Introduction:
Thrombotic events are potentially devastating complications of coronavirus disease 2019 (COVID-19) infection. Although less common than venous thromboembolism, arterial thrombosis has been reported in COVID-19 cohorts in almost 3% of patients. We describe a patient with COVID-19 infection and concurrent cerebral and noncerebral infarction.
Case Report:
A 53-year-old man with history of COVID-19 pneumonia was admitted to a primary stroke center for speech disturbances and left hemiplegia. Urgent laboratory tests showed a great increase of inflammatory and coagulation parameters as D-dimer, ferritin, interleukin-6 and C-reactive protein. Neuroimaging found occlusion of the M1 segment of the right middle cerebral artery with early signs of ischemic stroke. He received intravenous thrombolysis and mechanical thrombectomy. Abdominal computed tomography discovered a splenic infarction with hemorrhagic transformation and bilateral renal infarction. Urgent angiography showed an associated splenic pseudoaneurysm, which was embolized without complications. He was treated with intermediate-dose anticoagulation (1 mg subcutaneous enoxaparin/kg/24 h), acetylsalicylic acid 100 mg and 5 days of intravenous corticosteroids. In the following days, inflammatory markers decreased so anticoagulant treatment was stopped and acetylsalicylic acid 300 mg was prescribed. His condition improved and he was discharged to a rehabilitation facility on hospital day 30.
Conclusion:
In this case, a patient with multiple thrombotic events in the acute phase of COVID-19 infection, the delimitation of the inflammatory state through analytical markers as D-dimer helped to individualize the antithrombotic treatment (full anticoagulation or anticoagulation at intermediate doses plus antiplatelet treatment as used in our patient) and its duration. However, more data are needed to better understand the mechanisms and treatment of stroke in patients with COVID-19 infection.
Key Words: ischemic stroke, covid-19 infection, thrombolysis, mechanical thrombectomy, splenic infarction, renal infarction
Thrombotic events are potentially devastating complications of coronavirus disease 2019 (COVID-19) infection. Physiopathologic mechanisms remain uncertain but could be related to low-grade disseminated intravascular coagulation and localized pulmonary thrombotic microangiopathy. Although less common than venous thromboembolism, arterial thrombosis in COVID-19 cohorts has been reported in almost 3% of the patients.1 We describe a case of a patient with COVID-19 infection and concurrent cerebral and noncerebral infarction.
CASE REPORT
A 53-year-old man without relevant medical history was admitted to the emergency room on March 31, 2020, with fever, cough, and close contact with his father on March 24, 2020, who died days later of COVID-19 bilateral pneumonia. Vital signs were only remarkable for 37.6°C. Blood pressure, heart rate and respiratory rate were within the normal range, and his oxygen saturation was 95% on room air. Chest X-ray revealed right basal infiltrate suggesting viral pneumonia, urgent laboratory tests showed lymphopenia (580 cells/μL) and elevated C-reactive protein (CRP) (73 mg/L). Procalcitonin test was negative. Severe acute respiratory syndrome coronavirus 2 (SARS-COV-2) polymerase chain reaction (PCR) was also negative. As the SARS-COV-2 PCR was performed on the first day of symptoms, a false positive could not be ruled out, so considering the high prevalence of COVID-19 in the community and the high clinical, radiologic, and epidemiological suspicion he was diagnosed with probable COVID-19 infection. He was instructed to self-quarantine at home and treated with hydroxychloroquine, azithromycin and supportive care with antipyretics.
