Abstract
Periodic repetition of right heart catheterization (RHC) in pulmonary arterial hypertension (PAH) can be challenging. We evaluated the correlation between RHC and cardiopulmonary exercise test (CPET) aiming at CPET use as a potential noninvasive tool for hemodynamic burden evaluation. One hundred and forty‐four retrospective PAH patients who had performed CPET and RHC within 2 months were enrolled. The following analyses were performed: (a) CPET parameters in hemodynamic variables tertiles; (b) position of hemodynamic parameters in the peak end‐tidal carbon dioxide pressure (PETCO2) versus ventilation/carbon dioxide output (VE/VCO2) slope scatterplot, which is a specific hallmark of exercise respiratory abnormalities in PAH; (c) association between CPET and a hemodynamic burden score developed including mean pulmonary arterial pressure (mPAP), pulmonary vascular resistance (PVR), cardiac index, and right atrial pressure. VE/VCO2 slope and peak PETCO2 significantly varied in mPAP and PVR tertiles, while peak oxygen uptake (peak VO2) and O2 pulse varied in the tertiles of all hemodynamic parameters. PETCO2 versus VE/VCO2 slope showed a strong hyperbolic relationship (R 2 = 0.7627). Patients with peak PETCO2 > median (26 mmHg) and VE/VCO2 slope < median (44) presented lower mPAP and PVR (p < 0.005) than patients with peak PETCO2 < median and VE/VCO2 slope > median. Multivariate analysis individuated peak VO2 (p = 0.0158) and peak PETCO2 (p = 0.0089) as hemodynamic score independent predictors; the formula 11.584 − 0.0925 × peak VO2 − 0.0811 × peak PETCO2 best predicts the hemodynamic score value from CPET data. A significant correlation was found between estimated and calculated scores (p < 0.0001), with a precise match for patients with mild‐to‐moderate hemodynamic burden (76% of cases). The results of the present study suggest that CPET could allow to estimate the hemodynamic burden in PAH patients.
Keywords: cardiopulmonary exercise test, oxygen uptake, pulmonary arterial hypertension, right heart catheterization
INTRODUCTION
Pulmonary arterial hypertension (PAH) is a rare disease characterized by an increase in pulmonary vascular resistance (PVR) due to remodeling, fibrosis, and thrombosis in situ of pulmonary arterioles, with consequent pressure overload and right heart failure. 1 The main clinical manifestations of PAH are reduced exercise capacity and fluid retention; in the absence of treatment, the disease inevitably evolves to death. 2
According to European guidelines, right heart catheterization (RHC) is mandatory to confirm the diagnosis of PAH, and it is of main importance during patient follow‐up. Indeed, it should be considered 3–6 months after therapy changes and in case of clinical deterioration, moreover it can also be considered at regular intervals during follow‐up in stable patients, like some PAH centers do. 3
However, in light of the complexity of the disease and its course, in the last decade there has been a focus on the importance of a multiparametric evaluation of disease diagnosis, severity, prognosis, and follow‐up. Consequently, several kinds of noninvasive markers have been proposed. 3
Cardiopulmonary exercise test (CPET) is currently considered the gold standard for assessing the degree and causes of exercise intolerance, providing important information on gas exchange, ventilatory efficacy, and cardiac function during effort. 4 PAH patients show a typical CPET pattern, with marked hyperventilation, low end‐tidal partial pressure of carbon dioxide (PETCO2), high ventilatory equivalents for oxygen and carbon dioxide, and elevated ventilation/carbon dioxide production (VE/VCO2) relationship slope. 5 , 6 Indeed, the combination of low PETCO2 and high ventilatory equivalents for carbon dioxide at the anaerobic threshold (AT) has been considered as suggestive of pulmonary hypertension. 7 , 8 Moreover, some CPET parameters have shown a significant prognostic value in PAH, 9 , 10 , 11 and O2 consumption at exercise peak (peak VO2) and VE/VCO2 slope are currently included among the markers suggested by European guidelines for risk stratification in PAH patients. 3
The purpose of this study was to investigate the association between ergospirometric parameters and the invasive hemodynamic variables usually related to disease severity and prognosis in PAH, aiming at proposing a potential noninvasive tool useful for the decision‐making process.
METHODS
Study population
We conducted a multicenter retrospective study involving four Italian centers with sound expertize in performing CPET in PAH patients: Centro Cardiologico Monzino (CCM) of Milan, Sapienza University of Rome, IRCCS Maugeri of Milan, Ospedali Riuniti OO.RR. of Foggia. 6 , 12 , 13 , 14
Study inclusion criteria were: age over 18 years, diagnosis of Group 1 pulmonary hypertension confirmed by RHC (excluding PAH associated to congenital heart disease), optimized and individually tailored drug treatment. Patients suffering from pulmonary hypertension due to other causes including left heart disease, lung diseases and/or hypoxia, or chronic thromboembolic PH were excluded from study. Moreover, patients with pericardial disease, exercise‐induced angina, ST changes, and severe arrhythmias were also excluded, as were patients with any comorbidity directly affecting exercise performance.
Study protocol
All patients underwent a full instrumental and clinical assessment. Diagnosis of PAH was made according to the European guidelines. 3 Baseline evaluation included medical history, physical examination, 12‐lead electrocardiogram (ECG), complete transthoracic echocardiogram, lung function test, RHC, and CPET. To become familiar with the CPET procedure, patients had been previously trained to perform an exercise test.
