Abstract
Coronary artery disease continues to be a major cause of morbidity and mortality despite significant advances in risk stratification and management. This has prompted the search for alternative nonconventional risk factors that may provide novel therapeutic targets. Psychosocial stress, or mental stress, has emerged as an important risk factor implicated in a higher incidence of cardiovascular events, and although our understanding of this far ranging and interesting phenomenon has developed greatly over recent times, there is still much to be learned regarding how to measure mental stress and how it may impact physical health. With the current coronavirus disease 2019 global pandemic and its incumbent lockdowns and social distancing, understanding the potentially harmful biological effects of stress related to life-changing events and social isolation has become even more important. In the current review our multidisciplinary team discusses stress from a psychosocial perspective and aims to define psychological stress as rigorously as possible; discuss the pathophysiologic mechanisms by which stress may mediate cardiovascular disease, with a particular focus to its effects on vascular health; outline existing methods and approaches to quantify stress by means of a vascular biomarker; outline the mechanisms whereby psychosocial stressors may have their pathologic effects ultimately transduced to the vasculature through the neuroendocrine immunologic axis; highlight areas for improvement to refine existing approaches in clinical research when studying the consequences of psychological stress on cardiovascular health; and discuss evidence-based therapies directed at reducing the deleterious effects of mental stress including those that target endothelial dysfunction. To this end we searched PubMed and Google Scholar to identify studies evaluating the relationship between mental or psychosocial stress and cardiovascular disease with a particular focus on vascular health. Search terms included “myocardial ischemia,” “coronary artery disease,” “mental stress,” “psychological stress,” “mental∗ stress∗,” “psychologic∗ stress∗,” and “cardiovascular disease∗.” The search was limited to studies published in English in peer-reviewed journals between 1990 and the present day. To identify potential studies not captured by our database search strategy, we also searched studies listed in the bibliography of relevant publications and reviews.
Abbreviations and Acronyms: CAD, coronary artery disease; CBT, cognitive behavioral therapy; CVD, cardiovascular disease; FMD, flow-mediated dilatation; IL, interleukin; MI, myocardial infarction; MS, mental stress; MSIMI, mental stress induced myocardial ischemia; PAT, peripheral arterial tonometry; PED, peripheral endothelial dysfunction; PET, positron emission tomography; RH, reactive hyperemia; SES, socioeconomic status; TNF, tumor necrosis factor; VSMC, vascular smooth muscle cells
Article Highlights.
-
•
The importance of studying mental stress (MS) as a potential risk factor for cardiovascular disease has been brought to the fore with the coronavirus disease 2019 global pandemic and its associated public health policies.
-
•
Carefully defining MS involves distinguishing acute from chronic MS, accounting for coexisting biopsychosocial factors that are also associated with cardiovascular disease, and placing stress in its appropriate context — to this end, tools that quantify real-time physiologic reactivity to acute MS and the cumulative physiologic burden of chronic MS using measures that predict risk of disease will be of great value.
-
•
The pathologic cardiovascular effects of MS are transduced along the neuroendocrine immunologic axis to the vascular system by means of endothelial dysfunction and vascular inflammation.
-
•
Future MS studies should be more generalizable, must account for physiologic changes that occur temporally remote from the stressor, and should develop mechanistic models that include important coexisting biopsychosocial variables.
-
•
Although therapies targeting MS currently lack a robust evidence base, the pathologic effects of MS on the vascular system can be addressed using existing treatment strategies that target endothelial dysfunction.
Coronary artery disease (CAD) remains the leading causes of disease across the globe.1 Age-standardized life-years lived with ischemic heart disease decreased by 47% between 1990 and 2016,1 owing greatly to better cardiovascular disease (CVD) risk management and medical care.2 , 3 Nevertheless the number of people actually dying of CVD in the United States increased by 15% between 2006 and 2016 reaching 17.6 million deaths per year, of which 7.4 million were due to CAD.1 These mortality numbers continue to highlight the ongoing need to identify novel risk factors to target for CVD prevention and treatment. A risk factor of great interest and of which our understanding has developed significantly over recent years is psychosocial stress, or mental stress (MS).
Mental stress is a universal and shared experience of each of our lives. Estimates show that two-thirds of the general population has experienced MS within the past 2 weeks, with almost 50% rating their stress as “moderate or high.”4 Not just a facet of the fast-paced, globalized, and technologically advanced society of the 21st century, the nature and sources of stress have been contemplated since the time of the ancients. Six centuries before the birth of Christ, Confucius told his pupils “life is simple, but we insist on making it complicated” whereas almost a thousand years later, the Roman Emperor Marcus Aurelius wrote “if you are distressed by something, it is due to your own estimate of it.” Yet our understanding of the biological consequences of MS is still developing. Our research team has examined stress levels in a sample of more than 10,000 health care workers and reported that having a high level of perceived stress was associated with a poor quality of life and negative health behaviors.5 , 6 In addition, despite variations in study design, patient characteristics and measurements of stress and clinical outcomes, most studies have shown significant associations between stress and adverse health outcomes.7 , 8 Studies have also shown a link between MS and depression,9 diabetes mellitus,10 cancer,11 and CVDs including CAD, atrial fibrillation, and stroke.12, 13, 14, 15 In the large multinational INTERHEART study MS was associated with a greater than two-fold increased risk for myocardial infarction (MI) even after controlling for CVD risk factors; this effect persisted after stratifying by sex, prior CVD, socioeconomic status (SES), lifestyle factors, and geographic region.16 The potential link between MS and adverse health outcomes in general and CVD in particular has become of even greater importance in the context of the current coronavirus disease 2019 (COVID-19) global pandemic. This severe life-altering event has affected individuals at all levels of society across the world bringing with it a constellation of stressors en masse, including job loss or job and income insecurity with furlough schemes, illness and deaths of loved ones, dramatic changes to working and lifestyle habits, and (as a consequence of incumbent lockdown and social distancing policies) social isolation and loneliness. The long-term implications of the COVID-19 pandemic and its accompanying widespread social isolation on the risk of CVDs are yet to be realized but will form an essential area of future study.
Defining Stress
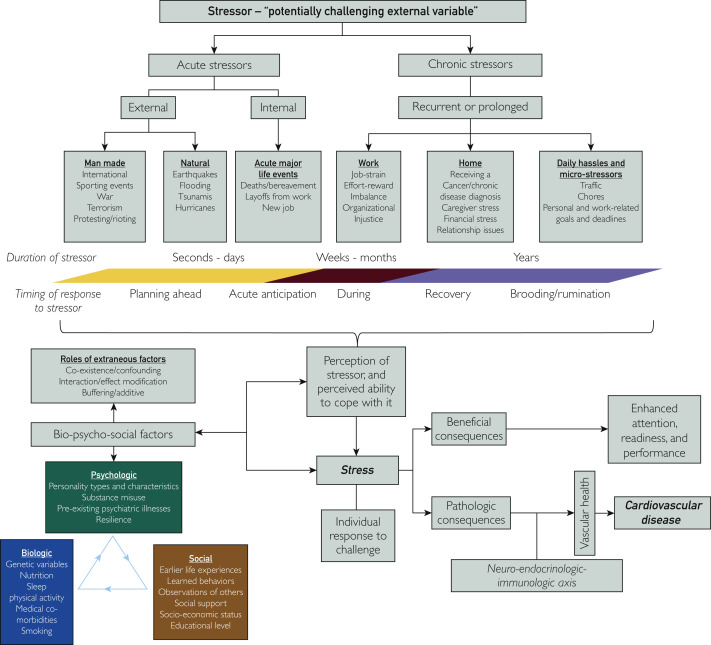
Stress has evolved into a variably used colloquialism. It is important to define this term carefully to study its role in disease and as a potential therapeutic target. An important distinction should be made between the stressor as a potentially challenging external variable, and stress as the individual’s response to the challenge. This response can be governed by several factors, only one of which is the external variable itself, with other important factors including the individual’s unique perception of the stressor, and his/her ability to cope with it.
Different personality types and characteristics have been shown to be risk factors for stress and CVD, including the type A behavior pattern (which is characterized as having an angry and hostile outlook),17 , 18 and the type D personality (which is characterized as having a tendency towards negative affectivity and social inhibition).19 These personality traits themselves may be further modulated by underlying genetic variables, substance use, nutrition, psychiatric and medical comorbidities, and sleep hygiene along with other environmental and sociological variables. Depression,20 anxiety,21 psychological distress,22 and post-traumatic stress disorder23 have all been shown to be risk factors for CVD. These variables are not synonymous with, but rather consequences of, stress and so, they often co-exist with stress. Individuals living in poverty show physiologic evidence of chronic stress,24 and although those of lower SES have a higher prevalence of CVD risk factors25 such as diabetes,26 hypertension,27 smoking,28 and unhealthy eating habits,29 adjusting for these factors does not entirely attenuate these associations.30 In fact, MS has been shown to have an attributable CVD risk similar to that of diabetes, hyperlipidemia, hypertension, and cigarette smoking.16 , 31
Therefore, the term stress itself forms a construct reflecting the synthesis of various biopsychosocial factors variably interacting and affecting an individual at a particular point in his/her life. In engineering, the application of stress to a material results in strain. Different types of stress applied for different periods leads to variable amounts of strain. This may lead to no change in the external form of the material but could result in unfavorable internal changes impairing the integrity of the material. Alternatively, that strain could lead to outwardly visible changes to the material’s shape that could be unfavorable, leading to break down, or desirable, leading to a new and more useful product. Thus, challenging new life circumstances may not necessarily be unpleasant, but may just require more attention, and readiness, that could result in better life outcomes such as good performances in exams, sporting competitions, and job interviews. Therefore, some stress may be beneficial, necessary, and even healthy. Thus, stress must always be seen in its appropriate context before drawing conclusions about its potential biological consequences.
Stress can be acute, lasting seconds to days or even weeks, or chronic, lasting months or even years. Acute stressors can be pinpointed to specific instances. These may be personal, including deaths in the family,32 and layoffs from work,33 , 34 both of which have been linked to CVD events, or impersonal, relating to natural disasters such as earthquakes,35 or manmade disasters such as terrorism,36 or even from watching World Cup Soccer.37 Chronic stressors may be discrete and identifiable, and along the lines of Freud’s maxim “happiness comes when one finds pleasure in love and work” can be separated into stressors at home, and those in the workplace (Figure 1 ). Work stress can be characterized using the job-strain model, the effort-reward balance model, and the organizational injustice model, all of which are associated with an increased risk of CAD.38 At home, marital stress has also been associated with recurrent CAD events.39 Further, higher stress levels have been reported in those who are divorced or separated compared with those who are married.40 Lacking life partnership is closely related to and overlaps with social isolation, although studies evaluating the interaction between social isolation and marital/relationship status on CVD are lacking. This matter is further complicated by the fact that marriage and relationships can be sources of stress in of themselves. Studies have also shown that stress associated with receiving a diagnosis of cancer also increases the risk of CVD,41 as does caregiving to ill family members at home.42 Financial stress may be considered as a bridging construct between stress at work and at home, and is also associated with cardiovascular events.43 Chronic stress may also refer to smaller and less easily characterized microstressors that include rush-hour traffic, performing household chores, social readjustment and isolation, as with the current COVID-19 global pandemic, and work or personal goals and deadlines.
Figure 1.
Schematic outline of the relationship between acute and chronic stressors and stress defined as the individual response to challenge, stress’s dynamic interaction with extraneous bio-psycho-social factors, and beneficial and pathologic consequences of stress.
Distinguishing acute from chronic stress is useful as acute stressors tend to trigger acute CVD events in those with established CVD, and chronic stressors contribute to disease progression and worse longer-term outcomes.44 Longer periods of stress or multiple separate episodes of stress may aggregate and lead to an accumulating burden of increased disease risk over time. However, separating acute from chronic stress arbitrarily can be problematic when considering that the psychologic fallout from a particular stressor may persist long after the event ends, such as ruminating on a subject of disagreement after fighting with a colleague. In such cases, the proximal stressor itself does not provide a good indicator of the potential disease-causing burden incurred by the patient.
Quantifying Stress
Although important, these terms may not provide useful information on the actual biological effects of MS. By contrast, physiologic changes and reactivity that occur with the cognitive appraisal of a stressor may provide a useful approach by which to quantify the pathologic consequences of MS. If valid and reliable, such tools would allow investigators to study the adverse impacts of stress in a homogenous way, permitting useful comparisons across studies from which more helpful conclusions could be drawn. Ideally, clinicians should be able to determine if a patient’s subjectively reported stress is associated with an exaggerated physiologic response, which can be measured in a way that is correlated with an increased risk for adverse outcomes. This could then provide a potential prognostic marker and therapeutic target. Currently, psychologists rely on patient-reported rating scales to quantitatively evaluate stress.45 Although such scales capture the inherent subjectivity of stress, they do not account for the various biopsychosocial factors that interact with and influence the degree to which the stressor may lead to stress that portends increased risk (see Figure 1). An individual’s perception of a stressor may also diverge from the neuropsychiatric, metabolic, and other physiologic manifestations of stress. Thus, patient questionnaires do not relate a subjective stressful experience to the pathophysiologic mechanisms that provide measurable indices of increased risk for adverse outcomes.
In an alternative approach, individuals could be exposed to a particular stressor while being observed and having measures of physiologic changes. This could be performed opportunistically by taking advantage of naturally occurring stressors in the environment such as earthquakes, with the obvious limitations of unpredictability and inability to control extraneous and confounding factors. Alternatively, stressors may be artificially simulated in a tightly controlled laboratory environment to produce reliable hemodynamic and sympathetic nervous system responses.46 These may include recalling an anger-provoking incident, structured public speaking tasks, number-letter recall challenge (spiral omnibus), and the Stroop word-color conflict test.47 , 48 Although these experimental stressors lack real-world ecologic validity, they are reliable in their ability to produce acute MS, allow for close control of experimental conditions, and provide an opportunity to study real-time pathophysiologic responses to stressors.
Studying the effects of chronic stress will require tools that assess the cumulative physiologic burden of multiple acute and chronic stressors over time. Given that single acute stressors play a role in triggering acute CVD events, and chronic stressors contribute to disease progression and worse longer term outcomes,44 the ideal measure(s) would quantify the real-time physiologic reactivity to acute MS as well as the cumulative physiologic burden of recurrent and chronic MS where possible using a single technique. By measuring both, the real-time mechanism identified in the former may provide insight into the cumulative mechanism contributing to the latter, and so would necessarily incorporate the adverse biologic effects of stress in a way that could predict risk.
Mechanisms Linking Mental Stress and Incident Cardiovascular Disease
What could be the accepted standard of such a physiologic measure(s)? To answer this question we systematically searched PubMed, and Google Scholar to identify prospective studies that examined the association between experimental MS and incident CVD and mortality, with the exception of studies that looked at incident hypertension and other CVD risk factors as we wished to focus on end-organ disease, and specifically included those that also included measure(s) of concurrent pathophysiologic mechanisms that may putatively play a role in mediating CVD. Search terms included “myocardial ischemia,” “coronary artery disease,” “mental stress,” “psychological stress,” “mental∗ stress∗,” “psychologic∗ stress∗,” and “cardiovascular disease∗.” The search was limited to studies published in English in peer-reviewed journals between 1990 and the present day, and included studies that had a prospective cohort design, a follow-up period of at least 6 months, and used a validated technique to experimentally induce MS. To identify potential studies not captured by our database search strategy, we also searched studies listed in the bibliography of relevant publications and reviews (Table 1 ).
Table 1.
