Abstract
Background
After renal transplantation, there is a need of immunosuppressive regimens that effectively prevent allograft rejection while minimizing cardiovascular complications. This substudy of the TRITON trial evaluated the cardiovascular effects of autologous bone marrow–derived mesenchymal stromal cells (MSCs) in renal transplant recipients.
Methods and Results
Renal transplant recipients were randomized to MSC therapy, infused at weeks 6 and 7 after transplantation, with withdrawal at week 8 of tacrolimus or standard tacrolimus dose. Fifty‐four patients (MSC group=27; control group=27) underwent transthoracic echocardiography at weeks 4 and 24 after transplantation and were included in this substudy. Changes in clinical and echocardiographic variables were compared. The MSC group showed a benefit in blood pressure control, assessed by a significant interaction between changes in diastolic blood pressure and the treatment group (P=0.005), and a higher proportion of patients achieving the predefined blood pressure target of <140/90 mm Hg compared with the control group (59.3% versus 29.6%, P=0.03). A significant reduction in left ventricular mass index was observed in the MSC group, whereas there were no changes in the control group (P=0.002). The proportion of patients with left ventricular hypertrophy decreased at 24 weeks in the MSC group (33.3% versus 70.4%, P=0.006), whereas no changes were noted in the control group (63.0% versus 48.1%, P=0.29). Additionally, MSC therapy prevented progressive left ventricular diastolic dysfunction, as demonstrated by changes in mitral deceleration time and tricuspid regurgitant jet velocity.
Conclusions
MSC strategy is associated with improved blood pressure control, regression of left ventricular hypertrophy, and prevention of progressive diastolic dysfunction at 24 weeks after transplantation.
Registration
URL: https://www.clinicaltrials.gov; Unique identifier: NCT03398681.
Keywords: immunosuppression, left ventricular diastolic function, left ventricular hypertrophy, mesenchymal stem cells, renal transplantation
Subject Categories: Secondary Prevention, Nephrology and Kidney, Stem Cells
Nonstandard Abbreviations and Acronyms
- CNI
calcineurin inhibitor
- DBP
diastolic blood pressure
- LVM
left ventricular mass
- MSC
mesenchymal stromal cell
- SBP
systolic blood pressure
Clinical Perspective
What Is New?
In renal transplant recipients, mesenchymal stromal cells therapy combined with early tacrolimus withdrawal safely improves blood pressure control, compared with a standard‐dose tacrolimus treatment, at 24 weeks after transplantation.
After renal transplantation, the combination of mesenchymal stromal cells therapy and early tacrolimus withdrawal attenuates adverse left ventricular remodeling, characterized by myocardial hypertrophy and diastolic dysfunction.
What Are the Clinical Implications?
Mesenchymal stromal cells therapy with complete discontinuation of tacrolimus appears a promising approach in renal transplant recipients, effective in the prevention of graft rejection, while exerting potential cardioprotective effects.
Further studies are warranted to determine the impact of this promising immunosuppressive regimen on long‐term cardiovascular outcomes.
Cardiovascular disease is the leading cause of morbidity and mortality in patients with chronic kidney disease and recipients of renal transplantation. 1 Maintenance immunosuppressive drugs, particularly calcineurin inhibitors (CNIs), such as tacrolimus, are important contributors to the elevated cardiovascular risk of renal transplant recipients. 2 CNI‐based therapy is associated with adverse cardiovascular effects including hypertension, dyslipidemia, and new‐onset diabetes. 2 Moreover, the CNI‐induced nephrotoxicity increases the risk of cardiovascular complications and chronic allograft dysfunction in renal transplant recipients. 3 Therefore, after renal transplantation, there is a clear need of immunosuppressive regimens that effectively prevent graft rejection while preserving renal function and minimizing cardiovascular side effects.
Mesenchymal stromal cells (MSCs) have immunosuppressive properties and contribute to tissue repair. Several experimental studies demonstrated that MSCs may increase levels of regulatory T cells and polarize the immune system toward tolerance. 4 , 5 Moreover, phase I studies have shown that MSCs are safe and effective in renal transplant recipients. 6 , 7 , 8 , 9 MSCs seem also to exert regenerative capacities of the myocardium after an ischemic damage and have been proposed as potential treatment for heart failure. 10 In addition, a growing body of evidence suggested that everolimus, a mammalian target of rapamycin inhibitor, may reduce cardiovascular complications after renal transplantation, 11 minimizing CNI exposure and possibly exerting direct cardioprotective effects. 12 , 13 , 14 , 15 , 16 , 17 The safety and feasibility of autologous bone marrow–derived MSC therapy combined with everolimus and complete tacrolimus withdrawal after renal transplantation was demonstrated in the TRITON trial. 18 However, the cardiovascular effects of such strategy in comparison with standard of care (CNI‐based therapy) have not been evaluated. In this substudy of the TRITON trial, we compared the cardiovascular effects of MSC therapy combined with everolimus and complete withdrawal of tacrolimus versus standard of care (tacrolimus‐based therapy).
