Abstract
The mammalian hematopoietic system is remarkably efficient in meeting an organism’s vital needs, yet is highly sensitive and exquisitely regulated. Much of the organismal control over hematopoiesis comes from the regulation of hematopoietic stem cells (HSCs) by specific microenvironments called niches in bone marrow (BM), where HSCs reside. The experimental studies of the last two decades using the most sophisticated and advanced techniques have provided important data on the identity of the niche cells controlling HSCs functions and some mechanisms underlying niche-HSC interactions. In this review we discuss various aspects of organization and functioning of the HSC cell niche in bone marrow. In particular, we review the anatomy of BM niches, various cell types composing the niche, niches for more differentiated cells, metabolism of HSCs in relation to the niche, niche aging, leukemic transformation of the niche, and the current state of HSC niche modeling in vitro.
Keywords: hematopoiesis, hematopoietic stem cells, niches, bone marrow, hypoxia, mesenchymal stromal cells, metabolism, aging, leukemogenesis, 3D modeling
1. Introduction
The hematopoietic system, with its ability to produce an estimated half a trillion new cells per day in humans [1], is arguably the most “prolific” system in a body. Yet this formidable number is generated thanks to only a wmqll tiny quantity—about one million [2], extremely rare HSCs residing in BM [3]. HSCs produce all mature blood and immune cells of the body, with the exception of a few special cell subpopulations such as tissue-resident macrophages and innate-like B and T lymphocytes [4,5,6].
The hematopoietic system, in addition to its utmost efficiency in mature cell production, is also highly adaptable yet very tightly regulated. HSCs are extremely quiescent cells that enter cell cycle quite rarely [7,8]. While in the state of quiescence, HSCs, however, remain on high alert, and in situations of dire organismal need such as major blood loss or microbial invasion, may enter the cell cycle to replenish exhausted progenitors and boost production of effector cells [9,10].
Due to quiescence of the majority of HSCs, homeostatic hematopoiesis is mostly performed through expansion of more differentiated progeny cells that are biased or committed to certain hematopoietic lineages [11,12]. As these cells have limited self-renewal ability and are not long-lived [13], the normal hematopoiesis appears as a succession of a large number of different clones [14]. HSCs themselves have an extensive self-renewal potential that is substantially higher than that of the immediate progeny but is not limitless as it declines with hematological stresses or age [15,16].
The purest and seemingly homogeneous HSC samples that can be obtained by cell sorting are nevertheless fairly heterogeneous, as evidenced by recent results employing lineage tracing and single cell RNA sequencing approaches. These sophisticated techniques revealed the existence in vivo of HSCs biased towards certain lineages, as well as non-classical differentiation routes bypassing multipotent progenitors and directly generating lineage-restricted progenitors [16,17]. In particular, these results indicate the existence of megakaryocyte-biased HSCs and the megakaryocyte differentiation pathway bypassing the stage of common myeloid precursors [18,19]. Thus, although the view of hematopoiesis as a hierarchically organized ensemble of developmentally related cell populations remains valid, the current models of hematopoiesis [20] allow for a substantially higher flexibility in cell fate decisions, as was previously considered possible [21].
2. Anatomy of the BM Niche
In 1978, Schofield, analyzing outcomes of hematopoietic transplantation experiments, proposed the concept of niche as a defined anatomical location that is required for HSCs in vivo to exist and fulfill their functions [22]. He further postulated that the niche has an instructive role in vital decisions of HCSs and their self-renewal is only possible within the niche, whereas HSCs leaving the niche embark on their differentiation journey. The general validity of Schofield’s concept was later confirmed first in Drosophila by identification of stem cell niches in gonads [23], and later in other cell systems.
HSCs during the adult mammalian life reside in BM, which thus serves as a macro-niche for HSCs. Bone cavities contain trabecula, a type of spongy bone tissue that is actively remodeled and in long bones is located in the epiphysis and metaphysis. The BM itself is located between the trabeculae and consists of a loose stroma permeated with vessels, stromal cells and various hematopoietic cells. The periosteal and feeding arteries supplying the BM pass into the bone marrow cavity of the long bones through the nutrient channel, giving rise to smaller arterioles [24,25]. The arterioles then connect to the vascular sinusoids, peculiar fenestrated capillaries with a wide lumen, which in turn connect to a longitudinal central venous sinus flowing into veins exiting through the nutrient channel. The arterioles are in close contact with sympathetic nerve fibers [26] and are also covered by perivascular mesenchymal cells and non-myelinating Schwann cells.
Although BM can be considered a macro-niche for HSCs in mammalians, this notion is lacking the necessary cellular detalization, and the need for detailed characterization of cellular and molecular mechanisms controlling HSC function spawned a race for identification of bona fide HSC niches in BM. Osteoblast located in the endosteal region were the first proposed candidate for the cellular component of the HSC niche as reported by two groups in 2003 [27,28]. In the study published next year, selective elimination of osteoblasts resulted in the decrease of HSC numbers with simultaneous activation of extramedullary hematopoiesis in the spleen and liver [29]. These works were, however, indirect and did not provide solid proof of physical contacts or close association between osteoblasts and HSCs. Subsequently, the attention of research teams was attracted to other cellular components of BM as potential components of the niche.
Major technical improvements, in particular identification of SLAM receptors as appropriate HSCs markers [30], development of mouse strains with HSCs or potential niche components marked by fluorescent proteins, and intravital microscopy—significantly advanced understanding of HSC niches in BM. However, despite intensive ongoing efforts, the nature of the hematopoietic niche remains controversial.
HSCs in the BM were shown to be located in hypoxia regions, with the lowest O2 levels in deeper peri-sinusoidal regions, while the endosteal regions are less hypoxic [31]. Some studies indicate that HSCs are located proximal to arterioles in the endosteal area [32,33], whereas other reports demonstrate that HSCs are located in the vicinity of sinusoids [34]. Yet another work demonstrated the most quiescent long-term repopulating subset of HSCs to reside close to both sinusoidal blood vessels and the endosteal surface [35]. A careful study revealed later that the frequency of apparent association HSCs with candidate niches correlated with the abundance of those niches in the BM. Importantly, the distribution of HSCs relative to these niches was not different from distribution of computationally generated dots randomly placed throughout the BM volume [36]. This work demonstrates therefore the absence of a preferential association of adult HSCs with anatomical locations in BM proposed as specific HSC niches. These unexpected findings suggest that misinterpretation of data on HSC-niche co-localization might be a source of discrepancies between the results obtained by different groups and demonstrate that appropriate controls are of primary importance for correct analysis of such extremely complex biological processes as hematopoiesis.
The discordant concepts placing HSC locations near arterioles or sinusoids may possibly be reconciled by studies suggesting that myeloid-biased HSCs may be localized near megakaryocytes in the vicinity of sinusoids, whereas lymphoid-biased HSCs seem to be located near arterioles [37,38]. If this concept is correct, it still remains to be elucidated whether the unbiased HSCs have a preference to specific anatomical locations in BM. It is also unclear whether the already pre-formed lineage-biased HSCs prefer the above-mentioned alternative locations, or if, vice versa, these locations induce lineage preference in the initially unbiased HSCs.
In general, the above studies demonstrate that there are a large number of functionally and anatomically similar locations in BM that HSCs may associate with, which implies that HSC niches are abundant and not a limiting factor in vivo. In accordance with this, transplantation of large quantities of HSCs into non-conditional hosts leads to their long-term engraftment while not replacing the recipient’s HSCs [39].
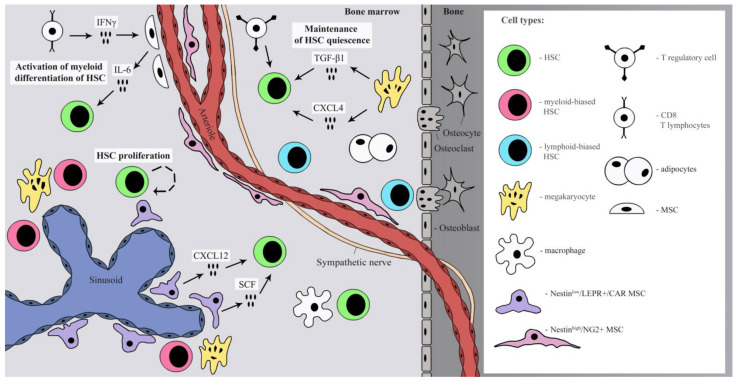
A general overview of BM niche anatomy is provided in Figure 1.
Figure 1.
Cellular and selected molecular components of the HSC niche in BM. The reader is referred to the Section 3 for more detailed information on properties and roles of specific niche cell types.
3. Cell Components of the Niche
3.1. Mesenchymal Stem/Stromal Cells
Mesenchymal stem/stromal cells (MSCs) are a group of cells with multipotent capabilities which play an important role in tissue repair. These cells are characterized by a set of positive (CD73, CD90, CD105) and negative (CD14, CD19, CD34, CD45, HLA-DR) markers, the ability to differentiate into other cell types, such as osteoblasts, chondrocytes, adipocytes, and the secretion of various growth factors, cytokines and chemokines [40,41]. Substances secreted by MSCs play a key role in immunomodulation, as well as cell migration, proliferation, and differentiation. MSCs in BM are a rare population of cells (~0.001–0.01% of the total number of nuclear cells in BM aspirates), located perivascularly, directly on blood vessel surfaces along with sympathetic nerves [42], and play a prominent role in HSCs support [43,44,45]. Using different marker characteristics of MSCs (nestin (Nes), SCF (Stem Cell Factor), CXCL12, NG2, leptin receptor (LEPR)) and genetically modified mouse models, it was possible to distinguish different MSC subpopulations [27,46,47]. According to Kunisaki et al., MSCs in the periarterial and perisinusoidal niches differ in phenotypes and transcriptional profiles: Nes-GFPbright and NG2+ MSCs were observed in the periarterial niche, while Nes-GFPdim cells, LEPR+ and CXCL12high MSCs are located in the perisinusoidal niche [32].
The regulatory role of BM MSCs towards HSCs was demonstrated by experiments with genetically modified mice. In particular, deletion of CXCL12 or SCF from all MSCs led to the depletion of the HSC population [28]. The MSCs populations in the periarterial and perisinusoidal niches have different effects on the preservation of HSC functionality: deletion of CXCL12 in the endosteal/periarterial niche NG2+ MSCs negatively affects the HSC population and leads to a redistribution of the remaining HSCs in BM away from arterioles, while deletion of CXCL12 in sinusoidal LEPR+ cells have no effect on HSC population. Results of Asada et al., however, indicate the importance of LEPR+ perisinusoidal cells rather than periarteriolar niche NG2+ cells as the main source of SCF needed to maintain HSC in the bone marrow [33]. The role of periarteriolar stromal cells in the resting state of HSCs was confirmed by a significant change in the distance between HSCs and arterioles after recovery from myelosuppression, polyinosin:polycytidylic acid (pIpC) treatment, or in Pml knockout mice, all of which led to excessive proliferation of HSCs, HSCs population exhaustion, and migration from arterioles [32].
It is interesting to note that the widely represented in the perisinusoidal niche MSC population with high expression of CXCL12, SCF, and LEPR (CAR cells) has a high expression of adipogenic and osteogenic factors, such as PPARy, Runx, and the ability to differentiate into osteoblasts and adipocytes [48,49]. Short-term ablation of CAR cells in mice led to a decrease in the HSC population in the niche [50], which was associated with reduced production of CXCL12 and SCF.
Nes+ MSCs are in close proximity to Schwann cells and the sympathetic nervous system involved in mobilization of HSCs. Removal of MSCs caused depletion of the HSC population in BM and increased their numbers in the spleen [45,51]. Co-cultivation of MSCs with HSCs increased survival and expansion of the latter [52].
MSCs secrete CXCL12 and a number of other factors such as SCF, VCAM-1, Angiopoietin-1, IL-6, IL-11, TPO (thrombopoietin), Flt-3 ligand, G-CSF, GM-CSF, M-CSF, and BMP4, which affect the HSC population and hematopoiesis in general [46,53,54]. The importance of SCF from MSC was confirmed by experiments on LepR-Cre mice [46]. The interaction of HSC and MSC is very multifaceted and is associated with the transmission of Wnt and Notch signals, the balance between which ensures self-renewal and maintenance of HSC. A member of another signaling pathway, BMP4, affects HSC during embryonic development, whereas high concentrations of this protein support the proliferation of HSCs from umbilical cord blood [55,56]. Conditional inactivation of the BMP4 receptor leads to an increase in HSC population in BM [27]. BMP4 can act directly on HSCs or through mediators like Shh (Sonic hedgehog). Shh in turn induces cytokine-dependent proliferation of HSCs [57]. Nes+ MSCs are involved in niche regulation via the beta-adrenergic receptor, which binds norepinephrine or adrenaline and affects physiological traffic of HSCs by modulating CXCL12 and SCF levels [58]. Of note, human CD45−CD105+CD146+ MSCs were able to create ectopic BM niche after subcutaneous or subrenal transplantation [59,60].
3.2. Endothelium
The endothelium in the niche is in direct contact with HSCs [30,47] and is represented by arteriolar and sinusoidal endothelial cells (ECs) [61]. These two cell groups differ in their functions: both groups produce SCF necessary for the survival of HSCs, although the sinusoidal ECs (SECs) synthesize only a small part of this factor [47,62]. At the same time, SECs produce large quantities of CXCL12 and E-selectin [63]. Arteriolar ECs (AECs), together with reticular cells, synthesize the extracellular matrix protein Del-1, which, through interactions with β3 integrin on HSCs, stimulates their proliferation and expansion, as well as differentiation towards myeloid lineage [64,65].
HSC self-renewal is stimulated by Notch ligands, which are synthesized by ECs [66]. In particular, Jagged-1 is an EC-produced Notch ligand that contributes to the maintenance of HSCs, since conditional deletion of Jagged-1 in ECs results in exhaustion of the HSC pool and severe decline of hematopoiesis [67]. Moreover, Jagged-1 participates in regeneration of hematopoiesis following myeloablation [66].
It should be noted that SECs are required for regeneratory hematopoiesis since lethal irradiation of BM produces their severe degeneration, while the regeneration of SECs is dependent on VEGFR2. Blocking SECs regeneration by neutralizing VE-cadherin or VEGFR2 antibodies prevents hematopoietic reconstitution as well [66,68]. A small subpopulation of Apln+ ECs also plays an important role in restoring the integrity of the niche [69]. The absence of these cells or deletion of the SCF and Vegfr2 genes disturbs vascular regeneration and maintenance of the HSC population following BM irradiation and HSC transplantation. This cell subset thus plays important role in the restoration of the vascular network [69].
While regeneration of SECs is critical for the restoration of HSC functions, SCF secreted by AECs also contributes to the regeneration of the HSC pool after irradiation [62,68]. EGF and some other factors secreted by Tie2+ ECs increase the survival and recovery of the HSC population after irradiation [70]. The importance of ECs is also demonstrated by the finding that erythromyeloid precursors can differentiate, if required, into ECs necessary for the restoration of the blood vessel network [71]. Co-cultivation of ECs with CD34+ hematopoietic progenitors contribute to an expansion of the latter [72]. Co-transplantation of endothelial progenitor cells with HSCs promotes endothelial cell recovery, as well as hematological and immune reconstitution [73].
Importantly, the Klf6 transcription factor expressed in ECs was shown to modulate HSC lodgment and expansion of HSCs in zebrafish via chemokine Ccl25b. Moreover, its mammalian ortholog Ccl21 was able to expand hematopoietic progenitors in the ex vivo system [74]. Of note, the loss of the vascular-endothelial-expressed Notch ligand DLL4 distorts bone marrow hematopoiesis towards significant transcriptional reprogramming and myeloid priming of HSC [75].
3.3. Osteoblasts
This cell type was the first one proposed to be a part of the hematopoietic niches [27,28,29]. In particular, long-term HSCs were found to be in contact with N-cadherin-positive (N-cad+) osteoblastic precursors lining the inner bone surface and supporting HSCs through, presumably, the BMP signaling pathway [27]. Ablation of N-cad+ osteoblasts caused irreversible changes in the function of HSC during homeostasis and regeneration [29]. N-cad+ osteoblasts were also suggested to control HSC state and functions via non-canonical Wnt signaling [76,77]. Despite this evidence, a number of subsequent studies downplayed a possible role of endosteal cells in the maintenance of HSCs. In particular, conditional deletion of important HSCs regulators CXCL12 or SCF in osteoblasts did not significantly affect HSCs [47,78].
However, recent study revived an interest to endosteal niches [79] by demonstrating that dormant HSCs that are resistant to myeloablation (reserve HSCs) were found to be in contact with N-cad+ mesenchymal progenitors in the endosteal region [80]. Furthermore, ablation of N-cad+ niche cells or inactivation of the SCF gene in them negatively affected reserve HSC maintenance during homeostasis and regeneration. In a recent in vitro co-culture study, adherence to osteoblasts favored self-renewal of HSCs [81].
In the context of possible involvement of endosteal cells in HSC support, it should be mentioned that osteoblasts synthesize many factors affecting HSC, such as TPO, angiopoietin-1 and osteopontin, which prevent an increase in the HSC population [82,83,84]. It has been shown that when osteoblasts are activated by parathyroid hormone, they produce high levels of the Notch ligand Jagged-1, expanding the HSC pool [28]. Osteoblasts were also reported to affect the state of silent HSCs through the signaling pathways Tie2/angiopoietin-1 and TPO/Mpl, contributing to the interaction of HSCs with niche components that promote quiescence and self-renewal of these cells [53,85].
Interestingly, osteoblasts affect not only HSCs, but also more differentiated descendants. In particular, selective elimination of osteoblasts negatively affects the pool of early lymphoid precursors, without at the same time affecting the HSCs themselves or the precursors of the myeloid line [86].
3.4. Megakaryocytes
Megakaryocytes (MKs) reside predominantly in BM and, through production of platelets, are a prominent regulator of hemostasis and vascular integrity, with key roles in thrombosis and inflammatory responses. However, MK role is not limited to platelet production. As a result of sophisticated experimental studies of recent years, MKs also emerged as key regulators of HSCs in BM. Bruns et al., demonstrated that megakaryocytes regulate HSC quiescence [87]. They found nearly 30% of HSCs in BM localized close to MKs, and selective depletion of MKs led to HSC expansion as a result of their quiescence loss. MKs were shown by this team to abundantly produce chemokine CXCL4, which induced quiescence in HSCs. Another study also demonstrated association of HSCs with MKs and reported that MK ablation resulted in HSC expansion [88]. This work, however, implicated TGFβ1 in maintenance of HSC quiescence by MKs. Importantly, under chemotherapeutic stress, MK functioned oppositely to support hematopoiesis reconstitution via transient production of FGF1, which stimulated HSC expansion. A third study corroborated findings that MK depletion resulted in a loss of HSC quiescence, and implicated TPO, secreted by megakaryocytes, in maintenance of HSC [89]. The Lectin-like receptor-2 C-type (CLEC-2) protein was found to stimulate production of TPO by MKs [90]. It should be noted, however, that the role of BM-produced TPO in HSC maintenance is currently being questioned [91].
In further development of the above studies, MK depletion was found to selectively expand myeloid-biased vWF1-positive HSCs [37] As a note of caution, a recent study demonstrated that although HSCs were frequently associated with MKs, their distribution did not differ from that of randomly placed dots [36]. However, since MKs appear to control HSC proliferation through secreted proteins, direct contact between MKs and HSCs may not be necessary.
3.5. Macrophages
Macrophages, like other niche components, have a direct effect on HSCs in BM niches. In particular, they promote retention of HSCs in their BM niches through Nes+ stromal cells, as evidenced by macrophage depletion in BM that results in HSC mobilization into bloodstream [92,93]. Interestingly, VCAM-1+ macrophages in zebrafish have similar functions and contribute to the homing and retention of HSCs in the vascular niche through integrin A4 [94].
Another function of macrophages is to suppress the cell cycle entry of quiescent HSCs through interaction of Duffy antigen/chemokine receptor CD234/DARC on macrophages with CD82 on HSCs. This interaction activates TGF-β1/Smad signaling, whereas CD82 knockout leads to loss of HSCs due to their proliferation and differentiation [95]. In addition, a rare population of BM monocytes and macrophages with high expression of α-smooth muscle actin and cyclooxygenase COX-2 acts to suppress stress-induced exhaustion of HSCs and progenitors by production of prostaglandin E(2) and activation of CXCL12 expression [96].
