Abstract
Oral squamous cell carcinoma (OSCC) is the most common type of oral cancer in which the consumption of tobacco and alcohol is considered to be the main aetiological factor. Salivary metabolome profiling could identify novel biochemical pathways involved in the pathogenesis of various diseases. This systematic review was designed to answer the question “Are salivary metabolites reliable for the diagnosis of oral squamous cell carcinoma?”. Following the inclusion and exclusion criteria, nineteen studies were included (according to PRISMA statement guidelines). In all included studies, the diagnostic material was unstimulated whole saliva, whose metabolome changes were determined by different spectroscopic methods. At the metabolic level, OSCC patients differed significantly not only from healthy subjects but also from patients with oral leukoplakia, lichen planus or other oral potentially malignant disorders. Among the detected salivary metabolites, there were the indicators of the impaired metabolic pathways, such as choline metabolism, amino acid pathways, polyamine metabolism, urea cycle, creatine metabolism, glycolysis or glycerolipid metabolism. In conclusion, saliva contains many potential metabolites, which can be used reliably to early diagnose and monitor staging in patients with OSCC. However, further investigations are necessary to confirm these findings and to identify new salivary metabolic biomarkers.
Keywords: saliva, metabolomics, metabolome, metabolites, oral squamous cell carcinoma, oral cancer, head and neck squamous cell carcinoma, head and neck cancer, biomarkers, oncological diagnostics
1. Introduction
Oral cancer, a subtype of head and neck cancer, refers to a group of neoplasms affecting lips, oral cavity and oropharynx [1]. It represents one of the most common cancers in the world, with 476,125 new cases and 225,900 deaths in 2020 [2]. Oral squamous cell carcinoma (OSCC) accounts for over 90% of all oral cancer cases. OSCC may arise de novo or from pre-existing oral lesions, such as lichen planus, leukoplakia, erythroplakia and oral submucosal fibrosis, collectively referred to as oral potentially malignant disorders. Moreover, non-healing mucosal ulcerations are linked with the development of OSCC [3,4]. The principal etiological factors for OSCC are tobacco use and alcohol consumption, which have a synergistic carcinogenic effect. In the case of alcohol, smoked tobacco and smokeless tobacco, the odds for OSCC occurrence may be more than 16 times higher compared to people without addictions [5]. In addition, infections of human-papillomavirus (HPV) and Epstein–Barr virus (EBV), poor oral hygiene, dietary and genetic factors are associated with oral cancer development [6,7,8].
Clinically, the most common sites for OSCC are the tongue, floor of the mouth and lips [9]. Although the oral cavity is a relatively accessible site for self-examination and medical inspection, some lesions at the early stage may remain unnoticed or ignored due to the nearly asymptomatic course. Therefore, about 50% of OSCC cases are detected at a late phase (in stages III or IV), which implies a worse prognosis and high mortality rate [10,11,12]. Furthermore, conventional biopsy followed by histopathological analysis is considered the gold standard for the diagnosis of OSCC. However, this diagnostic procedure involves several drawbacks, such as invasiveness, inaccurate sampling caused by tumour heterogeneity and is impractical for large population screening and periodic monitoring of treatment response. It also needs extensive experience and professional equipment [13,14]. Hence, novel, sensitive and less invasive diagnostic tools based on molecular markers are advisable.
Saliva is an important, complex biological fluid containing a broad spectrum of minerals, electrolytes, nucleic acids, proteins, peptides, hormones, enzymes, antimicrobial constituents and other molecules. It provides biomarkers of health and disease status and reflects not only oral cavity diseases, but also systematic conditions. Thus, saliva is referred to as the “mirror of the body”. Additionally, saliva collection is non-invasive, easy and free of stress, constituting a reliable diagnostic medium [15,16,17]. Salivary biomarkers seem to be attractive in oncological diagnostics, especially in oral cancers communicating indirectly with saliva [18].
Previous studies have focused on the analysis of the proteome and transcriptome in serum and saliva samples. Researchers are currently concentrating their attention on novel diagnostics of the metabolome, whose changes reflect the disturbances of the metabolic pathways caused by pathophysiological processes. As the substrates, intermediates and end products of biochemical reactions, the metabolites are small molecules with a molecular weight typically less than 1500 Da [19,20]. The main analytical techniques used for metabolomics investigations are nuclear magnetic resonance (NMR) spectroscopy and mass spectrometry (MS), in combination with gas chromatography (GC), capillary electrophoresis (CE) or high-performance liquid chromatography (HPLC) [21,22].
Extensive salivary metabolite profiling can identify novel biochemical pathways involved in the pathogenesis of various diseases. Salivary metabolomic analysis has provided essential information on oral diseases, especially periodontal disease and oral cancer, as well as on systemic conditions, such as different types of cancer, neurodegenerative disorders, diabetes mellitus, cardiovascular diseases and viral infections [23,24,25,26,27,28,29,30]. Consequently, salivary metabolites could be used as biomarkers for early detection of disease, predicting prognosis or evaluating response to the applied treatment. Moreover, metabolomics may be beneficial for the development of personalised medicine [31]. However, the search for possible markers should take into account possible differences in the composition of metabolites in saliva, which is secreted by the specific salivary glands [32].
The present systematic review was designed in order to answer the question “Are salivary metabolites reliable for the diagnosis of oral squamous cell carcinoma?”, formulated according to the PICO (“population”, “intervention”, “comparison”, “outcome”).
2. Results
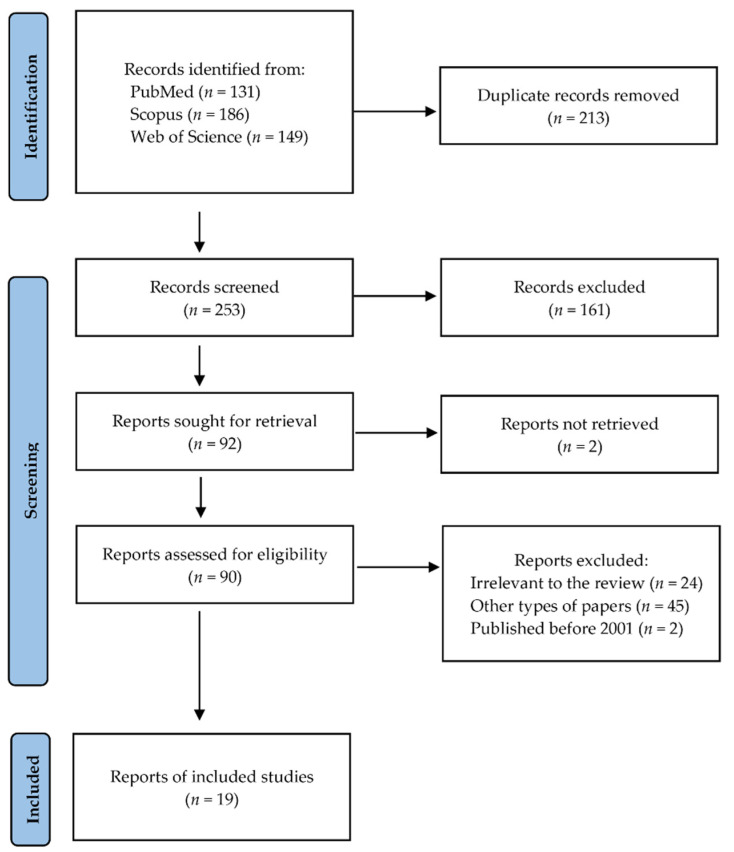
Following the search criteria, our systematic review included nineteen studies, demonstrating data collected in six different countries from a total of 799 participants with diagnosed oral cancer (including 273 females, 506 males and 20 patients without reported gender). Figure 1 shows the detailed selection strategy of the articles. The inclusion and exclusion criteria are presented in the Section 4.
Figure 1.
PRISMA flow diagram presenting search strategy.
From each eligible study included in the present systematic review, we collected data about its general characteristics, such as year of publication and setting, involved participants, diagnosis and tumour–node–metastasis (TNM) staging, inclusion and exclusion criteria and smoking status (Table 1). Table 2 presents the detailed characteristics considering types of saliva, methods of collection, centrifugation, storing and laboratory analysis, as well as potential salivary metabolites for oral cancer. All of the studies took into consideration unstimulated whole saliva samples. Saliva centrifugation methods were rather heterogeneous but the most frequent method of sample storing was freezing at −80 °C. Additionally, predictive parameters for most discriminant metabolites from included studies were reported in Table 3.
Table 1.
