Abstract
BACKGROUND
Heterotopic pancreas (HP) is an aberrant anatomic malformation that occurs most commonly in the upper gastrointestinal tract. While the majority of heterotopic pancreatic lesions are asymptomatic, many manifest severe clinical symptoms which require surgical or endoscopic intervention. Understanding of the clinical manifestations and symptoms of HP is limited due to the lack of large volume studies in the literature. The purpose of this study is to review symptomatic cases at a single center and compare these to a systematic review of the literature in order to characterize common clinical manifestations and treatment of this disease.
AIM
To classify the common clinical manifestations of heterotopic pancreas.
METHODS
A retrospective review was conducted of pathologic samples containing heterotopic pancreas from 2000-2018. Review was limited to HP of the upper gastrointestinal tract due to the frequency of presentation in this location. Symptomatic patients were identified from review of the medical records and clinical symptoms were tabulated. These were compared to a systematic review of the literature utilizing PubMed and Embase searches for papers pertaining to heterotopic pancreas. Publications describing symptomatic presentation of HP were selected for review. Information including demographics, symptoms, presentation and treatment were compiled and analyzed.
RESULTS
Twenty-nine patient were identified with HP at a single center, with six of these identified has having clinical symptoms. Clinical manifestations included, gastrointestinal bleeding, gastric ulceration with/without perforation, pancreatitis, and gastric outlet obstruction. Systemic review of the literature yielded 232 publications detailing symptomatic cases with only 20 studies describing ten or more patients. Single and multi-patient studies were combined to form a cohort of 934 symptomatic patients. The majority of patients presented with abdominal pain (67%) combined with one of the following clinical categories: (1) Dyspepsia, (n = 445, 48%); (2) Pancreatitis (n = 260, 28%); (3) Gastrointestinal bleeding (n = 80, 9%); and (4) Gastric outlet obstruction (n = 80, 9%). The majority of cases (n = 832, 90%) underwent surgical or endoscopic resection with 85% reporting resolution or improvement in their symptoms.
CONCLUSION
Heterotopic pancreas can cause significant clinical symptoms in the upper gastrointestinal tract. Better understanding and classification of this disease may result in more accurate identification and treatment of this malformation.
Keywords: Heterotopic pancreas, Ectopic pancreas, Aberrant pancreas, Pancreatic rest, Groove pancreatitis
Core Tip: This is a retrospective study that compares a single institution’s experience with that of a systematic literature review to evaluate the common clinical symptoms and presentation of heterotopic pancreas. To date, there have been numerous small volume studies in the literature reporting heterotopic pancreas. However, these studies predominantly report asymptomatic or incidental cases and there are few studies characterizing the symptomatic presentation of this disease. The goal of this study is to classify symptomatic cases of heterotopic pancreas to better identify the presentation and management of this disease.
INTRODUCTION
Heterotopic pancreas (HP) is an uncommon embryologic abnormality, defined as aberrant pancreatic tissue separated anatomically from the main body and blood supply of the pancreas. This tissue can be located throughout the gastrointestinal tract with a reported frequency of 1/500 in surgical specimens[1]. Most of these lesions are asymptomatic and are identified incidentally on pathologic specimens or at autopsy. However, some of these anatomic anomalies can manifest clinical symptoms. When this occurs, the clinical presentation can often be severe, requiring surgical intervention[2].
Knowledge of the clinical features of this disease process and the appropriate treatment is limited. While reports of clinically relevant cases are present in the literature dating back to 1727, the majority of the available literature consists of case reports, with few studies containing more than a handful of patients[3]. Thus, little is known about the “typical” clinical manifestations of HP due to small volume studies. Classification of this aberrancy is further limited by the diversity of possible symptoms which are often dictated by the anatomic location, size and functionality of ectopic lesion. While HP is most commonly located in the foregut, it has been identified in tissues including the small bowel, colon, gallbladder, spleen, esophagus and mediastinum[4]. This leads to further complexity in appropriate diagnosis and treatment of HP as it is often mimics other disease processes.
To address the deficit in description and classification of symptomatic HP we evaluated our center’s experience with symptomatic HP cases and compared it to a systematic review of the available literature. The scope of this study focuses on describing and characterizing the common clinical manifestations for HP within the stomach and duodenum as these locations represent the majority of cases. Cases identified in this study were compared with the results of a review of the available reported cases to identify the common symptoms, presentation, and treatment options for this disease process.
MATERIALS AND METHODS
A review of pathology specimens from 2000-2018 was conducted at a single academic institution. Approval was obtained from the institutional review board (#210811062). Specimens with pathologic confirmation of heterotopic pancreatic tissue were identified. Lesions found in the upper gastrointestinal tract were selected in adult patients over 18 for final review. Electronic medical records were analyzed and factors associated with the clinical presentation, symptoms, diagnosis and treatment were identified in each of the cases. Based upon the clinical history and presentation, patients were classified as symptomatic or asymptomatic. Symptomatic patients were defined as those who presented with upper gastrointestinal complaints which could be attributed to the identified pathology and ultimately resolved with treatment or removal of the ectopic tissue. Asymptomatic patients were classified as patients in whom the presence of heterotopic pancreas was identified in the workup or treatment of symptoms related to other clinically established pathology, or were found incidentally in a surgical pathologic specimen. Symptomatic patients were analyzed and categorized based upon their clinical features, presentation and treatment.
The cohort of patients with symptomatic manifestations from our institution was then compared that of published cases in the literature. A literature review of heterotopic pancreas using Embase and PubMED databases was conducted. The terms “heterotopic pancreas”, “ectopic pancreas”, and “aberrant pancreas” were searched. Publications printed in the English language were selected. Inclusion criteria were limited to publications reporting symptomatic cases of pathologically confirmed heterotopic pancreas located in stomach or duodenum. Publications which reported symptomatic and asymptomatic cases were excluded if they lacked characterization and description of clinical symptoms. Patients from the included publications were combined into a single cohort and categorized based on the presenting clinical symptoms, available demographic information and treatment. This sample was then compared with a series of patients identified at our institution for similarities. Descriptive statistics were employed to tabulate and report the results.
RESULTS
Institutional results
Twenty-nine pathology specimens were identified with pancreatic heterotopia in the upper gastrointestinal tract during the study period of January 2000 to December 2018. Of these, fourteen patients were female (48%). Six patients (20%) were identified as having symptoms related to an HP lesion. The remaining patients were identified as asymptomatic with incidentally noted lesions on pathology or discovered on endoscopy (Table 1). Five of the symptomatic patients underwent surgical treatment of their disease process and one declined surgery in favor of medical management.
Table 1.
Pathologic specimens identified with heterotopic pancreas; specimens are divided into symptomatic classification
|
Clinical features
|
n
|
Percent
|
| Total patients | 29 | 100% |
| Symptomatic HP | 6 | 20.0% |
| Pancreatitis | 2 | 7.0% |
| Bleeding/anemia | 2 | 7.0% |
| Peptic ulceration | 1 | 3.4% |
| Gastric outlet obstruction | 1 | 3.4% |
| Asymptomatic HP | 23 | 80.0% |
| Incidental finding on pathology | 5 | 17.2% |
| Noted on Endoscopy | 17 | 58.6% |
| Identified at time of surgery | 1 | 3.4% |
HP: Heterotopic pancreas.
Of the symptomatic patients, two (7%) presented with elevated lipase and evidence of pancreatitis within the heterotopic lesion, two patients presented with gastrointestinal bleeding or anemia, one patient (3.4%) presented with perforated gastric ulceration related to a heterotopic lesion, and one patient presented with gastric outlet obstruction from a HP lesion located at the pylorus. The six case presentations are briefly summarized below.
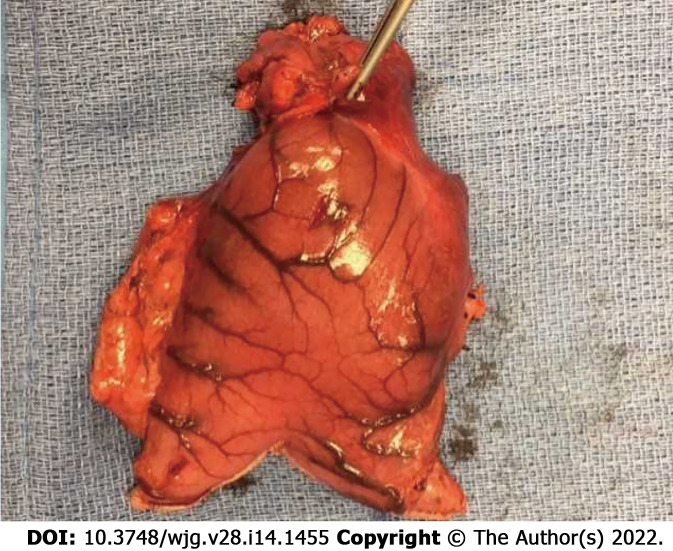
Case 1: 28-year-old male who presented as an outpatient with a history of mid-epigastric abdominal pain, nausea and daily vomiting for approximately 3 mo. He reported associated anorexia and a weight loss of approximately 40 Lbs. On exam he was noted to be cachectic with a BMI of 17 with moderate abdominal tenderness to palpation in the upper abdomen. Workup demonstrated an elevated lipase of 123 and an amylase of 141. Computed tomography revealed a 3.4cm hyper-enhancing mass near the pylorus on CT imaging. This was associated with a complex fluid collection within the wall of the stomach and evidence of inflammatory stranding, gastric thickening and edema. Endoscopy revealed a mass within the wall of the gastric antrum. Endoscopic ultrasound and biopsy demonstrated pancreatic acini. The patient was subsequently taken to the operating room for antrectomy and billroth II reconstruction. His pathology demonstrated heterotopic pancreatic tissue, 3.5 cm in size within the gastric antrum (Figure 1). In addition, there was an associated 4.3 cm pseudocyst within the gastric wall with surrounding inflammation. The patient was seen in follow up clinic one month later and was noted to be eating well with an increase in his weight of 14 Lbs since discharge.
Figure 1.
Image demonstrating the gastric antrum with an intramural mass identified within the wall of the stomach.
Case 2: 37-year-old male who presented with a chief complaint of melena, epigastric abdominal pain, anorexia and weight loss of approximately 20 Lbs. An esophagogastroduodenoscopy was performed which demonstrated a 2 cm submucosal mass with overlying ulcerated mucosa. An endoscopic ultrasound and FNA were obtained. Results demonstrated atypical cells. Radiographic imaging was obtained, however computed tomography scan was unable to identify or localize the mass. Surgical resection was offered, and the patient underwent a laparoscopic assisted gastric wedge resection. Pathology from the specimen was identified as a 2.5 cm × 2.0 cm mass consistent with multilobulated pancreatic tissue located between the gastric mucosa and muscularis propria with evidence of inflammation and ulcerated mucosa. The patient recovered without complication and with resolution of his anemia.
Case 3: 42-year-old male presented with acute abdominal pain and the presence of free air on upright abdominal film. He was noted to be tachycardic and with evidence of peritonitis on exam. A computed tomography scan confirmed the presence of free air and intraperitoneal fluid in the abdomen. His past medical history was notable only for a history of gastroesophageal reflux and indigestion that was refractory to years of proton pump inhibitor therapy. He was taken to the operating room for exploration where a large ulcer was identified upon opening the lesser sac in the posterior wall of the stomach approximately 6cm from the pylorus. A distal gastrectomy and reconstruction with a billroth II gastrojejunostomy was performed.
Pathology from the resected specimen demonstrated a transmural gastric perforation with evidence of associated pancreatic tissue within the gastric wall and along the edge of the ulcerated mucosa. The patient tolerated the procedure well and remained symptom free.
Case 4: 43-year-old male presented to the emergency department with chronic symptoms of non-bloody, non-billious vomiting and intolerance of oral intake. A detailed history revealed that the patient had suffered from intermittent vomiting and progressive oral intolerance over the prior 6 mo. A computed tomography scan was obtained which demonstrated a grossly distended stomach and a hyperenhancing mass as the level of the pylorus and first portion of the duodenum with an associated gastric outlet obstruction.
He was taken to surgery where an abnormal mass was palpated near the level of the pylorus. A distal gastrectomy was performed with a billroth II reconstruction. Final pathology demonstrated heterotopic pancreas at the level of the pylorus.
Case 5: 52-year-old female presented with anemia underwent upper endoscopy, which showed a submucosal mass along the lesser curve of the stomach near the incisura. EUS was performed, where a submucosal mas was identified with ulceration concerning for a gastrointestinal stromal tumor. Fine needle aspiration was non-diagnostic. She proceeded to surgery where a laparoscopic wedge resection was performed. Her pathology showed a 1.5 cm × 1.1 cm × 1.0 cm mass of pancreatic tissue. On post-operative follow up she was noted to have resolution of her anemia.
Case 6: 29-year-old male presented with abdominal pain and anorexia over the course of on year with a weight loss of 75 Lbs. Imaging with computed tomography showed 2.6 cm × 1.6 cm mass along the lesser curve of stomach and the patient was noted to have mild elevation in his amylase and lipase. Upper endoscopy with endoscopic ultrasound demonstrated a 2.3 cm subepithelial lesion along the lesser curve with surrounding gastritis. Biopsy demonstrated chronic gastritis and benign pancreatic tissue. The patient was placed on proton pump inhibitors and carafate with improvement in his symptoms. He was referred for surgical evaluation but declined intervention as his symptoms had improved with medical therapy.
Results of literature analysis
A systematic review of the literature was conducted to identify published cases of HP lesions. The initial search resulted in 1220 unique publications. Of these, 232 publications described cases of symptomatic lesions and met the inclusion criteria. The majority of these publications (n = 182) were single patient case studies. There were only 20 studies identified which contained more than 10 symptomatic patients in their series. The majority of the larger studies contained cases of both symptomatic and asymptomatic disease with few publications dedicated to describing symptomatic cases. In total, these studies included 1762 patients of which 934 patients were reported to have symptomatic lesions in the stomach or duodenum and comprised the total cohort of this evaluation. Demographic information was provided for 864 patients (94%) of which the mean age at symptom presentation was 43 years old and 564 were male (61%). In total, there were 556 patients with gastric lesions (59%) and 378 with duodenal lesions (41%).
A summary of the clinical cases of HP was performed by individual publication and were tabulated for review in Table 2. The majority of patients presented with epigastric abdominal pain (n = 620, 67%). In addition to pain, the remaining symptoms were noted to fall within one of 4 predominant diagnostic categories: (1) Dyspepsia, which included symptoms of epigastric pain, eructation, nausea or bloating with meals (n = 445, 48%); (2) Pancreatitis within the ectopic lesion (n = 260, 28%); (3) Gastrointestinal bleeding (n = 80, 9%); and (4) Gastric outlet obstruction (n = 80, 9%). There were also 37 patients who presented with jaundice and biliary obstruction due to periampullary lesions (4%), and 3 patients with perforated ulcers. The remaining patients presented with various symptoms which included fever, diarrhea, abscess, carcinoid syndrome and dysphagia but were in single cases or small numbers. The majority of patients who presented with pancreatitis were found to have radiographic evidence of pseudocyst formation or cystic degeneration of an ectopic lesion (n = 251, 97%). These lesions were commonly located within the wall of the stomach or duodenum or were characterized as “groove pancreatitis” with ectopic pancreatic tissue located between the medial duodenal wall and the head of the pancreas. Of the patients with clinical and radiographic evidence of pancreatitis, only 50 cases (19%) reported elevations in biochemical markers such as serum amylase or lipase to correlate with the clinical or radiographic evidence of pancreatitis. One hundred forty-five cases (56%) were reported to be associated with alcohol consumption as the inciting etiology.
Table 2.