It should finally be mentioned, although out of context of HSC niches, that macrophages play an exclusive and indispensable role in organization of BM erythroblastic islands, a highly specific niche for erythropoiesis containing a central specialized macrophage surrounded by differentiating erythroblasts [97]. In these structures, macrophages function to advance maturation of erythroblasts in various ways, including their mitochondrial clearance through tunnelling nanotubes [98].
3.6. Adipocytes
The reciprocal relationship between hematopoiesis and adipose tissue within the BM in humans has long been recognized. This notion is based on the fact that at birth bones contain red marrow essentially devoid of adipocytes and very active in hematopoiesis, while in adults red BM is replaced by yellow marrow rich in fat tissue and characterized by reduced hematopoietic activity [99]. The adipocytes, the most abundant stromal component in adult BM, were thus considered as negative regulators of hematopoiesis. This notion was experimentally supported by data indicating that in mice incapable of producing adipocytes, or after inhibition of adipogenesis by PPAR-γ receptor antagonist, hematopoiesis restoration after irradiation was significantly accelerated [100]. One of the relevant mechanisms might be a positive feedback loop in which adipocytes, via intense secretion of MCP-1, both stimulate differentiation of MSCs into new adipocytes and negatively affect HSCs [101].
Recent studies, however, have changed our perception of adipocytes as unambiguously negative HSC regulators [102]. First, adipocytes were shown to support in vitro HSC survival, proliferation, and differentiation for at least 5 weeks in culture. Importantly, in BM adipocytes production of factors with hematopoietic roles such as CXCL12, IL-8, CSF3, LIF was on par with that of MSCs [103]. Second, BM adipocytes were shown to promote regeneration of HSCs and hematopoiesis after irradiation or 5-FU treatment [104]. Adipocytes, as well as their precursors comprising a minor subpopulation of LEPR+ cells, produced SCF necessary for hematopoietic recovery. SCF from LEPR+ cells but not from endothelial or osteoblastic cells also activated regeneration, whereas conditional deletion of SCF in adipocytes inhibited hematopoietic regeneration. Thus, adipocytes formed under hematopoietic stress produce large amounts of SCF and seem to represent an emergency response providing HSCs with factors necessary for their survival and expansion [104]. Noteworthily, Wilson et al., have shown that PPARγ knockout mice lacking adipocytes exhibit severe extramedullary hematopoiesis [105]. This result might be associated with observed dysregulation of CXCL12/CXCR4 axis and suggests that adipocytes may be involved in HSC retention or mobilization.
Other reports indicate that some factors produced by adipocytes and thought to play a role in fat formation (adipokines) may be also involved in regulation of hematopoiesis. In particular, adiponectin was shown to be produced by components of the niche while its receptors are expressed by HSCs. Moreover, adiponectin increased proliferation of HSCs through a p38 MAPK-dependent pathway while maintaining their undifferentiated state, and adiponectin deficiency in mice caused negative changes in the restoration of hematopoiesis after chemotherapy [106].
Another adipocyte-secreted adipokine leptin, although alone, had little effect on survival or proliferation of mouse and human HSCs in vitro [107], synergized efficiently with SCF to stimulate the proliferation of primitive hematopoietic progenitors in vitro in colony formation by HSCs and progenitors in cultures [108]. The pleiotropy of leptin is also manifested in its effect on the differentiation of MSCs into fat cells in vivo [109].
3.7. Lymphoid Cells
Recent experimental evidence indicates that lymphoid cells, similar to other mature hematopoietic cells, participate in the HSCs niche function. FoxP3-positive regulatory T (Treg) cells were found to co-localize with HSCs and to play important role in protection of HSCs from immune attack in the niche. In particular, allogenic HSCs, similar to syngenic ones, are able to survive in non-irradiated mice after transplantation for at least a month, whereas depletion of Treg cells results in a rapid loss of allogenic stem cells [110]. BM niche is thus may be considered as an immunologically privileged site and a protective sanctuary for HSCs. Importantly, Treg depletion impairs function of both MSCs and HSCs and results in reduced hematopoiesis-supporting capacity of the niche [111]. Another study identified a CD150high subpopulation of Treg cells in BM that through CD39 cell surface ectoenzyme produced elevated levels of extracellular adenosine. Adenosine, in turn, potentiated Tregs, protected HSCs from oxidative stress and maintained HSCs quiescence. Moreover, co-tranplantation of this Treg subpopulation was found to promote a much better engraftment of HSCs in allogenic hosts compared to other Treg subsets [112]. Paradoxically, adenosine produced by LEPR+ perivascular MSC seems to work in the opposite direction by activating immunity and reducing immune privilege of the niche [113].
Less evidence exists concerning the potential role of B lymphocytes in the niche. However, neurotransmitter acetylcholine abundantly produced by B cells has recently been shown to limit hematopoiesis in vivo [114].
3.8. Nerve Fibers
The sympathetic nervous system (SNS) has been shown to innervate BM, wrapping around arterioles and contacting peri-arterial Nes-GFP+stromal cells [32,115] A study by Méndez-Ferrer et al., demonstrated that the SNS innervation plays a key role in the circadian mobilization of HSCs form BM [116]. It acts through β3 adrenergic receptors and downregulates CXCL12 levels during daytime, which activates HSC egress from BM. Moreover, a similar mechanism functions during chronic variable stress, which induces sympathetic nerve fibers to release more noradrenaline. This in turn leads to decreased CXCL12 levels, resulting in activation of HSCs and enhanced production of neutrophils and inflammatory monocytes [117].
Importantly, the autonomic cholinergic nervous system acts in the opposite direction, repressing the sympathetic noradrenergic system at night and thus reducing egress of HSCs [118]. In addition, cholinergic system through increased CXCL12 production acts to maintain HSC quiescence in the endosteal niche under proliferative stress [119].
3.9. Single Cell Analysis of Niche Heterogeneity
The studies described in previous chapters demonstrated the vast diversity of BM niche cells. The explosive development of new massive parallel methods of single cell analysis has added a new dimension to our understanding of niche complexity. The majority of the relevant data has so far been obtained using single cell RNA sequencing (scRNAseq) of fluorescently sorted BM cell populations, followed by comprehensive bioinformatics analysis. The work by Wolock et al. [120] studying transcriptomes of CD45−, Ter119−, CD31− BM cells used SPRING visualization algorithm [121] to reveal 7 stromal cell subsets, with MSCs sitting at the top of hierarchy and branching into adipo- and osteo/chondro-lineages. Another report by Baryawno et al. [122] analyzed substantially more cells of a broader Lin−, CD45− BM population and employed t-distributed stochastic neighbor embedding (t-SNE) algorithm for visualization [123]. This study discovered 17 distinct subsets of non-hematopoietic cells in BM, including LEPR+ MSCs, endothelial sinusoidal and arteriolar cells, five fibroblast subsets, as well as several osteolineage and chondrocyte subsets. The changes in BM cells associated with emerging acute myeloid leukemia (AML) were also identified.
Tikhonova et al. [75] employed a different approach to isolate starting cell populations using transgenic mice with fluorescently marked endothelial (VE-cad+), perivascular (LEPR+) or osteo-lineage (Col1a1+) populations, whose transcriptomes were analyzed separately, revealing 2, 4, and 3 distinct subsets, respectively. In addition, cellular sources of haematopoietic cytokines, chemokines, and membrane-bound ligands were identified in this study. Moreover, scRNAseq analysis of 5-FU chemotherapy effects revealed drastic elevation of adipogenesis-related pathways and downregulation of osteolineage signaling during stress hematopoiesis. Zhang et al. [124] aimed to focus their analysis on BM mesenchymal cells using transgenic mice with these cells specifically marked by fluorescent protein. They identified 22 cell subsets; however, only 9 groups were of mesenchymal lineage. Authors observed a novel adipogenic lineage population expressing adipocyte markers but not containing lipid droplets. These cells form a ubiquitous 3D network in BM, maintaining marrow vasculature and suppressing bone formation.
Baccin et al. [125] used an advanced approach combining scRNAseq analysis of sorted cell subsets with spatial transcriptomics of fixed, laser capture microdissected, small (200–300 cells) samples. This work identified two new subsets of CAR cells expressing osteogenic or adipogenic gene subsets (Osteo-CAR cells or Adipo-CAR cells, respectively).
Importantly, re-analysis of results of five mentioned scRNAseq studies using the Seurat pipeline [126] for comparing all datasets demonstrated a general yet relatively limited similarity of results obtained by different groups [127], with the highest similarity observed for Baryawno et al. [122] and Tikhonova et al. [75] data. This, most likely, is a result of differences in cell subsets analyzed, as well as in cell sample digestion and processing protocols. Of note, the sc transcriptome study by Addo et al. [128] exploiting similar approaches observed a very high heterogeneity of expression phenotypes and thus was unable to identify distinct stromal cell subsets using bulk RNA abundance patterns only. However, 14 non-overlapping cell populations were identified based on the clustering of transcripts encoding secreted factors participating in stromal-hematopoietic cell communication. The reasons for this divergence from other groups’ results remain unknown.
Finally, an important study by Severe et al. [129] needs to be mentioned. Authors used a single-cell mass cytometry, a cytometry method based on the use of transition element-labeled antibodies combined with single cell mass spectrometry. This approach allows one to analyze significantly more parameters at once than is possible with conventional fluorescent cytometry. Severe et al., were able to identify 28 distinct subsets of stromal cells in BM under steady-state conditions, although only half of them expressed hematopoiesis-relevant cytokines. Importantly, radiation conditioning resulted in a loss of most of stromal cell subpopulations including LEPR+ and Nes+ niche cells. However, CD73-positive cell subset was retained following irradiation, and ablation of CD73 resulted in defects of homing of transplanted HSCs and decreased hematopoiesis. Thus, the CD73+ stromal subset is likely to promote HSPC engraftment and acute hematopoietic recovery following radiation conditioning.
Table 1 presents a brief summary of various cell types contributing to the BM niches, whereas Table 2 reviews key regulators controlling HSCs.
Table 1.
Key cell types participating in regulation of HSCs activity in BM niches.
| Cells | Markers | Function | Main Molecules |
|---|---|---|---|
| Mesenchymal stem cells | CD73+, CD90+, CD105+, CD14−, CD19−, CD34−, CD45−, HLA−DR−, Terl119−, CD31−, CD51+, PDGFRa+, Sca1− | Support and regulation of HSC quiescence, proliferation, differentiation HSC mobilization |
CXCL12, SCF, angiopoietin-1, VCAM-1, osteopontin |
| Endothelial cells | CD45−, CD31+, CD144+, Terl119− | Support of HSC proliferation and expansion Hematopoietic regeneration after irradiation |
CXCL12, SCF, Notch ligands, E-selectin, Del-1, pleiotrophin |
| Osteoblasts | CD45−, Terl19−, CD31−, CD51+, PDGFRa−, Sca1− | Support of HSC quiescence | Osteopontin, N-cadherin, TPO, angiopoietin-1 |
| Megakaryocytes | CD41+, CXCR4, Mpl | Support of HSC quiescence HSC expansion after irradiation |
CXCL4, TGF-β, thrombopoietin, FGF1 |
| Macrophages | CD68+, CD169+ | HSC retention in niche Support of HSC quiescence |
VCAM-1, DARC, TGF-β |
| Adipocytes | ADIPOQ, FABP4, Leptin | Support of HSC survival, proliferation and differentiation Hematopoietic regeneration after irradiation |
MCP-1, CXCL12, SCF, IL-8, LIF, CSF3, adiponectin, leptin |
| Treg lymphocytes | FOXP3 | Protection of HSC from immune attack Support of HSC quiescence |
IL-10, CDC39, adenosine |
| Sympathetic nerve fibers | Tyrosine hydroxylase | HSCs mobilization | Noradrenaline |
| Parasympathetic nerve fibers | Choline acetyltransferase | HSC retention in niche, homing | Acetylcholine |
Table 2.
Key molecules participating in HSC regulation by BM niches.
| Molecule | Receptors on HSC Surface | Producing Cells | Regulatory Function |
|---|---|---|---|
| CXCL12 | CXCR4 | MSCs, ECs, ADs | HSC maintenance HSC homing and niche retention |
| SCF | KIT | MSCs, ECs, ADs | HSC survival HSC maintenance and proliferation HSC homing |
| TGF-β | TGFBR1 | MKs | HSC quiescence |
| Osteopontin | CD44 | OBs, MSCs | Suppression of HSC proliferation and expansion |
| Angiopoietin 1 | TIE2 | OBs, MSCs | HSC maintenance HSC quiescence |
| VCAM-1 | VLA4 | MSCs, Mϕ | HSC homing |
| G-CSF/CSF3 | CSF3R | MSCs | Myeloid differentiation HSC mobilization |
| M-CSF/CSF1 | CSF1R | MSCs | Myeloid differentiation |
| TPO | MPL | MKs, OBs | HSC maintenance and proliferation HSC quiescence |
| IGF-1 | IGF1R | OBs | HSC maintenance |
| Pleiotrophin | RPTPZ1 | ECs, MSCs | HSC maintenance |
| Jagged-1 | NOTCH | MSCs, ECs, OBs | HSC maintenance and self-renewal HSC expansion |
| EGF | EGFR | ECs | HSCs survival and maintenance |
| DARC | CD82 | Mϕ | HSC quiescence |
| CXCL4 | CXCR3B | MKs | HSC quiescence |
HSCs—hematopoietic stem cells, MSCs—mesenchymal stem/stromal cells, ECs—endothelial cells, ADs—adipocytes, MKs—megakaryocytes, OBs—osteoblasts, Mϕ—macrophages.
4. Niches of Committed Cells
The question arising after reviewing the specialized niches for HSCs in BM is where the potential niches for more differentiated cells are located. Ding et al., showed that while HSCs are maintained by a perivascular niche combining Lepr–cre- or Prx1–cre-expressing stromal cells and endothelium, early lymphoid precursors are located in the endosteal niche created by osteoblasts that support the proliferation and differentiation of lymphoid precursors in vitro [78]. Committed B-lineage progenitor cells are maintained by a different perivascular niche containing Prx1–cre-expressing stromal cells but no endothelial cells [78]. The removal of osteoblasts from the culture negatively affects the population of lymphoid precursors [29,86]. Moreover, the removal of G protein alpha from osteoblasts reduces the population of B lymphoid precursors [130].
The spatial distribution of hematopoietic progenitor cells is confirmed by the work of Comazzetto et al., showing that SCF from LEPR+ cells is generally required for HSCs and more differentiated progenitors [131]. Deletion of SCF from LEPR+ cells led to the depletion of common lymphoid and common myeloid progenitors as well as a number of more differentiated progenitors. At the same time, deletion of SCF from endothelial cells only negatively affected the HSC population, indicating that SCF produced by endothelial cells selectively supports HSCs.
A number of authors note that proliferating HSCs, precursors, and more differentiated cells are located in the sinusoid region [30,132]. Pinho et al., used a different cell labeling method to study the spatial distribution of cells in the niche [37] and showed that platelet- and myeloid-biased HSCs expressing von Willebrand factor (vWF) and CD150 are located near megakaryocytes, and the depletion of the latter expands the population of vWF+ HSCs, but reduces the ability to self-renew. At the same time, vWF lymphoid-biased HSCs in arteriolar niches are dependent on NG2+ cells. According to Winkler, multipotent precursors are located on the endosteal surface, whereas lymphoid precursors migrate from this region [133]. Balzano et al., demonstrated that HSCs and pro-B cells are often located in the same perisinusoidal niche next to LEPR+ cells [134].
5. Metabolic State of HSCs and Niche
Functional, transcriptomic, and proteomic studies demonstrated the importance of metabolic processes in biology of HSCs [135]. Most of the time HSCs are quiescent, which protects them against proliferative and genotoxic stress effects harmful to HSCs, while allowing them to maintain their self-renewal potential [136,137]. It is well documented that reactive oxygen species (ROS) cause damage to biomolecules, primarily DNA [138], representing a particular danger for HSCs that give rise to enormous numbers of progeny cells. Several studies established that HSCs in the BM are located in regions of severe hypoxia, with the lowest O2 levels in deeper peri-sinusoidal regions, while the endosteal regions are less hypoxic due to arteriole perfusion [31,139].
Quiescent HSCs in BM under these hypoxia conditions adapt their metabolism to switch to glycolysis [140] as energy source, with resulting decrease in oxidative phosphorylation (OxPhos) and production of ROS in mitochondria [136,141]. This switch is mediated by pyruvate dehydrogenase kinase (Pdk)-dependent mechanism [142]. In addition, the levels of transcription factors Meis1 and Hif-1α are highly elevated in long-term HSCs, while Meis1 positively regulates HSC glycolytic metabolism through transcriptional activation of Hif-1alpha [140]. The notion that hypoxia is necessary to maintain the functions of the HSCs is confirmed by the increasing expansion of colony-forming cells (CFCs) [143,144] or the restoration of quiescence in hypoxia ex vivo [145,146].
It should be noted, however, that ROS is an important mediator of intracellular signaling. In relation to HSCs biology, decrease in ROS below critical levels by inactivation of AKT1 and AKT2 kinases inhibits differentiation of HSCs [147]. In addition to AKT1 and AKT2, ROS levels in HSCs are regulated by ATM [148] and the Foxo transcription factor family [149,150]. The transition of HSCs to active state, including proliferation and differentiation into progenitor cells, is initiated by an increase in the concentration of ROS due to the activation of OxPhos [147,151], switching from glycolysis to the Krebs cycle to meet the rapidly growing energy needs of HSCs [135,141]. Apparently, this switch occurs through CD36-mediated uptake of free fatty acids [152] and involves histone demethylase Fbxl10 [153]. In addition, Maryanovich et al. [154] showed that loss of mitochondrial carrier homolog 2 (MTCH2) leads to an increase in OxPhos in mitochondria. This is accompanied by an increase in mitochondrial size and accumulation of ROS and ATP and, as a result, activation of HSC and an exit from the quiescent state.
The role of mitochondria is further emphasized by the finding that mitofusin 2 (Mfn2), a protein involved in mitochondrial fusion, is necessary for maintenance of lymphoid-biased but not myeloid-biased HSCs, the effect most likely mediated through intracellular accumulation of Ca2+ and NFAT signaling [155]. In addition, lowering the mitochondrial potential in HSCs favors their self-renewal at the expense of differentiation [156].
An interesting study demonstrates a critical function of p38MAPK family isoform p38α in transition from quiescence to proliferation [157]. After hematological stress, p38α is rapidly phosphorylated, which leads to elevated expression of IMPDH2 in HSPCs and activation of purine metabolism, whereas deletion of p38α results in suppression of recovery and delayed HSC proliferation.
Another important metabolic pathway, namely fatty acid oxidation (FAO), has also a prominent role in HSC biology. Ito et al., showed that the PML–PPAR-δ–FAO signaling controls the asymmetric division of HSCs, and its inhibition leads to symmetric divisions and loss of HSC maintenance [158]. Activation of PPAR-δ–FAO pathway leading to expansion of HSCs occurs through enhanced Parkin recruitment in mitochondria and induction of mitophagy [159]. Interestingly, depletion of PTPMT1, a PTEN-like mitochondrial phosphatase, affected mitochondrial metabolism and enhanced activation of mitochondrial uncoupling protein 2 by fatty acids, which was accompanied by block in differentiation and approximately 40-fold expansion of primitive HSC population [160].
Autophagy has also been implicated in the HSC maintenance. In particular, autophagy has a vital function in protecting HSCs from metabolic stress, and transcription factor Foxo3a is important for rapid induction of autophagy in HSCs upon starvation [161]. HSCs with suppressed autophagy accumulate mitochondria and acquire an activated metabolic state, which leads to accelerated myeloid differentiation and loss of regenerative potential, features characteristic for aged hematopoietic system. Thus, autophagy suppresses metabolism of HSCs to maintain their quiescence and stemness [162]. It should be noted, however, that excessive autophagy and mitophagy may be deleterious. For example, deletion of the mitochondrial membrane protein Atad3a (ATPase family, AAA domain containing 3A) causes hyperactivation of mitophagy, which in turn causes differentiation block with concomitant expansion of HSC pool [163].
Another work emphasizing important role of mitochondria in the HSC maintenance and function demonstrated that inactivation of histone deacetylase Sirtuin 7 (Sirt7) provokes cycling of HSCs and their decreased regenerative capacity [164]. These effects were due to suppression of mitochondrial unfolded protein response (UPRmt) in HSCs mediated by activation of nuclear respiratory factor 1 (Nrf1), a master regulator of mitochondria. Vice versa, upregulation of Sirt7 improved the functionality of aged HSCs. It has also been reported that UPRmt is activated in HSCs when they exit quiescent state [165].