METHODS
The data that support the findings of the present study are available from the corresponding author upon reasonable request. The protocol of TRITON trial was approved by the local ethics committee at the Leiden University Medical Center, Leiden, and by the Central Committee on Research Involving Human Subjects in the Netherlands. 18 Written informed consent was obtained from all participants.
Study Design and Population
In this echocardiographic substudy of the TRITON trial, a 24‐week investigator‐initiated, randomized, prospective, open‐label, single‐center clinical study, patients who underwent transthoracic echocardiography at 4 and 24 weeks after renal transplantation were included. The description of the trial and the main results have been previously published. 18 In brief, 70 adult recipients of a first kidney transplant from a living donor were enrolled between March 2014 and January 2020 and randomly assigned before transplantation to either MSC therapy with concomitant early withdrawal of tacrolimus or standard tacrolimus dose (control group) in a 1:1 ratio (Figure S1). Patients in the MSC group received 2 doses of autologous bone marrow–derived MSCs (1–2×106 per kilogram body weight) intravenously at weeks 6 and 7 after transplantation, in combination with everolimus and prednisolone. At the time of the second MSC infusion, the dose of tacrolimus was reduced to 50% and completely withdrawn 1 week later. Patients in the control group received everolimus, prednisolone, and tacrolimus. The tacrolimus target was 8 to 12 ng/mL the first 6 weeks after transplantation and lowered to 6 to 8 ng/mL in the control group 7 weeks after transplantation. Moreover, all patients enrolled in the study received alemtuzumab (anti‐CD52), 15 mg subcutaneously, at days 0 and 1, to minimize the risk for acute rejection.
Patients who did not receive the allocated treatment because of abnormal MSC growth, contraindication for prednisone usage, or for MSC infusion during the COVID‐19 pandemic, as well as patients from whom a baseline renal biopsy was not obtained and patients who withdrew informed consent were excluded (Figure 1).
Figure 1. Population included in the cardiovascular subanalysis of the TRITON study.
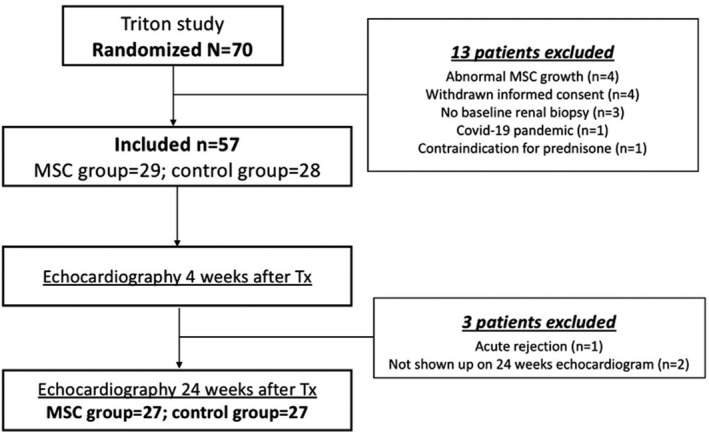
Of 70 renal transplant recipients randomly assigned to MSC therapy or standard tacrolimus regimen, 57 patients received the allocated treatment and were included in the TRITON trial. Patients who underwent transthoracic echocardiography at 4 and 24 weeks after renal transplantation (n=54) were selected for this substudy. MSC indicates mesenchymal stromal cells; and Tx, transplantation.
Changes in echocardiographic variables between 4 and 24 weeks after renal transplantation were evaluated and compared between the randomization arms.
Clinical Follow‐Up
All patients were followed‐up in a dedicated outpatient clinic and treated according to a predefined target of care: systolic blood pressure (SBP) <140 mm Hg and diastolic blood pressure (DBP) <90 mm Hg. The mean blood pressure was obtained by averaging 3 repeated measurements in a sitting position after a 5‐minute rest, using an automated monitor. Blood pressure, weight, estimated glomerular filtration rate (eGFR) calculated by the CKD‐EPI (Chronic Kidney Disease Epidemiology Collaboration) formula, 19 and concomitant medications were recorded 4 and 24 weeks after the renal transplantation.