Summary of All Published Prospective Cohort Studies Evaluating the Relationship Between Experimentally Induced Mental Stress and Cardiovascular Outcomes, in Addition to Measurements of a Putative Mediating Pathologic Mechanisma
| Reference | Year | Sample size N (no. of males) |
Population | Follow-up duration | Experimental stressor | CV outcome | Putative mediating pathologic mechanism | No. of events | Principle finding | Other finding(s) |
|---|---|---|---|---|---|---|---|---|---|---|
| 49 | 1992 | 13 (10) | Post MI | Average 57 mo (range: 39-64 mo) | Modified Stroop test on 2 occasions | Re-infarction and/or stroke | BP, HR, and venous plasma catecholamines | 5 | Patients with events had larger systolic and diastolic BP responses to Stroop test than patients who were event-free at follow-up | Catecholamine concentrations differed between groups during MS, but on only 1 of the 2 test days |
| Groups did not differ on baseline measurements, CV response to exercise testing, fasting serum lipid and glucose concentrations, age, or duration of follow-up | ||||||||||
| 50 | 1995 | 30 (30) | Stable angina pectoris and ischemia on stress MPI | 2 y | Mental arithmetic testing | Nonfatal MI, unstable angina | Continuous ambulatory LV function monitoring | 14 (4 nonfatal MIs, 10 unstable angina) | 15 developed transient LV dysfunction during MS | |
| At 2-year follow-up, 10 of 15 patients (67%) with MS-induced LV dysfunction had adverse events compared with only 4 of 15 (27%) with no MS-induced LV dysfunction (P=.025) | ||||||||||
| 51 | 1996 | 126 (112) | Documented CAD and exercise-induced myocardial ischemia | Mean/median 44 mo | Mental arithmetic, public speaking, mirror trace, reading, and type A structured interview | Hospitalization, cardiac revascularization, MI, cardiac death | RNV imaging and 48-h Holter monitor | 28 (2 cardiac deaths, 4 nonfatal MIs, 10 CABG, 17 angioplasty — 6 had multiple events) | Baseline MS-induced ischemia was associated with significantly higher rates of cardiac events (OR, 2.8; 95% CI, 1.0-7.7; P<.05) | The RR for ECG-defined ischemia during exercise testing was 1.9 (95% CI, 0.95-3.96; P=.07), and the RR for ambulatory ECG ischemia was 0.75 (95% CI, 0.35-1.64; P=.47). |
| LVEF change during MS was significantly related to event-free survival (RR, 2.4; 95% CI, 1. 12-5. 14; P=.02), controlling for age, history of prior MI, and baseline LVEF | ||||||||||
| This relationship remained significant after controlling for ECG-defined ischemia during exercise (RR, 2.2; 95% CI, 1.01-4.81; P<.05) | ||||||||||
| 52 | 1999 | 79 (76) | CAD as confirmed by previous MI or coronary angiography or a 90% probability of CAD determined by Bayesian analysis | Median 3.5 y (range: 2.7 to 7.3 y) | Mental arithmetic and a simulated public speech stress | Cardiac death, nonfatal MI, or revascularization procedures | New or worsened ischemic wall motion abnormalities were monitored using echocardiography or RNV | 28 (5 cardiac deaths, 9 MIs, 9 CABGs, and 5 angioplasties) | New or worsened LV wall motion abnormalities occurred in 61 patients (77%) | After controlling for baseline BP and study group status (echocardiography vs RNV), there was a higher RR of subsequent events for patients with high vs low peak stress-induced diastolic BP response (RR, 2.4; 95% CI, 1.1-5.2; P=.03) |
| Peak changes in BP and HR | Survival analysis showed that 20 of 45 patients with MS-ischemia (44%) experienced new cardiac events vs those without MS ischemia (8 of 34; 23%; P=.048) | |||||||||
| Type of cardiac event did not differ between MS ischemia– positive vs – negative patients | ||||||||||
| 53 | 2002 | 196 (170) | CAD (>50% narrowing in at least 1 major coronary artery or verified MI, or evidence of myocardial ischemia on an exercise treadmill test conducted off anti-ischemic medications) | Average 5.2±0.4 y (range: 5.2±0.4 y) | Speech on an assigned topic | Cardiac death | Bicycle exercise and MS testing with RNV imaging | 17 deaths | Of the 17 participants who died, new or worsened wall motion abnormalities during MS were present in 40% vs 19% of survivors (P=.04) and significantly predicted death (RR, 3.0; 95% CI, 1.04-8.36; P=.04) | EF changes during MS were similar in patients who died and in survivors (P=.9) and did not predict death even after adjusting for resting EF (P=.63) |
| Other indicators of ischemia during MS (ST-segment depression, chest pain) did not predict death, nor did psychological traits, hemodynamic responses to MS, or markers of the presence and severity of ischemia during daily life and exercise | ||||||||||
| 54 | 2010 | 138 (96) | Patients with stable CAD | Median 5.9 y | Mirror tracing and public speaking | Combined end point of MI and all-cause mortality | Ischemia on R-wave-synchronized, gated equilibrium RNV | 32 (17 nonfatal MIs and 15 deaths) | Of the 26 patients who exhibited myocardial ischemia during MS, 11 (42%) sustained subsequent clinical events, compared with 21 of 112 patients (19%) who showed no MS-induced ischemia | LVEF change during MS was related to the clinical events in a graded, continuous fashion, with each 4% decrease from the LVEF at rest associated with an adjusted HR of 1.7 (95% CI, 1.1-2.6, P=.011) |
| 55 | 2012 | 431 (229 females) | West of Scotland Twenty-07 Population Study | 16 y | Mental arithmetic test | CV mortality | Systolic and diastolic Blood Pressure response | Both systolic and diastolic blood pressure reactions were positively associated with CV mortality | ||
| 56 | 2012 | 1470 (746 males) | 1995 Nova Scotia Health Survey Population Based Study | 10 y | Interpersonally stressful interview designed to elicit anger and MS by asking participants about their characteristic responses to a variety of different situations | Fatal or non-fatal CV disease events | Systolic and diastolic BP response | 161 nonfatal and 10 fatal CV disease events | In an unadjusted model, those in the highest decile of systolic BP reactivity were more than twice as likely to have an incident CVD event vs those in the decile with no reactivity (HR, 2.33; 95% CI, 1.15-4.69; P=.02) | After adjusting for age and sex, and then also for FRS, BMI, and education, this relationship was attenuated and not statistically significant |
| Diastolic BP reactivity was not associated with CVD incidence in any model | ||||||||||
| 57 | 2015 | 100 (74) | Systolic HF | Median 48.5 mo | Structured public speech task | Mortality | BP and HR responses to MS | 31 deaths | Mortality rates were 2 times higher (HR, 2.04; 95% CI, 1.15-3.60; P=.014) among patients with the lowest diastolic BP responses (mean=-2.4 ± 5.4 mm Hg) to MS vs in patients with an intermediate diastolic BP response (mean = 7.3 ± 2.5 mm Hg), adjusting for covariates | Multivariate analyses showed that a high heart rate response (>6.3 beats/min) to acute MS was associated with a reduced mortality risk (HR, 0.40; 95% CI, 0.16 to 1.00; P=.051) vs those with intermediate responses |
| [31 patients had died (31%)] | High diastolic BP reactivity (mean = 16.3 ± 3.4 mm Hg) was not related to mortality (HR, 0.95; 95% CI, 0.55-1.66) | |||||||||
| Systolic BP responses showed a similar but nonsignificant association | ||||||||||
| 58 | 2017 | 224 (187) | Clinically stable CAD, NYHA functional class I | Median 4 y | Mental arithmetic, mirror tracing and anger recall public speech | Composite events that comprised all-cause mortality and/or nonfatal CV events, resulting in an unplanned hospitalization | MAV, specifically, diastolic early (e'), diastolic late (a'), and systolic (s') velocities | 86 patients experienced at least 1 composite event(s) | MS-induced changes in e' (HR, 0.73) and s' (HR, 0 .73) were significant (P<.05) predictors of composite events, and the change in a' (HR, 0.74) was marginal (P=.05). | Patients with a greater decrease in e' and/or s' velocity had a higher probability of experiencing a composite event, and the association of the change in a' and composite events was marginal (P=.05) |
| 59 | 2017 | 310 (257) | Stable adults with documented IHD | Median 4 y (maximum 6 y) | Mental arithmetic, mirror tracing, and anger recall public speech | First and total rate of MACE, defined as | Development or worsening of any wall motion abnormality, reduction of LVEF ≥ 8%, or ischemic ST-segment change on ECG (horizontal or downsloping depression ≥ 1 mm in two or more leads | 125 patients had at least 1 MACE (18 deaths, 220 hospitalizations due to CV causes including 24 nonfatal MIs, 81 unstable anginas, and 31 HF exacerbations) | The continuous variable of MS-induced LVEF change was significantly associated with both endpoints (all P<.05) | Indices of exercise-induced myocardial ischemia did not predict endpoints |
| all-cause mortality and hospitalizations for CV causes | For every reduction of 5% in LVEF induced by MS, patients had a 5% increase in the probability of a MACE at the median follow-up time and a 20% increase in the number of MACE endured over the follow-up period of 6 y | The incidence of MACE in MSIMI group was 9.85% higher than those without (P=.08) | ||||||||
| All-cause mortality was 7.46% with MSIMI and 7.14% with exercise induced myocardial ischemia (P=.19) | ||||||||||
| 60 | 2017 | 199 (135) | Outpatients diagnosed with HF, with EF ≤40% | Median 5 y | Public speaking task | Combined end point of death or CV hospitalization | Systolic and diastolic BP and HR reactivity | 155 first events (72 CV hospitalizations, and 83 deaths) | Both systolic and diastolic BP reactivity, quantified as continuous variables, were inversely related to risk of death or CV hospitalization (P<.01) after controlling for established risk factors, including HF disease severity and etiology | For diastolic BP, high reactivity was marginally associated with lower risk compared with intermediate reactivity (HR, 0 .767; 95% CI, .515-1.14; P=.193), whereas low diastolic BP reactivity was associated with greater risk (HR, 1.49; 95% CI, 1.027-2.155; P=.0359) |
| High systolic BP reactivity, compared with intermediate systolic BP reactivity, was associated with lower risk (HR, 0.498; 95% CI, .335-.742; P=.001), whereas low systolic BP reactivity did not differ from intermediate reactivity | No relationship of heart rate reactivity to outcome was identified | |||||||||
| 61 | 2019 | 549 (417) | Stable CAD | 3 y | Standardized public speaking stressor | CV death, MI, revascularization, and hospitalization for HF | PAT measurements during MS compared with baseline: | 24 all-cause deaths, 14 CV deaths, 24 MIs, 66 coronary revascularizations, 20 HF hospitalizations | After adjusting for demographic and CV risk factors, medications, and rate-pressure product change during MS, those with low stress PAT ratio were at significantly higher risk of adverse outcomes (HR, 1.77; 95% CI, 1.12-2.80) | |
| Stress PAT response ratio: pulse wave amplitude during MS/at baseline | ||||||||||
| Median ratio was 0.68 (IQR, 0.48-0.88), indicating 32% vasoconstriction with MS | ||||||||||
| 62 | 2019 | 569 (420) | Stable CAD | Median (IQR): 3.0 (2.9-3.1) y | Standardized public speaking stressor | Composite endpoint including CV death, MI, and unstable angina leading to revascularization and HF hospitalization | FMD was measured before and 30 min after MS | 74 patients experienced MACE (13 CV deaths, 15 MIs, 34 unstable angina, 12 hospitalizations for HF) | 360 participants (63.3%) developed transient PED (a decrease in FMD) | Risk discrimination statistics showed a significant model improvement after addition of either post-stress FMD (change in the AUC, 0.05; 95% CI, 0.01-0.09) or pre-stress plus change in FMD (change in the AUC, 0.04; 95% CI, 0.00-0.08) compared with conventional risk factors |
| Transient PED with MS was associated with a 78% increase (HR, 1.78; 95% CI, 1.15-2.76) in the incidence of MACE | ||||||||||
| Both the change in FMD (post – pre HR, 1.15; 95% CI, 1.03-1.27 for each 1% decline) and post-MS FMD (HR, 1.14 ;[95% CI, 1.04-1.24 for each 1% decline) were associated with MACE | ||||||||||
| 63 | 2020 | 148 (102) | Participants with stable CAD | Median 3 y | Series of standardized speech/arithmetic stressors | MACE - composite of CV death, MI, unstable angina with revascularization and HF hospitalization | Simultaneous brain imaging with high-resolution PET: rmPFC activation | 34 patients experienced MACE (2 CV deaths, 1 MI, 5 hospitalizations for HF, 26 cases of unstable angina with revascularization) | Each increase of 1 SD in rmPFC activation with MS was associated with a 21% increased risk of MACE (HR, 1.21; 95% CI, 1.08-1.37) | MS-induced IL-6 and high-frequency heart rate variability explained 15.5% and 32.5% of the relationship between rmPFC reactivity and MACE, respectively |
| IL-6 levels 90 min after stress | After adjustment for baseline demographics, risk factors, and baseline levels of IL-6 and high-frequency Heart Rate variability, higher rmPFC stress reactivity was independently associated with higher IL-6 and lower high-frequency HR variability with MS | Addition of rmPFC reactivity to conventional risk factors improved risk reclassification for MACE prediction, and C-statistic improved from 0.71 to 0.76 (P=.03) | ||||||||
| High-frequency HR variability during MS | ||||||||||
| 64 | 2020 | 148 (102) | Participants with stable CAD | 2 y | Series of standardized speech/arithmetic stressors | Angina (assessed with the Seattle Angina Questionnaire's angina frequency subscale) | High resolution PET imaging of the brain: blood flow to the inferior frontal lobe was evaluated as a ratio compared with whole brain flow for each scan | 54 patients experienced angina at follow-up (35 monthly, 19 daily or weekly) | For every doubling in the inferior frontal lobe activation, angina frequency was increased by 13.7 units at baseline (95% CI, 6.3-21.7; P=.008) and 11.6 units during follow-up (95% CI, 4.1-19.2; P=.01) in a model adjusted for baseline demographics | MS-induced ischemia and activation of other brain pain processing regions (thalamus, insula, and amygdala) accounted for 40.0% and 13.1% of the total effect of inferior frontal lobe activation on angina severity, respectively |
| 65 | 2020 | 562 (427) | Participants with stable CAD | Median 3 y | Standardized public speaking stressor | MACE was defined as a composite endpoint of CV death, MI, unstable angina with revascularization, and HF | IL-6, MCP-1, and MMP-9 | 71 patients experienced MACE (14 CV death, 22 MI, 19 HF, and 35 unstable angina with revascularization) | There was no significant association between inflammatory response to stress and risk of MACE | There were sex-based interactions for IL-6 (P=.001) and MCP-1 (P=.01) |
| Risk of MACE increased 56% (HR, 1.56; 95% CI, 1.21-2.01; P=.001) and 30% (HR, 1.30; 95% CI, 1.09-1.55; P=.004) for each SD increase in IL-6 and MCP-1 response to MS for women, but not in men | ||||||||||
| 66 | 2020 | 417 (383) | Patients hospitalized for ACS and received percutaneous coronary intervention | 1 y | Three different MS tasks of 6-min duration in random order (number-letter recall challenge of increasing length and complexity; number subtraction; Stroop word-color conflict) | A composite of ACS, rehospitalization, stroke, revascularization, CV death, and all-cause mortality | RH-PAT at baseline (baseline PEF) compared with RH-PAT following MS (post-MS PEF) | 82 MACE events (63 cardiac rehospitalizations, 49 revascularizations, 3 MIs) | Women were more likely to experience MACE in the year following ACS (RR, 2.42; 95% CI, 1.53-3.84; P=.044), and had a significantly lower stress PAT ratio compared with women who did not (1.0±0.17 vs 1.20±0.17; P=.04) | In multivariate analyses stratified by sex, baseline PED (EndoPAT<1.7) (χ=8.0, P=.005) and stress PAT ratio (χ=7.7, P=.006), were independently predictive of MACE in women, but not men |
| PAT measurements during MS compared with baseline: | ||||||||||
| Stress PAT response ratio: pulse wave amplitude during MS/at baseline |
ACS, acute coronary syndrome; AUC, area under the curve; BMI, body mass index; BP, blood pressure; CAD, coronary artery disease; CABG, coronary artery bypass graft; CV, cardiovascular; ECG, electrocardiogram; EF, ejection fraction; FMD, flow-mediated dilatation; FRS, Framingham risk score; HF, heart failure; IL, interleukin; IHD, ischemic heart disease; LV, left ventricle; MACE, major adverse cardiovascular event; MAV, myocardial annular velocity; MCP-1, monocyte chemoattractant protein 1; MI, myocardial infarction; MMP-9, matrix metalloproteinase 9; MPI, myocardial perfusion imaging; MS, mental stress; MSIMI, mental stress induced myocardial ischemia; NYHA, New York Heart Association; PAT, peripheral arterial tonometry; PEF, peripheral endothelial function; PET, positron emission tomography; RH-PAT, reactive hyperemia peripheral arterial tonometry; rmPFC, Rostromedial prefrontal cortex; RNV, radionuclide ventriculography
A variety of putative mediating pathophysiologic mechanisms were measured including the reactivity to MS of blood pressure, heart rate, catecholamines, inflammatory markers, and peripheral arterial tone; myocardial ischemia using Holter monitoring, and ventriculography; echocardiographic changes related to ischemia and diastology; functional brain imaging; and measures of peripheral endothelial function before and after MS using flow-mediated dilatation (FMD) and reactive hyperemia (RH). Given the significant heterogeneity in the study designs and experimental procedures, it is challenging to directly compare the prognostic utility of individual pathophysiologic measures against each other. Desirable measures include those capable of making measures that 1) predict risk of disease reliably and effectively, 2) are inexpensive and do not require the use of sophisticated technology and burdensome methodology, and 3) offer a modifiable therapeutic target that can be followed with repeat measures longitudinally. Techniques not included in these data, and which to-date have not been evaluated in prospective clinical outcome studies, include mobile health applications and wearable electrochemical biosensors that offer measures of specific physiologic responses to acute and chronic stressors that occur naturally in the lives of patients. These measures can be provided on a moment-to-moment basis, and can also provide summary data over longer periods,67 thus fulfilling the need to look at acute reactivity and cumulative chronic burden. Examples include the noninvasive monitoring of electrolytes and metabolites in sweat or saliva,67 spectroscopy biosensor assays to detect cortisol levels in saliva,68 and electrocardiograms (ECGs) from wearables to evaluate ischemia or arrhythmia.69
Measures of MS-induced myocardial ischemia (MSIMI) could provide a useful index when evaluating MS. Studies comparing exercise stress testing against MS testing have revealed interesting differences between the two. In one study, the investigators compared the frequency of self-reported angina and myocardial ischemia using positron emission tomography (PET) before and after bicycle ergometry and before and after 2 minutes of serial seven subtractions as a simulator of MS. After exercise, all patients reported symptoms of angina, ECG changes, and regional perfusion abnormalities. After MS, however, 75% of all patients developed regional perfusion abnormalities, among which only 33% also reported angina and had ECG changes; 17% had only ECG changes, and 50% had neither ECG changes nor angina.47 Thus, the perfusion abnormalities elicited by the mental stressor were more likely to be clinically silent in terms of symptoms and ECG findings. Similarly discordant findings have been reported in another study,70 although the significance of these differences is uncertain. It is unknown, for example, if these findings show that MS testing is simply less sensitive than exercise stress testing at eliciting ischemia, or if MS testing is in fact better at discriminating those at greatest risk. Certainly studies have shown that MSIMI has prognostic value, with one study demonstrating an almost three-fold increased risk in future cardiac events in those with MSIMI.51 However, measuring ischemia requires expensive testing equipment as well as specialist expertise for interpretation. Thus, at present, the role of testing for MSIMI in clinical practice remains undetermined, and further studies are required to address this question.
Vascular Mechanisms
Endothelial dysfunction is often described as the first step in the atherosclerotic process and is independently associated with adverse CVD events.71 Studies have shown that individuals with minimal traditional CVD risk factors who have peripheral endothelial dysfunction (PED) have a higher incidence of CVD events compared with those with normal peripheral endothelial function.71, 72, 73 Thus, an alternative and promising physiologic measure is peripheral and noninvasive measurements of vascular reactivity and PED in response to acute MS. An example approach includes reactive hyperemia–peripheral arterial tonometry (RH-PAT).74, 75, 76, 77 Peripheral endothelial function can be measured at baseline, using RH, and after the application of experimental MS tasks. Mental stress induces transient endothelial dysfunction,78 and so measured change in endothelial function may correspond to the vascular burden of MS. Further, PAT corresponding to pulse-wave velocity through the digital microcirculation can be measured during each MS task, and can be compared to the PAT at baseline to determine a stress PAT ratio that provides quantitative information on the real-time vascular reactivity to acute MS. Table 2 summarizes the results of all cross-sectional studies that have used measures of PED and/or stress PAT to quantify stress. In the following section, we outline why the vasculature is so important for the stress-induced physiological response, and why it could be the principal site of transduction of MS into CVD risk (Figure 2 ).
Table 2.
