Abstract
The main neurotransmitter in the brain responsible for the inhibition of neuronal activity is γ-aminobutyric acid (GABA). It plays a crucial role in circuit formation during development, both via its primary effects as a neurotransmitter and also as a trophic factor. The GABAB receptors (GABABRs) are G protein-coupled metabotropic receptors; on one hand, they can influence proliferation and migration; and, on the other, they can inhibit cells by modulating the function of K+ and Ca2+ channels, doing so on a slower time scale and with a longer-lasting effect compared to ionotropic GABAA receptors. GABABRs are expressed pre- and post-synaptically, at both glutamatergic and GABAergic terminals, thus being able to shape neuronal activity, plasticity, and the balance between excitatory and inhibitory synaptic transmission in response to varying levels of extracellular GABA concentration. Furthermore, given their subunit composition and their ability to form complexes with several associated proteins, GABABRs display heterogeneity with regard to their function, which makes them a promising target for pharmacological interventions. This review will describe (i) the latest results concerning GABABRs/GABABR-complex structures, their function, and the developmental time course of their appearance and functional integration in the brain, (ii) their involvement in manifestation of various pathophysiological conditions, and (iii) the current status of preclinical and clinical studies involving GABABR-targeting drugs.
Keywords: GABA receptors, GABAB receptors, development
1. Introduction
γ-aminobutyric acid (GABA) is the main neurotransmitter in the central nervous system that in adult age mediates the inhibition of neurons by acting on two classes of receptors. The activation of ionotropic receptors—GABAA and GABAC—leads to a flux of chloride ions in accordance with the driving force. Metabotropic GABAB receptors (GABABRs) are instead associated with Gi proteins, which lead to the inhibition of neuronal activity following activation.
During early development, many processes related to circuit formation must be dynamically fine-tuned and coordinated [1]. In this period, the inhibitory system undergoes prominent changes. The maturation of the inhibitory system is in fact a dynamic process that is crucial for correct brain functioning [2]. GABAergic neurons in the neocortex originate from the subpallium [3] and proceed to invade the pallium. Here, they shape the formation of—and integrate in—the existing circuit and, in rodents, they reach a mature state by postnatal day 30 [4,5]. Interneurons have an active role in development by influencing network activity [6,7], but their contribution is not limited to this aspect. In fact, GABA not only has an effect as a neurotransmitter, it also has a neurotrophic function on cell growth and network formation [8,9].
The role of GABAA receptors (GABAARs) in development has been revealed by many studies, as summarized by excellent reviews on this topic [2,10]. However, GABABRs have remained, in comparison, less investigated in this context despite the clear involvement they have in several processes such as learning and memory [11] or the shaping of neuronal circuits [12]. The dynamic adjustment of these receptors during development and how alterations in their function can affect brain growth are becoming emergent topics [13,14]. Recent technical advancements allow a detailed understanding of the structure of GABABRs. This will enable a more precise pharmacological modulation of GABABRs with the possibility of investigating them at a much deeper level [15]. This review article covers recent discoveries concerning GABABRs, their role during development, and current as well as potential future therapeutic applications.
2. GABABRs: Structure and Function
The inhibitory effect of GABA in the CNS has been observed and described for more than 50 years [16,17]. Dr. Norman Bowery was the first to describe a class of GABA receptors that could reduce the release of neurotransmitters and that were not sensitive to isoguvacine or bicuculline, thus distinguishing them from the established GABA receptor, naming the newly found GABAB and the previously known GABAA [18]. A functional GABABR is constituted of an obligatory heterodimer of the GABAB1 and GABAB2 subunits; GABAB1 is required for ligand-binding and GABAB2 is necessary for interactions with G proteins as well as increasing the affinity of GABAB1 to GABA [19,20]. The common domains between the two subunits are a C-terminal intracellular domain, a heptahelical transmembrane domain, and a Venus flytrap domain on the extracellular side that is connected by a stalk (Figure 1A). The GABAB1 receptor contains an endoplasmic reticulum (ER) retention tag that is masked following interaction with GABAB2, thus allowing for correct receptor transport [21]. Although GABAB1 possesses several splice variants, the most common are GABAB1a and GABAB1b. The main structural difference between the two is the presence of two sushi domains on GABAB1a [22], which influences the transport of the receptor and thus makes GABAB(1a,2) more stable in the pre-synaptic site and in the dendritic compartments, while GABAB(1b,2) is responsible for post-synaptic inhibition in spines (Figure 1A) [23].
Figure 1.
Composition and main functions of GABABRs. (A) Schematic representation of the subunit structure (top row) and of heterodimers (bottom row). (B) Diagram illustrating activation of a heterodimer, including a G protein and downstream effectors. The G protein subunits can inhibit the activity of adenylyl cyclase, thus reducing the levels of cAMP and of Ca2+ channels. Another consequence is the activation of GIRKs, which can be modulated by KCTDs. Associated proteins such as APP and PIANP are also included in the scheme (for details, see text).
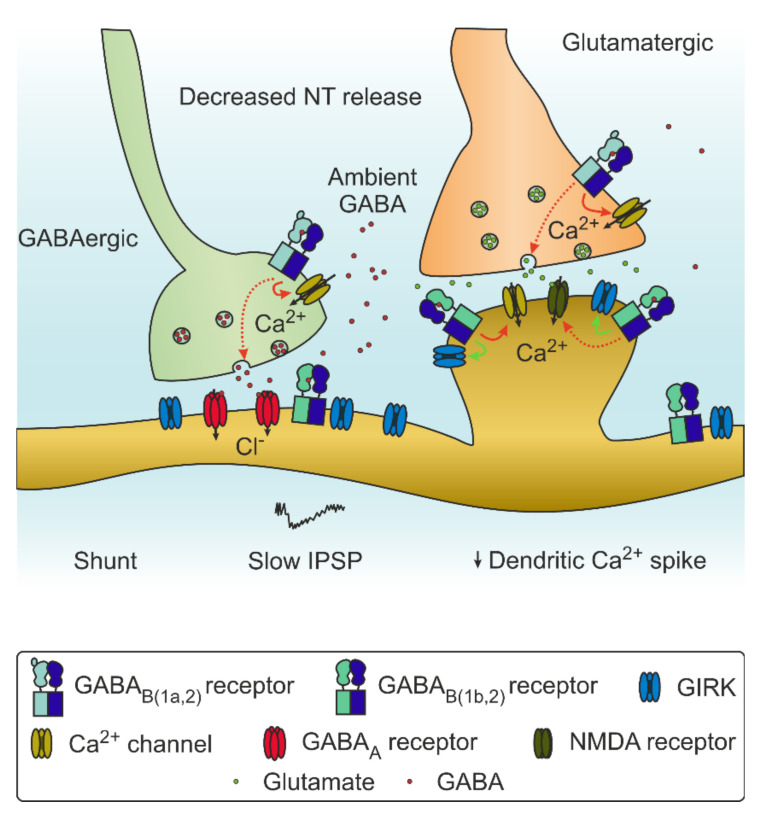
Recently, the structure of GABABRs in different conformations have been described using cryo-electron microscopy (unbound [24,25,26,27,28] and bound to agonist, modulators, and G protein [29]), allowing a greater understanding of their assembly and paving the way to the design of more advanced pharmacological tools for their modulation (structural results are reviewed in [30]). Activation by orthosteric binding results in a conformational shift which allows interaction with a heterotrimeric G protein. Upon binding, the G protein dissociates into Gα and Gβγ subunits. The Gα subunits most often associated to GABABRs are Gαi and Gαo. The Gαi/o subunit binds to adenylyl cyclase (AC), diminishing its activity and subsequently the levels of cAMP. Reduced cAMP levels in turn lead to a reduced probability of pre-synaptic neurotransmitter release. On the post-synaptic side, the protein kinase A pathway is also influenced by reduced AC activity, which results in decreased NMDAR conductance. The Gβγ fragment can also diminish vesicle fusion and the release of a neurotransmitter via inhibition of voltage-gated calcium channels. On the post-synaptic side, GABABRs activate G protein-coupled inward rectifying K+ channels (GIRK), which causes a hyperpolarizing slow inhibitory post-synaptic current that transiently inhibits the cell. The resulting increase in conductance contributes to the shunting effect on ongoing post-synaptic currents and shunts the backpropagation of the dendritic calcium spike [31] (Figure 1B and Figure 2).
Figure 2.
Functions of GABABRs in modulation of synaptic transmission. GABABRs are expressed pre-synaptically at both GABAergic (left, green) and glutamatergic (right, orange) synapses, where they can inhibit vesicle fusion and neurotransmitter release through inhibition of Ca2+ channels. They are also present post-synaptically, and they influence GIRK as well as the NMDA receptor function. Activation of GABABRs on the post-synaptic side leads to slow GIRK-channel-mediated IPSP and shunting inhibition, which can consequently inhibit dendritic calcium spike propagation. For details, see text.
Recent reports suggest that in cerebellar granule neuron cultures, GABABRs can influence synaptic strength and even provide an antiapoptotic effect, as they do not only couple to Gαi and Gαo, but they can also activate the Gα13 proteins. This action is performed at a much slower speed than those that are classical [32]. During development, GABABRs can also associate with Gαq, which enhances voltage-dependent calcium currents without Gαi/o [33]. Moreover, GABABRs can also present non-canonical effects, such as in the nucleus accumbens, where GABABRs activation likely inhibits glutamatergic pre-synaptic terminals by inhibiting the assembly of SNARE complexes [34].
GABABRs possess a multitude of regulatory mechanisms that can affect their functionality, for example heterodimers can dynamically associate to form oligomers [35] in which the ligand affinity decreases by interaction with neighboring receptors [36]. Many interacting proteins that can associate with GABABRs and alter their properties have been discovered. The K+ channel tetramerization domain containing proteins (KCTDs) KCTD8, KCTD12, KCTD12b, and KCTD16 are examples of auxiliary subunits of GABABRs [37]. The association of KCTDs with GABABRs influences the kinetics of GABABR-mediated responses, allowing for a faster interaction and consequently decreasing the rise time. In addition, KCTD12 can compete with Gβγ, thus inducing desensitization [38,39]. The KCTD 8 and the KCTD16 can prevent such desensitization [40], and different KCTDs can form hetero-oligomers with mixed effects on both the desensitization and the deactivation of K+ currents. The KCTDs therefore provide the possibility of precise fine-tuning of kinetics and the GABABR function [41], and this tuning can in turn be regulated depending on brain region and age. The KCTDs in fact display varying degrees of region and layer specificity, as well as temporal changes in expression [42]. A lack of KCTD12 or KCTD16 in mice can lead to alterations in fear processing and emotivity, thus further highlighting their importance for a correct regulation of GABABRs [43,44].
However, the possibilities of GABABR regulation are much larger. By using high-resolution proteomics, Schwenk and colleagues described a large amount of interacting proteins that participate in the formation of GABABR complexes, including effector proteins [45]. The exact composition of the complex can explain the heterogeneity of functions and constitute a promising target for future drug design (for review, see [46]). Between the described interacting proteins in GABABR complexes, the authors could find hyperpolarization activated cyclic nucleotide channel 2 (HCN2), which interacts with the complex through KCTD16. This interaction was shown to shorten the duration of IPSPs in dopaminergic neurons [45]. Furthermore, GABAB1Rs can inhibit the sensitization of transient receptor potential vanilloid 1 (TRPV1) channels in a G protein- and GABAB2-independent fashion [47]. Some components of the complex bind through the sushi domain of GABAB1a, including the β-amyloid precursor protein (APP, the precursor of β-amyloid peptides), PILRα-associated neural protein (PIANP), and adherence-junction associated protein 1 (AJAP-1), and they can influence the trafficking of GABABRs. These proteins have particular pathophysiological relevance (described in the following sections) [45]. Another factor to consider is the number of GABABRs on the cell surface. In fact, these receptors undergo constitutive endocytosis, which can be followed by either degradation or recycling to the membrane. Sustained glutamate-induced calcium influx can quickly and selectively diminish the rate of recycling, leading to a reduced GABABR-mediated inhibition [48]. Furthermore, additional mechanisms such as phosphorylation or ubiquitination have been shown to influence GABABR functions (for a review, see [49]).
Despite the relatively restricted number of subunits and isoforms, the family of functional GABABRs presents an extensive level of variety, which is granted by the formation of complexes, the oligomeric state, the phosphorylation state, and a large number of interacting proteins.
3. Spatial and Temporal Localization of GABABRs
In both rodent and in human adult brains, GABABRs are widely distributed across many brain areas, showing a distribution similar to that of GABAARs, albeit with a smaller number [50,51]. In rodents, the pharmacology and distribution of GABABRs varies during development in a region-specific manner [52,53,54]. In the neocortex, both subunits are expressed starting from embryonic stages [53]. During the first two postnatal weeks, the distribution of both subunits across the rodent brain varies almost independently, reaching a pattern of expression comparable to adults at around P20, with a general decrease thereafter [55]. The GABAB1 subunit appears near birth, mostly in superficial layer neurons with a Cajal–Retzius morphology. The GABAB2 subunit is also expressed early during development in the neocortex, especially in superficial layers, and it then becomes more uniformly distributed after P15 [53]. The GABAB1a and GABAB1b isoforms are also differentially regulated during development. GABAB1a is the predominant isoform at birth and it decreases over time, reaching adult levels after the end of the first postnatal month. On the other hand, the GABAB1b level at P0 is circa 50% of the adult level, and it undergoes a strong increase which peaks near P10 and decreases subsequently [56]. Near P10, the GABAB1 subunits in pyramidal neurons of the superficial cortical layers relocate from the soma and the dendrites to a more uniform spread across the whole cell membrane [53].
The distribution of GABABRs in the developing and the mature hippocampus has also been described in detail [54,57]; GABABRs are expressed at embryonic stages, they assemble both pre- and post-synaptically, and they show developmental regulation [58]. The distribution of GABAB1 and GIRKs is not homogeneous across cells, but rather arranged in a lamina-specific manner. The resulting GABABR-mediated potassium conductance is limited by the availability of both proteins [59]. Moreover, in the dentate gyrus, optogenetic stimulation of specific subtypes of interneurons demonstrated how the single stimulus GABAB-evoked GIRK response strength varies between different types of interneurons [60].