Other studies demonstrated important role in HSCs biology of AMP and mTOR pathways, which sense and coordinate nutrient level, energy status and cell growth. Thus, deletion of LKB1, a well-known regulator of AMPK, was shown to reduce mitochondrial membrane potential and ATP levels, induce HSC exit from quiescence and increased proliferation resulting in rapid depletion of HSC and all hematopoietic subpopulations [166,167]. Conditional deletion of TSC1 caused activation of the mTOR signaling pathway in HSCs and provoked their entry into cell cycle, leading to the depletion of the HSC pool and, as a result, dramatically reduced hematopoiesis and self-renewal of HSCs [168].
There is a relative paucity of data regarding the metabolic state of various components of BM niches. MSCs, similar to HSCs, exist in regions of deep hypoxia in BM niches and, therefore, use glycolysis for their energy needs [169]. In the state of normoxia, the proliferative properties of MSCs increase, but aging also increases [170], therefore hypoxia might serve as a way to prevent proliferation and aging of MSCs, maintaining self-renewing MSC population of BM.
Finally, in the context of mitochondria role in HSC function, it would be important to discuss the issue of mitochondrial transfer between cells in BM. According to numerous reports, MSCs, a vital component of HSC niches, are able to donate their mitochondria to various cell types in vitro, either through direct contact or via secretion of extracellular vesicles (reviewed in [171]). This process modulates properties of recipient cells and, frequently, results in restoring their functions. Moreover, MSCs may sense mitochondria released from injured cells and enhance their own mitochondrial biogenesis to combat cell injury [172]. It is thus possible that a similar functional restoration of HSCs containing damaged or exhausted mitochrondria by niche MSCs may occur in vivo. Of note, a reverse process of mitochondrial donation by healthy hematopoietic progenitor cells to damaged irradiated stromal cells during bone marrow transplantation has been reported [173].
6. Aging of the Niche
Age-related changes in the human body negatively affect hematopoiesis, leading to shifts in blood cell clonal composition, DNA damage and eventual development of hematological disorders [174,175,176]. The aging of the hematopoietic system involves two major components: the cell-intrinsic aging of the HSCs themselves, and age-related changes in the components of the BM niche that have a direct impact on the HSCs and hematopoiesis.
Aging affects HSCs in two ways: on the one hand, HSC functional capacity declines with accumulation of genetic, epigenetic, metabolic and homeostatic defects, on the other hand, the HSC pool expands, with a skewed differentiation towards the myeloid lineage at the expense of the lymphoid one [176,177,178]. Age-related changes are also associated with DNA and telomere damage; for aged HSCs, an increased level of DNA mutations was observed [179]. Telomere shortening leads to functional depletion of HSCs due to constitutive activation and asymmetric differentiation into megakaryocytes [180]. Accumulation of mutations in epigenetic regulator genes in HSCs with age leads to clonal hematopoiesis, enhanced inflammation and increased risk of cardiovascular diseases and malignancies [181,182]. The aging of HSCs is manifested at the organelle level as well. Various defects in the mitochondrial respiratory chain or mitochondrial biogenesis lead to disturbance of HSC quiescence and their dysfunction [164,168,183]. Among other prominent hallmarks of HSC aging are deregulation of autophagy [162,184] and modifications in epigenome [185,186]. Importantly, HSCs lose polarity with aging as a result of enhanced activity of the small RhoGTPase Cdc42 [187]. It should be noted that the biologically youngest HSC subset in the aged mice that retains cellular polarity, quiescence, and conserved regenerative potential was shown to be located in perisinusoidal niches [188].
HSCs do not age alone in BM; age-related changes occur in the BM niche components as well. In a number of adult stem cells, aging is accompanied by a continuous somatic DNA mutagenesis occurring at a rate of about 40 novel mutations per year [189]; the same is likely to occur in MSCs as well. The aging of MSCs is accompanied by an increase in ROS levels and DNA damage [190,191,192]. The lack of telomerase negatively impairs the ability of MSCs to differentiate [193]. Moreover, telomere dysfunction in Tert knockout mice enhanced myelopoiesis during aging at the expense of B lymphoid lineage, impaired MSC function and reduced their capacity to maintain functional HSCs, in particular their ability to support the engraftment of wild-type HSCs in KO animals [194].
The secretory ability and immunomodulatory properties of MSCs significantly weaken during aging in culture [195]. In line with in vitro data, Gnani et al., showed that MSCs isolated from aged donors displayed senescent phenotype, accumulation of DNA damage and increased secretion of multiple pro-inflammatory factors with concomitant reduction in immunomodulatory properties [196]. Factors secreted by aged MSCs activated expression of proinflammatory genes in young HSCs and reduced their clonogenic potential.
The vascular component also undergoes major alterations in the advanced age. Although the vascular volume remains fairly constant, a decrease in the number of ECs occurs [188]. With age, degeneration of arteries and arterioles occurs [197], they become disorganized, which impairs their ability to maintain HSC quiescence and affects function of the latter [188,198]. At the same, sinusoidal network undergoes little degradation [198]. Netrin-1 expressed by endothelial and periarteriolar stromal cells of the BM niche was recently reported to support HSC quiescence and self-renewal, while decline of its production with ageing negatively affects HSC maintenance [199]. It is worth noting that other genes in ECs have also been implicated in the aging of the niche. In particular, expression of mTOR and heme oxygenase-1 in BM ECs declines during aging, which impairs support of hematopoiesis [200,201].
Substantial alterations occur in the endosteal compartment with aging. The ability of MSCs to differentiate into osteoblasts decreases with aging, which is associated with decreased secretion of osteopontin, which, in turn, negatively affects the population of HSCs [83]. In aged mice, the bone matrix is reduced, and the number of mature osteoblasts diminished [202]. A decrease in the number of osteocytes in the niche leads to a shift in the differentiation of HSC towards myelopoiesis [203], which is further stimulated by the influx of plasma cells during aging [204].
The adipose component also changes with aging. Shift in MSCs during aging towards differentiation into adipose tissue results in niche remodeling associated with pathological accumulation of adipocytes, which disrupts hematopoietic regeneration [205]. It is assumed that accumulation of adipocytes in the niche can affect the immunomodulatory activity of MSCs, increasing or weakening the inflammatory state and contributing to a decrease in the population of HSCs [205,206,207].
In the aging BM, a decrease in sympathetic adrenergic nerve density and innervation was reported, with resulting increase in HSCs proliferation, loss of Cdc42 polarity, myeloid shift in differentiation and decrease in HCS transplantation potential [197]. However, a more recent study, on the contrary, reported an increase in sympathetic innervation with age, which promoted myeloid differentiation and increased HSC frequency [208].
One of the important negative changes in aging is development of the inflammatory process (inflammaging) in the niche [209]. Activation of signals associated with aging, such as NF-kB, β-catenin/WNT, elicits a pro-inflammatory shift in MSCs that provokes the expression and secretion of senescence-associated pro-inflammatory cytokines, chemokines and ligands, such as S100A8/A9, IL6, IL-1ß and others, into the niche [210]. Such a proinflammatory environment in the niche favors DNA damage, malignant transformation and the appearance of mutations in HSCs, which may lead to oncogenesis [196,211,212,213].
Finally, a number of interesting aging-related experiments were performed to elucidate the heterochronic interactions of HSCs with the hematopoietic niche. Transplantation of aged HSCs into young mice did not result in their conversion to a younger state and functionality, although a rejuvenation of HSC transcriptome, but not methylome, was observed [214,215]. Similarly, aged microenvironment had little pro-aging effect on transplanted young HSCs [214], although young HSC pool expansion biased towards myeloid differentiation was reported [216,217]. These data indicate that HSCs largely maintain their chronological age in the heterochronic environment.
Table 3 summarizes changes in HSC-niche system that occur during aging.
Table 3.
Phenotypic and functional aging-related alterations in HSCs and BM niche cells.
| Cell Types | Changes with Ageing |
|---|---|
| HSCs | ↑myeloid differentiation; ↓lymphoid differentiation; ↓regenerative potential; ↓HSC polarity; ↓autophagy; ↑deregulated mitochondrial activity; ↑epigenetic and genomic alterations |
| MSCs | ↓CFU-F clonogenicity; ↓Nes–GFP+ and NG2+ cells; ↓CXCR4 →↑ROS production ↑DNA damage→↓ HSCs support; ↑IL6 expression, ↑TGF-β expression →aged HSCs phenotype |
| ECs | ↓ECs number, vascular remodeling → loss of HSC quiescence; ↓key signaling pathways in ECs (mTOR, Jag1/Notch, CXCL12, SCF); ↓HO-1 expression →aged HSC phenotype |
| OBs | ↓ OBs number →↓OPN secretion → aged HSC phenotype; ↓osteogenic progenitor population |
| MKs | ↑ MKs number |
| Mϕ | ↑ Mϕ number; ↑IL-1 secretion→↑HSC myeloid differentiation |
| ADs | ↑ADs number →↓HSCs and progenitors numbers→↓repopulation capacity |
| Nerve fibers | ↓nerve density; ↑β2-adrenergic stimulation ↑→IL6 secretion by MSCs →↑HSC myeloid differentiation |
↓—decrease, ↑—increase, →—leads to, HSCs—hematopoietic stem cells, MSCs—mesenchymal stem/stromal cells, ECs—endothelial cells, ADs—adipocytes, MKs—megakaryocytes, OBs—osteoblasts, Mϕ—macrophages.
7. Niche Transformation in Leukemia
Numerous data indicate that during leukemia, malignant cells, in addition to perturbation of normal hematopoiesis, are able to substantially modify the BM niche components, and the hematopoietic microenvironment undergoes changes that facilitate disease progression. In particular, during chronic lymphocytic leukemia BM MSCs change their properties to give rise to drastically fewer fibroblast colony-forming units (CFU-Fs), proliferate slowly and behave as senescent cells [218]. Senescence was also observed for MSCs from AML BM [219]. Moreover, MSC senescence is reported for an in vitro leukemia niche model [220].
One of the leukemia hallmarks is enhanced angiogenesis, which is activated by proangiogenic factors secreted by tumor cells. This “superangiogenesis” allows rapidly proliferating malignant cells to spread throughout the body and is also an indicator of leukemia progress [221]. The key angiogenesis factor VEGF activates protection of leukemic cells from apoptosis by Hsp90-medited induction of anti-apoptotic factors BCL2 and MCL1 [222,223]. In co-cultures of endothelial cells with AML, an increase in secretion of IL-3, IL-6, G-CSF and GM-CS cytokines was observed, which stimulated AML growth and suppressed apoptosis [224].
In connection to the rapid proliferation of leukemic cells in BM, adipocytes acquire small size due to lipolysis [225]. As a result, a large amount of free fatty acids is formed, which are necessary to maintain the existence of malignant cells [226]. Transition from larger to small adipocytes during AML [227] was mediated by growth differentiation factor-15 (GDF15) secreted by leukemic cells, which enhanced expression of thermogenic and lipolytic genes stimulating lipolysis [228]. Notably, disruption of the adipocyte niche in leukemia BM negatively affects endogenous myelo-erythropoiesis [229]. Among other niche components, AML was shown to affect sympathetic nervous system in BM. The resulting neuropathy affects the Nes+ MSCs that support quiescent HSCs in the niche, inducing MSC differentiation into precursors primed for osteoblastic differentiation [230].
The BM microenvironment plays a substantial role in leukemia progression. Leukemia clones at first propagate locally in BM and then spread to other regions in bones. In particular, leukemia cells home to BM vasculature expressing CXCL12 and E-selectin [231]. CXCL12 deletion from vascular endothelial, but not perivascular, cells impeded T-cell acute lymphoblastic leukemia (T-ALL) [232]. Similar effects were reported for deletion of the CXCR4 (CXCL12 receptor) in T-ALL cells.
In the context of leukemia pathology, it would be important to mention that normal HSCs placed in the leukemia niche in vitro activate the cell cycle and start to intensely proliferate [220]. One of the major mechanisms responsible for this effect is the exosome-mediated intercellular communication between malignant and normal cells [233,234]. Leukemic cells capture and endocytose MSC-produced exosomes containing fibroblast growth factor 2 (FGF2), which protects against tyrosine kinase inhibitors [235]. Moreover, leukemic exosomes are able to stimulate the production of IL-8 by BM MSCs, which in turn supports the spread of leukemia [236]. The ability of leukemic cells to remodel the normal niches manifests in the fact that exosomes from these cells are able to enter and significantly modify the surrounding cells. By virtue of their RNA and protein load, exosomes modulate the secreting profiles of niche cells, accelerating leukemic growth and advancing leukemia progress [237,238,239]. Yet another mechanism was observed in the study by Marlein et al. [240], where production of superoxide by NADPH oxidase-2 in AML stimulated transfer of mitochondria from BM MSCs to AML cells through tunneling nanotubes.
It should be noted that the pathological remodeling of the niche by leukemia cells is not the only way of how the BM microenvironment may promote hematological disorders. Since the niche is thought to strictly control proliferation of HSCs and progenitors, genetic lesions of niche cells may have a disturbing effect on hematopoiesis. Thus, mice deficient in retinoic acid receptor gamma (RARγ) develop myeloproliferative syndrome with abnormal myeloid specification and proliferation, which is elicited solely by the defects in microenvironment [241]. Inactivation of Mind bomb-1, which participates in endocytosis of ligands of Notch receptor, results in myeloproliferative disease with accumulation of immature granulocytes that was due to abnormally functioning non-hematopietic BM cells [242]. Moreover, activating β-catenin mutation in osteoblasts results in upregulation of Notch ligand Jagged 1, which leads to leukemogenesis [243].
Finally, it would be important to discuss the significance of leukemic niche studies for development of new therapeutic strategies. Although the current anti-leukemia therapies target exclusively the malignant cells proper, the accumulating evidence for active and vital synergism between leukemic niche and leukemia cells suggests that this synergism might be a novel and promising therapeutic target. This field is still at its beginning, however, and it should be mentioned that the initial attempts to target vascular niche using anti-angiogenic drugs have been largely unsuccessful [244]. At the same time, targeting niche-leukemia interaction seems to hold more promise. In particular, blocking interaction of CXCR4 with its ligand CXCL12 by plerixafor demonstrated efficacy in several combination trials in AML [245,246,247]. Currently, a number of other substances blocking the CXCL12-CXCR4 axis, such as BL 8040, CX-01 and ulocuplumab, are in early phase trials [248,249,250].
Inhibition of E-selectin in vascular niche by uproleselan resulted in a promising outcome in a phase 1/2 clinical trial of relapsed/refractory AML [251]. Among the most promising targets is also an E-selectin ligand CD44, in particular its CD44v6 isoform expressed in AML SCs but not in normal HSCs. For detailed information on translational research in the field of leukemia-niche interactions, the reader is referred to recent excellent reviews [245,252,253].
8. Niche Modeling
HSC transplantation is one of the most powerful tools for the treatment of blood diseases, resulting in the restoration of functioning hematopoiesis in the body. However, HSC transplantation is limited by the availability of suitable HLA-matched donors and risks associated with GVHD development, insufficient graft quantity or its rejection [254]. These problems could be solved by developing an ex vivo system for expansion of donor HSCs in the quantities required for successful transplantation.
The developers of these ex vivo HSC expansion systems are facing a formidable problem of mimicking the complexity of the hematopoietic niche combined with a correct blend of external signals. To create a dynamic niche model, a combination of cells, growth factors/cytokine and sufficiently elastic biomaterials providing a 3D structure is being used [255,256]. The key supporting elements in making niche lookalikes are arguably the 3D scaffolds [257]. The ex vivo system should be functionally, if not structurally, similar to BM in order to ensure long-term HSC well-being and expansion. For the creation of 3D scaffolds, synthetic, natural and biomaterials are used that may provide a suitable framework for attachment, survival and free movement of cells.
The problem of biocompatibility sets certain restrictions on the choice of materials for 3D structures [258]. Hydrogels are among the most biocompatible materials and are being widely used to prepare scaffolds imitating BM structures [256,259,260,261]. Thus, Ravichandran et al., developed a model of BM adipose tissue by combining gelatin and a composite methacryloyl hydrogel/polycaprolactone scaffold with human BM MSCs subjected to mechanical stimulation [262]. Scaffolds from polydimethylsiloxane used as BM mimics allowed for the successful cultivation of HSCs, which, in the 3D culture, activated pathways maintaining cell pluripotency and promoting their expansion [263]. Studies by Ventura Ferreira et al., using co-cultures of cord blood CD34+ cells and MSCs on various 3D biomaterial scaffolds revealed that the fibrin-based scaffold provided best culture conditions and maximally contributed to the expansion of CD34+ cells [264]. The use of biocompatible zwitterion hydrogels made it possible to improve the properties of the scaffolds and achieve a multifold increase in the frequency of hematopoietic progenitor cells capable of long-term hematopoietic reconstitution in immunocompromised mice [265]. Other types of scaffolds are also being used to recreate the niche, and their low biocompatibility is improved by coating nanofibers with fibronectin [266,267], collagen [268] or by prior cultivation of MSCs [268]. A ceramic scaffold pre-populated with MSCs and osteoblasts, which secreted ECM and cytokines necessary for the maintenance of hematopoietic progenitors, successfully promoted the expansion of the latter [269].
Born et al., created a system imitating BM using the stromal vascular fraction (SVF) from human adipose tissue seeded on 3D osteoblastic niches formed by MSCs, in which SVF cells assembled into vascularized structures containing both endothelial and perivascular cells, and which provided a better support for HSCs than the non-vascularized 3D niches [270]. An artificial niche created from silk fibroin reproduced platelet biogenesis [271]. The use of natural materials—collagen and fibrin—in the design of the scaffold made it possible to support the proliferation and expansion of HSCs [259,264].
Another strategy for creation of biomimetic 3D scaffolds is the use of decellularized animal and human tissue. This enables one to simulate the architecture of the niche for cell cultivation as close to the natural environment as possible while eliciting a smaller host immune response during transplantation [272,273]. Properly performed decellularization of the tissue diminishes an unfavorable immune response [274,275]. Bianco et al., created a novel decellularized BM scaffold using a cell removal method that preserves the native 3D-structure of BM with its blood vessels and niches, thus supporting adhesion and proliferation of both stromal cells and HSCs [276]. Hashimoto et al., employed high hydrostatic pressure for bone decellularization, which allowed HSC recruitment after subcutaneous transplantation [277]. Lai et al., used yet another option for co-cultivation of HSCs and MSCs, namely, the decellularized bone-like matrix containing osteogenically differentiated MSCs encapsulated in collagen microspheres [278].
One of the options for modeling the BM niche is the cultivation of HSCs, MSCs and ECs in 3D multicellular spheroids using a magnetic levitation system [279]. This study demonstrated, in general agreement with previous data, that exposure of spheroids to hypoxia is beneficial for HSCs, resulting in enhanced cell proliferation and increased expression of CD34 and some other markers. Spheroid culture (hematosphere) from mononuclear blood cells demonstrated the possibility of maintaining and expansion of Lin−CD34+CD38− hematopoietic progenitors [280]. The creation of vascularized miniature bone/BM organoids in mice using umbilical cord blood and cord blood fibroblasts allowed for successful engraftment and maintenance of HSCs [281].
A highly promising way to create and monitor the niche as a constantly changing microenvironment is the organ-on-a-chip technology based on microfluidics. With the help of porous membranes or scaffolds and a system of microchannels, a suitable environment is created in which MSCs are seeded to prepare an appropriate niche for HSCs. Kefallinou et al., created such an organ-on-a-chip system using two chambers separated by a porous membrane, in one of which MSCs were cultivated [282]. In another application of organ-on-a-chip technology, a zirconium oxide scaffold coated with hydroxyapatite was used to co-cultivate MSCs and hematopoietic progenitors in the microfluidic chip [283]. This system created a microenvironment similar to BM that supported maintenance of CD34+ CD38− progenitors. Glaser et al., presented a microfluidic 3D model of BM combining two perivascular and endosteal compartments and a perfusable vascular network, which successfully maintained CD34+ cells and allowed for their differentiation and egress of neutrophils [284]. For a detailed study of multiple myeloma, a microfluidic system imitating BM stromal and sinusoidal endothelium components, as well as sinusoidal circulation, made it possible to study CXCL12-mediated egress of myeloma cells from BM stroma [285]. The use of microfluidic technology for niche modeling will undoubtedly undergo further development. For example, the application of the method of maskless photolithography to create more sophisticated microfluidic devices that can be adapted for specific research needs holds significant promise in further progress of organ-on-a-chip systems [286].
