Abstract
Acute pancreatitis (AP) is a common clinical abdominal emergency, with a high and increasing incidence each year. Severe AP can easily cause systemic inflammatory response syndrome, multiple organ dysfunction and other complications, leading to higher hospitalization rates and mortality. Currently, there is no specific treatment for AP. Thus, we still need to understand the exact AP pathogenesis to effectively cure AP. With the rise of transcriptomics, RNA molecules, such as microRNAs (miRNAs) transcribed from nonprotein-coding regions of biological genomes, have been found to be of great significance in the regulation of gene expression and to be involved in the occurrence and development of many diseases. Increasing evidence has shown that miRNAs, as regulatory RNAs, can regulate pancreatic acinar necrosis and apoptosis and local and systemic inflammation and play an important role in the development and thus potentially the diagnosis and treatment of AP. Therefore, here, the current research on the relationship between miRNAs and AP is reviewed.
Keywords: microRNAs, Acute pancreatitis, Pathogenic mechanism, Molecular markers, Treatment
Introduction
Acute pancreatitis (AP) is a very common acute disease of the digestive system [1] that is characterized by necrosis of pancreatic acinar cells and local and systemic inflammatory reactions [2, 3]. The incidence of AP is 13–45 per 100,000 people and is increasing [4]. The total mortality rate of AP is approximately 5%. When AP develops into severe acute pancreatitis (SAP), its mortality rate is as high as 20–40%, which seriously endangers people's lives and health [5–7]. Early AP can be cured by combination therapies, such as analgesia, nutritional support and protease inhibitors. However, without timely intervention, rapid development of AP will lead to SAP resulting in serious complications and even systemic multiple organ failure, which endangers the patient’s life. Because of the limited efficacy of conventional therapies and the lack of effective targets for treatment of AP, the prognosis of patients is often poor [8]. With the rapid development of high-throughput sequencing technology, researchers have found that microRNA (miRNA) intervention can change related physiological functions, causing inflammation cell infiltration, autoimmune diseases, cancer and other diseases [9, 10]. The role of miRNAs in inflammation provides a new direction for the treatment of AP. Targeting miRNAs to influence the progression and treatment of AP is a current research hotspot. To this end, this review discusses the research on miRNA mechanisms of action in AP and the potential use of miRNA for AP treatment, providing a theoretical reference for AP diagnosis, prognosis evaluation and targeted therapy.
miRNAs and inflammation
As small noncoding RNAs, miRNAs are approximately 19–25 nucleotides in length and plays an important regulatory role in epigenetics. Mature sequences are mostly located in the introns, exons or pre-mRNA introns of noncoding RNA [11]. By targeting the 3'UTR of target genes, miRNAs control the mRNA translation process or accelerate the degradation of mRNA, ultimately regulating the expression of target genes [12]. Studies have shown that a single miRNA regulates multiple signalling pathways in the human body by targeting different mRNAs, including phosphatase and tensin homologue (PTEN), nuclear factor kappa-B (NF-κB), wingless/β-catenin (Wnt/β-catenin) and Janus kinase/signal transducer and activator of trans (JAK/STAT). miRNAs are widely involved in various cell activities in organisms, including cell development, differentiation, metabolism and apoptosis [11], and plays an important role in the occurrence and development of many diseases, such as inflammation, kidney injury and tumours [13].
Studies have found that inflammation is involved in the occurrence and development of many diseases in the human body, such as liver cirrhosis, pancreatic cancer, diabetes and rheumatoid arthritis [14–16]. Inflammatory cells and inflammatory factors are involved in the occurrence and development of inflammation, but the relevant molecular mechanisms regulating inflammation are still unclear. With the understanding of miRNA function, miRNAs have been found to play an important role in the production of inflammatory factors and inflammatory cells. For example, Let-7adf promotes the inflammatory response, metabolic activity, and interleukin (IL)-6 production by M1-type macrophages by regulating Tet methylcytosine dioxygenase 2/the deubiquitinating enzyme A20 (TET2/A20) [17, 18]. miRNA-93 can mediate the Toll-like receptor 4 (TLR4)/NF-κB signalling pathway to reduce the production of the inflammatory factors tumour necrosis factor-α (TNF-α), IL-1β and IL-6; reduce inflammation; and improve cell apoptosis [19]. In addition, some miRNAs play proinflammatory roles, such as miR-34a [21], miR-27a [22], miR-200a [23], miR-495-3p [24] and miR-124-3p [25], while others exert anti-inflammatory effects, such as miR-21 [20], miR-138 [26], miR-342-3p [27], miR-873a-5p [28], miR-146a [29], miR-542-3p [30], miR-193b-3p [31], miR-140-5p [32] and miR-27a-3p [33]. A summary of studies of miRNAs that affect the production of inflammatory cells and inflammatory factors is shown in Table 1. The involvement of miRNAs in inflammation-mediated processes may allow intervention in the progression of inflammatory diseases and effectively improve the prognosis of patients. The unique mechanism of miRNAs in inflammation is expected to provide novel therapeutic targets for rapidly developing inflammatory diseases, such as AP.
Table 1.
Summary of studies of miRNAs that affect the production of inflammatory cells and inflammatory factors
| miRNAs | Expression | Targets | Effect on inflammation | Inflammation-related cells or factors | References |
|---|---|---|---|---|---|
| miR-let-7a/let-7d/let-7f | Up | Tet2, Lin28a/Sdha axis | Promotion | Macrophages, IL-6 | [17] |
| microRNA let-7 | Up | A20 | Promotion | Macrophages, TNF, IL-1β | [18] |
| miR-93 | Up | TLR4/NF-κB | Suppression | TNF-α, IL-6, IL-1β | [19] |
| miR-21 | Down | PDCD4/NF-κB | Suppression | Macrophages, TNF-α、IL-6 | [20] |
| miR-34a | Up | KLF4 | Promotion | Macrophages, TNF-α, IL-6, IL-1β, and MCP-1 | [21] |
| miR-27a | Down | TLR4/MyD88/NF-κB | Promotion | TNF-α, IL-6, IL-1β | [22] |
| miR-200a | Down | Keap1/Nrf2 | Promotion | TNF-α, L-1β | [23] |
| miR-495-3p | Up | IL5RA | Promotion | TNF-α | [24] |
| miR-124- 3p | Down | p65 | Promotion | TNF-α, IL-6, IL-1β | [25] |
| miR-138 | Up | VEGF/NF-κB | Suppression | TNF-α, IL-1β, IL-6 and IL-18 | [26] |
| miR-342-3p | Up | Rictor | Suppression |
Foxp3+ Regulatory T cells IL-17、IFN-γ and TNF-α |
[27] |
| miR-873a-5p | Up | NF-κB | Suppression | TNF-α, IL-1β, INOS and IL-6 | [28] |
| miR-146a | Up | TLR4/NF-κB | Suppression | TNF-α, IL-6, IL-8 and IL-1 β | [29] |
| miR-542-3p | Up | TLR4 | Suppression | TNF-α, IL-6 and MCP-1 | [30] |
| miR-193b-3p | Up | HDAC3/NF-κB p65 | Suppression | IL-1β, IL-6 and TNF-α | [31] |
| miR-140-5p | Up | HMGB1/PI3K/AKT | Suppression | TNF-α, IL-6, MMP1 and MMP3 | [32] |
| miR-27a-3p | Up | FOXO3/NAPDH/ROS | Suppression | IL-6, IL-8 | [33] |
“Promotion” indicates that miRNA upregulation or downregulation can promote AP progression. “Suppression” indicates that miRNA upregulation or downregulation can suppress AP progression
miRNAs and their role in AP progression
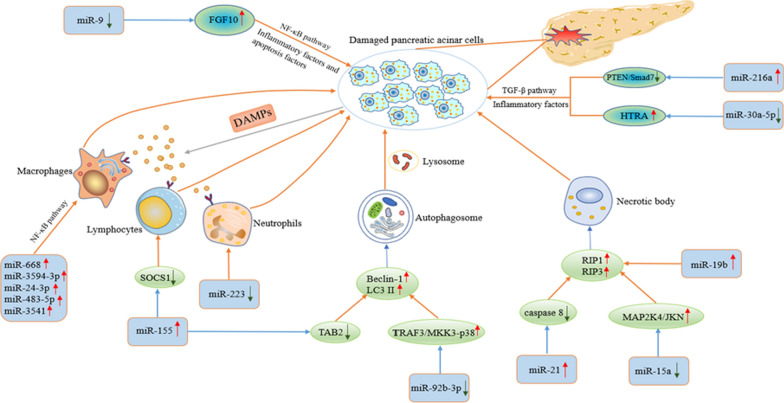
Although most AP patients are mildly ill and the illness is self-limiting, at least 20–30% of AP patients develop SAP within a short period, and better treatments do not exist, ultimately leading to a poor prognosis [8, 34]. Studies have shown that intervention of miRNA-mediated signalling pathways reduces the inflammatory response in AP and the apoptosis of pancreatic acinar cells, which affects the process of AP [35, 36]. This section introduces the research progress in miRNA-related mechanisms in AP. The mechanisms by which miRNAs regulate AP are shown in Fig. 1.
Fig. 1.
