Abstract
Alopecia areata (AA) is an autoimmune disease characterized by nonscarring hair loss. As a clinically heterogeneous disease, various classification systems have evolved for defining its severity. In this high-level review of the literature, we discuss the traditional classification systems for AA severity and their strengths and weaknesses. Most recent classifications have focused on the extent of scalp hair loss as a defining feature, but additional clinical aspects of the disease, including location, pattern, and duration of hair loss as well as impact on the patient’s quality of life, are also relevant. These various components have typically been used unidimensionally to classify patients. We propose a multidimensional framework to define AA severity that incorporates multiple patient- and illness-related domains. Using such a framework, dermatologists may better assess the severity of the disease for the individual patient beyond the extent of hair loss.
Keywords: Alopecia areata, Alopecia totalis, Alopecia universalis, Disease severity, Classification
Key Summary Points
| Why carry out this study? |
| Alopecia areata (AA) is an autoimmune, non-scarring hair loss disease with varying degrees of hair loss. |
| Current classification systems for defining AA severity are suboptimal and have limited utility. |
| What does this study add? |
| We propose a multidimensional framework that goes beyond the unidimensional measurement of scalp hair loss. |
| Dermatologists may find the framework helpful for understanding the holistic burden of AA and how to define severity of the disease. |
Introduction
Alopecia areata (AA) is an autoimmune disease characterized by nonscarring hair loss [1]. The typical presentation of AA is one or more coin-shaped or ovoid patches of alopecia, but there may be total scalp hair loss or total body hair loss [1]. This high-level review aims to highlight the traditional classification systems for AA severity, evaluate their strengths and weaknesses, and provide a possible framework that will improve diagnostic ascertainment of disease severity.
This article is based on previously conducted studies and does not contain any new studies with human participants or animals performed by any of the authors.
Diagnostic versus Classification Criteria
Although the terms diagnosis and classification are often used interchangeably, they differ in their objectives. Diagnostic criteria broadly define the disease to ensure that less common variants are appropriately identified, and criteria may be unrelated to etiology [2]. Classification criteria identify relatively homogeneous populations for the purpose of measurement against an external criterion. When a disease is homogeneous, diagnostic and classification criteria can be synchronous and interchangeable; however, heterogeneous disease can lead to multiple methods of classification [2]. In the context of AA, the heterogeneity of its clinical presentation has led to a number of different approaches for classifying patients and defining disease severity.
Early Classification Systems (Pre-2000)
In 1902, Besnier classified AA into three types based on pattern of hair loss and inflammation: AA vulgaris, AA decalvans, and AA ophiasis [3]. However, Ikeda (1965) proposed that this system was inadequate because it did not account for the course of the illness [4]. Subsequently, she reviewed the clinical history and course of 1989 patients who were treated within the skin clinic at Kyoto University over an 18-year period [4]. Using the course (pattern and persistence over time) as the external criterion, she based groupings primarily on duration of episode and onset of the disease. Comparing these groupings, she then observed differences in comorbidities to develop a four-category system: AA common (single patches that spontaneously regrow), AA prehypertensive (recurrent small patches and predisposition for hypertension), AA autoimmune (persistent alopecia with associated autoimmune conditions), and AA atopic (persistent alopecia with atopic comorbidity) [4].
An attempt to replicate these groupings was undertaken by Sharma et al. [5] through analyses of 356 patients who were grouped according to age and chronicity. Although their recommendation was to combine the AA prehypertensive type with the AA common type, they recognized that the overall validity of the groupings was poor, given that patients presented with overlapping features or that the groupings excluded consideration of other potentially relevant prognostic factors, such as family history [5].
Current Classifications (Post-2000)
Severity of Scalp Hair Loss
In more recent years, disease severity in AA has been anchored in the extent of total scalp hair loss. The Severity of Alopecia Tool (SALT) was developed to standardize the quantification of hair loss across the different quadrants of the head [6], and is commonly used for quantifying amount of scalp hair loss in clinical trials. In its initial development, severity was classified as S0 = no hair loss; S1 = 1–24% hair loss; S2 = 25–49% hair loss; S3 = 50–74% hair loss; S4 = 75–99% hair loss; and S5 = 100% hair loss. The category S4 was further subdivided into S4a = 75–95% hair loss and S4b = 96–99% hair loss, with the rationale that S4b has prognostically lower probability for regrowth than S4a. The SALT II, based on the original SALT, delineates scalp surface area into 1% segments, allowing for more refined assessment for smaller patches of alopecia, and it is utilized for calculating the ALODEX score [7]. Another variation of the SALT incorporates disease activity by quantifying density and shedding. In the Alopecia Areata Progression Index, the quantitative assessment of hair loss via the SALT is paired with indices of disease activity (hair pulling test and trichoscopy) to obtain a score that reflects activity, density, and extent of loss [8].
