Abstract
To determine if susceptibility testing of Streptococcus pneumoniae could detect those isolates that had one of the recognized mechanisms of fluoroquinolone resistance, 101 isolates were selected; the levofloxacin MIC for 28 of these isolates was ≥4 μg/ml. Only isolates with both parC and gyrA mutations or with no recognized resistance mechanisms were reliably identified by using these results. Isolates with only a parC mutation could not be detected reliably using any susceptibility testing method.
Decreased susceptibility of Streptococcus pneumoniae to fluoroquinolones has been documented (3, 8) and in one study was associated with increased fluoroquinolone use (3). Fluoroquinolones inhibit bacterial growth by trapping gyrase or topoisomerase IV on DNA as ternary drug-enzyme-DNA complexes (4).
Decreased susceptibility to fluoroquinolones may develop as a result of changes in the fluoroquinolone targets by mutations in the genes which encode ParC in topoisomerase IV and GyrA in DNA gyrase and/or by diminished drug accumulation by efflux (7, 9, 10, 17, 19, 20). Mutations in gyrB have not to date been associated with decreased susceptibility to the fluoroquinolones in S. pneumoniae, and whether mutations in parE alter the susceptibility is still controversial (1, 16, 17). Mutations in parC and gyrA occur in the quinolone resistance-determining region (QRDR) of the genes. In S. pneumoniae, the target to which the fluoroquinolone has the greatest binding affinity (primary target) depends on the fluoroquinolone (7, 9, 10, 17, 19). For levofloxacin and ciprofloxacin, the primary target is ParC. For gatifloxacin and moxifloxacin, the primary target is GyrA (18). Mutations in the primary target that result in a reduction in the affinity of the fluoroquinolone are associated with an increase in the MIC. A subsequent mutation in the original secondary target will result in a further increase in the MIC (7, 9, 10, 17, 19, 20). Laboratory identification of isolates with decreased susceptibility to fluoroquinolones is paramount for the effective treatment of patients with pneumococcal disease. Of equal importance may be the ability to detect isolates that have a fluoroquinolone resistance mechanism, even if the isolate is still susceptible, since this organism may more readily become resistant during therapy when a subsequent mutation occurs in the original secondary target. In this study, we compared the activities of ciprofloxacin and levofloxacin by broth microdilution, disk diffusion, and E-test susceptibility testing procedures against clinical isolates of S. pneumoniae for which the fluoroquinolone MICs varied in order to determine if the results could be used to reliably distinguish an isolate with one or more fluoroquinolone resistance mechanisms from one with no resistance mechanisms.
S. pneumoniae clinical isolates were from the Canadian Bacterial Surveillance Network; their collection and identification have been detailed elsewhere (1, 3). The initial in vitro broth microdilution testing and subsequent disk diffusion testing of ciprofloxacin (using a 5-μg disk; Oxoid, Nepean, Ontario, Canada) and levofloxacin (using a 5-μg disk; Becton Dickinson and Co., Cockeysville, Md.) were done according to National Committee for Clinical Laboratory Standards (NCCLS) (3, 12, 13). NCCLS disk diffusion testing breakpoints for levofloxacin are as follows: susceptible, ≥17 mm; intermediate >13 and <17 mm; and resistant, ≤13 mm. MIC breakpoints are as follows: susceptible, ≤2 μg/ml; intermediate, 4 μg/ml; and resistant, ≥8 μg/ml. The antimicrobials were provided by their respective manufacturers. The E-test was performed according to the manufacturer's instructions. Following storage at −70°C, isolates were thawed and subcultured onto blood agar (Oxoid) twice before susceptibility testing was performed. A total of 101 clinical isolates were selected for analysis; these included 75 isolates for which the ciprofloxacin MIC was ≥4 μg/ml (MICs of 64 μg/ml for 6 isolates, 32 μg/ml for 4, 16 μg/ml for 12, 8 μg/ml for 12, and 4 μg/ml for 41), 14 for which the MIC was 2 μg/ml, 3 for which the MIC was 1 μg/ml, and 9 for which the MIC was <1 μg/ml.
Crude cell lysates were used as DNA templates for PCR. Briefly, a loopful of overnight growth on Columbia nutrient agar supplemented with 5% sheep blood was suspended in 100 μl of lysis buffer (100 mM NaCl, 10 mM Tris-HCl [pH 8.3], 1 mM EDTA, 1% Triton X-100) and boiled for 10 min; 10 μl of the supernatant was used as a template in a 50-μl reaction volume. QRDR regions of parC and gyrA of all strains were amplified by PCR. Primers were based on published sequences (14, 16). Amplification products were purified using either the QIAquick PCR purification kit (Qiagen Inc., Mississauga, Ontario, Canada) or the Concert Rapid PCR Purification (Life Technologies, Burlington, Ontario, Canada).
