Abstract
There are two estrogen receptor (ER) genes (ESR1/ERα and ESR2/ERβ) in humans. Of those. ERβ, the second ER isotype identified in 1996, is differentially expressed in different phenotypes and molecular subtypes of breast cancer (BCa), and is highly expressed in ERα-negative BCa and triple-negative BCa (TNBC). This review summarizes the potential clinical relevance of ERβ in BCa and the challenges associated with studies on the role of ERβ in BCa. The experimental and clinical studies evaluating clinical outcomes and associations with clinical characteristics and responses to endocrine therapy on targeting ERβ reviewed herein indicate that ERβ is a clinically important biomarker in BCa. The reviewed studies also suggest that each ERβ isoform has a distinct role in BCa subtypes and the potential of novel- targeted therapies in BCa, especially ERα-negative BCa and TNBC. However, the findings of many studies on ERβ are inconsistent, and the exact role of ERβ in BCa remains elusive; this may potentially be attributed to the complexity of ERβ isoforms, but also to the lack of standardized testing protocol. Thus, successful clinical application of ERβ requires the development of standardized, reproducible, and objective measurement methods for ERβ that can be widely and routinely applied in clinical setting.
Keywords: Estrogen Receptor Beta, Patient Outcome Assessment, Prognosis, Survival Analysis, Therapeutic Uses
INTRODUCTION
There are two estrogen receptor (ER) genes (ESR1/ERα and ESR2/ERβ) in humans. Of those, the first isotype, ERα was discovered in the late 1950s and is a strong prognostic and predictive factor for the likelihood of response to endocrine therapies in breast cancer (BCa). ERβ, the second ER isotype was first identified in 1996 [1]. ERα and ERβ are members of the nuclear receptor, superfamily of transcription factors and share some structural similarities, including a high degree of homology (96%) in their DNA-binding regions. However, they differ in gene structure, tissue distribution, ligand selectivity, and binding to pharmacological agents, sharing only moderate homology in the ligand-binding region; further, they have considerably distinct NH2-terminal activation function-1 regions. ERα contains two distinct activation domains, namely ERα-AF-1 and ERα-AF-2, whereas ERβ contains a functional AF-2 and does not contain a strong AF-1 in its amino N-terminus but rather harbors a repressor domain. ERβ functions as a transdominant inhibitor of ERα transcriptional activity [2]. Furthermore, ERα and ERβ have been shown to form heterodimers. Thus, the relative levels of ERα and ERβ in BCa are likely to affect cell proliferation as well as the activities of diverse signaling pathways and their response to ER ligands and endocrine therapies in BCa [3]
ESR2 encodes different ERβ isoforms due to exon deletions or alternative splicing of the last coding exon (exon 8) truncated at the C-terminus. It has been shown that ERβ isoforms differ from each-other in their ligand- and DNA-binding abilities, as well as their ERα interaction properties. The full-length ERβ1 is the primary ERβ isoform that mediates gene expression and growth inhibition in response to estrogen or ERβ-selective ligands and is an obligatory partner in ERβ dimers, whereas the other ERβ isoforms function as variable dimer partners [4]. ERβcx isoform (identical to ERβ2) is the best-characterized isoform, and preferentially forms a heterodimer with ERα rather than with other ERβ isoforms, and shows a substantially dominant negative activity against ERα transactivation [5]. ERβ5 also has estrogen–independent transcriptional properties; it might be playing a significant role in BCa [6]. Thus, each isoform can contribute to tumor progression by activating the transcription of cancer-promoting genes, independent of estrogen or growth factors in BCa or by suppressing ERα transcriptional activity.
An increased understanding of the molecular pathways in BCa, and the development of molecularly based diagnostic and predictive tools have uncovered a substantial biological diversity in the phenotypic and molecular heterogeneity in BCa [7]. Hierarchical cluster analysis has identified BCa subtypes based on distinct gene expression profiles, including luminal A, luminal B, basal-like, and human epidermal growth factor receptor 2 (HER2) types. ERα-negative tumors constitute 30% of all BCa cases; and of them, triple-negative BCa (TNBC), which lacks ERα, progesterone receptor (PR), and Her2/neu accounts for 5%–20%of all BCa cases and has a poor clinical outcome and aggressive clinical behavior [8].
Currently, the primary treatment for ERα-positive BCa is endocrine therapy with selective ER modulators and aromatase inhibitors based on positive ERα nuclear expression. However, ERα is overexpressed in 60%–70% of BCa, with approximately 50% exhibiting de novo resistance to estrogen modulators [9]. Patients with ERα-negative BCa and TNBC do not benefit from endocrine therapy; however, some patients with TNBCs do respond to standard chemotherapy. Thus, novel treatments are needed for treating ERα-negative BCa and TNBC. As ERβ is highly expressed in ERα-negative BCa and TNBC, various studies have investigated the utility of ERβ as a prognostic and/or predictive marker, and a potential therapeutic target for BCa.
In my previous study [10], I found that high ERβ1 protein expression levels in ERα-negative BCa and TNBC were correlated with poor prognostic biomarkers; and thus ERβ can be considered a potentially significant marker for ERα-negative BCa and TNBC. This review summarizes studies on ERβ expression in BCa conducted during 1997 to 2021. These studies analyzed mRNA and/or protein expression levels of ERβ1, ERβ2 and/or ERβ5 isoforms and their associations with clinical characteristics, prognosis, endocrine therapy responses, and overall survival (OS) in the entire cohort as well as in different subgroups.
DETERMINATION AND EVALUATION METHODS OF THE mRNA AND PROTEIN EXPRESSION OF ERβ ISOFORMS
Most ERβ studies examining ERβ in BCa have focused mainly on ERβ1 protein expression and some have considered on ERβ mRNA expression with or without ERβ protein expression. The study subjects include the entire cohorts of patients with ERα-positive and ERα-negative BCa combined, ERα-positive alone, few ERα-negative BCa, TNBC or different molecular types. The number of subjects varied from 13 to 3,093; ERα-positive BCa with ERα -negative BCa combined cases were studied in 72.3% (47/65), ERα-positive BCa alone in 24.6% (16/65), and TNBC/ERα-negative BCa in 15.4% (10/65) cases, respectively. ERβ mRNA expression in BCa has been more frequently determined from 1997 to the 2000s, whereas ERβ protein expression in more recent years, reflecting the increased commercial availability of ERβ isoform antibodies. A few studies have investigated both ERβ mRNA and protein expression.
ERβ isoform protein expression by immunohistochemistry
The immunohistochemistry (IHC) studies have been conducted using a wide range of commercially available monoclonal and polyclonal ERβ antibodies, or in-house developed antibodies with or without in vivo validation (Tables 1 and 2). The validation methods of IHC involving primary and secondary antibodies, visualization system, equipment and controls are not consistent. Scoring systems also differ substantially from positive or negative, and from qualitative to semi-quantitative approaches by combining the percentage and intensity of positive immunoreactive cells. The cut-off threshold to define ERβ positivity varies significantly and the detection rate of ERβ positivity ranges from 15.9% to 92.0% (Tables 1 and 2). ERβ1 isoform protein expression has been analyzed using different ERβ1 antibodies singly or combined: clone PPG5/10 in 43.5% (20/46), 14C8 in 19.6% (9/46), PA313 in 2.1% (1/46), polyclonal ERβ1 (385p/AR385-10R) in 4.3% (2/46), both polyclonal and in-house-raised antibodies in 30.4% (14/46) of the studies, and in-house developed ERβtotal antibodies in 8.7% (4/46) of the studies. ERβ2 isoform has been analyzed using clone 57/3 in 15.2% (7/46) and other polyclonal ERβ2 antibodies in 21.7% (10/46) of studies. ERβ5 isoform has been investigated using clone 5/25 in 2.1% (1/46) and other ERβ5 antibodies in 6.5% (3/46) of the studies. Among the ERβ1 tested, the 14C8, PA1-313, and PPG5/10 ERβ1 antibodies reportedly yield high and specific detection levels of full-length ERβ, but seem to produce reliable results, only in some studies. Our laboratory studies revealed that clone PPG5/10 from two different vendors displayed inconsistent immune reaction and 14C8 presented lower levels of detection when compared with other ERβ1 antibodies. The polyclonal ERβ1 (385p/AR385-10R) and ERβ5 (5/25) antibodies produced intense nuclear staining as well as cytoplasmic staining. The differences in ERβ isoforms are toward the C-terminus of the molecule, whereas many commercially available ERβ antibodies are targeted against the N-terminus, making it unlikely that they will be able to distinguish wild-type ERβ from splice variant ERβ. Many ERβ isoform protein expression studies reviewed herein simultaneously analyzed more than one ERβ isoforms; ERβ1 was most frequently analyzed in 82.6% (38/46), and ERβ2, ERβ5, and ERβtotal protein expression in 39.1% (18/46), 6.5% (3/46) and 6.5% (3/46) of the studies, respectively.
