ABSTRACT
Background
There is concern that the PUFA composition of ready-to-use therapeutic food (RUTF) for the treatment of severe acute malnutrition (SAM) is suboptimal for neurocognitive recovery.
Objectives
We tested the hypothesis that RUTF made with reduced amounts of linoleic acid, achieved using high-oleic (HO) peanuts without added DHA (HO-RUTF) or with added DHA (DHA-HO-RUTF), improves cognition when compared with standard RUTF (S-RUTF).
Methods
A triple-blind, randomized, controlled clinical feeding trial was conducted among children with uncomplicated SAM in Malawi with 3 types of RUTF: DHA-HO-RUTF, HO-RUTF, and S-RUTF. The primary outcomes, measured in a subset of subjects, were the Malawi Developmental Assessment Tool (MDAT) global z-score and a modified Willatts problem-solving assessment (PSA) intention score for 3 standardized problems, measured 6 mo and immediately after completing RUTF therapy, respectively. MDAT domain z-scores, plasma fatty acid content, anthropometry, and eye tracking were secondary outcomes. Comparisons were made between the novel PUFA RUTFs and S-RUTF.
Results
Among the 2565 SAM children enrolled, mean global MDAT z-scores were –0.69 ± 1.19 and –0.88 ± 1.27 for children receiving DHA-HO-RUTF and S-RUTF, respectively (difference 0.19, 95% CI: 0.01, 0.38). Children receiving DHA-HO-RUTF had higher gross motor and social domain z-scores than those receiving S-RUTF. The PSA problem 3 scores did not differ by dietary group (OR: 0.92, 95% CI: 0.67, 1.26 for DHA-HO-RUTF). After 4 wk of treatment, plasma phospholipid EPA and α-linolenic acid were greater in children consuming DHA-HO-RUTF or HO-RUTF when compared with S-RUTF (for all 4 comparisons P values < 0.001), but only plasma DHA was greater in DHA-HO-RUTF than S-RUTF (P < 0.001).
Conclusions
Treatment of uncomplicated SAM with DHA-HO-RUTF resulted in an improved MDAT score, conferring a cognitive benefit 6 mo after completing diet therapy. This treatment should be explored in operational settings. This trial was registered at clinicaltrials.gov as NCT03094247.
Keywords: severe acute malnutrition, ready-to-use therapeutic food, PUFA, cognition, MDAT, DHA
Graphical Abstract
Graphical Abstract.
See corresponding editorial on page 1257.
Introduction
Severe acute malnutrition (SAM) is a global insult to the young child's developing mind and body. Therapeutic foods for SAM were largely designed to provide the nutrients known to affect recovery of anthropometry and muscle mass (1). Vegetable oil-rich, peanut-based ready-to-use therapeutic food (RUTF) is the standard of care for most SAM because it can be used safely at home in the context of utmost poverty (2). It has been assumed that the brain and other viscera would receive what is needed for recovery from standard RUTF (S-RUTF).
Dietary ω-3 PUFAs are essential to normal brain development and function. Brain accretion of the key neural structural component, ω-3 DHA, accelerates in the third trimester of gestation and continues until the age of 18 y (3, 4). Tissue DHA is derived from either preformed dietary DHA or a precursor, typically plant-derived α-linolenic acid (ALA). ALA is less efficient in supplying brain DHA than dietary DHA (5–7). Omega-6 linoleic acid (LA) is metabolized by the same desaturases (FADS2, FADS1) and elongases (ELOVL2, ELOVL5) as ω-3 PUFAs, and uses the same pathways to incorporate into membranes (8, 9). LA intake required to eliminate overt clinical abnormalities and support growth in infants is about 2% of energy or about 1% of fatty acids from diets deriving half their energy from fat (10). RUTF and breastmilk are examples of such diets. Suppression of ω-3 PUFA tissue accretion by excess dietary LA is an impactful clinical phenomenon (11). High LA dietary vegetable oils create a metabolic demand for all ω-3 PUFAs, thus requiring higher dietary DHA intake to produce similar tissue DHA concentrations (12).
Societal justice and stability require citizens with high-level mental functioning to support executive problem solving and affect regulation needed for resilient responses. Perinatal brain development depends on a balanced supply of brain-specific nutrients. Overwhelming amounts of LA in vegetable oils suppress trace amounts of ALA when the brain is rapidly accreting DHA. This inhibits neurobehavioral development at the levels of gene expression, hormonal balance, affect, and adaptive responses to environmental cues (13). Omega-3–deficient diets cause poor impulse control leading to potentiated stress response, increased depression and aggression, and poorer cognitive performance (14–16). Evidence-based guidelines include EPA and DHA supplements as a treatment of attention deficit hyperactivity disorder, autism spectrum disorder, and major depressive disorder (17, 18).
The potential effects of excess LA and limited ALA in RUTF on cognition have not been measured. Our previous study showed that RUTF with excess LA caused a 25% reduction in circulating DHA in 4 wk (19). This clinical trial tested the hypothesis that RUTF made with reduced amounts of LA, achieved by using high-oleic (HO) peanuts and palm oil without added DHA (HO-RUTF) or with added DHA (DHA-HO-RUTF), confers lasting cognition improvement when compared with S-RUTF. Cognition was measured by the Malawi Developmental Assessment Tool (MDAT), a standardized, validated battery of exercises and questions, and a modified Willatts problem-solving assessment (PSA) intention score.
Methods
Study design
This triple-blind, randomized controlled clinical trial compared cognition in children treated for uncomplicated SAM receiving 1 of 3 RUTFs: HO-RUTF, DHA-HO-RUTF, or S-RUTF. The primary outcomes were the MDAT global z-score and PSA intention scores. Secondary outcomes included MDAT domain z-scores, recovery rates, anthropometric growth rates, saccadic reaction time, visual paired comparison, novelty preference score, mean fixation during familiarization, and adverse events. Detailed descriptions of the study methods are provided in the Online Supplementary Material Protocol.
All sample size calculations were performed using G*Power (3.1.9.7) with 2-tailed testing at a significance level of 0.05 and power of 0.80 (20). A total of 300 children per group were required to detect a difference in MDAT global z-score of 0.25, assuming an SD of 1.1 (21–23). The difference of 0.25 z-scores was chosen based on prior nutrition-based effect sizes on developmental scores as well as MDAT-specific changes seen in association with illness (0.14–0.19) (24, 25). Assuming a 25% loss to follow-up, 400 participants per group were designated for testing. For the PSA, we assumed 20% of participants would not complete testing; thus, a total of 300 children per group were needed to detect a standardized effect size of 0.25. This effect size was chosen based on previous studies of healthy infants under controlled conditions, where standardized effect sizes ranged from 0.4–0.5 (26, 27). Given the extended age range relative to prior trials, the proximity of participants to SAM, and testing environments, we chose a smaller effect size for sample size calculations.
Sample size was also determined for the key secondary outcome, recovery. A total of 900 participants per group was required to detect an improvement in recovery of 4% assuming an expected recovery rate of 89% and default rate of 5%, both of which were determined based on prior operational experience in Malawi (28, 29). This sample size was chosen for the overall trial because it exceeded those required for the primary outcomes.
Participants were randomly assigned in a 1:1:1 ratio into the 3 intervention groups. Computer-generated block randomization lists were created by a remote study team member in blocks of 24 using 6 colors, 2 of which corresponded to each food. The SNOSE (sequentially numbered opaque sealed envelopes) method for allocation concealment was implemented. Cards with group color codes known only to the remote study team member were kept in opaque sealed envelopes prepared by a field research assistant not responsible for the enrollment or consenting of study participants. Participants were assigned to their food group when they drew an opaque sealed envelope which a study nurse then unsealed, revealing 1 of the colored cards.
Study foods were dispensed in identical packets aside from colored stickers. Food was prepared and labeled by study members who had no role in the distribution of study foods. The 3 study foods underwent extensive testing to minimize differences in appearance and taste. Except for 1 remote member of the study team, all research personnel, including nurses, investigators, and laboratory personnel, were blinded to the color code. The unblinded individual did not evaluate any of the study subjects, nor did she analyze the primary outcomes. The trial is registered at clinicaltrials.gov as NCT03094247.
Subjects and setting
The trial was conducted at 28 clinics in rural Southern Malawi between October 2017 and December 2020. Most families in the study farm for subsistence, with corn as the staple crop. Breastfeeding is ubiquitous throughout the first year of life. Homes are built from mud with thatch or corrugated metal sheet roofing. Plumbing and electricity are exceedingly rare, and water is obtained from boreholes, wells, or rivers.