Each patient's treatment was based on the severity of PAH, in accordance to European guidelines. 3
The investigation was approved by the CCM IRCCS Scientific Committee and notified, due to its retrospective nature and the anonymous use of medical data (CCM–PR182), to the CCM IRCCS Ethics Committee. All participants signed an informed consent for both RHC and CPET. The study was conducted in compliance with the declaration of Helsinki.
CPET
CPET was performed on an electronically braked cycle ergometer using a personalized ramp protocol that was chosen aiming at a test duration of 10 min. 15 All patients had previously performed at least one CPET and were carefully instructed by the medical staff about exercise procedures. The exercise was preceded by at least 3 min of rest gas exchange monitoring and by a short unloaded warm‐up period. During the exercise test, 12‐lead ECG, blood pressure, and heart rate were recorded, and oxygen saturation was monitored through a pulse oximeter. The participants either wore a nose clip and breathed through a mouthpiece, or used a facemask connected to a mass flow‐meter as they preferred. CPET was carried out and interpreted using a standard technique. 6 , 16 Specifically, subjects were asked to cycle at a pedaling rate of 60–70 rpm, and CPET was self‐terminated by the subjects when they claimed that maximal effort had been achieved. VO2, VE, and VCO2 were measured breath by breath and are reported as 20‐s averages. Linear regression was applied to the VE/VCO2 relationship from 1 min after the beginning of loaded pedaling to the end of the isocapnic buffering period. AT was calculated with the standard technique. 17 All tests were executed and evaluated by two cardiologists experienced in CPET. CCM was responsible for data collection and analysis, while individual investigators were responsible for their own records.
RHC
RHC was performed by an experienced operator, within a <2‐month time frame from CPET during which clinical and therapeutic stability was observed. Hemodynamic evaluation was made with standard technique: patients underwent RHC with a Swan‐Ganz triple‐lumen thermodilution catheter, in the supine position, with zero calibration at the center of the thorax and detection of pressure at the end of exhalation. Measurements performed included mean pulmonary artery pressure (mPAP), wedge pressure (WP), right atrial pressure (RAP), and cardiac output (CO). PVR was calculated with the formula (mPAP–WP)/CO; cardiac index (CI) was obtained by dividing CO by body surface area.
Hemodynamic impairment score
To stratify the degree of patients' hemodynamic burden, we created an arbitrary score according to the tertiles distribution of the four hemodynamic variables assessed at RHC: mPAP, RAP, CI, and PVR. These parameters were considered as routinely assessed in clinical practice and of recognized prognostic relevance in PAH. 3 We have chosen the subdivision into tertiles in order to have a homogeneous stratification of the impairment degree of the single parameter and we have chosen to integrate the 4 parameters into a score in order to have a global hemodynamic load marker.
We assigned a value of 1 to the lower tertile of hemodynamic impairment of each variable, a value of 2 to the intermediate tertile, and a value of 3 to the higher tertile of hemodynamic impairment. Therefore, adding the relative value for all four parameters, our final hemodynamic impairment score ranged between a minimum value of 4 (minimum degree of impairment for all four parameters) to a maximum value of 12 (maximum degree of impairment for all four parameters).
Statistical analysis
Continuous variables are expressed as mean ± SD and categorical variables are expressed as number and percentage. The differences of CPET parameters in the tertiles of hemodynamic variables were assessed using analysis of variance (ANOVA). The association between ergospirometric parameters and a score of hemodynamic impairment built from the 4 hemodynamic parameters (mPAP, PVR, CI, and RAP) was investigated by multivariate linear regression analysis with stepwise selection. The association between estimated and calculated score was assessed by Spearman correlation. The association between ergospirometric and hemodynamic values according to hemodynamic impairment scores merged into three wider groups was studied by ANOVA and p for trend.
Analyses were performed with the SAS statistical package v. 9.4 (SAS Institute Inc.), and all tests were two‐sided. p < 0.05 was considered as statistically significant.
RESULTS
Data of 144 patients with PAH were retrospectively collected. Clinical, hemodynamic, and ergospirometric characteristics of the study population are reported in Table 1. The majority of patients were female, in WHO Class III, had idiopathic PAH, and presented with moderate‐to‐severe pulmonary hypertension with significant impairment of exercise capacity.
Table 1.
Clinical, hemodynamic, and ergospirometric characteristic of study population
| Patients, n | 144 |
| Age (years) | 53 ± 16 |
| Gender, M/F | 61/82 |
| PAH etiology, n (%) | |
| Idiopathic PAH | 126 (87.5) |
| Hereditary PAH | 3 (2.1) |
| PAH associated to connective tissue disease | 10 (6.9) |
| Portopulmonary PAH | 4 (2.8) |
| PAH associated to HIV infection | 1 (0.7) |
| WHO, class | |
| I, n (%) | 2 (1.4) |
| II, n (%) | 45 (31.3) |
| III, n (%) | 93 (64.5) |
| IV, n (%) | 4 (2.8) |
| Hemodynamic parameters | |
| mPAP (mmHg) | 45 ± 15 |
| CI (l/min/m2) | 2.5 ± 0.7 |
| RAP (mmHg) | 7.5 ± 3.9 |
| PVR (WU) | 9.1 ± 5.5 |
| Ergospirometric parameters | |
| AT workload (watt) | 39 ± 22 |
| Peak workload (watt) | 68 ± 32 |
| AT HR (b/min) | 108 ± 18 |
| Peak HR (b/min) | 131 ± 24 |
| AT VO2 (ml/kg/min) | 11.2 ± 3.4 |
| Peak VO2 (ml/kg/min) | 15.5 ± 4.7 |
| Peak VO2 (% predicted) | 59 ± 19 |
| Peak O2 pulse (ml) | 8.8 ± 3.4 |
| Peak PETCO2 (mmHg) | 25.9 ± 6.1 |
| VE/VCO2 slope | 45.5 ± 14.4 |
| VO2/work slope | 9.7 ± 2.8 |
Note: Data are expressed as absolute numbers and percentages or mean ± SD. AT is not measurable in 18 patients.