As a final note, it would be important to mention the 3D printing technology, a potentially very powerful approach considered one of the major avenues for creation of artificial tissues and organs, despite tremendous technical and methodological hurdles that are lying ahead [287,288]. In the field of stem cell biology, this technology might eventually allow one to re-create sophisticated nature-like stem cell niches using biomaterials and various cell types [289]. Although this field is still in its infancy and no major breakthroughs have been yet reported, some steps towards achieving the above goal have already been made [290,291].
9. Concluding Remarks
The hematopoietic system is arguably unique among the body’s systems in the numeric cellular output and variety of cell types it generates. The sheer number of cells it produces daily presents a significant potential danger of malignant transformation or inability to correctly meet the various needs of organisms. Given this, it is no wonder that hematopoiesis is subjected to a very strict control, while HSCs are kept on a short leash and their behavior is tightly regulated by multiple neighbor cells that constitute the niche for HSCs. Unraveling the enigmas of different molecular and cellular mechanisms underlying HSC-niche interactions can thus be considered the Holy Grail of hematopoietic research. The most sophisticated techniques such as genetic lineage tracing and single cell transcriptome analysis are being increasingly used to understand the astonishing complexity of hematopoietic regulation by the BM microenvironment. Experiments performed until now have already yielded quite a few important discoveries in this field that are changing our paradigms. However, the unified and commonly accepted view of how HSCs interact with the BM microenvironment to fulfill their vital functions is still lacking. Although at least some of the major HSCs regulators produced by niche cells have been identified, we are as yet very far from obtaining an integral picture of how all numerous actors of the HSC-niche ecosystem work together to help HSCs to make correct decisions concerning their survival, division and differentiation in response to organism’s needs.
Future research will undoubtedly focus of these issues, and one may hope that it will not only advance our understanding of the complexities of HSC regulation by the niche, but also provide vital clues as to the identity of molecules and interactions that are required to maintain and multiply HSCs for clinical applications, as well as combat hematopoietic diseases.
Author Contributions
Conceptualization, A.B. and E.S.; Writing—Original draft preparation, O.K. and E.S.; Writing—Review and editing, A.B., O.K. and E.S. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Conflicts of Interest
The authors declare no conflict of interest.
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Kaushansky K. Lineage-specific hematopoietic growth factors. N. Engl. J. Med. 2006;354:2034–2045. doi: 10.1056/NEJMra052706. [DOI] [PubMed] [Google Scholar]
- 2.Watson C.J., Papula A.L., Poon G.Y.P., Wong W.H., Young A.L., Druley T.E., Fisher D.S., Blundell J.R. The evolutionary dynamics and fitness landscape of clonal hematopoiesis. Science. 2020;367:1449–1454. doi: 10.1126/science.aay9333. [DOI] [PubMed] [Google Scholar]
- 3.Wang J.C., Doedens M., Dick J.E. Primitive human hematopoietic cells are enriched in cord blood compared with adult bone marrow or mobilized peripheral blood as measured by the quantitative in vivo SCID-repopulating cell assay. Blood. 1997;89:3919–3924. doi: 10.1182/blood.V89.11.3919. [DOI] [PubMed] [Google Scholar]
- 4.Schulz C., Gomez Perdiguero E., Chorro L., Szabo-Rogers H., Cagnard N., Kierdorf K., Prinz M., Wu B., Jacobsen S.E., Pollard J.W., et al. A lineage of myeloid cells independent of Myb and hematopoietic stem cells. Science. 2012;336:86–90. doi: 10.1126/science.1219179. [DOI] [PubMed] [Google Scholar]
- 5.Hashimoto D., Chow A., Noizat C., Teo P., Beasley M.B., Leboeuf M., Becker C.D., See P., Price J., Lucas D., et al. Tissue resident macrophages self-maintain locally throughout adult life with minimal contribution from circulating monocytes. Immunity. 2013;38:792–804. doi: 10.1016/j.immuni.2013.04.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Beaudin A.E., Boyer S.W., Perez-Cunningham J., Hernandez G.E., Derderian S.C., Jujjavarapu C., Aaserude E., MacKenzie T., Forsberg E.C. A Transient Developmental Hematopoietic Stem Cell Gives Rise to Innate-like B and T Cells. Cell Stem Cell. 2016;19:768–783. doi: 10.1016/j.stem.2016.08.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Cheshier S.H., Morrison S.J., Liao X., Weissman I.L. In vivo proliferation and cell cycle kinetics of long-term self- renewing hematopoietic stem cells. Proc. Natl. Acad. Sci. USA. 1999;96:3120–3125. doi: 10.1073/pnas.96.6.3120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Cheng T., Rodrigues N., Shen H., Yang Y., Dombkowski D., Sykes M., Scadden D.T. Hematopoietic stem cell quiescence maintained by p21cip1/waf1. Science. 2000;287:1804–1808. doi: 10.1126/science.287.5459.1804. [DOI] [PubMed] [Google Scholar]
- 9.Wilson A., Laurenti E., Oser G., van der Wath R.C., Blanco-Bose W., Jaworski M., Offner S., Dunant C.F., Eshkind L., Bockamp E., et al. Hematopoietic stem cells reversibly switch from dormancy to self- renewal during homeostasis and repair. Cell. 2008;135:1118–1129. doi: 10.1016/j.cell.2008.10.048. [DOI] [PubMed] [Google Scholar]
- 10.Baldridge M.T., King K.Y., Boles N.C., Weksberg D.C., Goodell M.A. Quiescent haematopoietic stem cells are activated by IFN-gamma in response to chronic infection. Nature. 2010;465:793–797. doi: 10.1038/nature09135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Höfer T., Rodewald H.R. Differentiation-based model of hematopoietic stem cell functions and lineage pathways. Blood. 2018;132:1106–1113. doi: 10.1182/blood-2018-03-791517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Yamamoto R., Morita Y., Ooehara J., Hamanaka S., Onodera M., Rudolph K.L., Ema H., Nakauchi H. Clonal analysis unveils self-renewing lineage-restricted progenitors generated directly from hematopoietic stem cells. Cell. 2013;154:1112–1126. doi: 10.1016/j.cell.2013.08.007. [DOI] [PubMed] [Google Scholar]
- 13.Busch K., Klapproth K., Barile M., Flossdorf M., Holland-Letz T., Schlenner S.M., Reth M., Höfer T., Rodewald H.R. Fundamental properties of unperturbed haematopoiesis from stem cells in vivo. Nature. 2015;518:542–546. doi: 10.1038/nature14242. [DOI] [PubMed] [Google Scholar]
- 14.Sun J., Ramos A., Chapman B., Johnnidis J.B., Le L., Ho Y.-J., Klein A., Hofmann O., Camargo F.D. Clonal dynamics of native haematopoiesis. Nature. 2014;514:322–327. doi: 10.1038/nature13824. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Walter D., Lier A., Geiselhart A., Thalheimer F.B., Huntscha S., Sobotta M.C., Moehrle B., Brocks D., Bayindir I., Kaschutnig P., et al. Exit from dormancy provokes DNA-damage-induced attrition in haematopoietic stem cells. Nature. 2015;520:549–552. doi: 10.1038/nature14131. [DOI] [PubMed] [Google Scholar]
- 16.Yamamoto R., Wilkinson A.C., Ooehara J., Lan X., Lai C.-Y., Nakauchi Y., Pritchard J.K., Nakauchi H. Large-Scale Clonal Analysis Resolves Aging of the Mouse Hematopoietic Stem Cell Compartment. Cell Stem Cell. 2018;22:600–607.e4. doi: 10.1016/j.stem.2018.03.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Notta F., Zandi S., Takayama N., Dobson S., Gan O.I., Wilson G., Kaufmann K.B., McLeod J., Laurenti E., Dunant C.F., et al. Distinct routes of lineage development reshape the human blood hierarchy across ontogeny. Science. 2016;351:aab2116. doi: 10.1126/science.aab2116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Carrelha J., Meng Y., Kettyle L.M., Luis T.C., Norfo R., Alcolea V., Boukarabila H., Grasso F., Gambardella A., Grover A., et al. Hierarchically related lineage-restricted fates of multipotent haematopoietic stem cells. Nature. 2018;554:106–111. doi: 10.1038/nature25455. [DOI] [PubMed] [Google Scholar]
- 19.Rodriguez-Fraticelli A.E., Wolock S.L., Weinreb C.S., Panero R., Patel S.H., Jankovic M., Sun J., Calogero R.A., Klein A.M., Camargo F.D. Clonal analysis of lineage fate in native haematopoiesis. Nature. 2018;553:212–216. doi: 10.1038/nature25168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Qin P., Pang Y., Hou W., Fu R., Zhang Y., Wang X., Meng G., Liu Q., Zhu X., Hong N., et al. Integrated decoding hematopoiesis and leukemogenesis using single-cell sequencing and its medical implication. Cell Discov. 2021;7:2. doi: 10.1038/s41421-020-00223-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Bryder D., Rossi D.J., Weissman I.L. Hematopoietic stem cells: The paradigmatic tissue-specific stem cell. Am. J. Pathol. 2006;169:338–346. doi: 10.2353/ajpath.2006.060312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Schofield R. The relationship between the spleen colony-forming cell and the haemopoietic stem cell. Blood Cells. 1978;4:7–25. [PubMed] [Google Scholar]
- 23.Xie T., Spradling A.C. A niche maintaining germ line stem cells in the Drosophila ovary. Science. 2000;290:328–330. doi: 10.1126/science.290.5490.328. [DOI] [PubMed] [Google Scholar]
- 24.Iversen P.O. Blood flow to the haemopoietic bone marrow. Acta Physiol. Scand. 1997;159:269–276. doi: 10.1046/j.1365-201X.1997.00107.x. [DOI] [PubMed] [Google Scholar]
- 25.de Saint-Georges L., Miller S.C. The microcirculation of bone and marrow in the diaphysis of the rat hemopoietic long bones. Anat. Rec. 1992;233:169–177. doi: 10.1002/ar.1092330202. [DOI] [PubMed] [Google Scholar]
- 26.Cora M.C., Latimer K., Travlos G.C. Bone Marrow. In: Suttie A.W., editor. Boorman’s Pathology of the Rat. 2nd ed. Elsevier Inc Academic Press; Boston, MA, USA: 2018. pp. 495–519. [DOI] [Google Scholar]
- 27.Zhang J., Niu C., Ye L., Huang H., He X., Tong W.G., Ross J., Haug J., Johnson T., Feng J.Q., et al. Identification of the haematopoietic stem cell niche and control of the niche size. Nature. 2003;425:836–841. doi: 10.1038/nature02041. [DOI] [PubMed] [Google Scholar]
- 28.Calvi L.M., Adams G.B., Weibrecht K.W., Weber J.M., Olson D.P., Knight M.C., Martin R.P., Schipani E., Divieti P., Bringhurst F.R., et al. Osteoblastic cells regulate the haematopoietic stem cell niche. Nature. 2003;425:841–846. doi: 10.1038/nature02040. [DOI] [PubMed] [Google Scholar]
- 29.Visnjic D., Kalajzic Z., Rowe D.W., Katavic V., Lorenzo J., Aguila H.L. Hematopoiesis is severely altered in mice with an induced osteoblast deficiency. Blood. 2004;103:3258–3264. doi: 10.1182/blood-2003-11-4011. [DOI] [PubMed] [Google Scholar]
- 30.Kiel M.J., Yilmaz O.H., Iwashita T., Yilmaz O.H., Terhorst C., Morrison S.J. SLAM family receptors distinguish hematopoietic stem and progenitor cells and reveal endothelial niches for stem cells. Cell. 2005;121:1109–1121. doi: 10.1016/j.cell.2005.05.026. [DOI] [PubMed] [Google Scholar]
- 31.Spencer J.A., Ferraro F., Roussakis E., Klein A., Wu J., Runnels J.M., Zaher W., Mortensen L.J., Alt C., Turcotte R., et al. Direct measurement of local oxygen concentration in the bone marrow of live animals. Nature. 2014;508:269–273. doi: 10.1038/nature13034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Kunisaki Y., Bruns I., Scheiermann C., Ahmed J., Pinho S., Zhang D., Mizoguchi T., Wei Q., Lucas D., Ito K., et al. Arteriolar niches maintain haematopoietic stem cell quiescence. Nature. 2013;502:637–643. doi: 10.1038/nature12612. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Asada N., Kunisaki Y., Pierce H., Wang Z., Fernandez N.F., Birbrair A., Ma’ayan A., Frenette P.S. Differential cytokine contributions of perivascular haematopoietic stem cell niches. Nat. Cell. Biol. 2017;19:214–223. doi: 10.1038/ncb3475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Acar M., Kocherlakota K.S., Murphy M.M., Peyer J.G., Oguro H., Inra C.N., Jaiyeola C., Zhao Z., Luby-Phelps K., Morrison S.J. Deep imaging of bone marrow shows non-dividing stem cells are mainly perisinusoidal. Nature. 2015;526:126–130. doi: 10.1038/nature15250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Christodoulou C., Spencer J.A., Yeh S.A., Turcotte R., Kokkaliaris K.D., Panero R., Ramos A., Guo G., Seyedhassantehrani N., Esipova T.V., et al. Live-animal imaging of native haematopoietic stem and progenitor cells. Nature. 2020;578:278–283. doi: 10.1038/s41586-020-1971-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Kokkaliaris K.D., Kunz L., Cabezas-Wallscheid N., Christodoulou C., Renders S., Camargo F., Trumpp A., Scadden D.T., Schroeder T. Adult blood stem cell localization reflects the abundance of reported bone marrow niche cell types and their combinations. Blood. 2020;136:2296–2307. doi: 10.1182/blood.2020006574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Pinho S., Marchand T., Yang E., Wei Q., Nerlov C., Frenette P.S. Lineage-Biased Hematopoietic Stem Cells Are Regulated by Distinct Niches. Dev. Cell. 2018;44:634–641.e4. doi: 10.1016/j.devcel.2018.01.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Wu Q., Zhang J., Lucas D. Anatomy of Hematopoiesis and Local Microenvironments in the Bone Marrow. Where to? Front. Immunol. 2021;12:768439. doi: 10.3389/fimmu.2021.768439. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Shimoto M., Sugiyama T., Nagasawa T. Numerous niches for hematopoietic stem cells remain empty during homeostasis. Blood. 2017;129:2124–2131. doi: 10.1182/blood-2016-09-740563. [DOI] [PubMed] [Google Scholar]
- 40.Drela K., Stanaszek L., Snioch K., Kuczynska Z., Wrobel M., Sarzynska S., Legosz P., Maldyk P., Lukomska B. Bone marrow-derived from the human femoral shaft as a new source of mesenchymal stem/stromal cells: An alternative cell material for banking and clinical transplantation. Stem Cell Res. Ther. 2020;11:262. doi: 10.1186/s13287-020-01697-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Bhat S., Viswanathan P., Chandanala S., Prasanna S.J., Seetharam R.N. Expansion and characterization of bone marrow derived human mesenchymal stromal cells in serum-free conditions. Sci. Rep. 2021;11:3403. doi: 10.1038/s41598-021-83088-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Pittenger M.F., Mackay A.M., Beck S.C., Jaiswal R.K., Douglas R., Mosca J.D., Moorman M.A., Simonetti D.W., Craig S., Marshak D.R. Multilineage potential of adult human mesenchymal stem cells. Science. 1999;284:143–147. doi: 10.1126/science.284.5411.143. [DOI] [PubMed] [Google Scholar]
- 43.Greenbaum A., Hsu Y.M., Day R.B., Schuettpelz L.G., Christopher M.J., Borgerding J.N., Nagasawa T., Link D.C. CXCL12 in early mesenchymal progenitors is required for haematopoietic stem-cell maintenance. Nature. 2013;495:227–230. doi: 10.1038/nature11926. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Pinho S., Lacombe J., Hanoun M., Mizoguchi T., Bruns I., Kunisaki Y., Frenette P.S. PDGFR and CD51 mark human nestin+ sphere-forming mesenchymal stem cells capable of hematopoietic progenitor cell expansion. J. Exp. Med. 2013;210:1351–1367. doi: 10.1084/jem.20122252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Méndez-Ferrer S., Michurina T.V., Ferraro F., Mazloom A.R., Macarthur B.D., Lira S.A., Scadden D.T., Ma’ayan A., Enikolopov G.N., Frenette P.S. Mesenchymal and haematopoietic stem cells form a unique bone marrow niche. Nature. 2010;466:829–834. doi: 10.1038/nature09262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Sugiyama T., Kohara H., Noda M., Nagasawa T. Maintenance of the hematopoietic stem cell pool by CXCL12-CXCR4 chemokine signaling in bone marrow stromal cell niches. Immunity. 2006;25:977–988. doi: 10.1016/j.immuni.2006.10.016. [DOI] [PubMed] [Google Scholar]
- 47.Ding L., Saunders T.L., Enikolopov G., Morrison S.J. Endothelial and perivascular cells maintain haematopoietic stem cells. Nature. 2012;481:457–462. doi: 10.1038/nature10783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Zhou B.O., Yue R., Murphy M.M., Peyer J.G., Morrison S.J. Leptin-receptor-expressing mesenchymal stromal cells represent the main source of bone formed by adult bone marrow. Cell Stem Cell. 2014;15:154–168. doi: 10.1016/j.stem.2014.06.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Seike M., Omatsu Y., Watanabe H., Kondoh G., Nagasawa T. Stem cell niche-specific Ebf3 maintains the bone marrow cavity. Genes Dev. 2018;32:359–372. doi: 10.1101/gad.311068.117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Omatsu Y., Sugiyama T., Kohara H., Kondoh G., Fujii N., Kohno K., Nagasawa T. The essential functions of adipo-osteogenic progenitors as the hematopoietic stem and progenitor cell niche. Immunity. 2010;33:387–399. doi: 10.1016/j.immuni.2010.08.017. [DOI] [PubMed] [Google Scholar]
- 51.Sharma M., Ross C., Srivastava S. Ally to adversary: Mesenchymal stem cells and their transformation in leukaemia. Cancer Cell Int. 2019;19:139. doi: 10.1186/s12935-019-0855-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Darvish M., Payandeh Z., Soleimanifar F., Taheri B., Soleimani M., Islami M. Umbilical cord blood mesenchymal stem cells application in hematopoietic stem cells expansion on nanofiber three-dimensional scaffold. J. Cell Biochem. 2019;120:12018–12026. doi: 10.1002/jcb.28487. [DOI] [PubMed] [Google Scholar]
- 53.Kadomatsu T., Oike Y. Roles of angiopoietin-like proteins in regulation of stem cell activity. J. Biochem. 2019;165:309–315. doi: 10.1093/jb/mvz005. [DOI] [PubMed] [Google Scholar]
- 54.Dazzi F., Ramasamy R., Glennie S., Jones S.P., Roberts I. The role of mesenchymal stem cells in haemopoiesis. Blood Rev. 2006;20:161–171. doi: 10.1016/j.blre.2005.11.002. [DOI] [PubMed] [Google Scholar]
