Abstract
The presence of the multidrug efflux pump SmeDEF was assessed in a collection of clinical isolates of Stenotrophomonas maltophilia. All isolates encoded this pump, as demonstrated by PCR. Forty-seven percent of the strains overproduced a protein of the same size that was immunoreactive against an anti-SmeF antibody, and 33% overexpressed the gene semD when they were tested by reverse transcription-PCR. A correlation between smeDEF overexpression and antibiotic resistance was observed.
Stenotrophomonas maltophilia is an opportunistic pathogen that has been associated with different human pathologies (5) and that is considered an archetype of antibiotic-resistant bacteria. It has been demonstrated that S. maltophilia produces beta-lactamases (10, 13, 14), aminoglycoside acetyltransferases (7), and erythromycin-inactivating enzymes (1), with an obvious role in the antibiotic resistance phenotype of this bacterial species. Like all bacterial species studied so far (9, 11), S. maltophilia probably contains several multidrug resistance (MDR) efflux pumps, as demonstrated by the analysis of multidrug-resistant mutants that can be obtained upon antibiotic selective pressure (15). In a recent article, we have described smeDEF, the first MDR pump so far cloned and characterized in this bacterial species (2). In the present work, we have analyzed both the presence and the levels of expression of the SmeDEF pump in a collection of clinical isolates of S. maltophilia and correlated the results of those analyses with those obtained from the study of the susceptibilities of such isolates to antibiotics.
The clinical strains of S. maltophilia used in the present work were isolated from different sources at the Hospital of Mostoles (Madrid, Spain) (Table 1). S. maltophilia strain D457R is a single-step multidrug-resistant spontaneous mutant that is derived from S. maltophilia D457 and that overexpresses the SmeDEF multidrug efflux system (3). Bacteria were routinely cultured at 37°C in Luria-Bertani medium (4). The MICs of antibiotics were determined in Mueller-Hinton agar (4) by the E-test method (AB Biodisk), according to the manufacturer's instructions.
TABLE 1.
Clinical strains of S. maltophilia used in the present work
| Strain | Isolation date | Sample |
|---|---|---|
| D457 | January 1992 | Bronchial aspirate |
| C048 | November 1990 | Urine |
| C357 | March 1991 | Urine |
| D388 | December 1991 | Urine |
| E301 | October 1992 | Urine |
| E539 | January 1993 | Pus from a wound |
| E729 | March 1993 | Urine |
| E759 | March 1993 | Sputum |
| E824 | April 1993 | Blood |
| E923 | June 1993 | Sputum |
| E999 | July 1993 | Respiratory secretion |
| F227 | November 1993 | Blood |
| F375 | January 1994 | Blood |
| F861 | August 1994 | Sputum |
| G51 | November 1994 | Blood |
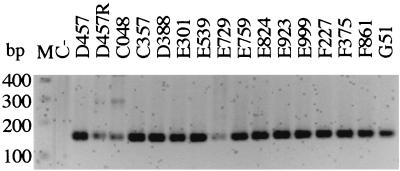
To assess the presence of the smeDEF efflux system among S. maltophilia isolates, chromosomal DNA (obtained with the Genome DNA Kit; Bio 101) from 15 clinical strains obtained from different clinical sources was subjected to PCR amplification with primers specific for the smeD gene. An internal fragment of 150 bp from the smeD gene was amplified with primers 1 (5′-CCAAGAGCCTTTCCGTCAT-3′) and 2 (5′-TCTCGGACTTCAGCGTGAC-3′). A total of 100 ng of chromosomal DNA was used as the template for each PCR. The reaction mixture (50 μl) contained each deoxynucleotide (dCTP, dGTP, dATP and dTTP) at a concentration of 0.2 mM, 0.5 μM each primer, 1.5 mM MgCl2, 10 mM Tris-HCl (pH 8.3), 50 mM KCl, and 1.0 U of Taq DNA polymerase. The mixture was heated at 94°C for 90 s, followed by 35 cycles of 30 s at 94°C, 60 s at 58°C, and 90 s at 72°C and, finally, one 10-min extension step at 72°C before the end of the reaction. PCR products were run on 2.0% agarose gels and stained with ethidium bromide. Figure 1 shows that a PCR product of the expected size was detected in all strains analyzed. This demonstrates that the smeDEF system was ubiquitously found in S. maltophilia isolates.
FIG. 1.