Sixteen days later, on April 15, 2020, he was admitted to a primary stroke center for speech disturbance and left hemiplegia 2 hours after the onset of symptoms. Initial vital signs revealed a low-grade fever (37.1°C) and decreased oxygen saturation (88% on room air). Neurological examination showed aphasia, left homonymous hemianopia and left hemiplegia, scoring 24 points in the National Institute of Health Stroke Scale (NIHSS). SARS-COV-2 PCR was negative and urgent laboratory tests showed elevated inflammatory and coagulation parameters [CRP 116 mg/L, ferritin 1164 mg/mL, interleukin-6 (IL-6) 21.5 pg/mL, D-dimer 3890 ng/dL]. A noncontrast brain computed tomography (CT) scan found early signs of ischemic stroke in the right frontal operculum, insular cortex, and putamen (Fig. 1A). The CT-angiogram confirmed occlusion of the M1 segment of the right middle cerebral artery (Fig. 1B). Chest CT showed bilateral pneumonia consistent with COVID-19 infection (Fig. 1C) and splenic infarction. He received intravenous thrombolysis and mechanical thrombectomy, achieving thrombolysis in cerebral infarction 3 recanalization 4 hours 45 minutes after the onset of symptoms (Fig. 1D).
FIGURE 1.
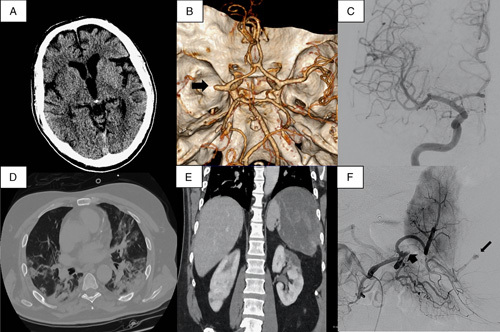
(A) Noncontrast brain computed tomography (CT) on admission. (B) CT-angiogram, black arrow shows right M1 occlusion. (C) Cerebral angiography after mechanical thrombectomy (thrombolysis in cerebral infarction 3 recanalization). (D) Chest CT showing bilateral pneumonia. (E) Abdominal CT with splenic hematoma and bilateral renal infarction. (F) Splenic angiography, short arrow points to splenic occlusion and large arrow points to distal pseudoaneurysm.
On April 18, 2020, COVID-19 enzyme-linked immunosorbent assays blood test showed negative immunoglobulin (Ig)M and positive IgG results, confirming the diagnosis of COVID-19 infection. The patient was treated with intermediate dose of low-molecular weight heparin (LMWH) (1 mg subcutaneous enoxaparin/kg/24 h) and acetylsalicylic acid (ASA) 100 mg/24 hours po. Because of the respiratory symptoms and increased inflammatory parameters, after excluding bacterial infection, intravenous corticosteroids (methylprednisolone) at a dose of 1 mg/kg/24 hours were prescribed for 5 days.
Ten days later, the patient still required high levels of supplemental oxygen therapy. A CT pulmonary angiogram ruled out pulmonary embolism but discovered a hemorrhagic transformation of the splenic infarct and new bilateral renal infarction (Fig. 1E). This was confirmed by an urgent abdominal CT-angiogram, so anticoagulation was stopped for 48 hours. Splenic angiography showed splenic thrombosis and discovered an associated pseudoaneurysm, which was embolized without complications (Fig. 1F). The spleen was partially embolized, so vaccination was not prescribed.
Etiologic work up of stroke was completed. No signs of carotid, intracranial or aortic arch atherosclerosis were found in CT-angiogram. Transthoracic echocardiography with agitated saline contrast excluded patent foramen ovale or other structural cardiopathies and electrocardiogram on arrival and 48 hours electrocardiogram monitoring were negative for atrial fibrillation. Other viral, neoplastic, autoimmune, and hypercoagulability tests were all normal. A previously undetected mild type 2 diabetes was diagnosed and treated.
In the following days oxygen saturation was normalized and oxygen therapy was discontinued. At day 28, his D-dimer level had decreased to 850 ng/dL, and other inflammatory parameters were in decline (CRP 7.8 mg/dL, fibrinogen 541 mg/dL, ferritin 499 ng/dL, IL-6 8.60 pg/dL) with normal renal function (Fig. 2). Thus, subcutaneous enoxaparin treatment was stopped, and ASA 300 mg was prescribed for secondary stroke prevention. On May 13, 2020, at day 30, he was discharged to a rehabilitation facility scoring 8 points in the NIHSS.
FIGURE 2.
