Abstract
Even more than 50 years after its initial description, bronchopulmonary dysplasia (BPD) remains one of the most important and lifelong sequelae following premature birth. Tremendous efforts have been undertaken since then to reduce this ever-increasing disease burden but a therapeutic breakthrough preventing BPD is still not in sight. The inflammatory response provoked in the immature lung is a key driver of distorted lung development and impacts the formation of alveolar, mesenchymal, and vascular structures during a particularly vulnerable time-period. During the last 5 years, new scientific insights have led to an improved pathomechanistic understanding of BPD origins and disease drivers. Within the framework of current scientific progress, concepts involving disruption of the balance of key inflammatory and lung growth promoting pathways by various stimuli, take center stage. Still today, the number of efficient therapeutics available to prevent BPD is limited to a few, well-established pharmacological interventions including postnatal corticosteroids, early caffeine administration, and vitamin A. Recent advances in the clinical care of infants in the neonatal intensive care unit (NICU) have led to improvements in survival without a consistent reduction in the incidence of BPD. Our update provides latest insights from both preclinical models and clinical cohort studies and describes novel approaches to prevent BPD.
Keywords: Chronic lung disease, Bronchopulmonary dysplasia, Preterm, Reactive oxygen species, Inflammation, Lung injury, Rodent, Therapeutic approach
Introduction
More than 50 years ago, bronchopulmonary dysplasia (BPD) was first described by Northway, Rosan, and Porter [1]. Even after more than five decades of scientific progress, the overall BPD disease burden is high in the newborn population but a dramatic shift in infants at high risk and BPD pathology occurred since the original publication. Although in the past, even comparatively mature preterm infants were vulnerable to the development of lung emphysema, fibrosis, and high mortality characteristic of the “old” BPD, the implementation of antenatal steroids and postnatal surfactant therapy in the 1980s and 1990s has led to both improved outcomes for late preterm infants and the increasing survival of infants born as early as 22–24 gestational age. This so-called new BPD is characterized by less severe tissue injury, but more or less distinct disturbance of further lung development in the late canalicular and saccular stage [2].
Large cohort studies have demonstrated that low gestational age at birth and low birthweight [3] constitute major determinants of the risk for BPD; however, genetic predisposition [4], including male sex and maternal disorders including pre-eclampsia/HELLP (hemolysis, elevated liver enzyme levels, low platelet count), intrauterine growth restriction [5], and nicotine consumption [6] also impact on the likelihood to develop BPD. Besides the need for mechanical ventilation and oxygen supplementation, intrauterine and postnatal infections nowadays constitute important contributors to BPD. The presence of a patent ductus arteriosus and NICU management variations including fluid and nutritional supply, antibiotic regimens, and measures to prevent postnatal infections and necrotizing enterocolitis (NEC) including the provision of human milk constitute further variables impacting on the risk for BPD [2, 7–9].
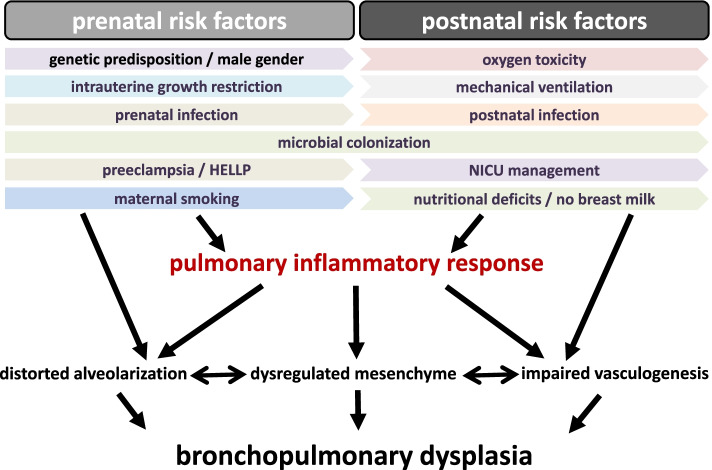
Nowadays, the gas exchange of most preterm infants can be stabilized after birth but most of these tiny infants experience a more or less dramatic deterioration of their lung function requiring escalation of respiratory support and oxygen fractions applied [10]. It is well accepted that the respiratory course is determined by the extent of the inflammatory response in the immature lung provoked or strengthened by the multiple pre- and postnatal risk factors and pro-inflammatory stimuli [10–12]. The ever-increasing number of publications on BPD reflects the need for further gain in knowledge [13]. It was already elaborated in the original description of BPD that all compartments of the lung, the epithelium, the endothelium, and the mesenchyme get affected during the evolution of BPD pathology. This leads to a reduced surface for gas exchange, rarefication of pulmonary vessels, and pathologic composition of the lung interstitium [1]. Therefore, addressing the BPD challenge requires a comprehensive pathomechanistic understanding presented within the second chapter of our review and needs to acknowledge the multifactorial origins of BPD summarized in Fig. 1.
Fig. 1.
Central pre- and postnatal risk factors for BPD development. The key contributors to the development of BPD are split into prenatal and postnatal risk factors (scheme developed from our previous review on the topic [12])
The pulmonary inflammatory response
Within the last two decades, the general concept of inflammation of the immature lung leading to BPD in preterm infants has not been changed (Fig. 2). Results obtained from various experimental models of BPD have demonstrated that infectious insults, oxygen toxicity, and mechanical ventilation cause the characteristic pathological features of BPD with distorted alveolar and vascular growth and unfavorable changes in the composition of the mesenchyme and mesenchymal cell transdifferentiation [14]. In rodents studied using a model of hyperoxia-induced BPD, the extent of lung developmental aberrations is determined by the fraction and duration of oxygen supplementation [15]. BPD pathology gets aggravated when hyperoxic episodes are combined with hypoxemic events that frequently occur in preterm infants and further boost reactive oxygen species production and subsequent inflammation [16].
Fig. 2.
The inflammatory response in the immature lung as key event leading to bronchopulmonary dysplasia. Mechanical ventilation, oxygen supply and infections induce a pro-inflammatory response in the immature lung. This pro-inflammatory overweight disrupts the balanced network of growth signal pathways and leads to characteristic features in the lung that affect the epithelium, the mesenchyme and the endothelium (scheme developed from our previous review on the topic [12])
Dysbalance of pro-inflammatory cytokines and of lung growth promoting factors
The inflammatory reaction is characterized by the excess production of pro-inflammatory cytokines including interleukin-1β (IL-1ß), interleukin-6 (IL-6), interleukin-8 (IL-8) and tumor necrosis factor-α (TNF-α), granulocyte colony-stimulating factor, macrophage inflammatory proteins, and monocyte chemoattractant proteins while anti-inflammatory cytokines like interleukin-10 get downregulated [12, 17]. Recent data from a study in very preterm infants indicate that the inflammatory response commences early after birth and precedes the deterioration of lung function [17]. For transforming growth factor-β (TGF-β), excess production was ascribed to pro-inflammatory activity and apoptosis induction but anti-inflammatory and immunosuppressive functions as well as growth promoting functions have also been confirmed [16, 18]. Further complexity arises when the inflammatory stimulus is not investigated within single hit models that do not reflect the clinical scenario. Either pretreatment with antenatal steroids or pre-exposure to ureaplasma species abrogated the intra-uterine inflammation induced by lipopolysaccharides [19, 20]. Therefore, the complexity of TGF-β signaling offers a prime example of how the role of a specific cytokine is not necessarily uniform and can be affected by both physiological and pathophysiological stimuli what will be detailed later in this section.
The overexpression of pro-inflammatory cytokines is counterbalanced by the suppression of growth promoting cytokines. Probably vascular endothelial growth factor A (VEGFA) constitutes the most prominent and best studied candidate for vascular development, while platelet-derived growth factor AA (PDGF-AA) was primarily related to mesenchymal stem cell (MSC) function and insulin like growth factor 1 (IGF-1) with alveolar epithelial and endothelial cells [21–23]. This cytokine and growth factor dysbalance is not restricted to the lung. Convincing evidence is available that describes a close correlation between pulmonary dysfunction and changes in systemic blood protein profile patterns [11].
