Abstract
Exosomes can be derived from mesenchymal stem cells (MSCs) and have recently been used to treat defects in the tendons. They may also have applications in treating osteoporosis. MSC-derived exosomes communicate with mesenchymal and osteogenic cells through endocrine or paracrine mechanisms and contribute factors involved in physiological and pathological orthopedic conditions associated with hypoxia and bone tumors. Also, generalized medical conditions, such as obesity, hyperglycemia, and degenerative diseases, can inhibit the osteogenic effect of MSC-derived exosomes. This review aims to present an update on the roles of MSCs and exosomes derived from MSCs in treating orthopedic diseases, such as osteoporosis, and in the repair of cartilage, tendons, and bone fractures.
Keywords: Bone Diseases, Exosomes, Mesenchymal Stem Cells
Background
Exosomes are small vesicles secreted by cells, which mediate cell-to-cell communication through paracrine and other means [1]. Exosomes derived from mesenchymal stem cells (MSCs) have become popular in the field of bone regeneration because they can selectively carry a large amount of bone-promoting cargo. In recent years, the direction of research has gradually shifted to the packaging modification of exosomes to achieve the artificial maximization of their function. However, exosomes are still affected by parent cells [2], and the carrying of these substances results in the exosomes from different sources having inherent and difficult-to-change parental characteristics [3]. In addition, the effect of pedigree-specific exosomes on the differentiation of MSCs was greater than that of the extracellular matrix, and osteoblast-derived exosomes containing osteogenic factors could reverse the adipogenic differentiation potential of MSCs cultured in adipogenic medium [4]. This makes the source of MSC-derived exosomes (MSC-Exos) an important aspect of the functional features of exosomes. Classified by source, MSCs can be obtained from prenatal tissue sources (umbilical cord and amniotic membrane), fetal sources (placenta, umbilical cord, and umbilical cord blood), and adult tissues (bone, muscle, fat, and peripheral blood) [5]. The above sources are exosomes are extracted from normal tissue and reflect some physiological functions and characteristics of the parent cells, making them an important tissue source in stem cell osteogenesis research [6]. However, the role of pathological tissue-derived exosomes in the field of osteogenesis has been rarely analyzed, and even tumor tissue-derived exosomes seem to play an auxiliary role in the scientific research of bone regeneration [7]. Moreover, it is undeniable that the production, formation, and secretion of MSC-Exos are affected by the external environment [8]. Analyzing how exosomes work in different pathological environments and what positive or negative effects they have on bone formation will provide a theoretical basis and practical reference for our use of exosomes. Therefore, this review aimed to present an update on the role of MSCs and exosomes derived from MSCs in treating orthopedic disease, such as osteoporosis, and in the repair of cartilage, tendons, and bone fractures.
Effect of Internal Environment on Exosomes
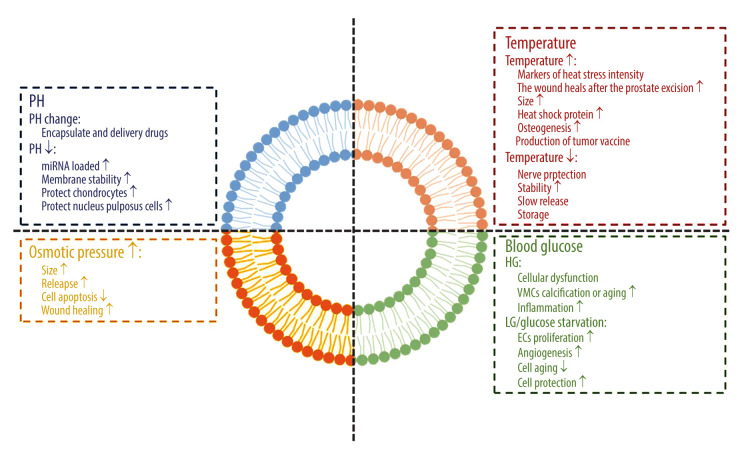
We have been looking forward to using exosomes from different tissue sources for disease detection, prevention, and treatment. Exosomes have indeed become the inevitable direction in the current cell-free trend. However, because stem cells have differentiation potential, the MSC environment plays a significant role in inducing the differentiation potential of stem cells. Before elucidating the characteristics and functions of exosomes in the pathological environment, we should first clarify the response of exosomes to environmental changes in the body (a schematic diagram of the impact of the internal environment on exosomes is shown in Figure 1).
Figure 1.
The effect of the internal environment on exosomes. Figure created with BioRender.com.
Potential of Hydrogen
An acidic environment is the starting or aggravating factor of many diseases. The fermentation and metabolism of glucose leads to the accumulation of lactic acid in the cytoplasm, which makes the tumor environment typically acidic. The acidic microenvironment selects the cell clones that are most suitable for survival in this harsh environment and for the transport of exosomes by tumor cells [9,10]. However, the stability of exosomes in an acidic environment becomes their advantage against the adverse environment.
In the separation of the urine-derived exosome, it was found that the acidic environment may be conducive to the separation of exosomes and maintain their stability and integrity. When the potential of hydrogen (pH) is 8, many exosomes are lost [11]. A reduction of pH can enhance the load of functional miRNA in exosomes [12]. When pH decreased to 2.5, the number of functional miRNA, siRNA, and ssRNA loaded by exosomes increased significantly, which was even similar to that of ultrasound and electroporation, without damaging unstable nucleic acid contents [13]. In degenerative diseases, intervertebral disc degeneration is caused by abnormal pH values in the intervertebral disc. Although stem cell transplantation is an effective method, MSC gene expression, proliferation, and activity are severely restricted when the pH is reduced to 5.7 [14]. In a pathologically acidic environment, MSC-Exos promote the expression of the extracellular matrix, collagen II, and proteoglycan in chondrocytes, reduce matrix degradation by downregulating matrix-degrading enzymes, and protect nucleus pulposus cells from apoptosis induced by acidic pH [15].
In addition, the method of taking advantage of the effect of changes in pH on exosomes has been used in the clinical transformation of exosome drug delivery. Holographic transferrin (containing a trivalent iron ion) can bind to transferrin receptors in a pH of 7.4. When the pH changes to 5.0, total transferrin releases trivalent iron ions to form apolipoprotein transferrin (without trivalent iron ions). However, apolipoprotein transferrin still binds to transferrin receptors until the pH value changes to 7.4 again. Because the pH value of the medium around the exosome changed from 5.0 (in polyvesicles) to 7.4 in extracellular space during its formation, it is a new idea to apply the pH response binding and dissociation characteristics of transferrin and transferrin receptors to the release of exosomes from superparamagnetic nanoparticles. Based on this method of superparamagnetic nanoparticle clusters, the accurate and mild separation of blood transferrin receptor–positive exosomes was realized for the first time, which promoted the clinical transformation of exosome-based drug delivery [16].
Temperature
Heat stress is a nonspecific physical response caused by a pathological increase in core body temperature, while the temperature set point does not rise. The contents of exosomes in different organs were detected at 40°C, 42°C, and 43°C. It was considered that the exosome was a marker of heat stress intensity [17]. However, epidermal growth factor receptor exosomes derived from thermal injury stromal cells can enhance prostate wound healing after thulium laser resection through epithelial-mesenchymal transition (EMT) and NF-κB signals [18]. Under heat stress, the efflux of exosomes was enhanced and the size of exosomes was larger. Exposure of naïve murine macrophages to exocrine released by 4T1 and EMT-6 cells after hyperthermia resulted in increased expression of specific macrophage activation markers. In addition, the exocrine released by hyperthermic cancer cells increased the content of heat shock protein 70. Taken together, these results suggest that exocrine bodies released from cancer cells treated with mild hyperthermia have potential immunogenicity.