Children aged 6–59 mo were eligible for enrollment if they had uncomplicated SAM, which was defined as a midupper arm circumference (MUAC) <11.5 cm, and/or weight-for-height z-score < –3, and/or bilateral pitting edema, with an adequate appetite as determined by a 30 g test feeding. Exclusion criteria included participation in a feeding program in the prior 1 mo, presence of developmental delay, a chronic debilitating medical condition, peanut allergy, or a hearing or vision problem.
The trial was approved by the Human Studies Committee of Washington University and the College of Medicine Research and Ethics Committee of the University of Malawi. Nurses fluent in Chichewa explained the trial to each child's caregiver and obtained verbal and written consent.
Participation
Upon enrollment, baseline demographic, socioeconomic, and health data were collected. Weight, length, and MUAC were measured. Caregivers received counseling, a 2-wk supply of RUTF, and a 7-d course of amoxicillin. Healthy twins of subjects were given an allotment of RUTF as well. Follow-up visits were scheduled at 2-wk intervals, at which time anthropometric measurements were repeated and caregivers were asked about adherence and symptoms. Children were fed until reaching a clinical outcome or for a maximum of 6 bi-weekly follow-up visits. The anthropometry methods and definitions of the clinical outcomes are described in the Online Supplementary Material Protocol (Section 6.10).
Beginning in February 2018, all participants aged <2 y were invited for PSA assessment within 4 wk of their clinical outcome. Beginning in March 2018, all participants under the age of 30 mo were asked to return to clinic 5–7 mo after SAM outcome for testing with the MDAT and eye-tracking testing using the Tobii X2-60 eye tracker.
Children that either missed fortnightly assessments during therapeutic feeding or neurocognitive testing were sought on 2 extra occasions by health surveillance assistants and asked to return for the scheduled treatment or testing.
Study foods
All 3 RUTF formulations were produced by Project Peanut Butter in Lunzu, Malawi, meeting all international quality and nutrient specifications. DHA-HO-RUTF and HO-RUTF were formulated to reduce LA and increase the ω-3 PUFA content (Table 1, Supplemental Tables 1 and 2, Online Supplementary Material Protocol Section 6.5). Reductions in LA were achieved by using high-oleic peanuts. Increases in ω-3 PUFAs were achieved by the addition of perilla oil. DHA was enhanced in one of the RUTFs by the addition of encapsulated fish oil. The formulation was demonstrated to be acceptable in a pilot trial (19).
TABLE 1.
Composition of study ready-to-use therapeutic foods1
| DHA-HO-RUTF | HO-RUTF | S-RUTF | |
|---|---|---|---|
| Ingredient | |||
| Milk powder, nonfat, dry, g | 22.0 | 22.0 | 27.3 |
| Milk, sweet whey powder, g | 6.1 | 6.1 | 0.0 |
| Palm oil, g | 14.0 | 14.0 | 16.0 |
| Canola oil, g | 0.0 | 0.0 | 5.1 |
| Sugar, g | 24.5 | 26.0 | 23.0 |
| High-oleic peanuts, g | 25.0 | 25.0 | 0.0 |
| Standard, high ω-6 peanuts, g | 0.0 | 0.0 | 23.7 |
| Perilla oil, g | 3.0 | 3.0 | 0.0 |
| Hydrogenated vegetable oil,2 g | 1.0 | 1.0 | 2.0 |
| Micronutrient powder, g | 2.9 | 2.9 | 2.9 |
| DHA-containing oil, g | 1.5 | 0.0 | 0.0 |
| Macronutrient | |||
| Energy, kcal | 532.0 | 531.0 | 541.0 |
| Protein, g | 15.2 | 15.2 | 15.7 |
| Protein, dairy, g | 8.9 | 8.9 | 9.8 |
| Lipids, g | 30.2 | 29.5 | 32.7 |
| Linoleic acid, g | 2.2 | 2.1 | 5.8 |
| α-Linolenic acid, g | 1.5 | 1.4 | 0.5 |
| DHA, mg | 72.0 | 0.0 | 0.0 |
| EPA, mg | 14.0 | 0.0 | 0.0 |
Content expressed in per 100 g. DHA-HO-RUTF, DHA added to RUTF made with high-oleic acid peanuts; HO-RUTF, RUTF made with high-oleic acid peanuts; RUTF, ready-to-use therapeutic food; S-RUTF, standard RUTF.
This was a proprietary commercial product, Trancendim 180 (Caravan Foods).
Neurocognitive testing
MDAT has been validated within the study context (30). We performed MDAT assessments between 5 and 7 mo after an SAM outcome was reached to assess the effect of the RUTFs on neurodevelopment. No intervention was performed between the SAM outcome and the MDAT testing. MDAT contains 136 items across 4 domains, 3 of which are assessed by direct observation of the child (gross motor, fine motor, and language), whereas the fourth (social) is assessed via caregiver interview. These domain scores are then combined into a global score and compared with normal population reference values to provide age-adjusted z-scores (30). In line with prior protocols, failure of any 6 consecutive items resulted in failure of that domain and skipping to the subsequent domain (21). Global z-score outliers were defined after evaluation of the score distribution, and values < –5 were excluded.
A modified version of the Willatts PSA was performed within 4 wk of the outcome from treatment of SAM (26). This time point was chosen to detect the immediate effect of the intervention RUTFs. The assessment was modified to accommodate testing a wider age range than those used in prior studies and so contained 3 problems of increasing complexity (27). The goal of the assessment was to judge intentionality in means-end task completion. All assessments were video recorded for subsequent coding.
Each problem contained a barrier between the child and the goal, a toy of her or his choosing. In problem 1, the toy was placed on a cloth and distanced from the child such that the cloth had to be pulled by the child to reach the toy. In problem 2, the toy was covered with an opaque cloth such that the child had to remove the cover to obtain the toy. Problem 3 was an amalgam of problems 1 and 2, wherein the toy was placed on a cloth and under a cover, such that the child had to pull the apparatus toward them and uncover the toy to obtain it.
Videos were coded using Behavioral Observation Research Interactive Software (31). Coders were trained to achieve >0.90 agreement by κ statistic for intention scores on a random sample of 20 test videos before coding for the trial. High reliability was maintained throughout the coding period by double-coding >50% of videos followed by a joint resolution of discrepancies. In a modification of the procedure of Willatts et al. problem-solving behaviors, problems were coded for 2 primary components, each on a scale of 0–2 and each assessing an aspect of intentionality: attentional orienting to each problem subgoal and the execution of behaviors on each subgoal. Problems 1 and 2 were scored 0–4, whereas problem 3 had 2 subgoals (moving the cloth and removing the cover) and thus was scored 0–8. “No score” was a possible outcome for all problems, and reasons for this result were recorded.
Two eye-tracking tests were performed as secondary cognitive outcomes: a visual paired comparison task and the infant orienting with attention task. Identical procedures to those previously described were used (22). The visual paired task consisted of 4 trials of African faces. The infant orienting task measured saccadic reaction time in a standardized manner. The testing sequence for paired comparison task and orienting attention task was randomized.
All neurocognitive assessors underwent extensive training and were required to pass periodic evaluations before administering the tests and throughout the trial. Please see the study Online Supplementary Material Protocol for further details (Section 11).
Plasma sampling and analysis
The purpose of blood sampling was to characterize the concentrations of fatty acids in the study groups and thus only a subset of children were sampled. Blood collection and analysis followed best practices (32). After receiving 4 wk of RUTF, blood was collected by venipuncture and placed in an EDTA tube. Plasma was then separated from red cells and held at –20°C until transferred to the laboratories of JT Brenna at the Dell Pediatric Research Institute in Austin, TX. Plasma phospholipids were chosen as the preferred lipid pool because they respond in a matter of weeks to changes in the diet compared with months for RBCs, but are stable to fatty acids from recent meals which are largely triacylglycerols. RBC PUFAs also degrade at –20°C via peroxidation catalyzed by iron release from ruptured cells (30).
For analyses, plasma phospholipids were isolated using an automated 3-phase liquid-liquid extraction method (33). Samples were then transmethylated to generate fatty acid methyl esters according to our routine methods (34). These fatty acid derivatives were detected and quantified by GC-flame ionization detection and structure confirmed by a specialized GC tandem MS method. Quantitative standards of 25 fatty acid methyl esters were then used to quantify the amounts of each detected. Results are reported as a weight percentage of total fatty acid identified. These methods are detailed in the Online Supplementary Material Protocol (Section 12).