In summary, GABABRs expression is regulated throughout development in the cortex and in the hippocampus, and their function can be further modulated via the control of the expression of effectors as well as auxiliary and interacting proteins. Indeed, in addition to the variation of GABABR subunit expression during development, it is also important to take into account how the interacting proteins in the receptor complex tend to display developmental variations [45]. The fact that both subunits of GABABRs are present so early during development, before the establishment of mature synaptic transmission, suggests they might play a part in developmental mechanisms.
4. Developmental Functions
While GABA regulates fundamental steps in CNS development–including cell migration during cortex formation [61,62], cell maturation, and network development [13,63]—more recently, the role of GABA on neurogenesis has been described, mostly with a focus on GABAA receptors [64]. However, GABABRs have also been shown to be able to influence adult neurogenesis in the hippocampus. In their niche, neural stem cells (NSCs) express functional GABABRs, which suppress their proliferation and their differentiation. Their effect is opposite to that of GABAARs, whose activation promotes differentiation and integration in the circuit. This suggests that both receptors may work together and balance each other antagonistically [64,65]. It is important to notice that adult born granule cells lack GIRKs, which appear only subsequently, after approximately three weeks of maturation [60].
In a recent study, with the aim of investigating the role of GABA receptors in early neurogenesis, GABAA- and GABABRs were transiently blocked between P6 and P11. The blockade of GABABRs, but not of GABAARs, reduced the number of proliferating NSCs and the intermediate progenitor cells in the dentate gyrus. Furthermore, GABABR blockade caused a decreased expression of neurotrophins which are associated with synaptic plasticity, such as brain-derived neurotrophic factor (BDNF), nerve growth factor (NGF), and neurotrophin 3 (NT-3) [66]. Contrarily, GABABR activation can trigger BDNF release and promote inhibitory synaptogenesis in the newborn hippocampus [67], which can affect the development of GABAergic transmission [68].
In contrast to neurotrophic factors, GABABRs can interact directly with transcription factors such as activating transcription factor 4 (ATF4), which contributes to synaptic plasticity [69]. This interaction undergoes changes in efficiency during postnatal development and it is shared by neurons and glial cells [70]. Interestingly, ATF4 itself can have effects over longer time scales as a regulator of GABABRs trafficking, by acting on GABAB1 subunits and promoting surface exposure [71].
GABABRs also play a developmental role in the framework of transient cellular population. Cajal-Retzius cells are a transient cellular population which are present at embryonal stages [72,73]. They disappear towards the end of the second postnatal week and play an important role in circuit formation and correct lamination [74]. Between P5 and P7, GABABRs, together with glial transporters, constitute an important feedback mechanism for controlling the excitability of those cells [75]. Cajal-Retzius cells are one of the sources of Reelin protein, which is released extracellularly and guides cellular migration. In a recent study, Reelin was found capable of modulating the amount of both GABAB1 and GABAB2 on the cell surface. Furthermore, agonist and antagonist treatment of GABABRs in the absence of Reelin had no effect on the presynaptic side [76]. These results highlight the tight connection between GABABRs and Reelin, which is not only a key player in cell migration, but is also receiving increasing attention for control of synaptic formation and function [77].
The metabotropic GABAB receptors, GABABRs, are not only expressed in neurons, but also in glial cells and their regulation has a developmental aspect. The importance of the role of astrocytes in the regulation of network activity is gaining growing attention [78]. Astrocytes communicate with GABAergic neurons, and they contribute to the regulation of synaptic transmission [79]. The activation of astrocytic GABABRs triggers a calcium transient via Ca2+ release from intracellular stores. In contrast to the calcium transients evoked by astrocytic GABAAR activation, GABABR-evoked responses show a change during development in hippocampal astrocytes. At P3 and P33 the percentage of cells showing such responses was 10%, but between P11 and P15 it was instead 60% [80]. In a similar way, neocortical astrocytes also respond to GABABR activation with calcium oscillations, albeit with only a slight decrease in the number of responding cells in slices from older animals [81]. Astrocytes, following activation with GABA, release glutamate that influences the activity of neighboring pyramidal neurons via the induction of slow inward currents [82]. Moreover, astrocytic GABABRs in the mouse hippocampus have been proposed to control the response to behavioral challenge through the regulation of the astrocytic release of BDNF [83].
Myelination in both the central nervous system and the peripheral nervous system is also influenced by GABABRs [84]. In fact, GABABR activation has a stimulating effect on the differentiation of oligodendroglial cells and it can boost the expression of myelin-related protein expression [85]. Transient GABABRs blockade between P6 and P11 can decrease the level of myelin basic protein and affect the proliferation of oligodendroglial cells in vivo [86].
Furthermore, the organization of the inhibitory circuit during development in the first two postnatal weeks requires the presence of GABABRs in microglial cells. The knockdown of GABAB1 selectively in microglial cells led to a significant increase of inhibitory synapses originating from parvalbumin-positive (PV) interneurons onto pyramidal cells but no changes in excitatory synapses, with a consequent decrease in the ratio between the excitatory and the inhibitory post-synaptic current frequency at P30 [87]. Interestingly, the same animals at P60 display an almost reversed pattern, with a reduction in inhibitory synapses and no changes in the excitatory system, presumably via compensatory mechanisms. A lack of correct microglial-dependent synaptic organization led to a slight reduction in exploratory behavior at P30 and hyperactivity in P60 animals [87].
To sum up, our understanding of the role of GABABRs during development is becoming increasingly multifaceted and rich. They are involved in a variety of functions, from neurogenesis (both in adult age and during development) to migration, and they include transient cell types and glial cells.
5. Crosstalk with GABAARs and Early Activity Patterns
Other than the aforementioned neurotrophic or activity-independent role, GABABRs are also in a central position to exert control on the network excitation level; for their role in synaptic transmission, assembly in heterodimers is required. In both the cortex and the hippocampus, the pre-synaptic and the post-synaptic components develop at a separate pace. Indeed, post-synaptic GABABR-mediated currents only appear in the second postnatal week [88,89]. On the other hand, functional activation of pre-synaptic GABABRs has been reported much earlier, at the end of the first postnatal week in the cortex and the CA1 region of the hippocampus [90,91], and even earlier (i.e., at birth) in the CA3 region [92].
The GABABRs display different mechanisms of crosstalk with other neurotransmitter systems including glutamate receptors [93] and GABAARs. For example, GABABRs activation is able to influence the decay of GABAAR-mediated currents as well as the mIPSC frequency [94]. This crosstalk is particularly relevant during early life. The development of the inhibitory system in the postnatal period is in fact a complex process [95] that includes strong changes in the function of GABA. In rodents, immature neurons regulate the intracellular chloride concentration mostly via the Na+-K+-Cl- cotransporter isoform 1 (NKCC1), which leads to a steady state higher chloride concentration. Therefore, the opening of GABAA receptors can result in an efflux of chloride ions, effectively depolarizing the cell. During development, the K-Cl cotransporter isoform 2 (KCC2) expression increases, reducing the intracellular Cl concentration to mature levels and thus influencing the GABA current reversing potential [6].
GABABRs associate with KCC2 in protein complexes, and they are therefore able to influence neuronal chloride regulation. Indeed, activation of GABABRs can reduce the efficacy of KCC2 as well as their surface expression. This mechanism allows elevated levels of extracellular GABA to influence the effect of the neurotransmitter itself, through the intracellular chloride concentration [96]. Conversely, chloride influx through GABAARs can modulate the reversal potential of GABABR GIRK-mediated IPSPs, thus reducing their magnitude [97]. Moreover, the level of GABABR activation can affect the tonic GABAAR-mediated inhibition by controlling the subunit composition of GABAARs mediating tonic inhibition [98,99]. The inhibition is regulated in a local manner across various dendrites, and it is dynamically regulated by activity and extracellular GABA levels in a homeostatic manner [100]. Moreover, the regulation of the GABABR function can also happen on a cell-subtype specific level, with different effects on different interneuron types. For example, in the hippocampus, the activation of GABABRs modulates GABA release from PV interneurons with significantly less efficacy than from other types of interneurons [101].
During early life, the combination of glutamatergic and GABAergic activity can lead to large synchronous activity, which can be observed in in vitro preparations of rodent immature hippocampus as large synchronous network discharges called giant depolarizing potentials (GDPs). They influence synaptic transmission and circuit formation [102], and they lead to the release of large quantities of GABA in the extracellular space, which is sufficient to activate GABABRs [103]. Thus, GABABR activation influences the duration of GDPs by promoting their termination [67,104].
In summary, the ability of GABABR to modulate both excitatory and inhibitory synaptic transmission, as well as cell excitability via tonic GABAergic inhibition, can influence early synchronous activity in the developing brain.
6. Circuit Mechanisms
The development of neuronal circuits in the brain is characterized by the emergence of specific activity patterns that reflect the maturation state, and it is influenced by thalamic as well as local activity [105]. GABABRs have been shown to be able to influence network activity [106] and its entrainment to specific frequencies [107] in the hippocampus. In the thalamus, thalamocortical relay neurons and thalamic reticular neurons, which contribute to the generation of thalamic rhythmic activity, express both GABABRs and KCTD16. The strength and the frequency of the oscillatory behavior is controlled by GABABRs [108]. In the cortex, persistent brain activity requires a precise and a dynamic control of the balance between excitation and inhibition [109]. The synchronous activation of a large number of cells during a network oscillation and the consequent elevated release of GABA is sufficient for the activation of extrasynaptic GABABRs in a relatively large volume. Indeed, GABABRs are involved in the termination of the state of synchronous network firing (UP states) [110]. In the medial entorhinal cortex of rats, the termination of the UP state can happen either through a spontaneous mechanism, which is mediated by activation of GABAB1a-containing GABABRs, or through the activation of layer 1, which requires GABAB1b-containing GABABRs [111]. Thus, GABABR activation can increase the variability of the oscillatory cycle, thereby having a desynchronizing effect on network activity, as opposed to the activation of GABAARs [112]. A lack of GABABR activation leads instead to a decrease in the complexity of brain activity [113].
The layer 1-dependent termination of the UP state is mediated by the release of GABA from neurogliaform cells (NGFCs), which are known to use volume transmission for the inhibition of a large number of target cells [114]. Thalamic activation of NGFCs can occur in a coordinated manner across the neocortex, thus providing synchronization of different brain areas for transition to the regime of low network firing (DOWN state), as happens in slow wave sleep [115]. The NGFCs play an important role in defining the length of the integration window of sensory inputs to the cortex by reducing the thalamic feedforward inhibition in layer 4 through GABABRs [116].
The complexity of GABABR actions can also result from input-specific mechanisms. As an example, in the piriform cortex superficial layers, two subtypes of glutamatergic cells, semilunar cells, and superficial pyramidal cells, receive inputs from the olfactory bulb. In addition, the latter population receives inputs from the former and from other brain areas. The activation of GABABRs can simultaneously decrease the excitability both post-synaptically in glutamatergic neurons and pre-synaptically in input terminals, providing disinhibition by reducing GABA release. A strong activation of GABABRs induces a biphasic response, consisting of inhibition followed by network excitation. This effect can be explained by the fact that superficial pyramidal cells display a stronger effect of GABABRs activation on pre-synaptic inhibitory terminals [117]. The anterior piriform cortex, together with the olfactory nucleus, in turn projects a feedback connection to the olfactory bulb, which is also controlled by GABABRs in an input-specific manner. Pre-synaptic GABABRs depress the inputs to the interneurons resident in the olfactory bulb, but not on the principal cells, thus their activation leads to a decrease in feedback inhibition on excitatory cells [118].
The strength of GABABR-mediated inhibition can also be modulated by activity [119] and by the emergence of sensory activity. The medial superior olive (MSO) is a nucleus in the auditory brainstem responsible for detecting sound direction based on interaural time difference. While before the hearing onset GABABRs mediate a strong inhibition of both excitatory and inhibitory inputs as well as post-synaptic inhibition, after the hearing onset only the inhibition of inhibitory inputs remains unchanged. The GABABR-mediated presynaptic suppression of excitatory inputs disappears over some weeks as well as the activation of GIRK-mediated currents in the post-synaptic side. Immunohistochemistry revealed how across the first month, the distribution of GABABRs in the MSO switches from mostly dendritic to prevalently somatic, mirroring the functional change [120].
Plasticity mechanisms are strongly influenced by GABABRs. For example, in the auditory cortex, transient activation of layer 4 neurons evokes a plastic response measurable 1 h afterwards, which consists of a specific strengthening of low gamma oscillations. This is achieved by both the enhancement and the suppression of the firing rate of individual cells in a layer-specific manner; GABABRs mediate both the strengthening of excitatory synapses from layer 4 to superficial layers and the inhibitory plasticity between layer 4 and layer 5. Thus, GABABR modulation of network activity can influence the sensory adaptation to the presentation of a repeated stimulus and the sharpening of the cortical output [121]. The role of GABABRs in modulating inhibitory inputs at pyramidal cells in the auditory cortex is known to be dependent on sensory experience and the developmental stage [122]. In the same area, GABABRs are necessary for the generation of long-term depression on the connection between PV interneurons and pyramidal neurons, thus playing a crucial role in plasticity during the critical period of circuit development [123]. Similarly, GABABR-mediated modulation could strongly influence the critical period for ocular dominance (OD) plasticity in cats in vivo. The activation of GABABRs could promote OD plasticity, and a blockade could prevent it. Interestingly, this effect showed a developmental pattern, as pharmacological manipulation of GABABRs did not affect OD plasticity in adult animals [124].
The effect of either activation or inhibition of GABABRs can therefore depend on the specific circuit being investigated, as they can be differentially expressed on specific cell types and vary during development.