Summary of systematic literature review listed by publication, number of symptomatic cases, demographics and clinical symptoms
|
Ref.
|
Year
|
Total patients
|
Symptomatic patients
|
Mean age
|
Gastric lesions
|
Duodenal lesions
|
Location other than foregut
|
Abdominal Pain
|
Dyspepsia
|
Gastrointestinal Bleeding
|
Pancreatitis
|
Gastric outlet obstruction
|
Perforation
|
Nausea
|
Jaundice
|
|
| Noh et al[5] | 2019 | 5 | 5 | 32 | 5 | 5 | 5 | |||||||||
| Zhou et al[6] | 2019 | 93 | 78 | 39.75 | 78 | 38 | 41 | 4 | ||||||||
| Betzler et al[7] | 2017 | 67 | 11 | 53 | 11 | 8 | 7 | |||||||||
| Jun et al[8] | 2017 | 165 | 79 | 45.2 | 46 | 18 | 15 | 64 | 64 | 64 | ||||||
| Zhang et al[9] | 2016 | 184 | 26 | 49 | 25 | 1 | 26 | 26 | 1 | |||||||
| Liao et al[10] | 2014 | 2 | 2 | 36 | 2 | 2 | ||||||||||
| Chou et al[11] | 2014 | 13 | 13 | 40.8 | 13 | 6 | 7 | |||||||||
| Endo et al[12] | 2014 | 2 | 2 | 74 | 1 | 1 | 1 | 1 | ||||||||
| Liu et al[13] | 2013 | 9 | 9 | 48.3 | 9 | 6 | 9 | 1 | ||||||||
| Zhong et al[14] | 2013 | 60 | 30 | 39 | 30 | 16 | 24 | 6 | ||||||||
| Shah et al[15] | 2011 | 30 | 4 | 53 | 1 | 1 | 2 | 1 | 1 | 1 | ||||||
| Park et al16] | 2011 | 26 | 9 | 34 | 9 | 4 | 9 | |||||||||
| Wei et al[17] | 2011 | 11 | 11 | 52 | 3 | 8 | 6 | 8 | 3 | 2 | ||||||
| Ryu et al18] | 2010 | 8 | 4 | 36 | 4 | 4 | ||||||||||
| Yuan et al[19] | 2009 | 2 | 2 | 61 | 1 | 1 | 1 | 1 | ||||||||
| Khashab et al[20] | 2009 | 2 | 2 | 32 | 2 | 1 | 1 | |||||||||
| Casetti et al[21] | 2009 | 58 | 58 | 44.7 | 58 | 46 | 1 | 58 | ||||||||
| Jovanovic et al[22] | 2008 | 13 | 13 | 41.5 | 13 | 10 | 6 | 1 | 7 | 4 | ||||||
| Chen, H et al[23] | 2008 | 39 | 15 | 46 | 12 | 1 | 2 | 9 | 9 | 5 | ||||||
| Chen, S et al[24] | 2008 | 20 | 13 | 39 | 12 | 1 | 9 | 4 | ||||||||
| Rebours et al[25] | 2007 | 105 | 105 | 46 | 105 | 91 | 5 | 105 | 30 | 13 | ||||||
| Tison et al[26] | 2007 | 9 | 9 | 49.6 | 9 | 8 | 9 | 9 | 1 | |||||||
| Ormarsson et al[4] | 2006 | 30 | 11 | 49.2 | 11 | 5 | 5 | 1 | ||||||||
| Pessaux et al[27] | 2006 | 12 | 12 | 42.4 | 12 | 11 | 1 | 12 | 8 | |||||||
| Ayantunde et al[28] | 2006 | 3 | 3 | 55 | 3 | 1 | 1 | 2 | 2 | |||||||
| Chatelain et al[29] | 2005 | 2 | 2 | 40.5 | 2 | 2 | 2 | 2 | ||||||||
| Zinkiewicz et al[30] | 2003 | 12 | 7 | 34 | 6 | 1 | 7 | 7 | ||||||||
| Shi et al[31] | 2002 | 7 | 7 | 41 | 1 | 4 | 2 | 2 | 1 | 1 | 3 | |||||
| Huang et al[32] | 2002 | 2 | 2 | 46 | 2 | 2 | ||||||||||
| Otani et al[33] | 2000 | 34 | 2 | 43 | 2 | 1 | 1 | |||||||||
| Hsia et al[34] | 1999 | 17 | 10 | 47.5 | 8 | 2 | 8 | 2 | ||||||||
| Fekete et al[35] | 1996 | 6 | 6 | 40 | 1 | 5 | 6 | 1 | 6 | |||||||
| Flejou et al[36] | 1993 | 10 | 10 | 41 | 1 | 9 | 7 | 1 | 10 | 2 | ||||||
| Claudon et al[37] | 1988 | 2 | 2 | 41 | 2 | 2 | 2 | |||||||||
| Pang et al[38] | 1988 | 32 | 14 | 44 | 3 | 4 | 7 | 6 | 5 | 2 | 1 | 1 | ||||
| Lai et al[39] | 1986 | 37 | 9 | 50 | 6 | 3 | 7 | 7 | 1 | 1 | ||||||
| Mollitt et al[40] | 1984 | 9 | 6 | 6.8 | 6 | 3 | 3 | 3 | ||||||||
| Armstrong et al[41] | 1981 | 34 | 13 | 51 | 5 | 5 | 3 | 11 | 2 | |||||||
| Thoeni et al[42] | 1980 | 9 | 6 | 43 | 6 | 1 | 1 | 1 | 1 | 1 | ||||||
| Yamagiwa et al[43] | 1977 | 64 | 26 | 45.2 | 23 | 3 | 17 | 15 | 4 | 8 | ||||||
| Dolan et al[44] | 1974 | 212 | 73 | 60 | 43 | 25 | 5 | 52 | 46 | 7 | 1 | |||||
| Eklof et al[45] | 1973 | 4 | 4 | 7 | 3 | 1 | 3 | 2 | 1 | |||||||
| Nebel et al[46] | 1973 | 3 | 3 | 28 | 1 | 2 | 3 | 3 | ||||||||
| McGarity et al[47] | 1971 | 2 | 2 | 33 | 2 | 2 | 1 | 2 | ||||||||
| Abrahams et al[48] | 1966 | 9 | 9 | 49.8 | 5 | 4 | 9 | 6 | ||||||||
| Tonkin et al[49] | 1962 | 4 | 4 | 44 | 2 | 2 | 3 | 4 | 1 | |||||||
| Dirks et al[50] | 1961 | 3 | 3 | 44.6 | 0 | 3 | 3 | 2 | 1 | |||||||
| Martinez et al[51] | 1958 | 51 | 28 | 43.1 | 21 | 7 | 16 | 19 | 2 | 3 | ||||||
| Waugh et al[52] | 1946 | 5 | 5 | 34.8 | 5 | 3 | 3 | 2 | ||||||||
| De Castro Barboso et al[1] | 1947 | 41 | 17 | 43 | 10 | 7 | 10 | 11 | 4 | |||||||
| Single case studies (n = 182)[3,53-233] | 182 | 182 | 39.1 | 126 | 56 | 96 | 66 | 21 | 38 | 51 | 3 | 57 | 12 | |||
| Total | 1761 | 978 | 43 | 556 | 378 | 44 | 620 | 445 | 80 | 260 | 80 | 3 | 188 | 37 | ||
The majority of cases (n = 832, 90%) underwent surgical or endoscopic management of their heterotopic pancreatic lesion, while 92 were observed or managed medically. Surgical therapy ranged from radical gastrectomy or pancreaticoduodenectomy to local excision or endoscopic resection. Available data on the therapies provided are listed in Table 3. Data on the effect of treatment on symptom control was available for 645 patients. Of the cases which reported post-intervention follow up, five hundred forty-nine patients demonstrated symptom resolution with treatment (85%), 63 were reported to have symptom improvement (9.7%) and 33 reported no improvement (5.1%). Based on the level of detail reported in the literature, there was no delineation of whether patients who did not improve were treated medically or via surgical or endoscopic intervention. Likewise, there was no study providing a direct comparison of treatment outcomes between medical versus surgical therapies nor was there a study comparing outcomes between different interventional therapies.
Table 3.
Treatment of heterotopic pancreas; listing of surgical or endoscopic procedures performed for patients with symptomatic heterotopic pancreas by procedure
|
Procedure
|
n
|
Percent (%)
|
| Distal gastrectomy | 84 | 10.50 |
| Billroth I reconstruction | 14 | 1.70 |
| Billroth II reconstruction | 64 | 8.00 |
| Roux-en-Y reconstruction | 6 | 0.75 |
| Gastric wedge resection | 81 | 10.00 |
| Subtotal gastrectomy | 18 | 2.25 |
| Total gastrectomy | 5 | 0.63 |
| Gastrotomy and local excision | 42 | 5.30 |
| Partial gastrectomy NOS | 215 | 27.00 |
| Pancreaticoduodenectomy | 168 | 21.00 |
| Trans-duodenal excision | 19 | 2.40 |
| Partial duodenectomy | 7 | 0.90 |
| Ampullectomy | 2 | 0.25 |
| Endoscopic submucosal excision | 158 | 19.77 |
| Total procedures | 799 | 100.00 |
DISCUSSION
Heterotopic pancreas is a congenital anomaly, often identified incidentally in asymptomatic patients, yet it has the ability to illicit significant clinical symptoms in some cases. These lesions can arise in tissues throughout the body including stomach, small bowel, gallbladder, esophagus and mediastinum, leading to a wide range of manifestations[234]. Characterization of symptomatic lesions is difficult due to the relative infrequency of this diagnosis and the variability in presentation. This often leads to misidentification and suboptimal management in many cases. Large volume studies characterizing the common presentation of HP are lacking and the aim of this study was to provide a conglomerate population to classify the typical clinical presentation within the foregut and aid in classification. With improved understanding and recognition of this disease process, clinicians can render more appropriate treatment decisions that may save patients morbidity from radical resection.
The clinical manifestations of HP are dictated in large part by two factors, the location and the functionality of the lesion. The anatomic location is often predetermined early in life by embryologic development. The exact mechanism by which this abnormality forms is not well understood however, it is believed to be related to a disruption in the normal embryologic migration of the pancreas. Formation of dorsal and ventral pancreatic buds results from endodermal outpouchings of the duodenum[235]. Aberrant separation of these tissues from the visceral endoderm may be responsible for retained pancreatic tissue within the developing gastrointestinal tract and subsequent migration as the embryo develops[235]. However, this theory does not fully account for the development of HP in all cases as this anomaly has also been reported to be found in tissues outside the gastrointestinal tract including the gallbladder, fallopian tube, umbilicus, mediastinum, omentum, mesentery and spleen[9,39]. An alternative explanation in these cases is the “metaplasia” theory in which totipotent endodermal tissues develop into pancreatic tissue during embryologic development and can present in various tissues throughout the body[235].
However, the majority of these lesions are located within the upper gastrointestinal tract with surgical and autopsy data reporting a frequency within the stomach of 25-52%, 27-36% in the duodenum and 15%-17% in the jejunum[9,44,235]. HP symptoms often mimic common upper gastrointestinal pathology and are difficult to identify. The location within the foregut often dictates symptom manifestation with peripyloric lesions more likely to lead to gastric outlet obstruction while periampullary lesions may cause biliary obstruction[17,80,164,236,237].
In addition to the anatomic location, the histologic location within the layers of the visceral wall may also have an effect on symptom presentation. HP occurs most frequently between the submucosa and lamina propria but can be found within all layers of the visceral wall[238]. Submucosal lesions may be more likely to cause ulceration with local gastritis or duodenitis while transmural lesions which involve all layers of the bowel wall can lead to chronic inflammation, and ultimately stricture or perforation[77].
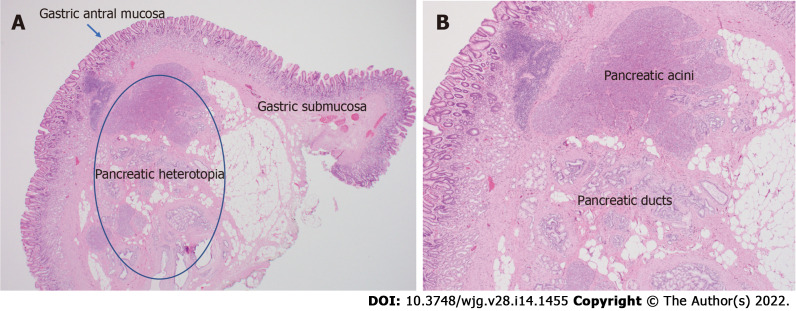
While location may often explain symptom presentation it does not fully account for why some lesions are symptomatic while others found in similar locations are not. The remaining factor can be potentially explained by the functionality of the lesion, and the ability to perform the normal exocrine and endocrine functions of the pancreas. Histopathologic evaluation by Heinrich in 1909 demonstrated differences in the composition of these lesions which were later revised by Fuentes in 1973[239,240]. These classification systems categorized HP lesions based on the presence of all the cellular components of functional pancreas tissue (Type I in both categories) versus presence of ducts, acinar tissue or islet cells alone (Fuentes classification types II, III and IV respectively)[240] (Figure 2). The presence or absence of histologic elements may affect the functionality of the lesion and contribute to its ability (or inability) to produce symptoms. It has been proposed that lesions with functional exocrine potential can produce local chemical irritation of surrounding tissues while those without appropriate ductal drainage may lead to pancreatitis and pseudocyst formation within the lesion[4,60].
Figure 2.
Histopathologic images. A: Histologic appearance of heterotopic pancreas in the stomach; B: High power view of heterotopic pancreas demonstrating pancreatic acinar and ductal architecture.
The clinical nature of HP may therefore have variable patterns of presentation which are highly dependent on both the location and the functional ability of the lesion. Since the majority of identified cases are located within the upper gastrointestinal tract, the scope of this study was restricted to identifying and characterizing symptoms within these locations. The combined experience from our center and the analysis of the published literature demonstrated that the majority of clinically significant cases can be categorized within 4 main groups. These categories are significant as they are common presentations of other pathology within the upper GI tract and can lead to difficulty in diagnosing and treating this pathology.
Dyspepsia
The majority of cases identified in this series reported symptoms consistent with epigastric abdominal pain, bloating, belching and nausea which are typically associated with eating. This category consisted predominantly of patients with gastric lesions. The manifestation of symptoms has been thought to be related to local chemical irritation of the gastric mucosa by products of the exocrine pancreas[87]. This acts as an inciting factor that then may lead to inflammatory mucositis and subsequent gastritis or duodenitis, with or without ulceration. As many of these patients present with identical symptoms to those with peptic ulcer disease (PUD) (and indeed, are often misdiagnosed as such), many authors argue that ulceration is the inciting factor and etiology of the majority of symptoms associated with HP[2,44]. In the series of 34 patients reported by Armstrong et al[41] the extent of mucosal involvement on pathology was correlated with symptoms suggesting a causal relationship. In the series published by Lai et al[39] comparing the histology of symptomatic and asymptomatic patients, 49% of symptomatic patients had mucosal ulcers while asymptomatic patients in their series lacked objective evidence of ulceration or gastritis.
Mucosal ulceration in some cases may progress to visceral perforation. This is thought to be the result of normal evolution of inflammation seen in peptic ulcer disease, with ulceration of the protective mucosal barrier, caused by heterotopic pancreas, acting as the initial catalyst. Perforation may also be related the degree of penetration within the gastric wall of the ectopic lesion. Extension of the HP lesions through the serosa can be seen in up to 4% of histologically examined specimens[84]. Lesions which penetrate deeper through the layers of the gastric wall can lead to transmural inflammation, necrosis and perforation of the stomach wall. This theory is supported by the histologic findings reported by Martinez et al[51] demonstrating inflammation and fibrosis extending through multiple layers of the gastric wall on tissue histology depending on the histologic depth of the lesion. Overall, this is a rare complication of HP, with only three identified publications describing visceral perforation related to HP[77,121,241]. We report the fourth known case above in Case 3 where the transmural gastric ulceration was clearly associated with HP. It is possible that additional cases exist but remain undocumented as they may have been treated with omental patching without resection or biopsy. Still, this remains an uncommon symptomatic presentation of HP compared to local mucosal ulceration.