General characteristics of included studies.
| Author, Year | Setting | Study Group (F/M); Age | Control Group (F/M); Age | Diagnosis | Inclusion Criteria | Exclusion Criteria | Smoking Status | TNM Stages |
|---|---|---|---|---|---|---|---|---|
| de Sá Alves et al., 2021 [33] | Brazil | 27 (8/19); 57 ± 13.87 (28–88) |
41 (20/21); 57.34 ± 11.66 (31–86) |
OSCC | OSCC: patients over 18 years of age concomitant with the diagnosis of OSCC; Ctrl: patients over 18 years of age, who wanted to participate in the research | OSCC: patients diagnosed with cancer anywhere on the body that had already undergone surgery, radiotherapy or chemotherapy; Ctrl: patients with some type of cancer during their lifetime | OSCC: 20 smokers; Ctrl: 8 smokers, 13 ex-smokers | I-15%, II-15%, III-22%, IV-48% |
| Ishikawa et al., 2016 [34] | Japan | 24 (10/14); 72 (23–94) |
44 (28/16); 68 (21–90) |
OSCC (n = 21), malignant melanoma (n = 2), unknown (n = 1) | NR | OC: prior chemotherapy or radiotherapy; Ctrl: history of prior malignancy or autoimmune disorders | OC: 14 smokers; Ctrl: 9 smokers | I-21%, II-25%, III-33%, IV-21% |
| Ishikawa et al., 2019 [35] | Japan | OSCC: 6 (0/6); 63.5 (49–83), OED: 10 (4/6); 69.0 (57–81), PSOML: 32 (11/21); 62.5 (21–86) |
- | OSCC, OED, PSOML | patients confirmed pathologically by open biopsy | prior chemotherapy or radiotherapy | NR | NR |
| Ishikawa et al., 2020 [36] | Japan | OSCC: 34 (14/20); 70.5 (29–87), OLP: 26 (21/5); 67.5 (34–98) |
- | OSCC, OLP | OSCC patients confirmed pathologically by incisional open biopsy | prior chemotherapy or radiotherapy | NR | I-41.2%, II-26.5%, III-5.9%, IV-26.5% |
| Ishikawa et al., 2022 [37] | Japan | training group: 35 (15/20); 65.0 (26–89), validation group: 37 (19/18); 69 (23–94) | - | OSCC | prior curative treatment, such as radical surgery or chemoradiotherapy, OSCC patients confirmed pathologically by incisional open biopsy | prior non-curative treatment, such as palliative treatment or symptomatic treatment | training group: 2 smokers; validation group: 6 smokers | training group: 0 (CIS)-5.7%, I-45.7%, II-17.1%, III-8.6%, IV-22.9%; validation group: 0 (CIS)-2.7%, I-21.6%, II-21.6%, III-27.0%, IV-27.0% |
| Lohavanichbutr et al., 2018 [38] | USA | First set: 79 (23/56); <50—14 (17.7%), 50–59—24 (30.4%), 60–69—22 (27.8%), >70—19 (24.1%); Second set: 80 (17/63); <50—16 (20%), 50–59—37 (46.3%), 60–69—17 (21.3%), >70—10 (12.5%) | First set: 20 (8/12); <50—13 (65.0%), 50–59—4 (20.0%), 60–69—3 (15.0%), >70—0; Second set: 20 (5/15); <50—13 (65.0%), 50–59—3 (15.0%), 60–69—4 (20.0%), >70—0 | OSCC | Ctrl: patients without OSCC who had oral surgery such as tonsillectomy at the same institutions where the OSCC patients were treated during the same period | NR | First set: 37 current smokers, 42 never/former smokers, Ctrl: 5 current smokers, 9 never/former smokers, 6 unknown; Second set: 28 current smokers, 51 never/former smokers, 1 unknown, Ctrl: 5 current smokers, 12 never/former smokers, 3 unknown | First set: T1/T2-50.6%, T3/T4-49.4%; Second set: T1/T2-68.0%, T3/T4-32.0% |
| Mikkonen et al., 2018 [39] | Brazil | 8 (0/8); 61.7 ± 9.6 (52–76) | 30; 54.4 ± 9.0 (42–74) | HNSCC: larynx (n = 5), oral cavity (n = 3) | NR | NR | HNSCC: 7 smokers; Ctrl: non-smokers | I-12.5%, II-0%, III-37.5%, IV-50% |
| Ohshima et al., 2017 [40] | Japan | 22 (9/13); 68 ± 13 | 21 (13/8); 56 ± 8 | OSCC | NR | OSCC: prior chemotherapy or radiotherapy, history of prior malignancy; Ctrl: history of mucosal diseases in the oral cavity, immunodeficiency, autoimmune disorders, hepatitis or HIV infection | NR | I-31.8%, II-31.8%, III-4.6%, IV-31.8% |
| Rai et al., 2007 [41] | India | 50 (25/25); 17–50 | 24 (11/13); 18–50 | OC | NR | NR | NR | III-100% |
| Shigeyama et al., 2019 [42] | Japan | 12 (7/5); F: 60 ± 16.8, M: 64 ± 19 |
8 (1/7); F: 27, M:28.3 ± 10.3 | OSCC | histologically diagnosed OSCC patients | OSCC: prior chemotherapy, radiotherapy, surgery or alternative remedies before sample collection; Ctrl: history of malignancy, immunodeficiency, underlying diseases | OSCC: 2 smokers, 1 ex-smoker; Ctrl: 1 smoker | I-41.7%, II-50.0%, III-0%, IV-8.33% |
| Song et al., 2020 [43] | China | discovery group: OSCC: 65 (30/35); 35–65, PML: 64 (30/34); 35–65, validation group: OSCC: 60 (30/30); 35–65, PML: 60 (30/30); 35–65 | discovery group: 64 (30/34); 30–60, validation group: 60 (30/30); 30–60 | OSCC, PML | NR | prior therapy | NR | discovery group: I-23.1%, II-32.3%, III-18.4%, IV-26.2%; validation group: I-23.3%, II-31.7%, III-18.3%, IV-26.7% |
| Sridharan et al., 2019 [44] | India | OSCC: 22 (4/18); 43 (39.5–54), OLK: 21 (2/19); 48 (38–54.5) |
21 (7/14); 32 (27.5–45.5) |
OSCC, OLK | OSCC: clinically and histopathologically confirmed OSCC; OLK: clinically diagnosed OLK; Ctrl: normal individuals without any oral lesions, tobacco habits and systemic illnesses | history of systemic illness and medications; history of therapy for OLK and OSCC and with recurrent oral lesions | OSCC: 2 smokers; OLK: 10 smokers | NR |
| Sugimoto et al., 2010 [45] | USA | OC: 69 (23/41/5 missing); 34–87 (59.5) (5 missing) | 87 (27/42/18 missing); 20–75 (43) (2 missing) | OC | diagnosed with primary disease without metastasis | prior chemotherapy, radiotherapy, surgery or alternative therapy, history of prior malignancy, immunodeficiency, autoimmune disorders, hepatitis or HIV infection | NR | NR |
| Supawat et al., 2021 [46] | Thailand | 15; 57.3 ± 8.9 (35–73) |
10; 50.5 ± 10.7 (21–60) |
OC | NR | Ctrl: history of cancer disease | OC: NR; Ctrl: non-smokers | NR |
| Taware et al., 2018 [47] | India | 32 (13/19); 60 (36–82) |
27 (12/15); 55 (33–75) |
OC | minimum 18 years old patient with histopathological confirmation of malignant lesion | OC: anticancer therapeutic intervention; Ctrl: hypertension, diabetes, any medication during last 3 months | OC: 8 smokers; Ctrl: 8 smokers | NR |
| Wang et al., 2014 [48] | China | 30 (5/25); 62 | 60 (25/35) | OSCC | clinical and histopathologic diagnosis | history of receiving medication, prior chemotherapy and radiotherapy | NR | I-23.3%, II-20%, III-6.7%, IV-50% |
| Wang et al., 2014 [49] | China | 30 (5/25); 55 (29–72) |
30 (5/25); 47 (25–69) |
OSCC | clinical and histopathologic diagnosis | history of receiving medication and surgical operation, prior chemotherapy and radiotherapy | NR | I-13.3%, II-30%, III-10%, IV-46.7% |
| Wang et al., 2014 [50] | China | 30 (5/25); 55 (29–72) |
30 (5/25); 47 (25–69) |
OSCC | clinical and histopathologic diagnosis | history of receiving medication and surgical operation, prior chemotherapy and radiotherapy | NR | I-13.3%, II-30%, III-10%, IV-46.7% |
| Wei et al., 2011 [51] | China | OSCC: 37 (11/26); 56 ± 11 (34–77), OLK: 32 (19/13); 60 ± 13 (34–80) | 34 (21/13); 43 ± 14 (21–73) |
OSCC, OLK | clinical and histopathologic diagnosis | history of receiving medication and treatment with topical or systemical steroids | OSCC: 10 smokers, OLK: 9 smokers, Ctrl: 6 smokers | I-24.3%, II-32.4%, III-16.2%, IV-27.1% |
Legend: USA, the United States of America; F, female; M, male; -, not applicable; NR, not reported; Ctrl, control group; OSCC, oral squamous cell carcinoma; OC, oral cancer; OED, oral epithelial dysplasia; PSOML, persistent suspicious oral mucosal lesions; OLP, oral lichen planus; HNSCC, head and neck squamous cell carcinoma; PML, premalignant lesions; OLK, oral leukoplakia; CIS, carcinoma in situ.