Mechanisms by which miRNAs regulate AP. The figure fully shows the relevant mechanisms by which miRNAs regulate AP, including regulation of inflammatory factors and inflammatory cells through related signal pathways, regulation of AP-related autophagy or necrosis, and promotion of immune cell infiltration and differentiation. These processes ultimately promote damage to pancreatic acinar cells and AP progression
miRNA/NF-κB and AP
As a stimulating transcription factor, NF-κB regulates immunity, inflammation and other processes that affect cell growth, differentiation and apoptosis, and plays an important role in inflammatory diseases and cancer [37, 38]. Studies have found that NF-κB aggravates AP progression by promoting the transcription of inflammatory cytokines [39]. It is known that miR-9 as a regulator can promote or suppress tumour progression, such as in liver cancer and pancreatic cancer [40, 41]. Previous studies have shown that miR-9 can affect the process of inflammation by regulating NF-κB. In caerulein-treated AR42J cells, the expression of miR-9 decreased, and the levels of the inflammatory factors IL-1β, IL-6 and TNF-α in the cells increased, while Bax and cleaved caspase 3 and 9, which are related to apoptosis, were also upregulated. Overexpression of miR-9 caused a reduction in the levels of the above factors and reduced the inflammation and apoptosis of AR42J cells. Through bioinformatics and dual-luciferase analysis, it was found that miR-9 affected the expression of related inflammatory factors and apoptotic factors by targeting fibroblast growth factor 10 (FGF10) to regulate the NF-κB pathway, thereby weakening the process of caerulein-treated AP [35]. Macrophages related to inflammation can produce various inflammatory mediators and cytokines, such as IL-6, IL-8 and TNF-α, causing damage to local tissues and even organs. The activation of macrophages may play a vital role in the occurrence and development of AP. Previous studies have found that the taurine-induced AP cell model activates the NF-κB and p38 MAP kinase (p38 MAPK) signalling pathways [42]. In this experiment, the supernatant of taurolithocholate (TLC)-treated AR42J cells significantly activated the NF-κB activation level in macrophages. Further studies have shown that miRNAs carried by exosomes or other vesicles are closely related to the activation of macrophages. Through relevant bioinformatics and qRT-PCR analysis, it has been found that differentially expressed miRNAs mainly participate in the activation of macrophages through the TNF receptor-associated factor 6-TGF-beta activated kinase 1/MAP3K7 binding protein 2-transforming growth factor beta activated kinase 1-NF-κB inducing kinase/IκB kinase (TRAF6-TAB2-TAK1-NIK/IKK)-NF-κB pathway, including miR-668, miR-3594-3p, miR-24-3p, miR-483-5p, and miR-3541, but further experiments are needed to verify this hypothesis [43]. In short, targeting miRNA/NF-κB may be able to effectively prevent the process of AP, especially in combination with macrophages and other inflammatory cells, and can provide a new approach for treatment of SAP with systemic inflammatory response syndrome, which merits further research in the future.
miRNA/TGF-β and AP
Due to the prominent role of TGF-β in inflammation, the immune response, and cell differentiation and proliferation, researchers have begun to extensively study its relationship with AP. Previous studies have reported that inhibiting the expression of TGF-β in an AP mouse model effectively alleviated the progression of AP, providing a theoretical basis for subsequent research [44]. It is known that miR-216a is highly expressed in the pancreas. To explore the specific mechanism of TGF-β in AP, Zhang et al. established a caerulein-induced AP mouse model and found that TGF-β expression was upregulated. After administration of TGF-β inhibitors, the levels of serum amylase, lipase, and the proinflammatory factors TNF-α and IL-6 were significantly lower than levels in the untreated group. Then, different concentrations of TGF-β were used to treat AR42J cells. RT-PCR analysis confirmed that TGF-β increased the expression of miR-216a in a dose-dependent manner, and online prediction tools (Targetscan 5.1, miRanda and PicTar) were used to predict the downstream target genes of miRNA. Further experiments have verified that miR-216a activates the PI3K/AKT and TGF-β signalling pathways through targeted regulation of PTEN and Smad7, and promotes the progression of AP [36]. For a long time, emodin has been widely used in the treatment of AP in China, but its exact mechanism and the target of drug action have not been elucidated. Previous studies have found that high-temperature requirement A (HTRA) can effectively prevent TGF-β1 from becoming mature. The inflammatory signal mediated by HTRA/TGF-β1 may be involved in the process by which pancreatic acinar cells are damaged [45, 46]. Xiang et al. conducted in vivo and in vitro experiments to verify that emodin reduces the number of sodium taurocholate (STC)-treated AP trypsin-positive cells, the release of amylase and the expression levels of the inflammatory mediators TNF-α, IL-6 and IL-1β, and these effects were realized through regulation of HTRA/TGF-β1 signalling pathway. Subsequently, bioinformatics analysis revealed miR-30a-5p as the upstream regulatory molecule of this signalling pathway, and that was verified by experiments such as the dual-luciferase assays detection report [47]. These studies may provide new directions for wide application of emodin and the treatment of AP.
miRNA/inflammatory cells and AP
Damaged pancreatic acinar cells release inflammatory signals through damage-related molecular patterns (DAMPs) and produce a large number of chemokines, adhesion molecules and cytokines, thereby activating and recruiting a large number of lymphocytes, macrophages and neutrophils. The migration of inflammatory cells to damaged regions further aggravates the inflammatory response [48, 49]. In this process, miRNAs play an important role. Imbalance of the IL-17-producing CD4+ T helper (Th17)/regulatory T (Treg) ratio is related to various autoimmune and inflammatory diseases [50]. An increase in this ratio induces an increase in a large number of cytokines and aggravates the progression of AP. Damage to the pancreas can lead to the accumulation of Th17 cells, forming a vicious cycle. Studies have found that miR-155 can induce inflammatory cells to produce TLR signals and promote systemic inflammation. To further explore the relationship between miR-155 and Th17/Treg ratio, Wang et al. isolated CD4+ T from AP patients, overexpressed miR-155, using flow cytometry found a significant increase in the percentage of IL-17+ cells; moreover, downregulation of suppressor of cytokine signalling 1(SOCS1) expression was confirmed via WB, indicating that miR-155 promotes the production of Th17 cells and inhibits the expression of SOCS1. Subsequent use of TargetScan software and dual-luciferase assay verified that SOCS1 is a direct target gene of miR-155 regulates the production of Th17 cells, and in vivo experiments verified the above conclusions. Therefore, targeting miR-155/SOCS1 can effectively interfere with the inflammatory response in AP [51]. In addition, Song et al. found that miR-361-5p can promote Th17 cells to secrete IL-17A and aggravate AP by targeting nuclear factor IA (NFIA) and hes family bHLH transcription factor 1 (Hes1), and these results further deepen the connection between Th17 cells and AP [52]. As previously reported, the activation of macrophages in AP can be mediated by the NF-κB pathway signalling pathway and that the miRNAs involved in this process include miR-668 and miR-3594-3p [43]. At present, there are few studies on the relationship between neutrophils and AP. Dey et al. demonstrated that miR-29a/b1 deletion aggravates pancreatic injury and impairs pancreatic regeneration in AP mice, a result that is consistent with the finding that miR-29a/b1 deficiency causes massive infiltration and activation of inflammatory cells such as neutrophils, and promotes the production of cytokines, such as IL-6, IL-10 and TGFβ1. TGFβ1-mediated pancreatic fibrosis is closely related, but the specific regulatory mechanism still needs to be studied in-depth [53]. The connection between miRNAs and inflammatory cells may effectively interfere with the vicious cycle in AP.
miRNA/autophagy and AP
As a mechanism to protect cells, autophagy can remove damaged, ageing and nonfunctional organelles or macromolecules, and provide energy for cell growth and proliferation. However, autophagy impairment is closely related to several diseases, such as inflammation, neurodegenerative diseases and tumours [54, 55]. Studies have shown that impaired autophagy is involved in the overactivation of acinar cell trypsinogen, abnormal function of organelles and activation of inflammation in AP, but the specific mechanism remains unclear [56, 57]. Due to the prominent role of miRNAs in AP, it is important to understand whether the pathogenesis of AP involves regulation of the autophagy process. Researchers have observed the effect of miR-155 on AP by injecting AVV-miR-155 and AVV-miR-155 sponges into a caerulein-induced AP mouse model. The results showed that MAP3K7 binding protein 2 (TAB2) expression in pancreatic tissue reduced after the injection of AVV-miR-155, which was in contrast to the results in the AVV-miR-155 sponge group, indicating that miR-155 regulates the expression of TAB2. The increase in TAB2 expression inhibited the increase in Beclin-1 levels and hindered autophagosome formation, while overexpression of miR-155 increased Beclin-1 expression, causing excessive accumulation of p62 and vacuolization in the cytoplasm (increase in microtubule-associated protein light chain 3 (LC3 II) levels), ultimately worsening the degree of autophagy impairment and promoting AP progression. This result was verified in an established SAP mouse model, and knocking down miR-155 significantly reduced the pathological damage to the pancreas and lungs in SAP mice [58]. In recent years, it has been discovered that miR-92b-3p, as a regulatory RNA, participates in a variety of cellular behaviour, including proliferation, migration, apoptosis and autophagy. Sun et al. found that during the formation of autophagosomes, miR-92b-3p inhibits AP autophagy by targeting tumour necrosis factor receptor-associated factor-3 (TRAF3) to regulate the phosphorylated mitogen-activated protein kinase kinase 3 (MKK3)-p38 signalling pathway. In caerulein-treated AR42J cells, the expression level of miR-92b-3p decreased, while the levels of Beclin-1 and LC3 II, which are related to the formation of TRAF3, increased. Knockdown of overexpressed miR-92b-3p or TRAF3 caused a decrease in the expression levels of Beclin-1 and LC3 II. A dual-luciferase assay revealed that miR-92b-3p affects protein translation by binding to the 3'UTR of TRAF3 mRNA and affects the expression of downstream genes. Then relevant molecular biology techniques verified that miR-92b-3p affects the formation of AP autophagosome-related proteins by regulating the TRAF3/MKK3-p38 signalling pathway and inhibits the progression of AP [59]. The regulatory relationship between miRNAs and autophagy provides a theoretical basis for elucidating the pathogenesis of AP and developing therapies targeting autophagy. However further research is still needed.
