Abstract
Neurokinin B (NKB) is a neuropeptide involved in the regulation of reproductive endocrine system of vertebrate animals, including fish. However, the pathway of NKB action in fish has not been clearly elucidated. In order to clarify the effect of NKB and NKF (neurokinin F) on gonadotropic hormone (GTH) gene expression in the pituitary, we studied the changes of LHβ and FSHβ gene expressions by using two different pituitary culture methods (whole pituitary culture or dispersed pituitary cell culture). Pituitaries were removed from mature female and male Nile tilapia. Changes of LHβ and FSHβ gene expressions were measured and compared after the treatment with NKB or NKF peptides at concentrations 0 to 1,000 nM. Expression of GTH genes in the whole pituitary cultures treated with NKB or NKF peptides did not show significant difference except in female at one concentration when treated with NKF. On the contrary, there were significant changes of GTH gene expressions in the dispersed pituitary cell cultures when treated with NKB and NKF peptides. These results suggest that dispersed pituitary cell culture is more relevant than whole pituitary culture in studying the function of pituitary, and that NKB and NKF could act directly on the pituitary to regulate the expression of GTH genes.
Keywords: Pituitary cell culture, Pituitary tissue culture, Neurokinin B (NKB), Neurokinin F (NKF), Gonadotropic hormone (GTH)
INTRODUCTION
Reproduction in teleost is controlled by a complex interplay of neuroendocrine system in HPG (hypothalamus-pituitary-gonad) axis. The pituitary in the HPG axis is a central regulator of reproduction as well as growth and endocrine physiology, conveying signals from the hypothalamus to various target organs. In particular, the pituitary secretes gonadotropic hormone (GTH), which leads to the development and maturation of fish gonad. GTH includes luteinizing hormone (LH) and follicle stimulating hormone (FSH), which stimulate the gonads to secrete sex hormones, leading to gonadal development and maturation. Gonadotropin releasing hormone (GnRH) has been known as a representative hormone that regulates the synthesis and secretion of GTH during this reproductive process (Trudeau, 1997). Recently, however, with the emergence of other neurohormones, studies on the regulation of the pituitary secretion of GTH have been actively conducted.
Neurokinin B (NKB), one of the neurohormones, has been shown to play an important role in the reproduction process in some fishes (Biran et al., 2012). NKB belongs to the tachykinin family, a neurohormone involved in vertebrate endocrine regulation, and has a common amino acid sequence in C-terminus called Phe-X-Gly-Leu-Met-NH2 (X is an aromatic or aliphatic amino acid) (Pennefather et al., 2004). NKB is short peptides produced by posttranslational proteolytic processing of preprotachykinins (PPTs), a precursor protein encoded by tac3 (Pennefather et al., 2004). In fish, unlike mammals, NKB-associated peptide (neurokinin B related peptide, NKBRP) contains neurokinin F (NKF), and the tac3 gene encodes both NKB and NKF (Zhou et al., 2012). NKF has also been suggested to be involved in the control of fish reproduction (Biran et al., 2012).
When NKB first appeared, it was found to be involved in the control of GnRH secretion (Rance et al., 2010). However, the pathway through which NKB is involved in regulating fish reproduction has not been fully understood. In fish, NKB has been suggested to interacts with GnRH via Kisspeptin (Zmora et al., 2017). In addition, pathways that act directly on the pituitary gland to affect GTH secretion (Biran et al., 2014), and pathways that directly act on the ovary to enhance the production of steroid hormones (Qi et al., 2016) have also been proposed. As such, various pathways of action have been identified, making it difficult to understand the role of NKB.
Many studies have shown that NKB affects the expression of GTH genes (Biran et al., 2012; Qi et al., 2015; Liu et al., 2019; Wang et al., 2021). However, it is not clear whether the NKB acts directly on the pituitary or not. In the pituitary tissue culture of Nile tilapia, there was no significant change of GTH when treated with NKB peptide, but GTH decreased with NKF peptide treatment ( Jin et al., 2016). On the contrary, treatment of NKB and NKF peptides in pituitary cell cultures has been reported to stimulate the secretion of GTH (Biran et al., 2014). In the pituitary cell culture of striped bass, treatment with NKB and NKF peptides did not affect the secretion of GTH (Zmora et al., 2017). Treatment of human NKB to European eel pituitary cell culture did not affect the expression of FSH, but LH expression was significantly decreased at specific concentrations (Campo et al., 2018). In addition, treatment of NKB and NKF peptide to the pituitary cell culture of orange-spotted grouper had no effect on GTH expression (Chen et al., 2018).
