Abstract
Introduction: Appendicitis can cause ventriculoperitoneal (VP) shunt infection. However, little data is available on the incidence of shunt infections and shunt revisions with appendicitis. Therefore, we sought to determine the rates of shunt infection and revision in patients with VP shunt and appendicitis using large database data and review the literature.
Methods: We used a de-identified database network (TriNetX) to retrospectively query via ICD-10 and current procedural terminology codes to evaluate all patients with the presence of a VP shunt and appendicitis. Primary outcomes included shunt infection and shunt revision at 90 days, with secondary outcomes of sepsis and seizure.
Results: 396 patients with VP shunt and subsequent appendicitis were identified. The average age was 27.02+-20.94 years. Shunt infection was identified in 43 (10.859%) patients within 90 days of appendicitis, and shunt externalization or revision was performed in 66 (16.667%) patients. Sepsis was identified in 49 (12.374%) patients and seizures occurred in 56 (14.141%) patients. The literature review revealed eight relevant articles, with 49 total patients. Ten (20.408%) patients had shunts externalized, four of which occurred after shunt infection was identified. Shunt infection occurred in a total of 11 (22.449%) patients. Two (4.082%) patients died, one of which had their shunt externalized pre-emptively, and the other after ventriculitis was identified. Shunt revisions were performed in 16 (32.653%)
Conclusion: Our results demonstrate that shunt externalization should be strongly considered in patients with appendicitis, given high shunt infection rates.
Keywords: mortality rate, shunt revision, outcomes, ventriculoperitoneal shunt, shunt, appendicitis, hydrocephalus, neurosurgery
Introduction
Infections are thought to be one of the most common causes of shunt failure [1]. Appendicitis is a particularly common abdominal infection, and acute appendicitis is the most common condition requiring emergency operation [2,3]. Rupture of an appendix can lead to spread of infection throughout the abdomen. Due to the shunt’s location in the abdomen, an abdominal infection can have especially grave consequences such as CNS infection, diminished shunt function or shunt failure, and increased intracranial pressure [4]. Yet there is little data connecting the incidence of appendicitis and ventriculoperitoneal (VP) shunt infection, or necessity of VP shunt revision or externalization. The purpose of this study is to leverage large database data to determine how common shunt infection and shunt revision is following appendicitis in order to guide practice.
Materials and methods
This was a retrospective comparative case-control study. We used a de-identified database network (TriNetX) to retrospectively query via ICD-10 and current procedural terminology codes to evaluate all patients with the presence of a VP shunt and appendicitis. Data came from 56 health care organizations (HCOs) spanning six countries and over 78 million patients. Data includes demographics, diagnoses, medications, laboratory values, genomics, and procedures. The identity of the HCOs and patients are not disclosed to comply with ethical guidelines against data re-identification. Because of the database's federated nature, an institutional review board (IRB) waiver has been granted. The data is updated daily. Our use of this database and its validity was informed by previous literature, and exact details of the network have been previously described [5-8].
Medical information included age at index date, as well as sex, race, and comorbidities of hypertension, acute kidney injury, diabetes, ischemic heart disease, heart failure, atrial fibrillation, disorders of lipoprotein metabolism and other lipidemias, obesity, history of nicotine dependence, chronic respiratory disease, cirrhosis, alcohol abuse or dependence, and peripheral vascular disease, recorded up to the date of the index date. Our primary outcomes included shunt infection and shunt revision at 90 days, with secondary outcomes of sepsis and seizure. Chi-square analysis was performed on categorical variables.
Results
396 patients were identified. Average age was 27.02+-20.94 years. 289 patients (72.613%) were white, 56 (14.07%) black or African American, and <10 patients (<2.513%) were Asian. 214 (53.769%) of patients were male. Data was unable to be collected as to whether the appendicitis was classified as ruptured or unruptured.
Baseline demographics and characteristics are shown in Table 1.
Table 1. Baseline demographics and characteristics.
