Abstract
Progressive improvements in perinatal care and respiratory management of preterm infants has resulted in increased survival of extremely low gestational age newborns over the past few decades. However, the incidence of bronchopulmonary dysplasia (BPD), the chronic lung disease after preterm birth, has not changed. Studies of the long-term follow-up of adults born preterm have shown persistent abnormalities of respiratory, cardiovascular, and cardiopulmonary function possibly leading to a lesser exercise capacity. The underlying causes of these abnormalities are incompletely known, but we hypothesize that dysanapsis, i.e., discordant growth and development, in the respiratory and cardiovascular systems is a central structural feature that leads to the lesser exercise capacity in young adults born preterm compared to those born at term. We discuss how the hypothesized system dysanapsis underscores the observed respiratory, cardiovascular, and cardiopulmonary limitations. Specifically, adults born preterm have 1) normal lung volumes but smaller airways that causes expiratory airflow limitation and abnormal respiratory mechanics but without impacts on pulmonary gas exchange efficiency; 2) normal total cardiac size but smaller cardiac chambers, and 3) in some cases, evidence of pulmonary hypertension particularly during exercise, suggesting a reduced pulmonary vascular capacity despite reduced cardiac output. We speculate that these underlying developmental abnormalities may accelerate the normal age-associated decline in exercise capacity, via an accelerated decline in respiratory, cardiovascular, and cardiopulmonary function. Finally, we suggest areas of future research, especially the need for longitudinal and interventional studies from infancy into adulthood to better understand how preterm birth alters exercise capacity across the lifespan.
Keywords: prematurity, extremely low gestational age newborns, lung development, cardiac development, bronchopulmonary dysplasia, pulmonary hypertension, exercise physiology
Graphical Abstract

Very preterm birth, i.e., birth occurring prior to completion of 32 weeks of gestation, creates challenges to neonatal life. The period of infancy and adolescences is characterized by abnormal development leading to pulmonary and cardiovascular dysanapsis. Various aspects of respiratory (e.g. lesser expiratory airflow, mechanical ventilatory constraints), cardiopulmonary (e.g. reduced pulmonary vascular capacity, greater pulmonary vascular pressures and resistance), and cardiovascular (e.g. reduced ventricular volumes and myocardial functional reserve) function are impaired. In combination, these aspects lead to a significantly reduced aerobic exercise capacity in adults born preterm. This topical review outlines the known and unknown physiologic factors that explain the observed aerobic exercise capacity.
INTRODUCTION
Preterm birth occurring “very” preterm (≤ 32 weeks gestation), and especially before 29 weeks gestation, presents significant health challenges including persistent respiratory and cardiovascular disease not only during neonatal life, but throughout childhood and into adulthood (Stoll et al., 2015; Thébaud et al., 2019). Bronchopulmonary dysplasia (BPD), the chronic lung disease that follows preterm birth, was first described over 50 years ago (Northway & Rosan, 1967). The characteristic pattern of BPD during this era involved relatively late gestation newborns at 32–34 weeks postmentrual age (PMA), and included four stages, beginning with acute respiratory distress syndrome (RDS; stage 1) with diffuse atelectasis and marked pulmonary edema due to high shunt across the ductus arteriosus (stage 2) to progressive chronic disease (stages 3 and 4). Death was due to severe respiratory failure with cor pulmonale and autopsy evidence of airways disease, distal lung inflammation, diffuse fibroproliferative changes, and hypertensive pulmonary vascular remodeling.
Since then, many advances in clinical care, including antenatal steroids, early surfactant therapy, improvements in respiratory care and mechanical ventilation, nutrition, and others, have improved the survival of extremely low gestational age newborns, especially those infants born between 24–28 weeks gestation. However, with this remarkable increase in survival over the last few decades, the incidence of BPD has not decreased over time and remains at ~40% for preterm infants born at ≤28 weeks gestation (Stoll et al., 2015; Abman et al., 2017b; Thébaud et al., 2019). BPD has traditionally been defined according to consensus criteria based on respiratory status at 36 weeks PMA as established by an NIH workshop (Jobe & Bancalari, 2001). The growing recognition of the importance of chronic lung disease in preterm infants that persist well beyond this timepoint throughout childhood has led to more recent efforts to re-define these late respiratory problems associated with the diagnosis of BPD (Steinhorn et al., 2017).
Although the risk for prolonged mechanical ventilation persists in an important subgroup of ventilator-dependent infants with severe BPD (Abman et al., 2017a), most preterm infants born in the “post-surfactant era” over the last 30 years often have milder form of BPD at 36 weeks PMA, yet these infants still remain at high risk for late respiratory illness in childhood and have late abnormalities of lung function (Martinez, 2016; Morrow et al., 2017). This so-called “new BPD” is characterized by abnormal lung development whereas “old BPD” was characterized primarily by disruption of normal lung development due to lung damage from hyperoxia and barotrauma from mechanical ventilation. The new BPD infants tend to have milder disease, yet early mortality in the NICU and severe BPD can persist as major clinical problems. Infants with the new BPD remain at high risk for prolonged NICU stays, extended need for ventilatory support, pulmonary hypertension, and late respiratory exacerbations.
Thus, despite major advances, many knowledge gaps toward enhancing late respiratory outcomes of preterm infants continue. In particular, more concerning endpoints after preterm birth beyond the BPD diagnosis are related to respiratory disease during childhood, i.e., risks for recurrent respiratory exacerbations, reactive airways disease, rehospitalizations, exercise intolerance, and others (Steinhorn et al., 2017). In addition, diverse physiologic mechanisms can contribute to oxygen dependency, such as the variable contributions of large airway disease, impaired distal lung development, pulmonary vascular disease, abnormal respiratory drive, and chest wall mechanics. Growing evidence has shown that preterm birth, even in the absence of having the diagnosis of BPD at 36 weeks PMA, can lead to late cardiopulmonary impairments in adulthood (Morrow et al., 2017).
Recent studies have shown that respiratory (Lovering et al., 2013, 2014; Duke et al., 2014, 2018; Molgat-Seon et al., 2019), cardiopulmonary (Laurie et al., 2018; Goss et al., 2018; Mulchrone et al., 2020), and cardiovascular (Lewandowski et al., 2013b; Huckstep et al., 2018, 2020), impairments persist into adulthood for preterms. These lead to high risk for early onset of chronic obstructive pulmonary disease (COPD), congestive heart failure, pulmonary hypertension and systemic vascular disease in adulthood (Carr et al., 2017; Goss et al., 2018; Crump et al., 2019b, 2019a, 2020, 2021; Mulchrone et al., 2020). In Table 1 we outline these known and unknown impairments that could and/or do contribute to a lesser aerobic exercise capacity in adults born preterm.
Table 1. Altered physiology in adults born preterm.
Review of the known and unknown aspects of altered physiology in adults born preterm that may or may not play a role in their lesser aerobic exercise capacity (i.e., VO2max) organized by physiologic system. Black text represents a factor that is normal and not impacted by preterm birth, red text represents a factor that is impaired in adults born preterm, and blue text represents a factor that is not known or has not been tested. Abbreviations: BPD = bronchopulmonary dysplasia; RM = respiratory muscles; RV = right ventricle.
| System | State | Aspect | Findings | References | Knowledge Gap(s) |
|---|---|---|---|---|---|
| Respiratory | Resting | Lung size | Normal | Halvorsen et al., 2004; Vrijlandt et al., 2006; Narang et al., 2009; Lovering et al., 2013, 2014; Clemm et al., 2014; Duke et al., 2014, 2018, 2019; Vollsæter et al., 2015; Farrell et al., 2015; Caskey et al., 2016; Laurie et al., 2018; Molgat-Seon et al., 2019; Huckstep et al., 2020 | 1) Static/dynamic compliance 2) Respiratory muscle strength 3) Airway mechanics 4) Work of breathing at rest and during exercise 5) Dimensions of breathlessness |
| Airway size | Smaller | Duke et al., 2018 | |||
| RM strength | ? | Jacob et al., 1998 (children only) | |||
| Compliance | ? | ||||
| Exercise | Expiratory flow limitation | Excessive | Lovering et al., 2014; MacLean et al., 2016; Duke et al., 2018; 2019 | ||
| Dynamic hyperinflation | Uncommon | Lovering et al., 2014; Duke et al., 2018; 2019 | |||
| Work of breathing | ? | ||||
| Excessive dyspnea | Unclear | Lovering et al., 2014; Duke et al., 2019 | |||
| Cardiopulmonary | Resting | Diffusing capacity | Normal/Low | Mitchell et al., 1998; Narang et al., 2009; Lovering et al., 2013; Duke et al., 2014 | 1) Direct measures of pulmonary pressure/resistance in larger cohorts 2) Measurement of left atrial pressure 3) Gas exchange in those with most severe preterm birth/BPD |
| Hemoglobin | Normal | Lovering et al., 2013; Duke et al., 2014 | |||
| Pulmonary pressure | Normal | Laurie et al., 2018; Goss et al., 2018 | |||
| Total pulmonary resistance | Normal/Increased | Laurie et al., 2018; Goss et al., 2018 (greater) | |||
| Pulmonary gas exchange efficiency | Normal | Lovering et al., 2013; Duke et al., 2014; Farrell et al., 2015 | |||
| Exercise | O2 delivery | Normal | Lovering et al., 2013; Duke et al., 2014; Farrell et al., 2015 | ||
| Pulmonary pressure | Increased | Laurie et al., 2018; Goss et al., 2018 | |||
| Total pulmonary resistance | Increased | Laurie et al., 2018 (Preterm with no BPD only); Goss et al., 2018 | |||
| Pulmonary Circulation/RV coupling | Normal? | Mohamed et al., 2020; Mulchrone et al., 2020 | |||
| Cardiovascular | Resting/Exercise | Ventricular Remodeling | Abnormal | Lewandowski et al., 2013a; 2013b; Telles et al., 2020 | 1) Direct measures of cardiac output/cardiopulmonary coupling 2) Myocardial functional reserve |
| Myocardial functional reserve | Reduced | Huckstep et al., 2018; Goss et al., 2018 | |||
| Vascular function | Abnormal | Lewandowski et al., 2015; Huckstep et al., 2018; Barnard et al., 2020 | |||
| Cardiac output | Reduced | Goss et al., 2018; Huckstep et al., 2018; 2020 | |||
| Musculoskeletal / Neuromuscular | Resting/Exercise | Dysfunction | ? | 1) Muscle biopsies for assement of metabolic function 2) Excitability of the motor pathway 3) Presence/magnitude of central/peripheral fatigue |
|
| Detraining | ? | ||||
| Neuromuscular function/fatiguability | ? |
As a result of this recent work, there has been a growing focus on the long-term health outcomes in adults born preterm with and without BPD, which has emerged as an important area of basic science, clinical, and applied research. Detrimental consequences of preterm birth exist in the respiratory and cardiovascular systems including impaired pulmonary function (Northway et al., 1990; Santuz et al., 1995; Jacob et al., 1998; Narang et al., 2009; Narang, 2010; Shah et al., 2012; Lovering et al., 2013, 2014; Duke et al., 2014, 2019), reduced exercise capacity, i.e., aerobic exercise or VO2max/peak, (Vrijlandt et al., 2006; Lovering et al., 2013, 2014; Clemm et al., 2014; Duke et al., 2014, 2018, 2019; Farrell et al., 2015; Caskey et al., 2016; Goss et al., 2018; Debevec et al., 2019; Haraldsdottir et al., 2019; Huckstep et al., 2020), varying degrees of pulmonary vascular disease (Laurie et al., 2018; Goss et al., 2018), early heart failure (Carr et al., 2017), and ischemic heart disease (Crump et al., 2019a).
The respiratory (mechanics, airways), cardiopulmonary (gas exchange, pulmonary hemodynamics), and cardiovascular (heart, systemic vessels) consequences of preterm birth have been well characterized in previous reviews over the last 5 years (Mazloum et al., 2014; Gibson & Doyle, 2014; Bolton et al., 2015; Islam et al., 2015; Davidson & Berkelhamer, 2017; Raju et al., 2017b; Malleske et al., 2018) and yet many of the underlying physiologic and pathophysiologic factors underlying these impairments, and their clinical implications, are just beginning to be understood. Likewise, the previous reviews listed above have focused upon providing descriptive information of resting conditions with a lesser focus upon the integrative systemic physiologic responses to exercise in adults born preterm. Exercise ability is negatively associated with morbidity and mortality in the general population (Myers et al., 2002; Gulati et al., 2003). The existing data strongly suggest that adult exercise ability is reduced in those born preterm (Svedenkrans et al., 2016; Crump et al., 2019b; Yang et al., 2021), but whether or not this reduced exercise ability is associated with an increased morbidity and mortality in those adults born preterm, as it is in other adult populations, is a critical knowledge gap. Our current lack of knowledge limits patient care, including optimizing therapeutic strategies for disease prevention, managing at-risk or symptomatic children, and designing appropriate interventions to develop successful clinical care programs. Accordingly, the goal of this review is to describe the physiologic factors causing impaired respiratory, cardiopulmonary, and cardiovascular function in adults born preterm and to highlight work investigating how these impariments contribute to reduced exercise capacity in this population, thereby identifying knowledge gaps in the process.