7. No Plan B: Dysfunction of GABABR-Mediated Inhibition in Pathology
Given the many processes GABABRs take part in, it is not surprising that reduced or absent GABABR function in mice has severe consequences. Lack of either of main subunits GABAB1 or GABAB2 leads to the development of seizures, which may cause death by the end of the first postnatal month [125,126], and to the development of hyperlocomotor activity, hyperalgesia and memory deficits [125,126,127]. Interestingly, mice lacking GABAB1a display impairments in hippocampal synaptic plasticity and memory that could not be detected in GABAB1b KO mice [23]. Furthermore, alteration of the GABABR system has been observed in several pathological conditions and can contribute to the manifestation of epilepsy and psychiatric disorders [11,128].
7.1. Epilepsy
While GABABRs may contribute to epileptogenesis, they traditionally received less attention than GABAARs in this context [129]. Analysis of tissue from temporal lobe epilepsy patients revealed alterations in the hippocampal levels of GABABRs [130], and a reduction in GABABR function was reported in the cortex of a rat model of absence epilepsy [131] and in human temporal lobe epilepsy tissue [132]. A possible mechanism of participation of GABABRs in seizure generation is via affecting the excitation to inhibition balance. Synapsin triple KO is a mouse model that generally displays alterations of GABAergic activity followed by epilepsy. In this model, reduced GABA release leads selectively to a weakening of GABABR-mediated presynaptic inhibition of glutamate release, thus shifting the excitation to inhibition (E/I) ratio towards excitation [133].
Another example is cortical dysplasia, which is often associated with non-pharmacologically tractable seizures. In vitro application of 4-aminopyridine, a blocker of K+ channels, to human tissue with different types of cortical dysplasia can lead to the generation of spontaneous discharges. It was recently shown how the activation of GABABRs can maintain the network in a less susceptible state, since GABABR blockade is required to induce ictal discharges [134]. However, the effects of the modulation of GABABR activity can be contextual. Cyclin dependent kinase like 5 (CDKL5) KO mice, a mouse model of CDKL5 deficiency disorder, exhibit seizures in early life, as well as intellectual disability later during development. In the perirhinal cortex of CDKL5 KO mice, the number of inhibitory synapses is increased compared to control animals, and long-term potentiation (LTP) is reduced. The blockade of GABABRs, but not of GABAARs, could rescue the morphological changes and the memory deficits in vivo, indicating that not only reduced but also enhanced GABABR activity can have a detrimental effect [135].
7.2. Autism Spectrum Disorders
Epilepsy is not the only pathological state in which GABABR-mediated inhibition plays a crucial role. Postmortem tissue from individuals with autistic spectrum disorders (ASD) display a reduced GABABRs expression [136,137]. The evaluation of the role of GABABRs in ASD involves investigation of possible underlying mechanisms in a range of heterogeneous mouse models. Fragile X syndrome (FXS) is a neurodevelopmental syndrome which is often associated with intellectual disability, ASD, and epilepsy. Fmr1-KO mice recapitulate some of the features observed in patients. In this model, a selective decrease in GABAB1a expression was observed in the hippocampus, accompanied by a reduced pre-synaptic inhibition of glutamatergic transmission. No changes could be seen on the inhibition of GABAergic inputs, leading to an imbalance in the E/I ratio, which could be rescued by administration of baclofen, a GABABR agonist [137]. However, using younger animals, a different group reported a pathway-specific disruption in feedforward inhibition in the hippocampus [138]. Those mice displayed increased power in high gamma activity, as measured by EEG. Treatment with baclofen normalized the aberrant activity as well as a subset of deficits, including sensory processing and working memory but not social interaction [139]. Indeed, the effects of baclofen on the social behavior of Fmrp1-KO mice has been mixed, and it may potentially depend on additional factors such as dose, animal stress level, or experimental paradigm [139,140,141,142]. A recent study in Fmr1-KO mice in the medial prefrontal cortex highlighted that the pattern of differences depends on the developmental stage; animals between P14 and P21 displayed an increased inhibitory drive, but between P36 and P42 the picture was instead reversed [143].
Similar developmental differences were found in the medial prefrontal cortex of a mouse model of tuberous sclerosis. Tuberous sclerosis is a monogenic syndrome with an elevated association with ASD, epilepsy, intellectual deficits, and alterations in synaptic transmission [144]. In Tsc2+/− mice, tonic GABABR-mediated inhibition on layer 2/3 pyramidal neurons is reduced at P25–30 but not earlier, and it is accompanied by increased excitability [145]. Interestingly, an increase in the E/I ratio is observable between P15 and P19, but only transiently, since the early increase in glutamatergic synaptic transmission is followed by a matched potentiation in inhibitory transmission as compared to control animals. [145]. Interestingly, the effect of baclofen on pre-synaptic GABABRs was found to be comparable at glutamatergic synapses but increased at GABAergic synapses, thus leading to a baclofen-mediated shift in the E/I ratio toward excitation [146].
Another protein that was shown to associate with GABABRs is PIANP [45]. The PIANP KO mice displayed a behavioral phenotype similar to ASD mice models, featuring increased anxiety, repetitive behavior, reduced explorative behavior, and abnormal social behavior [147]. In those mice, the effect of baclofen on mEPSC and mIPSC frequency was reduced, as well as the effect on high frequency stimulation, highlighting how GABABRs can potentially underlie some of the manifestations.
7.3. Alzheimer’s Disease
Not only neurodevelopmental, but also neurodegenerative disorders have been associated with alterations in GABABRs. For example, GABAB(1a,2) receptors have been shown to interact with APP through the sushi domains [45] and a decrease of the GABABR number has been shown in Alzheimer’s disease (AD) patients [148] and in AD animal models [149]. Only recently, a possible mechanism of GABABR involvement in this disease has been put forward. The interaction of GABAB(1a,2) with APP has the double effect of stabilizing GABABRs on the axonal cell surface and of preventing the cleavage of APP into Aβ. Therefore, stabilizing pre-synaptic GABABRs in AD could lead to both the reduction of increased glutamatergic transmission and the secretion of Aβ [150]. Furthermore, activation of GABABRs with baclofen in AD rats led to the stimulation of the PI3K/Akt pathway, as well as the rescue of the hippocampal atrophy and apoptosis levels [151]. Generally, modulation of GABABR activity has been shown to be able to lead to beneficial effects on cognition, learning, and spatial memory in the context of AD and dementia [151,152,153].
7.4. Long-Term Effects
Given the important role of GABABRs in development, it is reasonable to suggest that alterations in a specific time window, even if compensated later, may nevertheless lead to long-term consequences. The reduced functionality of GABABRs in the hippocampus may represent a consequence of epileptic seizures during early life. Epileptiform activity impairs GABABR-dependent pre-synaptic inhibition of GABAergic terminals [154], and it may have long term effects [155]. A single dose of GABABR antagonist administered at P15 could induce seizures that originate in the hippocampus and that have long-lasting effects, such as a decrease in paired pulse inhibition in CA1, which was measurable at P44 [156]. Similarly, early life systemic inflammation, as induced by a single injection of lipopolysaccharide at P14, leads to a reduced seizure threshold in P60 animals but not in those at P40. Concomitantly, it leads to a reduction in GABABR-mediated inhibition and a subsequent increase in the release probability at CA1 hippocampal synapses [157]. An increase in the baseline activation of GABABRs can have an effect on the behavior of the animals, for example, transient activation of GABABRs via daily injection of baclofen in mice between P14 and P28 led to the development of anxiety behavior in adult mice, which was tested between P60 and P80 [158]. The mechanism for such long-lasting effects might involve protein expression, circuit formation, or other unknown factors [66,159].
7.5. Stress
Long-term GABABR-dependent changes can also be triggered by other factors, such as stressors. Chronic stress affects GABABR function both pre- and post-synaptically in the hypothalamic periventricular nucleus, thus affecting the function of the hypothalamus–pituitary–adrenal axis [160]. Psychological stress leads to changes in GABABRs function in the prefrontal cortex in a cell-type specific manner, increasing the depression on parvalbumin interneurons and instead reducing that on somatostatin interneurons [161]. Interestingly, a lack of the GABAB1b subunit fosters resilience, while a lack of GABAB1a instead leads to increased susceptibility to anhedonia and social withdrawal following stress [162]. The involved mechanisms are still elusive, but it is hypothesized that the responsible circuit involves the ventral tegmental area-nucleus accumbens pathway, the dorsal raphe nucleus, and the hippocampus, and potentially adult neurogenesis and the serotoninergic system [163]. A recent study described how KCTD12 in the dentate gyrus can bidirectionally modulate the response to chronic social defeat stress in mice. Overexpression of KCTD12 in the dentate gyrus increased stress vulnerability, while downregulation could reverse stress-induced social avoidance [164], indicating that auxiliary proteins may play a crucial role in modulation of GABABR-mediated effects. Furthermore, activation of GABABRs in the nucleus accumbens can improve spatial memory in stress-exposed rats [165].
To sum up, GABABR activity has been shown to play an important role in the manifestation of various neuropsychiatric symptoms, including developmental and degenerative disorders. Alteration of their function may not manifest as a stable picture, but rather a dynamical rearrangement, especially during early development. Transient disturbances to their functionality in early life can lead to long-term consequences.
8. Pharmacological Modulation and Therapeutical Perspectives
In light of the previously discussed relevance of GABABRs in various pathological conditions, they represent a natural target for providing therapeutical support. Modulation of the GABABRs function has been taken into consideration for a wide range of disorders, such as depression [166], anxiety and mood disorders [167], substance use disorders [168], chronic pain [169], schizophrenia [170], and potential pro-cognitive aims [171].
8.1. Orthosteric Modulation
Even if several substances are available for modulation of their activity, baclofen is the only substance which has received FDA approval. It is currently used for the treatment of spasticity, and in particular, its most active form is the R-(-)-baclofen enantiomer (arbaclofen, [172]). Although several antagonists of GABABR have been routinely used in preclinical studies, only one entered clinical trials: CPG36742. Despite the fact that CPG36742 showed a potency to ameliorate AD symptoms in mild patient cases, further investigation was not pursued [173].
Activation of GABABRs has proven effective for the treatment of some of the symptoms in ASD mice models. For example, in the BTBR and C58 mice, baclofen treatment fixed stereotyped behavior and social interaction deficit, albeit it could not rescue every behavioral impairment [174]. In the 16p11.2 deletion mice—which in humans is characterized by intellectual disability, ASD, seizures, and anxiety—baclofen can rescue some cognitive deficits and social interaction [175]. Cntnap2 KO mice typically have impairments in behavior and auditory processing, which can be mostly remediated by treatment with R-baclofen [176]. A reduced NMDA receptor function is a common trait in schizophrenia, intellectual disability, and ASD. Mice with such phenotype display social and cognitive deficits, together with alterations in the EEG gamma band. Activation of GABABRs can rescue an altered E/I balance, gamma synchrony, and behavioral deficits following NMDAR hypofunction [140].
Arbaclofen is safe and well-tolerated in children and adolescents, and in an exploratory study it proved to be effective in several measures including the Aberrant Behavioral Checklist (ABC)—Irritability subscale and other social interaction measures [177]. A subsequent clinical trial failed to replicate the preliminary findings and to meet the expected effect on the ABC subscales, the primary outcome. Benefits were found on the Clinical Global Impression of Severity score, in which a subset of patients demonstrated strong improvement [178]. Arbaclofen has been shown in a recent clinical trial to have a positive effect as an adjuvant to risperidone in several subscales of ABC [179].
Baclofen treatment could improve impaired visual sensory processing in individuals with ASD; and, interestingly, it could impair visual processing in neurotypical subjects [180]. Currently, two randomized, double-blind, placebo-controlled studies are evaluating the effects of Arbaclofen on social function deficits in children and adolescents with ASD (NCT03887676 and NCT03682978).
Despite the efficiency of baclofen in preclinical studies in FXS (discussed in the previous section), where activation of GABABRs could rescue some aspects of the pathology, albeit with some controversy regarding the precise effect on social behavior [139,141,142], in a clinical trial for treatment of FXS arbaclofen failed to meet the primary outcome measure. It did, however, produce significant improvements on secondary measures [181].
8.2. Allosteric Modulation
As previously mentioned, GABABRs can interact with a variety of effectors, and they are embedded in many pathways. This makes orthosteric modulation prone to undesired consequences. Oral baclofen treatment in humans may cause dizziness, muscle weakness, sedation, nausea, fatigue [182], and it can less often lead to memory-related issues [183,184]. Therefore, a good amount of effort was devoted to the development of positive allosteric modulators (PAMs) of GABABRs. Introduced at the beginning of the 2000s, those compounds can influence the effect of GABA on GABABRs, but without complete activation of the receptor, therefore avoiding or minimizing potential side effects. The most well-known compounds are CGP7930 [185], GS39783 [186], and Rac-BHFF [187], which interact with the receptors by binding a pocket at the interface of the transmembrane domains of the active GABAB1/GABAB2 heterodimer [188]. What makes the use of positive allosteric modulation a valuable mechanism, besides the reduced amount of side effects [189], is their possible region- [190] as well as a pathway- and species-specificity [191], which could be leveraged for therapeutical advantage. Many PAMs have already shown positive effects on alcohol seeking behavior in animal models (e.g., [192]), and they are currently being investigated in clinical trials (see [193]).
Using CGP7930 as a starting point, CLH304a was developed, which can act as a negative allosteric modulator (NAM) [194]. This drug has been suggested to bind the GABAB2 subunit in the transmembrane domain and inhibit GABABRs constitutive activation [195]. Very recently, another negative allosteric modulator named COR758 was described, which likely binds a site in the GABAB1 subunit. It could successfully modulate GABABR activity in rat dopaminergic neurons [196]. However, especially for the NAMs, very little is known about in vivo responses and safety, and more investigation is required before considering a clinical use.
