Graphical abstract
Keywords: Vertical diffusion cell, Franz cell, In vitro release test, Transdermal patch, Semi-solid formulation, Quality control
Abstract
The vertical diffusion cell is an in vitro laboratory device for the study of drug release and permeation of semi-solid topical formulations and topical patches. Both static and dynamic versions of the diffusion cell are used in practice, the operation of which can be automated. The device is available at a reasonable cost for smaller, mainly transdermal patches, the amount of sampling fluid is replenished at the same time as the sample, in the same amount as the amount sampled without automation of the system, and simultaneous replenishment of acceptor fluid by hydrostatic pressure helps keeping the acceptor chamber bubble-free.
Specifications table
| Hardware name | Novel modified vertical diffusion cell |
|---|---|
| Subject area | Medical (pharmaceutical science) |
| Hardware type | Lab-scale pharmaceutical release testing unit operation system |
| Closest commercial analog | Vertical diffusion cell (Franz cell) |
| Open source license | Creative Commons Attribution-ShareAlike 4.0 International License (CC BY-SA 4.0) |
| Cost of hardware | 1400 Euro |
| Source file repository | http://doi.org/10.17605/OSF.IO/329TP |
Hardware in context
In vitro release studies of topical, semi-solid formulations are essential for formulation development, composition, manufacturing optimization, and batch to batch validation of the manufacturing process and batch testing [1]. The study also plays an important role in verifying the therapeutic efficacy of a drug - its therapeutic equivalence for generic products - and in establishing in vitro - in vivo correlation (IVIVC) [2], [3]. In this approach, therefore, the test method and apparatus used serve academic research, monitoring pharmaceutical development, and product quality control. The most commonly used instrument for in vitro release of the drug from dermal and other semi-solid formulations is the Franz vertical diffusion cell for both release (in vitro release test, IVRT) and permeation (in vitro permeation test, IVPT) tests [4]. The first description of the method is named after T. J. Franz [5], and has been used primarily to study dermal, transdermal systems, ophthalmic preparations, skincare products, and skin permeation.
There is no uniformly accepted and harmonized pharmacopoeial method for testing semi-solid preparations [6]. According to the American Association of Pharmaceutical Scientists (AAPS) and the Food and Drug Administration (FDA) guidelines, a Franz cell with different membranes is considered the most suitable device for the purpose but does not specify uniform requirements for drug release conditions [7], [8], [9]. Depending on the properties of the product, the targeted therapeutic indication, the biopharmaceutical quality of the active substance, several alternative devices are now available and can be divided into three main groups according to their principle of operation: a) Franz vertical diffusion cell [10], b) Immersion Cell Apparatus [11], c) Flow-Through Cell [12] (Fig. 1). The Franz cell [13], [14] is also used in most cases to study the drug release of transdermal patches, but the so-called also paddle method [15], [16]. The advantage of the Franz cell is that it can be used as a stand-alone device, but the system can be partially automated [17] and even 6 vessels can also be operated in parallel with the help of the programmed measurement protocol.
Fig. 1.
Schematic figure of the (a) Model A and (b) Model B versions of vertical diffusion cell.
In general, however, the bubble-free filling of the cells or keeping them free of bubbles during sampling can be a problem [18] and can ruin the given measurement cycle, thus degrading the reproducibility and representativeness of the assay.
The principle of operation of the proposed redesigned diffusion cell is similar to that of the currently available Franz cell, which consists of its main components of an acceptor and a donor component, where an inert mixer provides the mechanical agitation required for drug release. The advantage of the design of the static vertical diffusion device presented for the examination of topical, transdermal patches is that the amount of liquid sample is replenished simultaneously with the sample, the same amount as taken without automating the system so that the acceptor side space remains bubble-free. The increased acceptor side volume allows for a large number of samples without an excessive decrease in concentration. The thermocouple, which can also be placed in the acceptor-side liquid, allows the temperature of the release medium to be controlled directly without significantly disturbing the mixing by the geometry of the measuring unit. The device, similar to a traditional Franz cell, is easy to assemble and clean.
Hardware description
The modified vertical diffusion device consists essentially of three main glass components: a 90 ml donor chamber from the external environment, a drug release and diffusion receptor chamber, and a 25 ml equalization buffer to replenish the receptor fluid (Fig. 1a). In the supplemented embodiment, the device is expanded with two additional components (five in total): a double-walled tank of the primary water jacket designed to ensure a constant temperature of the receptor fluid and a thermocouple sensor that controls the temperature (Fig. 1b). The former component in the case of a simple technical design is a glass vessel also containing water as a heating medium, into which the diffusion cell or the thermostatic sensor is immersed.
