Abstract
Simple Summary
Organ graft type is an independent risk factor for the development of PTLD. Due to small cohort sizes caused by the rarity of the disease, most studies and trials are performed on populations of recipients of various organ graft types. Our study is a first direct comparison of the effect of different risk and prognostic factors between kidney and liver transplant recipients (KTRs and LTRs, respectively). We have demonstrated that the analysis of risk factors based on the general solid organ transplant recipient population is inconsistent with the results obtained through the analysis of single organ graft type cohorts. The risk of PTLD is lower in KTRs who are female, <45 years old at transplantation, or treated with cyclosporin. Based upon our findings, we are confident that the type of organ graft is an overriding factor in PTLDs’ development and course.
Abstract
Post-transplantation lymphoproliferative disorder (PTLD) is a life-threatening complication of solid organ transplantation (SOT). Its development risk varies among organ graft recipients. In this study, retrospective data were analyzed to compare PTLD’s risk and prognostic factors between adult kidney and liver transplant recipients (KTRs and LTRs, respectively). Over 15 years, 2598 KTRs and 1378 LTRs were under observation at our center. Sixteen KTRs (0.62%) and twenty-three LTRs (1.67%) were diagnosed with PTLD. PTLD developed earlier in LTRs (p < 0.001), SOT patients > 45 years old (p = 0.002), and patients receiving tacrolimus (p < 0.001) or not receiving cyclosporin (p = 0.03) at diagnosis. Tacrolimus use, male sex, and age > 45 years old significantly affected the time of PTLD onset in KTRs (hazard ratio (HR) = 18.6, 7.9 and 5.2, respectively). Survival was longer in LTRs < 45 years old (p < 0.009). LTRs were more likely than KTRs to achieve complete remission (p = 0.039). Factors affecting PTLD development and outcome differ between KTRs and LTRs; thus, these populations should be separately evaluated in future studies.
Keywords: kidney transplantation, liver transplantation, post-transplant lymphoproliferative disorder, solid organ transplantation
1. Introduction
Post-transplantation lymphoproliferative disorder (PTLD) was first described as a life-threatening complication of allotransplantation in 1969 by Penn et al. [1] in a series of five kidney graft recipients diagnosed with lymphoma. Currently, it is recognized as a heterogeneous group of lymphoid neoplasms occurring in hematopoietic stem cell and solid organ transplant (SOT) recipients. These groups of patients are at increased risk of carcinogenesis, with standardized incidence ratios nearing 10 for non-Hodgkin lymphoma (NHL) and 4 for classic HL [2]. According to the 2016 revision of the World Health Organization (WHO) classification of lymphoid neoplasms [3], the following histopathological PTLD subtypes are recognized: infectious mononucleosis, plasmacytic hyperplasia, florid follicular hyperplasia, polymorphic, monomorphic (B- and T/natural killer (NK)-cell subtypes with the exclusion of indolent lymphomas), and HL. Previous research has established a clear role of Epstein-Barr virus (EBV) in the development of early-onset PTLD (<12 months after transplantation), especially in pediatric graft recipients. It has been reported that primary EBV infection combined with the effects of T-cell depleting induction and maintenance immunosuppression (IS) can drive malignant B-cell transformation [4]. However, the pathogenesis of PTLD is multifactorial and not entirely understood, as in up to 50% of all cases, the disease is EBV-negative [5], up to 10% of neoplasms stem from T/NK cells [6,7], and another peak of onset is observed at 10–15 years post-transplantation [8,9]. To date, several other risk factors have been proposed or identified based on varying degrees of evidence [10], including the cumulative dose of immunosuppressants (induction, maintenance, and acute rejection treatment), use of certain immunosuppressive agents, primary diseases [11], and viral infections such as cytomegalovirus (CMV), hepatitis B (HBV), and C (HCV) [12,13,14,15].
Assembling a large cohort of single type organ graft patients with PTLD is difficult; therefore, most researchers have investigated PTLD in combined populations of different organ graft recipients, and found that the relative risk (RR) of disease development significantly varies according to the type of transplanted organ [16,17,18,19,20,21]. Multiorgan and intestinal graft recipients have the highest RR (RR = 239.5) in contrast to lung transplant recipients (RR = 58.6), pancreas transplant recipients (RR = 34.9), liver transplant recipients (LTRs) (RR = 29.9), heart transplant recipients (RR = 27.6), and kidney transplant recipients (KTRs) (RR = 12.6) [22].
This study identified the factors contributing to the development, course, response to treatment, and outcomes of PTLD and compared their impact on KTRs and LTRs.
2. Materials and Methods
2.1. Study Participants
This retrospective cohort study included 39 patients diagnosed with PTLD out of 2598 KTRs and 1378 LTRs followed up at our center between 2002 and 2017. The KTR group was defined as KTRs and non-simultaneous KTRs and LTRs who received a kidney graft first. The LTR group was defined analogically. The SOT group consisted of 39 patients (KTRs and LTRs). Both KTRs and LTRs included one non-simultaneous KTR and LTR each. No pediatric patients were included in the study; however, three KTRs and one LTR underwent transplantation at the age of 6–14 years and developed PTLD in adulthood. Identified cases were classified into PTLD subtypes according to the 2016 revision of the WHO classification of lymphoid neoplasms. In PTLD cases confirmed posthumously, the date of first cytological or imaging results suggestive of PTLD was considered the date of PTLD diagnosis. EBV positivity was determined to be either active EBV replication or the presence of latent membrane protein 1 in histopathological samples. The analyzed data were collected from the patients’ medical records. This study was approved by The Bioethics Committee of the Medical University of Warsaw, approval No. AKBE/90/2022.
2.2. Statistical Analyses
The statistical differences in clinical characteristics were assessed using the Mann–Whitney U test. The Welch’s t-test was performed to compare the mean parameters, and the log-rank test was used to compare Kaplan–Meier estimator curves between groups. Univariate and multivariate analyses of the effects of variables on the PTLD time of onset were calculated using the Cox proportional hazards model and Wald test. Data analyses were performed in R version 3.2.3. p < 0.05 was considered statistically significant.
3. Results
3.1. Clinical Characteristics
The clinical characteristics are summarized in Table 1. The prevalence of PTLD was 0.62% (16 patients) and 1.67% (23 patients) in KTRs and LTRs, respectively. The incidence rate of PTLD was 0.8 per 1000 patient-years for KTRs and 2.1 per 1000 patient-years for LTRs. There was no significant difference in sex distribution among the PTLD patients. The median age at first transplantation was 28 years for KTRs (range 6–70 years) and 48 for LTRs (range 12–60).
Table 1.