Attraction of innate immune cells to the immature lung
Upregulation in the expression of pro-inflammatory mediators attracts alveolar macrophages and neutrophils that constitute the prevailing leukocyte fractions of the innate immune response. The pro-inflammatory cytokines in the lung induce a switch from the initial anti-inflammatory M2 to the inflammatory M1 polarization status. The resulting predominance of the inflammatory M1 polarization status perpetuates and augments the inflammatory response among others by the release of further pro-inflammatory cytokines [24]. Neutrophils have also been shown to exert lung injury through the release of multiple proteases including metalloproteinases and elastase [12]. Neutrophil extracellular trap formation is one recently discovered hallmark leading to lung injury [25]. Furthermore, prevention of neutrophil influx following hyperoxia reduced reactive oxygen species (ROS) levels in the newborn rat lung and the severity of lung injury [26]. Besides macrophages and neutrophils, mast cells and reactive T cells were ascribed contributing functions to BPD pathology and it will be important to dissect the interplay between the different pro-inflammatory cell populations [12]. Plenty of observational data are available from biosample studies in preterm infants that were mostly performed around the turn of the century. The results of these studies have consistently demonstrated an association between excessive pro-inflammatory responses and suppression of growth factor signaling pathways [27]. Consultation of the upcoming and latest datasets from preclinical rodent models helps to further clarify the contribution of specific cytokines and targets to BPD pathology.
Dysbalance of the cytokine signaling network in the immature lung
Major advances within the last 5 years have confirmed that precise and complex signaling crosstalk occurs between the three compartments of the lung during both physiological conditions and BPD development. Excessive activation of TGF-β signaling pathways leads to the suppression of PDGF receptor α mediated pulmonary myofibroblast function and VEGFA secretion. In this way, excessive inflammation is linked with dysregulation of vascular development [22]. This relationship can also be illustrated by the finding that disturbance in fibroblast growth factor 10 (FGF10) function is implicated in unfavorable alterations in alveolar epithelial cell properties that resulted in the reduction of type II cells in the lung and impaired production of surfactant proteins B and C [28]. The interconnection of FGF10 deficiency with improper lung vascular development was documented in the identical animal model with both rarefication of the number of blood vessels and abnormal muscularization noted. These findings are reflective of the pathological phenotype of infants with BPD associated pulmonary hypertension [29]. Furthermore, spatial and temporal regulations of growth signaling need to be considered when putting the focus on lung development in the late canalicular and saccular stage that is beyond the scope of our review. Further details and the available data on the further pathways including sonic hedgehog and Wnt signaling have been superbly detailed elsewhere [13].
Phenotype distortion of lung resident mesenchymal stem cells
Although the essential role of lung MSCs in physiological lung development was identified as early as the 1990s, the therapeutic potential of exogenously applied umbilical cord derived MSCs has emerged as an exciting area of focus in BPD research [30]. All the studies on primary MSC from preterm infants confirmed the tight association between distorted lung resident mesenchymal stem cell phenotype and BPD [31, 32]. Again, the TGF-β and nuclear factor kappa-light-chain-enhancer of activated B cells (NFκB) signaling pathways were identified as key drivers of MSC pathology [32, 33]. As demonstrated, redirection of MSC phenotype alterations is in principle feasible but awaits further detailing in therapeutic intervention studies in in vivo models [32]. More sophisticated strategies than simply inhibiting the excessive NFκB response will be described later in this section.
Dual function of inflammatory cytokine signaling pathways during lung development and injury
As stated above, more and more cytokines and pathways get discovered that both contribute to physiologic lung development but can aggravate lung injury when overstimulation occurs. Perhaps the longest known connection is that of TGF-β signaling. While Smad-3 knockout mice displayed a normal lung phenotype at birth, morphological changes characterized by enlarged terminal air spaces became apparent on postnatal day 7. In the term born mouse, this time point corresponds approximately to the early phase of alveolarization that begins between 34 and 36 weeks postmenstrual age (PMA) in humans [18, 34]. This is in line with the previous observation of dynamic regulation of TGF-β signaling in mice and men during lung development [35]. Excess in TGF-β in tracheal aspirates of ventilated preterm infants was associated with BPD and lung epithelial TGF-β1 overexpression led to arrested lung development in the rodent model [36, 37]. Further details were recently provided from mice with a mesenchyme-specific deletion of TGF-β1. Here, embryonic lung branching morphogenesis was disrupted resulting in lung hypoplasia [38]. Inversely, the hyperactivation of the connective tissue growth factor (CTGF)-β-catenin signaling pathway downstream of TGF-β1 by hyperoxia was associated with the characteristic features of BPD lungs. Inhibition of this axis by CTGF neutralizing antibodies or pharmacological inhibition of β-catenin both attenuated aberrations in alveolar and vascular lung development [39, 40].
Similarly, the disruption of NFκB signaling resulted in distortion of further alveolar and vascular lung development [41]. The detailed examination of NFκB action revealed that the inhibitor of nuclear factor kappa-B kinase beta (IKKβ) but not IKKα was the predominant regulator of angiogenic function and pulmonary angiogenesis [42]. In line, knockout mice for TNF-α displayed reduced NFκB activation when subjected to mechanical ventilation in moderate hyperoxia and more aggravated lung inflammation and apoptosis induction [18]. The essential role of NFκB to preserve the lung from injury was extended to the situation of lipopolysaccharide (LPS)-induced inflammation mimicking gram-negative infections. Here, inhibition of NFκB aggravated the suppression of alveolarization and pulmonary angiogenesis, a finding that was attributed to increased expression of macrophage inflammatory protein 2 (MIP-2). This mediator acted directly to inhibit pulmonary endothelial cell function, a step that initiated the aberration of normal lung development [43]. As for the hyperoxic injury, inhibition of IKKβ prevented the pro-inflammatory response of macrophages [44]. Further complexity on the topic arises from one recent publication on macrophage derived IL-6 signaling. Here, the loss of IL-6 was associated with better preserved alveolar epithelial type II cell survival and elastic fiber assembly while myofibroblast differentiation was reduced [45]. These findings are in contrast to those published by another group that reported a more pronounced inflammatory response and increased severity of lung injury in IL-6 knockout mice exposed to hyperoxia [46]. The results are in line with the knowledge on the diverse functions of IL-6 that can exert anti-inflammatory and reparative effects or pro-inflammatory responses depending on the signal transduction pathway [47]. This ambiguity on IL-6 accentuates the concerns that pro-inflammatory cytokines in general have divergent functions during lung injury. Further evaluation is urgently needed whether pro-inflammatory cytokine targeting can evolve as a novel safe therapeutic approach.
From all these studies, it comes clear that the pro-inflammatory cytokines that activate NFκB or TGF-β signaling contribute to proper lung development under physiologic conditions but aggravate lung injury under excess activation. The key event of MSC phenotype alterations in this context as the origin of aberrant further lung development comes more and more into the focus of researchers [32, 33]. The original concept to block pro-inflammatory activity to overcome the deleterious effects on lung development needs to be substituted by the more sophisticated approach to restitute the physiologic situation of a balanced signaling network where excess activation of any partner involved leads to dysregulation and aggravation of lung injury.
Strategies to promote lung development and to attenuate the pro-inflammatory response
The tremendous progress in preclinical knowledge and the pathomechanistic understanding of the origins of BPD contrasts the factum that no novel therapeutic was added to the short list of medications to prevent BPD since our first review on the topic five years ago [12]. In this section, we will provide a short overview on key advances during the last 5 years and on actually ongoing research directions. For the level of statistical significance and confidence intervals, we refer the reader to the original publications.