Statistical analysis
Analyses were performed by a blinded investigator using a modified intention to treat methodology wherein children who were discovered not to meet enrollment criteria were excluded from analysis. Baseline characteristics were summarized as means ± SD, medians (IQR), or n (%). Anthropometric indices were calculated with WHO Anthro version 3.1 (WHO). Rates of weight, MUAC, and length gain were calculated over the entire period of feeding.
Outcomes from the DHA-HO-RUTF and HO-RUTF groups were compared to S-RUTF. Continuous outcomes were compared using Student's t-test. Categorical outcomes were compared using the chi-square test. PSA scores were analyzed using ordinal logistic regression with age as a covariate. An interaction term between intervention group and age was offered into the model but was not significant for any problem and thus was not included in the final model. The proportional odds assumption was assessed graphically and deemed not violated by producing a series of binary logistic regressions at cut-points for all outcome values >2. Significance testing was restricted to pairwise comparisons of the 2 primary outcomes and key secondary outcomes, namely the domain MDAT z-scores and recovery rates. In all other cases, differences with unadjusted 95% CIs were calculated and reported. Analysis was completed using R version 4.0.1 (R Foundation for Statistical Computing) and SPSS version 27 (SPSS Inc.) (35).
Results
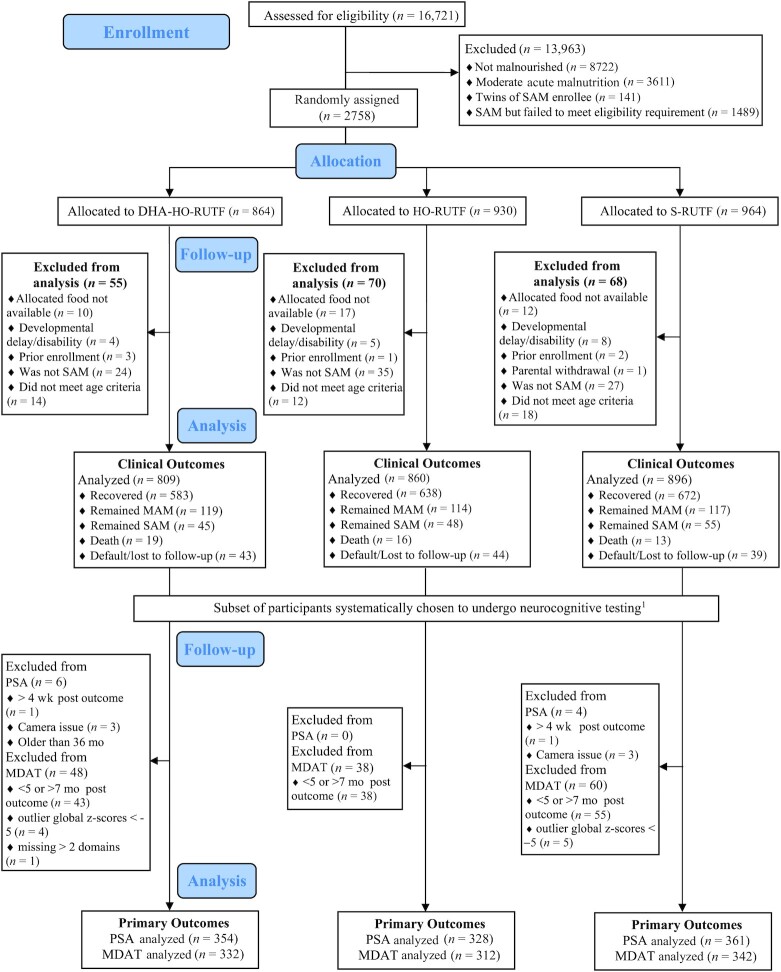
Of the 2758 children with SAM identified between October 2017 and December 2020, 2565 children were included in the study (Figure 1). In 2020, there were 3 stock-outs of RUTFs due to ingredient importation restrictions related to the COVID-19 pandemic. This precluded randomization as it was designed, and so subjects were randomly assigned among the available RUTF colors. Additionally, 39 participants who had been randomly assigned were not able to continue their study foods due to stock-outs and thus were excluded from analysis. This is described in Section 9 of the Online Supplementary Material Protocol. Baseline characteristics were similar among the 3 groups, both in the entire study sample and within the MDAT and problem-solving assessment subgroups (Table 2, Supplemental Tables 3 and 4).
FIGURE 1.
CONSORT diagram of study participation. DHA-HO-RUTF, high-oleic acid RUTF with added DHA; HO-RUTF, high-oleic acid RUTF; MAM, moderate acute malnutrition; MDAT, Malawi Developmental Assessment Tool; PSA, problem-solving assessment; RUTF, ready-to-use therapeutic food; SAM, severe acute malnutrition; S-RUTF, standard RUTF. 1For the PSA, beginning in February 2018, children aged <2 y at time of SAM outcome were invited to undergo neurocognitive testing within 4 wk of SAM outcome. For the MDAT, beginning in March 2018, children aged <30 mo at time of SAM outcome were invited to return 5–7 mo after their SAM outcome to undergo neurocognitive testing.
TABLE 2.
Baseline characteristics of Malawian children receiving study foods1
| DHA-HO-RUTF | HO-RUTF | S-RUTF | |
|---|---|---|---|
| (N = 809) | (N = 860) | (N = 896) | |
| Female sex, no. (%) | 452 (55.9) | 469 (54.5) | 527 (58.8) |
| Age, mo., median (IQR) | 12.1 (7.9–20.4) | 12.2 (7.8–21.2) | 12.7 (8.1–21.5) |
| Edematous, no. (%) | 274 (33.9) | 302 (35.1) | 293 (32.7) |
| Midupper arm circumference, cm | 11.6 ± 1.2 | 11.6 ± 1.2 | 11.6 ± 1.1 |
| MUAC <11.5 cm, no. (%) | 534 (66) | 545 (63.4) | 600 (67) |
| Weight-for-height z-score | −1.9 ± 1.2 | −1.9 ± 1.2 | −2.0 ± 1.2 |
| WHZ ≤ –3, no. (%) | 167 (20.6) | 181 (21.0) | 186 (20.8) |
| Height-for-age z-score | −3.3 ± 1.5 | −3.3 ± 1.4 | −3.2 ± 1.4 |
| HAZ ≤ –2, no./total no. (%) | 667/808 (82.5) | 726/858 (84.6) | 746/896 (83.3) |
| Fever in past 2 wk, no./total no. (%) | 461/805 (57.3) | 518/857 (60.4) | 554/893 (62.0) |
| Diarrhea in past 2 wk, no./total no. (%) | 446/805 (55.4) | 474/858 (55.2) | 484/891 (54.3) |
| Child breastfed, no./total no. (%) | 520/808 (64.4) | 565/858 (65.9) | 575/893 (64.4) |
| HIV-seropositive, no./total no. tested (%) | 19/265 (7.2) | 25/301 (8.3) | 23/311 (7.4) |
| Mother alive, no./total no. (%) | 783/807 (97) | 827/860 (96.2) | 876/895 (97.9) |
| Number of siblings, median (IQR) | 2 (0–3) | 2 (1–3) | 2 (0–3) |
| Thatch roof, no./total no. (%) | 607/803 (75.6) | 659/859 (76.7) | 689/890 (77.4) |
| Radio in home, no./total no. (%) | 178/807 (22.1) | 196/854 (23.0) | 168/893 (18.8) |
| Clean water source, no./total no. (%) | 761/807 (94.3) | 795/857 (92.8) | 834/895 (93.2) |
Plus-minus values are means ± SD. DHA-HO-RUTF, DHA added to ready-to-use therapeutic food made with high-oleic acid peanuts; HAZ, height-for-age z-score; HO-RUTF, RUTF with high-oleic acid peanuts; MUAC, midupper arm circumference. RUTF, ready-to-use therapeutic food.