Clinical characteristics.
| LTRs | KTRs | ||
|---|---|---|---|
| Patient Characteristics | n (%) | p-Value | |
| Number of patients | 23 (100) | 16 (100) | |
| PTLD incidence rate (per 1000 patient-years) Mean age at Tx (years) [SD] |
2.1 | 0.8 | |
| 43.7 (12.0) | 36.3 (20.7) | 0.21 | |
| Males Females |
13 (56.5) | 11 (68.8) | 0.456 |
| 10 (43.5) | 5 (31.3) | 0.456 | |
| KT+LTRs Retransplantation |
1 (4.4) | 1 (6.3) | 1.0 |
| 1 (4.4) | 1 (6.3) | 1.0 | |
| IS Treatment | |||
| Induction | |||
| Anti-CD25 | 12 (52.2) | 3 (18.8) | 0.076 |
| ATG | 0 (0) | 1 (6.3) | 0.853 |
| Acute rejection | |||
| Total | 8 (34.8) | 5 (31.3) | 0.329 |
| 1 (4.4) no data | 2 (12.5) no data | ||
| GCS treated | 8 (34.8) | 5 (31.3) | 0.984 |
| ATG treated | 1 (4.4) | 0 (0) | 0.456 |
| Maintenance | |||
| Monotherapy | 4 (17.4) | 0 (0) | 0.052 |
| Double drug therapy | 12 (52.2) | 6 (37.5) | 0.381 |
| Triple drug therapy | 7 (30.4) | 10 (62.5) | 0.052 |
| GCS | 14 (60.9) | 13 (81.3) | 0.428 |
| CsA | 2 (8.7) | 8 (50) | 0.004 * |
| TAC | 21 (91.3) | 6 (37.5) | <0.001 * |
| AZA | 1 (4.4) | 5 (31.3) | 0.025 * |
| MMF | 11 (47.8) | 10 (62.5) | 0.255 |
| Viral Replication | LTRs | KTRs | p-Value |
| EBV DNA | 10 (43.2) | 10 (62.5) | 0.053 |
| 5 (21.7) no data | 5 (31.3) no data | ||
| CMV DNA | 0 (0) | 2 (12.5) | 0.093 |
| HBV DNA | 2 (8.7) | 1 (6.3) | 0.805 |
| HCV RNA | 5 (21.7) | 1 (6.3) | 0.201 |
ATG—anti-thymocyte globulin, AZA—azathioprine, CMV—cytomegalovirus, CsA—cyclosporin, EBV—Epstein-Barr virus, GCs—glucocorticosteroids, HBV—hepatitis B virus, HCV—hepatitis C virus, KT+LTRs—non-simultaneous kidney and liver transplant recipients, KTRs—kidney transplant recipients, LTRs—liver transplant recipients, MMF—mycophenolate mofetil, TAC—tacrolimus (Mann–Whitney U-test for comparison between KTRs and LTRs), * p < 0.05.
Antibody induction was used in a total of 16 SOT recipients (41.03%). Anti-cluster of differentiation 25 (CD25) antibodies were administered to 12 of 23 LTRs (52.17%) and 3 of 16 KTRs (18.75%). Anti-thymocyte globulin was used in only one KTR (6.25%). A history of acute rejection was recorded in one-third of patients in both groups. Four LTRs received tacrolimus (TAC) monotherapy as maintenance IS. The remaining patients were either on two- or three-drug regimens composed of glucocorticosteroids, calcineurin inhibitor (cyclosporin (CsA) or TAC), and an anti-proliferative agent (azathioprine (AZA) or mycophenolate mofetil (MMF)). Compared to LTRs, a significantly larger number of KTRs received regimens containing CsA and AZA, whereas TAC-based IS was used in a significantly higher proportion of LTRs than KTRs (Supplement Table S1). No significant differences in viral replication and serological status for CMV, HBV, and HCV were detected. Only a difference in EBV positivity approached statistical significance (p = 0.053); however, in 31.25% of KTRs and 21.74% of LTRs, the data regarding EBV were unavailable. PTLD was mostly diagnosed at >12 months after transplantation (late onset), and its type was not significantly different between LTRs and KTRs (Table 2). Data regarding the location and dissemination of PTLD are presented in Supplement Table S2.
Table 2.
PTLD characteristics.
| LTRs | KTRs | ||
|---|---|---|---|
| PTLD Characteristics | n (%) | p-Value | |
| Focal disease | 11 (47.8) | 8 (50) | 0.908 |
| Disseminated disease Early onset |
12 (52.2) | 8 (50) | 0.908 |
| 5 (21.7) | 1 (6.3) | 0.16 | |
| Late onset Post-mortem diagnosis |
18 (78.3) | 15 (93.8) | 0.16 |
| 3 (13) | 0 (0) | 0.083 | |
| WHO 2016 Type | |||
| Florid follicular hyperplasia | 1 (4.4) | 0 (0) | 0.434 |
| Infectious mononucleosis | 0 (0) | 1 (6.3) | 0.251 |
| Plasmacytic hyperplasia | 6 (26.1) | 1 (6.3) | 0.122 |
| Polymorphic | 4 (17.4) | 1 (6.3) | 0.324 |
| Monomorphic B-cell | 11 (47.8) | 9 (56.3) | 0.182 |
| -DLBCL | 9 (39.1) | 3 (18.8) | 0.168 |
| -HGBL | 1 (4.4) | 1 (6.3) | 0.805 |
| -Lymphomatoid granulomatosis | 0 (0) | 2 (12.5) | 0.164 |
| -Plasmacytic myeloma | 0 (0) | 1 (6.3) | 0.333 |
| -MCL | 0 (0) | 1 (6.3) | 0.333 |
| -Other | 1 (4.4) | 1 (6.3) | 0.805 |
| Monomorphic T-cell | 1 (4.4) | 3 (18.8) | 0.167 |
| Classic Hodgkin lymphoma | 0 (0) | 1 (6.3) | 0.853 |
Early onset is <12 months after transplantation, late onset is >12 months after transplantation, DLBCL—diffuse large B-cell lymphoma, HGBL—high-grade B-cell lymphoma, KTRs—kidney transplant recipients, LTRs—liver transplant recipients, MCL—mantle cell lymphoma. p-values represent the Mann–Whitney U-test comparison between KTRs and LTRs.