While there are clear associations for mucosal ulceration with the symptomatic presentations of HP it does not account for all cases. Multiple series including that of Dolan et al[44] report on patients with symptoms of dyspepsia who did not exhibit evidence of ulceration or gastritis. This is also supported by reports such as that by Ormarsson et al[4] and Barbosa et al[242] which demonstrate resolution of symptoms in patients undergoing resection of HP with and without the presence of ulceration. In these patients, the etiology of abdominal pain and/or dyspepsia is not fully clear. It has been proposed that exocrine secretions into the stomach may affect the pH composition of the gastric lumen leading to alterations in the digestive process, gas formation and bloating, although confirmation of this via testing of gastric pH in the setting of HP has not been conducted to our knowledge[47]. In addition, it is hard to conceive that these lesions, unless the reach a large size, are able to secrete enough volume to significantly alter the pH composition of gastric juices.
The majority of patients with HP and dyspeptic symptoms are initially misdiagnosed and managed with medical therapy designed for reduction of acid secretion. It is unclear whether traditional treatments for gastritis and PUD is beneficial in these patients. If the inciting physiology is chemical irritation from pancreatic secretions, then traditionally prescribed therapies to reduce acid secretion would be unlikely to help alleviate symptoms. In patients who develop mucosal ulceration there may be some plausible benefit that acid reduction may prevent ulcer progression once breakdown of the mucosal barrier has already occurred. However, it is not clear whether this can promote adequate ulcer resolution in the setting of HP or prevent future recurrent ulceration since acid secretion is not believed to be the causative factor for inflammation or ulceration in this setting[41]. There were few symptomatic patients in the reviewed studies who were managed with observation or medical therapy and long term follow up was lacking. The single patient treated with medical therapy at our institution (Case 6), demonstrated symptomatic improvement and to date, has not returned with recurrent symptoms over six years of observation. However, an adequate evaluation of medical management compared to surgical or endoscopic resection is lacking and further study is needed to evaluate the optimal treatment modality.
Heterotopic pancreatitis
Manifestations of acute and chronic pancreatitis in HP tissues were the second most common presentation in our review. Clinical symptoms may include abdominal pain, nausea, vomiting, anorexia and weight loss[85,163]. Similar to orthotopic pancreatitis, the most common causes can be related to alcohol, medications, smoking, autoimmune disease or inadequate ductal drainage[243]. According to the histologic classification system proposed by Heinrich and Fuentes, heterotopic pancreas types I-III contain functional pancreatic acini and ductal components. Impaired drainage and outflow obstruction of these ducts may lead to pancreatitis and pseudocyst formation[241]. Cystic formation within the visceral wall of the stomach or duodenum was demonstrated radiographically in the majority of patients with pancreatitis in our series. This was often associated with radiographic signs of stranding, edema and inflammation. This pathophysiology is commonly seen in “groove pancreatitis” where ectopic pancreatic tissue between the duodenal wall and pancreas lack appropriate enteric drainage leading to inflamation and cystic degeneration[244,245].
Diagnosis of heterotopic pancreatitis is made through a combination of clinical presentation, radiographic imaging and biochemical evaluation[243,245]. Serum labs may yield mild elevations of amylase and lipase however, these levels are usually only modestly elevated, even in the setting of severe inflammation due to the relatively small volume of pancreatic tissue within the heterotopic lesion[235]. Leakage of enzyme-rich pancreatic fluid can cause pancreatic pseudocyst formation within the gastric wall producing symptoms of early satiety and gastric outlet obstruction[60]. This finding results in the appearance of a cystic structure within the wall of the stomach or bowel and is often mistaken for a duplication cyst or mucinous neoplasm[208].
Conservative treatment may be successful in mild cases of this disease process. Investigation of the inciting etiology should be performed with cessation of alcohol or smoking when applicable. However, in patients with repeated episodes of abdominal pain, oral intolerance and vomiting (as in Case 1 above), surgical intervention may be required. When accurately diagnosed, drainage of the pseudocyst and local excision is typically adequate. However, in patients with recurrent groove pancreatitis or cystic degeneration of the duodenal wall, more radical surgical intervention is often required. Groove pancreatitis may require pancreaticoduodenectomy in order to resolve recurrent flairs of pancreatitis and avoid complications such as duodenal stricturing and necrosis of the pancreatic head. The largest series of patients with groove pancreatitis and cystic degeneration of the duodenal wall was published by Rebours et al[25] and included 105 patients. In this series, 18% resolved with observation, 43% resolved with medical therapy, of which half of these required nutritional support and 39% required surgical intervention. The majority of patients underwent a pancreaticoduodenectomy (N=`17) with the remainder undergoing endoscopic cyst fenestration, biliary bypass or gastric bypass for symptom management[25].
Gastrointestinal bleeding
Gastrointestinal bleeding is a rare but potentially serious presentation of HP. Bleeding may manifest as chronic melena as in the two patients in our series (Case 2 and 5). However, there are also reports of massive gastrointestinal hemorrhage associated with large volume transfusion and hemodynamic instability[86,191]. Martinez et al[51] reported a rate of bleeding of 7% in gastric lesions which was supported by the experience of Pang et al[38] who also reported a bleeding rate of 7% in symptomatic patients. The largest series of HP published by Dolan et al[44] from the Mayo Clinic evaluated 212 cases of HP of which 40 were discovered due to clinical symptoms. Of these, 7 patients (17.5%), presented with anemia and or evidence of gastrointestinal bleeding. Overall, the rate of patients presenting with gastrointestinal bleeding in pooled analysis of the literature was 9%.
The etiology of bleeding in patients with heterotopic pancreas is believed to be related to ulceration of the gastric mucosa overlying the aberrant lesion. As described previously, chemical irritation and inflammation can lead to disruption of the mucosal barrier while pancreatic enzymes, such as elastase, may cause thinning and erosion into the walls of underlying vasculature. Ulceration as the source of gastrointestinal bleeding is supported by the series put forth by Pang and colleagues which reported ulcerated mucosa in pathologic specimens from patients with evidence of bleeding[38,86]. However, this does not account for all reported cases of bleeding related to HP as there are several cases of documented bleeding and anemia in which the mucosa overlying the resected lesion was found to be intact[246]. One proposed explanation for this is through the concept of hemorrhagic “suffusion”. This idea, originally proposed by Madinaveita and Loma, and later described by Hudock and colleagues speculates that chronic inflammation from heterotopic lesions leads to edema and congestion in the gastric submucosa[191,247]. Congestion of the fragile vasculature in the submucosa may lead to bleeding and diapedesis into the gastric lumen.
Definitive management of major bleeding caused by heterotopic pancreatic lesions is primarily through resection. There is no clear role for medical management in gastric bleeding related to HP. Even among those patients with bleeding related to gastric ulceration, traditional medical management for gastric ulceration is of questionable utility as the presumed etiology of mucosal ulceration is not related to acid secretion. However, there may be an argument that medical therapy may reduce the potential of gastric secretions to propagate ulceration in the setting of an already disrupted mucosal barrier from HP. In the setting of acute gastrointestinal bleeding, endoscopic therapies can be utilized to control hemorrhage but ultimately, resection of the offending lesion should be considered. There have been reports of endoscopic submucosal dissection and resection of HP in patients with melena and chronic anemia[14,18]. However, surgical resection remains the preferred management of a bleeding mass in most cases. This may be achieved with localized partial gastrectomy or duodenectomy in the setting of small lesions and more extensive resection and reconstructions can often be avoided.
Gastric outlet obstruction
Gastric outlet obstruction is the source of symptomatic presentation in 9% of patients based on our review. The most common location for lesions arising in the stomach is the distal antrum, accounting for 85-96% of cases[94]. Given the proximity to the pylorus, growth of these lesions may lead to obstruction of the pyloric channel. While complete gastric outlet obstruction is possible, the more common scenario is partial or intermittent obstruction from a ball-valve occlusion or protrusion of the mass into the pylorus[51]. Symptoms associated with this process include abdominal pain, nausea, intermittent vomiting, bloating and anorexia[81,162]. Gastric outlet obstruction is a common presentation in infants and pediatric patients where smaller lesions can easily occlude the pyloric channel[32,247]. Nevertheless, obstruction is also seen in adults where ectopic lesions grow to a size that can lead to mechanical obstruction as seen in Case 4[41]. Lesions that are small and otherwise asymptomatic for decades, may also become acutely symptomatic in setting of inflammation and edema[60,92]. Likewise, the development of cysts and fluid collections within the wall of the stomach may grow to a size that can impede gastric emptying[163]. Some have also proposed that HP lesions may cause functional obstruction due to pyloric spasm[28].
Definitive management involves gastric decompression with a nasogastric tube and surgical correction of the obstruction. Surgical resection in these scenarios will typically require a more extensive resection than local excision or wedge resection as lesions causing obstruction are typically large (> 2 cm) and located at or near the pylorus or duodenum. Distal gastrectomy with a Billroth I, Billroth II or Roux-en-Y reconstruction are the most common operations performed for gastric lesions causing obstruction while partial duodenectomy or pancreaticoduodenectomy may be required in the obstructing lesion in the duodenum. If the lesion is small with a large cystic component causing obstruction and the pathology of the lesion is definitively known prior to surgery, a less extensive resection can be accomplished with preservation of the pylorus[95]. In the case series published by Ayantunde et al[28], two out of three cases of gastric outlet obstruction underwent distal gastrectomy while one underwent an anterior gastrotomy and local resection of the submucosal lesion. The majority of studies reviewed in the literature however, reported more extensive surgical resection and reconstruction procedures[14,58,64,244].
Evaluation and treatment
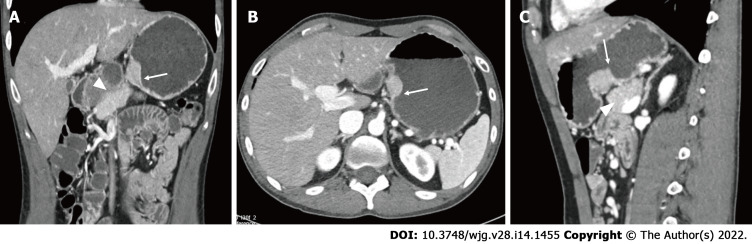
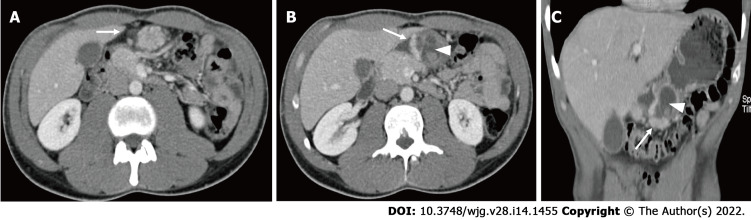
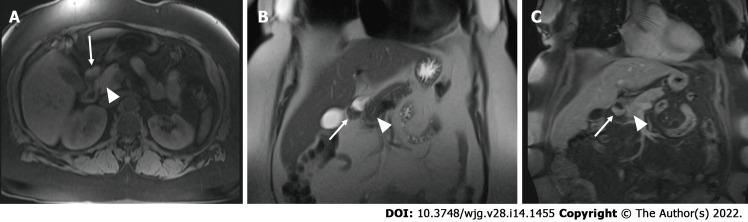
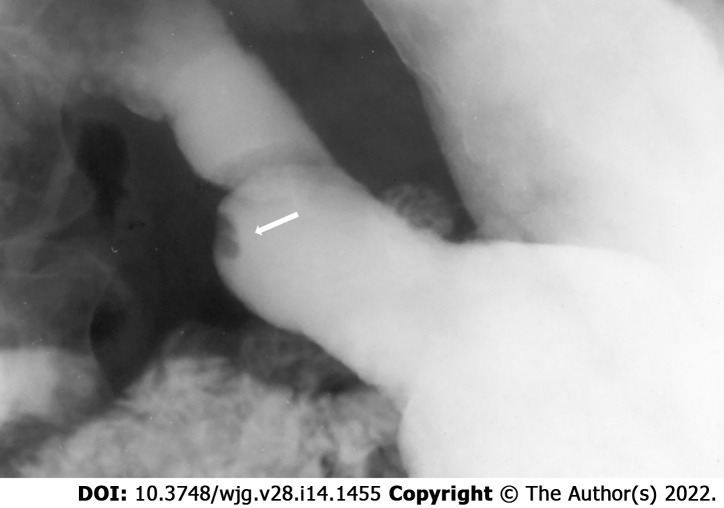
Regardless of the presenting symptoms, accurate diagnosis of HP can alter management and affect surgical decision making. Appropriate imaging and diagnostic studies must be ordered and evaluated carefully if suspicion for HP is high. Lesions of sufficient size can be identified with computed tomography, MRI, and fluoroscopy. When found within the stomach, HP tissue is typically seen along the greater curvature within 6 cm of the pylorus[238]. On CT, uncomplicated HP tissue will present as a soft tissue mass within the wall of the stomach, exhibiting similar enhancement and attenuation characteristics to that of normal pancreatic tissue[17] (Figure 3). However, the attenuation can be variable depending upon the dominant cellular type in the HP tissue, as there are pathologically different structures of heterotopic tissue described and characterized Heinrich's classification system[95,248]. Due to the variable enhancement of the HP tissue, it can sometimes be difficult to differentiate it from a gastrointestinal stromal tumor (GIST) or other common submucosal lesions[163]. In the setting of elevated pancreatic enzymes, sequelae of acute inflammation can be identified surrounding the ectopic tissue (Figure 4). On MRI the imaging of HP tissue will follow the signal intensity and enhancement characteristics of the native pancreas on all sequences. The HP tissue will demonstrate high signal intensity similar to that of the native pancreas on non-contrast T1 weighted imaging and demonstrate avid enhancement on post contrast images[234] (Figure 5). Barium fluoroscopy may also demonstrate a characteristic rounded filling defect with central indentation[241] (Figure 6).
Figure 3.
Coronal (A), axial (B), and (C) sagittal images of the abdomen and pelvis following the administration of IV contrast demonstrates enhancing heterotopic pancreatic tissue within the wall of the stomach along the lesser curvature (white arrows). The heterotopic pancreatic tissue is intramural in location and demonstrates similar attenuation characteristics of the adjacent pancreatic tissue seen on the coronal and sagittal images (white arrowhead).
Figure 4.
Computed tomography images. A: Axial image demonstrating heterotopic pancreatic tissue within the stomach (white arrow). B: Axial image demonstrating heterotopic pancreas (white arrow) with associated psudocyst (white arrowhead). C: Coronal reformatted image demonstrating heterotopic pancreas tissue (white arrow) with associated pseudocyst (arrowhead) causing gastric outlet obstruction.
Figure 5.
Non-contrast axial T1 weighted (A), coronal T2 weighted (B), and coronal postcontrast T1 weighted (C) images show a lesion within the first portion of the duodenum (white arrow) demonstrating T1 pre-contrast hyperintense signal similar to that of the adjacent pancreas (white arrowhead). This tissue shows similar imaging characteristics of the adjacent pancreatic tissue on the T2 coronal and T1 coronal post contrast images.
Figure 6.
Single spot image of the stomach on a barium fluoroscopic study demonstrates an intraluminal filling defect within the stomach with a central indentation (arrow) consistent with pancreatic heterotopia.
Further diagnostic workup may require biochemical evaluation of serum amylase, lipase or, in some cases, tumor markers and/or endoscopic investigation[249]. EGD will typically demonstrate the presence of a submucosal mass which may be associated with a central umbilication and or ulceration. At centers with expertise in endoscopic ultrasound, this modality may be helpful in determining the size, location and depth of penetration within the visceral wall. Final determination is achieved by tissue biopsy. This can often be difficult in the setting of ulceration, cystic degeneration and in lesions that are located within the outer wall of the viscera and EUS will may help facilitate accurate targeting and biopsy.