Table 1. Summary of ERβ isoform protein expression studies in BCas:1997–2009.
| Material | ERα status | Antibody/epitope location | ERβ isoform (% positivity) | High ERβ expression and outcome association with | References |
|---|---|---|---|---|---|
| 442 | ERα+/ERα− | pan-B, PPG5/10 | 1 (15) | Better survival in TAM treated postmenopausal and ERα-BCa and TNBC | Honma et al. [48] |
| ERα+/ERα− | 57/3 | 2 | None | ||
| 141 | ERα+/ERα− | 57/3 | 2 (33) | Better outcome in only ERα+ positive BCa | Vinayagam et al. [15] |
| 150 | ERα+/ERα− | poly ERβ1 | 1 (33) | Increased DFS and OS, small sized, node negative tumor | Sugiura et al. [64] |
| ERα+/ERα− | poly ERβ2 | 2 | High ERβ2 with low grade tumor | ||
| 62 | ERα+/ERα− | poly ERβ | 1 | High ERβ with reduced carcinogenesis or progression | Shaw et al. [18] |
| 936 | ERα+/ERα− | 14c8, PPG5/10 | 1 (11) | Increased DFS and predictive of TAM response in node-negative luminal A type but risk factor in node-positive luminal B type | Novelli et al. [61] |
| 27 | ERα+ | N-terminus | Total | High ERβt protein in TAM sensitive tumors | Murphy et al. [14] |
| 181 | ERα+/ERα− | PPG5/10, C-terminus | 1 (28) | Increased DFS, OS in ERα+ BCa, inverse relation to Her2/neu | Nakopoulou et al. [31] |
| Less favorable DFS and OS in ERα-BCa, increase of TopoIIa and P53 | |||||
| 353 | ERα+/ERα− | 14c8, PPG5/10 | 1 (30) | Increased OS and favorable in TAM treated ERα- BCa | Gruvberger-Saal et al. [51] |
| 145 | ERα+/ERα− | 14c8 | 1 | In ERα+/PR+, high ERb1with reduced fatal outcome; in ERα+/PR−; no association | Maehle et al. [37] |
| 226 | ERα+/ERα− | 385p/AR385 | 1 (67) | High expression of Her2/neu and P53 in ERα - BCa/TNBC | Choi and Pinto [10] |
| 210 | ERα− | 385p, polyclonal, 14C8 N-terminal | 1 (58) | ERβ1/T and high CK5/6, Ki-67, high tumor grade | Skliris et al. [65] |
| 2 (57) | ERβ2 with p-c-Jun and NF-kBp65 in ERα- BCa | ||||
| 138 | ERα+/ERα− | PPG5/10, C-terminus | 1 (46–92) | ERβ1 protein with trend of worse outcome in whole cohort and ERα+ TAM treated BCa; high KI-67 in ERα - BCa | O'Neill et al. [17] |
| ERβ1 mRNA in TAM treated ERα+ BCa with no correlation with outcome but large tumor in ERα - BCa | |||||
| 757 | ERα+/ERα− | 14c8, PPG5/10 | 1 | No correlation with clinical parameters | Shaaban et al. [43] |
| ERα+/ERα− | 57/3 | 2 | ERβ2 nuclear expression with inverse to metastasis, vascular invasion | ||
| ERβ2 cytoplasm expression with poor outcome | |||||
| ERα+/ERα− | ERβ5 (home raised) | 5 | Increased OS with high 65% cut-off points | ||
| 34 | ERα+/ERα− | 06-629, N-terminus | Total | High Ki-67+ proliferating cells, cyclin A+ | Jensen et al. [68] |
| 115 | ERα+ | 14c8, ERβ2 specific Ab | 2 | ERβ2+/PR− with poor response to TAM in ERα + BCa, inverse with PR expression | Saji et al. [39] |
| 36 | ERα+ | ERb1; MCA1974S, ERb2; MCA2279S | 1 | Not predictive of TAM therapy response | Miller et al. [47] |
| 512 | ERα+/ERα− | ERβ1 (EMRO2) | 1 (50) | Low ERβ with decreased DFS in ERα+ BCa and co-expression might predict better response but no correlation with clinical variables or outcomes | Borgquist et al. [30] |
ER = estrogen receptor; TAM = tamoxifen; BCa = breast cancer; TNBC = triple-negative breast cancer; DFS = disease-free survival; OS = overall survival; PR = progesterone receptor.
Table 2. Summary of ERβ protein expression studies in BCas: 2010–2021.
| Material | ERα status | Antibody/epitope location | ERβ isoform (% positivity) | Low or high ERβ isoform expression and association with | References |
|---|---|---|---|---|---|
| 139 | ERα+/ERα− | ERβ1 | 1 (40) | ERβ protein expression with smaller tumor | Kim et al. [16] |
| Branched chain QuantiGene (mRNA) | 1 | High ERβ mRNA with worse DFS, poorly differentiated tumor, LN+, PR− | |||
| 3093 | ERα+/ERα− | PPG5/10 | 1 | Inverse relation to Her2/neu, CK5/6, EGFR | Marotti et al. [24] |
| Higher in luminal type A and B type than HER2 or basal types | |||||
| Large tumor, LN+, high grade tumor with absent ERβ | |||||
| 162 | ERα+/ERα− | PPG5/10 | 1 (30) | Increased DFS in LN+ tumor, smaller tumor | Zhang et al. [26] |
| 57/3 | 2 | Absence ERβ2; small size tumor, good OS | |||
| 78 | ERα+/ERα− | 14c8 | 1 | Better TAM response in ERα + BCa | Madeira et al. [71] |
| 69 | ERα+/ERα− | 14c8, PPG5/10, 57/3 | 1 (79.3) | High ERβt, ERβ1 and ERβ2 in tumors with good chemotherapy responses | Wurster et al. [34] |
| 1026 | ERα+/ERα− | PPG5/10 | 1 | Favorable chemo therapy | Elebro et al. [33] |
| 718 | ERα+ | 1 (57) | DFS benefit; Low ERβ1 on exemestane therapy and high ERβ1 on TAM therapy; No benefit in the whole cohort | Speirs et al. [36] | |
| High ERβ1 with high grade tumor, low ERβ1 with small size tumor | |||||
| 689 | ERα+ | 2 | No difference of DFS and OS | ||
| 123 | ERα+/ERα− | 14C8, PPG5/10 | 1 (62) | ERβ1 nuclear expression; predictive of TAM therapy response | Yan et al. [58] |
| 57/3 | 2 | ERβ2 cytoplasmic expression; poor prognosis | |||
| 177 | ERα+ | PPG5/10 | 1 | ERβ1 nuclear expression with prolonged-recurrence free survival; in ERα expression, antiproliferative, high Ki67 in TNBC | Reese et al. [69] |
| 753 | ERα+/ERα− | TCGA protocol | 1, 2, 5 | In HR+ BCa, high expression of ERβ2 or ERβ5 with an overall better DFS but not OS. ERβ2 with no sig effect in whole cohort | Yan et al. [57] |
| In TNBC, ERβ2 with worse outcome, and upregulation of ERβ1 with increased tumor suppression, ERβ5 with no effect | |||||
| 81 | ERα+ | mB1C1, pB2 | 1, 2 | ERb1 with low tumor size and longer DFS but ERb2 with shorter DFS | Dhimolea et al. [62] |
| 459 | ERα+ | S105-ERb | High S105-ERb expression with better OS and low grade in TAM sensitive tumors but worse survival in TAM resistant tumors | Hamilton-Burke et al. [32] | |
| 492 | ERα+/ERα− | 1 | Both ER-β1 and SRAP could be predictive biomarkers of tamoxifen benefit in ER-α-negative premenopausal early BC | Yan et al. [52] | |
| 571 | TNBC | PPG5/10 | 1 | Prolonged OS, DFS and disease free metastasis in TNBC; ERb1+/pAKT(−) predicted favorable prognosis in TNBC | Wang et al. [53] |
| 689 | ERα+ | 2 (55) | No difference of DFS and OS | ||
| 41 | ERα+ | PPG5/10, M7292 | 1 | High ERβ and high ERα Ser167 phosphorylation with longer PFS | Motomura et al. [38] |
| 120 | ERα+/ERα− | Polyclonal ERβ | Total/1 | High ERβ1 with recurrent tumor and lymphatic metastasis | Chang et al. [67] |
| 195 | ERα+ | Poly pan ERβ | Reduced DFS with endocrine therapy | Guo et al. [40] | |
| 598 | ERα+/ERα− | ERβ | High ERβ with lower median tumor-free survival and no benefit of endocrine therapy | Guo et al. [41] | |
| 234 | TNBC+/TNBC− | Poly ERβ (BY-0201) | 1 (43) | poor disease progression free survival and poor prognosis in TNBC | Guo et al. [42] |
| Poly ERB2 | 2 (70) | Poor DFS with chemotherapy | |||
| 101 | ERα+/ERα− | PPG5/10 | 1 (80) | No significant effect on OS | Baek et al. [44] |
| 57/3 | 2 (50) | Worse DFS and OS | |||
| 60 | ERα+/ERα− | 14C8 | 1 (15.9) | Correlated with ERα + and low grade tumor | Bozkurt et al. [63] |
| 2000 | ERα+/ERα− | PPG5/10 | 1 (50) | Her2/neu + tumor | Wimberly et al. [55] |
| ERα+/ERα− | 5/25 | 5 (50) | ERβ5 expression with worse prognosis in TNBC and Her2/neu positive BCa | ||
| Increased co-expression in ERα + in BCa | |||||
| 19 | ERα− | PPG5/10 | 1 | Worse 5 year survival in AAW, increased IGF-2 with high ERβ1 in TNBC | Hamilton et al. [66] |
| 95 | ERα−, TNBC | ERβ1, C-terminus | 1 (70) | No association with OS | Chantzi et al. [56] |
| Poly ERB2 | 2 (70) | Early relapse and poor DFS in post-menopausal ERα - BCa and TNBC | |||
| 250 | TNBC | PPG5/10 | 1 (59.3) | Lower RFS, DMFS, high IGF2 in TNBC | Austin et al. [59] |
| High ERβ in AAW patients with TNBC | |||||
| 126 | TNBC | PA1-313 | 1 | High KI-67 in a subgroup of TNBC but no association with DFS or OS | Shanle et al. [46] |
ER = estrogen receptor; PR = progesterone receptor; LN = lymph node; EGFR = epidermal growth factor receptor; HER2 = human epidermal growth factor receptor 2; TAM = tamoxifen; AAW = African American women; PFS = progression-free survival; DMFS = distant metastasis -free survival; OS = overall survival; DFS = disease-free survival; TNBC = triple-negative breast cancer; RFS = relapse-free survival.
ERβ mRNA expression study
Reverse transcription-polymerase chain reaction has been mostly performed using fresh tumor tissues [11,12,13,14] as well as by using formalin-fixed paraffin embedded sections (FFPES) [15]. Kim et al. [16] demonstrated method for analyzing ERβ mRNA expression using FFPES by the branched chain QuantiGene2.0 assay. Among the ERβ mRNA expression studies, total ERβ/ERβ1 was most frequently analyzed in 75% (12/19) of the studies, and many studies analyzed more than one ERβ isoform; including ER β2, ERβΔ5, and ERβ5 mRNA in 15.7% (7/19), 10.5% (2/19) and 10.5% (2/19) of the studies, respectively (Table 3). Although ERβ mRNA measurement can provide more accurate determination of ERβ at the molecular level, ERβ mRNA analysis has drawbacks in terms of its routine application; mRNA may not necessarily reflect protein expression and can be degraded to undetectable levels during processing or become contaminated with stromal cells. Thus, the level of ERβ protein expression is not always consistent with that of ERβ mRNA expression [15,17].