Abbreviations: AT, anaerobic threshold; CI, cardiac index; HIV, human immunodeficiency virus; HR, heart rate; mPAP, mean pulmonary arterial pressure; PAH, pulmonary arterial hypertension; PETCO2, end‐tidal carbon dioxide pressure; pulse O2, oxygen pulse; PVR, pulmonary vascular resistance; RAP, right atrial pressure; VO2, oxygen uptake; VE/VCO2 slope, ventilation to carbon dioxide production slope; VO2/work: oxygen uptake to work slope; WHO, World Health Organization.
To investigate possible associations between CPET and RHC parameters, we first evaluated the mean values of major ergospirometric parameters in the tertiles of hemodynamic variables, considering I the tertile grouping patients with the lowest degree of RHC parameter impairment, and III the tertile related to the highest degree of impairment (Table 2). Analysis of variance reveals that ventilatory parameters (VE/VCO2 slope and PETCO2 peak) show statistically significant variations in the tertiles of hemodynamic parameters mainly associated to afterload changes (mPAP and PVR), while peak VO2 and O2 pulse significantly vary for all four hemodynamic parameters. Conversely, the values of cardiovascular efficiency slope (VO2/work slope) do not appear to vary significantly with the worsening of hemodynamic impairment.
Table 2.
Mean values of each ergospirometric parameter for the three tertiles of hemodynamic variables
| mPAP tertiles | ||||
|---|---|---|---|---|
| I | II | III | p | |
| ≤38 mmHg | >38 and ≤52 mmHg | >52 mmHg | ||
| VE/VCO2 slope | 41.0 ± 12.0 | 47.4 ± 15.0 | 48.5 ± 15.3* | 0.02 |
| Peak PETCO2 (mmHg) | 28.1 ± 6.1 | 24.8 ± 5.7** | 24.6 ± 6.0* | 0.006 |
| Peak VO2 (ml/min/kg) | 16.3 ± 5.1 | 15.7 ± 5.2 | 14.3 ± 3.5* | 0.007 |
| Peak O2 pulse (ml) | 9.7 ± 3.6 | 9.1 ± 3.6 | 7.4 ± 2.3*, *** | 0.003 |
| VO2/work slope (ml/min/W) | 10.1 ± 2.2 | 9.7 ± 2.1 | 9.6 ± 2.6 | NS |
| PVR tertiles | ||||
|---|---|---|---|---|
| I | II | III | p | |
| ≤5.9 WU | >5.9 and ≤10.1 WU | >10.1 WU | ||
| VE/VCO2 slope | 41.6 ± 13.3 | 46.1 ± 13.3 | 49.9 ± 15.7* | 0.022 |
| Peak PETCO2 (mmHg) | 28.0 ± 6.0 | 24.8 ± 5.7** | 24.3 ± 5.7* | 0.004 |
| Peak VO2 (ml/min/kg) | 16.9 ± 5.2 | 15.7 ± 4.4 | 13.8 ± 4.1* | 0.007 |
| Peak O2 pulse (ml) | 10.0 ± 3.6 | 9.3 ± 3.4 | 7.1 ± 2.2*, *** | 0.000 |
| VO2/work slope (ml/min/W) | 10.3 ± 2.2 | 9.7 ± 2.2 | 9.4 ± 2.5 | NS |
| CI tertiles | ||||
|---|---|---|---|---|
| I | II | III | p | |
| >2.8 L/min/m2 | >2.2 and ≤2.8 L/min/m2 | ≤2.2 L/min/m2 | ||
| VE/VCO2 slope | 45.2 ± 12.8 | 43.7 ± 14.6 | 48.2 ± 15.2 | NS |
| Peak PETCO2 (mmHg) | 26.2 ± 5.7 | 26.6 ± 6.0 | 24.5 ± 6.0 | NS |
| Peak VO2 (ml/min/kg) | 16.0 ± 4.0 | 16.8 ± 4.7 | 14.0 ± 5.0*** | 0.01 |
| Peak O2 pulse (ml) | 8.8 ± 3.1 | 9.0 ± 3.3** | 8.5 ± 3.7* | 0.000 |
| VO2/work slope (ml/min/W) | 9.9 ± 2.4 | 10.2 ± 2.2 | 9.3 ± 2.3 | NS |
| RAP tertiles | ||||
|---|---|---|---|---|
| I | II | III | p | |
| ≤6 mmHg | >6 and ≤8 mmHg | >8 mmHg | ||
| VE/VCO2 slope | 44.3 ± 14.2 | 42.5 ± 11.2 | 48.9 ± 16.3 | NS |
| Peak PETCO2 (mmHg) | 26.1 ± 5.6 | 27.3 ± 5.2 | 25.0 ± 7.1 | NS |
| Peak VO2 (ml/min/kg) | 16.8 ± 5.3 | 16.5 ± 4.2 | 13.5 ± 3.7*, *** | 0.002 |
| Peak O2 pulse (ml) | 9.0 ± 3.5 | 10.2 ± 3.5 | 7.6 ± 2.8*** | 0.024 |
| VO2/work slope (ml/min/W) | 9.8 ± 2.0 | 10.6 ± 2.0 | 9.4 ± 2.9 | NS |
Note: Data are expressed as mean ± SD.