The release and uptake of exosomes in the body is energy-dependent, not free diffusion [4]. By changing the ambient temperature of cultured cells (42°C and 37°C), it was found that the expression of exosome surface markers CD9 and CD63 in the 42°C group was more significant. It was found that the effect of using heat stress induced adipose-derived stem cell (ADSC) exosomes and using β-tricalcium phosphate (TCP) as carrier for bone regeneration was better than using β-TCP or using ADSCs and β-TCP alone [19]. Some studies have even collected exosome containing chemokines from heat-stressed adipocytes through the lipid raft-dependent pathway to attract and activate DC and T cells and induce an effective anti-tumor immune response as a new direction of tumor vaccine development.
Regarding low temperature, some studies have shown that in the brain protection of neurological deficit after deep hypothermic circulatory arrest cardiovascular surgery, exosomes can also stimulate the expression of phosphoprotein kinase B and Bcl-2 by carrying miRNA-214 to achieve neuroprotection in brain injury [20].
The decrease in temperature does not seem to change the role of the exosome itself, but it is gratifying to find that when an exosome is extracted from serum under various storage conditions (4°C, room temperature, and repeated freezing and thawing), it was found that the DNA in serum exosomes remained stable [21]. However, the best storage condition of the isolated exosome is still −80°C [22]. Using the principle of variable temperature, studies have been conducted on the combination of low temperature, exosomes, and biomaterials. The results have shown that when the macromonomer solution is frozen and annealed below 0°C, the polymer molecular chain separates from the solvent ice and crosslinks at a higher concentration. Because the reaction temperature is close to the glass transition temperature of macromonomers, the movement of molecular chains is inhibited, the self-assembly slows down, and the concentration of macromonomers is allowed to increase. Based on this principle, scientists have explored a new method of producing silk fibroin-based frozen sponges to achieve the continuous release of exosomes, the long-term stable release of exosomes derived from bone marrow mesenchymal stem cells (BMSCs), and the degradation of scaffolds [23].
Osmotic Pressure
The changes of osmotic pressure have adverse effects on water flux, cell volume, and signal pathways involved in cell proliferation, cell migration, and apoptosis. The change of exocrine body composition caused by stress is an effective adaptive mechanism, which can help cells regulate intracellular pressure conditions [24,25]. Osmotic pressure increases the size of exocrine bodies and enhances their release. Based on this principle, vimentin carried by adipose progenitor cell-derived exosomes is used to protect fibroblasts from osmotic pressure, thereby inhibiting apoptosis and promoting wound healing [26]. The research on the effect of osmotic pressure on exosomes is mostly focused on the direction of the wound. Changes in osmotic pressure, magnetic field, and other areas affect the role of exosomes in cell regeneration and apoptosis. At present, there are no significant results in the research on osteogenesis, but cell proliferation and regeneration are essential parts of the osteogenic mechanism, which seems to be regulated by this microenvironmental pressure. We aim to find a new direction to promote osteoblast proliferation and bone regeneration.
Blood Glucose
A high-glucose environment in the body can induce a variety of diseases, which are not only the direct manifestation of diabetes, but are also auxiliary factors and causes of other diseases, and the role of exosomes is also significantly affected in a high-glucose environment.
TGF-β1 mRNA is enriched in exosomes of glomerular endothelial cells treated with high glucose and may mediate EMT and podocyte dysfunction [27]. There is a close anatomical relationship between cardiomyocytes and endothelial cells, which is important for maintaining the normal development and function of the heart. Exosomes isolated from human umbilical vein endothelial cells (HG-HUVEC-Exos) stimulated by high glucose induce calcification, aging of vascular smooth muscle cells by increasing the expression of osteocalcin and p21, and the formation of mineralized nodules and SA-β-gal positive cells. This process is closely related to the notch and MTOR signal pathways [28]. When macrophage-derived exosomes were treated with high glucose of 30 μg/mL, exosomes could activate macrophages and induce inflammation through the NF-κB signal pathway [29], which seems to provide a theoretical supplement for the pathogenesis of diabetic nephropathy.
Under the condition of glucose starvation, immortalized H9C2 cardiomyocytes increased exosome secretion, and their incubation with endothelial cells induced endothelial cell proliferation and angiogenesis [30]. The exocrine produced by MSCs cultured in an anoxia and glucose environment had obvious therapeutic effects on brain injury after stroke in rats with transient focal cerebral ischemia [31]. However, the environment of oxygen and glucose deprivation in the body can lead to endothelial cell and neuronal damage, while MSC-Exos play a protective role in injured endothelium [32] and through miRNA-194 transfer, they target the Bach1 signal pathway to improve neuronal injury [33]. HG-HUVEC-Exos induce vascular smooth muscle cell senescence through the circRNA0077930-miR622-KrasCeRNA network, while circRNA-0077930-depleted exosomes lose the ability to promote vascular smooth muscle cell senescence [34]. Astrocyte-derived exosomes improved the differentiation and migration of oligodendrocyte precursor cells, while astrocyte-derived exosomes with severe hypoxia inhibited the proliferation of oligodendrocyte precursor cells. Other studies have shown that astrocyte-derived exosomes may be a potential therapeutic target for myelin regeneration and repair in white matter injury or other demyelinating-related diseases [35].
Osteogenesis of MSC-Exos in Pathological Environment
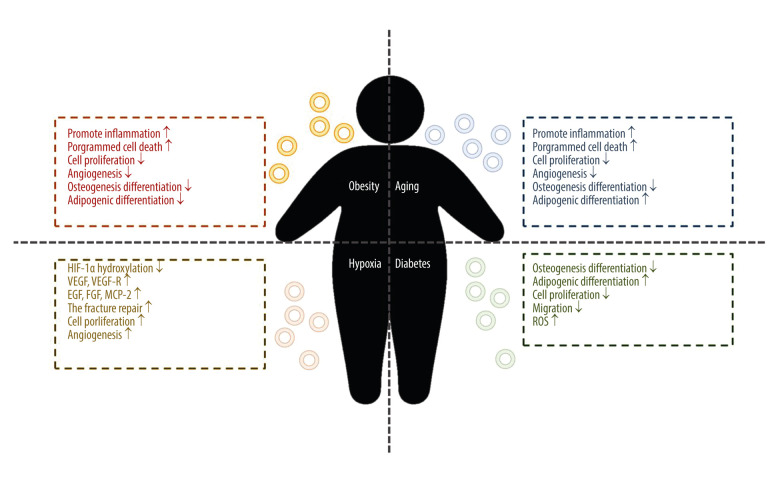
MSCs express and secrete a variety of factors that control immune response, angiogenesis, cell proliferation, migration, invasion, survival, and other factors that promote tissue repair, and thus participate in tissue repair, immune regulation, and anti-inflammation [36]. MSC-Exos retain most of the functions of MSC, and their role in osteogenesis can be classified into 4 aspects: (1) reducing cell apoptosis in an ischemic and necrotic microenvironment; (2) chemotaxis and proliferation of other stem cells or functional cells; (3) angiogenesis around new bone; and (4) directly promoting the osteogenic induction of MSCs [37]. MSCs are affected by different environments; therefore, exosomes with different contents can be released according to specific conditions to adapt to the effects of the environment on MSCs and the microenvironment (a schematic diagram of the effect of systemic disease environment on MSC-Exos is shown in Figure 2).
Figure 2.
The function of mesenchymal stem cell-derived exosomes from the systemic disease environment. Figure created with BioRender.com.