miRNA/necrosis and AP
In contrast to apoptosis, necrosis depends on the participation of receptor-interacting protein kinase 1 (RIP1/RIPK1) and receptor-interacting protein 3 (RIP3) and is related to many pathological conditions such as AP, ischaemia–reperfusion injury and neuropathy [60, 61]. Previous studies have found that necrosis is mainly manifested in the high-dose caerulein-induced AP mouse model, but apoptosis is rarely observed. The degree of necrosis is closely related to the severity of pancreatic injury [62]. To study whether oncogenic miR-21 can promote the occurrence of necrosis, researchers established an AP WT mouse model and obtained corresponding pancreatic tissue sections. The results showed that WT mice showed more obvious pancreatic oedema and acinar cell necrosis than the miR-21 konckdown group. The number of CD11b positive cells in the knockdown group was reduced, which significantly affected the infiltration of monocytes and macrophages in the pancreas, and ultimately reduced the severity of AP. Immunofluorescence experiments also confirmed this conclusion. In addition, silencing of miR-21 protects mice from TNF-α-induced systemic inflammatory response syndrome (SIRS), and this process involves silencing of miR-21 to increase the activity of caspase 8, and then downregulating the expression of RIP1/RIP3 to inhibit the formation of microsomes [63]. Hu et al. established an acute necrotizing pancreatitis (ANP) SD rat model. Using miRNA chips and RT-PCR, the expression of miR-19b in ANP was found to be upregulated. This result is consistent with that measured in taurolithocholic acid 3-sulfate disodium salt (TLC-S)-treated AR42J cells. The expression level of miR-19b is positively correlated with the necrosis rate of pancreatic acinar cells, thereby affecting the progression of AP [64]. In another study, baicalin, which has a tumour suppressor effect, significantly reduced the degree of necrosis in AP. This process is achieved through miR-15a targeting of the mitogen-activated protein kinase kinase 4 (MAP2K4)/c-Jun N-terminal kinase (JKN) signalling pathway [65]. The regulatory link between miRNA and necrosis is expected to provide a new therapeutic target for AP. A summary of studies of related miRNAs and their functional roles in the AP process is shown in Table 2.
Table 2.
Summary of studies of related miRNAs and their functional roles in the AP process
| miRNA | Target | Functional role | In vitro/in vivo | References |
|---|---|---|---|---|
| miR-9 | FGF10/NF-κB | Inhibits expression of the inflammatory factors IL-1β, IL-6 and TNF-α, as well as the apoptosis factors Bax and cl-caspase 3/9 | In vitro | [35] |
| miR-668, miR-3594-3p, miR-24-3p, miR-483-5p, miR-3541 | TRAF6-TAB2-TAK1-NIK/IKK-NF-κB pathway | Activates macrophages and promotes IL-1β, IL-6 and TNF-α production | In vitro | [43] |
| miR-216a | Akt and TGF-β Pathway | Promotes TNF-α and IL-6 production | Both | [36] |
| miR-30a-5p | HTRA/TGF-β1 | Inhibits the production of trypsin, amylase and the inflammatory factors TNF-α, IL-6 and IL-1β | Both | [47] |
| miR-155 | SOCS1 | Promotes Th17 cell production and IL-6, IL-13 and TNF-α expression | Both | [51] |
| miR-361-5p | NFIA and Hes1 | Promotes Th17 cells to secrete IL-17A | Both | [52] |
| miR-29a/b1 | – | Promotes infiltration of neutrophils and macrophages and release of IL-6, IL-10 and TGFβ1 | In vivo | [53] |
| miR-155 | TAB2 | Promote the production of Beclin-1 and LC3 II levels and worsens the degree of autophagy damage | In vivo | [58] |
| miR-92b-3p | TRAF3/MKK3-p38 | Increases levels of Beclin-1, LC3 II and autophagosome formation | In vitro | [59] |
| miR-21 | Caspase 8 | Upregulation of RIP1 and RIP3 expression promotes necrosome formation | In vivo | [63] |
| miR-19b | – | Promotes necrosis of pancreatic acinar cells | In vivo | [64] |
| miR-15a | MAP2K4/JKN | Inhibits the production of IL-1, TNF and IL-6 and reduces pancreatic tissue necrosis | Both | [65] |
miRNA and early diagnosis, severity assessment and prognosis of AP
Early diagnosis and accurate assessment of the severity of a patient's current condition are conducive to the treatment of AP and reduce the incidence of complications and the hospital stay. Although there are currently some biomarkers, such as serum amylase and lipase, and imaging methods (CT, MRI, etc.) for the diagnosis of AP, there is still no single gold standard for predicting the severity of AP [66], especially within 48h after the patient is admitted to the hospital. However, some AP score tables such as the Ranson standard and APACHE II score cannot achieve this goal due to complicated operations. miRNA may become a biomarker for early diagnosis and accurate prediction of the severity of AP due to its key role in the occurrence and development of AP. Liu et al. collected serum samples from 12 AP patients and 3 healthy individuals in Nanchang, China, and performed a microarray analysis of their total miRNAs. They found that there were several differentially expressed miRNAs between SAP and MAP cases, including miR-92b, miR-146b-5p and miR-7. Subsequent RT-PCR analysis quantitatively verified that downregulated miR-92b, miR-10a and miR-7 can be used for the early diagnosis of AP. Moreover, the expression of miR-551b-5p differed significantly between SAP and moderate acute pancreatitis (MAP) patients (p < 0.005), and was correlated with the serum calcium level and complication rate (p < 0.05), indicating that miR-551b-5p is important for predicting the severity of AP [67]. As a serious complication of AP, vascular dysfunction can cause serious organ damage. For this reason, differentially expressed miRNAs reflecting vascular endothelial dysfunction can be used to predict the severity of AP [68, 69]. In another study, by comparing the differential expression profiles of miRNAs in SAP and MAP, it was confirmed that miR-551-5p and miR-126a-5p, which are specifically related to the endothelium, are highly expressed in SAP and are closely related to the severity of AP (AUC 0.716, sensitivity 69.2%, specificity 72.6%, p < 0.001 and AUC 0.748, sensitivity 60.0%, specificity 87.1%, p < 0.001, respectively) [70]. Microarray analysis found that there were significant differences in the expression of serum miRNAs between SAP and moderately severe acute pancreatitis (MSAP) patients with triglycerides. Compared with healthy controls, miR-24-3p, miR-222-3p, miR-361-5p, miR-1246 and miR-181a-5p showed differential expression in the hypertriglyceridemia-induced acute pancreatitis (HTAP) group, and the detection of serum samples revealed that these miRNAs were associated with inflammatory factors (procalcitonin (PCT), IL-1β, IL-6). An ROC working curve confirmed that these miRNAs can accurately assess the progression of HTAP, but further experimental verification is required [71]. Lung injury is a serious complication of SAP, and early prediction is particularly important for improving the prognosis of patients. A bioinformatics analysis performed by Lu et al. found that there were 5 miRNAs (hsa-miR-22-3p, 1260b, 762, 23b and 23a) that were significantly upregulated in SAP patients with acute lung injury (ALI) compared with SAP patients without ALI, and the expression levels of 7 species (hsa-miR-550a*, 324-5p, 484, 331-3p, 22-3p, 140-3p, and 342-3p) were decreased. qRT-PCR verified this result, but the molecular mechanism of regulation still needs in-depth study [72]. In addition, miRNAs also have an important reference value for the prognostic prediction of AP. Li et al. evaluated the value of miR-146a and miR-146b in AP, and found that among patients with SAP, MSAP and MAP, the expression levels of miR-146a and miR-146b were highest in SAP patients and were closely related to the Ranson's score, APACHE II score, SOFA score and C-reactive protein (CRP) level. This increase in miR-146a and miR-146b was accompanied by an increase in the risk of hospital mortality in SAP patients. However, a larger sample size and more data are needed in the future to confirm this finding [73]. In summary, these findings indicate that miRNAs are closely related to the early diagnosis, severity assessment and prognosis of AP and are expected to play an important role in comprehensive treatment of AP. A summary of studies of the miRNA molecular markers related to early diagnosis, prognosis and evaluation of the severity of AP is shown in Table 3.
Table 3.
Summary of studies of the miRNA molecular markers related to the early diagnosis, prognosis and evaluation of the severity of AP
| Molecular marker | Expression | Patient | Clinical relevance | References |
|---|---|---|---|---|
| miR-92b, miR-10a, miR-7 | Up | AP | Diagnosis | [67] |
| miR-551-5p, miR-126a-5p | Up | AP | Severity assessment | [67, 70] |
| miR-24-3p, miR-222-3p, miR-361-5p and miR-1246 | Up | HTAP | Severity assessment | [71] |
| miR-181a-5p | Down | HTAP | Severity assessment | [71] |
| miR-22-3p, 1260b, miR-762, miR-23b and miR-23a | Up | AP with ALI | Diagnosis, prognosis | [72] |
| miR-550a*, miR-324-5p, miR-484, miR-331-3p, miR-22-3p, miR-140-3p and miR-342-3p | Down | AP with ALI | Diagnosis, prognosis | [72] |
| miR-146a, miR-146b | Up | AP | Prognosis | [73] |
| miR-192-5p | Down | AP with NAFLD | Diagnosis | [74] |
| miR-372 | Up | HTGAP | Diagnosis, prognosis | [75] |
| miR-29a | Up | AP | Severity assessment, prognosis | [76] |
| miR-7, miR-9, miR-122 and miR-141 | Up | AP | Diagnosis, prognosis | [77] |
| miR-155, miR-21 | Down | AP | Diagnosis, severity assessment, prognosis | [78] |
| miR-127 | Down | AP with ALI | Diagnosis, severity assessment, prognosis | [79] |
| miR-216 | Up | AP | Diagnosis, severity assessment | [80] |
miRNA and the treatment of AP
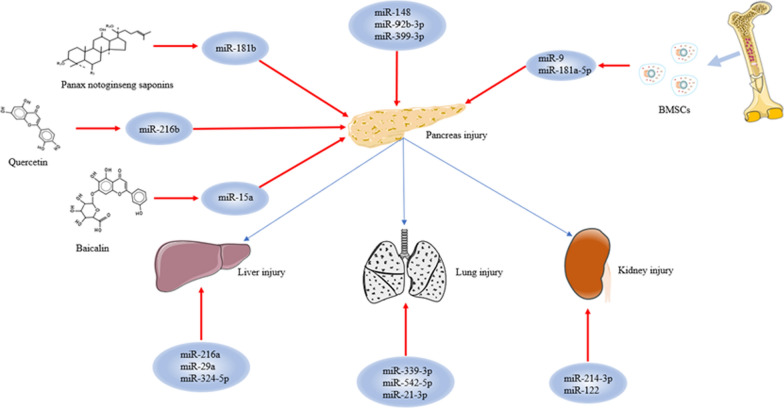
AP is still treated symptomatically. Although measures such as acid suppression, enzyme inhibition, anti-inflammatory drugs and fluid supplementation can play a certain role, their effect is relatively slow. Especially if early intervention is not timely, serious complications and long hospitalization times cannot be avoided [81]. As the roles of individual miRNAs in the progression of AP have been uncovered one by one, especially in the damage associated with distant organs, AP therapy targeting miRNAs has begun to be widely studied. Related miRNAs that could be directly or indirectly targeted for the treatment of AP and its complications are shown in Fig. 2.
