascending thoracic aortic aneurysms with bicuspid and tricuspid aortic valve. J. Biomech. 46, 1729–1738 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Di Giuseppe M., Farzaneh S., Zingales M., Pasta S., Avril S., Patient-specific computational evaluation of stiffness distribution in ascending thoracic aortic aneurysm. J. Biomech. 119, 110321 (2021). [DOI] [PubMed] [Google Scholar]
- 166.Wilson S. K., Hutchins G. M., Aortic dissecting aneurysms: Causative factors in 204 subjects. Arch. Pathol. Lab. Med. 106, 175–180 (1982). [PubMed] [Google Scholar]
- 167.Gore I., Pathogenesis of dissecting aneurysm of the aorta. A.M.A. Arch. Pathol. 53, 142–153 (1952). [PubMed] [Google Scholar]
- 168.Al Rstum Z., Tanaka A., Eisenberg S. B., Estrera A. L., Optimal timing of type A intramural hematoma repair. Ann. Cardiothorac. Surg. 8, 524–530 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Sundt T. M., Intramural hematoma and penetrating atherosclerotic ulcer of the aorta. Ann. Thorac. Surg. 83, S835–841; discussion S846-850 (2007). [DOI] [PubMed] [Google Scholar]
- 170.Haverich A., Boyle E. C., Aortic dissection is a disease of the vasa vasorum. JTCVS Open 5, 30–32 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Heistad D. D., Marcus M. L., Role of vasa vasorum in nourishment of the aorta. Blood Vessels 16, 225–238 (1979). [DOI] [PubMed] [Google Scholar]
- 172.Kessler K., Borges L. F., Ho-Tin-Noe B., Jondeau G., Michel J. B., Vranckx R., Angiogenesis and remodelling in human thoracic aortic aneurysms. Cardiovasc. Res. 104, 147–159 (2014). [DOI] [PubMed] [Google Scholar]
- 173.Li W., Li Q., Jiao Y., Qin L., Ali R., Zhou J., Ferruzzi J., Kim R. W., Geirsson A., Dietz H. C., Offermanns S., Humphrey J. D., Tellides G., Tgfbr2 disruption in postnatal smooth muscle impairs aortic wall homeostasis. J. Clin. Invest. 124, 755–767 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Agaimy A., Weyand M., Strecker T., Inflammatory thoracic aortic aneurysm (lymphoplasmacytic thoracic aortitis): A 13-year-experience at a German Heart Center with emphasis on possible role of IgG4. Int. J. Clin. Exp. Pathol. 6, 1713–1722 (2013). [PMC free article] [PubMed] [Google Scholar]
- 175.Chumachenko P. V., Postnov A. Y., Ivanova A. G., Afanasieva O. I., Afanasiev M. A., Ekta M. B., Sukhorukov V. N., Kheimets G. I., Sobenin I. A., Thoracic aortic aneurysm and factors affecting aortic dissection. J. Pers. Med. 10, 153 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Wu D., Choi J. C., Sameri A., Minard C. G., Coselli J. S., Shen Y. H., LeMaire S. A., Inflammatory cell infiltrates in acute and chronic thoracic aortic dissection. Aorta 1, 259–267 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.He R., Guo D. C., Estrera A. L., Safi H. J., Huynh T. T., Yin Z., Cao S. N., Lin J., Kurian T., Buja L. M., Geng Y. J., Milewicz D. M., Characterization of the inflammatory and apoptotic cells in the aortas of patients with ascending thoracic aortic aneurysms and dissections. J. Thorac. Cardiovasc. Surg. 131, 671–678 (2006). [DOI] [PubMed] [Google Scholar]
- 178.Kajino H., Goldbarg S., Roman C., Liu B. M., Mauray F., Chen Y. Q., Takahashi Y., Koch C. J., Clyman R. I., Vasa vasorum hypoperfusion is responsible for medial hypoxia and anatomic remodeling in the newborn lamb ductus arteriosus. Pediatr. Res. 51, 228–235 (2002). [DOI] [PubMed] [Google Scholar]