Summary of All Published Cross-Sectional Studies Evaluating the Relationship Between Experimentally Induced Mental Stress and Peripheral Vascular Reactivitya
| Reference | Year | Sample size N (no. of males) |
Population | Experimental stressor | Measure of peripheral vascular reactivity | Other measures | Principle finding | Other finding(s) |
|---|---|---|---|---|---|---|---|---|
| 79 | 1999 | 40 (21) | Healthy adults aged 25–44 y | Reaction time/shock avoidance, mirror trace, and anger interview | Brachial artery endothelial function measured by ultrasonography in response to RH (FMD) | BP and SVR | A high EDAD was associated with lower resting systolic and diastolic BP | SVR responses during MS testing were greater for individuals with lower EDAD responses |
| EDAD was not associated to BP response to MS | ||||||||
| 78 | 2000 | 18 (18) | 10 healthy males; 8 non–insulin-dependent diabetic males | Structured speech task | Brachial artery endothelial function measured by ultrasonography in response to RH (FMD) | Endothelial-independent function following infusion of nitroglycerin | In healthy subjects, FMD (5.0±2.1%) was significantly (P<.01) reduced at 30 and 90 min after MS (2.8±2.3% and 2.3±2.4%, respectively) and returned toward normal after 4 h (4.1±2.0%) | Diabetic subjects had lower FMD than controls (3.0±1.5% vs 5.0±2.1%, respectively; P=.02) but showed no changes in FMD (2.7±1.1% after 30 min, 2.8±1.9% after 90 min, and 3.1±2.3% after 240 min) or GTN responses after MS |
| MS had no effect on the response to GTN | ||||||||
| In studies without MS, FMD did not change | ||||||||
| 80 | 2002 | 23 | Healthy subjects without CV risk factors | Colored light response | Brachial artery endothelial function measured by ultrasonography in response to RH (FMD) | FMD before and after MS during intra-arterial infusion of a selective endothelin A receptor antagonist (BQ-123) | Endothelium-dependent vasodilation was reduced by half for about 45 min (8.0±1.1% vs 4.1±1.0%; P<.002), whereas endothelium-independent vasodilation to nitroglycerin | Intra-arterial infusion of the selective endothelin-A receptor antagonist, but not saline prevented the impairment of endothelium-dependent vasodilation (8.6±1.2 versus 9.4±1.3%; NS) |
| Endothelial-independent function following infusion of nitroglycerin | remained unaffected (15.6±1.6 vs 14.3±1.3%; NS) | Intra-arterial infusion of norepinephrine of similar duration as MS did not inhibit FMD | ||||||
| 81 | 2004 | 16 (16) | Previously diagnosed CAD with positive exercise tress tests | Mental arithmetic stress test with harassment | PAT measurements during MS compared to baseline | ERNA | In 8 patients both ERNA and PAT were abnormal | When considering an abnormal PAT tracing as indicative of MSIMI, concordance of the 2 methods was 88% |
| Considered abnormal when PAT decreased by ≥20% from baseline | Myocardial ischemia diagnosed when global EF fell ≥8% during MS or new/worsened focal wall motion abnormalities | In 6 patients both tests were negative | ||||||
| In 2 cases results were discordant | ||||||||
| 82 | 2006 | 16 (0) | Postmenopausal women with angina and normal coronary angiogram | Anger recall task (an incident that made patients angry and that involved interpersonal interactions) | Brachial artery endothelial function measured by ultrasonography in response to RH (FMD) | Technetium 99m methoxyisobutylisonitrile myocardial scintigraphy at rest, MS and exercise | During MS testing, 6 patients (group I) had reversible perfusion defects on myocardial scintigraphy; other 10 patients (group II) did not | No group I patients had ischemia on Holter monitoring; 2 of 10 group II patients had ischemia |
| 24-h ambulatory ECG recording (Holter monitor) | Group I patients exhibited PED more frequently than those in group II (83% vs 20%) | |||||||
| Myocardial scintigraphy showed anteroapical/septal ischemia in 5 patients and inferoapical ischemia in 1 other patient, with both types of stress | ||||||||
| In group II patients, none showed a reversible perfusion defect during physical or MS | ||||||||
| 83 | 2008 | 211 (134) | Patients with established stable CAD | Public speaking task | PAT measurements during MS compared with baseline | BP and heart rate were recorded during rest and MS | Stress PAT ratio was significantly higher in women (0.80±0.72) compared with men (0.59±0.48), P=.032 | MS induced significant changes in systolic BP, diastolic BP, heart rate, and double product compared with rest in all subjects, P<.001 |
| Stress PAT response ratio: pulse wave amplitude during MS/at baseline | Remained significant after controlling for confounders, P=.037 | Comparing hemodynamic responses with MS across sexes did not show differences in systolic BP, diastolic BP, heart rate, or double product | ||||||
| 84 | 2008 | 87 (34) | Healthy subjects | Three different MS tasks of 6-min duration in random order (number-letter recall challenge of increasing length and complexity; number subtraction; Stroop word-color conflict) | RH-PAT at baseline (baseline PEF) compared with RH-PAT following MS (post-MS PEF) | In response to MS, male subjects had an increase in RH-PAT compared with baseline RH-PAT compared with females, who showed a decline in PEF (13.7% vs −0.47%; P=.01) | Double product (systolic BP × heart rate) | Males had a greater double product response to MS (27.2+3.6% increase in double product vs 19.2+1.7%; P=.01) |
| PAT measurements during MS compared with baseline | Stress PAT ratio tended to be greater in males than females (0.79±0.07 vs 0.9±0.04, respectively; P=.07) | |||||||
| Stress PAT response ratio: pulse wave amplitude during MS/at baseline | Females who showed the least vasoreactivity to MS showed the greatest decline in PEF (−10.5+4% vs 17.4 + 6.3%; P<.001) | |||||||
| 85 | 2009 | 68 (60) | Patients with established stable CAD | Anger recall periods | PAT measurements during MS compared with baseline | Single PET-CT MPI concurrent with PAT testing during MS protocol | 26 developed a new perfusion defect during MS | Sensitivity/specificity of PAT ratio as an index of ischemia on PET-CT MPI was 0.62/0.63 |
| Stress PAT response ratio: pulse wave amplitude during MS/at baseline | Patients with a new perfusion defect with MS had a lower stress PAT ratio (0.76±0.04 vs 0.91±0.05, P= .03) | Among patients taking ACE-I the sensitivity and specificity increased to 0.86 and 0.73, respectively | ||||||
| 90% of patients without ischemia were correctly identified | ||||||||
| 86 | 2009 | 211 (134) | Patients with established stable CAD | Two phases of a public speaking task (stress anticipation and task performance) | PAT measurements during MS compared with baseline | Rest-stress MPI | Vascular response in the anticipation period (speech preparation) was more pronounced than during the actual speaking task | Stress PAT ratio during speech preparation had modest accuracy for predicting MSIMI on MPI(AUC, 0.63; 95% CI, 0.53-0.74; P=.015) |
| Stress PAT response ratio: pulse wave amplitude during MS/at baseline | Mean preparation stress PAT ratio 0.64±0.53; mean speech stress PAT ratio 0.72±0.60; P<.001) | |||||||
| 74 | 2010 | 26 (0) | 12 females with a history of ABS; 12 post-menopausal controls; 4 with history of MI | Three different MS tasks of 6-min duration in random order (number-letter recall challenge of increasing length and complexity; number subtraction; Stroop word-color conflict) | RH-PAT at baseline (baseline PEF) compared with RH-PAT following MS (post-MS PEF) | Plasma catecholamine levels at baseline and following MS tests | RH-PAT following MS was lower in patients with ABS vs with post-menopausal controls (P<.05) | Catecholamine levels were increased in patients with ABS vs in post-menopausal controls, following MS testing (P<.05) |
| PAT measurements during MS compared with baseline | Stress PAT ratios were lower in patients with ABS vs with patients with MI and post-menopausal controls (P<.05) | |||||||
| Stress PAT response ratio: pulse wave amplitude during MS/at baseline | No differences in stress PAT ratio in patients with MI vs post-menopausal controls | |||||||
| 87 | 2011 | 25 (6) | Healthy subjects | Three different MS tasks of 6-min duration in random order (number-letter recall challenge of increasing length and complexity; number subtraction; Stroop word-color conflict) | RH-PAT at baseline (baseline PEF) compared with RH-PAT following MS (post-MS PEF) | Arterial blood pressure signal amplitude using cuff attached to a pressure transducer (BIOPAC MP150 systems technology — a standard polygraph device used to detect deception during polygraph examinations in military or law enforcement applications) | No significant difference in RH-PAT and BIOPAC arterial blood pressure signal amplitude at rest or following MS (1.55±0.36 and 1.48±0.19; P=.38 and 1.44±0.29 and 1.47±0.21; P)=.61, respectively) | Lower stress PAT ratio vs BIOPAC stress ratio during each of the 3 MS tasks |
| PAT measurements during MS compared with baseline | Ratio of BIOPAC arterial blood pressure signal amplitude during MS to baseline arterial blood pressure signal amplitude | No differences in RH-PAT ratios between male and female subjects (P=.75) | No difference in stress PAT ratios between male and female subjects (P> .05) | |||||
| Stress PAT response ratio: pulse wave amplitude during MS/at baseline | ||||||||
| 88 | 2011 | 241 (126) | Healthy adolescents (mean age, 10 y) | Three different MS tasks of 6-min duration in random order (number-letter recall challenge of increasing length and complexity; number subtraction; Stroop word-color conflict) | PAT measurements during MS compared with baseline | Physical activity using a self-report questionnaire | In response to MS, male adolescents had a more vasoconstrictive response, followed by a less vasodilatory response, and needed longer time to return to baseline level than females | Adolescents who reported decreased physical activity over a 3-y period had increased arterial stiffness |
| Stress PAT response ratio: pulse wave amplitude during MS/at baseline | ||||||||
| 89 | 2013 | 384 (159) | Patients with angiographically documented CAD | Standardized public speaking task | PAT measurements during MS compared with baseline | 99mTc-sestamibi MPI at rest and following both MS and physical stress testing, performed on separate days | Stress PAT ratio was lower in those with vs without MSIMI on MPI (0.55±0.36 vs 0.76±0.52; P=.009) | CAD severity and extent scores were not significantly different between those with or without MSIMI, whereas they were greater in those with compared with without physical stress induced ischemia (P<.04 for all) |
| Stress PAT response ratio: pulse wave amplitude during MS/at baseline | In a multivariable analysis, stress PAT ratio was the only independent predictor of MSIMI on MPI (P=.009) | Angiographic severity and extent of CAD independently predicted physical stress induced myocardial ischemia | ||||||
| 90 | 2017 | 660 (482) | Patients with established stable CAD | Standardized public speaking task | RH-PAT at baseline (baseline PEF) compared with RH-PAT following MS (post-MS PEF) | 99mTc sestamibi MPI at rest, with MS, and with conventional (exercise/pharmacological) stress | 106 (16.1%) developed MSIMI, and 229 (34.7%) had conventional stress-induced myocardial ischemia | Only presence of ischemia during conventional stress (OR, 7.1; 95% CI, 4.2-11.9), high hemodynamic response (OR for RPP response ≥ vs < ROC cutoff of 1.8; 95% CI, 1.1-2.8), and high digital vasoconstriction (OR for stress PAT ratio < vs ≥ ROC cutoff of 2.1; 95% CI, 1.3-3.3) were independent predictors of MSIMI |
| Pulse wave velocity using PAT measurements | Rate-pressure-product (heart rate × systolic blood pressure) epinephrine levels | MS was associated with increases in SBP, DBP, HR, epinephrine levels, PWV, and significant decreases in FMD and stress PAT ratio denoting microvascular constriction | ||||||
| PAT measurements during MS compared with baseline | Patients with vs without MSIMI had higher hemodynamic and digital vasoconstrictive responses (P<.05 for both), but did not differ in epinephrine, endothelial (RH-PAT after MS) or macrovascular (FMD) responses | |||||||
| Stress PAT response ratio: pulse wave amplitude during MS/at baseline | ||||||||
| Endothelium-dependent FMD before and after MS | ||||||||
| 76 | 2018 | 62 (0) | 41 patients with coronary vascular dysfunction and 21 controls | Anger recall, mental arithmetic, and forehead cold pressor challenge | RH-PAT at baseline (baseline PEF) compared with RH-PAT following MS (post-MS PEF) | Emotional arousal was measured (Likert scale) | During MS 10% of controls reported chest pain vs 41% of subjects with coronary vascular dysfunction (P=.01) | Vasoconstriction inversely correlated with anxiety (r=-3.4, P=.03), frustration (r=-0.37, P=.02), and feeling challenged (r=-0.37, P=.02) in patients with coronary vascular dysfunction only |
| PAT measurements during MS compared with baseline | RH-PAT did not change significantly after MS in either group | |||||||
| Stress PAT response ratio: pulse wave amplitude during MS/at baseline | Subjects with coronary vascular dysfunction had lower stress PAT ratios vs controls during mental arithmetic (0.54; 95% CI, 0.15-1.46 vs 0.67; 95% CI, 0.36-1.8; P=.039), not evident in the other tasks | |||||||
| 91 | 2018 | 418 (210) | 306 (150 females) subjects who were hospitalized for MI in the previous 8 months | Standardized public speaking task | RH-PAT at baseline (baseline PEF) compared with RH-PAT following MS (post-MS PEF) | 99mTc-sestamibi MPI at rest, with MS and conventional (exercise/pharmacological) stress | Women in both groups showed a higher stress PAT ratio and a lower RH-PAT index after MS indicating enhanced microvascular dysfunction after MS | Rate of MSIMI was twice as high in women as in men (22% vs 11%, P=.009), and ischemia with conventional stress was similarly elevated (31% vs 16%, P=.002) |
| 112 community controls (58 females) frequency matched for sex and age | PAT measurements during MS compared with baseline | No sex differences in FMD with MS | Stress PAT ratio and RH-PAT index after MS were predictive of MSIMI in women only | |||||
| Stress PAT response ratio: pulse wave amplitude during MS/at baseline | ||||||||
| Endothelium-dependent FMD before and after MS | ||||||||
| 92 | 2018 | 678 (492) | Patients with established stable CAD | Standardized public speaking task | PAT measurements during MS compared with baseline | MPI before and during MS | Women (but not men) with vs without MSIMI had a significantly lower stress PAT ratio (0.5 vs 0.8) | Men (but not women) with vs without MSIMI had a higher rate-pressure product response (6500 vs 4800 mm Hg beats/min) |
| Stress PAT response ratio: pulse wave amplitude during MS/at baseline | SBP × HR – rate pressure product | Each 0.10-U decrease in stress PAT ratio was associated with 0.23% (95% CI, 0.11-0.35) increase in inducible myocardial ischemia in women | Each 1000-U increase in rate-pressure product response was associated with 0.32% (95% CI, 0.22-0.42) increase in inducible ischemia among men | |||||
| Ratios <1 indicate vasoconstrictive response | ||||||||
| 75 | 2018 | 38 (32) | Patients with stable CAD defined by an abnormal coronary angiogram demonstrating angiographic evidence of atherosclerosis with at least luminal irregularities | Mental arithmetic testing | PAT measurements during MS compared with baseline | Invasive endothelium-dependent and endothelium-independent coronary epicardial and microvascular responses were measured using intracoronary acetylcholine and nitroprusside, respectively, and after MS | MS increased the rate-pressure product by 22% (±23%) and constricted epicardial coronary arteries by median, -5.9%; IQR, -0.5% to -2.6%; P=.001, without changing CBF | Stress PAT ratio correlated with the demand-adjusted change in CBF during MS (r=-0.60, P=.004) |
| Stress PAT response ratio: pulse wave amplitude during MS/at baseline | Acetylcholine increased CBF by 38.5% (8.1%, 91.3%), P=.001, without epicardial coronary diameter change (0.1% [-10.9%, 8.2%], P=NS) | |||||||
| MS-induced CBF response correlated with endothelium-dependent CBF changes with acetylcholine (r=0.38; P=.03) but not with the response to nitroprusside | ||||||||
| 93 | 2019 | 18 (0) | 8 females with a history of ABS; 10 post-menopausal controls | Three different MS tasks of 6-min duration in random order (number-letter recall challenge of increasing length and complexity; number subtraction; Stroop word-color conflict) | PAT measurements during MS compared with baseline | Pain induced PAT ratio | Stress PAT ratio was lower in | Pain-induced PAT ratios were attenuated in patients with ABS: |
| Stress PAT response ratio: pulse wave amplitude during MS/at baseline | patients with ABS: Stroop test (0.79±0.30 vs 1.24±0.43; P=.01); | at baseline (0.94±0.08 vs 1.30±0.54; P<.05); and post-MS (0.87±0.19 vs 1.24±0.21; P=.01) | ||||||
| Arithmetic test (0.91±0.27 vs 1.36±0.57, P=.01). | Pain-induced PAT ratios correlated significantly with stress PAT ratios, both in arithmetic and Stroop test (P<.05) | |||||||
| 94 | 2019 | 59 (44) | Patients with a history of stable CAD | Mental arithmetic testing, and public speaking stressors | PAT measurements during MS compared with baseline | PET imaging of the brain | Stress response ratios below the median were associated with increased stress activation in insula and parietal cortex, and decreased activation in the medial prefrontal cortex with MS tasks compared with control tasks | |
| Stress PAT response ratio: pulse wave amplitude during MS/at baseline | ||||||||
| 95 | 2020 | 486 (350) | Patients with stable coronary atherosclerosis | Series of standardized speech/arithmetic stressors | PAT measurements during MS compared with baseline | 99mTc-sestamibi MPI at rest, with MS, and with conventional (exercise/pharmacological) stress | After multivariable adjustment MSIMI was associated with 21% and 20% slower completion of Trail-A and Trail-B, respectively (P for all <.01) | Ischemia with a conventional stress test was not associated with any of the cognitive tests over time |
| Stress PAT response ratio: pulse wave amplitude during MS/at baseline | Cognitive function assessed at baseline and at a 2-y follow-up using Trail Making Test parts A and B, and the verbal and visual memory subtests of the Wechsler Memory Scale | After a 2-y follow-up period, presence of MSIMI was associated with a 33% slower completion of Trail-B, denoting cognitive decline (B = 0.33; 95% CI, 0.04-0.62) | ||||||
| A lower stress PAT ratio, indicating greater vasoconstriction, mediated the association between MSIMI and worsening Trail-B performance by 18.2% |
ABS, apical ballooning syndrome; ACE-I, angiotensin-converting enzyme inhibitor; AUC, area under the curve; BP, blood pressure; CAD, coronary artery disease; CBF, coronary blood flow; CT, computed tomography; CV, cardiovascular; ECG, electrocardiogram; EDAD, endothelial-dependent arterial dilatation; EF, ejection fraction; ERNA, equilibrium radionucleotide angiocardiography; FMD, flow-mediated dilatation; GTN, sublingual glyceryl trinitrate; MI, myocardial infarction; MPI, myocardial perfusion imaging; MS, mental stress; MSIMI, mental stress–induced myocardial ischemia; NS, not significant; OR, odds ratio; PAT, peripheral arterial tonometry; PEF, peripheral endothelial function; PET, positron emission tomography; PWV, pulse wave velocity; RH-PAT, reactive hyperemia peripheral arterial tonometry; ROC, receiver operating characteristic curve; RPP, rate pressure product; SVR, systemic vascular resistance
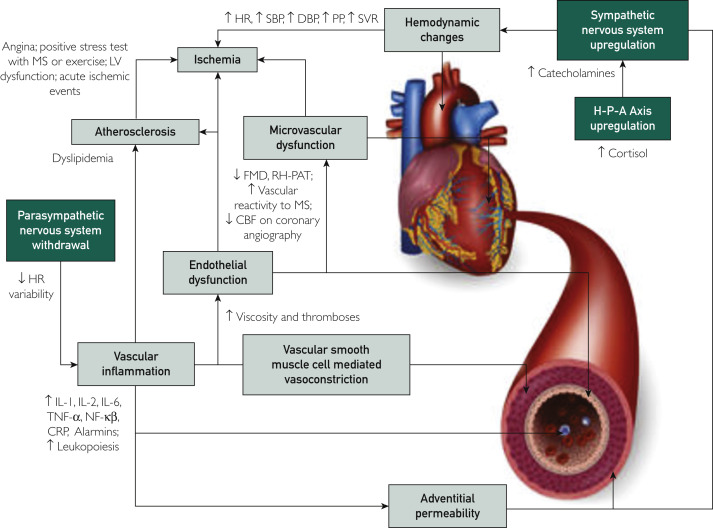
Figure 2.