| Code | Diagnosis | Cohort 1, n (%) |
| AI | Age at Index | 27.02 (100) |
| 2106-3 | White | 289 (72.613) |
| M | Male | 214 (53.769) |
| F | Female | 184 (46.231) |
| 2054-5 | Black or African American | 56 (14.07) |
| 2131-1 | Unknown Race | 45 (11.307) |
| 2028-9 | Asian | 10 (2.513) |
| I10-I16 | Hypertensive diseases | 117 (29.397) |
| R53 | Malaise and fatigue | 117 (29.397) |
| J40-J47 | Chronic lower respiratory diseases | 98 (24.623) |
| R63 | Symptoms and signs concerning food and fluid intake | 98 (24.623) |
| E78 | Disorders of lipoprotein metabolism and other lipidemias | 68 (17.085) |
| R13 | Aphagia and dysphagia | 60 (15.075) |
| R40 | Somnolence, stupor and coma | 50 (12.563) |
| N17-N19 | Acute kidney failure and chronic kidney disease | 46 (11.558) |
| E08-E13 | Diabetes mellitus | 41 (10.302) |
| F17 | Nicotine dependence | 37 (9.296) |
| I20-I25 | Ischemic heart diseases | 34 (8.543) |
| Z87.891 | Personal history of nicotine dependence | 32 (8.04) |
| I50 | Heart failure | 26 (6.533) |
| I48 | Atrial fibrillation and flutter | 12 (3.015) |
| K74 | Fibrosis and cirrhosis of liver | <10 (<2.513) |
| F10.1 | Alcohol abuse | <10 (<2.513) |
| F10.2 | Alcohol dependence | <10 (<2.513) |
| I73 | Other peripheral vascular diseases | <10 (<2.513) |
Table 2 shows outcomes. Shunt infection was identified in 43 (10.859%) of patients, with sepsis identified in 49 (12.374%) patients. Shunt revisions occurred in 66 (12.374%) of patients. Seizures occurred in 56 (14.141%) of patients.
Table 2. Outcomes.
| Outcome | Cohort 1, n (%) |
| Shunt infection | 43 (10.859) |
| Shunt revision | 66 (16.667) |
| Sepsis | 49 (12.374) |
| Seizures | 56 (14.141) |
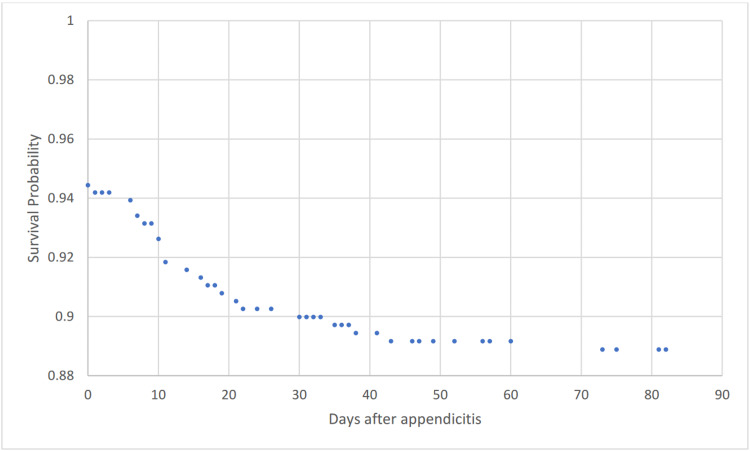
Figure 1 shows a Kaplan-Meier survival curve for outcome shunt infection to 90 days.
Figure 1. Kaplan-Meier survival analysis for outcome shunt infection.
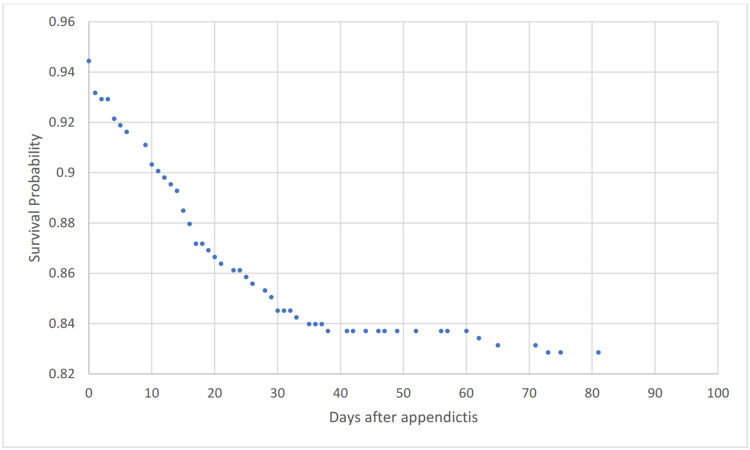
Figure 2 shows a Kaplan-Meier survival curve for outcome shunt revision to 90 days.
Figure 2. Kaplan-Meier survival analysis for outcome shunt revision.
Discussion
Few studies have come to a consensus on the necessity of VP shunt revision or externalization for acute appendicitis. The incidence of VP shunt infections from appendicitis is unknown. A literature review revealed eight relevant articles, with 49 total patients. Ten (20.408%) patients had shunts externalized, four of which occurred after shunt infection was identified. Shunt infection occurred in a total of 11 (22.449%) patients. Two (4.082%) patients died, one of which had their shunt externalized pre-emptively, and the other after ventriculitis was identified. Shunt revisions were performed in 16 (32.653%) patients [4,9-15].