Presence of the smeDEF efflux pump in the genomes of clinical isolates of S. maltophilia. Lane M, molecular size markers; lane C−, negative control, for which the same PCR reaction was performed but without the addition of DNA. A band of the predicted size was observed in all isolates.
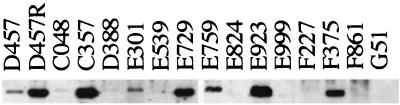
It has been described that overexpression of the outer membrane protein SmeF, the gene of which is a part of the smeDEF operon, is associated with increased levels of resistance for in vitro-selected multidrug-resistant mutants of S. maltophilia (2, 3). To test whether overexpression of the smeDEF operon was also associated with increased levels of resistance in clinical isolates of S. maltophilia, we analyzed both the expression of the SmeF protein by Western blotting and smeD RNA by reverse transcription-PCR (RT-PCR). Whole-cell extracts from clinical S. maltophilia isolates were electrophoresed on sodium dodedcyl sulfate (SDS)-polyacrylamide gels and immunoblotted as described previously with a polyclonal antibody raised against the outer membrane efflux component of SmeDEF, SmeF (2). As shown in Fig. 2, an immunoreactive band of the same size as SmeF was detected in 7 of 15 clinical isolates (strain D457R is not a clinical isolate but is a mutant derivative that has been included in the analysis as a control).
FIG. 2.
Expression of SmeF by clinical isolates of S. maltophilia. Whole-cell protein extracts of S. maltophilia were obtained and electrophoresed in SDS–10% polyacrylamide gels. Western blot analysis was performed with an anti-SmeF antibody.
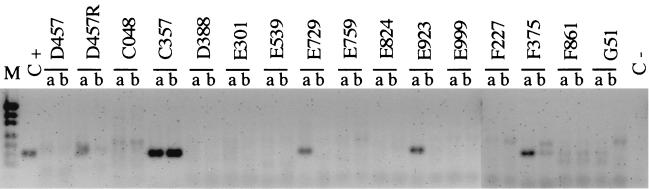
Some outer membrane components of efflux MDR pumps, such as OprM of Pseudomonas aeruginosa (8, 16) and TolC of Escherichia coli (6), are associated with more than one MDR system. In order to assess if SmeF expression was linked to the expression of the smeDEF operon, we analyzed the expression of smeD, the first gene of the operon, by RT-PCR analysis. RNA was obtained with the TriReagent-LS Kit (Molecular Research Center Inc.) according to the manufacturer's instructions. Residual DNA was eliminated with RNase-free DNase (Boehringer Mannheim), followed by acid phenol treatment and RNA precipitation with ethanol. The RNA concentration and purity were estimated by spectrophotometric analysis at 260 and 280 nm, and the RNA quality was analyzed by electrophoresis on 1% agarose gels under denaturing conditions (12). For RT-PCR analysis, a two-step reaction was carried out. First, 5 μg of RNA was incubated for 1 h at 37°C with primer 2 and avian myeloblastosis virus (AMV) reverse transcriptase (U.S. Biochemicals) to obtain cDNA. cDNA was then PCR amplified with primers 1 and 2 as described above. Amplicons were analyzed on 2% agarose gels and stained with ethidium bromide. The same reaction protocol performed for RT-PCR was carried out for each sample, but without AMV reverse transcriptase, as a control to detect DNA contamination in the RNA preparations, with negative results in all cases. The amount of smeD RNA was analyzed from bacteria grown to the early exponential or stationary growth phase because it was previously demonstrated that smeDEF expression is induced at the exponential phase of growth and is very low at the stationary phase of growth (2). As shown in Fig. 3, an RT-PCR product of the predicted size was found in 5 of the 15 isolates analyzed. All of the isolates with a positive RT-PCR result also expressed a protein immunoreactive against the anti-SmeF antiserum, indicating that expression of smeD and SmeF was linked in these strains. Two isolates (isolates E301 and E759), however, overproduced a protein that was of the same size as SmeF and that was immunoreactive against the SmeF antibody, but a RT-PCR band was not detected in our analysis. Two possibilities might explain these results: (i) the immunoreactive band detected in E301 and E759 was SmeF, but its expression was associated with other efflux systems in these strains, so that SmeF was expressed but smeD was not; (ii) the immunoreactive band detected in E301 and E759 was not SmeF but another protein which shares some structural features with SmeF and which is then recognized with the polyclonal anti-SmeF antibody, so that neither SmeF nor smeD was expressed in these isolates. In any case, it is thus clear that smeDEF is not expressed by these two strains.
