Abstract
Bipolar disorder is a mental illness that causes extreme mood swings and has a chronic course. However, the mechanism by which mood episodes with completely opposite characteristics appear repeatedly, or a mixture of symptoms appears, in patients with bipolar disorder remains unknown. Therefore, mood stabilizers are indicated only for single mood episodes, such as manic episodes and depressive episodes, and no true mood-stabilizing drugs effective for treating both manic and depressive episodes currently exist. Therefore, in this review, therapeutic targets that facilitate the development of mood stabilizers were examined by reviewing the current understanding of the neuromolecular etiology of bipolar disorder.
Keywords: Bipolar disorder, Drug therapy, Neurobiology
INTRODUCTION
The ancient Greeks were aware of bipolar disorder; however, medical research of bipolar disorder did not start until the 20th century after it became recognized as a disease. Efforts were made to investigate the etiology of bipolar disorder, with a focus on neurotransmitters. Recently, brain imaging research techniques have rapidly advanced neuromolecular biology research, enabling the discovery of novel findings involving altered brain structures or neurotransmission. In the present review, the etiology of bipolar disorder was evaluated by reviewing the literature to identify upcoming research trends. In particular, the neuromolecular therapeutic targets of mood stabilizers in terms of the pathophysiology of bipolar disorder were summarized and the potential of personalized medicine reviewed.
NEUROTRANSMITTERS
In bipolar disorder, neurotransmitters are abnormally regulated in the brain. Biogenic amine neurotransmission functions in the limbic system are impaired, influencing sleep, appetite, alertness, sexual function, endocrine function, and the regulation of emotions such as fear and anger [1]. Neurotransmitters have been the focus of numerous studies of the pathogenesis of bipolar disorder.
Noradrenaline
The noradrenaline level is usually low in subjects with bipolar disorder; however, the increase in noradrenaline secretion is greater than that in unipolar depression [2]. In addition, the level of 3-methoxy-4-hydroxyphenylglycol, the noradrenaline metabolite, increases during a manic episode [2]. These findings indicate that noradrenaline secre-tion and conversion increase in depression, and noradre-naline activities increase in manic episodes. The noradre-naline increase may be due to the low sensitivity of inhibi-tory α2-adrenaline receptors, although there is controversy as to whether the low sensitivity is specific to bipolar disorder pathophysiology because the low sensitivity of α2-adrenaline also occurs in panic disorder. Noradrenaline levels may indicate the degree of anxiety in general rather than characterizing specific syndromes such as depression or mania. However, the response to lithium is associated with the level of 3-methoxy-4-hydroxyphenylglycol; thus, noradrenaline changes likely reflect the clinical features of bipolar disorder to some degree [3,4].
Lithium is used to treat manic episodes and affects the noradrenaline system differently in various brain regions, showing two bimodal effects over time [5]. Lithium initially decreases b-adrenaline receptor functions due to noradrenaline, triggering inhibition of 3’,5’-cyclic adenosine monophosphate (cAMP) accumulation, and then acts on the presynaptic α2-autoreceptor. The presynaptic α2-autoreceptor inhibits noradrenaline secretion, thereby increasing noradrenaline secretion [6,7]. Carbamazepine can decrease noradrenaline conversion and upregulate the b-adrenaline receptor over an extended period; however, the adenylate cyclase (AC) activity triggered by the b-adrenaline receptor is decreased by direct suppression of AC catalyst subunits [8].
Serotonin
Much evidence supports a link between serotonin and mood disorders [9,10]. A relatively high cerebrospinal fluid (CSF) concentration of 5-hydroxyindoleacetic acid (5-HIAA), a serotonin metabolite, was associated with suicide occurrence and the appearance of aggressive behaviors [11]. This result was based on findings from patients with unipolar depression. Although studies on bipolar disorder exist, the study samples were small, and the influence of serotonin was not clearly explained.
Findings from previous studies vary regarding changes in the CSF level of 5-HIAA in patients with manic depressive episodes [12]. In some studies, a significant difference in the CSF level of 5-HIAA between patients with manic depressive episodes and those with depression was not found. In other studies, a higher correlation of CSF 5-HIAA with the frequency of suicide and aggressive behavior compared with depressive symptoms was reported. However, the decrease in the CSF 5-HIAA level was not evident in patients with bipolar disorder or patients who attempted suicide [13,14].
Dopamine
Mood changes (feelings of highs and lows), as well as changes in motivation and exercise patterns, are significant clinical signs observed in bipolar disorder. The dopamine system plays an essential role in activity level, motivation control, and compensation circuit in the midbrain [15]. In an animal model study of depression, decreased dopamine secretion in mesocorticolimbic neurons was reported [16]. The CSF level of homovanillic acid (HVA), a dopamine metabolite, is also decreased in depression but increased during a manic episode [17]. In patients in a depressive state, the CSF level of HVA is decreased. The change in psychomotor speed observed in depression is likely associated with dopamine abnormalities. In a previous study, the decrease in HVA level was greater in patients with psychomotor retardation than in patients with irritable depression [18]. Furthermore, in an observational study, whether the presence or absence of accompanying psychotic symptoms was associated with dopamine, as evidenced by the relatively greater increase in the HVA level in psychotic depression, was investigated [18]. Dopamine, more than other neurotrans-mitters, has been implicated in the transition of depression to a manic episode in bipolar disorder [19]. This theory is supported by the following evidence: manic episodes typically occur at the time of administration of the dopamine precursor L-dopa; amphetamine, a promoter of dopamine secretion and inhibitor of dopamine absorption, causes hypomanic episodes in patients with bipolar disorder and hypomanic-like episodes in healthy controls; antipsy-chotics, which block the dopamine receptor, are efficacious for treating manic episodes [20,21]. In addition, after administering amphetamine to patients with bipolar disorder, more significant behavioral changes were observed in the experimental group than in the control group, although no significant difference in dopamine secretion was found [22]. This indicates that dopamine reactivity should increase postsynaptically in patients with bipolar disorder [23]. Antipsychotic drugs can also provide an effective treatment for manic episodes. Drugs stimulating dopamine receptors (e.g., dopamine agonists and dopamine reuptake inhibitors) have antidepressant effects. Lithium, a mood stabilizer, increases dopamine conversion and decreases dopamine production in a dose-dependent manner while blocking receptor upregulation or hypersensitivity due to the administration of haloperidol [24]. Based on evidence showing that manic episodes are caused by the hypersensitivity of dopamine receptors, the reaction of the dopamine system to lithium may explain the mechanism of lithium’s ability to treat bipolar disorder [25].
Gamma-aminobutyric acid
Gamma-aminobutyric acid (GABA) is involved in neurotransmission via interneuronal synapses in brain regions controlling mood, such as the striatum, globus pallidus, and cerebral cortex [26]. A retrospective study on patients with depression demonstrated decreased activity of glutamic acid decarboxylase, an enzyme involved in GABA synthesis that potentially reduces GABA activity [26]. In addition, the serum GABA level was low in patients with depression, whereas an increased level was correlated with the treatment response to valproate in patients with manic episodes [27,28]. Long-term administration of a mood stabilizer to patients with bipolar disorder reduced the GABA turnover rate, reinforced GABA neurotransmission [29], upregulated GABA receptors in the frontal cortex and hippocampus, and downregulated GABA receptors in the hypothalamus [30,31]. In a different study, the administration of lithium or valproate reinforced the effects of GABA [32].
NEUROENDOCRINE SYSTEM
Hypothalamic-pituitary-adrenal Cortex Axis
Increased activity of the hypothalamic-pituitary-adrenal (HPA) cortex axis is a characteristic response to stress in mammals. Abnormal HPA axis activity is observed in psychiatric illnesses, including major depressive disorder. Overactivity of the HPA axis, such as increased corticotrophin-releasing hormone activity, has been reported in bipolar disorder [33], which may be associated with mixed manic episodes or depressive episodes more than with typical manic episodes [34]. In addition, the HPA axis abnormality in bipolar disorder is supported by the association between neuroendocrine abnormality and the severity of symptoms as well as between cortisol levels and the degree of anxiety, insomnia, or depression [34].