Systemic Disease Environment
Obesity
Obesity is a metabolic disease, the most direct manifestation of which is the increase of fat content in the blood. In the process of bone formation, fat is usually considered as a negative regulatory factor. One study showed that the BMSC-Exo lncRNA-H19 in an obesity model absorbed miRNA-188 through ceRNA inhibited the osteogenesis of mouse BMSC and turned to adipogenic differentiation; the authors also found that BMSC-exo lncRNA-H19 could competitively bind to miRNA-467 and inhibit the expression of Hoxa10, thus inhibiting osteogenesis [38]. The effect of obesity on the expression profile of miRNA in ADSC-Exos may have a direct impact on the function of exosomes. Studies have shown that the number of ADSC extracellular vesicles in patients with obesity is similar to that in patients without obesity, but the volume is smaller, while there are 8 upregulated miRNAs in ADSC extracellular vesicles of obese patients, which may be involved in the regulation of NFK, MAPK, and cytoskeleton formation, and apoptosis, and 75 downregulated, involved in cell cycle, angiogenesis, and the Wnt and MAPK pathways. The effect resulted in upregulation of the pro-inflammatory signal, programmed cell death miRNA, and inhibition of cell proliferation and angiogenesis pathway-related miRNA expression [39]. However, this principle is also being used to extract BMSC-Exo miRNA-29a to inhibit osteoclast formation by regulating PCAF-mediated RANKL and CXCL12, activating the Wnt pathway through Fzd4, and regulating osteogenic differentiation of BMSC in a high-fat environment [40].
Senescence
Previous studies have shown that the osteogenic activity of MSCs is not affected by age, but the latest studies have shown that MSC-Exo miRNA-126 can promote bone healing and is related to age [40]. An in-depth study of the mechanism showed that miRNA-183 clusters were the most differentially expressed in miRNA between 3 to 4 month-old rats and 24 to 28 month-old rats, in which miRNA-183-5p inhibited the proliferation of bone marrow cells with age in osteogenic extracellular vesicles and induced stem cell senescence. This change is also reflected in exosomes targeting heme oxygenase-1 protein levels and can increase cartilaginous production and promote bone resorption, age-related aging stem cell dysfunction, and reactive oxygen species [41]. It is well known that the interaction between monocytes, macrophages, osteoclasts, and BMSC-osteoblasts plays an important role in the pathological process of osteoarthritis. MiRNA-31a-5p can promote osteoclast formation and IL-2 expression through RhoA. SATB2 plays a role in regulating osteogenic differentiation in the process of tooth eruption; therefore, the role of MSC-miRNA31a-5p in the aging bone marrow microenvironment is worthy of further study. Results have shown that the exosomes released by BMSC in the aging environment was rich in miRNA31a-5p, which upregulated the expression of RhoA protein after osteochondral uptake and promoted the proliferation of osteoclasts [42]. MiRNA-31a-5p could also target to inhibit the expression of E2F2 (special AT-rich sequence-binging protein 2) and thus promote the expression of SAHF (senescence associated heterochromatin foci, aging-related heterochromosome focus), promote senescence, inhibit the expression of SATB2, and thus inhibit the promotion of Ob42. Therefore, the study suggests that it is particularly important to find miRNA-31a-5p antagomir [42]. Other studies have found that senile rat MSC-Exo is rich in miRNA-128-3p, which can target Smad5 through 3′UTR binding, thus inhibiting osteogenesis and causing senile-related fractures or osteogenesis inhibition; therefore, the long-term research direction should still focus on miRNA antagomir43. To improve the effect of aging on osteogenesis, we used exosomes in the serum of young rats to carry miRNA-196-3p and reduce the expression of PTEN, so as to reverse the bone micro-injury caused by fatigue load [44].
Hypoxia
A variety of diseases can lead to local hypoxia in the body, including moderate-, high-altitude conditions, and ischemia. Whether it is cardio-cerebrovascular disease [45], refractory wound [46], or a bone defect or fracture nonunion [47], the most characteristic and typical cell growth environment is the pathological environment characterized by hypoxia, inflammatory reaction, and oxidative stress.
It has been found that hypoxia-preconditioned ADSC-Exos can promote angiogenesis in fat grafts and thus promote ADSC and fat-graft survival [48]. Extracellular vesicles in a hypoxic environment is an important factor in regenerative medicine. This effect is induced by HIF-1α, which in a normoxic environment is hydroxylated and cleaved on proline residue by prolyl hydroxylase (PHD). Under hypoxia, PHD needs oxygen to interact with α-ketoglutarate and Fe, so HIF-1α hydroxylation is reduced, and HIF-1α is stable and translocated in the nucleus, activating hypoxia-sensitive genes and regulating cell cycle, apoptosis, and cell differentiation [49]. In addition, MSC under hypoxia showed decreased Mit activity, increased glucose depletion, decreased Mit autophagy, decreased ROS, and decreased telomerase shortening, which was generally characterized by the inhibition of cell senescence [50].
Osteogenic studies suggest that BMSC-Exos modified by mutant HIF-1 α can inhibit early steroid-induced SANFH in rabbits [51]. The transplantation of umbilical cord (U)MSC-Exos significantly enhanced the process of angiogenesis and bone healing in a rat model of femoral fracture. In that in vitro study, UMSC-Exos not only enhanced osteogenic differentiation, but also increased the expression of vascular endothelial growth factor (VEGF) and HIF-1α. UMSC-Exos were absorbed by HUVECs and enhanced their proliferation, migration, and tube formation. Finally, it was confirmed that HIF-1α, a factor that plays a role in vivo through hypoxia, can promote UMSC-Exo-induced VEGF expression, promote angiogenesis, and enhance fracture repair [52]. In other words, hypoxic preconditioning of MSCs can enhance their paracrine effect. MSCs preconditioned by hypoxia carry miRNA-126 through the exocrine and promote fracture healing by acting on the SPRED1/Ras/Erk signaling pathway. Taking MLO-Y4 as the model, under the environment of hypoxia and serum deprivation, ROS can induce apoptosis, and hADSC-Exos can inhibit the expression of RANKL, thus inhibiting osteoclast formation mediated by osteocytes and resisting apoptosis under hypoxia by upregulating Bcl-2/Bax, inhibiting ROS, and reducing the production of cytochrome enzymes [53]. Hyp-ADSC-Exo treatment significantly promoted the proliferation, migration, and tube formation of HUVECs. The expressions of VEGF, EGF, fibroblast growth factor, their receptors (VEGF-R2, VEGF-R3,) and monocyte chemoattractant protein 2 were upregulated. Immunohistochemical analysis showed that hyp-ADSC-Exo treatment significantly increased the expression of VEGF-R in transplanted tissue. Hypoxia-treated human ADSC exosomes have a higher ability to enhance graft angiogenesis [54].
It can be seen that the function and activity of MSCs can be inhibited after hypoxia preconditioning, but in order to maintain the original cell activity, MSCs produce exosomes that are resistant to hypoxia damage by the paracrine method. We can use this preconditioned exosome in hypoxia-induced diseases. The state of hypoxia can be achieved by atmospheric hypoxia, chemical hypoxia, or other methods.
Diabetes
Diabetes-induced bone loss has become a hot topic in recent years. It was found that the increase of miRNA-129-5p exosomes extracted from plasma of diabetic patients acts on FZD4 in BMSCs, thus inhibiting the β-catenin pathway and inhibiting jaw osteogenesis [55]. In addition, compared with ordinary BMSC-Exos, the osteogenic potential and repair ability of exosomes extracted from diabetic BMSC-Exos in vivo and in vitro are lower [56]. Studies have shown that miRNA-144-5P in DBMSC-EXO can inhibit bone regeneration by targeting Smad1. In patients with type I diabetes, the function of BMSC degrades. Studies suggest that there may be 2 mechanisms involved. First, the non-enzymatic reaction between AGEs (sugar residues) and Pro activates the TGF-β and p38MAPK pathways by binding to their receptors, preventing the differentiation of BMSC and inhibiting the proliferation and migration BMSC, promoting ROS-induced apoptosis; Second, the direct effect of high glucose on GSK 3β-mediated cyclin and CXCR-4 inhibits the migration and proliferation of BMSCS, inhibits the CXCL13 pathway to inhibit BMSCS osteogenesis by lncRNA AKO028326, or activates the Wnt11 pathway to promote adipogenesis and inhibit osteogenesis [57].