Accurate identification of HP in the diagnostic workup is an important factor in determining appropriate management. Unfortunately, many patients are misdiagnosed or assumed to have alternative pathology at the time of surgery. The range of clinical and radiographic findings makes proper pre-operative identification difficult, especially in the context of a relatively rare anatomic anomaly. Most lesions are mistakenly identified as malignant or premalignant pathology. This often affects the surgical decision making and may lead to a more extensive resection than would otherwise be required. This is evident in studies such as the one performed by Zhang et al[9] which reported that over 54% of patients with HP were misdiagnosed preoperatively. Many of the lesions were presumed to represent malignancy and underwent extensive resections. In our series, 18 patients underwent subtotal gastrectomy, 78 underwent distal gastrectomy and reconstruction and 168 underwent a pancreaticoduodenectomy. As the majority of HP lesions are benign and do not involve the entire visceral wall, many are amenable to local or endoscopic resection. Overall, we identified 158 cases of local endoscopic resection in our series. The largest series was published by Zhou et al[6] who reported endoscopic submucosal dissection in 78 symptomatic patients with good results. In this series, the majority of patients had lesions located in the submucosa or lamina propria and were less than 1cm in size. Likewise, the series published by Zhong et al[14] included 30 patients of which 90% were under 2cm in size. Thus, an endoscopic approach is a reasonable intervention in pathologically confirmed HP, < 2 cm in size in the submucosa.
Limitations
There are several limitations to this study. First, the relatively rare presentation of the disease process makes it difficult to identify a sufficiently large patient population to analyze and draw conclusions from. For this reason, we combined our single institution experience with that of a cohort derived from a literature review in order to evaluate a meaningful number of symptomatic cases. This type of analysis can be subject to inconsistency due to the differences in reporting, and classification of symptomatic cases in the literature. Likewise, a cohort derived from retrospective review of the literature may lead to selection bias as only those cases which are clearly symptomatic or associated with a higher level of severity are reported, potentially neglecting less symptomatic or milder cases. However, given the paucity of data and characterization of this abnormality, the reporting of the known literature and experience of HP in a conglomerate population may be important in the understanding and management of patients and to help guide future analysis and treatment decisions.
CONCLUSION
Heterotopic pancreatic tissue is a relatively rare anatomic anomaly that is found within the stomach and gastrointestinal tract. While numerous case reports of this pathology exist within the literature, there are few large volume studies that describe and characterize the clinical significance of this disease process. Review of a single institutional series and the available literature identified four categories of symptomatic features that occur within the upper gastrointestinal system. Many of these lesions are misdiagnosed and undergo invasive surgical resection. Better understanding of the clinical presentation and diagnosis of these lesions may lead to improved treatment and reduction of unnecessary surgical resection.
ARTICLE HIGHLIGHTS
Research background
Heterotopic pancreas (HP) is a rare embryologic anomaly that occurs when aberrant pancreatic tissue develops independently from the main body of the main body of the pancreas. This can occur throughout the body and develop in various tissues however, this most commonly occurs in the stomach and duodenum. While the majority of these lesions are asymptomatic, many have the potential to cause harmful clinical manifestations.
Research motivation
Symptomatic heterotopic pancreas is difficult to identify and often mimics common upper gastrointestinal pathology making diagnosis and treatment challenging. Symptomatic cases of HP often require surgical or endoscopic management however, the optimal treatment strategy is poorly understood at this time and patients often undergo radical resection.
Research objectives
The purpose of this study was to review the cases of symptomatic HP at a single, high volume academic institution and compare these with a cohort of patients derived from a systematic review of symptomatic cases in the English literature. This combined cohort was used to classify the common presentation and clinical symptoms for HP and identify the various treatment options.
Research methods
A retrospective review was conducted of pathologic specimens containing heterotopic pancreas at a single, high volume, academic medical center. Symptomatic cases were identified and analyzed for details related to presentation, symptoms and treatment. A systematic review was then conducted utilizing Embase and PubMed databases for publications in English which pertained to heterotopic pancreas. Publications which included symptomatic cases of HP were included for final literature analysis and results were tabulated into a final cohort for review.
Research results
Institutional review: Twenty-nine cases of HP were identified at a single institution, of which, six presented with symptoms (20%). Two patients (7%) presented with elevated lipase and evidence of pancreatitis within the heterotopic lesion, two patients presented with gastrointestinal bleeding or anemia, one patient (3.4%) presented with perforated gastric ulceration related to a heterotopic lesion, and one patient presented with gastric outlet obstruction from a HP lesion located at the pylorus.
Systematic review: Literature review resulted in identification of 1220 unique publications related to HP of which 232 publications met inclusion criteria and were reviewed. The final cohort of patients was 934 patients. Analysis of symptomatic cases revealed that the majority of cases could be classified into the following: (1) Dyspepsia, which included symptoms of epigastric pain, eructation, nausea or bloating with meals (n = 445, 48%); (2) Pancreatitis within the ectopic lesion (n = 260, 28%); (3) Gastrointestinal bleeding (n = 80, 9%); and (4) Gastric outlet obstruction (n = 80, 9%). 37 patients presented with jaundice and biliary obstruction due to periampullary lesions (4%), and 3 patients presented with perforated ulcers. The majority of patients underwent surgical or endoscopic resection (n = 832, 90%) and 85% of reported cases indicated symptom resolution. However, most patients underwent some form of formal anatomic resection (n = 490, 61%) most commonly related to improper diagnosis preoperatively and few benefited from local surgical or endoscopic resection.
Research conclusions
Heterotopic pancreas is a rare condition but has the ability to manifest severe clinical symptoms. These most commonly arise in the stomach and duodenum and can be classified within for main categories. Improved recognition and diagnosis of this disease process may help guide treatment recommendations and improve outcomes for patients.
Research perspectives
Prospective evaluation of medical and interventional therapies is needed to determine the most appropriate treatment for the different manifestations for heterotopic pancreas.
Footnotes
Institutional review board statement: This study was reviewed and approved by the institutional review board of Washington University of St. Louis. IRB number [#210811062].
Informed consent statement: Based on the study protocol and design, informed consent was determined to not be required and was therefore not obtained for this study. Patient information was collected and reviewed based on the parameters laid out by the IRB at our institution and was deidentified for analysis and reporting. The remainder of the study population was constructed based on a cohort of patients obtained through systematic review of the literature and was therefore based on patient information which had already been published and presented in the literature. For further questions or concerns please feel free to contact the corresponding author.
Conflict-of-interest statement: The authors of this study have no related conflicts of interest to disclose.
Data sharing statement: Technical data and information were provided by the corresponding author, Michael LeCompte who can be reached at michael_lecompte@med.unc.edu. Data from research subjects was anonymized and excluded any patient identifiers. Given the retrospective nature of this study, informed consent was deemed not necessary by the IRB. Additional data was collected and reviewed from datasets collected and published in the literature.
STROBE statement: The authors have read and revised the manuscript based on the STROBE guidelines.
Provenance and peer review: Unsolicited article; Externally peer reviewed.
Peer-review model: Single blind
Corresponding Author's Membership in Professional Societies: American Hepatopancreatobiliary Association; Society of Ameican Gastrointestinal and Endoscopic Surgeons; and American College of Surgeons.
Peer-review started: June 8, 2021
First decision: June 27, 2021
Article in press: March 4, 2022
Specialty type: Gastroenterology and hepatology
Country/Territory of origin: United States
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B, B, B
Grade C (Good): C, C
Grade D (Fair): D
Grade E (Poor): 0
P-Reviewer: Fujino Y, Japan; Mikulic D, Croatia; Wang J, China; Wu YH, , China; Yang CW, China S-Editor: Gong ZM L-Editor: A P-Editor: Gong ZM
Contributor Information
Michael T LeCompte, Department of Surgical Oncology, University of North Carolina, Raleigh, NC 27608, United States. mtleco01@gmail.com.
Brandon Mason, Department of Radiology, Stillwater Medical Center, Stillwater, OK 74074, United States.
Keenan J Robbins, Department of General Surgery, Washington University St. Louis, St. Louis, MO 63110-8109, United States.
Motoyo Yano, Department of Radiology, Mayo Clinic, Phoenix, AZ 8505, United States.
Deyali Chatterjee, Department of Pathology and Immunology, MD Anderson Cancer Center, Houston, TX 77030, United States.
Ryan C Fields, Department of Surgical Oncology, Washington University School of Medicine, St. Louis, MO 63110-8109, United States.
Steven M Strasberg, Section of Hepatobiliary-Pancreatic and GI Surgery, Washington University St. Louis, St. Louis, MO 63110, United States.
William G Hawkins, Section of Hepatobiliary-Pancreatic and GI Surgery, Washington University St. Louis, St. Louis, MO 63110, United States.
References
- 1.De Castro Barboso J, Waugh JM. Heterotopic pancreatic tissue, clinically significant, in the gastric wall of a boy six years of age. Proc Staff Meet Mayo Clin. 1947;22:25–30. [PubMed] [Google Scholar]
- 2.Eisenberger CF, Gocht A, Knoefel WT, Busch CB, Peiper M, Kutup A, Yekebas EF, Hosch SB, Lambrecht W, Izbicki JR. Heterotopic pancreas--clinical presentation and pathology with review of the literature. Hepatogastroenterology. 2004;51:854–858. [PubMed] [Google Scholar]
- 3.Guimarães M, Rodrigues P, Gonçalves G, Nora M, Monteiro MP. Heterotopic pancreas in excluded stomach diagnosed after gastric bypass surgery. BMC Surg. 2013;13:56. doi: 10.1186/1471-2482-13-56. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Ormarsson OT, Gudmundsdottir I, Mårvik R. Diagnosis and treatment of gastric heterotopic pancreas. World J Surg. 2006;30:1682–1689. doi: 10.1007/s00268-005-0669-6. [DOI] [PubMed] [Google Scholar]
- 5.Noh JH, Kim DH, Kim SW, Park YS, Na HK, Ahn JY, Jung KW, Lee JH, Choi KD, Song HJ, Lee GH, Jung HY. Endoscopic submucosal dissection as alternative to surgery for complicated gastric heterotopic pancreas. World J Clin Cases. 2020;8:4708–4718. doi: 10.12998/wjcc.v8.i20.4708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Zhou Y, Zhou S, Shi Y, Zheng S, Liu B. Endoscopic submucosal dissection for gastric ectopic pancreas: a single-center experience. World J Surg Oncol. 2019;17:69. doi: 10.1186/s12957-019-1612-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Betzler A, Mees ST, Pump J, Schölch S, Zimmermann C, Aust DE, Weitz J, Welsch T, Distler M. Clinical impact of duodenal pancreatic heterotopia - Is there a need for surgical treatment? BMC Surg. 2017;17:53. doi: 10.1186/s12893-017-0250-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Jun SY, Son D, Kim MJ, Kim SJ, An S, Park YS, Park SR, Choi KD, Jung HY, Kim SC, Yook JH, Kim BS, Hong SM. Heterotopic Pancreas of the Gastrointestinal Tract and Associated Precursor and Cancerous Lesions: Systematic Pathologic Studies of 165 Cases. Am J Surg Pathol. 2017;41:833–848. doi: 10.1097/PAS.0000000000000850. [DOI] [PubMed] [Google Scholar]
- 9.Zhang Y, Sun X, Gold JS, Sun Q, Lv Y, Li Q, Huang Q. Heterotopic pancreas: a clinicopathological study of 184 cases from a single high-volume medical center in China. Hum Pathol. 2016;55:135–142. doi: 10.1016/j.humpath.2016.05.004. [DOI] [PubMed] [Google Scholar]
- 10.Liao X, Zhan W, Cai LJ, Hu X, Gao Q, Jiao J, Liu J, Yang Q. The giant lower esophageal diverticulum within a heterotopic pancreas. Int J Clin Exp Med . 2016;9:5213–5217. [Google Scholar]
