Abstract
Cervical and trigeminal afferents innervate neighboring cranial territories, and their convergence on upper cervical dorsal horn neurons provides a potential substrate for pain referral in primary headache syndromes. Lamina I neurons are central to this mechanism, as they relay convergent nociceptive input to supraspinal pain centers. Unfortunately, little is known about the interactions between trigeminal and cervical afferents supplying Lamina I neurons. Here, we used rats of both sexes to show that cervical and trigeminal afferents interact via presynaptic inhibition, where monosynaptic inputs to Lamina I neurons undergo unidirectional as well as reciprocal presynaptic control. This means that afferent-driven presynaptic inhibition shapes the way trigeminal and cervical Aδ-fiber and C-fiber input reaches Lamina I projection neurons (PNs) and local-circuit neurons (LCNs). We propose that this inhibition provides a feedforward control of excitatory drive to Lamina I neurons that regulates their convergent and cervical-specific or trigeminal-specific processing modes. As a consequence, disruption of the trigeminal and cervical afferent-driven presynaptic inhibition may contribute to development of primary headache syndromes.
SIGNIFICANCE STATEMENT Cervical and trigeminal afferents innervate neighboring cranial territories, and their convergence on upper cervical dorsal horn neurons provides a potential substrate for pain referral in primary headache syndromes. Lamina I neurons are central to this mechanism as they relay convergent nociceptive input to supraspinal pain centers. Here, we show that cervical and trigeminal afferents interact via presynaptic inhibition, where inputs to Lamina I neurons undergo unidirectional as well as reciprocal control. The afferent-driven presynaptic inhibition shapes the trigeminocervical Aδ-fiber and C-fiber input to Lamina I neurons. This inhibition provides control of excitatory drive to Lamina I neurons that regulates their convergent and cervical-specific or trigeminal-specific processing modes. Disruption of this control may contribute to development of primary headache syndromes.
Keywords: C2 spinal nerve, dorsal root potentials, nociceptive afferents, presynaptic inhibition, trigeminal nerve, trigeminocervical complex
Introduction
The C2 spinal nerve (C2 SN) and trigeminal nerve (TN) innervate neighboring cranial territories that are involved in the pain associated with primary headache syndromes, such as migraine, cluster headache or tension-type headache (Bartsch and Goadsby, 2003a; Goadsby et al., 2017). Nociceptive C2 SN afferents supplying the back of the head and neck (Bogduk, 2001; Kemp et al., 2011) terminate in the superficial dorsal horn of C1–C3 cervical spinal segments (Escolar, 1948; Kerr, 1961; Pfaller and Arvidsson, 1988; García-Magro et al., 2018). Likewise, nociceptive TN afferents innervating the front of the head (i.e., facial skin, cranial dura mater or blood vessels; Kemp et al., 2012; Edvinsson et al., 2020) enter the brainstem at the level of the pons and terminate in the trigeminal nucleus caudalis and upper cervical dorsal horn (Kerr, 1961; Pfaller and Arvidsson, 1988; Marfurt and Rajchert, 1991; Luz et al., 2019).
These cervical and trigeminal afferents converge on dorsal horn neurons (Kerr, 1961; Pfaller and Arvidsson, 1988; Bartsch and Goadsby, 2003b; Fernandes et al., 2022) and this is thought to explain why pain in migraine and cluster headache is often perceived in the territories of both nerves. We have recently shown that Lamina I plays an important role in this trigeminocervical integration (Fernandes et al., 2022). Specifically, upper cervical Lamina I projection neurons (PNs) and local-circuit neurons (LCNs) receive diverse combinations of direct and polysynaptic inputs from cervical and trigeminal Aδ-fibers and C-fibers.
The processing of information carried on trigeminal and cervical afferents is complex. In addition to making contacts onto neurons in Lamina I, they also receive presynaptic inhibition from spinal interneurons. Indeed, presynaptic inhibition of primary afferents has been shown as powerful mechanism for spinal sensory control (Melzack and Wall, 1965; Rudomin and Schmidt, 1999; Willis, 1999; Boyle et al., 2019). Under this mechanism, axoaxonic synapses originating from spinal interneurons contact the central terminals of primary afferents. Release of GABA at these sites blocks invasion of action potentials or reduces their amplitude, which ultimately diminishes the likelihood of glutamate release at the primary afferent terminals (Segev, 1990; Graham and Redman, 1994; Thanawala and Regehr, 2013; Gradwell et al., 2020). Unfortunately, little is known about the presynaptic interactions between trigeminal and cervical afferents supplying upper cervical neurons.
GABAergic presynaptic inhibition is caused by a phenomena termed primary afferent depolarization (PAD) induced by Cl– efflux through anion channels of GABAA receptors (Alvarez-Leefmans et al., 1998; Rudomin and Schmidt, 1999; Willis, 1999). The antidromic spread of PAD along the afferent fiber to the dorsal root can be recorded as a dorsal root potential (DRP; Barron and Matthews, 1938; Lloyd and McIntyre, 1949). The afferent-driven presynaptic inhibition was first shown for large diameter muscle afferents and low-threshold cutaneous afferents (Koketsu, 1956; Eccles, 1964; Rudomin and Schmidt, 1999; Willis, 1999; Hughes et al., 2005, 2012; Fink et al., 2014; Boyle et al., 2019). Recently, this type of inhibition has also been described for small diameter afferents, specifically, unmyelinated C-afferents supplying Lamina I neurons in the lumbar cord (Fernandes et al., 2020). The lumbar C-afferents are subject to a complex pattern of presynaptic inhibition mediated by homosegmental and heterosegmental Aβ-afferents, Aδ-afferents, and C-afferents. This finding rises the possibility that cervical and trigeminal afferents supplying Lamina I neurons in the upper cervical cord are also under reciprocal control. Such afferent-driven presynaptic inhibition could shape the trigeminocervical nociceptive information flow to cervical cord and its alterations may cause primary headache syndromes. Accordingly, we ask whether cervical and trigeminal afferents inhibit each other via presynaptic mechanisms and whether this form of inhibition shapes nociceptive input to upper cervical Lamina I.
Materials and Methods
Ethical approval
Laboratory Wistar rats (at postnatal days 13–18, both sexes) were killed by decapitation in accordance with Portuguese national guidelines (Direcção Geral de Alimentação e Veterinária, Ministério da Agricultura) after anesthesia with Na+-pentobarbital (30 mg/kg, i.p.) and subsequent monitoring for lack of pedal withdrawal reflexes. The experiments were conducted according to the guidelines laid down by the institution's animal welfare committee (Comissão de Ética do Instituto de Biologia Molecular e Celular).
Ex vivo brainstem-cervical cord preparation
A tissue block comprising the brainstem and upper cervical spinal cord with the C2 SN and TN preserved was isolated in oxygenated artificial cerebrospinal fluid at room temperature (Luz et al., 2019). Briefly, the skull was opened and the cerebrum and cerebellum were exposed. The vertebral laminae and the part of the occipital bone at the spino-medullary junction were cut. The distal end of the TN was cut close to the trigeminal ganglion. The remaining cranial nerves were cut close to where they emerged from the brainstem. The spinal cord was transected at C5 and the C2 dorsal root was carefully dissected along with its ganglion, SN, dorsal ramus, and its major peripheral branch, the greater occipital nerve. The remaining dorsal and ventral roots were cut close to the cord. The pia mater over the upper cervical region was removed with forceps and fine scissors, to provide access for recording pipettes. The tissue block was glued (dorsolateral surface up) with cyanoacrylate adhesive to a plate made of gold and then transferred to the recording chamber. All measurements were made at 22–24°C to better preserve neural network viability (Szucs et al., 2009). Lamina I neurons were visualized in the region between the dorsolateral funiculus and the dorsal root entry zone (Pinto et al., 2010) using the oblique infrared light-emitting-diode (OSRAM, SFH 4550, 850 nm) illumination technique (Safronov et al., 2007; Szucs et al., 2009). Using this imaging technique, Lamina I neurons can be clearly distinguished from deeper Lamina II neurons because their larger somata are more loosely packed (Szucs et al., 2009).
Recording
Whole-cell recordings were obtained from neurons located in cervical segments C1–C3. Artificial cerebrospinal fluid contained (in mm): 115 NaCl, 3 KCl, 2 CaCl2, 1 MgCl2, 1 NaH2PO4, 25 NaHCO3, and 11 glucose (bubbled with 95% O2/5% CO2). Recording pipettes were pulled from thick-walled glass (BioMedical Instruments) and fire-polished (resistance, 4–6 MΩ). The pipette solution contained (in mm): 3 KCl, 150 K-gluconate, 1 MgCl2, 1 BAPTA, and 10 HEPES (pH 7.3 adjusted with KOH). The final [K+] was 160 mm and 0.5% biocytin was added to the internal solution for post hoc visualization of recorded neurons. Unless otherwise stated, all chemicals were obtained from Sigma-Aldrich. Recordings were made with a EPC10-Double amplifier (HEKA). Signals were sampled at 10 kHz and low-pass filtered at 2.9 kHz. Offset potentials were compensated before seal formation. Liquid junction potentials were calculated and corrected for using the compensation circuitry within the amplifier.
