Abstract
V-echinocandin (VER-002; LY303366) is a semisynthetic derivative of echinocandin B and a potent inhibitor of fungal (1, 3)-β-d-glucan synthase. We studied the antifungal efficacy, the concentrations in saliva and tissue, and the safety of VER-002 at escalating dosages against experimental oropharyngeal and esophageal candidiasis caused by fluconazole-resistant Candida albicans in immunocompromised rabbits. Study groups consisted of untreated controls, animals treated with VER-002 at 1, 2.5, and 5 mg/kg of body weight/day intravenously (i.v.), animals treated with fluconazole at 2 mg/kg/day i.v., or animals treated with amphotericin B at 0.3 mg/kg/day. VER-002-treated animals showed a significant dosage-dependent clearance of C. albicans from the tongue, oropharynx, esophagus, stomach, and duodenum in comparison to that for untreated controls. VER-002 also was superior to amphotericin B and fluconazole in clearing the organism from all sites studied. These in vivo findings are consistent with the results of in vitro time-kill assays, which demonstrated that VER-002 has concentration-dependent fungicidal activity. Esophageal tissue VER-002 concentrations were dosage proportional and exceeded the MIC at all dosages. Echinocandin concentrations in saliva were greater than or equal to the MICs at all dosages. There was no elevation of serum hepatic transaminase, alkaline phosphatase, bilirubin, potassium, or creatinine levels in VER-002-treated rabbits. In summary, the echinocandin VER-002 was well tolerated, penetrated the esophagus and salivary glands, and demonstrated dosage-dependent antifungal activity against fluconazole-resistant esophageal candidiasis in immunocompromised rabbits.
Esophageal candidiasis is one of the most common opportunistic fungal infections in immunocompromised patients, including human immunodeficiency virus (HIV)-positive patients and those who are immunosuppressed as a result of underlying diseases or medications (2, 35). A number of agents have been used to treat esophageal candidiasis, including nystatin, miconazole, ketoconazole, fluconazole, itraconazole, and amphotericin B. Fluconazole is frequently selected for systemic therapy because it is well tolerated and has excellent oral bioavailability. During the past several years, however, there have been increasing reports of fluconazole-resistant oropharyngeal and esophageal candidiasis (OPEC) (27). The emergence of fluconazole-resistant OPEC has heightened the need for development of new antifungal compounds with novel targets.
The echinocandins are semisynthetic lipopeptides with potent and broad-spectrum antifungal activity which act by inhibiting the synthesis of (1,3)-β-d-glucan, leading to cell wall damage and ultimately cell death (6, 7, 10, 16, 17). This novel mode of action and potent antifungal activity in vitro have led to the design of several new echinocandin compounds for potential clinical development.
V-echinocandin (VER-002; LY303366) is a semisynthetic echinocandin B derivative (9; I. Rajman, K. Desante, B. Hatcher, J. Hemingway, R. Lachno, S. Brooks, and M. Turik, Abstr. 37th Intersci. Conf. Antimicrob. Agents Chemother, abstr. F-74, p. 158, 1997) which demonstrates potent and non-cross-resistant antifungal activity against Candida albicans, Candida tropicalis, Candida glabrata, and other non-C. albicans species (8, 13, 14, 15, 24, 26, 29). Little is known, however, about the activity of VER-002 is treatment for fluconazole-resistant OPEC. We therefore investigated the concentrations in the esophageus and saliva the safety, and the antifungal efficacy of VER-002 in an immunosuppressed rabbit model of fluconazole-resistant OPEC.
MATERIALS AND METHODS
Organisms and MICs.
Two isolates (isolates NIH 105 and NIH 126) of fluconazole-resistant C. albicans were used for all experiments. Each isolate was obtained from an HIV-infected child who had fluconazole-resistant OPEC and who was monitored at the Pediatric Oncology Branch of the National Cancer Institute. MICs were determined by National Committee for Clinical Laboratory Standards reference methods as described previously (23). The MICs of fluconazole (Roerig-Pfizer, New York, N.Y.) and VER-002 (Eli Lilly & Company, Indianapolis, Ind.) were determined in morpholinepropanesulfonic acid (MOPS)-buffered RPMI 1640 (BioWhittaker, Walkersville, Md.), and the MICs of amphotericin B (Bristol-Myers Squibb Company, Princeton, N.J.), purchased as Fungizone, were measured in antibiotic medium 3 (AM3; National Institutes of Health Media Department, Bethesda, Md.). The MIC was defined as the concentration that resulted in complete optical clearance of fungal growth. The MICs for the isolates studied in these experiments are shown in Table 1.
TABLE 1.
MICs for isolates from rabbits with experimental fluconazole-resistant OPEC
| C. albicans isolate | MIC (μg/ml)
|
||
|---|---|---|---|
| Fluconazolea | VER-002a | Amphotericin Bb | |
| NIH 105 | >64 | 0.015 | 0.05 |
| NIH 126 | >64 | 0.004 | 0.05 |
MOPS-buffered RPMI 1640.
AM3.
Time-kill assay.