DNA sequencing was performed by ABI Prism Big Dye terminator cycle sequencing with the ABI 377 automated sequencer (PE Applied Biosystems, Mississauga, Ontario, Canada). Nucleotide and protein sequence comparisons were performed using the multiple alignment sequence function of Vector NTI Suite software (InforMax Inc., Bethesda, Md.). The GenBank accession numbers for wild-type sequences used for comparison purposes were Z67739 for parC and parE (15), AB010387 for gyrA, and Z67740 for gyrB (11).
Isolates were examined for active efflux by agar dilution on Mueller-Hinton agar containing 5% sheep blood in the presence of ciprofloxacin with or without 10 mg of the alkaloid reserpine (Sigma Chemical Co., St. Louis, Mo.) per ml (2). Strains for which the ciprofloxacin MIC decreased at least fourfold in the presence of reserpine were considered positive for active efflux.
Using two-by-two tables, a wide range of MICs or disk diffusion diameters were tested to find the threshold with the highest sensitivity and specificity for detecting resistance mechanisms. For positive and negative predictive values, the estimated prevalence of these resistance mechanisms in the 7,551 isolates from the Canadian Bacterial Surveillance Network was used (3).
There were 27 isolates with mutations in the QRDR of both parC and gyrA, 3 of which were also positive for efflux. Fifty isolates had an isolated parC mutation, and none had an isolated gyrA mutation. Of those with a parC mutation but no gyrA mutation, 14 were also positive for efflux. Finally, there were 24 isolates with neither a parC nor a gyrA mutation, 9 of which were positive for efflux, leaving 16 with no identified resistance mechanisms.
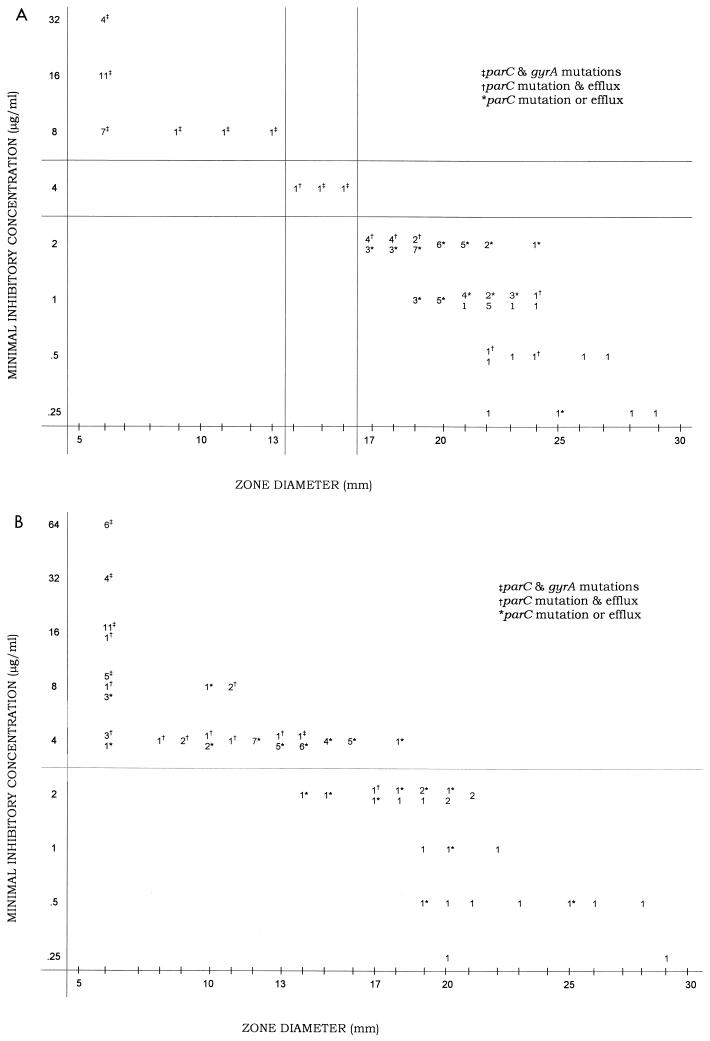
Figure 1A graphs the broth microdilution MICs versus the disk diffusion diameters for levofloxacin, with the breakpoints for each shown. There were no major, or very major, errors. Figure 1B graphs the broth microdilution MICs versus the disk diffusion diameters for ciprofloxacin. Table 1 summarizes the results for the three susceptibility testing methods. The NCCLS breakpoints for levofloxacin classified the 27 isolates with both parC and gyrA mutations as intermediate or resistant. There was only one isolate with a parC mutation but no gyrA mutation that was nonsusceptible (intermediate susceptibility). Interestingly, it did have a very active efflux mechanism (the ciprofloxacin MIC for this isolate decreased by 16-fold in the presence of reserpine). Other than for this isolate, efflux did not contribute to an increase in the levofloxacin MIC that resulted in a strain being classified as nonsusceptible.