- 179.Barker S. G., Talbert A., Cottam S., Baskerville P. A., Martin J. F., Arterial intimal hyperplasia after occlusion of the adventitial vasa vasorum in the pig. Arterioscler. Thromb. 13, 70–77 (1993). [DOI] [PubMed] [Google Scholar]
- 180.Kuwahara F., Kai H., Tokuda K., Shibata R., Kusaba K., Tahara N., Niiyama H., Nagata T., Imaizumi T., Hypoxia-inducible factor-1α/vascular endothelial growth factor pathway for adventitial vasa vasorum formation in hypertensive rat aorta. Hypertension 39, 46–50 (2002). [DOI] [PubMed] [Google Scholar]
- 181.Lopez-Cano C., Rius F., Sanchez E., Gaeta A. M., Betriu A., Fernandez E., Yeramian A., Hernandez M., Bueno M., Gutierrez-Carrasquilla L., Dalmases M., Lecube A., The influence of sleep apnea syndrome and intermittent hypoxia in carotid adventitial vasa vasorum. PLOS ONE 14, e0211742 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182.Davie N. J., Crossno J. T. Jr., Frid M. G., Hofmeister S. E., Reeves J. T., Hyde D. M., Carpenter T. C., Brunetti J. A., McNiece I. K., Stenmark K. R., Hypoxia-induced pulmonary artery adventitial remodeling and neovascularization: Contribution of progenitor cells. Am. J. Physiol. Lung Cell. Mol. Physiol. 286, L668–L678 (2004). [DOI] [PubMed] [Google Scholar]
- 183.Aplin A. C., Nicosia R. F., Hypoxia paradoxically inhibits the angiogenic response of isolated vessel explants while inducing overexpression of vascular endothelial growth factor. Angiogenesis 19, 133–146 (2016). [DOI] [PubMed] [Google Scholar]
- 184.Olszewska-Pazdrak B., Hein T. W., Olszewska P., Carney D. H., Chronic hypoxia attenuates VEGF signaling and angiogenic responses by downregulation of KDR in human endothelial cells. Am. J. Physiol. Cell Physiol. 296, C1162–C1170 (2009). [DOI] [PubMed] [Google Scholar]
- 185.Fercana G. R., Yerneni S., Billaud M., Hill J. C., VanRyzin P., Richards T. D., Sicari B. M., Johnson S. A., Badylak S. F., Campbell P. G., Gleason T. G., Phillippi J. A., Perivascular extracellular matrix hydrogels mimic native matrix microarchitecture and promote angiogenesis via basic fibroblast growth factor. Biomaterials 123, 142–154 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Zollikofer C. L., Redha F. H., Bruhlmann W. F., Uhlschmid G. K., Vlodaver Z., Castaneda-Zuniga W. R., Amplatz K., Acute and long-term effects of massive balloon dilation on the aortic wall and vasa vasorum. Radiology 164, 145–149 (1987). [DOI] [PubMed] [Google Scholar]
- 187.Connolly E. S. Jr., Huang J., Goldman J. E., Holtzman R. N., Immunohistochemical detection of intracranial vasa vasorum: A human autopsy study. Neurosurgery 38, 789–793 (1996). [PubMed] [Google Scholar]
- 188.Sekhar L. N., Heros R. C., Origin, growth, and rupture of saccular aneurysms: A review. Neurosurgery 8, 248–260 (1981). [DOI] [PubMed] [Google Scholar]
- 189.Stehbens W. E., Histopathology of cerebral aneurysms. Arch. Neurol. 8, 272–285 (1963). [DOI] [PubMed] [Google Scholar]
- 190.Suzuki J., Ohara H., Clinicopathological study of cerebral aneurysms. Origin, rupture, repair, and growth. J. Neurosurg. 48, 505–514 (1978). [DOI] [PubMed] [Google Scholar]