Table 3. Summary of ERβ mRNA expression and its association with prognostic markers and clinical characteristics in BCas.
| Materials | ERβ mRNA expression and association with | References |
|---|---|---|
| 62 | Down-regulation of ERβ in BCa in comparison with benign breast tissue | Shaw et al. [18] |
| 66 | Down-regulation of ERβ in BCa; Decrease of ERβ2 is the key reason of ERα+ breast carcinogenesis | Park et al. [35] |
| 18 | Down-regulation of ERβ and upregulation of ERα in ERα+ BCa | Leygue et al. [22] |
| 105 | ERβ2 and ERβ5 with better RFS; ERβ2 with better OS in patients with endocrine therapy | Davies et al. [29] |
| 141* | High ERβ2 mRNA with increased DFS and OS in whole cohort and LN - cases | Vinayagam et al. [15] |
| 150 | Better DFS and OS, ERβ2 mRNA is independent prognostic factor | Sugiura et al. [64] |
| 60 | Low ERβΔ5 splice variant with large tumor in ERα + tumor | Mandusic et al. [25] |
| High ERβ1 and ERβΔ5 splice variant in ERα-/PR-BCa | ||
| 74 | Higher ERβ1 in early onset BCa and ERβ2 in late-onset BCa, ERβ2 > ERβ1 with better DFS in TAM treated late onset BCa | Mandusic et al. [28] |
| 95† | High ERβ with worse DFS and poorly-differentiated BCa | Kim et al. [16] |
| 138 | ERα+ TAM treated cases: no correlation of outcomes | O'Neill et al. [17] |
| ERα−/ERβ+ BCa: high Ki-67, large tumor and high stage BCa | ||
| 121 | Poor outcome in ERα− BCa with chemotherapy | Markey et al. [12] |
| 134 | High ERβ1 and ERβ5 in ERα− BCa in African American women | Poola et al. [20] |
| 43 | ERβ exon 5 splice variant with grade 3 tumor | Poola et al. [11] |
| 17 | TAM-resistant breast tumors | Speirs et al. [19] |
| 53 | Higher ERβ2& 5 > ERβ1; associated with high grade and inflammation | Leygue et al. [45] |
| 32 | High ERβ with worse prognosis; aggressive, high grade, Ki-67, and LN+ tumor | Queslati et al. [70] |
| 24 | High ERβ in ERα-/PR-tumors | Iwao et al. [49] |
| 41 | ERβ mRNA 1, 2 or 5 expression and TAM responses: no correlation | Murphy et al. [14] |
ER = estrogen receptor; BCa = breast cancer; RFS = relapse-free survival; OS = overall survival; DFS = disease-free survival; TAM = tamoxifen; PR = progesterone receptor.
*Quantitative polymerase chain reaction in formalin fixed paraffin-embedded tissues; †Branched chain QunatiGene2.030 in formalin fixed paraffin-embedded tissue.
ERβ ISOFORM EXPRESSION IN NORMAL BREAST TISSUE AND BCas: DOWNREGULATION OF ERβ EXPRESSION IN BCas
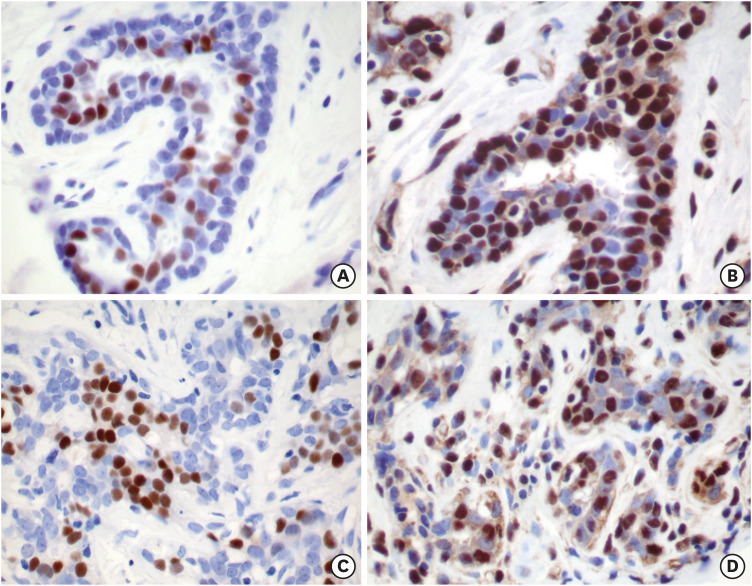
ERβ1, ERβ2, and ERβ5 protein is widely expressed [10,18,19] in the nuclei, and sometimes in the cytoplasm, - of luminal epithelial- and myoepithelial cells, fibroblasts, endothelial cells, and lymphocytes of normal and benign breast tissues, whereas ERα is expressed only in the nuclei of epithelial cells (Figure 1). In BCa, ERβ is expressed in the nuclei of neoplastic epithelial cells and stromal cells, whereas ERα is exclusively expressed in neoplastic epithelial cells. Expression of ERβ predominated in normal breast tissues and is downregulated during progression from pre-invasive neoplastic lesions to invasive BCa compare with that in benign breast tissues, whereas ERα expression is up-regulated in these breast tissues, and most breast tumors expressed ERα, either alone or in combination with ERβ [10,18,19,20,21,22]. The decline or silencing of ERβ is considered a result of DNA methylation [23] and has been considered a key factor in breast carcinogenesis and the independent growth of BCa cells.
Figure 1. Immunohistochemistry stains of ERα and ERβ protein expression in normal and benign breast tissue. ERα is expressed only in epithelial cells (A and C). ERβ expression (B and D) is expressed in benign epithelial cells and myoepithelial cells, stromal cells, and lymphocytes, and ERβ reaction is abundant and stronger than that of ERα (immunohistochemistry stain using polyclonal ERβ1 (385p/AR385-10R) antibody, original magnification ×20).
ER = estrogen receptor.
Absence or low expression of ERβ1 protein is associated with high cell proliferation, large tumor size, high histological grade, and lymph-node positive tumors [24]. A decrease in ERβ1 and ERβΔ5 mRNA levels was associated with larger tumor size in ERα-positive BCa [25]. In contrast, lack of ERβ2 protein expression is associated with good OS in patients with < 2 cm tumor in luminal subtype BCa [26].
DIFFERENTIAL EXPRESSION OF ERβ ISOFORM mRNA AND PROTEIN IN BCas
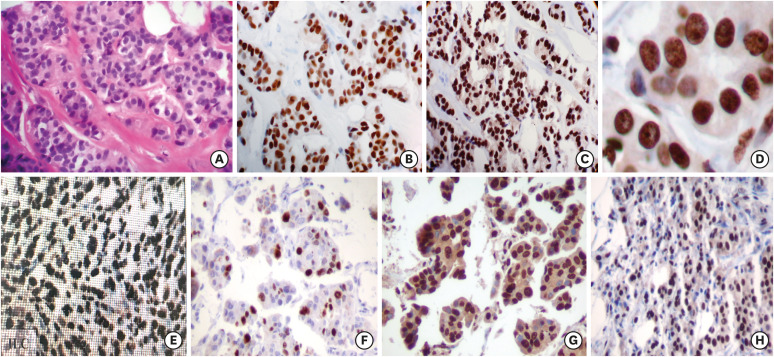
ERβ1 isoform is more frequently expressed than ERβ2 or ERβ5. ERβ isoform protein is differentially expressed in different BCa phenotypes and with clinical characteristics (Figure 2). ERβ1protein expression is higher in well-differentiated BCa than in poorly differentiated BCa, and is higher in ERα-positive BCa than in ERα-negative BCa or TNBC [10,20,22]. In lobular BCa, ERβ1 protein is diffusely expressed, but its expression is lost in late stages of lobular BCa leading to a highly proliferative ERα-positive BCa, whereas in infiltrating duct carcinoma, ERβ is gradually decreased from normal to proliferative and invasive BCa [27]. Further, a specific associations have been observed between the relative ERβ1and ERβ2 mRNA levels normalized with ERα and tumor size; an inverse correlation of tumor size with relative ERβ1 mRNA in 39 cases of early-onset BCa and with relative ERβ2 mRNA levels in 35 cases of late-onset BCa; a higher level of ERβ2 mRNA expression compared with that of ERβ1 was associated with a better outcome in late-onset BCa, indicating that different ERβ isoforms may be involved in BCa suppression of tumor growth in early- and late- onset BCa patients [28]. ERβ2 mRNA expression has been associated with significantly improved relapse-free survival (RFS) and OS, and ERβ5 mRNA has been associated with improved RFS in a subset of patients receiving tamoxifen (TAM), but ERβ1 expression was not associated with disease outcome; these findings suggest that splice variants of ERβ are more important predictors compared to ERβ1 alone [29]. ERβ1 and ERβ5 mRNA is highly expressed in ERα- negative BCa among African-American women [11]. A high level of ERβ exon 5 splice variant is associated with grade III tumors in post-menopausal women [20].
Figure 2. Hematoxylin and eosin stains of infiltrating duct carcinoma of BCa tissues (A) and immunohistochemistry stains of ERα (B) shows positive reaction only in neoplastic epithelial cells, whereas ERβ expression (C) shows strong and diffuse immunoreaction of neoplastic epithelial cells and stromal cells (original magnification ×20), (D) diffuse and intense staining of ERβ expression in nuclei (original magnification ×40), differential expression of ERβ in BCa types, (E) infiltrating lobular carcinoma, (F) apocrine carcinoma, (G) micropapillary carcinoma, (H) mucinous carcinoma (original magnification ×20).
BCa = breast cancer; ER = estrogen receptor.
ERβ ISOFORM EXPRESSION AND CLINICAL OUTCOMES: DIFFERENTIAL ASSOCIATION WITH BREAST CANCER SUBTYPES AND CLINICOPATHOLOGICAL PARAMETERS
Overall, the disease outcomes and clinical characteristics on ERβ expression are differentially associated with ERβ isoforms, their ERβ mRNA or protein expression and BCa subtypes. The disease outcomes or survival observed include both favorable as well as unfavorable prognostic biomarkers, survival outcomes, beneficial and poor responses to endocrine therapy, and inhibition and promotion of tumor growth and carcinogenesis. Thus, ERβ activating and inhibiting ligands have been suggested as a potential therapeutic agent.