- 55.Bhatia M., Bonnet D., Wu D., Murdoch B., Wrana J., Gallacher L., Dick J.E. Bone morphogenetic proteins regulate the developmental program of human hematopoietic stem cells. J. Exp. Med. 1999;189:1139–1148. doi: 10.1084/jem.189.7.1139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Crisan M., Kartalaei P., Vink C., Yamada-Inagawa T., Bollerot K., van IJcken W., van der Linden R., Chuva de Sousa Lopes S.M., Monteiro R., Mummery C., et al. BMP signalling differentially regulates distinct haematopoietic stem cell types. Nat. Commun. 2015;6:8040. doi: 10.1038/ncomms9040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Crisan M., Kartalaei P.S., Neagu A., Karkanpouna S., Yamada-Inagawa T., Purini C., Vink C.S., van der Linden R., van Ijcken W., Chuva de Sousa Lopes S.M., et al. BMP and Hedgehog Regulate Distinct AGM Hematopoietic Stem Cells Ex Vivo. Stem Cell Rep. 2016;6:383–395. doi: 10.1016/j.stemcr.2016.01.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Maestroni G.J.M. Adrenergic Modulation of Hematopoiesis. J. Neuroimmune Pharmacol. 2020;15:82–92. doi: 10.1007/s11481-019-09840-7. [DOI] [PubMed] [Google Scholar]
- 59.Sacchetti B., Funari A., Michienzi S., Di Cesare S., Piersanti S., Saggio I., Tagliafico E., Ferrari S., Robey P.G., Riminucci M., et al. Self-renewing osteoprogenitors in bone marrow sinusoids can organize a hematopoietic microenvironment. Cell. 2007;131:324–336. doi: 10.1016/j.cell.2007.08.025. [DOI] [PubMed] [Google Scholar]
- 60.Chan C.K.F., Chen C.-C., Luppen C.A., Kim J.-B., DeBoer A.T., Wei K., Helms J.A., Kuo C.J., Kraft D.L., Weissman I.L. Endochondral ossification is required for haematopoietic stem-cell niche formation. Nature. 2009;457:490–494. doi: 10.1038/nature07547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Man Y., Yao X., Yang T., Wang Y. Hematopoietic Stem Cell Niche During Homeostasis, Malignancy, and Bone Marrow Transplantation. Front. Cell Dev. Biol. 2021;9:621214. doi: 10.3389/fcell.2021.621214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Xu C., Gao X., Wei Q., Nakahara F., Zimmerman S.E., Mar J. Stem cell factor is selectively secreted by arterial endothelial cells in bone marrow. Nat. Commun. 2018;9:2449. doi: 10.1038/s41467-018-04726-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Hassanshahi M., Hassanshahi A., Khabbazi S., Su Y.W., Xian C.J. Bone marrow sinusoidal endothelium as a facilitator/regulator of cell egress from the bone marrow. Crit. Rev. Oncol. Hematol. 2019;137:43–56. doi: 10.1016/j.critrevonc.2019.01.024. [DOI] [PubMed] [Google Scholar]
- 64.Mitroulis I., Chen L.S., Singh R.P., Kourtzelis I., Economopoulou M., Kajikawa T., Troullinaki M., Ziogas A., Ruppova K., Hosur K., et al. Secreted proten Del-1 regulates myelopoiesis in the hematopoietic stem cell niche. J. Clin. Investig. 2017;127:3624–3639. doi: 10.1172/JCI92571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Chen L.S., Kourtzelis I., Singh R.P., Grossklaus S., Wielockx B., Hajishengallis G., Chavakis T., Mitroulis I. Endothelial cell-specific overexpression of Del-1 drives expansion of haematopoietic progenitor cells in the bone marrow. Thromb. Haemost. 2018;118:613–616. doi: 10.1055/s-0038-1624582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Butler J.M., Nolan D.J., Vertes E.L., Varnum-Finney B., Kobayashi H., Hooper A.T., Seandel M., Shido K., White I.A., Kobayashi M., et al. Endothelial cells are essential for the self-renewal and repopulation of Notch-dependent hematopoietic stem cells. Cell Stem Cell. 2010;6:251–264. doi: 10.1016/j.stem.2010.02.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Poulos M.G., Guo P., Kofler N.M., Pinho S., Gutkin M.C., Tikhonova A., Aifantis I., Frenette P.S., Kitajewski J., Rafii S., et al. Endothelial jagged-1 is necessary for homeostatic and regenerative hematopoiesis. Cell Rep. 2013;4:1022–1034. doi: 10.1016/j.celrep.2013.07.048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Hooper A.T., Butler J.M., Nolan D.J., Kranz A., Iida K., Kobayashi M., Kopp H.G., Shido K., Petit I., Yanger K., et al. Engraftment and reconstitution of hematopoiesis is dependent on VEGFR2-mediated regeneration of sinusoidal endothelial cells. Cell Stem Cell. 2009;4:263–274. doi: 10.1016/j.stem.2009.01.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Chen Q., Liu Y., Jeong H.W., Stehling M., Dinh V.V., Zhou B., Adams R.H. Apelin(+) Endothelial Niche Cells Control Hematopoiesis and Mediate Vascular Regeneration after Myeloablative Injury. Cell Stem Cell. 2019;25:768–783.e6. doi: 10.1016/j.stem.2019.10.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Doan P.L., Russell J.L., Himburg H.A., Helms K., Harris J.R., Lucas J., Holshausen K.C., Meadows S.K., Daher P., Jeffords L.B., et al. Tie2(+) bone marrow endothelial cells regulate hematopoietic stem cell regeneration following radiation injury. Stem Cells. 2013;31:327–337. doi: 10.1002/stem.1275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Plein A., Fantin A., Denti L., Pollard J.W., Ruhrberg C. Erythro-myeloid progenitors contribute endothelial cells to blood vessels. Nature. 2018;562:223–228. doi: 10.1038/s41586-018-0552-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Cary L., Noutai D., Salber R., Fadiyimu O., Gross A., Almeida-Porada G., Kidane Y., Whitnall M. Bone Marrow Endothelial Cells Influence Function and Phenotype of Hematopoietic Stem and Progenitor Cells after Mixed Neutron/Gamma Radiation. Int. J. Mol. Sci. 2019;20:1795. doi: 10.3390/ijms20071795. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Zeng L., Chen C., Song G., Yan Z., Xu S., Jia L., Ding S., Cao J., Chen W., Cheng H., et al. Infusion of endothelial progenitor cells accelerates hematopoietic and immune reconstitution, and ameliorates the graft-versus-host disease after hematopoietic stem cell transplantation. Cell Biochem. Biophys. 2012;64:213–222. doi: 10.1007/s12013-012-9387-5. [DOI] [PubMed] [Google Scholar]
- 74.Xue Y., Lv J., Zhang C., Wang L., Ma D., Liu F. The vascular niche regulates hematopoietic stem and progenitor cell lodgment and expansion via klf6a-ccl25b. Dev. Cell. 2017;42:349–362.e4. doi: 10.1016/j.devcel.2017.07.012. [DOI] [PubMed] [Google Scholar]
- 75.Tikhonova A.N., Dolgalev I., Hu H., Sivaraj K.K., Hoxha E., Cuesta-Domínguez Á., Pinho S., Akhmetzyanova I., Gao J., Witkowski M., et al. The bone marrow microenvironment at single-cell resolution. Nature. 2019;569:222–228. doi: 10.1038/s41586-019-1104-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Hosokawa K., Arai F., Yoshihara H., Iwasaki H., Hembree M., Yin T., Nakamura Y., Gomei Y., Takubo K., Shiama H., et al. Cadherin-based adhesion is a potential target for niche manipulation to protect hematopoietic stem cells in adult bone marrow. Cell Stem Cell. 2010;6:194–198. doi: 10.1016/j.stem.2009.04.013. [DOI] [PubMed] [Google Scholar]
- 77.Sugimura R., He X.C., Venkatraman A., Arai F., Box A., Semerad C., Haug J.S., Peng L., Zhong X.B., Suda T., et al. Noncanonical Wnt signaling maintains hematopoietic stem cells in the niche. Cell. 2012;150:351–365. doi: 10.1016/j.cell.2012.05.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Ding L., Morrison S.J. Haematopoietic stem cells and early lymphoid progenitors occupy distinct bone marrow niches. Nature. 2013;495:231–235. doi: 10.1038/nature11885. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Nakamura-Ishizu A., Suda T. Dynamic Changes in the Niche with N-Cadherin Revisited: The HSC “Niche Herein”. Cell Stem Cell. 2019;24:355–356. doi: 10.1016/j.stem.2019.02.007. [DOI] [PubMed] [Google Scholar]
- 80.Zhao M., Tao F., Venkatraman A., Li Z., Smith S.E., Unruh J., Chen S., Ward C., Qian P., Perry J.M., et al. N-Cadherin-Expressing Bone and Marrow Stromal Progenitor Cells Maintain Reserve Hematopoietic Stem Cells. Cell Rep. 2019;26:652–669.e6. doi: 10.1016/j.celrep.2018.12.093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Kajiume T., Kawahara Y., Yuge L., Kobayashi M. Osteoblastic adherence regulates hematopoietic stem cell self-renewal and differentiation: A conceptional in vitro and in vivo study. Stem Cell Investig. 2021;8:21. doi: 10.21037/sci-2021-019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Morrison S.J., Scadden D.T. The bone marrow niche for haematopoietic stem cells. Nature. 2014;505:327–334. doi: 10.1038/nature12984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Nilsson S.K., Johnston H.M., Whitty G.A., Williams B., Webb R.J., Denhardt D.T., Bertoncello I., Bendall L.J., Simmons P.J., Haylock D.N. Osteopontin, a key component of the hematopoietic stem cell niche and regulator of primitive hematopoietic progenitor cells. Blood. 2005;106:1232–1239. doi: 10.1182/blood-2004-11-4422. [DOI] [PubMed] [Google Scholar]
- 84.Haylock D.N., Nilsson S.K. Osteopontin: A bridge between bone and blood. Br. J. Haematol. 2006;134:467–474. doi: 10.1111/j.1365-2141.2006.06218.x. [DOI] [PubMed] [Google Scholar]
- 85.Arai F., Hirao A., Ohmura M., Sato H., Matsuoka S., Takubo K., Ito K., Koh G.Y., Suda T. Tie2/angiopoietin-1 signaling regulates hematopoietic stem cell quiescence in the bone marrow niche. Cell. 2004;118:149–161. doi: 10.1016/j.cell.2004.07.004. [DOI] [PubMed] [Google Scholar]
- 86.Zhu J., Garrett R., Jung Y., Zhang Y., Kim N., Wang J., Joe G.J., Hexner E., Choi Y., Taichman R.S., et al. Osteoblasts support B-lymphocyte commitment and differentiation from hematopoietic stem cells. Blood. 2007;109:3706–3712. doi: 10.1182/blood-2006-08-041384. [DOI] [PubMed] [Google Scholar]
- 87.Bruns I., Lucas D., Pinho S., Ahmed J., Lambert M.P., Kunisaki Y., Scheiermann C., Schiff L., Poncz M., Bergman A., et al. Megakaryocytes regulate hematopoietic stem cell quiescence through CXCL4 secretion. Nat. Med. 2014;20:1315–1320. doi: 10.1038/nm.3707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Zhao M., Perry J.M., Marshall H., Venkatraman A., Qian P., He X.C., Ahamed J., Li L. Megakaryocytes maintain homeostatic quiescence and promote post-injury regeneration of hematopoietic stem cells. Nat. Med. 2014;20:1321–1326. doi: 10.1038/nm.3706. [DOI] [PubMed] [Google Scholar]
- 89.Nakamura-Ishizu A., Takubo K., Fujioka M., Suda T. Megakaryocytes are essential for HSC quiescence through the production of thrombopoietin. Biochem. Biophys. Res. Commun. 2014;454:353–357. doi: 10.1016/j.bbrc.2014.10.095. [DOI] [PubMed] [Google Scholar]
- 90.Nakamura-Ishizu A., Takubo K., Kobayashi H., Suzuki-Inoue K., Suda T. CLEC-2 in megakaryocytes is critical for maintenance of hematopoietic stem cells in the bone marrow. J. Exp. Med. 2015;212:2133–2146. doi: 10.1084/jem.20150057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Decker M., Leslie J., Liu Q., Ding L. Hepatic thrombopoietin is required for bone marrow hematopoietic stem cell maintenance. Science. 2018;360:106–110. doi: 10.1126/science.aap8861. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Winkler I.G., Sims N.A., Pettit A.R., Barbier V., Nowlan B., Helwani F., Poulton I.J., van Rooijen N., Alexander K.A., Raggatt L.J., et al. Bone marrow macrophages maintain hematopoietic stem cell (HSC) niches and their depletion mobilizes HSCs. Blood. 2010;116:4815–4828. doi: 10.1182/blood-2009-11-253534. [DOI] [PubMed] [Google Scholar]
- 93.Chow A., Lucas D., Hidalgo A., Méndez-Ferrer S., Hashimoto D., Scheiermann C., Battista M., Leboeuf M., Prophete C., van Rooijen N., et al. Bone marrow CD169+ macrophages promote the retention of hematopoietic stem and progenitor cells in the mesenchymal stem cell niche. J. Exp. Med. 2011;208:261–271. doi: 10.1084/jem.20101688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Li D., Xue W., Li M., Dong M., Wang J., Wang X., Li X., Chen K., Zhang W., Wu S., et al. VCAM-1+ macrophages guide the homing of HSPCs to a vascular niche. Nature. 2018;564:119–124. doi: 10.1038/s41586-018-0709-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Hur J., Choi J.I., Lee H., Nham P., Kim T.W., Chae C.W., Yun J.Y., Kang J.A., Kang J., Lee S.E., et al. CD82/KAI1 Maintains the Dormancy of Long-Term Hematopoietic Stem Cells through Interaction with DARC-Expressing Macrophages. Cell Stem Cell. 2016;18:508–521. doi: 10.1016/j.stem.2016.01.013. [DOI] [PubMed] [Google Scholar]
- 96.Ludin A., Itkin T., Gur-Cohen S., Mildner A., Shezen E., Golan K., Kollet O., Kalinkovich A., Porat Z., D’Uva G., et al. Monocytes-macrophages that express α-smooth muscle actin preserve primitive hematopoietic cells in the bone marrow. Nat. Immunol. 2012;13:1072–1082. doi: 10.1038/ni.2408. [DOI] [PubMed] [Google Scholar]
- 97.May A., Forrester L.M. The erythroblastic island niche: Modeling in health, stress, and disease. Exp. Hematol. 2020;91:10–21. doi: 10.1016/j.exphem.2020.09.185. [DOI] [PubMed] [Google Scholar]
- 98.Yang C., Endoh M., Tan D.Q., Nakamura-Ishizu A., Takihara Y., Matsumura T., Suda T. Mitochondria transfer from early stages of erythroblasts to their macrophage niche via tunnelling nanotubes. Br. J. Haematol. 2021;193:1260–1274. doi: 10.1111/bjh.17531. [DOI] [PubMed] [Google Scholar]
- 99.Tratwal J., Rojas-Sutterlin S., Bataclan C., Blum S., Naveiras O. Bone marrow adiposity and the hematopoietic niche: A historical perspective of reciprocity, heterogeneity, and lineage commitment. Best Pract. Res. Clin. Endocrinol. Metab. 2021;35:101564. doi: 10.1016/j.beem.2021.101564. [DOI] [PubMed] [Google Scholar]
- 100.Naveiras O., Nardi V., Wenzel P.L., Hauschka P.V., Fahey F., Daley G.Q. Bone-marrow adipocytes as negative regulators of the haematopoietic microenvironment. Nature. 2009;460:259–263. doi: 10.1038/nature08099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Ferland-McCollough D., Maselli D., Spinetti G., Sambataro M., Sullivan N., Blom A., Madeddu P. MCP-1 Feedback Loop between Adipocytes and Mesenchymal Stromal Cells Causes Fat Accumulation and Contributes to Hematopoietic Stem Cell Rarefaction in the Bone Marrow of Patients with Diabetes. Diabetes. 2018;67:1380–1394. doi: 10.2337/db18-0044. [DOI] [PubMed] [Google Scholar]
- 102.Cuminetti V., Arranz L.J. Bone Marrow Adipocytes: The Enigmatic Components of the Hematopoietic Stem Cell Niche. J. Clin. Med. 2019;8:707. doi: 10.3390/jcm8050707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Mattiucci D., Maurizi G., Izzi V., Cenci L., Ciarlantini M., Mancini S., Mensa E., Pascarella R., Vivarelli M., Olivieri A., et al. Bone marrow adipocytes support hematopoietic stem cell survival. J. Cell. Physiol. 2018;233:1500–1511. doi: 10.1002/jcp.26037. [DOI] [PubMed] [Google Scholar]
- 104.Zhou B.O., Yu H., Yue R., Zhao Z., Rios J.J., Naveiras O., Morrison S.J. Bone marrow adipocytes promote the regeneration of stem cells and haematopoiesis by secreting SCF. Nat. Cell Biol. 2017;19:891–903. doi: 10.1038/ncb3570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Wilson A., Fu H., Schiffrin M., Winkler C., Koufany M., Jouzeau J.Y., Bonnet N., Gilardi F., Renevey F., Luther S.A., et al. Lack of Adipocytes Alters Hematopoiesis in Lipodystrophic Mice. Front. Immunol. 2018;9:2573. doi: 10.3389/fimmu.2018.02573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.DiMascio L., Voermans C., Uqoezwa M., Duncan A., Lu D., Wu J., Sankar U., Reya T. Identification of adiponectin as a novel hemopoietic stem cell growth factor. J. Immunol. 2007;178:3511–3520. doi: 10.4049/jimmunol.178.6.3511. [DOI] [PubMed] [Google Scholar]
- 107.Gainsford T., Willson T.A., Metcalf D., Handman E., McFarlane C., Ng A., Nicola N.A., Alexander W.S., Hilton D.J. Leptin can induce proliferation, differentiation, and functional activation of hemopoietic cells. Proc. Natl. Acad. Sci. USA. 1996;93:14564–14568. doi: 10.1073/pnas.93.25.14564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Umemoto Y., Tsuji K., Yang F.C., Ebihara Y., Kaneko A., Furukawa S., Nakahata T. Leptin stimulates the proliferation of murine myelocytic and primitive hematopoietic progenitor cells. Blood. 1997;90:3438–3443. doi: 10.1182/blood.V90.9.3438. [DOI] [PubMed] [Google Scholar]
- 109.Yue R., Zhou B.O., Shimada I.S., Zhao Z., Morrison S.J. Leptin Receptor Promotes Adipogenesis and Reduces Osteogenesis by Regulating Mesenchymal Stromal Cells in Adult Bone Marrow. Cell Stem Cell. 2016;18:782–796. doi: 10.1016/j.stem.2016.02.015. [DOI] [PubMed] [Google Scholar]
- 110.Fujisaki J., Wu J., Carlson A.L., Silberstein L., Putheti P., Larocca R., Gao W., Saito T.I., Lo Celso C., Tsuyuzaki H., et al. In vivo imaging of Treg cells providing immune privilege to the haematopoietic stem-cell niche. Nature. 2011;474:216–219. doi: 10.1038/nature10160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Camacho V., Matkins V.R., Patel S.B., Lever J.M., Yang Z., Ying L., Landuyt A.E., Dean E.C., George J.F., Yang H., et al. Bone marrow Tregs mediate stromal cell function and support hematopoiesis via IL-10. JCI Insight. 2020;5:e135681. doi: 10.1172/jci.insight.135681. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Hirata Y., Furuhashi K., Ishii H., Li H.W., Pinho S., Ding L., Robson S.C., Frenette P.S., Fujisaki J. CD150high Bone Marrow Tregs Maintain Hematopoietic Stem Cell Quiescence and Immune Privilege via Adenosine. Cell Stem Cell. 2018;22:445–453.e5. doi: 10.1016/j.stem.2018.01.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Kakiuchi M., Hirata Y., Robson S.C., Fujisaki J. Paradoxical Regulation of Allogeneic Bone Marrow Engraftment and Immune Privilege by Mesenchymal Cells and Adenosine. Transplant. Cell Ther. 2021;27:e1–e92. doi: 10.1016/j.bbmt.2020.09.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Schloss M.J., Hulsmans M., Rohde D., Lee I.H., Severe N., Foy B.H., Pulous F.E., Zhang S., Kokkaliaris K.D., Frodermann V., et al. B lymphocyte-derived acetylcholine limits steady-state and emergency hematopoiesis. Nat. Immunol. 2022;23:605–618. doi: 10.1038/s41590-022-01165-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Yamazaki K., Allen T.D. Ultrastructural morphometric study of efferent nerve terminals on murine bone marrow stromal cells, and the recognition of a novel anatomical unit: The “neuro-reticular complex”. Am. J. Anat. 1990;187:261–276. doi: 10.1002/aja.1001870306. [DOI] [PubMed] [Google Scholar]
- 116.Mendez-Ferrer S., Lucas D., Battista M., Frenette P.S. Haematopoietic stem cell release is regulated by circadian oscillations. Nature. 2008;452:442–447. doi: 10.1038/nature06685. [DOI] [PubMed] [Google Scholar]