Fig. 2.
Related miRNAs that could be directly or indirectly targeted for the treatment of AP and its complications. The figure shows that direct targeting of miRNA can attenuate the inflammatory response in AP or, combined with traditional Chinese medicine and MSCs, reverse the severity of AP. For AP with severe complications, therapies targeting miRNAs can also have a good effect
miRNAs and the treatment of simple AP
miRNA as a direct target
As a regulatory RNA, the mechanism of miRNAs in the progression of AP provides a research basis for AP-targeted therapies. Abnormal autophagy is an important part of the progression of AP, and how to inhibit this abnormal process has become a research hotspot. Miao et al. established a caerulein-treated AR42J cell model and found that the expression of miR-148a was downregulated. This result also appeared in the pancreatic tissue of AP mice, and the level of cell proliferation increased after overexpression of miR-148a. The corresponding pancreatic tissue was stained with haematoxylin–eosin (HE), and myeloperoxidase (MPO) expression was detected. It was found that the number of MPO-positive cells, the degree of degeneration of acinar cells, the infiltration and necrosis of inflammatory cells, and the vacuole area in cells were decreased in pancreatic tissue overexpressing miR-148a. Subsequent studies found that the expression levels of the autophagy-related proteins LC3-II, Beclin1, autophagy-related gene 4c (ATG4c) and autophagy-related gene 7 (ATG7) decreased with overexpression of miR-148a, and this process was mediated through the IL-6/STAT3 signalling pathway, thereby improving the pathological score of pancreatic tissue in AP mice [10]. miR-92b-3p, which is closely related to cancer, has also been found to play an important role in AP. Researchers established an AP cell model and detected increased expression of the proinflammatory factors TNF-α and IL-6 and the autophagy marker proteins Beclin1 and LC3-II/I. This increase was reversed by overexpression of miR-92b-3p, thereby improving the inflammation and autophagy in AP. Further experiments showed that overexpression of miR-92b-3p caused a decrease in TRAF3, subsequently inhibited the expression of proteins related to the MKK3-p38 signalling pathway (p-MKK3, MKK3, phosphorylated p38 (p-p38) and p38), and finally inhibited the progression of AP [59]. In another study, TRAF3 was shown to be regulated by miR-399-3p. miR-399-3p reduced the expression levels of inflammation-related factors (TNF-α, IL-1β and IL-6) and apoptosis factors (C-caspase3 and Bax), ultimately inhibiting inflammation and apoptosis in caerulein-treated AR42J cells [82]. In addition, Ge et al. found that a decreased abundance of miR-802, which maintains normal pancreatic acinar function, promoted acinar-to-ductal metaplasia (ADM) production and acinar cell proliferation, ultimately causing AP and exacerbating pancreatic injury. However, increasing the expression level of miR-802 effectively inhibited the occurrence and development of this event [83]. These studies provide new potential targets for the treatment of AP.
Associated active compounds of traditional Chinese medicine
Based on its unique theoretical system and effective treatment methods, Chinese medicine has become one of the most popular complementary and alternative therapies for the treatment of AP throughout the world. Moreover, increasing number of studies have verified that traditional Chinese medicine (such as curcumin, Camellia sinensis and Zingiber officinale roscoe) can reduce serum and urine amylase levels, inhibit the production of inflammatory factors, and reduce pancreatic damage. Due to the complexity of traditional Chinese medicine compounds, exploring the active compounds that exert anti-inflammatory effects and their regulatory mechanisms is the main direction of current research on AP therapy, especially combination therapy combined with modern medicine, such as combined miRNA targeted therapy for AP, which has become a research hotspot [84–86]. Panax notoginseng saponins (PNS), which are closely related to oxidative stress, are derived from the extract of Panax notoginseng Ledeb. The antioxidant properties of PNS may be effective in treating AP. Liu et al. established an SAP rat model using taurocholate, and the expression of miR-181b in rats treated with PNS was significantly increased. Subsequently, qRT-PCR and WB showed that the activity of the mammalian target of rapamycin (mTOR)/Akt pathway, which is related to autophagy activation, decreased, while the expression of LC3-II and Beclin1 decreased, leading to a reduction in the number of phagocytes, autophagosomes and autolysosomes. In addition, PNS induced increased apoptosis (increased expression of caspase-3, decreased expression of Blc-2) and significantly improved taurocholate-induced pancreatic injury [87]. Quercetin (QE), which has anticancer and anti-inflammatory effects, has also been shown to improve AP status. Through the establishment of AP cells and mouse models, related experiments have confirmed that QE can reduce the inflammatory factors TNF-α, IL-6 and IL-10. This result is achieved by upregulating miR-216b and inhibiting the MAP2K6/p38 pathway, which ultimately has a protective effect in AP [88]. In addition, the baicalin described above can target the MAP2K4/JKN signalling pathway through miR-15a, which can significantly reduce the necrosis in AP and aid in treatment of AP [65]. The active compounds of these traditional Chinese medicines and miRNAs provide a novel direction for combination therapy in AP.
Associated mesenchymal stem cells
As adult stem cells with low immunogenicity, marrow mesenchymal stem cells (MSCs) have the characteristics of self-renewal, immunosuppression, multidirectional differentiation, migration and paracrine activity, and an increasing number of studies have found that they have powerful anti-inflammatory and repair effects [89, 90]. For this reason, the treatment of severe AP with an infusion of MSCs has also attracted much attention. Qian et al. found that the expression of miR-9, which was low in the SAP group, was significantly increased by injection of bone marrow mesenchymal stem cells (BMSCs). The pathological sections also showed that pancreatic oedema, inflammatory infiltration, and necrosis levels decreased after injection of miR-9-modified BMSCs (pri-miR-9-BMSCs) compared with levels in the SAP group. The levels of amylase, lipase and inflammatory factors (TNF-α, IL-1β, IL-6) were all reduced, and pri-miR-9-BMSCs repaired damaged pancreatic tissue by inducing angiogenesis. This process includes promoting the expression of the angiogenesis-related protein angiopoietin-1 (Ang-1), soluble vascular endothelial tyrosine kinase receptor (TIE-2), C-X-C chemokine receptor type 4 (CXCR4) and p-AKT; targeting VE-cadherin and affecting the activity of the β훽-catenin signalling pathway; and recruiting more BMSCs to migrate to damaged tissues, ultimately promoting the regeneration of pancreatic tissue [91]. In another study, it was confirmed that miR-9 can be delivered to the damaged pancreas through pri-miR-9-BMSCs and miR-9 agomir, where it inhibits activation of the NF-κB signalling pathway, reduces proinflammatory factors (TNF-α, IL-1β, IL-6 and HMBG1) and increases the level of anti-inflammatory cytokines (IL-4, IL-10 and TGF-β). This provides an in-depth explanation of the molecular mechanism of AP treatment [92]. In addition, some researchers have found that BMSCs can repair damage in the pancreas by targeting miR-181a-5p. The main pathway involves BMSCs secretion of miR-181a-5p to target PTEN/Akt/TGF-β1 signalling and reduce inflammation and cell apoptosis, ultimately reducing the severity of AP [93]. At present, there is still little understanding of the regulatory mechanism between BMSCs and miRNAs, and further research in the future is expected to provide solid theoretical guidance for combination therapy for AP.
miRNAs and the treatment of AP with lung injury
If early intervention is not timely, AP will further develop and cause systemic inflammation, leading to serious complications. Lung injury is one of these complications. To explore the role of miRNA in SAP-ALI, Wu et al. established SAP-ALI mouse models and found that the expression of miR-339-3p was reduced in the lung tissue of SAP-ALI mice, while that of Annexin A3 (Anxa3) was the opposite. Bioinformatics predictions and subsequent experiments verified that miR-339-3p reduces inflammation and edema in SAP-ALI mice by targeting Anxa3 to inhibit Akt/mTOR signalling (decreased expression of TNF-α and IL-6) [94]. Some studies have found that miR-542-5p, which is related to late tumour lymphatic metastasis, vascular invasion and TNM staging, is expressed at lower levels in SAP-related ALI mice [95]. To further study the relationship between the two, miR-542-5p was overexpressed in SAP-ALI mice, and the serum amylase, wet-to-dry weight ratio of lung and pancreatic tissue, MPO activity, and severity of pathology all decreased in SAP-ALI mice after miR-542-5p overexpression. This process was mainly caused by a decrease in the expression of related inflammatory mediators and cytokines. The results of dual-luciferase assays revealed that overexpression of miR-542-5p reduced the expression of p21-activated kinase 1 (PAK1), and subsequently inhibited the activation of MAPK-related signalling pathways, including extracellular signal-regulated kinase 1/2 (ERK1/2), JNK and P38MAPK. Finally, miR-542-5p overexpression reduced the release of IL-1β, TNF-α, ICAM-1 and other factors, thereby improving the severity of SAP-ALI [96]. In a study performed by Wang et al., miR-21-3p was found that to be highly expressed in acute haemorrhagic necrotizing pancreatitis (AHNP), and its overexpression activated the transient receptor potential (TRP) signalling pathway; promoted the release of serum amylase, lipase and inflammatory factors; inhibited lung oxygenation; and aggravated pancreatic and lung damage. This result provides a new target for the treatment of SAP-ALI [97].