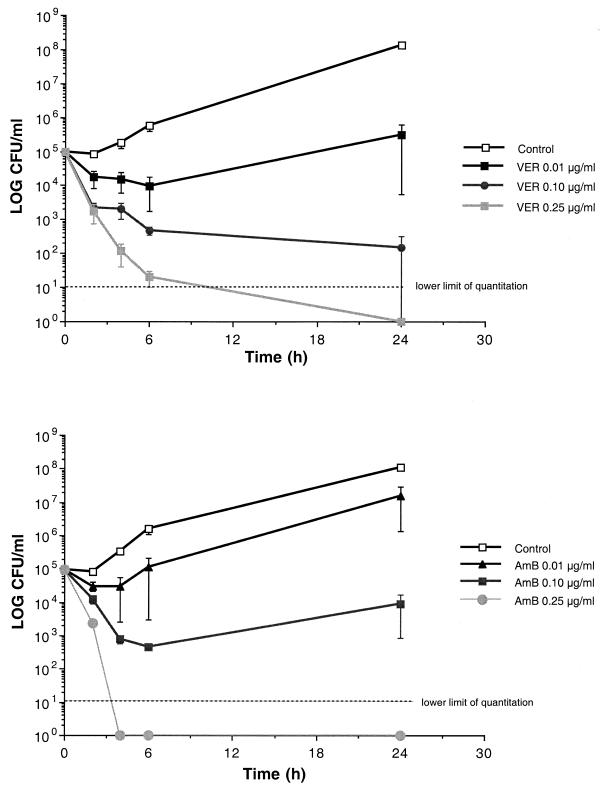
In order to characterize the in vitro pharmacodynamics and potential fungicidal activities of VER-002 and amphotericin B, time-kill assays were performed against C. albicans (NIH 105 and NIH 126). Three concentrations of VER-002 and amphotericin B (0.01, 0.1, and 0.25 μg/ml) were studied. These concentrations span the range of MICs of the two compounds. The inoculum for the time-kill assay was prepared by growing the isolate for 48 h at 37°C on Sabouraud glucose agar (SGA), inoculating colonies into a starter broth of 50 ml of Sabouraud glucose broth, and incubating the colonies in the broth for 2 h in a shaking water bath at 37°C. One milliliter of this suspension was transferred into 50 ml of fresh AM3 broth in each of four 250-ml Erienmeyer flasks, and the flasks were incubated at 37°C for 16 h in a shaking water bath in order to generate logarithmic phase growth. The suspension was centrifuged, washed three times, adjusted for concentration with a hemacytometer, and inoculated into 250-ml Erlenmeyer flasks containing 50 ml of AM3 broth alone (untreated growth control) and AM3 plus antifungal compound. The final baseline (time zero) concentration of approximately 3.0 × 105 CFU/ml was confirmed by quantitative culture. These flasks were incubated simultaneously in a shaking water bath at 37°C. The growth suspensions were sampled at time zero and at 2, 4, 6, and 24 h, and 100-μl aliquots were plated in dilutions of 10−2, 10−3, and 10−4 onto one SGA plate/per aliquot. The colonies were counted after 48 h of incubation at 37°C, and the calculated number of CFU/per milliliter was plotted for each time point. The lower limit of quantitation for the time-kill assay was 10 CFU/ml. Time-kill assays for all concentrations were performed in triplicate.
Animals.
Sixty-three female New Zealand White rabbits (Hazleton, Deutschland, Pa.) weighing 2.5 to 3.5 kg at the time of inoculation were used in all experiments. Rabbits were individually housed and were maintained according to National Institutes of Health guidelines for animal care and in fulfillment of American Association for Accreditation of Laboratory Animal Care standards (5). Vascular access was established in each rabbit by the surgical placement of a tunneled silastic central venous catheter (30). The silastic catheter permitted nontraumatic venous access for repeated blood sampling for study of biochemical safety parameters and pharmacokinetics in plasma, as well as administration of parenteral agents. Rabbits were euthanatized at the end of each experiment by injection of an intravenous (i.v.) bolus of pentobarbital (65 mg [1 ml]/kg of body weight; Veterinary Laboratories, Inc., Lenexa, Kans.).
Oral inoculation.
Organisms from stock isolates were stored in skim milk at −70°C. Cells from these suspensions were streaked onto SGA plates, and the plates were incubated at 37°C for 24 h and then maintained at 4°C. Five discrete colonies were then inoculated into 50 ml of Emmon's modified Sabouraud broth (pH 7) in a 250-ml Erlenmeyer flask, and the flask was incubated at 37°C for 16 h on a shaking incubator at 80 oscillations per minute. The Candida suspension was centrifuged at 3,000 × g for 10 min and was washed three times with sterile normal saline (Quality Biological, Inc., Gaithersburg, Md.). Candida blastoconidia were counted with a hemacytometer and diluted to the desired concentration. An inoculum of 2 × 108 blastoconidia was suspended in 1.5 ml of normal saline, and the suspension was administered per os from day 1 through day 7 of the experiment. There were no interisolate differences in the capacity to establish oropharyngeal or esophageal infection.
Immunosuppression and antibiotics.
Methylprednisolone (Abbott Laboratories, North Chicago, Ill.), at 5 mg/kg of body weight was administered from day 1 to day 14 of the experiment for suppression of mucosal cellular immunity. Gentamicin (Elkins-Sinn, Inc., Cherry Hill, N.J.) at 40 mg/liter and vancomycin (Abbott Laboratories) at 50 mg/liter were administered in the drinking water starting on day 1 and continuing through day 14 in order to reduce mucosal bacterial colonization competitive with C. albicans.
Antifungal compounds and treatment groups.
Therapy was initiated on day 8 of the experiment following per os inoculation and was continued throughout the course of the experiments for 7 days. Eli Lilly & Company provided VER-002 as a 10-mg/ml solution for parenteral administration. VER-002 was administered i.v. at dosages of 1 mg/kg/day (VER1) (n = 9), 2.5 mg/kg/day (VER2.5) (n = 9), and 5 mg/kg/day (VER5) (n = 9). The initial solution of 10 mg/ml was diluted with sterile 0.9% NaCl to a concentration of 1 mg/ml for the 1-mg/kg dosage and to a concentration of 2 mg/ml for the 2.5-mg/kg dosage. VER5 was administered in an initial solution of 10 mg/ml. Amphotericin B at 0.3 mg/kg/day (n = 9) was administered as a slow i.v. infusion (0.1 ml every 10) in order to assess the low-dose regimen of amphotericin B which is used in the management of esophageal candidiasis (22). Fluconazole was administered at 1 mg/kg (n = 9) twice daily i.v. Administration of compounds was initiated 24 h after the last inoculation on day 7 of the experiment. The untreated control group consisted of saline-treated infected rabbits that did not receive antifungal treatment (n = 18).
Outcome variables.
The rabbit model of fluconazole-resistant OPEC provides a strong in vitro-in vivo correlation of the therapeutic response to fluconazole (31). This system permits quantitative assessment of therapeutic response along clinically relevant sites of the upper alimentary tract: tongue, oropharynx, esophagus (proximal, middle, and distal), stomach, and duodenum. The tongue, oropharynx, and esophagus were resected en bloc postmortem. Segments of duodenum and stomach were resected separately. Antifungal efficacy was assessed by microbiologic clearance of C. albicans from tissue. Sections of the tongue, oropharynx, esophagus (proximal, middle, and distal), stomach, and duodenum were cultured by excision of a representative region. Each fragment was weighed and then homogenized in sterile reinforced polyethylene bags (Tekmar Corp., Cincinnati, Ohio) (34). Each tissue homogenate was serially diluted 100-fold from 10 to 10−4 in sterile 0.9% saline. A 0.1-ml quantity of undiluted homogenate and of each dilution was separately plated onto Emmon's modified SGA containing chloramphenicol and gentamicin. Culture plates were incubated at 37°C for 24 h, after which the members of CFU were counted and the number of CFU per gram of tissue was calculated for each organ. Previous studies with serial dilutions demonstrated that the method was sensitive to detection of ≥10 CFU/g. The culture-negative plates were counted as having 0 CFU/g. Data were graphed as the mean ± standard error of the mean (SEM) log10 (CFU per gram).
Pharmacokinetic and pharmacodynamic studies.