A ball joint clip is used to fix the donor chamber and the receptor chamber enclosing the sample to be tested and the semipermeable membrane in opposite directions. For studies, a cellulose-based membrane is usually used, but in the case of transdermal patches that come into contact with wound secretions during the application, other specific, e.g. heat-crosslinked polyvinyl alcohol membrane, can also be used [19].
On the wall of the receptor chamber, there is a threaded nozzle with a liquid-tight membrane/septum and a cap that can be sampled, and a threaded nozzle that can also be closed with a water-tight seal, through which the temperature control sensor is immersed in the receptor fluid. The latter threaded nozzle can be closed with a glass tap in the case of a simple technical design and helps to fill the system without bubbles, which is not suitable for receiving the temperature control unit.
There is an additional (three in total) abrasive-tipped nozzle on the receptor reservoir wall. In addition, a cylindrical abrasive-tipped dropping additional funnel containing the receptor fluid is attached as a buffer reservoir. There are glass taps on the drip funnel and the inlet nozzle to temporarily separate the liquid portions from each other as needed. The design of the custom-designed diffusion cell is not only complementary to the commonly used Franz cell in addition to the buffer tank [20], but also in that the receptor chamber has a larger volume and spherical symmetry, while the Franz cell is cylindrically symmetrical and of 5.7–6.0 ml volume (Fig. 2).
Fig. 2.
Schematic illustration for comparison of (a) Franz cell apparatus and (b) the novel modified vertical diffusion cell apparatus.
The device also includes a magnetic stirrer bar and a 0.5 ml or 1.0 ml sampling Hamilton syringe. The background system operating the diffusion cell is not discussed in detail in this publication, however, it should be noted that the agitation of the acceptor fluid is provided by a magnetic stirrer. In the simple technical design, the tempering medium of the diffusion cell is directly the water-filled glass vessel / beaker into which the cell is immersed and which is heated and controlled by an underlying heated magnetic stirrer while stirring the acceptor fluid. The temperature control unit then contacts the water jacket. In the case of the supplementary technical design, the diffusion cell is immersed directly in the double-walled glass vessel in which the secondary water jacket is located. The heat exchange required for the secondary water jacket is provided by the primary water jacket circulating in the double-walled vessel. Depending on the equipment of the laboratory, there are two possible solutions for heating and circulating the primary water stream: 1) using a circulating water bath, where the liquid is circulated, heated and the temperature is controlled by the water bath; thus, a nozzle designed for this purpose on the cell wall can be easily closed), 2) a peristaltic pump provides the circulation of the primary tempering liquid. A separate heated magnetic stirrer heats the liquid in the primary circuit of a larger beaker filled with water. It is possible to control the temperature by measuring the temperature of the acceptor fluid.
The modified vertical diffusion device (Model A) consists essentially of three main glass components: a 90 ml donor chamber from the external environment, a drug release and diffusion receptor chamber, and a 25 ml receptor fluid replacement volume equalization buffer tank (Fig. 1a). In the supplemented design (Model B), the device is expanded with two additional components (five in total): a double-walled tank of the primary water jacket designed to ensure a constant temperature of the receptor fluid and a thermocouple sensor that controls the temperature (Fig. 1b). The former component in the case of a simple technical design is a glass vessel also containing water as a heating medium, into which the diffusion cell or the thermostatic sensor is immersed. A ball joint clip is used to secure the donor chamber and the receptor chamber enclosing the sample to be tested and the semipermeable membrane in opposite directions. For tests, a cellulose-based membrane is usually used, but in the case of transdermal patches that come into contact with wound secretions during the application, other specific, e.g. heat-crosslinked polyvinyl alcohol membrane, can also be used [21]. Depending on the type of sample to be tested, a silicone insert (40 mm × 40 mm × 3 mm) and a glass plate (30 mm × 30 mm × 2 mm) can be inserted between the donor chamber and the receptor chamber. On the wall of the receptor chamber, there is a threaded nozzle with a liquid-tight membrane/septum and a cap that can be sampled, and a threaded nozzle that can also be closed with a water-tight seal, through which the temperature control sensor is immersed in the receptor fluid. The latter threaded nozzle can be closed with a glass pin in the case of a simple technical design and helps to fill the system without bubbles, which is not suitable for receiving the temperature control unit. There is an additional (three in total) abrasive-tipped nozzle on the receptor reservoir wall. A cylindrical abrasive-tipped dropping additional funnel containing the receptor fluid is attached as a buffer reservoir.