These discrepancies may be attributed to differences of fish species, sexes, sexual maturity, and in vitro system such as pituitary culture methods. Among these, the difference of in vitro systems means the difference between pituitary tissue culture and pituitary cell culture. Pituitary tissue culture requires the pituitary from many individuals for experiment because the fish pituitary is very small. Thus, differences between individuals can lead to a number of variations within or between experiments (Chen et al., 2010). Pituitary cell culture requires the process of separating and culturing pituitary cells, which is difficult to reproduce consistently, and recovery periods can affect specific gene expression (Chen et al., 2010).
In this study, the direct effects of NKB and NKF on the Nile tilapia pituitary gland were investigated using two in vitro systems. In addition, the two culture methods were compared to suggest which method is more relevant to study the function of the pituitary.
MATERIALS AND METHODS
1. Animals
Experimental fish were reared in a closed recirculating aquaculture system at 27±1℃ under a controlled photoperiod (14L:10D) and fed twice a day. They were anaesthetized with benzocaine (50 ppm) and measured (Table 1) before removing their pituitaries.
Table 1. Measurements of experimental fish.
| Sex | Body length (cm) | Body height (cm) | Body weigh (g) | GSI (%) | No. of fish |
|---|---|---|---|---|---|
| Female | 14.9±2.3 | 4.7±0.9 | 66.9±34.0 | 4.5±2.2 | 82 |
| Male | 15.5±1.8 | 4.9±0.7 | 69.8±28.4 | 0.7±0.6 | 87 |
2. Peptide synthesis
Nile tilapia NKB (EMDDIFIGLM-NH2) and NKF (YNDLDYDSFVGLM-NH2) were synthesized by Peptron (Daejeon, Korea). The purity of the synthesized peptide was confirmed to be over 95% by HPLC.
3. Experiments
1). Primary pituitary cell culture
Pituitary glands from tilapia were removed and pooled into the culture medium (Leibovitz L15 medium, 10% fetal bovine serum (FBS), 1% penicillin-streptomycin solution). A razor blade as used to cut the glands into small pieces. Small pieces were trypsinizd for 60 min at 27℃ in trypsin-EDTA solution. Trypsinization was terminated by addition of FBS. The supernatant was discarded by after centrifugation (1,000×g, 4℃, 10 min) to obtain cell pellet. Cells were cultured in the culture medium (Leibovitz L15 medium, 1% FBS, 1% penicillin-streptomycin solution) and the number of cells was counted using a hamocytometer. The cell culture medium was dispensed into a 24-well plate. The cells were cultured at 27℃ for 4 days in dark. The initial culture medium was replaced by fresh culture medium with NKB or NKF at concentrations ranging from 1 nM to 1,000 nM and cultured for 6 h. Cells were stored at -80℃ until total RNA is extracted.
2). Pituitary tissue culture (whole pituitary culture)
Pituitary from tilapia were removed and placed individually in a 96 multi well plate. The pituitaries were incubated in culture medium (Leibovitz L15 medium, 10% FBS, 1% penicillin-streptomycin) for 3 h at 27°C in a dark culture condition to stabilize them. The initial medium was replaced by fresh medium with different concentrations of NKB (1 nM, 10 nM, 100 nM, 1,000 nM), NKF (1 nM, 10 nM, 100 nM, 1,000 nM) peptide. After 6 h, incubated pituitaries were stored at -80℃ until total RNA is extracted.
3). RNA extraction and cDNA synthesis
Total RNA was extracted from the incubated pituitary cells of each sample or the incubated each whole pituitary tissue using Trizol® reagent (Ambion, Austin, TX, USA) according to the manufacturer’s protocol. Extracted total RNA was quantified using nanodrop 2000 (Thermo Scientific, Massachusetts, MA, USA) and cDNA was synthesized using TOPscript™RT DryMIX (Enzynomics, Daejeon, Korea).
