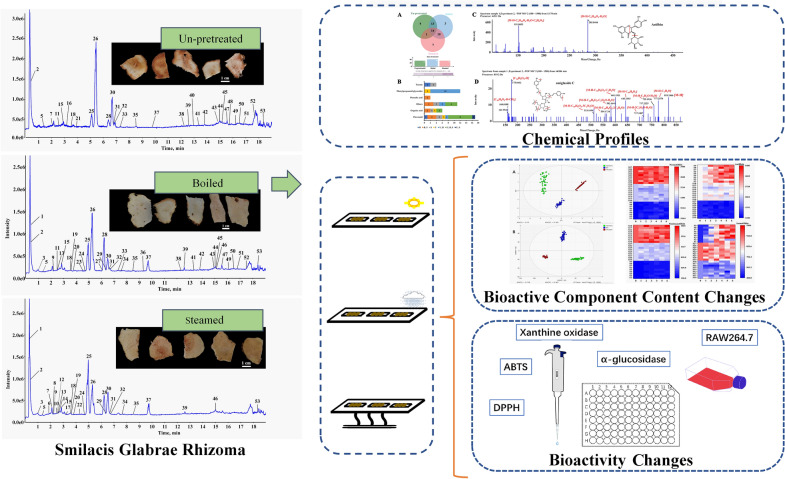
Abstract
Background
The processing of medicinal plant materials is one of the important factors influencing the components and biological activities of TCMs. Smilax glabra Roxb. is an herbal vine widely distributed in China, and its dried rhizome (Smilacis Glabrae Rhizoma, SGR) is often used in traditional medicines and functional foods. The processing methods of fresh cutting for SGR slices have been included in ancient Chinese herbal works, some local standards of TCMs, and the current Chinese Pharmacopoeia. Nevertheless, to date, the scientific basis for the processing of fresh medicinal materials for SGR slices has not been revealed.
Methods
To optimize the processing method for preparing SGR slices from the fresh rhizomes, the chemical compositions of the un-pretreated and pretreated (boiling, steaming) samples before and after drying (sun-drying, shade-drying, oven-drying), and the contents of astilbin isomers in dried SGR were analyzed by UHPLC-Q-TOF-MS/MS and UHPLC-DAD methods, respectively. Then, the antioxidant, anti-inflammatory, xanthine oxidase and α-glucosidase inhibitory activities of the prepared SGR slices were investigated by biological assays.
Results
A total of fifty-two compounds were identified from the un-pretreated and pretreated samples and a total of forty-nine compounds were identified from the subsequently dried samples. After pretreated by boiling and steaming, the contents of neoastilbin, neoisoastilbin, and isoastilbin in the prepared samples all increased. As a quality marker of SGR, the content of astilbin was unchanged or decreased slightly compared with that in the un-pretreated samples. During the drying process, the contents of the four astilbin stereoisomers in the un-pretreated samples increased significantly, while those in the pretreated samples had a slight increase or decrease. The effects of different processing methods were sorted according to the bioactivities of the prepared SGR. As a result, SGR slices prepared with no pretreatment followed by a sun-drying process have a higher astilbin content, better bioactivities and more energy savings, representing the optimum processing method for SGR slices.
Conclusions
This study reveals the scientific basis for the processing of fresh medicinal materials for SGR slices. The results provide scientific information for the quality control of SGR and its rational applications in herbal medicines and functional foods.
Graphical abstract
Supplementary Information
The online version contains supplementary material available at 10.1186/s13020-022-00614-7.
Keywords: Smilacis Glabrae Rhizoma, Pretreatment and drying method, UHPLC-Q-TOF-MS/MS and UHPLC-DAD analysis, Astilbin stereoisomers, Bioactivities
Background
Smilax glabra Roxb. (Smilacaceae) is an herbal vine that is widely distributed in China, India, Vietnam, Myanmar and Thailand [1]. The dried rhizome of Smilax glabra (Smilacis Glabrae Rhizoma, SGR) is often used as a traditional Chinese medicine (TCM), called “Tu Fu Ling” in Chinese. SGR is recorded in the Chinese Pharmacopoeia and is used to treat syphilis, furunculosis, eczema, dermatitis, nephritis, cystitis, and mercury poisoning [2]. SGR is also the main ingredient of “Gui Ling Gao” (turtle jelly), a well-known functional food popular in southern China [3, 4]. To date, modern scientific studies on SGR have demonstrated that it possesses multiple biological and pharmacological activities, including anti-inflammatory [5, 6], antioxidant [7], antipsoriasis [8], anticancer [9, 10], and antihyperuricemia [6] effects. In addition, according to the recent studies, SGR also exhibits strong inhibitory activity against α-glucosidase (α-Glu) [11] and has an antidiabetic effect [12].
It is generally believed that the multiple biological and pharmacological activities of TCMs are ascribed to their chemical components. The main compounds in SGR are flavonoids, phenylpropanoids and organic acids [13–16]. Flavonoids, such as astilbin and its stereoisomers are regarded as the main bioactive compounds in SGR [13, 14]. Astilbin has been used as a marker for SGR quality control according to the Chinese Pharmacopoeia [2].
Quality control of the main bioactive components is important for the safety, efficacy and consistency of TCMs and functional foods. The processing of plant materials is one of the important factors influencing their components and biological activities [17–19]. Traditionally, the processing method for SGR is very simple. The unsliced dried rhizome is washed and soaked with water and then sliced and dried. However, SGR needs a long time for soaking and rinsing before slicing, which might cause the loss of active components. Moreover, SGR is tough and fibrous, so fresh cutting of SGR is relatively easy, and the morphology of the slices changes minimally after drying. The processing methods of fresh cutting for SGR slices have been included in ancient Chinese herbal works, some local processing standards of TCMs, and the current Chinese Pharmacopoeia [2]. Nevertheless, to date, the scientific basis for the processing of fresh medicinal materials to prepare SGR slices has not been revealed [20].
Some Chinese herbal works recorded that the fresh rhizomatic materials of S. glabra could be pretreated by “boiling” and “steaming” and then sliced and dried [21]. However, hot water boiling of rhizomatic herbal materials could cause starch gelatinization and might inactivate glycoside enzymes in the plant materials [22, 23]. To date, no reports have indicated whether the effective ingredients and biological activities of SGR are affected by “boiling” and “steaming” pretreatments.
Drying technology is widely used in the fields of herbs and foods as an initial processing method for preservation and extending the shelf life [17, 18]. Some low-cost drying methods that are commonly used to process herbs and foods include sun-drying, shade-drying and oven-drying. As a result of different drying methods and treatment conditions, the drying process may prevent the loss of some components [24], or may cause the loss of some components. In previous studies on the processing of SGR, certain drying methods were studied, which were mainly energy-consuming drying methods [20, 25]. There was also a report about rational analysis of peeling or nonpeeling of SGR [26]. In addition, the sulfur fumigation of SGR and its effect on the quality of the herb have been studied [27]. However, the pretreatment methods recorded in some Chinese herbal works and the changes in chemical composition during the drying process have not yet been reported. The relationships between the drying methods and SGR bioactivity remain unclear.
To optimize the processing methods for SGR slices produced from the fresh rhizomes, the effects of “boiling” and “steaming” pretreatments and three drying methods, namely, sun-drying (Su.D), shade-drying (Sh.D) and oven-drying (Ov.D) treatments, on the chemical components and bioactivities of SGR were studied. The chemical profiles of pretreated and un-pretreated SGR samples and the subsequently dried samples dealt with different drying methods were analyzed and compared by ultra high-performance liquid chromatography/quadrupole time-of-flight mass spectrometry (UHPLC-Q-TOF-MS/MS). The contents of astilbin and its three stereoisomers in SGR were determined using ultra high-performance liquid chromatography coupled with diode array detection (UHPLC-DAD). The relationships between the different pretreatments and drying methods and the biological activities of the prepared dried SGR slices were also analyzed.
Methods
Materials and chemical reagents.
Rhizomes of S. glabra were collected from Lishui (Zhejiang Province, China) in November 2020 and identified by Professor Minjian Qin. The voucher specimen (No. TFL2020110501) was deposited at the Center of Herbarium, China Pharmaceutical University, Nanjing, China (Herbarium Code: CPU).
Standard reference compounds of astilbin, neoastilbin, neoisoastilbin, and isoastilbin were purchased from Chengdu Push Bio-technology Co., Ltd. (Chengdu, China), and their purity was more than 98%. 2,2′-Azino-bis (3-ethylbenzothiazoline-6-sulfonic acid) diammonium salt (ABTS, A1888), 1,1-diphenyl-2-picrylhydrazyl (DPPH), xanthine oxidase (XO, X1875), α-Glu (G5003), and lipopolysaccharide (LPS, L2880) were purchased from Sigma–Aldrich (Shanghai, China). The nitric oxide (NO) assay kit was provided by Beyotime Co., Ltd. (Shanghai, China). Chromatography grade acetonitrile and formic acid were purchased from Merck (Darmstadt, Germany) and Macklin (Shanghai, China), respectively. All the other chemicals and reagents used were of analytical grade.
Processes for pretreatment and drying
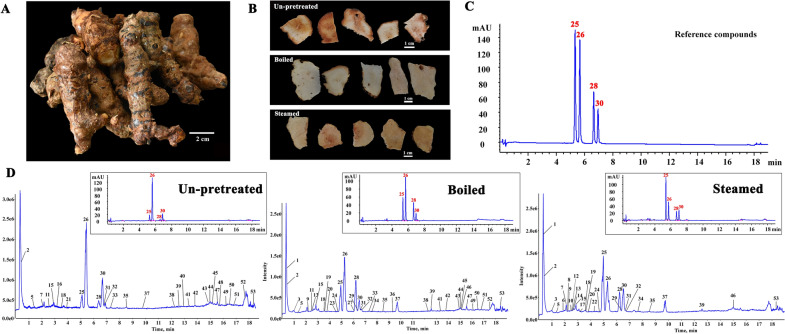
Fresh rhizomes of S. glabra were collected and sent to the laboratory within 24 h, and their fibrous roots were removed (Fig. 1A). Then, these fresh rhizomes were divided into three groups: in the un-pretreated group (un-pretreated), samples were sliced directly (with a slice thickness of 1 mm) and then dried; in the boiled group (boiled), samples were boiled in boiling water for 15 min prior to slicing and drying; and in the steamed group (steamed), samples were steamed at 121 °C for 30 min in an autoclave (GI54DS, Zealway Instrument Inc, Xiamen, Fujian, China) and then sliced and dried (Fig. 1B). The drying process included three conditions for each group: sun-drying (Su.D, dried under the sunlight, 18–20 °C, ventilation), shade-drying (Sh.D, dried in a room, 25 °C, ventilation), and oven-drying (Ov.D) at temperatures ranging from 45 °C to 105 °C at 10 °C intervals using a thermostatic oven (GZX-9246 MBE, Boxun Industrial Co., Ltd., Shanghai, China). The sampling intervals are summarized in Table 1 and Additional file 1: Table S1. At each sampling point, three aliquots (20.0 g) were randomly taken and placed in a desiccator for cooling and then pulverized in liquid nitrogen (20 mesh). The moisture content of each sample at different sampling points was measured using a digital moisture analyzer (Sartorius MA 35, Sartorius AG, Göttingen, Germany) at 105 °C according to the method established by our laboratory [28], and all assays were performed in triplicate. The remaining samples were stored at −80 °C to determine the content of bioactive components. The final sampling points of the processes were set as points at which the moisture content of the samples reached the desired standard, which is less than 15.0% according to the Chinese Pharmacopoeia [2].
Fig. 1.
Characterization and content analysis of Smilacis Glabrae Rhizoma (SGR) A Fresh SGR. B Slices of SGR. C UHPLC chromatograms of reference compounds at 291 nm. (25: neoastilbin, 26: astilbin, 28: neoisoastilbin, 30: isoastilbin). D TIC chromatogram of three pretreated SGR samples in negative ionization mode obtained by UHPLC-Q-TOF-MS/MS and UHPLC chromatograms of SGR samples at 291 nm. (25: neoastilbin, 26: astilbin, 28: neoisoastilbin, 30: isoastilbin)
Table 1.
Sampling interval, drying efficiency of SGR (n = 3)
| Drying methods | Sampling interval | Un-pretreated | Boiled | Steamed | |||
|---|---|---|---|---|---|---|---|
| No | Drying time (Final moisture %) | No | Drying time (Final moisture %) | No | Drying time (Final moisture%) | ||
| Su.D | 40 min | S1 | 200 min (12.91) | S10 | 200 min (12.28) | S19 | 200 min (9.63) |
| Sh.D | 4 h | S2 | 20 h (13.15) | S11 | 24 h (13.61) | S20 | 20 h (13.42) |
| Ov.D | |||||||
| 45 °C | 40 min | S3 | 200 min (7.21) | S12 | 160 min (13.85) | S21 | 160 min (8.42) |
| 55 °C | 20 min | S4 | 100 min (11.48) | S13 | 100 min (10.84) | S22 | 100 min (8.41) |
| 65 °C | 20 min | S5 | 100 min (7.34) | S14 | 80 min (12.62) | S23 | 80 min (12.61) |
| 75 °C | 15 min | S6 | 60 min (14.41) | S15 | 75 min (5.61) | S24 | 60 min (14.29) |
| 85 °C | 12 min | S7 | 60 min (5.01) | S16 | 60 min (3.97) | S25 | 48 min (13.28) |
| 95 °C | 10 min | S8 | 50 min (4.67) | S17 | 50 min (5.06) | S26 | 40 min (12.9) |
| 105 °C | 8 min | S9 | 40 min (6.57) | S18 | 40 min (8.38) | S27 | 40 min (5.09) |
The 189 samples prepared under different treatment conditions (three pretreatment methods combined with nine drying procedures) were used for determining the contents of four astilbin stereoisomers and the moisture content. The samples obtained at the final sampling points including 27 batches of dried SGR sliced samples were used to evaluate the antioxidant, XO inhibition, α-Glu inhibition, and anti-inflammatory activities.