3.2. Factors Associated with PTLD Development
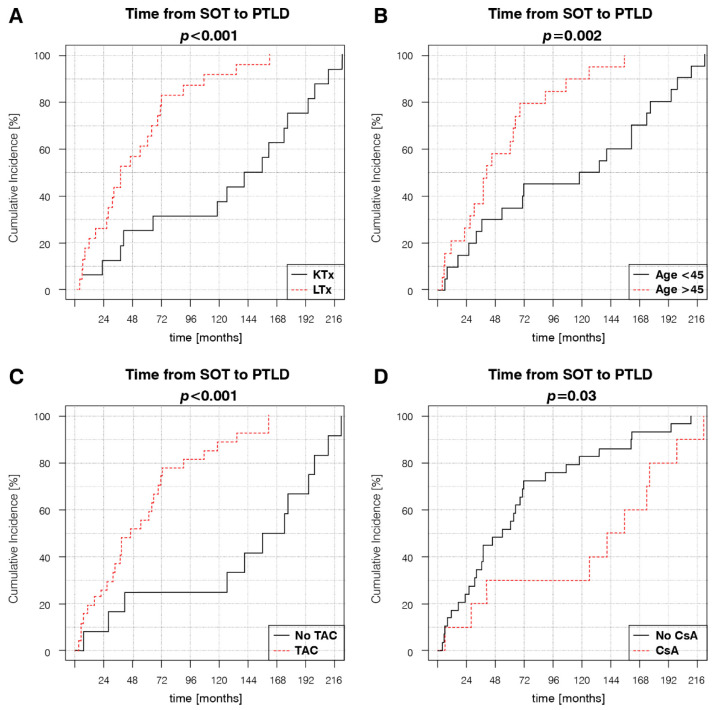
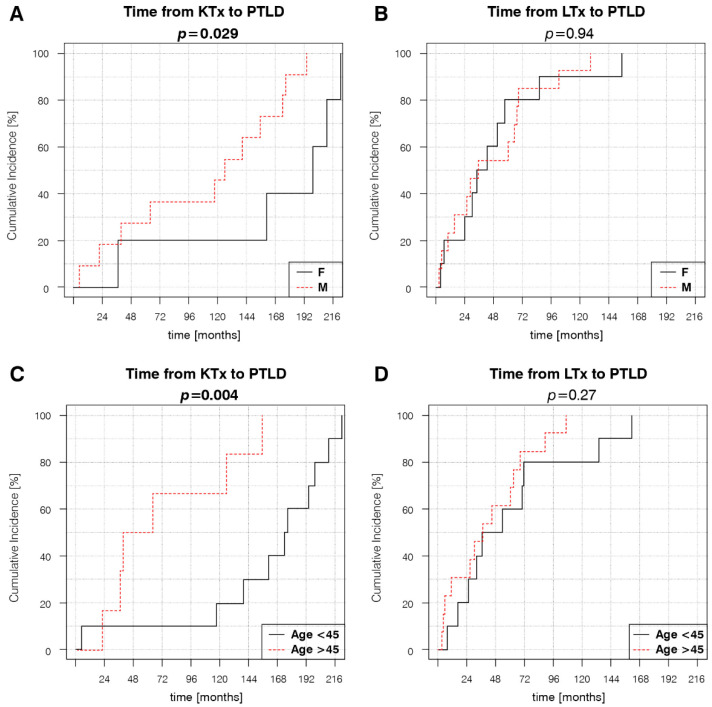
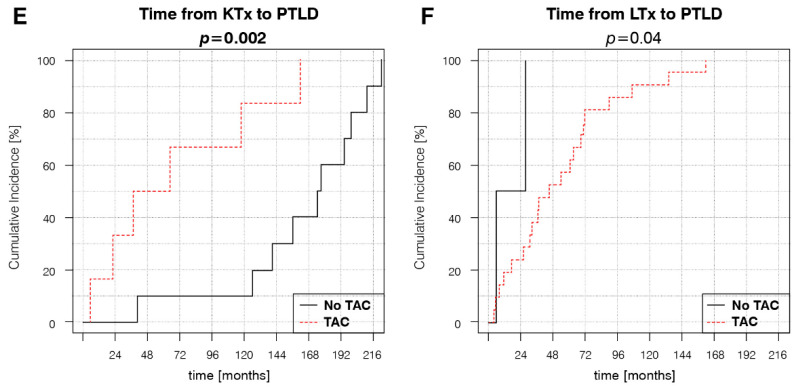
Kaplan–Meier estimator curves were plotted and compared using the log-rank test to determine the impact of different variables on the time of PTLD onset. The analyses revealed significantly different dynamics of disease development between KTRs and LTRs, with a mean time to PTLD diagnosis of 118.6 months (9.9 years) and 55.8 months (4.7 years), respectively (Figure 1A). Earlier PTLD development in the SOT population was associated with age at transplantation > 45 years, TAC-based, and non-CsA IS regimens (Figure 1B–D). Subsequent Kaplan–Meier estimator curve analyses for separate KTR and LTR groups showed that the significance of the aforementioned risk factors changed in relation to the type of transplanted organ. Age at transplantation > 45 years retained significance in KTRs, but not in LTRs (Figure 2C,D). TAC-based IS regimens retained significance in both KTRs and LTRs (Figure 2E,F). Female sex, a factor that was not significant in the SOT group, was significantly associated with later PTLD onset in the KTRs (Figure 2A,B).
Figure 1.
Factors influencing the dynamic of PTLD development in the SOT group: (A) organ type, (B) age at transplantation, (C) use of TAC in the maintenance IS regimen at the time of PTLD diagnosis, and (D) use of cyclosporin in maintenance IS at the time of PTLD diagnosis. p-values represent the log-rank analysis of the Kaplan–Meier estimator curves.
Figure 2.
Transplanted organ-dependent effect of different variables on PTLD development: (A) KTR sex, (B) LTR sex, (C) KTR age at transplantation, (D) LTR age at transplantation, (E) Presence of TAC in KTR IS regimens at PTLD diagnosis, (F) Presence of TAC in LTR IS regimens at PTLD diagnosis. p-values represent the log-rank analysis of the Kaplan–Meier estimator curves.
The results obtained through log-rank analyses of the Kaplan–Meier estimator curves were verified using univariate and multivariate Cox proportional hazards models (Table 3). In the SOT group, univariate analyses revealed a significantly increased hazard ratio (HR) of earlier PTLD development in patients older than 45 years at transplantation (HR = 1.03, 95% confidence interval (CI) = 1.01–1.05; p = 0.006), patients with TAC-based IS (HR = 5.09, 95% CI = 2.00–12.95; p < 0.001), and a decreased HR in patients treated with CsA-based IS regimens (HR = 0.44, 95% CI = 0.20–0.95; p = 0.036). In univariate analyses, significant increases in HR were detected in KTRs > 45 years old at transplantation (HR = 1.04, 95% CI = 1.00–1.07; p = 0.044), male KTRs (HR = 4.86, 95% CI = 1.03–22.81; p = 0.045), and KTRs receiving TAC (HR = 6.25, 95% CI = 1.68–23.21; p = 0.006).
Table 3.