4). Quantitative real-time PCR (qRT-PCR)
qRT-PCR was performed on a CFX96 Touch™ Real-Time PCR Detection System (Bio-Rad, Hercules, CA, USA) using the Topreal™ qPCR 2X PreMIX SYBR Green (Enzynomics). For each sample, 10 μL SYBR Green (Enzynomics), 5 μL cDNA (1:50 dilution), 3 μL nuclease free water (NFW) of each 10 pmole primer sets were used for amplification. Target gene were LHβ, FSHβ and Kiss2 and primers used for qRT-PCR are listed in Table 2. The reaction program was following: an initial denaturation at 95℃ for 10 min, followed by 40 cycles at 95℃ for 15 sec, 60℃ for 30 sec, and 72℃ for 30 sec. The transcript levels of target genes were normalized against β-actin transcript levels.
Table 2. Primers used for quantitative real-time PCR (qRT-PCR).
| Genes (Genbank accession No.) | Sequence | Product size | |
|---|---|---|---|
| LHβ (XM_025897714) | F | GCTGTCACCCAGTAGAGA | 79 bp |
| R | TTGCTGAATGGTATCTTGATGA | ||
| FSHβ (NM_001279743) | F | GGCTTCGTCGACACCACCAT | 148 bp |
| R | TGAAACCCCGTGGACATTGC | ||
| β-Actin (XM_003443127) | F | AGCATCCCGTCCTGCTCACA | 121 bp |
| R | AGCACAGCCTGGATGGCAAC |
4. Statistical analyses
All data for gene expression were expressed as mean values±SEM. Statistical differences between control group and NKB or NKF treated group were evaluated by using independent t-test. A probability level of less than 0.05 was used to indicate significance. All analyses were performed using the SPSS 18.0 software (Chicago, IL, USA).
RESULTS
1. Microscopic observation of pituitary cell culture
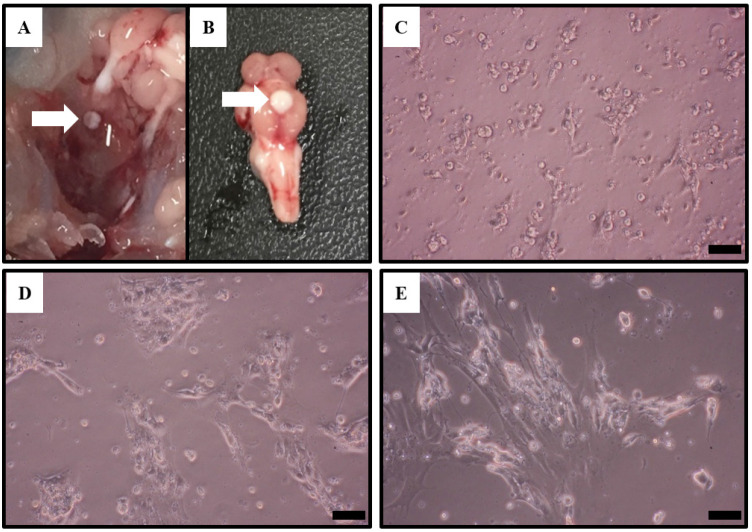
Cells were observed using a microscope during pituitary cell culture. Pituitary cells of Nile tilapia gradually adhered to the plate gradually after 24 h of cultivation since the beginning of culture. The cells tended to aggregate during the culture process (Fig. 1).
Fig. 1. Photographs of Nile tilapia pituitary and microscopic observation of pituitary cell culture.
(A) Location and isolation of Nile tilapia pituitary (arrow). (B) Location of the pituitary in Nile tilapia. (C–E) The pituitary of Nile tilapia was cultured for 24 (C), 48 (D) and 72 (E) h and then photographed by a camera. Scale bar=100 μm.