Outline of the various mechanisms by which stress pathologically affects vascular health (in boxes), and biomarkers available to measure these effects.
CBF, coronary blood flow; CRP, C-reactive protein; DBP, diastolic blood pressure; FMD, flow-mediated dilatation; H-P-A axis, hypothalamic-pituitary-adrenal axis; HR, heart rate; IL, interleukin; LV, left ventricular; MS, mental stress; MSIMI, mental stress induced myocardial ischemia; NF-κβ; nuclear factor kappa light chain enhancer of activated B cells; PP, pulse pressure; RH-PAT, reactive hyperemia – peripheral arterial tonometry; SBP, systolic blood pressure; SVR, systemic vascular resistance; TNF, tumor necrosis factor.
Effects on the Endothelium
The intima makes up the innermost layer of the arterial wall and is lined by longitudinally orientated endothelial cells comprising the endothelium. The endothelium has the following functions: 1) preserving vascular tone, 2) mediating thrombogenesis and fibrinolysis, and 3) regulating the proliferation of vascular smooth muscle cells (VSMCs) in the media.96 , 97 Vascular tone is maintained through the balance between vasodilation and vasoconstriction, which is achieved through the release of active substances from endothelial cells that act on the VSMCs. Nitric oxide is a vasodilatory substance synthesized from L-arginine, using the enzyme endothelial nitric oxide synthase, and is released in response to shear stress; other chemicals such as acetylcholine, bradykinin, or serotonin; the release of thrombin; and stimulation of the parasympathetic nervous system.98 , 99 Additionally, nitric oxide inhibits VSMC growth, platelet aggregation, and the adhesion, migration, and proliferation of white blood cells.100 Conversely, endothelin-1, produced by endothelial cells, VSMCs, and activated macrophages,101 is a potent vasoconstrictor in of itself, and enhances the vasoconstricting effects of other substances including angiotensin II, serotonin, and catecholamines.102 Given its location on the inside surface of the vascular system, the endothelium is most exposed to sources of injury, including MS. Monkeys exposed to a new social group, and the associated changes to social structure and environment, had increased endothelial cell damage and turnover in the thoracic aorta and coronary arteries,103 and reduced nitric oxide availability in arteries with atherosclerosis.104 Similarly, a public speaking task105 and anger provocation 106 were shown to be associated with an increase in circulating endothelial cell-derived microparticles, derived from the membranes of apoptotic endothelial cells. Mental stress may also adversely influence endothelial cell function. In animal studies, acute and chronic MS were associated with lower levels of nitric oxide synthase mRNA expression106 , 107 leading to endothelial dysfunction. Further, MS may lead to oxidative stress and the release of potent vasoconstrictors, such as endothelin108 and angiotensin II,109 which also contribute to endothelial dysfunction. In one study, high serum biomarkers of oxidative stress were shown during the Great East Japan Earthquake in individuals with disaster-related hypertension defined as a systolic blood pressure > 140 mm Hg.110 Indeed, in a recently published series of experiments, endothelial cells appear to play a key effector role in the pathophysiologic response to acute mental stress,111 which leads to the stimulation of peripheral sympathetic nerves increasing locally available norepinephrine. Norepinephrine exerts primary effects on endothelial cells via a-adrenoceptors, leading to upregulation of adhesion molecules and release of chemokines. These effects are secondarily enhanced via the binding of norepinephrine to surface receptors of macrophages and VSMCs that release chemokines that further stimulate the effector endothelial cells. These effects collectively lead to leucocyte recruitment and adhesion. Concomitant endothelial dysfunction and increased endothelial cell permeability may then result in vascular inflammation, fibrous cap thinning, plaque instability, and ensuing cardiovascular events. Thus, the endothelium may be the site where the physiologic effects of MS are transduced into a measurable index (Table 2).
Effects on the Adventitia
The next layer out from the media is the adventitia, comprising connective tissue, fibroblasts, macrophages, and mast cells. The adventitia is perfused by the vasa vasorum, a microvascular bed, and is innervated by autonomic nerves with endings at adventitial mast cells close to the border with the media.112 By releasing neurotransmitters that act on vascular smooth muscle, these nerve endings regulate vascular tone. Sympathetic nervous fibers release norepinephrine, which, via alpha-1 receptors, cause vasoconstriction.113 In a direct mechanism, MS is associated with increased circulating levels of norepinephrine, correlating with increases in mean arterial pressure.114 Healthy individuals with self-reported high levels of daily psychosocial stress had greater microvascular vasoconstriction as measured using laser Doppler flow and enhanced responsiveness to norepinephrine compared with those with low stress.115 In an indirect mechanism, MS leads to the release of corticotropin-releasing hormone and the related peptide urocortin in the amygdala and the hypothalamus, which leads to increased levels of catecholamines, and upregulation of the sympathetic nervous system.116 , 117 As well as causing the release of norepinephrine that directly causes receptor mediated vasoconstriction, sympathetic nervous fibers also release substance P and calcitonin gene-related peptide, which trigger mast cell degranulation and the release of the vasoactive substances histamine and leukotriene.118 , 119 This results in vasodilatation and increased microvascular permeability, which underpins characteristic stress-induced symptoms such as sweating, flushing, and gastrointestinal disturbances, and can be inhibited with histamine blocking medications.120 More than just uncomfortable symptomology, MS-induced vascular mast cell degranulation has been shown to lead to plaque destabilization with intraplaque hemorrhage in areas of existing atherosclerosis in the animal model,121 as well as intraplaque hemorrhage in advanced atherosclerosis122 and myocardial infarction (MI) in the infarct-related artery in humans.123
Vascular Inflammation
Parasympathetic nervous system activation leads to release of acetylcholine in various organs including the heart, gastrointestinal tract, liver, and spleen. Acetylcholine binds to macrophage surface receptors blocking release of inflammatory cytokines including interleukin (IL) -1, -2, and -6, tumor necrosis factor alpha (TNF-alpha), and nuclear factor kappa-beta.124, 125, 126 This efferent cholinergic arm of the so-called inflammatory reflex can be triggered centrally via muscarinic acetylcholine receptor binding with ligands, and acetylcholinesterase inhibitors such as galantamine.127 Therefore, MS- induced withdrawal of parasympathetic nervous activity leads to enhanced release of proinflammatory cytokines, underpinned by an important difference between MS and physical (exercise)–related stress, where in the latter there is increased parasympathetic nerve discharge.128 Conversely, catecholamines bind to the beta-adrenergic receptors of macrophages and induce the expression of cytokines such as C-reactive protein, IL-1, IL-6, and TNF-alpha in a process that is enhanced under conditions of chronic MS.129 Indeed, elevated plasma cortisol, IL-1 beta, IL-2, and soluble intracellular adhesion molecule were demonstrated in healthy males after a structured speaking task.130 Stress can also lead to increased bone marrow leukopoietic proliferation through the activation of beta-3-adrenergic receptors by norepinephrine on progenitor inflammatory cells and macrophages.131, 132, 133, 134 These newly released inflammatory cells then produce further inflammatory cytokines and manifest greater expression of immune response genes in a feed-forward loop.135 Norepinephrine binding to the beta-3-adrenergic receptors of bone marrow stromal cells reduces the production of C-X-C chemokine ligand 12 that functions ordinarily to retain leukocytes in the bone marrow.131 , 132 , 136 This heightened innate immune cellular output, combined with enhanced cytokine production, can contribute to accelerated atherosclerosis.131 , 134 Further, acute MS induces IL-6 release from brown adipocytes in a beta-3-adrenergic receptor dependent fashion in the mouse model,137 highlighting the potential role of brown adipose tissue as a stress-responsive organ, and therefore potential target, implicated in arterial inflammation and MS-related CVD.
The release of peripherally circulating biomarkers (that have come to be known collectively as alarmins such as high mobility group box 1 and IL-1) from ischemic brain after induced stoke was a critical mechanism in activating downstream inflammatory pathways to exacerbate atherosclerosis.138 These molecules are prevalent in acute ischemic events and are linked to both heightened vascular inflammation and atherosclerotic disease progression and plaque vulnerability.138 Indeed, vascular inflammation and endothelial dysfunction are tightly coupled, as shown in an animal model that was exposed to chronic mild stress, in which impaired endothelial-dependent smooth muscle dilatation was improved after treatment with the TNF-alpha inhibitor infliximab.139
Atherosclerosis
Increased rates of atherosclerosis have been observed in animals with chronically elevated levels of stress.140 Studies have shown that brief episodes of experimental MS are associated with prolonged impairment of endothelial-dependent relaxation using measures such as brachial artery FMD,75 , 78 and RH-PAT.74 , 86 , 87 Given that endothelial dysfunction represents the first stage of atherosclerosis, and is associated with plaque progression and vulnerability,141, 142, 143, 144, 145, 146 it follows that MS incurred on a repeated or ongoing basis may lead to the initiation, acceleration, and complication of atherosclerotic CVD. Indeed, atherosclerosis has a long preclinical period, during which multiple potentially injurious risk factors act on the arterial wall. Studies have shown greater carotid intima media thickness, a marker of early atherosclerotic disease, in individuals from lower SES,147 and those with anxiety148 and depression.149 Moreover, rats exposed to chronic stress showed increases in the serum concentration of total cholesterol, triglycerides, low-density lipoprotein cholesterol, very low-density lipoprotein cholesterol, and atherogenic index, without any change to high-density lipoprotein cholesterol concentrations.150 Chronic unpredictable stress combined with a high-fat diet weakens reverse cholesterol transport, a process that removes excess cholesterol, which in turn exacerbates atherosclerosis.151 Chronic stress also promotes visceral fat accumulation, as opposed to subcutaneous fat, with subsequent progression of atherosclerosis and incident CVD events.152
Hemodynamic Effects
Both physical stress and MS are associated with increases in heart rate and blood pressure, with incumbent increases in stroke volume and cardiac output.153 An important difference between the two types of stress is that physical stress is associated with peripheral vasodilatation, such that the augmented cardiac output can match increased demands, resulting in an increase in oxygen consumption. However, MS tends to either be associated with a slight decrease in systemic vascular resistance, characterized by a pre-emptive “fight or flight” response similar to that which precedes physical stress, or more commonly an increase in vascular resistance associated with no change in oxygen consumption.154 Changes in hemodynamic parameters associated with MS including blood pressure and left ventricular function are outlined in Table 1. These changes may represent acute triggers for CVD events, or may lead to CVD progression and worse longer-term prognosis.44 Repeated and prolonged episodes of stress lead to vasoconstriction, endothelial dysfunction, vascular hypertrophy, and changes in vessel architecture that include a decreased lumen diameter and reduced density of microvessels. This occurs in parallel with increased large arterial rigidity secondary to increased collagen deposition, and loss of elastin and glycosaminoglycans within the vascular matrix.155 This remodeling is associated with changes in the concentration of vasoactive substances such as nitric oxide, angiotensin II, and norepinephrine leading to the development and progression of hypertension,156 which itself is associated with increased physical stress on blood vessel walls and further remodeling. Elevations in pulse pressure, which result mainly from increased rigidity of large arteries, is an important risk factor for CVD morbidity and mortality independent of absolute blood pressure.157 Increased pulse pressure transmitted to the capillary endothelium also contributes further to endothelial cell injury.158 Finnish men with greater systolic and diastolic blood pressure reactivity to experimental MS had higher degrees of carotid intima media thickness at baseline,159 as well as greater increases in carotid intima media thickness at 4-years follow-up.160
Hemodynamic responses to stress can be influenced by a number of interacting factors. First, women respond predominantly with an increase in heart rate, whereas men respond with increases in diastolic blood pressure and peripheral vascular resistance. Women also have comparatively lower vascular resistance at rest and with MS.161 Second, African American men had higher peripheral vascular resistance than White men during public speaking tasks.161 Third, individuals with concurrent high levels of depressive symptoms have greater increases in systemic vascular resistance in response to mirror tracing.162 Fourth, individuals of lower SES have delayed blood pressure recovery after stress, and have greater carotid intima media thickness than individuals who have normal blood pressure recovery after stress or those in higher SES groups.163
Ischemia
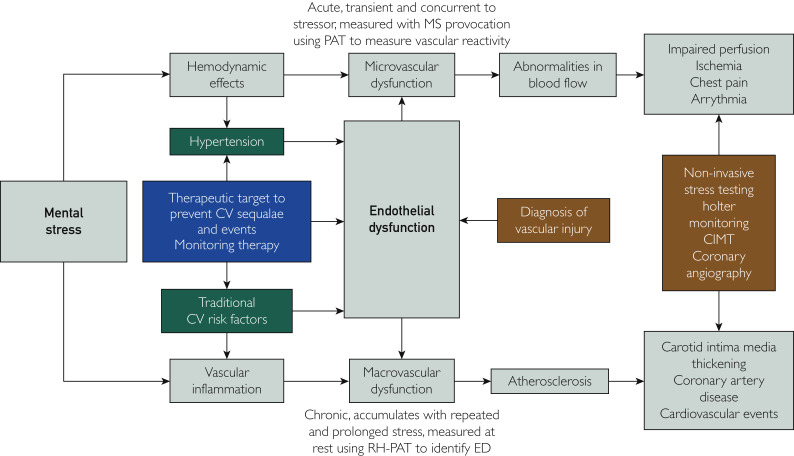
Myocardial blood flow is regulated through changes in microvascular resistance, which occurs through the sympathetic nervous system innervating the coronary arterioles via alpha-adrenergic receptors, and the myocardium via beta-adrenergic receptors.164 These receptors are the sites where the catecholamines epinephrine and norepinephrine bind and cause vasoconstriction. Thus, MSIMI is the downstream clinical manifestation of the combined physiologic effects of increased heart rate and blood pressure, endothelial dysfunction, and altered coronary blood flow70 in response to MS. Interestingly, angiographically normal epicardial coronary arteries show vasodilatation and increased coronary blood flow in the setting of MS.165 Conversely, MS is associated with local vasoconstriction in segments with epicardial atherosclerotic disease and stenosis, although the reductions in coronary blood flow are above and beyond that which can be explained by epicardial vasoconstriction alone, implicating the role of stress-induced increases in the resistance of the coronary microcirculation.166 This was confirmed in a study using coronary Doppler flow, which showed greater microvascular resistance in the setting of MS in patients with nonobstructive CAD.167 Further, patients with CAD exposed to MS while undergoing myocardial perfusion imaging had reduced coronary blood flow in territories without epicardial stenosis, suggesting the presence of heightened microvascular resistance.166 An important shortcoming of such diagnostic imaging techniques relates to the fact that 80% of the resistance to blood flow in the coronary circulation is regulated by the microvasculature, which comprises most of the endothelium. Noninvasive stress tests are poor predictors of endothelial-dependent and -independent coronary microvascular abnormalities168 as they lack the sensitivity to detect the subendocardial ischemia associated with these microvascular changes. They also rely on identifying areas of myocardium with relatively lower levels of perfusion compared with adjacent areas, whereas microvascular dysfunction typically produces diffuse ischemia.169 Additionally, imaging modalities such as echocardiography and myocardial perfusion imaging only provide a single snapshot of certain indices of function, such as left ventricular ejection fraction, at one cross-section in time. On the other hand, markers of endothelial function may provide an integrated index of vascular injury that has been accrued over time. Therefore, such indices may provide information more representative of the cumulative and dynamic impact of MS that could then form an index of risk and therapeutic target (see Figure 3 ).
Figure 3.
Outline of how mental stress (MS) leads to microvascular and macrovascular endothelial dysfunction (ED), how each of these can lead to pathologic and clinical manifestations of cardiovascular (CV) disease that can be measured, and how ED could be used as a diagnostic and therapeutic target when managing MS and its associated risk of CV disease.
CIMT, carotid intima media thickness; PAT, peripheral arterial tonometry; RH-PAT, reactive hyperemia–peripheral arterial tonometry.
The Connections Between Neuropsychiatry and Cardiovascular Disease
Our brains are continuously processing streams of stimuli, only a small fraction of which are selected for further processing in our salience network. Within this system, the amygdala plays an important role through its connections with the hypothalamus, and thence the pituitary gland in the hypothalamic-pituitary-adrenal (HPA) axis170 leading to the well-characterized “fight or flight” autonomic and hormonal stress responses. The amygdala also forms further connections to the periaqueductal gray which influences our behavior.171 In addition, the prefontal cortex is involved in cognitive appraisal of sensory stimuli and, through its downstream neuronal projections onto the amygdala and brainstem, it can further modulate the stress response with higher order regulation.172 Further, the dorsal anterior cingulate cortex also plays a role in regulating cardiovascular reactivity to cognitively demanding tasks,173 as well as the expression of proinflammatory cytokines and preclinical atherosclerosis after reappraisal of emotional stimuli.174 In contrast, greater social support levels are associated with lesser activity in the dorsal anterior cingulate cortex175 further supporting the notion of differential regional modulation of the neurally mediated stress response.