Furthermore, chronic stress can cause hippocampal atrophy, specifically atrophy of CA3 nerve cells as observed in a high glucocorticoid concentration environ-ment. This indicates the HPA axis plays an essential role in hippocampal atrophy when subjected to stress. Stress and glucocorticoids may cause atrophy directly or reduce cellular resilience by exposing vulnerable cells to various types of stimuli, such as ischemia, hypoglycemia, and excitatory amino acid toxicity. In addition, the HPA axis may be involved in the reduced brain-derived neurotro-phic factor (BDNF) expression caused by chronic stress. In previous studies, attempts were made to mitigate the symptoms of bipolar disorder by reducing the level of HPA activation. Administration of mifepristone (RU-486), a glucocorticoid receptor (GR) antagonist, caused a small but significant decrease in HPA activity, suggesting a potential treatment for bipolar disorder [35].
Gonadal and Thyroid Hormones
Gonadal hormones can trigger the development of mood disorders and are associated with the clinical stage of mood disorders. The incidence of major depression and frequency of depressive episodes are higher in females than males, although the prevalence is similar between the sexes. Gonadal hormones contribute partly to the sex differences in symptoms. Estrogen activates serotonin signaling for antidepressant activity by influencing various neurotransmitters such as noradrenaline, dopamine, and GABA. Estrogen is also involved in neuroplasticity by increasing BDNF expression and intercellular signaling by protein kinase C (PKC) [36,37].
The association between thyroid hormones and mood disorders is well known. Administration of thyroxine and triiodothyronine (T3) is useful for treatment-resistant depression or bipolar disorder because the degree of thyroid activity is associated with the prognosis of bipolar dis-order. Furthermore, thyroid hormones have neurotrophic effects. The T3-receptor complex can increase the expression of transcription factors or proteins responsible for intracellular signaling [38,39]. Thyroid hormones increase the level of cAMP response element-binding protein (CREB) and exert neurotrophic effects [40].
INTRACELLULAR SIGNALING
Theories have been proposed regarding the changes in neurotransmitters and neuroendocrine systems that partly explain the pathophysiological mechanisms of mood stabilizers and antidepressants in treating bipolar disorder. However, these theories need further validation. For example, the monoamine hypothesis does not explain why the clinical manifestations following administration of a mood stabilizer appear after only several weeks; neurotransmission and neuroendocrine system activity can change depending on the stage of depression or manic episodes but not of bipolar disorder [41]. In addition, new models are needed to explain periodicity, a clinical finding characteristic of bipolar disorder, as well as various clinical manifestations. Changes in the intracellular signal transduction system, including second messengers, are currently being investigated.
Changes in the Neuronal Membrane
Na-K ATPase plays a role in maintaining the electrochemical potential difference across the cell membrane by controlling intracellular ion concentrations. The Na-K ATPase concentration is decreased in the peripheral blood cells of patients with acute manic episodes but is increased intracellularly [42]. In rodent experiments, the characteristic activity of bipolar disorder increased or decreased in a dose-dependent manner after administering ouabain, a Na-K ATPase inhibitor. This change was associated with activation of the extracellular signal-regulated kinase pathway [43,44].
The barrier functions and permeable features of a cell membrane change depending on the location and phospholipid concentration. Experiments using nerve cells are limited, and red blood cells or platelets are frequently used to study intracellular signaling because they share similar characteristics with neuronal cell membranes [45]. In patients with bipolar disorder, the hydrophobic region in the cell membranes of red blood cells and lymphocytes is altered [46], and the phosphatidylcholine concentration is not within the normal limits in patients with schizophrenia and manic episodes [47].
G-proteins
Most neurotransmitters or neuromodulators binding to G-proteins display particular intracellular effects. G-proteins consist of α, β, and γ subunits and stimulate/inhibit the Gs and Gi endoenzymes. Endoenzymes such as AC and phospholipase C adenosine generate secondary signaling molecules (cAMP and diacylglycerol, respectively). cAMP and diacylglycerol reactivate protein kinase A (PKA) and PKC, which phosphorylate matrix proteins to regulate metabolism or activate transcription factors. PKA is associated mainly with CREB, which activates the expression of various genes. PKC influences various signaling systems, including the myristoylated alanine-rich C kinase substrate (MARCKS). Long-term clinical effects of antidepressants or mood regulators are hypothetically caused by secondary changes such as activation of various signaling systems and functional changes [48].
Gs/cAMP Signaling Pathway
The relationship between G-protein changes and the development of bipolar disorder have been investigated in previous studies. The peripheral blood cells in patients with bipolar disorder who did not receive medical treatment showed an increased level of a particular Gsα subtype but not Gsα mRNA, which indicates that Gsα only has an influence post-transcription [49,50]. In patients with manic episodes who did not take medication, the G-protein activity was increased in white blood cells, and in bipolar depressive episodes, both the Gsα and Giα levels were decreased in white blood cells [51,52].
In a postmortem study, AC activation associated with Gsα was reportedly increased in the temporal and occipital lobes, influencing intracellular signaling via cAMP in patients with bipolar disorder [53]. Furthermore, the cAMP response to the β adrenaline stimulus was decreased in depressed patients, and cAMP production caused by the forskolin stimulus in the temporal and occipital lobes was increased [54,55].
Changes in the PKA level may reflect the increased cAMP activity in patients with bipolar disorder. A decreased level of the regulatory subunit of PKA was observed in the postmortem brain [56]. In a previous study, the PKA catalytic activity in the temporal cortex was reportedly enhanced in patients with bipolar disorder [57]. A different study suggested that abnormal cAMP-stimulated endogenous phosphorylation is due to cAMP stimulation in the platelets of patients with stabilized bipolar disorder [58,59].
PKA activation is affected by mood change inducers, electroconvulsive therapy, and mood stabilizers [60]. In the ground state with no stimulation, lithium prevents Giα from binding to AC, increasing cAMP production. In contrast, when cAMP accumulates due to Gsα binding to AC caused by β-adrenaline stimulation, lithium induces separation of Gsα from AC and inhibits cAMP accumulation [61]. This bilateral reaction of lithium to cAMP indicates that lithium could have therapeutic effects on both manic and depressive episodes in bipolar disorder. In addition, the increase in PKA activity observed in the postmortem temporal lobe is similar to the cAMP increase seen after lithium treatment [62].
Phosphatidylinositol Pathway
In the phosphatidylinositol (PI) pathway, Gαq/11 controls phosphoinositol to influence intracellular signaling similar to Gsα control of cAMP via AC. In a postmortem study of bipolar disorder, the Gαq/11 activity in the occipital lobe was reduced; however, Gαq/11 binding was increased in the frontal lobe [63,64]. In addition, an increased PI 4,5-bisphosphate level was found in platelet cell membranes of a patient with manic episodes, indicating a significant increase in Pl pathway activity during manic episodes [65,66]. Furthermore, the level of inositol monophosphatase, an enzyme forming the myo-inositol precursor of the Pl pathway, was normal in patients with bipolar disorder, but the myo-inositol level was low [67].
In a postmortem study of patients with bipolar disorder, normal phospholipase C function was reported in the frontal, temporal, and occipital cortices. Conversely, PKC activity in the platelet cell membrane was increased more in patients with manic episodes who were not medically treated than in healthy controls or patients with schizophrenia [68,69]. Another postmortem study reported increased PKC activity and α, γ, and ε PKC isozyme activities and decreased ε PKC level in the frontal cortex [70]. Mood stabilizers normalized the increased PKC activity, as shown in previous studies in which various drugs influenced PKA [71].