ABNORMAL LUNG AND HEART FUNCTION IN THE PRETERM INFANT AND CHILD
The pathophysiology of BPD has evolved over the past 50 years to be characterized by arrested lung development due to intrauterine factors, as well as postnatal lung injury due to mechanical ventilation, hyperoxia and other adverse stimuli (Abman et al., 2017b). In addition to long-recognized abnormalites of airway structure and function, preclinical models and clinical autopsy specimens1 have identified striking decreases in alveolar and vascular growth in the distal lung, reflecting decreased surface area for gas exchange, as a major determinant of sustained impairment of lung function (Thébaud et al., 2019). For example, recent studies in rodents (Sprague-Dawley rats), have shown that antenatal models of preeclampsia (injection of soluble fms-like tyrosine kinase 1) and chorioamnionitis (injection of endotoxin; Escherichia coli), known as being strongly associated with risk for BPD in human infants, are sufficient to impair lung growth and cause pulmonary hypertension in offspring without exposures to postnatal stresses such as hyperoxia or mechanical ventilation (Tang et al., 2011; Mandell et al., 2015). Preterm infants have clinical courses characterized by recurrent wheezing, the need for emergency room visits or hospitalizations for respiratory exacerbations during the first two years of life, and the chronic need for asthma medication with worse clinical outcomes in those with BPD (Morrow et al., 2017).
Following preterm birth, lung function studies have consistently demonstrated variable degrees of airflow obstruction, lower residual volumes, and decreased lung compliance, which persist from the early postnatal period through 6 and 18 months of age (as reviewed in (Islam et al., 2015). In addition to airway disease, impaired postnatal alveolar growth has been demonstrated in BPD infants (Balinotti et al., 2010). Tepper and colleagues have uniquely performed studies of lung diffusion with carbon monoxide and report impaired distal lung surface area throughout infancy despite normal alveolar volumes (Balinotti et al., 2010), providing striking phyiologic evidence to support past histologic evidence of decreased alveolarization and vascular density in “old” BPD infants.
Additionally, lung dysanapsis, defined as discordant growth between airway diameter and lung size, can contribute to airflow limitation, air-trapping, and hyperinflation (Kennedy, 1999; Hove et al., 2014; Ronkainen et al., 2015; Hirata et al., 2017; Simpson et al., 2017; Urs et al., 2018). Altered resting lung function in preterm infants largely persist into adolescence and adulthood and is likely one of the links to lesser aerobic exercise capacity in adulthood. Doyle and coworkers showed progressively worse lung function over time in survivors of preterm birth with increased airways obstruction from 8 to 18 years of age, which was worse in those that developed BPD (Doyle et al., 2017). Lung function trajectories track from early childhood into adulthood with normal aging, and at least one study suggests that reduced lung function in early life is associated with a clinical presentation similar to COPD in adulthood (Bui et al., 2018).
In addition to the respiratory and cardiopulmonary sequelae in preterm infants and children, there are known cardiovascular abnormalities. During normal embryonic and fetal development, cardiac growth is driven primarily by cardiomyocyte hyperplasia (Bensley et al., 2010; Tan & Lewandowski, 2019). The dramatic hemodynamic shift, from low systemic vascular resistance due to the placental circulation in utero to high resistance systemic arterial circulation at birth, facilitates a switch in cardiac growth towards myocyte hypertrophy (Bensley et al., 2010). Within the first days to weeks post-delivery, the left ventricle (LV) takes over as the dominant pumping chamber to meet systemic blood flow demands, as the right ventricle (RV) remodels to being a thin-walled, crescent shaped chamber supplying the lower pressure pulmonary circulation. Preterm birth, even without BPD, is associated with significant cardiac remodeling, including functional and structural impairments in early life (Telles et al., 2020) that may be the result of dysanapsis within the cardiovascular system.
Importantly, these aforementioned infancy/childhood characteristics are the basis or starting point for the, potentially, lifelong cardiopulmonary challenges and a lesser aerobic exercise capacity in the absence of significant catch-up growth or rescue. Ongoing preclinical work has demonstrated a variety of mechanistic approaches to amerliorate the effects of hyperoxia-induced lung injury, i.e., animal models of BPD, and improve aerobic exercise capacity. For example, evidence in a rodent model of BPD (rat pups treated with 95% O2 from birth to postnatal day 14) has demonstrated that airway delivery of human umbilical cord-derived stem cells recovers pulmonary function and exercise ability (Pierro et al., 2013). Similarly, Willis et al. (Willis et al., 2020) demonstrated in a rodent model of BPD (FVP strain mouse pups treated with 75% O2 from birth to postnatal day 14) that venous administration of mesenchymal stromal cell-derived small extracellualr vesicles improved vasious aspects of hyperoxia-induced lung damage and resulted in functional improvements in exercise capacity and an attenuation of pulmonary hypertension. Finally, Hansmann et al. (Hansmann et al., 2012) used a rodent model of BPD (FVP strain mouse pups treated with 75% O2 from birth to postnatal day 14) to demonstrate that intravenous administration of cultured mouse bone marrow-derived mesenchymal stem cells partially reversed alveolar injury, which normalized lung function, and fully reversed pulmonary hypertension and right ventricular hypertrophy. Although they did not assess exercise capacity, prior work suggests that it would have been improved following treatment. These are just a few of the available studies using a variety of treatments in animal models of preterm birth and BPD to rescue cardiopulmonary function. Hopefully translation to human patients is something in the not-so-distant future.
IDENTIFYING POTENTIAL LIMITS TO EXERCISE CAPACITY IN ADULTS BORN PRETERM
Despite having similar levels of physical activity (Tikanmäki et al., 2017), adults born very preterm (gestational age ≤32 weeks) have a lower exercise capacity than term-born controls (Vrijlandt et al., 2006; Lovering et al., 2013, 2014; Clemm et al., 2014; Duke et al., 2014, 2018, 2019; Farrell et al., 2015; Caskey et al., 2016; Goss et al., 2018; Debevec et al., 2019; Haraldsdottir et al., 2019; Huckstep et al., 2020) (Figure 1). Exercise capacity decreases with age, making it important to better understand longitudinal changes in the physiologic aspects of why exercise is limited in young adults born preterm. Exercise capacity is predictive of all cause morbidity and mortality in the general population (Myers et al., 2002; Gulati et al., 2003), which suggests that understanding factors limiting exercise capacity at a younger age may have significant clinical implications. As a result, testing and developing optimal therapeutic strategies to improve exercise capacity in this population may substantially reduce morbidity and increase longevity.
Figure 1: Aerobic exercise capacity is reduced in young adults born preterm.
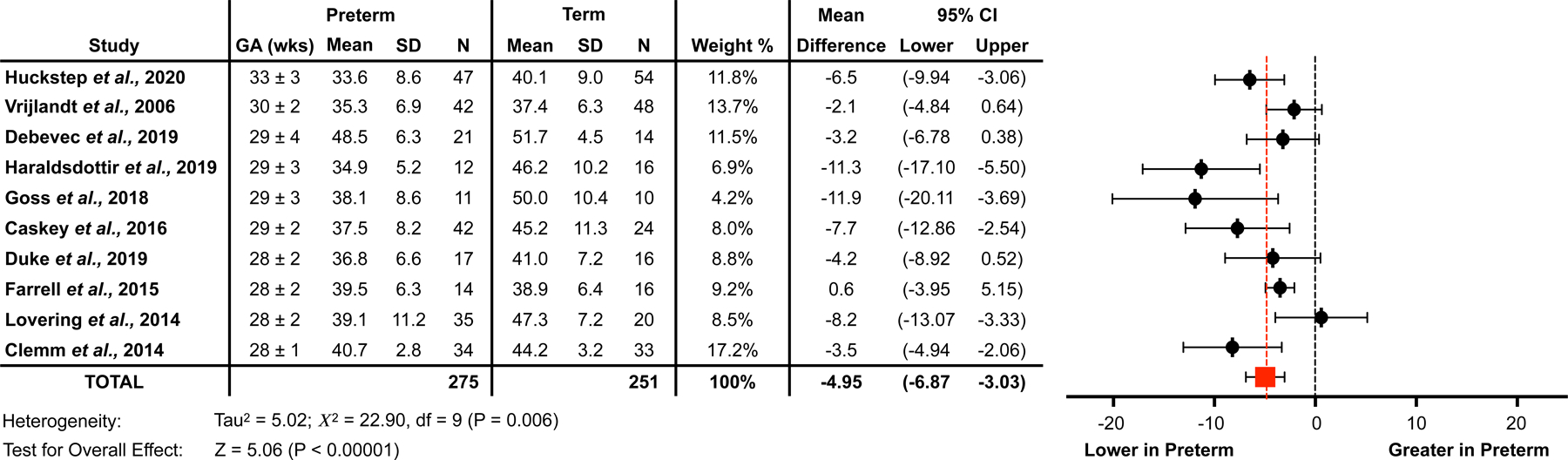
Forest plot generated via Review Manager demonstrating a lesser aerobic exercise capacity (VO2max; mL/kg/min) in young adults born preterm (preterm) compared to their counterparts born at term (term). These are pooled data from various studies in adults that reported VO2max in their results (Huckstep et al., 2020; Vrijlandt et al., 2006; Debevec et al., 2019; Haraldsdottir et al., 2019; Goss et al., 2018; Caskey et al., 2016; Duke et al., 2019; Farrell et al., 2015; Lovering et al., 2014; Clemm et al., 2014). Data are organized from latest to earliest mean gestational age reported in each study. Gestational age was averaged for those studies that separated the preterm group into those that did or did not have bronchopulmonary dysplasia. The N column represents the respective sample size of each study. The age range of the participants, at the time of study, of the included studies was 19–28 years, obtained from mean data reported. Mean differences and 95% confidence intervals were computed using the inverse variance weighting method. The red square and red dashed line represent the difference of pooled preterm and term-born data from studies included in the analysis. In the studies included, there was an overall statistical effect with preterm having a lower VO2max than term.
Exercise capacity is determined by the integrative response of multiple physiologic systems including the respiratory, cardiovascular, and musculoskeletal/neuromuscular systems. Therefore, exercise capacity can be reduced via several mechanisms including mechanical ventilatory constraints, abnormal pulmonary gas exchange, decreased pulmonary vascular capacity, reduced cardiac performance/function, impaired muscle O2 uptake/utilization and/or impaired neuromuscular coupling/function. We propose that adults born preterm have excessive cardiopulmonary dysanapsis, which is a primary underlying cause of their lower aerobic exercise capacity. This is outlined below and reflected schematically in Figure 2. Additionally, deconditioning/detraining from lack of physical activity could be a contributing factor to the lesser exercise capacity in adults born preterm. Although this has not yet been directly studied, lactate production is greater at a given cycling workload in adults born preterm compared to their counterparts born at term, which may suggest detraining and/or altered skeletal muscle metabolic function (Table 3; Lovering et al., 2013). In this regard, detraining would reduce exercise capacity beyond that due to the structural/physiologic impairments resulting from preterm birth. Interactions of these physiologic mechanisms and their potential contributions to changes in cardiopulmonary function in adults born preterm are discussed below.
Figure 2: Cardiopulmonary factors limiting aerobic exercise capacity in term and preterm adults.