Primary afferent inputs were evoked by stimulating the ipsilateral C2 SN and TN after the nerves had been carefully drawn into suction electrodes as described previously (Pinto et al., 2008, 2010). Stimulation was delivered by isolated pulse stimulators (2100, AM Systems). A square pulse (600 µA, 1-ms duration) was applied to the TN to recruit all Aβδ-fibers and C-fibers (Luz et al., 2019). A smaller pulse (200 µA, 1-ms duration) was found to be sufficient to recruit all Aβδ-fibers and C-fibers in the smaller diameter C2 SN. Monosynaptic EPSCs were identified on the basis of their low failure rates and small latency variations as described previously (Luz et al., 2010). The afferent conduction velocity (CV) was calculated by dividing the conduction distance by conduction time. The former included the length of the peripheral pathway, from the opening of the suction electrode to the entry zone, and the estimated central pathway, in the brainstem and spinal cord. The central pathway was determined as the rostrocaudal distance between the cell body and the TN or C2 dorsal root entry zone. The conduction time was calculated for a monosynaptic EPSC from its latency, after allowing 1 ms for synaptic transmission delay. Fibers mediating monosynaptic EPSCs were classified according to their CV. Fibers conducting at CVs > 0.45 m/s for the C2 SN and > 0.49 m/s for the TN were considered as Aδ-afferents, and those conducting at lower CVs as C-afferents. To study induction of PAD, DRPs, and trigeminal DRPs (TDRPs) were recorded via suction electrodes positioned near the C2 dorsal root and TN entry zones (Fig. 1A,B). The DRP and TDRP recordings were done in a hemisected preparation. Each suction electrode had its own reference electrode. We used the differential AC amplifier (1700, AM Systems) where the low cutoff filter was open to allow the DC recording. The signal was on-line low-pass filtered at 10 kHz and sampled using the A/D converter of the patch-clamp amplifier. Off-line filtering was conducted at 500 Hz. The DRPs and TDRPs were evoked at 10-s intervals (Fernandes et al., 2020).
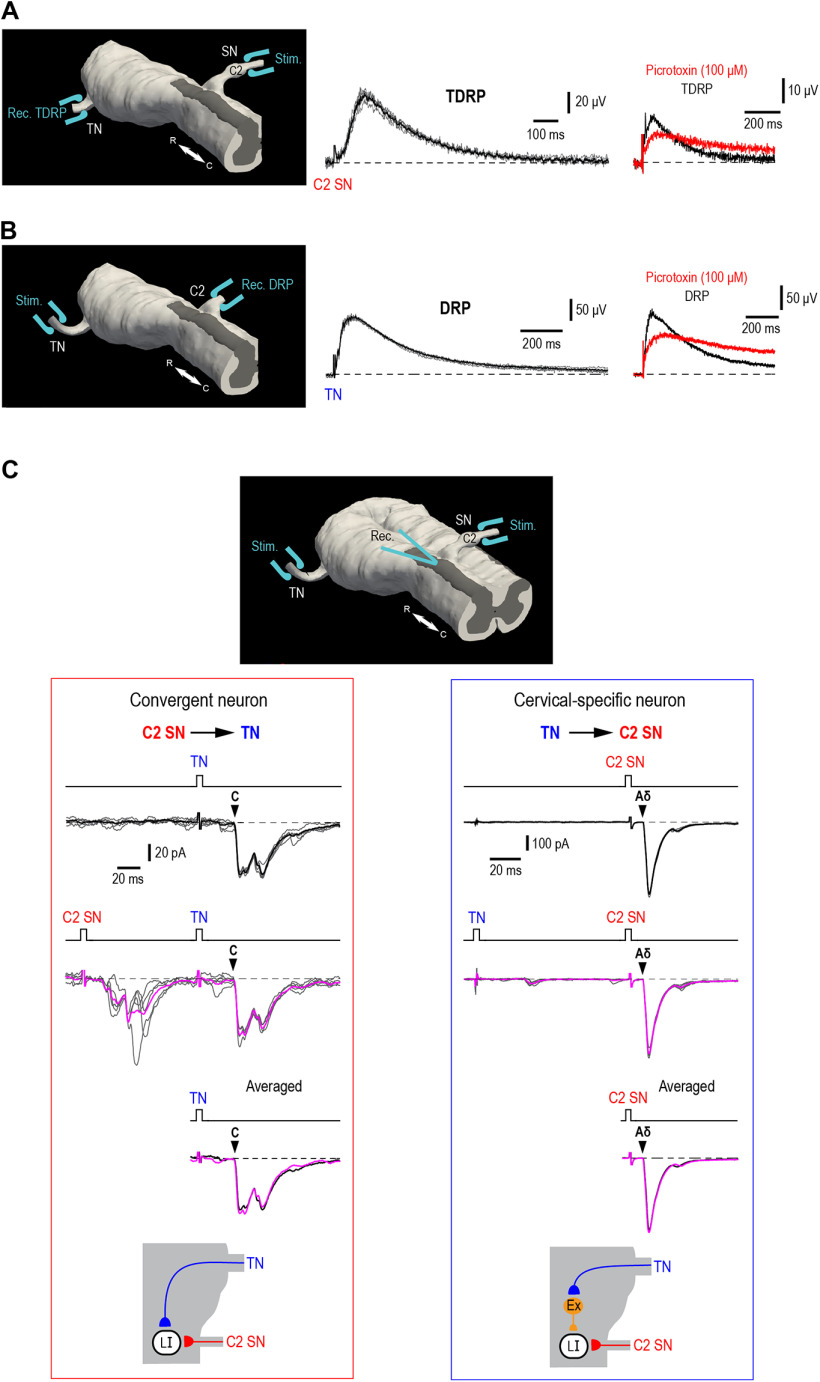
Figure 1.
Recordings in the ex vivo brainstem-cervical cord preparation. A, Left panel, Schematic of the preparation used for recording TDRPs. The TDRPs were evoked by stimulating the C2 SN. The recording electrode was positioned on the ipsilateral trigeminal dorsal root close to where it entered the brainstem. Note, the C2 SN has been reflected toward the contralateral side. Right, Effect of picrotoxin on the TDRP. B, C2 DRPs were evoked by stimulating the TN. The recording electrode was positioned where the C2 root entered the spinal cord. Right, Effect of picrotoxin on the DRP. Both TDRPs and DRPs were studied in the hemisected preparation. C, Recording of ipsilateral cervical and trigeminal afferent inputs to the upper cervical Lamina I neurons in the isolated brainstem-cervical cord preparation. Note, the C2 SN has been reflected toward the contralateral side to gain access to Lamina I neurons. Left panel, Example responses (5 traces with their average) in a convergent Lamina I (LI) neuron (cell 267). In this neuron, C2 SN conditioning did not affect the monosynaptic C-fiber input evoked by stimulation of the TN. Holding potential, −80 mV. Averaged superimposed traces are shown below. The schematic at the bottom of the panel illustrates two monosynaptic inputs, which are not affected by presynaptic inhibition. Filled arrowheads indicate monosynaptic components for the EPSCs evoked by the test stimuli. Right panel, Example responses in a cervical-specific neuron (cell 33). In this neuron, TN conditioning did not affect the monosynaptic Aδ-fiber EPSC evoked by stimulation of the C2 SN. Note that the TN contacts the recorded neuron via a polysynaptic excitatory input. Holding potential, −85 mV. R, rostral; C, caudal.
Paired pulse protocol was used to study the afferent-driven presynaptic inhibition of cervical and trigeminal inputs to Lamina I neurons (Fernandes et al., 2020). Degree of presynaptic inhibition was judged from the reduction in the amplitude of the monosynaptic EPSC evoked by the test stimulation of one nerve after the conditioning stimulation of the other nerve. The interval between these two stimuli was set to 100 ms based on our recordings of TDRP and DRP. The input was considered as affected by the presynaptic inhibition if at least one component of the monosynaptic EPSC was reduced by the conditioning stimulus.
Several precautions were taken to ensure that the EPSC reduction resulted from the afferent-driven presynaptic inhibition rather than postsynaptic feedforward inhibition. The analysis of presynaptic inhibition was done in the voltage-clamp mode at potentials near the Cl– equilibrium potential to maximally reduce IPSCs and their effect on evoked EPSCs. Since voltage-clamped currents (EPSCs and IPSCs) are additive, small remaining IPSCs could only shift the baseline but did not affect the amplitude of the monosynaptic EPSCs. We avoided using current-clamp, in which “invisible” at their reversal potential IPSPs could produce a drop in the membrane input resistance, and this way, shunt or partially reduce the monosynaptic EPSPs. Thus, analysis of effects on the monosynaptic EPSCs represents an adequate approach to document afferent-driven presynaptic inhibition of primary afferent input.
A component of monosynaptic input was considered as affected by the presynaptic inhibition if its amplitude was reduced by >10% and >5 pA after application of conditioning stimulus. This way, effect on small EPSCs (<50 pA) could be underestimated. Monosynaptic inputs reduced by 19–62% (Table 1) were classified as partially inhibited. Complete abolishment of the component was considered as its full presynaptic block. To estimate effect on the network, we did integration of the total inward, monosynaptic and polysynaptic, currents. In some cases, a clear reduction of the inward current was seen, but the integration could not be done, for example because of uncontrolled discharge of voltage-gated Na+ current (these cases are indicated in Table 1).
Table 1.