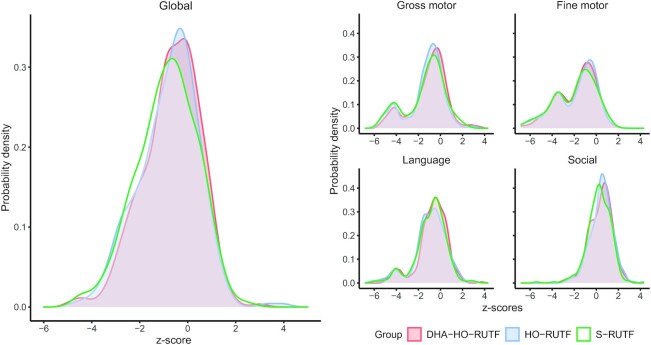
For the primary outcome analysis, 92% of children who were asked to undergo PSA testing within 4 wk of SAM outcome did so, and 82% of children who were asked to return for MDAT assessment 6 mo post SAM outcome returned for testing. Children receiving DHA-HO-RUTF had higher MDAT global z-scores than those receiving S-RUTF at ∼6 mo post SAM outcome, whereas children receiving HO-RUTF did not. Children receiving DHA-HO-RUTF had higher gross motor and social z-scores than those receiving S-RUTF, and children receiving HO-RUTF had higher social z-scores than those receiving S-RUTF at ∼6 mo post SAM outcome (Table 3, Supplemental Table 3). Probability density distributions for global and domain MDAT z-scores for each group are shown in Figure 2, with means shown in Supplemental Figure 1. When stratified by age, the probability density distributions of global MDAT z-scores show that children <12 mo at the time of SAM resemble healthy children, whereas children >18 mo at the time of SAM display scores ∼ –2 z-scores worse (Supplemental Figure 2).
TABLE 3.
Outcomes and adverse events, according to intervention group1
| DHA-HO-RUTF | HO-RUTF | S-RUTF | DHA-HO-RUTF vs. S-RUTF comparison2 | HO-RUTF vs. S-RUTF comparison2 | ||||
|---|---|---|---|---|---|---|---|---|
| N | N | N | ||||||
| Cognitive outcomes | ||||||||
| Malawi Developmental Assessment Tool z-scores | ||||||||
| Global | 332 | −0.69 ± 1.19 | 312 | −0.80 ± 1.25 | 342 | −0.88 ± 1.27 | 0.19 (0.01, 0.38)3 | 0.08 (−0.11, −0.27) |
| Gross motor domain | 331 | −1.08 ± 1.66 | 312 | −1.34 ± 1.67 | 342 | −1.37 ± 1.78 | 0.29 (0.03, 0.55)4 | 0.02 (−0.24, 0.29) |
| Fine motor domain | 323 | −1.81 ± 1.71 | 306 | −1.83 ± 1.80 | 337 | −1.91 ± 1.83 | 0.14 (−0.17, 0.37) | 0.07 (−0.21, 0.35) |
| Language domain | 331 | −0.73 ± 1.35 | 312 | −0.93 ± 1.51 | 342 | −0.90 ± 1.41 | 0.17 (−0.04, 0.38) | −0.03 (−0.25, 0.19) |
| Social domain | 332 | 0.40 ± 1.01 | 311 | 0.49 ± 1.00 | 342 | 0.24 ± 1.04 | 0.16 (0.00, 0.31)5 | 0.24 (0.09, 0.40)6 |
| Problem-solving assessment intention scores | ||||||||
| Problem 1, no. (%) with perfect score | 298 | 90 (30) | 281 | 88 (31) | 292 | 90 (31) | 0.99 (0.66, 1.19)7 | 0.89 (0.74, 1.31)7 |
| Problem 2, no. (%) with perfect score | 293 | 169 (58) | 283 | 151 (53) | 294 | 168 (57) | 0.97 (0.71, 1.33)7 | 0.88 (0.64, 1.21)7 |
| Problem 3, no. (%) with perfect score | 236 | 50 (21) | 241 | 54 (22) | 238 | 51 (21) | 0.92 (0.67, 1.26)7 | 1.01 (0.74, 1.38)7 |
| Eye-tracking | ||||||||
| Infant oriented with attention response time, ms | 217 | 430 ± 106 | 223 | 431 ± 106 | 254 | 418 ± 96 | 12 (−10, 34) | 13 (−9, 36) |
| Visual paired comparison novelty preference score | 239 | 0.60 ± 0.1 | 238 | 0.58 ± 0.1 | 290 | 0.59 ± 0.1 | 0.01 (−0.01, 0.03) | −0.01 (−0.03, 0.01) |
| Mean fixation duration, ms | 248 | 330 ± 152 | 244 | 358 ± 154 | 297 | 343 ± 162 | −13 (−45, 19) | 15 (−17, 47) |
| Programmatic outcomes | ||||||||
| Recovered, no. (%) | 809 | 583 (72.1) | 860 | 638 (74.2) | 896 | 672 (75.0) | −2.9 (−7.1, 1.3) | −0.8 (−4.9, 3.3) |
| Remained malnourished, no. (%) | 809 | 164 (20.3) | 860 | 162 (18.8) | 896 | 172 (19.2) | 1.1 (−2.7, 4.9) | −0.4 (−4.1, 3.3) |
| Improved to moderate acute malnutrition, no. (%) | 809 | 119 (14.7) | 860 | 114 (13.3) | 896 | 117 (13.1) | 1.6 (−1.7, 4.9) | 0.2 (−3.0, 3.4) |
| Remained severely malnourished, no. (%) | 809 | 45 (5.6) | 860 | 48 (5.6) | 896 | 55 (6.1) | 1.1 (−2.7, 1.7) | −0.5 (−2.7, 1.7) |
| Died, no. (%) | 809 | 19 (2.3) | 860 | 16 (1.9) | 896 | 13 (1.5) | 0.8 (−0.5, 2.1) | 0.4 (−0.8, 1.6) |
| Defaulted, no. (%) | 809 | 43 (5.3) | 860 | 44 (5.1) | 896 | 39 (4.4) | 0.9 (−1.1, 2.9) | 0.7 (−0.13, 2.7) |
| Anthropometric outcomes | ||||||||
| Rate of weight gain, g/kg/d | 786 | 3.7 ± 4.0 | 836 | 3.8 ± 3.8 | 875 | 4.1 ± 3.6 | −0.3 (−0.7, 0.0) | −0.3 (−0.6, 0.1) |
| Rate of midupper arm circumference gain, mm/d | 786 | 0.27 ± 0.27 | 836 | 0.26 ± 0.26 | 875 | 0.29 ± 0.27 | −0.02 (−0.04, 0.01) | −0.03 (−0.05, 0.00) |
| Rate of length gain, mm/d | 786 | 0.37 ± 0.36 | 836 | 0.36 ± 0.39 | 872 | 0.35 ± 0.33 | 0.02 (−0.01, 0.06) | 0.01 (−0.02, 0.05) |
| Adverse events, first 2 wk of intervention | ||||||||
| Fever, no. (%) | 785 | 209 (27) | 836 | 217 (26) | 875 | 269 (31) | −4.0 (−8.4, 0.4) | −5.0 (−9.3, −0.7) |
| Diarrhea, no. (%) | 784 | 242 (31) | 834 | 247 (30) | 874 | 292 (33) | −2.0 (−6.5, 2.5) | −3.0 (−7.4, 1.4) |
| Not eating well, no. (%) | 784 | 11 (1) | 836 | 17 (2) | 873 | 15 (2) | −1.0 (−2.2, 0.2) | 0.0 (−1.3, 1.3) |
Plus-minus values are means ± SD. Significance testing was performed for primary outcomes only. Malawi Developmental Assessment Tool and modified Willatts problem-solving assessment. Pairwise comparisons for continuous variables were performed with Student's t-test. Comparisons below P = 0.05 threshold are indicated with superscript numbers with values shown in this legend. DHA-HO-RUTF, DHA added to ready-to-use therapeutic food made with high-oleic acid peanuts; HO-RUTF, high-oleic acid ready-to-use therapeutic food; S-RUTF, standard ready-to-use therapeutic food.
All values are differences with 95% CIs unless otherwise noted.
P = 0.044
P = 0.031
P = 0.047
P = 0.002
This value is the OR with 95% CI.
FIGURE 2.
Probability density plots of Malawi Developmental Assessment Tool global and domain z-scores of children receiving RUTF made with high-oleic acid peanuts with added DHA (DHA-HO-RUTF), RUTF made with high-oleic acid peanuts but without added DHA (HO-RUTF), or standard RUTF (S-RUTF). Probability densities were constructed using kernel density estimation. Global nDHA-HO-RUTF = 332, nHO-RUTF = 312, nS-RUTF = 342. Gross motor domain nDHA-HO-RUTF = 331, nHO-RUTF = 312, nS-RUTF = 342. Fine motor nDHA-HO-RUTF = 323, nHO-RUTF = 306, nS-RUTF = 337. Language nDHA-HO-RUTF = 331, nHO-RUTF = 312, nS-RUTF = 342, nDHA-HO-RUTF = 332, nHO-RUTF = 311, nS-RUTF = 342. RUTF, ready-to-use therapeutic food.