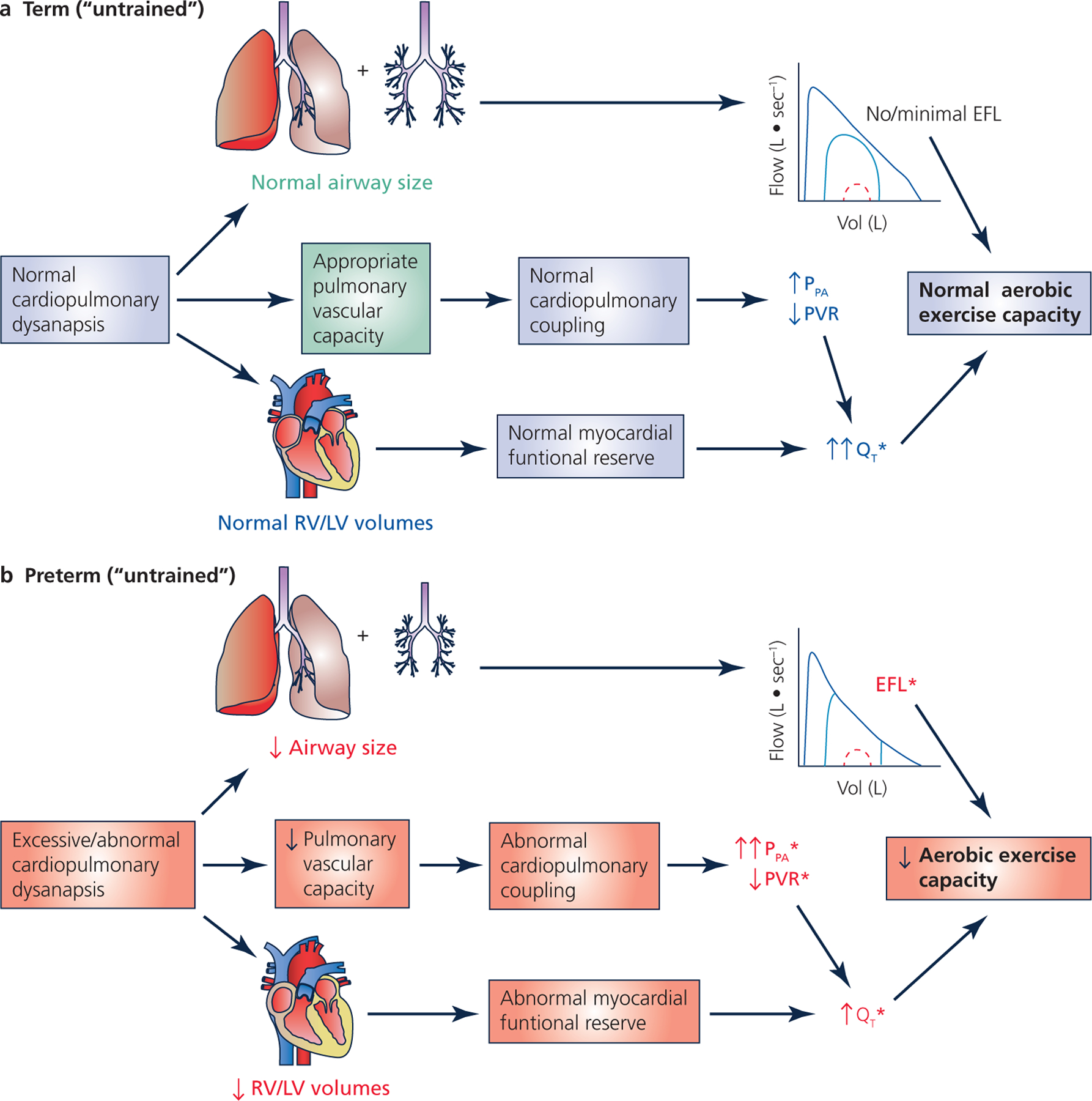
Schematic representation of the factors limiting aerobic exercise capacity (VO2max/peak in mL/kg/min) in (A) untrained adults born at term and (B) untrained adults born preterm. In those born at term, there is normal cardiopulmonary dysanapsis and, thus, an “overbuilt” pulmonary system (lungs + vasculature). This means that the respiratory system of the untrained, term-born adult is not a limiting factor as evidence by little/no mechanical ventilatory constraints and a minimal change in PPA and PVR, neither of which limit QT. Despite an appropriate cardiovascular system, QT limits aerobic exercise capacity in untrained adults born at term. Conversely, in the adult born preterm, excessive/abnormal respiratory, cardiopulmonary, and cardiovascular dysanapsis leads to multiple factors contributing (in heterogeneous proportions) to reduced aerobic exercise capacity. The * denotes a factor that impairs/limits aerobic exercise capacity. Green text represents a physiologic attribute that is exceptional/overbuilt, blue text represents an attribute that is considered to be normal, and red text represents an attribute that is reduced/impaired. For flow-volume loop schematic, the black line represents the maximum flow-volume loop generated from a forced expiration, the red dashed line represents the resting tidal flow-volume loop, and the blue line represents the exercise flow-volume loop. Abbreviations: EFL = expiratory flow limitation; LV = left ventricle; PPA = pulmonary artery pressure; PVR = pulmonary vascular resistance; QT = cardiac output; RV = right ventricle.
Respiratory Limitations to Exercise
Respiratory function is lower in adults born preterm compared to those born at term (Mazloum et al., 2014; Bolton et al., 2015; Islam et al., 2015; Gibson et al., 2015; Davidson & Berkelhamer, 2017; Raju et al., 2017a; Malleske et al., 2018). The clinical relevance of these respiratory abnormalities described above is reflected by the respiratory and cardiopulmonary responses to exercise. The potential underlying physiologic factors that explain why pulmonary function is worse in adults born preterm are described below. Physical properties governing airflow include lung size, driving pressure, and resistance. Previous work has noted no differences in lung volumes/capacities between adults born preterm and term-born controls when appropriately matched for sex and height (Halvorsen et al., 2004; Vrijlandt et al., 2006; Narang et al., 2009; Lovering et al., 2013, 2014; Clemm et al., 2014; Duke et al., 2014, 2018, 2019; Vollsæter et al., 2015; Farrell et al., 2015; Caskey et al., 2016; Laurie et al., 2018; Molgat-Seon et al., 2019; Huckstep et al., 2020). Thus, lower pulmonary function is likely the result of interactions between impaired expiratory driving pressure and increased airway resistance, as discussed below.
Expiratory driving pressure, i.e., alveolar pressure, is determined by expiratory muscle strength, inward elastic recoil of the lungs and is opposed (after the initial 20% of vital capacity is exhaled) by outward elastic recoil of the chest wall (Mead et al., 1967). Thus, altered compliance/elastance of the respiratory system and/or weak expiratory muscles would manifest as low expiratory airflow. Elastic recoil of the lungs and chest wall could be altered by preterm birth and lung injury, but pulmonary system compliance/elastance has not been measured in adults. Animal models of preterm birth and/or BPD, i.e., baboons delivered at 75% of gestation and lambs delivered at ~80% of gestation and mechanically ventilated for 3 weeks, (Coalson et al., 1995; Bland et al., 2003; Dahl et al., 2018) and infants with “old” BPD (Northway & Rosan, 1967) report fewer but larger alveoli, i.e., alveolar simplification, which could reduce lung recoil. In humans, there are a variety of methods that could provide direct [measurement of esophageal pressure during quasi-static relaxations; (Cross et al., 2021; Gideon et al., 2021)] and/or indirect (CT and/or forced oscillation technique) evidence of altered lung/chest wall compliance/elastance in adults born preterm. Previous research using CT, however, does not support the hypothesis that adults born preterm have impaired lung compliance (Margraf et al., 1991; Wong et al., 2008; Aukland et al., 2009; Caskey et al., 2016; Simpson et al., 2017). Similarly, data from the forced oscillation technique, and specifically assessing the reactance parameter, report that children born preterm may have a greater pulmonary compliance compared to term-born children (Udomittipong et al., 2008) or normal reference values (Vrijlandt et al., 2007). It is not possible to tease apart whether it was altered lung or chest wall compliance (or both) that explains the findings with the forced oscillation technique. Nevertheless, the greatest determinant of driving pressure is pressure generated by the respiratory (expiratory) muscles. Pressure-generating ability of the expiratory muscles is dependent upon their strength and the lung volume at which the effort is made. One study has assessed maximal expiratory pressure in survivors of preterm birth (Jacob et al., 1998). They found maximum expiratory pressure generation to be within the normal range for their age (10–12 years), but it is unknown if these findings extend into adulthood. Thus, direct measures of pulmonary system compliance/elastance and maximal expiratory pressure, throughout the lung volume continuum, in adults born preterm are needed to fill these knowledge gaps.
Airway resistance can be assessed simply by quantifying mid-expiratory airflow or more directly to provide information about tracheobronchial structure and dynamics. Children and adolescents born preterm have a greater airway resistance than term-born controls, as measured with plethysmography [calculated as the ratio between driving pressure (measured via mouth pressure as a correlate of alveolar pressure) and airflow (Halvorsen et al., 2004; Vrijlandt et al., 2006)]. Likewise, children born preterm have greater resistance in small or medium-sized airways compared with term-born controls (Malmberg et al., 2002; Vrijlandt et al., 2007; Udomittipong et al., 2008; Sarria et al., 2012) as measured by the forced oscillation technique or impulse oscillometry [measured using sinusoidal vibrations sent into the airways and drawing inference on airflow resistance based upon changes in airflow and mouth pressure (as a correlate of alveolar pressure)]. These methods, however, have not yet been employed in adults born preterm, which identifies an important knowledge gap.
Greater airflow resistance in adults born preterm could be caused by smaller airways due to structure, diffuse inflammation or bronchial hyperreactivity, or perhaps reduced airway tethering due to emphysema/alveolar simplification. A recent CT study reported that children born preterm had smaller distal airways (e.g., 3rd or 4th generation of airways) than term-born controls (Sarria et al., 2012). In adults born preterm, the dysanapsis ratio has been used as an index of relative airway diameter (Duke et al., 2018) as it reflects the non-synchronous growth of airways and lung parenchyma. Adults born preterm were found to have smaller airways (i.e., lower dysanapsis ratio) in comparison to term-born controls, with those diagnosed with BPD having the smallest airways (i.e., lowest dysanapsis ratio) (Duke et al., 2018). The dysanapsis ratio, as computed in previous work (Dominelli et al., 2011; Duke et al., 2018), is not without limitation. Particularly, using an estimated lung recoil pressure based upon age is problematic (Babb et al., 2012) as it eliminates an important, and physiologically relevant, source of inter-subject variability (Stickford et al., 2021). Nonetheless, the dysanapsis ratio incorporates several important aspects of the pulmonary system and, when computed with measured lung recoil pressure data, may be useful in helping to better understand the complex relationships between preterm birth, obstructive airflow, and distal lung development.
Prior work does not differentiate between airways that are morphologically small from airways that are narrowed due to bronchoconstriction (Duke et al., 2018). Previous work has examined whether or not adults born preterm have airway hyperreactivity (Halvorsen et al., 2004; Narang et al., 2009), are responsive to bronchodilators (Koumbourlis et al., 1996; Halvorsen et al., 2004, 2005; Lovering et al., 2013; Cardoen et al., 2019), and/or have a greater prevalence/incidence of asthma (Halvorsen et al., 2004, 2005; Narang et al., 2009). However, this work is equivocal and show no clear pattern of generalizable altered airway physiology in adults born preterm. Interestingly, the pattern of basal airflow obstruction with variable degrees of airway hyperresponsiveness as observed in adults born preterm was more likely due to structural sequelae rather than inflammation as observed in most patients with asthma (Halvorsen et al., 2005). Regardless, it may be reasonable to suspect that some but not most adults born preterm have both small and constricted airways. Further studies are needed to tease apart these different subgroups, i.e., those with normal airway physiology yet small airways, those with airway hyperresponsiveness and small airways, etc., within the population of adults born preterm to better understand specific mechanisms contributing to airflow obstruction that is observed in adults born preterm. Likewise, more studies are needed to determine the acute and long-term effects of different bronchodilators, i.e., short- or long-acting, inhaled steroid, etc., on airway function, in this population.
Mechanical Ventilatory Constraints.
A mechanical ventilatory constraint is any parameter that could alter the breathing pattern or limit the increase in ventilation during exercise (Whipp & Pardy, 2011). These include expiratory airflow limitation and/or dynamic hyperinflation, which would result in an excessive work of breathing. It has been shown in several populations, including highly-trained endurance athletes and COPD patients, that the presence of significant mechanical ventilatory constraints can impair exercise capacity (Harms et al., 1998, 2000; Amann et al., 2010; Dominelli et al., 2017) via a sympathetically-mediated redistribution of cardiac output termed the “metaboreflex” (Harms et al., 1998, 2000). This “metaboreflex” refers to a metabolite accumulation in the muscle beds during high intensity exercise, in this case the respiratory muscles, which elicits an increased blood flow demand from the respiratory muscles “stealing” blood from the exercising locomotor muscles. The presence of significant mechanical ventilatory constraints during exercise has been reported in adults born preterm (Lovering et al., 2014; MacLean et al., 2016; Duke et al., 2018, 2019).
Expiratory airflow limitation is present when expiratory airflow during a tidal breath meets the maximum attainable airflow for a given lung volume and is uncommon in healthy, non-sedentary individuals (Johnson et al., 1999). Conversely, expiratory airflow limitation during exercise is common in athletes and patients with lung disease. Adults and adolescents born preterm have a greater prevalence and magnitude of expiratory airflow limitation compared to term-born controls (Lovering et al., 2014; MacLean et al., 2016; Duke et al., 2018, 2019). How and why adults born preterm develop expiratory airflow limitation is not completely known, but is likely due to a lesser ventilatory capacity resulting from being born preterm, perhaps due to exaggerated dysanapsis. Exercise endurance increased significantly in adults born preterm when expiratory airflow limitation was lessened by breathing a helium-oxygen mixture (Duke et al., 2019). However, the increase in exercise endurance was not equal in magnitude to the decrease in expiratory airflow limitation in this study, suggesting that expiratory airflow limitation explains only a portion of the lower exercise capacity in adults born preterm.