2. Response to neurokinin B (NKB) peptide treatment
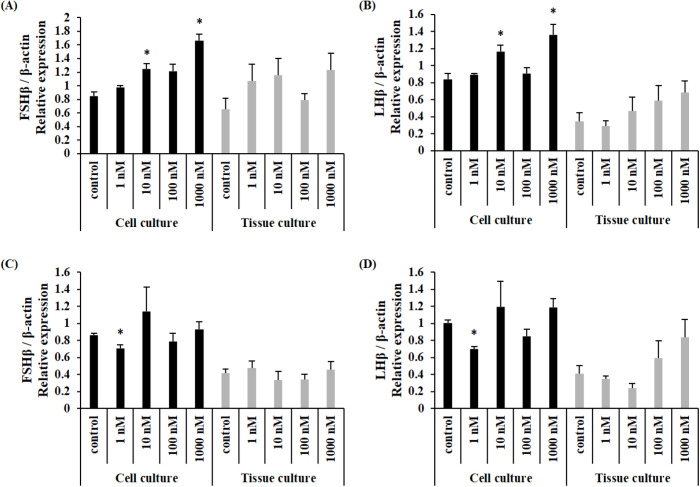
When NKB peptide was treated, the expression of FSHβ mRNA was significantly increased at 10 nM and 1,000 nM in cultured pituitary cells, but there was no significant difference in cultured tissue in female Nile tilapia (Fig. 2A). Similarly, the expression of LHβ mRNA was significantly increased at 10 nM and 1,000 nM in cultured cells, but there was no significant difference in cultured tissues (Fig. 2B). The expression of FSHβ mRNA in males was significantly decreased after 1 nM treatment in cultured pituitary cells, and there was no significant difference in cultured tissues in male Nile tilapia (Fig. 2C). The expression of LHβ was significantly decreased by 1 nM treatment in cultured cells, and there was no significant difference in cultured tissues (Fig. 2D).
Fig. 2. Expression levels of LHβ and FSH mRNA in the primary culture of pituitary cells (black bars) and whole pituitaries (grey bars) incubated with different concentrations of NKB peptide.
(A) FSHβ levels in female Nile tilapia, (B) LHβ levels in female Nile tilapia, (C) FSHβ levels in male Nile tilapia, (D) LHβ levels in male Nile tilapia. *Indicates significant difference (p<0.05) from control group. FSH, follicle stimulating hormone; LH, luteinizing hormone; NKB, neurokinin B.
3. Response to neurokinin F (NKF) peptide treatment
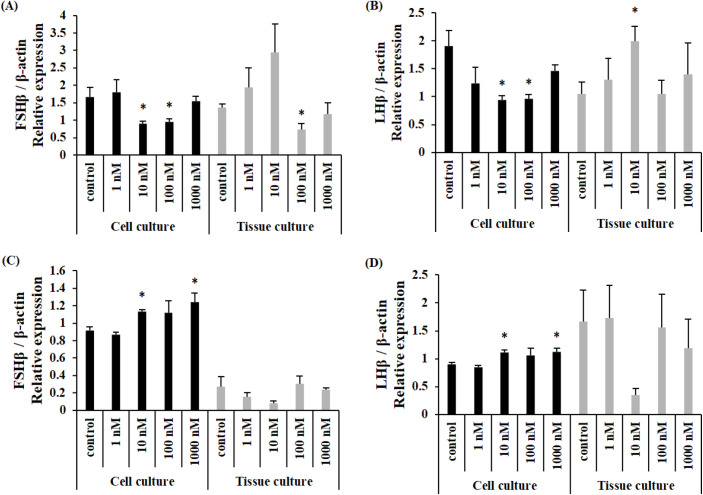
When NKF peptide was treated, the expression of FSHβ mRNA was significantly decreased when treated with 10 nM and 100 nM in cultured cells and significantly decreased when treated with 100 nM in cultured tissues in female Nile tilapia (Fig. 3A). The expression of LHβ mRNA was significantly decreased when treated with 10 nM and 100 nM in cultured cells and significantly increased when treated with 10 nM in cultured tissues (Fig. 3B). The expression of FSHβ mRNA was significantly increased after 10 nM and 1,000 nM treatment in cultured cells and there was no significant difference in cultured tissue (Fig. 3C) in male Nile tilapia. The expression of LHβ mRNA was significantly increased after 10 nM and 1,000 nM treatment in cultured cells. There was no significant difference in cultured tissue (Fig. 3D).
Fig. 3. Expression levels of LHβ and FSH mRNA in the primary culture of pituitary cells (black bars) and whole pituitaries (grey bars) incubated with different concentrations of NKF peptide.
(A) FSHβ levels in female Nile tilapia, (B) LHβ levels in female Nile tilapia, (C) FSHβ levels in male Nile tilapia, (D) LHβ levels in male Nile tilapia. *Indicates significant difference (p<0.05) from control group. FSH, follicle stimulating hormone; LH, luteinizing hormone; NKF, neurokinin F.