Abbreviations: ANOVA, analysis of variance; CI, cardiac index; mPAP, mean pulmonary arterial pressure; NS, nonsignificant; PAH, pulmonary arterial hypertension; PETCO2, end‐tidal carbon dioxide pressure; pulse O2, oxygen pulse; PVR, pulmonary vascular resistance; RAP, right atrial pressure; VO2, oxygen uptake; VE/VCO2 slope, ventilation to carbon dioxide production slope; VO2/work, oxygen uptake to work slope.
p for ANOVA < 0.05 III group versus I group
p for ANOVA < 0.05 II group versus I group
p for ANOVA < 0.05 III group versus II group.
Subsequently, we built a scatterplot of peak PETCO2 versus VE/VCO2 slope, choosing two parameters with peculiar behavior during exercise in PAH. 6 , 7 , 8 , 18 Although PETCO2 highest value is achieved during the isocapnic buffering period, in the present study we had chosen to consider PETCO2 value at exercise peak as both it is obtainable in all patients, while the isocapnic buffering period may be not identifiable in some patients, and it is associated with PAH typical effort hyperventilation.
The graph revealed a hyperbolic relationship between the two parameters (R 2 = 0.7627, Figure 1). We then divided the scatterplot into four quadrants, according to the median value of peak PETCO2 (26 mmHg) and the median value of VE/VCO2 slope (44). Most patients were included in Quadrant I (peak PETCO2 > median and VE/VCO2 slope < median, corresponding to mild impairment of both parameters) and in Quadrant III (peak PETCO2 < median and VE/VCO2 slope > median value, corresponding to high degree of impairment of both parameters) of the graph (63 [43% of cases] and 58 [41%] cases, respectively]). Only 14 (10% of cases) and 9 (6%) patients were included in Quadrant II and IV, respectively. The average value of RAP, mPAP, CI, and PVR in each quadrant of peak PETCO2– VE/VCO2 slope scatterplot is reported in Table 3. Compared to Quadrant III, Quadrant I was made up of patients with lower mPAP and PVR and higher CI, however a statistically significant difference of Quadrant III versus Quadrant I was in mPAP and PVR only.
Figure 1.
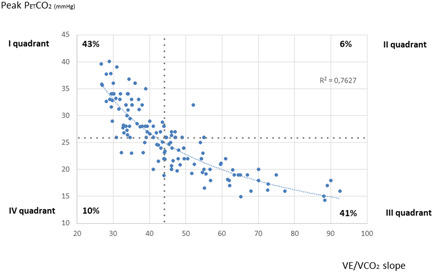
Scatterplot of VE/VCO2 slope versus peak PETCO2. PETCO2, end‐tidal carbon dioxide pressure; VE/VCO2 slope, ventilation to carbon dioxide production slope
Table 3.
Mean values of hemodynamic parameters in the four quadrants of peak PETCO2–VE/VCO2 slope scatterplot
| Quadrant | N pts (%) | mPAP (mmHg) | RAP (mmHg) | CI (L/min/m2) | PVR (WU) |
|---|---|---|---|---|---|
| I Peak PETCO2 > median–VE/VCO2 slope < median | 63 (48%) | 41 ± 15* | 7 ± 3 | 2.7 ± 0.7 | 7.9 ± 5.2* |
| II Peak PETCO2 and VE/VCO2 slope > median | 14 (10%) | 47 ± 18 | 8 ± 5 | 2.5 ± 0.4 | 8.4 ± 5.8 |
| III Peak PETCO2 < median–VE/VCO2 slope > median | 58 (41%) | 49 ± 14* | 8 ± 4 | 2.4 ± 0.7 | 10.7 ± 5.7* |
| IV Peak PETCO2 and VE/VCO2 slope < median | 9 (6%) | 47 ± 16 | 7 ± 5 | 2.4 ± 0.7 | 8.7 ± 4.0 |
Note: Data are expressed as mean ± SD.
Abbreviations: ANOVA, analysis of variance; CI, cardiac index; mPAP, mean pulmonary arterial pressure; PETCO2, end‐tidal carbon dioxide pressure; PVR, pulmonary vascular resistance; RAP, right atrial pressure; VE/VCO2 slope, ventilation to carbon dioxide production slope.
p for ANOVA < 0.05 III versus I quadrant.
Finally, we investigated the association between ergospirometric parameters and the hemodynamic burden score built from four hemodynamic parameters (mPAP, PVR, CI, and RAP). Univariate analysis identified peak VO2 (p = 0.0002), peak O2 pulse (p = 0.0012), peak PETCO2 (p = 0.0011), and VE/VCO2 slope (p = 0.0012) as significantly associated to hemodynamic score, while multivariate analysis individuated peak VO2 (p = 0.0158) and peak PETCO2 (p = 0.0089) as independent predictors of the score. Multivariate linear regression analysis provided the formula 11.584 − 0.0925 × peak VO2 − 0.0811 × peak PETCO2, which allows predicting the hemodynamic score value for each patient.