Preparation of the solution of standard reference compounds
Four standard reference compounds, namely, astilbin (1.96 mg), neoastilbin (1.94 mg), neoisoastilbin (2.06 mg), and isoastilbin (2.00 mg), were dissolved with methanol into a 1 mL volumetric flask to prepare stock solutions of the standard reference compounds. The mixed stock solution containing the four standard reference compounds was serially diluted for the construction of calibration curves and method validation.
Extraction of bioactive compounds from the SGR samples
To optimize the conditions for the extraction of bioactive components in SGR, response surface methodology (RSM) was carried out. The methods and results are shown in Additional file 2. According to the results of the RSM study, the extraction of bioactive components from the SGR samples was performed as follows. Powder of the prepared SGR sample (0.3 g DW, 20 mesh) was placed in a 50 mL centrifuge tube and mixed with 9 mL of 71% ethanol. The mixed solution was extracted twice by an ultrasonic bath (KH-300DB, Kunshan Ultrasonic Instrument Co., Ltd., Jiangsu, China), each time for 32 min. After ultrasonic extraction, the extract was centrifuged at 10,000 rpm for 10 min. The supernatant was collected in a 25 mL brown volumetric flask and diluted to volume with 71% ethanol. The samples were filtered through a 0.22 μm microfiltration membrane before UHPLC analysis.
UHPLC-Q-TOF-MS/MS and UHPLC-DAD conditions
UHPLC-Q-TOF-MS/MS analysis was performed on an Agilent Series 1290 UHPLC instrument (Agilent Technologies, Cambridge, MA, USA) and AB SCIEX TripleToF® 4600 mass analyzer (AB SCIEX, Concord, ON, Canada) operating separately in negative and positive ion modes. Chromatography was performed at 40 °C on an Agilent ZORBAX Eclipse Plus C18 column (2.1 × 50 mm, 1.8-Micro) with a flow rate of 0.4 mL/min; the injection volume was 2 μL. The mobile phase consisted of solvents A (0.1% aqueous formic acid, v/v) and B (acetonitrile). The gradient elution program was as follows: 0–1 min, 5–7% B; 1–2 min, 7–11% B; 2–6 min, 11–16% B; 6–9.5 min, 16–18% B; 9.5–10 min, 18–18% B; 10–14 min, 18–28% B; 14–14.5 min, 28–35% B; 14.5–15.5 min, 35–35% B; 15.5–16 min, 35–40% B; 16–17 min, 40–40% B; 17–17.5 min, 40–95% B; and 17.5–19 min, 95–95% B. The following parameter settings were also used: MS survey scan of 100–1500 Da (250 ms accumulation time) and MS/MS survey scan of 100–1500 Da (100 ms accumulation time) with declustering potential of 100 V; ion source voltage, 4500 V; ion source heater, 550 °C; and collision energy, 44 V. The MS/MS data were analyzed using PeakView®–Analyst® TF 1.6 software (AB SCIEX, Concord, ON, Canada).
The contents of astilbin and its three stereoisomers (neoastilbin, neoisoastilbin, and isoastilbin) in each SGR sample were determined using an Agilent Series 1290 UHPLC instrument (Agilent Technologies, Cambridge, MA, USA) equipped with a DAD detector (Cambridge, MA, USA). The chromatography conditions were the same as those used for UHPLC-Q-TOF-MS/MS analysis, and the DAD wavelength was set to 291 nm. Each sample was analyzed in triplicate.
Antioxidant activity assays
For the DPPH· radical activity assay [29], briefly, 50 μL of a serially diluted sample solution was added to 150 μL of DPPH (75 μM). The solutions were shaken vigorously and placed in the dark at room temperature (RT) for 30 min. The absorbance (OD) was measured at 517 nm using an Epoch microplate spectrophotometer (BioTek, USA). Triplicate measurements were performed in all experiments. Ascorbic acid was used as the positive control. The DPPH· radical scavenging activity was calculated using the following equation:
For the ABTS+ radical activity assay [30], ABTS+ was produced by reacting 7 mM ABTS aqueous solution with 2.45 mM potassium persulfate aqueous solution. After 12 h in the dark, the solution was diluted with PBS until the OD value was approximately 0.7 at 734 nm. The SGR sample solution (50 μL) was mixed with 150 μL of ABTS+ for 30 min and then measured at a wavelength of 734 nm. Triplicate measurements were performed in all experiments. Ascorbic acid was used as the positive control. The ABTS+ radical scavenging activity was calculated using the following equation:
Enzyme inhibition assay
XO inhibition assay
The inhibitory activity of the SGR sample against XO was assessed spectrophotometrically, following the method of Yin Wan et al. [31], with some modifications. XO, xanthine, and 4-nitro blue tetrazolium chloride (NBT) were dissolved in phosphate buffer (50 mM, pH 7.8). A 10 μL portion of the SGR extract and 10 μL of XO (0.6 U/mL) were added to 250 μL of phosphate buffer and incubated at 37 °C for 15 min. Subsequently, the solution was mixed with 20 μL of xanthine (30 mM) and 10 μL of NBT (0.6 mM) and incubated at 37 °C for 30 min, and then the absorbance was measured with a spectrophotometer at a wavelength of 560 nm. The sample without SGR extract was used as the control and allopurinol was used as a positive control. The percentage of XO inhibition was calculated as follows:
α-Glu inhibition assay
α-Glu inhibition was assessed according to the literature with modifications [32]. α-Glu (0.04 U/mL) and substrate p-nitrophenyl-α-d-glucopyranoside (PNPG, 0.4 mM) were dissolved in phosphate buffer (0.1 M, pH 6.8). Briefly, 50 μL of SGR at different concentrations was mixed with 70 μL of α-Glu solution and incubated at 37 °C for 5 min. Subsequently, 50 μL of PNPG was added to start the reaction, and then, the solution was incubated at 37 °C for 30 min. The reaction was stopped by the addition of 80 μL of Na2CO3 aqueous solution (0.2 M). After 5 min at RT, the absorbance of the sample was measured at a wavelength of 405 nm. Acarbose was used as a positive control, and α-Glu inhibitory activity was calculated using the following formula:
Anti-inflammatory activity
Cell culture and viability assay
RAW264.7 macrophages were obtained from the Department of Pharmacology of Traditional Chinese Medicines, China Pharmaceutical University (Nanjing, China), and were cultured in Dulbecco’s modified essential medium (DMEM, Gibco) containing 10% fetal bovine serum (FBS, Sigma–Aldrich) and penicillin and streptomycin (Biosharp) at 37 °C in a humidified 5% CO2 incubator (3111 type, Thermo Fisher Scientific). Cells were seeded in a 96-well culture plate at a density of 10 × 105 cells/well and allowed to attach at 37 °C for 24 h [33]. Cells were treated with SGR extracts (final concentration, low (L), medium (M) and high (H) dose of 0.53, 1.06, and 2.13 mg/mL, respectively) and LPS (final concentration, 1 µg/mL) for an additional 24 h. The formazan crystals were dissolved in 150 µL of dimethyl sulfoxide (DMSO) for 15 min, and the absorbance was detected at 570 nm.
Determination of NO concentrations
The RAW264.7 macrophages were plated at a density of 10 × 105 cells/mL and cultured for 24 h. Then, the cells were incubated with LPS (final concentration, 1 µg/mL) and SGR extracts (final concentration, low (L), medium (M) and high (H) dose of 0.53, 1.06, and 2.13 mg/mL, respectively) and cultured for 24 h at 37 °C. Then, the cell culture supernatant was collected for determination of the NO concentration using the manufacturer’s instructions. The detection wavelength was 540 nm and NaNO2 was used as the standard.
Statistical analysis
Data are presented as the mean ± standard deviation, and the mean values of the analysis and cell experiment were from three and six independent replicates, respectively. Statistical analysis was performed using Microsoft Excel 2016 (Microsoft Inc., Redmond WA, USA); the IC50 values were calculated using GraphPad Prism 7 (San Diego, CA, USA); heatmaps were generated using Origin 2019b software (OriginLab Corporation, Northampton, MA, USA); and SIMCA 13.0 software (Umetrics, Umea, Sweden) was used for principal component analysis (PCA) and orthogonal projections to latent structures discriminant analysis (OPLS-DA).
Results
Drying efficiencies
The initial moisture content of the un-pretreated SGR sample was 56.49 ± 1.66%. After pretreatment, the moisture content of the boiled and steamed samples was 55.14 ± 1.61% and 53.15 ± 1.8%, respectively. This result indicated that the moisture content was slightly reduced after performing the two pretreatment methods. The dried SGR slice samples and drying efficiencies are shown in Table 1.
The Sh.D samples required 20 h to be dried, and the final moisture contents reached the desired standard of less than 15.0% according to the Chinese Pharmacopoeia. However, the Su.D and Ov.D samples took less than 3 h to be dried within a moisture content of 15.0%. The drying efficiencies of Ov.D treatment were positively correlated with the temperature.
The contents of four astilbin stereoisomers determined by UHPLC-DAD
The contents of astilbin, neoastilbin, neoisoastilbin, and isoastilbin in the SGR samples were simultaneously determined by the UHPLC-DAD method. Validation of the UHPLC analysis method was carried out, with the established regression equations for determining the four astilbin stereoisomers shown in Table 2, and the limit of detection (LOD), limit of quantitation (LOQ), stability, precision, and reproducibility of each compound were determined (Additional file 1: Tables S2 and S3). UHPLC chromatograms of standard reference compounds are shown in Fig. 1C. All calibration curves showed good linear correlation (R2 > 0.9993) within the linear ranges. The results indicated that the method employed was rapid, accurate, and precise for determining the contents of the four major compounds in the SGR samples. The contents of the four astilbin stereoisomers are shown in Additional file 1: Tables S4–12.
Table 2.