DISCUSSION
1. Pituitary cell culture and whole pituitary culture (pituitary tissue culture)
The in vitro system is clearly advantageous in that interference from outside (brain) regulatory molecules is minimal, if not null. Therefore, in vitro experiments of pituitary culture have been actively conducted to understand endocrine phenomena in fish. However, there is a lack of information on what situations to use pituitary cell culture or tissue culture. In addition, there are not many confirmed cases of how each culture method affects the experimental results (Socha et al., 2003; Chen et al., 2010). Although pituitary culture experiments have been conducted in various fish to clarify the function of NKB and NKF, the findings further confuse the function of NKB and NKF. In this study, the expression of GTH genes in pituitary cultured cells treated with synthetic NKB and NKF peptides and pituitary tissues were different. The results of pituitary tissue culture showed large variations and there was no significant difference in most cases. On the other hand, GTH gene expression in pituitary cell culture showed significant differences.
All pituitary glands used in cell culture are pooled and dispensed into culture plates, respectively. On the contrary, in tissue culture, one pituitary tissue is used as a sample. In the dispersed cell culture, the biological difference of the sample between groups is small, resulting in small variation. On the other hand, in the whole pituitary tissue culture, the physiological difference of each individual could be reflected in the results, showing large variation in gene expression. Thus, the chances to obtain biased and unexpected results are high.
Therefore, pituitary cell cultures would be more effective than pituitary tissue cultures to clarify the effects of synthetic NKB and NKF peptide treatment on GTH gene expression through pituitary culture. Significant differences were observed in the cultured pituitary cells, in this study but not in concentration dependent manner. Previous studies using the Nile tilapia pituitary also failed to show concentration-dependent results (Biran et al., 2014; Jin et al., 2016). This may be due to individual differences between the pituitary used (in the case of tissue culture) or may be due to cell composition heterogeneity (in the case of cell culture) of the cultured cell population. Nevertheless, the results of this study suggest that the dispersed cell cultures are more suitable to study the effect of NKB and NKF.
2. Female and male
To date, few studies have considered the sex of fish when investigating the direct role of NKB and NKF in the pituitary. In previous studies, experiments were conducted with mixed sex groups (Hu et al., 2014, 2016; Jin et al., 2016) or with only one of sexes (Biran et al., 2014; Zmora et al., 2017; Campo et al., 2018; Zhang et al., 2019). Therefore, this study aimed to find out sexual difference of the pituitary response to NKB and NKF.
There was no clear difference of GTH gene expression between female and male in the culture of whole pituitary tissue. However, cultured pituitary cells showed differences of GTH gene expression between the sexes. In females, the expression of GTH was increased when NKB peptide was treated, whereas the changes of GTH expression in males not consistent showing significance at only one concentration (1 nM). When NKF peptide was treated, GTH gene expression in female tended to decrease and to increase in male. It appeared that the responses of pituitary cells to NKB or NKF were different to each other.
The reason why the female and male showed different results is not clear at the moment. In fish, the NKB system may be different between the sexes. In mammals, it has already suggested that the NKB system differs between the sexes. In rats (Ruiz-Pino et al., 2012) and sheep (Goubillon et al., 2000; Cheng et al., 2010), more NKB neurons were identified in female arcuate nucleus (ARC) than in males. Based on these results, it is possible that the NKB system has some differences between the sexes in fish as well, and the difference may have been reflected in GTH gene expression.
3. Neurokinin B and neurokinin F
NKB and NKF are known to have the same effect on the GTH expression (Biran et al., 2012, 2014; Zmora et al., 2017; Campo et al., 2018), but in teleost pituitary culture results, there are cases in which the pituitary responded differently to NKB and NKF peptides respectively ( Jin et al., 2016; Chen et al., 2018). In this study, GTH gene expression was increased by NKB peptide but decreased by NKF peptide in the culture of female pituitary cells. In addition, GTH gene expression was decreased by both NKB and NKF peptide in the culture of male pituitary cells. These results suggest the possibility of functional difference between NKB and NKF in fish, supporting a previous finding of structural diversity between NKB and NBBRPs (Wang et al., 2021).
