Abstract
Middle East respiratory syndrome (MERS) is a serious emerging zoonosis. It is characterized by severe infection of the respiratory tract in humans. Dromedary camels are considered to be the most probable origin of the pathogenic Middle East respiratory syndrome coronavirus (MERS-CoV). This cross-sectional survey was carried out to ascertain the seroprevalence of MERS-CoV in dromedary camels at Illela border and human patients in a secondary care hospital in Illela, Sokoto State, Nigeria from November 2016 to January 2017. Serum samples from 74 camels and 39 human patients were collected while a data form was administered to the camel handlers (40) and human patients to obtain information on zoographic characteristics of dromedary camels, demographic characteristics of camel handlers and human patients and some practices of both groups which are likely to predispose to MERS-CoV infection. The serum samples were analyzed for antibodies against MERS-CoV using the indirect Enzyme-Linked Immuno-Sorbent Assay (ELISA). All the camels sampled were seropositive against MERS-CoV and 74% of the human patients had antibodies against MERS. All handlers treated their camels without consulting veterinarians and there was little or no biosecurity measures undertaken. Age, sex, and occupation were not significant determinants for the presence of MERS-CoV antibody in human patients sampled. This study serves as a baseline for similar researches and due to the high seroprevalence obtained in this study for both camels and humans, there is need for trained personnel, surveillance and diagnostic tools at our border posts and animal markets.
Supplementary information
The online version contains supplementary material available at 10.1007/s00580-022-03351-3.
Keywords: MERS-CoV, Seroprevalence, Dromedary camels, Humans, Secondary care hospital
Background
Middle East Respiratory Syndrome (MERS), also known as camel flu (Bhatia et al. 2016; Alnemere 2020), is a lethal viral respiratory infection caused by the MERS coronavirus (MERS-CoV) and was first identified in humans in Saudi Arabia and Jordan in 2012 (Seddiq et al. 2017; Ramadan and Shaib 2019; Ugochukwu et al. 2019; Memish et al. 2020). The disease is typically more severe in individuals with underlining health problems and characterized by severe infection of the lower respiratory tract (Zumla et al. 2015; Ugochukwu et al. 2019). Although bats may act as a reservoir of MERS-CoV, it is unlikely that they are the immediate source for most human cases because human contact with bats is uncommon (Byrd and Roy 2015; Hu et al. 2015). Dromedary camels appear to be the major animal host for MERS-CoV (Mohd et al. 2016; Alhumaid et al. 2018; McIntosh 2019; Adney et al. 2020). Noteworthy is the fact that travel-related MERS cases have been reported around the world especially in Europe, Asia, Africa, and North-America. This virus is endemic in camel populations of East Africa and the Arabian Peninsula but worryingly, the zoonotic transmission of MERS-CoV is now well reported around the globe (Roess et al. 2016; Ugochukwu et al. 2019). The strongest evidence of camel-to-human transmission of MERS-CoV comes from a study in Saudi Arabia in which MERS-CoV was isolated from a man with fatal infection and from one of his camels; full-genome sequencing demonstrated that the viruses from both of them were identical (Azhar et al. 2014; Ramadan and Shaib 2019; Tolah et al. 2020). Case clusters in Britain, Tunisia, and Italy, and in hospitals in the Kingdom of Saudi Arabia, the United Arab Emirates, Iran, France, and South Korea strongly suggest that human-to-human transmission occurs (Abroug et al. 2014; Drosten et al. 2015; Guery et al. 2013; Park et al. 2015; World Health Organization (WHO) 2013, 2019; Mostafa et al. 2020).
Data suggest a direct zoonotic risk for MERS-CoV infection among persons who come in contact with camels, where MERS-CoV neutralizing antibodies in healthy persons who had daily occupational contact with dromedaries were detected but not in persons without such contact in Qatar (Mackay and Arden 2015; Reusken et al. 2015, 2016; Abbad et al. 2019).
There is maximum physical interaction between camels, other species of animals, camel handlers and individuals within Illela international market, this close interaction may predispose to the spread of infection. Despite the fact that camels are in abundance in the study area, there is dearth of information on MERS-CoV infection in this area. It is important to recognize that MERS-CoV has a high case fatality rate, at around 37% in the Middle East and 40% in Saudi Arabia where the disease originated from (Eric and Joerg 2015; Zumla et al. 2015; Singh 2016; Mobaraki and Ahmadzadeh 2019). Most of the camels found in the study area migrated across the Nigerian borders which are vast and largely unmanned. The infection has been established in the Middle East and most parts of North Africa from which most of these camels migrated from. The lack of effective surveillance (screening) at the border posts may lead to introduction of the infectious agent into Nigeria.
This study aimed to estimate the seroprevalence of MERS-CoV in dromedary camels and some patients in a secondary health care facility and to identify possible risk factors associated with MERS-CoV among camels, their handlers and patients in the secondary health care hospital using data form survey in Illela, Sokoto State.