Univariate and multivariate Cox proportional hazards models of the effect of variables on the PTLD time of onset.
| SOT—Univariate | KTR—Univariate | LTR—Univariate | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Variable | HR | 95% CI | p-Value | Variable | HR | 95% CI | p-Value | Variable | HR | 95% CI | p-Value |
| Male sex | 1.49 | (0.74–3.02) | 0.269 | Male sex | 4.86 | (1.03–22.81) | 0.045 * | Male sex | 1.03 | (0.44–2.44) | 0.939 |
| Age at Tx > 45 years | 1.03 | (1.01–1.05) | 0.006 * | Age at Tx > 45 years | 1.04 | (1.00–1.07) | 0.044 * | Age at Tx > 45 years | 1.02 | (0.98–1.05) | 0.357 |
| Retransplantation | 0.96 | (0.29–3.2) | 0.95 | Retransplantation | 2.46 | (0.29–21.12) | 0.412 | Retransplantation | 0.46 | (0.1–2.04) | 0.306 |
| Monotherapy | 2.18 | (0.81–5.84) | 0.12 | Monotherapy | NA | NA | NA | Monotherapy | 1.11 | (0.41–3.06) | 0.835 |
| Double drug therapy | 0.88 | (0.46–1.67) | 0.69 | Double drug therapy | 0.56 | (0.19–1.61) | 0.282 | Double drug therapy | 1.18 | (0.51–2.75) | 0.695 |
| Triple drug therapy | 0.97 | (0.51–1.86) | 0.934 | Triple drug therapy | 1.79 | (0.62–5.14) | 0.282 | Triple drug therapy | 0.91 | (0.35–2.36) | 0.851 |
| GCS | 0.87 | (0.43–1.78) | 0.703 | GCS | 0.68 | (0.18–2.59) | 0.574 | GCS | 1.66 | (0.7–3.94) | 0.247 |
| CsA | 0.44 | (0.20–0.95) | 0.036 * | CsA | 0.59 | (0.21–1.64) | 0.31 | CsA | 4.55 | (0.91–22.8) | 0.065 |
| TAC | 5.09 | (2.00–12.95) | <0.001 * | TAC | 6.25 | (1.68–23.21) | 0.006 * | TAC | 0.22 | (0.04–1.1) | 0.065 |
| AZA | 0.68 | (0.28–1.64) | 0.385 | AZA | 1.28 | (0.42–3.95) | 0.664 | AZA | 0.58 | (0.08–4.39) | 0.596 |
| MMF | 0.78 | (0.41–1.5) | 0.463 | MMF | 1.04 | (0.36–3.05) | 0.936 | MMF | 0.68 | (0.28–1.64) | 0.399 |
| ATG induction | 0.33 | (0.04–2.52) | 0.288 | ATG induction | 0.48 | (0.06–3.8) | 0.488 | ATG induction | NA | NA | NA |
| Anti-CD25 induction | 1.35 | (0.69–2.64) | 0.376 | Anti-CD25 induction | 0.74 | (0.2–2.67) | 0.645 | Anti-CD25 induction | 1.07 | (0.44–2.6) | 0.884 |
| AR treated with GCS | 0.75 | (0.37–1.51) | 0.413 | AR treated with GCS | 0.61 | (0.18–2.01) | 0.413 | AR treated with GCS | 0.72 | (0.3–1.76) | 0.476 |
| AR treated with ATG | 1.38 | (0.18–10.37) | 0.753 | AR treated with ATG | NA | NA | NA | AR treated with ATG | 0.9 | (0.12–6.93) | 0.922 |
| EBV DNA | 0.72 | (0.31–1.68) | 0.447 | EBV DNA | 0.7 | (0.08–6.03) | 0.744 | EBV DNA | 1.66 | (0.62–4.43) | 0.314 |
| CMV DNA | 0.6 | (0.14–2.51) | 0.482 | CMV DNA | 1.26 | (0.27–5.87) | 0.767 | CMV DNA | NA | NA | NA |
| HBV DNA | 0.86 | (0.26–2.83) | 0.802 | HBV DNA | 0.61 | (0.08–4.78) | 0.638 | HBV DNA | 1.32 | (0.3–5.85) | 0.718 |
| HCV RNA | 1.08 | (0.45–2.61) | 0.861 | HCV RNA | 0.48 | (0.06–3.8) | 0.488 | HCV RNA | 0.96 | (0.35–2.65) | 0.942 |
| SOT—Multivariate | KTR—Multivariate | LTR—Multivariate | |||||||||
| Variable | HR | 95% CI | p-Value | Variable | HR | 95% CI | p-Value | Variable | HR | 95% CI | p-Value |
| LTx | 1.75 | (0.63–4.83) | 0.281 | Male sex | 7.87 | (1.25–49.26) | 0.027 * | Male sex | 0.872 | (0.36–2.13) | 0.746 |
| Age at Tx > 45 years | 3.16 | (1.4–7.1) | 0.005 * | Age at Tx > 45 years | 5.21 | (1.04–26.25) | 0.045 * | Age at Tx > 45 years | 1.61 | (0.62–4.21) | 0.332 |
| TAC | 5.85 | (0.89–38.24) | 0.065 | TAC | 18.57 | (2.79–123.87) | 0.003 * | TAC | 0.24 | (0.04–1.35) | 0.107 |
| CsA | 1.91 | (0.39–9.31) | 0.423 |
AR—acute rejection, ATG—anti-thymocyte globulin, AZA—azathioprine, CMV—cytomegalovirus, CsA—cyclosporin, EBV—Epstein-Barr virus, GCs—glucocorticosteroids, HBV—hepatitis B virus, HCV—Hepatitis C virus, KT+LTRs—non-simultaneous kidney and liver transplant recipients, KTRs—kidney transplant recipients, LTRs—liver transplant recipients, LTx—liver transplantation, MMF—mycophenolate mofetil, SOT—solid organ transplantation group, TAC—tacrolimus, Tx—transplantation. Wald test’s p-values. * p < 0.05, NA—not applicable.
In multivariate analyses, only age at transplantation > 45 years remained a significant risk factor for all SOT patients (HR = 3.16, 95% CI = 1.4–7.1; p = 0.005). For KTRs, all three factors retained significance: age > 45 years at transplantation (HR = 5.21, 95% CI = 1.04–26.25; p = 0.045), male sex (HR = 7.87, 95% CI = 1.25–49.26; p = 0.027), and TAC-based IS (HR = 18.57, 95% CI = 2.79–123.87; p = 0.003).
3.3. Treatment and Outcomes
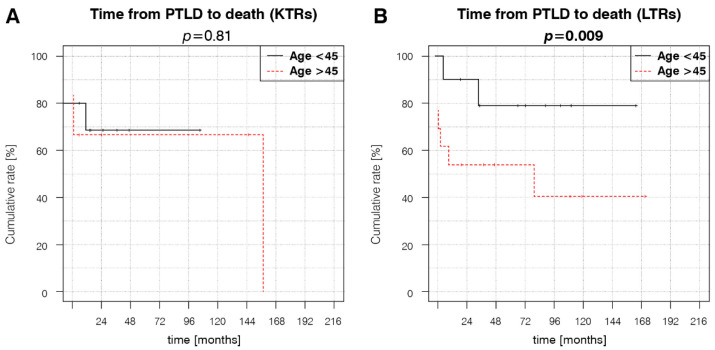
Both groups received similar PTLD treatment (Table 4). LTRs were more likely to achieve complete remission (CR) than KTRs, whereas other outcomes did not significantly differ between the groups (Table 5). Kaplan–Meier estimator curve analyses revealed that patient survival was superior in LTRs < 45 years old at transplantation (Figure 3B). In KTRs, age did not significantly affect survival (Figure 3A).