ERβ ISOFORM EXPRESSION AND BCa SUBTYPES
ERβ isoform expression in ERα-positive BCa
ERα and ERβ are often co-expressed in breast tumors and form ERα/β heterodimer; ERβ functions as modulator of ERα transcriptional activity and ERβ counteracts the stimulatory effects of ERα through heterodimerization of the two receptors, and ERα/β heterodimers are growth inhibitory in breast cells. Thus, the relative expression level of the two isoforms is a key determinant of cellular responses to agonists and antagonists at the physiological and pathological conditions [2,3]. Owing to increased ERα expression during tumorigenesis and decreased ERβ expression from normal breast through ductal hyperplasia and duct carcinoma in situ to invasive BCa, a significantly higher ERα: ERβ ratio has been observed in BCa compared with normal breast tissue. The ratio between the two ERs in BCa may thus be important in cell proliferation, the activity of diverse signaling pathways, and their responses to ER ligands and endocrine therapy in ERα-positive BCa cells [18,19,20,22,30].
Increased ERβ together with ERα expression in ERα-positive BCa was found to be associated with more favorable disease outcomes and better responses to endocrine therapy, with increased and high expression ERβ expression. High ERβ1 expression in ERα-positive BCa is significantly correlated with a favorable impact to disease-free survival (DFS) and OS in the subgroups of patients with stage 1 and II BCa [31]. ERβ2 and ERβ5 protein expression was inversely correlated with metastasis and vascular invasion, and co-expression of ERβ2 and ERα was associated with improved OS and DFS [21]. Higher ERβtotal protein expression in ERα-positive BCa more frequently observed in TAM-sensitive BCa than TAM-resistant BCa, and was associated with PR-positive tumors, and no disease progression [14]. Low or absent expression of ERβ in ERα-positive BCa was correlated with decreased DFS in ERα-positive BCa patients receiving endocrine treatment, suggesting co-expression of high ERβ with ERα might predict better response to endocrine therapy [30]. ERβ2 mRNA in ERα-positive BCa significantly is correlated with better RFS and OS, and better outcome in node- negative BCa [15]. High ERβ phosphorylated at serine 105-ERβ [32] and ERβ1 protein expression in ERα-positive BCa was prognostically more favorable in chemotherapeutic responses compared to absence of ERβ1 expression [33,34]. Park et al. [35] observed that, among ERβ isoforms, ERβ2 mRNA expression decreased significantly in ERα- positive BCa compared with that in the corresponding normal tissues, and that decreased ERβ2 mRNA was associated with development of ERα-expressing tumors, whereas ERβ5 mRNA expression increased significantly, especially in post-menopausal women with BCa. Speirs et al. [36] reported differential responses to endocrine therapy with different levels of ERβ1 protein expression in ERα-positive BCa; including DFS benefit of exemestane over TAM in the low ERβ1 subgroup, and the DFS benefit of TAM therapy with high ERβ1 expression. There was a reduced risk of fatal outcomes with increasing ERβ levels in patients with ERα- positive /PR- positive BCa [37]. High ERβ1 protein levels with high ERα-Ser 167 phosphorylation was associated with longer DFS [38].
On the contrary, high ERβ2 protein expression in ERα-positive BCa and reduced PR expression was correlated with poor response to TAM [39]. Higher ERβ1 protein expression level was associated with reduced DFS, and correlated with poor prognosis in TAM-treated ERα-positive BCa in post-menopausal women [40] and reduced DFS with endocrine therapy [41] and poor progression-free survival in patients with TNBC [42]. High levels of ERβ2 cytoplasmic expression alone or combined nuclear stains predicted worse OS [43]. Furthermore, high ERβ2 protein expression in patients with endocrine therapy was associated with worse DFS and OS [44] and high ERβ mRNA expression was associated with high grade tumors [16,45]. However, others have reported no significant survival outcomes or TAM therapeutic response in tumors expressing ERβ1 protein [30,35,43,46,47], in tumor expressing ERβ2 protein [36,48], in tumors expressing ERβtotal protein [39], and in tumor expressing high ERβ mRNA expression [14,17]. The findings indicate that each ERβ isoform protein or mRNA expression is associated with different clinical correlation and outcomes in this cohort of BCa.
ERβ expression in ERα-negative BCa and triple negative BCa
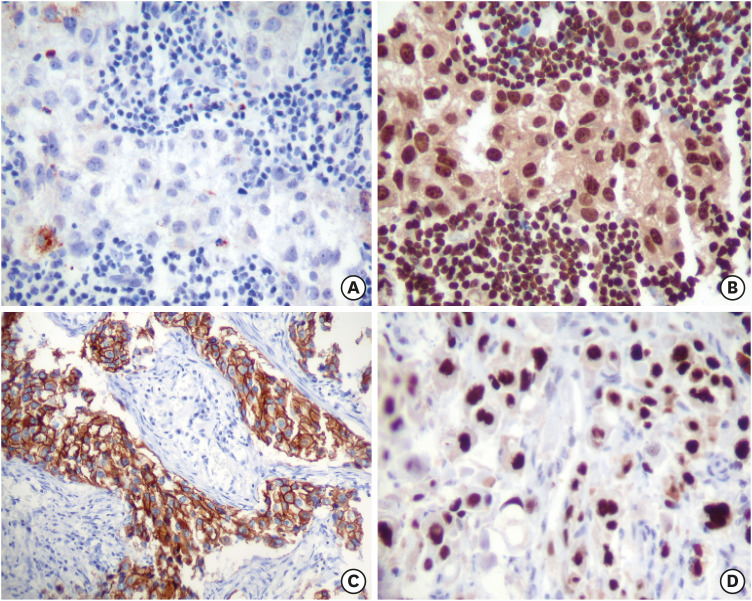
ERβ expression is lower in ERα-negative and TNBC than in benign breast tissues, and that of ERα-positive BCa, however, ERβ is significantly detectable in ERα-negative BCa [49] (Figure 3), TNBC, and HER2 type and basal-like molecular type BCa [24]. ERβ expression in the absence of ERα has been implicated in estrogen -independent growth of BCa and in gene transcription independent of estrogen or growth factors.
Figure 3. Negative ERα staining in ERα-negative BCa (A), high expression of ERβ (B), co-expression of Her-2/neu (C) and P53 (D) of the same ERα- negative BCa cells (original magnification ×20).
ER = estrogen receptor; BCa = breast cancer.
Overall, clinical studies on ERβ expression in ERα-negative BCa and TNBC have reported confounding results with favorable and unfavorable clinical outcomes, and tumor suppression or tumor development [50].
The favorable outcomes included increased OS, DFS and distant metastasis-free survival, as well as beneficial TAM responses in patients with tumors expressing ERβ1 protein [48,51,52,53]. ERβ1 protein expression with negative pAKT predicted favorable prognosis in TNBC [53]. Tan et al. [54] reported a significantly improved DFS and OS in patients with tumors expressing for ERβ1 and ERβ2 protein expression (not ERβ mRNA expression) in ERα- negative BCa. The unfavorable outcomes included poor OS, decreased RFS, high grade, large tumor size and higher-stage tumors, lymph-node positivity or poor prognostic biomarkers. High ERβ1 and ERβ5 protein expression is associated with large tumor size and lymph node-positive BCa, and worse outcome in TNBC [55]. High ERβ1 protein expression was associated with poor disease progression free survival and poor prognosis [42]. ERβ2 protein expression is associated with early disease relapse, poor DFS and cell proliferation in post-menopausal patients with ERα-negative BCa and TNBC [56]. In addition, high ERβ mRNA expression was associated with poor outcome in chemotherapy [12]. ERβ2 mRNA expression was associated with decreased DFS and OS as well as higher stage BCa in TNBC; upregulation of ERβ2 /ERβ5 expression increased cell proliferation, while up-regulation of ERβ1 increased tumor suppression [57]. Cytoplasmic ERβ2 expression without nuclear expression was correlated with shorter survival and poor response to chemotherapy regardless of the cut-off used in familial BCa [58]. ERβ1 activation significantly increased insulin-like growth factor 2 (IGF-2) secretion, and high ERβ1 and IGF2 expression in African-American women and Hispanic patients with TNBC resulted in significantly lower RFS, irrespective of chemotherapy [59]. ERβ5 mRNA expression has been considered to contribute to poor survival observed in ERα-negative BCa among African-American women [11]. These observation could be related to the estrogen–independent transcriptional properties of ERβ5 isoform. Thus, the pro-oncogenic activities of ERβ2 and ERβ5 can be considered the potential therapeutic targets using their specific antagonists, to their pro-oncogenic properties in TNBC, while ERβ1 plausibly acts as a tumor suppressor and its action could be used to limit tumor growth and spread as well as to increase the drug-sensitivities of TNBC [57,60].
ERβ expression in molecular types of BCas
ERβ1 protein expression was significantly related to molecular category of BCa; ERβ1 protein expression was more common in luminal types than basal-like or HER2 molecular types. However, ERβ1 protein was still expressed in 55% of HER2 type and 60% of basal-like cancers, indicating high ERβ expression in ERα-negative BCa or TNBC. ERβ expression in molecular types was inversely associated with Her2/neu, CK5/6 and epidermal growth factor receptor expression [24]. In luminal type BCa, ERβ1 protein expression was significantly associated with good DFS, especially in lymph-node positive BCa, whereas negative ERβ2 protein expression was associated with good OS in patients with < 2 cm tumor [26]. In contrast, ERβ1 expression (55.5%) was evenly distributed across the four molecular BCa subtypes confirming lack of correlation between ER-β and classical prognosticators. Furthermore, ERβ1 was a significant discriminating factor for DFS in node- negative luminal A type BCa, predictive of response to hormonal therapy, but it was a higher risk factor of relapse in node-positive luminal B type BCa. ERβ1 positivity was associated with more aggressive phenotypes such as HER2 type and TNBC or ERα/PR/Bcl2- tumors in node-positive BCa patients [61].