Transthoracic Echocardiography
Comprehensive transthoracic echocardiography was performed using a commercially available system (E95 System; General Electric‐Vingmed, Horten, Norway) equipped with the 4Vc‐D matrix cardiac probe. With the patients at rest in the left recumbent or supine position, 2‐dimensional and color, spectral continuous‐ and pulsed‐wave Doppler images were obtained from the parasternal, apical, and subcostal windows. All images were digitally stored for offline analysis (EchoPAC version 203; General Electric‐Vingmed).
The left ventricular (LV) linear dimensions were measured on the parasternal long‐axis view at end‐diastole and end‐systole. Subsequently, the LV mass was calculated according to the Devereux formula and indexed for body surface area. 20 LV hypertrophy was defined as an LV mass (LVM) index >115 g/m2 for men and >95 g/m2 for women. 21 In addition, from the apical 2‐ and 4‐chamber views zoomed on the left ventricle, the LV end‐diastolic and end‐systolic volumes were measured and indexed for body surface area, and LV ejection fraction was calculated using the biplane Simpson method. The left atrial volume was obtained from the apical 2‐ and 4‐chamber views using the biplane Simpson method and indexed for body surface area. From a zoomed parasternal long‐axis view, the LV outflow tract diameter was measured in early systole at the level of aortic cusp insertion. The LV outflow tract time‐velocity integral was recorded on the apical 5‐chamber view, with the sample volume positioned 5 mm proximal to the aortic valve. Stroke volume was calculated by multiplying the LV outflow tract area by the LV outflow tract time‐velocity integral and indexed for body surface area. 22
The assessment of LV diastolic function included the measurement of the ratio between the transmitral early (E wave) and late (A wave) diastolic filling velocities and deceleration time of the E wave on pulsed‐wave Doppler recording of the mitral inflow from the apical 4‐chamber view. 23 In addition, using pulsed‐wave tissue Doppler imaging, the early diastolic septal and lateral mitral annular velocities (E′) were measured and averaged. The ratio of the transmitral E wave to E′ (E/E′ ratio), a marker of LV filling pressures, was calculated. 24 Additionally, from color Doppler data of the tricuspid valve flow on a right ventricular‐focused 4‐chamber apical view, the presence of tricuspid regurgitation was assessed, and on continuous‐wave Doppler of the regurgitant jet, the peak jet velocity was measured. Pulmonary artery systolic pressure (PASP) was estimated from the tricuspid regurgitant jet velocity using the simplified Bernoulli equation and adding the estimated right atrial pressure: PASP=4(v)2+right atrial pressure, where v is the peak velocity (in meters per second) of the tricuspid regurgitant jet and right atrial pressure is estimated from diameter and respiratory changes of the inferior vena cava. 25
Statistical Analysis
Normally distributed continuous variables are presented as mean±standard deviation, whereas nonnormally distributed data are presented as median and interquartile range. Categorical variables are displayed as frequencies and percentages.
Changes in clinical and echocardiographic variables within each randomization arm were assessed by the paired Student t test (for normally distributed continuous variables), Wilcoxon test (for nonnormally distributed continuous variables), and McNemar test (for categorical variables). The treatment effect (MSC therapy versus tacrolimus‐based therapy) was evaluated for each clinical and echocardiographic variable using an ANCOVA model, adjusted for baseline variable. Additionally, a separate extension of the simple ANCOVA model was performed for echocardiographic variables that were statistically significant in the first model including baseline SBP, DBP, eGFR, weight, and the use of an angiotensin‐converting enzyme inhibitor/angiotensin II receptor blocker (ACE‐I/ARB) as covariates. Finally, a sensitivity analysis was conducted for the evaluation of treatment effect on changes in LVM index, excluding 1 patient in the control group who experienced a disproportionate worsening of the LV hypertrophy. All statistical analyses were 2‐sided, and P<0.05 was considered statistically significant. In addition, for the analysis of treatment effect on clinical and echocardiographic variables, Bonferroni correction for multiple comparisons was performed, and adjusted significant threshold has also been provided. Data analysis was performed using SPSS version 25.0 (IBM, Armonk, NY).
RESULTS
Of 57 patients randomized to MSC therapy (n=29) or to control (n=28), 54 patients underwent transthoracic echocardiography at 4 and 24 weeks after renal transplantation and were included in the present subanalysis (Figure 1). One patient randomized to MSC therapy developed acute rejection and tacrolimus was reintroduced, whereas 2 other patients (1 randomized to MSC therapy and 1 control) did not undergo the 24‐week echocardiogram. Patient survival during the study follow‐up was 100% in both groups. The baseline characteristics of the patients in each randomization arm are summarized in Table 1.