Neuroimaging modalities such as 18F-fluorodeoxyglucose (18F-FDG) PET/computed tomography (CT) are used to determine resting metabolic activity in different parts of the body, including the brain. Studies in primates have shown that increased amygdala uptake is associated with an anxious disposition in adults,176 and predicts future temperament in juveniles.177 In addition, functional magnetic resonance imaging (fMRI) and PET alone can provide insight into neural activity and connectivity between regions of the brain that can be correlated with the timing of stressors. Studies using both fMRI and PET have shown that stressful stimuli lead to the activation of the amygdala, which then through its connections with the hypothalamus results in activation of the HPA axis, enhanced sympathetic nervous activity and withdrawal of parasympathetic nervous activity, and stimulation of the renin-angiotensin-aldosterone system.170 , 178 , 179 Thus, through its connections with the limbic system, where the amygdala is located, the cerebral cortex transmits the experience of MS into a systemic physiological response mediated by catecholamines and stress hormones such as cortisol. Stress-induced catecholamine and cortisol underpin important hemodynamic, immunologic, and vascular changes that contribute not only to hypertension, adiposity, and insulin resistance,180 but also to atherosclerotic disease progression. In one study using fMRI, individuals with heightened amygdala activation and functional connectivity between the amygdala and anterior cingulate cortex had a greater burden of subclinical atherosclerosis.181 When using 18F-FDG PET/CT, amygdala activity, adjusted for temporal lobe or prefrontal cortex activity, was associated with CVD events that occurred with increased circulating inflammatory markers and leukopoietic activity, and greater degrees of atherosclerosis.182 In another study using 18F-FDG PET/CT with concurrent CT coronary angiography increased, adjusted amygdala activity was associated with greater bone marrow leukopoietic activity and atherosclerosis, including noncalcified coronary plaque volume.183 Further, increased adjusted amygdala activity was associated with incident diabetes independent of adiposity,184 as well as increased leukopoietic activity, abnormal myocardial perfusion, and decreased left ventricular function in female patients.185 Curiously, amygdala activity varies among individuals, even if all are uniformly exposed to stress; and individuals with lower amygdala activity despite exposure to stress were protected from CVD.186 Such neurobiological-resilient individuals may provide insight into how individual perceptions of one’s circumstance can influence regional brain activity and subsequent disease. In this way, the pathogenic effects of stress seem to be transduced along a neuroendocrine immunologic axis, terminating downstream on the vasculature where it then manifests as CVD.
Future Directions
Although there is evidence that stress is associated with CVD, there are critics who postulate that stress may in fact be a marker rather than a mechanism for CVD.187 This notion is based on the lack of high-quality robust evidence supporting the mechanism hypothesis. Indeed, current studies evaluating the relationship between stress and CVD have a number of limitations. Below we outline some suggested areas for improvement for future studies evaluating the biological effects of MS.
Generalizability
Studies using experimentally created MS tasks within the laboratory setting have questionable generalizability. Physiologic responses to stress are vulnerable to situation and individual factors,188 and these variables are often not easy to identify or control for. Opportunities for improving the real-life validity and generalizability of the effects of experimental MS include the following: 1) by using the aggregation of scores across multiple tasks and within tasks across all laboratory periods of stress including baseline, anticipation, reactivity, and recovery — each measurement contains random error that can be reduced by using multiple measurements, whereas aggregating scores enhances the diversity of situations sampled that may be more representative of real-life; 2) reducing error by performing multiday ambulatory assessments at home, similar to those used when assessing for hypertension — ambulatory blood pressure monitoring better predicts CVD than office blood pressure recordings189; and 3) social tasks may be more representative of daily life stressors than traditionally used cognitive activities. Type A interviews, discussing anger-provoking events, and listening to a competitor all show greater generalizability and ecologic validity.190, 191, 192
Timing of Stress
Pathologic effects of stress should be ascribed to the cumulative duration of the physiologic responses to stress, and not just to the temporal exposure to the stressor itself. Thus, pathogenicity increases not only with repeated or chronic exposure to a stressor, but also when cognitive and emotional processes perpetuate physiologic responses even in the temporal absence of that stressor, such as when ruminating. Indeed, recall of an anger-provoking event increased the difficulty in terminating ventricular tachycardia in arrhythmia-prone patients193 and reduced left ventricular fraction in patients with CVD.194 There is currently no accepted method for measuring anticipation or recovery from MS and most commonly the “change in scores from baseline” and “time to return to baseline” have been used.195 Delayed recovery in blood pressure from a laboratory psychological stressor has been shown to be predictive of incident hypertension even after accounting for initial blood pressure and traditional risk factors.196 , 197 Thus, future studies evaluating the impact of stress should develop models that also account for the physiologic effects of anticipating, recovering from, and ruminating over a stressor.
Other Interacting Factors
The causes of CVD are not completely understood by examining single factors in isolation, but instead through the consideration of a constellation of variably interacting factors such as behaviors, environment, and genetic predispositions. Epidemiologic studies have shown that migration from rural to urban environments is associated with higher blood pressures in those living in urban compared with rural areas.198, 199, 200 Although this effect was attributed to adopting Western lifestyle patterns, the changes in blood pressure were not the universal experience of all migrants, suggesting inter-individual differences in susceptibility. Pharmacogenetics studies have highlighted certain gene-environment interactions related to stress and CVD and, in one study, individuals with at least one copy of a particular allele had greater blood pressure reactivity to stress than those who lacked the allele.201 Studies using 18FDG-PET/CT imaging showed that individuals from lower SES neighborhoods202 and those exposed to transportation noise exposure203 had increased amygdala activity, increased atherosclerosis, and increased subsequent CVD events, suggesting that socioeconomic hardship can lead physiologically susceptible individuals to experience the effects of stress. Therefore, genetic, environmental, and other biopsychosocial risk factors should be evaluated in the context of the stressor to help better elucidate mechanistic pathways, identify individuals at risk, and target interventions appropriately.
Treatment Strategies
Stress Management, Resiliency, and Pharmacotherapy — Targeting Mental Stress
In the 2016 European clinical guidelines for the prevention of CVD, MS is acknowledged as a potential contributing factor to both development and progression of disease, for which a targeted rather than universal management strategy is recommended. The evidence for MS as a CVD risk factor is rated as class IIa, indicating that the weight of evidence favors efficacy such that managing stress as a risk factor “should be considered.” Thus, assessing and managing MS in individuals with established CVD or at high risk of disease is now guideline-recommended.204 Candidate therapeutic options targeting stress to reduce risk of CVD include nonpharmacological strategies (such as relaxation techniques, “mental exercise” including basic changes in philosophic outlook and empirically based cognitive behavioral therapy [CBT], meditation, graded adaptation to stressors, and physical activity) and pharmacological strategies. Although of considerable interest, mental training remains poorly defined and inadequately studied. Broadly speaking, meditation is designed to “improve concentration, increase awareness of the present moment, and familiarize a person with the nature of the mind.”205 In summary, current evidence shows the following: 1) nonrandomized studies suggest that meditative practices are beneficial in reducing the risk of CVD; 2) no firm conclusions can be made on the effects of meditation on endothelial function, subclinical atherosclerosis, or metabolic syndrome; and 3) although primary prevention studies report reductions in mortality, secondary prevention studies do not show any clear benefit.205 Indeed, current evidence is limited by small sample sizes, heterogeneous study populations, implementation of different strategies for stress reduction, and varying study endpoints, making it challenging to draw firm conclusions. Similarly, although evidence for the effectiveness of CBT is robust for psychological distress in the general population,206 effectiveness of CBT in CVD is unclear. Yet, given that meditation and CBT-based strategies are low-cost, low-risk approaches, and have the potential for widespread application, they could be considered as adjuncts to additional established lifestyle measures. As an example, a randomized controlled trial did show fewer adverse CVD events in individuals who underwent stress management in addition to cardiac rehabilitation, compared with cardiac rehabilitation alone, prompting the incorporation of stress management into cardiac rehabilitation programs.207 Nonetheless, the benefits of meditation and other nonpharmacological strategies remain to be clearly demonstrated, and adequately powered, well-designed randomized controlled trials are required. Further, given that exercise training improves endothelial function, reduces catecholamine release, and increases peripheral oxygen extraction,208 it is likely to also have benefits in stress management, although randomized trials here are also lacking. In both mental and physical training, digital health, wearables, and remote monitoring are likely to play important roles in study design as well as in developing individualized patient “prescriptions,” and for monitoring progress and efficacy of treatment. Along these lines, considerable attention and research activities have focused on virtual reality, high-density electroencephalogram neurofeedback and other methods to reduce MS.209 These tools offer objective measures of neural activity that can be observed in real-time during stress management activities and could provide the basis for goal-directed training and tracking longitudinal progress over time.
From a pharmacologic perspective, the serotonin reuptake inhibitor escitalopram has been studied in randomized controlled trials, including two 6-week long studies in patients with stable CAD and baseline MSIMI. Both studies showed that the incidence of MSIMI at follow-up was reduced in the experimental group compared with placebo.59 , 210 The mechanism of escitalopram’s actions in this context is unclear but may relate to the regulation and binding affinity of platelet serotonin receptors.59 , 210 Interestingly, in a study that included 152 adult outpatients with confirmed COVID-19 who were randomized to the serotonin reuptake inhibitor fluvoxamine vs placebo, those taking fluvoxamine had a lower likelihood of clinical deterioration within 15 days.211 These data have recently been replicated in high-risk outpatients in a study that included more than 1400 participants.212 Although the mechanism of this effect remains to be determined, unlike other serotonin reuptake inhibitors, fluvoxamine interacts strongly with the sigma-1 receptor, a protein inside cells that helps regulate the body's inflammatory response as well as reducing anxiety and depression.213 In this way the sigma-1 receptor could potentially offer a novel therapeutic target that could be the basis of future studies targeting the effects of MS. Further, given that the pathogenesis of stress-induced CVD and MSIMI is related to vasoconstriction and microcirculatory disease, alpha- and beta-blocking drugs as well as angiotensin II receptor blockers214 would seem to be useful agents, but randomized trials evaluating their efficacy in managing stress-related CVD are lacking. Given our evolving insight into the neuroendocrine immunologic axis mediating between MS and vascular disease, exploring the potential role of novel therapeutics targeting proinflammatory cytokines such as canakinumab,215 among others, may also be of great value.
CVD Risk Prevention — Targeting Endothelial Dysfunction
As the putative link between MS and CVD appears to be mediated by vascular injury and endothelial dysfunction, risk prevention strategies should also be targeted to the latter. In this way, increased vascular reactivity and endothelial dysfunction could be detected to diagnose the pathologic effects of MS on the vascular system (Figure 3). Endothelial dysfunction could then be used as a therapeutic target to prevent CVD and its consequences, and its measurements could be followed longitudinally to monitor treatment response. Traditional and other nonconventional CVD risk factors, such as unhealthy nutrition and sleep deprivation, also lead to endothelial dysfunction and contribute to CVD risk; therefore, the quantity of measured endothelial dysfunction may not consistently correspond to a given amount of MS per se. However, the identification of endothelial dysfunction as an integrated index of risk accumulated through the net effects of beneficial and harmful factors would signify the presence of vascular injury and increased cardiovascular risk, and thus would still prompt CVD preventive efforts. This could be achieved by not only screening for and managing MS itself to prevent vascular injury, but also by targeting the vascular injury itself. Traditional behavioral interventions known to prevent CVD including smoking cessation, diet modification, and physical activity could be implemented.216 Similarly, pharmacologic strategies such as statins that are known to improve endothelial dysfunction and the risk of CVD217 could also be used. Other traditional CVD risk factors that are known to separately contribute to vascular injury such as diabetes mellitus and hypertension should also be identified and treated in accordance with practice guidelines.218 Further, applying treatment strategies targeted to the adverse vascular effects of one risk factor may help to mitigate the deleterious effects of another risk factor without addressing the second risk factor directly. For example, regular aerobic exercise training has been shown to mitigate endothelial dysfunction related to insufficient sleep, and in doing so could reduce CVD risk associated with habitual insufficient nightly sleep.219 In this way, although therapies that target MS itself presently lack conclusive evidence and are continuing to be studied, patients with MS can still be effectively managed by harnessing our existing knowledge of how best to manage CVD risk factors using endothelial dysfunction as the integrated target that ultimately mediates CVD.
Conclusion
Conventional clinical practice often fails to incorporate an assessment of MS and its potential role as a risk factor for CVD. Our rapidly evolving understanding of the role that stress plays in CVD may change this. This has been brought even more greatly to the fore with the current COVID-19 global pandemic with its incumbent policies of social distancing, and our increasing awareness of the deleterious health consequences of life-changing events and loneliness. In fact, assessing and managing MS in individuals with established CVD or at high risk of disease is now guideline-recommended. Nevertheless, greater recognition of the dangers of MS and its impact on CVD will center on more carefully defining stress when communicating in scientific circles, and in making use of ways to quantify the physiologic responses and disease-causing consequences of stress, particularly with respect to endothelial function and vascular health where the downstream impact of stress appears to be transduced. Developing these methods for widespread clinical use will rely on improved study design as well as developing disease causative models that factor in additional biopsychosocial factors that also contribute to CVD. Greater attention must also be given to developing treatment strategies that target MS and endothelial dysfunction directly along the neuroendocrine immunologic axis in ways that effectively reduced the risk of CVD. Digital health and remote monitoring will likely play a role in this which, similar to stress, will undoubtedly continue being pervasive elements in all our lives.
Footnotes
Potential Competing Interests: The authors report no potential competing interests.
References
- 1.Global Burden of Disease 2016 Causes of Death Collaborators. Global, regional, and national age-sex specific mortality for 264 causes of death, 1980–2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet. 2017;390(10100):1151–1210. doi: 10.1016/S0140-6736(17)32152-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Ezzati M., Obermeyer Z., Tzoulaki I., Mayosi B.M., Elliott P., Leon D.A. Contributions of risk factors and medical care to cardiovascular mortality trends. Nat Rev Cardiol. 2015;12(9):508–530. doi: 10.1038/nrcardio.2015.82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Tzoulaki I., Elliott P., Kontis V., Ezzati M. Worldwide exposures to cardiovascular risk factors and associated health effects: current knowledge and data gaps. Circulation. 2016;133(23):2314–2333. doi: 10.1161/CIRCULATIONAHA.115.008718. [DOI] [PubMed] [Google Scholar]
- 4.Werneburg B.L.H.L., Preston H.R., Rausch S.M., Warren B.A., Olsen K.D., Clark M.M. Effectiveness of a multidisciplinary worksite stress reduction programme for women. Stress Health. 2011;27(5):356–364. [Google Scholar]
- 5.Clark M.M., Warren B.A., Hagen P.T., et al. Stress level, health behaviors, and quality of life in employees joining a wellness center. Am J Health Promot. 2011;26(1):21–25. doi: 10.4278/ajhp.090821-QUAN-272. [DOI] [PubMed] [Google Scholar]