Drug concentrations in plasma, saliva, and esophageal tissue were determined for all treated animals, and the relationships between drug levels and residual fungal burden were investigated for VER-002-treated rabbits. Plasma and saliva were sampled from each rabbit 2 h after administration of the seventh dose and after induction of salivation with 0.5 mg of pilocarpine (catalog no. 45H05181i Sigma), and esophageal tissue was obtained 24 h after administration of the seventh dose at autopsy. All samples were stored at −80°C until assay.
The concentrations of VER-002 were determined after solid-phase extraction by reversed-phase high-performance liquid chromatography (HPLC) as described previously (25). Esophageal tissue was homogenized prior to extraction in ice-cold normal saline (1:10 [wt/wt]) with using a tissue homogenizer (Tizzumizer; Tekmar Corp.), and drug concentrations in tissue were calculated for 1 g of tissue. External standards and quality controls were prepared separately for all matrices by spiking pooled normal rabbit serum, saliva, and esophageal homogenate with VER-002 and the internal standard. Eight-point standard curves were linear, with R2 values of ≥0.993. Accuracies were within ±5%, and intra-and interday variabilities (precisions) were <7%. The lower limit of quantitation of the assay was 20 ng/ml in plasma and saliva, respectively, and 200 ng/g in tissue.
Concentrations of fluconazole were determined after solid-phase extraction by reversed-phase HPLC (12) and detection at 260 nm. Esophageal tissue was homogenized prior to extraction in ice-cold normal saline (1:2 [wt/wt]), and drug concentrations were calculated for 1 g of tissue. External standards and quality controls were similarly prepared by spiking pooled normal rabbit serum, saliva, and esophageal homogenate with fluconazole. Eight-point standard curves were linear, with R2 values of ≥0.997. Accuracies were within ±7%, and intra- and interday variabilities (precisions) were <8%. The lower limit of quantitation of the assay was 0.5 μg/ml in plasma and saliva, respectively, and 1.5 μg/g in tissue.
Concentrations of amphotericin B were determined after liquid extraction by reversed-phase HPLC (3) and detection at 382 nm. Esophageal tissue was homogenized and extracted in ice-cold methanol (1:3 [wt/wt]), and drug concentrations were calculated for 1 g of tissue. External standards and quality controls were similarly prepared by spiking pooled normal rabbit serum, saliva, and esophageal homogenate with amphotericin B. Eight-point standard curves were linear, with R2 values of ≥0.999. Accuracies were within ±6.5%, and intra- and interday variabilities (precisions) were <12%. The lower limit of quantitation of the assay was 40 ng/ml in plasma and saliva, respectively, and 160 ng/g in tissue.
The relationships between concentration data and the residual fungal burden in tissue were determined by pharmacodynamic modeling. Experimental concentration-effect data were fit to an inhibitory effect sigmoidal maximum effect (Emax) model by iterative uniformly weighted nonlinear least-squares regression with the WinNonlin computer program (Scientific Consulting, Lexington, Ky.).
Histopathology.
Representative sections of the tongue were prepared for histological studies. Tissue specimens were excised and fixed in 10% neutral buffered formalin, embedded in paraffin, sectioned, and then stained with periodic acid-Schiff and Gomori methenamine silver stains.
Toxicity studies.
Chemical determinations of serum potassium, aspartyl aminotransaminase, alanine aminotransaminase, serum creatinine, alkaline phosphatase, and total bilirubin levels were performed with the penultimate sample drawn from each rabbit.
Statistical analysis.
Comparisons between groups were performed by the Kruskal-Wallis nonparametric analysis of variance test with Dunn's correction for multiple comparisons. All P values were two sided, and a P value of <0.05 was considered significant.
RESULTS
Time-kill assay.
Time-kill curves demonstrated the concentration-dependent fungicidal activities of VER-002 and amphotericin B (Fig. 1). At 4 to 6 h, ≥99.9% killing was achieved with VER-002 and amphotericin B at 0.1 and 0.25 μg/ml. The killing was sustained, and regrowth was not seen at 24 h at VER-002 concentrations of 0.1 and 0.25 μg/ml.
FIG. 1.
Time-kill assay of VER-002 and amphotericin B against C. albicans in antibiotic medium 3. Concentrations of VER-002 (VER) and amphotericin B (AmB) at 0.01, 0.1, and 0.25 μg/ml were studied in relation to a growth control. Data plotted are the mean ± SEM from three separate experiments for each growth curve, including the control and amphotericin B at 0.25 μg/ml. As the SEM was small for several time points, the error bars may not always be apparent in the time-kill curves.
Antifungal therapy.
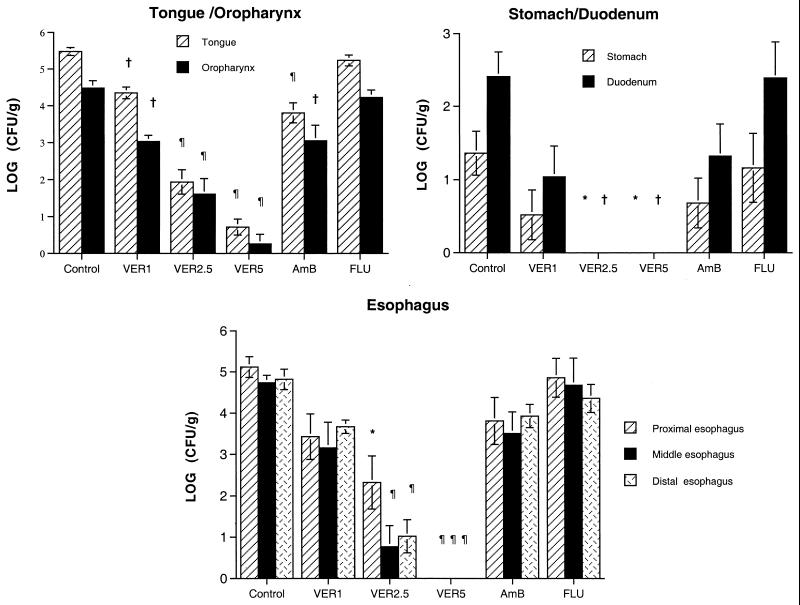
VER-002 showed a significant dosage-dependent antifungal effect as treatment for fluconazole-resistant OPEC. Rabbits treated with VER1, VER2.5, and VER5 showed significant dosage-dependent organism clearance from the tongue in comparison to the organism clearance for the untreated controls (P < 0.01) (Fig. 2). There was also a significant reduction of C. albicans the tongues of rabbits treated with amphotericin B in comparison to that for untreated controls (P < 0.001), while rabbits treated with fluconazole showed no reduction in the number of CFU per gram in this tissue.