Orthopedic Disease Environment
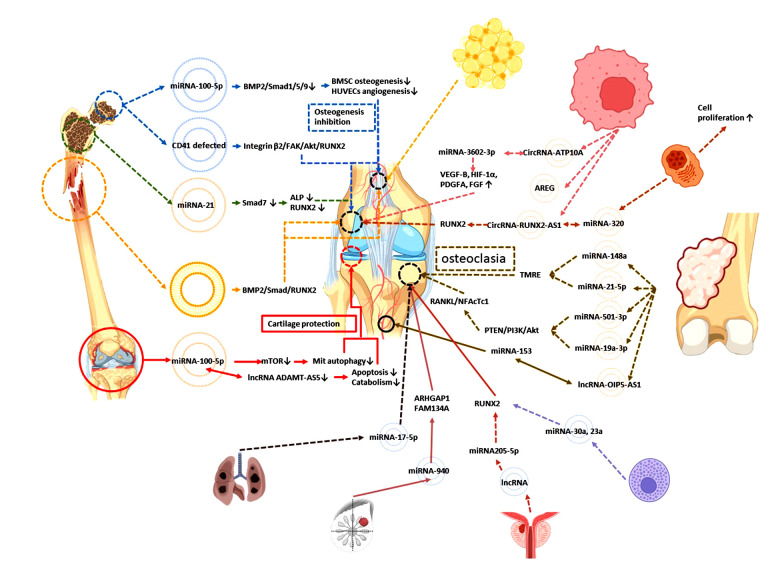
We are accustomed to extracting exosomes from MSCs, such as BMSCs and ADSCs, studying their carriers, and determining the mechanism of MSC-Exos in osteogenesis by detecting their target. We are eager to prepare MSC-Exos as a therapeutic tool that can replace stem cell therapy, but we are not clear about the difference between MSC-Exos in patients with orthopedic diseases and MSC-Exos in healthy people. We have also failed to fully control the changes in compliance or resistance of MSC-Exos under the influence of disease-related stimuli (the functions of exocrine bodies derived from mesenchymal stem cells in orthopedic or neoplastic disease environments are summarized in Figure 3).
Figure 3.
The function of mesenchymal stem cell-derived exosomes from the orthopedic or oncologic disease environment. Figure created with BioRender.com.
Osteoarthritis
The common fat supply site is the lower abdomen or medial thigh, which are routine areas of liposuction, but another source of ADSCs seems to have a unique feature in the pathological environment of osteoarthritis. ADSC-Exos in the subpatellar fat pad obtained by arthroscopy can downregulate mTOR through miRNA-100-5p, promote mitochondrial autophagy, and inhibit the osteoarthritis process in vivo. Inhibiting the expression of MMP13 and lncRNA-ADAMTS5 in vitro can inhibit apoptosis and catabolic factors and protect cartilage under the pathological environment of osteoarthritis [58].
ADSC treated with osteoarthritis synovial fluid showed changes in the expression of a variety of cytokines, including MMP inhibitor (TIMIP1,2), increased services, downregulated expression of IGFBP4,6 and SPP1 (induced release of decomposing factors from synoviocytes and chondrocytes in vitro), PLAUR (UPA receptor) inhibited proliferation, and CTSS (cathepsin) promoted cartilage extracellular matrix degradation, and also had changes in miRNA expression. Mirna-193b-3p, binding to the 3′UTR of MMP19, inhibited nitric oxide production. To encourage cartilage formation in vivo and identify the critical role of many kinds of miRNA, including miR-24-3p, in inhibiting inflammation and cartilage protection [59], MiRNA-24-3p promoted the proliferation of chondrocytes and inhibited the apoptosis of chondrocytes. At the same time, miRNA-92a-3p decreased in cartilage, thus enabling the acetylation and transcription of histone H3 in Col2A1 and avoiding the destruction of the extracellular matrix by proteolytic enzymes [60].
Osteonecrosis of the Femoral Head
Exosomes were extracted from necrotic bone of nontraumatic osteonecrosis of the femoral head (NONFH), and it was found that the expression of miRNA-100-5p was upregulated. Inhibiting the BMPR2/Smad1/5/9 pathway inhibited osteogenesis of BMSCs, inhibited angiogenesis of HUVECs, and promoted adipogenic differentiation of BMSCs [61], which identified a new gene target for inhibiting NONFH. CD41-deficient exosomes extracted from necrotic tissue of NONFH can inhibit osteogenic differentiation and migration of MSCs; the target pathway is the ONFH-Exo-CD41-integrin β3-FAK (focal adhesion kinase)-Akt-RUNX2 pathway [62]. By searching for suppressed miRNA in patients with ONFH, scientists found that miRNA-122-5p targeting SPRY2 (rapid development growth factor homologue 2) through the RTK/Ras/MAPK pathway promoted osteoblast proliferation and differentiation in vitro and inhibited the progression of BMSCs in vivo. BMSC-Exos can improve the immune status in steroid-induced osteonecrosis of the femoral head microenvironment and promote immune homeostasis [63].
Osteoporosis
Studies have shown that when Smad7 is overexpressed, the expression of ALP and RUNX2 is suppressed, and the purification of exosomes from the BMSCs of osteoporotic patients inhibits osteogenesis through the action of miRNA-21 on the Smad7 pathway [64].
Bone Nonunion
In a rat model of bone nonunion, nonunion-Exosome acted on osteoblasts, promoted the BMP2/Smad1/RUNX2 pathway, promoted osteogenesis, or acted on endothelial cells, promoting angiogenesis through the HIF-1a/VEGF pathway and promoting fracture healing through direct or indirect methods [65].
Oncologic Disease Environment
Tumors, as diseased tissue in the human body, present an urgent problem for us to overcome in disease research. However, at this stage, it is impossible to cure or prevent tumors. Therefore, it is important to study the osteogenic significance of tumor-related exosomes, which will broaden the study of the osteogenic mechanisms of exosomes.
Lipomas
Lipomas are the most common benign tumors in soft-tissue tumors. Lipoma-derived extracellular vesicles can promote the proliferation, migration, and adipogenic differentiation of ADSCs, recruit adipocytes in vivo, and promote angiogenesis; however, there is still a lack of research on the role of the osteogenic microenvironment in lipomas [7]. This study provides a new direction of exosome research, and the discarded lipoma tissue in clinical work may have greater value for use in exosome research [7].
Leukemia
In vivo and in vitro studies have confirmed that chronic myeloid leukemia can inhibit the osteogenic differentiation of BMSCs and chronic myelogenous leukemia.
MiRNA-320 exosomes can promote cell proliferation, but co-cultured with BMSCs, they can inhibit the expression of β-catenin, thus inhibiting osteogenesis. However, the selection of miRNA-320 is not random, but miRNA sorting is mediated by HNRNPA1 exons, which inhibits osteogenic differentiation. Although the study tends to the pathological bone marrow ecological environment of chronic myelogenous leukemia, it provides a new idea for the study of the mechanism of MSC-Exo-mediated osteogenesis [66].