Table 1.
Characteristics of Each Treatment Group at 4 Weeks After Transplantation
| Variable | MSC group, n=27 | Control group, n=27 |
|---|---|---|
| Age, y | 50.2±14.0 | 50.0±15.5 |
| Male sex, n (%) | 24 (88.9) | 20 (74.0) |
| Body weight, kg | 79.0±15.5 | 79.0±13.6 |
| BMI, kg/m2 | 25.8±3.6 | 25.0±4.0 |
| Primary diagnosis, n (%) | ||
| Hypertension | 3 (11.1) | 9 (33.3) |
| Polycistic kidney disease | 9 (33.3) | 3 (11.1) |
| IgA nephropaty | 6 (22.2) | 3 (11.1) |
| Diabetes | 5 (18.5) | 0 (0) |
| Reflux nephropathy | 0 (0) | 2 (7.4) |
| Membranous nephropathy | 1 (3.7) | 1 (3.7) |
| Lupus nephritis | 1 (3.7) | 0 (0) |
| Other | 1 (3.7) | 3 (11.1) |
| Unknown | 1 (3.7) | 6 (22.2) |
| SBP, mm Hg | 143.7±14.7 | 145.5±12.9 |
| DBP, mm Hg | 87.2±11.0 | 87.3±10.8 |
| MBP, mm Hg | 106.0±10.3 | 106.7±9.3 |
| BP >140/90 mm Hg | 18 (66.7) | 21 (77.8) |
| eGFR, mL/min per 1.73 m2 | 60.8±16.1 | 45.9±10.9 |
| Concomitant cardiovascular medications | ||
| Calcium‐channel blocker, n (%) | 18 (66.7) | 19 (70.4) |
| ACE‐I/ARB, n (%) | 11 (40.7) | 4 (14.8) |
| β‐Blocker, n (%) | 10 (37) | 10 (37) |
| Thiazide diuretic, n (%) | 0 (0) | 4 (14.8) |
| α‐Blocker, n (%) | 3 (11.1) | 1 (3.7) |
| Antihypertensive drugs ≥2, n (%) | 15 (55.6) | 11 (40.7) |
| Statin, n (%) | 7 (27.9) | 10 (37) |
| Insulin (%) | 6 (22.2) | 0 (0) |
ACE‐I indicates angiotensin‐converting enzyme inhibitor; ARB, angiotensin receptor blocker; BMI, body mass index; BP, blood pressure; DBP, diastolic blood pressure; eGFR, estimated glomerular filtration rate, calculated by the Chronic Kidney Disease Epidemiology Collaboration formula; IgA, immunoglobulin A; MBP, mean blood pressure; MSC, mesenchymal stromal cells; and SBP, systolic blood pressure.
Clinical Follow‐Up
Changes in clinical variables between 4 and 24 weeks after transplantation for each randomization group are presented in Table 2. At 24 weeks after transplantation, patients in the MSC group did not show significant changes in DBP, whereas patients in the control group had higher values of DBP compared with baseline, leading to a significant difference between groups (P=0.005). On the contrary, SBP values remained unchanged in both randomization groups. At 24 weeks after transplantation, a higher proportion of patients achieved the predefined blood pressure target of <140/90 mm Hg in the MSC therapy group compared with the control group (59.3% versus 29.6%, P=0.03). There were no significant differences in antihypertensive treatment used between the 2 treatment groups (Table S1).
Table 2.
Changes in Clinical Parameters Between 4 and 24 Weeks After Transplantation in Each Treatment Group
| Clinical variables | MSC group | Control group | P value* | ||
|---|---|---|---|---|---|
| 4 wk | 24 wk | 4 wk | 24 wk | ||
| SBP, mm Hg | 143.7±14.7 | 137±16.1 | 145.5±12.9 | 143.2±17.3 | 0.192 |
| DBP, mm Hg | 87.2±11.0 | 82.8±8.9 | 87.3±10.8 | 90.6±11.0 | 0.005 |
| MBP, mm Hg | 106.0±10.3 | 100.9±9.3 | 106.7±9.3 | 108.2±11.9 | 0.015 |
| Weight, kg | 77.3±12.1 | 81.5±12.5 | 81.4±13.5 | 83.5±15.1 | 0.737 |
| eGFR, mL/min per 1.73 m2 | 60.8±16.1 | 55.6±15.2 † | 45.9±10.9 | 46.5±15.6 | 0.345 |
DBP indicates diastolic blood pressure; eGFR, estimated glomerular filtration rate, calculated by the Chronic Kidney Disease Epidemiology Collaboration formula; MBP, mean blood pressure; MSC, mesenchymal stromal cells; and SBP, systolic blood pressure.