FIG. 2.
Response of experimental OPEC caused by fluconazole-resistant C. albicans in immunocompromised rabbits to antifungal therapy measured by mean log (CFU per gram) concentration of organism in the tongue, oropharynx, esophagus, stomach, and duodenum in untreated controls (control) (n = 18), rabbits treated with VER1 (n = 9), VER2.5 (n = 9), and VER5 (n = 9), rabbits treated with amphotericin B at 0.3 mg/kg/day (AmB) (n = 9), and rabbits treated with fluconazole at 1 mg/kg/day (FLU) (n = 9). Values are given as mean ± SEM. ∗, P < 0.05; †, P < 0.01; and ¶, P < 0.001, in comparison to untreated controls by the Kruskal-Wallis nonparametric analysis of variance test with Dunn's correction for multiple comparisons.
There was a significant reduction in C. albicans growth in the oropharynx from rabbits treated with VER1, VER2.5, and VER5 in comparison to the reductions for untreated controls and fluconazole-treated rabbits (P < 0.01) (Fig. 2). Rabbits treated with VER2.5 and VER5 mg/kg had significantly lower organism burdens in the oropharynx than rabbits treated with amphotericin B (P < 0.01).
Rabbits treated with VER-002 also showed a significant reduction of the concentration of C. albicans in the esophagus. There was complete clearance of organisms from all three segments of the esophagus in rabbits treated with VER5 (Fig. 2). In comparison to untreated controls, rabbits treated with VER2.5 showed a significant reduction in organism growth from the proximal middle (P < 0.001) and distal (P < 0.001) segments of the esophagus. There was no significant reduction in tissue burden in the esophageal tissue of rabbits treated with VER1, amphotericin B, and fluconazole in comparison to that for untreated control animals.
There also was significant organism clearance from the stomach and duodenum in rabbits treated with VER-002. Rabbits treated with VER2.5 and VER5 had no detectable C. albicans in the stomach or duodenum (Fig. 2). Rabbits receiving VER1 showed significant reductions in organism growth from the duodenum in comparison to that for untreated controls (P < 0.05), while rabbits treated with amphotericin B and fluconazole showed no reductions.
Concentrations of antifungal compounds in plasma, saliva, and esophageal tissue.
The concentrations of VER-002, fluconazole, and amphotericin B in esophageal tissue, saliva, and plasma at steady-state are depicted in Table 2. VER-002 exhibited dosage-proportional increases in concentrations at all three sites. Mean trough concentrations in esophageal tissue ranged from 0.564 to 2.402 μg/g and exceeded the MICs for the infecting isolates by more than 30-fold. Potentially therapeutic VER-002 concentrations, which were greater than or equal to the MICs, were also found in saliva. The mean levels in plasma 2 h after dosing ranged from 1.653 to 7.426 μg/ml. Fluconazole achieved concentrations in esophageal tissue and plasma that were subtherapeutic (below the MICs). The concentrations of amphotericin B in esophageal tissue were below the MICs for the C. albicans isolates. In comparison to VER-002, both fluconazole and amphotericin B were undetectable in saliva.
TABLE 2.
Concentrations of VER-002, fluconazole, and amphotericin B in esophageal tissue, saliva, and plasma after repeat dosing over 7 days
| Treatment groupa | Concn (μg/g or μg/ml)b
|
||
|---|---|---|---|
| Esophagus | Saliva | Plasma | |
| VER1 | 0.564 ± 0.149 | 0.017 ± 0.007 | 1.653 ± 0.365 |
| VER2.5 | 1.276 ± 0.273 | 0.024 ± 0.011 | 3.623 ± 0.882 |
| VER5 | 2.402 ± 0.401 | 0.046 ± 0.022 | 7.426 ± 1.616 |
| Fluconazole | 3.577 ± 0.787 | Not detectable | 1.357 ± 0.129 |
| Amphotericin B | 0.020 ± 0.007 | Not detectable | 0.995 ± 0.056 |
There were nine animals in each group.
All values are expressed as means ± SEMs. P was <0.001 by analysis of variance across the three VER-002 dosage groups. The lower limits of quantitation in plasma and saliva were 0.020 μg/ml for VER-002, 0.5 μg/ml for fluconazole, and 0.040 μg/ml for deoxycholate amphotericin B; the lower of limits of quantitation in tissues were 0.200 for VER-002, 1.5 μg/g for fluconazole, and 0.160 μg/g for amphotericin B.
Concentration-effect relationships.
The pharmacodynamic relationships between the concentrations of VER-002 versus the residual fungal burden in tissue are depicted in Table 3 and Fig. 3. The pharmacodynamic model was consistent with the linear pharmacokinetic and the linear dosage-response relationship of VER-002 in the infection model. The mean coefficient of determination between observed and estimated values (r) was 0.884 (range, 0.8397 to 0.9302) (Table 3) for the relationships between drug concentrations in plasma, saliva, and esophageal tissue versus residual fungal burden in tissue in the oropharynx and esophagus. Relationships between concentrations in plasma and saliva and residual fungal burden in stomach and duodenum were not as strong (r = 0.5371 to 0.7324; data not shown).
TABLE 3.
Pharmacodynamic relationships between concentrations of VER-002 versus residual fungal burden and goodness of fit of the model equationa
| Pharmacodynamic relationship | Emax [log (CFU/g)] | EC50 (μg/ml or μg/g)b | Gamma | Correlation (C) between observed and predicted values |
|---|---|---|---|---|
| C in plasma/log (CFU/g) for oropharynx | 5.22 ± 0.17 | 2.69 ± 0.22 | 2.11 ± 0.33 | 0.9297 |
| C in plasma/log (CFU/g) for esophagus | 4.87 ± 0.24 | 2.20 ± 0.28 | 2.08 ± 0.52 | 0.8781 |
| C in esophagus/log (CFU/g) for esophagus | 4.84 ± 0.19 | 0.81 ± 0.07 | 2.80 ± 0.55 | 0.9302 |
| C in saliva/log (CFU/g) for oropharynx | 5.20 ± 0.25 | 0.01 ± 0.00 | 1.62 ± 0.44 | 0.8464 |
| C in saliva/log (CFU/g) for esophagus | 4.89 ± 0.28 | 0.01 ± 0.00 | 1.83 ± 0.60 | 0.8397 |
All values are expressed as the mean ± standard deviation. The equation for the model follows the Emax function, where E = Emax · {(1) − [CGamma/(CGamma + EC50Gamma)]}, where E is the effect, Emax is the maximum effect, C is the concentration, EC50y is the 50% effective concentration, and Gamma is the slope in the central part of the curve.