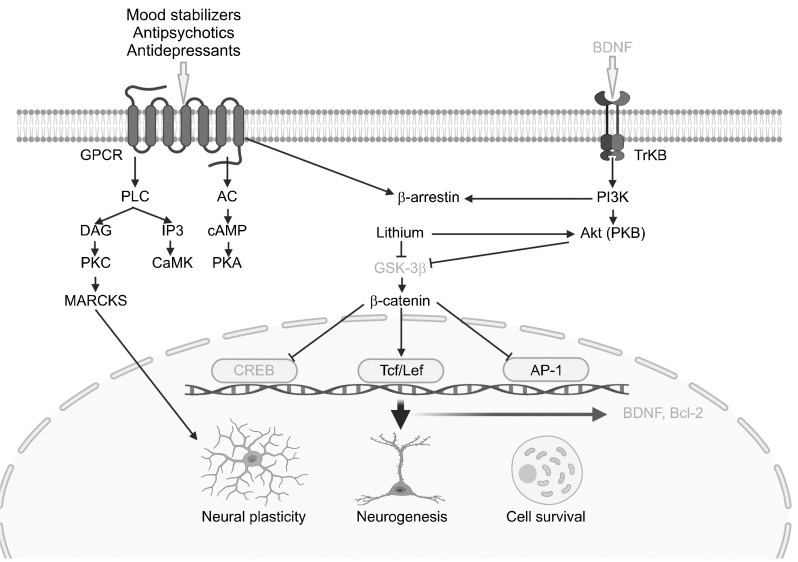
Pharmacologically, lithium firmly controls inositol-1- phosphatase and polyphosphate 1-phosphatase, which play significant roles in the Pl pathway [72]. Free inositol becomes depleted if these enzymes are inhibited. Conse-quently, the synthesis of constituent substances of the Pl pathway is reduced, decreasing intracellular transmission [73]. Manic episodes can be considered hyperactivity of particular endoneurial functions, in which the mood-stabilizing effects of lithium should be similar to the characteristics in manic episodes. In addition, lithium alters the Pl signaling pathway, modulates PKC-mediated pathways and gene expression, and induces long-term effects. Reportedly, long-term lithium administration decreased the concentrations of cytoplasmic PKC-a and cell membrane PI 4,5-bisphosphate [72,73]. MARCKS is a PKC substrate participating in multiple intracellular processes involved in synaptic plasticity. In a previous study, 4 weeks of lithium administration resulted in significantly decreased MARCKS expression in the hippocampus [73]. In addition, PKC activity was reported to play a vital role in the ability of lithium to act as a transcription factor (Fig. 1) [74].
Fig. 1.
Intrasignaling pathways as the mechanism of action for mood stabilizers, antipsychotics and antidepressants. AC, adenylyl cyclase; AP-1, activator protein-1; BDNF, brain-derived neurotrophic factor; CaMK, Ca2+/calmodulin-dependent kinase; cAMP, cyclic adenosine-3′,5′-monophosphate; CREB, cAMP-responsive element binding protein; DAG, diacylglycerol; GPCR, G protein-coupled receptor; GSK-3β, glycogen synthase kinase-3β; IP3, inositol 1,4,5-triphosphate; MARCKS, myristoylated alanine-rich C-kinase substrate; PI3K, phos-phoinositide 3-kinase; PLC, phospholipase C; PKA, protein kinase A; PKB, protein kinase B; PKC, protein kinase C; TrkB, tropomyosin receptor kinase B. Original illustration created with BioRender.com by MK Seo.
Furthermore, lithium and valproate show efficacy in bipolar disorder, and both act on PKC despite their different molecular structures. Researchers have investigated the efficacy of selective regulators of PKC as a treatment for bipolar disorder. Tamoxifen, a PKC inhibitor, was proven as a potential treatment for manic episodes in animal studies. In the latest double-blind placebo-controlled clinical study involving subjects with manic episodes, tamoxifen showed excellent treatment effects equivalent to those of lithium and valproate [75,76]. Efforts to develop new drugs targeting PKC should continue.
The increase in the intracellular calcium level is additional evidence supporting the hypothesis that the Pl pathway is hyperactivated in bipolar disorder. In patients with bipolar disorder who did not receive medical treatment, the baseline calcium concentration in the peripheral blood cells was increased [77]. In addition, the calcium response to stimuli was increased in patients who did not take medication, but reactivity decreased in stable patients who took lithium [78,79].
NEUROPLASTICITY
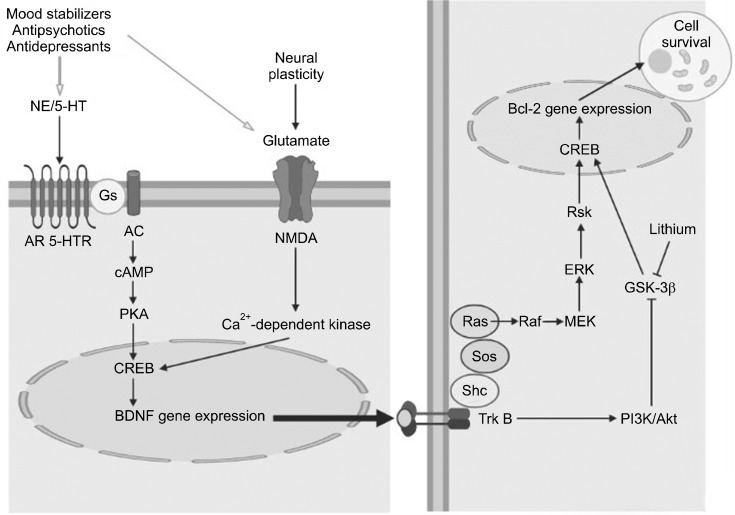
Recently, the effects of neuroplasticity and neuropro-tection have been investigated in multiple studies. Neuro-plasticity refers to a collective change in the brain’s interactions with the environment in the neural network. Neuroplasticity includes synaptic remodeling, long-term potentiation, axon sprouting, synaptogenesis, and neurogenesis [80]. Neuroprotection refers to the mechanism preventing neuronal cell death due to harmful stimuli such as ischemia, free radicals, and excess glutamate. Long-term administration of lithium can prevent neuronal apoptosis by blocking excitatory toxicity of N-methyl-D-aspartic acid [81], involving transcription factors such as CREB, neurotrophic factors such as BDNF, phosphorylation enzymes such as glycogen synthase kinase-3 (GSK-3), and mitogen-activated protein kinase (MAPK) (Fig. 2).
Fig. 2.
Effects of mood stabilizers, antipsychotics and antidepressants on neuroplasticity through cell signaling systems. 5-HT, serotonin; 5-HTR, serotonin receptor; AC, adenylyl cyclase; AR, adrenergic receptor; BDNF, brain-derived neurotrophic factor; cAMP, cyclic adenosine-3′,5′-monophosphate; CREB, cAMP-responsive element binding protein; ERK, extracellular signal-regulated kinase; Gs, G proteins; GSK-3β, glycogen synthase kinase-3β; MEK, Ras/Raf/Mitogen-activated protein kinase (MAP)/ERK kinase; NE, norepinephrine; NMDA, N-methyl-D-aspartate receptor; PI3K, phosphoinositide 3-kinase; PKA, protein kinase A; Shc, Src homology and containing protein; Sos, son of sevenless; TrkB, tropomyosin receptor kinase B. Original illustration created with BioRender.com by MK Seo.
CREB
CREB is a transcription factor involved in long-term functional control of nerve cells by affecting the transcription of various genes or BDNF function. Antidepressants can upregulate CREB, lithium may decrease or increase CREB expression depending on the specific brain region [82-84], and valproate increases CREB expression in different brain regions [85].
BDNF
BDNF is a growth factor essential for the survival and functional maintenance of nerve cells. BDNF acts on TrkB receptors to activate the MAPK pathway and increases the expression of neuroprotective proteins such as Bcl-2 [86]. BDNF is a target of CREB; increased CREB and BDNF levels are observed after antidepressant treatment, similar to the behavioral changes in animal models of depressive behaviors. Furthermore, the BDNF level increases after electroconvulsive therapy, and the administration of BDNF has similar characteristics to those of antidepressant administration in animal studies. BDNF and various endogenous growth factors stimulate the MAPK pathway, increasing transcription and BDNF activity, which involve roles of lithium and valproate [87]. Polymorphisms in BDNF genes could influence the cognitive ability of patients with bipolar disease or their age of development, a topic that is currently being actively researched [88].