In conclusion, results from this study suggest that dispersed pituitary cell culture are more relevant than whole pituitary culture in studying the function of pituitary, and that NKB and NKF could act directly on the pituitary to regulate the expression of GTH genes.
Acknowledgements
This research was supported by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (NRF- 2014R1A1A2058272).
ORCID
SeongHee Mun https://orcid.org/0000-0002-8810-5805
HyeonJi Oh https://orcid.org/0000-0001-8929-2983
JoonYeong Kwon https://orcid.org/0000-0001-8485-0878
Conflict of interests
The authors declare no potential conflict of interest.
Authors’ contributions
Conceptualization: Kwon JY.
Data curation: Kwon JY, Mun SH.
Formal analysis: Mun SH, Oh HJ.
Methodology: Kwon JY, Mun SH.
Software: Mun SH.
Validation: Kwon JY.
Investigation: Mun SH, Oh HJ.
Writing-original draft: Mun SH.
Writing-review & editing: Kwon JY, Mun SH.
Ethics approval
This article does not require IRB/IACUC approval because there are no human and animal participants.
REFERENCES
- Biran J, Golan M, Mizrahi N, Ogawa S, Parhar IS, Levavi-Sivan B. Direct regulation of gonadotropin release by neurokinin B in tilapia (Oreochromis niloticus) Endocrinology. 2014;155:4831–4842. doi: 10.1210/en.2013-2114. [DOI] [PubMed] [Google Scholar]
- Biran J, Palevitch O, Ben-Dor S, Levavi-Sivan B. Neurokinin Bs and neurokinin B receptors in zebrafish-potential role in controlling fish reproduction. Proc Natl Acad Sci USA. 2012;109:10269–10274. doi: 10.1073/pnas.1119165109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Campo A, Lafont AG, Lefranc B, Leprince J, Tostivint H, Kamech N, Dufour S, Rousseau K. Tachykinin-3 genes and peptides characterized in a basal teleost, the European eel: Evolutionary perspective and pituitary role. Front Endocrinol. 2018;9:304. doi: 10.3389/fendo.2018.00304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen H, Xiao L, Liu Y, Li S, Li G, Zhang Y, Lin H. Neurokinin B signaling in hermaphroditic species, a study of the orange-spotted grouper (Epinephelus coioides) Gen Comp Endocrinol. 2018;260:125–135. doi: 10.1016/j.ygcen.2018.01.009. [DOI] [PubMed] [Google Scholar]
- Chen MJ, Chiou PP, Liao YH, Lin CM, Chen TT. Development and characterization of five rainbow trout pituitary single-cell clone lines capable of producing pituitary hormones. J Endocrinol. 2010;205:69–78. doi: 10.1677/JOE-09-0315. [DOI] [PubMed] [Google Scholar]
- Cheng G, Coolen LM, Padmanabhan V, Goodman RL, Lehman MN. The kisspeptin/neurokinin B/dynorphin (KNDy) cell population of the arcuate nucleus: Sex differences and effects of prenatal testosterone in sheep. Endocrinology. 2010;151:301–311. doi: 10.1210/en.2009-0541. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goubillon ML, Forsdike RA, Robinson JE, Ciofi P, Caraty A, Herbison AE. Identification of neurokinin B-expressing neurons as an highly estrogen-receptive, sexually dimorphic cell group in the ovine arcuate nucleus. Endocrinology. 2000;141:4218–4225. doi: 10.1210/endo.141.11.7743. [DOI] [PubMed] [Google Scholar]
- Hu G, He M, Wong AOL. Novel functional role of NK3R expression in the potentiating effects on somatolactin α autoregulation in grass carp pituitary cells. Sci Rep. 2016;6:36102. doi: 10.1038/srep36102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hu G, Lin C, He M, Wong AOL. Neurokinin B and reproductive functions: “KNDy neuron” model in mammals and the emerging story in fish. Gen Comp Endocrinol. 2014;208:94–108. doi: 10.1016/j.ygcen.2014.08.009. [DOI] [PubMed] [Google Scholar]