Units are micrograms per milliliter for plasma and saliva; units are micrograms per gram for esophageal tissue.
FIG. 3.
Concentration-effect relationship for VER-002 in the treatment of experimental OPEC. The relationship follows an inhibitory effect sigmoidal Emax model with no effect at a concentration of 0 and complete inhibition at a concentration extrapolated to infinity. Depicted are the fitted curve and the scatter of the observed values: (A) Residual fungal burden in esophageal tissue versus near-peak concentrations of VER-002 in plasma; (B) residual fungal burden in esophageal tissue versus concentrations of VER-002 in esophageal tissue 24 h after dosing. The coefficients of determination between observed and predicted data (r) were 0.8781 and 0.9302, respectively. Note the steep negative slope over a small concentration range, indicating the presence of a narrow threshold concentration for antifungal efficacy.
Histopathology.
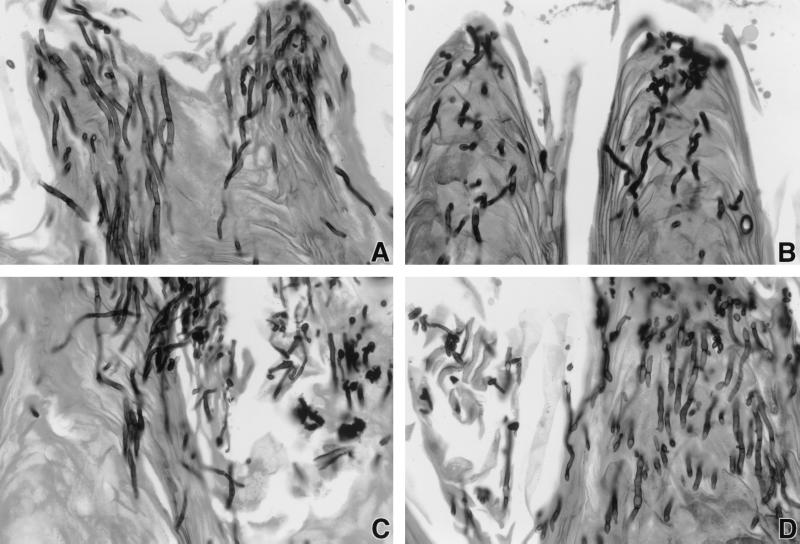
These histologic studies demonstrated a clear dose-dependent correlation between microscopic reduction of organisms in tissues and quantitative reduction of fungal burden in tissue (log CFU per gram). There was complete histologic eradication of the organisms with VER5. As shown in Fig. 4, there also were dose-dependent morphological changes from hyphae and pseudohyphae in untreated controls (Fig. 4A) to an appearance of yeast-like structures in VER2.5-treated rabbits (Fig. 4B). Amphotericin B- and fluconazole-treated rabbits had no significant changes (Fig. 4C and D) in cell wall morphology.
FIG. 4.
Antifungal effect on microscopic morphology of the cell structure of fluconazole-resistant C. albicans in glossal tissue of rabbits treated with VER2.5, amphotericin B at 0.3 mg/kg/day, and fluconazole at 2 mg/kg/day. (A) Untreated controls; (B) VER2.5; (C) amphotericin B; (D) fluconazole. Gomori methenamine silver stain magnification, ×428 was used. (original magnification, ×630). (A and B) Transition from predominantly hyphae and pseudohyphae in untreated controls to predominance of yeast-like structures in rabbits treated with VER2.5. Rabbits treated with amphotericin B and fluconazole had no significant changes (C and D) in cell wall morphology.
Safety.
Rabbits treated with VER-002, fluconazole, and amphotericin B and untreated controls had no detectable increase or decrease in serum creatinine, serum potassium, aspartyl amino transaminase alanine aminotransaminase alkaline phosphatase, or total bilirubin levels (Table 4).
TABLE 4.
Effects of VER-002, amphotericin B, and fluconazole on serum creatinine and potassium levels
| Treatment group | Serum creatinine level (mg/d)a | Serum potassium level (mmol/liter)a |
|---|---|---|
| Control (n = 15) | 0.43 ± 0.04 | 4.60 ± 0.15 |
| VER-002 (n = 27) | 0.47 ± 0.02 | 4.40 ± 0.09 |
| Amphotericin B (n = 9) | 0.50 ± 0.10 | 4.13 ± 0.07 |
| Fluconazole (n = 9) | 0.42 ± 0.04 | 4.20 ± 0.20 |
All values are expressed as means ± SEMs.
Mean serum creatinine and potasium levels for all dosage groups.
DISCUSSION
The semisynthetic echinocandin VER-002 demonstrated significant in vitro and in vivo activity against fluconazole-resistant OPEC due to C. albicans. The in vivo efficacy of VER-002 was dosage dependent and correlated with the in vitro fungicidal concentration-dependent effect of VER-002 against fluconazole-resistant C. albicans. The in vitro concentration-dependent fungicidal effects of VER-002 were similar to the fungicidal concentration-dependent effects of amphotericin B. Fluconazole had no effect in this rabbit model of fluconazole-resistant OPEC.
The in vitro time-kill antifungal activities of VER-002 observed in this study are consistent with those observed by Ernst and colleagues (8) and Klepser and colleagues (14). However, the fungicidal activity of the echinocandin observed in this study was more potent than that observed against most of the isolates studied by those investigators. The use of AM3 in our assays may account for this difference. Emst et al. (8) and Klepser et al. (14) indicate that RPMI 1640 is probably not a representative medium for assessment of the in vitro antifungal activities of echinocandins. On the basis of their subsequent studies, those investigators further recommend that AM3 may be a more suitable medium.
The model of OPEC described here reflects the profound impairment of mucosal immunity encountered in severely immunocompromised hosts, such as HIV-infected patients, corticosteroid-treated patients, and recipients of organ or bone marrow transplants. High-dose corticosteroids induce a profound impairment of mucosal immunity in rabbits (28). Specifically, the mucosa-associated lymphoid tissue in corticosteroid-treated rabbits reveals a profound depletion of lymphoid follicles and severe loss of B lymphocytes. The dome epithelium of mucosa-associated lymphoid tissue is also compromised, as evidenced by apoptosis of M cells and loss of intraepithelial lymphocytes. These immune impairments compromise surface immunoglobulin production and immunoregulation of mucosal host defenses against opportunistic pathogens. Such mucosal immunosuppression results in a burden of C. albicans in tissue that is higher than that in healthy animals. The refractoriness of this infection to fluconazole resembles that of azole-resistant candidiasis in humans (31).