Multiple myeloma
Angiogenesis is very important in multiple myeloma (MM). The expression of circ-ATP10A in patients with MM is significantly increased. Bioinformatics studies have found that circRNA-ATP10A can act as the sponge of hsa-miR-6758-3p/hsa-miR3977/hsa-miR-6804-3p/hsa-miR 1266-3p/hsa-miR-3620-3p and regulate downstream mRNA, thus upregulating the expression of VEGF-B, HIF-1α, platelet-derived growth factor A, and fibroblast growth factor to achieve angiogenesis in MM [67]. MM-Exos activate the EGF pathway in the bone microenvironment through rich double regulatory protein (amphiregulin, AREG), which leads to osteoclast formation. LncRNA RUNX2-AS1 contained in exosomes inhibits the expression of RUNX2, thus inhibiting osteogenesis [68]. From the abovementioned effects of MM-Exos on osteogenesis, it can be said that the function of osteogenesis and osteoclast is affected by different signal regulation, and the function is uncertain. Other studies found that MM-Exo lncRNA RUNX2-AS1 transferred to MSCs, promoting osteogenesis [69]. Therefore, some researchers believe that the use of lncRNA is unstable and the research prospect is uncertain.
Osteosarcoma
Exosomes carrying genetic material participate in the development of osteosarcoma by promoting angiogenesis, invasion and metastasis, immune escape, and drug resistance [70].
CircRNA-0056285 can promote the proliferation of osteosarcoma through sponge miR-1244 and enhance the expression of TRIM44, thus promoting the growth of osteosarcoma tumors in vivo [71]. Secreted by osteosarcoma cells and carrying miR-148a and miR-21-5p as exosomes of cargo, exosomes can participate in the establishment of the tumor microenvironment and stimulate endothelial cells to secrete more angiogenic factors and organize into tubular structures. Osteosarcoma-derived exosomes promote osteoclast differentiation and bone resorption activity, thus affecting the tumor microenvironment, which may be important for the changes of osteoclast phenotype and endothelial cell activity [72]. Osteosarcoma miRNA-5013p exosomes promote the differentiation of osteochondral cells and aggravate bone loss through the PTEN/PI3K/Akt pathway and upregulation of RANKL/NFAcTc1 [73]. MiRNA-19a-3p exosomes can also have a similar role [74]. Osteosarcoma lncRNAOIP5-AS1 exosomes can promote the proliferation, migration, and invasion of osteosarcoma cells, while there is a negative correlation between miRNA-153 and lncRNA OIP5-AS1, which can inhibit the metastasis and invasion of an osteosarcoma tumor. However, what is more important is the regulation of angiogenesis of these microRNAs and lncRNAs [75]. The ability of angiogenesis in tumors is much better than that in normal tissue. If this ability can be transferred to the angiogenesis of MSC-Exo osteogenesis, it seems it can improve the ability of osteogenesis. In addition, high mobility group box 4 (SOX4) associated with the sex determination region has been identified as the direct target gene of miR-25-3p. MiR-25-3p can inhibit the proliferation, migration, and invasion of osteosarcoma cells by targeting the expression of SOX4 in bone tissue, thus inhibiting cancer [76]. In addition, miR-253p embedded in exosomes derived from osteosarcoma cells can promote capillary formation and vascular endothelial cell invasion [77].
Systemic Mastocytosis
Osteoporosis and other bone diseases often occur in patients with systemic mastocytosis (SM), which is related to the presence of mast cell infiltration in bone marrow. SM extracellular vesicles released by tumorous mast cells and present in the serum of patients with SM block osteoblast differentiation and mineralization in vitro and the expression volume and microstructure of osteoblast markers and bone trabeculae in vivo. MiRNA-30a and miRNA-23a increased in SM-Exos and exosomes derived from tumor mast cells, which attenuated osteoblast maturation and aggravated bone mass loss by inhibiting the expression of RUNX2 and Smad1/5 [78].
Primary Tumors Prone to Bone Metastasis
Prostate cancer cell lncRNA nuclei exosomes are rich in transcript 1 by inhibiting miR-205-5p and SFPQ-PTBP2 pathways to inhibit RUNX2 and initiate osteogenic phenotypes in the bone metastasis microenvironment [79]. Hsa-miRNA-940 from breast cancer cells targets ARHGAP1 (including GTPase activating protein factor) and FAM134A, upregulates GTPase activity, and inactivates G protein, thus promoting osteogenic differentiation of human MSCs. This mechanism acts in the microenvironment of bone metastasis to induce the osteoblast phenotype [80]. Non-small cell lung cancer miRNA-17-5p exosomes target PTEN to promote osteochondral differentiation and bone destruction [81].
Research Significance and Future Prospects
Exosomes have favorable and unfavorable roles, by inhibiting tumor progression and playing an important role in tumorigenesis, angiogenesis, and metastasis [82]. However, in the aspect of osteogenesis of MSC-Exos, the exosomes from different pathological environments and tissue sources showed different effects. At present, most of the studies on MSC-Exos extracted from pathological environments or disease states have focused on the explanation of the disease formation mechanism. Future studies should focus on the positive or negative effects of these pathological factors on MSC-Exos, blocking the negative effects and magnifying the positive effects. Taking environmental factors as one of the important methods to modify MSC-Exos, the initialization and improvement of exosomes were realized to a great extent by means of hypoxia, the change of pH value, and the concentration of serum glucose. Combined with the advantages of biomaterials in different environments, this promoted the application of MSC-Exos in bone defects in vivo.
Conclusions
This review has presented an update on the role of MSCs and MSC-Exos in treating osteoporosis and orthopedic diseases, including the repair of cartilage, tendons, bone fractures. Future studies in this field may provide new approaches for treatment and disease prevention in the field of orthopedic medicine.
Footnotes
Conflict of interest: None declared
Declaration of Figures’ Authenticity
All figures submitted have been created by the authors who confirm that the images are original with no duplication and have not been previously published in whole or in part.