Another promising way of modulating the activity of GABABRs is through their interaction with the components of their complexes [45]. For example, Sereikaite and colleagues recently identified the binding epitope of the KCTD12 auxiliary proteins to GABABRs and via the use of deep mutational scan and an iterative screening procedure, they obtained peptides with a higher affinity to KCTD12 than GABAB2, which could reduce the interaction between the auxiliary protein and the receptor. This methodology does not only open the way for the study of the interaction of KCTDs and GABABRs, but it could be used to extend our knowledge of other GABABR -interacting proteins [197]. Recent development in protein–protein interaction modulation has made huge progress, as reflected by the increasing amount of clinical trials using such modulators [198]. In the context of treating brain pathologies, however, it will be necessary to evaluate the bioavailability of potential modulators and their ability to cross the blood–brain barrier.
9. Conclusions
This review highlights how, despite their apparent simplicity, GABABRs possess varied physiological effects. This property arises from the rich number of effector proteins they can affect as well as their precise position within a neural circuit. Much evidence points to the important role of GABABRs at early developmental stages, however, several details need to be further investigated. In the pathophysiological context, an increasing number of studies suggest that the temporal aspect should not be overlooked. In particular, it would be extremely useful to obtain a more precise description of the long-term effects of disturbances of GABABR activity during early development and by which mechanisms they are exerted. Such a perspective would provide valuable information for the investigation of different disease models, and it would provide indications on which developmental stage would be more useful to investigate. Thus, GABABRs represent a suitable target for treating a plethora of conditions which feature a decreased GABABR-mediated inhibition, or as a therapeutical tool to influence network activity. The recent description of GABABR structure in an unprecedented level of detail will likely foster advancements in the pharmacological methods that will be available for the investigation of their function, as well as the development of novel treatments such as allosteric modulators that exhibit pathway and/or area selectivity and reduced side effects. Similarly, the development of technologies to modulate protein–protein interaction will allow the investigation of the possible outcomes caused by modifications of the GABABR complex components.
Acknowledgments
I would like to express my gratitude to Heiko J. Luhmann, Sergei Kirischuk, and Werner Kilb for the valuable discussion and the help they provided and to Celine Gallagher for the highly appreciated comments on the manuscript. My sincerest apologies for those whose relevant work could not be included due to space limitations.
Abbreviations
| AC | adenylyl cyclase |
| AD | Alzheimer’s disease |
| APP | β-amyloid precursor protein |
| ASD | autism spectrum disorders |
| BDNF | brain-derived neurotrophic factor |
| FXS | fragile X syndrome |
| GABA | gamma aminobutyric acid |
| GABABR | GABAB receptor |
| GIRK | G protein-coupled inward rectifying K+ channel |
| PAM/NAM | positive/negative allosteric modulator |
| PV | parvalbumin |
Funding
This work was supported by the Deutsche Forschungsgemeinschaft (DFG, grant CRC1080-B10 to H.J.L.).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The author declares no conflict of interest.
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Silbereis J.C., Pochareddy S., Zhu Y., Li M., Sestan N. The Cellular and Molecular Landscapes of the Developing Human Central Nervous System. Neuron. 2016;89:248–268. doi: 10.1016/j.neuron.2015.12.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Tang X., Jaenisch R., Sur M. The Role of GABAergic Signalling in Neurodevelopmental Disorders. Nat. Rev. Neurosci. 2021;22:290–307. doi: 10.1038/s41583-021-00443-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Gelman D.M., Marín O. Generation of Interneuron Diversity in the Mouse Cerebral Cortex. Eur. J. Neurosci. 2010;31:2136–2141. doi: 10.1111/j.1460-9568.2010.07267.x. [DOI] [PubMed] [Google Scholar]
- 4.Lim L., Mi D., Llorca A., Marín O. Development and Functional Diversification of Cortical Interneurons. Neuron. 2018;100:294–313. doi: 10.1016/j.neuron.2018.10.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Luhmann H.J., Fukuda A. Can We Understand Human Brain Development from Experimental Studies in Rodents? Pediatr. Int. 2020;62:1139–1144. doi: 10.1111/ped.14339. [DOI] [PubMed] [Google Scholar]
- 6.Ben-Ari Y., Gaiarsa J.-L., Tyzio R., Khazipov R. GABA: A Pioneer Transmitter That Excites Immature Neurons and Generates Primitive Oscillations. Physiol. Rev. 2007;87:1215–1284. doi: 10.1152/physrev.00017.2006. [DOI] [PubMed] [Google Scholar]
- 7.Warm D., Schroer J., Sinning A. Gabaergic Interneurons in Early Brain Development: Conducting and Orchestrated by Cortical Network Activity. Front. Mol. Neurosci. 2022;14:344. doi: 10.3389/fnmol.2021.807969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Le Magueresse C., Monyer H. GABAergic Interneurons Shape the Functional Maturation of the Cortex. Neuron. 2013;77:388–405. doi: 10.1016/j.neuron.2013.01.011. [DOI] [PubMed] [Google Scholar]
- 9.Represa A., Ben-Ari Y. Trophic Actions of GABA on Neuronal Development. Trends Neurosci. 2005;28:278–283. doi: 10.1016/j.tins.2005.03.010. [DOI] [PubMed] [Google Scholar]
- 10.Deidda G., Bozarth I.F., Cancedda L. Modulation of GABAergic Transmission in Development and Neurodevelopmental Disorders: Investigating Physiology and Pathology to Gain Therapeutic Perspectives. Front. Cell. Neurosci. 2014;8:119. doi: 10.3389/fncel.2014.00119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Heaney C.F., Kinney J.W. Role of GABAB Receptors in Learning and Memory and Neurological Disorders. Neurosci. Biobehav. Rev. 2016;63:1–28. doi: 10.1016/j.neubiorev.2016.01.007. [DOI] [PubMed] [Google Scholar]
- 12.Gaiarsa J.-L., Kuczewski N., Porcher C. Contribution of Metabotropic GABAB Receptors to Neuronal Network Construction. Pharmacol. Ther. 2011;132:170–179. doi: 10.1016/j.pharmthera.2011.06.004. [DOI] [PubMed] [Google Scholar]
- 13.Gaiarsa J.-L., Porcher C. Emerging Neurotrophic Role of GABAB Receptors in Neuronal Circuit Development. Front. Cell. Neurosci. 2013;7:206. doi: 10.3389/fncel.2013.00206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Vlachou S. GABAB Receptors and cognitive processing in health and disease. In: Vlachou S., Wickman K., editors. Behavioral Neurobiology of GABAB Receptor Function. Springer International Publishing; Cham, Switzerland: 2022. pp. 291–329. Current Topics in Behavioral Neurosciences. [DOI] [PubMed] [Google Scholar]
- 15.Evenseth L.S.M., Gabrielsen M., Sylte I. The GABAB Receptor—Structure, Ligand Binding and Drug Development. Molecules. 2020;25:3093. doi: 10.3390/molecules25133093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Avoli M., Krnjević K. The Long and Winding Road to Gamma-Amino-Butyric Acid as Neurotransmitter. Can. J. Neurol. Sci. 2016;43:219–226. doi: 10.1017/cjn.2015.333. [DOI] [PubMed] [Google Scholar]
- 17.Krnjević K., Schwartz S. The Action of γ-Aminobutyric Acid on Cortical Neurones. Exp. Brain Res. 1967;3:320–336. doi: 10.1007/BF00237558. [DOI] [PubMed] [Google Scholar]
- 18.Hill D.R., Bowery N.G. 3H-Baclofen and 3H-GABA Bind to Bicuculline-Insensitive GABAB Sites in Rat Brain. Nature. 1981;290:149–152. doi: 10.1038/290149a0. [DOI] [PubMed] [Google Scholar]
- 19.Kaupmann K., Malitschek B., Schuler V., Heid J., Froestl W., Beck P., Mosbacher J., Bischoff S., Kulik A., Shigemoto R., et al. GABAB-Receptor Subtypes Assemble into Functional Heteromeric Complexes. Nature. 1998;396:683–687. doi: 10.1038/25360. [DOI] [PubMed] [Google Scholar]
- 20.White J.H., Wise A., Main M.J., Green A., Fraser N.J., Disney G.H., Barnes A.A., Emson P., Foord S.M., Marshall F.H. Heterodimerization Is Required for the Formation of a Functional GABAB Receptor. Nature. 1998;396:679–682. doi: 10.1038/25354. [DOI] [PubMed] [Google Scholar]
- 21.Margeta-Mitrovic M., Jan Y.N., Jan L.Y. A Trafficking Checkpoint Controls GABAB Receptor Heterodimerization. Neuron. 2000;27:97–106. doi: 10.1016/S0896-6273(00)00012-X. [DOI] [PubMed] [Google Scholar]
- 22.Kaupmann K., Huggel K., Heid J., Flor P.J., Bischoff S., Mickel S.J., McMaster G., Angst C., Bittiger H., Froestl W., et al. Expression Cloning of GABAB Receptors Uncovers Similarity to Metabotropic Glutamate Receptors. Nature. 1997;386:239–246. doi: 10.1038/386239a0. [DOI] [PubMed] [Google Scholar]
- 23.Vigot R., Barbieri S., Bräuner-Osborne H., Turecek R., Shigemoto R., Zhang Y.-P., Luján R., Jacobson L.H., Biermann B., Fritschy J.-M., et al. Differential Compartmentalization and Distinct Functions of GABAB Receptor Variants. Neuron. 2006;50:589–601. doi: 10.1016/j.neuron.2006.04.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Kim Y., Jeong E., Jeong J.-H., Kim Y., Cho Y. Structural Basis for Activation of the Heterodimeric GABAB Receptor. J. Mol. Biol. 2020;432:5966–5984. doi: 10.1016/j.jmb.2020.09.023. [DOI] [PubMed] [Google Scholar]
- 25.Mao C., Shen C., Li C., Shen D.-D., Xu C., Zhang S., Zhou R., Shen Q., Chen L.-N., Jiang Z., et al. Cryo-EM Structures of Inactive and Active GABAB Receptor. Cell Res. 2020;30:564–573. doi: 10.1038/s41422-020-0350-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Papasergi-Scott M.M., Robertson M.J., Seven A.B., Panova O., Mathiesen J.M., Skiniotis G. Structures of Metabotropic GABAB Receptor. Nature. 2020;584:310–314. doi: 10.1038/s41586-020-2469-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Park J., Fu Z., Frangaj A., Liu J., Mosyak L., Shen T., Slavkovich V.N., Ray K.M., Taura J., Cao B., et al. Structure of Human GABAB Receptor in an Inactive State. Nature. 2020;584:304–309. doi: 10.1038/s41586-020-2452-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Shaye H., Ishchenko A., Lam J.H., Han G.W., Xue L., Rondard P., Pin J.-P., Katritch V., Gati C., Cherezov V. Structural Basis of the Activation of a Metabotropic GABA Receptor. Nature. 2020;584:298–303. doi: 10.1038/s41586-020-2408-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Shen C., Mao C., Xu C., Jin N., Zhang H., Shen D.-D., Shen Q., Wang X., Hou T., Chen Z., et al. Structural Basis of GABAB Receptor–Gi Protein Coupling. Nature. 2021;594:594–598. doi: 10.1038/s41586-021-03507-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Shaye H., Stauch B., Gati C., Cherezov V. Molecular Mechanisms of Metabotropic GABAB Receptor Function. Sci. Adv. 2021;7:eabg3362. doi: 10.1126/sciadv.abg3362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Pérez-Garci E., Gassmann M., Bettler B., Larkum M.E. The GABAB1b Isoform Mediates Long-Lasting Inhibition of Dendritic Ca2+ Spikes in Layer 5 Somatosensory Pyramidal Neurons. Neuron. 2006;50:603–616. doi: 10.1016/j.neuron.2006.04.019. [DOI] [PubMed] [Google Scholar]
- 32.Wang Y., Gai S., Zhang W., Huang X., Ma S., Huo Y., Wu Y., Tu H., Pin J.-P., Rondard P., et al. The GABAB Receptor Mediates Neuroprotection by Coupling to G13. Sci. Signal. 2021;14:eaaz4112. doi: 10.1126/scisignal.aaz4112. [DOI] [PubMed] [Google Scholar]