Table 2.
Detailed characteristics of included studies considering methods of collection and analysis of saliva.
| Author, Year | Type of Saliva and Method of Collection | Centrifugation and Storing | Method of Analysis | Potential Discriminant Metabolites in Saliva |
|---|---|---|---|---|
| de Sá Alves et al., 2021 [33] | unstimulated whole saliva 3 mL collected in the plastic tubes, which were then hermetically closed, immersed in ice and transported within 1 h to the storage location | stored at −80 °C until analysis | GC-MS | 22 metabolites: up: malic acid, maltose, methionine, inosine, protocatechuic acid, dihydroxyacetone phosphate, galacturonic acid, uracil, isocitric acid, ribose 5-phosphate, o-phospho-serine, indole-3-acetic acid, 2-ketoglutaric acid, pantothenic acid and spermidine; down: lactose, catechol, 2-ketoadipic acid, urea, leucine, margaric acid, palmitic acid and maleic acid |
| Ishikawa et al., 2016 [34] | unstimulated whole saliva 400 μL collected for 5–10 min in a 50 mL Falcon tube on ice; between 8 a.m. and 12 noon | immediately stored at −80 °C | CE-TOF-MS | among 43 significantly elevated metabolites, 17 metabolites also in tissue: up: 3-phosphoglyceric acid, pipecolate, spermidine, methionine, S-adenosylmethionine, 2-aminobenzamide, tryptophan, valine, hypoxanthine, glycylglycine, trimethylamine N-oxide, guanine, guanosine, taurine, choline, cadaverine, threonine |
| Ishikawa et al., 2019 [35] | unstimulated whole saliva 4–5 mL collected for 5–15 min into 50 mL Falcon tubes in a paper cup filled with crushed ice | immediately stored at −80 °C | CE-TOF-MS | 6 metabolites: down: ornithine, carnitine, arginine, o-hydroxybenzoate, N-acetylglucosamine-1-phosphate and ribose 5-phosphate |
| Ishikawa et al., 2020 [36] | unstimulated whole saliva 3 mL collected for 5–10 min into 50 mL Falcon tubes in a paper cup filled with crushed ice | immediately stored at −80 °C | CE-TOF-MS | 14 metabolites: up: trimethylamine N-oxide, putrescine, creatinine, 5-aminovalerate, pipecolate, N-acetylputrescine, gamma-butyrobetaine, indole-3-acetate, N1-acetylspermine, 2’-deoxyinsine, ethanolamine phosphate and N-acetylglucosamine, down: N-acetylhistidine and o-acetylcarnitine |
| Ishikawa et al., 2022 [37] | unstimulated whole saliva 3 mL collected for 5 min into 50 mL Falcon tubes in a paper cup filled with crushed ice | stored at −80 °C | CE-TOF-MS | for predicting overall survival: in the training group identified proline, carnitine, 5-hydroxylysine, 3-methylhistidine, adenosine, inosine and N-acetylglucosamine, in the validation group only 3-methylhistidine (HR = 1.711) |
| Lohavanichbutr et al., 2018 [38] | unstimulated whole saliva into 50 mL sterile conical centrifuge tube and transferred on ice to the laboratory within two hours | centrifuged at 1300× g at 4 °C for 10 min; stored at −80 °C | NMR and LC-MS | 4 metabolites: citrulline and ornithine (only for T1/T2), proline and glycine |
| Mikkonen et al., 2018 [39] | unstimulated whole saliva sample collected into a sterile glass cup for 5 min; between 9 and 11 a.m. | centrifuged at 14,000 rpm for 6 min, stored at −20 °C | NMR spectroscopy | 3 metabolites: up: 1,2 propanediol and fucose, down: proline |
| Ohshima et al., 2017 [40] | unstimulated whole saliva 5 mL collected for 5–10 min into 50 mL tubes, which were placed in a Styrofoam cup filled with crushed; at 8 a.m. | centrifuged at 2600× g for 15 min at 4 °C, and spun for a further 20 min in case of incomplete separation | CE-TOF-MS | 25 metabolites: up: choline, p-hydroxyphenylacetic acid and 2-hydroxy-4-methylvaleric acid (p-value < 0.001), valine, 3-phenyllactic acid, leucine, hexanoic acid, octanoic acid, terephthalic acid, γ-butyrobetaine and 3-(4-hydroxyphenyl)propionic acid (p-value < 0.01), isoleucine, tryptophan, 3-phenylpropionic acid, 2-hydroxyvaleric acid, butyric acid, cadaverine, 2-oxoisovaleric acid, N6,N6,N6-trimethyllysine, taurine, glycolic acid, 3-hydroxybutyric acid, heptanoic acid and alanine (p-value < 0.05); down: urea (p-value < 0.05) |
| Rai et al., 2007 [41] | unstimulated whole saliva collected on ice | centrifuged and frozen at −20 °C until analysis |
HPLC | vitamins E and C (p-value < 0.001) |
| Shigeyama et al., 2019 [42] | unstimulated whole saliva 2 mL, collected in a 10 mL glass bottle over a period of 5–10 min; for at least a period of 5 days between 7 and 10 a.m. | immediately stored at −80 °C | thin-film microextraction based on a ZSM-5/PDMS hybrid film coupled with GC-MS | among 27 volatile metabolites, 12 top metabolites: up: 3-heptanone, 1,3-butanediol, 1,2-pentanediol and 1-hexadecanol, down: ethanol, 2-pentanone, phenol, hexadecanoic acid, undecane, 1-octanol, butyrolactone and benzyl alcohol |
| Song et al., 2020 [43] | unstimulated whole saliva 500 μL, collected into an EP tube | centrifuged at 5000 rpm for 3 min, frozen at −80 °C until analysis | CPSI-MS | among 116 metabolites, top 10 metabolites: up: putrescine, cadaverine, thymidine, adenosine and 5-aminopentoate, down: hippuric acid, phosphocholine, glucose, serine and adrenic acid |
| Sridharan et al., 2019 [44] | unstimulated whole saliva was collected under aseptic conditions by drooling method in a collecting jar | immediately centrifuged and stored at −80 °C before analysis | UPLC-QTOF-MS | 37 upregulated and 11 downregulated metabolites |
| Sugimoto et al., 2010 [45] | unstimulated whole saliva 5 mL for 5–10 min, spitted into 50 mL Falcon tubes, placed in a Styrofoam cup filled with crushed ice | centrifuged at 2600× g for 15 min at 4 °C and spun for 20 min in case of incomplete separation, transferred to two fresh tubes and frozen within 30 min | CE-TOF-MS | 28 metabolites: up: pyrroline hydroxycarboxylic acid, leucine plus isoleucine, choline, tryptophan, valine, threonine, histidine, pipecolic acid, glutamic acid, carnitine, alanine, piperideine, taurine, C4H9N and C8H9N (p-value < 0.001); piperidine, alpha-aminobutyric acid, phenylalanine and C6H6N2O2 (p-value < 0.01); betaine, serine, tyrosine, glutamine, beta-alanine, cadaverine and C5H14N5, down: C4H5N2O11P (p-value < 0.05) |
| Supawat et al., 2021 [46] | unstimulated whole saliva collected on a sterile container kept in an ice pack | immediately stored at −20 °C until analysis | NMR spectroscopy | 13 metabolites: up: trimethylamine N-oxide, taurine, glycine and aspartate, down: propionate, isobutyrate, fucose, cisaconitate, choline, trimethylamine N-oxide, methanol, acetoacetate and glycine |
| Taware et al., 2018 [47] | unstimulated whole saliva 2 mL collected in 10 mL sterilised glass vial with screw cap and immediately placed on ice; between 9 a.m. and 12 at noon | transported to the laboratory within 1 h and stored at −80 °C until analysis |
HS-SPME-GC-MS | among 27 volatile metabolites, 15 top metabolites: 1,4-dichlorobenzene, 1,2-decanediol, 2,5-Bis1,1-dimethylethylphenol, propanoic acid (ethyl ester), E-3-decen-2-ol, acetic acid, propanoic acid, ethyl acetate, 2,4-dimethyl-1-heptene, 1-chloro-2-propanol, 1-chloro-2-butanol, 2-propenoic acid, 2,3,3-trimethylpentane, ethanol, 1,2,3,4-tetrachlorobutane |
| Wang et al., 2014 [48] | unstimulated whole saliva 3 mL kept on ice | centrifuged at 12,000 rpm for 20 min at 4 °C and frozen at −40 °C until analysis |
UPLC-ESI-MS | 2 metabolites: L-phenylalanine and L-leucine |
| Wang et al., 2014 [49] | unstimulated whole saliva 2 mL; between 9 and 11 a.m. | centrifuged at 12,000 rpm for 20 min at 4 °C and frozen at −40 °C until analysis |
HILIC-UPLC-MS | 4 metabolites: choline, betaine, pipecolinic acid and L-carnitine |
| Wang et al., 2014 [50] | unstimulated whole saliva 3 mL; between 9 and 11 a.m. | centrifuged at 12,000 rpm for 20 min at 4 °C and frozen at −40 °C until analysis |
RP-UPLC-MS, HILIC-UPLC-MS | 14 metabolites: up: lactic acid, hydroxyphenyllactic acid, N-nonanoylglycine, 5-hydroxymethyluracil, succinic acid, ornithine, hexanoylcarnitine and propionylcholine; down: carnitine, 4-hydroxy-L-glutamic acid, acetylphenylalanine, sphinganine, phytosphingosine and S-carboxymethyl-L-cysteine |
| Wei et al., 2011 [51] | unstimulated whole saliva; between 9 and 10 a.m. | centrifuged at 3500× g for 20 min at 4 °C and immediately stored at −80 °C until analysis | UPLC-QTOF-MS | among 41 metabolites, 5 top: gamma-aminobutyric acid, phenylalanine, valine, n-eicosanoic acid and lactic acid |
Legend: GC-MS, gas chromatography-mass spectrometry; CE-TOF-MS, capillary electrophoresis time-of-flight mass spectrometry; NMR, nuclear magnetic resonance; LC-MS, liquid chromatography-mass spectrometry; HPLC, high-performance liquid chromatography; PDMS, polydimethylsiloxane; CPSI-MS, conductive polymer spray ionisation-mass spectrometry; UPLC-QTOF-MS, ultra-performance liquid chromatography coupled with quadrupole time of flight mass spectrometry; HS-SPME-GC-MS, headspace solid phase microextraction coupled with gas chromatography-mass spectrometry; UPLC-ESI-MS, ultra-performance liquid chromatography-electrospray ionisation-mass spectrometry; HILIC-UPLC-MS, ultra-performance liquid chromatography-mass spectrometry in hydrophilic interaction chromatography mode; RP-UPLC-MS, reversed-phase ultra-performance liquid chromatography-mass spectrometry; HR, hazard ratio.