Calculated using an ANCOVA model with baseline adjustment. Applying a Bonferroni correction for multiple comparisons (21 comparisons), P<0.0023 was the significance threshold.
P<0.05 vs baseline within each randomization arm.
Additionally, at 24 weeks after transplantation, patients in the MSC therapy group had a lower eGFR compared with baseline, whereas the eGFR remained unchanged in the control group (P=0.345).
Echocardiographic Follow‐Up
Changes in echocardiographic variables between 4 and 24 weeks after transplantation for each randomization group are presented in Table 3. Between 4 and 24 weeks after transplantation, a reduction in LVM index was observed in the MSC therapy group, whereas in the control group, there were no significant changes in the LVM index over time, leading to a significant difference between groups (P=0.002) (Figures 2 and 3, Figure S2). The association between changes in LVM index and treatment group remained significant after correcting for baseline SBP, DBP, weight, eGFR, and the use of ACE‐I/ARB (P=0.011). A sensitivity analysis performed excluding 1 outlier detected in the control group (Figure S2) confirmed these results (P=0.005 for the baseline model and P=0.032 for the model adjusted for baseline clinical variables). Additionally, the proportion of patients with LV hypertrophy reduced significantly at 24 weeks in the MSC therapy group (33.3% versus 70.4%, P=0.006), whereas it did not change in the control group (63.0% versus 48.1%, P=0.29).
Table 3.
Changes in Echocardiographic Parameters Between 4 and 24 Weeks After Transplantation in Each Treatment Group
| Echocardiographic variables | MSC group | Control group | P value* | ||
|---|---|---|---|---|---|
| 4 wk | 24 wk | 4 wk | 24 wk | ||
| LVEDD, mm | 49.2±6.9 | 49.4±6.2 | 49.8±5.3 | 51.8±6.6 † | 0.181 |
| LVESD, mm | 28.2±4.8 | 29.3±5.3 | 27.4±5.9 | 30.3±5.7 † | 0.393 |
| LV septum thickness, mm | 12±2.8 | 11.1±2.0 | 11.6±2.7 | 11.2±2.1 | 0.680 |
| LVPW thickness, mm | 12.8±2.5 | 11.3±1.7 † | 12.0±1.9 | 12.4±1.9 | 0.024 |
| LVM index, g/m2 | 124±26.4 | 108.9±23.1 † | 115.4±23.1 | 124±29.6 | 0.002 |
| LVEDV index, mL/m2 | 56.5±15.4 | 58.8±17.2 | 52.5±12.9 | 56.4±11.6 † | 0.857 |
| LVESV index, mL/m2 | 21.1±7.5 | 24.0±10.0 | 18.4±6.7 | 22.3±7.8 † | 0.886 |
| LVEF, % | 63.0±6.5 | 60.5±7.9 | 65.0±8.5 | 60.9±7.7 † | 0.973 |
| SV index, mL/m2 | 45.6±10.9 | 45.2±8.4 | 42.1±12.9 | 42.2±11.9 | 0.564 |
| LAV index, mL/m2 | 34.01±10.5 | 35.3±11.1 | 31.5±9.3 | 34.0±10.5 | 0.830 |
| Mitral E:A ratio | 0.9 (0.8–1.2) | 1.0 (0.8–1.3) | 0.8 (0.7–1.0) | 0.9 (0.7–1.2) † | 0.593 |
| E‐wave deceleration time, ms | 174.9±47.6 | 188.5±47.1 | 179.4±69.3 | 230.2±61.2 † | 0.004 |
| Mitral annular E′, cm/s | 11.0±3.2 | 12.3±3.8 † | 10.4±3.4 | 11.1±3.7 | 0.334 |
| Mitral E/E′ ratio | 7.2±2.6 | 7.1±2.8 | 7.0±2.8 | 6.9±2.1 | 0.582 |
| TR jet velocity, m/s | 2.5±0.4 | 2.3±0.3 | 2.1±0.3 | 2.4±0.3 | 0.001 |
| PASP, mm Hg | 29±9 | 25±4 | 21±6 | 26±5 | 0.272 |
LAV indicates left atrial volume; LV, left ventricular; LVEDD, left ventricular end‐diastolic diameter; LVEDV, left ventricular end‐diastolic volume; LVEF, left ventricular ejection fraction; LVESD, left ventricular end‐systolic diameter; LVESV, left ventricular end‐systolic volume; LVM, left ventricular mass; LVPW, left ventricular posterior wall; MSC, mesenchymal stromal cells; PASP, pulmonary artery systolic pressure; SV, stroke volume; and TR, tricuspid regurgitation.