Methods
Study area
Illela Local Government Area (LGA) shares a border with the Republic of Niger to the north, Gwadabawa LGA to the south, Gada LGA to the east, and Tangaza LGA to the west (Fig. 1). Illela’s geographical coordinates is between latitudes 13 40′ North and 5 20′ East. The Illela border is a gateway to Niger Republic, Benin Republic, and other countries in North and West Africa (Garba et al. 2019). The people of Illela mostly speak Hausa; however, other tribes within Nigeria and Republic of Niger are also found there. The inhabitants are majorly traders as goods are constantly moved in and out of Illela international border.
Fig. 1.
Map of Sokoto State showing Illela local government area
Study design and population
A cross-sectional study was used to survey for MERS-CoV in dromedary camels (at Illela international border and Illela international market) and some patients in Illela secondary care hospital, Sokoto State.
Sampling method
A non-randomized sampling method (convenience sampling) was used for the selection of camel batches and also human patients in the hospital. Sampled dromedaries at the study area were either transit or trade camels meant to be transported for slaughter or to a new household. These dromedaries were transported into Nigeria from Niger Republic.
Eligibility criteria
Inclusion
The criteria used to select camels, camel handlers, and patients in Illela secondary care hospital in this study included:
-
(i)
Consent obtained from camel owners, handlers, and patients in Illela secondary care hospital
-
(ii)
Availability of a reasonable number (≥ 66) of camels at the study area
-
(iii)
All age groups, sexes, apparently healthy and sick camels will be sampled.
-
(iv)
Patients sampled in Illela secondary care hospital must keep one or more animals.
Exclusion
The criteria used to exclude camels, camel handlers and patients in Illela secondary care hospital in this study included:
-
(i)
Batches of camels without consent
-
(ii)
Patients in the hospital suspected to have diseases like acquired immunodeficiency syndrome (AIDS) and all types of hepatitis infection
-
(iii)
Patients less than 12 years and greater than 60 years old were not sampled from the hospital.
Ethical consideration and consent
Ethical clearance was obtained before the commencement of this study from the Ministry of Health, Sokoto State. After proper restraint by the camel handlers, 5–10 ml of blood was collected from each camel and 2–3 ml from each human. Ethical clearance for research on “Transborder Survey of Middle East Respiratory Syndrome in dromedary camels and their herdsmen in Sokoto State, Nigeria was obtained with number SKHREC/020/016.
Sample size determination
The sample size for the dromedaries was determined by the formula described by Thrusfield (2007).
where: n is representing the Minimum sample size, z is representing the appropriate value for the standard normal deviation set at 95% confidence interval (1.96), p is the Prevalence 96% (Chu et al. 2015) while, q is Complementary probability (1-p) and d the Level of significance 5%.
With 10% non-response rate according to previous work done by Araoye, (2004) minimum sample size = 59.01/0.9. n = 66. However, 74 camel samples were collected to improve the chances of detection.
For humans: Sampling was purposive. Thirty-nine samples from outpatients with history of keeping one or more animals were collected in Illela secondary care hospital.
Data collection
A data form was made available to all consenting camel handlers and patients in Illela secondary care hospital. This was used to obtain information on the sources and population of camels and associated risk factors (sex, age, occupation, practice, and the presence of camels in patients’ houses). Face to face interview method was employed while the interviewer filled the forms appropriately. In all, 74, 40, and 39 data forms were filled for camels, camel handlers, and patients in the hospital respectively.
Sample collection and processing
Samples were collected and processed from the months of November 2016 to January 2017. After blood collection and proper storage during transportation from the field, all samples were processed at Usmanu Danfodiyo University, Sokoto Central laboratory in city campus where the sera were separated from the plasma by centrifugation at 2500 revolutions per minute (rpm) for 5 min. The sera were then appropriately transported to Zaria, frozen for 6 days at − 20 °C until it was time to run the ELISA test in the Public Health Laboratory of the Department of Veterinary Public Health and Preventive Medicine, Faculty of Veterinary Medicine, Ahmadu Bello University Zaria.
Sample analysis
Microplate ELISA reader of 450 nm wavelength was used to read the Optical density (OD) and absorbance of the entire plate using a single wavelength within 5 min after Stop Solution addition. Samples were then compared to the 1 U/ml Calibrator = Positive/Negative Cut-off which was used to calculate the threshold index that numerically discriminates Positives from Negatives: Each Sample OD was divided by the 1 U/ml calibrator OD and values obtained above the 1 U/ml value were a measure of positive antibody activity; below was negative for antibody. A ‘Cut-off’ line was drawn to indicate a threshold distinguishing between Positive/Negative.
Data analysis
The data obtained from serology and data forms were analyzed using SPSS: Statistical Package for the Social Sciences (SPSS version 20.0). Counts and frequencies for variables were presented using graphs and tables, and then summarized using percentages. Prevalence of MERS-CoV in camels and patients in the secondary care hospital were also calculated.
Sampling points, sources and destinations of camels were represented by spot maps. Statistics and data (STATA®) 14 was used to calculate 95% Confidence Interval (CI) for prevalence on MERS-CoV in dromedary camels and humans. Association between MERS-CoV and categorical variables was analyzed using Chi-square (χ2) test. Where significant association was observed, odds ratio was calculated to determine the magnitude and direction of the association. Values of p ≤ 0.05 were regarded as statistically significant.