Therapeutic interventions of proven efficacy within clinical trials
Still today, the well-studied medical interventions are limited to postnatal corticosteroids and postnatal caffeine and vitamin A [16, 30]. The clinical trials on inhaled nitric oxide to prevent BPD more than 10 years ago were the last series testing a new medication to prevent BPD but failed to display a benefit on mortality, the pulmonary and neurodevelopmental outcome [48]. In addition, the combination of inhaled nitric oxide (iNO) together with repeated surfactant application did not demonstrate any benefit [49]. These completely negative results despite highly promising perspectives from preclinical studies might have discouraged researchers to pursue the direction toward new therapeutics. Results of a recent phase 2 randomized controlled trial designed to evaluate the potential of rhIGF-1/rhIGFBP3 administration to reduce retinopathy of prematurity (ROP) in preterm infants born before 28 weeks gestational age (GA) (n = 121) showed that although there was no significant reduction in the primary outcome, this intervention was associated with a significant reduction in the incidence of severe BPD [50]. These results are not surprising taking into account the observed reduction of IGF-1 levels in preterm infants after birth and during the first weeks of life and the beneficial effects of IGF-1 therapy in rodent studies on lung development [21, 51, 52]. Of course, this initial study needs confirmation in adequately powered randomized controlled trials and the mitogen functions of IGF-1 need critical monitoring as is currently intended within an open label controlled three-arm phase 2b study on safety, optimal dosing, and efficacy (NCT03253263).
The avenue to further targeted interventions
Although the evidence for safety and efficacy is currently lacking, the results of the IGF-1/IGFBP3 pilot trial might be useful in encouraging the pursuit of therapeutic strategies involving lung growth promoting cytokines rather than previous approaches that were focused on the use of anti-inflammatory agents. During the recent years, several further candidates have been evaluated in detail in preclinical models. One of the most promising candidates was VEGFA, as disruption of pulmonary vessel formation is a hallmark of BPD pathology that was ascribed to the suppression of VEGFA. The application of VEGFA improved lung structures following hyperoxia but was accompanied by increased vessel leakage and pulmonary edema in the acute phase of injury. These results must be seen in the context of the physiologic function of VEGFA in vascular development that is to instigate branching of blood vessels by increasing permeability. Therefore, this activity needs to be counterbalanced by appropriate angiopoietin signaling that safeguards vascular integrity [53]. Confirming this interrelation of action, simultaneous application of angiopoietin-1 by gene transfer prevented the vascular vessel leakage [54, 55]. Further concerns about the therapeutic potential of modulating the hypoxia induced factor (HIF)-1α-VEGFA axis arose, when mice with stable overexpression of HIF-1α-subunit in the distal epithelium were exposed to hyperoxic injury. The stable overexpression of HIF-1α increased the levels of VEGFA family members and angiopoietins but failed to improve lung structures and resulted in poorer lung function [56]. Further promising growth factor candidates are currently thoroughly investigated for their therapeutic potential to prevent or treat BPD. The therapeutic potential of PDGF receptor-α signaling was confirmed in haploinsufficient mice where intrapulmonary treatment with PDGF-AA rescued the more pronounced lung injury by mechanical ventilation and oxygen toxicity [22]. Another highly promising candidate constitutes FGF-10. Detailed studies in transgenic newborn documented the beneficial effects on the lung epithelium and vessel formation [28, 29]. Research on further candidates comprise epidermal growth factor (EGF), keratinocyte growth factor (KGF) and hepatocyte growth factor (HGF) but are still at the onset of a thorough evaluation. One so far mostly neglected need is to establish not only preventive strategies but to document the therapeutic potential to counteract the arising pathologies of BPD as most preterm infants are already exposed to lung injuries in utero or shortly after birth.
Overall, the track to growth factor treatment to prevent or treat BPD is still a long journey maybe with the exception of IGF-1. It needs to be established whether the selective intervention into one cytokine pathway can be as effective as the broadly acting corticosteroids that are still today the choice of therapy to abrogate the inflammatory response in the immature lung provoked by oxygen, mechanical ventilation, and infection. Overall, postnatal corticosteroid application > 7 days of life are highly effective and at least for dexamethasone a clear benefit to prevent BPD at 36 weeks corrected age has been demonstrated [57]. In contrast, early corticosteroid application prior to 7 days of life poses dramatic risks for neurodevelopmental impairment and cerebral palsy and should therefore not be used [58, 59]. Administration of caffeine within the first week of life is another intervention with documented benefit to reduce the BPD burden that is supported by large scale randomized controlled trial data [60]. Although the action of caffeine in the prevention of BPD has been largely ascribed by clinicians to occur through the stabilization of respiratory drive [61], data from preclinical studies has indicated that benefits may also be derived through the antioxidant and anti-inflammatory properties of this now commonly used medication [62, 63]. These data are not surprising taking into account the contribution of ROS induction by infection and oxygen therapy to the proinflammatory response in the lungs of preterm infants and BPD [64–66].
Infections, pathologic microbiota structures, and probiotics application
In the context of inflammation, nosocomial infections represent another entity that increase the risk for BPD and act via the identical pathomechanisms. On the other hand, exposure to the powerful anti-inflammatory and immunomodulatory properties of human milk constitutes a key non-pharmacological intervention suggested to reduce the incidence of BPD [67]. These data should be expected when the pathomechanisms of BPD are taken into consideration. One further dimension came into the focus of research during the recent years, the shape of the preterm’s microbiome. It is highly accepted that the provision of human milk shapes the bacterial milieu in the preterm infant toward Bifidobacteria and Lactobacillus species while the predominance of potentially pathogenic germs is impeded [9]. Therefore, human milk feedings may act to reduce the incidence of BPD by protecting infants from nosocomial infections and NEC. But even in the absence of infection events, the shift of the microbiota structures toward an anti-inflammatory milieu might contribute to the risk reduction by human milk. In line, antibiotic exposure with the selection of pathogenic bacterial species increases the risk of BPD in clinical studies [68, 69]. Recent data from a prospective randomized controlled trial specify the risk toward exposures longer than the first 2 days of life [70]. While germ-free mice were partially protected from injury caused by hyperoxia [71], exposure to ampicillin during the pre-natal and immediate post-natal period was found to be associated with increased severity of hyperoxia induced alveolar simplification and dysregulated vasculogenesis [72]. In contrast to the benefits for sepsis and necrotizing enterocolitis, probiotic bacteria administration did not result in any benefit for the lung so far although specific microbial compositions of tracheal aspirates were associated with the development of BPD independent of the occurrence of pneumonia [9, 73]. This discrepancy might be explained by the fact that probiotics fail to stabilize the microbial milieu in the upper airway that seems to be of greater relevance for the lung than that in the gut [7]. Although so far not marked by success, a healthy microbiome and modulation of the bacterial milieu in utero and in the preterm infant constitutes a highly promising approach taking into account the tremendous disease burden arising from prematurity until aging [74, 75]. Recent data indicate that the approach to the lung and the gut might need different strategies although both entities arise from the identical bud [7, 73, 75].
Maternal risk factors and BPD
Altogether, these data indicate that external factors impact on the inflammatory milieu and the risk of BPD. Therefore, the focus on the environmental contributors might be another strategy toward efficient risk reduction for BPD. While diabetes in pregnancy does not increase the risk for BPD, maternal nicotine exposure does [76, 77]. Pre-pregnancy no smoking prevention programs and the avoidance of exposure of the preterm infant to the substances of tobacco smoke are further suited strategies. As intensification of smoking prevention programs will not terminate the intrauterine exposition, prenatal therapeutic interventions can constitute an additional approach to reduce the disease burden of BPD. Here, results from randomized controlled trials with prophylactic vitamin C supplementation for pregnant smoking women revealed improved pulmonary function and decreased wheezing episodes in mostly term born infants [78, 79].
Although a completely different pathomechanism with primary action on vascular development via vascular growth inhibiting factors, the prevention of preeclampsia/HELLP or strategies to prevent the injury by soluble Fms-like thyrosinkinase-1 (sFlt-1) constitute another area of perspective [80].