Calibration curve equations of astilbin, neoastilbin, neoisoastilbin, and isoastilbin
| Peak | Rt ( min) | Compounds | Regression equation | R2 |
|---|---|---|---|---|
| 24 | 4.565 | Neoastilbin | y = 12.926 × − 2.3048 | 0.9993 |
| 25 | 4.879 | Astilbin | y = 12.677 × − 2.2413 | 0.9993 |
| 27 | 5.757 | Neoisoastilbin | y = 11.606 × − 1.5159 | 0.9993 |
| 29 | 6.098 | Isoastilbin | y = 13.034 × − 1.2695 | 0.9993 |
Chemical profiles of the SGR samples after different pretreatments
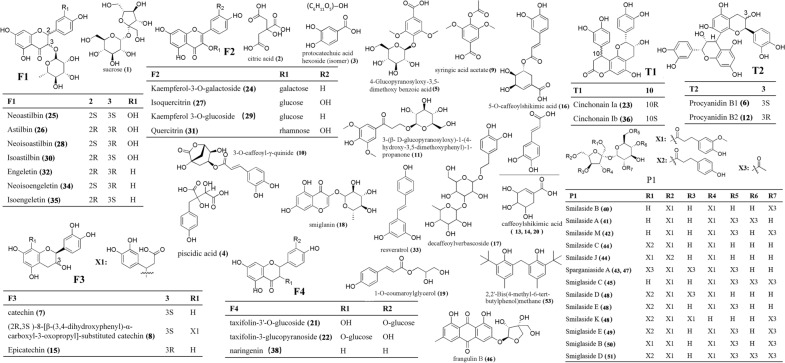
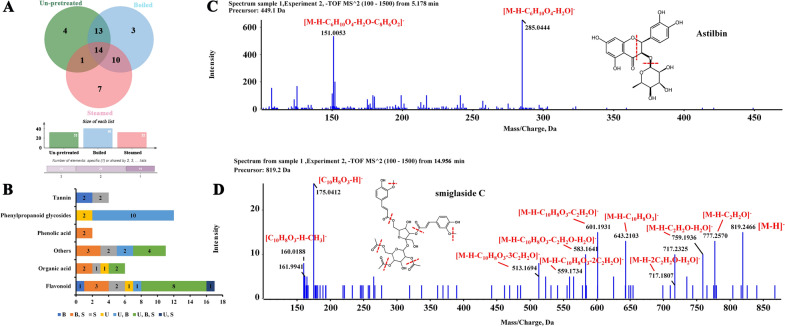
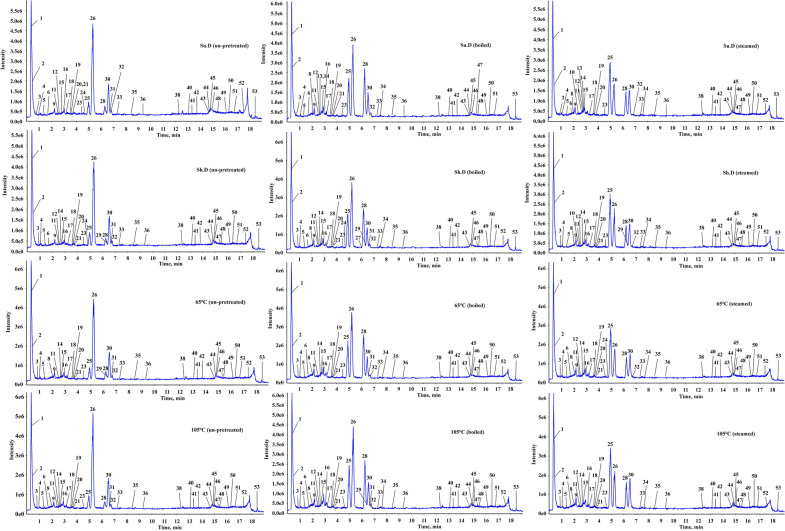
The un-pretreated samples and the samples pretreated by boiling and steaming prior to being dried (Table 3, 0 min) were analyzed by UHPLC-Q-TOF-MS/MS. Both positive and negative ion modes were used, but the peak shape and the numbers of peaks were better in negative ion mode. The total ion chromatograms (TICs) and MS data of the extracts of un-pretreated, boiled, and steamed SGR samples in negative ESI mode are shown in Fig. 1D. A total of fifty-two compounds were detected and identified from the three groups of samples (Fig. 2, Table 3), including seventeen flavonoids, twelve phenylpropanoid glycosides, six organic acids, two phenolic acids, four tannins, and eleven other compounds. Thirty-two compounds were identified from the un-pretreated SGR samples. Forty compounds were identified from the boiled SGR samples. Thirty-two compounds were identified from the steamed SGR samples (Fig. 3A). The three groups of samples shared fourteen compounds, eight of which were flavonoids, namely, epicatechin, neoastilbin, astilbin, neoisoastilbin, isoastilbin, quercitrin, engeletin, and isoengeletin. 5-O-Caffeoylshikimic acid, taxifolin-3'-O-glucoside, smilaside B and smilaside D (or smilaside E or smilaside K) were found only in un-pretreated samples, isoquercitrin, cinchonain Ia and cinchonain Ib were found only in boiled samples, and seven compounds including procyanidin B1, decaffeoylverbascoside, (2R,3S)-8-[β-(3,4-dihydroxyphenyl)-α-carboxyl-3-oxopropyl]-substituted catechin, 3-O-caffeoyl-γ-quinide, procyanidin B2, caffeoylshikimic acid III, and taxifolin-3-glucopyranoside were found only in steamed group samples (Fig. 3B, Table 3). In addition, two phenolic acids (protocatechuic acid hexoside (isomer) and syringic acid acetate) were detected only in boiled and steamed samples, and ten phenylpropanoid glycosides including smilaside A, smilaside M, sparganiaside A, smiglaside C, smilaside C (or smilaside J), smiglaside E, smiglaside B, and smiglaside D were detected only in un-pretreated and boiled groups of samples.
Table 3.
UHPLC-MS/MS chemical profiling of SGR after different pretreatments and drying
| Peak no. | Rt (min) | Selected ion | Observed/ calculate mass (m/z) | Error | Formula | MS/MS fragmentation patterns | 0 min | Su.D | Sh.D | Ov.D | Identification | Ref | Classification | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 65℃ | 105℃ | |||||||||||||
| 1 | 0.333 | [M-H]− | 341.1093/341.1089 | 1.1 | C12H22O11 | 179, 113, 101 | B, S | U, B, S | U, B, S | U, B, S | U, B, S | Sucrose | [34] | Others |
| 2 | 0.373 | [M-H]− | 191.0206/191.0197 | 4.5 | C6H8O7 | 111 | U, B, S | U, B, S | U, B, S | U, B, S | U, B, S | Citric acid | [35] | Organic acid |
| 3 | 0.967 | [M-H]− | 315.0726/315.0722 | 1.5 | C13H16O9 | 152, 109, 108 | B, S | U, B, S | U, B, S | U, B, S | U, B, S | Protocatechuic acid hexoside (isomer) | [36–38] | Phenolic acid |
| 4 | 1.037 | [M-H]− | 255.0519/255.051 | 3.4 | C11H12O7 | 179,165,125,107 | U, B, S | U, B, S | U, B, S | U, B, S | piscidic acid | [39] | Organic acid | |
| 5 | 1.36 | [M-H]− | 359.098/359.0984 | 2 | C15H20O10 | 197, 182, 153, 138, 123 | U, B, S | U, B, S | U, B, S | U, B, S | U, B, S | 4-glucopyranosyloxy-3,5-dimethoxy benzoic acid | [40] | Organic acid |
| 6 | 1.845 | [M-H]− | 577.1378/577.1352 | 4.6 | C30H26O12 | 425, 407, 381, 289, 245, 125 | S | U, B, S | U, B | B, S | U, B, S | Procyanidin B1 | [37] | Tannin |
| 7 | 2.111 | [M-H]− | 289.0722/289.0718 | 1.5 | C15H14O6 | 271, 245, 205, 179, 151 | U, S | Catechin | [14, 35] | Flavonoid | ||||
| 8 | 2.256 | [M-H]− | 469.1147/469.114 | 1.4 | C24H22O10 | 315, 289, 247, 205, 109 | S | B, S | B, S | U, B, S | U, B, S | (2R,3S)-8-[β-(3,4-dihydroxyphenyl)-α-carboxyl-3-oxopropyl]-substituted catechin | [14, 41] | Flavonoid |
| 9 | 2.262 | [M-H]− | 239.0556/239.0561 | − 2.1 | C11H12O6 | 149, 133, 107 | B, S | U, B, S | U, B, S | U, B, S | U, B, S | Syringic acid acetate | [42] | Phenolic acid |
| 10 | 2.445 | [M-H]− | 335.0783/335.0772 | 3.1 | C16H16O8 | 291, 179, 161, 135 | S | S | S | 3-O-caffeoyl-γ-quinide | [43] | Others | ||
| 11 | 2.556 | [M-H]− | 387.1290/387.1297 | -1.7 | C17H24O10 | 207, 192, 177 | U, B | U, B, S | U, B, S | U, B, S | U, B, S | 3-(β-d-glucopyranosyloxy)-1-(4-hydroxy-3,5-dimethoxyphenyl)-1-propanone | [14] | Others |
| 12 | 2.579 | [M-H]− | 577.1375/577.1352 | 4 | C30H26O12 | 425, 407, 381, 289, 245, 125 | S | U, B, S | U, B, S | S | U, B, S | Procyanidin B2 | [37] | Tannin |
| 13 | 2.626 | [M-H]− | 335.0760/335.0772 | − 3.6 | C16H16O8 | 291, 179, 161, 135 | B, S | B, S | (Trans) caffeoylshikimic acid II | [44] | Organic acid | |||
| 14 | 2.748 | [M-H]− | 335.0765/335.0772 | − 2.1 | C16H16O8 | 291, 179, 161, 135 | S | B, S | U, B, S | U, B, S | U, B, S | Caffeoylshikimic acid III | [44] | Organic acid |
| 15 | 2.874 | [M-H]− | 289.0716/289.0718 | − 0.5 | C15H14O6 | 205, 201, 187, 151, 123, 109 | U, B, S | U, B, S | U, B, S | U, B, S | U, B, S | Epicatechin | [35] | Flavonoid |
| 16 | 3.078 | [M-H]− | 335.0775/335.0772 | 0.8 | C16H14O8 | 291, 179, 161, 135 | U | U, B | U, B, S | U, B, S | U, B, S | 5-O-caffeoylshikimic acid | [14, 45] | Organic acid |
| 17 | 3.134 | [M-H]− | 461.1687/461.1665 | 4.8 | C20H30O12 | 415, 191, 149, 113 | S | U, B, S | U, B, S | U, B, S | U, B, S | Decaffeoylverbascoside | [46] | Others |
| 18 | 3.582 | [M-H]− | 339.0730/339.0722 | 2.5 | C15H16O9 | 193, 192 | U, B, S | U, B, S | U, B, S | U, B, S | U, B, S | Smiglanin | [14, 47] | Others |
| 19 | 3.608 | [M-H]− | 237.0770/237.0769 | 0.6 | C12H14O5 | 145,119,117 | B, S | U, B, S | U, B, S | U, B, S | U, B, S | 1-O-coumaroylglycerol | [48] | Others |
| 20 | 3.817 | [M-H]− | 335.0780/335.0772 | 2.3 | C16H16O8 | 291, 179, 161, 135 | B, S | U, B, S | U, B, S | U, B, S | U, B, S | Caffeoylshikimic acid IV | [44] | Organic acid |
| 21 | 3.899 | [M-H]− | 465.1047/465.1039 | 1.9 | C21H22O12 | 437, 303, 285, 179, 151 | U | U, B | U, B, S | U, B, S | U, B, S | Taxifolin-3'-O-glucoside | [36] | Flavonoid |
| 22 | 4.218 | [M-H]− | 465.1045/465.1039 | 1.4 | C21H22O12 | 303, 285, 275, 151 | S | Taxifolin-3-glucopyranoside | [49] | Flavonoid | ||||
| 23 | 4.774 | [M-H]− | 451.1050/451.1034 | 3.4 | C24H20O9 | 341 | B | U, B, S | U, B, S | U, B, S | U, B, S | Cinchonain Ia | [41] | Tannin |
| 24 | 4.925 | [M-H]− | 447.0931/447.0933 | − 0.5 | C21H20O11 | 283, 255, 239, 211 | B, S | U | U, B | S | Kaempferol-3-O-galactoside | [50] | Flavonoid | |
| 25 | 4.952 | [M-H]− | 449.1111/449.1089 | 4.9 | C21H22O11 | 303, 285, 151 | U, B, S | U, B, S | U, B, S | U, B, S | U, B, S | Neoastilbin | [14] | Flavonoid |
| 26 | 5.183 | [M-H]− | 449.1098/449.1089 | 1.9 | C21H22O11 | 303, 285, 151 | U, B, S | U, B, S | U, B, S | U, B, S | U, B, S | Astilbin | [45] | Flavonoid |
| 27 | 6.194 | [M-H]− | 463.0865/463.0882 | − 3.7 | C21H20O12 | 299, 271, 255, 243, 231 | B | B | Isoquercitrin | [51] | Flavonoid | |||
| 28 | 6.201 | [M-H]− | 449.1099/449.1089 | 2.1 | C21H22O11 | 303, 285, 151 | U, B, S | U, B, S | U, B, S | U, B, S | U, B, S | Neoisoastilbin | [14] | Flavonoid |
| 29 | 6.225 | [M-H]− | 447.0947/447.0933 | 0.9 | C21H20O11 | 285, 255, 239, 179, 159 | B, S | U, B, S | U | B | Kaempferol 3-O-glucoside | [50, 52] | Flavonoid | |
| 30 | 6.457 | [M-H]− | 449.1099/449.1089 | 2.1 | C21H22O11 | 303, 285, 151 | U, B, S | U, B, S | U, B, S | U, B, S | U, B, S | Isoastilbin | [14, 36] | Flavonoid |
| 31 | 6.634 | [M-H]− | 447.0947/447.0933 | 3.2 | C21H20O11 | 301, 283, 255, 239, 179, 151 | U, B, S | U | U, B | U, B | U, B | Quercitrin | [53] | Flavonoid |
| 32 | 6.658 | [M-H]− | 433.1151/433.1140 | 2.5 | C21H22O10 | 287, 269, 259, 178, 152 | U, B, S | U, B, S | U, B, S | U, B, S | U, B | Engeletin | [14, 45, 52] | Flavonoid |
| 33 | 7.297 | [M-H]− | 227.0722/227.0714 | − 4.7 | C14H12O3 | 185, 183, 159, 157, 143 | U, B | U, B, S | U, B, S | U, B, S | U, B, S | Resveratrol | [14, 52, 54] | Others |
| 34 | 7.615 | [M-H]− | 433.1143/433.1140 | 0.6 | C21H22O10 | 287, 269, 259, 180, 152 | B, S | B, S | B, S | B, S | B, S | Neoisoengeletin | [14, 52] | Flavonoid |
| 35 | 8.466 | [M-H]− | 433.1147/433.1140 | 1.6 | C21H22O10 | 287, 269, 259, 180, 152 | U, B, S | U, B, S | U, B, S | U, B, S | U, B, S | Isoengeletin | [14, 52] | Flavonoid |
| 36 | 9.389 | [M-H]− | 451.1023/451.1034 | − 2.6 | C24H20O9 | 341 | B | U, B, S | U, B, S | U, B, S | U, B, S | Cinchonain Ib | [41, 52] | Tannin |
| 37 | 9.59 | [M + CH3COOH-H]− | 723.5067/723.5053 | 2 | C41H72O10 | 677, 451, 225 | U, B, S | Unknown | Others | |||||
| 38 | 12.393 | [M-H]− | 271.0620/271.0612 | 3 | C15H12O5 | 177, 151, 119, 107 | U, B | U, B, S | U, B, S | U, B, S | U, B, S | Naringenin | [14] | Flavonoid |
| 39 | 12.521 | [M-H] + | 453.3441/453.3422 | 4.2 | C23H50O8 | 569, 343, 326, 227, 209, 111, 100 | U, B, S | Unknown | Others | |||||
| 40 | 13.11 | [M-H]− | 735.2151735.2142 | 1.2 | C34H40O18 | 559, 499, 193, 175, 160 | U | U, B, S | U, B, S | U, B, S | U, B, S | Smilaside B | [36] | Phenylpropanoid glycosides |
| 41 | 13.413 | [M-H]− | 777.2269/777.2247 | 2.8 | C36H42O19 | 735, 717, 601, 559, 499, 193, 175, 160 | U, B | U, B, S | U, B, S | U, B, S | U, B, S | Smilaside A | [36, 55] | Phenylpropanoid glycosides |
| 42 | 13.834 | [M-H]− | 777.2282/777.2247 | 4.4 | C36H42O19 | 735, 717, 601, 559, 193, 175, 160 | U, B | U, B, S | U, B, S | U, B, S | U, B, S | Smilaside M | [36, 56] | Phenylpropanoid glycosides |