- 117.Heidt T., Sager H.B., Courties G., Dutta P., Iwamoto Y., Zaltsman A., von Zur Muhlen C., Bode C., Fricchione G.L., Denninger J., et al. Chronic variable stress activates hematopoietic stem cells. Nat. Med. 2014;20:754–758. doi: 10.1038/nm.3589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.García-García A., Korn C., García-Fernández M., Domingues O., Villadiego J., Martín-Pérez D., Isern J., Bejarano-García J.A., Zimmer J., Pérez-Simón J.A., et al. Dual cholinergic signals regulate daily migration of hematopoietic stem cells and leukocytes. Blood. 2019;133:224–236. doi: 10.1182/blood-2018-08-867648. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Fielding C., García-García A., Korn C., Gadomski S., Fang Z., Reguera J.L., Pérez-Simón J.A., Göttgens B., Méndez-Ferrer S. Cholinergic signals preserve haematopoietic stem cell quiescence during regenerative haematopoiesis. Nat. Commun. 2022;13:543. doi: 10.1038/s41467-022-28175-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Wolock S.L., Krishnan I., Tenen D.E., Matkins V., Camacho V., Patel S., Agarwal P., Bhatia R., Tenen D.G., Klein A.M., et al. Mapping Distinct Bone Marrow Niche Populations and Their Differentiation Paths. Cell Rep. 2019;28:302–311.e5. doi: 10.1016/j.celrep.2019.06.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Weinreb C., Wolock S., Klein A.M. SPRING: A kinetic interface for visualizing high dimensional single-cell expression data. Bioinformatics. 2018;34:1246–1248. doi: 10.1093/bioinformatics/btx792. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Baryawno N., Przybylski D., Kowalczyk M.S., Kfoury Y., Severe N., Gustafsson K., Kokkaliaris K.D., Mercier F., Tabaka M., Hofree M., et al. A Cellular Taxonomy of the Bone Marrow Stroma in Homeostasis and Leukemia. Cell. 2019;177:1915–1932.e16. doi: 10.1016/j.cell.2019.04.040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.van der Maaten L., Hinton G. Visualizing data using t-SNE. J. Mach. Learn. Res. 2008;9:2579–2605. [Google Scholar]
- 124.Zhong L., Yao L., Tower R.J., Wei Y., Miao Z., Park J., Shrestha R., Wang L., Yu W., Holdreith N., et al. Single cell transcriptomics identifies a unique adipose lineage cell population that regulates bone marrow environment. Elife. 2020;9:e54695. doi: 10.7554/eLife.54695. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Baccin C., Al-Sabah J., Velten L., Helbling P.M., Grünschläger F., Hernández-Malmierca P., Nombela-Arrieta C., Steinmetz L.M., Trumpp A., Haas S. Combined single-cell and spatial transcriptomics reveal the molecular, cellular and spatial bone marrow niche organization. Nat. Cell Biol. 2020;22:38–48. doi: 10.1038/s41556-019-0439-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Stuart T., Butler A., Hoffman P., Hafemeister C., Papalexi E., Mauck W.M., 3rd, Hao Y., Stoeckius M., Smibert P., Satija R. Comprehensive Integration of Single-Cell Data. Cell. 2019;177:1888–1902.e21. doi: 10.1016/j.cell.2019.05.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Dolgalev I., Tikhonova A.N. Connecting the Dots: Resolving the Bone Marrow Niche Heterogeneity. Front.Cell Dev. Biol. 2021;9:622519. doi: 10.3389/fcell.2021.622519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Addo R.K., Heinrich F., Heinz G.A., Schulz D., Sercan-Alp Ö., Lehmann K., Tran C.L., Bardua M., Matz M., Löhning M., et al. Single-cell transcriptomes of murine bone marrow stromal cells reveal niche-associated heterogeneity. Eur. J. Immunol. 2019;49:1372–1379. doi: 10.1002/eji.201848053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Severe N., Karabacak N.M., Gustafsson K., Baryawno N., Courties G., Kfoury Y., Kokkaliaris K.D., Rhee C., Lee D., Scadden E.W., et al. Stress-Induced Changes in Bone Marrow Stromal Cell Populations Revealed through Single-Cell Protein Expression Mapping. Cell Stem Cell. 2019;25:570–583.e7. doi: 10.1016/j.stem.2019.06.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Wu J.Y., Purton L.E., Rodda S.J., Chen M., Weinstein L.S., McMahon A.P., Scadden D.T., Kronenberg H.M. Osteoblastic regulation of B lymphopoiesis is mediated by Gsα-dependent signaling pathways. Proc. Natl. Acad. Sci. USA. 2008;105:16976–16981. doi: 10.1073/pnas.0802898105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Comazzetto S., Murphy M.M., Berto S., Jeffery E., Zhao Z., Morrison S.J. Restricted Hematopoietic Progenitors and Erythropoiesis Require SCF from Leptin Receptor+ Niche Cells in the Bone Marrow. Cell Stem Cell. 2019;24:477–486.e6. doi: 10.1016/j.stem.2018.11.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Nombela-Arrieta C., Pivarnik G., Winkel B., Canty K.J., Harley B., Mahoney J.E., Park S.Y., Lu J., Protopopov A., Silberstein L.E. Quantitative imaging of haematopoietic stem and progenitor cell localization and hypoxic status in the bone marrow microenvironment. Nat. Cell Biol. 2013;15:533–543. doi: 10.1038/ncb2730. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Winkler I.G., Barbier V., Wadley R., Zannettino A.C., Williams S., Lévesque J.P. Positioning of bone marrow hematopoietic and stromal cells relative to blood flow in vivo: Serially reconstituting hematopoietic stem cells reside in distinct nonperfused niches. Blood. 2010;116:375–385. doi: 10.1182/blood-2009-07-233437. [DOI] [PubMed] [Google Scholar]
- 134.Balzano M., De Grandis M., Vu Manh T.P., Chasson L., Bardin F., Farina A., Sergé A., Bidaut G., Charbord P., Hérault L., et al. Nidogen-1 Contributes to the Interaction Network Involved in Pro-B Cell Retention in the Peri-sinusoidal Hematopoietic Stem Cell Niche. Cell Rep. 2019;26:3257–3271.e8. doi: 10.1016/j.celrep.2019.02.065. [DOI] [PubMed] [Google Scholar]
- 135.Ito K., Ito K. Metabolism and the control of cell fate decisions and stem cell renewal. Annu. Rev. Cell. Dev. Biol. 2016;32:399–409. doi: 10.1146/annurev-cellbio-111315-125134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Suda T., Takubo K., Semenza G.L. Metabolic regulation of hematopoietic stem cells in the hypoxic niche. Cell Stem Cell. 2011;9:298–310. doi: 10.1016/j.stem.2011.09.010. [DOI] [PubMed] [Google Scholar]
- 137.Rossi D.J., Jamieson C.H., Weissman I.L. Stems cells and the pathways to aging and cancer. Cell. 2008;132:681–696. doi: 10.1016/j.cell.2008.01.036. [DOI] [PubMed] [Google Scholar]
- 138.Zuo L., Zhou T., Pannell B.K., Ziegler A.C., Best T.M. Biological and physiological role of reactive oxygen species--the good, the bad and the ugly. Acta Physiol. 2015;214:329–348. doi: 10.1111/apha.12515. [DOI] [PubMed] [Google Scholar]
- 139.Parmar K., Mauch P., Vergilio J.A., Sackstein R., Down J.D. Distribution of hematopoietic stem cells in the bone marrow according to regional hypoxia. Proc. Natl. Acad. Sci. USA. 2007;104:5431–5436. doi: 10.1073/pnas.0701152104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Simsek T., Kocabas F., Zheng J., Deberardinis R.J., Mahmoud A.I., Olson E.N., Schneider J.W., Zhang C.C., Sadek H.A. The distinct metabolic profile of hematopoietic stem cells reflects their location in a hypoxic niche. Cell Stem Cell. 2010;7:380–390. doi: 10.1016/j.stem.2010.07.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Klimmeck D., Hansson J., Raffel S., Vakhrushev S.Y., Trumpp A., Krijgsveld J. Proteomic cornerstones of hematopoietic stem cell differentiation: Distinct signatures of multipotent progenitors and myeloid committed cells. Mol. Cell Proteom. 2012;11:286–302. doi: 10.1074/mcp.M111.016790. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Takubo K., Nagamatsu G., Kobayashi C.I., Nakamura-Ishizu A., Kobayashi H., Ikeda E., Goda N., Rahimi Y., Johnson R.S., Soga T., et al. Regulation of glycolysis by Pdk functions as a metabolic checkpoint for cell cycle quiescence in hematopoietic stem cells. Cell Stem Cell. 2012;12:49–61. doi: 10.1016/j.stem.2012.10.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Cipolleschi M.G., Dello Sbarba P., Olivotto M. The role of hypoxia in the maintenance of hematopoietic stem cells. Blood. 1993;82:2031–2037. doi: 10.1182/blood.V82.7.2031.2031. [DOI] [PubMed] [Google Scholar]
- 144.Ivanovic Z., Hermitte F., de la Grange P.B., Dazey B., Belloc F., Lacombe F., Vezon G., Praloran V. Simultaneous maintenance of human cord blood SCID-repopulating cells and expansion of committed progenitors at low O2 concentration (3%) Stem Cells. 2004;22:716–724. doi: 10.1634/stemcells.22-5-716. [DOI] [PubMed] [Google Scholar]
- 145.Hermitte F., de la Grange P.B., Belloc F., Praloran V., Ivanovic Z. Very low O2 concentration (0.1%) favors G0 return of dividing CD34+ cells. Stem Cells. 2006;24:65–73. doi: 10.1634/stemcells.2004-0351. [DOI] [PubMed] [Google Scholar]
- 146.Shima H., Takubo K., Tago N., Iwasaki H., Arai F., Takahashi T., Suda T. Acquisition of G0 state by CD34-positive cord blood cells after bone marrow transplantation. Exp. Hematol. 2010;38:1231–1240. doi: 10.1016/j.exphem.2010.08.004. [DOI] [PubMed] [Google Scholar]
- 147.Juntilla M.M., Patil V.D., Calamito M., Joshi R.P., Birnbaum M.J., Koretzky G.A. AKT1 and AKT2 maintain hematopoietic stem cell function by regulating reactive oxygen species. Blood. 2010;115:4030–4038. doi: 10.1182/blood-2009-09-241000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Ito K., Hirao A., Arai F., Matsuoka S., Takubo K., Hamaguchi I., Nomiyama K., Hosokawa K., Sakurada K., Nakagata N., et al. Regulation of oxidative stress by ATM is required for self-renewal of haematopoietic stem cells. Nature. 2004;431:997–1002. doi: 10.1038/nature02989. [DOI] [PubMed] [Google Scholar]
- 149.Miyamoto K., Araki K.Y., Naka K., Arai F., Takubo K., Yamazaki S., Matsuoka S., Miyamoto T., Ito K., Ohmura M., et al. Foxo3a is essential for maintenance of the hematopoietic stem cell pool. Cell Stem Cell. 2007;1:101–112. doi: 10.1016/j.stem.2007.02.001. [DOI] [PubMed] [Google Scholar]
- 150.Tothova Z., Kollipara R., Huntly B.J., Lee B.H., Castrillon D.H., Cullen D.E., McDowell E.P., Lazo-Kallanian S., Williams I.R., Sears C., et al. FoxOs are critical mediators of hematopoietic stem cell resistance to physiologic oxidative stress. Cell. 2007;128:325–339. doi: 10.1016/j.cell.2007.01.003. [DOI] [PubMed] [Google Scholar]
- 151.Owusu-Ansah E., Banerjee U. Reactive oxygen species prime Drosophila haematopoietic progenitors for differentiation. Nature. 2009;461:537–541. doi: 10.1038/nature08313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Mistry J.J., Hellmich C., Moore J.A., Jibril A., Macaulay I., Moreno-Gonzalez M., Di Palma F., Beraza N., Bowles K.M., Rushworth S.A. Free fatty-acid transport via CD36 drives β-oxidation-mediated hematopoietic stem cell response to infection. Nat. Commun. 2021;12:7130. doi: 10.1038/s41467-021-27460-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Ueda T., Nagamachi A., Takubo K., Yamasaki N., Matsui H., Kanai A., Nakata Y., Ikeda K., Konuma T., Oda H., et al. Fbxl10 overexpression in murine hematopoietic stem cells induces leukemia involving metabolic activation and upregulation of Nsg2. Blood. 2015;125:3437–3446. doi: 10.1182/blood-2014-03-562694. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Maryanovich M., Zaltsman Y., Ruggiero A., Goldman A., Shachnai L., Zaidman S.L., Porat Z., Golan K., Lapidot T., Gross A. An MTCH2 pathway repressing mitochondria metabolism regulates haematopoietic stem cell fate. Nat. Commun. 2015;6:7901. doi: 10.1038/ncomms8901. [DOI] [PubMed] [Google Scholar]
- 155.Luchsinger L.L., de Almeida M.J., Corrigan D.J., Mumau M., Snoeck H.W. Mitofusin 2 maintains haematopoietic stem cells with extensive lymphoid potential. Nature. 2016;529:528–531. doi: 10.1038/nature16500. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Vannini N., Girotra M., Naveiras O., Nikitin G., Campos V., Giger S., Roch A., Auwerx J., Lutolf M.P. Specification of haematopoietic stem cell fate via modulation of mitochondrial activity. Nat. Commun. 2016;7:13125. doi: 10.1038/ncomms13125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Karigane D.L., Kobayashi H., Morikawa T., Ootomo Y., Sakai M., Nagamatsu G., Kubota Y., Goda N., Matsumoto M., Nishimura E.K., et al. p38α Activates Purine Metabolism to Initiate Hematopoietic Stem/Progenitor Cell Cycling in Response to Stress. Cell Stem Cell. 2016;19:192–204. doi: 10.1016/j.stem.2016.05.013. [DOI] [PubMed] [Google Scholar]
- 158.Ito K., Carracedo A., Weiss D., Arai F., Ala U., Avigan D.E., Schafer Z.T., Evans R.M., Suda T., Lee C.H., et al. A PML–PPARδ pathway for fatty acid oxidation regulates hematopoietic stem cell maintenance. Nat. Med. 2012;18:1350–1358. doi: 10.1038/nm.2882. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Ito K., Turcotte R., Cui J., Zimmerman S.E., Pinho S., Mizoguchi T., Arai F., Runnels J.M., Alt C., Teruya-Feldstein J., et al. Self-renewal of a purified Tie2+ hematopoietic stem cell population relies on mitochondrial clearance. Science. 2016;354:1156–1160. doi: 10.1126/science.aaf5530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Yu W.M., Liu X., Shen J., Jovanovic O., Pohl E.E., Gerson S.L., Finkel T., Broxmeyer H.E., Qu C.K. Metabolic regulation by the mitochondrial phosphatase PTPMT1 is required for hematopoietic stem cell differentiation. Cell Stem Cell. 2013;12:62–74. doi: 10.1016/j.stem.2012.11.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Warr M.R., Binnewies M., Flach J., Reynaud D., Garg T., Malhotra R., Debnath J., Passegué E. FOXO3A directs a protective autophagy program in haematopoietic stem cells. Nature. 2013;494:323–327. doi: 10.1038/nature11895. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Ho T.T., Warr M.R., Adelman E.R., Lansinger O.M., Flach J., Verovskaya E.V., Figueroa M.E., Passegué E. Autophagy maintains the metabolism and function of young and old stem cells. Nature. 2017;543:205–210. doi: 10.1038/nature21388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Jin G., Xu C., Zhang X., Long J., Rezaeian A.H., Liu C., Furth M.E., Kridel S., Pasche B., Bian X.W., et al. Atad3a suppresses Pink1-dependent mitophagy to maintain homeostasis of hematopoietic progenitor cells. Nat. Immunol. 2018;19:29–40. doi: 10.1038/s41590-017-0002-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Mohrin M., Shin J., Liu Y., Brown K.L., Luo H., Xi Y., Haynes C.M., Chen D. Stem cell aging. A mitochondrial UPR-mediated metabolic checkpoint regulates hematopoietic stem cell aging. Science. 2015;347:1374–1377. doi: 10.1126/science.aaa2361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Mohrin M., Widjaja A., Liu Y., Luo H., Chen D. The mitochondrial unfolded protein response is activated upon hematopoietic stem cell exit from quiescence. Aging Cell. 2018;17:e12756. doi: 10.1111/acel.12756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Gurumurthy S., Xie S.Z., Alagesan B., Kim J., Yusuf R.Z., Saez B., Tzatsos A., Ozsolak F., Milos P., Ferrari F., et al. The Lkb1 metabolic sensor maintains haematopoietic stem cell survival. Nature. 2010;468:659–663. doi: 10.1038/nature09572. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Nakada D., Saunders T.L., Morrison S.J. Lkb1 regulates cell cycle and energy metabolism in haematopoietic stem cells. Nature. 2010;468:653–658. doi: 10.1038/nature09571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Chen C., Liu Y., Liu R., Ikenoue T., Guan K.L., Liu Y., Zheng P. TSC-mTOR maintains quiescence and function of hematopoietic stem cells by repressing mitochondrial biogenesis and reactive oxygen species. J. Exp. Med. 2008;205:2397–2408. doi: 10.1084/jem.20081297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Chen C.T., Shih Y.R., Kuo T.K., Lee O.K., Wei Y.H. Coordinated changes of mitochondrial biogenesis and antioxidant enzymes during osteogenic differentiation of human mesenchymal stem cells. Stem Cells. 2008;26:960–968. doi: 10.1634/stemcells.2007-0509. [DOI] [PubMed] [Google Scholar]
- 170.Pattappa G., Thorpe S.D., Jegard N.C., Heywood H.K., de Bruijn J.D., Lee D.A. Continuous and uninterrupted oxygen tension influences the colony formation and oxidative metabolism of human mesenchymal stem cells. Tissue Eng. Part C Methods. 2013;19:68–79. doi: 10.1089/ten.tec.2011.0734. [DOI] [PubMed] [Google Scholar]
- 171.Gomzikova M.O., James V., Rizvanov A.A. Mitochondria Donation by Mesenchymal Stem Cells: Current Understanding and Mitochondria Transplantation Strategies. Front. Cell Dev. Biol. 2021;9:653322. doi: 10.3389/fcell.2021.653322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Mahrouf-Yorgov M., Augeul L., Da Silva C.C., Jourdan M., Rigolet M., Manin S., Ferrera R., Ovize M., Henry A., Guguin A., et al. Mesenchymal stem cells sense mitochondria released from damaged cells as danger signals to activate their rescue properties. Cell Death Differ. 2017;24:1224–1238. doi: 10.1038/cdd.2017.51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.Golan K., Singh A.K., Kollet O., Bertagna M., Althoff M.J., Khatib-Massalha E., Petrovich-Kopitman E., Wellendorf A.M., Massalha H., Levin-Zaidman S., et al. Bone marrow regeneration requires mitochondrial transfer from donor Cx43-expressing hematopoietic progenitors to stroma. Blood. 2020;136:2607–2619. doi: 10.1182/blood.2020005399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Kandarakov O., Belyavsky A. Clonal Hematopoiesis, Cardiovascular Diseases and Hematopoietic Stem Cells. Int. J. Mol. Sci. 2020;21:7902. doi: 10.3390/ijms21217902. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Yahata T., Takanashi T., Muguruma Y., Ibrahim A.A., Matsuzawa H., Uno T., Sheng Y., Onizuka M., Ito M., Kato S., et al. Accumulation of oxidative DNA damage restricts the self-renewal capacity of human hematopoietic stem cells. Blood. 2011;118:2941–2950. doi: 10.1182/blood-2011-01-330050. [DOI] [PubMed] [Google Scholar]
- 176.Ito K., Hirao A., Arai F., Takubo K., Matsuoka S., Miyamoto K., Ohmura M., Naka K., Hosokawa K., Ikeda Y., et al. Reactive oxygen species act through p38 MAPK to limit the lifespan of hematopoietic stem cells. Nat. Med. 2006;12:446–451. doi: 10.1038/nm1388. [DOI] [PubMed] [Google Scholar]