A limitation of the SALT is that the label for each category is ordinal without interpretation. For example, S4a of 75–95% is less than S4b of 96–99% hair loss, but both categories lack a specific descriptor; that is, should these groups be considered moderate-to-severe versus severe OR severe versus very severe OR very severe versus extreme? Without having interpretive labels, the SALT S categories do not establish a common lexicon for the severity spectrum across the patient population.
Recently, an Investigator Global Assessment tool was developed to describe the severity spectrum for scalp hair loss [9]. Using both patient and physician interviews, agreement was reached across these stakeholders that an absolute SALT score ≤ 20, that is, less than or equal to 20% scalp hair loss, should be the therapeutic goal. There was also alignment on how SALT categories should be described: no hair loss = 0%; limited = 1–20%; moderate = 21–49%; severe = 50–94%; and very severe = 95–100% [9]. While these interpretative bands met psychometric concordance as clinically meaningful gradations in severity, these bands do not correspond exactly with the S bands as established in the original SALT development.
Amount of scalp hair loss often drives treatment decisions. A recent article proposed different interventions based on SALT score: SALT scores of 0–30%, 31–50%, and > 50% [10]. In particular, topical therapies and intralesional corticosteroids are favored for the lower thresholds, in part because intralesional corticosteroids are impracticable for scalp hair loss ≥ 50% [11]. Extent of loss of > 50% indicates the use of alternative therapies, such as contact immunotherapy or systemic intervention. Despite the usefulness of total scalp hair loss as a guide for treatment selection, the reliance on SALT score may be too narrow as the only criteria for defining severity of the disease because other signs and symptoms, including those observed in hair pull test or trichoscopy, may also impact the patient and/or have prognostic or therapeutic value.
Factors beyond Scalp Hair Loss
Besides severity of scalp hair loss, other clinical features have been incorporated into various classification systems. These factors include but are not limited to (1) location of hair loss, (2) pattern of hair loss, including body and nail involvement, and (3) duration of hair loss.
Location and Pattern of Hair Loss
Alopecia areata has been broadly subtyped as patchy AA, alopecia totalis (AT, total loss of scalp hair), and alopecia universalis (AU, total loss of body hair) [1]. Even within these categories, there may be disagreement about these definitions. For example, AU most often refers to total body hair loss but is sometimes applied to indicate total scalp hair loss plus some involvement of body hair loss. Furthermore, the amount of residual scalp hair that precludes a designation of AT or AU has not been clearly established; e.g., is AT only represented by SALT 100 or should patients with SALT 96–99 also be considered AT [1]? In addition, patients also describe eyebrow and/or eyelash hair loss and, for men, beard loss, as having their own unique physical and psychosocial consequences that make these areas an important part of disease severity assessment [12].
Complementing location of hair loss as a categorical indicator, AA subtypes have also been classified by pattern of hair loss. While patchy AA is most common, AA ophiasis describes a band-like pattern of hair loss, along the border of the temporal and occipital skull. The inverse pattern is AA sisaipho, which presents as significant hair loss except around the periphery of the scalp. While ophiasis pattern is often considered to be refractory to treatment, the usefulness of classification based on these patterns for clinical management has not been established [1].