We found a significant correlation between estimated and calculated score (p < 0.0001, R 2 = 0.37). Sixty‐nine out of 144 study patients (48% of cases) showed the same estimated and calculated score value or an estimated value ±1 from the calculated score, and 40 patients (28%) had an estimated score value ±2 from the calculated score. Thus, 76% of our study population presented an estimated score value within ±2 from the calculated score value. The remaining 35 patients showed a difference between estimated and calculated score value ≥3. Thirteen and 22 subjects presented an overestimation and an underestimation of the score value, respectively. The former belonged to patients with score values ranging between 4 and 6, and the latter to patients with score values between 11 and 12. In brief, an optimal match was observed for score values from 7 to 10, while a slight overestimation and underestimation was observed for lower and higher scores, respectively.
The estimated score values found in our population vary from a minimum of 5 to a maximum of 10, with the great majority of cases ranging between 7 and 9, shown to have the greatest calibration. To increase the sample size of the estimated hemodynamic score groups, we merged Scores 5, 6, and 7 into Group A (n = 31), and Scores 9 and 10 into Group C (n = 41); Group B comprehends 72 patients with Score 8 (Table 4). The increase in the hemodynamic score from Group A to Group C is associated with a progressive worsening of both hemodynamic and ergospirometric parameters (Table 4).
Table 4.
Ergospirometric and hemodynamic values according to hemodynamic impairment scores merged into three wider groups
| Score group | N pts | Peak VO2 (ml/min/kg) | Peak pulse O2 (ml) | Peak PETCO2 (mmHg) | VE/VCO2 slope | VO2/Work slope (ml/min/W) | mPAP (mmHg) | RAP (mmHg) | CI (L/min/m2) | PVR (WU) |
|---|---|---|---|---|---|---|---|---|---|---|
| A | 31 | 20.5 ± 5.3 | 10.5 ± 4.1 | 33.0 ± 4.0 | 33.3 ± 4.7 | 10.0 ± 4.1 | 38 ± 13 | 7 ± 3 | 2.6 ± 0.5 | 6.9 ± 4.3 |
| B | 72 | 15.5 ± 3.2 | 9.1 ± 3.0 | 26.5 ± 4.5 | 42.4 ± 8.5 | 10.1 ± 2.1 | 47 ± 16 | 7 ± 4 | 2.7 ± 0.7 | 8.7 ± 5.2 |
| C | 41 | 11.7 ± 2.4 | 6.9 ± 2.4 | 19.7 ± 3.0 | 60.2 ± 15.5 | 8.7 ± 2.5 | 48 ± 14 | 9 ± 5 | 2.2 ± 0.6 | 11.5 ± 6.2 |
| p for trend | <0.0001 | <0.0001 | <0.0001 | <0.0001 | 0.0531 (NS) | 0.0065 | 0.0182 | 0.0063 | 0.0004 | |
| p for ANOVA | <0.0001 | <0.0001 | <0.0001 | <0.0001 | 0.0457 | 0.0102 | 0.0066 | 0.001 | 0.0015 | |
| A versus B | <0.0001 | NS | <0.0001 | 0.0002 | NS | 0.0273 | NS | NS | NS | |
| A versus C | <0.0001 | <0.0001 | <0.0001 | <0.0001 | NS | 0.0134 | NS | 0.0376 | 0.0015 | |
| B versus C | <0.0001 | 0.0017 | <0.0001 | <0.0001 | 0.0489 | NS | 0.0058 | 0.0008 | 0.0244 | |
Note: Group A comprehends patients with scores 5, 6, and 7. Group B comprehends patients with score 8. Group C comprehends patients with scores 9 and 10. Data are expressed as absolute numbers and mean ± SD.
Abbreviations: ANOVA, analysis of variance; CI, cardiac index; mPAP, mean pulmonary arterial pressure; NS, nonsignificant; PETCO2, end‐tidal carbon dioxide pressure; pulse O2, oxygen pulse; PVR, pulmonary vascular resistance; RAP: mean right atrial pressure; VO2, oxygen uptake; VE/VCO2 slope, ventilation to carbon dioxide production slope; VO2/work, oxygen uptake to work slope.
DISCUSSION
Our study suggests that CPET can be supportive in the noninvasive stratification of hemodynamic impairment in PAH patients, but mainly in subjects with mild‐to‐moderate hemodynamic impairment.
The invasive evaluation of pulmonary hemodynamics by RHC provides useful information for risk stratification and prognosis at baseline and during follow‐up. 3 Mean PAP values have been traditionally used to define the hemodynamic severity of pulmonary hypertension, while CI, RAP, and PVR provide further insights on the hemodynamic impairment of PAH patients. Indeed, the prognostic role of pulmonary pressure was demonstrated more than three decades ago in the first registry of primary pulmonary hypertension by the United States National Institutes of Health (NIH). 19 Moreover, CI and PVR have been recently included in the United States Registry to Evaluate Early and Long‐Term PAH Disease Management registry (REVEAL) score, a valuable multiparametric tool increasingly used in clinical practice for the risk stratification of PAH patients, 20 while CI and RAP are currently included among the invasive markers suggested by European guidelines for risk stratification in PAH. 3
However, the periodic reassessment of PAH patients includes a multiparametric evaluation, and RHC, which usually requires hospitalization, may be frequently postponed due to center's facilities availability, patients' clinical condition and willingness, as well as economic resources. Further significant restrictions on routine medical care have been recently caused by the coronavirus disease‐19 pandemic to comply with public health guidance on public exposure and to help preserve or redirect limited resources, with potential negative impact on PAH patients' outcome.