- 11.Chou JW, Cheng KS, Ting CF, Feng CL, Lin YT, Huang WH. Endosonographic features of histologically proven gastric ectopic pancreas. Gastroenterol Res Pract. 2014;2014:160601. doi: 10.1155/2014/160601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Endo S, Saito R, Ochi D, Yamada T, Hirose M, Hiroshima Y, Yamamoto Y, Ueno T, Hasegawa N, Moriwaki T, Narasaka T, Kaneko T, Fukuda K, Suzuki H, Mizokami Y, Hyodo I. Effectiveness of an endoscopic biopsy procedure using EUS-FNA and EMR-C for diagnosing adenocarcinoma arising from ectopic pancreas: two case reports and a literature review. Intern Med. 2014;53:1055–1062. doi: 10.2169/internalmedicine.53.1420. [DOI] [PubMed] [Google Scholar]
- 13.Liu X, Wang G, Ge N, Wang S, Guo J, Liu W, Sun S. Endoscopic removal of symptomatic gastric heterotopic pancreas: a report of nine cases. Surg Innov. 2013;20:NP40–NP46. doi: 10.1177/1553350613499453. [DOI] [PubMed] [Google Scholar]
- 14.Zhong YS, Shi Q, Yao LQ, Zhou PH, Xu MD, Wang P. Endoscopic mucosal resection/endoscopic submucosal dissection for gastric heterotopic pancreas. Turk J Gastroenterol. 2013;24:322–329. [PubMed] [Google Scholar]
- 15.Shah H, Singh M, Heller SJ, Tokar JL, Haluszka O. Heterotopic pancreas: Five-year experience at single academic institution. Gastrointest Endosc . 2011;73:AB227. [Google Scholar]
- 16.Park SH, Kim GH, Park DY, Shin NR, Cheong JH, Moon JY, Lee BE, Song GA, Seo HI, Jeon TY. Endosonographic findings of gastric ectopic pancreas: a single center experience. J Gastroenterol Hepatol. 2011;26:1441–1446. doi: 10.1111/j.1440-1746.2011.06764.x. [DOI] [PubMed] [Google Scholar]
- 17.Wei R, Wang QB, Chen QH, Liu JS, Zhang B. Upper gastrointestinal tract heterotopic pancreas: findings from CT and endoscopic imaging with histopathologic correlation. Clin Imaging. 2011;35:353–359. doi: 10.1016/j.clinimag.2010.10.001. [DOI] [PubMed] [Google Scholar]
- 18.Ryu DY, Kim GH, Park DY, Lee BE, Cheong JH, Kim DU, Woo HY, Heo J, Song GA. Endoscopic removal of gastric ectopic pancreas: an initial experience with endoscopic submucosal dissection. World J Gastroenterol. 2010;16:4589–4593. doi: 10.3748/wjg.v16.i36.4589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Yuan Z, Chen J, Zheng Q, Huang XY, Yang Z, Tang J. Heterotopic pancreas in the gastrointestinal tract. World J Gastroenterol. 2009;15:3701–3703. doi: 10.3748/wjg.15.3701. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Khashab MA, Cummings OW, DeWitt JM. Ligation-assisted endoscopic mucosal resection of gastric heterotopic pancreas. World J Gastroenterol. 2009;15:2805–2808. doi: 10.3748/wjg.15.2805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Casetti L, Bassi C, Salvia R, Butturini G, Graziani R, Falconi M, Frulloni L, Crippa S, Zamboni G, Pederzoli P. "Paraduodenal" pancreatitis: results of surgery on 58 consecutives patients from a single institution. World J Surg. 2009;33:2664–2669. doi: 10.1007/s00268-009-0238-5. [DOI] [PubMed] [Google Scholar]
- 22.Jovanovic I, Alempijevic T, Lukic S, Knezevic S, Popovic D, Dugalic V, Micev M, Krstic M. Cystic dystrophy in heterotopic pancreas of the duodenal wall. Dig Surg. 2008;25:262–268. doi: 10.1159/000148133. [DOI] [PubMed] [Google Scholar]
- 23.Chen HL, Chang WH, Shih SC, Bair MJ, Lin SC. Changing pattern of ectopic pancreas: 22 years of experience in a medical center. J Formos Med Assoc. 2008;107:932–936. doi: 10.1016/S0929-6646(09)60016-4. [DOI] [PubMed] [Google Scholar]
- 24.Chen SH, Huang WH, Feng CL, Chou JW, Hsu CH, Peng CY, Yang MD. Clinical analysis of ectopic pancreas with endoscopic ultrasonography: an experience in a medical center. J Gastrointest Surg. 2008;12:877–881. doi: 10.1007/s11605-008-0476-0. [DOI] [PubMed] [Google Scholar]
- 25.Rebours V, Lévy P, Vullierme MP, Couvelard A, O'Toole D, Aubert A, Palazzo L, Sauvanet A, Hammel P, Maire F, Ponsot P, Ruszniewski P. Clinical and morphological features of duodenal cystic dystrophy in heterotopic pancreas. Am J Gastroenterol. 2007;102:871–879. doi: 10.1111/j.1572-0241.2007.01091.x. [DOI] [PubMed] [Google Scholar]
- 26.Tison C, Regenet N, Meurette G, Mirallié E, Cassagnau E, Frampas E, Le Borgne J. Cystic dystrophy of the duodenal wall developing in heterotopic pancreas: report of 9 cases. Pancreas. 2007;34:152–156. doi: 10.1097/01.mpa.0000246669.61246.08. [DOI] [PubMed] [Google Scholar]
- 27.Pessaux P, Lada P, Etienne S, Tuech JJ, Lermite E, Brehant O, Triau S, Arnaud JP. Duodenopancreatectomy for cystic dystrophy in heterotopic pancreas of the duodenal wall. Gastroenterol Clin Biol. 2006;30:24–28. doi: 10.1016/s0399-8320(06)73073-1. [DOI] [PubMed] [Google Scholar]
- 28.Ayantunde AA, Pinder E, Heath DI. Symptomatic pyloric pancreatic heterotopia: report of three cases and review of the literature. Med Sci Monit. 2006;12:CS49–CS52. [PubMed] [Google Scholar]
- 29.Chatelain D, Vibert E, Yzet T, Geslin G, Bartoli E, Manaouil D, Delcenserie R, Brevet M, Dupas JL, Regimbeau JM. Groove pancreatitis and pancreatic heterotopia in the minor duodenal papilla. Pancreas. 2005;30:e92–e95. doi: 10.1097/01.mpa.0000161885.79373.1d. [DOI] [PubMed] [Google Scholar]
- 30.Zinkiewicz K, Juśkiewicz W, Zgodziński W, Szumiło J, Cwik G, Furtak J, Maciejewski R, Wallner G. Ectopic pancreas: endoscopic, ultrasound and radiological features. Folia Morphol (Warsz) 2003;62:205–209. [PubMed] [Google Scholar]
- 31.Shi HQ, Zhang QY, Teng HL, Chen JC. Heterotopic pancreas: report of 7 patients. Hepatobiliary Pancreat Dis Int. 2002;1:299–301. [PubMed] [Google Scholar]
- 32.Huang YC, Chen HM, Jan YY, Huang TL, Chen MF. Ectopic pancreas with gastric outlet obstruction: report of two cases and literature review. Chang Gung Med J. 2002;25:485–490. [PubMed] [Google Scholar]
- 33.Otani Y, Ohgami M, Igarashi N, Kimata M, Kubota T, Kumai K, Kitajima M, Mukai M. Laparoscopic wedge resection of gastric submucosal tumors. Surg Laparosc Endosc Percutan Tech. 2000;10:19–23. [PubMed] [Google Scholar]
- 34.Hsia CY, Wu CW, Lui WY. Heterotopic pancreas: a difficult diagnosis. J Clin Gastroenterol. 1999;28:144–147. doi: 10.1097/00004836-199903000-00012. [DOI] [PubMed] [Google Scholar]
- 35.Fékété F, Noun R, Sauvanet A, Fléjou JF, Bernades P, Belghiti J. Pseudotumor developing in heterotopic pancreas. World J Surg. 1996;20:295–298. doi: 10.1007/s002689900047. [DOI] [PubMed] [Google Scholar]
- 36.Fléjou JF, Potet F, Molas G, Bernades P, Amouyal P, Fékété F. Cystic dystrophy of the gastric and duodenal wall developing in heterotopic pancreas: an unrecognised entity. Gut. 1993;34:343–347. doi: 10.1136/gut.34.3.343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Claudon M, Verain AL, Bigard MA, Boissel P, Poisson P, Floquet J, Regent D. Cyst formation in gastric heterotopic pancreas: report of two cases. Radiology. 1988;169:659–660. doi: 10.1148/radiology.169.3.3055030. [DOI] [PubMed] [Google Scholar]
- 38.Pang LC. Pancreatic heterotopia: a reappraisal and clinicopathologic analysis of 32 cases. South Med J. 1988;81:1264–1275. [PubMed] [Google Scholar]
- 39.Lai EC, Tompkins RK. Heterotopic pancreas. Review of a 26 year experience. Am J Surg. 1986;151:697–700. doi: 10.1016/0002-9610(86)90045-0. [DOI] [PubMed] [Google Scholar]
- 40.Mollitt DL, Golladay ES. Symptomatic gastroduodenal pancreatic rest in children. J Pediatr Surg. 1984;19:449–450. doi: 10.1016/s0022-3468(84)80273-0. [DOI] [PubMed] [Google Scholar]
- 41.Armstrong CP, King PM, Dixon JM, Macleod IB. The clinical significance of heterotopic pancreas in the gastrointestinal tract. Br J Surg. 1981;68:384–387. doi: 10.1002/bjs.1800680606. [DOI] [PubMed] [Google Scholar]
- 42.Thoeni RF, Gedgaudas RK. Ectopic pancreas: usual and unusual features. Gastrointest Radiol. 1980;5:37–42. doi: 10.1007/BF01888597. [DOI] [PubMed] [Google Scholar]
- 43.Yamagiwa H, Ishihara A, Sekoguchi T, Matsuzaki O. Heterotopic pancreas in surgically resected stomach. Gastroenterol Jpn. 1977;12:380–386. doi: 10.1007/BF02774535. [DOI] [PubMed] [Google Scholar]
- 44.Dolan RV, ReMine WH, Dockerty MB. The fate of heterotopic pancreatic tissue. A study of 212 cases. Arch Surg. 1974;109:762–765. doi: 10.1001/archsurg.1974.01360060032010. [DOI] [PubMed] [Google Scholar]
- 45.Eklöf O, Lassrich A, Stanley P, Chrispin AR. Ectopic pancreas. Pediatr Radiol. 1973;1:24–27. doi: 10.1007/BF00972820. [DOI] [PubMed] [Google Scholar]
- 46.Nebel OT, Farrell RL, Kirchner JP, Macionus RF. Aberrant pancreas--an endoscopic diagnosis. Am J Gastroenterol. 1973;60:295–300. [PubMed] [Google Scholar]
- 47.McGarity WC, Perry CW 3rd. Complications of gastric heterotopic pancreas: two case reports. Am Surg. 1971;37:77–79. [PubMed] [Google Scholar]
- 48.Abrahams JI. Heterotopic pancreas simulating peptic ulceration. Arch Surg. 1966;93:589–592. doi: 10.1001/archsurg.1966.01330040053009. [DOI] [PubMed] [Google Scholar]
- 49.Tonkin RD, Field TE, Wykes PR. Pancreatic heterotopia as a cause of dyspepsia. Gut. 1962;3:135–139. doi: 10.1136/gut.3.2.135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Dirks KR, Field TE. Heterotopic pancreas causing symptoms. J R Army Med Corps. 1961;107:173–175. [PubMed] [Google Scholar]
- 51.Martinez NS, Morlock CG, Dockerty MB, Waugh JM, Weber HM. Heterotopic pancreatic tissue involving the stomach. Ann Surg. 1958;147:1–12. doi: 10.1097/00000658-195801000-00001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Waugh TR, Harding EW. Heterotopic pancreatic tissue in the region of the pyloric orifice; a radiological and pathological analysis of five cases of clinically suspected peptic ulcer in which only pancreatic rests were found. Gastroenterology. 1946;6:417–435. [PubMed] [Google Scholar]
- 53.Barbu ST, Valeanu D, Muresan A, Munteanu D, Casoinic F. Cystic Dystrophy of the Duodenal Wall in Heterotopic Pancreas with Groove Pancreatitis: A Diagnostic and Therapeutic Challenge. Chirurgia (Bucur) 2018;113:418–423. doi: 10.21614/chirurgia.113.3.418. [DOI] [PubMed] [Google Scholar]
- 54.Jin HB, Lu L, Yang JF, Lou QF, Yang J, Shen HZ, Tang XW, Zhang XF. Interventional endoscopic ultrasound for a symptomatic pseudocyst secondary to gastric heterotopic pancreas. World J Gastroenterol. 2017;23:6365–6370. doi: 10.3748/wjg.v23.i34.6365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Desai GS, Phadke A, Kulkarni D. Cystic Dystrophy of the Duodenum Due to Heterotopic Pancreas - A Case Report and Review of Literature. J Clin Diagn Res. 2016;10:PD11–PD13. doi: 10.7860/JCDR/2016/20798.8498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Subasinghe D, Sivaganesh S, Perera N, Samarasekera DN. Gastric fundal heterotopic pancreas mimicking a gastrointestinal stromal tumour (GIST): a case report and a brief review. BMC Res Notes. 2016;9:185. doi: 10.1186/s13104-016-1995-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Baisakh MR, Mohapatra N, Baisakh P, Sahoo S, Kar M. Heterotopic pancreas in the stomach masquerading neoplasm. J Clin Diagn Res. 2014;8:FL01–FL02. doi: 10.7860/JCDR/2014/9142.5243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Rodriguez AA, Berquist W, Bingham D. Gastric outlet obstruction caused by heterotopic pancreas in an adolescent. Dig Dis Sci. 2015;60:835–837. doi: 10.1007/s10620-014-3314-0. [DOI] [PubMed] [Google Scholar]
- 59.Eskandari A, Choi E. Gastric heterotopic pancreas complicated by pancreatitis and pseudocyst formation: A rare complication of an uncommon lesion. Am J Gastroenterol . 2014;109:S454. [Google Scholar]
- 60.Bryan DS, Waxman I, Matthews JB. Gastric obstruction due to intramural pseudocyst associated with heterotopic pancreas. J Gastrointest Surg. 2014;18:1225–1226. doi: 10.1007/s11605-014-2511-7. [DOI] [PubMed] [Google Scholar]
- 61.Şişman G, Erzin Y, Tuncer M, Şentürk H. Endoscopic treatment of heterotopic pancreas localized in the duodenum. Turk J Gastroenterol. 2014;25:216–217. doi: 10.5152/tjg.2014.3415. [DOI] [PubMed] [Google Scholar]
- 62.Aly AM, Saied T. Cystic dystrophy of the duodenal wall in heterotopic pancreas: A case report. Egypt J Radiol Nucl Med . 2014;45:295–298. [Google Scholar]
- 63.Décarie PO, Paye F, Arrivé L. Cystic dystrophy of the gastroduodenal wall in heterotopic pancreas. Clin Res Hepatol Gastroenterol. 2013;37:551–552. doi: 10.1016/j.clinre.2013.03.009. [DOI] [PubMed] [Google Scholar]
- 64.Hasan MK, Hawes R, Varadarajulu S. Gastric heterotopic pancreas abscess leading to gastric outlet obstruction. Gastrointest Endosc. 2013;78:550–551. doi: 10.1016/j.gie.2013.03.1327. [DOI] [PubMed] [Google Scholar]
- 65.Carbonero-Celis MJ, Romero-Moriña H, Northrop-Sharp B, Umbría-Jiménez S, Argüelles-Martín F, Asensio-García J. Upper digestive tract hemorrhage in a child with heterotopic pancreas in a gastric diverticulum. Rev Esp Enferm Dig. 2013;105:54–55. doi: 10.4321/s1130-01082013000100012. [DOI] [PubMed] [Google Scholar]
- 66.Deprez FC, Pauls C, Coulier B. Heterotopic pancreas revealed by post-traumatic pancreatitis. JBR-BTR. 2012;95:83–86. doi: 10.5334/jbr-btr.100. [DOI] [PubMed] [Google Scholar]
- 67.Trifan A, Târcoveanu E, Danciu M, Huţanaşu C, Cojocariu C, Stanciu C. Gastric heterotopic pancreas: an unusual case and review of the literature. J Gastrointestin Liver Dis. 2012;21:209–212. [PubMed] [Google Scholar]
- 68.Kinoshita H, Yamaguchi S, Shimizu A, Sakata Y, Arii K, Mori K, Nasu T. Adenocarcinoma arising from heterotopic pancreas in the duodenum. Int Surg. 2012;97:351–355. doi: 10.9738/CC148.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Lhewa T, Zhang Z, Rozelle C, Terry A. Gastric antral diverticulum with heterotopic pancreas in a teenage patient. J Pediatr Gastroenterol Nutr. 2011;53:471. doi: 10.1097/MPG.0b013e31821b0cd3. [DOI] [PubMed] [Google Scholar]