These classification systems have been primarily unidimensional, which may be insufficient as classification criteria. One proposal has been to combine amount of hair loss, location of hair loss, and involvement of nails [6]. In this system, the amount of hair loss is assessed within the SALT categorical S groups. A notation regarding body involvement is added, in which B0 = no body hair loss, B1 = some body hair loss, and B2 = total body hair loss. A third code is then added to represent the degree of nail involvement [N0 = no involvement of nails; N1 = some of 20 nails show pitting or nail dystrophy (trachyonychia); N2 = all 20 nails show dense pitting or trachyonychia]. The category for nail N2 can be further subdivided to differentiate between dense pitting and trachyonychia: N2A = all 20 nails show dense pitting and N2B = all 20 nails show trachyonychia [7]. Thus, a patient classified as S3, B1, N1 would be a patient with a SALT score of 50–74% hair loss with some body hair loss and some nails showing pitting or trachyonychia. A recommendation to simplify this system has been to remove the nail designation and provide two scores—a SALT score and notation of body hair loss (i.e., absent loss/incomplete loss/complete loss) [13]. However, the presence and extent of nail involvement have been suggested to also be an indicator for disease severity [14].
Duration of Hair Loss
The natural history of AA is variable. Many people develop a single patch of AA that recovers spontaneously within 6–12 months. Others develop multiple patches or extensive hair loss, but still achieve a spontaneous remission within 6–12 months. Perhaps 30% of patients develop chronic relapsing AA, including alopecia totalis/universalis [15]. Disease and/or episode duration provides prognostic information, and so classification of AA as acute or chronic might also be considered in determining disease severity.
Burden of Illness
Although the burden of illness has not actually been incorporated formally into any disease severity classification, its clinical relevance is high: patches of scalp hair loss could be quite tolerable for one patient but devastating for another. Thus, from a clinical perspective, any discussion of disease severity has to incorporate the impact of the disease on the patient, regardless of location, duration, pattern, or amount of loss. In a qualitative interview study of patients with AA, a comprehensive model was derived from the interviews in which the impact of AA was shown to encompass signs and symptoms, physical impacts, emotions, and functioning, and each of these components was further subdivided into over 30 relevant areas of impact [16]. This study—hearing directly from patients—told of the impact of AA and how the disease experience can differ from one patient to another [16].
In assessing the burden of AA, there are a number of tools, but there are no consensus recommendations as has been done in other disease states, such as the Harmonizing Outcome Measures for Atopic Dermatitis initiative [17]. In clinical trials for AA, the SALT score is consistently the primary measure across programs, but programs differ in the assessment tools regarding quality of life (QoL). One patient-reported outcome measure is the Skindex-AA, which has been adapted to AA by replacing the term “skin condition” with “hair loss.” The Skindex-16 has been modified from the longer Skindex-29 in which responses are aggregated into Symptoms (four items), Emotions (seven items), and Functioning score (five items) [18]. One limitation of the Skindex 16 is that interpretative score ranges have not yet been established that could be used to externally validate severity of disease. Also, as the origination of the Skindex was based on a broad dermatological patient population and later adapted to AA, it may underestimate the impact of AA specifically. For example, of the three domains, patients with AA tend to score lowest on the Skindex-16 symptoms scale, which asks about itching, burning, stinging, and other sensations more associated with other skin conditions.
More recent health-related QoL measures have been designed specifically for AA, such as the AA Symptom Impact Scale, which assesses both frequency and impact of AA symptoms across a number of life domains [19]. This scale provides further insight specific to AA and has been demonstrated to be a reliable and valid measure, but the thresholds for clinically meaningful difference and interpretive bandwidths have not yet been established.
Alternatively, as AA is recognized to significantly impact self-image and psychological health, it may be that anxiety and depressive symptoms can be assessed independently as part of the overall life quality status. The Hospital Anxiety and Depression scale (HADS) has been adapted across a number of medical conditions to measure psychological symptoms [20]. The HADS consists of an Anxiety scale and a Depression scale; each scale consists of seven questions with scores ranging from 0 to 21 [21]. Comparison of HADS scores with psychiatric measures have established that scores ≥ 8 are indicative of notable anxiety or depression, and scores ≥ 11 are consistent with clinically significant cases [20]. Overall, however, while the impact of AA has been investigated on QoL and psychological comorbidities, these measures have not been used as criterion within an AA classification system.
Defining AA and Severity of Disease: a Framework
There is no broad agreement on a classification system in AA. This limits our ability to accurately and systematically assess and document AA severity across patients. Furthermore, with the emergence of targeted therapies for AA, AA classification will be important to inform treatment guidelines. Understanding AA severity more broadly will be important for aligning treatments with patients. In addition, rethinking the severity of AA beyond the amount of scalp hair loss recognizes the substantial morbidity of the disease.