Afferent-driven presynaptic inhibition of Lamina I neurons
| Cell | Type | C2 SN → TN |
TN → C2 SN |
Reciprocal (↔) or Unidirectional (→) | ||
|---|---|---|---|---|---|---|
| Mono | Network | Mono | Network | |||
| 25 | Convergent | AδF | — | n.t. | n.t. | |
| 32 | Convergent | — | + | — | 45.5% | C2 SN ↔ TN |
| 35 | Convergent* | Aδ23% | — | n.t. | n.t. | |
| 62 | Convergent | C23% | 47.6% | n.t. | n.t. | |
| 63 | Convergent | CF | 53.3% | — | — | C2 SN → TN |
| 70 | Convergent | Aδ22% | — | n.t. | n.t. | |
| 78 | Convergent | AδF | + | n.t. | n.t. | |
| 81 | Convergent | AδF | — | — | — | C2 SN → TN |
| 83 | Convergent | C26% | — | n.t. | n.t. | |
| 97 | Convergent PN* | Aδ34% | — | — | 16.9% | C2 SN ↔ TN |
| 98 | Convergent PN | CF | — | — | 44.7% | C2 SN ↔ TN |
| 100 | Convergent PN | n.t. | C26% | — | n.t. | |
| 102 | Convergent LCN | — | — | AδF | — | TN → C2 SN |
| 103 | Convergent | — | — | Aδ51%, CF | — | TN → C2 SN |
| 110 | Convergent* | CF | — | — | — | C2 SN → TN |
| 118 | Convergent | — | — | — | 31.0% | TN → C2 SN |
| 119 | Convergent | Aδ55% | — | — | — | C2 SN → TN |
| 170 | Convergent | AδF, Aδ27% | — | Aδ19% | — | C2 SN ↔ TN |
| 178 | Convergent PN | CF | — | — | 42.6% | C2 SN ↔ TN |
| 179 | Convergent | CF | — | — | — | C2 SN → TN |
| 182 | Convergent | n.t. | Aδ60% | 60.8% | n.t | |
| 185 | Convergent PN* | Aδ19% | — | — | — | C2 SN → TN |
| 198 | Convergent | n.t. | — | 33.2% | n.t. | |
| 203 | Convergent PN | n.t. | C23% | — | n.t. | |
| 215 | Convergent* | C36% | — | — | 20.6% | C2 SN ↔ TN |
| 255 | Convergent | n.t. | CF | — | n.t. | |
| 256 | Convergent | CF | 24.7% | CF | + | C2 SN ↔ TN |
| 262 | Convergent | — | — | CF | + | TN → C2 SN |
| 266 | Convergent* | CF | 70.5% | CF | 58.1% | C2 SN ↔ TN |
| 52 | Trigeminal-sp | — | — | — | ++ | TN → C2 SN |
| 80 | Trigeminal-sp | Aδ50% | — | n.t. | n.t. | |
| 251 | Trigeminal-sp* | C50% | 30.6% | n.t. | n.t. | |
| 47 | Cervical-sp | n.t. | CF | — | n.t. | |
| 55 | Cervical-sp | — | ++ | n.t. | n.t. | |
| 56 | Cervical-sp | — | ++ | CF | — | C2 SN ↔ TN |
| 58 | Cervical-sp* | — | 46.3% | — | 25.3% | C2 SN ↔ TN |
| 87 | Cervical-sp | — | ++ | — | — | C2 SN → TN |
| 99 | Cervical-sp PN | — | ++ | — | + | C2 SN ↔ TN |
| 111 | Cervical-sp LCN | — | — | Aδ31% | — | TN → C2 SN |
| 129 | Cervical-sp | n.t. | C62% | — | n.t. | |
| 132 | Cervical-sp | — | ++ | — | — | C2 SN → TN |
| 191 | Cervical-sp | n.t. | CF | — | n.t. | |
| 225 | Cervical-sp PN* | — | ++ | — | — | C2 SN → TN |
| 228 | Cervical-sp | — | — | — | 31.2% | TN → C2 SN |
| 253 | Cervical-sp | n.t. | — | 23.9% | n.t. | |
| 260 | Cervical-sp | — | 17.0% | CF | 45.5% | C2 SN ↔ TN |
This table describes the upper cervical Lamina I neurons (n = 46) affected by presynaptic inhibition. All these neurons were first tested for their monosynaptic inputs from the C2 SN and TN and classified as convergent, trigeminal-specific, or cervical-specific. C2 SN → TN, effect of the C2 SN conditioning on the TN input; TN → C2 SN, effect of the TN conditioning on the C2 SN input. Mono, effect on the monosynaptic component; F, full inhibition; a partial inhibition is described in percentage by which the component was reduced. Network, reduction in the integral of inward current containing monosynaptic and/or polysynaptic components is given in percentage; +, reduced polysynaptic EPSC that could not be integrated; ++, a full block of the input consisting of polysynaptic component only. -, no presynaptic effect or lack of input; n.t., effect of conditioning stimuli was not tested, therefore, no conclusion about the reciprocal or unidirectional nature of the inhibition could be done;
*, neurons tested for simultaneous stimulation of the C2 SN and TN; PN, projection neuron; LCN, local-circuit neuron.
Identification of Lamina I PNs and LCNs
Two approaches were used to label PNs and LCNs in Lamina I. In a part of the work, retrograde labeling of PNs was achieved by injecting tracer Fast DiI oil into the parabrachial nucleus, as described previously (Spike et al., 2003; Todd, 2010; Fernandes et al., 2020). In all cases, the accuracy of the injection was assessed by post hoc histologic analysis of brainstem sections as shown in our recent study (Fernandes et al., 2022). Retrogradely-labeled neurons were considered as PNs only if post hoc analysis confirmed that tracer was injected into the parabrachial area. Fluorescence optics was used to identify retrogradely-labeled PNs before recording. Lamina I neurons were also identified using the post hoc biocytin histochemistry. Successfully labeled neurons were classified as PNs or LCNs based on analysis of their axon structure/territory (Szucs et al., 2010, 2013; Fernandes et al., 2016). The axon of PNs crossed the midline and traveled to the contralateral anterolateral tract, whereas LCN axons branched extensively within the ipsilateral dorsal horn. In most upper cervical Lamina I neurons from this sample, however, the extent of the axonal tree labeling was not sufficient to characterize them as PNs or LCNs.
The neurons were classified as belonging to convergent, cervical-specific or trigeminal-specific processing lines based on their monosynaptic inputs from the C2 SN and TN (Fernandes et al., 2022). Convergent neurons received monosynaptic input from both nerves, whereas the cervical-specific and trigeminal-specific neurons received direct input only from the C2 SN and TN, respectively. Note that a trigeminal-specific neuron could receive a polysynaptic cervical input, and a cervical-specific neuron a polysynaptic trigeminal input.
Statistical analysis
Statistical analysis was performed with Prism 8.0 software (GraphPad). Data sets were compared using Student's t test and the difference was considered statistically significant when p < 0.05. All values are presented as mean ± SEM.
Results
Reciprocal induction of cervical and trigeminal PADs
Given that presynaptic inhibition is caused by PAD, we first studied its induction and time course in our preparation by recording TDRPs and DRPs evoked by stimulation of cervical and trigeminal afferents. The TDRP and C2 DRP evoked by stimulating the C2 SN and TN, respectively, typically exhibited slow kinetics (Fig. 1A,B). TDRPs and DRPs reached their peak amplitudes at 87.9 ± 6.2 ms (n = 15) and 83.4 ± 6.3 ms (n = 11) after afferent stimulation, respectively. Based on these measurements, we set the interval between conditioning and test stimuli in subsequent experiments at 100 ms. Bath application of the GABAA receptor blocker picrotoxin (100 μm) suppressed the initial phase of TDRP and C2 DRP (Fig. 1A,B) in a way similar to that described for the rat spinal cord (Kremer and Lev-Tov, 1998; Fernandes et al., 2020). When measured at the time moment of the peak response in control, the TDRP and C2 DRP were reduced by 44.1 ± 6.8% (n = 4) and 43.6 ± 4.6% (n = 4), respectively.
In summary, our recordings of TDRPs and DRPs have shown a reciprocal induction of PAD in both trigeminal and cervical afferents. This indicated that their inputs to the spinal dorsal horn neurons can be subject to presynaptic inhibition, which is mediated, at least in part, by GABAA receptors (Fernandes et al., 2020). The remaining part of PAD in both afferent systems could be mediated by ionotropic glutamate receptors (Russo et al., 2000; Zimmerman et al., 2019) as well as metabotropic glutamate or GABAB receptors. In agreement with this, the presynaptic inhibition of primary afferent inputs to some lumbar Lamina I neurons was not attenuated or abolished by picrotoxin (Fernandes et al., 2020), suggesting that it was mediated by receptors other than GABAA receptors.
Presynaptic inhibition affects a substantial population of Lamina I neurons
We next asked whether afferent-driven presynaptic inhibition affects cervical and trigeminal afferent inputs to Lamina I neurons. Whole-cell recordings were obtained from 83 upper cervical Lamina I neurons where we successfully tested whether they received inputs from both nerves (Fig. 1C). Based on the monosynaptic input, these neurons were classified as belonging to convergent, cervical or trigeminal processing lines (see Materials and Methods; Fernandes et al., 2022). The effect of conditioning C2 SN stimulation on TN input was studied in 57 neurons. Inhibition was observed in 30 of these neurons (i.e., 53%). TN conditioning had an effect on the C2 SN input in 28 out of 66 neurons tested (i.e., 42%). Thus, presynaptic interactions between cervical and trigeminal afferents were observed in about a half the recorded Lamina I neurons.
The lack of the presynaptic inhibition after the C2 SN conditioning (Fig. 1C, left) was observed in convergent (n = 14, 3 identified as PNs and 1 as an LCN), trigeminal-specific (n = 4, 1 identified as a PN) and cervical-specific neurons (n = 9, 1 identified as an LCN). After TN conditioning (Fig. 1C, right) presynaptic inhibition was not observed in 17 convergent (five identified as PNs) and 21 cervical-specific neurons (four identified as PNs and one as an LCN). This indicates that upper cervical Lamina I neurons belonging to all three processing lines can receive inputs that are not controlled by presynaptic interactions between cervical and trigeminal afferents. Hereafter, we focus exclusively on the neurons affected by presynaptic inhibition and these data are summarized in Table 1.
Presynaptic inhibition of direct Aδ-fiber and C-fiber inputs
We next analyzed how direct Aδ-fiber and C-fiber inputs to Lamina I PNs and LCNs, belonging to different processing lines (convergent, cervical or trigeminal), are affected by presynaptic inhibition induced by the C2 SN or TN volleys. Suppression of the monosynaptic components of the trigeminal and cervical input could be full or partial. A full block was considered when conditioning stimulation of one nerve (C2 SN, n = 11; TN, n = 10) completely suppressed one of the monosynaptic EPSC components evoked by stimulation of the other nerve (Fig. 2Aa). This effect was most likely caused by afferent-driven presynaptic inhibition of the parent afferent branch (Fernandes et al., 2020). C2 SN conditioning induced a full suppression of the TN-mediated EPSC component in 11 neurons, all of which were convergent, and two classified as PNs. In these neurons, monosynaptic trigeminal Aδ-fiber (n = 4) and C-fiber inputs (n = 7) were inhibited. In contrast, TN conditioning induced a full suppression of the C2 SN-mediated EPSC component in six convergent (one LCN) and four cervical-specific neurons. The trigeminal afferents mediated a full block of the components of monosynaptic input arising from the cervical Aδ-afferents (n = 1) and C-afferents (n = 9).
Figure 2.