CPET is currently considered the gold standard for assessing the degree and causes of exercise intolerance. 4 As effort dyspnea represents one of the main clinical features of PAH, the assessment of exercise capacity in PAH patients has gained a major role not only in the diagnostic phase, 7 but also at follow‐up for risk stratification and in assessing the response to treatment. 4 Indeed, several CPET parameters have shown prognostic value in PAH patients, above all peak VO2 and VE/VCO2 slope, but also O2 pulse at peak exercise. 7 , 8 , 9 , 21 , 22
In PAH, physical effort is characterized by a blunted CO increase and marked hyperventilation. There are two causes of hyperventilation in PAH: dead space increase and chemoreflex induced hyperventilation. The former is associated with a normal PaCO2 value, the latter with a reduced PaCO2 value. 18 Blunted CO increase during exercise leads to low peak VO2, peak O2 pulse and VO2/work slope, while hyperventilation leads to a typical pattern characterized by an increase of VE/VCO2 slope and ventilatory equivalents of CO2 and O2, and by a reduction in PETCO2. 5 These CPET features have been shown to be tightly associated with the hemodynamic impairment observed in PAH, resulting in a significant inverse correlation of both mPAP and PVR with peak VO2, and a direct correlation with VE/VCO2. 14 , 23 Specifically, the relationship between the ventilatory equivalent of CO2 and PETCO2 predicts the probability of pulmonary hypertension in subjects with dyspnea of unknown etiology. 7 , 8 Recently, Zhao et al. applied a CPET score combining VE/VCO2 slope and AT to improve the specificity of echocardiography in patients with suspected pulmonary hypertension undergoing RHC. 24
In the present study, we investigated the association between CPET parameters and the degree of hemodynamic impairment evaluated at RHC in PAH patients. The rationale for the reported measurements was strong, as mPAP and PVR reflects right ventricular (RV) afterload, CI reflects RV contractility adaptation to increased loading, and RAP reflects failure of this mechanism resulting in increased filling pressures.
Our results are the demonstration that the measure of CPET variables, such as peak VO2, VE/VCO2 slope, and peak PETCO2, and their physiologically meaningful combination are of clinical relevance for the assessment of RV hemodynamic burden. Peak VO2 is indeed a robust though load‐dependent measure of CO, and VE/VCO2 slope and peak PETCO2 reflect ventilation regulation. In severe pulmonary hypertension, the right ventricle adapts by increasing contractility to preserve CO. Thus, peak VO2 reflects the hemodynamic adaptation of the right ventricle, while increased VE/VCO2 slope and decreased PETCO2 reflect ventilation‐perfusion mismatch and ergo‐reflex activity increase.
Taken individually (Table 2), all analyzed CPET parameters except for VO2/work slope show significant differences in the tertiles of hemodynamic parameters: VE/VCO2 slope and PETCO2 at peak exercise for mPAP and PVR, peak VO2 and O2 pulse for all four hemodynamic parameters. Thus, CPET markers of PAH hyperventilation seem to be mainly related to the degree of afterload increase, rather than to hemodynamic indices of RV function, while CPET markers of cardiac performance are also related to RHC indices of RV function.
More interesting results emerge if we consider some combinations of parameters. Peak PETCO2–VE/VCO2 slope relationship (Figure 1, Table 3) seems useful to stratify patients according to mPAP, PVR, CI, and RAP values, revealing that PAH patients with VE/VCO2 slope under the median value and PETCO2 above the median value—that is, with a smaller effort ventilation impairment—are most likely to have lower afterload burden and filling pressure, and higher CI. Conversely, subjects with a greater effort ventilation impairment (VE/VCO2 slope above the median value and PETCO2 under the median value) are most likely to present with higher afterload increase and filling pressure, and lower CI. Statistical significance was reached only for mPAP and PVR, confirming the great impact of afterload on effort ventilatory impairment in PAH. Of note, the median value of VE/VCO2 slope of our study population (44) is close to the cut‐off value (45) proposed by European guidelines 3 for the definition of high‐risk patients according to CPET evaluation.
Finally, we sought to correlate CPET to a score including multiple RHC parameters indicative of the degree of PAH patients' global hemodynamic impairment. Multivariate analysis found peak VO2 and peak PETCO2 as independent predictors of the score, and regression provided an equation that allows us to predict the hemodynamic impairment score for each patient starting from these two noninvasive CPET‐derived measurements. These parameters are highly significant from both a pathophysiological and a clinical point of view, as they are indicative of two major effort alterations of the disease, that is, low CO and altered exercise ventilation; moreover, peak VO2 is the main CPET prognostic parameter for PAH patients.
Compared to the real score, the CPET‐derived hemodynamic impairment score shows some over/underestimation for low and high scores, respectively, and a good match for intermediate hemodynamic burden. Thus, the estimated score presented in this report seems supportive in noninvasive evaluation, particularly for patients with moderate hemodynamic impairment, who are both the most frequently observed PAH population and the population in which a properly tailored treatment has more efficacy.
Study limitations
This study has some relevant limitations that need to be acknowledged. First, it is a retrospective study. However, we believe that its results may be convincing since the study was multicentric with standardized hemodynamic and CPET assessments and results were analyzed with rigorous statistics. Second, we studied Group 1 PAH patients excluding patients with PAH associated to congenital heart disease. However, the great majority of cases were patients with idiopathic PAH and our results may not apply to all types of PAH. Third, the hemodynamic score we used to classify the severity of the hemodynamic burden has not been validated as a prognostic tool. It is built considering the tertiles of a few hemodynamic parameters recorded in the present population. The parameters were arbitrarily chosen but are all associated to prognosis in PAH albeit with a different power. Accordingly, this score should not be used as a prognostic tool but simply as a marker of the hemodynamic burden in PAH patients. Fourth, changes over time of CPET and RHC data were not assessed so that we do not know whether CPET allows to identify hemodynamic changes when they occur. Finally, whether CPET data in combination with other noninvasive measurements further improves CPET capability to predict the hemodynamic burden was not assessed.