The question then arises, “How should severity of disease be defined?” In general, symptoms, signs, and physiologic measures need to be incorporated for the purpose of reflecting patient experience, guiding treatment, or measuring treatment response. Furthermore, a classification system must be able to address the following questions:
What is the goal or purpose of the system?
- How will severity be defined?
-
i.Solely on signs? Inclusion of symptoms?
-
ii.Unidimensional factors or multidimensional?
-
iii.Should functional impact be consistently included in the classification?
-
i.
How easy will it be to obtain the necessary information for classification? Can it be captured and applied in both research and clinical settings? What is the burden of collecting this information for the dermatologist? Or for the patient?
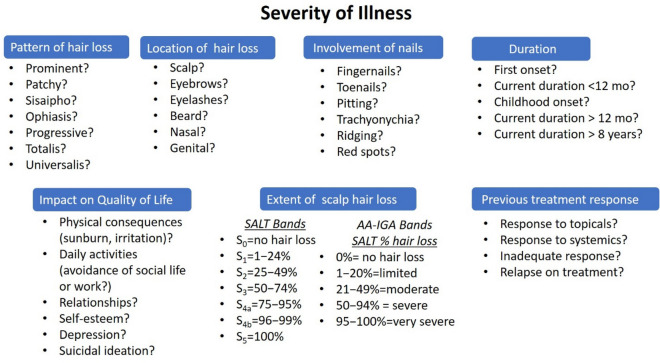
Emerging from the review of the most clinically relevant factors in AA, potential components of AA severity classification that can address these questions are presented in Fig. 1. This multidimensional framework suggests that dermatologists consider a number of patient illness variables as well as the QoL impact. Additionally, the framework indicates that clinical trials also need to assess beyond the amount of scalp hair loss and regrowth and that these additional factors, particularly the impact on the patient and the presence of eyebrow/eyelash, should be considered key secondary endpoints.
Fig. 1.
Components for assessing disease severity, including signs and symptoms of AA, impact on quality of life, and treatment history for qualitative evaluation in classification of severity. Questions under each component are attributes for consideration with potential greater implications of severity from top to bottom. AA-IGA Alopecia Areata Investigator Global Assessment, mo months, SALT Severity of Alopecia Tool
Within each component of the framework, questions are organized in the direction of indices of greater severity. This framework moves beyond the traditional components of AA (hair loss, location, pattern, duration) to formally incorporating the broad detrimental effect of the disease on patients’ overall well-being [22]. Other factors may impact the patient’s illness, such as the presence of autoimmune comorbidities, but were considered out of scope for defining AA severity within its own parameters and therefore were not included in the framework.
We propose this framework for a twofold purpose. First, to start a consensus discussion within the field about ascertaining the severity of AA as a disease through the inclusion of specific multiple patient- and illness-related domains. In this discussion, this framework represents a starting point that is grounded in both the medical and scientific literature as well as expert clinical perspectives. For such a purpose, there has been a parallel effort by US clinician researchers to develop a scale that proposes specific criteria for the severity spectrum of AA [23]. In this scale, the primary determination of disease severity is anchored in the amount of scalp hair loss and then modified depending on the presence of either non-scalp hair loss (eyebrows, eyelashes), inadequate treatment response, multifocal diffuse positive hair pull test (indicating poor prognosis), and negative impact on psychosocial functioning [23].
The second purpose of the framework is for application within the dermatological clinic. Using this framework, dermatologists can qualitatively evaluate the complexity of the patient’s illness to provide a holistic understanding of the disease. While many of these questions are routinely incorporated into the clinical interview, the framework also organizes questions to ensure that each aspect that may modify the physician’s perception of the disease severity is incorporated into the diagnostic judgment. Furthermore, inclusion of the emotional and quality of life aspects of the disease impact ensures the inclusion of the patient’s perspective regarding their burden. In a recent study, among a number of patient illness and disease characteristics, patient’s perception of their illness severity as moderate to severe was consistently associated with poorer QoL while the absolute SALT score did not predict QoL scores [24]. As more therapies become available, disease severity will be one aspect for identifying appropriate therapies for different patients. Using this framework or similarly developed scales, such as the AA scale [23], dermatologists may become more consistent in the determination of disease severity, which in turn can enhance our understanding of how these treatments work across the severity spectrum.
