Abstract
Frailty is very common in old age and often associated with adverse events. Transitioning between frailty states is possible in both directions (improvement and worsening) offering targets for interventions. Frailty is more prevalent in women, but little is known about the impact of gender on frailty transition. The aim of this study is to identify gender differences for frailty transition in older adults and to develop gender-stratified prognostic prediction models for frailty transition. We performed a longitudinal analyses of the Berlin Initiative (cohort) Study with a frailty follow-up of 2.1 years. Description of frailty transition using the frailty phenotype and development of prognostic prediction models using multivariable logistic regressions for transition (improvement or worsening) stratified by gender following the TRIPOD statement were performed. In total, the study population consisted of 1158 community-dwelling adults with a mean age of 84.4 years and of whom 55% were women. Out of 1158 participants 225 (19%) were robust, 532 (46%) prefrail and 401 (35%) frail. After 2.1 (IQR 2.0–2.3) years, half of the participants had transitioned between frailty states. Men worsened more often and those who were already frail died more often than women. Gender-stratified prediction models for frailty transition demonstrated that some predictors (age, self-rated health, cognitive impairment, baseline frailty status) were included in all models. While stroke, diabetes mellitus, smoking and glomerular filtration rate were unique predictors in the models for females, osteoarthritis, hospitalization and education were predictors in the models for males. There are gender differences in frailty transition rates, patterns and prediction. This supports the importance of considering gender when addressing frailty and targeting interventions in old age.
Subject terms: Geriatrics, Epidemiology
Introduction
Frailty has been described as one of the greatest challenges facing the aging populations1. It is described as a biological syndrome with accelerated decrease in physiological reserve and resistance to stressors2,3. Individuals degrade across multiple physiological systems and these cumulative declines result in an increased susceptibility to adverse events2. These adverse events include an increased risk of falls, hospitalization, disability in activities of daily living, need of nursing home, or mortality2,4. Among several different instruments to assess frailty one of the most frequently used is the frailty phenotype by Fried5,6. Using this instrument in populations 65 years and older the prevalence of frailty ranged between 4.0% and 17.0%6. With older age the prevalence of frailty increases further6,7. Overall, prevalence and incidence of frailty are higher in women than in men8,9.
Frailty is also a dynamic process and its status can continuously change by worsening or even improving over time10,11. Understanding the transition of frailty especially in very old age is important since it potentially offers timely approaches for interventions. A recent systematic review and meta-analysis showed that the frailty status of community-dwelling older adults worsened in about 30% within a mean follow-up of 3.9 years, whereas it improved in 13%12. To define specific targets for intervention, the factors associated with frailty transition have to be identified. Since frailty is more common in women, frailty transition—concerning its pattern and risk factors—could also be different for men and women. So far, most studies did not stratify by gender and identified older age, hospitalization, previous stroke, cognitive impairment, previous cancer, osteoarthritis, diabetes, socio economic status, COPD, congestive heart failure, polypharmacy, urinary incontinence or frailty baseline status to be associated with frailty worsening regardless of gender11,13–18.
Apparently, there is a lack of evidence regarding the gender impact on frailty transition19 and the little data available in old age are inconsistent12. Thus, the aim of this study was to analyze gender differences of frailty transition in a population of community-dwelling older adults by (1) describing the different transition patterns of improving, no change or worsening by gender and (2) to develop prognostic prediction models for frailty transition stratified by gender following the TRIPOD guidelines20.
Methods
The focus of this work was the analysis of frailty transition with a special emphasis on gender differences using a longitudinal approach with data from the Berlin Initiative Study (BIS).
Study population
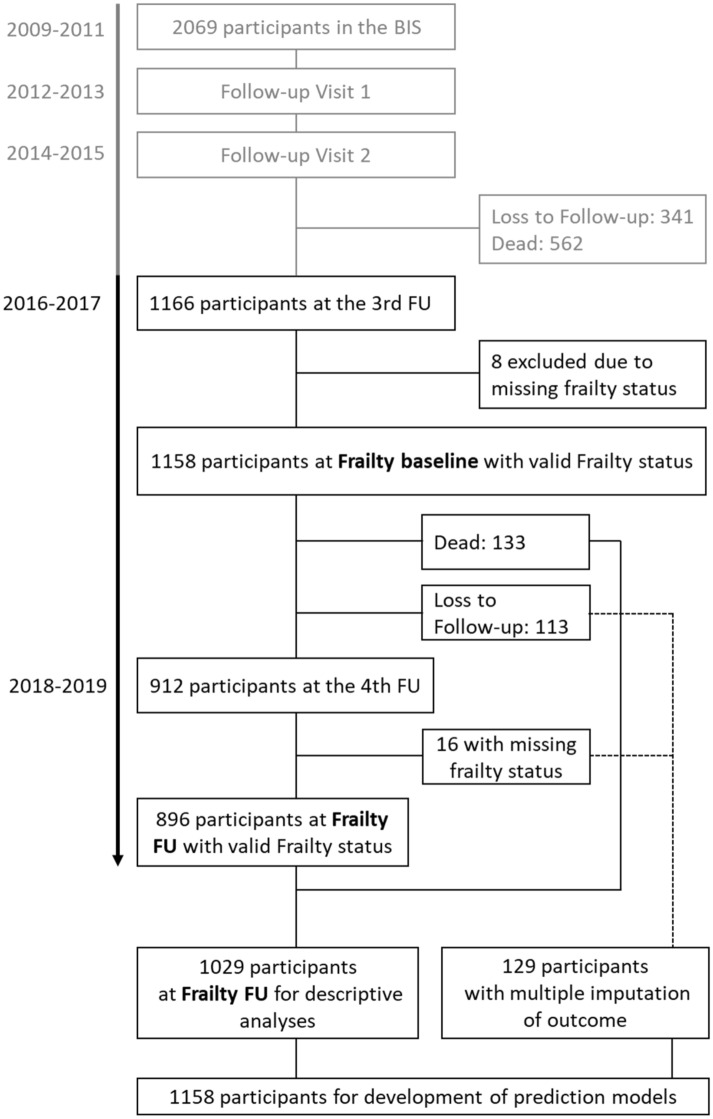
The BIS is a population-based cohort study of older adults initially enrolling 2069 participants between November 2009 and July 2011 aged 70 years and older. In order to be eligible, participants were not allowed to be dialysis patients, kidney transplant recipients or nursing home residents and had to be members of the AOK [“Allgemeine Ortskrankenkasse” (AOK)-Nordost—Berlin’s largest statutory health insurance fund]. Prior to the study, written informed consent was obtained from all participants. Participants were revisited every two years and those who were, e.g. for physical reasons, not able to come to a study site were visited at home. The study was approved by the ethics committee, Charité, Berlin, Germany (EA2/009/08). The study concept and design of the BIS have been described in detail21,22. The frailty questionnaire was implemented starting with the third follow-up BIS visit. For this analysis, we used data from the third and fourth BIS follow-up and will refer to these as baseline (3rd FU: 2016–2017) and follow-up (4th FU: 2018–2019), respectively (Fig. 1). The baseline study population consisted of 1166 participants. We excluded eight participants with missing data in the frailty score. The final study population consisted of 1158 eligible participants (Fig. 1). Closure of the data set was October 31st 2019.