ERβ ISOFORM EXPRESSION AND DIFFERENTIAL CORRELATION WITH CLINICOPATHOLOGICAL PARAMETERS
High ERβ1 protein expression was associated with smaller tumor [26,62] and low grade tumors [63]. High ERβ1 protein expression was associated with small tumor size, lymph-node negative tumor, and low histological grade, and ERβ2 protein expression was correlated with positive ERα status and low histological grade; ERβ1 and ERβ2 mRNA expression correlated with longer DFS [64]. In contrast, high ERβ1 and ERβ5 protein expression is associated with large tumor size and lymph node-positive BCa in TNBC [54]. ERβtotal and ERβ1 protein expression are associated with high expression of CK5/6 and Ki-67, and high-grade tumor, and ERβ2 protein expression levels are associated with p-c-Jun and NF-kBp65 expression in ERα-negative BCa [65]. High ERβ1 protein was associated with increased IGF-2 in TNBC [66]. High ERβ2/cx protein expression [63] and high ERβ1 [67] was associated with aggressive features and presence of lymph vascular invasion and was involved in progression to invasive carcinoma. ERβ1 positivity was associated with more aggressive phenotypes such as HER2 type and TNBC or ERα/PR/Bcl2- tumors in node-positive BCa patients [61]. Many studies demonstrated that high ERβ1 protein expression is correlated with increased Ki-67 > 15% [42,46,68,69]. High ERβ mRNA is correlated with high Ki-67 positivity, large tumors and higher stage BCa in ERα-negative /ERβ-positive BCa [12,17]. Further, co-expression of ERβ and ERα was associated with BCa aggressiveness including higher-grade and positive nodal status, and high Ki-67 positivity [70]. Findings of high proliferative markers of Ki-67 in tumors expressing ERβ isoforms support that ERβ may drive proliferation and involved in carcinogenesis.
POTENTIAL ROLE OF ERβ IN ENDOCRINE THERAPY
As summarized in Table 4, ERβ expression has shown to be associated with good or poor responses to endocrine therapies. High ERβ1 protein expression in patients with ERα-positive or ERα-negative BCa or TNBC tumors is predictive of a good response to TAM therapy [26,36,38,48,53,58,64,71]. ERβ2 mRNA expression is associated with a favorable TAM response in the entire cohort [15] and with significantly improved RFS and OS, and ERβ5 mRNA with improved RFS in a subset of patients receiving TAM [29]. High ERβ2 protein expression is associated with a favorable response in another study [64]. Speirs et al. [36] observed DFS benefit of exemestane in the low ERβ1 subgroup as well as DFS benefit of TAM therapy with high ERβ1 expression compared with the low ERβ1, but no significant difference was observed with treatment for ERß2 expression in either DFS or OS. Both ERβ1 protein and steroid receptor RNA activator protein (SRAP) expression was predictive markers of TAM benefit in ERα-negative BCa in postmenopausal women [52]. High ERβ 1 nuclear expression in tumors from familial BCa was predictive of TAM therapy response [58] and significant discriminating factor for DFS in node- negative luminal A type BCa predictive of response to hormonal therapy [61]. High ERβ protein or mRNA expression with Ki-67 > 15% [12,17,42,46,68,69] was considered that ERβ-dependent elevated Ki-67 level with a high tumor cell proliferation rate might render the cells more sensitive to TAM.
Table 4. Summary of ERβ expression and endocrine therapy responses in BCas.
| Materials(n) | ERα status* | Therapy | Antibody/Epitope location | ERβ isoform expression (% positivity) | ERβ isoform expression and association with | References |
|---|---|---|---|---|---|---|
| 27 | ERα+ | TAM | polyERβ, N-terminus | Total | High expression in TMA sensitive tumor | Murphy et al. [14] |
| 69 | ERα+/ERα− | AI, CHT | 14C8, PPG5/10, 57/3 | 1 (79.3) | Increase ERβt, ERβ1 and ERβ2 in tumors with good chemotherapy responses | Wurster et al. [34] |
| 181 | ERα+/ERα− | TAM, CHT | PPG5/10 | 1 (28) | Increased DFS, OS in ERα+ BC, inverse to Her2/neu positivity | Nakopoulou et al. [31] |
| 150 | ERα+/ERα− | TAM, CHT | Poly clonal ERβ1 and ERβ2 | 1 and 2 (33) | Increased DFS, OS; small size tumor, node-negative tumor and low grade tumor | Sugiura et al. [64] |
| No difference in TAM response with ERβ2 or ERβ1 mRNA expression | ||||||
| 141 | ERα+/ERα− | TAM, AI | 57/3 | 2 (33) | Better outcome only in ERα+ BCa | Vinayagam et al. [15] |
| 115 | ERα+ | TAM | 14c8, ERβ2 specific Ab | 2 | ERβ2+/PR- with poor response to TAM in ERα+ BCa, inverse with PR positivity | Saji et al. [39] |
| 442 | ERα+/ERα− | TAM | pan-B,PPG5/10 | 1 (15) | Better survival in TAM treated postmenopausal ERα-BCa and TNBC | Honma et al. [48] |
| 512 | ERα+/ERα− | TAM | ERβ1 (EMRO2) | 1 (50) | Low ERβ with decreased DFS in ERα+ BCa but no correlation with clinical parameters or outcomes | Borgquist et al. [30] |
| 718 | ERα+ | AI, TAM | PPG5/10 | 1 (57) | Better DFS withn low ERβ1 and exemestane therapy; High ERβ1 with TAM therapy | Speirs et al. [36] |
| 2 | No difference in DFS or OS | |||||
| 689 | ERα+ | TAM, AI | 57/3 | 2 (55) | ERβ2 with no difference of DFS and OS | |
| 162 | ERα+/ERα− | TAM, AI, CHT, RT | PPG5/10 | 1 (30) | Increased DFS in node+ tumor, smaller tumor | Zhang et al. [26] |
| 57/3 | 2 | Absence ERβ2 with OS | ||||
| 757 | ERα+/ERα− | TAM +zoladex | PPG5/10 | 1 | No correlation to clinical outcome | Shaaban et al. [43] |
| ERα+/ERα− | TAM +zoladex | 57/3 | 2 | ERβ2 nuclear+ with Inverse relation to metastasis and vascular invasion | ||
| 2 | ERβ2 Cytoplasam with poor outcome | |||||
| ERα+/ERα− | TAM + zoladex | ERβ5 (home raised) | 5 | Increased OS with high (65%) cut-off positivity | ||
| 936 | ERα+/ERα− | TAM, CHT | 14c8, PPG5/10 | 1 (11) | Increased DFSand TAM response in node-negative luminal A type and risk factor in node-positve luminla B type BCa | Novelli et al. [61] |
| 177 | ERα+ | TAM | PPG5/10 | 1 | ERβ1 nuclear expression with prolonged RFS in TAM treated ERα+ BCa; In ERα-negative BCa, ERβ agonist therapy result in antiproliferative | Reese et al. [69] |
| 753 | ERα+/ERα− | TAM | TCGA protocol | 1, 2, 5 | In HR+ BCa, high expression of ERβ2 or ERβ5 with an overall better DFS but not OS;In TNBC, ERβ2 with worse outcome, and upregulation of ERβ1 with inreased tumor suppression | Yan et al. [57] |
| 81 | ERα+ | AI/TAM, chemo | mB1C1,pB2 | 1, 2 | ERβ1 with low tumor size and longer DFS but ERβ2 with shorter DFS | Dhimolea et al. [62] |
| 459 | ERα+ | TAM | S105-ERb | High S105-ERβ expression with better OS and low grade in TAM-sensitive tumors but worse survival in TAM-resistant tumors | Hamilton-Burke et al. [32] | |
| 492 | ERα+/ERα− | TAM | 1 | Both ER-β1 and SRAP could be predictive biomarkers of tamoxifen benefit in ER-α-negative premenopausal early BC | Yan et al. [52] | |
| 101 | ERα+/ERα− | TAM | PPG5/10 | 1 (80) | No significant effect on OS | Baek et al. [44] |
| 57/3 | 2 (50) | Worse DFS and OS | ||||
| 195 | ERα+ | TAM | Poly pan ERβ | Reduced DFS after endocrine therapy | Guo et al. [40] | |
| 123 | ERα+/ERα− | TAM, CHT | PPG5/10 | 1 (36.9) | High ERβ1 nuclear expression; predictive of TAM therapy | Yan et al. [58] |
| 138 | ERα+/ERα− | TAM | PPG5/10 | 1 (46–92) | ERβ1mRNA with no association of TMA response, but large tumor in ERα- BCa | O'Neill et al. [17] |
| ERβ1 protein with worse outcome in the whole cohort and ERα+ BCa; High Ki-67 in ERα- BCa | ||||||
| 41 | ERα+ | TAM | PCR | 1, 2, 5 | ERβ1, 2 or 5 mRNA expression; no assocation with TAM response | Murphy et al. [14] |
| 27 | ERα+ | TAM | N terminus | Total | High ERβt protein in TAM sensitive tumors | Murphy et al. [14] |
| 36 | ERα+ | TAM | MCA1974S | 1 | Not predictive of TAM therapy responses | Miller et al. [47] |
| MCA2279S | 2 | None |
ER = estrogen receptor; BCa = breast cancer; TAM = tamoxfen; DFS = disease -free survival; RFS = recurrence-free survival; AI = aromatase inhibitor; OS = overall survival; SRAP = steroid receptor RNA activator protein; CHT = chemotherapy; RT = radiation therapy; PR = progesterone receptor.
*ERα expression = ERα + or − BCa
In contrast, ERβ protein expression was indicative of poor response or resistance to TAM therapy in patients with tumors expressing high ERβ1protein expression [17,42,68], tumors expressing high ERβ2 protein [44], and tumors expressing high ERβ2 protein with low PR in a neoadjuvant setting [39] ERβ mRNA expression was significantly up-regulated in the TAM-resistant group as compared with the TAM-sensitive group [13]. Co-expression of ERα and ERβ with over-expression of ERβ lead to the failure of antiestrogen therapy. The anti-estrogen–ERβ complex may inhibit gene transcription when bound to estrogen response elements, and may have agonistic effects resulting in low efficacy of hormonal therapy [19]. In a study by O’Neill et al. [17], ERβ1 mRNA expression showed no association with outcome but ERβ1 protein expression was associated with a trend of worse RFS outcome both within the groups as well as within the ERα–positive TAM treated cases. However, other studies showed no correlation between TMA responses in tumors expressing ERβ1 protein expression [30,39,43,47] or ERβ mRNA expression [14,17,64]. Yamashita et al [72] reported that expression of ERβ1 and ERβcx/β2 did not affect response to therapy.