Table 4.
PTLD treatment.
| LTRs | KTRs | ||
|---|---|---|---|
| PTLD Treatment | n (%) | p-Value | |
| RIS RAPA |
22 (95.7) | 14 (87.5) | 0.37 |
| 9 (39.1) | 3 (18.8) | 0.186 | |
| EVR Anti-CD20 |
0 (0) | 1 (6.3) | 0.251 |
| 6 (26.1) | 4 (25) | 0.955 | |
| Chemotherapy -R-CHOP |
14 (60.9) | 11 (68.8) | 0.631 |
| 7 (30.4) | 2 (12.5) | 0.204 | |
| -CHOP | 4 (17.4) | 2 (12.5) | 0.698 |
| -Other | 3 (13) | 7 (43.8) | 0.02 * |
| Surgery | 8 (34.8) | 5 (31.3) | 0.834 |
| Radiotherapy | 0 (0) | 2 (12.5) | 0.093 |
Chemotherapy—combined instances R-CHOP, CHOP, and other use, EVR—conversion to everolimus, KTRs—kidney transplant recipients, LTRs—liver transplant recipients, Other—chemotherapy protocols less frequently used in PTLD (e.g., doxorubicin, bleomycin, vinblastine, dacarbazine (ABVD), ifosfamide, carboplatin, etoposide (ICE), cyclophosphamide monotherapy), RAPA—conversion to sirolimus, RIS—reduction of immunosuppression, R-CHOP—anti-CD20 + CHOP. p-values represent the Mann–Whitney U-test comparison between KTRs and LTRs. * p < 0.05.
Table 5.
Outcomes.
| LTRs | KTRs | ||
|---|---|---|---|
| Outcomes | n (%) | p-Value | |
| CR | 12 (52.2) | 3 (18.8) | 0.039 * |
| Mean CR duration (months) (SD) Mean survival (months) (SD) |
73.7 (56.1) | 89.7 (61.7) | 0.711 |
| 56.1 (53.4) | 37.3 (51.9) | 0.28 | |
| Total deaths PTLD related deaths |
9 (39.1) | 6 (37.5) | 0.933 |
| 5 (21.7) | 4 (25) | 0.83 | |
| Alive | 12 (52.2) | 9 (56.3) | 0.817 |
| Lost to follow up | 2 (8.7) | 1 (6.3) | 0.805 |
CR—complete remission. KTRs—kidney transplant recipients, LTRs—liver transplant recipients. p-values represent the Mann-Whitney U-test comparison between KTRs and LTRs. * p < 0.05.
Figure 3.
Factors influencing patient survival. (A) KTR age at transplantation, (B) LTR age at transplantation. p-values represent the log-rank analysis of the Kaplan–Meier estimator curves.
4. Discussion
We identified the effect of specific IS agents on PTLD development in KTR and LTR populations. Our findings that CsA use or TAC avoidance in KTRs in female patients younger than 45 years are associated with later PTLD onset support the idea of tailored maintenance IS regimens as a risk-reduction strategy. These findings contradict the current consensus that the intensity and cumulative dose of IS, rather than particular maintenance immunosuppressive agents, affect PTLD development and course. Prospective validation of these findings is warranted.
We demonstrated that the significance of risk factors is dependent on the type of transplanted organ. Thus, extrapolation of the impact of PTLD risk factors in combined SOT recipient cohorts onto graft-specific populations may produce inaccurate results. For example, none of the three significant variables affecting the time of PTLD onset in our total SOT cohort retained significance in the LTR subgroup, and only two of three remained significant in the KTR group. Transplanted organ-related differences in the time of PTLD onset were recently reported by Lau et al. [23]. The authors found that the median time from transplantation to diagnosis was 0.49 years for LTRs and 4.0 years for KTRs, whereas, in our study, the mean time to PTLD diagnosis was 4.7 and 9.9 years for LTRs and KTRs, respectively. Such a difference may be attributed to the high proportion of pediatric patients in the cohort of Lau et al., as they require more IS and have a higher incidence of primary EBV infection than adults. Furthermore, a higher PTLD incidence among LTRs compared to KTRs in our cohort is in accordance with previous reports [24].
KTRs > 50 years of age at transplantation have an increased risk of late-onset PTLD (HR = 1.28) [25]. Our results show a marked risk increase (HR = 5.21) at a younger cut-off age of 45 years at kidney transplantation. Additionally, age > 55 years was previously reported to negatively impact the survival of KTRs with PTLD [26], which is not supported by our data in the KTR group. We consider the improved survival of LTRs < 45 years at liver transplantation a novel finding, as most studies analyzing the prognostic factors for LTRs are based on pediatric or mainly pediatric cohorts and describe an inverted age–PTLD association, where adult recipients demonstrate improved survival. In a paper by Fararjeh et al. [27], age was not a significant factor in the survival of adult LTRs.
To date, data on the role of patients’ sex in PTLD development in SOT recipients are limited. Moreover, sex analyses are mostly associated with patient survival [28]. The impact of different immunosuppressive agents on the PTLD time of onset and patients’ survival has been a subject of debate. Large registry studies have shown contradictory effects of TAC use with PTLD development in KTRs: Bustami et al. reported an increased risk of PTLD in first-time KTRs without antibody induction [29], whereas Francis et al. described a decreased risk of PTLD [30]. Additionally, a combination of MMF and CsA use is associated with a lower PTLD risk in KTRs [31]. Despite the statistical significance of TAC use in later PTLD onset among LTRs, it must be underlined that 21 out of 23 patients in this group received TAC and only 2 received CsA. However, the p-value of this analysis was p = 0.04. This observation should be validated on larger cohorts of LTRs taking TAC or CsA. Additionally, the significantly higher number of patients treated with TAC in the LTR group, who tend to develop PTLD earlier than KTRs in our cohort, might have affected the Kaplan–Meier estimator curve analysis of the effect of TAC on the time to PTLD diagnosis in the SOT group. We consider this as a possible limitation of this analysis.
The outcomes of PTLD treatment, particularly the 54% patient survival and the lack of a clear, statistically significant advantage of any treatment modality at this sample size, may be a result of the inclusion of patients treated before the rituximab era and of the high percentage of monomorphic T-cell PTLD cases. Initial reduction in IS treatment (RIS) was performed in 36 of 39 patients in our study and is concurrent with reports of withdrawal or reduction in AZA, MMF, or CNI or even temporary cessation of maintenance IS during chemotherapy [32]. However, the clinical outcome of RIS is highly variable and depends on the type and lineage of PTLD [33]. Contemporary treatment strategies yield higher efficacy, as confirmed by the results of the PTLD-1 prospective, international, multicenter phase II clinical trial. In that study, 88% of B-cell derived PTLD patients demonstrated a complete or partial response to rituximab induction, followed by rituximab monotherapy or an immunochemotherapy consolidation regimen consisting of rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP) [34]. The higher frequency of CR in the LTR than in KTR group in our study may be due to a relatively high number of non-diffuse large B-cell lymphoma monomorphic B-cell PTLD (e.g., lymphomatoid granulomatosis, plasmacytic myeloma) and monomorphic T-cell PTLD among KTRs. The observed higher rate of T-cell PTLD in KTRs is in accordance with previous studies—in their meta-analysis of 9 local and 147 reported cases of T-cell PTLD, Herreman and Dierickx found that this subtype of PTLD is most frequent in KTRs, who constituted 63% of all cases [35]. Although statistically insignificant, the higher number of PH PTLD patients in LTRs may reflect the shorter time to PTLD development in this group and lower intensity of IS treatment.