Figure 1.
Overview of the Berlin Initiative Study (BIS) population. The flowchart shows the composition of the study population and which participants were available for certain analyses. The light gray section of the figure indicates the part of the BIS study that preceded this study. The frailty assessment was implemented at the 3rd follow-up (FU) of the BIS, defining the baseline visit for this study.
Frailty assessment
Frailty status was determined by a modified Frailty score according to Fried2. Frailty was defined as having at least three of the following five items: shrinking, exhaustion, low physical activity, slowness and weakness. Participants with one or two of the five items were defined as prefrail, and participants with none of the five items were defined as robust. Further details are described in Supplement A.
Frailty transition
Primary outcome was the frailty transition state at follow-up (median follow up time 2.1 years). Frailty transition was defined as improvement (frail to prefrail, frail to robust, prefrail to robust), worsening (robust to prefrail, robust to frail, prefrail to frail, any status to death) or no change (staying robust, prefrail, or frail).
Co-variables assessment
At each study visit, extensive data on demographics, lifestyle variables, morbidity, medication, anthropometric and geriatric assessment were conducted using a standardized computer-based questionnaire. Additionally, blood and urine samples were taken for instant analysis. Laboratory processing and measurement have been described previously22,23. In addition, the study data were complemented with AOK claims data. By this unique approach, self-reported data on e.g. hospitalizations and morbidities could be supplemented with verified claims data (i.e. diagnoses coded by the 10h Revision of the International Statistical Classification of Diseases and Related Health Problems (ICD-10)), and information from participants who were no longer seen at follow up was available. Also, the exact date of death of all participants who had died during follow-up was ascertained with the claims data. The following covariates were derived from BIS data: age, sex, the short version of the CASMIN classification of education24, living alone (yes, no), cognitive impairment (yes, no)25, smoking status (never, ever), self-rated health in two categories (excellent/good vs. moderate/poor/very poor) and body mass index (< 24.9, 25–29.9 or ≥ 30 kg/m2). Information on glomerular filtration rate (GFR, < 60 or ≥ 60 ml/min per 1.73m2) estimated by the BIS2 Equation23 and albuminuria (albumin-to-creatinine ratio ≥ 30 mg/g) as well as polypharmacy (≥ 5 medications) and diabetes mellitus (hemoglobin A1c ≥ 6.5% or intake of any antidiabetic drug) were also derived from the BIS data. The Charlson Comorbidity Index (CCI)26, osteoarthritis (ICD-10: M15-M19), COPD (ICD-10: J41-J44) and congestive heart failure (ICD-10: I50, I11.0, I13.0, I13.2, I25) were ascertained by a combination of self-reported BIS data validated by ICD-10 coded medical reports and AOK claims data as were stroke (ICD-10: I61, I63, I64), cancer (ICD-10: C00-C97 except C44) and the number of all-cause hospitalizations two years prior to baseline.
Statistical analysis
The baseline characteristics of the study population as well as the transition states were described for the total population and stratified by gender. Descriptive analysis included absolute and relative frequencies for categorical variables, for continuous variables either means with standard deviations or medians with interquartile range.
To develop prognostic prediction models for the transition between frailty states (worsening or improvement) after a median duration of 2.1 years we used multivariable logistic regressions. It was not possible to develop prognostic models for no change. Candidate predictors were selected based on medical knowledge (Table 1). The number of candidate predictors was restricted by the number of events20,27. Multicollinearity was assessed using the variance inflation factor28. Prediction models for the two transition outcomes were also stratified by gender, resulting in 4 models in total. Multiple imputation by chained equation was used to address all missing values (Suppl. Table 2). All predictor candidates and frailty status at follow up were used in the multiple imputations to generate 10 imputed data sets. For variable selection, we combined multiple imputation and bootstrapping. From every imputed data set we drew 200 bootstrap samples and applied automatic backward selection based on AIC. A variable was finally selected, if the selection frequency was ≥ 50% of the 2000 samples29. The selected predictor sets were finally tested in the original imputed data; pooled results are reported. For model discrimination, the optimism-corrected AUC was estimated as a measurement of the concordance index (c-index)30. The c-index is the probability that a participant with a certain change in frailty status will be assigned in the model with a higher probability in experiencing that outcome than a participant who did not experience the outcome. The standard error of AUC for the estimation of the 95% confidence interval was estimated by 2000 bootstrap replications. To calculate the bias-corrected model calibration we drew 200 bootstrap samples from every imputed data set31. To illustrate the model calibration we plotted the observed probabilities of all 10 imputed data sets against the predicted probabilities.
Table 1.
Main baseline characteristics by gender.