Results
Prevalence of MERS-CoV amongst camels and humans at the study area
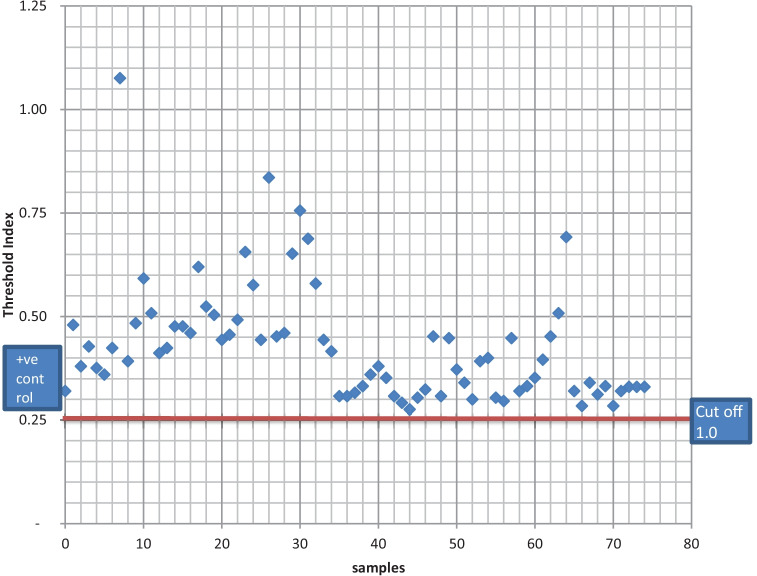
A total of 74 camels (adult males n = 52 (70%), adult females n = 10 (14%), young males n = 8 (11%) and young females n = 4 (5%) were sampled (Table 1). All the camels tested, were positive for MERS-CoV antibody (Fig. 2). Each sample was calculated in relation to 1 U/ml calibrator which had an optical density (OD) of 0.25/0.30 and represents the cut-off as indicated by the manufacturer (ADI®). The respiratory signs observed by handlers in their sampled camels was mainly coughing (n = 4; 10%) while 36 (90%) of the camels showed no respiratory signs in the past 2 weeks before the study. Likewise, according to the bio data forms, the handlers observed gastrointestinal signs in their camel herds 13 (32.5%) for diarrhea while 27 of the camel herders (67.5%) reported that they did not observe any gastrointestinal signs in the past 2 weeks before the study. It is important to note that information retrieved from the bio-data form showed that 75.7% (n = 56) of the sampled camels were sourced from Niger Republic.
Table 1.
Sex and AGe of dromedaries sampled at Illela LGA, Sokoto State
| Sampling point | Sub total | Sex | Age | Infection | |||
|---|---|---|---|---|---|---|---|
| M | F | Y | A | positive | |||
| Border | 32 | 25 | 7 | 9 | 23 | 32% | |
| Market | 42 | 35 | 7 | 3 | 39 | 42% | |
| Total | 74 | 60 | 14 | 12 | 62 | 74% | |
Legend- M, male; F, female; Y, young; A, adult
Fig. 2.
Serological result of dromedary camels sampled at Illela, Sokoto State
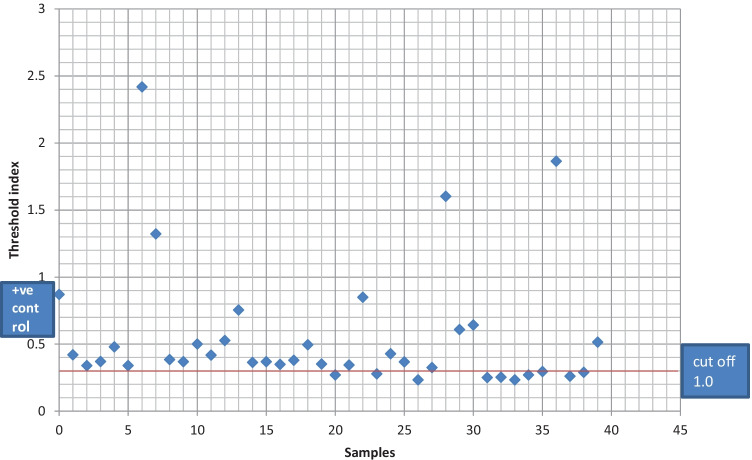
In humans tested at the facility, nine (23%) were negative while 29 (74%) were positive for MERS-CoV antibody (Fig. 3). The prevalence of MERS-CoV (74.4%) in sampled patients at the selected secondary care hospital had a confidence interval of 0.5787261–0.8696232 at 95% confidence limit.
Fig. 3.
Serological result of human participants at a secondary care hospital at Illela, Sokoto State
Demographic characteristics of camel handlers at Illela International market and border
All the camel handlers from sampled camels were males and their ages groupings were 31–40 years (48%), 20–30 years (33%), > 40 years (12%) and < 19 years (7%). About two-thirds were full time handlers (65%) while others (35%) part time. Data on trade practices revealed that 2 (5%) of the handlers had been practicing camel trade for < 1 year, 12 (30%) had been in the practice for > 10 years while 26 (65%) had been in the practice for 1–10 years. Handlers that rather treat their camels by self were 40 (100%) and the same percentage would not sanitize or disinfect their hands after handling camels. From the assessment of the 40 camel handlers, most of them have not noticed any respiratory (90%, n = 36) and/or gastrointestinal (67.5%, n = 27) symptoms in their batches for the past 2 weeks before the research.