- 33.Karls A., Mynlieff M. GABAB Receptors Couple to Gαq to Mediate Increases in Voltage-Dependent Calcium Current during Development. J. Neurochem. 2015;135:88–100. doi: 10.1111/jnc.13259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Manz K.M., Baxley A.G., Zurawski Z., Hamm H.E., Grueter B.A. Heterosynaptic GABAB Receptor Function within Feedforward Microcircuits Gates Glutamatergic Transmission in the Nucleus Accumbens Core. J. Neurosci. 2019;39:9277–9293. doi: 10.1523/JNEUROSCI.1395-19.2019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Calebiro D., Rieken F., Wagner J., Sungkaworn T., Zabel U., Borzi A., Cocucci E., Zürn A., Lohse M.J. Single-Molecule Analysis of Fluorescently Labeled G-Protein–Coupled Receptors Reveals Complexes with Distinct Dynamics and Organization. Proc. Natl. Acad. Sci. USA. 2013;110:743–748. doi: 10.1073/pnas.1205798110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Stewart G.D., Comps-Agrar L., Nørskov-Lauritsen L.B., Pin J.-P., Kniazeff J. Allosteric Interactions between GABAB1 Subunits Control Orthosteric Binding Sites Occupancy within GABAB Oligomers. Neuropharmacology. 2018;136:92–101. doi: 10.1016/j.neuropharm.2017.12.042. [DOI] [PubMed] [Google Scholar]
- 37.Schwenk J., Metz M., Zolles G., Turecek R., Fritzius T., Bildl W., Tarusawa E., Kulik A., Unger A., Ivankova K., et al. Native GABA B Receptors Are Heteromultimers with a Family of Auxiliary Subunits. Nature. 2010;465:231–235. doi: 10.1038/nature08964. [DOI] [PubMed] [Google Scholar]
- 38.Rajalu M., Fritzius T., Adelfinger L., Jacquier V., Besseyrias V., Gassmann M., Bettler B. Pharmacological Characterization of GABAB Receptor Subtypes Assembled with Auxiliary KCTD Subunits. Neuropharmacology. 2015;88:145–154. doi: 10.1016/j.neuropharm.2014.08.020. [DOI] [PubMed] [Google Scholar]
- 39.Turecek R., Schwenk J., Fritzius T., Ivankova K., Zolles G., Adelfinger L., Jacquier V., Besseyrias V., Gassmann M., Schulte U., et al. Auxiliary GABAB Receptor Subunits Uncouple G Protein Βγ Subunits from Effector Channels to Induce Desensitization. Neuron. 2014;82:1032–1044. doi: 10.1016/j.neuron.2014.04.015. [DOI] [PubMed] [Google Scholar]
- 40.Seddik R., Jungblut S.P., Silander O.K., Rajalu M., Fritzius T., Besseyrias V., Jacquier V., Fakler B., Gassmann M., Bettler B. Opposite Effects of KCTD Subunit Domains on GABAB Receptor-Mediated Desensitization. J. Biol. Chem. 2012;287:39869–39877. doi: 10.1074/jbc.M112.412767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Fritzius T., Turecek R., Seddik R., Kobayashi H., Tiao J., Rem P.D., Metz M., Kralikova M., Bouvier M., Gassmann M., et al. KCTD Hetero-Oligomers Confer Unique Kinetic Properties on Hippocampal GABAB Receptor-Induced K+ Currents. J. Neurosci. 2017;37:1162–1175. doi: 10.1523/JNEUROSCI.2181-16.2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Metz M., Gassmann M., Fakler B., Schaeren-Wiemers N., Bettler B. Distribution of the Auxiliary GABAB Receptor Subunits KCTD8, 12, 12b, and 16 in the Mouse Brain. J. Comp. Neurol. 2011;519:1435–1454. doi: 10.1002/cne.22610. [DOI] [PubMed] [Google Scholar]
- 43.Cathomas F., Stegen M., Sigrist H., Schmid L., Seifritz E., Gassmann M., Bettler B., Pryce C.R. Altered Emotionality and Neuronal Excitability in Mice Lacking KCTD12, an Auxiliary Subunit of GABAB Receptors Associated with Mood Disorders. Transl. Psychiatry. 2015;5:e510. doi: 10.1038/tp.2015.8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Cathomas F., Sigrist H., Schmid L., Seifritz E., Gassmann M., Bettler B., Pryce C.R. Behavioural Endophenotypes in Mice Lacking the Auxiliary GABAB Receptor Subunit KCTD16. Behav. Brain Res. 2017;317:393–400. doi: 10.1016/j.bbr.2016.10.006. [DOI] [PubMed] [Google Scholar]
- 45.Schwenk J., Pérez-Garci E., Schneider A., Kollewe A., Gauthier-Kemper A., Fritzius T., Raveh A., Dinamarca M.C., Hanuschkin A., Bildl W., et al. Modular Composition and Dynamics of Native GABAB Receptors Identified by High-Resolution Proteomics. Nat. Neurosci. 2016;19:233–242. doi: 10.1038/nn.4198. [DOI] [PubMed] [Google Scholar]
- 46.Fritzius T., Bettler B. The Organizing Principle of GABAB Receptor Complexes: Physiological and Pharmacological Implications. Basic Clin. Pharmacol. Toxicol. 2020;126:25–34. doi: 10.1111/bcpt.13241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Hanack C., Moroni M., Lima W.C., Wende H., Kirchner M., Adelfinger L., Schrenk-Siemens K., Tappe-Theodor A., Wetzel C., Kuich P.H., et al. GABA Blocks Pathological but Not Acute TRPV1 Pain Signals. Cell. 2015;160:759–770. doi: 10.1016/j.cell.2015.01.022. [DOI] [PubMed] [Google Scholar]
- 48.Terunuma M., Vargas K.J., Wilkins M.E., Ramirez O.A., Jaureguiberry-Bravo M., Pangalos M.N., Smart T.G., Moss S.J., Couve A. Prolonged Activation of NMDA Receptors Promotes Dephosphorylation and Alters Postendocytic Sorting of GABAB Receptors. Proc. Natl. Acad. Sci. USA. 2010;107:13918–13923. doi: 10.1073/pnas.1000853107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Terunuma M. Diversity of Structure and Function of GABAB Receptors: A Complexity of GABAB-Mediated Signaling. Proc. Jpn. Acad. Ser. B. 2018;94:390–411. doi: 10.2183/pjab.94.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Behuet S., Cremer J.N., Cremer M., Palomero-Gallagher N., Zilles K., Amunts K. Developmental Changes of Glutamate and GABA Receptor Densities in Wistar Rats. Front. Neuroanat. 2019;13:100. doi: 10.3389/fnana.2019.00100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Young A.B., Chu D. Distribution of GABAA and GABAB Receptors in Mammalian Brain: Potential Targets for Drug Development. Drug Dev. Res. 1990;21:161–167. doi: 10.1002/ddr.430210303. [DOI] [Google Scholar]
- 52.Gonchar Y., Pang L., Malitschek B., Bettler B., Burkhalter A. Subcellular Localization of GABAB Receptor Subunits in Rat Visual Cortex. J. Comp. Neurol. 2001;431:182–197. doi: 10.1002/1096-9861(20010305)431:2<182::AID-CNE1064>3.0.CO;2-K. [DOI] [PubMed] [Google Scholar]
- 53.López-Bendito G., Shigemoto R., Kulik A., Paulsen O., Fairén A., Luján R. Expression and Distribution of Metabotropic GABA Receptor Subtypes GABABR1 and GABABR2 during Rat Neocortical Development. Eur. J. Neurosci. 2002;15:1766–1778. doi: 10.1046/j.1460-9568.2002.02032.x. [DOI] [PubMed] [Google Scholar]
- 54.López-Bendito G., Shigemoto R., Kulik A., Vida I., Fairén A., Luján R. Distribution of Metabotropic GABA Receptor Subunits GABAB1a/b and GABAB2 in the Rat Hippocampus during Prenatal and Postnatal Development. Hippocampus. 2004;14:836–848. doi: 10.1002/hipo.10221. [DOI] [PubMed] [Google Scholar]
- 55.Fritschy J.-M., Sidler C., Parpan F., Gassmann M., Kaupmann K., Bettler B., Benke D. Independent Maturation of the GABAB Receptor Subunits GABAB1 and GABAB2 during Postnatal Development in Rodent Brain. J. Comp. Neurol. 2004;477:235–252. doi: 10.1002/cne.20188. [DOI] [PubMed] [Google Scholar]
- 56.Fritschy J.-M., Meskenaite V., Weinmann O., Honer M., Benke D., Mohler H. GABAB-Receptor Splice Variants GB1a and GB1b in Rat Brain: Developmental Regulation, Cellular Distribution and Extrasynaptic Localization. Eur. J. Neurosci. 1999;11:761–768. doi: 10.1046/j.1460-9568.1999.00481.x. [DOI] [PubMed] [Google Scholar]
- 57.Kulik Á., Vida I., Luján R., Haas C.A., López-Bendito G., Shigemoto R., Frotscher M. Subcellular Localization of Metabotropic GABAB Receptor Subunits GABAB1a/b and GABAB2 in the Rat Hippocampus. J. Neurosci. 2003;23:11026–11035. doi: 10.1523/JNEUROSCI.23-35-11026.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Khoshdel-Sarkarizi H., Hami J., Mohammadipour A., Sadr-Nabavi A., Mahmoudi M., Kheradmand H., Peyvandi M., Nourmohammadi E., Haghir H. Developmental Regulation and Lateralization of GABA Receptors in the Rat Hippocampus. Int. J. Dev. Neurosci. 2019;76:86–94. doi: 10.1016/j.ijdevneu.2019.07.006. [DOI] [PubMed] [Google Scholar]
- 59.Degro C.E., Kulik A., Booker S.A., Vida I. Compartmental Distribution of GABAB Receptor-Mediated Currents along the Somatodendritic Axis of Hippocampal Principal Cells. Front. Synaptic Neurosci. 2015;7:6. doi: 10.3389/fnsyn.2015.00006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Gonzalez J.C., Epps S.A., Markwardt S.J., Wadiche J.I., Overstreet-Wadiche L. Constitutive and Synaptic Activation of GIRK Channels Differentiates Mature and Newborn Dentate Granule Cells. J. Neurosci. 2018;38:6513–6526. doi: 10.1523/JNEUROSCI.0674-18.2018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Behar T.N., Smith S.V., Kennedy R.T., Mckenzie J.M., Maric I., Barker J.L. GABAB Receptors Mediate Motility Signals for Migrating Embryonic Cortical Cells. Cereb. Cortex. 2001;11:744–753. doi: 10.1093/cercor/11.8.744. [DOI] [PubMed] [Google Scholar]
- 62.Luhmann H.J., Fukuda A., Kilb W. Control of Cortical Neuronal Migration by Glutamate and GABA. Front. Cell. Neurosci. 2015;9:4. doi: 10.3389/fncel.2015.00004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Bony G., Szczurkowska J., Tamagno I., Shelly M., Contestabile A., Cancedda L. Non-Hyperpolarizing GABAB Receptor Activation Regulates Neuronal Migration and Neurite Growth and Specification by CAMP/LKB1. Nat. Commun. 2013;4:1800. doi: 10.1038/ncomms2820. [DOI] [PubMed] [Google Scholar]
- 64.Catavero C., Bao H., Song J. Neural Mechanisms Underlying GABAergic Regulation of Adult Hippocampal Neurogenesis. Cell Tissue Res. 2018;371:33–46. doi: 10.1007/s00441-017-2668-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Giachino C., Barz M., Tchorz J.S., Tome M., Gassmann M., Bischofberger J., Bettler B., Taylor V. GABA Suppresses Neurogenesis in the Adult Hippocampus through GABAB Receptors. Development. 2014;141:83–90. doi: 10.1242/dev.102608. [DOI] [PubMed] [Google Scholar]
- 66.Gustorff C., Scheuer T., Schmitz T., Bührer C., Endesfelder S. GABAB Receptor-Mediated Impairment of Intermediate Progenitor Maturation During Postnatal Hippocampal Neurogenesis of Newborn Rats. Front. Cell. Neurosci. 2021;15:295. doi: 10.3389/fncel.2021.651072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Fiorentino H., Kuczewski N., Diabira D., Ferrand N., Pangalos M.N., Porcher C., Gaiarsa J.-L. GABAB Receptor Activation Triggers BDNF Release and Promotes the Maturation of GABAergic Synapses. J. Neurosci. 2009;29:11650–11661. doi: 10.1523/JNEUROSCI.3587-09.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Porcher C., Medina I., Gaiarsa J.-L. Mechanism of BDNF Modulation in GABAergic Synaptic Transmission in Healthy and Disease Brains. Front. Cell. Neurosci. 2018;12:273. doi: 10.3389/fncel.2018.00273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.White J.H., McIllhinney R.A.J., Wise A., Ciruela F., Chan W.-Y., Emson P.C., Billinton A., Marshall F.H. The GABAB Receptor Interacts Directly with the Related Transcription Factors CREB2 and ATFx. Proc. Natl. Acad. Sci. USA. 2000;97:13967–13972. doi: 10.1073/pnas.240452197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Ritter B., Zschüntsch J., Kvachnina E., Zhang W., Ponimaskin E.G. The GABAB Receptor Subunits R1 and R2 Interact Differentially with the Activation Transcription Factor ATF4 in Mouse Brain during the Postnatal Development. Dev. Brain Res. 2004;149:73–77. doi: 10.1016/j.devbrainres.2003.12.006. [DOI] [PubMed] [Google Scholar]
- 71.Corona C., Pasini S., Liu J., Amar F., Greene L.A., Shelanski M.L. Activating Transcription Factor 4 (ATF4) Regulates Neuronal Activity by Controlling GABABR Trafficking. J. Neurosci. 2018;38:6102–6113. doi: 10.1523/JNEUROSCI.3350-17.2018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Causeret F., Moreau M.X., Pierani A., Blanquie O. The Multiple Facets of Cajal-Retzius Neurons. Development. 2021;148:dev199409. doi: 10.1242/dev.199409. [DOI] [PubMed] [Google Scholar]
- 73.Kirischuk S., Luhmann H.J., Kilb W. Cajal–Retzius Cells: Update on Structural and Functional Properties of These Mystic Neurons That Bridged the 20th Century. Neuroscience. 2014;275:33–46. doi: 10.1016/j.neuroscience.2014.06.009. [DOI] [PubMed] [Google Scholar]
- 74.Soriano E., del Río J.A. The Cells of Cajal-Retzius: Still a Mystery One Century After. Neuron. 2005;46:389–394. doi: 10.1016/j.neuron.2005.04.019. [DOI] [PubMed] [Google Scholar]