Table 3.
Determined predictive parameters for most discriminant metabolites from included studies.
| Study | Most Discriminant Metabolites | AUC | −95% CI | +95% CI | Sensitivity [%] | Specificity [%] |
|---|---|---|---|---|---|---|
| de Sá Alves et al., 2021 [33] | Malic acid | 0.981 | - | - | - | - |
| Lactose | 0.964 | - | - | - | - | |
| Catechol | 0.947 | - | - | - | - | |
| 2-Ketoadipic acid | 0.941 | - | - | - | - | |
| Maltose | 0.934 | - | - | - | - | |
| Methionine | 0.925 | - | - | - | - | |
| Urea | 0.925 | - | - | - | - | |
| Leucine | 0.923 | - | - | - | - | |
| Inosine | 0.922 | - | - | - | - | |
| Protocatechuic acid | 0.911 | - | - | - | - | |
| Ishikawa et al., 2016 [34] | 3-Phosphoglyceric acid | 0.767 | 0.635 | 0.899 | - | - |
| Pipecolate | 0.755 | 0.637 | 0.873 | - | - | |
| Spermidine | 0.751 | 0.626 | 0.876 | - | - | |
| Methionine | 0.744 | 0.628 | 0.861 | - | - | |
| S-adenosylmethionine | 0.743 | 0.613 | 0.874 | - | - | |
| S-adenosylmethionine + pipecolate | 0.827 | 0.726 | 0.928 | - | - | |
| Ishikawa et al., 2019 [35] | Ribose 5-phosphate ** | 0.714 | - | - | - | - |
| Carnitine ** | 0.704 | - | - | - | - | |
| Arginine ** | 0.689 | - | - | - | - | |
| N-Acetylglucosamine1-phosphate ** | 0.682 | - | - | - | - | |
| Ornithine ** | 0.676 | - | - | - | - | |
| Ornithine + o-hydroxybenzoate + ribose 5-phosphate ** |
0.871 | 0.760 | 0.982 | - | - | |
| Ishikawa et al., 2020 [36] | 5-Aminovalerate * | 0.786 | - | - | - | - |
| Indole-3-acetate * | 0.786 | - | - | - | - | |
| Creatinine * | 0.766 | - | - | - | - | |
| Putrescine * | 0.712 | - | - | - | - | |
| N-Acetylglucosamine * | 0.704 | - | - | - | - | |
| Indole-3-acetate + ethanolamine phosphate * | 0.856 | 0.762 | 0.950 | - | - | |
| Mikkonen et al., 2018 [39] | Fucose + glycine + methanol + proline | - | - | - | 87.5 | 93.3 |
| Shigeyama et al., 2019 [42] | 2-Pentanone + undecane + 1,3-butanediol + hexadecanoic acid |
- | - | - | 95.8 | 94.0 |
| Song et al., 2020 [43] | 62 metabolites | 0.992 | 0.978 | 1.000 | 90.0 | 98.3 |
| Sugimoto et al., 2010 [45] | Alanine + choline + “leucine + isoleucine” + glutamic acid + C8H9N + phenylalanine + alpha-aminobutyric acid + serine |
0.865 | - | - | - | - |
| Taware et al., 2018 [47] | 1,4-Dichlorobenzene | 0.998 | - | - | 100.0 | 100.0 |
| 1,2-Decanediol | 0.939 | - | - | 100.0 | 80.0 | |
| 2,5-Bis1,1-dimethylethylphenol | 0.913 | - | - | 90.0 | 80.0 | |
| E-3-Decen-2-ol | 0.889 | - | - | 80.0 | 80.0 | |
| Wang et al., 2014 [48] | L-Phenylalanine ^ | 0.695 | 0.560 | 0.830 | 84.6 | 61.7 |
| L-Leucine ^ | 0.863 | 0.747 | 0.979 | 84.6 | 81.7 | |
| L-Phenylalanine + L-leucine ^ | 0.871 | 0.767 | 0.974 | 92.3 | 81.7 | |
| L-Phenylalanine ^^ | 0.767 | 0.637 | 0.896 | 47.1 | 95.0 | |
| L-Leucine ^^ | 0.852 | 0.748 | 0.956 | 82.4 | 80.0 | |
| L-Phenylalanine + L-leucine^^ | 0.899 | 0.827 | 0.971 | 94.1 | 75.0 | |
| Wang et al., 2014 [49] | Choline ^ | 0.926 | 0.820 | 0.997 | 84.6 | 90.0 |
| Betaine ^ | 0.759 | 0.587 | 0.931 | 46.2 | 96.7 | |
| Pipecolinic acid ^ | 0.994 | 0.981 | 1.000 | 92.3 | 96.7 | |
| L-Carnitine ^ | 0.708 | 0.532 | 0.884 | 73.3 | 61.5 | |
| Choline + betaine + pipecolinic acid + L-carnitine ^ | 0.997 | 0.989 | 1.000 | 100.0 | 96.7 | |
| Choline ^^ | 0.898 | 0.781 | 1.000 | 82.4 | 96.7 | |
| Betaine ^^ | 0.665 | 0.501 | 0.828 | 47.1 | 80.0 | |
| Pipecolinic acid ^^ | 0.914 | 0.798 | 1.000 | 88.2 | 96.7 | |
| L-Carnitine ^^ | 0.731 | 0.563 | 0.900 | 96.7 | 52.9 | |
| Choline + betaine + pipecolinic acid + L-carnitine ^^ | 0.906 | 0.804 | 1.000 | 88.2 | 90.0 | |
| Wang et al., 2014 [50] | Propionylcholine ^ | 0.946 | 0.882 | 1.000 | 76.9 | 96.7 |
| S-carboxymethyl-L-cysteine ^ | 0.913 | 0.822 | 1.000 | 84.6 | 93.3 | |
| Phytosphingosine ^ | 0.910 | 0.816 | 1.000 | 92.3 | 83.3 | |
| Acetylphenylalanine ^ | 0.838 | 0.705 | 0.972 | 92.3 | 76.7 | |
| Sphinganine ^ | 0.818 | 0.660 | 0.976 | 84.6 | 83.3 | |
| Propionylcholine + acetylphenylalanine + sphinganine + phytosphingosine + S-carboxymethyl-L-cysteine ^ |
0.997 | - | - | 100.0 | 96.7 | |
| Propionylcholine + acetylphenylalanine + sphinganine + phytosphingosine + S-carboxymethyl-L-cysteine ^^ |
0.971 | - | - | 86.7 | 94.1 | |
| S-carboxymethyl-L-cysteine ^^ | 0.888 | 0.784 | 0.992 | 88.2 | 90.0 | |
| Phytosphingosine ^^ | 0.875 | 0.776 | 0.973 | 76.5 | 83.3 | |
| Lactic acid ^^ | 0.837 | 0.723 | 0.951 | 100.0 | 73.3 | |
| Propionylcholine ^^ | 0.788 | 0.655 | 0.921 | 64.7 | 80.0 | |
| Succinic acid ^^ | 0.786 | 0.658 | 0.914 | 88.2 | 66.7 | |
| Wei et al., 2011 [51] | Lactic acid | 0.800 | 0.700 | 0.904 | 73.0 | 70.6 |
| Gamma-Aminobutyric acid | 0.560 | 0.423 | 0.698 | 61.8 | 62.2 | |
| Valine | 0.810 | 0.706 | 0.911 | 82.4 | 75.7 | |
| Phenylalanine | 0.640 | 0.508 | 0.765 | 52.9 | 56.8 | |
| n-Eicosadienoic acid | 0.670 | 0.549 | 0.800 | 51.4 | 73.5 | |
| Lactic acid + valine | 0.890 | 0.813 | 0.972 | 86.5 | 82.4 | |
| Lactic acid *** | 0.820 | 0.724 | 0.918 | 73.0 | 75.0 | |
| gamma-Aminobutyric acid *** | 0.750 | 0.636 | 0.869 | 75.0 | 70.3 | |
| Valine *** | 0.830 | 0.736 | 0.925 | 78.1 | 75.8 | |
| Phenylalanine *** | 0.780 | 0.662 | 0.894 | 71.9 | 75.7 | |
| n-Eicosadienoic acid *** | 0.770 | 0.658 | 0.886 | 70.3 | 87.5 | |
| Lactic acid + valine + phenylalanine *** | 0.970 | 0.932 | 1.000 | 94.6 | 84.4 |
Legend: AUC, area under curve; CI, confidence interval; -, not reported; *, vs. oral lichen planus; **, vs. persistent suspicious oral mucosal lesions; ***, vs. leukoplakia; ^, OSCC I-II; ^^, OSCC III-IV.