Financial support: The study was supported by the National Natural Science Foundation of China (grant/award no. 51872332); Liaoning Provincial Science and Technology Innovation Leading Talents Program (grant/award no. XLYC2002103); Basic Research Project of the Education Department of Liaoning Province (grant/award no. LJKZ0740); National Natural Science Foundation of China (grant/award no. 51872332); Liaoning Provincial Science and Technology Innovation Leading Talents Program (grant/award no. XLYC2002103); and the Basic Research Project of the Education Department of Liaoning Province (grant/award no. LJKZ0740)
References
- 1.Tkach M, Thery C. Communication by extracellular vesicles: Ehere we are and where we need to go. Cell. 2016;164(6):1226–32. doi: 10.1016/j.cell.2016.01.043. [DOI] [PubMed] [Google Scholar]
- 2.Goldie BJ, Dun MD, Lin M, et al. Activity-associated miRNA are packaged in Map1b-enriched exosomes released from depolarized neurons. Nucleic Acids Res. 2014;42(14):9195–208. doi: 10.1093/nar/gku594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Baglio SR, Rooijers K, Koppers-Lalic D, et al. Human bone marrow- and adipose-mesenchymal stem cells secrete exosomes enriched in distinctive miRNA and tRNA species. Stem Cell Res Ther. 2015;6:127. doi: 10.1186/s13287-015-0116-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Narayanan K, Kumar S, Padmanabhan P, et al. Lineage-specific exosomes could override extracellular matrix mediated human mesenchymal stem cell differentiation. Biomaterials. 2018;182:312–22. doi: 10.1016/j.biomaterials.2018.08.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Brown C, McKee C, Bakshi S, et al. Mesenchymal stem cells: Cell therapy and regeneration potential. J Tissue Eng Regen Med. 2019;13(9):1738–55. doi: 10.1002/term.2914. [DOI] [PubMed] [Google Scholar]
- 6.Malekpour K, Hazrati A, Zahar M, et al. The potential use of mesenchymal stem cells and their derived exosomes for orthopedic diseases treatment. Stem Cell Rev Rep. 2021 doi: 10.1007/s12015-021-10185-z. [Online ahead of print] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Hong P, Xu X, Hu X, et al. Therapeutic potential of small extracellular vesicles derived from lipoma tissue in adipose tissue regeneration-an in vitro and in vivo study. Stem Cell Res Ther. 2021;12(1):222. doi: 10.1186/s13287-021-02291-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Yin Y, Chen H, Wang Y, et al. Roles of extracellular vesicles in the aging microenvironment and age-related diseases. J Extracell Vesicles. 2021;10(12):e12154. doi: 10.1002/jev2.12154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Logozzi M, Spugnini E, Mizzoni D, et al. Extracellular acidity and increased exosome release as key phenotypes of malignant tumors. Cancer Metastasis Rev. 2019;38(1–2):93–101. doi: 10.1007/s10555-019-09783-8. [DOI] [PubMed] [Google Scholar]
- 10.Parolini I, Federici C, Raggi C, et al. Microenvironmental pH is a key factor for exosome traffic in tumor cells. J Biol Chem. 2009;284(49):34211–22. doi: 10.1074/jbc.M109.041152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Zhao Y, Chen K, Li H, Wu H. Effect of pH on the isolation of urinary exosome. Int Urol Nephrol. 2017;49(1):165–69. doi: 10.1007/s11255-016-1408-7. [DOI] [PubMed] [Google Scholar]
- 12.Ban JJ, Lee M, Im W, Kim M. Low pH increases the yield of exosome isolation. Biochem Biophys Res Commun. 2015;461(1):76–79. doi: 10.1016/j.bbrc.2015.03.172. [DOI] [PubMed] [Google Scholar]
- 13.Jeyaram A, Lamichhane TN, Wang S, et al. Enhanced loading of functional miRNA cargo via pH gradient modification of extracellular vesicles. Mol Ther. 2020;28(3):975–85. doi: 10.1016/j.ymthe.2019.12.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Wang F, Shi R, Cai F, et al. Stem cell approaches to intervertebral disc regeneration: Obstacles from the disc microenvironment. Stem Cells Dev. 2015;24(21):2479–95. doi: 10.1089/scd.2015.0158. [DOI] [PubMed] [Google Scholar]
- 15.Li M, Li R, Yang S, et al. Exosomes derived from bone marrow mesenchymal stem cells prevent acidic pH-induced damage in human nucleus pulposus cells. Med Sci Monit. 2020;26:e922928. doi: 10.12659/MSM.922928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Yang L, Han D, Zhan Q, et al. Blood TfR+ exosomes separated by a pH-responsive method deliver chemotherapeutics for tumor therapy. Theranostics. 2019;9(25):7680–96. doi: 10.7150/thno.37220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Dou J, Khan A, Khan MZ, et al. Heat stress impairs the physiological responses and regulates genes coding for extracellular exosomal proteins in rat. Genes. 2020;11(3):306. doi: 10.3390/genes11030306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Shi F, Deng Z, Zhou Z, et al. Heat injured stromal cells-derived exosomal EGFR enhances prostatic wound healing after thulium laser resection through EMT and NF-κB signaling. Prostate. 2019;79(11):1238–55. doi: 10.1002/pros.23827. [DOI] [PubMed] [Google Scholar]
- 19.Akdeniz-Dogan Z, Sendur S, Karademir-Yilmaz B, et al. The role of extracellular vesicles secreted from thermal stress-induced adipose-derived stem cells on bone regeneration. J Craniofac Surg. 2021;32(6):2245–50. doi: 10.1097/SCS.0000000000007901. [DOI] [PubMed] [Google Scholar]
- 20.Shi J, Jiang X, Gao S, et al. Gene-modified exosomes protect the brain against prolonged deep hypothermic circulatory arrest. Ann Thorac Surg. 2021;111(2):576–85. doi: 10.1016/j.athoracsur.2020.05.075. [DOI] [PubMed] [Google Scholar]
- 21.Jin Y, Chen K, Wang Z, et al. DNA in serum extracellular vesicles is stable under different storage conditions. BMC Cancer. 2016;16(1):753. doi: 10.1186/s12885-016-2783-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Yuan F, Li YM, Wang Z. Preserving extracellular vesicles for biomedical applications: Consideration of storage stability before and after isolation. Drug Deliv. 2021;28(1):1501–9. doi: 10.1080/10717544.2021.1951896. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Sun M, Li Q, Yu H, et al. Cryo-self-assembled silk fibroin sponge as a biodegradable platform for enzyme-responsive delivery of exosomes. Bioact Mater. 2022;8:505–14. doi: 10.1016/j.bioactmat.2021.06.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Wang T, Jian Z, Baskys A, et al. MSC-derived exosomes protect against oxidative stress-induced skin injury via adaptive regulation of the NRF2 defense system. Biomaterials. 2020;257:120264. doi: 10.1016/j.biomaterials.2020.120264. [DOI] [PubMed] [Google Scholar]
- 25.Parvanian S, Yan F, Su D, et al. Exosomal vimentin from adipocyte progenitors accelerates wound healing. Cytoskeleton (Hoboken, NJ) 2020;77(10):399–413. doi: 10.1002/cm.21634. [DOI] [PubMed] [Google Scholar]
- 26.Parvanian S, Zha H, Su D, et al. Exosomal vimentin from adipocyte progenitors protects fibroblasts against osmotic stress and inhibits apoptosis to enhance wound healing. Int J Mol Sci. 2021;22(9):4678. doi: 10.3390/ijms22094678. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Wu X, Gao Y, Xu L, et al. Exosomes from high glucose-treated glomerular endothelial cells trigger the epithelial-mesenchymal transition and dysfunction of podocytes. Sci Rep. 2017;7(1):9371. doi: 10.1038/s41598-017-09907-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Lin X, Li S, Wang YJ, et al. Exosomal Notch3 from high glucose-stimulated endothelial cells regulates vascular smooth muscle cells calcification/aging. Life Sci. 2019;232:116582. doi: 10.1016/j.lfs.2019.116582. [DOI] [PubMed] [Google Scholar]