| Total | Women | Men | |
|---|---|---|---|
| n (%) | 1158 (100) | 637 (55) | 521 (45) |
| Age in years, mean (SD) | 84.4 (5.6) | 84.1 (5.6) | 84.8 (5.7) |
| Education (CASMIN-short), n (%) | |||
| Low | 686 (59) | 417 (66) | 269 (52) |
| Middle | 235 (20) | 148 (23) | 87 (17) |
| High | 232 (20) | 71 (11) | 161 (31) |
| Unknown | 5 (0.4) | 1 (0.2) | 4 (0.8) |
| Smoking, n (%) | |||
| Never | 622 (54) | 458 (72) | 164 (32) |
| Ever | 532 (46) | 176 (28) | 356 (68) |
| Unknown | 4 (0.3) | 3 (0.5) | 1 (0.2) |
| Self-rated health, n (%) | |||
| Excellent / good | 521 (45) | 267 (42) | 254 (49) |
| Moderate / poor / very poor | 634 (55) | 368 (58) | 266 (51) |
| Unknown | 3 (0.3) | 2 (0.3) | 1 (0.2) |
| Living alone, n (%) | 552 (48) | 393 (62) | 159 (31) |
| Unknown | 38 (3) | 25 (4) | 13 (2) |
| BMI in kg/m2, n (%) | |||
| < 25 | 377 (33) | 225 (35) | 152 (29) |
| 25–29.9 | 509 (44) | 251 (39) | 258 (50) |
| ≥ 30 | 255 (22) | 154 (24) | 101 (19) |
| Unknown | 17 (1) | 7 (1) | 10 (2) |
| Polypharmacy, n (%) | 848 (73) | 470 (74) | 378 (73) |
| Unknown | 1 (0.1) | 0 (0) | 1 (0.2) |
| Hospitalization, n (%) | |||
| 0 | 489 (42) | 284 (45) | 205 (39) |
| 1–2 | 448 (39) | 230 (36) | 218 (42) |
| ≥ 3 | 221 (19) | 123 (19) | 98 (19) |
| CCI, median (IQR) | 10 (7–12) | 9 (7–12) | 10 (8–13) |
| Cognitive impairment, n (%) | 95 (8) | 55 (9) | 40 (8) |
| Unknown | 68 (6) | 32 (5) | 36 (7) |
| Hypertension, n (%) | 958 (83) | 533 (84) | 425 (82) |
| Unknown | 2 (0.2) | 1 (0.2) | 1 (0.2) |
| Stroke, n (%) | 156 (14) | 75 (12) | 81 (16) |
| Congestive Heart failure, n (%) | 756 (65) | 390 (61) | 366 (70) |
| Diabetes mellitus, n (%) | 341 (29) | 171 (27) | 170 (33) |
| Unknown | 15 (1) | 8 (1) | 7 (1) |
| Cancer, n (%) | 315 (27) | 134 (21) | 181 (35) |
| Osteoarthritis, n (%) | 935 (81) | 558 (88) | 377 (72) |
| COPD, n (%) | 450 (39) | 254 (40) | 196 (38) |
| eGFRBIS2 < 60 ml/min/1.73m2, n (%) | 847 (73) | 468 (74) | 379 (73) |
| Unknown | 34 (3) | 22 (3) | 12 (2) |
| ACR ≥ 30 mg/g, n (%) | 306 (26) | 137 (22) | 169 (32) |
| Unknown | 77 (7) | 49 (8) | 28 (5) |
| Frailty status, n (%) | |||
| Robust | 225 (19) | 115 (18) | 110 (21) |
| Prefrail | 532 (46) | 282 (44) | 250 (48) |
| Frail | 401 (35) | 240 (38) | 161 (31) |
| Frailty transition after 2.1 years, n (%) | |||
| Improvement | 160 (14) | 101 (16) | 59 (11) |
| No change | 547 (47) | 304 (48) | 243 (47) |
| Worsening | 322 (28) | 162 (25) | 160 (31) |
| Unknown | 129 (11) | 70 (11) | 59 (11) |
SD standard deviation, BMI body mass index, CCI Charlson Comorbidity Index, COPD chronic obstructive lung disease, eGFR estimated glomerular filtration rate, ACR albumin to creatinine ratio.
All statistical analyses were conducted with IBM SPSS Statistics (Version 25.0; IBM Corp, Armonk, NY), or R (Version 3.6.1; R Foundation for Statistical Computing, Vienna, Austria).
Ethical approval
The study was approved by the ethics committee, Charité—Universitätsmedizin Berlin, Germany (EA2/009/08) and is in accordance with the 1964 Helsinki declaration and its later amendments. Written informed consent was obtained from all individual participants included in the study.
Results
Characteristics of the study sample
The main baseline characteristics of the 1158 study participants stratified by gender are displayed in Table 1. The mean age (SD) was 84.4 (5.6) years and 55% were women. About 60% had received a low education, half of the participants were living alone, and 45% rated their health as excellent or good. In the two years prior to baseline 58% of the participants had been hospitalized, 14% had experienced a stroke and 27% suffered from cancer. At baseline 8% were cognitively impaired, 29% had diabetes mellitus, and about three quarters had an impaired kidney function (GFRBIS2 < 60 ml/min/1.73 m2) or took ≥ 5 medications; with regard to frailty 19% were robust and 35% frail.
Women had received a lower education more often than men (66% vs. 52%), were more often never-smokers (72% vs. 32%) or living alone (62% vs. 31%), and exhibited less morbidities such as stroke (12% vs. 16%), heart failure (61% vs. 70%) and cancer (21% vs. 35%), but were diagnosed more often with osteoarthritis (88% vs. 72%) than men.
Frailty transition
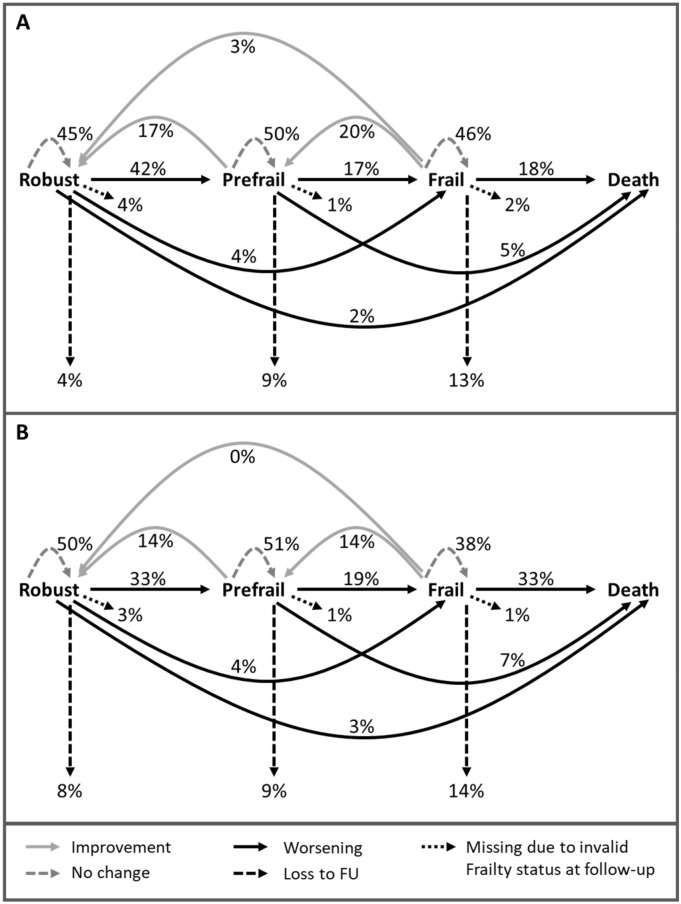
After 2.1 (2.0–2.3) years, frailty transition status could be determined for 1029 of the initial 1158 participants (Fig. 1). Out of 1029 participants 482 (42%) had transitioned between frailty states or death, with improvement in 160 (14%) and worsening in 322 (28%) participants (Table 1). Of those who worsened 133 (41%) had died (Supplement Table 1). Participants who were already frail at baseline, died more often (24%) compared to prefrail (6%) or robust (2%) participants. Transitions between adjacent frailty states were more frequent than those across several frailty states (Fig. 2). Overall, women improved slightly more often than men, while men worsened more often. More women than men who were frail remained frail (46% vs. 38%) and men who were frail died more frequently (33% men vs. 18% women).