Anti-inflammatory approaches with documented preclinical efficacy
Despite the long list of therapeutic strategies, that proved efficacious in rodent models but failed to translate into new efficient clinical therapeutics, it needs to be mentioned at this point that several anti-inflammatory strategies proved their potential as novel targeted therapies that deserve further evaluation within the preclinical setting [81]. Firstly, interleukin-1 receptor antagonists (IL-1Rα) reduced the overall pulmonary cellular inflammatory response and pro-inflammatory cytokine levels of IL-1β, IL-6, MIP-1α, MIP-1β, and MIP-2 [82]. Secondly, biochemical Ras-related C3 botulinum toxin substrate 1 (Rac-1) inhibition and the irreversible caspase-1 inhibitor Ac-YVAD-CMK both attenuated the inflammation-induced IL-1β release and inflammasome activity in the lung exposed to hyperoxia underlining the potential of such targeted approaches [83, 84]. Thirdly, targeting IL-6 release from inflammatory macrophages in the lung seems highly promising [45].
Targeting the pathologic processes downstream of the initiation of the inflammatory response constitutes another attractive approach toward prevention of lung injury. To give one example, elafin is the specific inhibitor of lung elastase that is a contributor to lung injury in the execution phase [85]. Either intrapulmonary elafin treatment in newborn mice or mice genetically modified to express elafin in their vascular endothelium were partially protected from the injurious insults and defective late lung development. Of notice, the pathologic activation of the pathways of TGF-β and NFκB, the influx of inflammatory cells, and apoptosis induction in the lung were partially prohibited [86, 87]. These data hint toward the potential success of strategies that aim at disrupting the vicious circle and self-reinforcement of inflammation. The continuation of studies on the therapeutic potential of elafin in newborn mice exposed to prolonged hyperoxia underlined its potential of repetitive application [88]. The results obtained from all experimental settings evaluating elafin are highly promising because they are consistent and reproducible. But VEGFA signaling and pulmonary vessel formation were not rescued by elafin treatment underlining the need for detailed studies on all three lung compartments when evaluating molecular mechanisms and targeted interventions [86].
Lastly, the upcoming research area on MSC to prevent BPD needs to be mentioned here. They are deemed to be particularly promising due to their broadly acting anti-inflammatory and growth promoting properties [30, 89]. Preclinical studies conducted using a variety of small rodent BPD models have demonstrated high therapeutic efficacy for MSC-based interventions even when treatment is applied after lung injury has occurred [24, 90]. MSC-derived exosomes (extracellular vesicles = EVs) are currently investigated as the next level of MSC research due to their lower immunogenicity and smaller size. These EVs contain all key anti-inflammatory and immunomodulatory mediators that explains their comparable efficacy to the direct MSC application [30]. One actual study in rodents further specified one main effect of EV action where EVs restored a non-inflammatory phenotype of lung myeloid cells by phenotypical and epigenetic reprogramming [91]. These data open the frame toward restoration of lung functional capacities even after intrauterine affection of the lung or immediate postnatal insults before a therapeutic intervention can be started. But as for many other therapies, the documentation of superiority in the clinical setting is still missing. Latest data from non-rodent models argue toward a careful consideration of aspects like cell preparation, timing, and dosing to reach finally a superiority in the clinical setting [30, 89]. The highly promising results obtained in rodents await confirmation in higher developed animal species like lambs and primates and clinical trials in preterm infants. So far, one phase 1 study intended for safety issues documented a benefit for BPD while the subsequent phase 2 study of the same group confirmed the safety but did not detect an effect on BPD [92, 93]. Future studies are ongoing that can advance the scientific knowledge of the therapeutic potential of MSC [94].
Outlook and research directions in ongoing clinical trials
Over the last 5 years, we have seen a tremendous gain in knowledge on the pathomechanisms of BPD and the complexity of its disease origins. But on a short-term perspective, we do not expect novel targeted interventions to revolutionize clinical therapy as many hurdles including the successful conductance of randomized controlled multicenter studies will take at least several years or even a decade before a benefit for the lung can be established. In the meantime, the optimization of established therapeutics including antenatal steroids to reduce the severity of respiratory distress after birth and studies on established therapeutics like vitamin A remain most promising within the pharmacologic approaches [95]. One just published smaller randomized controlled trial on oral vitamin A instead of the established approach of intramuscular application delivered disappointing results as high dose vitamin A did not reduce the overall BPD incidence and BPD severity distribution despite improving vitamin A blood levels [96, 97]. Therefore, still today as demonstrated during the last decade, optimization of ventilatory strategies, non-invasive surfactant application, and oxygen provision remain most promising to guide future treatment directions in the short run [66, 98, 99]. Besides the avoidance of mechanical ventilation, the stabilization of infants within the oxygen saturation targets is another promising strategy as hyperoxic and hypoxic episodes both aggravate lung injury. Several randomized controlled trials have been started into this direction. The OPTTIMMAL study aims to find the positive end-expiratory pressure (PEEP) level that is best suited to avoid intubation and mechanical ventilation within the first days of life when the immature lung is particularly vulnerable [100]. As another example, the Fi02C study investigates the automatically controlled titration of oxygen fractions by the respirator based on the actual oxygen saturation of the preterm infant during the total phase of respiratory support [101]. Furthermore, studies like the COSGOD III trial and the SafeBoosC III trial on tissue oxygenation of the brain during resuscitation in the delivery room and during respiratory support in the NICU respectively were designed to provide novel insights into the optimization of tissue oxygen saturation monitoring [102, 103]. In this context, the recent secondary analysis from the Canadian oxygen trial provided novel insights onto the association of the number and duration of hypoxemic episodes of preterm infants during their NICU stay and the later development of severe BPD [104]. Due to the observational character of the analysis, this study was not able to clarify the mechanistic link between hypoxemic episodes and BPD. The increased number of events can just reflect the more severe injury status of the lung in these infants. Here, the Pre-Vent study aims to precise the effects and mechanisms of ventilatory control to the adverse respiratory outcome [105].
From all these studies, we await novel insights into the origins and causes of BPD and which research directions are the most promising to further optimize the ventilatory strategies in preterm infants to reduce the burden of BPD.
Acknowledgments
We highly appreciate realization of figure layout by CE marketing & PR services, Germany. This work was supported by clinical research unit KFO309-2 (projects P6 and P7). Due to the limits in space, we had to select from the magnitude of important research works available on this topic to be included into our review.
Abbreviations
- BPD
Bronchopulmonary dysplasia
- CTGF
Connective tissue growth factor
- EGF
Epidermal growth factor
- EVs
Extracellular vesicles
- FGF10
Fibroblast growth factor 10
- GA
Gestational age
- HELLP
Hemolysis, Elevated Liver Enzyme Levels, Low Platelet Count
- HGF
Hepatocyte growth factor
- HIF
Hypoxia induced factor
- IGF-1
Insulin like growth factor 1
- IKKα
Inhibitor of nuclear factor kappa-B kinase alpha
- IKKβ
Inhibitor of nuclear factor kappa-B kinase beta
- IL-6
Interleukin-6
- IL-1Rα
Interleukin-1 receptor antagonists
- IL-1β
Interleukin-1β
- KGF
Keratinocyte growth factor
- LPS
Lipopolysaccharide
- MIP-1α
macrophage inflammatory protein 1α
- MIP-2
Macrophage inflammatory protein 2
- MIP-1β
Macrophage inflammatory protein 1β
- MSC
Mesenchymal stem cell
- NEC
Necrotizing enterocolitis
- NFκB
Nuclear factor kappa-light-chain-enhancer of activated B cells
- NICU
Neonatal intensive care unit
- iNO
Inhaled nitric oxide
- PDGF-AA
Platelet-derived growth factor AA
- PEEP
Positive end-expiratory pressure
- PMA
Postmenstrual age
- Rac-1
Ras-related C3 botulinum toxin substrate 1
- ROP
Retinopathy of prematurity
- ROS
Reactive oxygen species
- sFlt-1
Soluble Fms-like thyrosinkinase-1
- TGF-β
Transforming growth factor-β
- TNF-α
Tumor necrosis factor alpha
- VEGFA
Vascular endothelial growth factor A
Authors’ contributions
LH, TS, and HE drafted the manuscript and designed the figures. YD, LR, AS, BS, TL, AS, and SR provided valuable intellectual input. KPZ, JB, and SB edited and revised the manuscript. All authors read and approved the final version of the manuscript.