Dynamic hyperinflation occurs when expiration is prematurely terminated and the next inspiration begins at lung volumes that are closer to total lung capacity, i.e, an end-expiratory lung volume that is ≥0.5 L greater than functional residual capacity. This breathing strategy is common in patients with severe COPD (Aliverti et al., 2004; Eves et al., 2006; Guenette et al., 2012; Louvaris et al., 2012), but is less so (~1 in 10 participants) in adults born preterm (Lovering et al., 2014; Duke et al., 2018, 2019). To date, the mechanical work of breathing has not been measured in adults born preterm, but the presence of one or more mechanical ventilatory constraint would result in an excessive work of breathing for a given ventilation. Utilizing modified Campbell diagrams, which allows the work required to overcome airway resistance and tissue elasticity during inspiration and expiration to be quantified (Cross et al., 2021; Gideon et al., 2021), would provide substantive information about how dynamic respiratory mechanics of adults born preterm differ from term-born individuals. Likewise, quantifying maximum effective expiratory pressure, and determining whether it is met or exceeded during exercise, would provide additional insight. Collectively, this information would be of significant physiologic and clinical interest.
Cardiopulmonary Interactions and Limitations to Exercise
Pulmonary Gas Exchange Efficiency.
Maximization of pulmonary gas exchange efficiency requires optimal ventilation and perfusion matching, diffusing capacity and minimal right-to-left shunt. This is important because one potential candidate for limiting exercise capacity is exercise-induced arterial hypoxemia (Dempsey & Wagner, 1999), which reduces oxygen delivery leading to an earlier onset of fatigue (Romer et al., 2006; Amann et al., 2006). In healthy term-born individuals, the architecture of the lung ensures that pulmonary gas exchange efficiency, measured by the alveolar-to-arterial oxygen difference (AaDO2), is ~5–10 mmHg at rest and increases to ~25 mmHg at peak exercise (Dempsey & Wagner, 1999). Although the AaDO2 increases during exercise, arterial PO2 and oxygen saturation (SaO2) are maintained at near-resting levels (Dempsey & Wagner, 1999; Lovering et al., 2005). Thus, maximal pulmonary gas exchange efficiency is easily achieved so that exercise-induced arterial hypoxemia does not develop in healthy term born adults.
As detailed above, the preterm infant lung is characterized by abnormal alveolar and vascular growth and adults born preterm have reduced pulmonary function, with lower resting diffusing capacity for carbon monoxide (Mitchell et al., 1998; Narang et al., 2009; Duke et al., 2014). Abnormal lung development could be expected to compromise ventilation and perfusion matching and reduce diffusing capacity, possibly resulting in impaired pulmonary gas exchange efficiency and arterial hypoxemia at rest. This presumed impairment could also be expected to be exacerbated during exercise when ventilation and cardiac output are increased significantly. However, adults born preterm have normal resting arterial PO2, PCO2, and SaO2 (Lovering et al., 2013; Duke et al., 2014; Farrell et al., 2015). With exercise, the AaDO2 does not increase more in adults born preterm compared to term-born controls (Lovering et al., 2013; Duke et al., 2014; Farrell et al., 2015). Additionally, arterial PCO2 during near-maximal exercise also does not differ between adults born preterm and term-born controls (Lovering et al., 2013; Duke et al., 2014; Farrell et al., 2015), demonstrating an appropriate ventilatory response to exercise. Remarkably perhaps, this was also true in those adults born preterm with a clinically mild reduction in diffusing capacity when exercising while breathing hypoxic gas (Duke et al., 2014). Accordingly, the potential mechanisms of exercise limitations in adults born preterm do not appear to include arterial hypoxemia (Lovering et al., 2013; Duke et al., 2014), which may be due to their lower VO2max creating less of a demand on the lungs. Nevertheless, those with more severe lung disease resulting from prematurity may have normal resting pulmonary gas exchange efficiency that is compromised during exercise (Lovering et al., 2007). Continued studies of pulmonary gas exchange efficiency in larger cohorts with more severe lung disease are certainly warranted.
Blood flow through intrapulmonary arteriovenous anastomoses (i.e., shunts) could widen the AaDO2 as mentioned above, but their presence and physiologic importance remains controversial. Nonetheless, previous work in term-born individuals under rigorously controlled experimental conditions has demonstrated that increased blood flow through intrapulmonary arteriovenous anastomoses is associated with a significantly increased AaDO2 (Elliott et al., 2014). Our previous work found there to be no difference in the qualitative magnitude of blood flow through intrapulmonary arteriovenous anastomoses between adults born preterm and term-born individuals during exercise (Lovering et al., 2013; Duke et al., 2014). Importantly, the cohort studied was on the healthier side of the preterm severity spectrum with very few having severe BPD. In infants who perished from severe BPD there is histological evidence of intrapulmonary shunts (Galambos et al., 2013). Thus, it is possible that these intrapulmonary shunts play a role in the morbidity and mortality of BPD, and may also impair pulmonary gas exchange efficiency in more severe cases of BPD, but more work in this area is needed.
Pulmonary Hemodynamics.
Adults born preterm may have a reduced vascular capacity compared to term-born controls (Bhatt et al., 2001; Parker & Abman, 2003), suggesting dysanapsis within the pulmonary circulation. Reduced pulmonary vascular surface area/capacity may contribute to elevated pulmonary artery pressures at rest and/or an exaggerated response during exercise. In the past, preterm birth has generally not been considered to be associated with resting pulmonary arterial hypertension (pulmonary arterial pressure >25 mmHg (Galiè et al., 2015)) in adults (Laurie et al., 2018; Goss et al., 2018). Nevertheless, some adults born preterm (2 out of 11) do have resting pulmonary hypertension and others have pulmonary pressures in the border-line range (>20 but <25 mmHg) (Goss et al., 2018). Importantly, according to recent guidelines (Simonneau et al., 2018), the diagnosis of pulmonary hypertension now includes those with mean pulmonary artery pressure >20 mmHg. The basis for this change in definition is based on several studies of normal subjects and referral patients who underwent right heart catheterization, which demonstrated a continuum of morbidities and mortality that related to mean pulmonary artery pressure >20 mm Hg (Kovacs et al., 2009; Maron et al., 2016; Assad et al., 2017). These findings suggest that recent studies of “borderline” pulmonary hypertension in adults born preterm (Laurie et al., 2018; Goss et al., 2018) may be an important marker suggesting risk for later morbidities and early mortality.
Reduced pulmonary vascular capacity in adults born preterm may also contribute to exaggerated increases in pulmonary pressure during exercise as cardiac output increases. Indeed, there is an exaggerated increase in pulmonary arterial pressure during exercise in adults born preterm (Laurie et al., 2018; Goss et al., 2018). Interestingly, those adults born preterm but did not develop BPD had the largest increases in pulmonary pressure with exercise (Laurie et al., 2018). It is well known that pulmonary pressures at rest and during exercise increase with age (Kovacs et al., 2009, 2012) suggesting that the exaggerated increase in pulmonary pressure will likely get worse as preterm subjects age. Although the effect of biological sex on the pulmonary pressure response to exercise has not yet been determined, in the study by Laurie et al., the largest increases in pulmonary pressure tended to be women (see Figure 1 of that study), though due to low subject (n = 11 female out of n = 33 total born preterm) numbers this did not reach statistical significance (Laurie et al., 2018). However, it may be possible that exercise-induced pulmonary hypertension (Herve et al., 2015) may be more prevalent in adult women born preterm, but future work is needed. The potential effect of biological sex on pulmonary hemodynamic responses in adults born preterm is an important area of future exploration given that the prevalence of pulmonary hypertension is greater in women than men (Rich et al., 1987; Loyd et al., 1995; Badesch et al., 2010; Humbert et al., 2012; Pugh & Hemnes, 2014).
Likewise, pulmonary hypertension is associated with right heart failure, which may contribute to the greater risk of early heart failure in men and women born preterm (Carr et al., 2017; Crump et al., 2021). Indeed, in a population study of 2.6 million persons born between 1987–2012 followed until December 31st 2013, the adjusted incidence relative risk of heart failure compared to term-born individuals was 17.0 [95% CI, 7.96–36.3] for those born extremely preterm and 3.58 [95% CI, 1.57–8.14] for those born very preterm, with no increased risk in those born moderately preterm (1.36 [95% CI, 0.87–2.13]) (Carr et al., 2017). In a more recent population study of 4.1 million persons aged 18–43 years, heart failure incidence was greater in preterms compared to term born controls (adjusted hazard ratio, 1.42 [95% CI, 1.19–1.71]) without differences in heart failure cases for men and women (Crump et al., 2021).
With alveolar hypoxia, hypoxic pulmonary vasoconstriction and increased cardiac output result in an increase in pulmonary arterial pressure at rest. This increase in pulmonary pressure is further increased with exercise in low oxygen conditions. Preterm infants with severe BPD and low resting SaO2 values have resting pulmonary vasoconstriction and pulmonary hypertension that is decreased with administration of hyperoxia suggesting an exaggerated pulmonary vascular response to hypoxia (Abman et al., 1985). Interestingly with alveolar hypoxia (FIO2 = 0.12) in adults born preterm with mild-to-moderate severity lung disease, there does not appear to be an exaggerated pulmonary vasoconstriction and/or increase in pulmonary arterial pressure (Laurie et al., 2018; Goss et al., 2018).
In a recent study using echocardiography and cardiovascular MRI in adults born moderately preterm, RV changes, including smaller volumes and reduced systolic function, remained significant when adjusting for spirometry-derived pulmonary function parameters (Mohamed et al., 2020). Although preterm participants had increased pulmonary vascular resistance, the RV remained coupled to its pulmonary circulation. However, in a small study of adults born extremely preterm, despite lower RV ejection fractions from cardiovascular MRI, use of right heart catheterization showed that RV-pulmonary vascular coupling was impaired in those born preterm (Mulchrone et al., 2020), perhaps the result of cardiopulmonary dysanapsis. Given the much lower gestational age of the preterm group in this latter study, it is possible that early RV-pulmonary vascular uncoupling may be gestational age dependent and impacted by the presence of BPD and other perinatal complications. Additionally, the smaller LV cavity dimension and reduced resting diastology of the left heart that has been consistently observed in adults born preterm (Telles et al., 2020) may also lead to elevated left atrial and pulmonary venous pressure. Consequently, the exaggerated increase in pulmonary pressure with exercise in preterm subjects compared to term controls could lead to exercise-induced pulmonary hypertension as defined by Herve and colleagues (Herve et al., 2015). The resulting excessive right heart work combined with impaired RV-pulmonary vascular coupling could contribute to exercise intolerance (Fowler et al., 2011).
Animal studies, i.e., in thoroughbred horses, have established a strong relationship between pulmonary trans-capillary pressure and stress failure of the pulmonary capillaries (West et al., 1993). There is limited research in humans, but repetitive bouts of intensive exercise with large cardiac outputs under conditions of increased pulmonary vascular resistance (e.g., altitude) results in pulmonary capillary rupture (Eldridge et al., 2006). These studies suggest that adults born preterm could be at an increased risk of excessive pulmonary pressures during exercise with subsequent lung damage. For example, the lungs of thoroughbred horses with a history of repeated pulmonary capillary hemorrhage demonstrate significant vascular remodeling in the pulmonary veins (Williams et al., 2013). Thus, repeated injury in adults born preterm that results in vascular remodeling (Stenmark & Mecham, 1997), may result in an increased risk of pulmonary hypertension. Additional work in preclinical animal models of preterm birth and BPD are needed to test this supposition.
As highlighted in the previous sections, many of the aforementioned studies in adults born preterm include, mostly, those subjects with mild-to-moderate severity lung disease. Thus, those with more severe lung disease may have overt resting pulmonary hypertension and/or further exaggerated responses under more dynamic conditions like exercise. Accordingly, direct measures of pulmonary arterial and left atrial pressures at iso-cardiac outputs are required to determine whether or not pulmonary vascular resistance is greater in preterm subjects. Complimentary imaging of cardiac structure and function under these iso-cardiac output conditions would also greatly enhance our understanding of the cardiopulmonary coupling in preterm adults, especially if performed in larger cohorts that include preterm adults with more severe lung disease.
Cardiovascular Responses and Limitations to Exercise
A recent meta-analysis revealed that preterm birth is associated with significant functional and structural cardiac impairments across developmental stages (Telles et al., 2020). This includes RV systolic impairments, lower LV diastolic function that worsens with age, an accelerated rate of LV hypertrophy, and smaller ventricular dimensions. Cardiovascular MRI has revealed that reduced ventricular length is the main dimensional determinant of reduced end-diastolic volume in adults born preterm (Lewandowski et al., 2013a, 2013b). The emergence of these cardiac alterations during infancy, rather than during fetal development (Aye et al., 2017), supports the hypothesis that early exposure to the postnatal environment, including the hemodynamic disruption of the immature myocardium and postnatal hyperoxia exposure triggers altered cardiac remodeling (Bensley et al., 2010; Bertagnolli et al., 2018a) and may cause a reduction in their myocardial functional reserve.