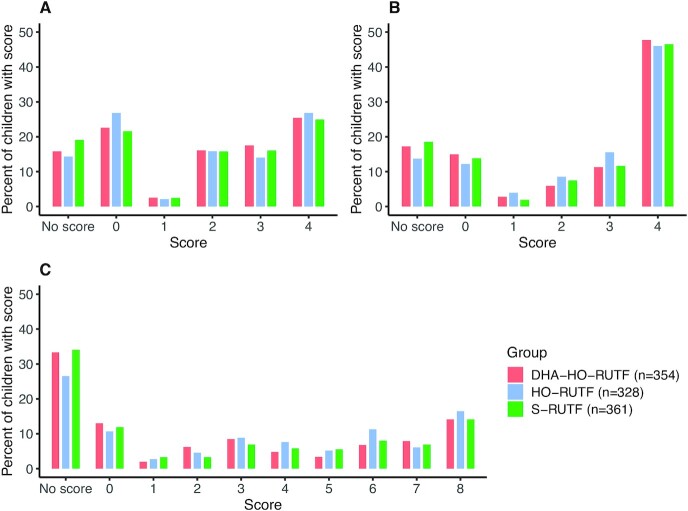
HO-RUTF or DHA-HO-RUTF did not lead to superior PSA scores compared with S-RUTF (Table 3, Figure 3). Inability to participate in problem 3 adequately to obtain a score occurred more often than anticipated and resulted in an underpowered analysis for PSA. Predicted probabilities from the ordinal logistic regression model showed that intention scores were strongly influenced by age and varied most in problem 3 (Supplemental Figure 3).
FIGURE 3.
Problem-solving assessment intention scores of children receiving RUTF made with high-oleic acid peanuts with added DHA (DHA-HO-RUTF), RUTF made with high-oleic acid peanuts but without added DHA (HO-RUTF), or standard RUTF (S-RUTF). Percentages of children in each intervention group with each score are shown. Overall, children in each study food had similar intention scores in all 3 problems; the differences between food groups were not significant. The lowest and highest scores possible in each problem were the most common results among children with scores. Children with “No Score” were unable to engage in the task. RUTF, ready-to-use therapeutic food.
Neither intervention RUTF led to superior eye-tracking results, anthropometric recovery, rates of gain in weight, length, or MUAC compared with S-RUTF (Table 3). Potential adverse events, including reports of fever, diarrhea, food intolerance, and poor appetite, were not higher in the intervention groups compared with S-RUTF, nor were default rates different between groups (Table 3).
Summary statistics for enrollment characteristics and analysis of primary and key secondary outcomes were repeated after excluding those children enrolled during each of the 3 stock-out periods (271 children excluded, Supplemental Tables 5–6). The groups remained balanced at baseline and the point estimates were similar for all primary and secondary outcomes. Exploratory analysis of MDAT results by subgroup is shown in Supplemental Figure 4. Linear and ordinal logistic regression models assessed the effects of DHA and HO in RUTF on MDAT global z-scores and PSA intentions scores, respectively. Results are shown in Supplemental Table 7.
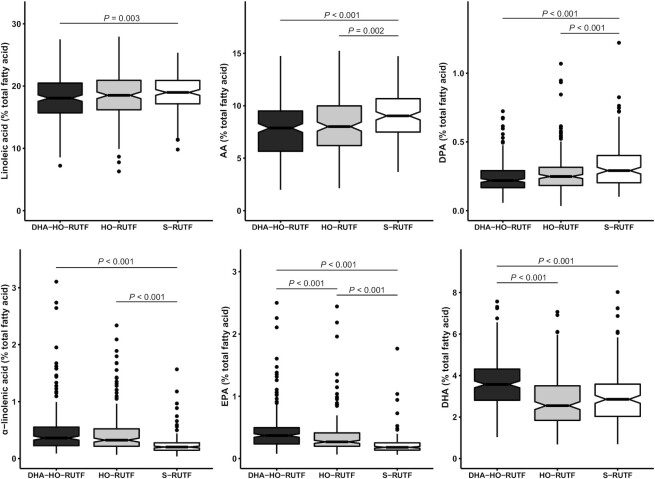
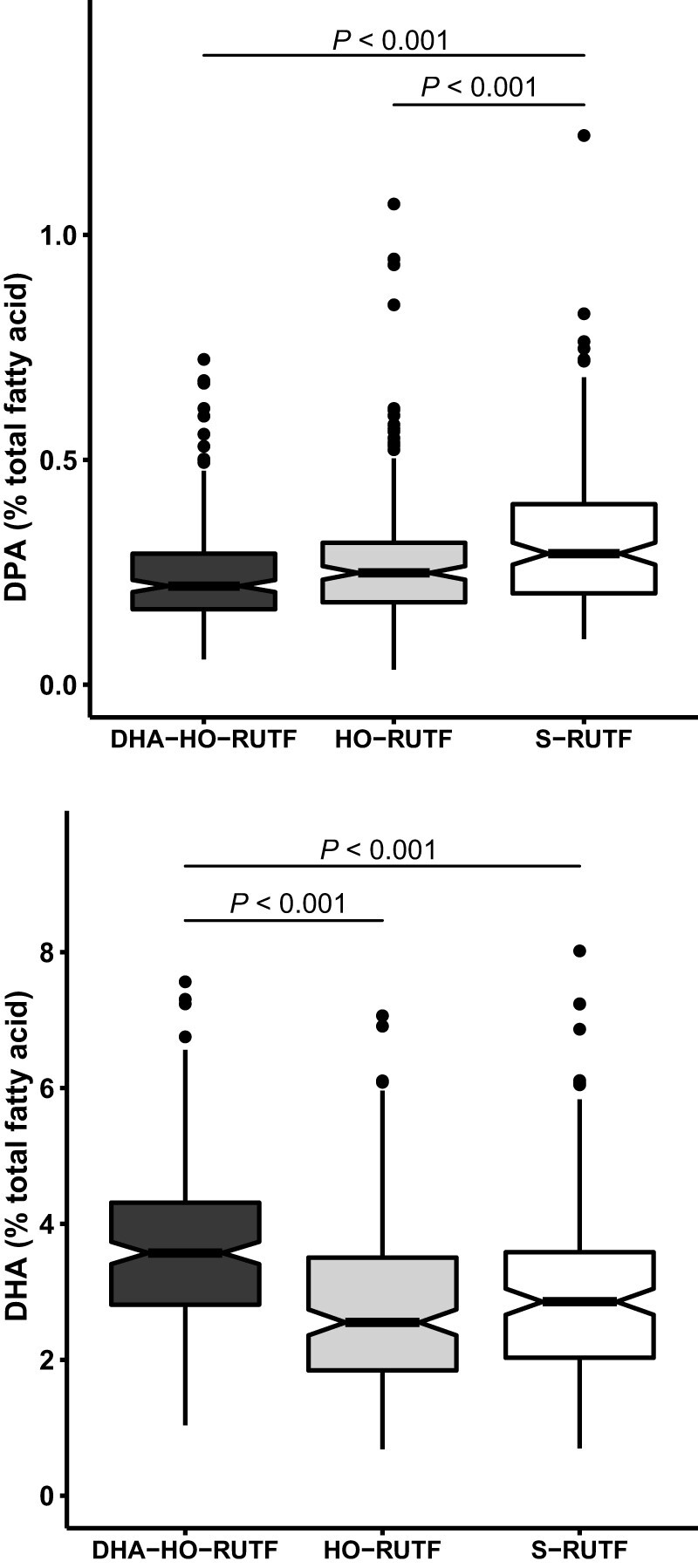
Six key plasma PUFA concentrations were selected to describe the biochemical changes induced by HO-RUTF and DHA-HO-RUTF (Figure 4). LA and ALA are the primary dietary ω-6 and ω-3 fatty acids, respectively, EPA and DHA are bioactive ω-3 fatty acids that affect mood and cognition, and arachidonic acid and docosapentaenoic acid are bioactive ω-6 fatty acids. Consumption of HO-RUTF and DHA-HO-RUTF resulted in greater amounts of plasma ALA, as well as more EPA, whereas DHA was increased only in the DHA-HO-RUTF group. S-RUTF resulted in more LA than DHA-HO-RUTF, as well as greater amounts of the bioactive ω-6 fatty acids. All measured plasma fatty acid concentrations from SAM children fed different RUTFs can be found in Supplemental Table 8.