Calculated using an ANCOVA model with baseline adjustment. Applying a Bonferroni correction for multiple comparisons (21 comparisons), P<0.0023 was the significance threshold.
P<0.05 vs baseline within each randomization arm.
Figure 2. Changes in left ventricular mass (LVM) index over time.
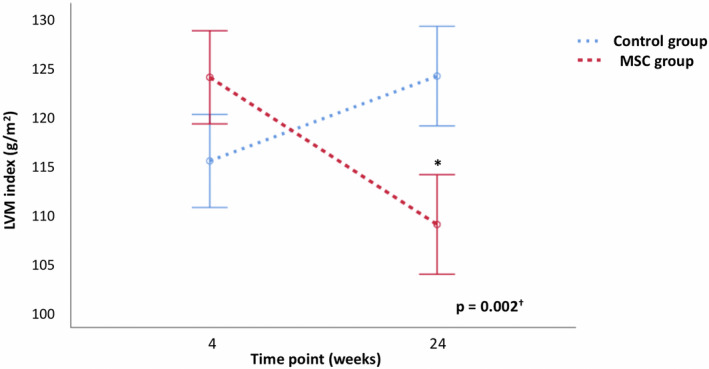
Changes in estimated marginal means of LVM index between 4 and 24 weeks after transplantation in each treatment group. Error bars denote the standard error of the mean. *P<0.05 vs baseline within each treatment group. † P value calculated using an ANCOVA model with baseline adjustment. MSC indicates mesenchymal stromal cells.
Figure 3. Example of the changes in left ventricular (LV) hypertrophy in each treatment group.
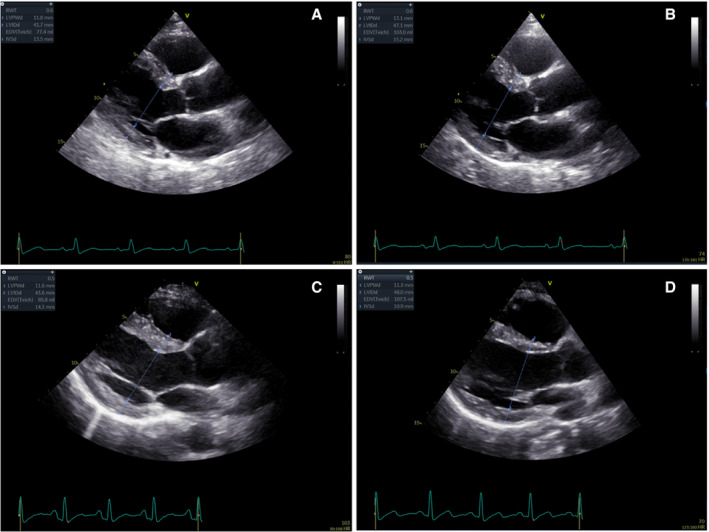
This figure illustrates an example of the changes in linear dimensions and LV hypertrophy between 4 and 24 weeks after transplantation in each treatment group. The upper panels (A and B) show the worsening of LV hypertrophy in a male patient of the control group (increase of left ventricular mass [LVM] index from 95 to 130 g/m2). The lower panels (C and D) display the regression of LV hypertrophy in a male patient of the mesenchymal stromal cells group (decrease of LVM index from 118 to 102 g/m2).
At 24 weeks after transplantation, patients randomized to the control group showed an increase in LV end‐diastolic volume index and LV end‐systolic volume index with a reduction in LV ejection fraction, whereas patients randomized to MSC therapy did not have relevant changes in these variables (Table 3). However, these differences were not statistically significant.
When evaluating LV diastolic function, patients in the control group presented a prolongation of E‐wave deceleration time at 24 weeks after transplantation, whereas E‐wave deceleration time did not change in the MSC therapy group, leading to a significant difference between groups (P=0.004). The association between changes in E‐wave deceleration time and treatment group remained significant after correcting for baseline SBP, DBP, eGFR, weight, and the use of ACE‐I/ARB (P=0.010). Furthermore, tricuspid regurgitant jet velocity reduced at 24 weeks after transplantation in the MSC therapy group and increased in the control group, and these opposite changes resulted in a significant difference between groups (P=0.001). Nevertheless, changes in tricuspid regurgitant jet velocity were not significantly different between groups when correcting for baseline clinical variables and the use of ACE‐I/ARB (P=0.433).
DISCUSSION
The present study showed that MSC therapy combined with early tacrolimus withdrawal in renal transplant recipients is associated with: (1) improved blood pressure control, (2) regression of LV hypertrophy, and (3) prevention of progressive LV diastolic dysfunction compared with a tacrolimus‐based regimen.