| 43 | 14.922 | [M-H]− | 819.2362/819.2353 | 1.0 | C38H44O20 | 777, 759, 583, 559, 193, 175, 160 | U, B | U, B, S | U, B, S | U, B, S | U, B, S | Sparganiaside A or Phenylpropanoid glycosides | [14, 15, 36] | Phenylpropanoid glycosides |
| 44 | 14.939 | [M-H]− | 839.2393/839.2404 | − 1.3 | C41H44O19 | 693, 663, 645, 483, 319, 217, 175, 145 | U, B | U, B, S | U, S | U, B, S | U, B, S | Smilaside C or smilaside J | [36] | Phenylpropanoid glycosides |
| 45 | 14.956 | [M-H]− | 819.2383/819.2353 | 3.6 | C38H44O20 | 777, 717, 601, 583, 513, 175 | U, B | U, B, S | U, B, S | U, B, S | U, B, S | Smiglaside C | [36] | Phenylpropanoid glycosides |
| 46 | 15.046 | [M-H]− | 401.0897/401.0878 | 4.7 | C20H18O9 | 313, 121 | B, S | U, B, S | U, B, S | U, B, S | U, B, S | Frangulin B | [57] | Others |
| 47 | 15.483 | [M-H]− | 819.2364/819.2353 | 1.3 | C38H44O20 | 777, 643, 601, 583, 513, 175 | U, B | B, S | U, B, S | U, B, S | U, B, S | Sparganiaside A or phenylpropanoid glycosides | [14, 15, 36] | Phenylpropanoid glycosides |
| 48 | 15.58 | [M-H]− | 881.2553/881.2510 | 4.9 | C43H46O20 | 735, 705, 687, 559, 483, 175 | U | U, B, S | U, B, S | U, B, S | U, B, S | Smilaside D or smilaside E orsmilaside K | [36] | Phenylpropanoid glycosides |
| 49 | 16.115 | [M-H]− | 923.2647/923.2615 | 3.4 | C45H48O21 | 881, 863, 777, 747, 483 | U, B | U, B, S | U, B, S | U, B, S | U, B, S | Smiglaside E | [15, 36] | Phenylpropanoid glycosides |
| 50 | 16.463 | [M-H]− | 953.2762/953.2721 | 4.3 | C46H50O22 | 911, 777, 759, 601 | U, B | U, B, S | U, B, S | U, B, S | U, B, S | Smiglaside B | [14, 15, 36] | Phenylpropanoid glycosides |
| 51 | 16.85 | [M-H]− | 965.2769/965.2721 | 5 | C47H50O22 | 923, 905, 789, 747, 483 | U, B | U, B, S | U, B, S | U, B, S | U, B, S | Smiglaside D | [14, 15, 36] | Phenylpropanoid glycosides |
| 52 | 17.592 | [M-H]− | 965.2726/965.2721 | 0.5 | C47H50O22 | 923, 905, 789, 777, 747, 559, 483, 175 | U, B | U, S | U, B, S | U, B, S | U, B, S | Phenylpropanoid glycosides | [36] | Phenylpropanoid glycosides |
| 53 | 18.32 | [M-H]− | 339.2344/339.2330 | 4.2 | C23H32O2 | 163 | U, B, S | U, B, S | U, B, S | U, B, S | U, B, S | 2,2'-Bis(4-methyl-6-tert-butylphenol)methane | [58] | Others |
U, un-pretreated; B, boiled; S, steamed; 0 min, the samples prior to being dried; Su.D, sun-drying; Sh.D, shade-drying; Ov.D 65 ℃, oven-drying at 65 ℃; Ov.D 105 ℃, oven-drying at 105 ℃
Fig. 2.
Chemical structures of the compounds identified in SGR samples
Fig. 3.
The chemical profiles of SGR after three pretreatment methods. A Venn diagrams of identified compounds in SGR. B Distribution of different compound types. (U, un-pretreated; B, boiled; S, steamed) C MS/MS spectra of astilbin in negative ionization mode. D MS/MS spectra of smiglaside C in negative ionization mode
Flavonoids are the main components of SGR; kaempferol-3-O-galactoside (24), isoquercitrin (27), kaempferol 3-O-glucoside (29) and quercitrin (31) are flavonols, naringenin (38) is a flavanone, and the other twelve flavonoids are flavanonols. Most of the identified flavonoids belong to the O-glycosyl types of flavonoid glycosides, except four flavonoid aglycones (compounds 7, 8, 15, and 38). For flavonoid O-glycosides, including compounds 21, 22, 24, 25, 26, 27, 28, 29, 30, 31, 32, 34 and 35, the most characteristic fragmentation behavior was C-O bond cleavage, which produced the fragments [M-H-162]− or [M-H-146] − due to the losses of glucose (162 Da) and galactose (162 Da) or rhamnose (146 Da). The four main flavanonols (compounds 25, 26, 28 and 30) were unambiguously identified by the same deprotonated ions at m/z 449 (C21H21O11) and the same product ions at m/z 285.0444 and m/z 151.0053, indicating that they are stereoisomers. The fragment ion at m/z 285.0444 corresponded to a loss of rhamnose and H2O from the [M-H]− moiety. The reverse Diels–Alder (RDA) reaction of the C-ring also generated characteristic fragmentation of flavonoids, and the fragment ion at m/z 151.0053 corresponded to a loss of C8H6O2 from the [M-H-C6H10O4-H2O]− moiety. The fragmentation is shown in the Fig. 3C. By comparison with the literature and the standard reference compounds, astilbin and its stereoisomers were identified.
Phenylpropanoid glycosides existed only in the un-pretreated and boiled groups of samples and shared similar fragmentations. For example, compound 45 gave an [M-H]− ion at m/z 819.2364 (C38H43O20) in negative ion mode. The fragment ion at m/z 777.2570 corresponded to the loss of an acetyl group (C2H2O) from the [M-H]− moiety, the fragment ion at m/z 759.1936 corresponded to the loss of H2O from 777.2570, the fragment ion at m/z 601.1931 corresponded to the loss of a phenyl propyl fragment (C10H8O3) and acetyl group (C2H2O) from the [M-H]− moiety, the fragment ion at m/z 513.1452 corresponded to the loss of a phenyl propyl fragment (C10H8O3) and three acetyl groups (3C2H2O) from [M-H]− moiety, and the fragment ion at m/z 175.0412 corresponded to a loss of H from the phenyl propyl fragment (C10H8O3) (Fig. 3D). Therefore, the fragment ions of compound 45 corresponded to the successive or simultaneous losses of acetyl, methyl, H2O, phenyl and propyl moieties. By comparison with the literature[36], compound 45 was tentatively identified as smiglaside C. Compounds 43 and 47 showed the same molecular ion at m/z 819.2364 (C38H43O20) and same MS/MS fragmentation and were tentatively identified as sparganiaside A or the other phenylpropanoid glycoside compounds. Similarly, compounds 41, 42, 44, 49, 50, 51 and smiglaside C were different in the number and substitution position of acetyl, methyl, phenyl and propyl groups, which were tentatively identified as smilaside A, smilaside M, smilaside C (or smilaside J), smiglaside E, smiglaside B and smiglaside D, respectively.
In this study, four tannins were identified. Compounds 6 and 12 were found only in the steamed group of samples and showed the same molecular ion at m/z 577.1352 (C30H25O12) in negative ionization mode, characterized by MS/MS ions at m/z 425.1055, 407.0860, and 289.0800. The fragment ion at m/z 425.1055 corresponded to a loss of C8H8O3 (152 Da) from the [M-H]− moiety, the fragment ion at m/z 407.0860 corresponded to a loss of C8H10O4 (170 Da) from the [M-H]− moiety, and the fragment ion at m/z 289.0800 (C15H13O6) corresponded to a loss of C15H12O6 (288 Da) from the [M-H]− moiety. The fragment ions at m/z 245.0858, 205.0501 and 179.031 indicated the existence of a catechin (C15H14O6) moiety, which implied that compounds 6 and 12 were dimers of catechin. Based on their retention time and literature data, compounds 6 and 12 were identified as procyanidin B1 and procyanidin B2, respectively. Compounds 23 and 36 were found only in the boiled group samples and showed the same molecular ion at m/z 451.1034 (C24H19O9) in negative ionization mode, characterized by MS/MS ions at m/z 341.0637, which corresponded to a loss of C6H6O2 (110 Da) from the [M-H]− moiety. Based on their retention time and literature data, compounds 23 and 36 were identified as cinchonain Ia and cinchonain Ib, respectively.
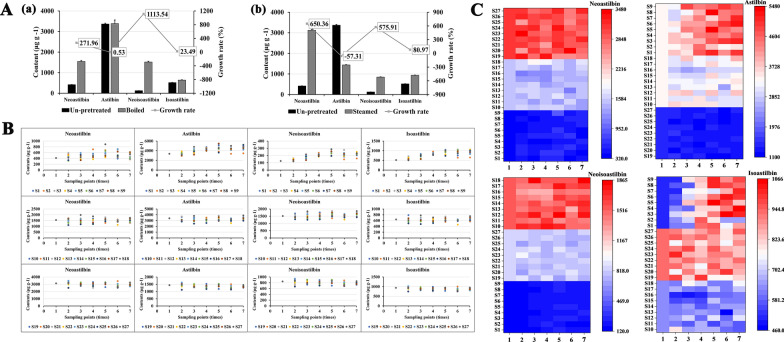
Influence of different pretreatments on the contents of astilbin stereoisomers in SGR
Before the drying process, the contents of astilbin and its three stereoisomers in the pretreated SGR samples changed significantly compared with those in the un-pretreated fresh sample. The contents of neoastilbin, neoisoastilbin, and isoastilbin all increased after pretreatment with “boiling” and “steaming”, while the astilbin content either remained the same as that of the un-pretreated sample or decreased (Table 4, Fig. 4A).
Table 4.
The content changes of the four astilbin isomers in SGR samples pretreated by different methods
| Pretreatment methods | Neoastilbin | Astilbin | Neoisoastilbin | Isoastilbin | ||||
|---|---|---|---|---|---|---|---|---|
| (μg/g) | (%) | (μg/g) | (%) | (μg/g) | (%) | (μg/g) | (%) | |
| Un-pretreated | 416.5 ± 6.96 | – | 3368.36 ± 35.51 | – | 125.05 ± 1.55 | – | 518.45 ± 7.18 | – |
| Boiled | 1549.23 ± 37.85 | + 271.96 | 3386.11 ± 174.17 | + 0.53 | 1517.53 ± 36.45 | + 1113.54 | 640.24 ± 13.12 | + 23.49 |
| Steamed | 3125.3 ± 29.42 | + 650.36 | 1437.91 ± 14.52 | − 57.31 | 845.22 ± 16.88 | + 575.91 | 938.22 ± 10.29 | + 80.97 |
Fig. 4.
Content changes of the four astilbin isomers in SGR samples prepared by different methods. A Changes in the contents of the four astilbin isomers in SGR by boiling (a) and steaming (b) pretreatments compared with un-pretreated SGR samples. B Scatter plot of the contents (μg/g) of the four astilbin isomers throughout the drying process at different sampling times. C Heatmaps of the contents (μg/g) of the four astilbin isomers throughout drying process at different sampling times
In the boiled and steamed samples, the neoastilbin content increased by 271.96% (Fig. 4Aa) and 650.36% (Fig. 4Ab), respectively; the neoisoastilbin content increased by 1113.54% and 575.91%; and the isoastilbin content increased by 23.49% and 80.97%. In contrast, the content of astilbin remained almost unchanged in the boiled samples and decreased by 57.31% in the steamed samples.
Influence of drying methods on the contents of astilbin stereoisomers in SGR
The contents of astilbin stereoisomers in SGR prepared by different drying methods are shown in Additional file 1: Table S13. During the drying process, the content of neoastilbin gradually decreased in the steamed group of samples. The neoastilbin content of the boiled group of samples fluctuated during the drying process and increased slightly after drying, while the change in neoastilbin content in the un-pretreated group of samples first increased and then decreased (Fig. 4B) but did not appear obvious compared with the pretreated SGR due to the small amounts (Fig. 4C).