The microbiological outcome variables in this model permit quantitative analysis of dosage-dependent antifungal responses in several sites of the upper alimentary track. Evaluation of different sites in the upper alimentary track is important, as the antifungal effect of different concentrations of antifungal agents in saliva and tissues may be site dependent. For example, one may observe patients with fluconazole-resistant OPEC who achieve a response in the oral cavity but who continue to have persistent esophageal disease.
The echinocandin VER-002 demonstrated concentration-dependent fungicidal activity in time-kill assays. As our preliminary findings demonstrated more potent activities of echinocandin and amphotericin B and a wider distribution of MICs for C. albicans in AM3 than in RPMI 1640, the MIC assays and time-kill assays were performed in AM3 throughout the study. The concentration-dependent effects of the echinocandin were consistent with those observed in tissues of the upper alimentary track. Both echinocandin and amphotericin B demonstrated similar profiles on time-kill curves, reflecting the concentration-dependent fungicidal properties of these compounds. Each concentration (0.01, 0.10, and 0.25 μg/ml) of VER-002 and amphotericin B with antifungal activity demonstrated a similar level of microbicidal activity. The concentration of 0.25 μg of amphotericin B per ml caused a more rapid decline in the number of organisms in the initial part of incubation. However, no significant differences in killing between VER-002 and amphotericin B were detected at 24 h. Although the mechanisms of action of amphotericin B and echinocandins are clearly different, with the former causing direct disruption of cell membrane integrity and the latter causing inhibition of cell wall biosynthesis, both mechanisms lead to a lethal effect against C. albicans. These concentration-dependent fungicidal properties of the echinocandin molecule demonstrated in the in vitro time-kill assays were reflected in the dosage-dependent and concentration-dependent fungicidal activities in the tongue, oropharynx, and esophagus in experimental fluconazole-resistant OPEC.
VER5 was most active in clearing C. albicans from the esophagus, stomach, and duodenum. The effects in the tongue and oropharynx also were greatest for VER5, with which a ≥104-fold reduction in the level of C. albicans was achieved. The different tissue sites (the tongue, oropharynx, and esophagus) appeared to have similar patterns of response to VER-002, amphotericin B, and fluconazole. The gastric and duodenal tissues demonstrated complete clearance of C. albicans with VER2.5 in comparison to the clearance seen for the other tissues, which were cleared with VER5. This greater response may be related to the lower burden of C. albicans at these sites compared to those in the tongue, oropharynx, and esophagus.
As little is known about the pharmacokinetics and pharmacodynamics of amphotericin B, fluconazole, and the echinocandin as treatment for esophageal candidiasis, we investigated the concentrations of these three compounds in plasma, tissue, and saliva. The concentrations of echinocandin demonstrated a dosage-proportional increase in the esophagus, saliva, and plasma. The concentrations in esophageal tissue exceeded the MIC for C. albicans by at least 37-fold when VER1 was used and by 160-fold when VER5 was used. By comparison, the concentrations of the echinocandin in saliva exceeded the MIC for C. albicans by as little as one- to threefold. These data suggest that the relatively large molecule of the echinocandin is not excreted well into saliva, perhaps due to barriers of transport from the capillaries through the basement membrane and into the epithelial cells of the salivary glands. Restrictions to interepithelial penetration by tight junctions may further limit transport of the echinocandin molecule into saliva. By comparison, the echinocandin molecule appears to penetrate relatively well into the capillary bed of esophageal tissue, where the barriers to penetration present in the salivary glands are not applicable. The linear dosage-proportional plasma and tissue pharmacokinetic properties of VER-002 contrast with those of its predecessor, cilofungin (LY121019) (11). The earlier echinocandin demonstrated nonlinear saturation plasma and tissue pharmacokinetics (19). The tissue cilofungin concentrations correlated with the antifungal response in a pattern similar to that observed for VER-002 (33).
Rabbits were treated with amphotericin B (0.3 mg/kg/day) in the well-known low-dose regimen, which has been advocated for management of esophageal candidiasis (22). The dosages of amphotericin B used in rabbits have pharmacokinetic profiles that are similar to those for the dosages used in humans. Comparison of the data for amphotericin B between rabbits and humans for the 1.0-mg/kg dosage level (1, 18) reveals values that are very similar to those for humans for the maximum concentration of drug in plasma (Cmax) and the area under the concentration-time curve from 0 to 24 h (AUC0–24) (4.7 versus 2.9 μg/ml and 31 versus 36 μg · h/ml, respectively), suggesting no fundamental differences in the level of exposure to amphotericin B between rabbits and humans. Further corroboration of these observations is a detailed analysis of conventional amphotericin B and amphotericin B lipid complex in rabbits (32). The pharmacokinetics of amphotericin B in the three-compartment model in rabbits are similar to those in humans across a broad dosage range. Thus, the results obtained for the key pharmacokinetic parameters (Cmax and AUC0–24) indicate that the effect of the dosage of 0.3 mg/kg/day in rabbit is likely to be similar to that in humans.
In previous studies performed in our laboratory, fluconazole at 1 mg/kg twice daily maintained concentrations in plasma above the MICs for fluconazole-sensitive isolates (≤0.125 μg/ml) throughout the dosing interval (31). Rabbits infected with fluconazole-sensitive isolates and treated with fluconazole had significant reductions in the levels of organisms in quantitative cultures of oral mucosal samples (P < 0.001) and burdens of C. albicans in the tongue, soft palate, and esophagus (P < 0.001) (31). As the half-life of fluconazole in the rabbit is shorter (the half-life is approximately 12 h in rabbits) than that in adult humans but similar to that in children (20) and since exposure over time has been shown to be important for antifungal efficacy (21), a twice-daily dosing regimen was used.
The ratio of the concentration in esophageal tissue to that in plasma of 0.02 for amphotericin B is substantially lower than the ratio of 0.3 achieved with echinocandin. Nevertheless, the concentration of amphotericin B in esophageal tissue is approximately four times higher than the MIC for C. albicans. This concentration appears to be sufficiently active to achieve an approximate 10-fold or 1-log reduction of Candida levels in tissue. Higher concentrations of amphotericin B would likely lead to greater fungicidal activity and to more complete clearance from tissue. These findings suggest that low-dose amphotericin B may not achieve complete microbiological eradication or result in a clinical response in profoundly compromised patients with esophageal candidiasis due to the relatively low concentrations of amphotericin B in tissue. To our knowledge, the study described here is the first in vivo investigation of the pharmacokinetics and pharmacodynamics of low-dose amphotericin B for the treatment of esophageal candidiasis. Further studies of the dosage-response relationships of amphotericin B in this model of experimental esophageal candidiasis may help provide an understanding of the correct dosing strategies for immunocompromised patients with esophageal candidiasis.