miRNAs and the treatment of AP with injury to other organs
Damaged to the liver and kidney is also a common complication of acute pancreatitis, but there are still few studies on the relationship between miRNAs and their interaction. In one study, an HP model complicated with acute renal injury was established through a long-term high-fat diet and sodium taurocholic injection, and serum amylase and blood lipid levels (total cholesterol (T-CHO), triglyceride (TG), low-density lipoprotein cholesterol (LDL-C)) were found to be increased. After miR-214-3p was injected, the expression effect of the above results was enhanced. HE and periodic acid-Schiff (PAS) staining of rat pancreas and kidney tissues revealed pancreatic acinar degeneration, interstitial inflammatory cell infiltration, vasodilation, bleeding, and increased necrosis in the miR-214-3p overexpression group. The degree of swelling of renal tubular epithelial cells and glomeruli was obvious. Sirius red staining revealed a significant increase in the degree of fibrosis in the kidneys and pancreas, while the anti-miR-214-3p group showed the opposite results. These effects were dependent on the activity of the miR-214-3p/PTEN/Akt axis [98]. In addition, studies have found that miR-122 is negatively correlated with the erythropoietin (EPO) level in an induced AP mouse model, and eventually causes anaemia. This may be related to miR-122-mediated kidney inflammatory damage, but relevant experimental verification is needed [99]. At present, there are still few studies on miRNAs as targets for the treatment of SAP complicated by liver injury. In a case–control study, by analysing the peripheral blood of AP patients with or without liver injury, researchers found that elevated miR-216a and miR-29a and reduced miR-324-5p are closely related to the Balthazar CT score, APACHE II score, CRP level and length of stay in the hospital, and the expression level of the above miRNAs can effectively predict the severity of liver injury, but further confirmation is needed [100]. In summary, the above studies clarify the application value of miRNAs in AP with liver and kidney damage and provide a new way to treat severe AP. A summary of studies of the mechanism of action of miRNAs in the treatment of AP and its complications is presented in Table 4.
Table 4.
Summary of studies of the mechanism of action of miRNAs in the treatment of AP and its complications
| miRNA | Mechanism | Disease type | Application | Animal model | References |
|---|---|---|---|---|---|
| miR-148a | Inhibits the production of the autophagy-related proteins LC3-II, Beclin1, ATG4c and ATG7 via the IL-6/STAT3 signalling pathway | AP | Suppress | Male BALB/c mice | [10] |
| miR-92b-3p | Suppresses the expression of proteins associated with the MKK3-p38 signalling pathway (p-MKK3, MKK3, p-p38 and p38) by reducing TRAF3 production | AP | Suppress | – | [59] |
| miR-399-3p | Inhibits the production of inflammatory and apoptotic factors by regulating TRAF3 | AP | Suppress | – | [82] |
| miR-802 | Inhibits ADM production and acinar cell proliferation | AP | Suppress | mir-802 fl/fl mice | [83] |
| miR-181b | Inhibits autophagy and increases apoptosis by regulating mTOR/Akt when combined with PNS | AP | Suppress | Male Sprague- Dawley rats | [87] |
| miR-216b | Inhibition of the MAP2K6/p38 pathway when combined with QE | AP | Suppress | C57BL/6 mice | [88] |
| miR-15a | Combined with baicalin to regulate the MAP2K4/JKN signalling pathway | AP | Suppress | – | [65] |
| miR-9 | Infusion of miR-9-modified BMSCs induces angiogenesis to repair damaged pancreatic tissue | AP | Suppress | Male SD rats | [91, 92] |
| miR-181a-5p | Regulation of PTEN/Akt/TGF-β1 via secretion of miR-181a-5p from BMSCs | AP | Suppress | Male SD rats | [93] |
| miR-339-3p | Inhibition of Akt/mTOR signaling by targeting Anxa3 | AP with lung injury | Suppress | Male mice | [94] |
| miR-542-5p | Inhibits the PAK1/MAPK signalling pathway and reduces the release of inflammatory factors | AP with lung injury | Suppress | Mice | [95, 96] |
| miR-21-3p | Promotes the release of serum amylase, lipase, and inflammatory factors and inhibits pulmonary oxygenation by activating the TRP signalling pathway | AP with lung injury | Promote | Wistar rats | [97] |
| miR-214-3p | Promotion of pancreatic acinar degeneration and renal tubular epithelial cell swelling via the PTEN/Akt axis | AP with kidney injury | Promote | Male Sprague–Dawley rats | [98] |
| miR-122 | AP promotes the secretion of miR-122 and reduces the level of renal EPO | AP with kidney injury | Promote | C57BL/6 mice | [99] |
| miR-216a, miR-29a, miR-324-5p | Predicts the severity of liver damage | AP with liver injury | Promote | – | [100] |
Summary and prospects
In recent years, the risk factors for AP have gradually increased, accompanied by high morbidity and mortality. Especially for SAP, it is difficult to avoid serious complications and recurrence with conventional diagnosis and treatment. Therefore, we urgently need to innovate early diagnosis, prognostic evaluation and treatment methods. With extensive research on miRNAs, their unique role in inflammation has laid a solid foundation for in-depth exploration of the regulatory mechanisms involved in the pathogenesis and progression of AP. For example, the upregulation or downregulation of miRNAs affects downstream inflammatory signals to regulate the expression of inflammatory factors and cytokines. In addition, miRNAs can also regulate apoptosis and necrosis-related molecules to promote or inhibit AP process. It is worth noting that the interactions between miRNAs and inflammatory cells, such as recruitment, activation, and induction of differentiation, have improved our understanding of the molecular mechanisms mediated by miRNAs in the pathogenesis of AP, making it possible that miRNAs could be a target for the treatment of AP.
Nevertheless, the current research on the relationship between miRNAs and AP progression is still in the initial stage, such as how miRNAs regulate the activation of trypsinogen, how exogenous miRNAs enter damaged cells through extracellular vesicles, and how miRNAs recruit and activate differentiation-related inflammatory cells. In addition, in future research we should pay more attention to how miRNAs can be used to treat AP, such as how miRNA-related mechanisms highlight the effects of Chinese medicine, how miRNAs can be combined with mesenchymal stem cells to treat SAP with severe complications, such as liver and kidney damage and how miRNAs regulate the activation of immune cells to effectively play immunotherapy role. Although these points will require a long time to be thoroughly studied, miRNA-related therapies can provide new methods and strategies for severe inflammatory diseases such as AP. In short, for targeted treatment of AP, miRNAs have broad application prospects.
Acknowledgements
Not applicable.
Abbreviations
- AP
Acute pancreatitis
- SAP
Severe acute pancreatitis
- miRNAs
MicroRNAs
- PTEN
Phosphatase and tensin homologue
- NF-κB
Nuclear factor kappa-B
- Wnt/β-catenin
Wnt (wingless) /β-catenin
- JAK/STAT
Janus kinase/signal transducer and activator of transcription
- TET2/A20
Tet methylcytosine dioxygenase 2/the deubiquitinating enzyme A20
- TLR4
Toll-like receptor 4
- PDCD4
Programmed cell death protein 4
- KLF4
Krüppel-like factor 4
- Keap1/Nrf2
Kelch-like ECH-associated protein 1/nuclear factor erythroid 2-related factor 2
- IL5RA
Interleukin 5 receptor
- VEGF
Vascular endothelial growth factor
- HMGB1/PI3K/AKT
High mobility group box 1/phosphatidylinositide 3-kinase/protein kinase B
- FOXO3/NAPDH/ROS
Forkhead box O3/nicotinamide adenine dinucleotide phosphate/ reactive oxygen species
- FGF10
Fibroblast growth factor 10
- p38 MAPK
P38 MAP kinase
- TLC
Taurolithocholate
- HDAC3
Histone deacetylase 3
- TRAF6-TAB2-TAK1-NIK/IKK
TNF receptor-associated factor 6-TGF-beta activated kinase 1/MAP3K7 binding protein 2-transforming growth factor beta activated kinase 1-NF-κB inducing kinase/IκB kinase
- HTRA
High-temperature requirement A
- STC
Sodium taurocholate
- DAMPs
Damage-associated molecular patterns
- Th17
IL-17-producing CD4+ T helper
- Treg
Regulatory T
- SOCS1
Suppressor of cytokine signalling 1
- NFIA
Nuclear factor IA
- TRAF3
Tumour necrosis factor receptor-associated factor-3
- RIP1/RIPK1
Receptor-interacting protein kinase 1
- MKK3
Phosphorylated mitogen-activated protein kinase kinase 3
- SIRS
Systemic inflammatory response syndrome
- ANP
Acute necrotizing pancreatitis
- TLC-S
Taurolithocholic acid 3-sulfate disodium salt
- MAP2K4
Mitogen-activated protein kinase kinase 4
- JKN
C-Jun N-terminal kinase
- MAP
Moderate acute pancreatitis
- NAFLD
Nonalcoholic fatty liver disease
- HTGAP
Hyperlipidaemic acute pancreatitis
- HE
Haematoxylin–eosin
- MPO
Myeloperoxidase
- LC3 II
Microtubule-associated protein light chain 3
- ATG4c
Autophagy-related gene 4c
- ATG7
Autophagy-related gene 7
- PNS
Panax notoginseng saponins
- mTOR
Mammalian target of rapamycin
- ADM
Acinar-to-ductal metaplasia
- QE
Quercetin
- MSCs
Mesenchymal stem cells
- BMSCs
Bone marrow mesenchymal stem cells
- PAK1
P21-activated kinase 1
- Anxa3
A higher level of Annexin A3
- ERK1/2
Extracellular signal-regulated kinase ½
- AHNP
Acute haemorrhagic necrotizing pancreatitis
- TRP
Transient receptor potential
- PAS
Periodic acid-Schiff
- T-CHO
Total cholesterol
- TG
Triglyceride
- LDL-C
Low-density lipoprotein cholesterol
- EPO
Erythropoietin
Authors' contributions
WZ: Writing—original draft preparation, supervision, project administration, funding acquisition. SD: Writing—original draft preparation, visualization. ZC, XL and WJ: Writing—review & editing, supervision. All authors read and approved the final manuscript.