The summarised quality assessment for each study is reported in Figure 2. The most frequently encountered risks of bias were the absence of data regarding sample size justification (eighteen studies), blinding (eighteen studies) and randomisation (fifteen studies). Critical appraisal was summarised by adding up the points for each criterion of potential risk (points: 1—low, 0.5—unspecified, 0—high). Six studies (31.6%) were classified as having “good” quality (≥80% total score) and thirteen (68.4%) as “intermediate” (≥60% total score).
Figure 2.
Quality assessment, including the main potential risk of bias (risk level: green—low, yellow—unspecified, red—high; quality score: green—good, yellow—intermediate, red—poor).
The level of evidence was assessed using the classification of the Oxford Centre for Evidence-Based Medicine levels for diagnosis [52]. All the included studies have the third or fourth level of evidence (in this 5-graded scale).
3. Discussion
Most of the included studies involved the detection of potential OSCC-specific metabolic markers compared to healthy subjects. The determined markers were diagnostic individually and in combination in patients with oral cancers of different stages.
Ohshima et al. [40] characterised the metabolic changes in saliva samples of Japanese patients with OSCC using capillary electrophoresis-mass spectrometry metabolome analysis. Among the potential twenty-five metabolites, choline showed the largest statistically significant difference between OSCC patients and healthy subjects. In addition to impaired choline metabolism, significant changes in other metabolic pathways were observed, e.g., branched-chain amino acid and aromatic amino acid pathways, polyamine metabolism, urea cycle, creatine metabolism and 3-hydroxybutyric acid metabolism. Virtually all observed changes in the salivary metabolome are associated with synthesis and degradation processes, which are reflected in the excessive proliferation of cancer cells. Moreover, Ishikawa et al. [34] explored salivary metabolites by profiling both saliva and tumour tissue samples for oral cancer screening. Among the detected metabolites, eighty-five were elevated in tumour tissue and forty-three in saliva, of which seventeen markers were common to both kinds of samples. Of these metabolites, based on the multivariate logistic regression modelling, the combination of S-adenosylmethionine and pipecolate demonstrated a high power to discriminate oral cancer patients from the controls. However, there were no significant differences in salivary metabolome according to the stage of OSCC.
In the latest study, Ishikawa et al. [37] evaluated the prognostic role of selected saliva metabolites. The authors randomly divided patients into training and validation groups in order to construct a Cox proportional hazards regression models. Two metabolites (5-hydroxylysine and 3-methylhistidine) were prognostically relevant for overall survival based on the training group. According to the analysis in the validation group, the statistically significant prognostic parameter for the OS was only 3-methylhistidine. For disease-free survival, N-acetylglucosamine appeared to be a potentially relevant prognostic factor based on the training group. However, no statistical significance was reached after adaptation to the model in the validation group. Kaplan–Meier survival curves were determined for the validation group. On this basis, OSCC patients with salivary concentrations of 3-methylhistidine above the median demonstrated significantly lower OS rates than those with the lower ones. Patients with increased salivary levels of N-acetylglucosamine had significantly higher DFS rates. The authors considered that salivary 3-methylhistidine (known as the indicator of sarcopenia status) might be an important factor in predicting the survival prognosis in OSCC patients.
Moreover, Sugimoto et al. [45] compared salivary metabolomes in patients with oral cancer, breast cancer, pancreatic cancer, periodontitis and healthy subjects, using capillary electrophoresis time-of-flight mass spectrometry. The multiple logistic regression model with nine potential metabolic markers for oral cancer patients had a good prediction determined by ROC analysis for differentiation from the controls, but lower than, e.g., the five-element model for pancreatic cancer.
In the study by Supawat et al. [46], salivary trimethylamine N-oxide and glycine were significantly higher in oral cancer patients. Similarly, Lohavanichbutr et al. [38] found the levels of glycine and proline to be significantly decreased in the saliva of OSCC patients. Additionally, four salivary metabolites, including glycine, proline, citrulline and ornithine, were related to early-stage OSCC (T1/T2) in both training and validation sets. However, there were no significant differences in the metabolome depending on the presence of lymph node metastases. Moreover, Mikkonen et al. [39] conducted multivariate discrimination function analysis to identify the combination of metabolites with maximal classification parameters for patients with head and neck squamous cell carcinoma. The highest discrimination power was achieved for fucose, glycine, methanol and proline. Decreased levels of amino acids indicate their overutilisation during carcinogenesis. Surfaces of cancer cells are affected by excessive fucosylation of glycoproteins which leads to uncontrolled tumour growth.
Interestingly, de Sá Alves et al. [33] conducted the first study focused on a group of Latin Americans with OSCC. The findings showed altered metabolic pathways, such as the malate–aspartate shuttle and the beta-alanine metabolism, as well as the Warburg effect. Above the threshold of AUC = 0.9, there were ten salivary metabolites as potential OSCC biomarkers: malic acid, lactose, catechol, 2-ketoadipic acid, maltose, methionine, urea, leucine, inosine and protocatechuic acid. Furthermore, Rai et al. [41] observed significantly lower levels of vitamins E and C in patients with oral cancer compared to healthy subjects. Given the contribution of these vitamins to protecting against free oxygen radicals, the authors recommend that oral cancer patients consider supplementing antioxidants to prevent cytotoxic effects and disease progression.
The specific group of metabolites seem to be the volatile organic metabolites. Volatile organic compounds are present in various biological fluids and can reflect the metabolic changes in response to pathological processes, such as inflammation, necrosis, degeneration or carcinogenesis. Using the method combining thin-film microextraction based on a ZSM-5/polydimethylsiloxane hybrid film coupled with gas chromatography-mass spectrometry, Shigeyama et al. [42] detected thirty-eight VOCs specifically from the OSCC group and thirty-five VOCs overlapping between OSCC patients and healthy subjects. Among the top ten, there were ketones, aldehydes and alcohols, which can be associated with the metabolic processes of cells during tumour formation, including oxidative reactions. The authors also proposed the diagnostic decision tree based on selected VOCs, suggesting that 2-pentanone over 2.11 × 105 is less probable to be related to oral cancer diagnosis. Taware et al. [47] identified altered VOCs levels associated with the metabolic pathways, such as glycolysis or gluconeogenesis, pyruvate metabolism, glycerolipid metabolism, sulphur metabolism, selenoamino acid metabolism, taurine and hypotaurine metabolism, tyrosine metabolism and nicotinate and nicotinamide metabolism. The authors observed four volatile organic metabolites (1,4-dichlorobenzene, 1,2-decanediol, 2,5-bis1,1-dimethylethylphenol and E-3-decen-2-ol) with the highest specificity and sensitivity for class segregation between OC patients and control subjects.
For metabolic dysregulation caused by progressive exacerbation of premalignant lesions to OSCC, Song et al. [43] determined five crucial altered pathways such as aminoacyl tRNA biosynthesis, arginine/proline metabolism, arginine biosynthesis, lysine degradation and histidine metabolism. Unfortunately, similar significant differences in salivary metabolome between different stages of OSCC (from stage I to stage IV) were not found. The authors propose the combination of conductive polymer spray ionisation mass spectrometry (CPSI-MS) and machine learning (ML) as a potentially fast and non-invasive method of early detection of OSCC, with high accuracy of molecular diagnostics.