- 6.Nyklicek I., Kuijpers K.F. Effects of mindfulness-based stress reduction intervention on psychological well-being and quality of life: is increased mindfulness indeed the mechanism? Ann Behav Med. 2008;35(3):331–340. doi: 10.1007/s12160-008-9030-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Dar T., Radfar A., Abohashem S., Pitman R.K., Tawakol A., Osborne M.T. Psychosocial stress and cardiovascular disease. Curr Treat Options Cardiovasc Med. 2019;21(5):23. doi: 10.1007/s11936-019-0724-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Perk J., De Backer G., Gohlke H., et al. European guidelines on cardiovascular disease prevention in clinical practice (version 2012): the fifth joint task force of the European Society of Cardiology and other societies on cardiovascular disease prevention in clinical practice (constituted by representatives of nine societies and by invited experts) Atherosclerosis. 2012;223(1):1–68. doi: 10.1016/j.atherosclerosis.2012.05.007. [DOI] [PubMed] [Google Scholar]
- 9.Madsen I.E.H., Nyberg S.T., Magnusson Hanson L.L., et al. Job strain as a risk factor for clinical depression: systematic review and meta-analysis with additional individual participant data. Psychol Med. 2017;47(8):1342–1356. doi: 10.1017/S003329171600355X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Hackett R.A., Steptoe A. Type 2 diabetes mellitus and psychological stress — a modifiable risk factor. Nat Rev Endocrinol. 2017;13(9):547–560. doi: 10.1038/nrendo.2017.64. [DOI] [PubMed] [Google Scholar]
- 11.Batty G.D., Russ T.C., Stamatakis E., Kivimaki M. Psychological distress in relation to site specific cancer mortality: pooling of unpublished data from 16 prospective cohort studies. BMJ. 2017;356:j108. doi: 10.1136/bmj.j108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Kivimaki M., Kawachi I. Work stress as a risk factor for cardiovascular disease. Curr Cardiol Rep. 2015;17(9):630. doi: 10.1007/s11886-015-0630-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Dragano N., Siegrist J., Nyberg S.T., et al. Effort-reward imbalance at work and incident coronary heart disease: a multicohort study of 90,164 individuals. Epidemiology. 2017;28(4):619–626. doi: 10.1097/EDE.0000000000000666. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Kivimaki M., Jokela M., Nyberg S.T., et al. Long working hours and risk of coronary heart disease and stroke: a systematic review and meta-analysis of published and unpublished data for 603,838 individuals. Lancet. 2015;386(10005):1739–1746. doi: 10.1016/S0140-6736(15)60295-1. [DOI] [PubMed] [Google Scholar]
- 15.Huang Y., Xu S., Hua J., et al. Association between job strain and risk of incident stroke: a meta-analysis. Neurology. 2015;85(19):1648–1654. doi: 10.1212/WNL.0000000000002098. [DOI] [PubMed] [Google Scholar]
- 16.Rosengren A., Hawken S., Ounpuu S., et al. Association of psychosocial risk factors with risk of acute myocardial infarction in 11,119 cases and 13,648 controls from 52 countries (the INTERHEART study): case-control study. Lancet. 2004;364(9438):953–962. doi: 10.1016/S0140-6736(04)17019-0. [DOI] [PubMed] [Google Scholar]
- 17.Hemingway H., Marmot M. Evidence based cardiology: psychosocial factors in the aetiology and prognosis of coronary heart disease. Systematic review of prospective cohort studies. BMJ. 1999;318(7196):1460–1467. doi: 10.1136/bmj.318.7196.1460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Chida Y., Steptoe A. The association of anger and hostility with future coronary heart disease: a meta-analytic review of prospective evidence. J Am Coll Cardiol. 2009;53(11):936–946. doi: 10.1016/j.jacc.2008.11.044. [DOI] [PubMed] [Google Scholar]
- 19.Grande G., Romppel M., Barth J. Association between type D personality and prognosis in patients with cardiovascular diseases: a systematic review and meta-analysis. Ann Behav Med. 2012;43(3):299–310. doi: 10.1007/s12160-011-9339-0. [DOI] [PubMed] [Google Scholar]
- 20.Gan Y., Gong Y., Tong X., et al. Depression and the risk of coronary heart disease: a meta-analysis of prospective cohort studies. BMC Psychiatry. 2014;14:371. doi: 10.1186/s12888-014-0371-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Celano C.M., Millstein R.A., Bedoya C.A., Healy B.C., Roest A.M., Huffman J.C. Association between anxiety and mortality in patients with coronary artery disease: a meta-analysis. Am Heart J. 2015;170(6):1105–1115. doi: 10.1016/j.ahj.2015.09.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Russ T.C., Stamatakis E., Hamer M., Starr J.M., Kivimaki M., Batty G.D. Association between psychological distress and mortality: individual participant pooled analysis of 10 prospective cohort studies. BMJ. 2012;345:e4933. doi: 10.1136/bmj.e4933. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Edmondson D., Kronish I.M., Shaffer J.A., Falzon L., Burg M.M. Posttraumatic stress disorder and risk for coronary heart disease: a meta-analytic review. Am Heart J. 2013;166(5):806–814. doi: 10.1016/j.ahj.2013.07.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Brunner E. Stress and the biology of inequality. BMJ. 1997;314(7092):1472–1476. doi: 10.1136/bmj.314.7092.1472. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kanjilal S., Gregg E.W., Cheng Y.J., et al. Socioeconomic status and trends in disparities in 4 major risk factors for cardiovascular disease among US adults, 1971–2002. Arch Intern Med. 2006;166(21):2348–2355. doi: 10.1001/archinte.166.21.2348. [DOI] [PubMed] [Google Scholar]
- 26.Kumari M., Head J., Marmot M. Prospective study of social and other risk factors for incidence of type 2 diabetes in the Whitehall II study. Arch Intern Med. 2004;164(17):1873–1880. doi: 10.1001/archinte.164.17.1873. [DOI] [PubMed] [Google Scholar]
- 27.Markovitz J.H., Matthews K.A., Whooley M., Lewis C.E., Greenlund K.J. Increases in job strain are associated with incident hypertension in the CARDIA study. Ann Behav Med. 2004;28(1):4–9. doi: 10.1207/s15324796abm2801_2. [DOI] [PubMed] [Google Scholar]
- 28.Kearns N.T., Carl E., Stein A.T., et al. Posttraumatic stress disorder and cigarette smoking: a systematic review. Depress Anxiety. 2018;35(11):1056–1072. doi: 10.1002/da.22828. [DOI] [PubMed] [Google Scholar]
- 29.Morera L.P., Marchiori G.N., Medrano L.A., Defago M.D. Stress, dietary patterns and cardiovascular disease: a mini-review. Front Neurosci. 2019;13:1226. doi: 10.3389/fnins.2019.01226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Sharma S., Malarcher A.M., Giles W.H., Myers G. Racial, ethnic and socioeconomic disparities in the clustering of cardiovascular disease risk factors. Ethn Dis. 2004;14(1):43–48. [PubMed] [Google Scholar]
- 31.Anand S.S., Islam S., Rosengren A., et al. Risk factors for myocardial infarction in women and men: insights from the INTERHEART study. Eur Heart J. 2008;29(7):932–940. doi: 10.1093/eurheartj/ehn018. [DOI] [PubMed] [Google Scholar]
- 32.Carey I.M., Shah S.M., DeWilde S., Harris T., Victor C.R., Cook D.G. Increased risk of acute cardiovascular events after partner bereavement: a matched cohort study. JAMA Intern Med. 2014;174(4):598–605. doi: 10.1001/jamainternmed.2013.14558. [DOI] [PubMed] [Google Scholar]
- 33.Smyth A., O'Donnell M., Lamelas P., et al. Physical activity and anger or emotional upset as triggers of acute myocardial infarction: the INTERHEART study. Circulation. 2016;134(15):1059–1067. doi: 10.1161/CIRCULATIONAHA.116.023142. [DOI] [PubMed] [Google Scholar]
- 34.Vahtera J., Kivimaki M., Pentti J., et al. Organisational downsizing, sickness absence, and mortality: 10-town prospective cohort study. BMJ. 2004;328(7439):555. doi: 10.1136/bmj.37972.496262.0D. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Kario K., McEwen B.S., Pickering T.G. Disasters and the heart: a review of the effects of earthquake-induced stress on cardiovascular disease. Hypertens Res. 2003;26(5):355–367. doi: 10.1291/hypres.26.355. [DOI] [PubMed] [Google Scholar]
- 36.Qureshi E.A., Merla V., Steinberg J., Rozanski A. Terrorism and the heart: implications for arrhythmogenesis and coronary artery disease. Card Electrophysiol Rev. 2003;7(1):80–84. doi: 10.1023/a:1023607527038. [DOI] [PubMed] [Google Scholar]
- 37.Wilbert-Lampen U., Leistner D., Greven S., et al. Cardiovascular events during World Cup soccer. N Engl J Med. 2008;358(5):475–483. doi: 10.1056/NEJMoa0707427. [DOI] [PubMed] [Google Scholar]
- 38.Sara J.D., Prasad M., Eleid M.F., Zhang M., Widmer R.J., Lerman A. Association between work-related stress and coronary heart disease: a review of prospective studies through the job strain, effort-reward balance, and organizational justice models. J Am Heart Assoc. 2018;7(9) doi: 10.1161/JAHA.117.008073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Orth-Gomer K., Wamala S.P., Horsten M., Schenck-Gustafsson K., Schneiderman N., Mittleman M.A. Marital stress worsens prognosis in women with coronary heart disease: the Stockholm Female Coronary Risk study. JAMA. 2000;284(23):3008–3014. doi: 10.1001/jama.284.23.3008. [DOI] [PubMed] [Google Scholar]
- 40.Albert M.A., Durazo E.M., Slopen N., et al. Cumulative psychological stress and cardiovascular disease risk in middle aged and older women: Rationale, design, and baseline characteristics. Am Heart J. 2017;192:1–12. doi: 10.1016/j.ahj.2017.06.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Fang F., Fall K., Mittleman M.A., et al. Suicide and cardiovascular death after a cancer diagnosis. N Engl J Med. 2012;366(14):1310–1318. doi: 10.1056/NEJMoa1110307. [DOI] [PubMed] [Google Scholar]
- 42.Schulz R., Beach S.R. Caregiving as a risk factor for mortality: the Caregiver Health Effects study. JAMA. 1999;282(23):2215–2219. doi: 10.1001/jama.282.23.2215. [DOI] [PubMed] [Google Scholar]
- 43.Carlsson A.C., Starrin B., Gigante B., Leander K., Hellenius M.L., de Faire U. Financial stress in late adulthood and diverse risks of incident cardiovascular disease and all-cause mortality in women and men. BMC Public Health. 2014;14:17. doi: 10.1186/1471-2458-14-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Kivimaki M., Steptoe A. Effects of stress on the development and progression of cardiovascular disease. Nat Rev Cardiol. 2018;15(4):215–229. doi: 10.1038/nrcardio.2017.189. [DOI] [PubMed] [Google Scholar]
- 45.Cohen S., Kamarck T., Mermelstein R. A global measure of perceived stress. J Health Soc Behav. 1983;24(4):385–396. [PubMed] [Google Scholar]
- 46.Steptoe A., Fieldman G., Evans O., Perry L. Cardiovascular risk and responsivity to mental stress: the influence of age, gender and risk factors. J Cardiovasc Risk. 1996;3(1):83–93. [PubMed] [Google Scholar]
- 47.Deanfield J.E., Shea M., Kensett M., et al. Silent myocardial ischaemia due to mental stress. Lancet. 1984;2(8410):1001–1005. doi: 10.1016/s0140-6736(84)91106-1. [DOI] [PubMed] [Google Scholar]
- 48.Jern S., Pilhall M., Jern C., Carlsson S.G. Short-term reproducibility of a mental arithmetic stress test. Clin Sci (Lond) 1991;81(5):593–601. doi: 10.1042/cs0810593. [DOI] [PubMed] [Google Scholar]
- 49.Manuck S.B., Olsson G., Hjemdahl P., Rehnqvist N. Does cardiovascular reactivity to mental stress have prognostic value in postinfarction patients? A pilot study. Psychosom Med. 1992;54(1):102–108. doi: 10.1097/00006842-199201000-00003. [DOI] [PubMed] [Google Scholar]
- 50.Jain D., Burg M., Soufer R., Zaret B.L. Prognostic implications of mental stress-induced silent left ventricular dysfunction in patients with stable angina pectoris. Am J Cardiol. 1995;76(1):31–35. doi: 10.1016/s0002-9149(99)80796-6. [DOI] [PubMed] [Google Scholar]
- 51.Jiang W., Babyak M., Krantz D.S., et al. Mental stress–induced myocardial ischemia and cardiac events. JAMA. 1996;275(21):1651–1656. doi: 10.1001/jama.275.21.1651. [DOI] [PubMed] [Google Scholar]
- 52.Krantz D.S., Santiago H.T., Kop W.J., Bairey Merz C.N., Rozanski A., Gottdiener J.S. Prognostic value of mental stress testing in coronary artery disease. Am J Cardiol. 1999;84(11):1292–1297. doi: 10.1016/s0002-9149(99)00560-3. [DOI] [PubMed] [Google Scholar]
- 53.Sheps D.S., McMahon R.P., Becker L., et al. Mental stress-induced ischemia and all-cause mortality in patients with coronary artery disease: results from the Psychophysiological Investigations of Myocardial Ischemia study. Circulation. 2002;105(15):1780–1784. doi: 10.1161/01.cir.0000014491.90666.06. [DOI] [PubMed] [Google Scholar]
- 54.Babyak M.A., Blumenthal J.A., Hinderliter A., et al. Prognosis after change in left ventricular ejection fraction during mental stress testing in patients with stable coronary artery disease. Am J Cardiol. 2010;105(1):25–28. doi: 10.1016/j.amjcard.2009.08.647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Carroll D., Ginty A.T., Der G., Hunt K., Benzeval M., Phillips A.C. Increased blood pressure reactions to acute mental stress are associated with 16-year cardiovascular disease mortality. Psychophysiology. 2012;49(10):1444–1448. doi: 10.1111/j.1469-8986.2012.01463.x. [DOI] [PubMed] [Google Scholar]
- 56.Shaffer J.A., Wasson L.T., Davidson K.W., Schwartz J.E., Kirkland S., Shimbo D. Blood pressure reactivity to an anger provocation interview does not predict incident cardiovascular disease events: the Canadian Nova Scotia Health Survey (NSHS95) prospective population study. Int J Hypertens. 2012;2012:658128. doi: 10.1155/2012/658128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Kupper N., Denollet J., Widdershoven J., Kop W.J. Cardiovascular reactivity to mental stress and mortality in patients with heart failure. J Am Coll Cardiol HF. 2015;3(5):373–382. doi: 10.1016/j.jchf.2014.12.016. [DOI] [PubMed] [Google Scholar]
- 58.Alenezi F., Brummett B.H., Boyle S.H., et al. Usefulness of myocardial annular velocity change during mental stress to predict cardiovascular outcome in patients with coronary artery disease (from the Responses of Mental Stress-Induced Myocardial Ischemia to Escitalopram Treatment trial) Am J Cardiol. 2017;120(9):1495–1500. doi: 10.1016/j.amjcard.2017.07.039. [DOI] [PubMed] [Google Scholar]
- 59.Sun J.L., Boyle S.H., Samad Z., et al. Mental stress–induced left ventricular dysfunction and adverse outcome in ischemic heart disease patients. Eur J Prev Cardiol. 2017;24(6):591–599. doi: 10.1177/2047487316686435. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Sherwood A., Hill L.K., Blumenthal J.A., et al. Blood pressure reactivity to psychological stress is associated with clinical outcomes in patients with heart failure. Am Heart J. 2017;191:82–90. doi: 10.1016/j.ahj.2017.07.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Kim J.H., Almuwaqqat Z., Hammadah M., et al. Peripheral vasoconstriction during mental stress and adverse cardiovascular outcomes in patients with coronary artery disease. Circ Res. 2019;125(10):874–883. doi: 10.1161/CIRCRESAHA.119.315005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Lima B.B., Hammadah M., Kim J.H., et al. Association of transient endothelial dysfunction induced by mental stress with major adverse cardiovascular events in men and women with coronary artery disease. JAMA Cardiol. 2019;4(10):988–996. doi: 10.1001/jamacardio.2019.3252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Moazzami K., Wittbrodt M.T., Lima B.B., et al. Higher activation of the rostromedial prefrontal cortex during mental stress predicts major cardiovascular disease events in individuals with coronary artery disease. Circulation. 2020;142(5):455–465. doi: 10.1161/CIRCULATIONAHA.119.044442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Moazzami K., Wittbrodt M.T., Alkhalaf M., et al. Association between mental stress–induced inferior frontal cortex activation and angina in coronary artery disease. Circ Cardiovasc Imaging. 2020;13(8) doi: 10.1161/CIRCIMAGING.120.010710. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Sullivan S., Young A., Hammadah M., et al. Sex differences in the inflammatory response to stress and risk of adverse cardiovascular outcomes among patients with coronary heart disease. Brain Behav Immun. 2020;90:294–302. doi: 10.1016/j.bbi.2020.09.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Widmer R.J., Prasad M., Gomaa M., et al. Vascular reactivity to mental stress is associated with poor cardiovascular disease outcomes in females following acute coronary syndrome. Coron Artery Dis. 2020;31(3):300–305. doi: 10.1097/MCA.0000000000000831. [DOI] [PubMed] [Google Scholar]
- 67.Bandodkar A.J., Wang J. Non-invasive wearable electrochemical sensors: a review. Trends Biotechnol. 2014;32(7):363–371. doi: 10.1016/j.tibtech.2014.04.005. [DOI] [PubMed] [Google Scholar]
- 68.Mitchell J.S., Lowe T.E., Ingram J.R. Rapid ultrasensitive measurement of salivary cortisol using nano-linker chemistry coupled with surface plasmon resonance detection. Analyst. 2009;134(2):380–386. doi: 10.1039/b817083p. [DOI] [PubMed] [Google Scholar]
- 69.Lampert R., Jamner L., Burg M., et al. Triggering of symptomatic atrial fibrillation by negative emotion. J Am Coll Cardiol. 2014;64(14):1533–1534. doi: 10.1016/j.jacc.2014.07.959. [DOI] [PubMed] [Google Scholar]
- 70.Rozanski A., Bairey C.N., Krantz D.S., et al. Mental stress and the induction of silent myocardial ischemia in patients with coronary artery disease. N Engl J Med. 1988;318(16):1005–1012. doi: 10.1056/NEJM198804213181601. [DOI] [PubMed] [Google Scholar]
- 71.Bonetti P.O., Lerman L.O., Lerman A. Endothelial dysfunction: a marker of atherosclerotic risk. Arterioscler Thromb Vasc Biol. 2003;23(2):168–175. doi: 10.1161/01.atv.0000051384.43104.fc. [DOI] [PubMed] [Google Scholar]
- 72.Flammer A.J., Anderson T., Celermajer D.S., et al. The assessment of endothelial function: from research into clinical practice. Circulation. 2012;126(6):753–767. doi: 10.1161/CIRCULATIONAHA.112.093245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Matsuzawa Y., Kwon T.G., Lennon R.J., Lerman L.O., Lerman A. Prognostic value of flow-mediated vasodilation in brachial artery and fingertip artery for cardiovascular events: a systematic review and meta-analysis. J Am Heart Assoc. 2015;4(11) doi: 10.1161/JAHA.115.002270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Martin E.A., Prasad A., Rihal C.S., Lerman L.O., Lerman A. Endothelial function and vascular response to mental stress are impaired in patients with apical ballooning syndrome. J Am Coll Cardiol. 2010;56(22):1840–1846. doi: 10.1016/j.jacc.2010.03.107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Hammadah M., Kim J.H., Al Mheid I., et al. Coronary and peripheral vasomotor responses to mental stress. J Am Heart Assoc. 2018;7(10) doi: 10.1161/JAHA.118.008532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Mehta P., Hermel M., Nelson M.D., et al. Mental stress peripheral vascular reactivity is elevated in women with coronary vascular dysfunction: Results from the NHLBI-sponsored Cardiac Autonomic Nervous System (CANS) study. Int J Cardiol. 2018;251:8–13. doi: 10.1016/j.ijcard.2017.10.061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Hammadah M., Al Mheid I., Wilmot K., et al. Association between high-sensitivity cardiac troponin levels and myocardial ischemia during mental stress and conventional stress. J Am Coll Cardiol Img. 2018;11(4):603–611. doi: 10.1016/j.jcmg.2016.11.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Ghiadoni L., Donald A.E., Cropley M., et al. Mental stress induces transient endothelial dysfunction in humans. Circulation. 2000;102(20):2473–2478. doi: 10.1161/01.cir.102.20.2473. [DOI] [PubMed] [Google Scholar]