- 75.Kirmse K., Kirischuk S. Ambient GABA Constrains the Strength of GABAergic Synapses at Cajal-Retzius Cells in the Developing Visual Cortex. J. Neurosci. 2006;26:4216–4227. doi: 10.1523/JNEUROSCI.0589-06.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Hamad M.I.K., Jbara A., Rabaya O., Petrova P., Daoud S., Melliti N., Meseke M., Lutz D., Petrasch-Parwez E., Schwitalla J.C., et al. Reelin Signaling Modulates GABAB Receptor Function in the Neocortex. J. Neurochem. 2021;156:589–603. doi: 10.1111/jnc.14990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Faini G., Del Bene F., Albadri S. Reelin Functions beyond Neuronal Migration: From Synaptogenesis to Network Activity Modulation. Curr. Opin. Neurobiol. 2021;66:135–143. doi: 10.1016/j.conb.2020.10.009. [DOI] [PubMed] [Google Scholar]
- 78.Poskanzer K.E., Yuste R. Astrocytes Regulate Cortical State Switching in Vivo. Proc. Natl. Acad. Sci. USA. 2016;113:E2675–E2684. doi: 10.1073/pnas.1520759113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Lia A., Zonta M., Requie L.M., Carmignoto G. Dynamic Interactions between GABAergic and Astrocytic Networks. Neurosci. Lett. 2019;689:14–20. doi: 10.1016/j.neulet.2018.06.026. [DOI] [PubMed] [Google Scholar]
- 80.Meier S.D., Kafitz K.W., Rose C.R. Developmental Profile and Mechanisms of GABA-Induced Calcium Signaling in Hippocampal Astrocytes. Glia. 2008;56:1127–1137. doi: 10.1002/glia.20684. [DOI] [PubMed] [Google Scholar]
- 81.Mariotti L., Losi G., Sessolo M., Marcon I., Carmignoto G. The Inhibitory Neurotransmitter GABA Evokes Long-Lasting Ca2+ Oscillations in Cortical Astrocytes. Glia. 2016;64:363–373. doi: 10.1002/glia.22933. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Durkee C.A., Covelo A., Lines J., Kofuji P., Aguilar J., Araque A. Gi/o Protein-Coupled Receptors Inhibit Neurons but Activate Astrocytes and Stimulate Gliotransmission. Glia. 2019;67:1076–1093. doi: 10.1002/glia.23589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Liu J.-H., Li Z.-L., Liu Y.-S., Chu H.-D., Hu N.-Y., Wu D.-Y., Huang L., Li S.-J., Li X.-W., Yang J.-M., et al. Astrocytic GABAB Receptors in Mouse Hippocampus Control Responses to Behavioral Challenges through Astrocytic BDNF. Neurosci. Bull. 2020;36:705–718. doi: 10.1007/s12264-020-00474-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Faroni A., Melfi S., Castelnovo L.F., Bonalume V., Colleoni D., Magni P., Araúzo-Bravo M.J., Reinbold R., Magnaghi V. GABA-B1 Receptor-Null Schwann Cells Exhibit Compromised In Vitro Myelination. Mol. Neurobiol. 2019;56:1461–1474. doi: 10.1007/s12035-018-1158-x. [DOI] [PubMed] [Google Scholar]
- 85.Serrano-Regal M.P., Luengas-Escuza I., Bayón-Cordero L., Ibarra-Aizpurua N., Alberdi E., Pérez-Samartín A., Matute C., Sánchez-Gómez M.V. Oligodendrocyte Differentiation and Myelination Is Potentiated via GABAB Receptor Activation. Neuroscience. 2020;439:163–180. doi: 10.1016/j.neuroscience.2019.07.014. [DOI] [PubMed] [Google Scholar]
- 86.Pudasaini S., Friedrich V., Bührer C., Endesfelder S., Scheuer T., Schmitz T. Postnatal Myelination of the Immature Rat Cingulum Is Regulated by GABAB Receptor Activity. Dev. Neurobiol. 2022;82:16–28. doi: 10.1002/dneu.22853. [DOI] [PubMed] [Google Scholar]
- 87.Favuzzi E., Huang S., Saldi G.A., Binan L., Ibrahim L.A., Fernández-Otero M., Cao Y., Zeine A., Sefah A., Zheng K., et al. GABA-Receptive Microglia Selectively Sculpt Developing Inhibitory Circuits. Cell. 2021;184:4048–4063.e32. doi: 10.1016/j.cell.2021.06.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Luhmann H.J., Prince D.A. Postnatal Maturation of the GABAergic System in Rat Neocortex. J. Neurophysiol. 1991;65:247–263. doi: 10.1152/jn.1991.65.2.247. [DOI] [PubMed] [Google Scholar]
- 89.Nurse S., Lacaille J.-C. Late Maturation of GABAB Synaptic Transmission in Area CA1 of the Rat Hippocampus. Neuropharmacology. 1999;38:1733–1742. doi: 10.1016/S0028-3908(99)00122-7. [DOI] [PubMed] [Google Scholar]
- 90.Discenna P.G., Nowicky A.V., Teyler T.J. The Development of GABAB-Mediated Activity in the Rat Dentate Gyrus. Dev. Brain Res. 1994;77:295–298. doi: 10.1016/0165-3806(94)90206-2. [DOI] [PubMed] [Google Scholar]
- 91.Fukuda A., Mody I., Prince D.A. Differential Ontogenesis of Presynaptic and Postsynaptic GABAB Inhibition in Rat Somatosensory Cortex. J. Neurophysiol. 1993;70:448–452. doi: 10.1152/jn.1993.70.1.448. [DOI] [PubMed] [Google Scholar]
- 92.Gaiarsa J.L., Tseeb V., Ben-Ari Y. Postnatal Development of Pre- and Postsynaptic GABAB-Mediated Inhibitions in the CA3 Hippocampal Region of the Rat. J. Neurophysiol. 1995;73:246–255. doi: 10.1152/jn.1995.73.1.246. [DOI] [PubMed] [Google Scholar]
- 93.Kantamneni S. Cross-Talk and Regulation between Glutamate and GABAB Receptors. Front. Cell. Neurosci. 2015;9:135. doi: 10.3389/fncel.2015.00135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Martinello K., Sciaccaluga M., Morace R., Mascia A., Arcella A., Esposito V., Fucile S. Loss of Constitutive Functional γ-Aminobutyric Acid Type A-B Receptor Crosstalk in Layer 5 Pyramidal Neurons of Human Epileptic Temporal Cortex. Epilepsia. 2018;59:449–459. doi: 10.1111/epi.13991. [DOI] [PubMed] [Google Scholar]
- 95.Kilb W. Development of the GABAergic System from Birth to Adolescence. Neuroscientist. 2012;18:613–630. doi: 10.1177/1073858411422114. [DOI] [PubMed] [Google Scholar]
- 96.Wright R., Newey S.E., Ilie A., Wefelmeyer W., Raimondo J.V., Ginham R., Mcllhinney R.A.J., Akerman C.J. Neuronal Chloride Regulation via KCC2 Is Modulated through a GABAB Receptor Protein Complex. J. Neurosci. 2017;37:5447–5462. doi: 10.1523/JNEUROSCI.2164-16.2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Lopantsev V., Schwartzkroin P.A. GABAA-Dependent Chloride Influx Modulates Reversal Potential of GABAB-Mediated IPSPs in Hippocampal Pyramidal Cells. J. Neurophysiol. 2001;85:2381–2387. doi: 10.1152/jn.2001.85.6.2381. [DOI] [PubMed] [Google Scholar]
- 98.Connelly W.M., Fyson S.J., Errington A.C., McCafferty C.P., Cope D.W., Giovanni G.D., Crunelli V. GABAB Receptors Regulate Extrasynaptic GABAA Receptors. J. Neurosci. 2013;33:3780–3785. doi: 10.1523/JNEUROSCI.4989-12.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Gerrow K., Triller A. GABAA Receptor Subunit Composition and Competition at Synapses Are Tuned by GABAB Receptor Activity. Mol. Cell. Neurosci. 2014;60:97–107. doi: 10.1016/j.mcn.2014.04.001. [DOI] [PubMed] [Google Scholar]
- 100.Laviv T., Riven I., Dolev I., Vertkin I., Balana B., Slesinger P.A., Slutsky I. Basal GABA Regulates GABABR Conformation and Release Probability at Single Hippocampal Synapses. Neuron. 2010;67:253–267. doi: 10.1016/j.neuron.2010.06.022. [DOI] [PubMed] [Google Scholar]
- 101.Liu Y., Yang X.J., Xia H., Tang C.-M., Yang K. GABA Releases from Parvalbumin-Expressing and Unspecific GABAergic Neurons onto CA1 Pyramidal Cells Are Differentially Modulated by Presynaptic GABAB Receptors in Mouse Hippocampus. Biochem. Biophys. Res. Commun. 2019;520:449–452. doi: 10.1016/j.bbrc.2019.10.044. [DOI] [PubMed] [Google Scholar]
- 102.Griguoli M., Cherubini E. Early Correlated Network Activity in the Hippocampus: Its Putative Role in Shaping Neuronal Circuits. Front. Cell. Neurosci. 2017;11:255. doi: 10.3389/fncel.2017.00255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.McLean H.A., Caillard O., Khazipov R., Ben-Ari Y., Gaiarsa J.L. Spontaneous Release of GABA Activates GABAB Receptors and Controls Network Activity in the Neonatal Rat Hippocampus. J. Neurophysiol. 1996;76:1036–1046. doi: 10.1152/jn.1996.76.2.1036. [DOI] [PubMed] [Google Scholar]
- 104.Khalilov I., Minlebaev M., Mukhtarov M., Juzekaeva E., Khazipov R. Postsynaptic GABA(B) Receptors Contribute to the Termination of Giant Depolarizing Potentials in CA3 Neonatal Rat Hippocampus. Front. Cell. Neurosci. 2017;11:179. doi: 10.3389/fncel.2017.00179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Hanganu-Opatz I.L., Butt S.J.B., Hippenmeyer S., De Marco García N.V., Cardin J.A., Voytek B., Muotri A.R. The Logic of Developing Neocortical Circuits in Health and Disease. J. Neurosci. 2021;41:813–822. doi: 10.1523/JNEUROSCI.1655-20.2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Scanziani M. GABA Spillover Activates Postsynaptic GABAB Receptors to Control Rhythmic Hippocampal Activity. Neuron. 2000;25:673–681. doi: 10.1016/S0896-6273(00)81069-7. [DOI] [PubMed] [Google Scholar]
- 107.Brown J.T., Davies C.H., Randall A.D. Synaptic Activation of GABAB Receptors Regulates Neuronal Network Activity and Entrainment. Eur. J. Neurosci. 2007;25:2982–2990. doi: 10.1111/j.1460-9568.2007.05544.x. [DOI] [PubMed] [Google Scholar]
- 108.Ulrich D., Lalanne T., Gassmann M., Bettler B. GABAB Receptor Subtypes Differentially Regulate Thalamic Spindle Oscillations. Neuropharmacology. 2018;136:106–116. doi: 10.1016/j.neuropharm.2017.10.033. [DOI] [PubMed] [Google Scholar]
- 109.Haider B. Neocortical Network Activity In Vivo Is Generated through a Dynamic Balance of Excitation and Inhibition. J. Neurosci. 2006;26:4535–4545. doi: 10.1523/JNEUROSCI.5297-05.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Mann E.O., Kohl M.M., Paulsen O. Distinct Roles of GABAA and GABAB Receptors in Balancing and Terminating Persistent Cortical Activity. J. Neurosci. 2009;29:7513–7518. doi: 10.1523/JNEUROSCI.6162-08.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Craig M.T., Mayne E.W., Bettler B., Paulsen O., McBain C.J. Distinct Roles of GABAB1a- and GABAB1b-Containing GABAB Receptors in Spontaneous and Evoked Termination of Persistent Cortical Activity. J. Physiol. 2013;591:835–843. doi: 10.1113/jphysiol.2012.248088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Perez-Zabalza M., Reig R., Manrique J., Jercog D., Winograd M., Parga N., Sanchez-Vives M.V. Modulation of Cortical Slow Oscillatory Rhythm by GABAB Receptors: An in Vitro Experimental and Computational Study. J. Physiol. 2020;598:3439–3457. doi: 10.1113/JP279476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Barbero-Castillo A., Mateos-Aparicio P., Porta L.D., Camassa A., Perez-Mendez L., Sanchez-Vives M.V. Impact of GABAA and GABAB Inhibition on Cortical Dynamics and Perturbational Complexity during Synchronous and Desynchronized States. J. Neurosci. 2021;41:5029–5044. doi: 10.1523/JNEUROSCI.1837-20.2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Oláh S., Füle M., Komlósi G., Varga C., Báldi R., Barzó P., Tamás G. Regulation of Cortical Microcircuits by Unitary GABA-Mediated Volume Transmission. Nature. 2009;461:1278–1281. doi: 10.1038/nature08503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Hay Y.A., Deperrois N., Fuchsberger T., Quarrell T.M., Koerling A.-L., Paulsen O. Thalamus Mediates Neocortical Down State Transition via GABAB-Receptor-Targeting Interneurons. Neuron. 2021;109:2682–2690.e5. doi: 10.1016/j.neuron.2021.06.030. [DOI] [PubMed] [Google Scholar]
- 116.Chittajallu R., Pelkey K.A., McBain C.J. Neurogliaform Cells Dynamically Regulate Somatosensory Integration via Synapse-Specific Modulation. Nat. Neurosci. 2013;16:13–15. doi: 10.1038/nn.3284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Gerrard L.B., Tantirigama M.L.S., Bekkers J.M. Pre- and Postsynaptic Activation of GABAB Receptors Modulates Principal Cell Excitation in the Piriform Cortex. Front. Cell. Neurosci. 2018;12:28. doi: 10.3389/fncel.2018.00028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Mazo C., Lepousez G., Nissant A., Valley M.T., Lledo P.-M. GABAB Receptors Tune Cortical Feedback to the Olfactory Bulb. J. Neurosci. 2016;36:8289–8304. doi: 10.1523/JNEUROSCI.3823-15.2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Orts-Del’Immagine A., Pugh J.R. Activity-Dependent Plasticity of Presynaptic GABAB Receptors at Parallel Fiber Synapses. Synapse. 2018;72:e22027. doi: 10.1002/syn.22027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Hassfurth B., Grothe B., Koch U. The Mammalian Interaural Time Difference Detection Circuit Is Differentially Controlled by GABAB Receptors during Development. J. Neurosci. 2010;30:9715–9727. doi: 10.1523/JNEUROSCI.1552-10.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Ainsworth M., Lee S., Kaiser M., Simonotto J., Kopell N.J., Whittington M.A. GABAB Receptor-Mediated, Layer-Specific Synaptic Plasticity Reorganizes Gamma-Frequency Neocortical Response to Stimulation. Proc. Natl. Acad. Sci. USA. 2016;113:E2721–E2729. doi: 10.1073/pnas.1605243113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Takesian A.E., Kotak V.C., Sanes D.H. Presynaptic GABAB Receptors Regulate Experience-Dependent Development of Inhibitory Short-Term Plasticity. J. Neurosci. 2010;30:2716–2727. doi: 10.1523/JNEUROSCI.3903-09.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Vickers E.D., Clark C., Fratzl A., Bettler B., Schneggenburger R. Spike-Timing Dependent Plasticity of Inhibition Gates Critical Period Remodeling in Auditory Cortex. Social Science Research Network; Rochester, NY, USA: 2018. [Google Scholar]