Funding
Open Access funding enabled and organized by Projekt DEAL.
Availability of data and materials
Not applicable.
Declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that there is no conflict of interest regarding the publication of this paper.
Footnotes
Lena Holzfurtner and Tayyab Shahzad both share first authorship.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Lena Holzfurtner and Tayyab Shahzad contributed equally to this work.
References
- 1.Northway WH, Rosan RC, Porter DY. Pulmonary disease following respirator therapy of hyaline-membrane disease. Bronchopulmonary dysplasia. N Engl J Med. 1967;276:357–368. doi: 10.1056/NEJM196702162760701. [DOI] [PubMed] [Google Scholar]
- 2.Thébaud B, Goss KN, Laughon M, et al. Bronchopulmonary dysplasia. Nat Rev Dis Primers. 2019;5:78. doi: 10.1038/s41572-019-0127-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Laughon MM, Langer JC, Bose CL, et al. Prediction of bronchopulmonary dysplasia by postnatal age in extremely premature infants. Am J Respir Crit Care Med. 2011;183:1715–1722. doi: 10.1164/rccm.201101-0055OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Bhandari V, Bizzarro MJ, Shetty A, et al. Familial and genetic susceptibility to major neonatal morbidities in preterm twins. Pediatrics. 2006;117:1901–1906. doi: 10.1542/peds.2005-1414. [DOI] [PubMed] [Google Scholar]
- 5.Pierro M, Villamor-Martinez E, van Westering-Kroon E et al (2021) Association of the dysfunctional placentation endotype of prematurity with bronchopulmonary dysplasia: a systematic review, meta-analysis and meta-regression. Thorax. 10.1136/thoraxjnl-2020-216485 [DOI] [PMC free article] [PubMed]
- 6.Morrow LA, Wagner BD, Ingram DA, et al. Antenatal determinants of bronchopulmonary dysplasia and late respiratory disease in preterm infants. Am J Respir Crit Care Med. 2017;196:364–374. doi: 10.1164/rccm.201612-2414OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Lauer T, Behnke J, Oehmke F et al (2020) Bacterial colonization within the first six weeks of life and pulmonary outcome in preterm infants <1000 g. J Clin Med 9. 10.3390/jcm9072240 [DOI] [PMC free article] [PubMed]
- 8.Thiess T, Lauer T, Woesler A, et al. Correlation of early nutritional supply and development of bronchopulmonary dysplasia in preterm infants <1,000 g. Front Pediatr. 2021;9:741365. doi: 10.3389/fped.2021.741365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Staude B, Oehmke F, Lauer T, et al. The microbiome and preterm birth: a change in paradigm with profound implications for pathophysiologic concepts and novel therapeutic strategies. Biomed Res Int. 2018;2018:7218187. doi: 10.1155/2018/7218187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Laughon M, Bose C, Allred EN, et al. Antecedents of chronic lung disease following three patterns of early respiratory disease in preterm infants. Arch Dis Child Fetal Neonatal Ed. 2011;96:F114–F120. doi: 10.1136/adc.2010.182865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Laughon M, Bose C, Allred EN, et al. Patterns of blood protein concentrations of ELGANs classified by three patterns of respiratory disease in the first 2 postnatal weeks. Pediatr Res. 2011;70:292–296. doi: 10.1203/PDR.0b013e3182274f35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Shahzad T, Radajewski S, Chao C-M, et al. Pathogenesis of bronchopulmonary dysplasia: when inflammation meets organ development. Mol Cell Pediatr. 2016;3:23. doi: 10.1186/s40348-016-0051-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Lignelli E, Palumbo F, Myti D, et al. Recent advances in our understanding of the mechanisms of lung alveolarization and bronchopulmonary dysplasia. Am J Phys Lung Cell Mol Phys. 2019;317:L832–L887. doi: 10.1152/ajplung.00369.2019. [DOI] [PubMed] [Google Scholar]
- 14.Torday JS, Torres E, Rehan VK. The role of fibroblast transdifferentiation in lung epithelial cell proliferation, differentiation, and repair in vitro. Pediatr Pathol Mol Med. 2003;22:189–207. doi: 10.1080/pdp.22.3.189.207. [DOI] [PubMed] [Google Scholar]
- 15.Nardiello C, Mižíková I, Silva DM, et al. Standardisation of oxygen exposure in the development of mouse models for bronchopulmonary dysplasia. Dis Model Mech. 2017;10:185–196. doi: 10.1242/dmm.027086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Choi Y, Rekers L, Dong Y et al (2021) Oxygen toxicity to the immature lung-part I: pathomechanistic understanding and preclinical perspectives. Int J Mol Sci 22. 10.3390/ijms222011006 [DOI] [PMC free article] [PubMed]
- 17.Leroy S, Caumette E, Waddington C, et al. A time-based analysis of inflammation in infants at risk of bronchopulmonary dysplasia. J Pediatr. 2018;192:60–65.e1. doi: 10.1016/j.jpeds.2017.09.011. [DOI] [PubMed] [Google Scholar]
- 18.Ehrhardt H, Pritzke T, Oak P, et al. Absence of TNF-α enhances inflammatory response in the newborn lung undergoing mechanical ventilation. Am J Phys Lung Cell Mol Phys. 2016;310:L909–L918. doi: 10.1152/ajplung.00367.2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Collins JJP, Kunzmann S, Kuypers E, et al. Antenatal glucocorticoids counteract LPS changes in TGF-β pathway and caveolin-1 in ovine fetal lung. Am J Phys Lung Cell Mol Phys. 2013;304:L438–L444. doi: 10.1152/ajplung.00251.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Collins JJP, Kallapur SG, Knox CL, et al. Repeated intrauterine exposures to inflammatory stimuli attenuated transforming growth factor-β signaling in the ovine fetal lung. Neonatology. 2013;104:49–55. doi: 10.1159/000350548. [DOI] [PubMed] [Google Scholar]
- 21.Seedorf G, Kim C, Wallace B, et al. rhIGF-1/BP3 preserves lung growth and prevents pulmonary hypertension in experimental bronchopulmonary dysplasia. Am J Respir Crit Care Med. 2020;201:1120–1134. doi: 10.1164/rccm.201910-1975OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Oak P, Pritzke T, Thiel I, et al. Attenuated PDGF signaling drives alveolar and microvascular defects in neonatal chronic lung disease. EMBO Mol Med. 2017;9:1504–1520. doi: 10.15252/emmm.20160730. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Hilgendorff A, Reiss I, Ehrhardt H, et al. Chronic lung disease in the preterm infant. Lessons learned from animal models. Am J Respir Cell Mol Biol. 2014;50:233–245. doi: 10.1165/rcmb.2013-0014TR. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Willis GR, Fernandez-Gonzalez A, Anastas J, et al. Mesenchymal stromal cell exosomes ameliorate experimental bronchopulmonary dysplasia and restore lung function through macrophage immunomodulation. Am J Respir Crit Care Med. 2018;197:104–116. doi: 10.1164/rccm.201705-0925OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Sun Y, Chen C, Zhang X, et al. Heparin improves alveolarization and vascular development in hyperoxia-induced bronchopulmonary dysplasia by inhibiting neutrophil extracellular traps. Biochem Biophys Res Commun. 2020;522:33–39. doi: 10.1016/j.bbrc.2019.11.041. [DOI] [PubMed] [Google Scholar]
- 26.Yi M, Jankov RP, Belcastro R, et al. Opposing effects of 60% oxygen and neutrophil influx on alveologenesis in the neonatal rat. Am J Respir Crit Care Med. 2004;170:1188–1196. doi: 10.1164/rccm.200402-215OC. [DOI] [PubMed] [Google Scholar]