- 70.Karaca G, Taş A, Pekceci R, Özer H, Köklü S. Unusual Cause of Severe Nausea and Vomiting in an Elderly Man: Heterotopic Pancreas. International Journal of Gerontology 2011; 5: 133-134. [Google Scholar]
- 71.Reggoug S, Errabih I, Ouazzani L, Benzzoubeir N, Krami H, Raiss M, Hrora A, Ahallat M, Ouazzani H. Heterotopic pancreas: an unusual cause of epigastric pain. Gastroenterol Clin Biol. 2010;34:726–727. doi: 10.1016/j.gcb.2010.09.005. [DOI] [PubMed] [Google Scholar]
- 72.Patel N, Berzin T. Intraductal papillary mucinous neoplasm arising in a heterotopic pancreas: a case report. Am J Gastroenterol. 2010;105:2513–2514. doi: 10.1038/ajg.2010.298. [DOI] [PubMed] [Google Scholar]
- 73.Gokhale UA, Nanda A, Pillai R, Al-Layla D. Heterotopic pancreas in the stomach: a case report and a brief review of the literature. JOP. 2010;11:255–257. [PubMed] [Google Scholar]
- 74.Payeras G, Castellón C, De Jaime J, Barranco R, Calvache A, Castro P. Heterotopic pancreas: a difficult diagnosis. Endoscopy. 2010;42 Suppl 2:E121. doi: 10.1055/s-0029-1243988. [DOI] [PubMed] [Google Scholar]
- 75.Inoue Y, Hayashi M, Arisaka Y, Higuchi K, Egashira Y, Tanigawa N. Adenocarcinoma arising in a heterotopic pancreas (Heinrich type III): a case report. J Med Case Rep. 2010;4:39. doi: 10.1186/1752-1947-4-39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Yoshida N, Nakayama H, Hemmi A, Suzuki T, Takayama T. Duodenal stenosis caused by cystic dystrophy in heterotopic pancreas: report of a case. Surg Today. 2009;39:803–806. doi: 10.1007/s00595-009-3939-3. [DOI] [PubMed] [Google Scholar]
- 77.Gurocak B, Gokturk HS, Kayacetin S, Bakdik S. A rare case of heterotopic pancreas in the stomach which caused closed perforation. Neth J Med. 2009;67:285–287. [PubMed] [Google Scholar]
- 78.Galloro G, Napolitano V, Magno L, Diamantis G, Nardone G, Bruno M, Mollica C, Persico G. Diagnosis and therapeutic management of cystic dystrophy of the duodenal wall in heterotopic pancreas. A case report and revision of the literature. JOP. 2008;9:725–732. [PubMed] [Google Scholar]
- 79.Rimal D, Thapa SR, Munasinghe N, Chitre VV. Symptomatic gastric heterotopic pancreas: clinical presentation and review of the literature. Int J Surg. 2008;6:e52–e54. doi: 10.1016/j.ijsu.2007.02.002. [DOI] [PubMed] [Google Scholar]
- 80.Jiang LX, Xu J, Wang XW, Zhou FR, Gao W, Yu GH, Lv ZC, Zheng HT. Gastric outlet obstruction caused by heterotopic pancreas: A case report and a quick review. World J Gastroenterol. 2008;14:6757–6759. doi: 10.3748/wjg.14.6757. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Margolin DJ. Endoscopy-assisted laparoscopic resection of gastric heterotopic pancreas. Am Surg. 2008;74:829–831. [PubMed] [Google Scholar]
- 82.Lopez-Pelaez MS, Hoyos FB, Isidro MG, Unzurrunzaga EA, Lopez Ede V, Collazo YQ. Cystic dystrophy of heterotopic pancreas in stomach: radiologic and pathologic correlation. Abdom Imaging. 2008;33:391–394. doi: 10.1007/s00261-007-9275-4. [DOI] [PubMed] [Google Scholar]
- 83.Stefanescu C, Vullierme MP, Couvelard A, Bretagnol F, Amouyal P, Maire F, Rebours V, Hammel P, Ruszniewski P, Lévy P. Cystic dystrophy in gastric heterotopic pancreas complicated by intracystic hemorrhage and fistulisation in the stomach - a pediatric case. Gastroenterol Clin Biol. 2008;32:645–648. doi: 10.1016/j.gcb.2008.02.032. [DOI] [PubMed] [Google Scholar]
- 84.Christodoulidis G, Zacharoulis D, Barbanis S, Katsogridakis E, Hatzitheofilou K. Heterotopic pancreas in the stomach: a case report and literature review. World J Gastroenterol. 2007;13:6098–6100. doi: 10.3748/wjg.v13.45.6098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Wilde GE, Gakhal M, Sartip KA, Corso MJ, Butt WG. Pancreatitis in initially occult gastric heterotopic pancreas. Clin Imaging. 2007;31:356–359. doi: 10.1016/j.clinimag.2007.05.007. [DOI] [PubMed] [Google Scholar]
- 86.Wall I, Shah T, Tangorra M, Li JJ, Tenner S. Giant heterotopic pancreas presenting with massive upper gastrointestinal bleeding. Dig Dis Sci. 2007;52:956–959. doi: 10.1007/s10620-006-9481-x. [DOI] [PubMed] [Google Scholar]
- 87.Biswas A, Husain EA, Feakins RM, Abraham AT. Heterotopic pancreas mimicking cholangiocarcinoma. Case report and literature review. JOP. 2007;8:28–34. [PubMed] [Google Scholar]
- 88.Kobayashi S, Okayama Y, Hayashi K, Sano H, Shiraki S, Goto K, Ohara H, Joh T. Heterotopic pancreas in the stomach which caused obstructive stenosis in the duodenum. Intern Med. 2006;45:1137–1141. doi: 10.2169/internalmedicine.45.1814. [DOI] [PubMed] [Google Scholar]
- 89.Emerson L, Layfield LJ, Rohr LR, Dayton MT. Adenocarcinoma arising in association with gastric heterotopic pancreas: A case report and review of the literature. J Surg Oncol. 2004;87:53–57. doi: 10.1002/jso.20087. [DOI] [PubMed] [Google Scholar]
- 90.Tolentino LF, Lee H, Maung T, Stabile BE, Li K, French SW. Islet cell tumor arising from a heterotopic pancreas in the duodenal wall with ulceration. Exp Mol Pathol. 2004;76:51–56. doi: 10.1016/j.yexmp.2003.08.005. [DOI] [PubMed] [Google Scholar]
- 91.Ormarsson OT, Haugen SE, Juul I. Gastric outlet obstruction caused by heterotopic pancreas. Eur J Pediatr Surg. 2003;13:410–413. doi: 10.1055/s-2003-44733. [DOI] [PubMed] [Google Scholar]
- 92.Ikematsu Y, Nishiwaki Y, Kida H, Iwaoka Y, Nagashima S, Ozawa T, Hasegawa S, Okawada T, Waki S. Gastric outlet obstruction caused by a heterotopic pancreas in a pregnant woman: report of a case. Surg Today. 2003;33:952–955. doi: 10.1007/s00595-003-2614-3. [DOI] [PubMed] [Google Scholar]
- 93.Griffin AC, Brost BC, Staren ED. Symptomatic heterotopic pancreas following seat belt injury. Injury. 2003;34:944–945. doi: 10.1016/s0020-1383(02)00044-x. [DOI] [PubMed] [Google Scholar]
- 94.Haj M, Shiller M, Loberant N, Cohen I, Kerner H. Obstructing gastric heterotopic pancreas: case report and literature review. Clin Imaging. 2002;26:267–269. doi: 10.1016/s0899-7071(01)00408-9. [DOI] [PubMed] [Google Scholar]
- 95.Hazzan D, Peer G, Shiloni E. Symptomatic heterotopic pancreas of stomach. Isr Med Assoc J. 2002;4:388–389. [PubMed] [Google Scholar]
- 96.Halkic N, Nordback P. Soft-tissue images. Malignant degeneration of heterotopic pancreas. Can J Surg. 2001;44:407. [PMC free article] [PubMed] [Google Scholar]
- 97.Shaib YH, Rabaa E, Feddersen RM, Jamal MM, Qaseem T. Gastric outlet obstruction secondary to heterotopic pancreas in the antrum: case report and review. Gastrointest Endosc. 2001;54:527–530. doi: 10.1067/mge.2001.116461. [DOI] [PubMed] [Google Scholar]
- 98.Chetty R, Weinreb I. Gastric neuroendocrine carcinoma arising from heterotopic pancreatic tissue. J Clin Pathol. 2004;57:314–317. doi: 10.1136/jcp.2003.013557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Bethel CA, Luquette MH, Besner GE. Cystic degeneration of heterotopic pancreas. Pediatr Surg Int. 1998;13:428–430. doi: 10.1007/s003830050358. [DOI] [PubMed] [Google Scholar]
- 100.Lucena JF, Alvarez OA, Gross GW. Endoscopic resection of heterotopic pancreas of the minor duodenal papilla: case report and review of the literature. Gastrointest Endosc. 1997;46:69–72. doi: 10.1016/s0016-5107(97)70214-1. [DOI] [PubMed] [Google Scholar]
- 101.Mönig SP, Selzner M, Raab M, Eidt S. Heterotopic pancreas. A difficult diagnosis. Dig Dis Sci. 1996;41:1238–1240. doi: 10.1007/BF02088243. [DOI] [PubMed] [Google Scholar]
- 102.Allison JW, Johnson JF 3rd, Barr LL, Warner BW, Stevenson RJ. Induction of gastroduodenal prolapse by antral heterotopic pancreas. Pediatr Radiol. 1995;25:50–51. doi: 10.1007/BF02020846. [DOI] [PubMed] [Google Scholar]
- 103.Ueno S, Ishida H, Hayashi A, Kamagata S, Morikawa M. Heterotopic pancreas as a rare cause of gastrointestinal hemorrhage in the newborn: report of a case. Surg Today. 1993;23:269–272. doi: 10.1007/BF00309240. [DOI] [PubMed] [Google Scholar]
- 104.Jeng KS, Yang KC, Kuo SH. Malignant degeneration of heterotopic pancreas. Gastrointest Endosc. 1991;37:196–198. doi: 10.1016/s0016-5107(91)70687-1. [DOI] [PubMed] [Google Scholar]
- 105.Farrar WB, Scott M, O'Dwyer PJ. Heterotopic pancreas. Ir J Med Sci. 1990;159:19–20. doi: 10.1007/BF02937209. [DOI] [PubMed] [Google Scholar]
- 106.Burke GW, Binder SC, Barron AM, Dratch PL, Umlas J. Heterotopic pancreas: gastric outlet obstruction secondary to pancreatitis and pancreatic pseudocyst. Am J Gastroenterol. 1989;84:52–55. [PubMed] [Google Scholar]
- 107.Laughlin EH, Keown ME, Jackson JE. Heterotopic pancreas obstructing the ampulla of Vater. Arch Surg. 1983;118:979–980. doi: 10.1001/archsurg.1983.01390080081020. [DOI] [PubMed] [Google Scholar]
- 108.O'Reilly DJ, Craig RM, Lorenzo G, Yokoo H. Heterotopic pancreas mimicking carcinoma of the head of the pancreas: a rare cause of obstructive jaundice. J Clin Gastroenterol. 1983;5:165–168. doi: 10.1097/00004836-198304000-00014. [DOI] [PubMed] [Google Scholar]
- 109.Goldfarb WB, Bennett D, Monafo W. Carcinoma in heterotopic gastric pancreas. Ann Surg. 1963;158:56–58. doi: 10.1097/00000658-196307000-00011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Laird EG. Heterotopic pancreas of the duodenum diagnosed pre-operatively. Del Med J. 1963;35:149–151. [PubMed] [Google Scholar]
- 111.Alastal Y, Khalil B, Singh S, Almadani SB. Ectopic Pancreas in the Gastric Antrum Wall Complicated by Ectopic Pancreatitis and Persistent Gastric Abscess. ACG Case Rep J. 2018;5:e34. doi: 10.14309/crj.2018.34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Štor Z, Hanžel J. Gastric ectopic pancreas mimicking a gastrointestinal stromal tumour: A case report. Int J Surg Case Rep. 2018;53:348–350. doi: 10.1016/j.ijscr.2018.11.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Ning X, Liang B, Liu Q, Chen X, Xu M. Gastric inflammatory myofibroblastic tumor identified as ectopic pancreas and treated by endoscopic submucosal dissection. Gastrointest Endosc. 2017;86:921–922. doi: 10.1016/j.gie.2017.05.037. [DOI] [PubMed] [Google Scholar]
- 114.Parra V, Acero F, Alvarez E, Aponte DM, Sabbagh LC. A case of mucinous cystic neoplasm from a gastric ectopic pancreas. Gastrointest Endosc. 2017;85:1096–1097. doi: 10.1016/j.gie.2016.06.010. [DOI] [PubMed] [Google Scholar]
- 115.Passos ID, Chatzoulis G, Milias K, Tzoi E, Christoforakis C, Spyridopoulos P. Gastric duplication cyst (gdc) associated with ectopic pancreas: Case report and review of the literature. Int J Surg Case Rep. 2017;31:109–113. doi: 10.1016/j.ijscr.2017.01.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Fujii S, Tanaka H, Ono K, Onizawa Y, Miyazono Y, Sakamoto N, Gotoh C, Urita Y, Shinkai T, Takayasu H, Sumazaki R, Masumoto K. Gastric outlet obstruction caused by an ectopic pancreas in a neonate: A case report. Journal of Pediatric Surgery Case Reports 2015; 3(12): 573-576. [Google Scholar]
- 117.Gong EJ, Kim DH, Cho CJ, Lee SS, Jung HY, Yoo SH. Endoscopic submucosal dissection of ectopic pancreas with pancreatitis and pseudocyst formation. Gastrointest Endosc. 2015;82:1126. doi: 10.1016/j.gie.2015.06.029. [DOI] [PubMed] [Google Scholar]
- 118.Lee SL, Ku YM, Lee HH, Cho YS. Gastric ectopic pancreas complicated by formation of a pseudocyst. Clin Res Hepatol Gastroenterol. 2014;38:389–391. doi: 10.1016/j.clinre.2014.02.011. [DOI] [PubMed] [Google Scholar]
- 119.Kalaitzakis E, Elebro J, Johansson J. Gastric ectopic pancreas: not always an innocent bystander. JOP. 2013;14:684–685. doi: 10.6092/1590-8577/1991. [DOI] [PubMed] [Google Scholar]
- 120.Kishiki T, Abe N, Sugiyama M, Tokutsu T, Matsuoka H, Yanagida O, Masaki T, Mori T. Inflammatory mass formation caused by gastric ectopic pancreas: report of a case. Surg Today. 2013;43:1448–1451. doi: 10.1007/s00595-012-0317-3. [DOI] [PubMed] [Google Scholar]
- 121.O'Malley RB, Maturen KE, Al-Hawary MM, Mathur AK. Case of the season: ectopic pancreas. Semin Roentgenol. 2013;48:188–191. doi: 10.1053/j.ro.2013.03.011. [DOI] [PubMed] [Google Scholar]
- 122.Fukino N, Oida T, Mimatsu K, Kida K, Kawasaki A, Kuboi Y, Kano H. Diffuse Peritonitis due to Perforated Gastric Ectopic Pancreas. Case Rep Gastroenterol. 2012;6:689–694. doi: 10.1159/000345382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Lee YT, Lin H, Guo JC, Yan SL, Hou HJ, Lai YS, Liu YH, Wu HS, Hwang MH. Laparoscopy-assisted billroth I gastrectomy for ectopic pancreas in the prepyloric region. Case Rep Gastroenterol. 2012;6:712–719. doi: 10.1159/000345388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Uğur Kantar F, Akarsu M, Atılla K, Sağol O, Soytürk M, Akpinar H. Ectopic pancreas presenting with intractable diarrhea: case report. Turk J Gastroenterol. 2011;22:426–429. [PubMed] [Google Scholar]
- 125.Goto S, Okazaki T, Koga H, Miyano G, Arakawa A, Yao T, Lane GJ, Yamataka A. Ectopic pancreas presenting as a submucosal gastric tumor: case report and literature review. Pediatr Surg Int. 2011;27:107–109. doi: 10.1007/s00383-010-2717-z. [DOI] [PubMed] [Google Scholar]
- 126.Sharma DK, Agarwal S, Saran RK, Agarwal AK. Pseudocyst of ectopic pancreas of the duodenal wall masquerading as malignant cystic tumor of pancreas. Saudi J Gastroenterol. 2009;15:271–273. doi: 10.4103/1319-3767.56101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Surov A, Hainz M, Hinz L, Holzhausen HJ, Finke R, Spielmann RP, Kunze C. Case report: ectopic pancreas with pseudocyst and pseudoaneurysm formation. Clin Radiol. 2009;64:734–737. doi: 10.1016/j.crad.2009.01.013. [DOI] [PubMed] [Google Scholar]
- 128.Kim KJ. Endoscopic resection of ectopic pancreas in the minor papilla. Endoscopy. 2009;41 Suppl 2:E12. doi: 10.1055/s-0028-1103467. [DOI] [PubMed] [Google Scholar]