There are glass taps on the drip funnel and the inlet nozzle to temporarily separate the liquid parts from each other as needed. The design of the custom-designed diffusion cell differs not only in the addition of the buffer tank compared to the commonly used Franz cell [20] but also in that the receptor chamber has a larger volume and spherically symmetrical geometry, while the Franz cell is cylindrically symmetrical and of 5.7–6.0 ml volume (Fig. 2). The device also includes a magnetic stirrer bar and a 0.5 ml or 1.0 ml sampling Hamilton syringe. The background system operating the diffusion cell is not discussed in detail in this publication; however, it should be noted that a magnetic stirrer provides the agitation of the acceptor fluid. In the simple technical design, the tempering medium of the diffusion cell is directly the water-filled glass vessel / beaker into which the cell is immersed and heated and controlled by an underlying heated magnetic stirrer while stirring the acceptor fluid. The temperature control unit then contacts the water jacket. In the case of the supplementary technical design, the diffusion cell is immersed directly in the double-walled glass vessel in which the secondary water jacket is located. Thus, the heat exchange required for the secondary water jacket is provided by the primary water jacket circulating in the double-walled vessel. Depending on the equipment of the laboratory, there are two possible solutions for heating and circulating the primary water stream: 1) using a circulating water bath, where the liquid is circulated, heated and the temperature is controlled by the water bath; thus, the nozzle formed for this purpose on the cell wall can be easily closed), 2) the circulation of the primary tempering liquid is provided by a peristaltic pump, where the liquid is heated by a separate heated magnetic stirrer in the primary circuit of a larger beaker filled with water. Then, it is possible to control the temperature by measuring the temperature of the acceptor fluid.
In summary, the modified vertical diffusion device shown can be used with the following advantages:.
-
•
The amount of fluid sampled at the time of sampling is replenished at the same time as the sample, in
-
•
exactly the same amount as the amount sampled without automation of the system.
-
•
Simultaneous replenishment of acceptor fluid by hydrostatic pressure helps keep the acceptor chamber bubble-free.
-
•
The increased acceptor side volume allows for many samples without an excessive decrease in concentration.
-
•
For ‘Model B’, temperature control based on direct temperature measurement of the acceptor fluid is possible.
-
•
Tempering of ‘Model B’ can be provided using several alternative background systems depending on the laboratory equipment.
-
•
The device, similar to a traditional Franz cell, is easy to assemble and clean.
Design files summary
The central element of the modified vertical diffusion cell is the spherically symmetrical glass acceptor chamber (in this case, the diameter of the container is 53 mm), which can be made from a three-necked spherical round-bottomed flask by a unique glass conversion. The drip funnel (buffer tank), the sampling syringe, the thermocouple, and the donor chamber can be further connected to this central element with threaded nozzles.
The main metric parameters of the ‘Model A’ and ‘Model B’ devices are shown in Fig. 3, and the technical drawing and rotatable 3D drawing are available on the online repository. Of these devices, the actual technical implementation was only for the ‘Model A’. The cell can be attached to a Bunsen stand using a simple flask holder, but in most cases, the magnetic stirrer required to stir the acceptor fluid also includes a metal rod to secure the cell. In the case of ‘Model B’, a separate unit of the apparatus is the tempering glass vessel (see Fig. 3 c, d), which is to be placed directly on the surface of the magnetic stirrer so that it does not need to be fixed separately. In the case of ‘Model B’, the length of the threaded nozzles is longer than that of the nozzles on ‘Model A’, so that they do not impede the immersion of the cell in the secondary tempering fluid and sampling can be carried out without interruption. The attachment of the nozzle to the acceptor chamber, to which the buffer tank is connected, is reinforced by a glass stiffening bridge.
Fig. 3.
Three-dimensional views of various constructions of modified vertical diffusion cell ((a) Model A, (c) Model B) and technical drawings of these devices ((b) Model A, (d) Model B).