FIG. 3.
Expression of smeDEF RNA by clinical isolates of S. maltophilia. The expression of smeDEF was analyzed by RT-PCR. RNAs obtained at the exponential (lanes a) and stationary (lanes b) growth phases were analyzed. A clear band of the predicted size was observed for strains D457R, C357, E729, E923, and F375; and a very faint band was observed for strain D457. Note that strain D457R is not a clinical isolate but a multidrug-resistant mutant derived from strain D457 (3), and it has been included in the present analysis as a control for smeDEF expression. Lane M, molecular size markers (from top to bottom, 404, 331, 242, 190, 157, 147, and 112 bp); C+, positive control, for which PCR was performed with chromosomal DNA from S. maltophilia D457 as the template; C−, negative control, for which PCR was performed without DNA.
The expression of smeDEF was correlated with the susceptibilities to antibiotics of the collection of clinical isolates described in Table 1. The MICs of tetracycline, chloramphenicol, erythromycin, and the quinolones were, overall, higher for the strains in which expression of SmeF was detectable by Western blotting and in which smeD was detectable by RT-PCR (33% of the isolates) than for the isolates that did not express the protein at detectable levels. This relationship was not observed for the MICs of amikacin or ticarcillin-clavulanic acid. Noteworthy increased levels of resistance to tetracycline, chloramphenicol, erythromycin, and the quinolones, with no effect on the MIC of amikacin or ticarcillin-clavulanic acid, were observed in the multidrug-resistant mutant S. maltophilia strain D457R (which overexpresses SmeF) compared to its isogenic wild-type S. maltophilia strain D457 (Table 2). The same substrate range was observed when smeDEF was cloned in a low-copy-number plasmid and introduced in the heterologous host E. coli (3).
TABLE 2.
Antibiotic susceptibilities of S. maltophilia clinical strains
| Strain | MIC (μg/ml)a
|
|||||||
|---|---|---|---|---|---|---|---|---|
| TET | CHL | AMK | TIC | ERY | NAL | NOR | OFX | |
| D457 | 6 | 1.5 | 24 | 4 | 48–64 | 8 | 6 | 3 |
| D457R | 16 | 12 | 16 | 6 | >256 | 128 | 64 | >32 |
| C048 | 4 | 1.5 | 6 | 0.75 | 128 | 12 | 3 | 2 |
| C357 | 32 | 96 | 4 | 24 | >256 | 128 | 256 | >32 |
| D388 | 16 | 12 | 96 | 32 | 96 | 8 | 8 | 2 |
| E301 | 12 | 4 | 3 | 16 | 128 | 12 | 3 | 1.0 |
| E539 | 8 | 4 | >256 | 32 | 96 | 12 | 4 | 2 |
| E729 | 32 | 16 | 16 | 1 | 256 | 32 | >256 | 8 |
| E759 | 16 | 16 | 24 | >256 | >256 | 12 | 8 | 4 |
| E824 | 4 | 8 | 16 | 4 | 192 | 3 | 2 | 0.38 |
| E923 | 12 | 64 | 12 | 96 | >256 | 16 | 16 | 3 |
| E999 | 12 | 12 | 256 | 8 | >256 | 16 | 8 | 3 |
| F227 | 4 | 4 | 24 | 48 | 16 | 4 | 3 | 0.38 |
| F375 | 12 | 16 | 128 | 16 | >256 | 128 | 32 | 16 |
| F861 | 3 | 2 | 32 | 96 | 24 | 4 | 2 | 0.5 |
| G51 | 6 | 3 | 12 | 192 | 96 | 16 | 2 | 1 |
TET, tetracycline; CHL, chloramphenicol; AMK, amikacin; TIC, ticarcillin-clavulanic acid; ERY, erythromycin; NAL, nalidixic acid; NOR, norfloxacin; OFX, ofloxacin.
Our data indicate that smeDEF is overexpressed by 33% of the clinical isolates analyzed in our work and suggest that its overexpression might contribute to increased levels of resistance to tetracycline, chloramphenicol, erythromycin, and quinolones in clinical isolates of S. maltophilia.
Acknowledgments
Thanks are given to Ignacio Alós for the gift of the clinical isolates used in the present work.
The present research was aided in part by grant 08.2/022/98 from CAM. A. Alonso was a recipient of a fellowship from Gobierno Vasco.