Astilbin is a dominant bioactive compound in SGR, and it has also been found in many other herbs, fruits, and plant-based foods [59]. The astilbin content was the highest in the un-pretreated SGR group of samples, followed by the boiled group samples, and the steamed group samples had the lowest content (Fig. 4B, Fig. 4C). During the drying process, the astilbin content in the un-pretreated group of samples increased from 3368.36 ± 35.51 μg/g to the maximum content, 5264.62 ± 97.45 μg/g DW (Ov.D treatment at 75 °C), increasing by approximately 50%. The un-pretreated group of samples under Su.D or Ov.D treatment at 45 °C and 85 °C also showed better astilbin-increasing effects. The astilbin content in the boiled and the steamed groups of samples showed fluctuating changes and increased or decreased slightly with the drying process.
The content of neoisoastilbin in the boiled group samples was higher than that in the steamed group samples, while the content in the un-pretreated group samples was the lowest. During the drying process, in the un-pretreated group samples, the neoisoastilbin content increased from 125.05 ± 1.55 μg/g DW to the maximum amount, 255.25 ± 5.54 μg/g DW (Ov.D at 55 °C), increasing by approximately 104% (Fig. 4B). Because of the relatively small amounts, the changes in neoisoastilbin content in the un-pretreated group samples were not obvious (Fig. 4C), while the boiled and steamed groups of samples showed a fluctuating increase.
The isoastilbin content in the un-pretreated and two pretreated groups of samples before drying was different (Figs. 4B, C). During the drying process, in the un-pretreated group samples, the isoastilbin content increased significantly, and the content of some samples even increased by 80%; for example, the content in samples subjected to Ov.D treatment at 55 °C increased from 518.45 ± 7.18 μg/g DW to 1070.25 ± 27.55 μg/g DW. Both the boiled and steamed groups of samples showed fluctuating changes in the isoastilbin content during the drying processes, but the final content in the dried samples barely changed compared with that before drying. Prior to drying of the pretreated samples, the initial order of the isoastilbin content was steamed > boiled > un-pretreated, and after the samples were dried, the order of the isoastilbin content was un-pretreated > steamed > boiled.
Influence of drying methods on the chemical profiles of the SGR slices
UHPLC-MS/MS method was also used to analyze the chemical profiles of SGR slices obtained by three drying methods (sun-drying, shade-drying and oven-drying at 65 °C and 105 °C). As shown in Table 3 and Fig. 5, a total of 49 compounds were identified from the dried samples. All the compounds were presented in the samples before drying except piscidic acid which was newly detected after drying. As mentioned above, the chemical compositions of the undried samples with different pretreatments were obviously different and only 14 compounds were common among the samples. However, in the sun-drying, shade-drying, oven-drying at 65 °C and 105 °C samples, there were 36, 40, 40 and 41 compounds common among the samples with different pretreatments, respectively. Some compounds could be detected in all three pretreatment samples after drying which were only detected in one or two pretreatment samples before drying. In particular, phenylpropanoid glycosides were common in the un-pretreated and boiled samples before drying, but all of them can also be detected in the steamed samples after drying. Therefore, the chemical compositions of the samples after drying tended to be consistent. Previous studies have shown that different pretreatment methods have a certain impact on the content of total phenols in thyme [60]. It could be interred that the pretreatment methods in the present study changed the content of chemical components in SGR, especially the isomers of astilbin [61–63]. Moreover, degradation of the cell wall and membrane function during drying process might lead to an increase in the extractability of the active compounds in the samples [64]. Thus, the compounds with low content in the samples before drying could be detected after drying.
Fig. 5.
TIC chromatogram of dried SGR samples (sun-drying, shade-drying, and oven-drying at 65 °C and 105 °C) in negative ionization mode obtained by UHPLC-Q-TOF-MS/MS
Comprehensive analysis of the influences of pretreatment and drying methods on the bioactive components contents in SGR
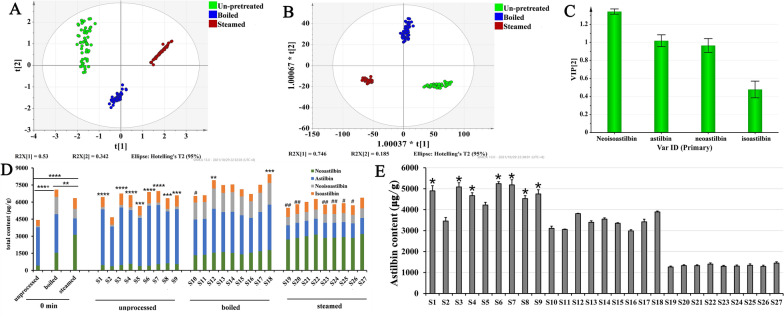
We conducted PCA of the content data of the four astilbin stereoisomers for the 189 samples from the experimental design (Fig. 6A). In the PCA score plot of SGR slice samples, the samples could be separated into three clusters corresponding to the pretreatment methods. The values of the established PCA model fit parameters R2X (cum) and Q2 (cum) were 0.998 and 0.987, respectively, which indicated that the model is robust. To further investigate the potential differential compounds, sample content data were then subjected to supervised discriminant analysis, OPLS-DA. The results showed that the OPLS-DA model exhibited good fitness (Q2 (cum) 0.978) and predictability (R2X (cum) 0.931, R2Y (cum) 0.98). As presented in Fig. 6B, the slice samples are clearly distinguished. The variable importance in projection (VIP) scores (Fig. 6C) generated from OPLS-DA indicated the contents of neoisoastilbin and astilbin (VIP ≥ 1) conducive to distinguishing groups of SGR slice samples.
Fig. 6.
Content changes of bioactive compounds in the prepared SGR samples. A PCA score plot for SGR at different sampling times. B OPLS-DA score plot for SGR at different sampling times. C VIP plot for SGR at different sampling times. D The total contents of the four isomers under different pretreatment and drying conditions (Compared with those at 0 min, significantly increased contents were described as *, and significantly decreased contents were described as #). E The astilbin content of the prepared SGR samples. (*means that the astilbin content of the prepared SGR sample meets the requirement of the Chinese Pharmacopoeia)
This result indicated that the pretreatment methods had significant influences on the contents of the four astilbin stereoisomers in the prepared SGR slice samples, which determined the content ratio of the four individual astilbin stereoisomers in these SGR slices. The results showed that the ratios of the contents of neoastilbin, astilbin, neoisoastilbin and isoastilbin to the total content of the four astilbin stereoisomers were 7.17%, 76.02%, 3.18%, and 13.63%, respectively, in the un-pretreated SGR sample; the ratios in the boiled sample were 21.84%, 47.74%, 21.39% and 9.03%, respectively, and those of the steamed sample were 49.24%, 22.66%, 13.32% and 14.78%. This result indicated that pretreatment changed the dominant astilbin stereoisomers in SGR. In the un-pretreated and boiled SGR samples, astilbin was dominant, while after steaming, neoastilbin became the dominant constituent in SGR.
There was a significant difference in the total content of the four astilbin stereoisomers in the un-pretreated and two pretreated groups of samples before drying (Fig. 6D). The order of the three groups of samples in terms of the total contents of astilbin stereoisomers was boiled > steamed > un-pretreated. The trend of the total contents of astilbin stereoisomers after drying was in the order boiled > un-pretreated > steamed. During the drying process, the total contents of the four astilbin stereoisomers in the un-pretreated sample group all significantly increased with decreasing moisture content, and those in the boiled and steamed sample groups slightly increased or decreased.
However, in the present study, we found that only seven treatment conditions in the un-pretreated group could meet the requirement of the Chinese Pharmacopoeia that the content of astilbin be no less than 0.45% DW (Fig. 6E).
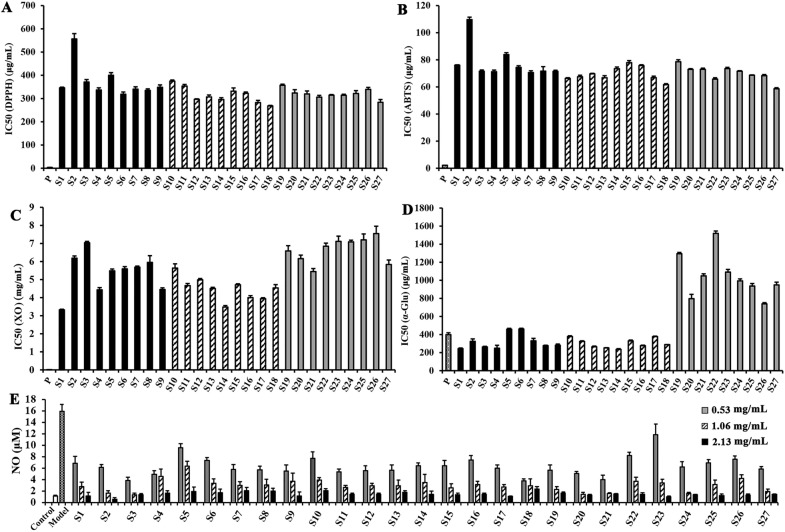
Antioxidant activities
The DPPH· radical scavenging ability of SGR samples prepared by the three pretreatment methods showed little change (Fig. 7A); however, the activity of SGR samples was inferior to that of the positive control drug VC (IC50 = 3.32 ± 0.03 μg/mL). Except for the Sh.D samples in the un-pretreated group, the other SGR samples possessed strong DPPH radical scavenging abilities. Changing the drying method had a slight impact on the antioxidant activity of SGR.
Fig. 7.
Bioactivities of the dried SGR samples under different pretreatments and drying conditions. A IC50 of the DPPH scavenging ability of SGR under different pretreatment and drying conditions. B IC50 of the ABTS+ scavenging ability of SGR under different pretreatment and drying conditions. C IC50 of XO inhibitory ability of SGR under different pretreatment and drying conditions. D IC50 of α-Glu inhibitory ability of SGR under different pretreatment and drying conditions. E Effects of different amounts of dried SGR on suppressing the production of NO (n = 6) when RAW264.7 cells were stimulated by LPS
The trends in ABTS+ radical scavenging ability of the SGR samples (Fig. 7B) were similar to those of the DPPH radical scavenging ability; however, the ABTS+ radical scavenging ability values were closer to that of the positive drug VC (IC50 = 2.15 ± 0.03 μg/mL).
Enzyme inhibition
SGR is used to treat gout in traditional Chinese medicines, and the XO inhibition is an important biochemical index to evaluate its therapeutic effect. The XO inhibitory effects of the SGR samples under different treatment conditions varied greatly (Fig. 7C). The XO inhibitory effects of the steamed group samples were generally poor. The un-pretreated SGR group samples had better effects than those of the steamed group. Overall, the XO inhibitory effects of the boiled SGR group samples were the best among the three groups of samples, while the Su.D samples of the un-pretreated group had the best effect. However, the XO inhibitory activities of the SGR samples were weaker than that of the positive control allopurinol (IC50 = 15.54 ± 0.35 μg/mL).
The results of in vitro bioactivity assays demonstrated that all SGR samples obtained from different pretreatment and drying methods showed good α-Glu inhibitory activities. Almost all of the IC50 values of the un-pretreated and boiled samples were less than that of the positive control drug acarbose (IC50 = 398.20 ± 21.63 μg/mL). However, the α-Glu inhibitory activities of the steamed group of samples were significantly worse than those of the un-pretreated and boiled samples. There were no significant differences in α-Glu inhibitory activities between the un-pretreated and boiled group samples, both of which had strong α-Glu inhibitory activities (Fig. 7D).
Anti-inflammatory activity
The concentration of NO was measured to explore and compare the anti-inflammatory activities of different dried SGR samples. After treatment with SGR at concentrations of 0.53, 1.06, and 2.13 mg/mL, the viability of RAW264.7 cells exceeded 90%. As shown in Fig. 7E, the anti-inflammatory activities of steamed group samples were slightly worse than those of the other two groups of samples. Overall, the prepared SGR samples showed an obvious treatment effect on suppressing the production of NO when RAW264.7 cells were stimulated by LPS. Therefore, the results of low-concentration SGR treatment were chosen for subsequent analysis.
Comprehensive evaluation of pretreatments and drying methods for preparing SGR slices
In this study, the biological activities of SGR under different pretreatment and drying conditions were investigated. According to the literature, astilbin and its three stereoisomers have strong antioxidant, XO and α-Glu inhibitory activities with little difference [65]. Therefore, the total content of astilbin stereoisomers showed a close correlation with the bioactivity of SGR. Additionally, it has been reported that other polar constituents in SGR have stronger XO [66] and α-Glu [67–71] inhibitory activities than astilbin stereoisomers and phenylpropanoid glycosides. This might be one of the reasons for the lower bioactivities of the steamed group samples than those of the other two groups of samples.
Entropy weight and technique for order preference by similarity to ideal solution (TOPSIS) analysis are widely used in various fields for decision-making or evaluation [72], such as in engineering, environmental applications, and other fields where precision is required [73–75]. In this study, according to the results of the entropy weight and TOPSIS analysis based on the antioxidant, XO and α-Glu inhibitory, and anti-inflammatory activities (Additional file 1: S14, Table S15), the samples of boiled SGR dried at 65 °C and un-pretreated SGR dried in the sun ranked the first and second, respectively. The un-pretreated SGR samples dried in the shade ranked 20th, indicating that the effect of shade drying is far inferior to that of sun drying. Furthermore, the Chinese Pharmacopoeia requests that the content of astilbin be at least 0.45% in SGR [2], and the un-pretreated sun-dried SGR has a higher astilbin content, better activity and more energy savings. The results further confirmed the rationality of this traditional processing method for SGR. Therefore, no pretreatment of SGR followed by drying in the sun represents the optimum treatment and drying method for preparing SGR slices from the fresh rhizomatic material of S. glabra.