INCONSISTENT CLINICAL OUTCOMES BETWEEN ERβ mRNA AND ERβ PROTEIN EXPRESSION IN BCas
When ERβ protein and mRNA expression was simultaneously investigated in the same study, the levels of ERβ mRNA expression correlated with ERβ protein levels, only in 34%–54% of cases. Moreover, their clinical outcomes were not always consistent [14,15,16,17,18], although few studies demonstrated consistent findings [64].In a study by Kim et al. [16], ERβ mRNA was associated with worse DFS and poorly-differentiated and lymph node-positive, and PR-negative tumors; ERβ mRNA is thus considered an independent predictor of disease recurrence in ERα-positive BCa, whereas ERβ1 protein was associated with smaller tumor. High ERβ2 mRNA is associated with a favorable TAM response and improved survival in node-negative BCa, ERα-positive BCa and the entire cohort of TAM -treated patients, whereas ERβ2 protein levels are associated with better outcome only in ERα- positive BCa [15]. High ERβtotal protein expression is associated with TAM- sensitive tumors, whereas ERβ mRNA expression of ERβ isoform 1, 2 and/or 5 are not [14]. Such findings have been reported by the same research group in two different studies; in the first study, they reported TAM resistance in tumors expressing high ERβ mRNA [13], and in the second study, they demonstrated better DFS in patients on exemestane therapy with low ERβ1 protein expression as well as better DFS in patients on TAM therapy in tumors expressing with high ERβ1protein expression [36].
When ERβ protein or mRNA is separately investigated in different studies, the clinical outcome findings of ERβ isoform protein or mRNA expression are not consistent. Among reviewed ERβ protein expression studies, 56.5% (26/46) of ERβ isoform expression demonstrated favorable outcomes; ERβ1, ERβ2, and ERβ5 protein expression in 15.2% (7/46), 10.9% (5/46), and 4.3% (2/46), respectively. The unfavorable outcomes were observed with ERβ1, ERβ2, and ERβ5 protein expression in 15.2% (7/46), 4.3% (2/46), and 2.1% (1/46), respectively. Similarly, the studies on ERβ mRNA expression showed favorable outcomes in 47.3% (9/19) of the studies and the unfavorable outcomes in 53.3% (10/19). Thus, the studies on ERβ isoform protein or mRNA expression demonstrated both favorable as well as unfavorable findings.
CONCLUSION
The results of many studies reviewed herein on ERβ mRNA or protein expression in BCa are inconsistent. Thus, the clinical significance of ERβ in BCa remains elusive. The studies reviewed herein showed that ERβ isoform mRNA or protein expression in different BCa subtypes including ERα-negative BCa and TNBC is associated with both favorable or unfavorable clinical outcomes, good or poor clinical parameters and good or poor endocrine therapy responses. Each ERβ isoform seems to have different associations with clinical outcomes and clinical correlation. This may result from the complexities of ERβ isoforms, however, may also related to the lack of standardized testing guidelines for ERβ mRNA or ERβ protein expression, the heterogeneity of study subjects and varying assessment methods. Testing methods for ERβ mRNA or protein expression need to be standardized, and reproducible. Objectively measurable methods to determine ERβ as a prognostic, predictive and therapeutic target are therefore required [73]. Although IHC is the method of choice and routinely and widely applicable for ERβ protein expression analysis, the use of commercially available ERβ antibodies can produce varying results [74]. ERβ protein assays may need to be adopted as per recommended guidelines and strict validation to develop the standardized protocols. The usage of highly selective and specific antibodies is essential to ensure accurate measurement of ERβ isoforms when applied to protein immunoassay as that for ERα [75,76]. Recent studies on quantitative in situ measurement of ERα mRNA in FFPES [77] have provided insights into the potential application of ERβ mRNA analysis in FFPES.
ERβ is generally thought to have anti-proliferative roles in disease progression and is considered to be tumor suppressive in BCa [78]. However, it also exerts proliferative effects with high Ki-67 positivity in the absence of ERα. and may have a potential therapeutic implication in this cohorts. Thus, ERβ is not simply a surrogate marker for ERα but it may affect the growth and proliferation of BCa cells. The ER testing profile of BCa may thus benefit to include ERβ and its variants together with ERα. Further investigation of ERβ isoforms in BCa subtypes, particularly in a large cohort of ERα-negative BCa and TNBC is thus warranted to explore the role of ERβ as prognostic and/or predictive factor and a potential therapeutic target in this cohort of BCa. Furthermore, it remains a challenge to develop strategies for standardizing the widely and routinely applicable analytical methods and protocols for measuring ERβ expression to evaluate the clinical relevance of ERβ isoforms in BCa in depth.
ACKNOWLEDGMENTS
The authors would like to thank the Editage (www.editage.com) for English language editing.
Footnotes
Conflict of Interest: The author declares that they have no competing interests.
References
- 1.Mosselman S, Polman J, Dijkema R. ER β: identification and characterization of a novel human estrogen receptor. FEBS Lett. 1996;392:49–53. doi: 10.1016/0014-5793(96)00782-x. [DOI] [PubMed] [Google Scholar]
- 2.Hall JM, McDonnell DP. The estrogen receptor β-isoform (ERbeta) of the human estrogen receptor modulates ERalpha transcriptional activity and is a key regulator of the cellular response to estrogens and antiestrogens. Endocrinology. 1999;140:5566–5578. doi: 10.1210/endo.140.12.7179. [DOI] [PubMed] [Google Scholar]
- 3.Powell E, Shanle E, Brinkman A, Li J, Keles S, Wisinski KB, et al. Identification of estrogen receptor dimer selective ligands reveals growth-inhibitory effects on cells that co-express ERα and ERβ. PLoS One. 2012;7:e30993. doi: 10.1371/journal.pone.0030993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Leung YK, Mak P, Hassan S, Ho SM. Estrogen receptor (ER)-beta isoforms: a key to understanding ER-beta signaling. Proc Natl Acad Sci U S A. 2006;103:13162–13167. doi: 10.1073/pnas.0605676103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Zhao C, Matthews J, Tujague M, Wan J, Ström A, Toresson G, et al. Estrogen receptor β2 negatively regulates the transactivation of estrogen receptor alpha in human breast cancer cells. Cancer Res. 2007;67:3955–3962. doi: 10.1158/0008-5472.CAN-06-3505. [DOI] [PubMed] [Google Scholar]
- 6.Poola I, Abraham J, Baldwin K, Saunders A, Bhatnagar R. Estrogen receptors beta4 and beta5 are full length functionally distinct ERbeta isoforms: cloning from human ovary and functional characterization. Endocrine. 2005;27:227–238. doi: 10.1385/ENDO:27:3:227. [DOI] [PubMed] [Google Scholar]
- 7.Sørlie T, Perou CM, Tibshirani R, Aas T, Geisler S, Johnsen H, et al. Gene expression patterns of breast carcinomas distinguish tumor subclasses with clinical implications. Proc Natl Acad Sci U S A. 2001;98:10869–10874. doi: 10.1073/pnas.191367098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Foulkes WD, Smith IE, Reis-Filho JS. Triple-negative breast cancer. N Engl J Med. 2010;363:1938–1948. doi: 10.1056/NEJMra1001389. [DOI] [PubMed] [Google Scholar]
- 9.Clarke R, Skaar TC, Bouker KB, Davis N, Lee YR, Welch JN, et al. Molecular and pharmacological aspects of antiestrogen resistance. J Steroid Biochem Mol Biol. 2001;76:71–84. doi: 10.1016/s0960-0760(00)00193-x. [DOI] [PubMed] [Google Scholar]
- 10.Choi Y, Pinto M. Estrogen receptor β in breast cancer: associations between ERbeta, hormonal receptors, and other prognostic biomarkers. Appl Immunohistochem Mol Morphol. 2005;13:19–24. doi: 10.1097/00129039-200503000-00004. [DOI] [PubMed] [Google Scholar]
- 11.Poola I, Fuqua SA, De Witty RL, Abraham J, Marshallack JJ, Liu A. Estrogen receptor α-negative breast cancer tissues express significant levels of estrogen-independent transcription factors, ERbeta1 and ERbeta5: potential molecular targets for chemoprevention. Clin Cancer Res. 2005;11:7579–7585. doi: 10.1158/1078-0432.CCR-05-0728. [DOI] [PubMed] [Google Scholar]
- 12.Markey GC, Cullen R, Diggin P, Hill AD, Mc Dermott EW, O'Higgins NJ, et al. Estrogen receptor-beta mRNA is associated with adverse outcome in patients with breast cancer. Tumour Biol. 2009;30:171–175. doi: 10.1159/000236409. [DOI] [PubMed] [Google Scholar]
- 13.Speirs V, Malone C, Walton DS, Kerin MJ, Atkin SL. Increased expression of estrogen receptor β mRNA in tamoxifen-resistant breast cancer patients. Cancer Res. 1999;59:5421–5424. [PubMed] [Google Scholar]