Figure 2.
Transitions between frailty states and death after 2.1 years (A) Women (N = 637): at baseline 115 (18%) were robust, 282 (44%) prefrail and 240 (38%) frail. (B) Men (N = 521): at baseline 110 (21%) were robust, 250 (48%) prefrail and 161 (31%) frail. The relative frequencies of the frailty status transition are displayed by gender and refer to the baseline status. In total, all frequencies add up to 300%.
Baseline characteristics by frailty transition and stratified by gender
Participants who worsened were older, self-rated their health more often as moderate or (very) poor, were more often cognitively impaired, demonstrated more often a decreased kidney function (GFR < 60 ml/min/1.73m2) or kidney damage (ACR ≥ 30 mg/g) and had a higher prevalence of morbidities such as stroke or heart failure compared to those who improved or remained in their frailty status (Table 2).
Table 2.
Main baseline characteristics by frailty transition status after 2.1 years and stratified by gender.
| Improvement | No Change | Worsening | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Women | Men | Total | Women | Men | Total | Women | Men | Total | |
| n | 101 | 59 | 160 | 304 | 243 | 547 | 162 | 160 | 322 |
| Age in year, mean (SD) | 82.9 (5.1) | 83.4 (5.2) | 83.1 (5.1) | 83.7 (5.4) | 84.3 (5.5) | 84.0 (5.4) | 85.0 (6.1) | 86.0 (6.0) | 85.5 (6.1) |
| Education (CASMIN-short), n (%) | |||||||||
| Low | 69 (68) | 26 (44) | 95 (59) | 197 (65) | 124 (51) | 321 (59) | 104 (64) | 88 (55) | 192 (60) |
| Middle | 22 (22) | 16 (27) | 38 (24) | 76 (25) | 41 (17) | 117 (21) | 36 (22) | 22 (14) | 58 (18) |
| High | 9 (9) | 17 (29) | 26 (16) | 31 (10) | 78 (32) | 109 (20) | 22 (14) | 47 (29) | 69 (21) |
| Unknown | 1 (1) | 0 (0) | 1 (0.6) | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 3 (2) | 3 (0.9) |
| Smoking, n (%) | |||||||||
| Never | 73 (72) | 17 (29) | 90 (56) | 223 (73) | 81 (33) | 304 (56) | 106 (65) | 50 (31) | 156 (48) |
| Ever | 28 (28) | 42 (71) | 70 (44) | 80 (26) | 162 (67) | 242 (44) | 54 (33) | 110 (69) | 164 (51) |
| Unknown | 0 (0) | 0 (0) | 0 (0) | 1 (0.3) | 0 (0) | 1 (0.2) | 2 (1) | 0 (0) | 2 (0.6) |
| Self-rated health, n (%) | |||||||||
| Excellent / good | 46 (46) | 31 (53) | 77 (48) | 129 (42) | 131 (54) | 260 (48) | 61 (38) | 68 (43) | 129 (40) |
| Moderate /poor / very poor | 55 (55) | 28 (48) | 83 (52) | 173 (57) | 111 (46) | 284 (52) | 101 (62) | 92 (58) | 193 (60) |
| Unknown | 0 (0) | 0 (0) | 0 (0) | 2 (0.7) | 1 (0.4) | 3 (0.5) | 0 (0) | 0 (0) | 0 (0) |
| Living alone, n (%) | 56 (55) | 16 (27) | 72 (45) | 180 (59) | 77 (32) | 257 (47) | 113 (70) | 46 (29) | 159 (49) |
| Unknown | 3 (3) | 2 (3) | 5 (3) | 12 (4) | 5 (2) | 17 (3) | 7 (4) | 5 (3) | 12 (4) |
| BMI in kg/m2, n (%) | |||||||||
| < 25 | 32 (32) | 15 (25) | 47 (29) | 107 (35) | 62 (26) | 169 (31) | 61 (38) | 53 (33) | 114 (35) |
| 25–29.9 | 39 (39) | 32 (54) | 71 (44) | 122 (40) | 124 (51) | 246 (45) | 64 (40) | 72 (45) | 136 (42) |
| ≥ 30 | 28 (28) | 12 (20) | 40 (25) | 74 (24) | 52 (21) | 126 (23) | 36 (22) | 32 (20) | 68 (21) |
| Unknown | 2 (2) | 0 (0) | 2 (1) | 1 (0.3) | 5 (2) | 6 (1) | 1 (0.6) | 3 (2) | 4 (1) |
| Polypharmacy, n (%) | 73 (72) | 41 (70) | 114 (71) | 223 (73) | 174 (72) | 397 (73) | 125 (77) | 121 (76) | 246 (76) |
| Hospitalization, n (%) | |||||||||
| 0 | 45 (45) | 28 (48) | 73 (46) | 141 (46) | 104 (43) | 245 (45) | 67 (41) | 48 (30) | 115 (36) |
| 1–2 | 36 (36) | 20 (34) | 56 (35) | 104 (34) | 98 (40) | 202 (37) | 61 (38) | 72 (45) | 133 (41) |
| ≥ 3 | 20 (20) | 11 (19) | 31 (19) | 59 (19) | 41 (17) | 100 (18) | 34 (21) | 40 (25) | 74 (23) |
| CCI, median (IQR) | 8 (6–11) | 10 (8–12) | 9 (7–11) | 9 (7–12) | 10 (7–13) | 9 (7–12) | 10 (7–13) | 12 (9–14) | 11 (8–13) |
| Cognitive impairment, n (%) | 2 (2) | 1 (2) | 3 (2) | 22 (7) | 12 (5) | 34 (6) | 22 (14) | 16 (10) | 38 (12) |
| Unknown | 3 (3) | 1 (2) | 4 (3) | 10 (3) | 10 (4) | 20 (4) | 11 (7) | 17 (11) | 28 (9) |
| Hypertension, n (%) | 86 (85) | 49 (83) | 135 (84) | 253 (83) | 199 (82) | 452 (83) | 139 (86) | 133 (83) | 272 (85) |
| Unknown | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 1 (0.6) | 0 (0) | 1 (0.3) |
| Stroke, n (%) | 7 (7) | 9 (15) | 16 (10) | 35 (12) | 27 (11) | 62 (11) | 26 (16) | 31 (19) | 57 (18) |
| Congestive Heart failure, n (%) | 45 (45) | 40 (68) | 85 (53) | 186 (61) | 161 (66) | 347 (63) | 112 (69) | 127 (79) | 239 (74) |
| Diabetes mellitus, n (%) | 18 (18) | 15 (25) | 33 (21) | 82 (27) | 79 (33) | 161 (29) | 50 (31) | 56 (35) | 106 (33) |
| Unknown | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 4 (3) | 2 (1) | 6 (2) |
| Cancer, n (%) | 16 (16) | 27 (46) | 43 (27) | 62 (20) | 75 (31) | 137 (25) | 35 (22) | 61 (38) | 96 (30) |
| Osteoarthritis, n (%) | 92 (91) | 43 (73) | 135 (84) | 267 (88) | 168 (69) | 435 (80) | 142 (88) | 127 (79) | 269 (84) |