In three similar studies, Wang et al. [48,49,50] investigated potential salivary metabolic biomarkers to facilitate the early diagnosis of OSCC, using the ultra-performance liquid chromatography-electrospray ionisation-mass spectrometry (UPLC-ESI-MS). All selected metabolites differed in salivary concentrations between OSCC patients and healthy subjects. The two constructed combinations of biomarkers (choline, betaine, pipecolinic acid and L-carnitine; propionylcholine, N-acetyl-L-phenylalanine, sphinganine, phytosphingosine and S-carboxymethyl-L-cysteine) achieved satisfactory predictive parameters, such as accuracy, sensitivity and specificity in the discrimination of early stages of OSCC (I-II) from the controls. Excessive tumour cell proliferation requires an increased level of choline metabolism due to phosphorylation processes. Altered concentrations of betaine and L-carnitine are associated with downregulated fatty acid metabolism, as well as pipecolinic acid with upregulated lysine metabolism. Sphinganine and phytosphingosine are involved in the synthesis and metabolism of ceramide, which participates in the cellular signalling of apoptosis inducing. Considering single metabolic markers, L-leucine could have better predictive power for OSCC with T1/T2, and L-phenylalanine for OSCC with T3/T4. L-leucine stimulates protein synthesis and reduces protein breakdown, and L-phenylalanine is an essential precursor of tyrosine and catecholamines. When combined, both amino acids showed improved sensitivity and specificity for early detection of OSCC. The successfully presented innovative possibility of using salivary metabolites in a non-invasive and simple way can allow creating the clinical screening tool for the early diagnosis of OSCC.
Several studies have focused on the attempt to differentiate OSCC from other lesions of the oral mucosa, which may be considered oral potentially malignant disorders, based on changes in salivary metabolome. This is particularly important for oncological prevention and early detection of lesions. Wei et al. [51] selected the panel of five salivary metabolites (γ-aminobutyric acid, phenylalanine, valine, n-eicosanoic acid and lactic acid) and assessed their stratification value by ROC curves analyses. On this basis, 2–3-item combinations were created, the predictive power for OSCC was satisfactory in relation to patients with oral leukoplakia (OLK) and no changes in the oral mucosa. For OLK, the combination of valine, lactic acid and phenylalanine had higher parameters discriminating from OSCC (accuracy, sensitivity, specificity, positive predictive value). Higher levels of lactic acid and lower levels of amino acids are associated with increased glycolysis and impaired tricarboxylic acid cycle present in the cancer tissues during cell proliferation. The authors suggest that these findings could help complement the clinical differentiation of OSCC from OLK, improving prognosis with earlier detection. Similarly, Sridharan et al. [44] evaluated the clinical utility of salivary metabolites in oral leukoplakia and OSCC diagnosis. Both in OLK and OSCC patients, salivary metabolites such as sphinganine-1-phosphate, pseudouridine, 4-nitroquinolone-1-oxide, inositol 1,3,4-triphosphate, 2-phospho-glycerate, 1-methylhistidine, 2-oxoarginine, norcocaine nitroxide, L-isoleucine, gamma-aminobutyryllysine, L-homocysteic acid and ubiquinone were significantly upregulated. Compared to oral leukoplakia, a significant upregulation of D-glycerate-2-phosphate, estrone-3-glucoronide, 4-nitroquinolone-1-oxide, sphinganine-1-phosphate, 1-methylhistidine, inositol 1,3,4-triphosphate, d-glycerate-2-phosphate, 2-oxoarginine, norcocaine nitroxide and pseudouridine was observed for OSCC.
Ishikawa et al. [35] determined salivary metabolite markers to differentiate patients with oral squamous cell carcinoma and oral epithelial dysplasia (OSCC/OED) from those with persistent suspicious oral mucosal lesions (PSOML). From six significantly elevated metabolites in PSOML, ornithine, o-hydroxybenzoate and ribose 5-phosphate were selected in the multivariate logistic regression model, which together resulted in a high AUC value for the ROC curve discriminating from OSCC/OED patients. Ornithine, as the intermediate metabolite in the urea cycle, is the precursor of polyamines (e.g., putrescine), which are well known metabolic markers for various neoplasms. The Warburg effect is indicated by the reduction in intermediate metabolites in the pentose phosphate pathway, such as ribose 5-phosphate. According to the authors’ knowledge, it was the first study comparing both types of lesions on the oral mucosa.
Moreover, Ishikawa et al. [36] conducted another study to identify salivary metabolites for the discrimination of OSCC from oral lichen planus. Among fourteen metabolites significantly different between both groups, the combination of indole-3-acetate and ethanolamine phosphate showed a high AUC for discriminating OSCC from OLP. The first is produced by cancer tissue during the growth of malignant cells, and the second is the intermediate metabolite of the phospholipid metabolism involved in tumour progression. The authors suggest the potential of salivary metabolites for non-invasive screening of OSCC versus OLP.
Our systematic review collects and discusses the most recent information on the very fast-growing and promising diagnosis of salivary metabolites in oral cancer patients. Many metabolic markers still have an unknown significance for physiological and pathological processes. There is no doubt that saliva is a diagnostic material that is easy to collect and relatively simple to process for analytical diagnostics. However, changes in saliva composition, particularly metabolites, can be very dynamic and depend on many factors such as oral health status, the current microbiome activity and dietary habits. The stability of the salivary metabolome may also be affected by external factors such as collection or processing temperature and duration. Therefore, when testing such dynamic components, there is a risk of impaired diagnostic accuracy. Limitations of our systematic review include the heterogeneity of the included studies in terms of study design, clinical characteristics of patients with tumour progression, and laboratory methods. Not all authors explicitly stated the histopathological diagnosis of OSCC, although it can be assumed that the majority of these patients had the most common form of oral cancers. Similarly, despite the diversity of detected markers that disrupt numerous metabolic pathways, only some researchers reported statistical parameters to assess the diagnostic accuracy for these metabolites, which only allowed for the qualitative analysis of the extracted data.
4. Materials and Methods
4.1. Search Strategy and Data Extraction
A systematic review was conducted up to 7 February 2022, according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement guidelines [53], using the databases PubMed, Scopus and Web of Science. The search formulas included:
-
-
For PubMed: (((oral OR (head and neck)) AND (cancer OR carcinoma)) OR OSCC) AND saliva AND (metabolite OR metabolomics);
-
-
For Scopus: TITLE-ABS-KEY((((oral OR “head and neck”) AND (cancer OR carcinoma)) OR OSCC) AND saliva AND (metabolite OR metabolomics));
-
-
For Web of Science: TS = ((((oral OR (head and neck)) AND (cancer OR carcinoma)) OR OSCC) AND saliva AND (metabolite OR metabolomics)).
Records were screened by the title, abstract and full text by two independent investigators. Studies included in this review matched all the predefined criteria according to PICOS (“Population”, “Intervention”, “Comparison”, “Outcomes” and “Study design”), as shown in Table 4. A detailed search flowchart is presented in the Section 2. The study protocol was registered in International prospective register of systematic reviews PROSPERO (CRD42022312946).
Table 4.
Inclusion and exclusion criteria according to the PICOS.
| Parameter | Inclusion Criteria | Exclusion Criteria |
|---|---|---|
| Population | patients with oral cancer—aged from 0 to 99 years, both sexes | patients with other neoplasms |
| Intervention | not applicable | |
| Comparison | not applicable | |
| Outcomes | salivary metabolites as markers | other salivary components as markers |
| Study design | case-control, cohort and cross-sectional studies | literature reviews, case reports, expert opinion, letters to the editor, conference reports |
| published after 2000 | not published in English |
4.2. Quality Assessment and Critical Appraisal for the Systematic Review of Included Studies
The risk of bias in each individual study was assessed according to the “Study Quality Assessment Tool” issued by the National Heart, Lung, and Blood Institute within the National Institute of Health [54]. These questionnaires were answered by two independent investigators, and any disagreements were resolved by discussion between them.
5. Conclusions
According to our systematic review, saliva contains many potential metabolites, which can be used reliably to early diagnose and monitor staging in patients with OSCC. However, further investigations are necessary to confirm these findings and to identify new salivary metabolic biomarkers.
Author Contributions
Conceptualisation, K.N.; methodology, K.N.; formal analysis, K.N. and D.G.; investigation and data curation, K.N., D.G. and D.K.; writing—original draft preparation, K.N. and D.G.; writing—review and editing, K.N. and A.S.; visualisation, K.N.; supervision, A.S. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Data are available on request from the corresponding author. The data are not publicly available due to this is a systematic review (not an original article), so the database is in Excel and contains the data already displayed in most Tables in our manuscript.
Conflicts of Interest
The authors declare no conflict of interest.