Bill of materials summary
| Designator | Component | Number | Cost per unit -EUR | Total cost - EUR |
Source of materials | Material type |
|---|---|---|---|---|---|---|
| A | Customized spherical glass cover | 1 | 42.69 | 42.69 |
N.A. (Custom order for glass technique) |
Inorganic |
| B3 | Translucent silicon rubber sheet (978–8578) | 1 | 8.53 | 8.53 | https://int.rsdelivers.com | Polymer |
| B4 | Glass sheet (40 mm × 40 mm × 3 mm) | 1 | 5.69 | 5.69 | N.A. (Custom order for glass technique) |
Inorganic |
| C | Stainless Steel Ball Joint Clip (35/20) (NC-13799) | 1 | 16.86 | 16.86 | https://www.sciencecompany.com/ | Metal |
| D1 | Customized spherical round bottle flask | 1 | 241.91 | 241.91 | N.A. (Custom order for glass technique) |
Inorganic |
| D2/1 | Screw caps with hole, GL 45 (L994.1) | 2 | 7.15 | 14.3 | www.carlroth.com | Polymer |
| D2/2 | Seal / Septa GL 45 with PTFE coating (10 units per pack) (KCP9.1) | 2 | 54.4 | 108.8 | www.carlroth.com | Polymer |
| E | Magnetic stirring bar, PTFE, oval (L15 mm) (Z329215) | 1 | 71.15 | 71.15 | www.sigmaaldrich.com | Metal and Polymer |
| F | Hamilton® Syringe 1001LTN, 1 ml (20740-U) | 1 | 187.27 | 187.27 | www.sigmaaldrich.com | Other |
| G1 | Dropping additional funnel (25 ml) (1895002) | 1 | 78.88 | 78.88 | www.bovimex.com | Inorganic |
| H |
Tempering Beaker Made of Glass; Reaction Vessel, Cylindrical, with thermostatic jacket |
1 | 608.29 | 608.29 | www.bovimex.com | Inorganic |
| O1, O2 | Heated magnetic stirrer with thermo sensor (IKA® RET/PT100) (4216) | 1 | 535.5 | 535.5 | www.ebay.com | Non-specific |
| O4 | Perisctaltic pump | 1 | 397.85 | 397.85 | www.ebay.com | Non-specific |
| O5 | Circulating water bath (Thermo Haake® K20/C20) | 1 | 417.67 | 417.67 |
www.ebay.com |
Non-specific |
Items O1 – O5 are non-specific major parts of the operational background system.
Build instructions
’Model A’ device construction
Model A (Fig. 4): For Model A, modify the selected three-necked spherical ball (D1) of at least 50 ml (80 ml for this version, 53 mm in diameter) underlying the acceptor chamber so that the first inlet (D5) and the opposite a ground glass stopper (D4, D6), possibly a Teflon stopper, should be placed on the second inlet stub (D3). An essential instruction for glass engineering work is that it is necessary to thin the diameter of said stubs to 4–5 mm during design. It should be noted that the donor chamber and the outlet nozzles can also be formed from a simple (single-necked) spherical round-bottomed flask by also individual glass technology work, during which the pre-processed nozzles are subsequently soldered. The first inlet must be ground (D7). Glass pins D4 and D6 should be placed on the stubs so that they are approximately flush with the spherical opening of the flask. The third inlet nozzle is shorter than the previous two and has a threaded end on which a plastic closure cap (D2) with a sampling septum is placed and through which sampling takes place. When using the cell, connect the ground drip funnel (buffer tank) (G1) to the first inlet (D5), which is equipped with a glass tap (G2) between the tank and the ground. Part (A) of the cell donor chamber is a spherically shaped counterpart of the spherical neck of the round bottom flask, which is ∼ 35 mm in diameter, similar to the acceptor chamber. The donor chamber is supplied with a glass plate or cover plate (B4) 3 mm thick and 40 mm × 40 mm in area and a translucent silicon rubber sheet (B3) of the same size, which may be omitted depending on the type of test. The alignment of the donor chamber and the acceptor chamber is secured by a stainless steel ball joint clip (C). The modified vertical diffusion cell is basically a desktop device that can be easily attached to the appropriate mobile stand with a flask holder, and its position can be easily adjusted.
Fig. 4.
Photo about ‘Model A’ version of the modified vertical diffusion cell. The inset shows in more detail what is the right combination of the donor (A1) and the acceptor (D8) compartments. The figure also shows the essential parts of cell: donor chamber (A), membrane (B1), sample (B2), silicone insert (B3), glass plate or cover plate (B4) stainless steel ball joint clip (C), acceptor chamber (D1), sampling inlet (D2), second overflow inlet (D3), second inlet tap (D4), first inlet (D5), first inlet tap (D6), ground inlet (D7), spherical edge of the acceptor chamber (D8), magnetic stirrer bar (E), Hamilton syringe (F), drip funnel (buffer tank) (G1), glass tap (G2).