- 14.Murphy LC, Leygue E, Niu Y, Snell L, Ho SM, Watson PH. Relationship of coregulator and oestrogen receptor isoform expression to de novo tamoxifen resistance in human breast cancer. Br J Cancer. 2002;87:1411–1416. doi: 10.1038/sj.bjc.6600654. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Vinayagam R, Sibson DR, Holcombe C, Aachi V, Davies MP. Association of oestrogen receptor β 2 (ER β 2/ER β cx) with outcome of adjuvant endocrine treatment for primary breast cancer--a retrospective study. BMC Cancer. 2007;7:131. doi: 10.1186/1471-2407-7-131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kim TJ, Lee A, Choi YJ, Song BJ, Yim HW, Kang CS. Prognostic significance of high expression of ER-beta in surgically treated ER-positive breast cancer following endocrine therapy. J Breast Cancer. 2012;15:79–86. doi: 10.4048/jbc.2012.15.1.79. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.O’Neill PA, Davies MP, Shaaban AM, Innes H, Torevell A, Sibson DR, et al. Wild-type oestrogen receptor β (ERbeta1) mRNA and protein expression in Tamoxifen-treated post-menopausal breast cancers. Br J Cancer. 2004;91:1694–1702. doi: 10.1038/sj.bjc.6602183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Shaw JA, Udokang K, Mosquera JM, Chauhan H, Jones JL, Walker RA. Oestrogen receptors alpha and beta differ in normal human breast and breast carcinomas. J Pathol. 2002;198:450–457. doi: 10.1002/path.1230. [DOI] [PubMed] [Google Scholar]
- 19.Speirs V, Parkes AT, Kerin MJ, Walton DS, Carleton PJ, Fox JN, et al. Coexpression of estrogen receptor alpha and beta: poor prognostic factors in human breast cancer? Cancer Res. 1999;59:525–528. [PubMed] [Google Scholar]
- 20.Poola I, Abraham J, Liu A. Estrogen receptor β splice variant mRNAs are differentially altered during breast carcinogenesis. J Steroid Biochem Mol Biol. 2002;82:169–179. doi: 10.1016/s0960-0760(02)00185-1. [DOI] [PubMed] [Google Scholar]
- 21.Shaaban AM, Jarvis C, Moore F, West C, Dodson A, Foster CS. Prognostic significance of estrogen receptor beta in epithelial hyperplasia of usual type with known outcome. Am J Surg Pathol. 2005;29:1593–1599. doi: 10.1097/01.pas.0000184807.38037.75. [DOI] [PubMed] [Google Scholar]
- 22.Leygue E, Dotzlaw H, Watson PH, Murphy LC. Altered estrogen receptor alpha and beta messenger RNA expression during human breast tumorigenesis. Cancer Res. 1998;58:3197–3201. [PubMed] [Google Scholar]
- 23.Zhao C, Lam EW, Sunters A, Enmark E, De Bella MT, Coombes RC, et al. Expression of estrogen receptor β isoforms in normal breast epithelial cells and breast cancer: regulation by methylation. Oncogene. 2003;22:7600–7606. doi: 10.1038/sj.onc.1207100. [DOI] [PubMed] [Google Scholar]
- 24.Marotti JD, Collins LC, Hu R, Tamimi RM. Estrogen receptor-β expression in invasive breast cancer in relation to molecular phenotype: results from the Nurses’ Health Study. Mod Pathol. 2010;23:197–204. doi: 10.1038/modpathol.2009.158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Mandusić V, Nikolić-Vukosavljević D, Tanić N, Kanjer K, Neskovic-Konstantinović Z, Celeketić D, et al. Expression of estrogen receptor beta wt isoform (ERbeta1) and ERbetaDelta5 splice variant mRNAs in sporadic breast cancer. J Cancer Res Clin Oncol. 2007;133:571–579. doi: 10.1007/s00432-007-0209-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Zhang H, Zhang Z, Xuan L, Zheng S, Guo L, Zhan Q, et al. Evaluation of ER-α, ER-B1 and ER-B2 expression and correlation with clinicopathologic factors in invasive luminal subtype breast cancers. Clin Transl Oncol. 2012;14:225–231. doi: 10.1007/s12094-012-0788-0. [DOI] [PubMed] [Google Scholar]
- 27.Huang B, Omoto Y, Iwase H, Yamashita H, Toyama T, Coombes RC, et al. Differential expression of estrogen receptor α, β1, and β2 in lobular and ductal breast cancer. Proc Natl Acad Sci U S A. 2014;111:1933–1938. doi: 10.1073/pnas.1323719111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Mandusic V, Dimitrijevic B, Nikolic-Vukosavljevic D, Neskovic-Konstantinovic Z, Kanjer K, Hamann U. Different associations of estrogen receptor β isoforms, ERβ1 and ERβ2, expression levels with tumor size and survival in early- and late-onset breast cancer. Cancer Lett. 2012;321:73–79. doi: 10.1016/j.canlet.2012.02.022. [DOI] [PubMed] [Google Scholar]
- 29.Davies MP, O’Neill PA, Innes H, Sibson DR, Prime W, Holcombe C, et al. Correlation of mRNA for oestrogen receptor β splice variants ERbeta1, ERbeta2/ERbetacx and ERbeta5 with outcome in endocrine-treated breast cancer. J Mol Endocrinol. 2004;33:773–782. doi: 10.1677/jme.1.01574. [DOI] [PubMed] [Google Scholar]
- 30.Borgquist S, Holm C, Stendahl M, Anagnostaki L, Landberg G, Jirström K. Oestrogen receptors alpha and beta show different associations to clinicopathological parameters and their co-expression might predict a better response to endocrine treatment in breast cancer. J Clin Pathol. 2008;61:197–203. doi: 10.1136/jcp.2006.040378. [DOI] [PubMed] [Google Scholar]
- 31.Nakopoulou L, Lazaris AC, Panayotopoulou EG, Giannopoulou I, Givalos N, Markaki S, et al. The favourable prognostic value of oestrogen receptor β immunohistochemical expression in breast cancer. J Clin Pathol. 2004;57:523–528. doi: 10.1136/jcp.2003.008599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Hamilton-Burke W, Coleman L, Cummings M, Green CA, Holliday DL, Horgan K, et al. Phosphorylation of estrogen receptor β at serine 105 is associated with good prognosis in breast cancer. Am J Pathol. 2010;177:1079–1086. doi: 10.2353/ajpath.2010.090886. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Elebro K, Borgquist S, Rosendahl AH, Markkula A, Simonsson M, Jirström K, et al. High estrogen receptor β expression is prognostic among adjacent chemotherapy-treated patients-results from a population-based breast cancer cohort. Clin Cancer Res. 2017;23:766–777. doi: 10.1158/1078-0432.CCR-16-1095. [DOI] [PubMed] [Google Scholar]
- 34.Wurster M, Ruoff A, Meisner C, Seeger H, Vogel U, Juhasz-Böss I, et al. Evaluation of ERalpha, PR and ERbeta isoforms in neoadjuvant treated breast cancer. Oncol Rep. 2010;24:653–659. [PubMed] [Google Scholar]
- 35.Park BW, Kim KS, Heo MK, Yang WI, Kim SI, Kim JH, et al. The changes of estrogen receptor-β variants expression in breast carcinogenesis: decrease of estrogen receptor-β2 expression is the key event in breast cancer development. J Surg Oncol. 2006;93:504–510. doi: 10.1002/jso.20336. [DOI] [PubMed] [Google Scholar]
- 36.Speirs V, Viale G, Mousa K, Palmieri C, Reed SN, Nicholas H, et al. Prognostic and predictive value of ERβ1 and ERβ2 in the intergroup exemestane study (IES)-first results from PathIES†. Ann Oncol. 2015;26:1890–1897. doi: 10.1093/annonc/mdv242. [DOI] [PubMed] [Google Scholar]
- 37.Maehle BO, Collett K, Tretli S, Akslen LA, Grotmol T. Estrogen receptor β--an independent prognostic marker in estrogen receptor α and progesterone receptor-positive breast cancer? APMIS. 2009;117:644–650. doi: 10.1111/j.1600-0463.2009.02510.x. [DOI] [PubMed] [Google Scholar]
- 38.Motomura K, Ishitobi M, Komoike Y, Koyama H, Nagase H, Inaji H, et al. Expression of estrogen receptor beta and phosphorylation of estrogen receptor alpha serine 167 correlate with progression-free survival in patients with metastatic breast cancer treated with aromatase inhibitors. Oncology. 2010;79:55–61. doi: 10.1159/000319540. [DOI] [PubMed] [Google Scholar]
- 39.Saji S, Omoto Y, Shimizu C, Warner M, Hayashi Y, Horiguchi S, et al. Expression of estrogen receptor (ER) (beta)cx protein in ER(alpha)-positive breast cancer: specific correlation with progesterone receptor. Cancer Res. 2002;62:4849–4853. [PubMed] [Google Scholar]
- 40.Guo L, Zhang YU, Yilamu D, Liu S, Guo C. ERβ overexpression results in endocrine therapy resistance and poor prognosis in postmenopausal ERα-positive breast cancer patients. Oncol Lett. 2016;11:1531–1536. doi: 10.3892/ol.2016.4095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Guo L, Zhang Y, Zhang W, Yilamu D. Correlation between estrogen receptor β expression and the curative effect of endocrine therapy in breast cancer patients. Exp Ther Med. 2014;7:1568–1572. doi: 10.3892/etm.2014.1634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Guo L, Zhu Q, Aisimutuola M, Yilamu D, Liu S, Jakulin A. Expression and prognostic value of estrogen receptor β in patients with triple-negative and triple-positive breast cancer. Exp Ther Med. 2015;9:2147–2150. doi: 10.3892/etm.2015.2380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Shaaban AM, Green AR, Karthik S, Alizadeh Y, Hughes TA, Harkins L, et al. Nuclear and cytoplasmic expression of ERbeta1, ERbeta2, and ERbeta5 identifies distinct prognostic outcome for breast cancer patients. Clin Cancer Res. 2008;14:5228–5235. doi: 10.1158/1078-0432.CCR-07-4528. [DOI] [PubMed] [Google Scholar]
- 44.Baek JM, Chae BJ, Song BJ, Jung SS. The potential role of estrogen receptor β2 in breast cancer. Int J Surg. 2015;14:17–22. doi: 10.1016/j.ijsu.2014.10.007. [DOI] [PubMed] [Google Scholar]
- 45.Leygue E, Dotzlaw H, Watson PH, Murphy LC. Expression of estrogen receptor beta1, beta2, and beta5 messenger RNAs in human breast tissue. Cancer Res. 1999;59:1175–1179. [PubMed] [Google Scholar]
- 46.Shanle EK, Onitilo AA, Huang W, Kim K, Zang C, Engel JM, et al. Prognostic significance of full-length estrogen receptor beta expression in stage I-III triple negative breast cancer. Am J Transl Res. 2015;7:1246–1259. [PMC free article] [PubMed] [Google Scholar]