- 129.Hsu SD, Wu HS, Kuo CL, Lee YT. Robotic-assisted laparoscopic resection of ectopic pancreas in the posterior wall of gastric high body: case report and review of the literature. World J Gastroenterol. 2005;11:7694–7696. doi: 10.3748/wjg.v11.i48.7694. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Mizuno Y, Sumi Y, Nachi S, Ito Y, Marui T, Saji S, Matsutomo H. Acinar cell carcinoma arising from an ectopic pancreas. Surg Today. 2007;37:704–707. doi: 10.1007/s00595-006-3384-5. [DOI] [PubMed] [Google Scholar]
- 131.Ogata M, Chihara N, Matsunobu T, Koizumi M, Yoshino M, Shioya T, Watanabe M, Tokunaga A, Tajiri T, Matsumoto K. Case of intra-abdominal endocrine tumor possibly arising from an ectopic pancreas. J Nippon Med Sch. 2007;74:168–172. doi: 10.1272/jnms.74.168. [DOI] [PubMed] [Google Scholar]
- 132.Phillips J, Katz A, Zopolsky P. Intraductal papillary mucinous neoplasm in an ectopic pancreas located in the gastric wall. Gastrointest Endosc. 2006;64:814–5; discussion 815. doi: 10.1016/j.gie.2006.05.017. [DOI] [PubMed] [Google Scholar]
- 133.Chou SJ, Chou YW, Jan HC, Chen VT, Chen TH. Ectopic pancreas in the ampulla of vater with obstructive jaundice. A case report and review of literature. Dig Surg. 2006;23:262–264. doi: 10.1159/000096158. [DOI] [PubMed] [Google Scholar]
- 134.Ertem D, Tutar E, Cam S, Ugras M, Pehlivanoglu E. Severe gastric outlet obstruction in a child with ectopic pancreas: is there a role for Helicobacter pylori? J Pediatr Gastroenterol Nutr. 2006;43:385–387. doi: 10.1097/01.mpg.0000221895.51748.44. [DOI] [PubMed] [Google Scholar]
- 135.Hsu SD, Chan DC, Hsieh HF, Chen TW, Yu JC, Chou SJ. Ectopic pancreas presenting as ampulla of Vater tumor. Am J Surg. 2008;195:498–500. doi: 10.1016/j.amjsurg.2007.01.043. [DOI] [PubMed] [Google Scholar]
- 136.Tanemura A, Yano T, Tamaki H, Sanda T, Ohashi N, Ishihara A, Fukutome K, Shiraki K. Ectopic pancreas in the minor duodenal papilla presenting as upper-GI bleeding. Gastrointest Endosc. 2005;62:324–326. doi: 10.1016/s0016-5107(05)01571-3. [DOI] [PubMed] [Google Scholar]
- 137.De Vogelaere K, Buydens P, Reynaert H, Vanhoey M, Delvaux G. Laparoscopic wedge resection for gastric ectopic pancreas. Surg Laparosc Endosc Percutan Tech. 2005;15:166–168. doi: 10.1097/01.sle.0000166991.05099.b4. [DOI] [PubMed] [Google Scholar]
- 138.Mulholland KC, Wallace WD, Epanomeritakis E, Hall SR. Pseudocyst formation in gastric ectopic pancreas. JOP. 2004;5:498–501. [PubMed] [Google Scholar]
- 139.Sharma SB, Gupta V. Ectopic pancreas as a cause of gastric outlet obstruction in an infant. Indian J Gastroenterol. 2004;23:219. [PubMed] [Google Scholar]
- 140.Fragoso AC, Correia-Pinto J, Carvalho JL, Dias JA, Troncoso MP, Estevão-Costa J. Ectopic pancreas and foveolar hyperplasia in a newborn: a unifying etiopathogenesis for gastric outlet obstruction. J Pediatr Gastroenterol Nutr. 2004;39:92–94. doi: 10.1097/00005176-200407000-00018. [DOI] [PubMed] [Google Scholar]
- 141.Verma R, Abraham DT, Joseph P, Nayak S, Agarwal S. Ectopic pancreas mimicking superior mesenteric artery syndrome. Indian J Gastroenterol. 2003;22:105–106. [PubMed] [Google Scholar]
- 142.Jeong HY, Yang HW, Seo SW, Seong JK, Na BK, Lee BS, Song GS, Park HS, Lee HY. Adenocarcinoma arising from an ectopic pancreas in the stomach. Endoscopy. 2002;34:1014–1017. doi: 10.1055/s-2002-35836. [DOI] [PubMed] [Google Scholar]
- 143.Paolucci P, Brasesco OE, Rosin D, Saber AA, Avital S, Berho M, Rosenthal RJ. Laparoscopic resection of ectopic pancreas in the gastric antrum: case report and literature review. J Laparoendosc Adv Surg Tech A. 2002;12:139–141. doi: 10.1089/10926420252939691. [DOI] [PubMed] [Google Scholar]
- 144.Ozcan C, Celik A, Güçlü C, Balik E. A rare cause of gastric outlet obstruction in the newborn: Pyloric ectopic pancreas. J Pediatr Surg. 2002;37:119–120. doi: 10.1053/jpsu.2002.29443. [DOI] [PubMed] [Google Scholar]
- 145.Chen CH, Yang CC, Yeh YH, Chou DA, Kuo CL. Ectopic pancreas located in the major duodenal papilla: case report and review. Gastrointest Endosc. 2001;53:121–123. doi: 10.1067/mge.2001.111396. [DOI] [PubMed] [Google Scholar]
- 146.Hayes-Jordan A, Idowu O, Cohen R. Ectopic pancreas as the cause of gastric outlet obstruction in a newborn. Pediatr Radiol. 1998;28:868–870. doi: 10.1007/s002470050485. [DOI] [PubMed] [Google Scholar]
- 147.Kawashima H, Iwanaka T, Matsumoto M, Takayasu H, Aihara T, Kishimoto H, Imaizumi S. Pyloric stenosis caused by noncystic duodenal duplication and ectopic pancreas in a neonate. J Pediatr Gastroenterol Nutr. 1998;27:228–229. doi: 10.1097/00005176-199808000-00020. [DOI] [PubMed] [Google Scholar]
- 148.Blais C, Massé S. Preoperative ultrasound diagnosis of a gastric duplication cyst with ectopic pancreas in a child. J Pediatr Surg. 1995;30:1384–1386. doi: 10.1016/0022-3468(95)90516-2. [DOI] [PubMed] [Google Scholar]
- 149.Laurent T, Fournier D, Doenz F, Karaaslan T, Wassmer FA. Complex lesion of the gastric wall: an unusual presentation of ectopic pancreas. J Clin Ultrasound. 1995;23:438–441. doi: 10.1002/jcu.1870230709. [DOI] [PubMed] [Google Scholar]
- 150.Dawson KP. Ectopic pancreas and cystic fibrosis. Aust N Z J Med. 1988;18:788–789. doi: 10.1111/j.1445-5994.1988.tb00181.x. [DOI] [PubMed] [Google Scholar]
- 151.Chung JP, Lee SI, Kim KW, Chi HS, Jeong HJ, Moon YM, Kang JK, Park IS. Duodenal ectopic pancreas complicated by chronic pancreatitis and pseudocyst formation--a case report. J Korean Med Sci. 1994;9:351–356. doi: 10.3346/jkms.1994.9.4.351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Lucaya J, Ochoa JB. Ectopic pancreas in the stomach. J Pediatr Surg. 1976;11:101–102. doi: 10.1016/0022-3468(76)90181-0. [DOI] [PubMed] [Google Scholar]
- 153.Clark RE, Teplick SK. Ectopic pancreas causing massive upper gastrointestinal hemorrhage. Report of a case diagnosed arteriographically. Gastroenterology. 1975;69:1331–1333. [PubMed] [Google Scholar]
- 154.Weiss GN, Marek FH. Surgical intervention for ectopic pancreas in the pylorus. J La State Med Soc. 1953;105:389–393. [PubMed] [Google Scholar]
- 155.Munroe CA, Ross E, Bolinger B. Don't Mess With the Pancreas (Wherever It May Be): Acute Pancreatic Rest"itis" Presenting as a Submucosal Mass With Gastric Outlet Obstruction. Gastroenterology. 2018;155:e1–e2. doi: 10.1053/j.gastro.2017.12.034. [DOI] [PubMed] [Google Scholar]
- 156.Polavarapu AD, Philipose J, Daoud M, Zheng X, Opitz L, Andrawes S. Low grade pre-malignant lesion of pancreatic rest in stomach, surveillance a dilemma. Am J Gastroenterol . 2017;112:S1425–S1426. [Google Scholar]
- 157.Masoodi I, Al-Lehibi A, Almohaimeed K, Hussain S. Pancreatic Rest - An Unusual Cause of Dyspepsia: A Case Report with Literature Review. Saudi J Med Med Sci. 2016;4:225–228. doi: 10.4103/1658-631X.188261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Peng JS, Ritter K, Hamadeh F, Seifarth FG. Asymmetric Hypertrophic Pyloric Stenosis with Concurrent Pancreatic Rest Presenting as Gastric Outlet Obstruction. J Pediatr. 2016;174:273–273.e1. doi: 10.1016/j.jpeds.2016.03.079. [DOI] [PubMed] [Google Scholar]
- 159.Pancreatic/Biliary. Am J Gastroenterol. 2014;109 Suppl 2:S59–S101. doi: 10.1038/ajg.2014.275. [DOI] [PubMed] [Google Scholar]
- 160.Faigel DO, Gopal D, Weeks DA, Corless C. Cap-assisted endoscopic submucosal resection of a pancreatic rest. Gastrointest Endosc. 2001;54:782–784. doi: 10.1067/mge.2001.116620. [DOI] [PubMed] [Google Scholar]
- 161.Wan Abdul Rahman WF, Che Jalil NA, Samsudin H, Hi Merican SR, Lam AK. Duodenal Mass from Heterotopic Pancreas: A Unique Cause of Septic Shock. Ann Acad Med Singap. 2016;45:421–423. [PubMed] [Google Scholar]
- 162.Matsumoto T, Tanaka N, Nagai M, Koike D, Sakuraoka Y, Kubota K. A case of gastric heterotopic pancreatitis resected by laparoscopic surgery. Int Surg. 2015;100:678–682. doi: 10.9738/INTSURG-D-14-00182.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Sarsenov D, Tırnaksız MB, Doğrul AB, Tanas Ö, Gedikoglu G, Abbasoğlu O. Heterotopic pancreatic pseudocyst radiologically mimicking gastrointestinal stromal tumor. Int Surg. 2015;100:486–489. doi: 10.9738/INTSURG-D-13-00242.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Filip R, Walczak E, Huk J, Radzki RP, Bieńko M. Heterotopic pancreatic tissue in the gastric cardia: a case report and literature review. World J Gastroenterol. 2014;20:16779–16781. doi: 10.3748/wjg.v20.i44.16779. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Kalkan İH, Öztaş E, Beyazit Y, Suvak B, Yildiz H, Ibiş M. An unusual case of double heterotopic pancreatic tissues in the same localization. JOP. 2012;13:243–244. [PubMed] [Google Scholar]
- 166.Karpińska MS, Nienartowicz M, Markowska-Woyciechowska A, Budrewicz-Czapska K. Heterotopic pancreas in the stomach (type II according to Heinrich) - literature review and case report. Pol Przegl Chir. 2011;83:171–174. doi: 10.2478/v10035-011-0026-4. [DOI] [PubMed] [Google Scholar]
- 167.Almashat S, Sepehr A. Obstructive and inflammatory gastric heterotopic pancreatic tissue. Arch Iran Med. 2011;14:357–358. [PubMed] [Google Scholar]
- 168.Beleta E, Peghin M, Guarner L. Cystic dystrophy of the pancreas in patient with only one previous cyst. Rev Esp Enferm Dig. 2011;103:95–97. doi: 10.4321/s1130-01082011000200010. [DOI] [PubMed] [Google Scholar]
- 169.Sinha A, Saluja SS, Gamanagatti S. Gastric duplication cyst with macroscopic serosal heterotopic pancreas. JOP. 2010;11:470–473. [PubMed] [Google Scholar]
- 170.Agale SV, Agale VG, Zode RR, Grover S, Joshi S. Heterotopic pancreas involving stomach and duodenum. J Assoc Physicians India. 2009;57:653–654. [PubMed] [Google Scholar]
- 171.Zheng LD, Tong QS, Tang ST, Du ZY, Ruan QL. Duodenal heterotopic pancreas in a child. World J Pediatr. 2009;5:146–148. doi: 10.1007/s12519-009-0029-y. [DOI] [PubMed] [Google Scholar]
- 172.Jarry J Jr, Rault A, Sa Cuhna A, Collet D, Masson B. Acute recurrent pancreatitis by heterotopic fundic mucosa at the ampulla of vater. Pancreas. 2009;38:351–353. doi: 10.1097/MPA.0b013e3181888feb. [DOI] [PubMed] [Google Scholar]
- 173.Sukumar N, Teoh CM. Heterotopic pancreas in the stomach. Med J Malaysia. 2004;59:541–543. [PubMed] [Google Scholar]
- 174.Rubbia-Brandt L, Huber O, Hadengue A, Frossard JL. An unusual case of gastric heterotopic pancreas. JOP. 2004;5:484–487. [PubMed] [Google Scholar]
- 175.Jovanovic I, Knezevic S, Micev M, Krstic M. EUS mini probes in diagnosis of cystic dystrophy of duodenal wall in heterotopic pancreas: a case report. World J Gastroenterol. 2004;10:2609–2612. doi: 10.3748/wjg.v10.i17.2609. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Obermaier R, Walch A, Kurtz C, von Dobschuetz E, Adam U, Neeff H, Benz S, Hopt UT. Heterotopic pancreatitis with obstruction of the major duodenal papill--a rare trigger of obstructive orthotopic pancreatitis. Pancreatology. 2004;4:244–248. doi: 10.1159/000078435. [DOI] [PubMed] [Google Scholar]
- 177.Hammock L, Jorda M. Gastric endocrine pancreatic heterotopia. Arch Pathol Lab Med. 2002;126:464–467. doi: 10.5858/2002-126-0464-GEPH. [DOI] [PubMed] [Google Scholar]
- 178.Chetty R, Salahshor S, Bapat B, Berk T, Croitoru M, Gallinger S. Intraductal papillary mucinous neoplasm of the pancreas in a patient with attenuated familial adenomatous polyposis. J Clin Pathol. 2005;58:97–101. doi: 10.1136/jcp.2004.020925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Indinnimeo M, Cicchini C, Stazi A, Ghini C, Laghi A, Memeo L, Iannaccone R, Teneriello FL, Mingazzini PL. Duodenal pancreatic heterotopy diagnosed by magnetic resonance cholangiopancreatography: report of a case. Surg Today. 2001;31:928–931. doi: 10.1007/s005950170038. [DOI] [PubMed] [Google Scholar]
- 180.Osanai M, Miyokawa N, Tamaki T, Yonekawa M, Kawamura A, Sawada N. Adenocarcinoma arising in gastric heterotopic pancreas: clinicopathological and immunohistochemical study with genetic analysis of a case. Pathol Int. 2001;51:549–554. doi: 10.1046/j.1440-1827.2001.01240.x. [DOI] [PubMed] [Google Scholar]
- 181.Hackett TR, Memon MA, Fitzgibbons RJ Jr, Mixter CG. Laparoscopic resection of heterotopic gastric pancreatic tissue. J Laparoendosc Adv Surg Tech A. 1997;7:307–312. doi: 10.1089/lap.1997.7.307. [DOI] [PubMed] [Google Scholar]
- 182.Steyaert H, Voigt JJ, Brouet P, Vaysse P. Uncommon complication of gastric duplication in a three-year-old child. Eur J Pediatr Surg. 1997;7:243–244. doi: 10.1055/s-2008-1071104. [DOI] [PubMed] [Google Scholar]
- 183.Nopajaroonsri C. Mucus retention in heterotopic pancreas of the gastric antrum. A lesion mimicking mucinous carcinoma. Am J Surg Pathol. 1994;18:953–957. doi: 10.1097/00000478-199409000-00011. [DOI] [PubMed] [Google Scholar]
- 184.Kesieme EB, Dongo AE, Osime CO, Olomu SC, Awe OO, Eze GI, Eluehike SU. Gastric duplication: a rare cause of massive lower gastrointestinal haemorrhage, chest wall mass, and enterocutaneous fistula. Case Rep Gastrointest Med. 2012;2012:250890. doi: 10.1155/2012/250890. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 185.Teixeira CR, Haruma K, Shimamoto T, Tsuda T, Okamoto S, Sumii K, Kajiyama G. Heterotopic pancreas diagnosed by endoscopic ultrasonography and endoscopic injection of ethanol to make a histologic diagnosis. J Clin Gastroenterol. 1992;15:52–54. doi: 10.1097/00004836-199207000-00012. [DOI] [PubMed] [Google Scholar]
- 186.Visentin M, Vaysse P, Guitard J, Fedacou F, Juskiewenski S. Pyloric obstruction with complicated pancreatic heterotopia. Eur J Pediatr Surg. 1991;1:247–248. doi: 10.1055/s-2008-1042500. [DOI] [PubMed] [Google Scholar]