’Model B’ device construction
The Model B design (Fig. 3c, d) differs from the Model A design in that the inlet nozzles fitting the acceptor chambers are longer so that the cell can be surrounded by the secondary tempering fluid over a larger area and as close as possible to the test sample (higher tempering fluid levels are available). This extension ensures that the pins and joints remain above the liquid surface and that sampling and handling of the pins can be carried out without interruption. In the case of Model B, the end of the second inlet nozzle is threaded, on which the cap with the sealing ring can be placed, and through which the temperature control thermocouple is immersed in the acceptor medium. In addition, a glass bridge can be soldered between the first inlet nozzle for the buffer tank and the acceptor chamber, which increases the load capacity of the nozzle. In the case of the Model B, the device is supplied with a tempering glass vessel with an internal floor area of 65 mm and a height of 50 mm to 60 mm, in which the primary tempering medium circulates and in which the diffusion cell is located.
Operation instructions
’Model A’ device (Fig. 4)
-
1.
Attach the acceptor chamber (D1) vertically to the stand on the magnetic stirrer or to a separate table stand.
-
2.
Apply a thin layer of silicone grease to the spherical edge of the acceptor chamber (D8).
-
3.
Connect the drip funnel (buffer tank) (G1) to the first ground inlet (D7) of the acceptor chamber and check that the pins (D4, D6, G2) are on the device in the open position.
-
4.
Screw the septum cap onto the third sampling inlet (D2).
-
5.
Place the magnetic stirrer bar (E) in the acceptor chamber (D1).
-
6.
Fill the device with acceptor fluid through the buffer tank (G1) and close the first inlet tap (D6) when the fluid level reaches the edge of the acceptor chamber spherical opening (D8) and the upper section of the second overflow inlet (D3).
-
7.
Close the tap on the second inlet (D4).
-
8.
Replace the acceptor fluid in the buffer tank (G1) so that the tank is at least half full.
-
9.
Carefully loosen the cap on the third inlet port (D2) until you notice a slight leak when fitting the cap, then close it immediately (wipe off excess fluid from the device's wall). The fluid level then drops slightly.
-
10.
Partially open the first inlet tap (D6) and close it as soon as the acceptor fluid level reaches the edge of the spherical opening of the chamber again (D8) and form a slight bulge due to surface tension (carefully wipe off any excess fluid from the device).
-
11.
If testing a transdermal patch, prepare the membrane (B1), the sample to be tested (B2), the silicone insert (B3) and the glass cover plate (B4) in the order of placement shown in Fig. 4. As the first step in preparation, use silicone grease to assemble the glass cover plate (B4) and the silicone insert (B3) with the donor chamber (A) in the order shown in Fig. 4.
-
12.
Place the assembly (A, B) described in point 11 on the acceptor chamber (D1). At the same time, while gradually pushing elements A, B completely into the acceptor chamber (D1), open the pin of the first inlet (D6) and finally secure the joint with the metal clamp (C).
-
13.
Close the front intake pin (D6).
-
14.
Start magnetic stirring at the desired speed.
-
15.
At the required sampling times, use a Hamilton syringe (F) to take the required but uniform volume of sample through the sampling septum (D2). Open immediately before sampling close the first inlet tap (D6) and close after sampling.
To supplement the above description, the silicones insert (B3) and the glass cover plate (B4) are not required for testing non-transdermal patches (eg, gels, ointments, creams). In this case, the operation according to point 10 is followed by the following operational steps:.
-
11.
Apply a thin layer of silicone grease to the spherical rim of the donor chamber (A1) and carefully place the membrane (B2) on it under tension.
-
12.
Assemble the donor chamber (A) and the acceptor chamber (D1). At the same time, while gradually pushing the assembly A, B2 completely into the acceptor chamber (D1), open the pin of the first inlet (D6) and finally secure the joint with the metal clamp (C)
-
13.
Close the front intake manifold pin (D6).
-
14.
Start magnetic stirring at the desired speed.
-
15.
Place the test sample on the membrane (B2) through the free lead-in of the donor chamber.
-
16.
At the required sampling times, use a Hamilton syringe (F) to take the required but uniform sample volume through the sampling septum (D2). Open immediately before the sampling of the first inlet tap (D6) and close after sampling.