Demographic characteristics of sampled human patients at Illela secondary facility
A total of 39 patients were sampled. 26 (67%) respondents were males while 13 (33%) were females. Based on the age categories, 48.72% of the patients sampled were between the ages of 20 and 30 years, 33.3% were between 31 and 40 years, 15.38% were above 40 years of age and only 1 (2.56%) was less than 19 years. The distribution of animals they housed in relation to their test result was; camels: 8, 1 and 1, donkeys: 10, 3 and 0, cattle: 6, 5 and 0, goats: 8, 3, and 0, sheep: 9, 2 and 0, dogs: 1, 0 and 0 and chickens: 6, 3 and 0 for positive, negative and invalid, respectively. The distribution of status by occupation of sampled patients had civil servants, farmers, housewives, paramilitary, students, and traders for positive, negative and invalid group, respectively (Table 2). The categorized ages of the patients in relation to their status were distributed into age groups 20–30, 31–40 and more than age 40 for positive, negative, and invalid group, respectively (Table 2). Male and female patients were grouped into positive, negative, and invalid groups, respectively (Table 3).
Table 2.
Assessment of the serological status of patients at Illela secondary care hospital with their age and occupation in Sokoto State
| Demographic variables | Positive (%) | Negative (%) | df | p-value | OR (95% CI on OR) |
|---|---|---|---|---|---|
| Age | 3 | 0.855 | |||
| 20–30 | 14 (73.7) | 4 (21.1) | 2.969 (0.136–64.962) | ||
| 31–40 | 9 (69.2) | 4 (30.8) | 2.374 (0.226–24.967) | ||
| > 40 | 5 (83.3) | 1 (16.7) | |||
| Occupation | 1 | 0.615 | |||
| Civil servant | 1 (25) | 3 (75) | 0.078 (0.004–1.428) | ||
| Housewife | 7 (77.8) | 2 (22.2) | 0.979 (0.090–10.675) | ||
| Student | 7 (77.8) | 2 (22.2) | 0.510 (0.044–5.948) | ||
| Trader | 9 (75) | 2 (16.7) |
SPSS Logistic Regression. The df and p-value for age are 3 and 0.86, respectively, while the values for occupation are 5 and 0.61, respectively
Legend: df, degree of freedom; OR, Odds Ratio; CI, Confidence Interval
Table 3.
Sex distribution of patients who were sampled at Illela secondary care hospital
| Sex | Total | |||
|---|---|---|---|---|
| Male | Female | |||
| Status | Intermediate | 0 | 1 | 1 |
| Negative | 3 | 6 | 9 | |
| Positive | 10 | 19 | 29 | |
| Total | 13 | 26 | 39 | |
Assessment of patient status based on some demographics in Illela secondary health facility
The different categories of age groups of patients in the hospital were compared with the ELISA test results, and there has been a high association between age and MERS seroprevalence; more than 40 years with 20–30 years (OR = 2.969; 95% CI 0.136–64.962) and more than 40 years with 31–40 years (OR = 2.374; 95% CI 0.226–24.967) (Table 2). From the assessment of occupation of patients, Traders with Civil servants (OR = 0.078; 95% CI 0.004–1.428), Traders with Housewives (OR = 0.979; 95% CI 0.090–10.675) and that for Traders with Students was (OR = 0.510; 95% CI 0.044–5.948) (Table 2). Patients’ antibody status whose collective percentage did not add up to 100% had invalid (Indeterminate) counts and Age (less than 19 years).
Discussion
Camels converging at various sampling points if infected can be a source of spread to animals and traders likewise, the risk of spread in health facilities.
Information retrieved from the bio-data form showed that majority of the sampled camels were sourced from Niger Republic. Nigeria has an extensive border land with Niger Republic, approximately 1,408.5 km in distance and extends from Kamba local government in Kebbi State through Sokoto, Katsina, Jigawa, Yobe States and ends at Damasak town in Borno State. The percentage of camels coming in through the Republic of Niger is probably higher compared to other sources like about 8.1% of these camels from Niger Republic, come into Niger Republic originally from Algeria while 5.4% of them from Mali. However, the case may be different as it is not all camels that are said to have been sourced from Niger Republic by the handlers is completely true; some might have been imported from other countries that share border with Niger Republic. This is in agreement with Chu et al. (2015), in a study in Kano abattoir where he reported Niger Republic and Mali among the likeliest sources of slaughtered camels in Nigeria. Camels from Gada LGA were most definitely from Niger Republic since it shares a porous border with this country.
Female camels during this study were possibly fewer in trade owing to reproductive reasons of being kept for breeding purposes. In addition, season might be an important factor as camel calving season occurs in the winter months (between late October and late February), this will reduce the female population on transit and may be a time when there is increased risk of human infection spill-overs due to new infections among the handlers (MacKay and Arden 2015). This study was carried out in November which falls within the dry season.