- 124.Cai S., Fischer Q.S., He Y., Zhang L., Liu H., Daw N.W., Yang Y. GABAB Receptor-Dependent Bidirectional Regulation of Critical Period Ocular Dominance Plasticity in Cats. PLoS ONE. 2017;12:e0180162. doi: 10.1371/journal.pone.0180162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Gassmann M. Redistribution of GABAB(1) Protein and Atypical GABAB Responses in GABAB(2)-Deficient Mice. J. Neurosci. 2004;24:6086–6097. doi: 10.1523/JNEUROSCI.5635-03.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Prosser H.M., Gill C.H., Hirst W.D., Grau E., Robbins M., Calver A., Soffin E.M., Farmer C.E., Lanneau C., Gray J., et al. Epileptogenesis and Enhanced Prepulse Inhibition in GABAB1-Deficient Mice. Mol. Cell. Neurosci. 2001;17:1059–1070. doi: 10.1006/mcne.2001.0995. [DOI] [PubMed] [Google Scholar]
- 127.Schuler V., Lüscher C., Blanchet C., Klix N., Sansig G., Klebs K., Schmutz M., Heid J., Gentry C., Urban L., et al. Epilepsy, Hyperalgesia, Impaired Memory, and Loss of Pre- and Postsynaptic GABAB Responses in Mice Lacking GABAB(1) Neuron. 2001;31:47–58. doi: 10.1016/S0896-6273(01)00345-2. [DOI] [PubMed] [Google Scholar]
- 128.Vertkin I., Styr B., Slomowitz E., Ofir N., Shapira I., Berner D., Fedorova T., Laviv T., Barak-Broner N., Greitzer-Antes D., et al. GABAB Receptor Deficiency Causes Failure of Neuronal Homeostasis in Hippocampal Networks. Proc. Natl. Acad. Sci. USA. 2015;112:E3291–E3299. doi: 10.1073/pnas.1424810112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Fritschy J.-M. Epilepsy, E/I Balance and GABAA Receptor Plasticity. Front. Mol. Neurosci. 2008;1:5. doi: 10.3389/neuro.02.005.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Sheilabi M.A., Battacharyya D., Caetano L., Thom M., Reuber M., Duncan J.S., Princivalle A.P. Quantitative Expression and Localization of GABAB Receptor Protein Subunits in Hippocampi from Patients with Refractory Temporal Lobe Epilepsy. Neuropharmacology. 2018;136:117–128. doi: 10.1016/j.neuropharm.2017.08.001. [DOI] [PubMed] [Google Scholar]
- 131.Inaba Y., D’Antuono M., Bertazzoni G., Biagini G., Avoli M. Diminished Presynaptic GABAB Receptor Function in the Neocortex of a Genetic Model of Absence Epilepsy. Neurosignals. 2009;17:121–131. doi: 10.1159/000197864. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Teichgräber L.A., Lehmann T.-N., Meencke H.-J., Weiss T., Nitsch R., Deisz R.A. Impaired Function of GABAB Receptors in Tissues from Pharmacoresistant Epilepsy Patients. Epilepsia. 2009;50:1697–1716. doi: 10.1111/j.1528-1167.2009.02094.x. [DOI] [PubMed] [Google Scholar]
- 133.Valente P., Farisello P., Valtorta F., Baldelli P., Benfenati F. Impaired GABAB-Mediated Presynaptic Inhibition Increases Excitatory Strength and Alters Short-Term Plasticity in Synapsin Knockout Mice. Oncotarget. 2017;8:90061–90076. doi: 10.18632/oncotarget.21405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Levinson S., Tran C.H., Barry J., Viker B., Levine M.S., Vinters H.V., Mathern G.W., Cepeda C. Paroxysmal Discharges in Tissue Slices From Pediatric Epilepsy Surgery Patients: Critical Role of GABAB Receptors in the Generation of Ictal Activity. Front. Cell. Neurosci. 2020;14:54. doi: 10.3389/fncel.2020.00054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Gennaccaro L., Fuchs C., Loi M., Roncacè V., Trazzi S., Ait-Bali Y., Galvani G., Berardi A.C., Medici G., Tassinari M., et al. A GABAB Receptor Antagonist Rescues Functional and Structural Impairments in the Perirhinal Cortex of a Mouse Model of CDKL5 Deficiency Disorder. Neurobiol. Dis. 2021;153:105304. doi: 10.1016/j.nbd.2021.105304. [DOI] [PubMed] [Google Scholar]
- 136.Fatemi S.H., Folsom T.D., Reutiman T.J., Thuras P.D. Expression of GABAB Receptors Is Altered in Brains of Subjects with Autism. Cerebellum. 2009;8:64–69. doi: 10.1007/s12311-008-0075-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Kang J.-Y., Chadchankar J., Vien T.N., Mighdoll M.I., Hyde T.M., Mather R.J., Deeb T.Z., Pangalos M.N., Brandon N.J., Dunlop J., et al. Deficits in the Activity of Presynaptic γ-Aminobutyric Acid Type B Receptors Contribute to Altered Neuronal Excitability in Fragile X Syndrome. J. Biol. Chem. 2017;292:6621–6632. doi: 10.1074/jbc.M116.772541. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Wahlstrom-Helgren S., Klyachko V.A. GABAB Receptor-Mediated Feed-Forward Circuit Dysfunction in the Mouse Model of Fragile X Syndrome. J. Physiol. 2015;593:5009–5024. doi: 10.1113/JP271190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Sinclair D., Featherstone R., Naschek M., Nam J., Du A., Wright S., Pance K., Melnychenko O., Weger R., Akuzawa S., et al. GABA-B Agonist Baclofen Normalizes Auditory-Evoked Neural Oscillations and Behavioral Deficits in the Fmr1 Knockout Mouse Model of Fragile X Syndrome. eNeuro. 2017;4 doi: 10.1523/ENEURO.0380-16.2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Gandal M.J., Sisti J., Klook K., Ortinski P.I., Leitman V., Liang Y., Thieu T., Anderson R., Pierce R.C., Jonak G., et al. GABAB-Mediated Rescue of Altered Excitatory–Inhibitory Balance, Gamma Synchrony and Behavioral Deficits Following Constitutive NMDAR-Hypofunction. Transl. Psychiatry. 2012;2:e142. doi: 10.1038/tp.2012.69. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Henderson C., Wijetunge L., Kinoshita M.N., Shumway M., Hammond R.S., Postma F.R., Brynczka C., Rush R., Thomas A., Paylor R., et al. Reversal of Disease-Related Pathologies in the Fragile X Mouse Model by Selective Activation of GABAB Receptors with Arbaclofen. Sci. Transl. Med. 2012;4:152ra128. doi: 10.1126/scitranslmed.3004218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Qin M., Huang T., Kader M., Krych L., Xia Z., Burlin T., Zeidler Z., Zhao T., Smith C.B. R-Baclofen Reverses a Social Behavior Deficit and Elevated Protein Synthesis in a Mouse Model of Fragile X Syndrome. Int. J. Neuropsychopharmacol. 2015;18:pyv034. doi: 10.1093/ijnp/pyv034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Kramvis I., van Westen R., Lammertse H.C.A., Riga D., Heistek T.S., Loebel A., Spijker S., Mansvelder H.D., Meredith R.M. Dysregulated Prefrontal Cortex Inhibition in Prepubescent and Adolescent Fragile X Mouse Model. Front. Mol. Neurosci. 2020;13:88. doi: 10.3389/fnmol.2020.00088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Bassetti D., Luhmann H.J., Kirischuk S. Effects of Mutations in TSC Genes on Neurodevelopment and Synaptic Transmission. Int. J. Mol. Sci. 2021;22:7273. doi: 10.3390/ijms22147273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Bassetti D., Lombardi A., Kirischuk S., Luhmann H.J. Haploinsufficiency of Tsc2 Leads to Hyperexcitability of Medial Prefrontal Cortex via Weakening of Tonic GABAB Receptor-Mediated Inhibition. Cereb. Cortex. 2020;30:6313–6324. doi: 10.1093/cercor/bhaa187. [DOI] [PubMed] [Google Scholar]
- 146.Bassetti D., Luhmann H.J., Kirischuk S. Presynaptic GABAB Receptor–Mediated Network Excitation in the Medial Prefrontal Cortex of Tsc2+/- Mice. Pflüg. Arch.-Eur.J. Physiol. 2021;473:1261–1271. doi: 10.1007/s00424-021-02576-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Winkler M., Biswas S., Berger S.M., Küchler M., Preisendörfer L., Choo M., Früh S., Rem P.D., Enkel T., Arnold B., et al. Pianp Deficiency Links GABAB Receptor Signaling and Hippocampal and Cerebellar Neuronal Cell Composition to Autism-like Behavior. Mol. Psychiatry. 2020;25:2979–2993. doi: 10.1038/s41380-019-0519-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Iwakiri M., Mizukami K., Ikonomovic M.D., Ishikawa M., Hidaka S., Abrahamson E.E., DeKosky S.T., Asada T. Changes in Hippocampal GABABR1 Subunit Expression in Alzheimer’s Patients: Association with Braak Staging. Acta Neuropathol. 2005;109:467–474. doi: 10.1007/s00401-005-0985-9. [DOI] [PubMed] [Google Scholar]
- 149.Martín-Belmonte A., Aguado C., Alfaro-Ruíz R., Moreno-Martínez A.E., de la Ossa L., Martínez-Hernández J., Buisson A., Shigemoto R., Fukazawa Y., Luján R. Density of GABAB Receptors Is Reduced in Granule Cells of the Hippocampus in a Mouse Model of Alzheimer’s Disease. Int. J. Mol. Sci. 2020;21:2459. doi: 10.3390/ijms21072459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Dinamarca M.C., Raveh A., Schneider A., Fritzius T., Früh S., Rem P.D., Stawarski M., Lalanne T., Turecek R., Choo M., et al. Complex Formation of APP with GABAB Receptors Links Axonal Trafficking to Amyloidogenic Processing. Nat. Commun. 2019;10:1331. doi: 10.1038/s41467-019-09164-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Sun Z., Sun L., Tu L. GABA B Receptor-Mediated PI3K/Akt Signaling Pathway Alleviates Oxidative Stress and Neuronal Cell Injury in a Rat Model of Alzheimer’s Disease. J. Alzheimers Dis. 2020;76:1513–1526. doi: 10.3233/JAD-191032. [DOI] [PubMed] [Google Scholar]
- 152.Kumar K., Kaur H., Deshmukh R. Neuroprotective Role of GABAB Receptor Modulation against Streptozotocin-Induced Behavioral and Biochemical Abnormalities in Rats. Neuroscience. 2017;357:67–74. doi: 10.1016/j.neuroscience.2017.05.054. [DOI] [PubMed] [Google Scholar]
- 153.Pilipenko V., Narbute K., Beitnere U., Rumaks J., Pupure J., Jansone B., Klusa V. Very Low Doses of Muscimol and Baclofen Ameliorate Cognitive Deficits and Regulate Protein Expression in the Brain of a Rat Model of Streptozocin-Induced Alzheimer’s Disease. Eur. J. Pharmacol. 2018;818:381–399. doi: 10.1016/j.ejphar.2017.11.012. [DOI] [PubMed] [Google Scholar]
- 154.Tosetti P., Ferrand N., Brun I.C.-L., Gaïarsa J.L. Epileptiform Activity Triggers Long-Term Plasticity of GABAB Receptor Signalling in the Developing Rat Hippocampus. J. Physiol. 2005;568:951–966. doi: 10.1113/jphysiol.2005.094631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Leung L.S. Long-Lasting Changes in Hippocampal GABAB-Receptor Mediated Inhibition Following Early-Life Seizures in Kindling-Prone but Not Kindling-Resistant Rats. Brain Res. Bull. 2019;150:231–239. doi: 10.1016/j.brainresbull.2019.06.006. [DOI] [PubMed] [Google Scholar]
- 156.Tsai M.-L., Shen B., Leung L.S. Seizures Induced by GABAB-Receptor Blockade in Early-Life Induced Long-Term GABAB Receptor Hypofunction and Kindling Facilitation. Epilepsy Res. 2008;79:187–200. doi: 10.1016/j.eplepsyres.2008.02.001. [DOI] [PubMed] [Google Scholar]
- 157.Gomez C.D., Acharjee S., Lewis M.L., Read J., Pittman Q.J. Increased Excitatory Synaptic Transmission Associated with Adult Seizure Vulnerability Induced by Early-Life Inflammation in Mice. J. Neurosci. 2021;41:4367–4377. doi: 10.1523/JNEUROSCI.2667-20.2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Sweeney F.F., O’Leary O.F., Cryan J.F. Activation but Not Blockade of GABAB Receptors during Early-Life Alters Anxiety in Adulthood in BALB/c Mice. Neuropharmacology. 2014;81:303–310. doi: 10.1016/j.neuropharm.2013.08.039. [DOI] [PubMed] [Google Scholar]
- 159.Bolton M.M., Heaney C.F., Murtishaw A.S., Sabbagh J.J., Magcalas C.M., Kinney J.W. Postnatal Alterations in GABAB Receptor Tone Produce Sensorimotor Gating Deficits and Protein Level Differences in Adulthood. Int. J. Dev. Neurosci. 2015;41:17–27. doi: 10.1016/j.ijdevneu.2014.10.001. [DOI] [PubMed] [Google Scholar]