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Inchingolo F., Santacroce L., Ballini A., Topi S., Dipalma G., Haxhirexha K., Bottalico L., Charitos I.A. Oral Cancer: A Historical Review. Int. J. Environ. Res. Public Health. 2020;17:3168. doi: 10.3390/ijerph17093168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Sung H., Ferlay J., Siegel R.L., Laversanne M., Soerjomataram I., Jemal A., Bray F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021;71:209–249. doi: 10.3322/caac.21660. [DOI] [PubMed] [Google Scholar]
- 3.Chen X., Yu D. Metabolomics Study of Oral Cancers. Metab. Off. J. Metab. Soc. 2019;15:22. doi: 10.1007/s11306-019-1483-8. [DOI] [PubMed] [Google Scholar]
- 4.Gilligan G.-M., Panico R.-L., Di Tada C., Piemonte E.-D., Brunotto M.-N. Clinical and Immunohistochemical Epithelial Profile of Non-Healing Chronic Traumatic Ulcers. Med. Oral Patol. Oral Cir. Bucal. 2020;25:e706–e713. doi: 10.4317/medoral.23729. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Mello F.W., Melo G., Pasetto J.J., Silva C.A.B., Warnakulasuriya S., Rivero E.R.C. The Synergistic Effect of Tobacco and Alcohol Consumption on Oral Squamous Cell Carcinoma: A Systematic Review and Meta-Analysis. Clin. Oral Investig. 2019;23:2849–2859. doi: 10.1007/s00784-019-02958-1. [DOI] [PubMed] [Google Scholar]
- 6.Dhanuthai K., Rojanawatsirivej S., Thosaporn W., Kintarak S., Subarnbhesaj A., Darling M., Kryshtalskyj E., Chiang C.-P., Shin H.-I., Choi S.-Y., et al. Oral Cancer: A Multicenter Study. Med. Oral Patol. Oral Cir. Bucal. 2018;23:e23–e29. doi: 10.4317/medoral.21999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Anwar N., Pervez S., Chundriger Q., Awan S., Moatter T., Ali T.S. Oral Cancer: Clinicopathological Features and Associated Risk Factors in a High Risk Population Presenting to a Major Tertiary Care Center in Pakistan. PLoS ONE. 2020;15:e0236359. doi: 10.1371/journal.pone.0236359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Markopoulos A.K. Current Aspects on Oral Squamous Cell Carcinoma. Open Dent. J. 2012;6:126–130. doi: 10.2174/1874210601206010126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Rivera C. Essentials of Oral Cancer. Int. J. Clin. Exp. Pathol. 2015;8:11884–11894. [PMC free article] [PubMed] [Google Scholar]
- 10.Pillai J., Chincholkar T., Dixit R., Pandey M. A Systematic Review of Proteomic Biomarkers in Oral Squamous Cell Cancer. World J. Surg. Oncol. 2021;19:315. doi: 10.1186/s12957-021-02423-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Scott S.E., Grunfeld E.A., McGurk M. The Idiosyncratic Relationship between Diagnostic Delay and Stage of Oral Squamous Cell Carcinoma. Oral Oncol. 2005;41:396–403. doi: 10.1016/j.oraloncology.2004.10.010. [DOI] [PubMed] [Google Scholar]
- 12.Gómez I., Seoane J., Varela-Centelles P., Diz P., Takkouche B. Is Diagnostic Delay Related to Advanced-Stage Oral Cancer? A Meta-Analysis. Eur. J. Oral Sci. 2009;117:541–546. doi: 10.1111/j.1600-0722.2009.00672.x. [DOI] [PubMed] [Google Scholar]
- 13.Yakob M., Fuentes L., Wang M.B., Abemayor E., Wong D.T.W. Salivary Biomarkers for Detection of Oral Squamous Cell Carcinoma-Current State and Recent Advances. Curr. Oral Health Rep. 2014;1:133–141. doi: 10.1007/s40496-014-0014-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Singh P., Verma J.K., Singh J.K. Validation of Salivary Markers, IL-1β, IL-8 and Lgals3bp for Detection of Oral Squamous Cell Carcinoma in an Indian Population. Sci. Rep. 2020;10:7365. doi: 10.1038/s41598-020-64494-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Nijakowski K., Surdacka A. Salivary Biomarkers for Diagnosis of Inflammatory Bowel Diseases: A Systematic Review. Int. J. Mol. Sci. 2020;21:7477. doi: 10.3390/ijms21207477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Motamayel F.A., Davoodi P., Dalband M., Hendi S.S. Saliva as a Mirror of the Body Health. Avicenna J. Dent. Res. 2018;1:41–55. [Google Scholar]
- 17.Chiappin S., Antonelli G., Gatti R., De Palo E.F. Saliva Specimen: A New Laboratory Tool for Diagnostic and Basic Investigation. Clin. Chim. Acta Int. J. Clin. Chem. 2007;383:30–40. doi: 10.1016/j.cca.2007.04.011. [DOI] [PubMed] [Google Scholar]
- 18.Spielmann N., Wong D.T. Saliva: Diagnostics and Therapeutic Perspectives. Oral Dis. 2011;17:345–354. doi: 10.1111/j.1601-0825.2010.01773.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Chaleckis R., Meister I., Zhang P., Wheelock C.E. Challenges, Progress and Promises of Metabolite Annotation for LC-MS-Based Metabolomics. Curr. Opin. Biotechnol. 2019;55:44–50. doi: 10.1016/j.copbio.2018.07.010. [DOI] [PubMed] [Google Scholar]
- 20.Hynne H., Sandås E.M., Elgstøen K.B.P., Rootwelt H., Utheim T.P., Galtung H.K., Jensen J.L. Saliva Metabolomics in Dry Mouth Patients with Head and Neck Cancer or Sjögren’s Syndrome. Cells. 2022;11:323. doi: 10.3390/cells11030323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Iwasaki Y., Sawada T., Hatayama K., Ohyagi A., Tsukuda Y., Namekawa K., Ito R., Saito K., Nakazawa H. Separation Technique for the Determination of Highly Polar Metabolites in Biological Samples. Metabolites. 2012;2:496–515. doi: 10.3390/metabo2030496. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Segers K., Declerck S., Mangelings D., Heyden Y.V., Eeckhaut A.V. Analytical Techniques for Metabolomic Studies: A Review. Bioanalysis. 2019;11:2297–2318. doi: 10.4155/bio-2019-0014. [DOI] [PubMed] [Google Scholar]
- 23.Ussher J.R., Elmariah S., Gerszten R.E., Dyck J.R.B. The Emerging Role of Metabolomics in the Diagnosis and Prognosis of Cardiovascular Disease. J. Am. Coll. Cardiol. 2016;68:2850–2870. doi: 10.1016/j.jacc.2016.09.972. [DOI] [PubMed] [Google Scholar]
- 24.Hyvärinen E., Savolainen M., Mikkonen J.J.W., Kullaa A.M. Salivary Metabolomics for Diagnosis and Monitoring Diseases: Challenges and Possibilities. Metabolites. 2021;11:587. doi: 10.3390/metabo11090587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Yilmaz A., Geddes T., Han B., Bahado-Singh R.O., Wilson G.D., Imam K., Maddens M., Graham S.F. Diagnostic Biomarkers of Alzheimer’s Disease as Identified in Saliva Using 1H NMR-Based Metabolomics. J. Alzheimers Dis. 2017;58:355–359. doi: 10.3233/JAD-161226. [DOI] [PubMed] [Google Scholar]
- 26.Meoni G., Lorini S., Monti M., Madia F., Corti G., Luchinat C., Zignego A.L., Tenori L., Gragnani L. The Metabolic Fingerprints of HCV and HBV Infections Studied by Nuclear Magnetic Resonance Spectroscopy. Sci. Rep. 2019;9:4128. doi: 10.1038/s41598-019-40028-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Citterio F., Romano F., Meoni G., Iaderosa G., Grossi S., Sobrero A., Dego F., Corana M., Berta G.N., Tenori L., et al. Changes in the Salivary Metabolic Profile of Generalized Periodontitis Patients after Non-Surgical Periodontal Therapy: A Metabolomic Analysis Using Nuclear Magnetic Resonance Spectroscopy. J. Clin. Med. 2020;9:3977. doi: 10.3390/jcm9123977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.de Oliveira L.R.P., Martins C., Fidalgo T.K.S., Freitas-Fernandes L.B., de Oliveira Torres R., Soares A.L., Almeida F.C.L., Valente A.P., de Souza I.P.R. Salivary Metabolite Fingerprint of Type 1 Diabetes in Young Children. J. Proteome Res. 2016;15:2491–2499. doi: 10.1021/acs.jproteome.6b00007. [DOI] [PubMed] [Google Scholar]