CONCLUSIONS
The results of our study show that CPET could be a potential noninvasive tool for the assessment of hemodynamic burden in PAH patients with mild‐to‐moderate hemodynamic impairment, that could be considered as an alternative to invasive RHC during follow‐up in selected patients. Further investigation is needed to confirm and possibly strengthen our results in a larger prospective population, also including serial evaluation during follow‐up to assess the prognostic impact of this noninvasive tool over time.
AUTHOR CONTRIBUTORS
Each author of this paper has made substantial contributions to conception and design, acquisition, analysis, and interpretation of data; has drafted the submitted article or revised it critically for important intellectual content; has read the manuscript and provided final approval of the version to be published; has participated sufficiently in the work to take public responsibility for appropriate portions of the content.
CONFLICTS OF INTEREST
P. Agostoni reports grants and/or financial support from Bayer and Actelion; R. Badagliacca reports fees from United Therapeutics, Dompè, Ferrer, Bayer, Merck Sharp & Dohme, and OP Orphan Pharmaceuticals AG. The other authors: nothing to disclose.
ETHICS STATEMENT
As reported in the study protocol section, the investigation was approved by the Centro Cardiologico Monzino IRCCS Scientific Committee and notified, due to its retrospective nature and the anonymous use of medical data (CCM ‐ PR182), to the Centro Cardiologico Monzino IRCCS Ethics Committee. All participants signed an informed consent for both RHC and CPET. The study was conducted in compliance with the declaration of Helsinki.
Pezzuto B, Badagliacca R, Muratori M, Farina S, Bussotti M, Correale M, Bonomi A, Vignati C, Sciomer S, Papa S, Palazzo Adriano E, Agostoni P. Role of cardiopulmonary exercise test in the prediction of hemodynamic impairment in patients with pulmonary arterial hypertension. Pulmonary Circulation. 2022;12:e12044. 10.1002/pul2.12044
REFERENCES
- 1. Humbert M, Guignabert C, Bonnet S, Dorfmüller P, Klinger JR, Nicolls MR, Olschewski AJ, Pullamsetti SS, Schermuly RT, Stenmark KR, Rabinovitch M. Pathology and pathobiology of pulmonary hypertension: state of the art and research perspectives. Eur Respir J. 2019;53(1):1801887. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. McLaughlin VV, McGoon MD. Pulmonary arterial hypertension. Circulation. 2006;114(13):1417–31. [DOI] [PubMed] [Google Scholar]
- 3. Galiè N, Humbert M, Vachiery J‐L, Gibbs S, Lang I, Torbicki A, Simonneau G, Peacock A, Vonk Noordegraaf A, Beghetti M, Ghofrani A, Gomez Sanchez MA, Hansmann G, Klepetko W, Lancellotti P, Matucci M, McDonagh T, Pierard LA, Trindade PT, Zompatori M, Hoeper M, ESC Scientific Document Group. 2015 ESC/ERS Guidelines for the diagnosis and treatment of pulmonary hypertension. Eur Heart J. 2016;37(1):67–119. [DOI] [PubMed] [Google Scholar]
- 4. Balady GJ, Arena R, Sietsema K, Myers J, Coke L, Fletcher GF, Forman D, Franklin B, Guazzi M, Gulati M, Keteyian SJ, Lavie CJ, Macko R, Mancini D, Milani RV, American Heart Association Exercise, Cardiac Rehabilitation, and Prevention Committee of the Council on Clinical Cardiology, Council on Epidemiology and Prevention P, Council on Peripheral Vascular Disease D, Interdisciplinary Council on Quality of Care and Outcomes Research. Clinician's guide to cardiopulmonary exercise testing in adults: a scientific statement from the American Heart Association. Circulation. 2010;122(2):191–225. [DOI] [PubMed] [Google Scholar]
- 5. Sun XG, Hansen JE, Oudiz RJ, Wasserman K. Exercise pathophysiology in patients with primary pulmonary hypertension. Circulation. 2001;104(4):429–35. [DOI] [PubMed] [Google Scholar]
- 6. Farina S, Correale M, Bruno N, Paolillo S, Salvioni E, Badagliacca R, Agostoni P, “Right and Left Heart Failure Study Group” of the Italian Society of Cardiology. The role of cardiopulmonary exercise tests in pulmonary arterial hypertension. Eur Respir Rev. 2018;27(148):170134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Yasunobu Y, Oudiz RJ, Sun XG, Hansen JE, Wasserman K. End‐tidal PCO2 abnormality and exercise limitation in patients with primary pulmonary hypertension. Chest. 2005;127(5):1637–46. [DOI] [PubMed] [Google Scholar]
- 8. Ferrazza AM, Martolini D, Valli G, Palange P. Cardiopulmonary exercise testing in the functional and prognostic evaluation of patients with pulmonary diseases. Respiration. 2009;77(1):3–17. [DOI] [PubMed] [Google Scholar]