| COPD, n (%) | 35 (35) | 21 (36) | 56 (35) | 121 (40) | 87 (36) | 208 (38) | 67 (41) | 60 (38) | 127 (39) |
| eGFRBIS2 < 60 | |||||||||
| ml/min/1.73m2, n (%) | 68 (67) | 43 (73) | 111 (69) | 218 (72) | 170 (70) | 388 (71) | 128 (79) | 127 (79) | 255 (79) |
| Unknown | 4 (4) | 0 (0) | 4 (3) | 6 (2) | 2 (0.8) | 8 (2) | 5 (3) | 4 (3) | 9 (3) |
| ACR ≥ 30 mg/g, n (%) | 15 (15) | 19 (32) | 34 (21) | 57 (19) | 69 (28) | 126 (23) | 50 (31) | 65 (41) | 115 (36) |
| Unknown | 7 (7) | 1 (2) | 8 (5) | 22 (7) | 10 (4) | 32 (6) | 11 (7) | 10 (6) | 21 (7) |
| Frailty status, n (%) | |||||||||
| Robust | n.a | n.a | n.a | 52 (17) | 55 (23) | 107 (20) | 55 (34) | 43 (27) | 98 (30) |
| Prefrail | 48 (48) | 36 (61) | 84 (53) | 142 (47) | 127 (52) | 269 (49) | 64 (40) | 64 (40) | 128 (40) |
| Frail | 53 (53) | 23 (39) | 76 (48) | 110 (36) | 61 (25) | 171 (31) | 43 (27) | 53 (33) | 96 (30) |
SD standard deviation, n.a. not applicable, BMI body mass index, CCI Charlson Comorbidity Index, COPD chronic obstructive lung disease, eGFR estimated glomerular filtration rate, ACR albumin to creatinine ratio.
While men who worsened were more likely to have received a lower education compared to those who improved their frailty status (55% vs. 44%), education did not differ between transition groups in women (64% vs. 68%). About one third of men were living alone independent of frailty transition. In women who worsened 70% were living alone compared to 59% who remained or 55% of those who improved in their frailty status. Prior hospitalizations for women did not differ between the frailty transition outcomes. However, men who worsened were more often previously hospitalized (70%) compared to men who maintained (57%) or improved (52%) their frailty status (Table 2).
Prediction of improvement
An improvement of the frailty status could be observed in 116 out of 522 women (22%) and in 70 out of 411 men (17%) after multiple imputation of all missing values (Table 3, Supplement Table 2 and 3). Out of the 18 candidate predictor variables considered, after the bootstrap selection process for the logistic regression models eight variables remained for women and seven for men. Both gender-specific models included the predictors age, self-rated health, cognitive impairment, frailty baseline status, and cancer. For women, the model additionally included heart failure (OR: 0.41; 95%-CI: 0.25–0.68), stroke (OR: 0.46; 95%-CI: 0.19–1.14), but not statistically significant when tested in the original imputed data set, and diabetes mellitus (OR: 0.50; 95%-CI: 0.27–0.90) with reduced odds between 50 and 59% for improvement if one had these comorbidities. The c-index (95%-CI) for discrimination was 0.74 (0.72–0.76). The calibration intercept ranged from -0.16 to -0.05 and the calibration slope from 0.84 – 0.90 (Supplement Fig. 1A).
Table 3.
Prognostic prediction models for frailty improvement or worsening after 2.1 years in community-dwelling older adults stratified for gender.
| Improvement | Worsening | |||
|---|---|---|---|---|
| Women | Men | Women | Men | |
| N | 522 | 411 | 637 | 521 |
| Number of events | 116 | 70 | 174 | 173 |
| Predictor Candidates* | Odds Ratio (95% CI) | Odds Ratio (95% CI) | Odds Ratio (95% CI) | Odds Ratio (95% CI) |
|---|---|---|---|---|
| Age (1 year) | 0.93 (0.88–0.98) | 0.87 (0.82–0.93) | 1.06 (1.01–1.12) | 1.09 (1.05–1.14) |
| Self-rated health, moderate-very poor | 0.56 (0.32–0.97) | 0.42 (0.22–0.82) | 1.95 (1.24–3.07) | 1.67 (1.07–2.62) |
| Cognitive impairment | 0.14 (0.03–0.73) | 0.18 (0.02–1.40) | 3.70 (1.73–7.92) | 2.43 (1.20–4.93) |
| Frailty status at baseline | ||||
| Robust | n.a | n.a | 30.11 (13.73–66.06) | 8.03 (3.68–17.51) |
| Prefrail | 0.23 (0.13–0.41) | 0.29 (0.15–0.57) | 4.57 (2.53–8.28) | 1.95 (1.14–3.35) |
| Frail | Ref | Ref | Ref | Ref |
| Congestive Heart failure | 0.41 (0.25–0.68) | – | 1.60 (1.00–2.57) | 1.63 (0.98–2.72) |
| Stroke | 0.46 (0.19–1.14) | – | 1.65 (0.90–3.02) | – |
| Diabetes mellitus | 0.50 (0.27–0.90) | – | 1.43 (0.90–2.27) | |
| Cancer | 0.72 (0.38–1.36) | 1.65 (0.91–3.01) | – | – |
| Living alone | – | 0.63 (0.33–1.21) | 1.57 (0.95–2.58) | – |
| Education (CASMIN-short) | ||||
| Low | – | Ref | – | – |
| Middle | – | 1.98 (0.96–4.10) | – | – |
| High | – | 1.15 (0.58–2.31) | – | – |
| ACR ≥ 30 mg/g | – | – | 1.74 (1.05–2.89) | 1.85 (1.12–3.06) |
| Smoking | – | – | 1.59 (1.01–2.50) | – |
| eGFRBIS2 < 60 ml/min/1.73 m2 | – | – | 1.59 (0.90–2.79) | – |
| Osteoarthritis | – | – | – | 1.44 (0.90–2.31) |
| Hospitalization | ||||
| 0 | – | – | – | Ref |
| 1–2 | – | – | – | 1.36 (0.83–2.25) |
| ≥ 3 | – | – | – | 1.72 (0.96–3.08) |
| Model discrimination | ||||
| c-Index | 0.74 (0.72–0.76) | 0.68 (0.66–0.70) | 0.76 (0.75–0.77) | 0.66 (0.65–0.67) |
*Predictor Candidates not included in any model: BMI, Polypharmacy, COPD.