Recently, evidence emerged that the epidemiology of PTLD is evolving, and with current antiviral prophylaxis and stricter donor matching for EBV-seronegative recipients, there is an observable trend towards an increase in late-onset, EBV-negative PTLD [36,37,38,39]. Conversely, the field of oncological and immunological research in the immunocompromised population is expanding, as novel clinicopathological entities such as EBV-positive mucocutaneous ulcer [40] are still being discovered, and new modes of therapy are being proposed. Regarding PTLD patients, the most promising results are from studies investigating adoptive immunotherapy such as EBV-specific cytotoxic T cells [41] and chimeric antigen receptor T cells [42,43]. Both treatments can provide a sustained anti-PTLD response by T-cell engraftment, reducing the risk of disease relapse and serving as an end of the line intervention for rituximab or R-CHOP non-responders [44,45]. Recently, various NK-cell therapies have been suggested as possible PTLD treatment based on studies conducted on immunocompetent non-Hodgkin lymphoma patients [46]. In our study, no significant differences in the viral DNA and serological status for EBV were detected.
To the best of our understanding, this study had two main limitations. First, the sample size was relatively small, leading to wide CIs in the multivariate analyses. Second, the data regarding EBV status were missing in one-quarter of the patients, resulting in a nearly significant discrepancy in KTR and LTR group composition. However, the major advantages of our study were the 15-year follow-up period of a large cohort of 3976 SOT recipients and homogenous post-transplant medical care.
5. Conclusions
In summary, CsA use in female KTRs < 45 years at transplantation postpones PTLD development. Despite the cohort size, multivariate analyses supported this claim, as statistically significant changes in HR were found for these variables. The data suggest that graft type might be an overriding risk determinant, as the dynamics of PTLD onset and the impact of multiple other risk and prognostic factors significantly differ in relation to the type of transplanted organ. Further analyses are necessary to identify subsets of patients who might benefit from specific maintenance treatment modalities. Such an approach seems invaluable to achieve tailored diagnostic tools and treatment modalities for SOT recipients.
Acknowledgments
We would like to thank Magdalena Zielenkiewicz from the Faculty of Mathematics, Informatics and Mechanics, University of Warsaw for her valuable suggestions and support for the development of the study and revision of the manuscript.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/cancers14081953/s1, Table S1: History of immunosuppressive treatment in our cohort of PTLD patients; Table S2: PTLD location and dissemination in the SOT group (n = 39).
Author Contributions
Conceptualization: K.M., R.S.; methodology: K.M., R.S.; data curation: K.M., R.S., B.F., B.Z.-W., M.K., S.N., B.N., J.R.-W., K.Z. and L.P.; writing—original draft preparation: R.S.; writing—review and editing: K.M., R.S., B.F. and L.P. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
The Institutional Review Board/Bioethics Committee was informed that the data have been collected and analyzed for the purposes this study and that the results were submitted for publication. The IRB/BC No. AKBE/90/2022.
Informed Consent Statement
Patient consent was waived due to the retrospective nature of data collected from existing medical documentation.
Data Availability Statement
The data presented in this study are available upon request by the authors.
Conflicts of Interest
The authors declare no conflict of interest.
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Penn I., Hammond W., Brettschneider L., Starzl T.E. Malignant lymphomas in transplantation patients. Transplant. Proc. 1969;1:106–112. [PMC free article] [PubMed] [Google Scholar]
- 2.Grulich A.E., Van Leeuwen M.T., Falster M.O., Vajdic C.M. Incidence of cancers in people with HIV/AIDS compared with immunosuppressed transplant recipients: A meta-analysis. Lancet. 2007;370:59–67. doi: 10.1016/S0140-6736(07)61050-2. [DOI] [PubMed] [Google Scholar]
- 3.Swerdlow S.H., Campo E., Pileri S.A., Harris N.L., Stein H., Siebert R., Advani R., Ghielmini M., Salles G.A., Zelenetz A.D., et al. The 2016 revision of the World Health Organization classification of lymphoid neoplasms. Blood. 2016;127:2375–2390. doi: 10.1182/blood-2016-01-643569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Mynarek M., Schober T., Behrends U., Maecker-Kolhoff B. Posttransplant lymphoproliferative disease after pediatric solid organ transplantation. Clin. Dev. Immunol. 2013;2013:814973. doi: 10.1155/2013/814973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Luskin M.R., Heil D.S., Tan K.S., Choi S., Stadtmauer E.A., Schuster S.J., Porter D.L., Vonderheide R.H., Bagg A., Heitjan D.F., et al. The Impact of EBV Status on Characteristics and Outcomes of Posttransplantation Lymphoproliferative Disorder. Am. J. Transplant. 2015;15:2665–2673. doi: 10.1111/ajt.13324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Al-Mansour Z., Nelson B.P., Evens A.M. Post-transplant lymphoproliferative disease (PTLD): Risk factors, diagnosis, and current treatment strategies. Curr. Hematol. Malig. Rep. 2013;8:173–183. doi: 10.1007/s11899-013-0162-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Fang Y., Pinkney K.A., Lee J.C., Gindin T., Weiner M.A., Alobeid B., Bhagat G. T-cell acute lymphoblastic leukaemia after liver transplantation: Post-transplant lymphoproliferative disorder or coincidental de novo leukaemia? Hematol. Oncol. 2013;31:49–53. doi: 10.1002/hon.2016. [DOI] [PubMed] [Google Scholar]
- 8.Le J., Durand C.M., Agha I., Brennan D.C. Epstein-Barr virus and renal transplantation. Transplant. Rev. 2017;31:55–60. doi: 10.1016/j.trre.2016.12.001. [DOI] [PubMed] [Google Scholar]