Discussion
SGR is often used in traditional medicines and functional foods, and SGR slices have been included in ancient Chinese herbal works, some local processing standards of TCMs, and the current Chinese Pharmacopoeia. In this research, the scientific basis for the processing of fresh medicinal materials for SGR slices was revealed. After pretreatment with “boiling” and “steaming”, the contents of the compounds changed greatly, while the chemical profiles were slightly changed compared with those of un-pretreated SGR. During the subsequent drying process, the contents of the four astilbin isomers in the un-pretreated SGR increased significantly under the action of enzymes, while the boiled and steamed SGR showed a slight increase or decrease, respectively. The boiled SGR with different drying conditions had a richer chemical composition, higher total content of four astilbin stereoisomers and strong biological activities. But according to the requirement of the Chinese Pharmacopoeia that the astilbin content be no less than 0.45% DW, only seven un-pretreated dried SGR samples were qualified medicinal materials, and the un-pretreated SGR sample also had strong biological activities. Based on the TOPSIS analysis results, no pretreatment followed by a sun-drying process was the optimum treatment and drying method for preparing SGR slices.
After “boiling” and “steaming” pretreatments, the content of neoastilbin increased significantly, because this compound is the most stable of the four stereoisomers [62]. The “boiling” and “steaming” pretreated SGR samples showed a great change in the content ratio of the four individual astilbin stereoisomers during pretreatment, which might be due to the special metabolism variation of the plant response to the stress resistance and the influences of physical and chemical factors [76–78]. The “boiling” and “steaming” pretreated SGRs retained some vitality at the beginning of the treatment, and the high temperature increased the amount of flavonoids and phenolic compounds [78, 79]. However, as the treatment time was prolonged, particular enzymes in the samples were inactivated, and the biochemical reaction of the plant itself was blocked. It has been reported that the four astilbin stereoisomers in the ethanol extract of SGR can interconvert through a chalcone intermediate, and the ratio of trans (neoastilbin and astilbin) to cis (neoisoastilbin and isoastilbin) stereoisomers (trans/cis ratio of astilbin isomers) can reach an equilibrium under certain physical and chemical conditions [62, 63]. It is deduced that the four astilbin stereoisomers in the “nonliving” prepared SGR slice could also interconvert through chalcone intermediates and finally reach an equilibrium between the amount of trans- and cis- astilbin stereoisomers. In this study, in the steamed sample, the content ratio between neoastilbin and isoastilbin was approximately 3.3:1, and that between astilbin and neoisoastilbin was approximately 1.7:1. These content ratios were close to previously reported results in which that the stable content ratio between neoastilbin and isoastilbin was 3.4:1, and that between astilbin and neoisoastilbin was 1.6:1 under incubation of an ethanol extract of SGR in a water bath at 80 °C [63]. During the drying process, the trans/cis ratio of the astilbin isomer in un-pretreated group samples varied and decreased significantly, while those in the “nonliving” steamed group samples remained almost unchanged, and the ratio of the boiled group samples decreased slightly (Additional file 1: Fig. S1). It is implied that the content of cis isomers of astilbin might increase at a faster rate (%) during the pretreatment and drying processes of SGR. This possibility provides a certain basis for the optimization of pretreatment and drying methods that are conducive to the accumulation of active components of SGR.
It is speculated that the un-pretreated rhizome of S. glabra remained physiologically alive after harvest and that the enzymes involved in the biosynthetic pathways of the compounds were also active [79]. During the drying process, un-pretreated rhizomes may experience a series of stress resistance reactions. These environmental stresses including solar radiation, high temperature and dewatering might induce its physiological and biochemical responses, and many reactive oxygen species (ROS) might be produced. Excess ROS might cause oxidative stress, which can result in injury to plants at both the molecular and cellular levels [80]. To avoid oxidative injury to the plant itself, ROS might induce many biochemical reactions through the action of enzymes occurring in the plant, resulting in an increased amount of flavonoids and phenolic compounds that might be used as antioxidants [76, 77]. Thus, excess ROS amounts might be particularly scavenged by antioxidant metabolites of plants [78].
Hyperuricemia and diabetes are both prevalent lifestyle-related diseases, and hyperuricemia is also a predictive factor for the development of type 2 diabetes mellitus (T2DM) [81, 82]. Hyperuricemia promotes the occurrence and development of diseases by regulating molecular signals, such as the inflammatory response, oxidative stress, and insulin resistance/diabetes [83]. Furthermore, SGR has been demonstrated to possess multiple biological and pharmacological activities, including anti-inflammatory [5, 6], antioxidant [7], antihyperuricemia [6] and antidiabetic [12] effects. To better evaluate the different pretreatment and drying methods, the antioxidant, anti-inflammatory, and XO and α-Glu inhibitory activities of SGR with different pretreatments and drying conditions were studied. This research provided data on the activities of SGR under different pretreatment and drying conditions. However, more in vivo activity differences under different processing conditions need to be further investigated.
In this study, pretreatment methods had significant effects on the content of bioactive components of SGR, which further affected the chemical characteristics of SGR. The content of astilbin in the un-pretreated SGR was the highest, the total content of the four isomers in boiled SGR was the highest, and the content of neoastilbin in steamed SGR was the highest. The antioxidant and anti-inflammatory activities of SGR in the un-pretreated and pretreated SGR were not significantly different, while the XO and α-Glu inhibitory activities of steamed SGR were weaker than those of boiled and un-pretreated SGR. It is noteworthy that there were no qualified medicinal materials before the drying of SGR, which showed that the processing of fresh SGR was an important means to achieve qualified SGR slices. However, drying in the shade, as a common drying method in the production area, is the most unfavorable drying method for the accumulation of astilbin. The Chinese Pharmacopoeia notes that SGR should be “thinly sliced while fresh, dried” and does not specify specific drying conditions. In the actual drying process, drying in the shade is a very common drying method. Therefore, we suggest that the Chinese Pharmacopoeia should clarify that SGR should not be dried in the shade. No pretreatment followed by a sun-drying process was the optimum treatment and drying method for preparing SGR slices. This conclusion has guiding significance for processing and drying in the production area.
Conclusion
In this paper, the influences of different pretreatments and drying methods on the chemical compositions and bioactivities of Smilacis Glabrae Rhizoma were systematically studied for the first time. The chemical profiles and contents of the four individual astilbin stereoisomers were markedly changed by different pretreatments. After the “boiling” and “steaming” pretreatments, the contents of neoastilbin, neoisoastilbin, and isoastilbin all increased, while the astilbin content either remained the same as that in the un-pretreated sample or decreased. During the drying process, the contents of the four astilbin stereoisomers in the un-pretreated group of samples were all significantly increased; in particular, the content of astilbin, the quality marker of SGR, increased by approximately 50%. However, the content in the boiled and steamed groups of samples showed a slight increase or decrease.
The prepared SGR samples under different drying conditions exhibited strong antioxidant, XO and α-Glu inhibitory activities. It is noteworthy that the α-Glu inhibition activities of the un-pretreated and boiled groups of samples were significantly better than that of the positive control acarbose.
These results suggested that no pretreatment followed by a sun-drying process was the optimum treatment and drying method for preparing SGR slices. This study provides scientific information for the quality control of SGR and its rational applications in herbal medicines and functional foods.
Supplementary Information
Additional file 1: Table S1. Sampling schedules during drying process of pretreated SGR. Table S2. The results of precision and reputability tests. Table S3. The results of linear ranges, LODs, LOQs and recovery tests. Table S4. Content of moisture and analytes in SGR during sun-drying. Table S5. Content of moisture and analytes in SGR during shade-drying. Table S6. Content of moisture and analytes in SGR during oven-drying at 45 °C. Table S7. Content of moisture and analytes in SGR during oven-drying at 55 °C. Table S8. Content of moisture and analytes in SGR during oven-drying at 65 °C. Table S9. Content of moisture and analytes in SGR during oven-drying at 75 °C. Table S10. Content of moisture and analytes in SGR during oven-drying at 85 °C. Table S11. Content of moisture and analytes in SGR during oven-drying at 95 °C. Table S12. Content of moisture and analytes in SGR during oven-drying at 105 °C. Table S13. Bioactive ingredient content and DPPH·, ABTS·+, XO and α-Glu(n = 3) inhibitory effects of the 27 dried SGR. Table S14. The operation steps and equations of entropy weight and TOPSIS analysis. Table S15 Comprehensive evaluation result of entropy weight and TOPSIS model. Figure S1. The content rations of trans to cis isomers of SGR throughout drying processing at different sampling times.
Additional file 2: S1. Optimization of Extraction Processing for Bioactive Ingredients. Table S1. The Box-Behnken design experiment scheme. Table S2. ANOVA of quadratic model terms of three factor variables on responses. Table S3. Predicted and experimental values of the responses obtained under the optimal extraction conditions. Figure S1. Result of the single factor experiments. Figure S2. Response surface plots corresponding to the desirability function.
Acknowledgements
This work was supported by Experimental Teaching Center of Traditional Chinese Medicine and Natural Medicine (China Pharmaceutical University).
Abbreviations
- SGR
Smilacis Glabrae Rhizoma
- Su.D
Sun-drying
- Sh.D
Shade-drying
- Ov.D
Oven-drying
- UHPLC-Q-TOF-MS/MS
Ultra high-performance liquid chromatography coupled with electrospray ionization quadrupole time-of-flight tandem mass spectrometry
- UHPLC
Ultra high-performance liquid chromatography
- DAD
Diode array detector
- RSM
Response surface methodology
- PCA
Principal component analysis
- OPLS-DA
Orthogonal projections to latent structures discriminant analysis
- DMSO
Dimethyl sulfoxide
- DPPH
1,1-D-diphenyl-2-picrylhydrazyl
- LPS
Lipopolysaccharide
- ABTS
2,2′-Azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) diammonium salt
- XO
Xanthine oxidase
- α-Glu
α-Glucosidase
- NO
Nitric oxide
- TOPSIS
Technique for order preference by similarity to ideal solution
Author contributions
MJQ and JJQ designed the study. JJQ conducted the experiments and statistical analyses, drafted the manuscript. GYL, WLW and TMZ supervised the study. GW helped carry out the drying experiments. HL and GYX contributed to the UHPLC-Q-TOF-MS/MS analysis. MJQ revised the manuscript. All authors read and approved the final manuscript.
Funding
This work is supported by the Subsidy Funds for Medical Service and Security Capacity (Inheritance and Development of Traditional Chinese Medicine) “National Survey on Chinese Materia Medica Resources” [Department of Social Security, Ministry of Finance of the People's Republic of China (2019) 39].
Availability of data and materials
Not applicable.
Declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Juanjuan Qiao, Email: kfq6310@163.com.
Gengyu Lu, Email: njlugengyu@126.com.
Gang Wu, Email: woosmail@163.com.
Hui Liu, Email: liu_huiabc@163.com.
Wanli Wang, Email: wanliw23@163.com.
Tianmao Zhang, Email: 1319263688@qq.com.
Guoyong Xie, Email: guoyongxie321@163.com.
Minjian Qin, Email: minjianqin@163.com.
References
- 1.Lee J, Li C, Surayot U, Yelithao K, Lee S, Park W, Tabarsa M, You S. Molecular structures, chemical properties and biological activities of polysaccharide from Smilax glabra rhizome. Int J Biol Macromol. 2018;120(Pt B):1726–1733. doi: 10.1016/j.ijbiomac.2018.09.138. [DOI] [PubMed] [Google Scholar]
- 2.Chinese Pharmacopoeia Commission. Smilacis Glabrae Rhizoma. In: Pharmacopoeia of the People's Republic of China. 2020 edition Vol I. Beijing: China Medical Science Press; 2020. p. 19.