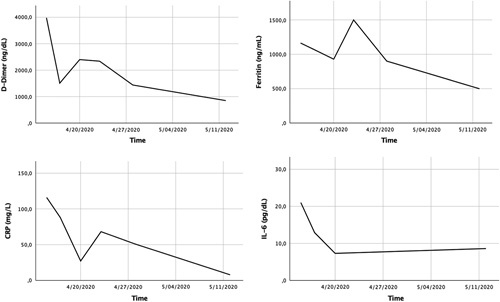
Trend of the inflammatory markers: D-dimer, C-reactive protein (CRP), ferritin and interleukin 6 (IL-6).
DISCUSSION
We present a paradigmatic case of stroke and COVID-19 infection: a patient with no relevant medical history except a moderate-severe COVID-19 infection, elevated inflammation and hypercoagulability markers, and multiple cerebral and noncerebral thrombosis.
COVID-19–related stroke was first reported in 5% of patients with severe infection.2 Later, in larger series, there was a 2.5% of stroke incidence in all hospitalized patients.3 In a recent review of COVID-19 infection and vascular complications, patients hospitalized with COVID-19, only 1.7% suffered myocardial infarction or acute coronary syndrome and 1.2% suffered a stroke.1
Our patient was admitted with stroke on day 16 after the beginning of respiratory symptoms. A classification of COVID-19 stages was proposed4 with 3 different and progressive phases: stage I (mild)-early infection, stage II (moderate)-pulmonary involvement and stage III (severe)-systemic hyperinflammation. Regarding the clinical course, other study suggests an increased inflammatory response during the second week after onset of COVID-19 symptoms affecting the lungs and other organs.5 This inflammatory response could remain during the third week, which explains persistently high inflammatory markers. This data supports the probable hyperinflammatory stage of our patient in his third week of illness when thrombotic events (either venous or arterial) could occur more frequently.
Although SARS-COV-2 PCR was negative upon admission to the Stroke Unit, the epidemiological data (close contact with a COVID-19 patient), clinical time course with persistent fever, typical COVID-19 pneumonia in chest CT concomitant to thrombotic events and the support of SARS-COV-2 IgG antibodies suggest COVID-19 infection was presented at arrival.
The physiopathologic mechanism is not clear, and 2 different mechanisms have been postulated.6 First, a low-grade intravascular disseminated coagulation, related to the increased D-dimer and fibrinogen levels, without significant changes in the number of platelets or prolongation of clotting time parameters, evident primarily in patients with severe disease.7 The other plausible mechanism observed in a series of autopsies is thrombotic microangiopathy in multiple organs, especially in the lungs, possibly mediated by intense complement activation.8,9 Our patient was admitted with highly altered inflammatory parameters, remarkably a D-dimer level of almost 4000 ng/dL; however, he had no other coagulation alterations such as thrombocytopenia or prolongation of prothrombin time.
The optimal specific treatment is not yet known and includes antiplatelets, parenteral or oral anticoagulants, and even a combination of both.10,11 Some studies recommend anticoagulant treatment based on the inflammatory markers, particularly the D-dimer increase.12,13 In April 2020, our hospital COVID-19 protocol recommends use intermediate dose of LMWH (usually subcutaneous enoxaparin) at 1 mg/kg every 24 hours for patients in an inflammatory state (defined by CRP >150 mg/dL, ferritin >1000 ng/dL, IL-6 >40 pg/dL, or D-dimer >3000 ng/dL) as prophylaxis of thromboembolic complications.
This protocol is based in the increased risk of thrombotic events in patients with moderate to severe COVID-19 infection, where prophylactic doses of heparin do not fully protect against vascular complications. In our department, in patients with recent stroke and COVID-19 infection, antithrombotic treatment used depends on the increase of markers of systemic inflammation, presence of other possible etiologies that require specific treatment, stroke severity and the risk of hemorrhagic transformation. Finally, after individualize risk-benefit of antithrombotic treatment, we choose between full doses of anticoagulant treatment or intermediate doses associated with antiplatelet treatment.