- 79.Sherwood A., Johnson K., Blumenthal J.A., Hinderliter A.L. Endothelial function and hemodynamic responses during mental stress. Psychosom Med. 1999;61(3):365–370. doi: 10.1097/00006842-199905000-00017. [DOI] [PubMed] [Google Scholar]
- 80.Spieker L.E., Hurlimann D., Ruschitzka F., et al. Mental stress induces prolonged endothelial dysfunction via endothelin-A receptors. Circulation. 2002;105(24):2817–2820. doi: 10.1161/01.cir.0000021598.15895.34. [DOI] [PubMed] [Google Scholar]
- 81.Goor D.A., Sheffy J., Schnall R.P., et al. Peripheral arterial tonometry: a diagnostic method for detection of myocardial ischemia induced during mental stress tests: a pilot study. Clin Cardiol. 2004;27(3):137–141. doi: 10.1002/clc.4960270307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Peix A., Trapaga A., Asen L., et al. Mental stress-induced myocardial ischemia in women with angina and normal coronary angiograms. J Nucl Cardiol. 2006;13(4):507–513. doi: 10.1016/j.nuclcard.2006.03.016. [DOI] [PubMed] [Google Scholar]
- 83.Hassan M., Li Q., Brumback B., et al. Comparison of peripheral arterial response to mental stress in men versus women with coronary artery disease. Am J Cardiol. 2008;102(8):970–974. doi: 10.1016/j.amjcard.2008.06.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Martin E.A., Tan S.L., MacBride L.R., Lavi S., Lerman L.O., Lerman A. Sex differences in vascular and endothelial responses to acute mental stress. Clin Auton Res. 2008;18(6):339–345. doi: 10.1007/s10286-008-0497-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Burg M.M., Graeber B., Vashist A., et al. Noninvasive detection of risk for emotion-provoked myocardial ischemia. Psychosom Med. 2009;71(1):14–20. doi: 10.1097/PSY.0b013e318187c035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Hassan M., York K.M., Li H., et al. Usefulness of peripheral arterial tonometry in the detection of mental stress-induced myocardial ischemia. Clin Cardiol. 2009;32(9):E1–E6. doi: 10.1002/clc.20515. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Martin E.A., Nelson R.E., Felmlee-Devine M.D., Brown T.E., Lerman A. Comparing EndoPAT and BIOPAC measurement of vascular responses to mental stress. Cell Biochem Funct. 2011;29(4):272–278. doi: 10.1002/cbf.1745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Chen Y., Dangardt F., Osika W., Berggren K., Gronowitz E., Friberg P. Age- and sex-related differences in vascular function and vascular response to mental stress. Longitudinal and cross-sectional studies in a cohort of healthy children and adolescents. Atherosclerosis. 2012;220(1):269–274. doi: 10.1016/j.atherosclerosis.2011.10.030. [DOI] [PubMed] [Google Scholar]
- 89.Ramadan R., Sheps D., Esteves F., et al. Myocardial ischemia during mental stress: role of coronary artery disease burden and vasomotion. J Am Heart Assoc. 2013;2(5) doi: 10.1161/JAHA.113.000321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Hammadah M., Alkhoder A., Al Mheid I., et al. Hemodynamic, catecholamine, vasomotor and vascular responses: determinants of myocardial ischemia during mental stress. Int J Cardiol. 2017;243:47–53. doi: 10.1016/j.ijcard.2017.05.093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Vaccarino V., Sullivan S., Hammadah M., et al. Mental stress–induced myocardial ischemia in young patients with recent myocardial infarction: sex differences and mechanisms. Circulation. 2018;137(8):794–805. doi: 10.1161/CIRCULATIONAHA.117.030849. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Sullivan S., Hammadah M., Al Mheid I., et al. Sex differences in hemodynamic and microvascular mechanisms of myocardial ischemia induced by mental stress. Arterioscler Thromb Vasc Biol. 2018;38(2):473–480. doi: 10.1161/ATVBAHA.117.309535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Sun T., Widmer R.J., Matsuzawa Y., et al. Pain-induced peripheral artery tonometry scores in the control arm are impaired in patients with apical ballooning syndrome. Medicine (Baltimore) 2019;98(1) doi: 10.1097/MD.0000000000013841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Shah A., Chen C., Campanella C., et al. Brain correlates of stress-induced peripheral vasoconstriction in patients with cardiovascular disease. Psychophysiology. 2019;56(2) doi: 10.1111/psyp.13291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Moazzami K., Sullivan S., Lima B.B., et al. Mental stress-induced myocardial ischemia and cognitive impairment in coronary atherosclerosis. J Psychosom Res. 2020;141:110342. doi: 10.1016/j.jpsychores.2020.110342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Davignon J., Ganz P. Role of endothelial dysfunction in atherosclerosis. Circulation. 2004;109(23 suppl 1):III27–III32. doi: 10.1161/01.CIR.0000131515.03336.f8. [DOI] [PubMed] [Google Scholar]
- 97.Creager M.A., Luscher T.F., Cosentino F., Beckman J.A. Diabetes and vascular disease: pathophysiology, clinical consequences, and medical therapy: part I. Circulation. 2003;108(12):1527–1532. doi: 10.1161/01.CIR.0000091257.27563.32. [DOI] [PubMed] [Google Scholar]
- 98.Moncada S., Higgs A. The L-arginine-nitric oxide pathway. N Engl J Med. 1993;329(27):2002–2012. doi: 10.1056/NEJM199312303292706. [DOI] [PubMed] [Google Scholar]
- 99.Broten T.P., Miyashiro J.K., Moncada S., Feigl E.O. Role of endothelium-derived relaxing factor in parasympathetic coronary vasodilation. Am J Physiol. 1992;262(5 pt 2):H1579–H1584. doi: 10.1152/ajpheart.1992.262.5.H1579. [DOI] [PubMed] [Google Scholar]
- 100.Ross R. Atherosclerosis — an inflammatory disease. N Engl J Med. 1999;340(2):115–126. doi: 10.1056/NEJM199901143400207. [DOI] [PubMed] [Google Scholar]
- 101.Drexler H.G. Responsiveness of B-cll cells to growth factors. Blood. 1988;72(4):1435–1436. [PubMed] [Google Scholar]
- 102.Kinlay S., Behrendt D., Wainstein M., et al. Role of endothelin-1 in the active constriction of human atherosclerotic coronary arteries. Circulation. 2001;104(10):1114–1118. doi: 10.1161/hc3501.095707. [DOI] [PubMed] [Google Scholar]
- 103.Strawn W.B., Bondjers G., Kaplan J.R., et al. Endothelial dysfunction in response to psychosocial stress in monkeys. Circ Res. 1991;68(5):1270–1279. doi: 10.1161/01.res.68.5.1270. [DOI] [PubMed] [Google Scholar]
- 104.Williams J.K., Kaplan J.R., Manuck S.B. Effects of psychosocial stress on endothelium-mediated dilation of atherosclerotic arteries in cynomolgus monkeys. J Clin Invest. 1993;92(4):1819–1823. doi: 10.1172/JCI116772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Shimbo D., Rosenberg L.B., Chaplin W., et al. Endothelial cell activation, reduced endothelial cell reparative capacity, and impaired endothelial-dependent vasodilation after anger provocation. Int J Cardiol. 2013;167(3):1064–1065. doi: 10.1016/j.ijcard.2012.10.069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Custodis F., Gertz K., Balkaya M., et al. Heart rate contributes to the vascular effects of chronic mental stress: effects on endothelial function and ischemic brain injury in mice. Stroke. 2011;42(6):1742–1749. doi: 10.1161/STROKEAHA.110.598607. [DOI] [PubMed] [Google Scholar]
- 107.Chung I.M., Kim Y.M., Yoo M.H., Shin M.K., Kim C.K., Suh S.H. Immobilization stress induces endothelial dysfunction by oxidative stress via the activation of the angiotensin II/its type I receptor pathway. Atherosclerosis. 2010;213(1):109–114. doi: 10.1016/j.atherosclerosis.2010.08.052. [DOI] [PubMed] [Google Scholar]
- 108.Friedman E.H. Increased activation of sympathetic nervous system and endothelin by mental stress in normotensive offspring of hypertensive patients. Circulation. 1997;95(6):1667–1668. [PubMed] [Google Scholar]
- 109.Kosunen K.J. Plasma renin activity, angiotensin II, and aldosterone after mental arithmetic. Scand J Clin Lab Invest. 1977;37(5):425–429. [PubMed] [Google Scholar]
- 110.Shiraishi Y., Kujiraoka T., Hakuno D., Masaki N., Tokuno S., Adachi T. Elevation of derivatives of reactive oxygen metabolites elevated in young "disaster responders" in hypertension due to great East Japan earthquake. Int Heart J. 2016;57(1):61–66. doi: 10.1536/ihj.15-303. [DOI] [PubMed] [Google Scholar]
- 111.Sara J.D.S., Lerman L.O., Lerman A. The endothelium is a key player in the vascular response to acute mental stress. Eur Heart J. 2021;42(39):4089–4091. doi: 10.1093/eurheartj/ehab510. [DOI] [PubMed] [Google Scholar]
- 112.Laine P., Naukkarinen A., Heikkila L., Penttila A., Kovanen P.T. Adventitial mast cells connect with sensory nerve fibers in atherosclerotic coronary arteries. Circulation. 2000;101(14):1665–1669. doi: 10.1161/01.cir.101.14.1665. [DOI] [PubMed] [Google Scholar]
- 113.Soufer R., Jain H., Yoon A.J. Heart-brain interactions in mental stress-induced myocardial ischemia. Curr Cardiol Rep. 2009;11(2):133–140. doi: 10.1007/s11886-009-0020-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Freyschuss U., Hjemdahl P., Juhlin-Dannfelt A., Linde B. Cardiovascular and sympathoadrenal responses to mental stress: influence of beta-blockade. Am J Physiol. 1988;255(6 pt 2):H1443–H1451. doi: 10.1152/ajpheart.1988.255.6.H1443. [DOI] [PubMed] [Google Scholar]
- 115.Greaney J.L., Surachman A., Saunders E.F.H., Alexander L.M., Almeida D.M. Greater daily psychosocial stress exposure is associated with increased norepinephrine-induced vasoconstriction in young adults. J Am Heart Assoc. 2020;9(9) doi: 10.1161/JAHA.119.015697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Jamieson P.M., Li C., Kukura C., Vaughan J., Vale W. Urocortin 3 modulates the neuroendocrine stress response and is regulated in rat amygdala and hypothalamus by stress and glucocorticoids. Endocrinology. 2006;147(10):4578–4588. doi: 10.1210/en.2006-0545. [DOI] [PubMed] [Google Scholar]
- 117.Hashimoto K., Nishiyama M., Tanaka Y., et al. Urocortins and corticotropin releasing factor type 2 receptors in the hypothalamus and the cardiovascular system. Peptides. 2004;25(10):1711–1721. doi: 10.1016/j.peptides.2004.05.024. [DOI] [PubMed] [Google Scholar]
- 118.Kalsner S., Richards R. Coronary arteries of cardiac patients are hyperreactive and contain stores of amines: a mechanism for coronary spasm. Science. 1984;223(4643):1435–1437. doi: 10.1126/science.6701530. [DOI] [PubMed] [Google Scholar]
- 119.Allen S., Dashwood M., Morrison K., Yacoub M. Differential leukotriene constrictor responses in human atherosclerotic coronary arteries. Circulation. 1998;97(24):2406–2413. doi: 10.1161/01.cir.97.24.2406. [DOI] [PubMed] [Google Scholar]
- 120.Lytinas M., Kempuraj D., Huang M., Boucher W., Esposito P., Theoharides T.C. Acute stress results in skin corticotropin-releasing hormone secretion, mast cell activation and vascular permeability, an effect mimicked by intradermal corticotropin-releasing hormone and inhibited by histamine-1 receptor antagonists. Int Arch Allergy Immunol. 2003;130(3):224–231. doi: 10.1159/000069516. [DOI] [PubMed] [Google Scholar]
- 121.Lagraauw H.M., Wezel A., van der Velden D., Kuiper J., Bot I. Stress-induced mast cell activation contributes to atherosclerotic plaque destabilization. Sci Rep. 2019;9(1):2134. doi: 10.1038/s41598-019-38679-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Bot I., de Jager S.C., Bot M., et al. The neuropeptide substance P mediates adventitial mast cell activation and induces intraplaque hemorrhage in advanced atherosclerosis. Circ Res. 2010;106(1):89–92. doi: 10.1161/CIRCRESAHA.109.204875. [DOI] [PubMed] [Google Scholar]
- 123.Laine P., Kaartinen M., Penttila A., Panula P., Paavonen T., Kovanen P.T. Association between myocardial infarction and the mast cells in the adventitia of the infarct-related coronary artery. Circulation. 1999;99(3):361–369. doi: 10.1161/01.cir.99.3.361. [DOI] [PubMed] [Google Scholar]
- 124.Steptoe A., Willemsen G., Owen N., Flower L., Mohamed-Ali V. Acute mental stress elicits delayed increases in circulating inflammatory cytokine levels. Clin Sci (Lond) 2001;101(2):185–192. [PubMed] [Google Scholar]
- 125.Black P.H., Garbutt L.D. Stress, inflammation and cardiovascular disease. J Psychosom Res. 2002;52(1):1–23. doi: 10.1016/s0022-3999(01)00302-6. [DOI] [PubMed] [Google Scholar]
- 126.Altavilla D., Guarini S., Bitto A., et al. Activation of the cholinergic anti-inflammatory pathway reduces NF-kappab activation, blunts TNF-alpha production, and protects againts splanchic artery occlusion shock. Shock. 2006;25(5):500–506. doi: 10.1097/01.shk.0000209539.91553.82. [DOI] [PubMed] [Google Scholar]
- 127.Pavlov V.A., Tracey K.J. The vagus nerve and the inflammatory reflex — linking immunity and metabolism. Nat Rev Endocrinol. 2012;8(12):743–754. doi: 10.1038/nrendo.2012.189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Tracey K.J. Physiology and immunology of the cholinergic antiinflammatory pathway. J Clin Invest. 2007;117(2):289–296. doi: 10.1172/JCI30555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Camell C.D., Sander J., Spadaro O., et al. Inflammasome-driven catecholamine catabolism in macrophages blunts lipolysis during ageing. Nature. 2017;550(7674):119–123. doi: 10.1038/nature24022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Heinz A., Hermann D., Smolka M.N., et al. Effects of acute psychological stress on adhesion molecules, interleukins and sex hormones: implications for coronary heart disease. Psychopharmacology (Berl) 2003;165(2):111–117. doi: 10.1007/s00213-002-1244-6. [DOI] [PubMed] [Google Scholar]
- 131.Dutta P., Courties G., Wei Y., et al. Myocardial infarction accelerates atherosclerosis. Nature. 2012;487(7407):325–329. doi: 10.1038/nature11260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Hodes G.E., Pfau M.L., Leboeuf M., et al. Individual differences in the peripheral immune system promote resilience versus susceptibility to social stress. Proc Natl Acad Sci U S A. 2014;111(45):16136–16141. doi: 10.1073/pnas.1415191111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Heidt T., Sager H.B., Courties G., et al. Chronic variable stress activates hematopoietic stem cells. Nat Med. 2014;20(7):754–758. doi: 10.1038/nm.3589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Bernberg E., Ulleryd M.A., Johansson M.E., Bergstrom G.M. Social disruption stress increases IL-6 levels and accelerates atherosclerosis in ApoE-/- mice. Atherosclerosis. 2012;221(2):359–365. doi: 10.1016/j.atherosclerosis.2011.11.041. [DOI] [PubMed] [Google Scholar]
- 135.Dantzer R., O'Connor J.C., Freund G.G., Johnson R.W., Kelley K.W. From inflammation to sickness and depression: when the immune system subjugates the brain. Nat Rev Neurosci. 2008;9(1):46–56. doi: 10.1038/nrn2297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Nahrendorf M., Swirski F.K. Lifestyle effects on hematopoiesis and atherosclerosis. Circ Res. 2015;116(5):884–894. doi: 10.1161/CIRCRESAHA.116.303550. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Qing H., Desrouleaux R., Israni-Winger K., et al. Origin and function of stress-induced IL-6 in murine models. Cell. 2020;182(6):1660. doi: 10.1016/j.cell.2020.08.044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Roth S., Singh V., Tiedt S., et al. Brain-released alarmins and stress response synergize in accelerating atherosclerosis progression after stroke. Sci Transl Med. 2018;10(432) doi: 10.1126/scitranslmed.aao1313. [DOI] [PubMed] [Google Scholar]
- 139.Demirtas T., Utkan T., Karson A., Yazir Y., Bayramgurler D., Gacar N. The link between unpredictable chronic mild stress model for depression and vascular inflammation? Inflammation. 2014;37(5):1432–1438. doi: 10.1007/s10753-014-9867-4. [DOI] [PubMed] [Google Scholar]
- 140.Rozanski A., Blumenthal J.A., Kaplan J. Impact of psychological factors on the pathogenesis of cardiovascular disease and implications for therapy. Circulation. 1999;99(16):2192–2217. doi: 10.1161/01.cir.99.16.2192. [DOI] [PubMed] [Google Scholar]
- 141.Lerman A., Zeiher A.M. Endothelial function: cardiac events. Circulation. 2005;111(3):363–368. doi: 10.1161/01.CIR.0000153339.27064.14. [DOI] [PubMed] [Google Scholar]
- 142.Reriani M.K., Flammer A.J., Jama A., Lerman L.O., Lerman A. Novel functional risk factors for the prediction of cardiovascular events in vulnerable patients following acute coronary syndrome. Circ J. 2012;76(4):778–783. doi: 10.1253/circj.cj-12-0248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Suwaidi J.A., Hamasaki S., Higano S.T., Nishimura R.A., Holmes D.R., Jr., Lerman A. Long-term follow-up of patients with mild coronary artery disease and endothelial dysfunction. Circulation. 2000;101(9):948–954. doi: 10.1161/01.cir.101.9.948. [DOI] [PubMed] [Google Scholar]
- 144.Targonski P.V., Bonetti P.O., Pumper G.M., Higano S.T., Holmes D.R., Jr., Lerman A. Coronary endothelial dysfunction is associated with an increased risk of cerebrovascular events. Circulation. 2003;107(22):2805–2809. doi: 10.1161/01.CIR.0000072765.93106.EE. [DOI] [PubMed] [Google Scholar]
- 145.Yoon M.H., Reriani M., Mario G., et al. Long-term endothelin receptor antagonism attenuates coronary plaque progression in patients with early atherosclerosis. Int J Cardiol. 2013;168(2):1316–1321. doi: 10.1016/j.ijcard.2012.12.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Lavi S., Bae J.H., Rihal C.S., et al. Segmental coronary endothelial dysfunction in patients with minimal atherosclerosis is associated with necrotic core plaques. Heart. 2009;95(18):1525–1530. doi: 10.1136/hrt.2009.166017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Lynch J., Kaplan G.A., Salonen R., Cohen R.D., Salonen J.T. Socioeconomic status and carotid atherosclerosis. Circulation. 1995;92(7):1786–1792. doi: 10.1161/01.cir.92.7.1786. [DOI] [PubMed] [Google Scholar]
- 148.Paterniti S., Zureik M., Ducimetiere P., Touboul P.J., Feve J.M., Alperovitch A. Sustained anxiety and 4-year progression of carotid atherosclerosis. Arterioscler Thromb Vasc Biol. 2001;21(1):136–141. doi: 10.1161/01.atv.21.1.136. [DOI] [PubMed] [Google Scholar]