GSK-3
The GSK-3 enzyme that mediates various intracellular signaling pathways, significantly contributing to neuroplasticity and cell elasticity regulation. GSK-3 deactivates transcription factors or cytoskeleton-forming proteins by phosphorylation, acting on axonal microtubules and synaptic vesicle functions. GSK-3 activity promotes apoptosis, which is prevented by inhibiting GSK-3 [89,90].
In addition, GSK-3 activation is involved in hypoxia-induced apoptosis [90]. GSK-3 activity is inhibited by PKC and BDNF, as mentioned above, controlling Bcl-2 protein expression via p53 and inhibiting transcription via CREB [87]. GSK-3 is involved in multiple signaling systems. Various growth factors, such as BDNF, have neuropro-tective functions partly due to GSK-3. Therefore, GSK-3 inhibitors may be therapeutic for bipolar disorder; lithium and valproate can inhibit GSK-3 [91]. The capability of atypical antipsychotic drugs, such as olanzapine, to inhibit GSK-3 was also shown (Fig. 1) [92]. Based on imaging studies, drugs such as lithium and valproate exert neuroprotective effects by acting on multiple intracellular signaling and gene transcription factors. In a patient with a family history of bipolar disorder, the prefrontal cortex volume was decreased by 40%. In patients with bipolar disorder who received long-term lithium treatment, a reduction in the prefrontal cortex volume was not observed (the volume was similar to that in normal controls) [93,94].
CIRCADIAN RHYTHM
The circadian rhythm of mammals is thought to be responsible for the suprachiasmatic nucleus. The circadian rhythm is involved in physiological controls such as mobility, the sleep−wake cycle, body temperature, and hormone secretion. Circadian rhythm dysregulation may be associated with the development of mood disorders such as bipolar disorder and clinical features related to disease periodicity [95]. The relationship between the circadian rhythm and bipolar disorder was confirmed in animal models and human studies; the sleep cycle was delayed in patients with type 1 bipolar disorder [96], but the delay may have been caused by the patients’ drug treatments. Furthermore, the appearance of circadian rhythm changes was confirmed in human patients in the clinical setting. In addition, sleep deprivation resulted in therapeutic effects in unipolar or bipolar depression but caused manic episodes in patients with bipolar disorder [97,98]. A change in the sleep cycle may be associated with dopamine receptors in the brain. In another study, sleep deprivation increased the expression of genes associated with neuroplasticity [99].
In addition, various clock genes, including CLOCK, BMAL1, mPer1, mPer2, mPer3, mTim, mCry1, and mCry2, play vital roles in circadian rhythm control. Circadian rhythm genes are implicated in the sensitization mechanism of cocaine-induced behaviors, a typical bipolar disorder model [100]. In addition, in mPer1 and mPer2 mutant mice, behaviors such as conditioned place preference were increased or decreased, and these behaviors were shown to be controlled by clock genes. Lithium may induce a phase delay in the circadian rhythm and extend the rhythm cycle by acting directly on suprachiasmatic nucleus cells [101].
EPIGENETIC MECHANISMS
Among the mechanisms that can cause long-lasting protein expression and functional changes, epigenetics has been the focus of the latest research because of its influence on the pathophysiology of depression and mood disorders [102,103]. Epigenetic changes include DNA methylation, which alters DNA covalent bonding, translational repair after acetylation or methylation of the histone N terminal, and transcriptional gene expression [102,103]. A long-term epigenetic change can explain some phenomena in depression that have been difficult to understand otherwise. Examples that can be explained by epigenetic changes include the following: a difference in the incidence of depression development in monozygotic twins, different depressive symptoms in an inbred rodent animal model, chronic recurrence of depression in women, and a higher prevalence of depression in women [104].
An epigenetic change is an alteration in genetic function subject to environmental influences with no modification of the DNA sequence. Epigenetic changes can explain the inconsistent results of previous depression studies. The influence that DNA sequence polymorphisms exert on depression pathophysiology may not be sub-stantial. Despite the involvement of epigenetic changes in mental disorders, only two classes of chromatin modifica-tion processes have been investigated. The first is DNA methylation, which plays an important role in how the behavior of the mother influences her children’s emotional development and adult behavior. In mouse pups who did not breastfeed well and thus did not receive appropriate care, anxiety behavior increased when the pups became adults compared with pups who received sufficient maternal care. In addition, GR expression was decreased in the hippocampus. A decrease in GR expression is caused by increased methylation of the GR gene promoter. How-ever, a long-lasting molecular wound that occurs during the first week of life can be repaired simply by maternal care such as good licking and grooming [105]. The increase in methylation was attenuated after administration of a histone deacetylase inhibitor. The second chromatin modification process is histone acetylation, which involves activation of protein transcription and chromatin relaxation, and histone acetylation is thought to play an essential role in the action of antidepressants. Improving histone acetylation at the BDNF gene promoter in the hippocampus may cause depressive symptoms in socially discouraged subjects. Histone acetylation inhibitors showed effects similar to those of antidepressants in studies investigating social discouragement and other behaviors [106]. Researchers are currently attempting to elucidate diverse information regarding chromatin regulatory proteins and genes. Before these research findings are used to treat patients, clinicians should be aware that these results were obtained from in vivo experiments and not human studies. However, the findings indicate a possible association between local chromatin and the antidepressant treatment response in depression.
CONCLUSION
In summary, the neurobiological etiology of bipolar disorder is multifactorial. However, it can be estimated from an integrated point of view that mood episodes, treatment responses, and medical diseases resulting from the sequelae of bipolar disorder might be originated from the neuromolecular pathophysiology of bipolar disorder.
Improved understanding of bipolar disorder can only be attained by continued research efforts on the neurobiology of bipolar disorder. Molecular clock gene dysfunction appears repetitively in bipolar disorder, and circadian gene dysfunction occurs both when a mood disorder episode occurs and during the remission of signs and symptoms. Future studies should be performed to determine how circadian gene abnormalities are modified, facilitating the modification of gene abnormalities to control mood. In addition, discovery of proteins and genes associated with neuroplasticity is important to create novel applications for mood control. Finally, identifying the neurobiological etiology of bipolar disorder can aid customized treatment or targeted therapy development.
Footnotes
Funding
This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korea government (MSIT) (NRF-2021R1A2C1003591 to J.G. Lee).
Conflicts of Interest
No potential conflict of interest relevant to this article was reported.
Author Contributions
Conceptualization: Won-Myong Bahk. Data acquisition & Formal analysis: Young Sup Woo, Sung Woo Park, Dae-Hyun Seog, Mi Kyoung Seo. Funding: Jung Goo Lee. Supervision: Won-Myong Bahk. Writing−original draft: Jung Goo Lee. Writing−review & editing: Won-Myong Bahk.