- 29.Gonzalez-Covarrubias V., Martínez-Martínez E., del Bosque-Plata L. The Potential of Metabolomics in Biomedical Applications. Metabolites. 2022;12:194. doi: 10.3390/metabo12020194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Rapado-González Ó., Majem B., Muinelo-Romay L., López-López R., Suarez-Cunqueiro M.M. Cancer Salivary Biomarkers for Tumours Distant to the Oral Cavity. Int. J. Mol. Sci. 2016;17:1531. doi: 10.3390/ijms17091531. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Klupczyńska A., Dereziński P., Kokot Z.J. Metabolomics in Medical Sciences-Trends, Challenges and Perspectives. Acta Pol. Pharm. 2015;72:629–641. [PubMed] [Google Scholar]
- 32.Meleti M., Quartieri E., Antonelli R., Pezzi M.E., Ghezzi B., Viani M.V., Setti G., Casali E., Ferrari E., Ciociola T., et al. Metabolic Profiles of Whole, Parotid and Submandibular/Sublingual Saliva. Metabolites. 2020;10:318. doi: 10.3390/metabo10080318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.de Sá Alves M., de Sá Rodrigues N., Bandeira C.M., Chagas J.F.S., Pascoal M.B.N., Nepomuceno G.L.J.T., da Silva Martinho H., Alves M.G.O., Mendes M.A., Dias M., et al. Identification of Possible Salivary Metabolic Biomarkers and Altered Metabolic Pathways in South American Patients Diagnosed with Oral Squamous Cell Carcinoma. Metabolites. 2021;11:650. doi: 10.3390/metabo11100650. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Ishikawa S., Sugimoto M., Kitabatake K., Sugano A., Nakamura M., Kaneko M., Ota S., Hiwatari K., Enomoto A., Soga T., et al. Identification of Salivary Metabolomic Biomarkers for Oral Cancer Screening. Sci. Rep. 2016;6:31520. doi: 10.1038/srep31520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Ishikawa S., Wong D.T.W., Sugimoto M., Gleber-Netto F.O., Li F., Tu M., Zhang Y., Akin D., Iino M. Identification of Salivary Metabolites for Oral Squamous Cell Carcinoma and Oral Epithelial Dysplasia Screening from Persistent Suspicious Oral Mucosal Lesions. Clin. Oral Investig. 2019;23:3557–3563. doi: 10.1007/s00784-018-2777-3. [DOI] [PubMed] [Google Scholar]
- 36.Ishikawa S., Sugimoto M., Edamatsu K., Sugano A., Kitabatake K., Iino M. Discrimination of Oral Squamous Cell Carcinoma from Oral Lichen Planus by Salivary Metabolomics. Oral Dis. 2020;26:35–42. doi: 10.1111/odi.13209. [DOI] [PubMed] [Google Scholar]
- 37.Ishikawa S., Sugimoto M., Konta T., Kitabatake K., Ueda S., Edamatsu K., Okuyama N., Yusa K., Iino M. Salivary Metabolomics for Prognosis of Oral Squamous Cell Carcinoma. Front. Oncol. 2022;11:789248. doi: 10.3389/fonc.2021.789248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Lohavanichbutr P., Zhang Y., Wang P., Gu H., Nagana Gowda G.A., Djukovic D., Buas M.F., Raftery D., Chen C. Salivary Metabolite Profiling Distinguishes Patients with Oral Cavity Squamous Cell Carcinoma from Normal Controls. PLoS ONE. 2018;13:e0204249. doi: 10.1371/journal.pone.0204249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Mikkonen J.J.W., Singh S.P., Akhi R., Salo T., Lappalainen R., González-Arriagada W.A., Ajudarte Lopes M., Kullaa A.M., Myllymaa S. Potential Role of Nuclear Magnetic Resonance Spectroscopy to Identify Salivary Metabolite Alterations in Patients with Head and Neck Cancer. Oncol. Lett. 2018;16:6795–6800. doi: 10.3892/ol.2018.9419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Ohshima M., Sugahara K., Kasahara K., Katakura A. Metabolomic Analysis of the Saliva of Japanese Patients with Oral Squamous Cell Carcinoma. Oncol. Rep. 2017;37:2727–2734. doi: 10.3892/or.2017.5561. [DOI] [PubMed] [Google Scholar]
- 41.Rai B., Kharb S., Jain R., Anand S.C. Salivary Vitamins E and C in Oral Cancer. Redox Rep. 2007;12:163–164. doi: 10.1179/135100007X200245. [DOI] [PubMed] [Google Scholar]
- 42.Shigeyama H., Wang T., Ichinose M., Ansai T., Lee S.-W. Identification of Volatile Metabolites in Human Saliva from Patients with Oral Squamous Cell Carcinoma via Zeolite-Based Thin-Film Microextraction Coupled with GC-MS. J. Chromatogr. B Analyt. Technol. Biomed. Life. Sci. 2019;1104:49–58. doi: 10.1016/j.jchromb.2018.11.002. [DOI] [PubMed] [Google Scholar]
- 43.Song X., Yang X., Narayanan R., Shankar V., Ethiraj S., Wang X., Duan N., Ni Y.-H., Hu Q., Zare R.N. Oral Squamous Cell Carcinoma Diagnosed from Saliva Metabolic Profiling. Proc. Natl. Acad. Sci. USA. 2020;117:16167–16173. doi: 10.1073/pnas.2001395117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Sridharan G., Ramani P., Patankar S., Vijayaraghavan R. Evaluation of Salivary Metabolomics in Oral Leukoplakia and Oral Squamous Cell Carcinoma. J. Oral Pathol. Med. Off. Publ. Int. Assoc. Oral Pathol. Am. Acad. Oral Pathol. 2019;48:299–306. doi: 10.1111/jop.12835. [DOI] [PubMed] [Google Scholar]
- 45.Sugimoto M., Wong D.T., Hirayama A., Soga T., Tomita M. Capillary Electrophoresis Mass Spectrometry-Based Saliva Metabolomics Identified Oral, Breast and Pancreatic Cancer-Specific Profiles. Metab. Off. J. Metab. Soc. 2010;6:78–95. doi: 10.1007/s11306-009-0178-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Supawat B., Aye K.T., Ritpanja J., Nueangwong W., Kothan S., Pan J., Tungjai M. Differences in Spectroscopic Properties of Saliva Taken From Normal Subjects and Oral Cancer Patients: Comparison Studies. J. Fluoresc. 2021;31:747–754. doi: 10.1007/s10895-021-02707-2. [DOI] [PubMed] [Google Scholar]
- 47.Taware R., Taunk K., Pereira J.A.M., Shirolkar A., Soneji D., Câmara J.S., Nagarajaram H.A., Rapole S. Volatilomic Insight of Head and Neck Cancer via the Effects Observed on Saliva Metabolites. Sci. Rep. 2018;8:17725. doi: 10.1038/s41598-018-35854-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Wang Q., Gao P., Cheng F., Wang X., Duan Y. Measurement of Salivary Metabolite Biomarkers for Early Monitoring of Oral Cancer with Ultra Performance Liquid Chromatography-Mass Spectrometry. Talanta. 2014;119:299–305. doi: 10.1016/j.talanta.2013.11.008. [DOI] [PubMed] [Google Scholar]
- 49.Wang Q., Gao P., Wang X., Duan Y. Investigation and Identification of Potential Biomarkers in Human Saliva for the Early Diagnosis of Oral Squamous Cell Carcinoma. Clin. Chim. Acta Int. J. Clin. Chem. 2014;427:79–85. doi: 10.1016/j.cca.2013.10.004. [DOI] [PubMed] [Google Scholar]
- 50.Wang Q., Gao P., Wang X., Duan Y. The Early Diagnosis and Monitoring of Squamous Cell Carcinoma via Saliva Metabolomics. Sci. Rep. 2014;4:6802. doi: 10.1038/srep06802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Wei J., Xie G., Zhou Z., Shi P., Qiu Y., Zheng X., Chen T., Su M., Zhao A., Jia W. Salivary Metabolite Signatures of Oral Cancer and Leukoplakia. Int. J. Cancer. 2011;129:2207–2217. doi: 10.1002/ijc.25881. [DOI] [PubMed] [Google Scholar]
- 52.OCEBM Levels of Evidence. [(accessed on 22 August 2020)]. Available online: https://www.cebm.net/2016/05/ocebm-levels-of-evidence/
- 53.Page M.J., McKenzie J.E., Bossuyt P.M., Boutron I., Hoffmann T.C., Mulrow C.D., Shamseer L., Tetzlaff J.M., Akl E.A., Brennan S.E., et al. The PRISMA 2020 Statement: An Updated Guideline for Reporting Systematic Reviews. BMJ. 2021;372:n71. doi: 10.1136/bmj.n71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Study Quality Assessment Tools | NHLBI, NIH. [(accessed on 22 August 2020)]; Available online: https://www.nhlbi.nih.gov/health-topics/study-quality-assessment-tools.
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Data are available on request from the corresponding author. The data are not publicly available due to this is a systematic review (not an original article), so the database is in Excel and contains the data already displayed in most Tables in our manuscript.