In support of this, exercise stress echocardiography data show that LV ejection fraction was lower in adults born moderately preterm compared to term-born controls during moderate- and high-intensity exercise, with lower cardiac index emerging from low intensities (Huckstep et al., 2018). This impairment in LV ejection fraction elevation during exercise was shown to be a significant predictor of the lower exercise capacity and slower heart rate recovery in adolescents and adults born preterm (Huckstep et al., 2020). Similar cardiac functional impairments may exist in adults born extremely preterm, with a reduced ability to increase RV stroke volume and RV cardiac index during moderate intensity exercise (Goss et al., 2018). This finding did not reach statistical significance (p = 0.07), but it is worthwhile to keep in mind that these were highly invasive studies, i.e., right heart catheterization, undertaken in a relatively small sample size (n = 11 preterm) (Goss et al., 2018). Though further work is needed to explore causal pathways, this reduction in myocardial functional reserve may make them more susceptible to heart failure and push them into a chronic cardiovascular disease state when faced with significant acute and chronic stressors, such as hypertension (Leeson & Lewandowski, 2017).
This reduction in myocardial functional reserve makes the added increased risk of hypertension in individuals born preterm (Crump et al., 2020) of particular concern. While elevated resting blood pressure in individuals born preterm is evident as early as childhood (Parkinson et al., 2013), the underlying mechanisms remain unknown. Evidence from animal and human studies has shown that preterm birth impairs normal vascular development (Norman, 2008; Nuyt & Alexander, 2009). Interrupted vascular tree development, including vessel structure and function, has been shown to be central to retinopathy of prematurity (Sapieha et al., 2010), BPD (Thébaud et al., 2019), and other prematurity-related complications (Lewandowski & Leeson, 2014). The lumen and length of the aorta is smaller in individuals born preterm compared to those born at term (Boardman et al., 2015), which may be due to the reduction in preterm early postnatal aortic growth compared to normal intrauterine development (Schubert et al., 2011). Further to this, the in vitro proliferative capacity of endothelial progenitor cells collected from neonatal cord blood (Ligi et al., 2011) and from adult venous blood of individuals born preterm has been shown to be reduced (Bertagnolli et al., 2018b). An imbalance in angiogenic molecular pathways within the systemic circulation is further supported by studies in adults born preterm (Lewandowski et al., 2015). Levels of antiangiogenic circulating blood markers, specifically soluble endoglin, are elevated in preterm-born adults and associate with an increase in systolic blood pressure, which is mediated by systemic capillary rarefaction (Lewandowski et al., 2015).
Blood pressure alterations in preterm-born adults exist both at rest and during exercise. Huckstep et al. (Huckstep et al., 2018) demonstrated that diastolic blood pressure during peak exercise was greater in normotensive adults born preterm than term-born controls. Similarly, Barnard et al. (Barnard et al., 2020) used invasive arterial measures to assess blood pressure responses to exercise under normoxic and hypoxic conditions. They found higher systolic, diastolic, and pulse pressures in adults born preterm compared to term-born controls throughout normoxic exercise. During hypoxic exercise, diastolic blood pressure declined in the preterm group only, suggestive of impaired vascular reactivity. Alterations to the autonomic nervous system may play a role in the development of the preterm hypertensive phenotype as the sympathetic nervous system is overactive, with a lesser parasympathetic tone, in infants (Smith et al., 2005). Although long-term data on the autonomic nervous system is limited in the preterm population, preterm-born adolescents have been shown to have reductions in parasympathetic tone compared to term-born adolescents that may affect blood pressure regulation (Haraldsdottir et al., 2018).
While it is evident that potentially adverse cardiac changes exist in those born preterm, larger studies including longitudinal repeated measures, i.e., echocardiography, systemic and skeletal muscle vascular resistance, etc., are needed to further explore these findings, the underlying mechanisms, as well as cardiovascular coupling during exercise. We suggest these cardiovascular responses, i.e., a smaller heart yet greater blood pressure, represents dysanapsis within the cardiovascular system.
SUMMARY AND FUTURE DIRECTIONS
Advances in neonatal care have allowed for improved survival rates, which means that the population of adults born preterm will continue to increase. Thus, gaining a better understanding of the long-term health outcomes associated with preterm birth is of significant importance. Specifically, obtaining a complete understanding of the systems physiologic aspects that underlie the impaired respiratory, cardiopulmonary, and cardiovascular function in these survivors is important to understand why their exercise capacity is reduced. It would also be valuable to understand the developmental basis for cardiopulmonary dysanapsis and whether discordant growth of the heart and lung contributes to cardiopulmonary sequelae of adults after preterm birth. There exists some degree of dysanapsis within the cardiopulmonary systems of all individuals regardless of birth status. However, we suggest that dysanapsis across multiple systems is exaggerated in those born preterm. It is possible that this is an underlying explanation for much of the altered physiology in adults born preterm.
Another interesting consideration and future direction, involves the use of assisted reproductive technology (ART). Individuals born via ART are at a higher risk of late impairments in cardiovascular and respiratory function in childhood up to young adulthood (Scherrer et al., 2012; Guo et al., 2017). Many of these changes mirror what is seen in the preterm population. Additionally, the risk of preterm delivery risk is up to 80% higher with ART compared to naturally occurring pregnancies (Cavoretto et al., 2018). Further work is warranted but, the present findings add further support to the concept that antenatal and perinatal factors can have life-long consequences for late structural and physiological limitations of the cardiovascular and respiratory systems.
Additionally, more work is needed to characterize the impact of normal age-associated decline in respiratory and cardiovascular function in individuals born preterm. This is particularly true as an important consideration for future work is that of the long-term outcomes of adults born preterm (Duke et al., 2020; Hurst et al., 2020). In general, the lungs are not believed to be a limiting factor to aerobic exercise capacity in healthy older individuals (Duke & Lovering, 2021), but this may not be the case in individuals born preterm who have a lower peak lifetime pulmonary function. Additionally, with underlying impaired cardiovascular function in young adulthood, the aged preterm individual may have mechanisms of exercise impairment similar to a COPD patient (Amann et al., 2010), though further work is needed to investigate this. To fill this important gap, continuation of important longitudinal work beyond early adulthood is critical. Specifically, it would be valuable to perform the same measurements using the same methods in the same individuals over time, such that direct comparisons can be made. Likewise, studies of preterms that include the entire spectrum of lung disease severities and gestational ages are needed to begin establishing the characteristics of this population to help guide perinatal clinical practice.
Acknowledgements:
The authors express their gratitude to Jyotika Erram, MS for her assistance with figure preparation.
Funding:
The authors’ work reported in this manuscript was funded in part by the Medical Research Foundation of Oregon Early Clinical Investigator Award (JWD), Ohio University Research Committee Award (JWD), the National Institutes of Health (R15HL148850) (JWD), AJL is funded by a British Heart Foundation Intermediated Research Fellowship (FS/18/3/33292), the National Institutes of Health (R01HL145679) (SHA), the American Heart Association Scientist Development Grant (#2280238) (ATL), and the Giles F. Filley Memorial Award for Excellence in Respiratory Physiology and Medicine (ATL).
Footnotes
Competing Interests: The authors have no conflicts of interest.
For preclinical studies, comparisons always refer to BPD versus control animals; for clinical autopsy studies, it is BPD versus preterms who have died without BPD for comparisons.
REFENCES
- Abman SH et al. (2017a). Interdisciplinary Care of Children with Severe Bronchopulmonary Dysplasia. J Pediatrics 181, 12–28.e1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Abman SH, Bancalari E & Jobe A (2017b). The Evolution of Bronchopulmonary Dysplasia after 50 Years. Am J Respir Crit Care Med 195, 421–424. [DOI] [PubMed] [Google Scholar]
- Abman SH, Wolfe RR, Accurso FJ, Koops BL, Bowman CM & Wiggins JW (1985). Pulmonary vascular response to oxygen in infants with severe bronchopulmonary dysplasia. Pediatrics 75, 80–84. [PubMed] [Google Scholar]
- Aliverti A, Stevenson N, Dellacà RL & Mauro AL (2004). Regional chest wall volumes during exercise in chronic obstructive pulmonary disease. Thorax 59, 210–216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Amann M, Eldridge MW, Lovering AT, Stickland MK, Pegelow DF & Dempsey JA (2006). Arterial oxygenation influences central motor output and exercise performance via effects on peripheral locomotor muscle fatigue in humans. J Physiol 575, 937–952. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Amann M, Regan MS, Kobitary M, Eldridge MW, Boutellier U, Pegelow DF & Dempsey JA (2010). Impact of pulmonary system limitations on locomotor muscle fatigue in patients with COPD. Am J Physiol Regu Integr Comp Physiol 299, R314–R324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Assad TR, Maron BA, Robbins IM, Xu M, Huang S, Harrell FE, Farber-Eger EH, Wells QS, Choudhary G, Hemnes AR & Brittain EL (2017). Prognostic Effect and Longitudinal Hemodynamic Assessment of Borderline Pulmonary Hypertension. JAMA Cardiol 2, 1361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aukland SM, Rosendahl K, Owens CM, Fosse KR, Eide GE & Halvorsen T (2009). Neonatal bronchopulmonary dysplasia predicts abnormal pulmonary HRCT scans in long-term survivors of extreme preterm birth. Thorax 64, 405–410. [DOI] [PubMed] [Google Scholar]
- Aye CYL, Lewandowski AJ, Lamata P, Upton R, Davis E, Ohuma EO, Kenworthy Y, Boardman H, Wopperer S, Packham A, Adwani S, McCormick K, Papageorghiou AT & Leeson P (2017). Disproportionate cardiac hypertrophy during early postnatal development in infants born preterm. Pediatr Res 82, 36–46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Babb TG, Beck KC & Johnson BD (2012). Dysanapsis: importance of measured lung static recoil pressure. Med Sci Sports Exerc 44, 1194. [DOI] [PubMed] [Google Scholar]
- Badesch DB, Raskob GE, Elliott CG, Krichman AM, Farber HW, Frost AE, Barst RJ, Benza RL, Liou TG, Turner M, Giles S, Feldkircher K, Miller DP & McGoon MD (2010). Pulmonary Arterial Hypertension Baseline Characteristics From the REVEAL Registry. Chest 137, 376–387. [DOI] [PubMed] [Google Scholar]