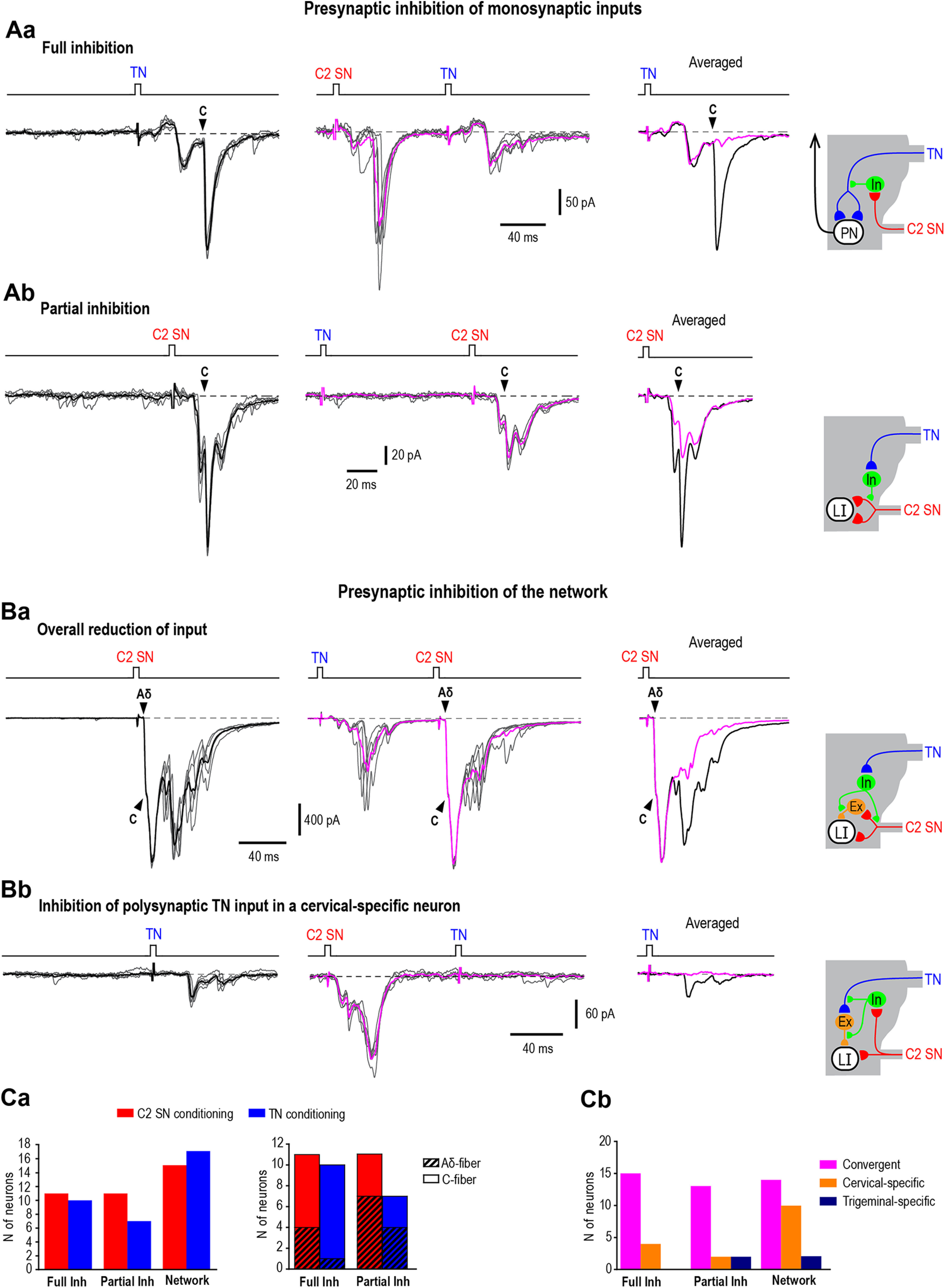
Responses modulated by presynaptic inhibition. Aa, Ab show afferent-driven presynaptic inhibition of the components of monosynaptic input (filled arrowheads). Aa, A full block of a monosynaptic trigeminal C-fiber EPSC by C2 SN conditioning (cell 178 from Table 1, note that the monosynaptic input to this convergent PN from the C2 SN is not indicated). Schematic shows how presynaptic inhibition affects the parent branch of the central axon. In, inhibitory interneuron. Holding potential, −85 mV. Ab, A partial block of the monosynaptic cervical C-fiber EPSC by TN conditioning (cell 129 from Table 1). Schematic illustrates how presynaptic inhibition affects the terminal branch of the afferent. Holding potential, −80 mV. Note the TN did not show monosynaptic or polysynaptic inputs to this cervical-specific neuron, but induced presynaptic inhibition of its monosynaptic cervical input. Ba, Bb, Inhibitory effects on the neuronal network. Ba, An overall reduction of the C2 SN-evoked test response after the TN conditioning (cell 198 from Table 1, the monosynaptic input to this convergent neuron from the TN is not indicated). Note the lack of the effect on monosynaptic Aδ-fiber-mediated and C-fiber-mediated EPSCs but a substantial reduction of the polysynaptic component. Schematic shows presynaptic inhibition at the cervical afferent branch supplying an intercalated excitatory neuron (Ex) or/and at the axon terminal of the intercalated neuron. Holding potential, −85 mV. Bb, A complete inhibition of the polysynaptic component in a neuron lacking monosynaptic input. C2 SN conditioning abolishes polysynaptic TN-mediated EPSCs in a cervical-specific Lamina I neuron (cell 132 from Table 1). Schematic shows presynaptic inhibition at the trigeminal afferent branch supplying an intercalated excitatory neuron (Ex) or/and at the axon terminal of the intercalated neuron. Holding potential, −70 mV. For each type of response, five traces are shown with their average. Averaged responses to the test stimuli are shown superimposed. Filled arrowheads indicate monosynaptic components only for the EPSCs evoked by the test stimuli. Ca, Numbers of Lamina I neurons affected by different types of presynaptic inhibition (left) and by the full or partial suppression of Aδ-fiber and C-fiber components of input (right). Data are shown for C2 SN (red) and TN (blue) conditioning. Cb, Numbers of convergent, cervical-specific and trigeminal-specific Lamina I neurons with inputs controlled by different forms of afferent-driven inhibition.
We also observed a partial reduction in the monosynaptic component of the response induced by conditioning stimulation of the C2 SN (n = 11) or TN (n = 7; Fig. 2Ab). This partial block could result from presynaptic inhibition of one of the afferent terminal branches as discussed previously (Fernandes et al., 2020). Such block of trigeminal input was induced by C2 SN conditioning in nine convergent (two PNs) and two trigeminal-specific Lamina I neurons. This type of inhibition was seen for the trigeminal Aδ-afferent (n = 7) and C-afferent inputs (n = 4). The TN conditioning induced a partial block of direct cervical inputs in five convergent (two PNs) and two cervical-specific (one LCN) neurons. The TN-induced partial block affected both Aδ-fiber (n = 4) and C-fiber cervical inputs (n = 3). Together, these data suggest that the presynaptic inhibition driven by cervical and trigeminal afferents can suppress (completely or partially) components of nociceptive Aδ-fiber and C-fiber input to Lamina I PNs and LCNs processing convergent or specific trigeminocervical information.
It should be noted that since the test stimulation in our experiments activated a population of primary afferents (rather than a single afferent fiber) supplying a given Lamina I neuron, it is also possible that the full versus partial inhibition simply reflects whether the conditioning stimulation drives presynaptic inhibition in all, or rather in a subset, of the primary afferent inputs associated with the test stimulation. However, regardless of the mechanisms underlying these phenomena, full and partial inhibitions represent two forms of the afferent-driven control of monosynaptic primary afferent input to Lamina I.
In some cases, conditioning differently affected two components of monosynaptic inputs arising from one nerve. This was seen for Aδ-fiber and C-fiber inputs from the C2 SN (cell 103; Table 1), two Aδ-inputs from the TN (cell 170; Table 1), and Aδ-fiber and C-fiber inputs from the TN (cell 266; Table 1; Fig. 4, left). Thus, the afferent-driven presynaptic inhibition has the ability to selectively affect transmitter release from some primary afferent terminals synapsing onto a given Lamina I neuron.
Figure 4.
Reciprocal presynaptic inhibition. An example of reciprocal afferent-driven presynaptic inhibition in a convergent Lamina I neuron (cell 256 from Table 1). C2 SN conditioning abolished the monosynaptic C-fiber-mediated EPSC component of the TN input, and TN conditioning completely abolished the monosynaptic C-fiber EPSC component of the C2 SN input. Note, both conditioning stimuli reduced the polysynaptic component activated by the test stimuli. Holding potential, −80 mV. Schematic depicting how an inhibitory neuron (In) can mediate presynaptic inhibition of afferents from both nerves. Five traces are shown for each response and the averaged responses are superimposed. Filled arrowheads indicate monosynaptic components only for the EPSCs evoked by the test stimuli.
Inhibition of the neuronal network supplying Lamina I neurons
We next undertook detailed examination of 26 upper cervical Lamina I neurons in which the trigeminal (n = 15) and/or cervical (n = 17) afferent input was affected by inhibition of the neuronal network. In these neurons, afferent-driven inhibition of the network was observed as a reduction in the polysynaptic inputs (Fig. 2Ba,Bb).
In most cases (25 of 32), conditioning produced a partial reduction of the response (Fig. 2Ba; Table 1, network, indicated by % or +). The partial reduction of the TN-mediated EPSCs by C2 SN conditioning was observed in six convergent, one trigeminal-specific, and two cervical-specific neurons. The TN conditioning reduced the input in 11 convergent (three PNs) and five cervical-specific (one PN) neurons. The mean reduction of the area under the EPSCs was by 38.5 ± 0.1% (C2 SN conditioning, n = 7; TN conditioning, n = 13). This indicated that both trigeminal and cervical afferents can control the overall input to convergent and cervical-specific Lamina I neurons in the upper cervical segments.
Another form of inhibitory action on the neuronal network was observed in the remaining seven cases. In six cervical-specific neurons, the C2 SN conditioning completely abolished the TN input consisting of only a polysynaptic component (Fig. 2Bb; Table 1, indicated by ++). This C2 SN-driven suppression of polysynaptic TN input may function as a mechanism protecting cervical-specific Lamina I neurons from the trigeminal information flow. In a similar way, the TN conditioning completely inhibited the C2 SN polysynaptic input in one trigeminal-specific neuron.