Renal transplant recipients have an increased risk of cardiovascular morbidity and mortality. 1 Poor systemic blood pressure control, LV diastolic dysfunction, and atherosclerosis‐related complications are frequently observed in renal transplant recipients and may impair renal function, accelerating allograft failure. 26
Current immune‐suppressive treatment is effective in preventing rejection but contributes to the increased risk of cardiovascular complications that ultimately may have an impact on the clinical outcomes of kidney transplant recipients. CNIs increase systemic arterial hypertension through several mechanisms, including endothelin‐mediated systemic vasoconstriction, impaired vasodilatation secondary to a reduction in nitric oxide, and afferent arteriolar vasoconstriction, mediated by the upregulation of the renin–angiotensin–aldosterone system and the sympathetic nervous system. 27 Poor blood pressure control has been associated with chronic graft failure in renal transplant recipients. 28 , 29 Moreover, systemic arterial hypertension is the main risk factor for both worsening and de novo occurrence of LV hypertrophy after renal transplantation. 30 LV hypertrophy is a common finding among kidney transplant recipients, with a prevalence ranging from 50% to 70% of patients 31 , 32 , 33 , 34 and is a strong predictor of both all‐cause mortality and new‐onset heart failure. 30 However, it is important to note that CNI‐withdrawal immunosuppressive regimens are effective in reducing the prevalence of systemic arterial hypertension with a subsequent improvement in LV function but with an increased rate of graft rejection. 35 These results underscore the need for alternative immunosuppressive treatments that minimize cardiovascular effects while preventing graft rejection.
In the present study, a better control of arterial blood pressure was achieved in the group of patients treated with MSCs and CNI discontinuation as compared with the control group. This was accompanied by a significant reduction in LVM index and in the prevalence of LV hypertrophy at 24 weeks after transplantation without an increased rejection rate. 18 Additionally, MSC therapy seemed to prevent the progressive deterioration of LV diastolic function, as assessed by changes in mitral deceleration time and tricuspid regurgitant jet velocity. The regression of LV hypertrophy observed in the MSC group was the result of the decrease in LV thickness, mostly of the posterior wall, whereas there were no significant changes in the LV end‐diastolic diameter.
Patients in the MSC group showed a higher eGFR at baseline in comparison with controls and had a reduction of eGFR during the study period of 24 weeks. On the other hand, a post hoc analysis of the TRITON study 18 demonstrated that renal function remains stable in the MSC group up to 5 years, whereas it gradually declined in the control group. Notably, the association between changes in LVM index and treatment group remained statistically significant after correcting for baseline clinical variables, including eGFR values.
The achievement of better blood pressure control in the MSC group may, at least partially, explain the more favorable LV remodeling, in comparison with the control group. Nevertheless, whether a direct antiremodeling action of MSCs or CNI withdrawal contributed to the regression of LV hypertrophy remains to be ascertained. Studies conducted in animal models of surgically induced renovascular hypertension showed that MSCs are effective in reducing LV hypertrophy and myocardial fibrosis. 36 , 37 Besides their immunosuppressive and anti‐inflammatory properties, MSCs could exert an antifibrotic action through several mechanisms, such as improving the myocardial regional blood flow through the upregulation of vascular endothelial growth factor, 38 , 39 downregulating the myocardial expression of angiotensin II receptor type 1, and stimulating the secretion of antifibrotic cytokines such as adrenomedullin and metalloproteinases. 40 , 41 Moreover, preclinical and clinical studies showed that the intracoronary or intramyocardial administration of MSCs may exert reparative capacities of the myocardium after an ischemic injury. 10 MSCs have been suggested to stimulate angiogenesis and myocardial regeneration through the paracrine activation of resident stem cells in the myocardium. 42 However, clinical trials that tested the use of MSCs in patients with ischemic heart failure have not shown consistent outcome benefits. 10 Therefore, the beneficial effects of MSC therapy in cardiovascular disease needs to be further clarified by larger randomized controlled studies.