Funding
This work was supported by the Science and Technology Projects of Chengguan District in Lanzhou (2020-2-11-4) and Traditional Chinese Medicine Scientific Research Project of Gansu Province (GZKP-2020-28).
Declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Wence Zhou and Shi Dong contributed equally to this work
References
- 1.van Dijk SM, Hallensleben NDL, van Santvoort HC, et al. Acute pancreatitis: recent advances through randomized trials. Gut. 2017;66(11):2024–2032. doi: 10.1136/gutjnl-2016-313595. [DOI] [PubMed] [Google Scholar]
- 2.Lugea A, Waldron RT, Mareninova OA, et al. Human pancreatic acinar cells: proteomic characterization, physiologic responses, and organellar disorders in ex vivo pancreatitis. Am J Pathol. 2017;187(12):2726–2743. doi: 10.1016/j.ajpath.2017.08.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Gukovskaya AS, Pandol SJ, Gukovsky I. New insights into the pathways initiating and driving pancreatitis. Curr Opin Gastroenterol. 2016;32(5):429–435. doi: 10.1097/MOG.0000000000000301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Lankisch PG, Apte M, Banks PA. Acute pancreatitis [published correction appears in Lancet. 2015;386(10008):2058] Lancet. 2015;386(9988):85–96. doi: 10.1016/S0140-6736(14)60649-8. [DOI] [PubMed] [Google Scholar]
- 5.van Santvoort HC, Bakker OJ, Bollen TL, et al. A conservative and minimally invasive approach to necrotizing pancreatitis improves outcome. Gastroenterology. 2011;141(4):1254–1263. doi: 10.1053/j.gastro.2011.06.073. [DOI] [PubMed] [Google Scholar]
- 6.Bang JY, Wilcox CM, Arnoletti JP, et al. Superiority of endoscopic interventions over minimally invasive surgery for infected necrotizing pancreatitis: meta-analysis of randomized trials. Dig Endosc. 2020;32(3):298–308. doi: 10.1111/den.13470. [DOI] [PubMed] [Google Scholar]
- 7.Schepers NJ, Bakker OJ, Besselink MG, et al. Impact of characteristics of organ failure and infected necrosis on mortality in necrotising pancreatitis. Gut. 2019;68(6):1044–1051. doi: 10.1136/gutjnl-2017-314657. [DOI] [PubMed] [Google Scholar]
- 8.Pagliari D, Brizi MG, Saviano A, et al. Clinical assessment and management of severe acute pancreatitis: a multi-disciplinary approach in the XXI century. Eur Rev Med Pharmacol Sci. 2019;23(2):771–787. doi: 10.26355/eurrev_201901_16892. [DOI] [PubMed] [Google Scholar]
- 9.Wei JW, Huang K, Yang C, et al. Non-coding RNAs as regulators in epigenetics (Review) Oncol Rep. 2017;37(1):3–9. doi: 10.3892/or.2016.5236. [DOI] [PubMed] [Google Scholar]
- 10.Miao B, Qi WJ, Zhang SW, et al. miR-148a suppresses autophagy by down-regulation of IL-6/STAT3 signaling in cerulein-induced acute pancreatitis. Pancreatology. 2019;19(4):557–565. doi: 10.1016/j.pan.2019.04.014. [DOI] [PubMed] [Google Scholar]
- 11.Saliminejad K, Khorram Khorshid HR, Soleymani Fard S, et al. An overview of microRNAs: biology, functions, therapeutics, and analysis methods. J Cell Physiol. 2019;234(5):5451–5465. doi: 10.1002/jcp.27486. [DOI] [PubMed] [Google Scholar]
- 12.Pham H, Rodriguez CE, Donald GW, et al. miR-143 decreases COX-2 mRNA stability and expression in pancreatic cancer cells. Biochem Biophys Res Commun. 2013;439(1):6–11. doi: 10.1016/j.bbrc.2013.08.042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Li X, Jiang W, Gan Y, et al. The application of exosomal microRNAs in the treatment of pancreatic cancer and its research progress. Pancreas. 2021;50(1):12–16. doi: 10.1097/MPA.0000000000001713. [DOI] [PubMed] [Google Scholar]
- 14.Yeung YT, Aziz F, Guerrero-Castilla A, et al. Signaling pathways in inflammation and anti-inflammatory therapies. Curr Pharm Des. 2018;24(14):1449–1484. doi: 10.2174/1381612824666180327165604. [DOI] [PubMed] [Google Scholar]
- 15.Melisi D, Garcia-Carbonero R, Macarulla T, et al. TGFβ receptor inhibitor galunisertib is linked to inflammation- and remodeling-related proteins in patients with pancreatic cancer. Cancer Chemother Pharmacol. 2019;83(5):975–991. doi: 10.1007/s00280-019-03807-4. [DOI] [PubMed] [Google Scholar]
- 16.Hunter P. The inflammation theory of disease. The growing realization that chronic inflammation is crucial in many diseases opens new avenues for treatment. EMBO Rep. 2012;13(11):968–970. doi: 10.1038/embor.2012.142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Jiang S, Yan W, Wang SE, et al. Dual mechanisms of posttranscriptional regulation of Tet2 by Let-7 microRNA in macrophages. Proc Natl Acad Sci USA. 2019;116(25):12416–12421. doi: 10.1073/pnas.1811040116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kumar M, Sahu SK, Kumar R, et al. MicroRNA let-7 modulates the immune response to Mycobacterium tuberculosis infection via control of A20, an inhibitor of the NF-κB pathway. Cell Host Microbe. 2015;17(3):345–356. doi: 10.1016/j.chom.2015.01.007. [DOI] [PubMed] [Google Scholar]
- 19.Ding Y, Wang L, Zhao Q, et al. MicroRNA-93 inhibits chondrocyte apoptosis and inflammation in osteoarthritis by targeting the TLR4/NF-κB signaling pathway. Int J Mol Med. 2019;43(2):779–790. doi: 10.3892/ijmm.2018.4033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Zhou W, Su L, Duan X, et al. MicroRNA-21 down-regulates inflammation and inhibits periodontitis. Mol Immunol. 2018;101:608–614. doi: 10.1016/j.molimm.2018.05.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Pan Y, Hui X, Hoo RLC, et al. Adipocyte-secreted exosomal microRNA-34a inhibits M2 macrophage polarization to promote obesity-induced adipose inflammation. J Clin Invest. 2019;129(2):834–849. doi: 10.1172/JCI123069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Ju M, Liu B, He H, et al. MicroRNA-27a alleviates LPS-induced acute lung injury in mice via inhibiting inflammation and apoptosis through modulating TLR4/MyD88/NF-κB pathway. Cell Cycle. 2018;17(16):2001–2018. doi: 10.1080/15384101.2018.1509635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Zhao XJ, Yu HW, Yang YZ, et al. Polydatin prevents fructose-induced liver inflammation and lipid deposition through increasing miR-200a to regulate Keap1/Nrf2 pathway [published correction appears in Redox Biol. 2019 Apr;22:101101] Redox Biol. 2018;18:124–137. doi: 10.1016/j.redox.2018.07.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Lin X, Lin Q. MiRNA-495–3p attenuates TNF-α induced apoptosis and inflammation in human nucleus pulposus cells by targeting IL5RA. Inflammation. 2020;43(5):1797–1805. doi: 10.1007/s10753-020-01254-5. [DOI] [PubMed] [Google Scholar]
- 25.Liang Y, Xie J, Che D, et al. MiR-124-3p helps to protect against acute respiratory distress syndrome by targeting p65. Biosci Rep. 2020;40(5):BSR20192132. doi: 10.1042/BSR20192132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Zhang A, Wang G, Jia L, et al. Exosome-mediated microRNA-138 and vascular endothelial growth factor in endometriosis through inflammation and apoptosis via the nuclear factor-κB signaling pathway. Int J Mol Med. 2019;43(1):358–370. doi: 10.3892/ijmm.2018.3980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Kim D, Nguyen QT, Lee J, et al. Anti-inflammatory roles of glucocorticoids are mediated by Foxp3+ regulatory T cells via a miR-342-dependent mechanism. Immunity. 2020;53(3):581–596.e5. doi: 10.1016/j.immuni.2020.07.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Long X, Yao X, Jiang Q, et al. Astrocyte-derived exosomes enriched with miR-873a-5p inhibit neuroinflammation via microglia phenotype modulation after traumatic brain injury. J Neuroinflamm. 2020;17(1):89. doi: 10.1186/s12974-020-01761-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Zheng L, Su J, Zhang Z, et al. Salidroside regulates inflammatory pathway of alveolar macrophages by influencing the secretion of miRNA-146a exosomes by lung epithelial cells. Sci Rep. 2020;10(1):20750. doi: 10.1038/s41598-020-77448-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Cai G, Cai G, Zhou H, et al. Mesenchymal stem cell-derived exosome miR-542–3p suppresses inflammation and prevents cerebral infarction. Stem Cell Res Ther. 2021;12(1):2. doi: 10.1186/s13287-020-02030-w. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 31.Lai N, Wu D, Liang T, et al. Systemic exosomal miR-193b-3p delivery attenuates neuroinflammation in early brain injury after subarachnoid hemorrhage in mice. J Neuroinflamm. 2020;17(1):74. doi: 10.1186/s12974-020-01745-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Wang Y, Shen S, Li Z, et al. MIR-140–5p affects chondrocyte proliferation, apoptosis, and inflammation by targeting HMGB1 in osteoarthritis. Inflamm Res. 2020;69(1):63–73. doi: 10.1007/s00011-019-01294-0. [DOI] [PubMed] [Google Scholar]