- 191.Zhong W., Su W., Li T., Tan X., Chen C., Wang Q., Wang D., Su W., Wang Y., Aneurysm wall enhancement in unruptured intracranial aneurysms: A histopathological evaluation. J. Am. Heart Assoc. 10, e018633 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Wen D., Kieran N. W., Yu Z., Liu X., Xiao Y., Li H., You C., Yang M., Ma L., Presence of vasa vasorum in human intracranial aneurysms. Acta Neurochir. 162, 2283–2293 (2020). [DOI] [PubMed] [Google Scholar]
- 193.Zhao L., Moos M. P., Grabner R., Pedrono F., Fan J., Kaiser B., John N., Schmidt S., Spanbroek R., Lotzer K., Huang L., Cui J., Rader D. J., Evans J. F., Habenicht A. J., Funk C. D., The 5-lipoxygenase pathway promotes pathogenesis of hyperlipidemia-dependent aortic aneurysm. Nat. Med. 10, 966–973 (2004). [DOI] [PubMed] [Google Scholar]
- 194.Miyata H., Imai H., Koseki H., Shimizu K., Abekura Y., Oka M., Kawamata T., Matsuda T., Nozaki K., Narumiya S., Aoki T., Vasa vasorum formation is associated with rupture of intracranial aneurysms. J. Neurosurg. 133, 655–663 (2019). [DOI] [PubMed] [Google Scholar]
- 195.Montani D., Perros F., Gambaryan N., Girerd B., Dorfmuller P., Price L. C., Huertas A., Hammad H., Lambrecht B., Simonneau G., Launay J. M., Cohen-Kaminsky S., Humbert M., C-kit–positive cells accumulate in remodeled vessels of idiopathic pulmonary arterial hypertension. Am. J. Respir. Crit. Care Med. 184, 116–123 (2011). [DOI] [PubMed] [Google Scholar]
- 196.Nijmeh H., Balasubramaniam V., Burns N., Ahmad A., Stenmark K. R., Gerasimovskaya E. V., High proliferative potential endothelial colony-forming cells contribute to hypoxia-induced pulmonary artery vasa vasorum neovascularization. Am. J. Physiol. Lung Cell. Mol. Physiol. 306, L661–L671 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197.Strassheim D., Karoor V., Nijmeh H., Weston P., Lapel M., Schaack J., Sullivan T., Dempsey E. C., Stenmark K. R., Gerasimovskaya E., c-Jun, Foxo3a, and c-Myc transcription factors are key regulators of ATP-mediated angiogenic responses in pulmonary artery vasa vasorum endothelial cells. Cell 9, 416 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 198.Verin A. D., Batori R., Kovacs-Kasa A., Cherian-Shaw M., Kumar S., Czikora I., Karoor V., Strassheim D., Stenmark K. R., Gerasimovskaya E. V., Extracellular adenosine enhances pulmonary artery vasa vasorum endothelial cell barrier function via Gi/ELMO1/Rac1/PKA-dependent signaling mechanisms. Am. J. Physiol. Cell Physiol. 319, C183–C193 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199.McFadyen J. D., Stevens H., Peter K., The emerging threat of (micro)thrombosis in COVID-19 and its therapeutic implications. Circ. Res. 127, 571–587 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 200.Daisley H. Jr., Rampersad A., Daisley M., Ramdin A., Acco O., Narinesingh F., Humphrey O., James E., The vasa vasorum of the large pulmonary vessels are involved in COVID-19. Autops. Case Rep. 11, e2021304 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201.Boyle E. C., Haverich A., Microvasculature dysfunction as the common thread between atherosclerosis, Kawasaki disease, and severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2)-associated multi-system inflammatory syndrome in children. Eur. J. Cardio Thorac. Surg. 58, 1109–1110 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 202.Daisley H. Jr., Rampersad A., Daisley M., Ramdin A., Acco O., Narinesingh F., Humphrey O., COVID-19: A closer look at the pathology in two autopsied cases. Is the pericyte at the center of the pathological process in COVID-19? Autops. Case