The above experiments show that the mechanism by which the conditioning stimulation acts on the neuronal network is complex. Although some contribution of the slow postsynaptic action on the intercalated neurons (e.g., conditioning-evoked metabotropic receptor-mediated changes of excitability) cannot be excluded, the presynaptic action was likely to play a major role. This assertion is based on our observations that: (1) the long duration (>100 ms) of the effect correlated with the time course of the DRPs and TDRPs (Fig. 1A,B); and (2) inhibition occurred when the postsynaptic response evoked by the conditioning stimuli has already terminated (see Luz et al., 2019). Thus, the presynaptic inhibition of the primary afferents supplying intercalated neurons and/or of the axons of intercalated neurons synapsing on Lamina I neurons is likely to reflect a complex presynaptic control of the superficial dorsal horn network.
Taken together, our results show that diverse forms of presynaptic inhibition can shape monosynaptic and polysynaptic trigeminal and cervical afferent input to the upper cervical Lamina I neurons (Fig. 2Ca,Cb).
Unidirectional presynaptic inhibition
We next tested whether cervical and trigeminal inputs to a Lamina I neuron could be subject to unidirectional or reciprocal presynaptic control. Unidirectional presynaptic inhibition was observed in 16 out of 40 (i.e., 40%) upper cervical Lamina I neurons tested for effects evoked by conditioning stimuli applied to both nerves (Table 1; Fig. 3). C2 SN conditioning induced unidirectional presynaptic inhibition of the TN-mediated EPSC components in neurons from convergent (n = 6, 1 PN; Fig. 3A) and cervical-specific (n = 3, 1 PN) processing lines (Table 1). The inhibition was observed for both monosynaptic Aδ-fiber-mediated (n = 3) and C-fiber-mediated EPSCs (n = 3) as well as for the polysynaptic input (n = 4). In all these neurons, the TN conditioning did not change the C2 SN input.
Figure 3.
Unidirectional presynaptic inhibition. A, Example of unidirectional inhibition of monosynaptic trigeminal C-fiber input to a convergent Lamina I neuron by C2 SN conditioning (cell 110 from Table 1). Note, the TN conditioning had no effect on the monosynaptic C2 SN input. Holding potential, −80 mV. B, unidirectional inhibition of the polysynaptic C2 SN input to a trigeminal-specific Lamina I neuron by TN conditioning (cell 52 from Table 1). Note, the C2 SN conditioning had no effect on monosynaptic trigeminal Aδ-fiber and C-fiber inputs. Holding potential, −80 mV. Schematic showing that presynaptic inhibition occurs at the cervical afferent terminal supplying an intercalated excitatory neuron (Ex) or/and at the axon terminal of the intercalated neuron. Five traces are shown with averaged traces superimposed. Averaged responses to the test stimuli are shown superimposed below. Filled arrowheads indicate monosynaptic components only for the EPSCs evoked by the test stimuli.
Unidirectional inhibition of cervical afferent input was observed in seven upper cervical Lamina I neurons (Table 1), of which four were convergent (one LCN), two were cervical-specific (one LCN), and one was trigeminal-specific (shown in Fig. 3B). In these cells, the TN conditioning caused presynaptic inhibition of the monosynaptic (Aδ-fiber, n = 3; C-fiber, n = 2) and polysynaptic (n = 4) C2 SN input (Table 1). In all these neurons, the C2 SN conditioning did not affect the TN input.
Thus, the unidirectional presynaptic inhibition of the upper cervical Lamina I neurons could be induced by either the C2 SN or TN conditioning.
Reciprocal presynaptic inhibition
Reciprocal presynaptic inhibition of cervical and trigeminal afferent inputs was observed in 12 out of 40 (30%) neurons tested (in the remaining 12 neurons, the afferent-driven presynaptic inhibition was not seen). Reciprocal inhibition affected inputs in convergent (n = 8, one of these neurons is shown in Fig. 4) and cervical-specific (n = 4) Lamina I neurons; this sample contained four identified PNs (Table 1). In these neurons, the C2 SN conditioning induced suppression of the TN-mediated monosynaptic (Aδ-fiber, n = 3; C-fiber, n = 5) and polysynaptic (n = 7) EPSC components. In contrast, TN conditioning induced presynaptic inhibition of the monosynaptic (Aδ-fiber, n = 1; C-fiber, n = 4) and polysynaptic (n = 10) components of the EPSCs arising from the C2 SN.
Thus, reciprocal presynaptic inhibition affects direct Aδ-fiber and C-fiber inputs to convergent and cervical-specific Lamina I neurons. Besides, it controls direct trigeminal and cervical input to PNs and has an effect on the neuronal network supplying them.
Simultaneous activation of cervical and trigeminal inputs
Because conditioning stimulation of the C2 SN and/or TN induces a presynaptic inhibition of the EPSCs arising from the other nerve, we next asked how these responses mix when the stimuli to both nerves are applied simultaneously. These experiments were done in a heterogeneous population of 14 Lamina I neurons (convergent, n = 11; cervical-specific, n = 2; trigeminal-specific, n = 1), nine of which are listed in Table 1 (indicated by asterisks) and the remaining five either did not show presynaptic inhibition or were not tested for it. In these neurons, the overall monosynaptic and polysynaptic EPSCs evoked by simultaneous C2 SN and TN stimulation were compared with the summed response evoked by stimulating each nerve individually (Fig. 5). This was assessed by comparing the integrals for the simultaneous stimulation of each nerve versus the sum of the two individual responses. The responses were similar (p = 0.8, n = 14, difference 13.8 ± 2.3%) as shown in the plots in Figure 5. Thus, simultaneously evoked inputs from the C2 SN and TN show a substantial degree of additivity. In this case, timing plays a critical role in induction of trigeminal and cervical afferent-driven presynaptic inhibition, and it does not significantly affect summation of simultaneously arriving overall inputs.
Figure 5.
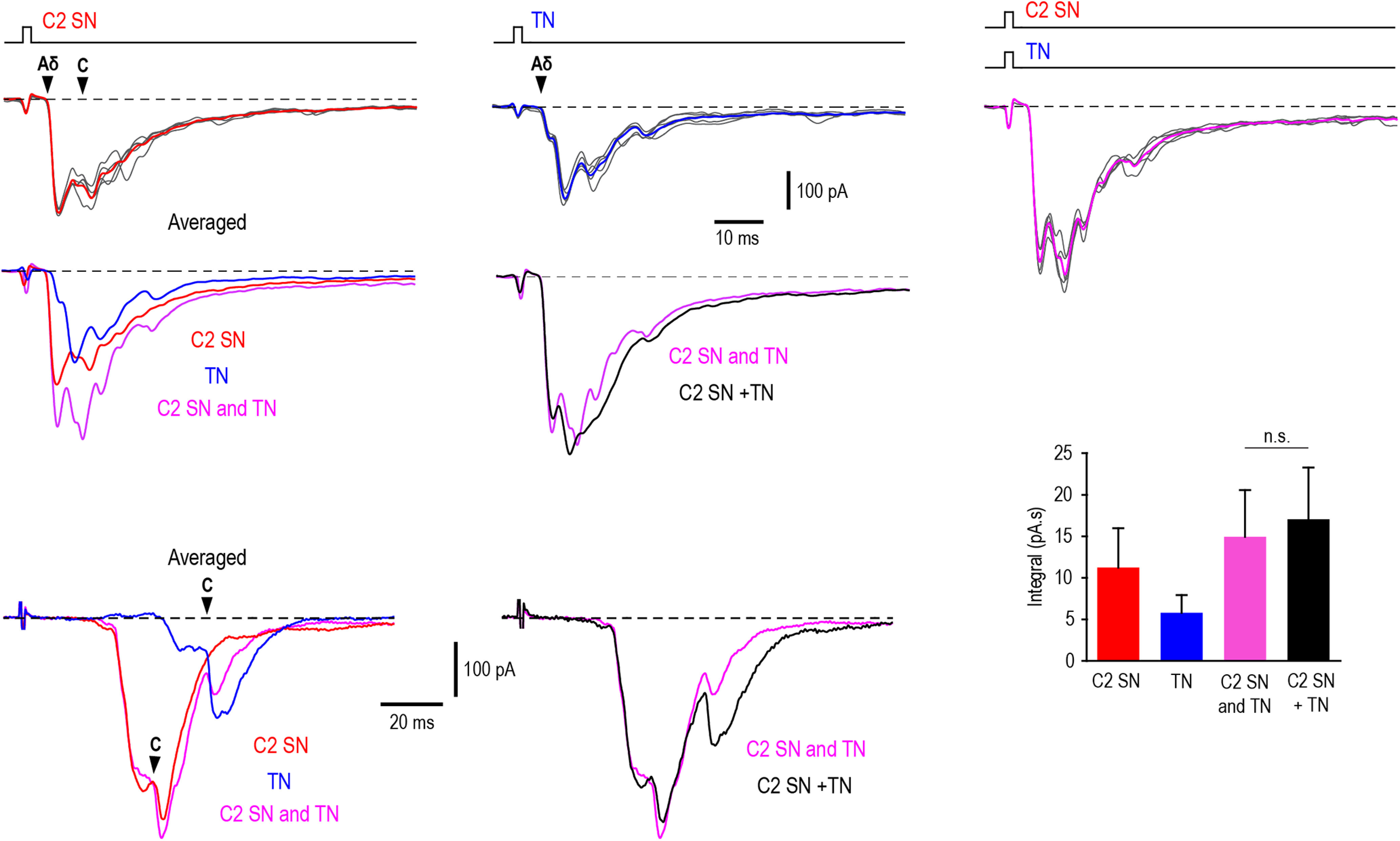
Inputs evoked by simultaneous stimulation of C2 SN and TN. The effect is illustrated for two convergent Lamina I neurons differently affected by presynaptic inhibition. Upper part, A PN (cell 185; Table 1) showing a partial (19%) unidirectional presynaptic inhibition of its monosynaptic Aδ-input from the TN. Top traces, EPSCs evoked by stimulating the C2 SN (left), TN (middle), and both nerves at the same time (right). Filled arrowheads indicate monosynaptic components. Five traces (gray) are shown for each type of stimulation and their averages are given in red, blue, and magenta, respectively. The averaged traces are shown superimposed below (left). C2 SN and TN, averaged response evoked by simultaneously stimulating both nerves. Right, The EPSC evoked by simultaneously stimulating both nerves is superimposed with the sum of the EPSCs evoked by the C2 SN and TN stimulations (C2 SN + TN, black). Holding potential, −80 mV. Bottom part, A convergent Lamina I neuron (cell 266; Table 1) showing a reciprocal inhibition of monosynaptic C components and a substantial effect on the network. Left, The averaged EPSCs are compared. Filled arrowheads indicate monosynaptic components. Right, The EPSC evoked by simultaneously stimulating both nerves is superimposed with the sum of the EPSCs evoked by the C2 SN and TN stimulations. Holding potential, −85 mV. The histogram shows the area under the EPSC (i.e., integral) evoked by stimulating the C2 SN, TN and both nerves simultaneously (C2 SN and TN), compared with the calculated sum of the C2 SN and TN inputs (C2 SN + TN). Pooled data from 14 neurons. Note, the integrals for simultaneously evoked inputs and the sum of two individual inputs are similar. N.s., nonsignificant.