References
- 1.Goodwin FK, Jamison KR. Manic-depressive illness. Oxford University Press; Oxford: 1990. pp. 402–502. [Google Scholar]
- 2.Potter WZ, Manji HK. Catecholamines in depression: an update. Clin Chem. 1994;40:279–287. doi: 10.1093/clinchem/40.2.279. [DOI] [PubMed] [Google Scholar]
- 3.Nutt DJ. Altered central alpha 2-adrenoceptor sensitivity in panic disorder. Arch Gen Psychiatry. 1989;46:165–169. doi: 10.1001/archpsyc.1989.01810020067011. [DOI] [PubMed] [Google Scholar]
- 4.Swann AC, Stokes PE, Secunda SK, Maas JW, Bowden CL, Berman N, et al. Depressive mania versus agitated depression: biogenic amine and hypothalamic-pituitary-adrenocortical function. Biol Psychiatry. 1994;35:803–813. doi: 10.1016/0006-3223(94)91143-6. [DOI] [PubMed] [Google Scholar]
- 5.Lenox RH, Hahn CG. Overview of the mechanism of action of lithium in the brain: fifty-year update. J Clin Psychiatry. 2000;61 Suppl 9:5–15. [PubMed] [Google Scholar]
- 6.Rowe MK, Chuang DM. Lithium neuroprotection: molecular mechanisms and clinical implications. Expert Rev Mol Med. 2004;6:1–18. doi: 10.1017/S1462399404008385. [DOI] [PubMed] [Google Scholar]
- 7.Quiroz JA, Machado-Vieira R, Zarate CA, Jr, Manji HK. Novel insights into lithium's mechanism of action: neurotrophic and neuroprotective effects. Neuropsychobiology. 2010;62:50–60. doi: 10.1159/000314310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Chen G, Pan B, Hawver DB, Wright CB, Potter WZ, Manji HK. Attenuation of cyclic AMP production by carbamazepine. J Neurochem. 1996;67:2079–2086. doi: 10.1046/j.1471-4159.1996.67052079.x. [DOI] [PubMed] [Google Scholar]
- 9.Muneer A. Kynurenine pathway of tryptophan metabolism in neuropsychiatric disorders: pathophysiologic and therapeutic considerations. Clin Psychopharmacol Neurosci. 2020;18:507–526. doi: 10.9758/cpn.2020.18.4.507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Pourhamzeh M, Moravej FG, Arabi M, Shahriari E, Mehrabi S, Ward R, et al. doi: 10.1007/s10571-021-01064-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Goodwin FK. Suicide, aggression, and depression. A theoretical framework for future research. Ann N Y Acad Sci. 1986;487:351–355. doi: 10.1111/j.1749-6632.1986.tb27913.x. [DOI] [PubMed] [Google Scholar]
- 12.Meltzer HY, Arora RC, Baber R, Tricou BJ. Serotonin uptake in blood platelets of psychiatric patients. Arch Gen Psychiatry. 1981;38:1322–1326. doi: 10.1001/archpsyc.1981.01780370024002. [DOI] [PubMed] [Google Scholar]
- 13.Asberg M, Nordström P, Träskman-Bendz L. Cerebrospinal fluid studies in suicide. An overview. Ann N Y Acad Sci. 1986;487:243–255. doi: 10.1111/j.1749-6632.1986.tb27903.x. [DOI] [PubMed] [Google Scholar]
- 14.Hughes JH, Dunne F, Young AH. Effects of acute tryptophan depletion on mood and suicidal ideation in bipolar patients symptomatically stable on lithium. Br J Psychiatry. 2000;177:447–451. doi: 10.1192/bjp.177.5.447. [DOI] [PubMed] [Google Scholar]
- 15.Berk M, Dodd S, Kauer-Sant'anna M, Malhi GS, Bourin M, Kapczinski F, et al. Dopamine dysregulation syndrome: implications for a dopamine hypothesis of bipolar disorder. Acta Psychiatr Scand Suppl. 2007;(434):41–49. doi: 10.1111/j.1600-0447.2007.01058.x. [DOI] [PubMed] [Google Scholar]
- 16.Yadid G, Friedman A. Dynamics of the dopaminergic system as a key component to the understanding of depression. Prog Brain Res. 2008;172:265–286. doi: 10.1016/S0079-6123(08)00913-8. [DOI] [PubMed] [Google Scholar]
- 17.Manji HK, Potter WZ. Monoaminergic systems. In: Young LT, Joffe RT, editors. Bipolar disorder: biological models and their clinical application. Marcel Dekker; New York: 1997. pp. 1–40. [Google Scholar]
- 18.Shah PJ, Ogilvie AD, Goodwin GM, Ebmeier KP. Clinical and psychometric correlates of dopamine D2 binding in depression. Psychol Med. 1997;27:1247–1256. doi: 10.1017/S0033291797005382. [DOI] [PubMed] [Google Scholar]
- 19.Lykouras L, Markianos M, Hatzimanolis J, Malliaras D, Stefanis C. Association of biogenic amine metabolites with symptomatology in delusional (psychotic) and nondelusional depressed patients. Prog Neuropsychopharmacol Biol Psychiatry. 1995;19:877–887. doi: 10.1016/0278-5846(95)00117-E. [DOI] [PubMed] [Google Scholar]
- 20.Murphy DL, Brodie HK, Goodwin FK, Bunney WE., Jr Regular induction of hypomania by L-dopa in "bipolar" manic-de-pressive patients. Nature. 1971;229:135–136. doi: 10.1038/229135a0. [DOI] [PubMed] [Google Scholar]
- 21.Jacobs D, Silverstone T. Dextroamphetamine-induced arousal in human subjects as a model for mania. Psychol Med. 1986;16:323–329. doi: 10.1017/S0033291700009132. [DOI] [PubMed] [Google Scholar]
- 22.Anand A, Darnell A, Miller HL, Berman RM, Cappiello A, Oren DA, et al. Effect of catecholamine depletion on lithium-induced long-term remission of bipolar disorder. Biol Psychiatry. 1999;45:972–978. doi: 10.1016/S0006-3223(98)00293-5. [DOI] [PubMed] [Google Scholar]
- 23.Cousins DA, Butts K, Young AH. The role of dopamine in bipolar disorder. Bipolar Disord. 2009;11:787–806. doi: 10.1111/j.1399-5618.2009.00760.x. [DOI] [PubMed] [Google Scholar]
- 24.Berggren U, Ahlenius S, Engel J. Effects of acute lithium administration on conditioned avoidance behavior and monoamine synthesis in rats. J Neural Transm. 1980;47:1–10. doi: 10.1007/BF01256634. [DOI] [PubMed] [Google Scholar]
- 25.Bunney WE, Jr, Garland BL. Possible receptor effects of chronic lithium administration. Neuropharmacology. 1983;22(3 Spec No):367–372. doi: 10.1016/0028-3908(83)90185-5. [DOI] [PubMed] [Google Scholar]
- 26.Massat I, Souery D, Mendlewicz J, Papadimitriou GN. The GABAergic hypothesis of mood disorders. In: Soares JC, Gershon S, editors. Bipolar disorders: basic mechanisms and therapeutic implications. Marcel Dekker; New York: 2000. pp. 143–165. [Google Scholar]
- 27.Shiah IS, Yatham LN. GABA function in mood disorders: an update and critical review. Life Sci. 1998;63:1289–1303. doi: 10.1016/S0024-3205(98)00241-0. [DOI] [PubMed] [Google Scholar]
- 28.Prosser J, Hughes CW, Sheikha S, Kowatch RA, Kramer GL, Rosenbarger N, et al. Plasma GABA in children and adolescents with mood, behavior, and comorbid mood and behavior disorders: a preliminary study. J Child Adolesc Psychopharmacol. 1997;7:181–199. doi: 10.1089/cap.1997.7.181. [DOI] [PubMed] [Google Scholar]
- 29.Brambilla P, Perez J, Barale F, Schettini G, Soares JC. GABAergic dysfunction in mood disorders. Mol Psychiatry. 2003;8:721–737. 715. doi: 10.1038/sj.mp.4001362. [DOI] [PubMed] [Google Scholar]