FIGURE 4.
Box and whisker plots of plasma fatty acid content of 6 selected fatty acids in severely malnourished children receiving RUTF made with high-oleic acid peanuts with added DHA (DHA-HO-RUTF), RUTF made with high-oleic acid peanuts (HO-RUTF), or standard RUTF (S-RUTF). The boxes represent the IQR of the distribution with a heavy midline median value. The whiskers extend to 1.5 times the IQR, values outside of these designations are plotted as points. The statistical comparisons were made using a Wilcoxon Rank Sum test. The number of participants sampled in each group were nDHA-HO-RUTF = 208, nHO-RUTF = 189, and nS-RUTF = 162. AA, arachidonic acid; DHA, docosahexaenoic acid; DPA, docosapentaenoic acid; EPA, eicosapentaenoic acid; RUTF, ready-to-use therapeutic food.
Discussion
This trial found that SAM children who consumed DHA-HO-RUTF achieved superior global MDAT z-scores than children who consumed S-RUTF. HO-RUTF did not confer the same benefit. DHA-HO-RUTF caused a positive shift in the distribution of MDAT z-scores measured ∼6 mo after an SAM outcome with no intervening DHA supplementation, suggesting that the cognitive benefits were seen in the entire population of SAM treated children and were sustained well after treatment was complete. However, PSA scores were similar in all dietary groups. The expected improvements in plasma fatty acid content were observed; children receiving DHA-HO-RUTF had greater concentrations of DHA. Of note, only in the social cognitive domain was HO-RUTF superior to S-RUTF, and this corresponds with higher plasma EPA concentrations in children receiving HO-RUTF (36).
Anthropometric recovery was nonsignificantly lower in both DHA-HO-RUTF and HO-RUTF groups compared with S-RUTF. This could be the consequence of differences in the nonlipid nutrient content of the study foods, or simply by chance. No adverse effects or negative preferences against DHA-HO-RUTF were observed in this large trial.
This trial demonstrated a clear discrepancy between anthropometric recovery and cognitive status in SAM. Children fed S-RUTF show inferior cognitive performance when compared with population norms, an insult of 0.9 MDAT z-scores. This deleterious effect is observed in all domains of the MDAT, except for the social domain. The magnitude of the insult suggests that a frameshift in thinking and approach may well be warranted with respect to SAM treatment and recovery. The focus on cognitive recovery must rise to equal importance as anthropometric recovery to enable the child to thrive. The distribution of MDAT scores in children aged <1 y mirrors a healthy population (Supplemental Figure 2) and offers hope that improvements in treatment may avert long-term disabilities. The increase seen in MDAT by the inclusion of less LA and more DHA in RUTF is encouraging and actionable. Differences seen between dietary groups were not dramatic (Supplemental Figure 1) when compared with the SAM insult. Our results report a functional benefit that extends the importance of our previous biochemical results, indicating that children recovering from SAM require a source of DHA to ameliorate the cognitive insult (19, 37).
The MDAT was developed in a rural Malawian setting, is fully validated, reliable, and predictive of later intellectual performance. For these reasons, MDAT was chosen as a primary outcome and Malawi was chosen as the study location. MDAT has revealed cognitive deficits in acutely malnourished populations in Malawi and Burkina Faso. In a previous study, MDAT domain scores were reported in Malawian SAM children upon discharge from the hospital (23). All 4 domain scores were remarkably lower among hospitalized SAM children than in our population. The MUAC of these hospitalized children was 11.5 cm, similar to that in our population, although the clinical status due to infectious complications was likely worse. These findings lead us to speculate that significant improvements in MDAT occur after discharge and that maximizing the potential for cognitive recovery should be a primary goal in malnutrition programs.
The PSA scores did not differ between food groups. PSA problem 3 has been used to demonstrate cognitive differences among healthy infants aged 9–12 mo receiving DHA-enriched infant formulas (27). In this study, half of the healthy infants achieved a perfect score on the PSA on all 3 attempts. This is in contrast to our results, wherein only 15% of children achieved a perfect score on any attempt. This suggests the presence of a substantial cognitive deficit at the time of anthropometric recovery. Our PSA was conducted using this very same problem and protocol, and with a sample size increased 3-fold, as previously reported, so it is unlikely the failure to detect differences is the result of methodological flaws. Rather, PSA problem 3 was beyond the intellectual capacity of most children tested. Subsequent use of PSA in SAM should explore its timing relative to anthropometric recovery and the potential utility of repeated testing.
Two previous, smaller studies in SAM children were done using blood measures of fatty acid content (19, 37). Our findings are consonant with these; increases in DHA were only seen when fish oil was added to the diet, and reduction in dietary LA resulted in greater EPA content.
The food formulations of HO-RUTF and DHA-HO-RUTF were achieved by Project Peanut Butter via ingredient changes, without alterations in the mixing or packaging processes. HO peanuts can be purchased in the major peanut production markets worldwide. DHA is now encapsulated and available from the same ingredient producers as the micronutrient premix. Encapsulation is very durable and obscures the taste of the fish oil exceedingly well, in addition to limiting oxidative degradation. Nongenetically modified, high-oleic vegetable oils are available at a cost of about 10% more than traditional vegetable oils. They were developed for the purposes of increasing the shelf-life of the oil and have been shown to reduce the risk of heart disease in adults.
Our study has multiple limitations. The study population did not habitually consume fish, and the positive effect of DHA-HO-RUTF in a fish-consuming population might differ. However, most children recovering from SAM worldwide consume little else but RUTF, and RUTF should meet the needs of such children. Our study population developed SAM largely because of food insecurity. Children in whom chronic illness or excessive inflammation precipitated SAM might not realize similar cognitive benefits of DHA-HO-RUTF. The use of different cognitive assessments at single time points limited the ability to interpret the dynamic nature of cognitive recovery. Food stock-outs resulted in imbalanced randomization, potentially introducing bias. Reanalysis of the sample enrolled outside of these stock-outs did not show evidence of bias. Finally, there was loss to follow-up between SAM outcome and MDAT assessment 6 mo later. We do not suspect this loss to follow-up differentially affected the intervention groups.
We estimate that during treatment, SAM children were consuming about 240 g RUTF/d. For the DHA-HO-RUTF group, DHA intake averaged 173 mg/d or about 0.24% w/w DHA. The global breast milk DHA reference level is 0.32% w/w (38). Fat constitutes half the energy in breast milk and RUTF. The DHA content of DHA-HO-RUTF and breast milk was similar. Importantly, the effective intervention was to supply RUTF with DHA and with limited LA when recovery food was supplying calories and protein needed to restart a normal or even accelerated trajectory of neurocognitive development. The brain substitutes ω-6 docosapentaenoic acid (DPA6) for DHA when dietary LA is surfeit and ω-3 is limiting. DPA does not support neurocognitive function similarly to DHA and thus our results may point to slow replacement of DHA for DPA in the months after recovery (13).
LA antagonism of ω-3 PUFAs is understood from a molecular and genetic basis (39–41). Large dietary amounts of LA and little ALA given to pregnant animals results in offspring with increased aggression and impulsivity, reduced executive function, and impaired visual function (42). Confusion over the interrelation of dietary LA and ALA has led to futile attempts to increase circulating/tissue DHA by increasing dietary ALA without reducing LA. Although the dietary ω-6 to ω-3 ratio, usually cast as [LA]/[ALA], is widely quoted as a parameter defining ω-3 tissue accretion, it is the excess of ω-6 over ω-3, i.e. [LA]–[ALA], that controls DHA availability (43, 44). Clinical studies show that additional ALA does not increase circulating or breast milk DHA (45). Moreover, no amount of supplementary ω-3 EPA increases DHA. To increase DHA, 2 dietary interventions are well established: lowering dietary LA and increasing dietary DHA.
This study provides the first direct evidence that reduction in LA and addition of DHA in RUTF enhances cognition in SAM children. This finding is consonant with a body of evidence that extends over many decades, methodologies, and species. Perhaps the most compelling evidence is found in the composition of breast milk. The need to enhance cognitive recovery in SAM is substantial, even crucial, as this insult affects tens of millions of children annually. At present, ∼85 million children worldwide will develop SAM in the first 5 y of their life, a number roughly equal to the burden of all childhood developmental delays. Changing the composition of RUTF to reduce the insult of SAM is safe, feasible, and effective. Further research is needed to optimize the amount and duration of DHA supplementation, but in the meantime, there is a clear course of action to help SAM children worldwide: provision of RUTF with preformed DHA and reduced amounts of LA.