- 29.Zhu M, Sun X, Qi X, Xia L, Wu Y. Exosomes from high glucose-treated macrophages activate macrophages andinduce inflammatory responses via NF-κB signaling pathway in vitro and in vivo. Int Immunopharmacol. 2020;84:106551. doi: 10.1016/j.intimp.2020.106551. [DOI] [PubMed] [Google Scholar]
- 30.Garcia NA, Ontoria-Oviedo I, González-King H, et al. Glucose starvation in cardiomyocytes enhances exosome secretion and promotes angiogenesis in endothelial cells. PLoS One. 2015;10(9):e0138849. doi: 10.1371/journal.pone.0138849. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Nalamolu KR, Venkatesh I, Mohandass A, et al. Exosomes secreted by the cocultures of normal and oxygen-glucose-deprived stem cells improve post-stroke outcome. Neuromolecular Med. 2019;21(4):529–39. doi: 10.1007/s12017-019-08540-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Kong LY, Liang MY, Liu JP, et al. Mesenchymal stem cell-derived exosomes rescue oxygen-glucose deprivation-induced injury in endothelial cells. Curr Neurovasc Res. 2020;17(2):155–63. doi: 10.2174/1567202617666200214103950. [DOI] [PubMed] [Google Scholar]
- 33.Li X, Zhang X, Liu Y, et al. Exosomes derived from mesenchyml stem cells ameliorate oxygen-glucose deprivation/reoxygenation-induced neuronal injury via transferring MicroRNA-194 and targeting Bach1. Tissue Cell. 2021;73:101651. doi: 10.1016/j.tice.2021.101651. [DOI] [PubMed] [Google Scholar]
- 34.Wang S, Zhan J, Lin X, et al. CircRNA-0077930 from hyperglycaemia-stimulated vascular endothelial cell exosomes regulates senescence in vascular smooth muscle cells. Cell Biochem Funct. 2020;38(8):1056–68. doi: 10.1002/cbf.3543. [DOI] [PubMed] [Google Scholar]
- 35.Xu Y, Tian Y, Wang Y, et al. Exosomes derived from astrocytes after oxygen-glucose deprivation promote differentiation and migration of oligodendrocyte precursor cells in vitro. Mol Biol Rep. 2021;48(7):5473–84. doi: 10.1007/s11033-021-06557-w. [DOI] [PubMed] [Google Scholar]
- 36.Heo JS, Choi Y, Kim HO. Adipose-derived mesenchymal stem cells promote M2 macrophage phenotype through exosomes. Stem Cells Int. 2019;2019:7921760. doi: 10.1155/2019/7921760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Li W, Liu Y, Zhang P, et al. Tissue-engineered bone immobilized with human adipose stem cells-derived exosomes promotes bone regeneration. ACS Appl Mater Interfaces. 2018;10(6):5240–54. doi: 10.1021/acsami.7b17620. [DOI] [PubMed] [Google Scholar]
- 38.Wang Y, Chen W, Zhao L, et al. Obesity regulates miR-467/HoxA10 axis on osteogenic differentiation and fracture healing by BMSC-derived exosome LncRNA H19. J Cell Mol Med. 2021;25(3):1712–24. doi: 10.1111/jcmm.16273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Eirin A, Meng Y, Zhu XY, et al. The micro-RNA cargo of extracellular vesicles released by human adipose tissue-derived mesenchymal stem cells is modified by obesity. Front Cell Dev Biol. 2021;9:660851. doi: 10.3389/fcell.2021.660851. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Lu GD, Cheng P, Liu T, Wang Z. BMSC-derived exosomal miR-29a promotes angiogenesis and osteogenesis. Front Cell Dev Biol. 2020;8:608521. doi: 10.3389/fcell.2020.608521. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Davis C, Dukes A, Drewry M, et al. MicroRNA-183-5p increases with age in bone-derived extracellular vesicles, suppresses bone marrow stromal (stem) cell proliferation, and induces stem cell senescence. Tissue Eng Part A. 2017;23(21–22):1231–40. doi: 10.1089/ten.tea.2016.0525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Xu R, Shen X, Si Y, et al. MicroRNA-31a-5p from aging BMSCs links bone formation and resorption in the aged bone marrow microenvironment. Aging Cell. 2018;17(4):e12794. doi: 10.1111/acel.12794. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Xu T, Luo Y, Wang J, et al. Exosomal miRNA-128-3p from mesenchymal stem cells of aged rats regulates osteogenesis and bone fracture healing by targeting Smad5. J Nanobiotechnology. 2020;18(1):47. doi: 10.1186/s12951-020-00601-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Xun J, Li C, Liu M, et al. Serum exosomes from young rats improve the reduced osteogenic differentiation of BMSCs in aged rats with osteoporosis after fatigue loading in vivo. Stem Cell Res Ther. 2021;12(1):424. doi: 10.1186/s13287-021-02449-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Hou K, Li G, Zhao J, et al. Bone mesenchymal stem cell-derived exosomal microRNA-29b-3p prevents hypoxic-ischemic injury in rat brain by activating the PTEN-mediated Akt signaling pathway. J Neuroinflammation. 2020;17(1):46. doi: 10.1186/s12974-020-1725-8. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 46.Qiu X, Liu J, Zheng C, et al. Exosomes released from educated mesenchymal stem cells accelerate cutaneous wound healing via promoting angiogenesis. Cell Prolif. 2020;53(8):e12830. doi: 10.1111/cpr.12830. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Liu W, Li L, Rong Y, et al. Hypoxic mesenchymal stem cell-derived exosomes promote bone fracture healing by the transfer of miR-126. Acta Biomater. 2020;103:196–212. doi: 10.1016/j.actbio.2019.12.020. [DOI] [PubMed] [Google Scholar]
- 48.Han YD, Bai Y, Yan XL, et al. Co-transplantation of exosomes derived from hypoxia-preconditioned adipose mesenchymal stem cells promotes neovascularization and graft survival in fat grafting. Biochem Biophys Res Commun. 2018;497(1):305–12. doi: 10.1016/j.bbrc.2018.02.076. [DOI] [PubMed] [Google Scholar]
- 49.Imanirad P, Solaimani Kartalaei P, Crisan M, et al. HIF1α is a regulator of hematopoietic progenitor and stem cell development in hypoxic sites of the mouse embryo. Stem Cell Res. 2014;12(1):24–35. doi: 10.1016/j.scr.2013.09.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Noronha NC, Mizukami A, Caliári-Oliveira C, et al. Priming approaches to improve the efficacy of mesenchymal stromal cell-based therapies. Stem Cell Res Ther. 2019;10(1):131. doi: 10.1186/s13287-019-1224-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Li H, Liu D, Li C, et al. Exosomes secreted from mutant-HIF-1α-modified bone-marrow-derived mesenchymal stem cells attenuate early steroid-induced avascular necrosis of femoral head in rabbit. Cell Biol Int. 2017;41(12):1379–90. doi: 10.1002/cbin.10869. [DOI] [PubMed] [Google Scholar]
- 52.Zhang Y, Hao Z, Wang P, et al. Exosomes from human umbilical cord mesenchymal stem cells enhance fracture healing through HIF-1α-mediated promotion of angiogenesis in a rat model of stabilized fracture. Cell Prolif. 2019;52(2):e12570. doi: 10.1111/cpr.12570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Ren L, Song ZJ, Cai QW, et al. Adipose mesenchymal stem cell-derived exosomes ameliorate hypoxia/serum deprivation-induced osteocyte apoptosis and osteocyte-mediated osteoclastogenesis in vitro. Biochem Biophys Res Commun. 2019;508(1):138–44. doi: 10.1016/j.bbrc.2018.11.109. [DOI] [PubMed] [Google Scholar]
- 54.Han Y, Ren J, Bai Y, Pei X, Han Y. Exosomes from hypoxia-treated human adipose-derived mesenchymal stem cells enhance angiogenesis through VEGF/VEGF-R. Int J Biochem Cell Biol. 2019;109:59–68. doi: 10.1016/j.biocel.2019.01.017. [DOI] [PubMed] [Google Scholar]
- 55.Wang J, Xia Y, Li J, Wang W. miR-129-5p in exosomes inhibits diabetes-associated osteogenesis in the jaw via targeting FZD4. Biochem Biophys Res Commun. 2021;566:87–93. doi: 10.1016/j.bbrc.2021.05.072. [DOI] [PubMed] [Google Scholar]