- 33.Shang J, Wang L, Tan L, et al. MiR-27a-3p overexpression mitigates inflammation and apoptosis of lipopolysaccharides-induced alveolar epithelial cells by targeting FOXO3 and suppressing the activation of NAPDH/ROS. Biochem Biophys Res Commun. 2020;533(4):723–731. doi: 10.1016/j.bbrc.2020.07.126. [DOI] [PubMed] [Google Scholar]
- 34.Heckler M, Hackert T, Hu K, et al. Severe acute pancreatitis: surgical indications and treatment. Langenbecks Arch Surg. 2021;406(3):521–535. doi: 10.1007/s00423-020-01944-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Shen Y, Xue C, You G, et al. miR-9 alleviated the inflammatory response and apoptosis in caerulein-induced acute pancreatitis by regulating FGF10 and the NF-κB signaling pathway. Exp Ther Med. 2021;22(2):795. doi: 10.3892/etm.2021.10227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Zhang J, Ning X, Cui W, et al. Transforming growth factor (TGF)-β-induced microRNA-216a promotes acute pancreatitis via Akt and TGF-β pathway in mice. Dig Dis Sci. 2015;60(1):127–135. doi: 10.1007/s10620-014-3261-9. [DOI] [PubMed] [Google Scholar]
- 37.Lawrence T. The nuclear factor NF-kappaB pathway in inflammation. Cold Spring Harb Perspect Biol. 2009;1(6):a001651. doi: 10.1101/cshperspect.a001651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Perkins ND. The diverse and complex roles of NF-κB subunits in cancer. Nat Rev Cancer. 2012;12(2):121–132. doi: 10.1038/nrc3204. [DOI] [PubMed] [Google Scholar]
- 39.Jin HZ, Yang XJ, Zhao KL, et al. Apocynin alleviates lung injury by suppressing NLRP3 inflammasome activation and NF-κB signaling in acute pancreatitis. Int Immunopharmacol. 2019;75:105821. doi: 10.1016/j.intimp.2019.105821. [DOI] [PubMed] [Google Scholar]
- 40.Han D, Li J, Wang H, et al. Circular RNA circMTO1 acts as the sponge of microRNA-9 to suppress hepatocellular carcinoma progression. Hepatology. 2017;66(4):1151–1164. doi: 10.1002/hep.29270. [DOI] [PubMed] [Google Scholar]
- 41.Wu Y, Tang Y, Xie S, et al. Chimeric peptide supramolecular nanoparticles for plectin-1 targeted miRNA-9 delivery in pancreatic cancer. Theranostics. 2020;10(3):1151–1165. doi: 10.7150/thno.38327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Chen Z, Chen Y, Pan L, et al. Dachengqi decoction attenuates inflammatory response via inhibiting HMGB1 mediated NF-κB and P38 MAPK signaling pathways in severe acute pancreatitis. Cell Physiol Biochem. 2015;37(4):1379–1389. doi: 10.1159/000430403. [DOI] [PubMed] [Google Scholar]
- 43.Zhao Y, Wang H, Lu M, et al. Pancreatic acinar cells employ miRNAs as mediators of intercellular communication to participate in the regulation of pancreatitis-associated macrophage activation. Mediat Inflamm. 2016;2016:6340457. doi: 10.1155/2016/6340457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Wildi S, Kleeff J, Mayerle J, et al. Suppression of transforming growth factor beta signalling aborts caerulein induced pancreatitis and eliminates restricted stimulation at high caerulein concentrations. Gut. 2007;56(5):685–692. doi: 10.1136/gut.2006.105833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Shiga A, Nozaki H, Yokoseki A, et al. Cerebral small-vessel disease protein HTRA1 controls the amount of TGF-β1 via cleavage of proTGF-β1. Hum Mol Genet. 2011;20(9):1800–1810. doi: 10.1093/hmg/ddr063. [DOI] [PubMed] [Google Scholar]
- 46.Rani R, Smulian AG, Greaves DR, et al. TGF-β limits IL-33 production and promotes the resolution of colitis through regulation of macrophage function. Eur J Immunol. 2011;41(7):2000–2009. doi: 10.1002/eji.201041135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Xiang H, Tao X, Xia S, et al. Emodin alleviates sodium taurocholate-induced pancreatic acinar cell injury via MicroRNA-30a-5p-mediated inhibition of high-temperature requirement A/transforming growth factor beta 1 inflammatory signaling. Front Immunol. 2017;8:1488. doi: 10.3389/fimmu.2017.01488. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Kang R, Lotze MT, Zeh HJ, et al. Cell death and DAMPs in acute pancreatitis. Mol Med. 2014;20(1):466–477. doi: 10.2119/molmed.2014.00117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Hoque R, Malik AF, Gorelick F, et al. Sterile inflammatory response in acute pancreatitis. Pancreas. 2012;41(3):353–357. doi: 10.1097/MPA.0b013e3182321500. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Venkatesha SH, Dudics S, Weingartner E, et al. Altered Th17/Treg balance and dysregulated IL-1β response influence susceptibility/resistance to experimental autoimmune arthritis. Int J Immunopathol Pharmacol. 2015;28(3):318–328. doi: 10.1177/0394632015595757. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Wang D, Tang M, Zong P, et al. MiRNA-155 regulates the Th17/Treg ratio by targeting SOCS1 in severe acute pancreatitis. Front Physiol. 2018;9:686. doi: 10.3389/fphys.2018.00686. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Song M, Wang Y, Zhou P, et al. MicroRNA-361–5p aggravates acute pancreatitis by promoting interleukin-17A secretion via impairment of nuclear factor IA-dependent Hes1 downregulation. J Med Chem. 2021;64(22):16541–16552. doi: 10.1021/acs.jmedchem.1c01110. [DOI] [PubMed] [Google Scholar]
- 53.Dey S, Udari LM, RiveraHernandez P, et al. Loss of miR-29a/b1 promotes inflammation and fibrosis in acute pancreatitis. JCI Insight. 2021;6(19):e149539. doi: 10.1172/jci.insight.149539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Levine B, Kroemer G. Biological functions of autophagy genes: a disease perspective. Cell. 2019;176(1–2):11–42. doi: 10.1016/j.cell.2018.09.048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Kocaturk NM, Akkoc Y, Kig C, et al. Autophagy as a molecular target for cancer treatment. Eur J Pharm Sci. 2019;134:116–137. doi: 10.1016/j.ejps.2019.04.011. [DOI] [PubMed] [Google Scholar]
- 56.Biczo G, Vegh ET, Shalbueva N, et al. Mitochondrial dysfunction, through impaired autophagy, leads to endoplasmic reticulum stress, deregulated lipid metabolism, and pancreatitis in animal models. Gastroenterology. 2018;154(3):689–703. doi: 10.1053/j.gastro.2017.10.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Mei Q, Zeng Y, Huang C, et al. Rapamycin alleviates hypertriglyceridemia-related acute pancreatitis via restoring autophagy flux and inhibiting endoplasmic reticulum stress. Inflammation. 2020;43(4):1510–1523. doi: 10.1007/s10753-020-01228-7. [DOI] [PubMed] [Google Scholar]
- 58.Wan J, Yang X, Ren Y, et al. Inhibition of miR-155 reduces impaired autophagy and improves prognosis in an experimental pancreatitis mouse model. Cell Death Dis. 2019;10(4):303. doi: 10.1038/s41419-019-1545-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Sun H, Tian J, Li J. MiR-92b-3p ameliorates inflammation and autophagy by targeting TRAF3 and suppressing MKK3-p38 pathway in caerulein-induced AR42J cells. Int Immunopharmacol. 2020;88:106691. doi: 10.1016/j.intimp.2020.106691. [DOI] [PubMed] [Google Scholar]
- 60.Villatoro E, Mulla M, Larvin M. Antibiotic therapy for prophylaxis against infection of pancreatic necrosis in acute pancreatitis. Cochrane Database Syst Rev. 2010;2010(5):CD002941. doi: 10.1002/14651858.CD002941.pub3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Garg PK, Meena D, Babu D, et al. Endoscopic versus laparoscopic drainage of pseudocyst and walled-off necrosis following acute pancreatitis: a randomized trial. Surg Endosc. 2020;34(3):1157–1166. doi: 10.1007/s00464-019-06866-z. [DOI] [PubMed] [Google Scholar]
- 62.Mareninova OA, Sung KF, Hong P, et al. Cell death in pancreatitis: caspases protect from necrotizing pancreatitis. J Biol Chem. 2006;281(6):3370–3381. doi: 10.1074/jbc.M511276200. [DOI] [PubMed] [Google Scholar]
- 63.Ma X, Conklin DJ, Li F, et al. The oncogenic microRNA miR-21 promotes regulated necrosis in mice. Nat Commun. 2015;6:7151. doi: 10.1038/ncomms8151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Hu MX, Zhang HW, Fu Q, et al. Functional role of MicroRNA-19b in acinar cell necrosis in acute necrotizing pancreatitis. J Huazhong Univ Sci Technolog Med Sci. 2016;36(2):221–225. doi: 10.1007/s11596-016-1570-2. [DOI] [PubMed] [Google Scholar]
- 65.Zhen J, Chen W, Liu Y, et al. Baicalin protects against acute pancreatitis involving JNK signaling pathway via regulating miR-15a. Am J Chin Med. 2021;49(1):147–161. doi: 10.1142/S0192415X21500087. [DOI] [PubMed] [Google Scholar]
- 66.Lippi G, Valentino M, Cervellin G. Laboratory diagnosis of acute pancreatitis: in search of the Holy Grail. Crit Rev Clin Lab Sci. 2012;49(1):18–31. doi: 10.3109/10408363.2012.658354. [DOI] [PubMed] [Google Scholar]
- 67.Liu P, Xia L, Zhang WL, et al. Identification of serum microRNAs as diagnostic and prognostic biomarkers for acute pancreatitis. Pancreatology. 2014;14(3):159–166. doi: 10.1016/j.pan.2014.03.019. [DOI] [PubMed] [Google Scholar]