Conclusions
Current approaches to classification for AA indicate a lack of consensus. While the amount and location of hair loss is the primary criterion, there is no broad agreement regarding how these values translate into the more clinician-friendly concepts of “mild,” “moderate,” “severe,” or “very severe” disease. Furthermore, psychosocial impact is not routinely included despite the clear psychosocial morbidity of this disease.
Diagnostic consensus is established for the clinical features of AA, but heterogeneity of AA has resulted in multiple approaches in identifying subtypes or in defining severity within the disease. As our understanding of disease pathogenesis evolves and treatments advance, guidance is needed, which will require a broader view of disease. In addition, consensus on the best tools can also be helpful as we try to capture the most relevant impacts. We provide a framework for conceptualizing AA severity classification based on multiple patient and disease factors. This framework can serve as a scientific and professional guide for a conversation about the conceptualization of AA disease severity beyond scalp hair loss. It can also serve as useful tool for dermatologists to systemically assess the holistic burden of the disease using a multidimensional approach.
Acknowledgements
Funding
This work and associated article processing charges, including the journal’s Rapid Service Fee, were fully funded by Eli Lilly and Company.
Medical Writing, Editorial, and Other Assistance
The authors would like to thank Dr. Marsha Tracey, an employee of Eli Lilly and Company, for help with project management, editing, and formatting the manuscript.
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Author Contributions
Brett A. King has made substantial contributions to the interpretation of data for the work and the critical revision of the manuscript for important intellectual content. Maryanne M. Senna has made substantial contributions to the conception and design of the work, interpretation of data and the critical revision of the manuscript for important intellectual content. Manabu Ohyama has made substantial contributions to the interpretation of data for the work, the drafting and critical revision of the manuscript for important intellectual content. Antonella Tosti has made substantial contributions to the interpretation of data for the work and the critical revision of the manuscript for important intellectual content. Rodney Sinclair has made substantial contributions to the interpretation of data for the work and the critical revision of the manuscript for important intellectual content. Susan Ball has made substantial contributions to the conception and design of the work, interpretation of data and the drafting of the manuscript. Justin M. Ko has made substantial contributions to the interpretation of data for the work and the critical revision of the manuscript for important intellectual content. Marc Glashofer has made substantial contributions to the analysis of data for the work and the critical revision of the manuscript for important intellectual content. Rodrigo Pirmez has made substantial contributions to the conception of the work, analysis of data for the work. interpretation of data and the critical revision of the work for important intellectual content. Jerry Shapiro has made substantial contributions to the acquisition of data for the work, analysis and interpretation of data and the critical revision of the manuscript for important intellectual content.
Disclosures
Brett A. King reported personal and other fees from Eli Lilly and Company, Pfizer, and Concert Pharmaceuticals and, personal fees from Bristol-Meyers Squibb, Dermavant, Celgene, Aclaris, Arena, Regeneron, Sanofi Genzyme, Viela Bio, TWi Biotechnology, outside the submitted work. Maryanne M. Senna reported clinical trial funding and SAB consulting fees from Eli Lilly and Company, during the conduct of the study and reported clinical trial funding from Concert Pharmaceuticals and SAB consulting fees from Pfizer, outside the submitted work. Manabu Ohyama is a scientific advisor for Eli Lilly Japan, Taisho Pharmaceutical Co., Pfizer Japan Inc., Janssen Pharmaceutical K.K., and ROHTO Pharmaceutical Co. and