Rep. 11, e2021262 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.Vasuri F., Ciavarella C., Collura S., Mascoli C., Valente S., Degiovanni A., Gargiulo M., Capri M., Pasquinelli G., Adventitial microcirculation is a major target of SARS-CoV-2-mediated vascular inflammation. Biomolecules 11, 1063 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 204.Faa G., Gerosa C., Fanni D., Barcellona D., Cerrone G., Orru G., Scano A., Marongiu F., Suri J., Demontis R., Nioi M., D’Aloja E., La Nasa G., Saba L., Aortic vulnerability to COVID-19: Is the microvasculature of vasa vasorum a key factor? Eur. Rev. Med. Pharmacol. Sci. 25, 6439–6442 (2021). [DOI] [PubMed] [Google Scholar]
- 205.Shekhar H., Rowan J. S., Doyley M. M., Combining subharmonic and ultraharmonic modes for intravascular ultrasound imaging: A preliminary evaluation. Ultrasound Med. Biol. 43, 2725–2732 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 206.Yu F. T., Villanueva F. S., Chen X., Radial modulation contrast imaging using a 20-MHz single-element intravascular ultrasound catheter. IEEE Trans. Ultrason. Ferroelectr. Freq. Control 61, 779–791 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 207.Magnoni M., Coli S., Marrocco-Trischitta M. M., Melisurgo G., De Dominicis D., Cianflone D., Chiesa R., Feinstein S. B., Maseri A., Contrast-enhanced ultrasound imaging of periadventitial vasa vasorum in human carotid arteries. Eur. J. Echocardiogr. 10, 260–264 (2009). [DOI] [PubMed] [Google Scholar]
- 208.Chen Q., Yu J., Lukashova L., Latoche J. D., Zhu J., Lavery L., Verdelis K., Anderson C. J., Kim K., Validation of ultrasound super-resolution imaging of vasa vasorum in rabbit atherosclerotic plaques. IEEE Trans. Ultrason. Ferroelectr. Freq. Control 67, 1725–1729 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 209.You X., Huang P., Zhang C., Wang M., Zhang Y., Hong Y., Wei S., Liu C., Pu Z., Zhang J., Chen S., Relationship between enhanced intensity of contrast enhanced ultrasound and microvessel density of aortic atherosclerostic plaque in rabbit model. PLOS ONE 9, e92445 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 210.Moguillansky D., Leng X., Carson A., Lavery L., Schwartz A., Chen X., Villanueva F. S., Quantification of plaque neovascularization using contrast ultrasound: A histologic validation. Eur. Heart J. 32, 646–653 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 211.Schinkel A. F., Krueger C. G., Tellez A., Granada J. F., Reed J. D., Hall A., Zang W., Owens C., Kaluza G. L., Staub D., Coll B., Ten Cate F. J., Feinstein S. B., Contrast-enhanced ultrasound for imaging vasa vasorum: Comparison with histopathology in a swine model of atherosclerosis. Eur. J. Echocardiogr. 11, 659–664 (2010). [DOI] [PubMed] [Google Scholar]
- 212.Drinane M., Mollmark J., Zagorchev L., Moodie K., Sun B., Hall A., Shipman S., Morganelli P., Simons M., Mulligan-Kehoe M. J., The antiangiogenic activity of rPAI-1(23) inhibits vasa vasorum and growth of atherosclerotic plaque. Circ. Res. 104, 337–345 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 213.Stefanadis C., Toutouzas K., Tsiamis E., Vavuranakis M., Stefanadi E., Kipshidze N., First-in-man study with bevacizumab-eluting stent: A new approach for the inhibition of atheromatic plaque neovascularisation. EuroIntervention 3, 460–464 (2008). [DOI] [PubMed] [Google Scholar]