For men, the model additionally included living alone (OR: 0.63; 95%-CI: 0.33–1.21) and education. The c-index (95%-CI) of this logistic model was 0.68 (0.66–0.70). The calibration intercept ranged from − 0.32 to − 0.21 and the calibration slope from 0.77 to 0.84 (Supplement Fig. 1B).
Prediction of worsening
A worsening of the frailty status could be observed in 174 of 637 (27%) women and in 173 of 521 (33%) men after multiple imputation of all missing values (Table 3, Supplement Table 2 and 3). Out of the 18 candidate predictor variables considered, after the bootstrap selection process for the logistic regression models 11 remained for women and 8 for men (Table 3). Both gender-specific prediction models included age, self-rated health, heart failure, kidney damage (ACR), cognitive impairment (women: OR 3.70, 95%-CI: 1.73–7.92; men: OR 2.43, 95%-CI: 1.20–4.93) and frailty baseline status, the latter two being the variables with the highest odds for worsening. The model for women additionally included stroke, diabetes mellitus, living alone, smoking and kidney function (GFR). The c-index (95%-CI) for discrimination was 0.76 (0.75–0.77). The calibration intercept ranged from -0.09 to -0.07 and the calibration slope from 0.88 to 0.90 (Supplement Fig. 1C). The model for men additionally included osteoarthritis and hospitalization. The c-index (95%-CI) of this logistic model was 0.66 (0.65–0.67). The calibration intercept ranged from − 0.09 to − 0.07 and the calibration slope from 0.84 to 0.88 (Supplement Fig. 1D).
Discussion
In this study, we were able to demonstrate that over the course of 2.1 years, half of the participants transitioned into a different frailty status and of those two thirds were worsening. Men worsened more often than women, and if already frail at baseline men died more often than women. Prediction models for frailty improvement or worsening stratified by gender included age, self-rated health, cognitive impairment, heart failure, living alone, ACR, cancer and baseline frailty as common predictors for both genders. Osteoarthritis, hospitalization, and education however, were unique to men, whereas stroke, diabetes, smoking or eGFR were unique to women.
To our knowledge, this is the first study to report gender differences in frailty transition among German older adults. Studies on frailty have been conducted in a variety of settings and have shown that the prevalence of frailty is higher in women than in men6 consistent with our results. Our focus was on frailty transition over time. A recent meta-analysis showed that about half of the older adults (mean age between 64 and 79 years) maintain their frailty status over about four years with no significant gender differences in transition rates12. In contrast to their findings, we observed more improvement in women but more worsening in men, with identical rates of no change. Reason for this may lie in the high degree of heterogeneity within the studies included in the gender-specific meta-analysis12. The overall observation length was longer, participants were younger and by pooling the data from 16 studies they had a bigger sample size.
Studies investigating gender-specific frailty transition are scarce. The few studies investigating frailty transition by gender often only reported the transition rates18,32,33. We characterized older adults by their transition pattern and thereby identified gender differences for education, living alone, BMI, the number of hospitalizations, cancer and osteoarthritis. Furthermore, we developed gender-specific prognostic prediction models following the TRIPOD guidelines20 to identify predictors for frailty transition. We included the above variables and factors identified in studies mostly not stratifying by gender11,13,15–18,33–36. The prediction models for females included more variables and had a higher discrimination ability compared to the models in men that only demonstrated possibly helpful discrimination ability. These findings are in concordance with a study by Lee et al. (2014), where also more variables were included in the models for females, suggesting gender-specific predictors in men have not all yet been identified13. However the study did not report discrimination measures. With respect to model calibration, all four prediction models were well calibrated, those for women slightly better than those for men. We acknowledge that our models need to be validated externally before deriving clinical implications37,38.
The prediction models could help to identify older adults likely to worsen within the following two years to initiate preventive interventions or to improve the basis for shared decision making. So far, exercise and nutrition-based interventions have shown the highest level of evidence to delay worsening39, although there is also evidence that multifactorial interventions including cognitive training may be beneficial19,40,41. This aligns well with our findings that impaired cognitive function predicts frailty worsening. Gender differences in both, transition patterns as well as transition predictors support approaches to take a gender aspect into account, especially in multifactorial frailty interventions. Studies comparing the clinical effectiveness of frailty intervention by gender, however, are still missing39.
The strengths of our study include a very old study population, a detailed phenotyping and its longitudinal design. This is the first time that in a high-aged German cohort prediction models for frailty transition stratified by gender were developed. Another strength is the thorough methodological approach used for developing the prediction models following the recommended TRIPOD guidelines20. By linking primary (BIS) and secondary (AOK claims) data, we have compiled a large, complimentary and valid health data set including date of death facilitating the imputation of missing data.
With regard to limitations, one may argue that 2.1 years of follow-up is short. However, in high-aged persons an observation of one year seemed sufficient to analyze transition effectively12. Although we may have missed intermediate transitions between visits, our transition rates fit in well with numbers published for up to 4 years follow-up. The 10% loss to follow-up during the observation period may have introduced selection bias. Regardless of gender, frail older adults were more often lost to follow-up. By using the claims data source, our study could utilize claims data for those participants to support the imputation of missing data for the prediction models. Therefore, we consider our models, based on a solid and well-defined dataset, as reliable. Secondly, we did not use the identical exclusion criteria as Fried, who excluded conditions which potentially present frailty characteristics as a consequence of a single disease, e.g. Parkinson2. This would have applied to only 26 individuals (2.2%) in our study. However, the aim of this study was not to report a representative prevalence for frailty but to describe gender-specific frailty transition and identify predictors. Since we investigated the dynamic of frailty transition using the same instrument over time, we assume our deviation from the Fried criteria to be negligible. Thirdly, with our methodological approach, models for the prediction of no change in frailty could not be developed, probably because many of the factors predict both worsening and improvement but in opposite directions. Lastly, limited by our transition numbers we were not able to include all a priori identified knowledge-based predictors in the modelling. It can be postulated that some of them might improve the prediction models especially for men. Furthermore, few variables identified in the bootstrap selection process did not show statistical significance when finally applied in the original imputed data set, presumably because they are weaker or less frequent predictors. Before potentially implementing the models in practice they require external validation.