- Balinotti JE, Chakr VC, Tiller C, Kimmel R, Coates C, Kisling J, Yu Z, Nguyen J & Tepper RS (2010). Growth of lung parenchyma in infants and toddlers with chronic lung disease of infancy. Am J Resp Crit Care 181, 1093–1097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barnard CR, Peters M, Sindler AL, Farrell ET, Baker KR, Palta M, Stauss HM, Dagle JM, Segar J, Pierce GL, Eldridge MW & Bates ML (2020). Increased aortic stiffness and elevated blood pressure in response to exercise in adult survivors of prematurity. Physiol Rep 8, e14462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bensley JG, Stacy VK, Matteo RD, Harding R & Black MJ (2010). Cardiac remodelling as a result of pre-term birth: implications for future cardiovascular disease. Eur Heart J 31, 2058–2066. [DOI] [PubMed] [Google Scholar]
- Bertagnolli M, Huyard F, Cloutier A, Anstey Z, Huot-Marchand J-É, Fallaha C, Paradis P, Schiffrin EL, deBlois D & Nuyt AM (2018a). Transient Neonatal High Oxygen Exposure Leads to Early Adult Cardiac Dysfunction, Remodeling, and Activation of the Renin–Angiotensin System. Hypertension 63, 143–150. [DOI] [PubMed] [Google Scholar]
- Bertagnolli M, Xie LF, Paquette K, He Y, Cloutier A, Fernandes RO, Béland C, Sutherland MR, Delfrate J, Curnier D, Bigras J, Rivard A, Thébaud B, Luu TM & Nuyt AM (2018b). Endothelial Colony-Forming Cells in Young Adults Born Preterm: A Novel Link Between Neonatal Complications and Adult Risks for Cardiovascular Disease. J Am Heart Assoc 7, e009720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bhatt AJ, Pryhuber GS & Huyck H (2001). Disrupted pulmonary vasculature and decreased vascular endothelial growth factor, Flt-1, and TIE-2 in human infants dying with bronchopulmonary dysplasia. Am J Respir Crit Care Med 164, 1971–1980. [DOI] [PubMed] [Google Scholar]
- Bland RD, Albertine KH, Pierce RA, Starcher BC & Carlton DP (2003). Impaired alveolar development and abnormal lung elastin in preterm lambs with chronic lung injury: potential benefits of retinol treatment. Biol Neonate 84, 101–102. [DOI] [PubMed] [Google Scholar]
- Boardman H, Birse K, Davis EF, Whitworth P, Aggarwal V, Lewandowski AJ & Leeson P (2015). Comprehensive multi-modality assessment of regional and global arterial structure and function in adults born preterm. Hypertens Res 39, 39–45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bolton CE, Bush A, Hurst JR, Kotecha S & McGarvey L (2015). Lung consequences in adults born prematurely. Thorax 70, 574–580. [DOI] [PubMed] [Google Scholar]
- Bui DS et al. (2018). Childhood predictors of lung function trajectories and future COPD risk: a prospective cohort study from the first to the sixth decade of life. Lancet Respir Med 6, 535–544. [DOI] [PubMed] [Google Scholar]
- Cardoen F, Vermeulen F, Proesmans M, Moens M & Boeck KD (2019). Lung function evolution in children with old and new type bronchopulmonary dysplasia: a retrospective cohort analysis. Eur J Pediatr 178, 1859–1866. [DOI] [PubMed] [Google Scholar]
- Carr H, Cnattingius S, Granath F, Ludvigsson JF & Bonamy A-KE (2017). Preterm Birth and Risk of Heart Failure Up to Early Adulthood. J Am Coll Cardiol 69, 2634–2642. [DOI] [PubMed] [Google Scholar]
- Caskey S, Gough A, Rowan S, Gillespie S, Clarke J, Riley M, Megarry J, Nicholls P, Patterson C, Halliday HL, Shields MD & McGarvey L (2016). Structural and Functional Lung Impairment in Adult Survivors of Bronchopulmonary Dysplasia. Ann Am Thorac Soc 13, 1262–1270. [DOI] [PubMed] [Google Scholar]
- Cavoretto P, Candiani M, Giorgione V, Inversetti A, Abu-Saba MM, Tiberio F, Sigismondi C & Farina A (2018). Risk of spontaneous preterm birth in singleton pregnancies conceived after IVF/ICSI treatment: meta-analysis of cohort studies. Ultrasound Obst Gyn 51, 43–53. [DOI] [PubMed] [Google Scholar]
- Clemm HH, Vollsæter M, Røksund OD, Eide GE, Markestad T & Halvorsen T (2014). Exercise capacity after extremely preterm birth. Development from adolescence to adulthood. Ann Am Thorac Soc 11, 537–545. [DOI] [PubMed] [Google Scholar]
- Coalson JJ, Winter V & deLemos RA (1995). Decreased alveolarization in baboon survivors with bronchopulmonary dysplasia. Am J Respir Crit Care Med 152, 640–646. [DOI] [PubMed] [Google Scholar]
- Cross TJ, Gideon EA, Morris SJ, Coriell CL, Hubbard CD & Duke JW (2021). A comparison of methods used to quantify the work of breathing during exercise. J Appl Physiol 131, 1123–1133. [DOI] [PubMed] [Google Scholar]
- Crump C, Groves A, Sundquist J & Sundquist K (2021). Association of Preterm Birth With Long-term Risk of Heart Failure Into Adulthood. JAMA Pediatr 175, 689–697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crump C, Howell EA, Stroustrup A, McLaughlin MA, Sundquist J & Sundquist K (2019a). Association of Preterm Birth With Risk of Ischemic Heart Disease in Adulthood. JAMA Pediatr 173, 736. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crump C, Sundquist J & Sundquist K (2020). Risk of hypertension into adulthood in persons born prematurely: a national cohort study. Eur Heart J 41, 1542–1550. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crump C, Winkleby MA, Sundquist J & Sundquist K (2019b). Prevalence of Survival Without Major Comorbidities Among Adults Born Prematurely. JAMA 322, 1580–1588. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dahl MJ, Bowen S, Aoki T, Rebentisch A, Dawson E, Pettet L, Emerson H, Yu B, Wang Z, Yang H, Zhang C, Presson AP, Joss-Moore L, Null DM, Yoder BA & Albertine KH (2018). Former-preterm lambs have persistent alveolar simplification at 2 and 5 months corrected postnatal age. Am J Physiol Lung Cell Mol Physiol 315, L816–L833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davidson LM & Berkelhamer SK (2017). Bronchopulmonary Dysplasia: Chronic Lung Disease of Infancy and Long-Term Pulmonary Outcomes. J Clin Med 6, 4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Debevec T, Pialoux V, Millet GP, Martin A, Mramor M & Osredkar D (2019). Exercise Overrides Blunted Hypoxic Ventilatory Response in Prematurely Born Men. Front Physiol 10, 437. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dempsey JA & Wagner PD (1999). Exercise-induced arterial hypoxemia. J Appl Physiol 87, 1997–2006. [DOI] [PubMed] [Google Scholar]
- Dominelli PB, Archiza B, Ramsook AH, Mitchell RA, Peters CM, Molgat-Seon Y, Henderson WR, Koehle MS, Boushel R & Sheel AW (2017). Effects of respiratory muscle work on respiratory and locomotor blood flow during exercise. Exp Physiol 102, 1535–1547. [DOI] [PubMed] [Google Scholar]
- Dominelli PB, Guenette JA, Wilkie SS, Foster GE & Sheel AW (2011). Determinants of expiratory flow limitation in healthy women during exercise. Med Sci Sports Exerc 43, 1666–1674. [DOI] [PubMed] [Google Scholar]
- Doyle LW, Adams A-M, Robertson C, Ranganathan S, Davis NM, Lee KJ, Cheong JL & Group VICS (2017). Increasing airway obstruction from 8 to 18 years in extremely preterm/low-birthweight survivors born in the surfactant era. Thorax 72, 712–719. [DOI] [PubMed] [Google Scholar]
- Duke JW, Elliott JE, Laurie SS, Beasley KM, Mangum TS, Hawn JA, Gladstone IM & Lovering AT (2014). Pulmonary gas exchange efficiency during exercise breathing normoxic and hypoxic gas in adults born very preterm with low diffusion capacity. J Appl Physiol 117, 473–481. [DOI] [PubMed] [Google Scholar]
- Duke JW, Gladstone IM, Sheel AW & Lovering AT (2018). Premature birth affects the degree of airway dysanapsis and mechanical ventilatory constraints. Exp Physiol 103, 261–275. [DOI] [PubMed] [Google Scholar]
- Duke JW & Lovering AT (2021). Exercise and Physical Activity for Older Adults. In Pulmonary Changes, 1st edn., ed. Bouchard DR, pp. 97–110. Human Kinetics, Champaign, IL, USA. [Google Scholar]
- Duke JW, Lovering AT & Goss KN (2020). Premature Aging and Increased Risk of Adult Cardiorespiratory Disease after Extreme Preterm Birth. Getting to the Heart (and Lungs) of the Matter. Am J Resp Crit Care 202, 319–320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Duke JW, Zidron AM, Gladstone IM & Lovering AT (2019). Alleviating mechanical constraints to ventilation with heliox improves exercise endurance in adult survivors of very preterm birth. Thorax 74, 302–304. [DOI] [PubMed] [Google Scholar]
- Eldridge MW, Braun RK, Yoneda KY & Walby WF (2006). Effects of altitude and exercise on pulmonary capillary integrity: evidence for subclinical high-altitude pulmonary edema. J Appl Physiol 100, 972–980. [DOI] [PubMed] [Google Scholar]
- Elliott JE, Duke JW, Hawn JA, Halliwill JR & Lovering AT (2014). Increased cardiac output, not pulmonary artery systolic pressure, increases intrapulmonary shunt in healthy humans breathing room air and 40% O2. J Physiol 592, 4537–4553. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eves ND, Petersen SR, Haykowsky MJ, Wong EY & Jones RL (2006). Helium-Hyperoxia, Exercise, and Respiratory Mechanics in Chronic Obstructive Pulmonary Disease. Am J Respir Crit Care Med 174, 763–771. [DOI] [PubMed] [Google Scholar]
- Farrell ET, Bates ML, Pegelow DF, Palta M, Eickhoff JC, O’Brien MJ & Eldridge MW (2015). Pulmonary Gas Exchange and Exercise Capacity in Adults Born Preterm. Ann Am Thorac Soc 12, 1130–1137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fowler RM, Maiorana AJ, Jenkins SC, Gain KR, O’Driscoll G & Gabbay E (2011). Implications of exercise-induced pulmonary arterial hypertension. Med Sci Sports Exerc 43, 983–989. [DOI] [PubMed] [Google Scholar]
- Galambos C, Sims-Lucas S & Abman SH (2013). Histologic evidence of intrapulmonary anastomoses by three-dimensional reconstruction in severe bronchopulmonary dysplasia. Ann Am Thorac Soc 10, 474–481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Galiè N, Humbert M, Vachiery JL, Gibbs S, Lang I, Torbicki A, Simonneau G, Peacock A, Vonk Noordegraaf A, Beghetti M, Ghofrani A, Gomez Sanchez MA, Hansmann G, Klepetko W, Lancellotti P, Matucci M, McDonagh T, Pierard LA, Trindade PT, Zompatori M, & Hoeper M. (2016). 2015 ESC/ERS Guidelines for the diagnosis and treatment of pulmonary hypertension. Eur Heart J 37, 67–119. [DOI] [PubMed] [Google Scholar]
- Gibson A-M & Doyle LW (2014). Respiratory outcomes for the tiniest or most immature infants. Semin Fetal Neonatal Med 19, 105–111. [DOI] [PubMed] [Google Scholar]
- Gibson A-M, Reddington C, McBride L, Callanan C, Robertson C & Doyle LW (2015). Lung function in adult survivors of very low birth weight, with and without bronchopulmonary dysplasia. Pediatr Pulmonol 50, 987–994. [DOI] [PubMed] [Google Scholar]
- Gideon EA, Cross TJ, Coriell CL & Duke JW (2021). The effect of estimating chest wall compliance on the work of breathing during exercise as determined via the modified Campbell diagram. Am J Physiol Regu Integr Comp Physiol 320, R268–R275. [DOI] [PubMed] [Google Scholar]
- Goss KN, Beshish AG, Barton GP, Haraldsdottir K, Levin TS, Tetri LH, Battiola TJ, Mulchrone AM, Pegelow DF, Palta M, Lamers LJ, Watson AM, Chesler NC & Eldridge MW (2018). Early Pulmonary Vascular Disease in Young Adults Born Preterm. Am J Respir Crit Care Med 198, 1549–1558. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guenette JA, Webb KA & O’Donnell DE (2012). Does dynamic hyperinflation contribute to dyspnoea during exercise in patients with COPD? Eur Respir J 40, 322–329. [DOI] [PubMed] [Google Scholar]
- Gulati M, Pandey DK, Arnsdorf MF, Lauderdale DS, Thisted RA, Wicklund RH, Al-Hani AJ & Black HR (2003). Exercise Capacity and the Risk of Death in Women. Circulation 108, 1554–1559. [DOI] [PubMed] [Google Scholar]
- Guo X-Y, Liu X-M, Jin L, Wang T-T, Ullah K, Sheng J-Z & Huang H-F (2017). Cardiovascular and metabolic profiles of offspring conceived by assisted reproductive technologies: a systematic review and meta-analysis. Fertil Steril 107, 622–631.e5. [DOI] [PubMed] [Google Scholar]
- Halvorsen T, Skadberg BT, Eide GE, Røksund O, Aksnes L & Øymar K (2005). Characteristics of asthma and airway hyper-responsiveness after premature birth. Pediatr Allergy Immunol 16, 487–494. [DOI] [PubMed] [Google Scholar]