- 27.Speer CP. Chorioamnionitis, postnatal factors and proinflammatory response in the pathogenetic sequence of bronchopulmonary dysplasia. Neonatology. 2009;95:353–361. doi: 10.1159/000209301. [DOI] [PubMed] [Google Scholar]
- 28.Chao C-M, Yahya F, Moiseenko A, et al. Fgf10 deficiency is causative for lethality in a mouse model of bronchopulmonary dysplasia. J Pathol. 2017;241:91–103. doi: 10.1002/path.4834. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Chao C-M, Moiseenko A, Kosanovic D, et al. Impact of Fgf10 deficiency on pulmonary vasculature formation in a mouse model of bronchopulmonary dysplasia. Hum Mol Genet. 2019;28:1429–1444. doi: 10.1093/hmg/ddy439. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Goetz MJ, Kremer S, Behnke J et al (2021) MSC based therapies to prevent or treat BPD—a narrative review on advances and ongoing challenges. Int J Mol Sci 22. 10.3390/ijms22031138 [DOI] [PMC free article] [PubMed]
- 31.Hennrick KT, Keeton AG, Nanua S, et al. Lung cells from neonates show a mesenchymal stem cell phenotype. Am J Respir Crit Care Med. 2007;175:1158–1164. doi: 10.1164/rccm.200607-941OC. [DOI] [PubMed] [Google Scholar]
- 32.Reicherzer T, Häffner S, Shahzad T, et al. Activation of the NF-κB pathway alters the phenotype of MSCs in the tracheal aspirates of preterm infants with severe BPD. Am J Phys Lung Cell Mol Phys. 2018;315:L87–L101. doi: 10.1152/ajplung.00505.2017. [DOI] [PubMed] [Google Scholar]
- 33.Popova AP, Bozyk PD, Goldsmith AM, et al. Autocrine production of TGF-beta1 promotes myofibroblastic differentiation of neonatal lung mesenchymal stem cells. Am J Phys Lung Cell Mol Phys. 2010;298:L735–L743. doi: 10.1152/ajplung.00347.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Chen H, Sun J, Buckley S, et al. Abnormal mouse lung alveolarization caused by Smad3 deficiency is a developmental antecedent of centrilobular emphysema. Am J Phys Lung Cell Mol Phys. 2005;288:L683–L691. doi: 10.1152/ajplung.00298.2004. [DOI] [PubMed] [Google Scholar]
- 35.Alejandre-Alcázar MA, Michiels-Corsten M, Vicencio AG, et al. TGF-beta signaling is dynamically regulated during the alveolarization of rodent and human lungs. Dev Dyn. 2008;237:259–269. doi: 10.1002/dvdy.21403. [DOI] [PubMed] [Google Scholar]
- 36.Zhou L, Dey CR, Wert SE, et al. Arrested lung morphogenesis in transgenic mice bearing an SP-C-TGF-beta 1 chimeric gene. Dev Biol. 1996;175:227–238. doi: 10.1006/dbio.1996.0110. [DOI] [PubMed] [Google Scholar]
- 37.Kotecha S, Wangoo A, Silverman M, et al. Increase in the concentration of transforming growth factor beta-1 in bronchoalveolar lavage fluid before development of chronic lung disease of prematurity. J Pediatr. 1996;128:464–469. doi: 10.1016/s0022-3476(96)70355-4. [DOI] [PubMed] [Google Scholar]
- 38.Noe N, Shim A, Millette K, et al. Mesenchyme-specific deletion of Tgf-β1 in the embryonic lung disrupts branching morphogenesis and induces lung hypoplasia. Lab Investig. 2019;99:1363–1375. doi: 10.1038/s41374-019-0256-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Rong M, Chen S, Zambrano R, et al. Inhibition of β-catenin signaling protects against CTGF-induced alveolar and vascular pathology in neonatal mouse lung. Pediatr Res. 2016;80:136–144. doi: 10.1038/pr.2016.52. [DOI] [PubMed] [Google Scholar]
- 40.Alapati D, Rong M, Chen S, et al. Connective tissue growth factor antibody therapy attenuates hyperoxia-induced lung injury in neonatal rats. Am J Respir Cell Mol Biol. 2011;45:1169–1177. doi: 10.1165/rcmb.2011-0023OC. [DOI] [PubMed] [Google Scholar]
- 41.Iosef C, Alastalo T-P, Hou Y, et al. Inhibiting NF-κB in the developing lung disrupts angiogenesis and alveolarization. Am J Phys Lung Cell Mol Phys. 2012;302:L1023–L1036. doi: 10.1152/ajplung.00230.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Iosef C, Liu M, Ying L, et al. Distinct roles for IκB kinases alpha and beta in regulating pulmonary endothelial angiogenic function during late lung development. J Cell Mol Med. 2018;22:4410–4422. doi: 10.1111/jcmm.13741. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Hou Y, Liu M, Husted C, et al. Activation of the nuclear factor-κB pathway during postnatal lung inflammation preserves alveolarization by suppressing macrophage inflammatory protein-2. Am J Phys Lung Cell Mol Phys. 2015;309:L593–L604. doi: 10.1152/ajplung.00029.2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.McKenna S, Butler B, Jatana L, et al. Inhibition of IκBβ/NFκB signaling prevents LPS-induced IL1β expression without increasing apoptosis in the developing mouse lung. Pediatr Res. 2017;82:1064–1072. doi: 10.1038/pr.2017.182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Hirani D, Alvira CM, Danopoulos S et al (2021) Macrophage-derived IL-6 trans-signaling as a novel target in the pathogenesis of bronchopulmonary dysplasia. Eur Respir J. 10.1183/13993003.02248-2020 [DOI] [PMC free article] [PubMed]
- 46.Li H, Wang G, Lin S, et al. Loss of interleukin-6 enhances the inflammatory response associated with hyperoxia-induced lung injury in neonatal mice. Exp Ther Med. 2019;17:3101–3107. doi: 10.3892/etm.2019.7315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Scheller J, Chalaris A, Schmidt-Arras D, et al. The pro- and anti-inflammatory properties of the cytokine interleukin-6. Biochim Biophys Acta. 2011;1813:878–888. doi: 10.1016/j.bbamcr.2011.01.034. [DOI] [PubMed] [Google Scholar]
- 48.Barrington KJ, Finer N, Pennaforte T. Inhaled nitric oxide for respiratory failure in preterm infants. Cochrane Database Syst Rev. 2017;1:CD000509. doi: 10.1002/14651858.CD000509.pub5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Ballard RA, Keller RL, Black DM, et al. Randomized trial of late surfactant treatment in ventilated preterm infants receiving inhaled nitric oxide. J Pediatr. 2016;168:23–29.e4. doi: 10.1016/j.jpeds.2015.09.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Ley D, Hallberg B, Hansen-Pupp I, et al. rhIGF-1/rhIGFBP-3 in preterm infants: a phase 2 randomized controlled trial. J Pediatr. 2019;206:56–65.e8. doi: 10.1016/j.jpeds.2018.10.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Löfqvist C, Hellgren G, Niklasson A, et al. Low postnatal serum IGF-I levels are associated with bronchopulmonary dysplasia (BPD) Acta Paediatr. 2012;101:1211–1216. doi: 10.1111/j.1651-2227.2012.02826.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Beardsall K, Vanhaesebrouck S, Frystyk J, et al. Relationship between insulin-like growth factor I levels, early insulin treatment, and clinical outcomes of very low birth weight infants. J Pediatr. 2014;164:1038–1044.e1. doi: 10.1016/j.jpeds.2013.12.046. [DOI] [PubMed] [Google Scholar]
- 53.Hato T, Kimura Y, Morisada T, et al. Angiopoietins contribute to lung development by regulating pulmonary vascular network formation. Biochem Biophys Res Commun. 2009;381:218–223. doi: 10.1016/j.bbrc.2009.02.030. [DOI] [PubMed] [Google Scholar]
- 54.Thébaud B, Ladha F, Michelakis ED, et al. Vascular endothelial growth factor gene therapy increases survival, promotes lung angiogenesis, and prevents alveolar damage in hyperoxia-induced lung injury: evidence that angiogenesis participates in alveolarization. Circulation. 2005;112:2477–2486. doi: 10.1161/CIRCULATIONAHA.105.541524. [DOI] [PubMed] [Google Scholar]