Supplementary Material
Acknowledgments
We are grateful to the Wiley Companies for the generous gift of AlaskOmega® ω-3 concentrate derived from Alaskan Pollock (Gadus chalcogrammus).
The authors’ responsibilities were as follows—JTB, MC-G, KM, DRW, RLC, and MJM: designed the research; RLC and KM: designed the neurocognitive testing and scoring; MN, MG, RL, IH-C, KM, and MC-G: conducted the clinical research, assessed the children's cognition, and collected the samples; JTB, HGP, and RJSL: conducted the basic laboratory work; KS, MC-G, JTB, RJSL, RLC, and MJM: analyzed and interpreted the data; KS and MC-G: created the figures and graphically analyzed the data; MC-G, KS, and MJM: wrote the first draft of the manuscript with input from JTB; MJM: has primary responsibility for final content; and all authors: read and approved the final manuscript. MJM is an associate editor at The American Journal of Clinical Nutrition. All other authors report no conflicts of interest.
Notes
Funding was generously provided by Unorthodox Philanthropy, Open Philanthropy, the Hickey Family Foundation, and the Children's Discovery Institute. Wiley Companies supported the work with a donation of ingredients for one of the study foods. Funders played no role in study design, implementation, analysis, or interpretation.
Supplementary Material Protocol, Supplemental Tables 1–8, and Supplemental Figures 1–4 are available from the “Supplementary data” link in the online posting of the article and from the same link in the online table of contents at https://academic.oup.com/ajcn/.
KS and MC-G considered as first authors.
JTB and MJM considered as senior authors.
Abbreviations used: ALA, α-linolenic acid; DHA-HO-RUTF, high-oleic acid RUTF with added DHA; DPA, docosapentaenoic acid; HO, high-oleic; HO-RUTF, high-oleic acid RUTF; LA, linoleic acid; MDAT, Malawi Developmental Assessment Tool; MUAC, midupper arm circumference; PSA, problem-solving assessment; RUTF, ready-to-use therapeutic food; SAM, severe acute malnutrition; S-RUTF, standard RUTF.
Contributor Information
Kevin Stephenson, Department of Medicine, Washington University, St. Louis, MO, USA.
Meghan Callaghan-Gillespie, Department of Pediatrics, Washington University, St. Louis, MO, USA.
Kenneth Maleta, Department of Public Health, School of Public Health & Family Medicine, Kamuzu University of Health Sciences, Blantyre, Malawi.
Minyanga Nkhoma, Department of Public Health, School of Public Health & Family Medicine, Kamuzu University of Health Sciences, Blantyre, Malawi.
Matthews George, Department of Public Health, School of Public Health & Family Medicine, Kamuzu University of Health Sciences, Blantyre, Malawi.
Hui Gyu Park, Department of Pediatrics, University of Texas at Austin, Austin, TX, USA.
Reginald Lee, Department of Pediatrics, Washington University, St. Louis, MO, USA.
Iona Humphries-Cuff, Project Peanut Butter, Lunzu, Malawi.
R J Scott Lacombe, Department of Pediatrics, University of Texas at Austin, Austin, TX, USA.
Donna R Wegner, Department of Pediatrics, Washington University, St. Louis, MO, USA.
Richard L Canfield, Department of Pediatrics, University of Texas at Austin, Austin, TX, USA.
J Thomas Brenna, Department of Pediatrics, University of Texas at Austin, Austin, TX, USA; Division of Nutritional Sciences, Cornell University, Ithaca, NY, USA.
Mark J Manary, Department of Pediatrics, Washington University, St. Louis, MO, USA; Department of Public Health, School of Public Health & Family Medicine, Kamuzu University of Health Sciences, Blantyre, Malawi; USDA/Agricultural Research Service Children's Nutrition Research Center, Houston, TX, USA.
Data Availability
Data described in the manuscript, code book, and analytic code will be made available upon request pending application to the corresponding author and approval.
References
- 1. Chase RP, Kerac M, Grant A, Manary M, Briend A, Opondo C, Bailey J. Acute malnutrition recovery energy requirements based on mid-upper arm circumference: secondary analysis of feeding program data from 5 countries, combined protocol for acute malnutrition study (ComPAS) stage 1. PLoS One. 2020;15(6):e0230452. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. World Health Organization, the World Food Programme, the United Nations System Standing Committee on Nutrition and the United Nations Children's Fund . Community-based management of severe acute malnutrition. [Internet]. 2007. [Accessed July 2021]. Available from: https://www.who.int/publications/i/item/9789280641479.
- 3. Martinez M. Tissue levels of polyunsaturated fatty acids during early human development. J Pediatr. 1992;120(4 Pt 2):S129–38. [DOI] [PubMed] [Google Scholar]
- 4. Carver JD, Benford VJ, Han B, Cantor AB. The relationship between age and the fatty acid composition of cerebral cortex and erythrocytes in human subjects. Brain Res Bull. 2001;56(2):79–85. [DOI] [PubMed] [Google Scholar]
- 5. Lacombe RJS, Chouinard-Watkins R, Bazinet RP. Brain docosahexaenoic acid uptake and metabolism. Mol Aspects Med. 2018;64:109–34. [DOI] [PubMed] [Google Scholar]
- 6. Sinclair AJ, Crawford MA. The incorporation of linolenic acid and docosahexaenoic acid into liver and brain lipids of developing rats. FEBS Lett. 1972;26(1):127–9. [DOI] [PubMed] [Google Scholar]
- 7. Su HM, Bernardo L, Mirmiran M, Ma XH, Corso TN, Nathanielsz PW, Brenna JT. Bioequivalence of dietary alpha-linolenic and docosahexaenoic acids as sources of docosahexaenoate accretion in brain and associated organs of neonatal baboons. Pediatr Res. 1999;45(1):87–93. [DOI] [PubMed] [Google Scholar]
- 8. Lattka E, Illig K B, Heinrich J. Genetic variants of the FADS1 FADS2 gene cluster as related to fatty acid metabolism. Curr Opin Lipidol. 2010;21(1):64–9. [DOI] [PubMed] [Google Scholar]
- 9. Zhang JY, Kothapalli K, Brenna JT. Desaturase and elongase-limiting endogenous long-chain polyunsaturated fatty acid biosynthesis. Curr Opin Clin Nutr Metab Care. 2016;19(2):103–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Holman RT, Caster WO, Wiese HF. The essential fatty acid requirement of infants and the assessment of their dietary intake of linoleate by serum fatty acid analysis. Am J Clin Nutr. 1964;14(2):70–5. [DOI] [PubMed] [Google Scholar]
- 11. Holman RT. The slow discovery of the importance of omega 3 essential fatty acids in human health. J Nutr. 1998;128(2 Suppl):427S33S. [DOI] [PubMed] [Google Scholar]
- 12. Hibbeln JR, Nieminen LR, Blasbalg TL, Riggs JA, Lands WEM. Healthy intakes of n-3 and n-6 fatty acids: estimations considering worldwide diversity. Am J Clin Nutr. 2006;83(6 Suppl):1483S–93S. [DOI] [PubMed] [Google Scholar]
- 13. Brenna JT. Animal studies of the functional consequences of suboptimal polyunsaturated fatty acid status during pregnancy, lactation and early post-natal life. Matern Child Nutr. 2011;7:(Suppl 2):59–79. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Mathieu G, Denis S, Lavialle M, Vancassel S. Synergistic effects of stress and omega-3 fatty acid deprivation on emotional response and brain lipid composition in adult rats. Prostaglandins Leukot Essent Fatty Acids. 2008;78(6):391–401. [DOI] [PubMed] [Google Scholar]
- 15. DeMar JC Jr, Ma K, Bell JM, Igarashi M, Greenstein D, Rapoport SI. One generation of n-3 polyunsaturated fatty acid deprivation increases depression and aggression test scores in rats. J Lipid Res. 2006;47(1):172–80. [DOI] [PubMed] [Google Scholar]
- 16. Hafandi A, Begg DP, Premaratna SD, Sinclair AJ, Jois M, Weisinger RS. Dietary repletion with omega3 fatty acid or with COX inhibition reverses cognitive effects in F3 omega3 fatty-acid-deficient mice. Comp Med. 2014;64(2):106–9. [PMC free article] [PubMed] [Google Scholar]