- 30.Petty F, Rush AJ, Davis JM, Calabrese JR, Kimmel SE, Kramer GL, et al. Plasma GABA predicts acute response to divalproex in mania. Biol Psychiatry. 1996;39:278–284. doi: 10.1016/0006-3223(95)00141-7. [DOI] [PubMed] [Google Scholar]
- 31.Post RM, Weiss SR, Chuang DM. Mechanisms of action of anticonvulsants in affective disorders: comparisons with lithium. J Clin Psychopharmacol. 1992;12(1 Suppl):23S–35S. doi: 10.1097/00004714-199202001-00005. [DOI] [PubMed] [Google Scholar]
- 32.Motohashi N. GABA receptor alterations after chronic lithium administration. Comparison with carbamazepine and sodium valproate. Prog Neuropsychopharmacol Biol Psychiatry. 1992;16:571–579. doi: 10.1016/0278-5846(92)90062-J. [DOI] [PubMed] [Google Scholar]
- 33.Vieta E, Martínez-De-Osaba MJ, Colom F, Martínez-Arán A, Benabarre A, Gastó C. Enhanced corticotropin response to corticotropin-releasing hormone as a predictor of mania in euthymic bipolar patients. Psychol Med. 1999;29:971–978. doi: 10.1017/S0033291799008727. [DOI] [PubMed] [Google Scholar]
- 34.Rybakowski JK, Twardowska K. The dexamethasone/corticotropin-releasing hormone test in depression in bipolar and unipolar affective illness. J Psychiatr Res. 1999;33:363–370. doi: 10.1016/S0022-3956(99)00014-X. [DOI] [PubMed] [Google Scholar]
- 35.Gallagher P, Watson S, Elizabeth Dye C, Young AH, Nicol Ferrier I. Persistent effects of mifepristone (RU-486) on cortisol levels in bipolar disorder and schizophrenia. J Psychiatr Res. 2008;42:1037–1041. doi: 10.1016/j.jpsychires.2007.12.005. [DOI] [PubMed] [Google Scholar]
- 36.Halbreich U, Kahn LS. Role of estrogen in the aetiology and treatment of mood disorders. CNS Drugs. 2001;15:797–817. doi: 10.2165/00023210-200115100-00005. [DOI] [PubMed] [Google Scholar]
- 37.Payne JL. The role of estrogen in mood disorders in women. Int Rev Psychiatry. 2003;15:280–290. doi: 10.1080/0954026031000136893. [DOI] [PubMed] [Google Scholar]
- 38.Bauer M, London ED, Silverman DH, Rasgon N, Kirchheiner J, Whybrow PC. Thyroid, brain and mood modulation in affective disorder: insights from molecular research and functional brain imaging. Pharmacopsychiatry. 2003;36 Suppl 3:S215–S221. doi: 10.1055/s-2003-45133. [DOI] [PubMed] [Google Scholar]
- 39.Carlson PJ, Singh JB, Zarate CA, Jr, Drevets WC, Manji HK. Neural circuitry and neuroplasticity in mood disorders: insights for novel therapeutic targets. NeuroRx. 2006;3:22–41. doi: 10.1016/j.nurx.2005.12.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Manji HK, Quiroz JA, Payne JL, Singh J, Lopes BP, Viegas JS, et al. The underlying neurobiology of bipolar disorder. World Psychiatry. 2003;2:136–146. [PMC free article] [PubMed] [Google Scholar]
- 41.Cuellar AK, Johnson SL, Winters R. Distinctions between bipolar and unipolar depression. Clin Psychol Rev. 2005;25:307–339. doi: 10.1016/j.cpr.2004.12.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Looney SW el-Mallakh RS, author. Meta-analysis of erythrocyte Na,K-ATPase activity in bipolar illness. Depress Anxiety. 1997;5:53–65. doi: 10.1002/(SICI)1520-6394(1997)5:2<53::AID-DA1>3.0.CO;2-6. [DOI] [PubMed] [Google Scholar]
- 43.El-Mallakh RS, El-Masri MA, Huff MO, Li XP, Decker S, Levy RS. Intracerebroventricular administration of ouabain as a model of mania in rats. Bipolar Disord. 2003;5:362–365. doi: 10.1034/j.1399-5618.2003.00053.x. [DOI] [PubMed] [Google Scholar]
- 44.Kim SH, Yu HS, Park HG, Jeon WJ, Song JY, Kang UG, et al. Dose-dependent effect of intracerebroventricular injection of ouabain on the phosphorylation of the MEK1/2-ERK1/2-p90RSK pathway in the rat brain related to locomotor activity. Prog Neuropsychopharmacol Biol Psychiatry. 2008;32:1637–1642. doi: 10.1016/j.pnpbp.2008.05.027. [DOI] [PubMed] [Google Scholar]
- 45.Soares JC, Mallinger AG. Cell membrane abnormalities in bipolar disorder. In: Soares JC, Gershon S, editors. Bipolar disorders: basic mechanisms and therapeutic implications. Marcel Dekker; New York: 2000. pp. 167–177. [Google Scholar]
- 46.Pettegrew JW, Nichols JS, Minshew NJ, Rush AJ, Stewart RM. Membrane biophysical studies of lymphocytes and erythrocytes in manic-depressive illness. J Affect Disord. 1982;4:237–247. doi: 10.1016/0165-0327(82)90008-8. [DOI] [PubMed] [Google Scholar]
- 47.Hitzemann RJ, Hirschowitz J, Garver DL. On the physical properties of red cell ghost membranes in the affective disorders and psychoses. A fluorescence polarization study. J Affect Disord. 1986;10:227–232. doi: 10.1016/0165-0327(86)90009-1. [DOI] [PubMed] [Google Scholar]
- 48.Shelton RC. Intracellular mechanisms of antidepressant drug action. Harv Rev Psychiatry. 2000;8:161–174. doi: 10.1080/hrp_8.4.161. [DOI] [PubMed] [Google Scholar]
- 49.Manji HK, Chen G, Shimon H, Hsiao JK, Potter WZ, Belmaker RH. Guanine nucleotide-binding proteins in bipolar affective disorder. Effects of long-term lithium treatment. Arch Gen Psychiatry. 1995;52:135–144. doi: 10.1001/archpsyc.1995.03950140053007. [DOI] [PubMed] [Google Scholar]
- 50.Young LT, Asghari V, Li PP, Kish SJ, Fahnestock M, Warsh JJ. Stimulatory G-protein alpha-subunit mRNA levels are not increased in autopsied cerebral cortex from patients with bipolar disorder. Brain Res Mol Brain Res. 1996;42:45–50. doi: 10.1016/S0169-328X(96)00112-X. [DOI] [PubMed] [Google Scholar]
- 51.Schreiber G, Avissar S, Danon A, Belmaker RH. Hyperfunctional G proteins in mononuclear leukocytes of patients with mania. Biol Psychiatry. 1991;29:273–280. doi: 10.1016/0006-3223(91)91289-4. [DOI] [PubMed] [Google Scholar]
- 52.Avissar S, Nechamkin Y, Barki-Harrington L, Roitman G, Schreiber G. Differential G protein measures in mononuclear leukocytes of patients with bipolar mood disorder are state dependent. J Affect Disord. 1997;43:85–93. doi: 10.1016/S0165-0327(96)01400-0. [DOI] [PubMed] [Google Scholar]
- 53.Young LT, Li PP, Kish SJ, Siu KP, Kamble A, Hornykiewicz O, et al. Cerebral cortex Gs alpha protein levels and forskolin-stimulated cyclic AMP formation are increased in bipolar affective disorder. J Neurochem. 1993;61:890–898. doi: 10.1111/j.1471-4159.1993.tb03600.x. [DOI] [PubMed] [Google Scholar]
- 54.Mann JJ, Brown RP, Halper JP, Sweeney JA, Kocsis JH, Stokes PE, et al. Reduced sensitivity of lymphocyte beta-adrenergic receptors in patients with endogenous depression and psychomotor agitation. N Engl J Med. 1985;313:715–720. doi: 10.1056/NEJM198509193131202. [DOI] [PubMed] [Google Scholar]
- 55.Rahman S, Li PP, Young LT, Kofman O, Kish SJ, Warsh JJ. Reduced [3H]cyclic AMP binding in postmortem brain from subjects with bipolar affective disorder. J Neurochem. 1997;68:297–304. doi: 10.1046/j.1471-4159.1997.68010297.x. [DOI] [PubMed] [Google Scholar]