- 9. Deboeck G, Scoditti C, Huez S, Vachiéry JL, Lamotte M, Sharples L, Melot C, Naeije R. Exercise testing to predict outcome in idiopathic versus associated pulmonary arterial hypertension. Eur Respir J. 2012;40(6):1410–19. [DOI] [PubMed] [Google Scholar]
- 10. Wensel R, Francis DP, Meyer FJ, Opitz CF, Bruch L, Halank M, Winkler J, Seyfarth HJ, Gläser S, Blumberg F, Obst A, Dandel M, Hetzer R, Ewert R. Incremental prognostic value of cardiopulmonary exercise testing and resting haemodynamics in pulmonary arterial hypertension. Int J Cardiol. 2013;167(4):1193–8. [DOI] [PubMed] [Google Scholar]
- 11. Ferreira EV, Ota‐Arakaki JS, Ramos RP, Barbosa PB, Almeida M, Treptow EC, Valois FM, Nery LE, Neder JA. Optimizing the evaluation of excess exercise ventilation for prognosis assessment in pulmonary arterial hypertension. Eur J Prev Cardiol. 2014;21(11):1409–19. [DOI] [PubMed] [Google Scholar]
- 12. Badagliacca R, Papa S, Valli G, Pezzuto B, Poscia R, Reali M, Manzi G, Giannetta E, Berardi D, Sciomer S, Palange P, Fedele F, Naeije R, Vizza CD. Right ventricular dyssynchrony and exercise capacity in idiopathic pulmonary arterial hypertension. Eur Respir J. 2017;49(6):1601419. [DOI] [PubMed] [Google Scholar]
- 13. Paolillo S, Farina S, Bussotti M, Iorio A, Filardi PP, Piepoli MF, Agostoni P. Exercise testing in the clinical management of patients affected by pulmonary arterial hypertension. Eur J Prev Cardiol. 2012;19(5):960–71. [DOI] [PubMed] [Google Scholar]
- 14. Correale M, Tricarico L, Ferraretti A, Monaco I, Concilio M, Padovano G, Acanfora G, Di Biase M, Brunetti ND. Cardiopulmonary exercise test predicts right heart catheterization. Eur J Clin Invest. 2017;47(12):e12851. [DOI] [PubMed] [Google Scholar]
- 15. Agostoni P, Bianchi M, Moraschi A, Palermo P, Cattadori G, La Gioia R, Bussotti M, Wasserman K. Work‐rate affects cardiopulmonary exercise test results in heart failure. Eur J Heart Fail. 2005;7(4):498–504. [DOI] [PubMed] [Google Scholar]
- 16. Agostoni P, Dumitrescu D. How to perform and report a cardiopulmonary exercise test in patients with chronic heart failure. Int J Cardiol. 2019;288:107–13. [DOI] [PubMed] [Google Scholar]
- 17. Beaver WL, Wasserman K, Whipp BJ. A new method for detecting anaerobic threshold by gas exchange. J Appl Physiol (1985). 1986;60(6):2020–27. [DOI] [PubMed] [Google Scholar]
- 18. Farina S, Bruno N, Agalbato C, Contini M, Cassandro R, Elia D, Harari S, Agostoni P. Physiological insights of exercise hyperventilation in arterial and chronic thromboembolic pulmonary hypertension. Int J Cardiol. 2018;259:178–82. [DOI] [PubMed] [Google Scholar]
- 19. D'Alonzo GE. Survival in patients with primary pulmonary hypertension. Results from a national prospective registry. Ann Intern Med. 1991;115(5):343–9. [DOI] [PubMed] [Google Scholar]
- 20. Benza RL, Miller DP, Gomberg‐Maitland M, Frantz RP, Foreman AJ, Coffey CS, Barst RJ, Badesch DB, Elliot CG, Liou TG, McGoon MD. Predicting survival in pulmonary arterial hypertension: insights from the Registry to Evaluate Early and Long‐Term Pulmonary Arterial Hypertension Disease Management (REVEAL). Circulation. 2010;122(2):164–72. [DOI] [PubMed] [Google Scholar]
- 21. Badagliacca R, Papa S, Valli G, Pezzuto B, Poscia R, Manzi G, Giannetta E, Sciomer S, Palange P, Naeije R, Fedele F, Vizza CD. Echocardiography combined with cardiopulmonary exercise testing for the prediction of outcome in idiopathic pulmonary arterial hypertension. Chest. 2016;150(6):1313–22. [DOI] [PubMed] [Google Scholar]
- 22. Badagliacca R, Papa S, Poscia R, Valli G, Pezzuto B, Manzi G, Torre R, Gianfrilli D, Sciomer S, Palange P, Naeije R, Fedele F, Vizza CD. The added value of cardiopulmonary exercise testing in the follow‐up of pulmonary arterial hypertension. J Heart Lung Transplant. 2019;38(3):306–14. [DOI] [PubMed] [Google Scholar]
- 23. Nishio R, Tanaka H, Tsuboi Y, Kinutani H, Taniguchi Y, Shigeru M, Toh R, Miura Y, Sakai Y, Emoto N, Kawai H, Hirata K. Differences in hemodynamic parameters and exercise capacity between patients with pulmonary arterial hypertension and chronic heart failure. J Cardiopulm Rehabil Prev. 2012;32(6):379–85. [DOI] [PubMed] [Google Scholar]
- 24. Zhao QH, Wang L, Pudasaini B, Jiang R, Yuan P, Gong SG, Guo J, Xiao Q, Liu H, Wu C, Jing ZC, Liu JM. Cardiopulmonary exercise testing improves diagnostic specificity in patients with echocardiography‐suspected pulmonary hypertension. Clin Cardiol. 2017;40(2):95–101. [DOI] [PMC free article] [PubMed] [Google Scholar]