- 47.Miller WR, Anderson TJ, Dixon JM, Saunders PT. Oestrogen receptor β and neoadjuvant therapy with tamoxifen: prediction of response and effects of treatment. Br J Cancer. 2006;94:1333–1338. doi: 10.1038/sj.bjc.6603082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Honma N, Horii R, Iwase T, Saji S, Younes M, Takubo K, et al. Clinical importance of estrogen receptor-β evaluation in breast cancer patients treated with adjuvant tamoxifen therapy. J Clin Oncol. 2008;26:3727–3734. doi: 10.1200/JCO.2007.14.2968. [DOI] [PubMed] [Google Scholar]
- 49.Iwao K, Miyoshi Y, Egawa C, Ikeda N, Tsukamoto F, Noguchi S. Quantitative analysis of estrogen receptor-alpha and -beta messenger RNA expression in breast carcinoma by real-time polymerase chain reaction. Cancer. 2000;89:1732–1738. doi: 10.1002/1097-0142(20001015)89:8<1732::AID-CNCR13>3.0.CO;2-2. [DOI] [PubMed] [Google Scholar]
- 50.Sellitto A, D’Agostino Y, Alexandrova E, Lamberti J, Pecoraro G, Memoli D, et al. Insights into the role of estrogen receptor β in triple-negative breast cancer. Cancers (Basel) 2020;12:1477. doi: 10.3390/cancers12061477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Gruvberger-Saal SK, Bendahl PO, Saal LH, Laakso M, Hegardt C, Edén P, et al. Estrogen receptor β expression is associated with tamoxifen response in ERalpha-negative breast carcinoma. Clin Cancer Res. 2007;13:1987–1994. doi: 10.1158/1078-0432.CCR-06-1823. [DOI] [PubMed] [Google Scholar]
- 52.Yan Y, Li X, Blanchard A, Bramwell VH, Pritchard KI, Tu D, et al. Expression of both estrogen receptor-beta 1 (ER-β1) and its co-regulator steroid receptor RNA activator protein (SRAP) are predictive for benefit from tamoxifen therapy in patients with estrogen receptor-alpha (ER-α)-negative early breast cancer (EBC) Ann Oncol. 2013;24:1986–1993. doi: 10.1093/annonc/mdt132. [DOI] [PubMed] [Google Scholar]
- 53.Wang J, Zhang C, Chen K, Tang H, Tang J, Song C, et al. ERβ1 inversely correlates with PTEN/PI3K/AKT pathway and predicts a favorable prognosis in triple-negative breast cancer. Breast Cancer Res Treat. 2015;152:255–269. doi: 10.1007/s10549-015-3467-3. [DOI] [PubMed] [Google Scholar]
- 54.Tan W, Li Q, Chen K, Su F, Song E, Gong C. Estrogen receptor beta as a prognostic factor in breast cancer patients: a systematic review and meta-analysis. Oncotarget. 2016;7:10373–10385. doi: 10.18632/oncotarget.7219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Wimberly H, Han G, Pinnaduwage D, Murphy LC, Yang XR, Andrulis IL, et al. ERβ splice variant expression in four large cohorts of human breast cancer patient tumors. Breast Cancer Res Treat. 2014;146:657–667. doi: 10.1007/s10549-014-3050-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Chantzi NI, Tiniakos DG, Palaiologou M, Goutas N, Filippidis T, Vassilaros SD, et al. Estrogen receptor beta 2 is associated with poor prognosis in estrogen receptor alpha-negative breast carcinoma. J Cancer Res Clin Oncol. 2013;139:1489–1498. doi: 10.1007/s00432-013-1467-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Yan S, Dey P, Ziegler Y, Jiao X, Kim SH, Katzenellenbogen JA, et al. Contrasting activities of estrogen receptor beta isoforms in triple negative breast cancer. Breast Cancer Res Treat. 2021;185:281–292. doi: 10.1007/s10549-020-05948-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Yan M, Rayoo M, Takano EA, Fox SB kConFab Investigators. Nuclear and cytoplasmic expressions of ERβ1 and ERβ2 are predictive of response to therapy and alters prognosis in familial breast cancers. Breast Cancer Res Treat. 2011;126:395–405. doi: 10.1007/s10549-010-0941-9. [DOI] [PubMed] [Google Scholar]
- 59.Austin D, Hamilton N, Elshimali Y, Pietras R, Wu Y, Vadgama J. Estrogen receptor-beta is a potential target for triple negative breast cancer treatment. Oncotarget. 2018;9:33912–33930. doi: 10.18632/oncotarget.26089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Monaco A, Licitra F, Di Gisi M, Galasso G, Di Donato M, Giovannelli P, et al. ERβ in triple-negative breast cancer: emerging concepts and therapeutic possibilities. Endocrines. 2021;2:356–365. [Google Scholar]
- 61.Novelli F, Milella M, Melucci E, Di Benedetto A, Sperduti I, Perrone-Donnorso R, et al. A divergent role for estrogen receptor-β in node-positive and node-negative breast cancer classified according to molecular subtypes: an observational prospective study. Breast Cancer Res. 2008;10:R74. doi: 10.1186/bcr2139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Dhimolea E, Tiniakos DG, Chantzi NI, Goutas N, Vassilaros SD, Mitsiou DJ, et al. Estrogen receptors β1 and β2 are associated with distinct responses of estrogen receptor α-positive breast carcinoma to adjuvant endocrine therapy. Cancer Lett. 2015;358:37–42. doi: 10.1016/j.canlet.2014.12.022. [DOI] [PubMed] [Google Scholar]
- 63.Bozkurt KK, Kapucuoğlu N. Investigation of immunohistochemical ERα, ERβ and ERβcx expressions in normal and neoplastic breast tissues. Pathol Res Pract. 2012;208:133–139. doi: 10.1016/j.prp.2011.12.015. [DOI] [PubMed] [Google Scholar]
- 64.Sugiura H, Toyama T, Hara Y, Zhang Z, Kobayashi S, Fujii Y, et al. Expression of estrogen receptor β wild-type and its variant ERbetacx/β2 is correlated with better prognosis in breast cancer. Jpn J Clin Oncol. 2007;37:820–828. doi: 10.1093/jjco/hym114. [DOI] [PubMed] [Google Scholar]
- 65.Skliris GP, Leygue E, Curtis-Snell L, Watson PH, Murphy LC. Expression of oestrogen receptor-β in oestrogen receptor-α negative human breast tumours. Br J Cancer. 2006;95:616–626. doi: 10.1038/sj.bjc.6603295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Hamilton N, Márquez-Garbán D, Mah V, Fernando G, Elshimali Y, Garbán H, et al. Biologic roles of estrogen receptor-β and insulin-like growth factor-2 in triple-negative breast cancer. BioMed Res Int. 2015;2015:925703. doi: 10.1155/2015/925703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Chang J, Liu J, Li H, Li J, Mu Y, Feng B. Expression of ERβ gene in breast carcinoma and the relevance in neoadjuvant therapy. Oncol Lett. 2017;13:1641–1646. doi: 10.3892/ol.2017.5659. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Jensen EV, Cheng G, Palmieri C, Saji S, Mäkelä S, Van Noorden S, et al. Estrogen receptors and proliferation markers in primary and recurrent breast cancer. Proc Natl Acad Sci U S A. 2001;98:15197–15202. doi: 10.1073/pnas.211556298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Reese JM, Suman VJ, Subramaniam M, Wu X, Negron V, Gingery A, et al. ERβ1: characterization, prognosis, and evaluation of treatment strategies in ERα-positive and -negative breast cancer. BMC Cancer. 2014;14:749. doi: 10.1186/1471-2407-14-749. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Oueslati M, Bittaieb I, Sassi N, Jemaa AB, Gamoudi A, Rahal K, et al. ERα and ERβ co-expression: an indicator of aggressive tumors and hormonal sensitivity. Oncol Lett. 2017;14:1675–1682. doi: 10.3892/ol.2017.6314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Madeira M, Mattar A, Logullo AF, Soares FA, Gebrim LH. Estrogen receptor alpha/beta ratio and estrogen receptor beta as predictors of endocrine therapy responsiveness-a randomized neoadjuvant trial comparison between anastrozole and tamoxifen for the treatment of postmenopausal breast cancer. BMC Cancer. 2013;13:425. doi: 10.1186/1471-2407-13-425. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Yamashita H, Nishio M, Kobayashi S, Ando Y, Sugiura H, Zhang Z, et al. Phosphorylation of estrogen receptor α serine 167 is predictive of response to endocrine therapy and increases postrelapse survival in metastatic breast cancer. Breast Cancer Res. 2005;7:R753–R764. doi: 10.1186/bcr1285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Califf RM. Biomarker definitions and their applications. Exp Biol Med (Maywood) 2018;243:213–221. doi: 10.1177/1535370217750088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Nelson AW, Groen AJ, Miller JL, Warren AY, Holmes KA, Tarulli GA, et al. Comprehensive assessment of estrogen receptor β antibodies in cancer cell line models and tissue reveals critical limitations in reagent specificity. Mol Cell Endocrinol. 2017;440:138–150. doi: 10.1016/j.mce.2016.11.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Torlakovic EE, Cheung CC, D’Arrigo C, Dietel M, Francis GD, Gilks CB, et al. Evolution of quality assurance for clinical immunohistochemistry in the era of precision medicine. Part 3: technical validation of immunohistochemistry (IHC) assays in clinical IHC laboratories. Appl Immunohistochem Mol Morphol. 2017;25:151–159. doi: 10.1097/PAI.0000000000000470. [DOI] [PubMed] [Google Scholar]
- 76.Allison KH, Hammond MEH, Dowsett M, McKernin SE, Carey LA, Fitzgibbons PL, et al. Estrogen and progesterone receptor testing in breast cancer: ASCO/CAP guideline update. J Clin Oncol. 2020;38:1346–1366. doi: 10.1200/JCO.19.02309. [DOI] [PubMed] [Google Scholar]
- 77.Thomsen C, Nielsen S, Nielsen BS, Pedersen SH, Vyberg M. Estrogen receptor-α quantification in breast cancer: concordance between immunohistochemical assays and mRNA-in situ hybridization for ESR1 gene. Appl Immunohistochem Mol Morphol. 2020;28:347–353. doi: 10.1097/PAI.0000000000000760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Zhou Y, Liu X. The role of estrogen receptor beta in breast cancer. Biomark Res. 2020;8:39. doi: 10.1186/s40364-020-00223-2. [DOI] [PMC free article] [PubMed] [Google Scholar]