- 56.Fields A, Li PP, Kish SJ, Warsh JJ. Increased cyclic AMP-dependent protein kinase activity in postmortem brain from patients with bipolar affective disorder. J Neurochem. 1999;73:1704–1710. doi: 10.1046/j.1471-4159.1999.731704.x. [DOI] [PubMed] [Google Scholar]
- 57.Chang A, Li PP, Warsh JJ. cAMP-Dependent protein kinase (PKA) subunit mRNA levels in postmortem brain from patients with bipolar affective disorder (BD) Brain Res Mol Brain Res. 2003;116:27–37. doi: 10.1016/S0169-328X(03)00211-0. [DOI] [PubMed] [Google Scholar]
- 58.Perez J, Zanardi R, Mori S, Gasperini M, Smeraldi E, Racagni G. Abnormalities of cAMP-dependent endogenous phosphorylation in platelets from patients with bipolar disorder. Am J Psychiatry. 1995;152:1204–1206. doi: 10.1176/ajp.152.8.1204. [DOI] [PubMed] [Google Scholar]
- 59.Nestler EJ, Terwilliger RZ, Duman RS. Chronic antidepressant administration alters the subcellular distribution of cyclic AMP-dependent protein kinase in rat frontal cortex. J Neurochem. 1989;53:1644–1647. doi: 10.1111/j.1471-4159.1989.tb08564.x. [DOI] [PubMed] [Google Scholar]
- 60.Newman M, Klein E, Birmaher B, Feinsod M, Belmaker RH. Lithium at therapeutic concentrations inhibits human brain noradrenaline-sensitive cyclic AMP accumulation. Brain Res. 1983;278:380–381. doi: 10.1016/0006-8993(83)90278-0. [DOI] [PubMed] [Google Scholar]
- 61.Mathews R, Li PP, Young LT, Kish SJ, Warsh JJ. Increased G alpha q/11 immunoreactivity in postmortem occipital cortex from patients with bipolar affective disorder. Biol Psychiatry. 1997;41:649–656. doi: 10.1016/S0006-3223(96)00113-8. [DOI] [PubMed] [Google Scholar]
- 62.Friedman E, Wang HY. Receptor-mediated activation of G proteins is increased in postmortem brains of bipolar affective disorder subjects. J Neurochem. 1996;67:1145–1152. doi: 10.1046/j.1471-4159.1996.67031145.x. [DOI] [PubMed] [Google Scholar]
- 63.Brown AS, Mallinger AG, Renbaum LC. Elevated platelet membrane phosphatidylinositol-4,5-bisphosphate in bipolar mania. Am J Psychiatry. 1993;150:1252–1254. doi: 10.1176/ajp.150.8.1252. [DOI] [PubMed] [Google Scholar]
- 64.Soares JC, Mallinger AG. Intracellular signal transduction dysfunction in bipolar disorder. In: Soares JC, Gershon S, editors. Bipolar disorders: basic mechanisms and therapeutic implications. Marcel Dekker; New York: 2000. pp. 179–200. [Google Scholar]
- 65.Shimon H, Agam G, Belmaker RH, Hyde TM, Kleinman JE. Reduced frontal cortex inositol levels in postmortem brain of suicide victims and patients with bipolar disorder. Am J Psychiatry. 1997;154:1148–1150. doi: 10.1176/ajp.154.8.1148. [DOI] [PubMed] [Google Scholar]
- 66.Friedman E, Levinson D, Connell TA, Singh H Hoau-Yan-Wang, author. Altered platelet protein kinase C activity in bipolar affective disorder, manic episode. Biol Psychiatry. 1993;33:520–525. doi: 10.1016/0006-3223(93)90006-Y. [DOI] [PubMed] [Google Scholar]
- 67.Wang HY, Friedman E. Enhanced protein kinase C activity and translocation in bipolar affective disorder brains. Biol Psychiatry. 1996;40:568–575. doi: 10.1016/0006-3223(95)00611-7. [DOI] [PubMed] [Google Scholar]
- 68.Hahn CG, Friedman E. Abnormalities in protein kinase C signaling and the pathophysiology of bipolar disorder. Bipolar Disord. 1999;1:81–86. doi: 10.1034/j.1399-5618.1999.010204.x. [DOI] [PubMed] [Google Scholar]
- 69.Nahorski SR, Ragan CI, Challiss RA. Lithium and the phosphoinositide cycle: an example of uncompetitive inhibition and its pharmacological consequences. Trends Pharmacol Sci. 1991;12:297–303. doi: 10.1016/0165-6147(91)90581-C. [DOI] [PubMed] [Google Scholar]
- 70.Ikonomov OC, Manji HK. Molecular mechanisms underlying mood stabilization in manic-depressive illness: the phenotype challenge. Am J Psychiatry. 1999;156:1506–1514. doi: 10.1176/ajp.156.10.1506. [DOI] [PubMed] [Google Scholar]
- 71.Manji HK, Bebchuk JM, Moore GJ, Glitz D, Hasanat KA, Chen G. Modulation of CNS signal transduction pathways and gene expression by mood-stabilizing agents: therapeutic implications. J Clin Psychiatry. 1999;60 Suppl 2:27–39. discussion 40–41. [PubMed] [Google Scholar]
- 72.Soares JC, Chen G, Dippold CS, Wells KF, Frank E, Kupfer DJ, et al. Concurrent measures of protein kinase C and phosphoinositides in lithium-treated bipolar patients and healthy individuals: a preliminary study. Psychiatry Res. 2000;95:109–118. doi: 10.1016/S0165-1781(00)00175-X. [DOI] [PubMed] [Google Scholar]
- 73.Watson DG, Lenox RH. Chronic lithium-induced down-regulation of MARCKS in immortalized hippocampal cells: potentiation by muscarinic receptor activation. J Neurochem. 1996;67:767–777. doi: 10.1046/j.1471-4159.1996.67020767.x. [DOI] [PubMed] [Google Scholar]
- 74.Chen G, Masana MI, Manji HK. Lithium regulates PKC- mediated intracellular cross-talk and gene expression in the CNS in vivo. Bipolar Disord. 2000;2(3 Pt 2):217–236. doi: 10.1034/j.1399-5618.2000.20303.x. [DOI] [PubMed] [Google Scholar]
- 75.Einat H, Yuan P, Szabo ST, Dogra S, Manji HK. Protein kinase C inhibition by tamoxifen antagonizes manic-like behavior in rats: implications for the development of novel therapeutics for bipolar disorder. Neuropsychobiology. 2007;55:123–131. doi: 10.1159/000106054. [DOI] [PubMed] [Google Scholar]
- 76.Yildiz A, Guleryuz S, Ankerst DP, Ongür D, Renshaw PF. Protein kinase C inhibition in the treatment of mania: a double-blind, placebo-controlled trial of tamoxifen. Arch Gen Psychiatry. 2008;65:255–263. doi: 10.1001/archgenpsychiatry.2007.43. [DOI] [PubMed] [Google Scholar]
- 77.Förstner U, Bohus M, Gebicke-Härter PJ, Baumer B, Berger M, van Calker D. Decreased agonist-stimulated Ca2+ response in neutrophils from patients under chronic lithium therapy. Eur Arch Psychiatry Clin Neurosci. 1994;243:240–243. doi: 10.1007/BF02191581. [DOI] [PubMed] [Google Scholar]
- 78.van Calker D, Förstner U, Bohus M, Gebicke-Härter P, Hecht H, Wark HJ, et al. Increased sensitivity to agonist stimulation of the Ca2+ response in neutrophils of manic-depressive patients: effect of lithium therapy. Neuropsychobiology. 1993;27:180–183. doi: 10.1159/000118977. [DOI] [PubMed] [Google Scholar]
- 79.Dubovsky SL, Thomas M, Hijazi A, Murphy J. Intracellular calcium signalling in peripheral cells of patients with bipolar affective disorder. Eur Arch Psychiatry Clin Neurosci. 1994;243:229–234. doi: 10.1007/BF02191579. [DOI] [PubMed] [Google Scholar]
- 80.Muresanu DF. Neuroprotection and neuroplasticity- a holistic approach and future perspectives. J Neurol Sci. 2007;257:38–43. doi: 10.1016/j.jns.2007.01.041. [DOI] [PubMed] [Google Scholar]