receives research grants not related to this study from Shiseido Co., Maruho Co., Sun Pharma Japan Ltd. Antonella Tosti is a compensated consultant/advisory board member for Eli Lilly, sponsor of the study and reported consultancy/advisory board member fees, during the conduct of the study. She reported consultancy fees from DS Laboratories, Monat Global, Almirall, Thirty Madison, Bristol Myers Squibb, P&G, Leo Pharmaceuticals, outside the submitted work and is serving as the President of the American Hair Research Society. Rodney Sinclair reported serving as a consultant or paid speaker for or participating in clinical trials sponsored by LEO, Pharma, Amgen, Inc, Novartis Pharmaceuticals Corporation, Merck & Co, Celgene Corporation, Coherus BioSciences, Janssen Global Services, LLC, Regeneron Pharmaceuticals Inc, MedImmune, LLC, GlaxoSmithKline, Cutanea, Samson Clinical, Boehringer Ingelheim, Pfizer, Inc, Merck Sharpe & Dohme, Oncobiologics, Inc, F. Hoffman–La Roche, Ltd, Eli Lilly and Company, and Bayer AG and is serving as the current President of the Australasian Hair and Wool Research Society. Susan Ball is an employee and a shareholder of Eli Lilly and Company. Justin M. Ko has served on advisory boards and is a consultant and clinical investigator for Eli Lilly and Company he has served as a clinical investigator and/or consultant for AbbVie, Sanofi, Regeneron, Dermira, BMS and Arena Pharmaceuticals. He has received consulting fees from Eli Lilly and Company, Concert Pharmaceuticals, and Arena Pharmaceuticals. Marc Glashofer reported serving as a consultant for Eli Lilly and Company. Rodrigo Pirmez reported a consultancy fee from Eli Lilly and Company, during the conduct of the study and a consultancy fee from Pfizer, outside the submitted work. Jerry Shapiro reported serving as a consultant for Eli Lilly and Company and serving as a consultant and investigator for Pfizer Inc, and receiving honorarium from Pfizer Inc., Eli Lilly and Company, Applied Biology and DS Laboratories. He served as an investigator for Regenlab and received stock options for Eirion and Replicel Life Sciences.
Compliance with Ethics Guidelines
This article is based on previously conducted studies and does not contain any new studies with human participants or animals performed by any of the authors.
Data Availability
Data sharing is not applicable to this article as no datasets were generated or analyzed during the current study.
References
- 1.Strazzulla LC, Wang EHC, Avila L, Lo Sicco K, Brinster N, Christiano AM, et al. Alopecia areata: disease characteristics, clinical evaluation, and new perspectives on pathogenesis. J Am Acad Dermatol. 2018;78:1–12. doi: 10.1016/j.jaad.2017.04.1141. [DOI] [PubMed] [Google Scholar]
- 2.Aggarwal R, Ringold S, Khanna D, Neogi T, Johnson SR, Miller A, et al. Distinctions between diagnostic and classification criteria? Arthritis Care Res (Hoboken). 2015;67:891–897. doi: 10.1002/acr.22583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Besnier E, Brocq L, Jacquet L. La pratique dermatologique. In la pratique dermatologique,1902. p. 920.
- 4.Ikeda T. A new classification of alopecia areata. Dermatology. 1965;131:421–445. doi: 10.1159/000254503. [DOI] [PubMed] [Google Scholar]
- 5.Sharma VK, Muralidhar S, Kumar B. Reappraisal of Ikeda's classification of alopecia areata: analysis of 356 cases from Chandigarh, India. J Dermatol. 1998;25:108–111. doi: 10.1111/j.1346-8138.1998.tb02359.x. [DOI] [PubMed] [Google Scholar]
- 6.Olsen EA, Hordinsky MK, Price VH, Roberts JL, Shapiro J, Canfield D, et al. Alopecia areata investigational assessment guidelines--part ii. National alopecia areata foundation. J Am Acad Dermatol. 2004;51:440–447. [DOI] [PubMed]
- 7.Olsen EA, Canfield D. Salt II: a new take on the severity of alopecia tool (salt) for determining percentage scalp hair loss. J Am Acad Dermatol. 2016;75:1268–1270. doi: 10.1016/j.jaad.2016.08.042. [DOI] [PubMed] [Google Scholar]
- 8.Jang YH, Moon SY, Lee WJ, Lee SJ, Lee WK, Park BC, et al. Alopecia areata progression index, a scoring system for evaluating overall hair loss activity in alopecia areata patients with pigmented hair: a development and reliability assessment. Dermatology. 2016;232:143–149. doi: 10.1159/000442816. [DOI] [PubMed] [Google Scholar]