In the ‘Model A’ design, the acceptor fluid is tempered by placing a glass vessel (e.g., a crystallization cup) and a heatable magnetic stirrer directly below the diffusion cell fixed in position. Fill the glass vessel with the tempering liquid (water) so that the liquid level does not cover the taps (D4, D6). Before steps 12–16, wait for the required temperature to set before proceeding. A cover layer made of aluminum foil can reduce the evaporation of the tempering medium and unnecessary heat exchange with the external environment.
‘Model B’ device (Fig. 5)
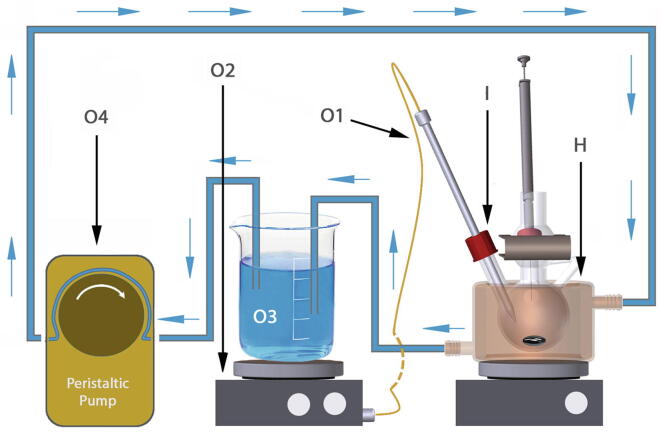
Fig. 5.
Schematic figure of operational system which contains the necessary main background instruments: double-walled vessel (H), sealed cap (I), temperature control thermocouple (O1), heated magnetic stirrer (O2), primary tempering fluid in baker, peristaltic pump (O3).
Each operation step of the ‘Model B’ diffusion cell shown in Fig. 3c, d differs from the operation steps described when using the ‘Model A’ device. The difference is because, in contrast to the design of the second inlet nozzle on the 'Model A' (Fig. 4: D4), the second inlet nozzle of the 'Model B' has no glass pin and the end of the nozzle has a threaded design that can be closed with a sealing ring (I) and on which the temperature control thermocouple (O1) is immersed in the acceptor fluid. When filling the acceptor cell with liquid, the function of the glass tap (Fig. 4: D4) of the second inlet nozzle shown on the ‘Model A’ device is thus performed by the closing cap (I). Accordingly, steps 7 and 13 are modified as follows:.
Screw on the second inlet's sealed cap (I) and pass the thermocouple (O1) through the pre-cut seal so that its liquid-immersed end is in the acceptor chamber (Fig. 4: D1) in front of an imaginary vertical axis.
13. Carefully loosen the cap (I) on the second inlet until you notice a slight leak when fitting the cap, and then close it immediately. Then close the tap on the first inlet (Fig. 4: D6), and wipe off any excess liquid from the device's wall.
The ‘Model B’ diffusion cell is supplied with a double-walled tempering glass vessel (H) which has the same function as the glass vessel described in the ‘Model A’ case. However, in this assembly, the liquid in direct contact with the acceptor chamber (secondary tempering fluid) is heated by the primary tempering fluid (O3) (water) flowing through the double-walled vessel (H). The primary tempering fluid circulates in the flow circuit schematically shown in Fig. 5 using a peristaltic pump (O4). The tempering liquid is heated in a beaker covered with aluminum foil on a heated magnetic stirrer (O2) specially used for this purpose. The thermocouple (O1) can be connected to control the heated magnetic stirrer (O2). Note that it is sufficiently stable and to provide a dynamically controlled tempering temperature, the flow rate should be adjusted to 50 ml to 100 ml/min and the dead volume of the flow circuit should be minimized by using short jumper lines.