- 149.Haas D.C., Davidson K.W., Schwartz D.J., et al. Depressive symptoms are independently predictive of carotid atherosclerosis. Am J Cardiol. 2005;95(4):547–550. doi: 10.1016/j.amjcard.2004.10.032. [DOI] [PubMed] [Google Scholar]
- 150.Meng L.B., Qi R., Xu L., et al. The more critical murderer of atherosclerosis than lipid metabolism: chronic stress. Lipids Health Dis. 2018;17(1):143. doi: 10.1186/s12944-018-0795-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Wang S., Xiaoling G., Pingting L., Shuqiang L., Yuaner Z. Chronic unpredictable mild stress combined with a high-fat diets aggravates atherosclerosis in rats. Lipids Health Dis. 2014;13:77. doi: 10.1186/1476-511X-13-77. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Peters A., McEwen B.S. Stress habituation, body shape and cardiovascular mortality. Neurosci Biobehav Rev. 2015;56:139–150. doi: 10.1016/j.neubiorev.2015.07.001. [DOI] [PubMed] [Google Scholar]
- 153.Astrand P.O., Cuddy T.E., Saltin B., Stenberg J. Cardiac output during submaximal and maximal work. J Appl Physiol. 1964;19:268–274. doi: 10.1152/jappl.1964.19.2.268. [DOI] [PubMed] [Google Scholar]
- 154.Sherwood A., Allen M.T., Obrist P.A., Langer A.W. Evaluation of beta-adrenergic influences on cardiovascular and metabolic adjustments to physical and psychological stress. Psychophysiology. 1986;23(1):89–104. doi: 10.1111/j.1469-8986.1986.tb00602.x. [DOI] [PubMed] [Google Scholar]
- 155.Et-Taouil K., Schiavi P., Levy B.I., Plante G.E. Sodium intake, large artery stiffness, and proteoglycans in the spontaneously hypertensive rat. Hypertension. 2001;38(5):1172–1176. doi: 10.1161/hy1101.96740. [DOI] [PubMed] [Google Scholar]
- 156.Oparil S., Zaman M.A., Calhoun D.A. Pathogenesis of hypertension. Ann Intern Med. 2003;139(9):761–776. doi: 10.7326/0003-4819-139-9-200311040-00011. [DOI] [PubMed] [Google Scholar]
- 157.Miura K., Dyer A.R., Greenland P., et al. Pulse pressure compared with other blood pressure indexes in the prediction of 25-year cardiovascular and all-cause mortality rates: the Chicago Heart Association Detection Project in Industry study. Hypertension. 2001;38(2):232–237. doi: 10.1161/01.hyp.38.2.232. [DOI] [PubMed] [Google Scholar]
- 158.Davies P.F. Flow-mediated endothelial mechanotransduction. Physiol Rev. 1995;75(3):519–560. doi: 10.1152/physrev.1995.75.3.519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Kamarck T.W., Everson S.A., Kaplan G.A., et al. Exaggerated blood pressure responses during mental stress are associated with enhanced carotid atherosclerosis in middle-aged Finnish men: findings from the Kuopio Ischemic Heart Disease Study. Circulation. 1997;96(11):3842–3848. doi: 10.1161/01.cir.96.11.3842. [DOI] [PubMed] [Google Scholar]
- 160.Jennings J.R., Kamarck T.W., Everson-Rose S.A., Kaplan G.A., Manuck S.B., Salonen J.T. Exaggerated blood pressure responses during mental stress are prospectively related to enhanced carotid atherosclerosis in middle-aged Finnish men. Circulation. 2004;110(15):2198–2203. doi: 10.1161/01.CIR.0000143840.77061.E9. [DOI] [PubMed] [Google Scholar]
- 161.McAdoo W.G., Weinberger M.H., Miller J.Z., Fineberg N.S., Grim C.E. Race and gender influence hemodynamic responses to psychological and physical stimuli. J Hypertens. 1990;8(10):961–967. doi: 10.1097/00004872-199010000-00012. [DOI] [PubMed] [Google Scholar]
- 162.Baldwin R., Jeffries S., Jackson A., et al. Neurological findings in late-onset depressive disorder: comparison of individuals with and without depression. Br J Psychiatry. 2005;186:308–313. doi: 10.1192/bjp.186.4.308. [DOI] [PubMed] [Google Scholar]
- 163.Steptoe A., Donald A.E., O'Donnell K., Marmot M., Deanfield J.E. Delayed blood pressure recovery after psychological stress is associated with carotid intima-media thickness: Whitehall psychobiology study. Arterioscler Thromb Vasc Biol. 2006;26(11):2547–2551. doi: 10.1161/01.ATV.0000242792.93486.0d. [DOI] [PubMed] [Google Scholar]
- 164.Soufer R., Burg M.M. The heart-brain interaction during emotionally provoked myocardial ischemia: implications of cortical hyperactivation in CAD and gender interactions. Cleve Clin J Med. 2007;74(suppl 1):S59–S62. doi: 10.3949/ccjm.74.suppl_1.s59. [DOI] [PubMed] [Google Scholar]
- 165.Yeung A.C., Vekshtein V.I., Krantz D.S., et al. The effect of atherosclerosis on the vasomotor response of coronary arteries to mental stress. N Engl J Med. 1991;325(22):1551–1556. doi: 10.1056/NEJM199111283252205. [DOI] [PubMed] [Google Scholar]
- 166.Arrighi J.A., Burg M., Cohen I.S., et al. Myocardial blood-flow response during mental stress in patients with coronary artery disease. Lancet. 2000;356(9226):310–311. doi: 10.1016/S0140-6736(00)02510-1. [DOI] [PubMed] [Google Scholar]
- 167.Dakak N., Quyyumi A.A., Eisenhofer G., Goldstein D.S., Cannon R.O., 3rd Sympathetically mediated effects of mental stress on the cardiac microcirculation of patients with coronary artery disease. Am J Cardiol. 1995;76(3):125–130. doi: 10.1016/s0002-9149(99)80043-5. [DOI] [PubMed] [Google Scholar]
- 168.Panting J.R., Gatehouse P.D., Yang G.Z., et al. Abnormal subendocardial perfusion in cardiac syndrome X detected by cardiovascular magnetic resonance imaging. N Engl J Med. 2002;346(25):1948–1953. doi: 10.1056/NEJMoa012369. [DOI] [PubMed] [Google Scholar]
- 169.Maseri A., Crea F., Kaski J.C., Crake T. Mechanisms of angina pectoris in syndrome X. J Am Coll Cardiol. 1991;17(2):499–506. doi: 10.1016/s0735-1097(10)80122-6. [DOI] [PubMed] [Google Scholar]
- 170.Wang S.S., Yan X.B., Hofman M.A., Swaab D.F., Zhou J.N. Increased expression level of corticotropin-releasing hormone in the amygdala and in the hypothalamus in rats exposed to chronic unpredictable mild stress. Neurosci Bull. 2010;26(4):297–303. doi: 10.1007/s12264-010-0329-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Koutsikou S., Watson T.C., Crook J.J., et al. The periaqueductal gray orchestrates sensory and motor circuits at multiple levels of the neuraxis. J Neurosci. 2015;35(42):14132–14147. doi: 10.1523/JNEUROSCI.0261-15.2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.McEwen B.S., Morrison J.H. The brain on stress: vulnerability and plasticity of the prefrontal cortex over the life course. Neuron. 2013;79(1):16–29. doi: 10.1016/j.neuron.2013.06.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.Silvestrini N. Psychological and neural mechanisms associated with effort-related cardiovascular reactivity and cognitive control: an integrative approach. Int J Psychophysiol. 2017;119:11–18. doi: 10.1016/j.ijpsycho.2016.12.009. [DOI] [PubMed] [Google Scholar]
- 174.Gianaros P.J., Marsland A.L., Kuan D.C., et al. An inflammatory pathway links atherosclerotic cardiovascular disease risk to neural activity evoked by the cognitive regulation of emotion. Biol Psychiatry. 2014;75(9):738–745. doi: 10.1016/j.biopsych.2013.10.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Eisenberger N.I., Taylor S.E., Gable S.L., Hilmert C.J., Lieberman M.D. Neural pathways link social support to attenuated neuroendocrine stress responses. Neuroimage. 2007;35(4):1601–1612. doi: 10.1016/j.neuroimage.2007.01.038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Oler J.A., Fox A.S., Shelton S.E., et al. Amygdalar and hippocampal substrates of anxious temperament differ in their heritability. Nature. 2010;466(7308):864–868. doi: 10.1038/nature09282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Fox A.S., Oler J.A., Shelton S.E., et al. Central amygdala nucleus (Ce) gene expression linked to increased trait-like Ce metabolism and anxious temperament in young primates. Proc Natl Acad Sci U S A. 2012;109(44):18108–18113. doi: 10.1073/pnas.1206723109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Shin L.M., Liberzon I. The neurocircuitry of fear, stress, and anxiety disorders. Neuropsychopharmacology. 2010;35(1):169–191. doi: 10.1038/npp.2009.83. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Shin L.M., Wright C.I., Cannistraro P.A., et al. A functional magnetic resonance imaging study of amygdala and medial prefrontal cortex responses to overtly presented fearful faces in posttraumatic stress disorder. Arch Gen Psychiatry. 2005;62(3):273–281. doi: 10.1001/archpsyc.62.3.273. [DOI] [PubMed] [Google Scholar]
- 180.Girod J.P., Brotman D.J. Does altered glucocorticoid homeostasis increase cardiovascular risk? Cardiovasc Res. 2004;64(2):217–226. doi: 10.1016/j.cardiores.2004.07.006. [DOI] [PubMed] [Google Scholar]
- 181.Gianaros P.J., Hariri A.R., Sheu L.K., Muldoon M.F., Sutton-Tyrrell K., Manuck S.B. Preclinical atherosclerosis covaries with individual differences in reactivity and functional connectivity of the amygdala. Biol Psychiatry. 2009;65(11):943–950. doi: 10.1016/j.biopsych.2008.10.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182.Tawakol A., Ishai A., Takx R.A., et al. Relation between resting amygdalar activity and cardiovascular events: a longitudinal and cohort study. Lancet. 2017;389(10071):834–845. doi: 10.1016/S0140-6736(16)31714-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Goyal A., Dey A.K., Chaturvedi A., et al. Chronic stress-related neural activity associates with subclinical cardiovascular disease in psoriasis: a prospective cohort study. J Am Coll Cardiol Img. 2020;13(2 pt 1):465–477. doi: 10.1016/j.jcmg.2018.08.038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.Osborne M.T., Ishai A., Hammad B., et al. Amygdalar activity predicts future incident diabetes independently of adiposity. Psychoneuroendocrinology. 2019;100:32–40. doi: 10.1016/j.psyneuen.2018.09.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 185.Fiechter M., Haider A., Bengs S., et al. Sex-dependent association between inflammation, neural stress responses, and impaired myocardial function. Eur J Nucl Med Mol Imaging. 2020;47(8):2010–2015. doi: 10.1007/s00259-019-04537-8. [DOI] [PubMed] [Google Scholar]
- 186.Dar T., Osborne M.T., Abohashem S., et al. Greater neurobiological resilience to chronic socioeconomic or environmental stressors associates with lower risk for cardiovascular disease events. Circ Cardiovasc Imaging. 2020;13(8) doi: 10.1161/CIRCIMAGING.119.010337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Schwartz A.R., Gerin W., Davidson K.W., et al. Toward a causal model of cardiovascular responses to stress and the development of cardiovascular disease. Psychosom Med. 2003;65(1):22–35. doi: 10.1097/01.psy.0000046075.79922.61. [DOI] [PubMed] [Google Scholar]
- 188.Christenfeld N., Glynn L.M., Kulik J.A., Gerin W. The social construction of cardiovascular reactivity. Ann Behav Med. 1998;20(4):317–325. doi: 10.1007/BF02886381. [DOI] [PubMed] [Google Scholar]
- 189.Verdecchia P., Porcellati C., Schillaci G., et al. Ambulatory blood pressure. An independent predictor of prognosis in essential hypertension. Hypertension. 1994;24(6):793–801. doi: 10.1161/01.hyp.24.6.793. [DOI] [PubMed] [Google Scholar]
- 190.Ewart C.K., Kolodner K.B. Predicting ambulatory blood pressure during school: effectiveness of social and nonsocial reactivity tasks in black and white adolescents. Psychophysiology. 1993;30(1):30–38. doi: 10.1111/j.1469-8986.1993.tb03202.x. [DOI] [PubMed] [Google Scholar]
- 191.Linden W., Con A. Laboratory reactivity models as predictors of ambulatory blood pressure and heart rate. J Psychosom Res. 1994;38(3):217–228. doi: 10.1016/0022-3999(94)90117-1. [DOI] [PubMed] [Google Scholar]
- 192.Light K.C., Turner J.R., Hinderliter A.L., Sherwood A. Race and gender comparisons: II. Predictions of work blood pressure from laboratory baseline and cardiovascular reactivity measures. Health Psychol. 1993;12(5):366–375. doi: 10.1037//0278-6133.12.5.366. [DOI] [PubMed] [Google Scholar]
- 193.Lampert R., Jain D., Burg M.M., Batsford W.P., McPherson C.A. Destabilizing effects of mental stress on ventricular arrhythmias in patients with implantable cardioverter-defibrillators. Circulation. 2000;101(2):158–164. doi: 10.1161/01.cir.101.2.158. [DOI] [PubMed] [Google Scholar]
- 194.Ironson G., Taylor C.B., Boltwood M., et al. Effects of anger on left ventricular ejection fraction in coronary artery disease. Am J Cardiol. 1992;70(3):281–285. doi: 10.1016/0002-9149(92)90605-x. [DOI] [PubMed] [Google Scholar]
- 195.Christenfeld N., Glynn L.M., Gerin W. On the reliable assessment of cardiovascular recovery: an application of curve-fitting techniques. Psychophysiology. 2000;37(4):543–550. [PubMed] [Google Scholar]
- 196.Stewart J.C., France C.R. Cardiovascular recovery from stress predicts longitudinal changes in blood pressure. Biol Psychol. 2001;58(2):105–120. doi: 10.1016/s0301-0511(01)00105-3. [DOI] [PubMed] [Google Scholar]
- 197.Treiber F.A., Musante L., Kapuku G., Davis C., Litaker M., Davis H. Cardiovascular (CV) responsivity and recovery to acute stress and future CV functioning in youth with family histories of CV disease: a 4-year longitudinal study. Int J Psychophysiol. 2001;41(1):65–74. doi: 10.1016/s0167-8760(00)00183-5. [DOI] [PubMed] [Google Scholar]
- 198.Poulter N.R., Khaw K.T., Hopwood B.E., et al. The Kenyan Luo migration study: observations on the initiation of a rise in blood pressure. BMJ. 1990;300(6730):967–972. doi: 10.1136/bmj.300.6730.967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199.Nadim A., Amini H., Malek-Afzali H. Blood pressure and rural-urban migration in Iran. Int J Epidemiol. 1978;7(2):131–138. doi: 10.1093/ije/7.2.131. [DOI] [PubMed] [Google Scholar]
- 200.Salmond C.E., Prior I.A., Wessen A.F. Blood pressure patterns and migration: a 14-year cohort study of adult Tokelauans. Am J Epidemiol. 1989;130(1):37–52. doi: 10.1093/oxfordjournals.aje.a115321. [DOI] [PubMed] [Google Scholar]
- 201.Williams R.B., Marchuk D.A., Gadde K.M., et al. Central nervous system serotonin function and cardiovascular responses to stress. Psychosom Med. 2001;63(2):300–305. doi: 10.1097/00006842-200103000-00016. [DOI] [PubMed] [Google Scholar]
- 202.Tawakol A., Osborne M.T., Wang Y., et al. Stress-associated neurobiological pathway linking socioeconomic disparities to cardiovascular disease. J Am Coll Cardiol. 2019;73(25):3243–3255. doi: 10.1016/j.jacc.2019.04.042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.Osborne M.T., Radfar A., Hassan M.Z.O., et al. A neurobiological mechanism linking transportation noise to cardiovascular disease in humans. Eur Heart J. 2020;41(6):772–782. doi: 10.1093/eurheartj/ehz820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 204.Piepoli M.F., Hoes A.W., Agewall S., et al. 2016 European guidelines on cardiovascular disease prevention in clinical practice. Rev Esp Cardiol (Engl Ed) 2016;69(10):939. doi: 10.1016/j.rec.2016.09.009. [DOI] [PubMed] [Google Scholar]
- 205.Levine G.N., Lange R.A., Bairey-Merz C.N., et al. Meditation and cardiovascular risk reduction: a scientific statement from the American Heart Association. J Am Heart Assoc. 2017;6(10) doi: 10.1161/JAHA.117.002218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 206.Cuijpers P., Berking M., Andersson G., Quigley L., Kleiboer A., Dobson K.S. A meta-analysis of cognitive-behavioural therapy for adult depression, alone and in comparison with other treatments. Can J Psychiatry. 2013;58(7):376–385. doi: 10.1177/070674371305800702. [DOI] [PubMed] [Google Scholar]
- 207.Blumenthal J.A., Sherwood A., Smith P.J., et al. Enhancing cardiac rehabilitation with stress management training: a randomized, clinical efficacy trial. Circulation. 2016;133(14):1341–1350. doi: 10.1161/CIRCULATIONAHA.115.018926. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 208.Anderson L., Oldridge N., Thompson D.R., et al. Exercise-based cardiac rehabilitation for coronary heart disease: Cochrane systematic review and meta-analysis. J Am Coll Cardiol. 2016;67(1):1–12. doi: 10.1016/j.jacc.2015.10.044. [DOI] [PubMed] [Google Scholar]
- 209.Dessy E., Mairesse O., van Puyvelde M., Cortoos A., Neyt X., Pattyn N. Train your brain? Can we really selectively train specific EEG frequencies with neurofeedback training. Front Hum Neurosci. 2020;14:22. doi: 10.3389/fnhum.2020.00022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 210.Jiang W., Velazquez E.J., Kuchibhatla M., et al. Effect of escitalopram on mental stress-induced myocardial ischemia: results of the REMIT trial. JAMA. 2013;309(20):2139–2149. doi: 10.1001/jama.2013.5566. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 211.Lenze E.J., Mattar C., Zorumski C.F., et al. Fluvoxamine vs placebo and clinical deterioration in outpatients with symptomatic COVID-19: a randomized clinical trial. JAMA. 2020;324(22):2292–2300. doi: 10.1001/jama.2020.22760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 212.Reis G., Dos Santos Moreira-Silva E.A., Silva D.C.M., et al. Effect of early treatment with fluvoxamine on risk of emergency care and hospitalisation among patients with COVID-19: the TOGETHER randomised, platform clinical trial. Lancet Glob Health. 2022;10(1):e42–e51. doi: 10.1016/S2214-109X(21)00448-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 213.Hayashi T., Su T.P. Sigma-1 receptor ligands: potential in the treatment of neuropsychiatric disorders. CNS Drugs. 2004;18(5):269–284. doi: 10.2165/00023210-200418050-00001. [DOI] [PubMed] [Google Scholar]
- 214.Jiang W. Emotional triggering of cardiac dysfunction: the present and future. Curr Cardiol Rep. 2015;17(10):91. doi: 10.1007/s11886-015-0635-3. [DOI] [PubMed] [Google Scholar]
- 215.Ridker P.M., Everett B.M., Thuren T., et al. Antiinflammatory therapy with canakinumab for atherosclerotic disease. N Engl J Med. 2017;377(12):1119–1131. doi: 10.1056/NEJMoa1707914. [DOI] [PubMed] [Google Scholar]
- 216.Dong C., Rundek T., Wright C.B., Anwar Z., Elkind M.S., Sacco R.L. Ideal cardiovascular health predicts lower risks of myocardial infarction, stroke, and vascular death across whites, blacks, and hispanics: the northern Manhattan study. Circulation. 2012;125(24):2975–2984. doi: 10.1161/CIRCULATIONAHA.111.081083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 217.Good C.B., Hernandez I., Parekh N. Statins for primary prevention of cardiovascular disease. Ann Intern Med. 2019;171(1):72. doi: 10.7326/L19-0257. [DOI] [PubMed] [Google Scholar]
- 218.Goff D.C., Jr., Lloyd-Jones D.M., Bennett G., et al. 2013 ACC/AHA guideline on the assessment of cardiovascular risk: a report of the American College of Cardiology/American Heart Association task force on practice guidelines. J Am Coll Cardiol. 2014;63(25 pt B):2935–2959. doi: 10.1016/j.jacc.2013.11.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 219.Stockelman K.A., Bain A.R., Dow C.A., et al. Regular aerobic exercise counteracts endothelial vasomotor dysfunction associated with insufficient sleep. Am J Physiol Heart Circ Physiol. 2021;320(3):H1080–H1088. doi: 10.1152/ajpheart.00615.2020. [DOI] [PMC free article] [PubMed] [Google Scholar]