FIG. 1.
Scattergram showing correlations among levofloxacin (A) and ciprofloxacin (B) MICs and zone diameters around 5-μg disks. Numbers are the datum points at each location (101 isolates tested).
TABLE 1.
Isolates with and without gyrA and/or parC mutations and their susceptibility category classification according to NCCLS breakpoints for levofloxacin using three different testing methods
| Mutation(s) (no. of isolates) | No. of isolates with indicated test results
|
||||||||
|---|---|---|---|---|---|---|---|---|---|
| Broth microdilution MIC (μg/ml) of:
|
E-test MIC (μg/ml) of:
|
Disk diffusion (mm) of:
|
|||||||
| ≤2 | 4 | ≥8 | ≤2 | 4 | ≥8 | ≥17 | 14–16 | ≤13 | |
| parC and gyrA (27) | 0 | 2 | 25 | 0 | 4 | 23 | 0 | 2 | 25 |
| parC only (50) | 49 | 1 | 0 | 49 | 1 | 0 | 49 | 1 | 0 |
| None (24) | 24 | 0 | 0 | 24 | 0 | 0 | 24 | 0 | 0 |
Table 2 summarizes the test characteristics of the three different susceptibility testing methods for detecting isolates with mutations in both parC and gyrA. MIC and disk diffusion values that favored a higher sensitivity were chosen. The prevalence used to determine the positive and negative predictive value is 0.4% (27 of 7,551 isolates). For detecting isolates with both gyrA and parC mutations, the levofloxacin E-test and disk diffusion tests were the most sensitive and specific. Using the levofloxacin E-test, a MIC of >2 μg/ml had a sensitivity of 100% and a specificity of 99%. The levofloxacin disk diffusion test had the same sensitivity and specificity using a disk diameter of less than 17 mm. These MIC and disk diffusion values are the same as the NCCLS breakpoints.
TABLE 2.
Test characteristics of three susceptibility testing methods for detecting isolates with both parC and gyrA mutations
| Test and resultsa | Sensitivity (%) | Specificity (%) | PPVb (%) | NPVc (%) |
|---|---|---|---|---|
| Broth microdilution | ||||
| Cipro MIC > 4 μg/ml | 96 | 89 | 3 | 100 |
| Levo MIC > 2 μg/ml | 100 | 99 | 29 | 100 |
| E-test | ||||
| Cipro MIC > 16 μg/ml | 92 | 89 | 4 | 100 |
| Levo MIC > 2 μg/ml | 100 | 99 | 29 | 100 |
| Disk diffusion | ||||
| Cipro; diameter < 8 mm | 96 | 88 | 3 | 100 |
| Levo; diameter < 17 mm | 100 | 99 | 29 | 100 |
Cipro, ciprofloxacin; levo, levofloxacin.
PPV, positive predictive value.
NPV, negative predictive value.
Table 3 considers only the levofloxacin-susceptible isolates (levofloxacin MIC of ≤2 μg/ml, n = 73) and summarizes the test characteristics of the three different susceptibility testing methods for detecting isolates with only a mutation in parC from among these isolates. No one MIC or disk diffusion value had a high sensitivity and specificity. For all testing methods, the negative predictive value was 100%. The prevalence used to calculate positive and negative predictive values was 1.0% (77 of 7,551 isolates). For detecting isolates with either a parC mutation or an active efflux mechanism among susceptible isolates, the test characteristics could be slightly improved. For example, in the best case, a disk with a diameter of <22 mm for levofloxacin had a sensitivity of 79% and a specificity of 93% (other data not shown).
TABLE 3.