Ein and colleagues studied eight children with appendicitis. Three patients had perforated appendicitis (37.5%) and five had non-perforated appendicitis (62.5%). All eight patients underwent an appendectomy immediately following diagnosis. The VP shunt was externalized for the three perforated cases. The shunts were not externalized in the non-perforated cases. Follow-up ranged from one to 30 years. Four children (three with acute appendicitis and one with ruptured appendix) had one-to-three shunt revisions from three months to four years after appendectomy. Ein and colleagues concluded that the VP shunt must be temporarily exteriorized in the case of appendiceal rupture [10].
Häussler and colleagues described a series of 21 children with appendicitis. Seven patients had a non-perforated appendix, five patients with ulcerous appendicitis, five patients with chronic appendicitis, and four patients had a perforated appendix. Nine (42.9%) patients developed a cerebrospinal fluid (CSF) infection, including 100% of the patients with perforated appendicitis. None of the patients were initially externalized. One patient out of the seven patients with a non-perforated appendix developed a CSF infection and had their shunt externalized. Three patients out of the four patients that had a perforated appendix had their shunts externalized. Two patients developed pseudocysts. One patient died. According to their findings, there seems to be a slightly higher risk of VP shunt infection following a ruptured appendix [14].
Pumberger and colleagues presented a series of six children, three with a perforated appendix (50%), and three with a non-perforated appendix (50%). None underwent shunt externalization, and none of the patients showed shunt complications related to the surgery or appendicitis. The authors concluded that the VP shunt system may be left in place in the case of appendicitis due to the low risk of infection [9].
Barina et al. looked at five adults, four with perforated appendicitis and one with gangrenous appendicitis. One patient’s VP shunt was discontinued following discovery of a Gram-positive cocci in the peritoneal fluid. No shunt infections occurred, and the authors concluded that the presence of a VP shunt does not increase risk of postoperative complication in patients undergoing appendectomy for appendicitis. The authors did recommend that several precautions be taken to reduce risk of shunt infection, such as taking care to eliminate any concurrent infection prior to elective surgery or administering perioperative antibiotic prophylaxis [4].
Dalfino and colleagues describe a series of seven patients with acute peritonitis, two of whom had a ruptured appendix. During the follow-up interval of 18.5 months no patients developed a VP shunt infection. However, all patients were treated with antibiotics. The authors suggest that externalization of the shunt should be considered only if it presents less risk to the patient than leaving it in place [11].
Hadani and colleagues followed two patients, one with a perforated appendix and one with a phlegmonous appendix. Both patients’ shunts were externalized, and the authors assert that shunt externalization in patients who exhibit acute abdominal symptoms is essential and may prevent unnecessary laparotomy. One of the patients died after developing septicemia. The CSF culture showed growth of Escherichia coli [15].
Although the literature shows differing rates of infection and shunt externalization practices, our results demonstrate significant rates of shunt infection with acute appendicitis. Although shunt externalization is not without its risks, externalizing shunts may be able to prevent serious infection, and might be the safest option for these patients [16].
Our analysis was not without limitations. The major limitation of this study was that it was retrospective. Furthermore, due to the nature of the database, we were unable to collect patient-level data on specific outcomes. We were unable to report on radiology information. We do not have information on the type of diagnostic test used for confirmation of disease. The data collected was for billing purposes, not for clinical use, and thus much clinical information is missing. In addition, some misidentification is inevitable in database studies.
Conclusions
There is a limitation of not being able to determine if the appendix was ruptured or unruptured in this study. Nevertheless, our results suggests that shunt externalization should be strongly considered in patients with appendicitis given high rates of shunt infection. Because shunt externalization is not without its risks, this decision should be made within the clinical context of each individual patient.
The content published in Cureus is the result of clinical experience and/or research by independent individuals or organizations. Cureus is not responsible for the scientific accuracy or reliability of data or conclusions published herein. All content published within Cureus is intended only for educational, research and reference purposes. Additionally, articles published within Cureus should not be deemed a suitable substitute for the advice of a qualified health care professional. Do not disregard or avoid professional medical advice due to content published within Cureus.
The authors have declared that no competing interests exist.
Human Ethics
Consent was obtained or waived by all participants in this study
Animal Ethics
Animal subjects: All authors have confirmed that this study did not involve animal subjects or tissue.