In conclusion, our study demonstrates gender differences in the pattern of worsening and improvement of frailty as well as in predictors of transition: Women, initially more frequently frail, experience less worsening and more improvement compared to men. Women and men have different predictors of worsening. Thus, these newly developed prediction models may trigger further clinical investigation and model evaluation.
Supplementary Information
Acknowledgements
We thank the colleagues at the 13 study sites in Berlin for supporting our work and providing the necessary infrastructure for the study. We are indebted to the participants of the BIS for their participation and commitment. We also thank the health insurance fund AOK Nordost-Die Gesundheitskasse for its continuous cooperation and technical support.
Author contributions
N.M. and E.S. conceived and designed the study. N.M., E.S., N.E. and D.H. participated in acquisition and processing of the data. A.S. and N.M. contributed to data analysis. N.M. wrote the original draft. N.M., A.S., N.E., D.H., E.S. substantively revised the manuscript. All authors have read and approved the manuscript. The sponsors were not involved in the design and conduct of the study; the collection, management, analysis, and interpretation of the data; or the preparation, review, or approval of the manuscript.
Funding
Open Access funding enabled and organized by Projekt DEAL. This study was supported by the Kuratorium für Dialyse und Nierentransplantation (KfH) Foundation of Preventive Medicine (http://www.kfh-stiftung-praeventivmedizin.de) and the DDnÄ – Institut für Disease Management e.V. (all granted to E.S.).
Data availability
There are no linked research data sets for this study. The data are available from the corresponding author OR from the study PI upon reasonable request.
Competing interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. NM and AS have nothing to disclose. DH received travel reimbursement for analysis project meetings from Actelion Pharmaceuticals, Boehringer Ingelheim and Shire. NE received honoraria from Siemens Healthineers. ES received honoraria for lectures from Siemens Healtineers and Fresenius Kabi.
Footnotes
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
The online version contains supplementary material available at 10.1038/s41598-022-11358-7.
References
- 1.Feng, Z., et al.; Risk factors and protective factors associated with incident or increase of frailty among community-dwelling older adults A systematic review of longitudinal studies. PLoS One 2017;12(6):0178383. Doi: 10.1371/journal.pone.0178383. [DOI] [PMC free article] [PubMed]
- 2.Fried LP, et al. Frailty in older adults: evidence for a phenotype. J. Gerontol. A Biol. Sci. Med. Sci. 2001;56(3):M146–M157. doi: 10.1093/gerona/56.3.M146. [DOI] [PubMed] [Google Scholar]
- 3.Clegg A, Young J, Iliffe S, Rikkert MO, Rockwood K. Frailty in elderly people. Lancet. 2013;381(9868):752–762. doi: 10.1016/S0140-6736(12)62167-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Bandeen-Roche K, et al. Phenotype of frailty: characterization in the women's health and aging studies. J. Gerontol. A Biol. Sci. Med. Sci. 2006;61(3):262–266. doi: 10.1093/gerona/61.3.262. [DOI] [PubMed] [Google Scholar]
- 5.Bouillon K, et al. Measures of frailty in population-based studies: an overview. BMC Geriatr. 2013;13(1):64. doi: 10.1186/1471-2318-13-64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Collard RM, Boter H, Schoevers RA, Oude Voshaar RC. Prevalence of frailty in community-dwelling older persons: a systematic review. J. Am. Geriatr. Soc. 2012;60(8):1487–1492. doi: 10.1111/j.1532-5415.2012.04054.x. [DOI] [PubMed] [Google Scholar]
- 7.Siriwardhana DD, Hardoon S, Rait G, Weerasinghe MC, Walters KR. Prevalence of frailty and prefrailty among community-dwelling older adults in low-income and middle-income countries: a systematic review and meta-analysis. BMJ Open. 2018;8(3):e018195. doi: 10.1136/bmjopen-2017-018195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ofori-Asenso R, et al. Global incidence of frailty and prefrailty among community-dwelling older adults: a systematic review and meta-analysis. JAMA Netw. Open. 2019;2(8):e198398. doi: 10.1001/jamanetworkopen.2019.8398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Gordon EH, et al. Sex differences in frailty: a systematic review and meta-analysis. Exp. Gerontol. 2017;89:30–40. doi: 10.1016/j.exger.2016.12.021. [DOI] [PubMed] [Google Scholar]
- 10.Gill TM, Gahbauer EA, Allore HG, Han L. Transitions between frailty states among community-living older persons. Arch. Int. Med. 2006;166(4):418–423. doi: 10.1001/archinte.166.4.418. [DOI] [PubMed] [Google Scholar]
- 11.Lorenzo-Lopez L, et al. Changes in frailty status in a community-dwelling cohort of older adults: the VERISAUDE study. Maturitas. 2019;119:54–60. doi: 10.1016/j.maturitas.2018.11.006. [DOI] [PubMed] [Google Scholar]
- 12.Kojima G, Taniguchi Y, Iliffe S, Jivraj S, Walters K. Transitions between frailty states among community-dwelling older people: a systematic review and meta-analysis. Ageing Res. Rev. 2019;50:81–88. doi: 10.1016/j.arr.2019.01.010. [DOI] [PubMed] [Google Scholar]
- 13.Lee JS, Auyeung TW, Leung J, Kwok T, Woo J. Transitions in frailty states among community-living older adults and their associated factors. J. Am. Med. Dir. Assoc. 2014;15(4):281–286. doi: 10.1016/j.jamda.2013.12.002. [DOI] [PubMed] [Google Scholar]
- 14.Alencar MA, Dias JMD, Figueiredo LC, Dias RC. Transitions in frailty status in community-dwelling older adults. Top. Geriatr. Rehabil. 2015;31(2):105–112. doi: 10.1097/TGR.0000000000000055. [DOI] [Google Scholar]
- 15.Espinoza SE, Jung I, Hazuda H. Frailty transitions in the San Antonio Longitudinal Study of Aging. J. Am. Geriatr. Soc. 2012;60(4):652–660. doi: 10.1111/j.1532-5415.2011.03882.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Thompson MQ, Theou O, Adams RJ, Tucker GR, Visvanathan R. Frailty state transitions and associated factors in South Australian older adults. Geriatr. Gerontol. Int. 2018;18(11):1549–1555. doi: 10.1111/ggi.13522. [DOI] [PubMed] [Google Scholar]