- 177.Sudo K., Ema H., Morita Y., Nakauchi H. Age-associated characteristics of murine hematopoietic stem cells. J. Exp. Med. 2000;192:1273–1280. doi: 10.1084/jem.192.9.1273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Liang Y., Van Zant G., Szilvassy S.J. Effects of aging on the homing and engraftment of murine hematopoietic stem and progenitor cells. Blood. 2005;106:1479–1487. doi: 10.1182/blood-2004-11-4282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Holstege H., Pfeiffer W., Sie D., Hulsman M., Nicholas T.J., Lee C.C., Ross T., Lin J., Miller M.A., Ylstra B., et al. Somatic mutations found in the healthy blood compartment of a 115-yr-old woman demonstrate oligoclonal hematopoiesis. Genome Res. 2014;24:733–742. doi: 10.1101/gr.162131.113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Thongon N., Ma F., Santoni A., Marchesini M., Fiorini E., Rose A., Adema V., Ganan-Gomez I., Groarke E.M., Gutierrez-Rodrigues F., et al. Hematopoiesis under telomere attrition at the single-cell resolution. Nat. Commun. 2021;12:6850. doi: 10.1038/s41467-021-27206-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181.Genovese G., Kähler A.K., Handsaker R.E., Lindberg J., Rose S.A., Bakhoum S.F., Chambert K., Mick E., Neale B.M., Fromer M., et al. Clonal hematopoiesis and blood-cancer risk inferred from blood DNA sequence. N. Engl. J. Med. 2014;371:2477–2487. doi: 10.1056/NEJMoa1409405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182.Jaiswal S., Fontanillas P., Flannick J., Manning A., Grauman P.V., Mar B.G., Lindsley R.C., Mermel C.H., Burtt N., Chavez A., et al. Age-related clonal hematopoiesis associated with adverse outcomes. N. Engl. J. Med. 2014;371:2488–2498. doi: 10.1056/NEJMoa1408617. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Qian P., He X.C., Paulson A., Li Z., Tao F., Perry J.M., Guo F., Zhao M., Zhi L., Venkatraman A., et al. The Dlk1-Gtl2 locus preserves LT-HSC function by inhibiting the PI3K-mTOR pathway to restrict mitochondrial metabolism. Cell Stem Cell. 2016;18:214–228. doi: 10.1016/j.stem.2015.11.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.Dong S., Wang Q., Kao Y.R., Diaz A., Tasset I., Kaushik S., Thiruthuvanathan V., Zintiridou A., Nieves E., Dzieciatkowska M., et al. Chaperone-mediated autophagy sustains haematopoietic stem-cell function. Nature. 2021;591:117–123. doi: 10.1038/s41586-020-03129-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 185.Grigoryan A., Guidi N., Senger K., Liehr T., Soller K., Marka G., Vollmer A., Markaki Y., Leonhardt H., Buske C., et al. LaminA/C regulates epigenetic and chromatin architecture changes upon aging of hematopoietic stem cells. Genome Biol. 2018;19:189. doi: 10.1186/s13059-018-1557-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Adelman E.R., Huang H.T., Roisman A., Olsson A., Colaprico A., Qin T., Lindsley R.C., Bejar R., Salomonis N., Grimes H.L., et al. Aging Human Hematopoietic Stem Cells Manifest Profound Epigenetic Reprogramming of Enhancers That May Predispose to Leukemia. Cancer Discov. 2019;9:1080–1101. doi: 10.1158/2159-8290.CD-18-1474. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Florian M.C., Dörr K., Niebel A., Daria D., Schrezenmeier H., Rojewski M., Filippi M.D., Hasenberg A., Gunzer M., Scharffetter-Kochanek K., et al. Cdc42 activity regulates hematopoietic stem cell aging and rejuvenation. Cell Stem Cell. 2012;10:520–530. doi: 10.1016/j.stem.2012.04.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188.Sacma M., Pospiech J., Bogeska R., de Back W., Mallm J.P., Sakk V., Soller K., Marka G., Vollmer A., Karns R., et al. Haematopoietic stem cells in perisinusoidal niches are protected from ageing. Nat. Cell. Biol. 2019;21:1309–1320. doi: 10.1038/s41556-019-0418-y. [DOI] [PubMed] [Google Scholar]
- 189.Blokzijl F., de Ligt J., Jager M., Sasselli V., Roerink S., Sasaki N., Huch M., Boymans S., Kuijk E., Prins P., et al. Tissue-specific mutation accumulation in human adult stem cells during life. Nature. 2016;538:260–264. doi: 10.1038/nature19768. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Stolzing A., Jones E., McGonagle D., Scutt A. Age-related changes in human bone marrow-derived mesenchymal stem cells: Consequences for cell therapies. Mech. Ageing Dev. 2008;129:163–173. doi: 10.1016/j.mad.2007.12.002. [DOI] [PubMed] [Google Scholar]
- 191.Ganguly P., El-Jawhari J.J., Burska A.N., Ponchel F., Giannoudis P.V., Jones E.A. The analysis of in vivo aging in human bone marrow mesenchymal stromal cells using colony-forming unit-fibroblast assay and the CD45(low)CD271(+) Phenotype. Stem Cells Int. 2019;2019:5197983. doi: 10.1155/2019/5197983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Zhang D.Y., Wang H.J., Tan Y.Z. Wnt/beta-catenin signaling induces the aging of mesenchymal stem cells through the DNA damage response and the p53/p21 pathway. PLoS ONE. 2011;6:e21397. doi: 10.1371/journal.pone.0021397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193.Liu L., DiGirolamo C.M., Navarro P.A., Blasco M.A., Keefe D.L. Telomerase deficiency impairs differentiation of mesenchymal stem cells. Exp. Cell Res. 2004;294:1–8. doi: 10.1016/j.yexcr.2003.10.031. [DOI] [PubMed] [Google Scholar]
- 194.Ju Z., Jiang H., Jaworski M., Rathinam C., Gompf A., Klein C., Trumpp A., Rudolph K.L. Telomere dysfunction induces environmental alterations limiting hematopoietic stem cell function and engraftment. Nat. Med. 2007;13:742–747. doi: 10.1038/nm1578. [DOI] [PubMed] [Google Scholar]
- 195.Yang M., Lin J., Tang J., Chen Z., Qian X., Gao W.Q., Xu H. Decreased immunomodulatory and secretory capability of aging human umbilical cord mesenchymal stem cells in vitro. Biochem. Biophys. Res. Commun. 2020;525:633–638. doi: 10.1016/j.bbrc.2020.02.125. [DOI] [PubMed] [Google Scholar]
- 196.Gnani D., Crippa S., Volpe L.D., Rossella V., Conti A., Lettera E., Rivis S., Ometti M., Fraschini G., Bernardo M.E., et al. An early-senescence state in aged mesenchymal stromal cells contributes to hematopoietic stem and progenitor cell clonogenic impairment through the activation of a pro-inflammatory program. Aging Cell. 2019;18:e12933. doi: 10.1111/acel.12933. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197.Maryanovich M., Zahalka A.H., Pierce H., Pinho S., Nakahara F., Asada N., Wei Q., Wang X., Ciero P., Xu J., et al. Adrenergic nerve degeneration in bone marrow drives aging of the hematopoietic stem cell niche. Nat. Med. 2018;24:782–791. doi: 10.1038/s41591-018-0030-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 198.Kusumbe A.P., Ramasamy S.K., Itkin T., Mäe M.A., Langen U.H., Betsholtz C., Lapidot T., Adams R.H. Age-Dependent Modulation of Vascular Niches for Haematopoietic Stem Cells. Nature. 2016;532:380–384. doi: 10.1038/nature17638. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199.Renders S., Svendsen A.F., Panten J., Rama N., Maryanovich M., Sommerkamp P., Ladel L., Redavid A.R., Gibert B., Lazare S., et al. Niche Derived Netrin-1 Regulates Hematopoietic Stem Cell Dormancy via its Receptor Neogenin-1. Nat. Commun. 2021;12:608. doi: 10.1038/s41467-020-20801-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 200.Ramalingam P., Poulos M.G., Gutkin M.C., Katsnelson L., Freire A.G., Lazzari E., Butler J.M. Endothelial mTOR Maintains Hematopoiesis During Aging. J. Exp. Med. 2020;217:e20191212. doi: 10.1084/jem.20191212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201.Szade K., Zukowska M., Szade A., Nowak W., Skulimowska I., Ciesla M., Bukowska-Strakova K., Gulati G.C., Kachamakova-Trojanowska N., Kusienicka A., et al. Heme Oxygenase-1 Deficiency Triggers Exhaustion of Hematopoietic Stem Cells. EMBO Rep. 2020;21:e47895. doi: 10.15252/embr.201947895. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 202.Frisch B.J., Hoffman C.M., Latchney S.E., LaMere M.W., Myers J., Ashton J., Li A.J., Saunders J., 2nd, Palis J., Perkins A.S., et al. Aged marrow macrophages expand platelet-biased hematopoietic stem cells via Interleukin1B. JCI Insight. 2019;5:e124213. doi: 10.1172/jci.insight.124213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.Fulzele K., Krause D.S., Panaroni C., Saini V., Barry K.J., Liu X., Lotinun S., Baron R., Bonewald L., Feng J.Q., et al. Myelopoiesis is regulated by osteocytes through Gsα-dependent signaling. Blood. 2013;121:930–939. doi: 10.1182/blood-2012-06-437160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 204.Pioli P.D., Casero D., Montecino-Rodriguez E., Morrison S.L., Dorshkind K. Plasma cells are obligate effectors of enhanced myelopoiesis in aging bone marrow. Immunity. 2019;51:351–366.e6. doi: 10.1016/j.immuni.2019.06.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 205.Ambrosi T.H., Scialdone A., Graja A., Gohlke S., Jank A.M., Bocian C., Woelk L., Fan H., Logan D.W., Schürmann A., et al. Adipocyte accumulation in the bone marrow during obesity and aging impairs stem cell-based hematopoietic and bone regeneration. Cell Stem Cell. 2017;20:771–784. doi: 10.1016/j.stem.2017.02.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 206.Badimon L., Cubedo J. Adipose tissue depots and inflammation: Effects on plasticity and resident mesenchymal stem cell function. Cardiovasc. Res. 2017;113:1064–1073. doi: 10.1093/cvr/cvx096. [DOI] [PubMed] [Google Scholar]
- 207.Tuljapurkar S.R., McGuire T.R., Brusnahan S.K., Jackson J.D., Garvin K.L., Kessinger M.A., Lane J.T., O’Kane B.J., Sharp J.G. Changes in Human Bone Marrow Fat Content Associated With Changes in Hematopoietic Stem Cell Numbers and Cytokine Levels With Aging. J. Anat. 2011;219:574–581. doi: 10.1111/j.1469-7580.2011.01423.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 208.Ho Y.-H., Del Toro R., Rivera-Torres J., Rak J., Korn C., Garcıa-Garc A., Macías D., González-Gómez C., Del Monte A., Wittner M., et al. Remodeling of Bone Marrow Hematopoietic Stem Cell Niches Promotes Myeloid Cell Expansion During Premature or Physiological Aging. Cell Stem Cell. 2019;25:407–418.e6. doi: 10.1016/j.stem.2019.06.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 209.Kovtonyuk L.V., Fritsch K., Feng X., Manz M.G., Takizawa H. Inflamm-Aging of Hematopoiesis, Hematopoietic Stem Cells, and the Bone Marrow Microenvironment. Front. Immunol. 2016;7:502. doi: 10.3389/fimmu.2016.00502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 210.Helbling P.M., Piñeiro-Yáñez E., Gerosa R., Boettcher S., Al-Shahrour F., Manz M.G., Nombela-Arrieta C. Global Transcriptomic Profiling of the Bone Marrow Stromal Microenvironment During Postnatal Development, Aging, and Inflammation. Cell Rep. 2019;29:3313–3330.e4. doi: 10.1016/j.celrep.2019.11.004. [DOI] [PubMed] [Google Scholar]
- 211.Zambetti N.A., Ping Z., Chen S., Kenswil K.J.G., Mylona M.A., Sanders M.A., Hoogenboezem R.M., Bindels E.M.G., Adisty M.N., Van Strien P.M.H., et al. Mesenchymal inflammation drives genotoxic stress in hematopoietic stem cells and predicts disease evolution in human pre-leukemia. Cell Stem Cell. 2016;19:613–627. doi: 10.1016/j.stem.2016.08.021. [DOI] [PubMed] [Google Scholar]
- 212.Dong L., Yu W.M., Zheng H., Loh M.L., Bunting S.T., Pauly M., Huang G., Zhou M., Broxmeyer H.E., Scadden D.T., et al. Leukaemogenic effects of Ptpn11 activating mutations in the stem cell microenvironment. Nature. 2016;539:304–308. doi: 10.1038/nature20131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 213.Hellmich C., Moore J.A., Bowles K.M., Rushworth S.A. Bone Marrow Senescence and the Microenvironment of Hematological Malignancies. Front. Oncol. 2020;10:230. doi: 10.3389/fonc.2020.00230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 214.Ho T.T., Dellorusso P.V., Verovskaya E.V., Bakker S.T., Flach J., Smith L.K., Ventura P.B., Lansinger O.M., Hérault A., Zhang S.Y., et al. Aged hematopoietic stem cells are refractory to bloodborne systemic rejuvenation interventions. J. Exp. Med. 2021;218:e20210223. doi: 10.1084/jem.20210223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 215.Kuribayashi W., Oshima M., Itokawa N., Koide S., Nakajima-Takagi Y., Yamashita M., Yamazaki S., Rahmutulla B., Miura F., Ito T., et al. Limited rejuvenation of aged hematopoietic stem cells in young bone marrow niche. J. Exp. Med. 2021;218:e20192283. doi: 10.1084/jem.20192283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 216.Guidi N., Sacma M., Ständker L., Soller K., Marka G., Eiwen K., Weiss J.M., Kirchhoff F., Weil T., Cancelas J.A., et al. Osteopontin Attenuates Aging-Associated Phenotypes of Hematopoietic Stem Cells. EMBO J. 2017;36:840–853. doi: 10.15252/embj.201694969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 217.Ergen A.V., Boles N.C., Goodell M.A. Rantes/Ccl5 influences hematopoietic stem cell subtypes and causes myeloid skewing. Blood. 2012;119:2500–2509. doi: 10.1182/blood-2011-11-391730. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 218.Janel A., Dubois-Galopin F., Bourgne C., Berger J., Tarte K., Boiret-Dupré N., Boisgard S., Verrelle P., Déchelotte P., Tournilhac O., et al. The chronic lymphocytic leukemia clone disrupts the bone marrow microenvironment. Stem Cells Dev. 2014;23:2972–2982. doi: 10.1089/scd.2014.0229. [DOI] [PubMed] [Google Scholar]
- 219.Desbourdes L., Javary J., Charbonnier T., Ishac N., Bourgeais J., Iltis A., Chomel J.C., Turhan A., Guilloton F., Tarte K., et al. Alteration Analysis of Bone Marrow Mesenchymal Stromal Cells from De Novo Acute Myeloid Leukemia Patients at Diagnosis. Stem Cells Dev. 2017;26:709–722. doi: 10.1089/scd.2016.0295. [DOI] [PubMed] [Google Scholar]
- 220.Vanegas N.P., Vernot J.P. Loss of quiescence and self-renewal capacity of hematopoietic stem cell in an in vitro leukemic niche. Exp. Hematol. Oncol. 2017;6:2. doi: 10.1186/s40164-016-0062-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 221.Negaard H.F., Iversen N., Bowitz-Lothe I.M., Sandset P.M., Steinsvik B., Ostenstad B., Iversen P.O. Increased bone marrow microvascular density in haematological malignancies is associated with differential regulation of angiogenic factors. Leukemia. 2009;23:162–169. doi: 10.1038/leu.2008.255. [DOI] [PubMed] [Google Scholar]
- 222.Dias S., Shmelkov S.V., Lam G., Rafii S. VEGF(165) promotes survival of leukemic cells by Hsp90-mediated induction of Bcl-2 expression and apoptosis inhibition. Blood. 2002;99:2532–2540. doi: 10.1182/blood.V99.7.2532. [DOI] [PubMed] [Google Scholar]
- 223.Katoh O., Takahashi T., Oguri T., Kuramoto K., Mihara K., Kobayashi M., Hirata S., Watanabe H. Vascular endothelial growth factor inhibits apoptotic death in hematopoietic cells after exposure to chemotherapeutic drugs by inducing MCL1 acting as an antiapoptotic factor. Can. Res. 1998;58:5565–5569. [PubMed] [Google Scholar]
- 224.Hatfield K., Ryningen A., Corbascio M., Bruserud O. Microvascular endothelial cells increase proliferation and inhibit apoptosis of native human acute myelogenous leukemia blasts. Int. J. Cancer. 2006;119:2313–2321. doi: 10.1002/ijc.22180. [DOI] [PubMed] [Google Scholar]
- 225.Lu W., Weng W., Zhu Q., Zhai Y., Wan Y., Liu H., Yang S., Yu Y., Wei Y., Shi J. Small bone marrow adipocytes predict poor prognosis in acute myeloid leukemia. Haematologica. 2018;103:e21–e24. doi: 10.3324/haematol.2017.173492. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 226.Shafat M.S., Oellerich T., Mohr S., Robinson S.D., Edwards D.R., Marlein C.R., Piddock R.E., Fenech M., Zaitseva L., Abdul-Aziz A., et al. Leukemic blasts program bone marrow adipocytes to generate a protumoral microenvironment. Blood. 2017;129:1320–1332. doi: 10.1182/blood-2016-08-734798. [DOI] [PubMed] [Google Scholar]
- 227.Lu W., Wan Y., Li Z., Zhu B., Yin C., Liu H., Yang S., Zhai Y., Yu Y., Wei Y., et al. Growth differentiation factor 15 contributes to marrow adipocyte remodeling in response to the growth of leukemic cells. J. Exp. Clin. Cancer Res. 2018;37:66. doi: 10.1186/s13046-018-0738-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 228.Chrysovergis K., Wang X., Kosak J., Lee S.H., Kim J.S., Foley J.F., Travlos G., Singh S., Baek S.J., Eling T.E. NAG-1/GDF-15 prevents obesity by increasing thermogenesis, lipolysis and oxidative metabolism. Int. J. Obes. 2014;38:1555–1564. doi: 10.1038/ijo.2014.27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 229.Boyd A.L., Reid J.C., Salci K.R., Aslostovar L., Benoit Y.D., Shapovalova Z., Nakanishi M., Porras D.P., Almakadi M., Campbell C.J.V., et al. Acute myeloid leukaemia disrupts endogenous myelo-erythropoiesis by compromising the adipocyte bone marrow niche. Nat. Cell Biol. 2017;19:1336–1347. doi: 10.1038/ncb3625. [DOI] [PubMed] [Google Scholar]
- 230.Hanoun M., Zhang D., Mizoguchi T., Pinho S., Pierce H., Kunisaki Y., Lacombe J., Armstrong S.A., Duhrsen U., Frenette P.S. Acute myelogenous leukemia-induced sympathetic neuropathy promotes malignancy in an altered hematopoietic stem cell niche. Cell Stem Cell. 2014;15:365–375. doi: 10.1016/j.stem.2014.06.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 231.Sipkins D.A., Wei X., Wu J.W., Runnels J.M., Côté D., Means T.K., Luster A.D., Scadden D.T., Lin C.P. In vivo imaging of specialized bone marrow endothelial microdomains for tumour engraftment. Nature. 2005;435:969–973. doi: 10.1038/nature03703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 232.Pitt L.A., Tikhonova A.N., Hu H., Trimarchi T., King B., Gong Y., Sanchez-Martin M., Tsirigos A., Littman D.R., Ferrando A.A., et al. CXCL12-producing vascular endothelial niches control acute T cell leukemia maintenance. Cancer Cell. 2015;27:755–768. doi: 10.1016/j.ccell.2015.05.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 233.Melo S.A., Sugimoto H., O’Connell J.T., Kato N., Villanueva A., Vidal A., Qiu L., Vitkin E., Perelman L.T., Melo C.A., et al. Cancer exosomes perform cell-independent microRNA biogenesis and promote tumorigenesis. Cancer Cell. 2014;26:707–721. doi: 10.1016/j.ccell.2014.09.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 234.Hoshino A., Costa-Silva B., Shen T.L., Rodrigues G., Hashimoto A., Tesic Mark M., Molina H., Kohsaka S., Di Giannatale A., Ceder S., et al. Tumour exosome integrins determine organotropic metastasis. Nature. 2015;527:329–335. doi: 10.1038/nature15756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 235.Javidi-Sharifi N., Martinez J., English I., Joshi S.K., Scopim-Ribeiro R., Viola S.K., Edwards D.K.T., Agarwal A., Lopez C., Jorgens D., et al. FGF2-FGFR1 signaling regulates release of leukemia-protective exosomes from bone marrow stromal cells. Elife. 2019;8:e40033. doi: 10.7554/eLife.40033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 236.Corrado C., Raimondo S., Saieva L., Flugy A.M., De Leo G., Alessandro R. Exosome-mediated crosstalk between chronic myelogenous leukemia cells and human bone marrow stromal cells triggers an interleukin 8-dependent survival of leukemia cells. Cancer Lett. 2014;348:71–76. doi: 10.1016/j.canlet.2014.03.009. [DOI] [PubMed] [Google Scholar]