- 81.Nonaka S, Hough CJ, Chuang DM. Chronic lithium treatment robustly protects neurons in the central nervous system against excitotoxicity by inhibiting N-methyl-D-aspartate receptor-mediated calcium influx. Proc Natl Acad Sci U S A. 1998;95:2642–2647. doi: 10.1073/pnas.95.5.2642. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Thome J, Sakai N, Shin K, Steffen C, Zhang YJ, Impey S, et al. cAMP response element-mediated gene transcription is upregulated by chronic antidepressant treatment. J Neurosci. 2000;20:4030–4036. doi: 10.1523/JNEUROSCI.20-11-04030.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Chen B, Wang JF, Hill BC, Young LT. Lithium and valproate differentially regulate brain regional expression of phosphorylated CREB and c-Fos. Brain Res Mol Brain Res. 1999;70:45–53. doi: 10.1016/S0169-328X(99)00125-4. [DOI] [PubMed] [Google Scholar]
- 84.Manji HK, Moore GJ, Rajkowska G, Chen G. Neuroplasticity and cellular resilience in mood disorders. Mol Psychiatry. 2000;5:578–593. doi: 10.1038/sj.mp.4000811. [DOI] [PubMed] [Google Scholar]
- 85.Yuan PX, Huang LD, Jiang YM, Gutkind JS, Manji HK, Chen G. The mood stabilizer valproic acid activates mitogen-activated protein kinases and promotes neurite growth. J Biol Chem. 2001;276:31674–31683. doi: 10.1074/jbc.M104309200. [DOI] [PubMed] [Google Scholar]
- 86.Pap M, Cooper GM. Role of glycogen synthase kinase-3 in the phosphatidylinositol 3-Kinase/Akt cell survival pathway. J Biol Chem. 1998;273:19929–19932. doi: 10.1074/jbc.273.32.19929. [DOI] [PubMed] [Google Scholar]
- 87.Beurel E, Jope RS. The paradoxical pro- and anti-apoptotic actions of GSK3 in the intrinsic and extrinsic apoptosis signaling pathways. Prog Neurobiol. 2006;79:173–189. doi: 10.1016/j.pneurobio.2006.07.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Min HJ, Cho HS, Kim SJ, Seok JH, Lee E, Jon DI. Association of the brain-derived neurotrophic factor gene and clinical features of bipolar disorder in Korea. Clin Psychopharmacol Neurosci. 2012;10:163–167. doi: 10.9758/cpn.2012.10.3.163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Chen G, Hasanat KA, Bebchuk JM, Moore GJ, Glitz D, Manji HK. Regulation of signal transduction pathways and gene expression by mood stabilizers and antidepressants. Psychosom Med. 1999;61:599–617. doi: 10.1097/00006842-199909000-00004. [DOI] [PubMed] [Google Scholar]
- 90.Eldar-Finkelman H. Glycogen synthase kinase 3: an emerging therapeutic target. Trends Mol Med. 2002;8:126–132. doi: 10.1016/S1471-4914(01)02266-3. [DOI] [PubMed] [Google Scholar]
- 91.Muneer A. Wnt and GSK3 signaling pathways in bipolar disorder: clinical and therapeutic implications. Clin Psycho-pharmacol Neurosci. 2017;15:100–114. doi: 10.9758/cpn.2017.15.2.100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Kim NR, Park SW, Lee JG, Kim YH. Protective effects of olanzapine and haloperidol on serum withdrawal-induced apoptosis in SH-SY5Y cells. Prog Neuropsychopharmacol Biol Psychiatry. 2008;32:633–642. doi: 10.1016/j.pnpbp.2007.11.002. [DOI] [PubMed] [Google Scholar]
- 93.Drevets WC, Price JL, Simpson JR, Jr, Todd RD, Reich T, Vannier M, et al. Subgenual prefrontal cortex abnormalities in mood disorders. Nature. 1997;386:824–827. doi: 10.1038/386824a0. [DOI] [PubMed] [Google Scholar]
- 94.Drevets WC. Neuroimaging studies of mood disorders. Biol Psychiatry. 2000;48:813–829. doi: 10.1016/S0006-3223(00)01020-9. [DOI] [PubMed] [Google Scholar]
- 95.Chung JK, Lee KY, Kim SH, Kim EJ, Jeong SH, Jung HY, et al. Circadian rhythm characteristics in mood disorders: comparison among bipolar I disorder, bipolar II disorder and recurrent major depressive disorder. Clin Psychopharmacol Neurosci. 2012;10:110–116. doi: 10.9758/cpn.2012.10.2.110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Ahn YM, Chang J, Joo YH, Kim SC, Lee KY, Kim YS. Chrono-type distribution in bipolar I disorder and schizophrenia in a Korean sample. Bipolar Disord. 2008;10:271–275. doi: 10.1111/j.1399-5618.2007.00573.x. [DOI] [PubMed] [Google Scholar]
- 97.Barbini B, Colombo C, Benedetti F, Campori E, Bellodi L, Smeraldi E. The unipolar-bipolar dichotomy and the response to sleep deprivation. Psychiatry Res. 1998;79:43–50. doi: 10.1016/S0165-1781(98)00020-1. [DOI] [PubMed] [Google Scholar]
- 98.Wehr TA, Sack DA, Rosenthal NE. Sleep reduction as a final common pathway in the genesis of mania. Am J Psychiatry. 1987;144:201–204. doi: 10.1176/ajp.144.2.201. [DOI] [PubMed] [Google Scholar]
- 99.Demontis MG, Fadda P, Devoto P, Martellotta MC, Fratta W. Sleep deprivation increases dopamine D1 receptor antagonist [3H]SCH 23390 binding and dopamine-stimulated adenylate cyclase in the rat limbic system. Neurosci Lett. 2339;117:224–227. doi: 10.1016/0304-3940(90)90148-3. [DOI] [PubMed] [Google Scholar]
- 100.Abarca C, Albrecht U, Spanagel R. Cocaine sensitization and reward are under the influence of circadian genes and rhythm. Proc Natl Acad Sci U S A. 2002;99:9026–9030. doi: 10.1073/pnas.142039099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Iwahana E, Akiyama M, Miyakawa K, Uchida A, Kasahara J, Fukunaga K, et al. Effect of lithium on the circadian rhythms of locomotor activity and glycogen synthase kinase-3 protein expression in the mouse suprachiasmatic nuclei. Eur J Neurosci. 2004;19:2281–2287. doi: 10.1111/j.0953-816X.2004.03322.x. [DOI] [PubMed] [Google Scholar]
- 102.Seo MK, Ly NN, Lee CH, Cho HY, Choi CM, Nhu LH, et al. Early life stress increases stress vulnerability through BDNF gene epigenetic changes in the rat hippocampus. Neuro-pharmacology. 2016;105:388–397. doi: 10.1016/j.neuropharm.2016.02.009. [DOI] [PubMed] [Google Scholar]
- 103.Joo EJ, Ahn YM, Park M, Kim SA. Significant shortening of leukocyte telomere length in Korean patients with bipolar disorder 1. Clin Psychopharmacol Neurosci. 2021;19:559–563. doi: 10.9758/cpn.2021.19.3.559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Tsankova N, Renthal W, Kumar A, Nestler EJ. Epigenetic regulation in psychiatric disorders. Nat Rev Neurosci. 2007;8:355–367. doi: 10.1038/nrn2132. [DOI] [PubMed] [Google Scholar]
- 105.Sarkisova KY, Gabova AV. Maternal care exerts disease- modifying effects on genetic absence epilepsy and comorbid depression. Genes Brain Behav. 2018;17:e12477. doi: 10.1111/gbb.12477. [DOI] [PubMed] [Google Scholar]
- 106.Szyf M, Weaver I, Meaney M. Maternal care, the epigenome and phenotypic differences in behavior. Reprod Toxicol. 2007;24:9–19. doi: 10.1016/j.reprotox.2007.05.001. [DOI] [PubMed] [Google Scholar]