Validation and characterization
The validation of the vertical diffusion cell was verified by the reproducibility of the instrument test, the precision and recovery (accuracy or recovery) of the chromatographic method for the analysis of the samples. The reproducibility of using the modified vertical diffusion cell shown was investigated by the in vitro drug release of electrospun colistin sulfate-loaded PVA nanofiber wound patch. A PVA scaffold consisting of nanofibers with an average diameter of 0.5 ± 0.1 mm per membrane was used, crosslinked by heat treatment. Before diffusion measurement, a liquid chromatographic method was developed to quantify the drug on a Waters Acquity UPLC H-Class system, which was validated. Accuracy and recovery tests were performed during validation. With colistin sulfate solution, three parallel measurements determined accuracy and recovery at five concentration levels (0.3, 0.5, 1, 5, and 50 μg/ml). For the recovery assay, the drug solution was spiked with the PVA polymer solution. The drug recovery ranged from 97% to 108% (RSD < 2.4%). As a result of the validation, it was proved that the developed method measures with documented precision in the applied concentration range. One ml sample was taken from the acceptor fluid at 16 different time points (1, 3, 5, 7, 10, 13, 16, 20, 25, 30, 45, 60, 90, 120, 180, 240 min) to investigate the release of drug from a donor patch of 35 mm in diameter with an average content of 1 mg colistin sulfate. It was found that the drug release from simple patches showed similar profiles, and in each case, 70–73% of the drug was in solution at 240 min. This study demonstrates that the modified vertical diffusion cell is suitable for in vitro study of drug release from transdermal patches. However, it should be noted that the regulations for in vitro release testing methods and devices are currently not completely uniform, so that the appropriate level of representativeness of a given method depends to a large extent on the properties of the given preparation. The representativeness can be determined with greater accuracy by comparison with in vivo studies [19], [21].
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgements
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Biographies

István Sebe graduated from Eötvös Loránd University in 2012 as a chemical-pharmaceutical researcher and then began his PhD studies and research in the field of pharmaceutical technology at the Faculty of Pharmacy of Semmelweis University. He received his Ph.D. in 2018, and in the same year, he won the Albert Szent-Györgyi Young Investigator Award from the New York Hungarian Scientific Society. Since 2015, he has been a product development project manager at Egis Pharmaceuticals Ltd. and a research fellow at Semmelweis University in Budapest. He is a founding and board member of the Hungarian Association for Medical Microbiology, and since 2017 he has been a board member of the Pharmaceutical Industrial Organization of the Hungarian Pharmaceutical Society.

Romána Zelkó Ph.D., D.Sc. is a full-time professor and the former dean of the Faculty of Pharmacy of the Semmelweis University, Budapest. Her research work focuses on polymeric drug delivery systems, physical aging of polymers, microstructural characterization of dosage forms associated with their functionality-related characteristics. She is the author of several scientific (more than 200 journal papers, 6 patents) and expert works. She serves as an editor, associate editor and editorial board member of internationally recognized journals. Her expertise covers the planning, development, solid-state characterization, and quality assurance of different dosage forms, including novel nanofiber-based scaffolds for various pharmaceutical and biomedical purposes.
Contributor Information
István Sebe, Email: sebe.istvan@pharma.semmelweis-univ.hu.
Romána Zelkó, Email: zelko.romana@pharma.semmelweis-univ.hu.
References
- 1.Thakker K.D., Chern W.H. Development and validation of in vitro release tests for semisolid dosage forms – case study. Dissolut. Technol. 2003;10(2):10–15. [Google Scholar]
- 2.Ghosh P., Milewski M., Paudel K. In vitro/in vivo correlations in transdermal product development. Ther. Deliv. 2015;6(9):1117–1124. doi: 10.4155/tde.15.72. [DOI] [PubMed] [Google Scholar]
- 3.Berner B., Raney S.G., Franz T.J. In: Drug Delivery Approaches: Perspective from Pharmacokinetics and Pharnacodynamics. Benson A.E., Roberts M.S., Gordi T., Berner B., editors. Wiley; Hoboken: 2021. Topical Delivery: Toward an IVIVC; pp. 241–252. [DOI] [Google Scholar]
- 4.Raghavan L., Brown M., Michniak-Kohn B., Ng S., Sammeta S. In: The Role of Microstructure in Topical Drug Product Development, AAPS Advances in the Pharmaceutical Sciences Series. Langley N., Michniak-Kohn B., Osborne D., editors. Springer; Cham: 2019. In Vitro Release Tests as a Critical Quality Attribute in Topical Product Development; pp. 47–87. [DOI] [Google Scholar]
- 5.Franz T. Percutaneous absorbtion. On the relevance of in vitro data. J. Invest. Dermatol. 1975;64(3):190–195. doi: 10.1111/1523-1747.ep12533356. [DOI] [PubMed] [Google Scholar]
- 6.The United States Pharmacopoeia (USP) XXIII (1995).