It could also have been that all the camels sampled had come into contact with an infected host sometime in their lifetime. This result is in agreement with the study conducted in Kano abattoir where on a daily basis, the seroprevalence ranged from 82 to 100% giving an overall seroprevalence of 96% (Chu et al. 2015). Similarly, it also agrees with Reusken et al. (2014), who obtained 94% seroprevalence in stored sera collected in Adamawa, Borno, Kano and Sokoto States. The high prevalence may be suggestive of our porous borders where no screening is undertaken for MERS-CoV, no proper diagnostic facilities and no animal health trained personnel at the market to ensure and ascertain the health status of traded animals.
Hand hygiene is essential in the transmission of MERS-CoV infection. The reports by 65% of the camel handlers who have been in the practice for 1 to 10 years as compared to those who have been handlers for less than 1 year (5%) and more than 10 years (30%) may be an indicator that the handlers and their immediate community are at risk of MERS-CoV exposure since 100% of the sampled camels were positive for MERS-CoV antibodies and the handlers in turn associate with the community members; hence, secondary spread is a possibility. The overall practices toward self-treatment of their camels and do nothing in terms of hand hygiene after handling camels; this warrants attention of public health and extension workers. Hence, a public health sensitization might be necessary as transmission is chiefly via contact with infected camels. It is believed that humans could be infected through direct or indirect connection with infected dromedary camels (Martins 2015). O’Keefe (2016), Alshukairi et al. 2018 and Kiyong'a et al. (2020) are of the opinion that those who handle camels are at higher risk of MERS-CoV infection than people who do not have regular close contacts with camels and the assessment of the 40 camel handlers, where most of them have not noticed any respiratory and/or gastrointestinal symptoms in the past 2 weeks before the research. This could be due to the infection being asymptomatic in most camels (Assiri et al. 2013).
In other studies, outside Nigeria; as early as 2013, Saudi Arabia reported 90% seroprevalence in their camels after the index case (Hemida et al. 2013). Also, in agreement with this present survey is that 100% blood serum collected from Omani camels had protein-specific antibodies against the MERS-CoV spike protein (Reusken et al. 2013). Similarly, findings of the study by Bennett (2014) is in agreement with our findings where all animals from one southern province of Tunisia were positive for MERS-CoV antibody. The high prevalence obtained in these studies might be due to previous infection that the camels had overcome, thereby evident by the presence of antibodies against MERS-CoV.
Prevalence did significantly differ by age although there were no significant statistical difference observed in the occupation and sex of human patients and this is in agreement with Kamau et al. (2015). However, the result was 100% positive for crop farmers and may reflect a relationship with occupational diseases when these farmers use animals for traction. The high prevalence in humans can possibly depict an ongoing non-pathogenic strain of the virus circulating in the study area. The virus does not appear to be easily transmitted except there is proximate contact such as providing clinical care to an infected patient while not applying strict hygiene measures. This has been seen among family members, patients, and health care workers where MERS-CoV human cases have been reported where strict biosafety measures were not applied (World Health Organization (WHO) 2015). The majority of cases have resulted from human-to-human transmission in health care settings. Infected persons with no symptoms have been found because they were tested for MERS-CoV during follow-up studies of contacts of people with MERS infection (World Health Organization (WHO) 2015).
However, there have been no human cases of MERS diagnosed in Sokoto State. This could be due to limited clinical or epidemiological surveillance for the virus where infections have gone unrecognized (Eric and Joerg 2015). It is not known if the MERS-CoV in Nigeria has similar zoonotic potential to other strains currently circulating in the Arabian Peninsula. Other explanations for the lack of zoonotic MERS-CoV infection in Africa include differences in patterns of exposure to infected animals or animal products or differences in human susceptibility to MERS-CoV (Chu et al. 2015).
Despite the fact that this was the first MERS-CoV research that would involve humans in Nigeria, there were few limitations namely, the kit used could have had cross reactivity with other genus of Coronaviruses; hence, the high seropositivity. Again, the sample size for humans was not calculated while the number of participants recruited were few; however, the sample size that was calculated for the dromedaries, although we couldn’t hit the 100 mark because of the constraints relating to consent from the herders.
Conclusions
This study serves as a baseline for similar researches. It confirms through serology that MER-CoV is zoonotic as both camels and human samples had a high percentage of sero-positives and this lends support to the fact that MERS-CoV had existed in Nigeria in the past unnoticed. It then raises these questions; why are we having low CFR of infections from MERS-CoV in Nigeria? Are the strains introduced in Nigeria less virulent? Have we developed some sort of natural immunity against MERS-CoV and other coronaviruses? The practice of treating camels by their handlers with lack of personal protective equipment during contact with camels may predispose these handlers to MERS-CoV infection. All the camels screened were positive for MERS-CoV antibody regardless of their source while more than half of the patients recruited at the secondary care facility were also positive for the virus. We recommend that Nigerian borders be provided with screening equipment and well-trained personnel to test animals for various zoonotic infections and those diseases of economic importance. Further molecular diagnostic research is needed to better understand the epidemiology of MERS-CoV in Nigeria.
Supplementary information
Below is the link to the electronic supplementary material.