- 9.Morton M., Coupes B., Roberts S.A., Klapper P.E., Byers R.J., Vallely P.J., Ryan K., Picton M.L. Epidemiology of posttransplantation lymphoproliferative disorder in adult renal transplant recipients. Transplantation. 2013;95:470–478. doi: 10.1097/TP.0b013e318276a237. [DOI] [PubMed] [Google Scholar]
- 10.Dharnidharka V.R. Comprehensive review of post-organ transplant hematologic cancers. Am. J. Transplant. 2018;18:537–549. doi: 10.1111/ajt.14603. [DOI] [PubMed] [Google Scholar]
- 11.Abu-Shanab A., Ged Y., Ullah N., Houlihan D., McCormick A. Increased Incidence of Post-transplant Lymphoproliferative Disorder in Autoimmune Liver Disease: An Irish National Experience. J. Clin. Exp. Hepatol. 2018;8:42–49. doi: 10.1016/j.jceh.2017.05.209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Yu F., Huang Y., Wang Y., Yu Z., Li X., Dong J. Very late onset post-transplant diffuse large B cell lymphoma in a liver transplant recipient with hepatitis B: A case report. Medicine. 2018;97:e13063. doi: 10.1097/MD.0000000000013063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Kinch A., Baecklund E., Backlin C., Ekman T., Molin D., Tufveson G., Fernberg P., Sundström C., Pauksens K., Enblad G. A population-based study of 135 lymphomas after solid organ transplantation: The role of Epstein-Barr virus, hepatitis C and diffuse large B-cell lymphoma subtype in clinical presentation and survival. Acta Oncol. 2014;53:669–679. doi: 10.3109/0284186X.2013.844853. [DOI] [PubMed] [Google Scholar]
- 14.Burra P., Buda A., Livi U., Rigotti P., Zanus G., Calabrese F., Caforio A., Menin C., Canova D., Farinati F., et al. Occurrence of post-transplant lymphoproliferative disorders among over thousand adult recipients: Any role for hepatitis C infection? Eur. J. Gastroenterol. Hepatol. 2006;18:1065–1070. doi: 10.1097/01.meg.0000231752.50587.ae. [DOI] [PubMed] [Google Scholar]
- 15.Mihaila R.G. Hepatitis C virus-associated B cell non-Hodgkin’s lymphoma. World J. Gastroenterol. 2016;22:6214–6223. doi: 10.3748/wjg.v22.i27.6214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Karuturi M., Shah N., Frank D., Fasan O., Reshef R., Ahya V.N., Bromberg M., Faust T., Goral S., Schuster S.J., et al. Plasmacytic post-transplant lymphoproliferative disorder: A case series of nine patients. Transpl. Int. 2013;26:616–622. doi: 10.1111/tri.12091. [DOI] [PubMed] [Google Scholar]
- 17.Bishnoi R., Bajwa R., Franke A.J., Skelton W.P., 4th, Wang Y., Patel N.M., Slayton W.B., Zou F., Dang N.H. Post-transplant lymphoproliferative disorder (PTLD): Single institutional experience of 141 patients. Exp. Hematol. Oncol. 2017;6:26. doi: 10.1186/s40164-017-0087-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Reshef R., Vardhanabhuti S., Luskin M.R., Heitjan D.F., Hadjiliadis D., Goral S., Krok K.L., Goldberg L.R., Porter D.L., Stadtmauer E.A., et al. Reduction of immunosuppression as initial therapy for posttransplantation lymphoproliferative disorder. Am. J. Transplant. 2011;11:336–347. doi: 10.1111/j.1600-6143.2010.03387.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Yoon S.O., Yu E., Cho Y.M., Suh C., Kim K.M., Han D.J., Lee S.G., Huh J. Post-transplant lymphoproliferative disorders: Clinicopathological analysis of 43 cases in a single center, 1990–2009. Clin. Transplant. 2012;26:67–73. doi: 10.1111/j.1399-0012.2010.01392.x. [DOI] [PubMed] [Google Scholar]
- 20.Martinez-Calle N., Alfonso A., Rifon J., Herrero I., Errasti P., Rábago G., Merino J., Panizo Á., Pardo J., Prósper F., et al. First-line use of rituximab correlates with increased overall survival in late post-transplant lymphoproliferative disorders: Retrospective, single-centre study. Eur. J. Haematol. 2017;98:38–43. doi: 10.1111/ejh.12782. [DOI] [PubMed] [Google Scholar]
- 21.Montanari F., Radeski D., Seshan V., Alobeid B., Bhagat G., O’Connor O.A. Recursive partitioning analysis of prognostic factors in post-transplant lymphoproliferative disorders (PTLD): A 120 case single institution series. Br. J. Haematol. 2015;171:491–500. doi: 10.1111/bjh.13621. [DOI] [PubMed] [Google Scholar]
- 22.Dierickx D., Habermann T.M. Post-Transplantation Lymphoproliferative Disorders in Adults. N. Engl. J. Med. 2018;378:549–562. doi: 10.1056/NEJMra1702693. [DOI] [PubMed] [Google Scholar]
- 23.Lau E., Moyers J.T., Wang B.C., Jeong I.S.D., Lee J., Liu L., Kim M., Villicana R., Kim B., Mitchell J., et al. Analysis of Post-Transplant Lymphoproliferative Disorder (PTLD) Outcomes with Epstein-Barr Virus (EBV) Assessments-A Single Tertiary Referral Center Experience and Review of Literature. Cancers. 2021;13:899. doi: 10.3390/cancers13040899. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Abbas F., El Kossi M., Shaheen I.S., Sharma A., Halawa A. Post-transplantation lymphoproliferative disorders: Current concepts and future therapeutic approaches. World J. Transplant. 2020;10:29–46. doi: 10.5500/wjt.v10.i2.29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Quinlan S.C., Pfeiffer R.M., Morton L.M., Engels E.A. Risk factors for early-onset and late-onset post-transplant lymphoproliferative disorder in kidney recipients in the United States. Am. J. Hematol. 2011;86:206–209. doi: 10.1002/ajh.21911. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Caillard S., Porcher R., Provot F., Dantal J., Choquet S., Durrbach A., Morelon E., Moal V., Janbon B., Alamartine E., et al. Post-transplantation lymphoproliferative disorder after kidney transplantation: Report of a nationwide French registry and the development of a new prognostic score. J. Clin. Oncol. 2013;31:1302–1309. doi: 10.1200/JCO.2012.43.2344. [DOI] [PubMed] [Google Scholar]
- 27.Fararjeh F.A., Mahmood S., Tachtatzis P., Yallop D., Devereux S., Patten P., Agrawal K., Suddle A., O’Grady J., Heaton N., et al. A retrospective analysis of post-transplant lymphoproliferative disorder following liver transplantation. Eur. J. Haematol. 2018;100:98–103. doi: 10.1111/ejh.12988. [DOI] [PubMed] [Google Scholar]