- 68.Baillargeon JD, Orav J, Ramagopal V, et al. Hemoconcentration as an early risk factor for necrotizing pancreatitis. Am J Gastroenterol. 1998;93(11):2130–2134. doi: 10.1111/j.1572-0241.1998.00608.x. [DOI] [PubMed] [Google Scholar]
- 69.Mao EQ, Fei J, Peng YB, et al. Rapid hemodilution is associated with increased sepsis and mortality among patients with severe acute pancreatitis. Chin Med J. 2010;123(13):1639–1644. [PubMed] [Google Scholar]
- 70.Kuśnierz-Cabala B, Nowak E, Sporek M, et al. Serum levels of unique miR-551–5p and endothelial-specific miR-126a-5p allow discrimination of patients in the early phase of acute pancreatitis. Pancreatology. 2015;15(4):344–351. doi: 10.1016/j.pan.2015.05.475. [DOI] [PubMed] [Google Scholar]
- 71.An F, Zhan Q, Xia M, et al. From moderately severe to severe hypertriglyceridemia induced acute pancreatitis: circulating miRNAs play role as potential biomarkers. PLoS ONE. 2014;9(11):e111058. doi: 10.1371/journal.pone.0111058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Lu XG, Kang X, Zhan LB, et al. Circulating miRNAs as biomarkers for severe acute pancreatitis associated with acute lung injury. World J Gastroenterol. 2017;23(41):7440–7449. doi: 10.3748/wjg.v23.i41.7440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Li XY, Wang YF, Li N. Circulating microRNA-146a and microRNA-146b exhibit potential to serve as markers for acute pancreatitis management and prognosis. Eur Rev Med Pharmacol Sci. 2020;24(24):12770–12780. doi: 10.26355/eurrev_202012_24177. [DOI] [PubMed] [Google Scholar]
- 74.Hu Y, Yu Y. Dysregulation of miR-192-5p in acute pancreatitis patients with nonalcoholic fatty liver and its functional role in acute pancreatitis progression. Biosci Rep. 2020;40(5):BSR20194345. doi: 10.1042/BSR20194345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Shan Y, Kong W, Zhu A, et al. Increased levels of miR-372 correlate with disease progression in patients with hyperlipidemic acute pancreatitis. Exp Ther Med. 2020;19(6):3845–3850. doi: 10.3892/etm.2020.8609. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Gao M, Bian E, Li H, et al. Up-regulation of circulating miR-29a in patients with acute pancreatitis and is positively correlated with disease severity and poor prognosis. Xi Bao Yu Fen Zi Mian Yi Xue Za Zhi. 2018;34(10):931–936. [PubMed] [Google Scholar]
- 77.Lu P, Wang F, Wu J, et al. Elevated serum miR-7, miR-9, miR-122, and miR-141 are noninvasive biomarkers of acute pancreatitis. Dis Markers. 2017;2017:7293459. doi: 10.1155/2017/7293459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Hu L, Han D, Yu D, et al. Circulating blood miR-155 and miR-21 promote the development of acute pancreatitis and can be used to assess the risk stratification of pancreatitis. J Healthc Eng. 2021;2021:2064162. doi: 10.1155/2021/2064162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Shi N, Deng L, Chen W, et al. Is microRNA-127 a novel biomarker for acute pancreatitis with lung injury? Dis Markers. 2017;2017:1204295. doi: 10.1155/2017/1204295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Zhang XX, Deng LH, Chen WW, et al. Circulating microRNA 216 as a marker for the early identification of severe acute pancreatitis. Am J Med Sci. 2017;353(2):178–186. doi: 10.1016/j.amjms.2016.12.007. [DOI] [PubMed] [Google Scholar]
- 81.Hines OJ, Pandol SJ. Management of severe acute pancreatitis. BMJ. 2019;367:l6227. doi: 10.1136/bmj.l6227. [DOI] [PubMed] [Google Scholar]
- 82.Wang Q, Liu S, Han Z. miR-339–3p regulated acute pancreatitis induced by caerulein through targeting TNF receptor-associated factor 3 in AR42J cells. Open Life Sci. 2020;15(1):912–922. doi: 10.1515/biol-2020-0084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Ge W, Goga A, He Y, et al. miR-802 suppresses acinar-to-ductal reprogramming during early pancreatitis and pancreatic carcinogenesis. Gastroenterology. 2022;162(1):269–284. doi: 10.1053/j.gastro.2021.09.029. [DOI] [PubMed] [Google Scholar]
- 84.Zhu SF, Chen WW, Xiang J, et al. Pharmacokinetic and pharmacodynamic comparison of Chinese herbal ointment Liu-he-Dan and micron Liu-he-Dan ointment in rats with acute pancreatitis. Evid Based Complement Alternat Med. 2014;2014:389576. doi: 10.1155/2014/389576. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Wang J, Guo Y, Li GL. Current status of standardization of traditional Chinese medicine in China. Evid Based Complement Alternat Med. 2016;2016:9123103. doi: 10.1155/2016/9123103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Qiong W, Yiping W, Jinlin Y, et al. Chinese medicinal herbs for acute pancreatitis. Cochrane Database Syst Rev. 2005;1:CD003631. doi: 10.1002/14651858.CD003631.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Liu MW, Wei R, Su MX, et al. Effects of Panax notoginseng saponins on severe acute pancreatitis through the regulation of mTOR/Akt and caspase-3 signaling pathway by upregulating miR-181b expression in rats. BMC Complement Altern Med. 2018;18(1):51. doi: 10.1186/s12906-018-2118-8. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 88.Sheng B, Zhao L, Zang X, et al. Quercetin inhibits caerulein-induced acute pancreatitis through regulating miR-216b by targeting MAP2K6 and NEAT1. Inflammopharmacology. 2021;29(2):549–559. doi: 10.1007/s10787-020-00767-7. [DOI] [PubMed] [Google Scholar]
- 89.Ankrum JA, Ong JF, Karp JM. Mesenchymal stem cells: immune evasive, not immune privileged. Nat Biotechnol. 2014;32(3):252–260. doi: 10.1038/nbt.2816. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Fu X, Liu G, Halim A, et al. Mesenchymal stem cell migration and tissue repair. Cells. 2019;8(8):784. doi: 10.3390/cells8080784. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Qian D, Song G, Ma Z, et al. MicroRNA-9 modified bone marrow-derived mesenchymal stem cells (BMSCs) repair severe acute pancreatitis (SAP) via inducing angiogenesis in rats. Stem Cell Res Ther. 2018;9(1):282. doi: 10.1186/s13287-018-1022-y. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 92.Qian D, Wei G, Xu C, et al. Bone marrow-derived mesenchymal stem cells (BMSCs) repair acute necrotized pancreatitis by secreting microRNA-9 to target the NF-κB1/p50 gene in rats. Sci Rep. 2017;7(1):581. doi: 10.1038/s41598-017-00629-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Li HY, He HC, Song JF, et al. Bone marrow-derived mesenchymal stem cells repair severe acute pancreatitis by secreting miR-181a-5p to target PTEN/Akt/TGF-β1 signaling. Cell Signal. 2020;66:109436. doi: 10.1016/j.cellsig.2019.109436. [DOI] [PubMed] [Google Scholar]
- 94.Wu XM, Ji KQ, Wang HY, et al. MicroRNA-339-3p alleviates inflammation and edema and suppresses pulmonary microvascular endothelial cell apoptosis in mice with severe acute pancreatitis-associated acute lung injury by regulating Anxa3 via the Akt/mTOR signaling pathway [retracted in: J Cell Biochem. 2021 Nov;122 Suppl 1:S107] J Cell Biochem. 2018;119(8):6704–6714. doi: 10.1002/jcb.26859. [DOI] [PubMed] [Google Scholar]
- 95.He RQ, Li XJ, Liang L, et al. The suppressive role of miR-542–5p in NSCLC: the evidence from clinical data and in vivo validation using a chick chorioallantoic membrane model. BMC Cancer. 2017;17(1):655. doi: 10.1186/s12885-017-3646-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Wu XM, Ji KQ, Wang HY, et al. microRNA-542–5p protects against acute lung injury in mice with severe acute pancreatitis by suppressing the mitogen-activated protein kinase signaling pathway through the negative regulation of P21-activated kinase 1 [retracted in: J Cell Biochem. 2021 Nov;122 Suppl 1:S117] J Cell Biochem. 2019;120(1):290–304. doi: 10.1002/jcb.27356. [DOI] [PubMed] [Google Scholar]
- 97.Wang T, Jiang L, Wei X, et al. MiR-21–3p aggravates injury in rats with acute hemorrhagic necrotizing pancreatitis by activating TRP signaling pathway. Biomed Pharmacother. 2018;107:1744–1753. doi: 10.1016/j.biopha.2018.08.164. [DOI] [PubMed] [Google Scholar]
- 98.Yan Z, Zang B, Gong X, et al. MiR-214–3p exacerbates kidney damages and inflammation induced by hyperlipidemic pancreatitis complicated with acute renal injury. Life Sci. 2020;241:117118. doi: 10.1016/j.lfs.2019.117118. [DOI] [PubMed] [Google Scholar]
- 99.Rivkin M, Simerzin A, Zorde-Khvalevsky E, et al. Inflammation-induced expression and secretion of microRNA 122 leads to reduced blood levels of kidney-derived erythropoietin and anemia. Gastroenterology. 2016;151(5):999–1010. doi: 10.1053/j.gastro.2016.07.031. [DOI] [PubMed] [Google Scholar]
- 100.Han YY, Wang CY, Yang L, et al. Significance of microRNA 216a, 324–5p and 29a expression in peripheral blood in patients with acute pancreatitis and their correlation with liver injury. Zhonghua Yi Xue Za Zhi. 2020;100(27):2126–2131. doi: 10.3760/cma.j.cn112137-20200103-00016. [DOI] [PubMed] [Google Scholar]