- Jin YH, Park JW, Kim JH, Kwon JY. Neurokinin B-related peptide suppresses the expression of GnRH Ⅰ, Kiss2 and tac3 in the brain of mature female Nile tilapia Oreochromis niloticus. Dev Reprod. 2016;20:51–61. doi: 10.12717/DR.2016.20.1.051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu Y, Wang Q, Wang X, Meng Z, Liu Y, Li S, Zhang Y, Lin H. NKB/NK3 system negatively regulates the reproductive axis in sexually immature goldfish (Carassius auratus) Gen Comp Endocrinol. 2019;281:126–136. doi: 10.1016/j.ygcen.2019.05.020. [DOI] [PubMed] [Google Scholar]
- Pennefather JN, Lecci A, Candenas ML, Patak E, Pinto FM, Maggi CA. Tachykinins and tachykinin receptors: A growing family. Life Sci. 2004;74:1445–1463. doi: 10.1016/j.lfs.2003.09.039. [DOI] [PubMed] [Google Scholar]
- Qi X, Salem M, Zhou W, Sato-Shimizu M, Ye G, Smitz J, Peng C. Neurokinin B exerts direct effects on the ovary to stimulate estradiol production. Endocrinology. 2016;157:3355–3365. doi: 10.1210/en.2016-1354. [DOI] [PubMed] [Google Scholar]
- Qi X, Zhou W, Li S, Liu Y, Ye G, Liu X, Peng C, Zhang Y, Lin H. Goldfish neurokinin B: Cloning, tissue distribution, and potential role in regulating reproduction. Gen Comp Endocrinol. 2015;221:267–277. doi: 10.1016/j.ygcen.2014.10.017. [DOI] [PubMed] [Google Scholar]
- Rance NE, Krajewski SJ, Smith MA, Cholanian M, Dacks PA. Neurokinin B and the hypothalamic regulation of reproduction. Brain Res. 2010;1364:116–128. doi: 10.1016/j.brainres.2010.08.059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ruiz-Pino F, Navarro VM, Bentsen AH, Garcia-Galiano D, Sanchez-Garrido MA, Ciofi P, Steiner RA, Mikkelsen JD, Pinilla L, Tena-Sempere M. Neurokinin B and the control of the gonadotropic axis in the rat: Developmental changes, sexual dimorphism, and regulation by gonadal steroids. Endocrinology. 2012;153:4818–4829. doi: 10.1210/en.2012-1287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Socha M, Sokołowska-Mikołajczyk M, Mikołajczyk T, Chyb J, Epler P. Naltrexone stimulates LH secretion in vitro from the common carp (Cyprinus carpio L.) pituitary gland. Fish Aquat Life. 2003;11:5–16. [Google Scholar]
- Trudeau VL. Neuroendocrine regulation of gonadotrophin Ⅱ release and gonadal growth in the goldfish, Carassius auratus. Rev Reprod. 1997;2:55–68. doi: 10.1530/ror.0.0020055. [DOI] [PubMed] [Google Scholar]
- Wang B, Cui A, Zhang Y, Xu Y, Wang W, Jiang Y, Liu X. Neurokinin B in a flatfish species, the half-smooth tongue sole (Cynoglossus semilaevis), and its potential role in reproductive functions. Aquac Rep. 2021;20:100651. doi: 10.1016/j.aqrep.2021.100651. [DOI] [Google Scholar]
- Zhang Z, Wen H, Li Y, Li Q, Li W, Zhou Y, Wang L, Liu Y, Lyu L, Qi X. Tac3 gene products regulate brain and digestive system gene expression in the spotted sea bass (Lateolabrax maculatus) Front Endocrinol. 2019;10:556. doi: 10.3389/fendo.2019.00556. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zmora N, Wong TT, Stubblefield J, Levavi-Sivan B, Zohar Y. Neurokinin B regulates reproduction via inhibition of kisspeptin in a teleost, the striped bass. Endocrinology. 2017;233:159–174. doi: 10.1530/JOE-16-0575. [DOI] [PubMed] [Google Scholar]
- Zhou W, Li S, Liu Y, Qi X, Chen H, Cheng CHK, Liu X, Zhang Y, Lin H. The evolution of tachykinin/tachykinin receptor (TAC/TACR) in vertebrates and molecular identification of the TAC3/TACR3 system in zebrafish (Danio rerio) Mol Cell Endocrinol. 2012;361:202–212. doi: 10.1016/j.mce.2012.04.007. [DOI] [PubMed] [Google Scholar]