Discussion
We have revealed different forms of presynaptic interactions between cervical and trigeminal afferents supplying upper cervical Lamina I neurons (Fig. 6). Trigeminocervical afferent-driven presynaptic inhibition (1) controls input to a substantial population of Lamina I neurons; (2) suppresses monosynaptic and polysynaptic Aδ-fiber-mediated and C-fiber-mediated inputs; (3) can function in a unidirectional (driven by the C2 SN or TN) and a reciprocal mode; (4) modulates input to convergent, cervical-specific and trigeminal-specific Lamina I neurons; and (5) depends on timing, being insignificant for the overall inputs arriving simultaneously. This presynaptic inhibition shapes trigeminocervical input to Lamina I PNs and LCNs. We therefore propose that disruption of afferent-driven presynaptic inhibition may contribute to pain referral to cervical and TN territories observed in patients with primary headache syndromes.
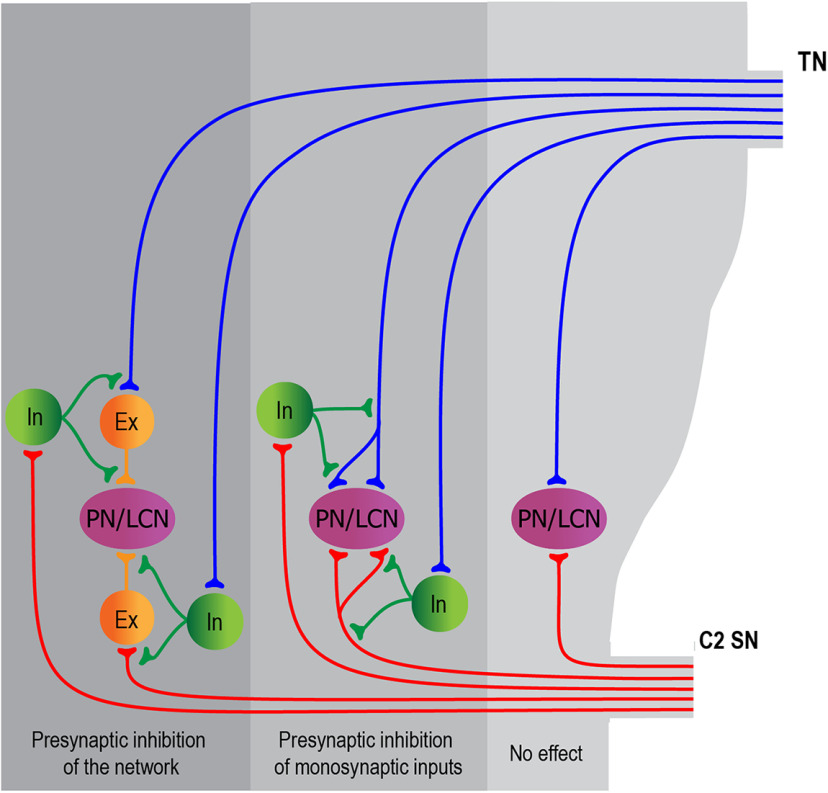
Figure 6.
Presynaptic interactions between trigeminal and cervical afferents supplying upper cervical Lamina I. Schematic illustrating the major forms of presynaptic interactions between cervical and trigeminal Aδ-afferents and C-afferents supplying Lamina I neurons. Presynaptic inhibition of monosynaptic inputs can be reciprocal or unidirectional and affect both the parent or terminal branches of the afferent fiber. Presynaptic inhibition of the neuronal network regulates polysynaptic input to Lamina I neurons. This can be evoked by inhibiting the afferent axon, which supplies an intercalated excitatory neuron (Ex), or the axon terminal of the intercalated neuron. In some Lamina I neurons, no interaction between cervical and trigeminal afferent fibers was detected (no effect). PN, projection neuron; LCN, local-circuit neuron; In, inhibitory interneuron. Note, the precise laminar location of the inhibitory interneurons and excitatory intercalated neurons is not known.
Ex vivo brainstem-cervical cord preparation
We used our ex vivo brainstem-cervical cord preparation, with preserved C2 SN and TN, for visually guided recordings from upper cervical Lamina I neurons (Luz et al., 2019; Fernandes et al., 2022). This preparation offers several advantages as the entire brainstem and upper cervical cord are preserved along with afferent pathways from both nerves. Moreover, spinal interneurons mediating presynaptic inhibition and the dendritic structure of the receiving Lamina I neuron remain intact. The present study shows that our preparation can provide insight into functional presynaptic interactions between trigeminal and cervical nociceptors supplying cervical neurons. It can also be used for studying the pharmacology of neuronal networks controlling synaptic transmission in primary nociceptors (Kilinc et al., 2017).
The PAD
Presynaptic inhibition is caused by the PAD because of Cl– efflux through the pore of GABAA receptors (Alvarez-Leefmans et al., 1998; Rudomin and Schmidt, 1999; Willis, 1999). The antidromic spread of the PAD to the dorsal root can be recorded as a DRP (Barron and Matthews, 1938; Lloyd and McIntyre, 1949). Here, we show that the C2 SN and TN afferents evoke reciprocal PADs. Both the C2 SN-induced TDRP and the TN-induced C2 DRP have a slow time course. Importantly, this is similar to that described in a number of reports for spinal and trigeminal afferents (Barron and Matthews, 1938; Lloyd and McIntyre, 1949; Baldissera et al., 1967; Yu and Avery, 1974; Nakamura et al., 1977; Fernandes et al., 2020). Therefore, one could expect that cervical and trigeminal afferents undergo a broad reciprocal presynaptic control. Our whole-cell recordings revealed that this control affects the Aδ-fiber and C-fiber afferents supplying the upper cervical Lamina I PNs and LCNs as well as their network.
Presynaptic inhibition of Aδ-fibers and C-fibers
Presynaptic inhibition of primary afferents is an important mechanism of spinal sensory processing (Melzack and Wall, 1965; Rudomin and Schmidt, 1999; Willis, 1999; Boyle et al., 2019). The afferent-driven inhibition was originally described for large diameter muscle afferents supplying motoneurons and for low-threshold cutaneous afferents terminating in the deep dorsal horn (Koketsu, 1956; Eccles, 1964; Rudomin and Schmidt, 1999; Willis, 1999; Hughes et al., 2005, 2012; Fink et al., 2014; Boyle et al., 2019). Such afferent-driven presynaptic inhibition has recently been shown for unmyelinated C-fibers supplying Lamina I neurons in the lumbar cord (Fernandes et al., 2020). The lumbar C-fibers are controlled by homosegmental and heterosegmental Aβ-afferents, Aδ-afferents, and C-afferents. Here, we further show that, in the upper cervical cord, input to Lamina I neurons from cervical nerves and TNs undergoes unidirectional and reciprocal control. Trigeminal and cervical afferent stimulation induced a full or a partial suppression of the components of monosynaptic input mediated by Aδ-fibers and C-fibers. This full or partial block of direct inputs could be caused by inhibition of the parent or terminal afferent branches, respectively (Fernandes et al., 2020). In addition, the C2 SN and TN afferent-driven presynaptic inhibition could interfere with the neuronal network by reducing the polysynaptic component of the input, which occurred most likely because of the inhibition of the afferents supplying intercalated neurons or the axons of intercalated neurons (Fig. 6). In the present study, we did not identify the types of afferent (Aβ, Aδ, or C) that mediates the presynaptic effects. Since all three afferent types can suppress nociceptive inputs to Lamina I neurons (Fernandes et al., 2020), a special study will be needed to clarify this issue. Thus, at present, we can only say that the afferent-driven presynaptic inhibition controls direct and indirect Aδ-fiber and C-fiber inputs to Lamina I neurons and shapes the trigeminocervical nociceptive information inflow in the cervical cord.
Control of convergent and specific inputs
Lamina I neurons in the upper cervical cord receive diverse patterns of trigeminal and cervical afferent supply. They can be classified as convergent, cervical-specific, or trigeminal-specific on the basis of their monosynaptic input, which reflects a direct connection of the neuron to the corresponding primary afferent system (Fernandes et al., 2022). Here, we show that both monosynaptic and polysynaptic components of the peripheral input are modulated by afferent-driven inhibition. In convergent neurons, direct trigeminal Aδ-fiber and C-fiber inputs are controlled by cervical afferents and vice versa. Monosynaptic C2 SN inputs to cervical-specific neurons are inhibited by trigeminal afferents, whereas the direct TN inputs to the trigeminal-specific neurons are controlled by cervical afferents. Thus, the monosynaptic input from one nerve is regulated by afferents from the other, even if that nerve does not supply the neuron directly. This organization allows the control of monosynaptic input to cervical-specific and trigeminal-specific neurons.