Acknowledgements
We wish to appreciate Sokoto State Ministries of Health and Animal Health for the permission to carry out this research. We also want to thank the staff of the Federal Livestock Department (FLD) at Illela border post, the staff of the secondary health care facility in Illela town, and the animal handlers at the market for their immense cooperation. We acknowledge the assistance proffered by the Central laboratory, Usmanu Danfodiyo University Sokoto and the Department of Veterinary Public Health and Preventive Medicine laboratory, Ahmadu Bello University Zaria. We also recognize the contribution Alpha Diagnostics (ADI®) made through subsidizing their kits.
Abbreviations
- ADI
Alpha Diagnostics International
- CI
Confidence Interval
- ELISA
Enzyme-Linked Immunosorbent Assay
- HIV
Human Immunodeficiency Virus
- HRP
Horseradish peroxidase
- IgA
Immunoglobulin A
- IgG
Immunoglobulin G
- IgM
Immunoglobulin M
- Igs
Immunoglobulins
- LGA
Local Government Area
- MERS-CoV
Middle East Respiratory Syndrome Coronavirus
- OD
Optical density
- OR
Odds Ratio
- S1
Spike protein
- SPSS
Statistical Package for the Social Sciences
- STATA
Statistics and data
- TMB
Tetramethylbenzidine
- WHO
World Health Organization
Author contribution
SPS and GSNK conceived, performed the study and initial draft: SPS, GSNK, OOF and ICIU designed, contributed to data analysis and result interpretation, made substantial contribution to the review of the draft manuscript for intellectual content. All authors read and approved the final manuscript.
Availability of data and material
Necessary data and materials associated with this research work have been submitted with work in Figures and Table and the others are available for submission on the request of the editors.
Declarations
Ethics approval
This study was a surveillance of MERS-CoV. Approval was granted by Sokoto State Ministry of Health with Reference No. SKHREC/020)016). Written informed consent was obtained from all participants in this study prior to sample and data collection.
Consent for publication
The authors of this manuscript hereby give our unreserved consent for the peer-reviewing and subsequent publication of this manuscript. The human patients gave their consent following the Ethical approval being obtained but insisted on anonymity and we have their express permission to publish the findings of this research. However, written individual consent to publish was not applicable to this research work.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- Abbad A, Perera RAPM, Anga L, Faouzi A, Minh NNT, Malik SMMR, Lounes N, Maaroufi A, Van KMD, Peiris M, Nourlil J. Middle East respiratory syndrome coronavirus (MERS-CoV) neutralising antibodies in a high-risk human population, Morocco, November 2017 to January 2018. Eurosurv. 2019;24:48. doi: 10.2807/1560-7917.ES.2019.24.48.1900244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Abroug F, Slim A. Ouanes-Besbes L (2014) Family cluster of Middle East respiratory syndrome coronavirus infections, Tunisia. Emerg Infect Dis. 2013;20(9):1527–1530. doi: 10.3201/eid2009.140378. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Adney DR, Clancy CS, Bowen RA, Munster VJ. Camelid inoculation with Middle East respiratory syndrome coronavirus: experimental models of reservoir host infection. Vir. 2020;12(12):1370. doi: 10.3390/v12121370. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alhumaid S, Tobaiqy M, Albagshi M, Alrubaya M, Algharib F, Aldera A, Alali J. MERS-CoV transmitted from animal-to-human vs MERSCoV transmitted from human-to-human: comparison of virulence and therapeutic outcomes in a Saudi hospital. Trop J Pharmaceut Res. 2018;17(6):1155–1164. doi: 10.4314/tjpr.v17i6.23. [DOI] [Google Scholar]
- Alnemere AK. Middle East respiratory syndrome – what every otolaryngologist should know: a review. Int J Gen Med. 2020;13:483–489. doi: 10.2147/IJGM.S252796. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alshukairi AN, Zheng J, Zhao J, Nehdi A, Baharoon SA, Layqah L, Bokhari A, Al Johani SM, Samman N, Boudjelal M, Ten Eyck P, Al-Mozaini MA, Zhao J, Perlman S, Alagaili AN (2018) High prevalence of MERS-CoV infection in camel workers in Saudi Arabia. mBio 9(5):e01985–18 [DOI] [PMC free article] [PubMed]
- Araoye M (2004) Research methodology with statistics for health and social sciences.1st Edition. Natherdex Publishers, Ilorin, Nigeria