- 3.Li F, Wu JH, Wang QH, Shu YL, Wan CW, Chan CO, Kam-Wah Mok D, Chan SW. Gui-ling-gao, a traditional Chinese functional food, prevents oxidative stress-induced apoptosis in H9c2 cardiomyocytes. Food Funct. 2013;4(5):745–753. doi: 10.1039/c3fo30182f. [DOI] [PubMed] [Google Scholar]
- 4.Zhang H, Wu MY, Guo DJ, Wan CW, Lau CC, Chan CO, Mok DK, Chan SW. Gui-ling-gao (turtle jelly), a traditional Chinese functional food, exerts anti-inflammatory effects by inhibiting iNOS and pro-inflammatory cytokine expressions in splenocytes isolated from BALB/c mice. J Funct Foods. 2013;5(2):625–632. [Google Scholar]
- 5.Shi Y, Tian C, Yu X, Fang Y, Zhao X, Zhang X, Xia D. Protective effects of Smilax glabra Roxb. against lead-induced renal oxidative stress, inflammation and apoptosis in weaning rats and HEK-293 cells. Front Pharmacol. 2020;11:556248. doi: 10.3389/fphar.2020.556248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Wang S, Fang Y, Yu X, Guo L, Zhang X, Xia D. The flavonoid-rich fraction from rhizomes of Smilax glabra Roxb. ameliorates renal oxidative stress and inflammation in uric acid nephropathy rats through promoting uric acid excretion. Biomed Pharmacother. 2019;111:162–168. doi: 10.1016/j.biopha.2018.12.050. [DOI] [PubMed] [Google Scholar]
- 7.Park G, Kim TM, Kim JH, Oh MS. Antioxidant effects of the sarsaparilla via scavenging of reactive oxygen species and induction of antioxidant enzymes in human dermal fibroblasts. Environ Toxicol Pharmacol. 2014;38(1):305–315. doi: 10.1016/j.etap.2014.06.009. [DOI] [PubMed] [Google Scholar]
- 8.Zhang C, Xu Q, Tan X, Meng L, Wei G, Liu Y, Zhang C. Astilbin decreases proliferation and improves differentiation in HaCaT keratinocytes. Biomed Pharmacother. 2017;93:713–720. doi: 10.1016/j.biopha.2017.05.127. [DOI] [PubMed] [Google Scholar]
- 9.Kwon OY, Ryu S, Choi JK, Lee SH. Smilax glabra Roxb. inhibits collagen induced adhesion and migration of PC3 and LNCaP prostate cancer cells through the inhibition of Beta 1 integrin expression. Molecules. 2020;25(13):3006. doi: 10.3390/molecules25133006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Sa F, Gao JL, Fung KP, Zheng Y, Lee SM, Wang YT. Anti-proliferative and pro-apoptotic effect of Smilax glabra Roxb. extract on hepatoma cell lines. Chem Biol Interact. 2008;171(1):1–14. doi: 10.1016/j.cbi.2007.08.012. [DOI] [PubMed] [Google Scholar]
- 11.Nguyen PTM, Ngo QV, Nguyen MTH, Quach LT, Pyne SG. Hypoglycemic activity of the ethyl acetate extract from Smilax glabra Roxb. in mice: biochemical and histopathological studies. Iran J Basic Med Sci. 2020;23(12):1558–1564. doi: 10.22038/ijbms.2020.46658.10763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Ansari S, Bari A, Ullah R, Mathanmohun M, Veeraraghavan VP, Sun ZW. Gold nanoparticles synthesized with Smilax glabra rhizome modulates the anti-obesity parameters in high-fat diet and streptozotocin induced obese diabetes rat model. J Photochem Photobiol B. 2019;201:111643. doi: 10.1016/j.jphotobiol.2019.111643. [DOI] [PubMed] [Google Scholar]
- 13.Xie J, Li L, Shi Y, Chen R, Liu G, Wang M, Zheng M, Zhang N. Simultaneous ultra-performance liquid chromatography-tandem mass spectrometry determination of six components in rat plasma after oral administration of Smilacis glabrae Roxb. extract. Biomed Chromatogr. 2019;33(12):e4680. doi: 10.1002/bmc.4680. [DOI] [PubMed] [Google Scholar]
- 14.Chen SD, Lu CJ, Zhao RZ. Qualitative and quantitative analysis of Rhizoma Smilacis glabrae by ultra high performance liquid chromatography coupled with LTQ OrbitrapXL hybrid mass spectrometry. Molecules. 2014;19(7):10427–10439. doi: 10.3390/molecules190710427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Chen T, Li JX, Xu Q. Phenylpropanoid glycosides from Smilax glabra. Phytochemistry. 2000;53(8):1051–1055. doi: 10.1016/s0031-9422(99)00522-1. [DOI] [PubMed] [Google Scholar]
- 16.Yang G, Zhao X, Wen J, Zhou T, Fan G. Simultaneous fingerprint, quantitative analysis and anti-oxidative based screening of components in Rhizoma Smilacis Glabrae using liquid chromatography coupled with Charged Aerosol and Coulometric array Detection. J Chromatogr B Analyt Technol Biomed Life Sci. 2017;1049–1050:41–50. doi: 10.1016/j.jchromb.2017.02.012. [DOI] [PubMed] [Google Scholar]
- 17.Ren Z, Yu X, Yagoub AEA, Fakayode OA, Ma H, Sun Y, Zhou C. Combinative effect of cutting orientation and drying techniques (hot air, vacuum, freeze and catalytic infrared drying) on the physicochemical properties of ginger (Zingiber officinale Roscoe) Lwt. 2021;144:111238. [Google Scholar]
- 18.Panagopoulou EA, Chiou A, Nikolidaki EK, Christea M, Karathanos VT. Corinthian raisins (Vitis vinifera L., var. Apyrena) antioxidant and sugar content as affected by the drying process: a 3-year study. J Sci Food Agric. 2019;99(2):915–922. doi: 10.1002/jsfa.9263. [DOI] [PubMed] [Google Scholar]
- 19.Şahin S, Elhussein E, Bilgin M, Lorenzo JM, Barba FJ, Roohinejad S. Effect of drying method on oleuropein, total phenolic content, flavonoid content, and antioxidant activity of olive (Olea europaea ) leaf. J Food Process Pres. 2018;42(5):e13604. [Google Scholar]
- 20.Jiang G, He SQ, Liu SZ. Effect of different drying methods on the quality of Smilax glabra Roxb. J Guangdong Pharma University. 2013;29(3):258–261. [Google Scholar]
- 21.Yang JJ, Lu TL. Analysis on the historical evolution and current situation of the processing of Smilax glabra rhizoma. J Chinese Med Mater. 2016;39(4):767–769. [Google Scholar]
- 22.Cai C, Wei C. In situ observation of crystallinity disruption patterns during starch gelatinization. Carbohydr Polym. 2013;92(1):469–478. doi: 10.1016/j.carbpol.2012.09.073. [DOI] [PubMed] [Google Scholar]
- 23.Xu B, Wang LK, Miao WJ, Wu QF, Liu YX, Sun Y, Gao C. Thermal versus microwave inactivation kinetics of lipase and lipoxygenase from wheat germ. J Food Process Eng. 2016;39(3):247–255. [Google Scholar]
- 24.Maurya VK, Gothandam KM, Ranjan V, Shakya A, Pareek S. Effect of drying methods (microwave vacuum, freeze, hot air and sun drying) on physical, chemical and nutritional attributes of five pepper (Capsicum annuum var. annuum) cultivars. J Sci Food Agric. 2018;98(9):3492–3500. doi: 10.1002/jsfa.8868. [DOI] [PubMed] [Google Scholar]
- 25.Yang JJ, Huang ZY, Li L, Ji D, Lu TL. Study on the process optimization of the integration of the habitat processing and the processing of Smilax glabra and Pharmacological validation. Lishizhen Med Mater Medica Res. 2018;29(9):2165–2168. [Google Scholar]
- 26.Yang JJ, Zhang YL, Wang ZT, Lu TL. Rational analysis on peeling in babitat processing of Smilacis Glabrae Rhizoma. Chinnese J Exp Tradit Med Formulae. 2017;23(3):1–5. [Google Scholar]
- 27.Yuan MH. Study on the influence of quality in Smilax glabra Roxb. By sulfer-fumigated process. Southwest Minzu University, 2018.
- 28.Yuan J, Hao LJ, Wu G, Wang S, Duan JA, Xie GY, Qin MJ. Effects of drying methods on the phytochemicals contents and antioxidant properties of chrysanthemum flower heads harvested at two developmental stages. J Funct Foods. 2015;19(Part A):786–795. [Google Scholar]
- 29.Zhang Y, Liu W, Wei Z, Yin B, Man C, Jiang Y. Enhancement of functional characteristics of blueberry juice fermented by Lactobacillus plantarum. Lwt. 2021;139:110590. [Google Scholar]
- 30.El-Sabagh OA, El-Toumy SA, Mounir R, Farag MA, Mahrous EA. Metabolite profiles of Pulicaria crispa and P. incisa in relation to their in-vitro/ in-vivo antioxidant activity and hepatoprotective effect: a comparative mass spectrometry-based metabolomics. J Pharm Biomed Anal. 2021;194:113804. doi: 10.1016/j.jpba.2020.113804. [DOI] [PubMed] [Google Scholar]
- 31.Wan Y, Zou B, Zeng H, Zhang L, Chen M, Fu G. Inhibitory effect of verbascoside on xanthine oxidase activity. Int J Biol Macromol. 2016;93(Pt A):609–614. doi: 10.1016/j.ijbiomac.2016.09.022. [DOI] [PubMed] [Google Scholar]
- 32.Xu Y, Niu X, Liu N, Gao Y, Wang L, Xu G, Li X, Yang Y. Characterization, antioxidant and hypoglycemic activities of degraded polysaccharides from blackcurrant (Ribes nigrum L.) fruits. Food Chem. 2018;243:26–35. doi: 10.1016/j.foodchem.2017.09.107. [DOI] [PubMed] [Google Scholar]
- 33.Xie GY, Jin SY, Li HT, Ai MK, Han F, Dai YQ, Tao W, Zhu Y, Zhao YC, Qin MJ. Chemical constituents and antioxidative, anti-inflammatory and anti-proliferative activities of wild and cultivated Corydalis Saxicola. Ind Crop Prod. 2021;169:113647. [Google Scholar]
- 34.Zhao L, Zhang Y, Wang L, Liu X, Zhang J, He Z. Stereoselective metabolomic and lipidomic responses of lettuce (Lactuca sativa L.) exposing to chiral triazole fungicide tebuconazole. Food Chem. 2022;371:131209. doi: 10.1016/j.foodchem.2021.131209. [DOI] [PubMed] [Google Scholar]
- 35.Sun Y, Qin Y, Li H, Peng H, Chen H, Xie H, Deng Z. Rapid characterization of chemical constituents in Radix Tetrastigma, a functional herbal mixture, before and after metabolism and their antioxidant/antiproliferative activities. J Funct Foods. 2015;18(Part A):300–318. [Google Scholar]
- 36.He L, Zhang Z, Liu Y, Chen D, Yuan M, Dong G, Luo P, Yan Z. Rapid discrimination of raw and sulfur-fumigated Smilax glabra based on chemical profiles by UHPLC-QTOF-MS/MS coupled with multivariate statistical analysis. Food Res Int. 2018;108:226–236. doi: 10.1016/j.foodres.2018.03.047. [DOI] [PubMed] [Google Scholar]
- 37.Wang H, Ye YH, Wang HH, Liu J, Liu YJ, Jiang BW. HPLC-QTOF-MS/MS profiling, antioxidant, and α-glucosidase inhibitory activities of Pyracantha fortuneana fruit extracts. J Food Biochem. 2019;43(5):e12821. doi: 10.1111/jfbc.12821. [DOI] [PubMed] [Google Scholar]
- 38.de Freitas MA, Andrade JC, Alves AIS, Dos Santos FAG, Leite-Andrade MC, Sales DL, Nunes M, Ribeiro PRV, Melo Coutinho HD, Morais-Braga MFB, Neves RP. Use of the natural products from the leaves of the fruitfull tree Persea americana against Candida sp. biofilms using acrylic resin discs. Sci Total Environ. 2020;703:134779. doi: 10.1016/j.scitotenv.2019.134779. [DOI] [PubMed] [Google Scholar]
- 39.Elshamy AI, Farrag ARH, Ayoub IM, Mahdy KA, Taher RF, Gendy AEGE, Mohamed TA, Al-Rejaie SS, Ei-Amier YA, Abd-EIGawad AM, Farag MA. UPLC-qTOF-MS phytochemical profile and antiulcer potential of Cyperus conglomeratus Rottb. Alcoholic Extract Mol. 2020;25(18):4234. doi: 10.3390/molecules25184234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Jia J, Liu M, Wen Q, He MZ, Ouyang H, Chen LY, Li JM, Feng YL, Zhong GY, Yang SL. Screening of anti-complement active ingredients fromEucommia ulmoides Oliv. branches and their metabolism in vivo based on UHPLC-Q-TOF/MS/MS. J Chromatogr B. 2019;1124:26–36. doi: 10.1016/j.jchromb.2019.05.029. [DOI] [PubMed] [Google Scholar]
- 41.Li X, Zhang YF, Yang L, Feng Y, Deng YH, Liu YM, Zeng X. Chemical profiling of constituents of Smilacis glabrae using ultra-high pressure liquid chromatography coupled with LTQ Orbitrap mass spectrometry. Nat Prod Commun. 2012;7(2):181–184. [PubMed] [Google Scholar]
- 42.Cabañas-García E, Areche C, Jáuregui-Rincón J, Cruz-Sosa F, Pérez-Molphe BE. Phytochemical Profiling of Coryphantha macromeris (Cactaceae) growing in greenhouse conditions using ultra-high-performance liquid chromatography-tandem mass spectrometry. Molecules. 2019;24(4):705. doi: 10.3390/molecules24040705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Frank O, Blumberg S, Krümpel G, Hofmann T. Structure determination of 3-O-caffeoyl-epi-gamma-quinide, an orphan bitter lactone in roasted coffee. J Agric Food Chem. 2008;56(20):9581–9585. doi: 10.1021/jf802210a. [DOI] [PubMed] [Google Scholar]
- 44.Abu-Reidah IM, Ali-Shtayeh MS, Jamous RM, Arráez-Román D, Segura-Carretero A. Comprehensive metabolite profiling of Arum palaestinum (Araceae) leaves by using liquid chromatography–tandem mass spectrometry. Food Res Int. 2015;70:74–86. [Google Scholar]