References
- 1.Ventriculoperitoneal shunt complications: a review. Paff M, Alexandru-Abrams D, Muhonen M, Loudon W. Interdiscip Neurosurg Adv Tech Case Manag. 2018;13:66–70. [Google Scholar]
- 2.Appendicitis in children in the managed care era. Adolph VR, Falterman KW, Lund DP, et al. J Pediatr Surg. 1996;31:1035–1037. doi: 10.1016/s0022-3468(96)90080-9. [DOI] [PubMed] [Google Scholar]
- 3.Risk factors for adverse outcomes after the surgical treatment of appendicitis in adults. Margenthaler JA, Longo WE, Virgo KS, et al. Ann Surg. 2003;238:59–66. doi: 10.1097/01.SLA.0000074961.50020.f8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Appendectomy for appendicitis in patients with a prior ventriculoperitoneal shunt. Barina AR, Virgo KS, Mushi E, Bahadursingh AM, Johnson FE. J Surg Res. 2007;141:40–44. doi: 10.1016/j.jss.2007.02.039. [DOI] [PubMed] [Google Scholar]
- 5.6-month neurological and psychiatric outcomes in 236 379 survivors of COVID-19: a retrospective cohort study using electronic health records. Taquet M, Geddes JR, Husain M, Luciano S, Harrison PJ. Lancet Psychiatry. 2021;8:416–427. doi: 10.1016/S2215-0366(21)00084-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Using a federated network of real-world data to optimize clinical trials operations. Topaloglu U, Palchuk MB. JCO Clin Cancer Inform. 2018;2:1–10. doi: 10.1200/CCI.17.00067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Outcomes of COVID-19 in patients with rheumatoid arthritis: a multicenter research network study in the United States. Raiker R, DeYoung C, Pakhchanian H, Ahmed S, Kavadichanda C, Gupta L, Kardeş S. Semin Arthritis Rheum. 2021;51:1057–1066. doi: 10.1016/j.semarthrit.2021.08.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Cerebral venous thrombosis and portal vein thrombosis: a retrospective cohort study of 537,913 COVID-19 cases. Taquet M, Husain M, Geddes JR, Luciano S, Harrison PJ. EClinicalMedicine. 2021;39:101061. doi: 10.1016/j.eclinm.2021.101061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Appendicitis in children with a ventriculoperitoneal shunt. Pumberger W, Löbl M, Geissler W. Pediatr Neurosurg. 1998;28:21–26. doi: 10.1159/000028613. [DOI] [PubMed] [Google Scholar]
- 10.Appendicitis in the child with a ventriculo-peritoneal shunt: a 30-year review. Ein SH, Miller S, Rutka JT. J Pediatr Surg. 2006;41:1255–1258. doi: 10.1016/j.jpedsurg.2006.03.004. [DOI] [PubMed] [Google Scholar]
- 11.Conservative management of ventriculoperitoneal shunts in the setting of abdominal and pelvic infections. Dalfino JC, Adamo MA, Gandhi RH, Boulos AS, Waldman JB. J Neurosurg Pediatr. 2012;9:69–72. doi: 10.3171/2011.10.PEDS1189. [DOI] [PubMed] [Google Scholar]
- 12.Acute appendicitis with abscess in the setting of a ventriculoperitoneal shunt, requiring shunt explanation due to meningitis. Chambers JG 4th. Am Surg. 2020;86:1559–1560. doi: 10.1177/0003134820936620. [DOI] [PubMed] [Google Scholar]
- 13.Does appendicitis in a child with a ventriculoperitoneal shunt necessitate shunt revision? Johnstone PD, Jayamohan J, Kelly DF, Drysdale SB. Arch Dis Child. 2019;104:607–609. doi: 10.1136/archdischild-2018-316265. [DOI] [PubMed] [Google Scholar]
- 14.Ventriculoperitoneal shunt infection and appendicitis in children. Häussler B, Menardi G, Hausberger K, Hager J. https://pubmed.ncbi.nlm.nih.gov/11848056/ Eur J Pediatr Surg. 2001;11:0–6. [PubMed] [Google Scholar]
- 15.Acute appendicitis in children with a ventriculoperitoneal shunt. Hadani M, Findler G, Muggia-Sullam M, Sahar A. Surg Neurol. 1982;18:69–71. doi: 10.1016/0090-3019(82)90023-4. [DOI] [PubMed] [Google Scholar]
- 16.A meta-analysis of ventriculostomy-associated cerebrospinal fluid infections. Ramanan M, Lipman J, Shorr A, Shankar A. BMC Infect Dis. 2015;15 doi: 10.1186/s12879-014-0712-z. [DOI] [PMC free article] [PubMed] [Google Scholar]