- 237.Huan J., Hornick N.I., Shurtleff M.J., Skinner A.M., Goloviznina N.A., Roberts C.T., Jr., Kurre P. RNA trafficking by acute myelogenous leukemia exosomes. Cancer Res. 2013;73:918–929. doi: 10.1158/0008-5472.CAN-12-2184. [DOI] [PubMed] [Google Scholar]
- 238.Huan J., Hornick N.I., Goloviznina N.A., Kamimae-Lanning A.N., David L.L., Wilmarth P.A., Mori T., Chevillet J.R., Narla A., Roberts C.T., Jr., et al. Coordinate regulation of residual bone marrow function by paracrine trafficking of AML exosomes. Leukemia. 2015;29:2285–2295. doi: 10.1038/leu.2015.163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 239.Kumar B., Garcia M., Weng L., Jung X., Murakami J.L., Hu X., McDonald T., Lin A., Kumar A.R., DiGiusto D.L., et al. Acute myeloid leukemia transforms the bone marrow niche into a leukemia-permissive microenvironment through exosome secretion. Leukemia. 2018;32:575–587. doi: 10.1038/leu.2017.259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 240.Marlein C.R., Zaitseva L., Piddock R.E., Robinson S.D., Edwards D.R., Shafat M.S., Zhou Z., Lawes M., Bowles K.M., Rushworth S.A. NADPH oxidase-2 derived superoxide drives mitochondrial transfer from bone marrow stromal cells to leukemic blasts. Blood. 2017;130:1649–1660. doi: 10.1182/blood-2017-03-772939. [DOI] [PubMed] [Google Scholar]
- 241.Walkley C.R., Olsen G.H., Dworkin S., Fabb S.A., Swann J., McArthur G.A., Westmoreland S.V., Chambon P., Scadden D.T., Purton L.E. A microenvironment-induced myeloproliferative syndrome caused by retinoic acid receptor gamma deficiency. Cell. 2007;129:1097–1110. doi: 10.1016/j.cell.2007.05.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 242.Kim Y.W., Koo B.K., Jeong H.W., Yoon M.J., Song R., Shin J., Jeong D.C., Kim S.H., Kong Y.Y. Defective Notch activation in microenvironment leads to myeloproliferative disease. Blood. 2008;112:4628–4638. doi: 10.1182/blood-2008-03-148999. [DOI] [PubMed] [Google Scholar]
- 243.Kode A., Manavalan J.S., Mosialou I., Bhagat G., Rathinam C.V., Luo N., Khiabanian H., Lee A., Murty V.V., Friedman R., et al. Leukaemogenesis induced by an activating β-catenin mutation in osteoblasts. Nature. 2014;506:240–244. doi: 10.1038/nature12883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 244.Mosteo L., Storer J., Batta K., Searle E.J., Duarte D., Wiseman D.H. The Dynamic Interface Between the Bone Marrow Vascular Niche and Hematopoietic Stem Cells in Myeloid Malignancy. Front. Cell. Dev. Biol. 2021;9:635189. doi: 10.3389/fcell.2021.635189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 245.Cooper T.M., Sison E.A.R., Baker S.D., Li L., Ahmed A., Trippett T., Gore L., Macy M.E., Narendran A., August K., et al. A phase 1 study of the CXCR4 antagonist plerixafor in combination with high-dose cytarabine and etoposide in children with relapsed or refractory acute leukemias or myelodysplastic syndrome: A Pediatric Oncology Experimental Therapeutics Investigators’ Consortium study (POE 10-03) Pediatr. Blood Cancer. 2017;64:e26414. doi: 10.1002/pbc.26414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 246.Uy G.L., Rettig M.P., Motabi I.H., McFarland K., Trinkaus K.M., Hladnik L.M., Kulkarni S., Abboud C.N., Cashen A.F., Stockerl-Goldstein K.E., et al. A phase 1/2 study of chemosensitization with the CXCR4 antagonist plerixafor in relapsed or refractory acute myeloid leukemia. Blood. 2012;119:3917–3924. doi: 10.1182/blood-2011-10-383406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 247.Roboz G.J., Ritchie E.K., Dault Y., Lam L., Marshall D.C., Cruz N.M., Hsu H.C., Hassane D.C., Christos P.J., Ippoliti C., et al. Phase I trial of plerixafor combined with decitabine in newly diagnosed older patients with acute myeloid leukemia. Haematologica. 2018;103:1308–1316. doi: 10.3324/haematol.2017.183418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 248.Borthakur G., Ofran Y., Tallman M.S., Foran J., Uy G.L., DiPersio J.F., Showel M.M., Shimoni A., Nagler A., Rowe J.M., et al. BL-8040 CXCR4 antagonist is safe and demonstrates antileukemic activity in combination with cytarabine for the treatment of relapsed/refractory acute myelogenous leukemia: An open-label safety and efficacy phase 2a study. Cancer. 2021;127:1246–1259. doi: 10.1002/cncr.33338. [DOI] [PubMed] [Google Scholar]
- 249.Kovacsovics T.J., Mims A., Salama M.E., Pantin J., Rao N., Kosak K.M., Ahorukomeye P., Glenn M.J., Deininger M.W.N., Boucher K.M., et al. Combination of the low anticoagulant heparin CX-01 with chemotherapy for the treatment of acute myeloid leukemia. Blood Adv. 2018;2:381–389. doi: 10.1182/bloodadvances.2017013391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 250.Becker P.S., Foran J.M., Altman J.K., Yacoub A., Castro J.E., Sabbatini P., Dilea C., Wade M., Xing G., Gutierrez A., et al. Targeting the CXCR4 Pathway: Safety, Tolerability and Clinical Activity of Ulocuplumab (BMS-936564), an Anti-CXCR4 Antibody, in Relapsed/Refractory Acute Myeloid Leukemia. Blood. 2014;124:386. doi: 10.1182/blood.V124.21.386.386. [DOI] [Google Scholar]
- 251.DeAngelo D.J., Jonas B.A., Liesveld J.L., Bixby D.L., Advani A.S., Marlton P., O’Dwyer M.E., Fogler W.E., Wolfgang C.D., Magnani J.L., et al. Uproleselan (GMI-1271), an E-selectin antagonist, improves the efficacy and safety of chemotherapy in relapsed/refractory (R/R) and newly diagnosed older patients with acute myeloid leukemia: Final, correlative, and subgroup analyses. Blood. 2018;132:331. doi: 10.1182/blood-2018-99-114286. [DOI] [Google Scholar]
- 252.Maganti H., Visram A., Shorr R., Fulcher J., Sabloff M., Allan D.S. Plerixafor in combination with chemotherapy and/or hematopoietic cell transplantation to treat acute leukemia: A systematic review and metanalysis of preclinical and clinical studies. Leuk. Res. 2020;97:106442. doi: 10.1016/j.leukres.2020.106442. [DOI] [PubMed] [Google Scholar]
- 253.Woods K., Guezguez B. Dynamic Changes of the Bone Marrow Niche: Mesenchymal Stromal Cells and Their Progeny During Aging and Leukemia. Front. Cell Dev. Biol. 2021;9:714716. doi: 10.3389/fcell.2021.714716. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 254.Shono Y., Ueha S., Wang Y., Abe J., Kurachi M., Matsuno Y., Sugiyama T., Nagasawa T., Imamura M., Matsushima K. Bone marrow graft-versus-host disease: Early destruction of hematopoietic niche after MHC-mismatched hematopoietic stem cell transplantation. Blood. 2010;115:5401–5411. doi: 10.1182/blood-2009-11-253559. [DOI] [PubMed] [Google Scholar]
- 255.Holst J., Watson S., Lord M.S., Eamegdool S.S., Bax D.V., Nivison-Smith L.B., Kondyurin A., Ma L., Oberhauser A.F., Weiss A.S., et al. Substrate elasticity provides mechanical signals for the expansion of hemopoietic stem and progenitor cells. Nat. Biotechnol. 2010;28:1123–1128. doi: 10.1038/nbt.1687. [DOI] [PubMed] [Google Scholar]
- 256.Li J., Chen T., Huang X., Zhao Y., Wang B., Yin Y., Dai J., Cui Y., Zhao Y., Zhang R., et al. Substrate-independent immunomodulatory characteristics of mesenchymal stem cells in three-dimensional culture. PLoS ONE. 2018;13:e0206811. doi: 10.1371/journal.pone.0206811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 257.Caiazzo M., Okawa Y., Ranga A., Piersigilli A., Tabata Y., Lutolf M.P. Defined three-dimensional microenvironments boost induction of pluripotency. Nat. Mater. 2016;15:344–351. doi: 10.1038/nmat4536. [DOI] [PubMed] [Google Scholar]
- 258.O’Brien F.J. Biomaterials scaffolds for tissue engineering. Mater. Today. 2011;14:88–95. doi: 10.1016/S1369-7021(11)70058-X. [DOI] [Google Scholar]
- 259.Leisten I., Kramann R., Ventura Ferreira M.S., Bovi M., Neuss S., Ziegler P., Wagner W., Knüchel R., Schneider R.K. 3D co-culture of hematopoietic stem and progenitor cells and mesenchymal stem cells in collagen scaffolds as a model of the hematopoietic niche. Biomaterials. 2012;33:1736–1747. doi: 10.1016/j.biomaterials.2011.11.034. [DOI] [PubMed] [Google Scholar]
- 260.Kandow C.E., Georges P.C., Janmey P.A., Beningo K.A. Polyacrylamide hydrogels for cell mechanics: Steps toward optimization and alternative uses. Methods Cell Biol. 2007;83:29–46. doi: 10.1016/S0091-679X(07)83002-0. [DOI] [PubMed] [Google Scholar]
- 261.Carthew J., Frith J.E., Forsythe J.S., Truong V.X. Polyethylene glycol-gelatin hydrogels with tuneable stiffness prepared by horseradish peroxidase-activated tetrazine-norbornene ligation. J. Mater. Chem. B. 2018;6:1394–1401. doi: 10.1039/C7TB02764H. [DOI] [PubMed] [Google Scholar]
- 262.Ravichandran A., Meinert C., Bas O., Hutmacher D.W., Bock N. Engineering a 3D bone marrow adipose composite tissue loading model suitable for studying mechanobiological questions. Mater. Sci. Eng. C. 2021;128:112313. doi: 10.1016/j.msec.2021.112313. [DOI] [PubMed] [Google Scholar]
- 263.Marx-Blümel L., Marx C., Sonnemann J., Weise F., Hampl J., Frey J., Rothenburger L., Cirri E., Rahnis N., Koch P., et al. Molecular characterization of hematopoietic stem cells after in vitro amplification on biomimetic 3D PDMS cell culture scaffolds. Sci. Rep. 2021;11:21163. doi: 10.1038/s41598-021-00619-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 264.Ventura Ferreira M.S., Jahnen-Dechent W., Labude N., Bovi M., Hieronymus T., Zenke M., Schneider R.K., Neuss S. Cord blood-hematopoietic stem cell expansion in 3D fibrin scaffolds with stromal support. Biomaterials. 2012;33:6987–6997. doi: 10.1016/j.biomaterials.2012.06.029. [DOI] [PubMed] [Google Scholar]
- 265.Bai T., Li J., Sinclair A., Imren S., Merriam F., Sun F., Delaney C., O’Kelly M.B., Nourigat C., Jain P., et al. Expansion of primitive human hematopoietic stem cells by culture in a zwitterionic hydrogel. Nat. Med. 2019;25:1566–1575. doi: 10.1038/s41591-019-0601-5. [DOI] [PubMed] [Google Scholar]
- 266.Mousavi S.H., Abroun S., Soleimani M., Mowla S.J. 3-Dimensional nano-fibre scaffold for ex vivo expansion of cord blood haematopoietic stem cells. Artif. Cells Nanomed. Biotechnol. 2018;46:740–748. doi: 10.1080/21691401.2017.1337026. [DOI] [PubMed] [Google Scholar]
- 267.Kang Y.G., Shin J.W., Park S.H., Kim Y.M., Gu S.R., Wu Y., Ban H.Y., Shin J.W. A three-dimensional hierarchical scaffold fabricated by a combined rapid prototyping technique and electrospinning process to expand hematopoietic stem/progenitor cells. Biotechnol. Lett. 2016;38:175–181. doi: 10.1007/s10529-015-1952-8. [DOI] [PubMed] [Google Scholar]
- 268.Tavakol D.N., Bonini F., Tratwal J., Genta M., Brefie-Guth J., Braschler T., Naveiras O. Cryogel-based Injectable 3D Microcarrier Co-culture for Support of Hematopoietic Progenitor Niches. Curr. Protoc. 2021;1:e275. doi: 10.1002/cpz1.275. [DOI] [PubMed] [Google Scholar]
- 269.Bourgine P.E., Klein T., Paczulla A.M., Shimizu T., Kunz L., Kokkaliaris K.D. In vitro biomimetic engineering of a human hematopoietic niche with functional properties. Proc. Natl. Acad. Sci. USA. 2018;115:5689–5693. doi: 10.1073/pnas.1805440115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 270.Born G., Nikolova M., Scherberich A., Treutlein B., García-García A., Martin I. Engineering of fully humanized and vascularized 3D bone marrow niches sustaining undifferentiated human cord blood hematopoietic stem and progenitor cells. J. Tissue Eng. 2021;12:20417314211044855. doi: 10.1177/20417314211044855. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 271.Di Buduo C.A., Laurent P.A., Zaninetti C., Lordier L., Soprano P.M., Ntai A., Barozzi S., La Spada A., Biunno I., Raslova H., et al. Miniaturized 3D bone marrow tissue model to assess response to Thrombopoietin-receptor agonists in patients. ELife. 2021;10:e58775. doi: 10.7554/eLife.58775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 272.Badylak S.F., Gilbert T.W. Immune Response to Biologic Scaffold Materials. Semin. Immunol. 2008;20:109–116. doi: 10.1016/j.smim.2007.11.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 273.Duisit J., Orlando G., Debluts D., Maistriaux L., Xhema D., Bisthoven Y.J., Galli C., Peloso A., Behets C., Lengelé B., et al. Decellularization of the Porcine Ear Generates a Biocompatible, Nonimmunogenic Extracellular Matrix Platform for Face. Ann. Surg. 2018;267:1191–1201. doi: 10.1097/SLA.0000000000002181. [DOI] [PubMed] [Google Scholar]
- 274.Rieder E., Kasimir M.T., Silberhumer G., Seebacher G., Wolner E., Simon P., Weigel G. Decellularization protocols of porcine heart valves differ importantly in efficiency of cell removal and susceptibility of the matrix to recellularization with human vascular cells. J. Thorac. Cardiovasc. Surg. 2004;127:399–405. doi: 10.1016/j.jtcvs.2003.06.017. [DOI] [PubMed] [Google Scholar]
- 275.Zheng M.H., Chen J., Kirilak Y., Willers C., Xu J., Wood D. Porcine Small Intestine Submucosa (SIS) Is Not an Acellular Collagenous Matrix and Contains Porcine DNA: Possible Implications in Human Implantation. J. Biomed. Mater. Res. 2005;73:61–67. doi: 10.1002/jbm.b.30170. [DOI] [PubMed] [Google Scholar]
- 276.Bianco J.E.R., Rosa R.G., Congrains-Castillo A., Joazeiro P.P., Waldman S.D., Weber J.F., Saad S.T.O. Characterization of a novel decellularized bone marrow scaffold as an inductive environment for hematopoietic stem cells. Biomater. Sci. 2019;7:1516–1528. doi: 10.1039/C8BM01503A. [DOI] [PubMed] [Google Scholar]
- 277.Hashimoto Y., Funamoto S., Kimura T., Nam K., Fujisato T., Kishida A. The effect of decellularized bone/bone marrow produced by high-hydrostatic pressurization on the osteogenic differentiation of mesenchymal stem cells. Biomaterials. 2011;32:7060–7067. doi: 10.1016/j.biomaterials.2011.06.008. [DOI] [PubMed] [Google Scholar]
- 278.Lai W.Y., Li Y.Y., Mak S.K., Ho F.C., Chow S.T., Chooi W.H., Chan B.P. Reconstitution of bone-like matrix in osteogenically differentiated mesenchymal stem cell—Collagen constructs: A three-dimensional in vitro model to study hematopoietic stem cell niche. J. Tissue Eng. 2013;4:2041731413508668. doi: 10.1177/2041731413508668. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 279.Barreto-Duran E., Mejia-Cruz C.C., Jaramillo-Garcia L.F., Leal-Garcia E., Barreto-Prieto A., Rodriguez-Pardo V.M. 3D Multicellular Spheroid for the Study of Human Hematopoietic Stem Cells: Synergistic Effect Between Oxygen Levels, Mesenchymal Stromal Cells and Endothelial Cells. J. Blood Med. 2021;12:517–528. doi: 10.2147/JBM.S305319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 280.Hur J., Park J., Lee S.E., Yoon C.H., Jang J.H., Yang J.M., Lee T.K., Choi J.I., Yang H.M., Lee E.J., et al. Human peripheral blood-born hematosphere as a niche for hematopoietic stem cell expansion. Cell Res. 2011;45:987–990. doi: 10.1038/cr.2011.69. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 281.Pievani A., Sacchetti B., Corsi A., Rambaldi B., Donsante S., Scagliotti V., Vergani P., Remoli C., Biondi A., Robey P.G., et al. Human umbilical cord blood-borne fibroblasts contain marrow niche precursors that form a bone/marrow organoid in vivo. Development. 2017;144:1035–1044. doi: 10.1242/dev.142836. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 282.Kefallinou D., Grigoriou M., Boumpas D.T., Gogolides E., Tserepi A. Fabrication of a 3D Microfluidic Cell Culture Device for Bone Marrow-On-A-Chip. Micro Nano Eng. 2020;9:100075. doi: 10.1016/j.mne.2020.100075. [DOI] [Google Scholar]
- 283.Sieber S., Wirth L., Cavak N., Koenigsmark M., Marx U., Lauster R., Rosowski M. Bone marrow-on-a-chip: Long-term culture of human haematopoietic stem cells in a three-dimensional microfluidic environment. J. Tissue Eng. Regen. Med. 2018;12:479–489. doi: 10.1002/term.2507. [DOI] [PubMed] [Google Scholar]
- 284.Glaser D.E., Curtis M.B., Sariano P.A., Rollins Z.A., Shergill B.S., Anand A., Deely A.M., Shirure V.S., Anderson L., Lowen J.M., et al. Organ-on-a-chip model of vascularized human bone marrow niches. Biomaterials. 2022;280:121245. doi: 10.1016/j.biomaterials.2021.121245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 285.Sui C., Zilberberg J., Lee W. Microfluidic device engineered to study the trafficking of multiple myeloma cancer cells through the sinusoidal niche of bone marrow. Sci. Rep. 2022;12:1439. doi: 10.1038/s41598-022-05520-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 286.Souquet B., Opitz M., Vianay B., Brunet S., Théry M. Manufacturing a Bone Marrow-On-A-Chip Using Maskless Photolithography. Methods Mol. Biol. 2021;2308:263–278. doi: 10.1007/978-1-0716-1425-9_20. [DOI] [PubMed] [Google Scholar]
- 287.Tavafoghi M., Darabi M.A., Mahmoodi M., Tutar R., Xu C., Mirjafari A., Billi F., Swieszkowski W., Nasrollahi F., Ahadian S., et al. Multimaterial bioprinting and combination of processing techniques towards the fabrication of biomimetic tissues and organs. Biofabrication. 2021;13:042002. doi: 10.1088/1758-5090/ac0b9a. [DOI] [PubMed] [Google Scholar]
- 288.Tasnim N., De la Vega L., Anil Kumar S., Abelseth L., Alonzo M., Amereh M., Joddar B., Willerth S.M. 3D Bioprinting Stem Cell Derived Tissues. Cell Mol. Bioeng. 2018;11:219–240. doi: 10.1007/s12195-018-0530-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 289.Su X., Wang T., Guo S. Applications of 3D printed bone tissue engineering scaffolds in the stem cell field. Regen. Ther. 2021;16:63–72. doi: 10.1016/j.reth.2021.01.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 290.Teixeira B.N., Aprile P., Mendonça R.H., Kelly D.J., da Silva Moreira Thiré R.M. Evaluation of bone marrow stem cell response to PLA scaffolds manufactured by 3D printing and coated with polydopamine and type I collagen. J. Biomed. Mater. Res. B Appl. Biomater. 2019;107:37–49. doi: 10.1002/jbm.b.34093. [DOI] [PubMed] [Google Scholar]
- 291.Grigoryan B., Paulsen S.J., Corbett D.C., Sazer D.W., Fortin C.L., Zaita A.J., Greenfield P.T., Calafat N.J., Gounley J.P., Ta A.H., et al. Multivascular networks and functional intravascular topologies within biocompatible hydrogels. Science. 2019;464:458–464. doi: 10.1126/science.aav9750. [DOI] [PMC free article] [PubMed] [Google Scholar]