- 45.Zhang QF, Guo YX, Shangguan X, Zheng G, Wang WJ. Identification and quantification of polyphenols in Rhizoma Smilacis Chinae by HPLC/DAD/ESI-MS/MS. J Liq Chromatogr Relat Technol. 2013;36(16):2251–2260. [Google Scholar]
- 46.Amessis-Ouchemoukh N, Abu-Reidah IM, Quirantes-Piné R, Madani K, Segura-Carretero A. Phytochemical profiling, in vitro evaluation of total phenolic contents and antioxidant properties of Marrubium vulgare (horehound) leaves of plants growing in Algeria. Ind Crop Prod. 2014;61:120–129. [Google Scholar]
- 47.Li Z, Li D, Owen NL, Len Z, Cao Z, Yi Y. Structure determination of a new chromone glycoside by 2D INADEQUATE NMR and molecular modeling. Magn Reson Chem. 1996;34(7):512–517. [Google Scholar]
- 48.Li Y, Li M, Liu J, Zheng W, Zhang Y, Xu T, Gao B, Yu L. Chemical composition profiling and biological activities of phenolic compounds in eleven red sorghums. J Agric Food Chem. 2021;69(32):9407–9418. doi: 10.1021/acs.jafc.1c03115. [DOI] [PubMed] [Google Scholar]
- 49.Sakushima A, Ohno K, Coskun M, Seki K, Ohkura K. Separation and identification of Taxifolin 3-O-glucoside isomers from Chamaecyparis obtusa (Cupressaceae) Nat Prod Lett. 2002;16(6):383–387. doi: 10.1080/10575630290033141. [DOI] [PubMed] [Google Scholar]
- 50.Sun L, Tao S, Zhang S. Characterization and quantification of polyphenols and triterpenoids in thinned young fruits of ten pear varieties by UPLC-Q TRAP-MS/MS. Molecules. 2019;24(1):159. doi: 10.3390/molecules24010159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Fang HL, Liu ML, Li SY, Song WQ, Ouyang H, Xiao ZP, Zhu HL. Identification, potency evaluation, and mechanism clarification of α-glucosidase inhibitors from tender leaves of Lithocarpus polystachyus Rehd. Food Chem. 2021;371:131128. doi: 10.1016/j.foodchem.2021.131128. [DOI] [PubMed] [Google Scholar]
- 52.Gu WY, Li N, Leung EL, Zhou H, Luo GA, Liu L, Wu JL. Metabolites software-assisted flavonoid hunting in plants using ultra-high performance liquid chromatography-quadrupole-time of flight mass spectrometry. Molecules. 2015;20(3):3955–3971. doi: 10.3390/molecules20033955. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Qin Y, Gao B, Shi H, Cao J, Yin C, Lu W, Yu L, Cheng Z. Characterization of flavonol mono-, di-, tri- and tetra-O-glycosides by ultra-performance liquid chromatography-electrospray ionization-quadrupole time-of-flight mass spectrometry and its application for identification of flavonol glycosides in Viola tianschanica. J Pharm Biomed Anal. 2017;142:113–124. doi: 10.1016/j.jpba.2017.05.007. [DOI] [PubMed] [Google Scholar]
- 54.Zengin G, Diuzheva A, Jekő J, Cziáky Z, Bulut G, Dogan A, Haznedaroglu MZ, Rengasamy KRR, Lobine D, Bahadori MB, Mahomoodally MF. HPLC–MS/MS-based metabolic profiling and pharmacological properties of extracts and infusion obtained from Amelanchier parviflora var. dentata. Ind Crop Prod. 2018;124:699–706. [Google Scholar]
- 55.Kuo YH, Hsu YW, Liaw CC, Lee JK, Huang HC, Kuo LM. Cytotoxic phenylpropanoid glycosides from the stems of Smilax china. J Nat Prod. 2005;68(10):1475–1478. doi: 10.1021/np050109q. [DOI] [PubMed] [Google Scholar]
- 56.Sun TT, Zhang DW, Han Y, Dong FY, Wang W. Smilasides M and N, two new phenylpropanoid glycosides from Smilax riparia. J Asian Nat Prod Res. 2012;14(2):165–170. doi: 10.1080/10286020.2011.641536. [DOI] [PubMed] [Google Scholar]
- 57.Wang X, Zhong XJ, Zhou N, Cai N, Xu JH, Wang QB, Li JJ, Liu Q, Lin PC, Shang XY. Rapid characterizaiton of chemical constituents of the tubers of gymnadenia conopsea by UPLC-Orbitrap-MS/MS Analysis. Molecules. 2020;25(4):898. doi: 10.3390/molecules25040898. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Du BB, Shen MJ, Zeng LX, Zhu CY, Luo D, Zeng LX. Trace analysis of multiple synthetic phenolic antioxidants in foods by liquid chromatography–tandem mass spectrometry with complementary ue of electrospray ionization and atmospheric pressure chemical ionization. Food Chem. 2021;375:131663. doi: 10.1016/j.foodchem.2021.131663. [DOI] [PubMed] [Google Scholar]
- 59.Zhang QF, Fu YJ, Huang ZW, Shangguang XC, Guo YX. Aqueous stability of astilbin: effects of pH, temperature, and solvent. J Agric Food Chem. 2013;61(49):12085–12091. doi: 10.1021/jf404420s. [DOI] [PubMed] [Google Scholar]
- 60.Lahnine L, Idlimam A, Mahrouz M, Mghazli S, Hidar N, Hanine H, Koutit A. Thermophysical characterization by solar convective drying of thyme conserved by an innovative thermal-biochemical process. Renew Energy. 2016;94:72–80. [Google Scholar]
- 61.Huang L, Zhu Y, Peng T, Qiu J, Rao Q, Song J, Xiao S, Li Y, Tang L. Drying methods and structure-activity relationships of hydroxycinnamic acid derivatives in Idesia polycarpa Maxim. Leaves Food Funct. 2021;12(4):1651–1661. doi: 10.1039/d0fo01813a. [DOI] [PubMed] [Google Scholar]
- 62.Gaffield W, Waiss AC, Jr, Tominaga T. Structural relations and interconversions of isomeric astilbins. J Org Chem. 1975;40(8):1057–1061. [Google Scholar]
- 63.Zheng D, Zhang L, Zhang QF. Isomerization of astilbin and its application for preparation of the four stereoisomers from Rhizoma Smilacis Glabrae. J Pharm Biomed Anal. 2018;155:202–209. doi: 10.1016/j.jpba.2018.04.005. [DOI] [PubMed] [Google Scholar]
- 64.Martínez-Las Heras R, Heredia A, Castelló ML, Andrés A. Influence of drying method and extraction variables on the antioxidant properties of persimmon leaves. Food Biosci. 2014;6:1–8. [Google Scholar]
- 65.Zhao X, Chen R, Shi Y, Zhang X, Tian C, Xia D. Antioxidant and anti-inflammatory activities of six flavonoids from Smilax glabra Roxb. Molecules. 2020;25(22):5295. doi: 10.3390/molecules25225295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Xu M, Xu D. Anti-uric acid constituents from Rhizoma Smilacis Glabrae. Nat Prod Res Devel. 2020;32(11):1860–1865. [Google Scholar]
- 67.Nguyen PTM, Ngo QV, Nguyen MTH, Maccarone AT, Pyne SG. α-Glucosidase inhibitory activity of the extracts and major phytochemical components of Smilax glabra Roxb. Nat Prod J. 2020;10(1):26–32. [Google Scholar]
- 68.Pérez-Nájera VC, Gutiérrez-Uribe JA, Antunes-Ricardo M, Hidalgo-Figueroa S, Del-Toro-Sánchez CL, Salazar-Olivo LA, Lugo-Cervantes E. Smilax aristolochiifolia root extract and its compounds chlorogenic acid and astilbin inhibit the activity of α-amylase and α-glucosidase enzymes. Evid Based Complement Alternat Med. 2018;2018:6247306. doi: 10.1155/2018/6247306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Kim DS, Lim SB. Semi-continuous subcritical water extraction of flavonoids from Citrus unshiu peel: their antioxidant and enzyme inhibitory activities. Antioxidants (Basel) 2020;9(5):360. doi: 10.3390/antiox9050360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Masuoka N. Stilbene compounds are specific inhibitors of the superoxide anion generation catalyzed by xanthine oxidase. Food Chem X. 2021;12:100146. doi: 10.1016/j.fochx.2021.100146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Kumar D, Gupta N, Ghosh R, Gaonkar RH, Pal BC. α-Glucosidase and α-amylase inhibitory constituent of Carex baccans: Bio-assay guided isolation and quantification by validated RP-HPLC–DAD. J Funct Foods. 2013;5(1):211–218. [Google Scholar]
- 72.Salehi V, Zarei H, Shirali GA, Hajizadeh K. An entropy-based TOPSIS approach for analyzing and assessing crisis management systems in petrochemical industries. J Loss Prevent Proc. 2020;67:104241. [Google Scholar]
- 73.Chodha, V, Dubey, R, Kumar, R, Singh, S, Kaur, S. Selection of industrial arc welding robot with TOPSIS and Entropy MCDM techniques. Mater. Today-Proc. 2021.
- 74.Kumar, R, Gandotra, N, Suman. A novel pythagorean fuzzy entropy measure using MCDM application in preference of the advertising company with TOPSIS approach. Mater. Today-Proc. 2021.
- 75.Li H, Huang J, Hu Y, Wang S, Liu J, Yang L. A new TMY generation method based on the entropy-based TOPSIS theory for different climatic zones in China. Energy. 2021;231:120723. [Google Scholar]
- 76.Hartmann T. From waste products to ecochemicals: fifty years research of plant secondary metabolism. Phytochemistry. 2007;68(22–24):2831–2846. doi: 10.1016/j.phytochem.2007.09.017. [DOI] [PubMed] [Google Scholar]
- 77.Qin MJ, Ji WL, Liu J, Zhao J, Yu GD. Scavenging effects on radicals of isoflavones from rhizome of Belamcanda chinensis. Chinese Tradit Herbal Drugs. 2003;34(7):640–641. [Google Scholar]
- 78.Golldack D, Li C, Mohan H, Probst N. Tolerance to drought and salt stress in plants: unraveling the signaling networks. Front Plant Sci. 2014;5:151. doi: 10.3389/fpls.2014.00151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Zhu Y, Pu BQ, Xie GY, Tian M, Xu FY, Qin MJ. Dynamic changes of flavonoids contents in the different parts of rhizome of Belamcanda chinensis during the thermal drying process. Molecules. 2014;19(7):10440–10454. doi: 10.3390/molecules190710440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Dat J, Vandenabeele S, Vranová E, Van Montagu M, Inzé D, Van Breusegem F. Dual action of the active oxygen species during plant stress responses. Cell Mol Life Sci. 2000;57(5):779–795. doi: 10.1007/s000180050041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Borghi C, Agabiti-Rosei E, Johnson RJ, Kielstein JT, Lurbe E, Mancia G, Redon J, Stack AG, Tsioufis KP. Hyperuricaemia and gout in cardiovascular, metabolic and kidney disease. Eur J Intern Med. 2020;80:1–11. doi: 10.1016/j.ejim.2020.07.006. [DOI] [PubMed] [Google Scholar]
- 82.Shi H, Xiang S, Huang X, Wang L, Hua F, Jiang X. Development and validation of a nomogram for predicting the risk of obstructive sleep apnea in patients with type 2 diabetes. Ann Transl Med. 2020;8(24):1675. doi: 10.21037/atm-20-6890. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Yu W, Cheng JD. Uric Acid and cardiovascular disease: an update from molecular mechanism to clinical perspective. Front Pharmacol. 2020;11:582680. doi: 10.3389/fphar.2020.582680. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Additional file 1: Table S1. Sampling schedules during drying process of pretreated SGR. Table S2. The results of precision and reputability tests. Table S3. The results of linear ranges, LODs, LOQs and recovery tests. Table S4. Content of moisture and analytes in SGR during sun-drying. Table S5. Content of moisture and analytes in SGR during shade-drying. Table S6. Content of moisture and analytes in SGR during oven-drying at 45 °C. Table S7. Content of moisture and analytes in SGR during oven-drying at 55 °C. Table S8. Content of moisture and analytes in SGR during oven-drying at 65 °C. Table S9. Content of moisture and analytes in SGR during oven-drying at 75 °C. Table S10. Content of moisture and analytes in SGR during oven-drying at 85 °C. Table S11. Content of moisture and analytes in SGR during oven-drying at 95 °C. Table S12. Content of moisture and analytes in SGR during oven-drying at 105 °C. Table S13. Bioactive ingredient content and DPPH·, ABTS·+, XO and α-Glu(n = 3) inhibitory effects of the 27 dried SGR. Table S14. The operation steps and equations of entropy weight and TOPSIS analysis. Table S15 Comprehensive evaluation result of entropy weight and TOPSIS model. Figure S1. The content rations of trans to cis isomers of SGR throughout drying processing at different sampling times.
Additional file 2: S1. Optimization of Extraction Processing for Bioactive Ingredients. Table S1. The Box-Behnken design experiment scheme. Table S2. ANOVA of quadratic model terms of three factor variables on responses. Table S3. Predicted and experimental values of the responses obtained under the optimal extraction conditions. Figure S1. Result of the single factor experiments. Figure S2. Response surface plots corresponding to the desirability function.
Data Availability Statement
Not applicable.