- Assiri A, Al-Tawfiq J, Al-Rabeeah A, Al-Rabiah F, Al-Hajjar S, Al-Barrak A, Flemban H, Al-Nassir W, Balkhy H, Al-Hakeem R, Makhdoom H, Memish Z, Zumla A. Epidemiological, demographic, and clinical characteristics of 47 cases of Middle East respiratory syndrome coronavirus disease from Saudi Arabia: a descriptive study. Lancet Inf Dis. 2013;13(9):752–761. doi: 10.1016/S1473-3099(13)70204-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Azhar EI, El-Kafrawy SA, Farraj SA, Hassan AM, Al-Saeed MS, Hashem AM, Madani TA. Evidence for camel-to-human transmission of MERS coronavirus. N Engl J Med. 2014;370(26):2499–2505. doi: 10.1056/NEJMc1409847. [DOI] [PubMed] [Google Scholar]
- Bennett S (2014) African Camels Show MERS virus more widespread than believed. http://bloomberg.com/news. Accessed 5 May 2019
- Bhatia PK, Sethi P, Gupta N, Biyani G. Middle East respiratory syndrome: a new global threat. Ind J Anaesth. 2016;60(2):85–88. doi: 10.4103/0019-5049.176286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Byrd RP, Jr, Roy TM. Identification and management of Middle East respiratory syndrome. Fed Pract. 2015;32(3):12–16. [Google Scholar]
- Chu D, Oladipo J, Perera R, Kurang S, Chan S, Poon L, Peiris M (2015) Middle East respiratory syndrome coronavirus (MERS-CoV) in dromedary camels in Nigeria. Eurosurv 20(49). 10.2807/1560-7917.ES.2015.20.49.30086 [DOI] [PubMed]
- Drosten C, Muth D. Corman VM (2015) An observational, laboratory-based study of outbreaks of Middle East respiratory syndrome coronavirus in Jeddah and Riyadh, Kingdom of Saudi Arabia. Clin Inf Dis. 2014;60(3):369–377. doi: 10.1093/cid/ciu812. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eric F, Joerg J (2015) Studying African camels is key to learning more about the MERS virus. http://theconversation.com/studying-african-camels-is-key-to-learning-more-about-the-MERS-virus-48761. Accessed 8 May 2019
- Garba M, Abubakar M, Ibrahim BS. Implications of Nigeria-Niger boundary demarcation. Int J Curr Res. 2019;11(10):7704–7707. [Google Scholar]
- Guery B, Poissy J, El Mansouf L. Clinical features and viral diagnosis of two cases of infection with Middle East Respiratory Syndrome coronavirus: a report of nosocomial transmission. Lancet. 2013;381(9885):2265–2272. doi: 10.1016/S0140-6736(13)60982-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hemida M, Perera R, Wang P, Alhammadi M, Siu L, Li M, Poon L, Saif L, Alnaeem A (2013) Middle East Respiratory Syndrome (MERS) coronavirus seroprevalence in domestic livestock in Saudi Arabia, 2010 to 2013. Eurosurv 18(50). 10.2807/1560-7917.ES2013.18.50.20659 [DOI] [PubMed]
- Hu B, Ge X, Wang LF, Shi Z. Bat origin of human coronaviruses. Virol J. 2015;12:221. doi: 10.1186/s12985-015-0422-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kamau EM, Ongus J, Gitau G, Galgalo T, Orinde A, Munyau P (2015) High seroprevalence of Middle East Respiratory Syndrome Coronavirus in camels from a slaughter house in Kenya. 6th AFENET and 1st ECOWAS Regional Center for Disease Control Scientific Conference
- Kiyong'a AN, Cook EAJ, Okba NMA, Kivali V, Reusken C, Haagmans BL, Fèvre EM. Middle East Respiratory Syndrome Coronavirus (MERS-CoV) seropositive camel handlers in Kenya. Vir. 2020;12(4):396. doi: 10.3390/v12040396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mackay IM, Arden KE (2015) MERS coronavirus: diagnostics, epidemiology and transmission. Virol J 12(222). 10.1186/s12985-015-0439-5 [DOI] [PMC free article] [PubMed]
- Martins I (2015) Nigeria must act to prevent MERS’, an interview by ThisDay Newspaper. 2015. [Online] http://www.latestnigeriannews.com/news/1686891/.html. Accessed 12 Jun 2018
- McIntosh K (2019) Middle East respiratory syndrome coronavirus: virology, pathogenesis and epidemiology. http://www.uptodate.com/contents/middle-east-respiratory-syndrome-coronavirus-virology-pathogenesis-and-epidemiology. Accessed 12 Aug 2019
- Memish ZA, Perlman S, Van Kerkhove MD, Zumla A. Middle East respiratory syndrome. Lancet. 2020;395:1063–1077. doi: 10.1016/S0140-6736(19)33221-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mobaraki K, Ahmadzadeh J (2019) Current epidemiological status of Middle East respiratory syndrome coronavirus in the world from 1.1.2017 to 17.1.2018: a cross-sectional study. BMC Inf Dis 19:351. 10.1186/s12879-019-3987 [DOI] [PMC free article] [PubMed]
- Mohd HA, Al-Tawfiq JA, Memish ZA. Middle East Respiratory Syndrome Coronavirus (MERS-CoV) origin and animal reservoir. Virol J. 2016;13:87. doi: 10.1186/s12985-016-0544-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mostafa A, Kandeil A, Shehata M, El Shesheny R, Samy AM, Kayali G, Ali MA. Middle East Respiratory Syndrome Coronavirus (MERS-CoV): state of the science. Microorg. 2020;8(7):991. doi: 10.3390/microorganisms8070991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- O’Keefe L. Middle East respiratory syndrome coronavirus (MERS-CoV) – Saudi Arabia. Wrkpl Hlth Safe. 2016;64(5):184–186. doi: 10.1177/2165079915607497. [DOI] [PubMed] [Google Scholar]