- 55.Kunig AM, Balasubramaniam V, Markham NE, et al. Recombinant human VEGF treatment transiently increases lung edema but enhances lung structure after neonatal hyperoxia. Am J Phys Lung Cell Mol Phys. 2006;291:L1068–L1078. doi: 10.1152/ajplung.00093.2006. [DOI] [PubMed] [Google Scholar]
- 56.Tibboel J, Groenman FA, Selvaratnam J, et al. Hypoxia-inducible factor-1 stimulates postnatal lung development but does not prevent O2-induced alveolar injury. Am J Respir Cell Mol Biol. 2015;52:448–458. doi: 10.1165/rcmb.2014-0037OC. [DOI] [PubMed] [Google Scholar]
- 57.Doyle LW, Cheong JL, Hay S, et al. Late (≥ 7 days) systemic postnatal corticosteroids for prevention of bronchopulmonary dysplasia in preterm infants. Cochrane Database Syst Rev. 2021;11:CD001145. doi: 10.1002/14651858.CD001145.pub5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Doyle LW, Cheong JL, Hay S, et al. Early (< 7 days) systemic postnatal corticosteroids for prevention of bronchopulmonary dysplasia in preterm infants. Cochrane Database Syst Rev. 2021;10:CD001146. doi: 10.1002/14651858.CD001146.pub6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Barrington KJ. The adverse neuro-developmental effects of postnatal steroids in the preterm infant: a systematic review of RCTs. BMC Pediatr. 2001;1:1. doi: 10.1186/1471-2431-1-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Schmidt B, Roberts RS, Davis P, et al. Caffeine therapy for apnea of prematurity. N Engl J Med. 2006;354:2112–2121. doi: 10.1056/NEJMoa054065. [DOI] [PubMed] [Google Scholar]
- 61.Henderson-Smart DJ, de Paoli AG (2010) Methylxanthine treatment for apnoea in preterm infants. Cochrane Database Syst Rev:CD000140. 10.1002/14651858.CD000140.pub2 [DOI] [PMC free article] [PubMed]
- 62.Endesfelder S, Strauß E, Scheuer T, et al. Antioxidative effects of caffeine in a hyperoxia-based rat model of bronchopulmonary dysplasia. Respir Res. 2019;20:88. doi: 10.1186/s12931-019-1063-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Weichelt U, Cay R, Schmitz T, et al. Prevention of hyperoxia-mediated pulmonary inflammation in neonatal rats by caffeine. Eur Respir J. 2013;41:966–973. doi: 10.1183/09031936.00012412. [DOI] [PubMed] [Google Scholar]
- 64.Kim SK, Romero R, Chaiworapongsa T, et al. Evidence of changes in the immunophenotype and metabolic characteristics (intracellular reactive oxygen radicals) of fetal, but not maternal, monocytes and granulocytes in the fetal inflammatory response syndrome. J Perinat Med. 2009;37:543–552. doi: 10.1515/JPM.2009.106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Banks BA, Ischiropoulos H, McClelland M, et al. Plasma 3-nitrotyrosine is elevated in premature infants who develop bronchopulmonary dysplasia. Pediatrics. 1998;101:870–874. doi: 10.1542/peds.101.5.870. [DOI] [PubMed] [Google Scholar]
- 66.Behnke J, Dippel CM, Choi Y et al (2021) Oxygen toxicity to the immature lung-part II: the unmet clinical need for causal therapy. Int J Mol Sci 22. 10.3390/ijms221910694 [DOI] [PMC free article] [PubMed]
- 67.Huang J, Zhang L, Tang J, et al. Human milk as a protective factor for bronchopulmonary dysplasia: a systematic review and meta-analysis. Arch Dis Child Fetal Neonatal Ed. 2019;104:F128–F136. doi: 10.1136/archdischild-2017-314205. [DOI] [PubMed] [Google Scholar]
- 68.Fajardo C, Alshaikh B, Harabor A. Prolonged use of antibiotics after birth is associated with increased morbidity in preterm infants with negative cultures. J Matern Fetal Neonatal Med. 2019;32:4060–4066. doi: 10.1080/14767058.2018.1481042. [DOI] [PubMed] [Google Scholar]
- 69.Cantey JB, Huffman LW, Subramanian A, et al. Antibiotic exposure and risk for death or bronchopulmonary dysplasia in very low birth weight infants. J Pediatr. 2017;181:289–293.e1. doi: 10.1016/j.jpeds.2016.11.002. [DOI] [PubMed] [Google Scholar]
- 70.Kim CS, Grady N, Derrick M, et al. Effect of antibiotic use within first 48 hours of life on the preterm infant microbiome: a randomized clinical trial. JAMA Pediatr. 2021;175:303–305. doi: 10.1001/jamapediatrics.2020.4916. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Dolma K, Freeman AE, Rezonzew G, et al. Effects of hyperoxia on alveolar and pulmonary vascular development in germ-free mice. Am J Phys Lung Cell Mol Phys. 2020;318:L421–L428. doi: 10.1152/ajplung.00316.2019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Willis KA, Siefker DT, Aziz MM, et al. Perinatal maternal antibiotic exposure augments lung injury in offspring in experimental bronchopulmonary dysplasia. Am J Phys Lung Cell Mol Phys. 2020;318:L407–L418. doi: 10.1152/ajplung.00561.2018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Villamor-Martínez E, Pierro M, Cavallaro G et al (2017) Probiotic supplementation in preterm infants does not affect the risk of bronchopulmonary dysplasia: a meta-analysis of randomized controlled trials. Nutrients 9. 10.3390/nu9111197 [DOI] [PMC free article] [PubMed]
- 74.Vuong HE, Pronovost GN, Williams DW, et al. The maternal microbiome modulates fetal neurodevelopment in mice. Nature. 2020;586:281–286. doi: 10.1038/s41586-020-2745-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Dermyshi E, Wang Y, Yan C, et al. The "golden age" of probiotics: a systematic review and meta-analysis of randomized and observational studies in preterm infants. Neonatology. 2017;112:9–23. doi: 10.1159/000454668. [DOI] [PubMed] [Google Scholar]
- 76.González-Luis GE, van Westering-Kroon E, Villamor-Martinez E, et al. Tobacco Smoking During Pregnancy Is Associated With Increased Risk of Moderate/Severe Bronchopulmonary Dysplasia: A Systematic Review and Meta-Analysis. Front Pediatr. 2020;8:160. doi: 10.3389/fped.2020.00160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Persson M, Shah PS, Rusconi F, et al. Association of maternal diabetes with neonatal outcomes of very preterm and very low-birth-weight infants: an international cohort study. JAMA Pediatr. 2018;172:867–875. doi: 10.1001/jamapediatrics.2018.1811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.McEvoy CT, Shorey-Kendrick LE, Milner K et al (2020) Vitamin C to pregnant smokers persistently improves infant airway function to 12 months of age: a randomised trial. Eur Respir J. 10.1183/13993003.02208-2019 [DOI] [PMC free article] [PubMed]
- 79.McEvoy CT, Schilling D, Clay N, et al. Vitamin C supplementation for pregnant smoking women and pulmonary function in their newborn infants: a randomized clinical trial. JAMA. 2014;311:2074–2082. doi: 10.1001/jama.2014.5217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Wallace B, Peisl A, Seedorf G, et al. Anti-sFlt-1 Therapy preserves lung alveolar and vascular growth in antenatal models of bronchopulmonary dysplasia. Am J Respir Crit Care Med. 2018;197:776–787. doi: 10.1164/rccm.201707-1371OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Morty RE. Using Experimental Models to Identify Pathogenic Pathways and Putative Disease Management Targets in Bronchopulmonary Dysplasia. Neonatology. 2020;117:233–239. doi: 10.1159/000506989. [DOI] [PubMed] [Google Scholar]