REFERENCES
- 1.Alonso A, Sanchez P, Martinez J L. Stenotrophomonas maltophilia D457R contains a cluster of genes from gram-positive bacteria involved in antibiotic and heavy metal resistance. Antimicrob Agents Chemother. 2000;44:1778–1782. doi: 10.1128/aac.44.7.1778-1782.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Alonso A, Martinez J L. Cloning and characterization of SmeDEF, a novel multidrug efflux pump from Stenotrophomonas maltophilia. Antimicrob Agents Chemother. 2000;44:3079–3086. doi: 10.1128/aac.44.11.3079-3086.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Alonso A, Martínez J L. Multiple antibiotic resistance in Stenotrophomonas maltophilia. Antimicrob Agents Chemother. 1997;41:1140–1142. doi: 10.1128/aac.41.5.1140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Atlas R M. Handbook of microbiological media. London, United Kingdom: CRC Press, Inc.; 1993. [Google Scholar]
- 5.Denton M, Kerr K G. Microbiological and clinical aspects of infection associated with Stenotrophomonas maltophilia. Clin Microbiol Rev. 1998;11:57–80. doi: 10.1128/cmr.11.1.57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Fralick J A. Evidence that TolC is required for functioning of the Mar/AcrAB efflux pump of Escherichia coli. J Bacteriol. 1996;178:5803–5805. doi: 10.1128/jb.178.19.5803-5805.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Lambert T, Ploy M C, Denis F, Courvalin P. Characterization of the chromosomal aac(6′)-Iz gene of Stenotrophomonas maltophilia. Antimicrob Agents Chemother. 1999;43:2366–2371. doi: 10.1128/aac.43.10.2366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Mine T, Morita Y, Kataoka A, Mizushima T, Tsuchiya T. Expression in Escherichia coli of a new multidrug efflux pump, MexXY, from Pseudomonas aeruginosa. Antimicrob Agents Chemother. 1999;43:415–417. doi: 10.1128/aac.43.2.415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Nikaido H. Multiple antibiotic resistance and efflux. Curr Opin Microbiol. 1998;1:516–523. doi: 10.1016/s1369-5274(98)80083-0. [DOI] [PubMed] [Google Scholar]
- 10.Paton R, Miles R S, Amyes S G. Biochemical properties of inducible beta-lactamases produced from Xanthomonas maltophilia. Antimicrob Agents Chemother. 1994;38:2143–2149. doi: 10.1128/aac.38.9.2143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Saier M H, Paulsen I T, Sliwinski M K, Pao S S, Skurray R A, Nikaido H. Evolutionary origins of multidrug and drug-specific efflux pumps in bacteria. FASEB J. 1998;12:265–274. doi: 10.1096/fasebj.12.3.265. [DOI] [PubMed] [Google Scholar]
- 12.Sambrook J, Fritsch E F, Maniatis T. Molecular cloning: a laboratory manual. 2nd ed. Cold Spring Harbor, N.Y: Cold Spring Harbor Laboratory Press; 1989. [Google Scholar]
- 13.Walsh T R, MacGowan A P, Bennett P M. Sequence analysis and enzyme kinetics of the L2 serine beta-lactamase from Stenotrophomonas maltophilia. Antimicrob Agents Chemother. 1997;41:1460–1464. doi: 10.1128/aac.41.7.1460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Walsh T R, Hall L, Assinder S J, Nichols W W, Cartwright S J, MacGowan A P, Bennett P M. Sequence analysis of the L1 metallo-beta-lactamase from Xanthomonas maltophilia. Biochim Biophys Acta. 1994;1218:199–201. doi: 10.1016/0167-4781(94)90011-6. [DOI] [PubMed] [Google Scholar]
- 15.Zhang L, Li X Z, Poole K. Multiple antibiotic resistance in Stenotrophomonas maltophilia: involvement of a multidrug efflux system. Antimicrob Agents Chemother. 2000;44:287–293. doi: 10.1128/aac.44.2.287-293.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Zhao Q, Li X Z, Srikumar R, Poole K. Contribution of outer membrane efflux protein OprM to antibiotic resistance in Pseudomonas aeruginosa independent of MexAB. Antimicrob Agents Chemother. 1998;42:1682–1688. doi: 10.1128/aac.42.7.1682. [DOI] [PMC free article] [PubMed] [Google Scholar]