In a recent study, we exposed Streptococcus pneumoniae isolates to subinhibitory concentrations of six fluoroquinolones (FQs) (1). This subculturing led to resistance development. None of the FQs, however, was associated with a clearly lower potential to cause resistance development, measured as an increase in MIC-doubling dilutions. However, based on the limited data available, i.e., six isolates, further studies were needed to clarify whether resistance development is strain dependent or dichotomous due to differences in the FQ composition.
Thus, we repeatedly exposed 70 clinical S. pneumoniae strains for 6 days to ciprofloxacin (CIP), gatifloxacin (GAT), gemifloxacin (GEM), levofloxacin (LEV), and moxifloxacin (MOX) (1). Mean MICs (in micrograms per milliliter) for the original isolates were as follows: GEM, 0.017; MOX, 0.091; GAT, 0.175; LEV, 0.707; CIP, 0.788.
In order to analyze the rate of resistance development, we converted the MICs for all clinical strains and selected mutant isolates to a log scale (base 2). Initial values at time zero (original MICs) were subtracted from MICs on days 1 through 6. The slope of the MIC increase over the 6-day period was computed by linear regression and compared by analysis of variance with the Student-Newman-Keuls posthoc test; differences were considered statistically significant if P was <0.05. Judging from the squared correlation coefficients R2 (means ± standard errors: CIP, 0.916 ± 0.007; GAT, 0.754 ± 0.018; GEM, 0.926 ± 0.008; LEV, 0.899 ± 0.009; MOX, 0.755 ± 0.019), the linearity of the individual lines was good, meaning that the regression coefficients were a reasonable measure of the resistance development rate. The relatively low R2 values of GAT and MOX reflect low slope values rather than lack of linearity.
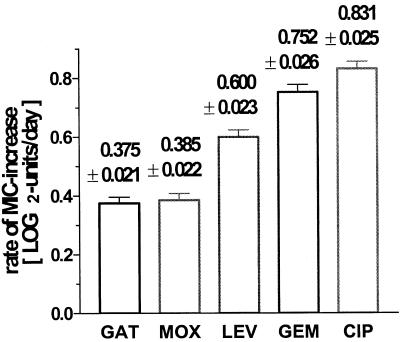
Nearly identical lines were found for CIP and GEM, both reaching rather high log2 MIC increases after 6 days (4.6 ± 0.14 and 4.3 ± 0.15 μg/ml), while much lower increases were measured for LEV (2.99 ± 0.14 μg/ml), MOX (2.20 ± 0.11 μg/ml), and GAT (2.16 ± 0.11 μg/ml). The rates of resistance development are statistically different between the drugs tested (Fig. 1). These differences, measured on the log scale, correspond to x-fold increases in the initial MICs per day: GAT, 1.30-fold; MOX, 1.30-fold; LEV, 1.52-fold; GEM, 1.68-fold; CIP, 1.78-fold. Thus, GAT and MOX, the two FQs with a methoxy moiety at the C-8 position, displayed the lowest propensity for causing resistance development.
FIG. 1.
The rate of resistance development as measured by the slopes of individual time-dependent increases in MICs of five FQs. The data are means with standard errors (n = 70 strains). GAT exhibits the lowest MIC increase, with a mean rate of <0.4 log2 units per day (corresponding to a 1.30-fold increase per day), while CIP shows the highest mean slope, with >0.8 log2 units per day (corresponding to a 1.78-fold increase per day).
Our data are supported by reports from the laboratory of Drlica and coworkers, who showed that GAT was less affected by changes in either DNA gyrase or topoisomerase IV (2, 5–8). Similar results were reported by A. Dalhoff for MOX (8th Int. Cong. Infect. Dis., poster 47.003, 1998; 37th Annu. Meet. Infect. Dis. Soc. Am., poster 102, 1999; 1st Int. Symp. Resist. Gram-Positive Infect., 2000). Data reported by other authors demonstrate that the molecular entity selected for the C-8 position of an FQ can enhance the drug's antibacterial activity (3, 4). In summary, the clinical relevance of a potentially lower propensity of an FQ with the C-8-methoxy moiety to develop resistance should be analyzed in animal studies as well as in surveillance studies to investigate FQ MIC increases for S. pneumoniae in combination with the use of different FQs.
REFERENCES
- 1.Boos M, Mayer S, Fischer A, Köhrer K, Scheuring S, Heisig P, Verhoef J, Fluit A C, Schmitz F-J. In-vitro development of resistance to six quinolones in Streptococcus pneumoniae, Streptococcus pyogenes, and Staphylococcus aureus. Antimicrob Agents Chemother. 2001;45:938–942. doi: 10.1128/AAC.45.3.938-942.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Dong Y, Xu C, Zhao X, Domagala J, Drlica K. Fluoroquinolone action against mycobacteria: effects of C-8 substituents on growth, survival, and resistance. Antimicrob Agents Chemother. 1998;42:2978–2984. doi: 10.1128/aac.42.11.2978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Ito T, Matsumoto M, Nishino T. Improved bactericidal activity of Q-35 against quinolone-resistant staphylococci. Antimicrob Agents Chemother. 1995;39:1522–1525. doi: 10.1128/aac.39.7.1522. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Kitamura A, Hoshino K, Kimura Y, Hayakawa I, Sato K. Contribution of the C-8 substituent of DU-6859a, a new potent fluoroquinolone, to its activity against DNA gyrase mutants of Pseudomonas aeruginosa. Antimicrob Agents Chemother. 1995;39:1467–1471. doi: 10.1128/aac.39.7.1467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Lu T, Zhao X, Drlica K. Gatifloxacin activity against quinolone-resistant gyrase: allele-specific enhancement of bacteriostatic and bactericidal activities by the C-8-methoxy group. Antimicrob Agents Chemother. 1999;43:2969–2974. doi: 10.1128/aac.43.12.2969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Zhao B Y, Pine R, Domagala J, Drlica K. Fluoroquinolone action against clinical isolates of Mycobacterium tuberculosis: effects of a C-8 methoxyl group on survival in liquid media and in human macrophages. Antimicrob Agents Chemother. 1999;43:661–666. doi: 10.1128/aac.43.3.661. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Zhao X, Wang J Y, Xu C, Dong Y, Zhou J, Domagala J, Drlica K. Killing of Staphylococcus aureus by C-8-methoxy fluoroquinolones. Antimicrob Agents Chemother. 1998;42:956–958. doi: 10.1128/aac.42.4.956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Zhao X, Xu C, Domagala J, Drlica K. DNA topoisomerase targets of the fluoroquinolones: a strategy for avoiding bacterial resistance. Proc Natl Acad Sci USA. 1997;94:13991–13996. doi: 10.1073/pnas.94.25.13991. [DOI] [PMC free article] [PubMed] [Google Scholar]