- 82.Nold MF, Mangan NE, Rudloff I, et al. Interleukin-1 receptor antagonist prevents murine bronchopulmonary dysplasia induced by perinatal inflammation and hyperoxia. Proc Natl Acad Sci U S A. 2013;110:14384–14389. doi: 10.1073/pnas.1306859110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Dapaah-Siakwan F, Zambrano R, Luo S, et al. Caspase-1 inhibition attenuates hyperoxia-induced lung and brain injury in neonatal mice. Am J Respir Cell Mol Biol. 2019;61:341–354. doi: 10.1165/rcmb.2018-0192OC. [DOI] [PubMed] [Google Scholar]
- 84.Hummler JK, Dapaah-Siakwan F, Vaidya R, et al. Inhibition of Rac1 signaling downregulates inflammasome activation and attenuates lung injury in neonatal rats Exposed to Hyperoxia. Neonatology. 2017;111:280–288. doi: 10.1159/000450918. [DOI] [PubMed] [Google Scholar]
- 85.Watterberg KL, Carmichael DF, Gerdes JS, et al. Secretory leukocyte protease inhibitor and lung inflammation in developing bronchopulmonary dysplasia. J Pediatr. 1994;125:264–269. doi: 10.1016/S0022-3476(94)70209-8. [DOI] [PubMed] [Google Scholar]
- 86.Hilgendorff A, Parai K, Ertsey R, et al. Neonatal mice genetically modified to express the elastase inhibitor elafin are protected against the adverse effects of mechanical ventilation on lung growth. Am J Phys Lung Cell Mol Phys. 2012;303:L215–L227. doi: 10.1152/ajplung.00405.201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Hilgendorff A, Parai K, Ertsey R, et al. Inhibiting lung elastase activity enables lung growth in mechanically ventilated newborn mice. Am J Respir Crit Care Med. 2011;184:537–546. doi: 10.1164/rccm.201012-2010OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Han W, Li X, Zhang H, et al. Recombinant human elafin promotes alveologenesis in newborn mice exposed to chronic hyperoxia. Int J Biochem Cell Biol. 2017;92:173–182. doi: 10.1016/j.biocel.2017.08.004. [DOI] [PubMed] [Google Scholar]
- 89.Augustine S, Cheng W, Avey MT, et al. Are all stem cells equal? Systematic review, evidence map, and meta-analyses of preclinical stem cell-based therapies for bronchopulmonary dysplasia. Stem Cells Transl Med. 2020;9:158–168. doi: 10.1002/sctm.19-0193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.O'Reilly M, Möbius MA, Vadivel A, et al. Late rescue therapy with cord-derived mesenchymal stromal cells for established lung injury in experimental bronchopulmonary dysplasia. Stem Cells Dev. 2020;29:364–371. doi: 10.1089/scd.2019.0116. [DOI] [PubMed] [Google Scholar]
- 91.Willis GR, Reis M, Gheinani AH, et al. Extracellular vesicles protect the neonatal lung from hyperoxic injury through the epigenetic and transcriptomic reprogramming of myeloid cells. Am J Respir Crit Care Med. 2021;204:1418–1432. doi: 10.1164/rccm.202102-0329OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Ahn SY, Chang YS, Lee MH, et al. Stem cells for bronchopulmonary dysplasia in preterm infants: A randomized controlled phase II trial. Stem Cells Transl Med. 2021;10:1129–1137. doi: 10.1002/sctm.20-0330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Chang YS, Ahn SY, Yoo HS, et al. Mesenchymal stem cells for bronchopulmonary dysplasia: phase 1 dose-escalation clinical trial. J Pediatr. 2014;164:966–972.e6. doi: 10.1016/j.jpeds.2013.12.011. [DOI] [PubMed] [Google Scholar]
- 94.Pierro M, Thébaud B, Soll R. Mesenchymal stem cells for the prevention and treatment of bronchopulmonary dysplasia in preterm infants. Cochrane Database Syst Rev. 2017;11:CD011932. doi: 10.1002/14651858.CD011932.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Meyer S, Gortner L. Early postnatal additional high-dose oral vitamin A supplementation versus placebo for 28 days for preventing bronchopulmonary dysplasia or death in extremely low birth weight infants. Neonatology. 2014;105:182–188. doi: 10.1159/000357212. [DOI] [PubMed] [Google Scholar]
- 96.Tyson JE, Wright LL, Oh W, et al. Vitamin A supplementation for extremely-low-birth-weight infants. National Institute of Child Health and Human Development Neonatal Research Network. N Engl J Med. 1999;340:1962–1968. doi: 10.1056/NEJM199906243402505. [DOI] [PubMed] [Google Scholar]
- 97.Rakshasbhuvankar AA, Simmer K, Patole SK et al (2021) Enteral vitamin A for reducing severity of bronchopulmonary dysplasia: a randomized trial. Pediatrics 147. 10.1542/peds.2020-009985 [DOI] [PubMed]
- 98.Göpel W, Kribs A, Härtel C, et al. Less invasive surfactant administration is associated with improved pulmonary outcomes in spontaneously breathing preterm infants. Acta Paediatr. 2015;104:241–246. doi: 10.1111/apa.12883. [DOI] [PubMed] [Google Scholar]
- 99.Isayama T, Iwami H, McDonald S, et al. Association of noninvasive ventilation strategies with mortality and bronchopulmonary dysplasia among preterm infants: a systematic review and meta-analysis. JAMA. 2016;316:611–624. doi: 10.1001/jama.2016.10708. [DOI] [PubMed] [Google Scholar]
- 100.Waitz M, Engel C, Schloesser R, et al. Application of two different nasal CPAP levels for the treatment of respiratory distress syndrome in preterm infants-"The OPTTIMMAL-Trial"-Optimizing PEEP To The IMMAture Lungs: study protocol of a randomized controlled trial. Trials. 2020;21:822. doi: 10.1186/s13063-020-04660-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Maiwald CA, Niemarkt HJ, Poets CF, et al. Effects of closed-loop automatic control of the inspiratory fraction of oxygen (FiO2-C) on outcome of extremely preterm infants - study protocol of a randomized controlled parallel group multicenter trial for safety and efficacy. BMC Pediatr. 2019;19:363. doi: 10.1186/s12887-019-1735-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Pichler G, Baumgartner S, Biermayr M, et al. Cerebral regional tissue oxygen saturation to guide oxygen delivery in preterm neonates during immediate transition after birth (COSGOD III): an investigator-initiated, randomized, multi-center, multi-national, clinical trial on additional cerebral tissue oxygen saturation monitoring combined with defined treatment guidelines versus standard monitoring and treatment as usual in premature infants during immediate transition: study protocol for a randomized controlled trial. Trials. 2019;20:178. doi: 10.1186/s13063-019-3258-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Hansen ML, Pellicer A, Gluud C, et al. Cerebral near-infrared spectroscopy monitoring versus treatment as usual for extremely preterm infants: a protocol for the SafeBoosC randomised clinical phase III trial. Trials. 2019;20:811. doi: 10.1186/s13063-019-3955-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Jensen EA, Whyte RK, Schmidt B, et al. Association between Intermittent hypoxemia and severe bronchopulmonary dysplasia in preterm infants. Am J Respir Crit Care Med. 2021;204:1192–1199. doi: 10.1164/rccm.202105-1150OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Dennery PA, Di Fiore JM, Ambalavanan N, et al. Pre-Vent: the prematurity-related ventilatory control study. Pediatr Res. 2019;85:769–776. doi: 10.1038/s41390-019-0317-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not applicable.