Test characteristics of three susceptibility testing methods for detecting isolates with parC mutations (n = 49) from among all 73 isolates susceptible to levofloxacin
| Test and resultsa | Sensitivity (%) | Specificity (%) | PPVb (%) | NPVc (%) |
|---|---|---|---|---|
| Broth microdilution | ||||
| Cipro MIC ≥ 4 μg/ml | 78 | 63 | 2 | 100 |
| Levo MIC ≥ 2 μg/ml | 69 | 88 | 6 | 100 |
| E-test | ||||
| Cipro MIC ≥ 2 μg/ml | 88 | 58 | 2 | 100 |
| Levo MIC ≥ 1 μg/ml | 90 | 50 | 2 | 100 |
| Disk diffusion | ||||
| Cipro; diameter < 20 mm | 94 | 50 | 2 | 100 |
| Levo; diameter < 20 mm | 49 | 96 | 11 | 100 |
Cipro, ciprofloxacin; levo, levofloxacin.
PPV, positive predictive value.
NPV, negative predictive value.
S. pneumoniae isolates with mutations in the QRDR regions of both parC and gyrA genes are often not only resistant to levofloxacin but also nonsusceptible to moxifloxacin and gatifloxacin (3). We were able to reliably predict those isolates with both parC and gyrA mutations as well as those with no resistant mechanisms. However, it may be more important clinically and epidemiologically to be able to detect those isolates with first-step mutations. The likelihood of a wild-type isolate acquiring mutations in both of these genes simultaneously is on the order of 1 in 1012 to 1 in 1014 (6). However, if an infecting organism has a mutation in one of the targets, the likelihood of a mutation occurring in the other target is on the order of 1 in 106 to 1 in 107 (6). In pneumococcal pneumonia, the bacterial burden is >1010, making the likelihood of a spontaneous mutation in the second target high (5). Unfortunately, no test had both a sensitivity and a specificity adequate for reliably identifying susceptible isolates with single mutations. Future studies should address this deficiency.
The large number of clinical isolates in our study enhances the validity of our findings with recognized fluoroquinolone-resistant mechanisms. A weakness of the study is that all the mechanisms of resistance were selected for during a time when the only fluoroquinolones in clinical use had preferential affinity for ParC over GyrA (i.e., norfloxacin, ofloxacin, ciprofloxacin, and levofloxacin). To further validate our findings, it may be necessary to assess clinical pneumococcal isolates with fluoroquinolone resistance mechanisms that have been selected for both by agents that have preferential ParC affinity and the newer agents with preferential GyrA affinity (i.e., moxifloxacin and gatifloxacin). Finally, these results are based primarily on a selected collection of isolates for most of which the ciprofloxacin MICs were ≥4 μg/ml, and the prevalence values could only be estimated from this small collection.
Acknowledgments
This study was supported in part by a grant from the Canadian Bacterial Diseases Network.
REFERENCES
- 1.Bast D J, Low D E, Duncan C L, Kilburn L, Mandell L A, Davidson R J, de Azavedo J C. Fluoroquinolone resistance in clinical isolates of Streptococcus pneumoniae: contributions of type II topoisomerase mutations and efflux to levels of resistance. Antimicrob Agents Chemother. 2000;44:3049–3054. doi: 10.1128/aac.44.11.3049-3054.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Brenwald N P, Gill M J, Wise R. Prevalence of a putative efflux mechanism among fluoroquinolone-resistant clinical isolates of Streptococcus pneumoniae. Antimicrob Agents Chemother. 1998;42:2032–2035. doi: 10.1128/aac.42.8.2032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Chen D, McGeer A, de Azavedo J C, Low D E The Canadian Bacterial Surveillance Network. Decreased susceptibility of Streptococcus pneumoniae to fluoroquinolones in Canada. N Engl J Med. 1999;341:233–239. doi: 10.1056/NEJM199907223410403. [DOI] [PubMed] [Google Scholar]