- 7.Siewert M., Dressman J., Brown C.K., Shah V.P. Guidelines to dissolution/in vitro release testing of novel/special dosage forms. AAPS Pharm. Sci. Tech. 2003;4(1):43–52. doi: 10.1208/pt040107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.FDA Guidance for Industry: Topical Dermatological Drug Product NDAs and ANDAs – In Vivo Bioavailability, Bioequivalence, In Vitro Release, and Asssociated Studies SUPAC-SS, U.S. Department of Health and Human Services, Food and Drug Administration, Center for Drug Evaluation and Research (CDER), June (1998).
- 9.FDA Guidance for Industry: Nonsterile Semisolid Dosage Forms, Scale-Up and Postapproval Changes: Chemistry, Manufacturing, and Controls; In Vitro Release Testing and In Vivo Bioequivalence Documentation SUPAC-SS, U.S. Department of Health and Human Services, Food and Drug Administration, Center for Drug Evaluation and Research (CDER), May (1997).
- 10.Ng S.-F., Rouse J.J., Sanderson F.D., Meidan V., Eccleston G.M. Validation of a static Franz diffusion cell system for in vitro permeation studies. AAPS PharmSciTech. 2010;11(3):1432–1441. doi: 10.1208/s12249-010-9522-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Kulkarni M., Potdar S., Date A.A., Marfatiya A. In vitro release testing of acyclovir topical formulations using immersion cells. Assay Drug Dev. Techn. 2021;19(2):75–84. doi: 10.1089/adt.2020.995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Kanfer I., Rath S., Purazi P., Mudyahoto N.A. In vitro release testing of semi-solid dosage forms. Dissolut. Technol. 2017;24(3):52–60. [Google Scholar]
- 13.Mali A.D., Bathe R., Patil M. An updated review on transdermal drug delivery systems. Int. J. Adv. Sci. Res. 2015;1(06):244–254. [Google Scholar]
- 14.Simon A., Amaro M.I., Healy A.M., Cabral L.M., de Sousa V.P. Development of a discriminative in vitro release test for rivastigmine transdermal patches using pharmacopeial apparatuses: USP 5 and USP 6. AAPS PharmSciTech. 2017;18(7):2561–2569. doi: 10.1208/s12249-017-0738-9. [DOI] [PubMed] [Google Scholar]
- 15.Shah V.P., Tymes N.W., Yamamoto L.A., Skelly J.P. In vitro dissolution profile of transdermal nitroglycerin patches using paddle method. Int. J. Pharm. 1986;32(2-3):243–250. [Google Scholar]
- 16.Ming M., Connie C., Lee P.H., Broman C.T., Cleary G.W. New improved paddle method for determining the in vitro drug release profiles of transdermal delivery systems. J. Controlled Release. 1993;27(1):59–68. [Google Scholar]
- 17.Alves A.C., Ramos I.I., Nunes C., Magalhães L.M., Sklenářová H., Segundo M.A., Lima J.L.F.C., Reis S. On-line automatedevaluationoflipidnanoparticlestransdermal permeation usingFranzdiffusioncellandlow-pressure chromatography. Talanta. 2016;146:369–374. doi: 10.1016/j.talanta.2015.08.070. [DOI] [PubMed] [Google Scholar]
- 18.Chattaraj S.C., Swarbrick J., Kanfer I. A simple diffusion cell to monitor drug release from semi-solid dosage forms. Int. J. Pharm. 1995;120(1):119–124. [Google Scholar]
- 19.Sebe I., Ostorházi E., Bodai Zs., Eke Zs., Szakács J., Kovács N.K., Zelkó R. In vitro and in silico characterization of fibrous scaffolds comprising alternate colistin sulfate-loaded and heat-treated polyvinyl alcohol nanofibrous sheets. Int. J. Pharm. 2017;523(1):151–158. doi: 10.1016/j.ijpharm.2017.03.044. [DOI] [PubMed] [Google Scholar]
- 20.Vertical Diffusion Cell, Semisolid Drug Products, Performance Tests, in: The United States Pharmacopoeia and National Formulary USP 37–NF 32, Chapter 1724, The United States Pharmacopoeial Convention, MD, Rockville, 2014, 1273–84.
- 21.Sebe I., Ostorházi E., Fekete Zs.A., Kovács N.K., Zelkó R., Kovalszky I., Wenyi L., Wade J.D., Szabó D., Ötvös L., Jr Polyvinyl alcohol nanofiber formulation of the designer antimicrobial APO sterilizes Acinetobacter baumannii-infected skin wounds in mice. Amino Acids. 2016;48:203–211. doi: 10.1007/s00726-015-2080-4. [DOI] [PubMed] [Google Scholar]