The afferent-driven presynaptic inhibition of the dorsal horn neuronal network, that is seen as an overall reduction of the monosynaptic and polysynaptic EPSC components, further shapes input to convergent neurons. This mechanism could have a substantial impact on the long-lasting discharge of spinoparabrachial Lamina I neurons that is driven by strong polysynaptic inputs (Agashkov et al., 2019). Interestingly, C2 SN conditioning completely abolished the polysynaptic input from the TN in cervical-specific neurons, thus preventing them from being excited by the trigeminal afferent volley. Clinically, disinhibition of this pathway could “open the gate” for ectopic trigeminal signaling in a neuron processing occipitocervical information and cause pain referral and spread from trigeminal to cervical and occipital nerve territories.
The trigeminocervical afferent-driven presynaptic inhibition can be unidirectional or reciprocal. The unidirectional effect was mostly observed in convergent neurons and could occur in either direction. This mechanism could represent a trigeminocervical form of presynaptic lateral inhibition, where one peripheral input suppresses another that arises from neighboring territory. The reciprocal control of trigeminal and cervical afferent inputs was observed in convergent and cervical-specific neurons. It can function as a reciprocal form of lateral presynaptic inhibition for the afferents innervating neighboring cranial territories. Under normal conditions, these mechanisms can prevent a Lamina I PN from receiving nonspecific nociceptive input. Thus, diverse forms of presynaptic control, unidirectional and reciprocal, can affect direct inputs and neuronal network and shape the pattern of the trigeminocervical afferent supply to Lamina I neurons in the upper cervical cord.
Trigeminal and cervical afferents and pain referral
The C2 SN provides sensory innervation to various cervical and occipital structures that are recognized as sources of neck and head pain (Bogduk, 2001; Bartsch and Goadsby, 2003a; Kemp et al., 2011). The major peripheral branch of this SN, the greater occipital nerve, supplies the back of the head. Pain originating in occipital and cervical structures innervated by the C2 SN is often projected to the front of the head, which is the territory of the TN (Bogduk and Govind, 2009). The TN supplying craniofacial structures, such as the facial skin, cranial dura mater or blood vessels, is involved in many types of headaches and facial pain pathologies (Yoon et al., 2010; Kemp et al., 2012; Edvinsson et al., 2020). Pain of trigeminal origin, e.g., in migraine and cluster headache, is also projected to the territory of the greater occipital nerve (Bartsch, 2005). These patterns of pain referral and spread reflect a broad overlap in the termination fields of cervical and trigeminal afferents in the upper cervical dorsal horn as well as their convergence on dorsal horn neurons (Kerr, 1961; Pfaller and Arvidsson, 1988; Bartsch and Goadsby, 2002, 2003a, b; Bogduk and Govind, 2009; García-Magro et al., 2020; Fernandes et al., 2022).
Lamina I is the major spinal pain-projection area where most neurons are nociceptive-specific and targeted by Aδ-afferents and C-afferents of somatic and visceral origin (Cervero and Iggo, 1980; Brown, 1982; Cervero and Tattersall, 1987; Willis and Coggeshall, 2004; Luz et al., 2015). Thus, cervical Lamina I plays a crucial role in trigeminocervical processing (Fernandes et al., 2022). Convergent or line-specific PNs integrate or segregate occipitocervical and trigeminal Aδ-fiber and C-fiber inputs. An excitatory drive to PNs is balanced by convergent inhibitory LCNs (Fernandes et al., 2022). Here, we show that processing in the upper cervical Lamina I is additionally shaped by diverse forms of afferent-driven presynaptic inhibition which can fine-tune nociceptive information flow. Disruption of this balance could contribute to diverse pain syndromes of trigeminocervical origin.
Footnotes
This work was supported by Fundo Europeu de Desenvolvimento Regional (FEDER) funds through the COMPETE 2020-Operacional Programme for Competitiveness and Internationalization (POCI), Portugal 2020; by Portuguese funds through FCT, Fundacão para a Ciência e a Tecnologia/Ministério da Ciência, Tecnologia e Ensino Superior in the framework of the Project PTDC/NEU-NMC/1259/2014 (POCI-01-0145-FEDER-016588); and by Prémio MELO e CASTRO from SCML. E.C.F. and L.L.L. were supported by FCT Fellowships SFRH/BD/118129/2016 and SFRH/BPD/120097/2016, respectively. We thank Dr. Robert J. Callister (the University of Newcastle, Australia) for constructive discussions and critical reading of the manuscript.
The authors declare no competing financial interests.
References
- Agashkov K, Krotov V, Krasniakova M, Shevchuk D, Andrianov Y, Zabenko Y, Safronov BV, Voitenko N, Belan P (2019) Distinct mechanisms of signal processing by lamina I spino-parabrachial neurons. Sci Rep 9:19231. 10.1038/s41598-019-55462-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alvarez-Leefmans FJ, Nani A, Marquez S (1998) Chloride transport, osmotic balance, and presynaptic inhibition. In: Presynaptic inhibition and neural control (Rudomin P, Romo R, Mendel LM, eds), pp 50–79. New York: Oxford University Press. [Google Scholar]
- Baldissera F, Broggi G, Mancia M (1967) Depolarization of trigeminal afferents induced by stimulation of brain-stem and peripheral nerves. Exp Brain Res 4:1–17. 10.1007/BF00235213 [DOI] [PubMed] [Google Scholar]
- Barron DH, Matthews BH (1938) The interpretation of potential changes in the spinal cord. J Physiol 92:276–321. 10.1113/jphysiol.1938.sp003603 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bartsch T (2005) Migraine and the neck: new insights from basic data. Curr Pain Headache Rep 9:191–196. 10.1007/s11916-005-0061-0 [DOI] [PubMed] [Google Scholar]
- Bartsch T, Goadsby PJ (2002) Stimulation of the greater occipital nerve induces increased central excitability of dural afferent input. Brain 125:1496–1509. 10.1093/brain/awf166 [DOI] [PubMed] [Google Scholar]
- Bartsch T, Goadsby PJ (2003a) The trigeminocervical complex and migraine: current concepts and synthesis. Curr Pain Headache Rep 7:371–376. [DOI] [PubMed] [Google Scholar]
- Bartsch T, Goadsby PJ (2003b) Increased responses in trigeminocervical nociceptive neurons to cervical input after stimulation of the dura mater. Brain 126:1801–1813. 10.1093/brain/awg190 [DOI] [PubMed] [Google Scholar]
- Bogduk N (2001) Cervicogenic headache: anatomic basis and pathophysiologic mechanisms. Curr Pain Headache Rep 5:382–386. 10.1007/s11916-001-0029-7 [DOI] [PubMed] [Google Scholar]
- Bogduk N, Govind J (2009) Cervicogenic headache: an assessment of the evidence on clinical diagnosis, invasive tests, and treatment. Lancet Neurol 8:959–968. 10.1016/S1474-4422(09)70209-1 [DOI] [PubMed] [Google Scholar]
- Boyle KA, Gradwell MA, Yasaka T, Dickie AC, Polgar E, Ganley RP, Orr DPH, Watanabe M, Abraira VE, Kuehn ED, Zimmerman AL, Ginty DD, Callister RJ, Graham BA, Hughes DI (2019) Defining a spinal microcircuit that gates myelinated afferent input: implications for tactile allodynia. Cell Rep 28:526–540.e6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown AG (1982) The dorsal horn of the spinal cord. Q J Exp Physiol 67:193–212. 10.1113/expphysiol.1982.sp002630 [DOI] [PubMed] [Google Scholar]
- Cervero F, Iggo A (1980) The substantia gelatinosa of the spinal cord: a critical review. Brain 103:717–772. 10.1093/brain/103.4.717 [DOI] [PubMed] [Google Scholar]
- Cervero F, Tattersall JE (1987) Somatic and visceral inputs to the thoracic spinal cord of the cat: marginal zone (lamina I) of the dorsal horn. J Physiol 388:383–395. 10.1113/jphysiol.1987.sp016620 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eccles JC (1964) Presynaptic inhibition in the spinal cord. Prog Brain Res 12:65–91. 10.1016/s0079-6123(08)60618-4 [DOI] [PubMed] [Google Scholar]
- Edvinsson JCA, et al. (2020) The fifth cranial nerve in headaches. J Headache Pain 21:65. 10.1186/s10194-020-01134-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Escolar J (1948) The afferent connections of the 1st, 2nd, and 3rd cervical nerves in the cat; an analysis by Marchi and Rasdolsky methods. J Comp Neurol 89:79–92. [DOI] [PubMed] [Google Scholar]
- Fernandes EC, Luz LL, Mytakhir O, Lukoyanov NV, Szucs P, Safronov BV (2016) Diverse firing properties and Aβ-, Aδ-, and C-afferent inputs of small local circuit neurons in spinal lamina I. Pain 157:475–487. 10.1097/j.pain.0000000000000394 [DOI] [PubMed] [Google Scholar]
- Fernandes EC, Pechincha C, Luz LL, Kokai E, Szucs P, Safronov BV (2020) Primary afferent-driven presynaptic inhibition of C-fiber inputs to spinal lamina I neurons. Prog Neurobiol 188:101786. 10.1016/j.pneurobio.2020.101786 [DOI] [PubMed] [Google Scholar]
- Fernandes EC, Carlos-Ferreira J, Luz LL, Kokai E, Meszar Z, Szucs P, Safronov BV (2022) Processing of trigeminocervical nociceptive afferent input by neuronal circuity in the upper cervical lamina I. Pain 163:362–375. 10.1097/j.pain.0000000000002342 [DOI] [PubMed] [Google Scholar]
- Fink AJ, Croce KR, Huang ZJ, Abbott LF, Jessell TM, Azim E (2014) Presynaptic inhibition of spinal sensory feedback ensures smooth movement. Nature 509:43–48. 10.1038/nature13276 [DOI] [PMC free article] [PubMed] [Google Scholar]