In this patient, the underlying stroke etiology was undetermined but probably related to COVID-19-associated coagulopathy. Given the risk of cerebral hemorrhage of full dose anticoagulation in the early stage of a medium-sized infarction we decided to use intermediate doses of subcutaneous LMWH (1 mg/kg/24 h) and ASA 100 mg/24 h. This treatment was maintained as long as the D-dimer level was >1000 ng/dL.
In the case reported here, a patient with multiple thrombotic events in the acute phase of COVID-19 infection, the delimitation of the inflammatory state through analytical markers as D-dimer helped to individualize the antithrombotic treatment and its duration (full anticoagulation or anticoagulation at intermediate doses plus antiplatelet treatment as used in our patient) and secondarily avoid thrombotic recurrences. However, more data are needed to better understand the mechanisms and treatment of stroke in patients with COVID-19 infection.
Footnotes
The authors declare no conflict of interest.
Contributor Information
Ricardo Rigual, Email: rrigualb@gmail.com.
Gerardo Ruiz-Ares, Email: gruizares@gmail.com.
Jorge Rodriguez-Pardo, Email: jorge.rpdd@gmail.com.
Andrés Fernández-Prieto, Email: andresfdezp@gmail.com.
Pedro Navia, Email: pnavia1@gmail.com.
Joan R. Novo, Email: Joan.novo.torres@gmail.com.
María Alonso de Leciñana, Email: malecinanacases@salud.madrid.org.
Pablo Alonso-Singer, Email: palonsosinger@gmail.com.
Blanca Fuentes, Email: blfuentesg@hotmail.com.
Exuperio Díez-Tejedor, Email: exuperio.diez@salud.madrid.org.
REFERENCES
- 1.Pellicori P, Doolub G, Wong CM, et al. COVID-19 and its cardiovascular effects: a systematic review of prevalence studies. Cochrane Database Syst Rev. 2021;3:CD013879. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Mao L, Jin H, Wang M, et al. Neurologic manifestations of hospitalized patients with coronavirus disease 2019 in Wuhan, China. JAMA Neurol. 2020;77:683–690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Lodigiani C, Iapichino G, Carenzo L, et al. Venous and arterial thromboembolic complications in COVID-19 patients admitted to an academic hospital in Milan, Italy. Thromb Res. 2020;191:9–14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Siddiqi HK, Mehra MR. COVID-19 illness in native and immunosuppressed states: a clinical–therapeutic staging proposal. J Heart Lung Transplant. 2020;39:405–407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Lin Y, Wu Y, Zhong P, et al. A clinical staging proposal of the disease course over time in non-severe patients with coronavirus disease 2019. Sci Rep. 2021;11:10681. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Levi M, Thachil J, Iba T, et al. Coagulation abnormalities and thrombosis in patients with COVID-19. Lancet Haematol. 2020;7:e438–e440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Iadecola C, Anrather J, Kamel H. Effects of COVID-19 on the nervous system. Cell. 2020;183:16.e1–27.e1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Magro C, Mulvey JJ, Berlin D, et al. Complement associated microvascular injury and thrombosis in the pathogenesis of severe COVID-19 infection: a report of five cases. Transl Res. 2020;220:1–13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Piazza G, Morrow DA. Diagnosis, management, and pathophysiology of arterial and venous thrombosis in covid-19. JAMA. 2020;324:2548–2549. [DOI] [PubMed] [Google Scholar]
- 10.Wang Z, Yang Y, Liang X, et al. COVID-19 associated ischemic stroke and hemorrhagic stroke: incidence, potential pathological mechanism, and management. Front Neurol. 2020;11:571996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Moroni F, Baldetti L. COVID-19 and arterial thrombosis: a potentially fatal combination. Int J Cardiol. 2021;322:286–290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Violi F, Pastori D, Cangemi R, et al. Hypercoagulation and antithrombotic treatment in coronavirus 2019: a new challenge. Thromb Haemost. 2020;120:949–956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Connors JM, Levy JH. COVID-19 and its implications for thrombosis and anticoagulation. Blood. 2020;135:2033–2040. [DOI] [PMC free article] [PubMed] [Google Scholar]