- Halvorsen T, Skadberg BT, Eide GE, Røksund OD, Carlsen KH & Bakke P (2004). Pulmonary outcome in adolescents of extreme preterm birth: a regional cohort study. Acta Paediatr 93, 1294–1300. [PubMed] [Google Scholar]
- Hansmann G, Fernandez-Gonzalez A, Aslam M, Vitali SH, Martin T, Mitsialis SA & Kourembanas S (2012). Mesenchymal stem cell-mediated reversal of bronchopulmonary dysplasia and associated pulmonary hypertension. Pulm Circ 2, 170–181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haraldsdottir K, Watson AM, Beshish AG, Pegelow DF, Palta M, Tetri LH, Brix MD, Centanni RM, Goss KN & Eldridge MW (2019). Heart rate recovery after maximal exercise is impaired in healthy young adults born preterm. Eur J Appl Physiol 119, 857–866. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haraldsdottir K, Watson AM, Goss KN, Beshish AG, Pegelow DF, Palta M, Tetri LH, Barton GP, Brix MD, Centanni RM & Eldridge MW (2018). Impaired autonomic function in adolescents born preterm. Physiol Rep 6, e13620. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harms CA, Wetter TJ, Croix CMS, Pegelow DF & Dempsey JA (2000). Effects of respiratory muscle work on exercise performance. J Appl Physiol 89, 131–138. [DOI] [PubMed] [Google Scholar]
- Harms CA, Wetter TJ, McClaran SR, Pegelow DF, Nickele GA, Nelson WB, Hanson P & Dempsey JA (1998). Effects of respiratory muscle work on cardiac output and its distribution during maximal exercise. J Appl Physiol 85, 609–618. [DOI] [PubMed] [Google Scholar]
- Herve P, Lau EM, Sitbon O, Savale L, Montani D, Godinas L, Lador F, Jaïs X, Parent F, Günther S, Humbert M, Simonneau G & Chemla D (2015). Criteria for diagnosis of exercise pulmonary hypertension. Eur Respir J 46, 728–737. [DOI] [PubMed] [Google Scholar]
- Hirata K, Nishihara M, Kimura T, Shiraishi J, Hirano S, Kitajima H & Fujimura M (2017). Longitudinal impairment of lung function in school-age children with extremely low birth weights: Early Lung Function Deterioration in ELBW Children. Pediatr Pulm 52, 779–786. [DOI] [PubMed] [Google Scholar]
- vom Hove M, Prenzel F, Uhlig HH & Robel-Tillig E (2014). Pulmonary Outcome in Former Preterm, Very Low Birth Weight Children with Bronchopulmonary Dysplasia: A Case-Control Follow-Up at School Age. J Pediatr 164, 40–45.e4. [DOI] [PubMed] [Google Scholar]
- Huckstep OJ, Burchert H, Williamson W, Telles F, Tan CMJ, Bertagnolli M, Arnold L, Mohamed A, McCormick K, Hanssen H, Leeson P & Lewandowski AJ (2020). Impaired myocardial reserve underlies reduced exercise capacity and heart rate recovery in preterm-born young adults. European Hear J Cardiovasc Imaging 22, 572–580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huckstep OJ, Williamson W, Telles F, Burchert H, Bertagnolli M, Herdman C, Arnold L, Smillie R, Mohamed A, Boardman H, McCormick K, Neubauer S, Leeson P & Lewandowski AJ (2018). Physiological Stress Elicits Impaired Left Ventricular Function in Preterm-Born Adults. J Am Coll Cardiol 71, 1347–1356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Humbert M, Sitbon O, Chaouat A, Bertocchi M, Habib G, Gressin V, Yaici A, Weitzenblum E, Cordier J-F, Chabot F, Dromer C, Pison C, Reynaud-Gaubert M, Haloun A, Laurent M, Hachulla E & Simonneau G (2012). Pulmonary Arterial Hypertension in France. Am J Resp Crit Care 173, 1023–1030. [DOI] [PubMed] [Google Scholar]
- Hurst JR, Beckmann J, Ni Y, Bolton CE, McEniery CM, Cockcroft JR & Marlow N (2020). Respiratory and Cardiovascular Outcomes in Survivors of Extremely Preterm Birth at 19 Years. Am J Resp Crit Care; DOI: 10.1164/rccm.202001-0016oc. [DOI] [PMC free article] [PubMed]
- Islam JY, Keller RL, Aschner JL, Hartert TV & Moore PE (2015). Understanding the Short- and Long-Term Respiratory Outcomes of Prematurity and Bronchopulmonary Dysplasia. Am J Respir Crit Care Med 192, 134–156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jacob SV, Coates AL, Lands LC & MacNeish CF (1998). Long-term pulmonary sequelae of severe bronchopulmonary dysplasia. J Pediatr 133, 193–200. [DOI] [PubMed] [Google Scholar]
- Jobe AH & Bancalari E (2001). Bronchopulmonary dysplasia. Am J Respir Crit Care Med 163, 1723–1729. [DOI] [PubMed] [Google Scholar]
- Johnson BD, Weisman IM, Zeballos RJ & Beck KC (1999). Emerging concepts in the evaluation of ventilatory limitation during exercise: the exercise tidal flow-volume loop. Chest 116, 488–503. [DOI] [PubMed] [Google Scholar]
- Kennedy J (1999). Lung function outcome in children of premature birth: Lung function outcome in children of premature birth. J Paediatr Child H 35, 516–521. [DOI] [PubMed] [Google Scholar]
- Koumbourlis AC, Motoyama EK, Mutich RL, Mallory GB, Walczak SA & Fertal K (1996). Longitudinal follow-up of lung function from childhood to adolescence in prematurely born patients with neonatal chronic lung disease. Pediatr Pulmonol 21, 28–34. [DOI] [PubMed] [Google Scholar]
- Kovacs G, Berghold A, Scheidl S & Olschewski H (2009). Pulmonary arterial pressure during rest and exercise in healthy subjects: a systematic review. Eur Respir J 34, 888–894. [DOI] [PubMed] [Google Scholar]
- Kovacs G, Olschewski A, Berghold A & Olschewski H (2012). Pulmonary vascular resistances during exercise in normal subjects: a systematic review. Eur Respir J 39, 319–328. [DOI] [PubMed] [Google Scholar]
- Laurie SS, Elliott JE, Beasley KM, Mangum TS, Goodman RD, Duke JW, Gladstone IM & Lovering AT (2018). Exaggerated Increase in Pulmonary Artery Pressure during Exercise in Adults Born Preterm. Am J Resp Crit Care 197, 821–823. [DOI] [PubMed] [Google Scholar]
- Leeson P & Lewandowski AJ (2017). A New Risk Factor for Early Heart Failure: Preterm Birth. J Am Coll Cardiol 69, 2643–2645. [DOI] [PubMed] [Google Scholar]
- Lewandowski AJ, Augustine D, Lamata P, Davis EF, Lazdam M, Francis J, McCormick K, Wilkinson AR, Singhal A, Lucas A, Smith NP, Neubauer S & Leeson P (2013a). Preterm heart in adult life: cardiovascular magnetic resonance reveals distinct differences in left ventricular mass, geometry, and function. Circulation 127, 197–206. [DOI] [PubMed] [Google Scholar]
- Lewandowski AJ, Bradlow WM, Augustine D, Davis EF, Francis J, Singhal A, Lucas A, Neubauer S, McCormick K & Leeson P (2013b). Right ventricular systolic dysfunction in young adults born preterm. Circulation 128, 713–720. [DOI] [PubMed] [Google Scholar]
- Lewandowski AJ, Davis EF, Yu G, Digby JE, Boardman H, Whitworth P, Singhal A, Lucas A, McCormick K, Shore AC & Leeson P (2015). Elevated blood pressure in preterm-born offspring associates with a distinct antiangiogenic state and microvascular abnormalities in adult life. Hypertension 65, 607–614. [DOI] [PubMed] [Google Scholar]
- Lewandowski AJ & Leeson P (2014). Preeclampsia, prematurity and cardiovascular health in adult life. Early Hum Dev 90, 725–729. [DOI] [PubMed] [Google Scholar]
- Ligi I, Simoncini S, Tellier E, Vassallo PF, Sabatier F, Guillet B, Lamy E, Sarlon G, Quemener C, Bikfalvi A, Marcelli M, Pascal A, Dizier B, Simeoni U, Dignat-George F & Anfosso F (2011). A switch toward angiostatic gene expression impairs the angiogenic properties of endothelial progenitor cells in low birth weight preterm infants. Blood 118, 1699–1709. [DOI] [PubMed] [Google Scholar]
- Louvaris Z, Zakynthinos S, Aliverti A, Habazettl H, Vasilopoulou M, Andrianopoulos V, Wagner H, Wagner P & Vogiatzis I (2012). Heliox increases quadriceps muscle oxygen delivery during exercise in COPD patients with and without dynamic hyperinflation. J Appl Physiol 113, 1012–1023. [DOI] [PubMed] [Google Scholar]
- Lovering AT, Elliott JE, Laurie SS, Beasley KM, Gust CE, Mangum TS, Gladstone IM & Duke JW (2014). Ventilatory and sensory responses in adult survivors of preterm birth and bronchopulmonary dysplasia with reduced exercise capacity. Ann Am Thorac Soc 11, 1528–1537. [DOI] [PubMed] [Google Scholar]
- Lovering AT, Haverkamp HC & Eldridge MW (2005). Responses and limitations of the respiratory system to exercise. Clin Chest Med 26, 439–57–vi. [DOI] [PubMed] [Google Scholar]
- Lovering AT, Laurie SS, Elliott JE, Beasley KM, Yang X, Gust CE, Mangum TS, Goodman RD, Hawn JA & Gladstone IM (2013). Normal pulmonary gas exchange efficiency and absence of exercise-induced arterial hypoxemia in adults with bronchopulmonary dysplasia. J Appl Physiol 115, 1050–1056. [DOI] [PubMed] [Google Scholar]
- Lovering AT, Romer LM, Haverkamp HC, Hokanson JS & Eldridge MW (2007). Excessive gas exchange impairment during exercise in a subject with a history of bronchopulmonary dysplasia and high altitude pulmonary edema. High Alt Med Biol 8, 62–67. [DOI] [PubMed] [Google Scholar]
- Loyd JE, Butler MG, Foroud TM, Conneally PM, JAP 3rd & Newman JH (1995). Genetic anticipation and abnormal gender ratio at birth in familial primary pulmonary hypertension. Am J Resp Crit Care 152, 93–97. [DOI] [PMC free article] [PubMed] [Google Scholar]
- MacLean JE, DeHaan K, Fuhr D, Hariharan S, Kamstra B, Hendson L, Adatia I, Majaesic C, Lovering AT, Thompson RB, Nicholas D, Thebaud B & Stickland MK (2016). Altered breathing mechanics and ventilatory response during exercise in children born extremely preterm. Thorax 71, 1012–1019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Malleske DT, Chorna O & Maitre NL (2018). Pulmonary sequelae and functional limitations in children and adults with bronchopulmonary dysplasia. Paediatr Respir Rev 26, 55–59. [DOI] [PubMed] [Google Scholar]
- Malmberg LP, Pelkonen A, Poussa T, Pohianpalo A, Haahtela T & Turpeinen M (2002). Determinants of respiratory system input impedance and bronchodilator response in healthy Finnish preschool children. Clin Physiol Funct Imaging 22, 64–71. [PubMed] [Google Scholar]
- Mandell E, Powers KN, Harral JW, Seedorf GJ, Hunter KS, Abman SH & Dodson RB (2015). Intrauterine endotoxin-induced impairs pulmonary vascular function and right ventricular performance in infant rats and improvement with early vitamin D therapy. Am J Physiol Lung Cell Mol Physiol 309, L1438–L1446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Margraf inda R, JFT Jr., Bruce MC & Dahms BB (1991). Morphometric Analysis of the Lung in Bronchopulmonary Dysplasia. Am Rev Respir Dis 143, 391–400. [DOI] [PubMed] [Google Scholar]
- Maron BA et al. (2016). Association of Borderline Pulmonary Hypertension With Mortality and Hospitalization in a Large Patient Cohort: Insights From the Veterans Affairs Clinical Assessment, Reporting, and Tracking Program. Circulation 133, 1240–1248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martinez FD (2016). Lung growth through the “life course” and predictors and determinants of chronic respiratory disorders. In Fetal and neonatal lung development. Clinical correlates and technologies for the future, ed. Jobe AH, Whitsett JA & Abman SH, pp. 286–302. Cambridge University Press, New York, NY. [Google Scholar]
- Mazloum DE, Moschino L, Bozzetto S & Baraldi E (2014). Chronic lung disease of prematurity: long-term respiratory outcome. Neonatology 105, 352–356. [DOI] [PubMed] [Google Scholar]
- Mead J, Turner JM, Macklem PT & Little JB (1967). Significance of the relationship between lung recoil and maximum expiratory flow. J Appl Physiol 22, 95–108. [DOI] [PubMed] [Google Scholar]
- Mitchell SH & Teague WG (1998). Reduced Gas Transfer at Rest and during Exercise in School-age Survivors of Bronchopulmonary Dysplasia. Am J Resp Crit Care 157, 1406–1412. [DOI] [PubMed] [Google Scholar]