- 187.Lim PH, Chng HC, Ho JM. Transendoscopic removal of heterotopic pancreatic tissue in the stomach--a case report. Singapore Med J. 1987;28:366–368. [PubMed] [Google Scholar]
- 188.Rohrmann CA Jr, Delaney JH Jr, Protell RL. Heterotopic pancreas diagnosed by cannulation and duct study. AJR Am J Roentgenol. 1977;128:1044–1045. doi: 10.2214/ajr.128.6.1044. [DOI] [PubMed] [Google Scholar]
- 189.Barrocas A, Fontenelle LJ, Williams MJ. Gastric heterotopic pancreas: a case report and review of literature. Am Surg. 1973;39:361–365. [PubMed] [Google Scholar]
- 190.McCorriston JR. Heterotopic pancreatic tissue in wall of duodenum associated with bleeding duodenal ulcer. Can J Surg. 1958;1:153–156. [PubMed] [Google Scholar]
- 191.Hudock JJ, Wanner H, Reilly CJ. Acute massive gastro-intestinal hemorrhage associated with pancreatic heterotopic tissue of the stomach. Ann Surg. 1956;143:121–125. doi: 10.1097/00000658-195601000-00017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Foote EL. Heterotopic pancreas in stomach; case report. J Maine Med Assoc. 1955;46:277–279. [PubMed] [Google Scholar]
- 193.Wagner M. Clinical significance of heterotopic pancreas in wall of stomach; report of a case and review of the literature. AMA Arch Surg. 1952;64:840–846. doi: 10.1001/archsurg.1952.01260010860016. [DOI] [PubMed] [Google Scholar]
- 194.Stich MH. Heterotopic pancreatic tissue in the duodenum. N Y State J Med. 1952;52:1057. [PubMed] [Google Scholar]
- 195.Busard JM, Walters W. HETEROTOPIC PANCREATIC TISSUE: Report of a Case Presenting Symptoms of Ulcer and Review of the Recent Literature. Arch Surg . 1950;60:674–682. [Google Scholar]
- 196.Kim SB, Oh MJ, Lee SH. Gastric subepithelial lesion complicated with abscess: Case report and literature review. World J Gastroenterol. 2015;21:6398–6403. doi: 10.3748/wjg.v21.i20.6398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197.Carvalho R, Freire P, Portela F, Gomes D, Andrade P, Bento A, Batista H, Cipriano MA, Sofia C. Ectopic pancreas as a rare cause of obstructive jaundice. Rev Esp Enferm Dig. 2014;106:557–558. [PubMed] [Google Scholar]
- 198.Lemaire J, Delaunoit T, Molle G. Adenocarcinoma arising in gastric heterotopic pancreas. Case report and review of the literature. Acta Chir Belg. 2014;114:79–81. [PubMed] [Google Scholar]
- 199.Verdin V, Putzeys V, Delbecque K, Demarche M. Pancreatic ectopia: a case report. Acta Chir Belg. 2013;113:449–451. [PubMed] [Google Scholar]
- 200.Ginori A, Vassallo L, Butorano MA, Bettarini F, Di Mare G, Marrelli D. Pancreatic adenocarcinoma in duodenal ectopic pancreas: a case report and review of the literature. Pathologica. 2013;105:56–58. [PubMed] [Google Scholar]
- 201.Rao RN, Kamlesh Y, Pallav G, Singla N. Ectopic pancreas presenting as periampullary tumor with obstructive jaundice and pruritus is a rare diagnostic and therapeutic dilemma. A case report. JOP. 2011;12:607–609. [PubMed] [Google Scholar]
- 202.Bini R, Voghera P, Tapparo A, Nunziata R, Demarchi A, Capocefalo M, Leli R. Malignant transformation of ectopic pancreatic cells in the duodenal wall. World J Gastroenterol. 2010;16:1293–1295. doi: 10.3748/wjg.v16.i10.1293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.Yi SY. Ectopic pancreas in duodenal bulb. Clin Gastroenterol Hepatol. 2010;8:e30. doi: 10.1016/j.cgh.2009.10.005. [DOI] [PubMed] [Google Scholar]
- 204.Filippou DK, Vezakis A, Filippou G, Condilis N, Rizos S, Skandalakis P. A rare case of ectopic pancreas in the ampulla of Vater presented with obstructive jaundice. Ann Ital Chir. 2006;77:517–519. [PubMed] [Google Scholar]
- 205.Wagle PK, Shetty GS, Sampat M, Patel K. Ectopic pancreatic tissue mimicking ampullary tumor. Indian J Gastroenterol. 2005;24:265–266. [PubMed] [Google Scholar]
- 206.Sloots CE, de Brauw LM, Bot FJ, Greve JW. False-positive cytology in diagnostic laparoscopy due to ectopic pancreas. Dig Surg. 1999;16:434–436. doi: 10.1159/000018762. [DOI] [PubMed] [Google Scholar]
- 207.Kaneda M, Yano T, Yamamoto T, Suzuki T, Fujimori K, Itoh H, Mizumoto R. Ectopic pancreas in the stomach presenting as an inflammatory abdominal mass. Am J Gastroenterol. 1989;84:663–666. [PubMed] [Google Scholar]
- 208.Jain AS, Patel AM, Jain SR, Thakkar A. Accessory pancreatic lobe with gastric duplication cyst: diagnostic challenges of a rare congenital anomaly. BMJ Case Rep. 2015;2015 doi: 10.1136/bcr-2014-207751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 209.Kumagai H, Yokoyama K, Shimamura W, Miura Y, Yamagata T. Endoscopic ultrasonography appearance of pancreatic rest. Pediatr Int. 2014;56:654. doi: 10.1111/ped.12364. [DOI] [PubMed] [Google Scholar]
- 210.Deshpande VP, Raghunath BV, Sarin YK, Sinha S. Heterotopic pancreas causing duodenal obstruction in a patient previously treated for choledochal cyst. J Indian Assoc Pediatr Surg. 2012;17:40–42. doi: 10.4103/0971-9261.91088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 211.Shah A, Gordon AR, Ginsberg GG, Furth EE, Levine MS. Case report: ectopic pancreatic rest in the proximal stomach mimicking gastric neoplasms. Clin Radiol. 2007;62:600–602. doi: 10.1016/j.crad.2007.01.001. [DOI] [PubMed] [Google Scholar]
- 212.Friedland S, Yu A, Rouse RV, Wong D, Nino-Murcia M, Soetikno RM. Gastric pancreatic rest leading to pyogenic liver abscess. Gastrointest Endosc. 2002;56:438–440. doi: 10.1016/s0016-5107(02)70058-8. [DOI] [PubMed] [Google Scholar]
- 213.Shalaby M, Kochman ML, Lichtenstein GR. Heterotopic pancreas presenting as dysphagia. Am J Gastroenterol. 2002;97:1046–1049. doi: 10.1111/j.1572-0241.2002.05627.x. [DOI] [PubMed] [Google Scholar]
- 214.De Roche K, Gilbert M. Heterotopic pancreas presenting as hematemesis. Am J Gastroenterol . 2016;111:S1123. [Google Scholar]
- 215.Sharma A, Mohapatra S, Lim KJ, Chaubal A. Broder A. Gastric Outlet Obstruction Caused by a Triple Lesion: Eosinophilic Gastroenteritis, Brunner's Gland Hyperplasia, and Pancreatic Heterotopia in Unison. J Surg . 2017;5:43–46. [Google Scholar]
- 216.Pakala T, Wu G. An Unusual Location for Pancreatic Heterotopia: The Gastroesophageal Junction. Am J Gastroenterol . 2016;111:S802. [Google Scholar]
- 217.Bashir S, Borum M. Co-existent heterotopic pancreas and brunner gland hyperplasia causing gastric outlet obstruction and mimicking a pancreatic head tumor. Am J Gastroenterol. 2012;107:S380. [Google Scholar]
- 218.Matsushita M, Hajiro K, Takakuwa H. Acute pancreatitis occurring in gastric aberrant pancreas accompanied by paralytic ileus. Am J Gastroenterol. 1997;92:2121–2122. [PubMed] [Google Scholar]
- 219.Tanemura H, Uno S, Suzuki M, Miyauchi Y, Shimokawa K, Saji S, Ohashi H, Sakata K. Heterotopic gastric mucosa accompanied by aberrant pancreas in the duodenum. Am J Gastroenterol. 1987;82:685–688. [PubMed] [Google Scholar]
- 220.Waku T, Uetsuka H, Watanabe N, Mori T, Shiiki S, Nakai H, Orita Y, Harafuji I. A Case of Mucin-Producing Duodenal Carcinoma Arising from the Aberrant Pancreas. Jap J Gastroenterol Surg . 1996;29:2289–2293. [Google Scholar]
- 221.Nam JY, Lee SI, Chung JP, Choi SH, Lee DY, Choi JP, Lee JI, Lee SJ, Lee KS, Kang JK, Kim KW, Lim BJ, Park CI. [A case of duodenal adenocarcinoma arising from the heterotopic pancreas] Korean J Gastroenterol. 2003;42:164–167. [PubMed] [Google Scholar]
- 222.Matsuki M, Gouda Y, Ando T, Matsuoka H, Morita T, Uchida N, Kuriyama S. Adenocarcinoma arising from aberrant pancreas in the stomach. J Gastroenterol. 2005;40:652–656. doi: 10.1007/s00535-004-1601-9. [DOI] [PubMed] [Google Scholar]
- 223.Stock C, Keutgen XM, Pisapia D, Crawford C, Zarnegar R. Heterotopic pancreatic neoplasm presenting as an obstructing mass at the fourth portion of the duodenum. JOP. 2011;12:241–243. [PubMed] [Google Scholar]
- 224.Okamoto H, Kawaoi A, Ogawara T, Fujii H. Invasive ductal carcinoma arising from an ectopic pancreas in the gastric wall: a long-term survival case. Case Rep Oncol. 2012;5:69–73. doi: 10.1159/000335870. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 225.Mehra R, Pujahari AK, Jaiswal SS. Duodenal heterotopic pancreatic tissue: a case report and literature review. Gastroenterol Rep (Oxf) 2015;3:262–265. doi: 10.1093/gastro/gou049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 226.Priyathersini N, Sundaram S, Senger JL, Rajendiran S, Balamurugan TD, Kanthan R. Malignant Transformation in Gastric Pancreatic Heterotopia A Case Report and Review of the Literature. JOP . 2017;18:73–77. [Google Scholar]
- 227.Fujii M, Kawamoto H, Nagahara T, Tsutsumi K, Kato H, Shinoura S, Seno S, Okada H, Yamamoto K. A case of gastric aberrant pancreas diagnosed with EUS-guided FNA. Gastrointest Endosc. 2011;74:407–409. doi: 10.1016/j.gie.2011.03.1124. [DOI] [PubMed] [Google Scholar]
- 228.Tanaka T, Omote R, Okazaki N, Yanai H, Yoshino T. Gastric neuroendocrine tumor arising from heterotopic pancreas. Clin J Gastroenterol. 2018;11:34–37. doi: 10.1007/s12328-017-0795-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 229.Li Y, Zhou Y, Gu W, Nie Y. Obstructive Jaundice Caused by Heterotopic Pancreas. Am J Gastroenterol. 2017;112:1777. doi: 10.1038/ajg.2017.356. [DOI] [PubMed] [Google Scholar]
- 230.Sahni S, Mbachu JC, Stallwood C. Can we rest on just a rest? Am J Gastroenterol. 2017;112:S1380. [Google Scholar]
- 231.Shiwani MH. Laparoscopic excision of heterotopic pancreas of stomach. J Coll Physicians Surg Pak. 2010;20:620–621. [PubMed] [Google Scholar]
- 232.Plier M, Durez P, Komuta M, Raptis A. Severe panniculitis and polyarthritis caused by acinar cell carcinoma arising from an ectopic pancreas. BMJ Case Rep. 2017;2017:bcr2017222241. doi: 10.1136/bcr-2017-222241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 233.Boyaci C, Pasaoglu E, Okcu O, Dursun N. Gastric heterotopic pancreas with low grade dysplasia of the ducts. Pathol Int. 2016;66:645–647. doi: 10.1111/pin.12465. [DOI] [PubMed] [Google Scholar]
- 234.Rezvani M, Menias C, Sandrasegaran K, Olpin JD, Elsayes KM, Shaaban AM. Heterotopic Pancreas: Histopathologic Features, Imaging Findings, and Complications. Radiographics. 2017;37:484–499. doi: 10.1148/rg.2017160091. [DOI] [PubMed] [Google Scholar]
- 235.Jennings RE, Berry AA, Strutt JP, Gerrard DT, Hanley NA. Human pancreas development. Development. 2015;142:3126–3137. doi: 10.1242/dev.120063. [DOI] [PubMed] [Google Scholar]
- 236.Papageorge M, Waghray A, Chen L, Yoo J. Pedunculated small bowel lipoma with heterotopic pancreas causing intussusception. Clin Case Rep. 2018;6:1885–1886. doi: 10.1002/ccr3.1710. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 237.Molinari M, Ong A, Farolan MJ, Helton WS, Espat NJ. Pancreatic heterotopia and other uncommon causes of non-malignant biliary obstruction. Surg Oncol. 2000;9:135–142. doi: 10.1016/s0960-7404(00)00036-0. [DOI] [PubMed] [Google Scholar]
- 238.Kim JY, Lee JM, Kim KW, Park HS, Choi JY, Kim SH, Kim MA, Lee JY, Han JK, Choi BI. Ectopic pancreas: CT findings with emphasis on differentiation from small gastrointestinal stromal tumor and leiomyoma. Radiology. 2009;252:92–100. doi: 10.1148/radiol.2521081441. [DOI] [PubMed] [Google Scholar]
- 239. von Heinrich H. Ein Beitrag zur Histologie des sogen. akzessorischen Pankreas. Virchows Archiv für pathologische Anatomie und Physiologie und für klinische Medizin 1909; 198(3): 392-401 . [Google Scholar]
- 240.Gaspar Fuentes A, Campos Tarrech JM, Fernández Burgui JL, Castells Tejón E, Ruíz Rossello J, Gómez Pérez J, Armengol Miró J. [Pancreatic ectopias] Rev Esp Enferm Apar Dig. 1973;39:255–268. [PubMed] [Google Scholar]
- 241.Mandal S, Mandal AK. Heterotopic pancreas presenting as a tumour nodule along with perforation and peritonitis is rare: report of two cases. ANZ J Surg. 2008;78:725. doi: 10.1111/j.1445-2197.2008.04634.x. [DOI] [PubMed] [Google Scholar]
- 242.De Castro Barbosa JJ, Dockerty MB, Waugh JM. Pancreatic heterotopia; review of the literature and report of 41 authenticated surgical cases, of which 25 were clinically significant. Surg Gynecol Obstet. 1946;82:527–542. [PubMed] [Google Scholar]
- 243.Elwir S, Glessing B, Amin K, Jensen E, Mallery S. Pancreatitis of ectopic pancreatic tissue: a rare cause of gastric outlet obstruction. Gastroenterol Rep (Oxf) 2017;5:237–240. doi: 10.1093/gastro/gov037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 244.Jani B, Rzouq F, Saligram S, Nawabi A, Nicola M, Dennis K, Ernst C, Abbaszadeh A, Bonino J, Olyaee M. Groove Pancreatitis: A Rare form of Chronic Pancreatitis. N Am J Med Sci. 2015;7:529–532. doi: 10.4103/1947-2714.170624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 245.Raman SP, Salaria SN, Hruban RH, Fishman EK. Groove pancreatitis: spectrum of imaging findings and radiology-pathology correlation. AJR Am J Roentgenol. 2013;201:W29–W39. doi: 10.2214/AJR.12.9956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 246.Razi MD. Ectopic pancreatic tissue of esophagus with massive upper gastrointestinal bleeding. Arch Surg. 1966;92:101–104. doi: 10.1001/archsurg.1966.01320190103024. [DOI] [PubMed] [Google Scholar]
- 247.Madinaveitia JM, Madinaveitia M, Loma V. [Aberrant pancreas] Rev Esp Enferm Apar Dig Nutr. 1951;10:31–42. [PubMed] [Google Scholar]
- 248.Sathyanarayana SA, Deutsch GB, Bajaj J, Friedman B, Bansal R, Molmenti E, Nicastro JM, Coppa GF. Ectopic pancreas: a diagnostic dilemma. Int J Angiol. 2012;21:177–180. doi: 10.1055/s-0032-1325119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 249.Chou JW, Chang KP, Wu YH. A Rare Cause of Elevated Serum CA19-9. Gastroenterology. 2021;160:31–32. doi: 10.1053/j.gastro.2020.08.031. [DOI] [PubMed] [Google Scholar]