- 9.Wyrwich KW, Kitchen H, Knight S, Aldhouse NVJ, Macey J, Nunes FP, et al. The alopecia areata investigator global assessment scale: a measure for evaluating clinically meaningful success in clinical trials. Br J Dermatol. 2020;183:702–709. doi: 10.1111/bjd.18883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Meah N, Wall D, York K, Bhoyrul B, Bokhari L, Sigall DA, et al. The Alopecia Areata Consensus of Experts (ACE) study: results of an international expert opinion on treatments for alopecia areata. J Am Acad Dermatol. 2020;83:123–130. doi: 10.1016/j.jaad.2020.03.004. [DOI] [PubMed] [Google Scholar]
- 11.Meah N, Wall D, York K, Bhoyrul B, Bokhari L, Sigall DA, et al. The Alopecia Areata Consensus of Experts (ACE) study part II: results of an international expert opinion on diagnosis and laboratory evaluation for alopecia areata. J Am Acad Dermatol. 2020. [DOI] [PubMed]
- 12.Liu LY, King BA, Ko JM. Eyebrows are important in the treatment of alopecia areata. J Investig Dermatol Symp Proc. 2020;20:S37–s40. doi: 10.1016/j.jisp.2020.04.006. [DOI] [PubMed] [Google Scholar]
- 13.Wambier CG, King BA. Rethinking the classification of alopecia areata. J Am Acad Dermatol. 2019;80:e45. [DOI] [PubMed]
- 14.Roest YBM, van Middendorp HT, Evers AWM, van de Kerkhof PCM, Pasch MC. Nail involvement in alopecia areata: a questionnaire-based survey on clinical signs, impact on quality of life and review of the literature. Acta Derm Venereol. 2018;98:212–217. doi: 10.2340/00015555-2810. [DOI] [PubMed] [Google Scholar]
- 15.Cranwell WC, Lai VW, Photiou L, Meah N, Wall D, Rathnayake D, et al. Treatment of alopecia areata: an Australian expert consensus statement. Australas J Dermatol. 2019;60:163–170. doi: 10.1111/ajd.12941. [DOI] [PubMed] [Google Scholar]
- 16.Aldhouse NVJ, Kitchen H, Knight S, Macey J, Nunes FP, Dutronc Y, et al. "'You lose your hair, what's the big deal?' I was so embarrassed, I was so self-conscious, I was so depressed:" A qualitative interview study to understand the psychosocial burden of alopecia areata. J Patient Rep Outcomes. 2020;4:76. doi: 10.1186/s41687-020-00240-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Thomas KS, Apfelbacher CA, Chalmers JR, Simpson E, Spuls PI, Gerbens LAA, et al. Recommended core outcome instruments for health-related quality of life, long-term control and itch intensity in atopic eczema trials: results of the HOME VII consensus meeting. Br J Dermatol. 2021;185:139–146. doi: 10.1111/bjd.19751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Chren MM. The skindex instruments to measure the effects of skin disease on quality of life. Dermatol Clin. 2012;30:231–236, xiii. [DOI] [PMC free article] [PubMed]
- 19.Mendoza TR, Osei J, Duvic M. The utility and validity of the alopecia areata symptom impact scale in measuring disease-related symptoms and their effect on functioning. J Investig Dermatol Symp Proc. 2018;19:S41–s46. doi: 10.1016/j.jisp.2017.10.009. [DOI] [PubMed] [Google Scholar]
- 20.Silverberg JI, Gelfand JM, Margolis DJ, Boguniewicz M, Fonacier L, Grayson MH, et al. Measurement properties of the hospital anxiety and depression scale used in atopic dermatitis in adults. J Invest Dermatol. 2019;139:1388–1391. doi: 10.1016/j.jid.2018.11.029. [DOI] [PubMed] [Google Scholar]
- 21.Zigmond AS, Snaith RP. The hospital anxiety and depression scale. Acta Psychiatr Scand. 1983;67:361–370. doi: 10.1111/j.1600-0447.1983.tb09716.x. [DOI] [PubMed] [Google Scholar]
- 22.US Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research. The voice of the patient: a series of reports from the U.S. Food and Drug Administration's (FDA's) patient‐focused drug development initiative. Alopecia areata. 2018.
- 23.King BA, Mesinkovska NA, Craiglow B, Kindred C, Ko J, McMichael A, et al. Development of the alopecia areata scale for clinical use: results of an academic–industry collaborative effort. J Am Acad Dermatol. 2021. [DOI] [PubMed]
- 24.Senna MM, Ko J, Glashofer M, Walker C, Ball S, Heredia E, et al. Predictors of quality of life in patients with alopecia areata. Manuscript under review, 2021.
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Data sharing is not applicable to this article as no datasets were generated or analyzed during the current study.