- García-Magro N, Martin YB, Negredo P, Avendaño C (2018) The greater occipital nerve and its spinal and brainstem afferent projections: a stereological and tract-tracing study in the rat. J Comp Neurol 526:3000–3019. 10.1002/cne.24511 [DOI] [PubMed] [Google Scholar]
- García-Magro N, Negredo P, Martin YB, Nuñez A, Avendaño C (2020) Modulation of mechanosensory vibrissal responses in the trigeminocervical complex by stimulation of the greater occipital nerve in a rat model of trigeminal neuropathic pain. J Headache Pain 21:96. 10.1186/s10194-020-01161-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goadsby PJ, Holland PR, Martins-Oliveira M, Hoffmann J, Schankin C, Akerman S (2017) Pathophysiology of migraine: a disorder of sensory processing. Physiol Rev 97:553–622. 10.1152/physrev.00034.2015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gradwell MA, Callister RJ, Graham BA (2020) Reviewing the case for compromised spinal inhibition in neuropathic pain. J Neural Transm (Vienna) 127:481–503. 10.1007/s00702-019-02090-0 [DOI] [PubMed] [Google Scholar]
- Graham B, Redman S (1994) A simulation of action potentials in synaptic boutons during presynaptic inhibition. J Neurophysiol 71:538–549. 10.1152/jn.1994.71.2.538 [DOI] [PubMed] [Google Scholar]
- Hughes DI, Mackie M, Nagy GG, Riddell JS, Maxwell DJ, Szabó G, Erdélyi F, Veress G, Szucs P, Antal M, Todd AJ (2005) P boutons in lamina IX of the rodent spinal cord express high levels of glutamic acid decarboxylase-65 and originate from cells in deep medial dorsal horn. Proc Natl Acad Sci U S A 102:9038–9043. 10.1073/pnas.0503646102 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hughes DI, Sikander S, Kinnon CM, Boyle KA, Watanabe M, Callister RJ, Graham BA (2012) Morphological, neurochemical and electrophysiological features of parvalbumin-expressing cells: a likely source of axo-axonic inputs in the mouse spinal dorsal horn. J Physiol 590:3927–3951. 10.1113/jphysiol.2012.235655 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kemp WJ 3rd, Tubbs RS, Cohen-Gadol AA (2011) The innervation of the scalp: a comprehensive review including anatomy, pathology, and neurosurgical correlates. Surg Neurol Int 2:178. 10.4103/2152-7806.90699 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kemp WJ 3rd, Tubbs RS, Cohen-Gadol AA (2012) The innervation of the cranial dura mater: neurosurgical case correlates and a review of the literature. World Neurosurg 78:505–510. 10.1016/j.wneu.2011.10.045 [DOI] [PubMed] [Google Scholar]
- Kerr FW (1961) Structural relation of the trigeminal spinal tract to upper cervical roots and the solitary nucleus in the cat. Exp Neurol 4:134–148. 10.1016/0014-4886(61)90036-x [DOI] [PubMed] [Google Scholar]
- Kilinc E, Guerrero-Toro C, Zakharov A, Vitale C, Gubert-Olive M, Koroleva K, Timonina A, Luz LL, Shelukhina I, Giniatullina R, Tore F, Safronov BV, Giniatullin R (2017) Serotonergic mechanisms of trigeminal meningeal nociception: implications for migraine pain. Neuropharmacology 116:160–173. 10.1016/j.neuropharm.2016.12.024 [DOI] [PubMed] [Google Scholar]
- Koketsu K (1956) Intracellular potential changes of primary afferent nerve fibers in spinal cords of cats. J Neurophysiol 19:375–392. 10.1152/jn.1956.19.5.375 [DOI] [PubMed] [Google Scholar]
- Kremer E, Lev-Tov A (1998) GABA-receptor-independent dorsal root afferents depolarization in the neonatal rat spinal cord. J Neurophysiol 79:2581–2592. 10.1152/jn.1998.79.5.2581 [DOI] [PubMed] [Google Scholar]
- Lloyd DP, McIntyre AK (1949) On the origins of dorsal root potentials. J Gen Physiol 32:409–443. 10.1085/jgp.32.4.409 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luz LL, Szucs P, Pinho R, Safronov BV (2010) Monosynaptic excitatory inputs to spinal lamina I anterolateral-tract-projecting neurons from neighbouring lamina I neurons. J Physiol 588:4489–4505. 10.1113/jphysiol.2010.197012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luz LL, Fernandes EC, Sivado M, Kokai E, Szucs P, Safronov BV (2015) Monosynaptic convergence of somatic and visceral C-fiber afferents on projection and local circuit neurons in lamina I: a substrate for referred pain. Pain 156:2042–2051. 10.1097/j.pain.0000000000000267 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luz LL, Fernandes EC, Dora F, Lukoyanov NV, Szucs P, Safronov BV (2019) Trigeminal Aδ- and C-afferent supply of lamina I neurons in the trigeminocervical complex. Pain 160:2612–2623. 10.1097/j.pain.0000000000001659 [DOI] [PubMed] [Google Scholar]
- Marfurt CF, Rajchert DM (1991) Trigeminal primary afferent projections to “non-trigeminal” areas of the rat central nervous system. J Comp Neurol 303:489–511. 10.1002/cne.903030313 [DOI] [PubMed] [Google Scholar]
- Melzack R, Wall PD (1965) Pain mechanisms: a new theory. Science 150:971–979. 10.1126/science.150.3699.971 [DOI] [PubMed] [Google Scholar]
- Nakamura Y, Murakami T, Kikuchi M, Kubo Y, Ishimine S (1977) Primary afferent depolarization in the trigeminal spinal nucleus of cats. Exp Brain Res 29:45–56. 10.1007/BF00236874 [DOI] [PubMed] [Google Scholar]
- Pfaller K, Arvidsson J (1988) Central distribution of trigeminal and upper cervical primary afferents in the rat studied by anterograde transport of horseradish peroxidase conjugated to wheat germ agglutinin. J Comp Neurol 268:91–108. 10.1002/cne.902680110 [DOI] [PubMed] [Google Scholar]
- Pinto V, Szucs P, Derkach VA, Safronov BV (2008) Monosynaptic convergence of C- and Adelta-afferent fibres from different segmental dorsal roots on to single substantia gelatinosa neurones in the rat spinal cord. J Physiol 586:4165–4177. 10.1113/jphysiol.2008.154898 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pinto V, Szucs P, Lima D, Safronov BV (2010) Multisegmental Aδ- and C-fiber input to neurons in lamina I and the lateral spinal nucleus. J Neurosci 30:2384–2395. 10.1523/JNEUROSCI.3445-09.2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rudomin P, Schmidt RF (1999) Presynaptic inhibition in the vertebrate spinal cord revisited. Exp Brain Res 129:1–37. 10.1007/s002210050933 [DOI] [PubMed] [Google Scholar]
- Russo RE, Delgado-Lezama R, Hounsgaard J (2000) Dorsal root potential produced by a TTX-insensitive micro-circuitry in the turtle spinal cord. J Physiol 528 Pt 1:115–122. 10.1111/j.1469-7793.2000.00115.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Safronov BV, Pinto V, Derkach VA (2007) High-resolution single-cell imaging for functional studies in the whole brain and spinal cord and thick tissue blocks using light-emitting diode illumination. J Neurosci Methods 164:292–298. 10.1016/j.jneumeth.2007.05.010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Segev I (1990) Computer study of presynaptic inhibition controlling the spread of action potentials into axonal terminals. J Neurophysiol 63:987–998. 10.1152/jn.1990.63.5.987 [DOI] [PubMed] [Google Scholar]
- Spike RC, Puskár Z, Andrew D, Todd AJ (2003) A quantitative and morphological study of projection neurons in lamina I of the rat lumbar spinal cord. Eur J Neurosci 18:2433–2448. 10.1046/j.1460-9568.2003.02981.x [DOI] [PubMed] [Google Scholar]
- Szucs P, Pinto V, Safronov BV (2009) Advanced technique of infrared LED imaging of unstained cells and intracellular structures in isolated spinal cord, brainstem, ganglia and cerebellum. J Neurosci Methods 177:369–380. 10.1016/j.jneumeth.2008.10.024 [DOI] [PubMed] [Google Scholar]
- Szucs P, Luz LL, Lima D, Safronov BV (2010) Local axon collaterals of lamina I projection neurons in the spinal cord of young rats. J Comp Neurol 518:2645–2665. 10.1002/cne.22391 [DOI] [PubMed] [Google Scholar]
- Szucs P, Luz LL, Pinho R, Aguiar P, Antal Z, Tiong SY, Todd AJ, Safronov BV (2013) Axon diversity of lamina I local-circuit neurons in the lumbar spinal cord. J Comp Neurol 521:2719–2741. 10.1002/cne.23311 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thanawala MS, Regehr WG (2013) Presynaptic calcium influx controls neurotransmitter release in part by regulating the effective size of the readily releasable pool. J Neurosci 33:4625–4633. 10.1523/JNEUROSCI.4031-12.2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Todd AJ (2010) Neuronal circuitry for pain processing in the dorsal horn. Nat Rev Neurosci 11:823–836. 10.1038/nrn2947 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Willis WD Jr (1999) Dorsal root potentials and dorsal root reflexes: a double-edged sword. Exp Brain Res 124:395–421. [DOI] [PubMed] [Google Scholar]
- Willis WD, Coggeshall RE (2004) Sensory mechanisms of the spinal cord, Ed 3. New York: Kluwer Academic/Plenum Publishers. [Google Scholar]
- Yoon MS, Mueller D, Hansen N, Poitz F, Slomke M, Dommes P, Diener HC, Katsarava Z, Obermann M (2010) Prevalence of facial pain in migraine: a population-based study. Cephalalgia 30:92–96. 10.1111/j.1468-2982.2009.01899.x [DOI] [PubMed] [Google Scholar]
- Yu HH, Avery JK (1974) Primary afferent depolarization: direct evidence in the trigeminal system. Brain Res 75:328–333. 10.1016/0006-8993(74)90756-2 [DOI] [PubMed] [Google Scholar]
- Zimmerman AL, Kovatsis EM, Pozsgai RY, Tasnim A, Zhang Q, Ginty DD (2019) Distinct modes of presynaptic inhibition of cutaneous afferents and their functions in behavior. Neuron 102:420–434.e8. 10.1016/j.neuron.2019.02.002 [DOI] [PMC free article] [PubMed] [Google Scholar]