- Mohamed A, Lamata P, Williamson W, Alsharqi M, Tan CMJ, Burchert H, Huckstep OJ, Suriano K, Francis JM, Pelado JL, Monteiro C, Neubauer S, Levy PT, Leeson P & Lewandowski AJ (2020). Multimodality Imaging Demonstrates Reduced Right-Ventricular Function Independent of Pulmonary Physiology in Moderately Preterm-Born Adults. JACC Cardiovasc Imaging 13, 2046–2048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Molgat-Seon Y, Dominelli PB, Peters CM, Guenette JA, Sheel AW, Gladstone IM, Lovering AT & Duke JW (2019). Analysis of maximal expiratory flow-volume curves in adult survivors of preterm birth. Am J Phsyiol Regu Integr Comp Physiol 317, R588–R596. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morrow LA, Wagner BD, Ingram DA, Poindexter BB, Schibler K, Cotten CM, Dagle J, Sontag MK, Mourani PM & Abman SH (2017). Antenatal Determinants of Bronchopulmonary Dysplasia and Late Respiratory Disease in Preterm Infants. Am J Resp Crit Care 196, 364–374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mulchrone A, Bellofiore A, Douwes JM, Duong N, Beshish AG, Barton GP, Francois CJ, Eldridge MW, Goss KN & Chesler NC (2020). Impaired Right Ventricular-Vascular Coupling in Young Adults Born Preterm. Am J Resp Crit Care 201, 615–618. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Myers J, Prakash M, Froelicher V, Do D, Partington S & Atwood JE (2002). Exercise capacity and mortality among men referred for exercise testing. N Engl J Med 346, 793–801. [DOI] [PubMed] [Google Scholar]
- Narang I (2010). Review Series: What goes around, comes around: childhood influences on later lung health?: Long-term follow-up of infants with lung disease of prematurity. Chron Respir Dis 7, 259–269. [DOI] [PubMed] [Google Scholar]
- Narang I, Bush A & Rosenthal M (2009). Gas transfer and pulmonary blood flow at rest and during exercise in adults 21 years after preterm birth. Am J Respir Crit Care Med 180, 339–345. [DOI] [PubMed] [Google Scholar]
- Norman M (2008). Low birth weight and the developing vascular tree: a systematic review. Acta Paediatr 1992 97, 1165–1172. [DOI] [PubMed] [Google Scholar]
- Northway WH, Moss RB, Carlisle KB, Parker BR, Popp RL, Pitlick PT, Eichler I, Lamm RL & Brown BW (1990). Late pulmonary sequelae of bronchopulmonary dysplasia. N Engl J Med 323, 1793–1799. [DOI] [PubMed] [Google Scholar]
- Northway WH & Rosan RC (1967). Pulmonary disease following respirator therapy of hyaline-membrane disease: bronchopulmonary dysplasia. N Engl J Med 276, 357–368. [DOI] [PubMed] [Google Scholar]
- Nuyt AM & Alexander BT (2009). Developmental programming and hypertension. Curr Opin Nephrol Hy 18, 144–152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parker TA & Abman SH (2003). The pulmonary circulation in bronchopulmonary dysplasia. Semin Neonatol 8, 51–61. [DOI] [PubMed] [Google Scholar]
- Parkinson JRC, Hyde MJ, Gale C, Santhakumaran S & Modi N (2013). Preterm birth and the metabolic syndrome in adult life: a systematic review and meta-analysis. Pediatrics 131, e1240–63. [DOI] [PubMed] [Google Scholar]
- Pierro M, Ionescu L, Montemurro T, Vadivel A, Weissmann G, Oudit G, Emery D, Bodiga S, Eaton F, Péault B, Mosca F, Lazzari L & Thébaud B (2013). Short-term, long-term and paracrine effect of human umbilical cord-derived stem cells in lung injury prevention and repair in experimental bronchopulmonary dysplasia. Thorax 68, 475. [DOI] [PubMed] [Google Scholar]
- Pugh ME & Hemnes AR (2014). Pulmonary hypertension in women. Expert Rev Cardiovasc Ther 8, 1549–1558. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Raju TNK, Buist AS, Blaisdell CJ, Moxey-Mims M & Saigal S (2017a). Adults born preterm: a review of general health and system-specific outcomes. Acta Paediatr 106, 1409–1437. [DOI] [PubMed] [Google Scholar]
- Raju TNK, Pemberton VL, Saigal S, Blaisdell CJ, Moxey-Mims M, Buist S & Discussants ABPCS and (2017b). Long-Term Healthcare Outcomes of Preterm Birth: An Executive Summary of a Conference Sponsored by the National Institutes of Health. J Pediatr 181, 309–318.e1. [DOI] [PubMed] [Google Scholar]
- Rich S, Dantzker DR, Ayres SM, Bergofsky EH, Brundage BH, Detre KM, Fishman AP, Goldring RM, Groves BM & Koerner SK (1987). Primary Pulmonary Hypertension: A National Prospective Study. Ann Intern Med 107, 216. [DOI] [PubMed] [Google Scholar]
- Romer LM, Haverkamp HC, Lovering AT, Pegelow DF & Dempsey JA (2006). Effect of exercise-induced arterial hypoxemia on quadriceps muscle fatigue in healthy humans. Am J Physiol Regu Integr Comp Physiol 290, R365–75. [DOI] [PubMed] [Google Scholar]
- Ronkainen E, Dunder T, Peltoniemi O, Kaukola T, Marttila R & Hallman M (2015). New BPD predicts lung function at school age: Follow-up study and meta-analysis. Pediatr Pulm 50, 1090–1098. [DOI] [PubMed] [Google Scholar]
- Santuz P, Baraldi E, Zaramella P, Filippone M & Zacchello F (1995). Factors limiting exercise performance in long-term survivors of bronchopulmonary dysplasia. Am J Respir Crit Care Med 152, 1284–1289. [DOI] [PubMed] [Google Scholar]
- Sapieha P, Joyal J-S, Rivera JC, Kermorvant-Duchemin E, Sennlaub F, Hardy P, Lachapelle P & Chemtob S (2010). Retinopathy of prematurity: understanding ischemic retinal vasculopathies at an extreme of life. J Clin Invest 120, 3022–3032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sarria EE, Mattiello R, Rao L, Tiller CJ, Poindexter B, Applegate KE, Granroth-Cook J, Denski C, Nguyen J, Yu Z, Hoffman E & Tepper RS (2012). Quantitative assessment of chronic lung disease of infancy using computed tomography. Eur Respir J 39, 992–999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scherrer U, Rimoldi SF, Rexhaj E, Stuber T, Duplain H, Garcin S, de Marchi SF, Nicod P, Germond M, Allemann Y & Sartori C (2012). Systemic and Pulmonary Vascular Dysfunction in Children Conceived by Assisted Reproductive Technologies. Circulation 125, 1890–1896. [DOI] [PubMed] [Google Scholar]
- Schubert U, Müller M, Bonamy A-KE, Abdul-Khaliq H & Norman M (2011). Aortic growth arrest after preterm birth: a lasting structural change of the vascular tree. J Dev Orig Hlth Dis 2, 218–225. [DOI] [PubMed] [Google Scholar]
- Shah PS et al. (2012). Outcomes of preterm infants <29 weeks gestation over 10-year period in Canada: a cause for concern|[quest]|. J Perinatol 32, 132–138. [DOI] [PubMed] [Google Scholar]
- Simonneau G, Montani D, Celermajer DS, Denton CP, Gatzoulis MA, Krowka M, Williams PG & Souza R (2018). Haemodynamic definitions and updated clinical classification of pulmonary hypertension. Eur Respir J 53, 1801913. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Simpson SJ, Logie KM, O’Dea CA, Banton GL, Murray C, Wilson AC, Pillow JJ & Hall GL (2017). Altered lung structure and function in mid-childhood survivors of very preterm birth. Thorax 72, 702–711. [DOI] [PubMed] [Google Scholar]
- Smith SL, Doig AK & Dudley WN (2005). Impaired parasympathetic response to feeding in ventilated preterm babies. Archives Dis Child Fetal Neonatal Ed 90, F505–F508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steinhorn R, Davis JM, Göpel W, Jobe A, Abman S, Laughon M, Bancalari E, Aschner J, Ballard R, Greenough A, Storari L, Thomson M, Ariagno RL, Fabbri L, Turner MA & Consortium IN (2017). Chronic Pulmonary Insufficiency of Prematurity: Developing Optimal Endpoints for Drug Development. J Pediatr 191, 15–21.e1. [DOI] [PubMed] [Google Scholar]
- Stenmark KR & Mecham RP (1997). Cellular and molecular mechanisms of pulmonary vascular remodeling. Ann Rev Physiol 59, 89–144. [DOI] [PubMed] [Google Scholar]
- Stickford JL, Wilhite DP, Bhammar DM, Balmain BN & Babb TG (2021). Dysanapsis in men and women with obesity. J Appl Physiol 131, 496–503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stoll BJ et al. (2015). Trends in Care Practices, Morbidity, and Mortality of Extremely Preterm Neonates, 1993–2012. JAMA 314, 1039–1051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Svedenkrans J, Kowalski J, Norman M & Bohlin K (2016). Low Exercise Capacity Increases the Risk of Low Cognitive Function in Healthy Young Men Born Preterm: A Population-Based Cohort Study. Plos One 11, e0161314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tan CMJ & Lewandowski AJ (2019). The Transitional Heart: From Early Embryonic and Fetal Development to Neonatal Life. Fetal Diagn Ther 47, 373–386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tang JR, Karumanchi SA, Seedorf G, Markham N & Abman SH (2011). Excess soluble vascular endothelial growth factor receptor-1 in amniotic fluid impairs lung growth in rats: linking preeclampsia with bronchopulmonary dysplasia. Am J Physiol Lung Cell Mol Physiol 302, L36–L46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Telles F, McNamara N, Nanayakkara S, Doyle MP, Williams M, Yaeger L, Marwick TH, Leeson P, Levy PT & Lewandowski AJ (2020). Changes in the Preterm Heart From Birth to Young Adulthood: A Meta-analysis. Pediatrics 146, e20200146. [DOI] [PubMed] [Google Scholar]
- Thébaud B, Goss KN, Laughon M, Whitsett JA, Abman SH, Steinhorn RH, Aschner JL, Davis PG, McGrath-Morrow SA, Soll RF & Jobe AH (2019). Bronchopulmonary dysplasia. Nat Rev Dis Primers 5, 78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tikanmäki M, Kaseva N, Tammelin T, Sipola-Leppänen M, Matinolli H-M, Eriksson JG, Järvelin M-R, Vääräsmäki M & Kajantie E (2017). Leisure Time Physical Activity in Young Adults Born Preterm. J Pediatr 189, 135–142.e2. [DOI] [PubMed] [Google Scholar]
- Udomittipong K, Sly PD, Patterson HJ, Gangell CL, Stick SM & Hall GL (2008). Forced oscillations in the clinical setting in young children with neonatal lung disease. Eur Respir J 31, 1292–1299. [DOI] [PubMed] [Google Scholar]
- Urs R, Kotecha S, Hall GL & Simpson SJ (2018). Persistent and progressive long-term lung disease in survivors of preterm birth. Paediatr Respir Rev 28, 87–94. [DOI] [PubMed] [Google Scholar]
- Vollsæter M, Clemm HH, Satrell E, Eide GE, Røksund OD, Markestad T & Halvorsen T (2015). Adult respiratory outcomes of extreme preterm birth. A regional cohort study. Ann Am Thorac Soc 12, 313–322. [DOI] [PubMed] [Google Scholar]
- Vrijlandt EJLE, Boezen HM, Gerritsen J, Stremmelaar EF & Duiverman EJ (2007). Respiratory health in prematurely born preschool children with and without bronchopulmonary dysplasia. J Pediatr 150, 256–261. [DOI] [PubMed] [Google Scholar]
- Vrijlandt EJLE, Gerritsen J, Boezen HM, Grevink RG & Duiverman EJ (2006). Lung Function and Exercise Capacity in Young Adults Born Prematurely. Am J Respir Crit Care Med 173, 890–896. [DOI] [PubMed] [Google Scholar]
- West JB, Mathieu-Costello O, Jones JH, Birks EK, Logemann RB, Pascoe JR & Tyler WS (1993). Stress failure of pulmonary capillaries in racehorses with exercise-induced pulmonary hemorrhage. J Appl Physiol 75, 1097–1109. [DOI] [PubMed] [Google Scholar]
- Whipp BJ & Pardy RL (2011). Breathing During Exercise. Compr Physiol 605–629.
- Williams KJ, Robinson NE, DeFeijter-Rupp H, Millerick-May M, Stack A, Hauptman J & Derksen FJ (2013). Distribution of venous remodeling in exercise-induced pulmonary hemorrhage of horses follows reported blood flow distribution in the equine lung. J Appl Physiol 114, 869–878. [DOI] [PubMed] [Google Scholar]
- Willis GR, Fernandez-Gonzalez A, Reis M, Yeung V, Liu X, Ericsson M, Andrews NA, Mitsialis SA & Kourembanas S (2020). Mesenchymal stromal cell-derived small extracellular vesicles restore lung architecture and improve exercise capacity in a model of neonatal hyperoxia-induced lung injury. J Extracell Vesicles 9, 1790874. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wong PM, Lees AN, Louw J, Lee FY, French N, Gain K, Murray CP, Wilson A & Chambers DC (2008). Emphysema in young adult survivors of moderate-to-severe bronchopulmonary dysplasia. Eur Respir J 32, 321–328. [DOI] [PubMed] [Google Scholar]
- Yang J, Epton MJ, Harris SL, Horwood J, Kingsford RA, Troughton R, Greer C & Darlow BA (2021). Reduced Exercise Capacity in Adults Born Very Low Birth Weight: A Population-Based Cohort Study. Am J Resp Crit Care; DOI: 10.1164/rccm.202103-0755oc. [DOI] [PubMed]