- 4.Fournier B, Zhao X, Lu T, Drlica K, Hooper D C. Selective targeting of topoisomerase IV and DNA gyrase in Staphylococcus aureus: different patterns of quinolone-induced inhibition of DNA synthesis. Antimicrob Agents Chemother. 2000;44:2160–2165. doi: 10.1128/aac.44.8.2160-2165.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Frisch A W, Tripp J W, Barrett C D, Pidgeon B E. The specific polysaccharide content of pneumonic lungs. J Exp Med. 1942;76:505–510. doi: 10.1084/jem.76.6.505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Fukuda H, Hiramatsu K. Primary targets of fluoroquinolones in Streptococcus pneumoniae. Antimicrob Agents Chemother. 1999;43:410–412. doi: 10.1128/aac.43.2.410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Gootz T D, Zaniewski R, Haskell S, Schmieder B, Tankovic J, Girard D, Courvalin P, Polzer R J. Activity of the new fluoroquinolone trovafloxacin (CP-99,219) against DNA gyrase and topoisomerase IV mutants of Streptococcus pneumoniae selected in vitro. Antimicrob Agents Chemother. 1996;40:2691–2697. doi: 10.1128/aac.40.12.2691. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ho P L, Que T L, Tsang D N, Ng T K, Chow K H, Seto W H. Emergence of fluoroquinolone resistance among multiply resistant strains of Streptococcus pneumoniae in Hong Kong. Antimicrob Agents Chemother. 1999;43:1310–1313. doi: 10.1128/aac.43.5.1310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Janoir C, Zeller V, Kitzis M D, Moreau N J, Gutmann L. High-level fluoroquinolone resistance in Streptococcus pneumoniae requires mutations in parC and gyrA. Antimicrob Agents Chemother. 1996;40:2760–2764. doi: 10.1128/aac.40.12.2760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Jorgensen J H, Weigel L M, Ferraro M J, Swenson J M, Tenover F C. Activities of newer fluoroquinolones against Streptococcus pneumoniae clinical isolates including those with mutations in the gyrA, parC, and parE loci. Antimicrob Agents Chemother. 1999;43:329–334. doi: 10.1128/aac.43.2.329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Munoz R, Bustamante M, De La Campa A G. Ser-127-to-Leu substitution in the DNA gyrase B subunit of Streptococcus pneumoniae is implicated in novobiocin resistance. J Bacteriol. 1995;177:4166–4170. doi: 10.1128/jb.177.14.4166-4170.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.National Committee for Clinical Laboratory Standards. Methods for dilution antimicrobial susceptibility tests for bacteria that grow aerobically. Approved standard M7–A5. NCCLS document M100–S10/M7. Wayne, Pa: National Committee for Clinical Laboratory Standards; 2000. [Google Scholar]
- 13.National Committee for Clinical Laboratory Standards. Performance standards for antimicrobial disk susceptibility tests. Approved standard. NCCLS document M2–A7. Wayne, Pa: National Committee for Clinical Laboratory Standards; 2000. [Google Scholar]
- 14.Pan X S, Ambler J, Mehtar S, Fisher L M. Involvement of topoisomerase IV and DNA gyrase as ciprofloxacin targets in Streptococcus pneumoniae. Antimicrob Agents Chemother. 1996;40:2321–2326. doi: 10.1128/aac.40.10.2321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Pan X S, Fisher L M. Cloning and characterization of the parC and parE genes of Streptococcus pneumoniae encoding DNA topoisomerase IV: role in fluoroquinolone resistance. J Bacteriol. 1996;178:4060–4069. doi: 10.1128/jb.178.14.4060-4069.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Perichon B, Tankovic J, Courvalin P. Characterization of a mutation in the parE gene that confers fluoroquinolone resistance in Streptococcus pneumoniae. Antimicrob Agents Chemother. 1997;41:1166–1167. doi: 10.1128/aac.41.5.1166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Pestova E, Beyer R, Cianciotto N P, Noskin G A, Peterson L R. Contribution of topoisomerase IV and DNA gyrase mutations in Streptococcus pneumoniae to resistance to novel fluoroquinolones. Antimicrob Agents Chemother. 1999;43:2000–2004. doi: 10.1128/aac.43.8.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Pestova E, Millichap J J, Noskin G A, Peterson L R. Intracellular targets of moxifloxacin: a comparison with other fluoroquinolones. J Antimicrob Chemother. 2000;45:583–590. doi: 10.1093/jac/45.5.583. [DOI] [PubMed] [Google Scholar]
- 19.Tankovic J, Perichon B, Duval J, Courvalin P. Contribution of mutations in gyrA and parC genes to fluoroquinolone resistance of mutants of Streptococcus pneumoniae obtained in vivo and in vitro. Antimicrob Agents Chemother. 1996;40:2505–2510. doi: 10.1128/aac.40.11.2505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Zhanel G G, Walkty A, Vercaigne L, Karlowsky J A, Embil J, Gin A S, Hoban D J. The new fluoroquinolones: a critical review. Can J Infect Dis. 1999;10:207–238. doi: 10.1155/1999/378394. [DOI] [PMC free article] [PubMed] [Google Scholar]