The clinical and microbiological responses in our immunocompromised patients with fluconazole-resistant OPEC have been variable, prompting us to use higher amphotericin B dosages (0.5 to 0.6 mg/kg/day) (4). This lack of response may reflect profoundly impaired mucosal immunity as well as relatively low levels of amphotericin B in esophageal tissue.
The tissue fluconazole concentration of 3.6 μg/ml and the plasma fluconazole concentration of 1.36 μg/ml are substantially lower than the MICs (>64 mg/ml). These findings further demonstrate a relatively high ratio of the concentration in tissue to that in plasma (approximately 3:1). The excellent response achieved with fluconazole against esophageal candidiasis caused by susceptible organisms may be related to its favorable esophageal penetration (31).
The concentrations of an antimicrobial compound in tissue are the sum of the concentrations in the intravascular, the intestitial, and the intracellular tissue compartments. However, it is not clear which of these compartments is important as a target tissue for invasive fungal infections. The pseudohyphae and hyphae of Candida spp., for example, invade cells, blood vessels, and interstitial compartments of tissues. Furthermore, the pathogenic hallmark of tissue-invasive fungal infections is tissue necrosis and inflammation, in which different equilibria of drug concentrations are likely to prevail, suggesting that sampling of the entire tissue homogenate may be more reflective of the pathogenesis of invasive mycoses.
Our data show a similar concentration-response relationship of the concentrations of VER-002 in tissue and the levels of VER-002 in plasma with the residual fungal burden. This similarity further validates the relationship between plasma pharmacokinetic parameters as a surrogate for the concentrations of echinocandins in the alimentary tract. Whether these pharmacokinetic relationships are valid for other tissue sites such as the central nervous system remain to be explored.
In summary, this study demonstrates that the echinocandin VER-002 exerts concentration-dependent and dosage-dependent fungicidal activity against experimental fluconazole-resistant OPEC. The new group of echinocandins, as exemplified by VER-002, represents a potentially important therapeutic advance in the management of esophageal candidiasis and warrants further investigation in clinical trials of treatments for this infection.
ACKNOWLEDGMENTS
We are grateful to Myrna Candelario and Aida Field-Ridley for assistance in the laboratory animal facility, to Joanne Peter for performing MIC determinations, to Diana Mickiene for performing HPLC, and to Jeniffer Rabb for expert secretarial assistance in preparation of the manuscript.
REFERENCES
- 1.Amantea M A, Bowden R A, Forrest A, Working P K, Newman M S, Mamelok R D. Population pharmacokinetics and renal function-sparing effects of amphotericin B colloidal dispersion in patients receiving bone marrow transplants. Antimicrob Agents Chemother. 1995;39:2042–2047. doi: 10.1128/aac.39.9.2042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Barbaro G, Barbarini G, Calderon W, Grisorio B, Alcini P, Di Lorenzo G. Fluconazole versus itraconazole for Candida esophagitis in acquired immunodeficiency syndrome. Gastroenterology. 1996;111:1169–1177. doi: 10.1053/gast.1996.v111.pm8898629. [DOI] [PubMed] [Google Scholar]
- 3.Brassinne C, Laduron C, Coune A, Sculier J P, Hollaert C, Collette N, Meunier F. High-performance liquid chromatographic determination of amphotericin B in human serum. J Chromatogr. 1987;7:401–407. doi: 10.1016/0378-4347(87)80307-9. [DOI] [PubMed] [Google Scholar]
- 4.Chiou C C, Groll A H, Gonzalez C E, Callender D, Venzon D, Pizzo P A, Wood L, Walsh T J. Esophageal candidiasis in pediatric acquired immunodeficiency syndrome: clinical manifestations and risk factors. Pediatr Infect Dis J. 2000;19:729–734. doi: 10.1097/00006454-200008000-00011. [DOI] [PubMed] [Google Scholar]
- 5.Committee on the Care and Use of Laboratory Animals of the Institute of Laboratory Animal Resources, Commission on Life Sciences, National Research Council. Guide for the care and use of laboratory animals. Washington, D.C.: National Academy Press; 1996. [Google Scholar]
- 6.Debono M, Gordee R S. Antibiotics that inhibit fungal cell wall development. Annu Rev Microbiol. 1994;48:471–497. doi: 10.1146/annurev.mi.48.100194.002351. [DOI] [PubMed] [Google Scholar]
- 7.Debono M, Turner W W, LaGrandeur L, Burkhardt F J, Nissen J S, Nichols K K, Rodriguez M J, Zweifel M J, Zeckner D J, Gordee R S, Tang J, Parr T R., Jr Semisynthetic chemical modification of the antifungal lipopeptide echinocandin B (ECB): structure-activity studies of the lipophilic and geometric parameters of polyarylated acyl analogs of ECB. J Med Chem. 1995;38:3271–3281. doi: 10.1021/jm00017a012. [DOI] [PubMed] [Google Scholar]
- 8.Ernst M E, Klepser M E, Wolfe E J, Pfaller A. Antifungal dynamics of LY303366, an investigational echinocandin B analog, against Candida spp. Diagn Microbiol Infect Dis. 1996;26:125–131. doi: 10.1016/s0732-8893(96)00202-7. [DOI] [PubMed] [Google Scholar]
- 9.Fromtling R A. LY303366. Drugs Future. 1994;19:338–342. [Google Scholar]
- 10.Georgopapadakou N H, Tkacz J S. The fungal cell wall as a drug target. Trends Microbiol. 1995;3:98–104. doi: 10.1016/s0966-842x(00)88890-3. [DOI] [PubMed] [Google Scholar]
- 11.Gordee R S, Zeckner D J, Howard L C, Alborn W E, DeBono M. Anti-Candida activity and toxicology of LY121019, a novel semisynthetic polypeptide antifungal antibiotic. Ann N Y Acad Sci. 1988;544:294–309. doi: 10.1111/j.1749-6632.1988.tb40415.x. [DOI] [PubMed] [Google Scholar]
- 12.Inagaki K, Takagi J, Lor E, Okamoto M P, Gill M A. Determination of fluconazole in human serum by solid-phase extraction and reversed-phase high-performance liquid chromatography. Ther Drug Monit. 1992;14:306–311. doi: 10.1097/00007691-199208000-00008. [DOI] [PubMed] [Google Scholar]