- Park YS, Lee C, Kim KM. The first case of the 2015 Korean Middle East Respiratory Syndrome outbreak. Epid Hlth. 2015;37:e2015049. doi: 10.4178/epih/e2015049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ramadan N, Shaib H (2019) Middle East respiratory syndrome coronavirus (MERS-CoV): a review. Germs 9(1):35–42. 10.18683/germs.2019.1155 [DOI] [PMC free article] [PubMed]
- Reusken C, Haagmans BL, Müller MA, Gutierrez C, Godeke GJ, Meyer B, Muth D, Raj VS, Smits-De Vries L, Corman VM, Drexler JF, Smits SL, El Tahir YE, De Sousa R, van Beek J, Nowotny N, van Maanen K, Hidalgo-Hermoso E, Bosch BJ, Rottier P, Koopmans MPG. Middle East respiratory syndrome coronavirus neutralizing serum antibodies in dromedary camels: a comparative serological study. Lancet Inf Dis. 2013;13(10):859–866. doi: 10.1016/S1473-3099.2813.2970164-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reusken CB, Chantal BEM, Messadi L, Ashenafi F, Ularamu H, Gert-Jan G, Agom D, Fufa D, Mohamed J, Simenew M, Shamaki D, Yusuf W, Yiltawe W, Endrias ZG, Ilse Z, Berend-Jan B, Bart LH, Marion PGK. Geographic distribution of MERS coronavirus among dromedary camels. Africa Emerg Inf Dis. 2014;20(8):1370–1374. doi: 10.3201/eid2008.140590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reusken C, Elmoubasher ABA, Farag SA, Bart L, Haagmans VS, Khaled A, Mohran KA, Gert-Jan G, Stalin G, Farhoud A, Salih AA, Hamad EA, Mohamed A, Berend-Jan B, Annemiek AV, Ahmed ME, Adel KI, Al-Molawi N, Marcel A, Marion PGK. Occupational exposure to dromedaries and risk for MERS-CoV infection, Qatar, 2013–2014. Emerg Inf Dis. 2015;21(8):1422–1425. doi: 10.3201/eid2108.150481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reusken CB, Raj VS, Koopmans MP, Haagmans BL. Cross host transmission in the emergence of MERS coronavirus. Curr Opin Virol. 2016;16:55–62. doi: 10.1016/j.coviro.2016.01.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roess AA, Carruth L, Lahm SA, Salman S. Camels, MERS-CoV, and other emerging infections in east Africa. Lancet Inf Dis. 2016;16(1):14–15. doi: 10.1016/S1473-3099(15)00471-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seddiq N, Al-Qahtani M, Al-Tawfiq JA, Bukamal N. First confirmed case of Middle East Respiratory Syndrome Coronavirus infection in the Kingdom of Bahrain: in a Saudi gentleman after cardiac bypass surgery. Case Rep Inf Dis. 2017 doi: 10.1155/2017/1262838. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Singh SK. Middle East Respiratory Syndrome virus pathogenesis. Sem Resp Crit Care Med. 2016;37(4):572–577. doi: 10.1055/s-0036-1584796. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thrusfield M. Veterinary Epidemiology. 3. Oxford: Blackwell Science Ltd; 2007. [Google Scholar]
- Tolah AM, Al Masaudi SB, El-Kafrawy SA, Mirza AA, Harakeh SM, Hassan AM, Alsaadi MA, Alzahrani AA, Alsaaidi GA, Amor NMS, Alagaili AN, Hashem AM, Azhar EI. Cross-sectional prevalence study of MERS-CoV in local and imported dromedary camels in Saudi Arabia, 2016–2018. PLoS ONE. 2020;15(5):e0232790. doi: 10.1371/journal.pone.0232790. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ugochukwu ICI, Agina OA, Ugochukwu EI. Middle East Respiratory Syndrome: its occurrence, aetiology, epidemiology, associated clinicopathological findings, diagnosis, prevention and control in humans and animals: An Overview. Not Sci Biol. 2019;11(3):320–324. doi: 10.15835/nsb11310406. [DOI] [Google Scholar]
- World Health Organization (WHO) (2013) Novel coronavirus summary and literature update – as of 17 May 2013. 2013. http://www.who.int/csr/disease/coronavirus_infections/update_20130517/en/index.html. Accessed 12 Jun 2019
- World Health Organization (WHO) Frequently asked questions on Middle East respiratory syndrome coronavirus (MERS-CoV) Co-Investigator n. 2015;2015:2019. doi: 10.1017/CBO9781107415324.004.Accessed12Jun. [DOI] [Google Scholar]
- World Health Organization (WHO) (2019) Middle East respiratory syndrome coronavirus (MERS-CoV): summary of current situation, literature update and risk assessment. 2019. http://apps.who.int/iris/bitstream/10665/179184/2/WHO_MERS_RA_15.1_eng.pdfyuiop. Accessed 12 Jun 2019
- Zumla A, Hui DS, Perlman S. Middle East respiratory syndrome. The Lancet. 2015;386(9997):995–1007. doi: 10.1016/S0140-6736(15)60454-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Necessary data and materials associated with this research work have been submitted with work in Figures and Table and the others are available for submission on the request of the editors.