- 28.Bishnoi R., Minish J., Franke A.J., Skelton W.P., 4th, Shah C.P., Wang Y., Dang N.H. Single-institution Retrospective Analysis of Prognostic Factors Influencing Very Late-onset Post-transplant Lymphoproliferative Disorder. Cureus. 2020;12:e6912. doi: 10.7759/cureus.6912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Bustami R.T., Ojo A.O., Wolfe R.A., Merion R.M., Bennett W.M., McDiarmid S.V., Leichtman A.B., Held P.J., Port F.K. Immunosuppression and the risk of post-transplant malignancy among cadaveric first kidney transplant recipients. Am. J. Transplant. 2004;4:87–93. doi: 10.1046/j.1600-6135.2003.00274.x. [DOI] [PubMed] [Google Scholar]
- 30.Francis A., Johnson D.W., Teixeira-Pinto A., Craig J.C., Wong G. Incidence and predictors of post-transplant lymphoproliferative disease after kidney transplantation during adulthood and childhood: A registry study. Nephrol. Dial. Transplant. 2018;33:881–889. doi: 10.1093/ndt/gfx356. [DOI] [PubMed] [Google Scholar]
- 31.Sampaio M.S., Cho Y.W., Shah T., Bunnapradist S., Hutchinson I.V. Association of immunosuppressive maintenance regimens with posttransplant lymphoproliferative disorder in kidney transplant recipients. Transplantation. 2012;93:73–81. doi: 10.1097/TP.0b013e31823ae7db. [DOI] [PubMed] [Google Scholar]
- 32.Taylor E., Jones M., Hourigan M.J., Johnson D.W., Gill D.S., Isbel N., Hawley C.M., Marlton P., Gandhi M.K., Campbell S.B., et al. Cessation of immunosuppression during chemotherapy for post-transplant lymphoproliferative disorders in renal transplant patients. Nephrol. Dial. Transplant. 2015;30:1774–1779. doi: 10.1093/ndt/gfv260. [DOI] [PubMed] [Google Scholar]
- 33.Mucha K., Foroncewicz B., Ziarkiewicz-Wróblewska B., Krawczyk M., Lerut J., Paczek L. Post-transplant lymphoproliferative disorder in view of the new WHO classification: A more rational approach to a protean disease? Nephrol. Dial. Transplant. 2010;25:2089–2098. doi: 10.1093/ndt/gfq231. [DOI] [PubMed] [Google Scholar]
- 34.Trappe R.U., Dierickx D., Zimmermann H., Morschhauser F., Mollee P., Zaucha J.M., Dreyling M.H., Dührsen U., Reinke P., Verhoef G., et al. Response to Rituximab Induction Is a Predictive Marker in B-Cell Post-Transplant Lymphoproliferative Disorder and Allows Successful Stratification into Rituximab or R-CHOP Consolidation in an International, Prospective, Multicenter Phase II Trial. J. Clin. Oncol. 2017;35:536–543. doi: 10.1200/JCO.2016.69.3564. [DOI] [PubMed] [Google Scholar]
- 35.Herreman A., Dierickx D., Morscio J., Camps J., Bittoun E., Verhoef G., De Wolf-Peeters C., Sagaert X., Tousseyn T. Clinicopathological characteristics of posttransplant lymphoproliferative disorders of T-cell origin: Single-center series of nine cases and meta-analysis of 147 reported cases. Leuk. Lymphoma. 2013;54:2190–2199. doi: 10.3109/10428194.2013.775436. [DOI] [PubMed] [Google Scholar]
- 36.Tsai D.E., Bagley S., Reshef R., Shaked A., Bloom R.D., Ahya V., Goldberg L., Chung A., Debonera F., Schuster S.J., et al. The changing face of adult posttransplant lymphoproliferative disorder: Changes in histology between 1999 and 2013. Am. J. Hematol. 2018;93:874–881. doi: 10.1002/ajh.25116. [DOI] [PubMed] [Google Scholar]
- 37.Peters A.C., Akinwumi M.S., Cervera C., Mabilangan C., Ghosh S., Lai R., Iafolla M., Doucette K., Preiksaitis J.K. The Changing Epidemiology of Posttransplant Lymphoproliferative Disorder in Adult Solid Organ Transplant Recipients Over 30 Years: A Single-center Experience. Transplantation. 2018;102:1553–1562. doi: 10.1097/TP.0000000000002146. [DOI] [PubMed] [Google Scholar]
- 38.Gwon J.G., Kim Y.H., Han D.J. Different causes of early and late-onset post transplant lymphoproliferative disorder in kidney transplantation patients after 2000. Asian J. Surg. 2019;42:551–556. doi: 10.1016/j.asjsur.2018.09.002. [DOI] [PubMed] [Google Scholar]
- 39.Ville S., Imbert-Marcille B.M., Coste-Burel M., Garandeau C., Meurette A., Cantarovitch D., Giral M., Hourmant M., Blancho G., Dantal J. Impact of antiviral prophylaxis in adults Epstein-Barr Virus-seronegative kidney recipients on early and late post-transplantation lymphoproliferative disorder onset: A retrospective cohort study. Transpl. Int. 2018;31:484–494. doi: 10.1111/tri.13085. [DOI] [PubMed] [Google Scholar]
- 40.Roberts T.K., Chen X., Liao J.J. Diagnostic and therapeutic challenges of EBV-positive mucocutaneous ulcer: A case report and systematic review of the literature. Exp. Hematol. Oncol. 2015;5:13. doi: 10.1186/s40164-016-0042-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Bieling M., Tischer S., Kalinke U., Blasczyk R., Buus S., Maecker-Kolhoff B., Eiz-Vesper B. Personalized adoptive immunotherapy for patients with EBV-associated tumors and complications: Evaluation of novel naturally processed and presented EBV-derived T-cell epitopes. Oncotarget. 2018;9:4737–4757. doi: 10.18632/oncotarget.23531. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Ghobadi A. Chimeric antigen receptor T cell therapy for non-Hodgkin lymphoma. Curr. Res. Transl. Med. 2018;66:43–49. doi: 10.1016/j.retram.2018.03.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Skorka K., Ostapinska K., Malesa A., Giannopoulos K. The Application of CAR-T Cells in Haematological Malignancies. Arch. Immunol. Ther. Exp. 2020;68:34. doi: 10.1007/s00005-020-00599-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Prockop S., Doubrovina E., Suser S., Heller G., Barker J., Dahi P., Perales M.A., Papadopoulos E., Sauter C., Castro-Malaspina H., et al. Off-the-shelf EBV-specific T cell immunotherapy for rituximab-refractory EBV-associated lymphoma following transplant. J. Clin. Investig. 2019;130:733–747. doi: 10.1172/JCI121127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Schuster S.J., Svoboda J., Chong E.A., Nasta S.D., Mato A.R., Anak Ö., Brogdon J.L., Pruteanu-Malinici I., Bhoj V., Landsburg D., et al. Chimeric Antigen Receptor T Cells in Refractory B-Cell Lymphomas. N. Engl. J. Med. 2017;377:2545–2554. doi: 10.1056/NEJMoa1708566. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Nakid-Cordero C., Baron M., Guihot A., Vieillard V. Natural Killer Cells in Post-Transplant Lymphoproliferative Disorders. Cancers. 2021;13:1836. doi: 10.3390/cancers13081836. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The data presented in this study are available upon request by the authors.