- 17.Herr M, et al. Factors associated with changes of the frailty status after age 70: Findings in the MAPT study. Ann. Epidemiol. 2019;34:65–70 e61. doi: 10.1016/j.annepidem.2019.03.008. [DOI] [PubMed] [Google Scholar]
- 18.Trevisan C, et al. Factors influencing transitions between frailty states in elderly adults: the Progetto Veneto Anziani Longitudinal Study. J. Am. Geriatr. Soc. 2017;65(1):179–184. doi: 10.1111/jgs.14515. [DOI] [PubMed] [Google Scholar]
- 19.Gordon EH, Hubbard RE. Differences in frailty in older men and women. Med. J. Aust. 2020;212(4):183–188. doi: 10.5694/mja2.50466. [DOI] [PubMed] [Google Scholar]
- 20.Moons KG, et al. Transparent reporting of a multivariable prediction model for individual prognosis or diagnosis (TRIPOD): explanation and elaboration. Ann. Int. Med. 2015;162(1):W1–73. doi: 10.7326/M14-0698. [DOI] [PubMed] [Google Scholar]
- 21.Schaeffner ES, et al. The Berlin initiative study: the methodology of exploring kidney function in the elderly by combining a longitudinal and cross-sectional approach. Eur. J. Epidemiol. 2010;25(3):203–210. doi: 10.1007/s10654-010-9424-x. [DOI] [PubMed] [Google Scholar]
- 22.Ebert N, et al. Prevalence of reduced kidney function and albuminuria in older adults: the Berlin Initiative Study. Nephrol. Dial. Transplant. 2017;32(6):997–1005. doi: 10.1093/ndt/gfw079. [DOI] [PubMed] [Google Scholar]
- 23.Schaeffner ES, et al. Two novel equations to estimate kidney function in persons aged 70 years or older. Ann. Int. Med. 2012;157(7):471–481. doi: 10.7326/0003-4819-157-7-201210020-00003. [DOI] [PubMed] [Google Scholar]
- 24.Brauns H, Scherer S, Steinmann S. The CASMIN educational classification in international comparative research. In: Hoffmeyer-Zlotnik JHP, Wolf C, editors. Advances in cross-national comparison. New York: Kluwer; 2003. pp. 221–244. [Google Scholar]
- 25.Schnitzer S, et al. Risk profiles for care dependency: cross-sectional findings of a population-based cohort study in Germany. J. Aging Health. 2020;32(5–6):352–360. doi: 10.1177/0898264318822364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Charlson ME, Pompei P, Ales KL, MacKenzie CR. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J. Chronic Dis. 1987;40(5):373–383. doi: 10.1016/0021-9681(87)90171-8. [DOI] [PubMed] [Google Scholar]
- 27.Peduzzi P, Concato J, Kemper E, Holford TR, Feinstein AR. A simulation study of the number of events per variable in logistic regression analysis. J. Clin. Epidemiol. 1996;49(12):1373–1379. doi: 10.1016/s0895-4356(96)00236-3. [DOI] [PubMed] [Google Scholar]
- 28.Kim JH. Multicollinearity and misleading statistical results. Korean J. Anesthesiol. 2019;72(6):558–569. doi: 10.4097/kja.19087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Heymans MW, van Buuren S, Knol DL, van Mechelen W, de Vet HC. Variable selection under multiple imputation using the bootstrap in a prognostic study. BMC Med. Res. Methodol. 2007;7:33. doi: 10.1186/1471-2288-7-33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Steyerberg E. Clinical Prediction Models: A Practical Approach to Development, Validation, and Updating. Springer Nature Switzerland AG; 2019. [Google Scholar]
- 31.Harrell FE. Regression modeling strategies with applications to linear models, logistic and ordinal regression, and survival analysis. New York: Springer; 2015. [Google Scholar]
- 32.Borrat-Besson, C., Ryser, V.-A., & Wernli, B. Transitions between frailty states—a European comparison. Active ageing and solidarity between generations in Europe: De Gruyter, 175–186 (2013)
- 33.Ye B, et al. Changes in frailty among community-dwelling Chinese older adults and its predictors: evidence from a two-year longitudinal study. BMC Geriatr. 2020;20(1):130. doi: 10.1186/s12877-020-01530-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Ahmad NS, et al. Prevalence, transitions and factors predicting transition between frailty states among rural community-dwelling older adults in Malaysia. PLoS ONE. 2018;13(11):e0206445. doi: 10.1371/journal.pone.0206445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Pollack LR, Litwack-Harrison S, et al. Patterns and predictors of frailty transitions in older men: the osteoporotic fractures in men study. J. Am. Geriatr. Soc. 2017;65(11):2473–2479. doi: 10.1111/jgs.15003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Cawthon PM, et al. Frailty in older men: prevalence, progression, and relationship with mortality. J. Am. Geriatr. Soc. 2007;55(8):1216–1223. doi: 10.1111/j.1532-5415.2007.01259.x. [DOI] [PubMed] [Google Scholar]
- 37.Altman DG, Vergouwe Y, Royston P, Moons KG. Prognosis and prognostic research: validating a prognostic model. BMJ. 2009;338:605. doi: 10.1136/bmj.b605. [DOI] [PubMed] [Google Scholar]
- 38.Altman DG, Royston P. What do we mean by validating a prognostic model? Stat. Med. 2000;19(4):453–473. doi: 10.1002/(sici)1097-0258(20000229)19:4<453::aid-sim350>3.0.co;2-5. [DOI] [PubMed] [Google Scholar]
- 39.Apostolo J, et al. Effectiveness of interventions to prevent pre-frailty and frailty progression in older adults: a systematic review. JBI Database Syst. Rev. Implement Rep. 2018;16(1):140–232. doi: 10.11124/JBISRIR-2017-003382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Luger E, et al. Effects of a home-based and volunteer-administered physical training, nutritional, and social support program on malnutrition and frailty in older persons a randomized controlled trial. J. Am. Med. Dir. Assoc. 2016;17(7):671 e679–671 e616. doi: 10.1016/j.jamda.2016.04.018. [DOI] [PubMed] [Google Scholar]
- 41.Romera-Liebana L, et al. Effects of a primary care-based multifactorial intervention on physical and cognitive function in frail, elderly individuals: a randomized controlled trial. J. Gerontol. A Biol. Sci. Med. Sci. 2018;73(12):1688–1674. doi: 10.1093/gerona/glx259. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
There are no linked research data sets for this study. The data are available from the corresponding author OR from the study PI upon reasonable request.