- 56.Zhang D, Wu Y, Li Z, et al. MiR-144-5p, an exosomal miRNA from bone marrow-derived macrophage in type 2 diabetes, impairs bone fracture healing via targeting Smad1. J Nanobiotechnology. 2021;19(1):226. doi: 10.1186/s12951-021-00964-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Zhu Y, Jia Y, Wang Y, et al. Impaired bone regenerative effect of exosomes derived from bone marrow mesenchymal stem cells in type 1 diabetes. Stem Cells Transl Med. 2019;8(6):593–605. doi: 10.1002/sctm.18-0199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Wu J, Kuang L, Chen C, et al. miR-100-5p-abundant exosomes derived from infrapatellar fat pad MSCs protect articular cartilage and ameliorate gait abnormalities via inhibition of mTOR in osteoarthritis. Biomaterials. 2019;206:87–100. doi: 10.1016/j.biomaterials.2019.03.022. [DOI] [PubMed] [Google Scholar]
- 59.Ragni E, Colombini A, Vigano M, et al. Cartilage protective and immunomodulatory features of osteoarthritis synovial fluid-treated adipose-derived mesenchymal stem cells secreted factors and extracellular vesicles-embedded miRNAs. Cells. 2021;10(5):1072. doi: 10.3390/cells10051072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Lv Q, Deng J, Chen Y, Wang Y, et al. Engineered human adipose stem-cell-derived exosomes loaded with miR-21-5p to promote diabetic cutaneous wound healing. Mol Pharm. 2020;17(5):1723–33. doi: 10.1021/acs.molpharmaceut.0c00177. [DOI] [PubMed] [Google Scholar]
- 61.Yang W, Zhu W, Yang Y, et al. Exosomal miR-100-5p inhibits osteogenesis of hBMSCs and angiogenesis of HUVECs by suppressing the BMPR2/Smad1/5/9 signalling pathway. Stem Cell Res Ther. 2021;12(1):390. doi: 10.1186/s13287-021-02438-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Li L, Wang Y, Yu X, et al. Bone marrow mesenchymal stem cell-derived exosomes promote plasminogen activator inhibitor 1 expression in vascular cells in the local microenvironment during rabbit osteonecrosis of the femoral head. Stem Cell Res Ther. 2020;11(1):480. doi: 10.1186/s13287-020-01991-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Fang S, Li Y, Chen P. Osteogenic effect of bone marrow mesenchymal stem cell-derived exosomes on steroid-induced osteonecrosis of the femoral head. Drug Des Dev Ther. 2018;13:45–55. doi: 10.2147/DDDT.S178698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Jiang LB, Tian L, Zhang CG. Bone marrow stem cells-derived exosomes extracted from osteoporosis patients inhibit osteogenesis via microRNA-21/SMAD7. Eur Rev Med Pharmacol Sci. 2018;22(19):6221–29. doi: 10.26355/eurrev_201810_16028. [DOI] [PubMed] [Google Scholar]
- 65.Zhang L, Jiao G, Ren S, et al. Exosomes from bone marrow mesenchymal stem cells enhance fracture healing through the promotion of osteogenesis and angiogenesis in a rat model of nonunion. Stem Cell Res Ther. 2020;11(1):38. doi: 10.1186/s13287-020-1562-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Gao X, Wan Z, Wei M, et al. Chronic myelogenous leukemia cells remodel the bone marrow niche via exosome-mediated transfer of miR-320. Theranostics. 2019;9(19):5642–56. doi: 10.7150/thno.34813. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Yu M, Yu J, Zhang Y, et al. A novel circRNA-miRNA-mRNA network revealed exosomal circ-ATP10A as a biomarker for multiple myeloma angiogenesis. Bioengineered. 2021;13(1):667–83. doi: 10.1080/21655979.2021.2012553. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Raimondo S, Saieva L, Vicario E, et al. Multiple myeloma-derived exosomes are enriched of amphiregulin (AREG) and activate the epidermal growth factor pathway in the bone microenvironment leading to osteoclastogenesis. J Hematol Oncol. 2019;12(1):2. doi: 10.1186/s13045-018-0689-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Li B, Xu H, Han H, et al. Exosome-mediated transfer of lncRUNX2-AS1 from multiple myeloma cells to MSCs contributes to osteogenesis. Oncogene. 2018;37(41):5508–19. doi: 10.1038/s41388-018-0359-0. [DOI] [PubMed] [Google Scholar]
- 70.Zhang XB, Zhang RH, Su X, et al. Exosomes in osteosarcoma research and preclinical practice. Am J Transl Res. 2021;13(3):882–97. [PMC free article] [PubMed] [Google Scholar]
- 71.Huo S, Dou D. Circ_0056285 regulates proliferation, apoptosis and glycolysis of osteosarcoma cells via miR-1244/TRIM44 axis. Cancer Manag Res. 2021;13:1257–70. doi: 10.2147/CMAR.S290645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Raimondi L, De Luca A, Gallo A, et al. Osteosarcoma cell-derived exosomes affect tumor microenvironment by specific packaging of microRNAs. Carcinogenesis. 2020;41(5):666–77. doi: 10.1093/carcin/bgz130. [DOI] [PubMed] [Google Scholar]
- 73.Lin L, Wang H, Guo W, et al. Osteosarcoma-derived exosomal miR-501-3p promotes osteoclastogenesis and aggravates bone loss. Cell Signal. 2021;82:109935. doi: 10.1016/j.cellsig.2021.109935. [DOI] [PubMed] [Google Scholar]
- 74.Luo T, Zhou X, Jiang E, et al. Osteosarcoma cell-derived small extracellular vesicles enhance osteoclastogenesis and bone resorption through transferring MicroRNA-19a-3p. Front Oncol. 2021;11:618662. doi: 10.3389/fonc.2021.618662. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Li Y, Lin S, Xie X, et al. Highly enriched exosomal lncRNA OIP5-AS1 regulates osteosarcoma tumor angiogenesis and autophagy through miR-153 and ATG5. Am J Transl Res. 2021;13(5):4211–23. [PMC free article] [PubMed] [Google Scholar]
- 76.Wu X, Zhou H, Yue B, et al. Upregulation of microRNA-25-3p inhibits proliferation, migration and invasion of osteosarcoma cells in vitro by directly targeting SOX4. Mol Med Rep. 2017;16(4):4293–300. doi: 10.3892/mmr.2017.7103. [DOI] [PubMed] [Google Scholar]
- 77.Yoshida A, Fujiwara T, Uotani K, et al. Clinical and functional significance of intracellular and extracellular microRNA-25-3p in osteosarcoma. Acta Medica Okayama. 2018;72(2):165–74. doi: 10.18926/AMO/55857. [DOI] [PubMed] [Google Scholar]
- 78.Kim DK, Bandara G, Cho YE, et al. Mastocytosis-derived extracellular vesicles deliver miR-23a and miR-30a into pre-osteoblasts and prevent osteoblastogenesis and bone formation. Nat Commun. 2021;12(1):2527. doi: 10.1038/s41467-021-22754-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Mo C, Huang B, Zhuang J, et al. LncRNA nuclear-enriched abundant transcript 1 shuttled by prostate cancer cells-secreted exosomes initiates osteoblastic phenotypes in the bone metastatic microenvironment via miR-205-5p/runt-related transcription factor 2/splicing factor proline- and glutamine-rich/polypyrimidine tract-binding protein 2 axis. Clin Transl Med. 2021;11(8):e493. doi: 10.1002/ctm2.493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Hashimoto K, Ochi H, Sunamura S, et al. Cancer-secreted hsa-miR-940 induces an osteoblastic phenotype in the bone metastatic microenvironment via targeting ARHGAP1 and FAM134A. Proc Natl Acad Sci USA. 2018;115(9):2204–9. doi: 10.1073/pnas.1717363115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Wang M, Zhao M, Guo Q, et al. Non-small cell lung cancer cell-derived exosomal miR-17-5p promotes osteoclast differentiation by targeting PTEN. Exp Cell Res. 2021;408(1):112834. doi: 10.1016/j.yexcr.2021.112834. [DOI] [PubMed] [Google Scholar]
- 82.Vakhshiteh F, Atyabi F, Ostad SN. Mesenchymal stem cell exosomes: A two-edged sword in cancer therapy. Int J Nanomedicine. 2019;14:2847–59. doi: 10.2147/IJN.S200036. [DOI] [PMC free article] [PubMed] [Google Scholar]