- 160.Gao Y., Zhou J.-J., Zhu Y., Wang L., Kosten T.A., Zhang X., Li D.-P. Neuroadaptations of Presynaptic and Postsynaptic GABAB Receptor Function in the Paraventricular Nucleus in Response to Chronic Unpredictable Stress. Br. J. Pharmacol. 2017;174:2929–2940. doi: 10.1111/bph.13924. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Liu L., Ito W., Morozov A. GABAb Receptor Mediates Opposing Adaptations of GABA Release From Two Types of Prefrontal Interneurons After Observational Fear. Neuropsychopharmacology. 2017;42:1272–1283. doi: 10.1038/npp.2016.273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.O’Leary O.F., Felice D., Galimberti S., Savignac H.M., Bravo J.A., Crowley T., Yacoubi M.E., Vaugeois J.-M., Gassmann M., Bettler B., et al. GABAB(1) Receptor Subunit Isoforms Differentially Regulate Stress Resilience. Proc. Natl. Acad. Sci. USA. 2014;111:15232–15237. doi: 10.1073/pnas.1404090111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.O’Leary O.F., Cryan J.F. Chapter 5—GABAB receptors, depression, and stress resilience: A tale of two isoforms. In: Chen A., editor. Stress Resilience. Academic Press; Cambridge, MA, USA: 2020. pp. 63–79. [Google Scholar]
- 164.Deng S.-L., Hu Z.-L., Mao L., Gao B., Yang Q., Wang F., Chen J.-G. The Effects of Kctd12, an Auxiliary Subunit of GABAB Receptor in Dentate Gyrus on Behavioral Response to Chronic Social Defeat Stress in Mice. Pharmacol. Res. 2021;163:105355. doi: 10.1016/j.phrs.2020.105355. [DOI] [PubMed] [Google Scholar]
- 165.Sahraei H., Askaripour M., Esmaeilpour K., Shahsavari F., Rajabi S., Moradi-Kor N. GABAB Receptor Activation Ameliorates Spatial Memory Impairments in Stress-Exposed Rats. Neuropsychiatr. Dis. Treat. 2019;15:1497–1506. doi: 10.2147/NDT.S205951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Alexander R.C. The Potential Efficacy of GABAB Antagonists in Depression. Curr. Opin. Pharmacol. 2017;35:101–104. doi: 10.1016/j.coph.2017.07.009. [DOI] [PubMed] [Google Scholar]
- 167.Felice D., O’Leary O.F., Cryan J.F. Targeting the GABAB receptor for the treatment of depression and anxiety disorders. In: Colombo G., editor. GABAB Receptor. Springer International Publishing; Cham, Switzerland: 2016. pp. 219–250. The Receptors. [Google Scholar]
- 168.Filip M., Frankowska M., Sadakierska-Chudy A., Suder A., Szumiec Ł., Mierzejewski P., Bienkowski P., Przegaliński E., Cryan J.F. GABAB Receptors as a Therapeutic Strategy in Substance Use Disorders: Focus on Positive Allosteric Modulators. Neuropharmacology. 2015;88:36–47. doi: 10.1016/j.neuropharm.2014.06.016. [DOI] [PubMed] [Google Scholar]
- 169.Malcangio M. GABAB Receptors and Pain. Neuropharmacology. 2018;136:102–105. doi: 10.1016/j.neuropharm.2017.05.012. [DOI] [PubMed] [Google Scholar]
- 170.Xu M., Wong A.H.C. GABAergic Inhibitory Neurons as Therapeutic Targets for Cognitive Impairment in Schizophrenia. Acta Pharmacol. Sin. 2018;39:733–753. doi: 10.1038/aps.2017.172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Serrats J., Cunningham M.O., Davies C.H. GABAB Receptor Modulation—to B or Not to Be B a pro-Cognitive Medicine? Curr. Opin. Pharmacol. 2017;35:125–132. doi: 10.1016/j.coph.2017.09.012. [DOI] [PubMed] [Google Scholar]
- 172.Curtis D.R., Lodge D., Bornstein J.C., Peet M.J. Selective Effects of (−)-Baclofen on Spinal Synaptic Transmission in the Cat. Exp. Brain Res. 1981;42:158–170. doi: 10.1007/BF00236902. [DOI] [PubMed] [Google Scholar]
- 173.Froestl W., Gallagher M., Jenkins H., Madrid A., Melcher T., Teichman S., Mondadori C.G., Pearlman R. SGS742: The First GABAB Receptor Antagonist in Clinical Trials. Biochem. Pharmacol. 2004;68:1479–1487. doi: 10.1016/j.bcp.2004.07.030. [DOI] [PubMed] [Google Scholar]
- 174.Silverman J.L., Pride M.C., Hayes J.E., Puhger K.R., Butler-Struben H.M., Baker S., Crawley J.N. GABA B Receptor Agonist R-Baclofen Reverses Social Deficits and Reduces Repetitive Behavior in Two Mouse Models of Autism. Neuropsychopharmacology. 2015;40:2228–2239. doi: 10.1038/npp.2015.66. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Stoppel L.J., Kazdoba T.M., Schaffler M.D., Preza A.R., Heynen A., Crawley J.N., Bear M.F. R -Baclofen Reverses Cognitive Deficits and Improves Social Interactions in Two Lines of 16p11.2 Deletion Mice. Neuropsychopharmacology. 2018;43:513–524. doi: 10.1038/npp.2017.236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Möhrle D., Wang W., Whitehead S.N., Schmid S. GABAB Receptor Agonist R-Baclofen Reverses Altered Auditory Reactivity and Filtering in the Cntnap2 Knock-Out Rat. Front. Integr. Neurosci. 2021;15:710593. doi: 10.3389/fnint.2021.710593. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Erickson C.A., Veenstra-Vanderweele J.M., Melmed R.D., McCracken J.T., Ginsberg L.D., Sikich L., Scahill L., Cherubini M., Zarevics P., Walton-Bowen K., et al. STX209 (Arbaclofen) for Autism Spectrum Disorders: An 8-Week Open-Label Study. J. Autism Dev. Disord. 2014;44:958–964. doi: 10.1007/s10803-013-1963-z. [DOI] [PubMed] [Google Scholar]
- 178.Veenstra-VanderWeele J., Cook E.H., King B.H., Zarevics P., Cherubini M., Walton-Bowen K., Bear M.F., Wang P.P., Carpenter R.L. Arbaclofen in Children and Adolescents with Autism Spectrum Disorder: A Randomized, Controlled, Phase 2 Trial. Neuropsychopharmacol. Off. Publ. Am. Coll. Neuropsychopharmacol. 2017;42:1390–1398. doi: 10.1038/npp.2016.237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Mahdavinasab S.-M., Saghazadeh A., Motamed-Gorji N., Vaseghi S., Mohammadi M.-R., Alichani R., Akhondzadeh S. Baclofen as an Adjuvant Therapy for Autism: A Randomized, Double-Blind, Placebo-Controlled Trial. Eur. Child Adolesc. Psychiatry. 2019;28:1619–1628. doi: 10.1007/s00787-019-01333-5. [DOI] [PubMed] [Google Scholar]
- 180.Huang Q., Pereira A.C., Velthuis H., Wong N.M.L., Ellis C.L., Ponteduro F.M., Dimitrov M., Kowalewski L., Lythgoe D.J., Rotaru D., et al. GABAB Receptor Modulation of Visual Sensory Processing in Adults with and without Autism Spectrum Disorder. Sci. Transl. Med. 2022;14:eabg7859. doi: 10.1126/scitranslmed.abg7859. [DOI] [PubMed] [Google Scholar]
- 181.Berry-Kravis E., Hagerman R., Visootsak J., Budimirovic D., Kaufmann W.E., Cherubini M., Zarevics P., Walton-Bowen K., Wang P., Bear M.F., et al. Arbaclofen in Fragile X Syndrome: Results of Phase 3 Trials. J. Neurodev. Disord. 2017;9:3. doi: 10.1186/s11689-016-9181-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182.Ertzgaard P., Campo C., Calabrese A. Efficacy and Safety of Oral Baclofen in the Management of Spasticity: A Rationale for Intrathecal Baclofen. J. Rehabil. Med. 2017;49:193–203. doi: 10.2340/16501977-2211. [DOI] [PubMed] [Google Scholar]
- 183.Sandyk R., Gillman M.A. Baclofen-Induced Memory Impairment. Clin. Neuropharmacol. 1985;8:294–295. doi: 10.1097/00002826-198509000-00011. [DOI] [PubMed] [Google Scholar]
- 184.Zeman A., Hoefeijzers S., Milton F., Dewar M., Carr M., Streatfield C. The GABAB Receptor Agonist, Baclofen, Contributes to Three Distinct Varieties of Amnesia in the Human Brain—A Detailed Case Report. Cortex. 2016;74:9–19. doi: 10.1016/j.cortex.2015.10.005. [DOI] [PubMed] [Google Scholar]
- 185.Urwyler S., Mosbacher J., Lingenhoehl K., Heid J., Hofstetter K., Froestl W., Bettler B., Kaupmann K. Positive Allosteric Modulation of Native and Recombinant γ-Aminobutyric AcidB Receptors by 2,6-Di-Tert-Butyl-4-(3-Hydroxy-2,2-Dimethyl-Propyl)-Phenol (CGP7930) and Its Aldehyde Analog CGP13501. Mol. Pharmacol. 2001;60:963–971. doi: 10.1124/mol.60.5.963. [DOI] [PubMed] [Google Scholar]
- 186.Urwyler S., Pozza M.F., Lingenhoehl K., Mosbacher J., Lampert C., Froestl W., Koller M., Kaupmann K. N,N′-Dicyclopentyl-2-Methylsulfanyl-5-Nitro-Pyrimidine-4,6-Diamine (GS39783) and Structurally Related Compounds: Novel Allosteric Enhancers of γ-Aminobutyric AcidB Receptor Function. J. Pharmacol. Exp. Ther. 2003;307:322–330. doi: 10.1124/jpet.103.053074. [DOI] [PubMed] [Google Scholar]
- 187.Malherbe P., Masciadri R., Norcross R.D., Knoflach F., Kratzeisen C., Zenner M.-T., Kolb Y., Marcuz A., Huwyler J., Nakagawa T., et al. Characterization of (R,S)-5,7-Di-Tert-Butyl-3-Hydroxy-3-Trifluoromethyl-3H-Benzofuran-2-One as a Positive Allosteric Modulator of GABAB Receptors. Br. J. Pharmacol. 2008;154:797–811. doi: 10.1038/bjp.2008.135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188.Liu L., Fan Z., Rovira X., Xue L., Roux S., Brabet I., Xin M., Pin J.-P., Rondard P., Liu J. Allosteric Ligands Control the Activation of a Class C GPCR Heterodimer by Acting at the Transmembrane Interface. eLife. 2021;10:e70188. doi: 10.7554/eLife.70188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 189.Cryan J.F., Kelly P.H., Chaperon F., Gentsch C., Mombereau C., Lingenhoehl K., Froestl W., Bettler B., Kaupmann K., Spooren W.P.J.M. Behavioral Characterization of the Novel GABAB Receptor-Positive Modulator GS39783 (N,N′-Dicyclopentyl-2-Methylsulfanyl-5-Nitro-Pyrimidine-4,6-Diamine): Anxiolytic-Like Activity without Side Effects Associated with Baclofen or Benzodiazepines. J. Pharmacol. Exp. Ther. 2004;310:952–963. doi: 10.1124/jpet.104.066753. [DOI] [PubMed] [Google Scholar]
- 190.Hensler J.G., Advani T., Burke T.F., Cheng K., Rice K.C., Koek W. GABAB Receptor-Positive Modulators: Brain Region-Dependent Effects. J. Pharmacol. Exp. Ther. 2012;340:19–26. doi: 10.1124/jpet.111.186577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191.Sturchler E., Li X., de Lourdes Ladino M., Kaczanowska K., Cameron M., Griffin P.R., Finn M.G., Markou A., McDonald P. GABAB Receptor Allosteric Modulators Exhibit Pathway-Dependent and Species-Selective Activity. Pharmacol. Res. Perspect. 2017;5:e00288. doi: 10.1002/prp2.288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Maccioni P., Fara F., Lorrai I., Acciaro C., Mugnaini C., Corelli F., Colombo G. Suppressing Effect of CMPPE, a New Positive Allosteric Modulator of the GABAB Receptor, on Alcohol Self-Administration and Reinstatement of Alcohol Seeking in Rats. Alcohol. 2019;75:79–87. doi: 10.1016/j.alcohol.2018.05.015. [DOI] [PubMed] [Google Scholar]
- 193.Augier E. Recent Advances in the Potential of Positive Allosteric Modulators of the GABAB Receptor to Treat Alcohol Use Disorder. Alcohol Alcohol. 2021;56:139–148. doi: 10.1093/alcalc/agab003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Chen L.-H., Sun B., Zhang Y., Xu T.-J., Xia Z.-X., Liu J.-F., Nan F.-J. Discovery of a Negative Allosteric Modulator of GABAB Receptors. ACS Med. Chem. Lett. 2014;5:742–747. doi: 10.1021/ml500162z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195.Sun B., Chen L., Liu L., Xia Z., Pin J.-P., Nan F., Liu J. A Negative Allosteric Modulator Modulates GABAB-Receptor Signalling through GB2 Subunits. Biochem. J. 2016;473:779–787. doi: 10.1042/BJ20150979. [DOI] [PubMed] [Google Scholar]
- 196.Porcu A., Mostallino R., Serra V., Melis M., Sogos V., Beggiato S., Ferraro L., Manetti F., Gianibbi B., Bettler B., et al. COR758, a Negative Allosteric Modulator of GABAB Receptors. Neuropharmacology. 2021;189:108537. doi: 10.1016/j.neuropharm.2021.108537. [DOI] [PubMed] [Google Scholar]
- 197.Sereikaite V., Fritzius T., Kasaragod V.B., Bader N., Maric H.M., Schindelin H., Bettler B., Strømgaard K. Targeting the γ-Aminobutyric Acid Type B (GABAB) Receptor Complex: Development of Inhibitors Targeting the K+ Channel Tetramerization Domain (KCTD) Containing Proteins/GABAB Receptor Protein–Protein Interaction. J. Med. Chem. 2019;62:8819–8830. doi: 10.1021/acs.jmedchem.9b01087. [DOI] [PubMed] [Google Scholar]
- 198.Lu H., Zhou Q., He J., Jiang Z., Peng C., Tong R., Shi J. Recent Advances in the Development of Protein–Protein Interactions Modulators: Mechanisms and Clinical Trials. Signal Transduct. Target. Ther. 2020;5:213. doi: 10.1038/s41392-020-00315-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not applicable.