- 17. Chang JP, Su KP. Nutritional neuroscience as mainstream of psychiatry: the evidence-based treatment guidelines for using omega-3 fatty acids as a new treatment for psychiatric disorders in children and adolescents. Clin Psychopharmacol Neurosci. 2020;18(4):469–83. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Hallahan B, Ryan T, Hibbeln JR, Murray IT, Glynn S, Ramsden CE, SanGiovanni JP, Davis JM. Efficacy of omega-3 highly unsaturated fatty acids in the treatment of depression. Br J Psychiatry. 2016;209(3):192–201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Hsieh JC, Liu L, Zeilani M, Ickes S, Trehan I, Maleta K, Craig C, Thakwalaka C, Singh L, Brenna JTet al. High-oleic ready-to-use therapeutic food maintains docosahexaenoic acid status in severe malnutrition. J Pediatr Gastroenterol Nutr. 2015;61(1):138–43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Faul F, Erdfelder E, Buchner A, Lang AG. Statistical power analyses using G*Power 3.1: tests for correlation and regression analyses. Behav Res Methods. 2009;41(4):1149–60. [DOI] [PubMed] [Google Scholar]
- 21. Olsen MF, Iuel-Brockdorff AS, Yaméogo CW, Chicon B, Fabiansen C, Filteau D, Phelan K, Ouedraogo A, Michaelsen KF, Gladstone Met al. Impact of food supplements on early child development in children with moderate acute malnutrition: a randomised 2×2×3 factorial trial in Burkina Faso. PLoS Med. 2020;17(12):e1003442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Prado EL, Maleta K, Caswell BL, George M, Oakes LM, DeBolt MC, Bragg MG, Arnold CD, Ioanotti L, Lutter CKet al. Early child development outcomes of a randomized trial providing 1 egg per day to children age 6 to 15 months in Malawi. J Nutr. 2020;150(7):1933–42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. van den Heuvel M, Voskuijl W, Chidzalo K, Kerac M, Reijneveld SA, Bandsma R, Gladstone M. Developmental and behavioural problems in children with severe acute malnutrition in Malawi: a cross-sectional study. J Glob Health. 2017;7(2):020416. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Larson LM, Yousafzai AK. A meta-analysis of nutrition interventions on mental development of children under-two in low- and middle-income countries. Matern Child Nutr. 2017;13(1):e12229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Olsen MF, Iuel-Brockdorff AS, Yaméogo CW, Chicon B, Fabiansen C, Filteau D, Phelan K, Ouedraogo A, Wells JC, Briend Aet al. Early development in children with moderate acute malnutrition: a cross-sectional study in Burkina Faso. Matern Child Nutr. 2020;16(2):e12928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Willatts P, Forsyth JS, DiModugno MK, Varma S, Colvin M. Influence of long-chain polyunsaturated fatty acids on infant cognitive function. Lipids. 1998;33(10):973–80. [DOI] [PubMed] [Google Scholar]
- 27. Drover J, Hoffman DR, Castañeda YS, Morale SE, Birch EE. Three randomized controlled trials of early long-chain polyunsaturated fatty acid supplementation on means-end problem solving in 9-month-olds. Child Dev. 2009;80(5):1376–84. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Trehan I, Goldbach HS, Lagrone LN, Meuli GJ, Wang RJ, Maleta KM, Manary MJ. Antibiotics as part of the management of severe acute malnutrition. N Engl J Med. 2013;368(5):425–35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Linneman Z, Matilsky D, Ndekha M, Manary MJ, Maleta K, Manary MJ. A large-scale operational study of home-based therapy with ready-to-use therapeutic food in childhood malnutrition in Malawi. Matern Child Nutr. 2007;3(3):206–15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Gladstone M, Lancaster GA, Umar E, Nyirenda M, Kayira E, van den Broek N, Smyth RL. The Malawi Developmental Assessment Tool (MDAT): the creation, validation, and reliability of a tool to assess child development in rural African settings. PLoS Med. 2010;7(5):e1000273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Friard O, Gamba M. BORIS: a free, versatile open-source event-logging software for video/audio coding and live observations. Methods in Ecology and Evolution. 2016;7(11):1325–30. [Google Scholar]
- 32. Brenna JT, Plourde M, Stark KD, Jones PJ, Yu-Hong L. Best practices for the design, laboratory analysis, and reporting of trials involving fatty acids. Am J Clin Nutr. 2018;108(2):211–27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Park HG, McDonald JG, Thomson BM, Vale G, Brenna T. A robotic system for high throughput isolation of phospholipids from non-polar lipids. WP 520, The 67th ASMS Annual Conference on Mass Spectrometry and Allied Topics. 2019; Atlanta, Georgia. [Google Scholar]
- 34. Zhou Y, Nijland M, Miller M, Ford S, Nathanielsz JTB. The influence of maternal early to mid-gestation nutrient restriction on long chain polyunsaturated fatty acids in fetal sheep. Lipids. 2008;43(6):525–31. [DOI] [PubMed] [Google Scholar]
- 35. R: a language and environment for statistical computing . R Foundation for Statistical Computing, Vienna (Austria), [program], 2017. https://www.R-project.org/. [Google Scholar]
- 36. Sublette ME, Ellis SP, Geant AL, Mann JJ. Meta-analysis of the effects of eicosapentaenoic acid (EPA) in clinical trials in depression. J Clin Psychiatry. 2011;72(12):1577–84. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Jones KD, Ali R, Khasira MA, Odera D, West AL, Koster G, Akomo P, Talbert AWA, Gross VM, Ngari Met al. Ready-to-use therapeutic food with elevated n-3 polyunsaturated fatty acid content, with or without fish oil, to treat severe acute malnutrition: a randomized controlled trial. BMC Med. 2015;13(1):93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Brenna JT, Varamini B, Jensen RG, Diersen-Schade DA, Boettcher JA, Arterbuen LM. Docosahexaenoic and arachidonic acid concentrations in human breast milk worldwide. Am J Clin Nutr. 2007;85(6):1457–64. [DOI] [PubMed] [Google Scholar]
- 39. Kothapalli KSD, Park HG, Brenna JT. Polyunsaturated fatty acid biosynthesis pathway and genetics. Implications for interindividual variability in prothrombotic, inflammatory conditions such as COVID-19 ✰,✰✰,★,★★. Prostaglandins Leukot Essent Fatty Acids. 2020;162:102183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Koletzko B, Reischl E, Tanjung C, Gonzalez-Casanova I, Ramakrishnan U, Meldrum S, Simmer K, Heinrich J, Demmelmai H. FADS1 and FADS2 polymorphisms modulate fatty acid metabolism and dietary impact on health. Annu Rev Nutr. 2019;39(1):21–44. [DOI] [PubMed] [Google Scholar]
- 41. Reynolds LM, Howard TD, Ruczinski I, Kanchin K, Seeds MC, Mathias RA, Chilton FH. Tissue-specific impact of FADS cluster variants on FADS1 and FADS2 gene expression. PLoS One. 2018;13(3):e0194610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Anderson GJ, Neuringer M, Lin DS, Connor WE. Can prenatal N-3 fatty acid deficiency be completely reversed after birth? Effects on retinal and brain biochemistry and visual function in rhesus monkeys. Pediatr Res. 2005;58(5):865–72. [DOI] [PubMed] [Google Scholar]
- 43. Gibson RA, Neumann MA, Lien EL, Boyd KA, Tu WC. Docosahexaenoic acid synthesis from alpha-linolenic acid is inhibited by diets high in polyunsaturated fatty acids. Prostaglandins Leukot Essent Fatty Acids. 2013;88(1):139–46. [DOI] [PubMed] [Google Scholar]
- 44. Brenna JT, Salem N, Sinclair AJ, Cunnane SC. Alpha-linolenic acid supplementation and conversion to n-3 long-chain polyunsaturated fatty acids in humans. Prostaglandins Leukot Essent Fatty Acids. 2009;80(2–3):85–91. [DOI] [PubMed] [Google Scholar]
- 45. Francois CA, Connor SL, Bolewicz LC, Connor WE. Supplementing lactating women with flaxseed oil does not increase docosahexaenoic acid in their milk. Am J Clin Nutr. 2003;77(1):226–33. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Data described in the manuscript, code book, and analytic code will be made available upon request pending application to the corresponding author and approval.