- 13.Karlowsky J A, Harding G A J, Zelenitsky S A, Hoban D J, Kabani A, Balko T V, Turik M, Zhanel G G. In vitro kill curves of a new semisynthetic echinocandin, LY-303366, against fluconazole-sensitive and -resistant Candida species. Antimicrob Agents Chemother. 1997;41:2576–2578. doi: 10.1128/aac.41.11.2576. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Klepser M E, Ernst E J, Ernst M E, Messer S A, Pfaller M A. Evaluation of endpoints for antifungal susceptibility determinations with LY303366. Antimicrob Agents Chemother. 1998;42:1387–1391. doi: 10.1128/aac.42.6.1387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Krishnarao T V, Galgiani N J. Comparison of the in vitro activities of the echinocandin LY303366, the pneumocandin MK-0991, and fluconazole against Candida species and Cryptococcus neoformans. Antimicrob Agents Chemother. 1997;41:1957–1960. doi: 10.1128/aac.41.9.1957. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kurtz M B. New antifungal drug targets: a vision for future. ASM News. 1997;64:31–39. [Google Scholar]
- 17.Kurtz M B, Douglas C M. Lipopeptide inhibitors of fungal glucan synthase. J Med Vet Mycol. 1997;35:79–86. doi: 10.1080/02681219780000961. [DOI] [PubMed] [Google Scholar]
- 18.Lee J W, Amantea M A, Francis P A, Navarro E E, Bacher J, Pizzo P A, Walsh T J. Pharmacokinetics and safety of a unilamellar liposomal formulation of amphotericin B (AmBisome) in rabbits. Antimicrob Agents Chemother. 1994;38:713–718. doi: 10.1128/aac.38.4.713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Lee J W, Kelly P, Lecciones J, Coleman D, Gordee R, Pizzo P A, Walsh T J. Cilofungin ( LY121019) shows non-linear plasma pharmacokinetics and tissue penetration in rabbits. Antimicrob Agents Chemother. 1990;34:2240–2245. doi: 10.1128/aac.34.11.2240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Lee J W, Seibel N T, Amantea M A, Whitcomb P, Pizzo P A, Walsh T J. Safety, tolerance, and pharmacokinetics of fluconazole in children with neoplastic diseases. J Pediatr. 1992;120:987–993. doi: 10.1016/s0022-3476(05)81975-4. [DOI] [PubMed] [Google Scholar]
- 21.Louie A, Banerjee P, Drusano G L, Shayegani M, Miller M H. Interaction between fluconazole and amphotericin B in mice with systemic infection due to fluconazole-susceptible or -resistant strains of Candida albicans. Antimicrob Agents Chemother. 1999;43:2841–2847. doi: 10.1128/aac.43.12.2841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Medoff G, Dismukes W E, Meade R H, Moses J M. A new therapeutic approach to Candida infections. A preliminary report. Arch Intern Med. 1972;130:241–245. [PubMed] [Google Scholar]
- 23.National Committee for Clinical Laboratory Standards. Reference method for broth dilution antifungal susceptibility testing of yeasts. Tentative standard. NCCLS document M27-A. Wayne, Pa: National Committee for Clinical Laboratory Standards; 1997. [Google Scholar]
- 24.Petraitiene R, Petraitis V, Groll A H, Candelario M, Sein T, Bell A, Lyman C A, McMillian C L, Bacher J, Walsh T J. Antifungal activity of LY303366, a novel echinocandin B, in experimental disseminated candidiasis in rabbits. Antimicrob Agents Chemother. 1999;43:2148–2155. doi: 10.1128/aac.43.9.2148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Petraitis V, Petraitiene R, Groll A H, Bell A, Callender D P, Sein T, Schaufele R L, McMillian C L, Bacher J, Walsh T J. Antifungal efficacy, safety, and single-dose pharmacokinetics of LY303366, a novel echinocandin B, in experimental pulmonary aspergillosis in persistently neutropenic rabbits. Antimicrob Agents Chemother. 1998;42:2898–2905. doi: 10.1128/aac.42.11.2898. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Pfaller M A, Messer S A, Coffman S. In vitro susceptibilities of clinical yeast isolates to a new echinocandin derivative, LY303366, and other antifungal agents. Antimicrob Agents Chemother. 1997;41:763–766. doi: 10.1128/aac.41.4.763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Rex J H, Rinaldi M G, Pfaller M A. Resistance of Candida species to fluconazole. Antimicrob Agents Chemother. 1995;39:1–8. doi: 10.1128/aac.39.1.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Roy M J, Walsh T J. Histopathological and immunohistochemical changes in gut-associated lymphoid tissues following treatment of rabbits with dexamethasone. Lab Investig. 1992;64:437–443. [PubMed] [Google Scholar]
- 29.Uzun O, Kocagoz S, Cetinkaya Y, Arikan S, Unal S. In vitro activity of a new echinocandin, LY303366, compared with those of amphotericin B and fluconazole against clinical yeast isolates. Antimicrob Agents Chemother. 1997;41:1156–1157. doi: 10.1128/aac.41.5.1156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Walsh T J, Bacher J, Pizzo P A. Chronic silastic central venous catheterization for induction, maintenance, and support of persistent granulocytopenia in rabbits. Lab Anim Med. 1988;38:467–470. [PubMed] [Google Scholar]
- 31.Walsh T J, Gonzalez C E, Piscitelli S, Bacher J D, Peter J, Torres R, Shetti D, Katsov V, Kligys K, Lyman C A. Correlation between in vitro and in vivo antifungal activity in experimental fluconazole-resistant oropharyngeal and esophageal candidiasis. J Clin Microbiol. 2000;38:2369–2373. doi: 10.1128/jcm.38.6.2369-2373.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Walsh T J, Jackson A J, Lee J W, Amantea M, Sein T, Bacher J, Zech L. Dose-dependent pharmacokinetics of amphotericin B lipid complex in rabbits. Antimicrob Agents Chemother. 2000;44:2068–2076. doi: 10.1128/aac.44.8.2068-2076.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Walsh T J, Lee J W, Kelly P, Bacher J, Lecclones J, Thomas V, Lyman C, Coleman D, Gordee R, Pizzo P A. The antifungal effects of the non-linear pharmacokinetics of cilofungin, a 1,3-β-glucan synthase inhibitor, during continuous vs. intermittent infusion of cilofungin in treatment of experimental disseminated candidiasis. Antimicrob Agents Chemother. 1991;35:1321–1328. doi: 10.1128/aac.35.7.1321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Walsh T J, McEntee C, Dixon D M. Tissue homogenization with sterile reinforced polyethylene bags for quantitative culture of Candida albicans. J Clin Microbiol. 1987;25:931–932. doi: 10.1128/jcm.25.5.931-932.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Wilcox C M, Straub R F, Alexander L N, Clark W S. Etiology of esophageal disease in human immunodeficiency virus-infected patients who fail antifungal therapy. Am J Med. 1996;101:599–604. doi: 10.1016/s0002-9343(96)00303-8. [DOI] [PubMed] [Google Scholar]