On the other hand, there are conflicting data about the effect of CNIs on cardiac hypertrophy. In experimental models, calcineurin inhibition seemed to prevent the development of LV hypertrophy, 43 , 44 , 45 although other studies failed to confirm these data. 46 Moreover, a recent study in recipients of cardiac transplantation reported that the combination of low‐dose tacrolimus and everolimus, compared with standard‐dose tacrolimus, attenuates LV hypertrophy, with a reduction in myocardial fibrosis measured on late gadolinium‐enhanced magnetic resonance and improvement in myocardial strain at 1 year after transplantation. 47 Of note, among patients treated with a standard dose of tacrolimus, there was an increase in LV hypertrophy and fibrosis, despite no changes in blood pressure, suggesting a direct adverse remodeling effect of CNIs. 47 Additionally, a strategy of late CNI withdrawal has been associated with prevention of progressive deterioration of LV diastolic function at 2 years after renal transplantation, but without relevant changes in LVM index. 48 However, most of the patients enrolled in the study did not have LV hypertrophy at the time of the randomization, 48 thus challenging the observation of any significant variations in LVM index.
Finally, it is important to note that the proportion of patients using an ACE‐I/ARB at 4 weeks after transplantation was higher in the MSC group than in the control group. The treatment with ACE‐I has been previously associated with regression of LV hypertrophy in renal transplant recipients. 49 Nevertheless, the association between reduction of LVM index and MSC therapy remained significant after correcting for the use of an ACE‐I/ARB at baseline.
Study Limitations
The present study was conducted in a single center, with a small‐sized population and a short follow‐up period. Therefore, our results need to be confirmed in larger trials, with long‐term follow‐up. Moreover, this study was not designed to discriminate between the effects of MSC therapy and CNI withdrawal on the blood pressure or the myocardium itself or both. The assessment of myocardial fibrosis with cardiac magnetic resonance would have strengthened our results. However, this goal was beyond the aim of the present study. More generally, the open‐label design of the TRITON trial, required by the procedure of bone marrow aspiration and MSCs infusion, introduces a risk for bias in management (ie, antihypertensive therapies).
CONCLUSIONS
The present study provides evidence that MSC therapy in combination with early CNI discontinuation is associated with better blood pressure control, regression of LV hypertrophy, and prevention of progressive diastolic dysfunction after renal transplantation without increased risk of graft rejection.
Sources of Funding
None.
Disclosures
The Department of Cardiology, Heart Lung Center, Leiden University Medical Center received research grants from Abbott Vascular, Bayer, Bioventrix, Medtronic, Biotronik, Boston Scientific, GE Healthcare and Edwards Lifesciences. Drs Bax and Marsan received speaking fees from Abbott Vascular. Dr Delgado received speaking fees from Abbott Vascular, Medtronic, Edwards Lifesciences, MSD, and GE Healthcare. The remaining authors have no disclosures to report.
Supporting information
Table S1
Figures S1–S2
Supplementary Material for this article is available at https://www.ahajournals.org/doi/suppl/10.1161/JAHA.121.023300
For Sources of Funding and Disclosures, see page 9.
REFERENCES
- 1. Ying T, Shi B, Kelly PJ, Pilmore H, Clayton PA, Chadban SJ. Death after kidney transplantation: an analysis by era and time post‐transplant. J Am Soc Nephrol. 2020;31:2887–2899. doi: 10.1681/ASN.2020050566 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Jardine AG, Gaston RS, Fellstrom BC, Holdaas H. Prevention of cardiovascular disease in adult recipients of kidney transplants. Lancet. 2011;378:1419–1427. doi: 10.1016/S0140-6736(11)61334-2 [DOI] [PubMed] [Google Scholar]
- 3. Chapman JR, O’Connell PJ, Nankivell BJ. Chronic renal allograft dysfunction. J Am Soc Nephrol. 2005;16:3015. doi: 10.1681/ASN.2005050463 [DOI] [PubMed] [Google Scholar]
- 4. Aggarwal S, Pittenger MF. Human mesenchymal stem cells modulate allogeneic immune cell responses. Blood. 2005;10:1815–1822. doi: 10.1182/blood-2004-04-1559 [DOI] [PubMed] [Google Scholar]
- 5. Galleu A, Riffo‐Vasquez Y, Trento C, Lomas C, Dolcetti L, Cheung TS, von Bonin M, Barbieri L, Halai K, Ward S, et al. Apoptosis in mesenchymal stromal cells induces in vivo recipient‐mediated immunomodulation. Sci Transl Med. 2017;9:eaam7828. doi: 10.1126/scitranslmed.aam7828 [DOI] [PubMed] [Google Scholar]
- 6. Perico N, Casiraghi F, Introna M, Gotti E, Todeschini M, Cavinato RA, Capelli C, Rambaldi A, Cassis P, Rizzo P, et al. Autologous mesenchymal stromal cells and kidney transplantation: a pilot study of safety and clinical feasibility. Clin J Am Soc Nephrol. 2011;6:412–422. doi: 10.2215/CJN.04950610 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Reinders MEJ, de Fijter JW, Roelofs H, Bajema IM, de Vries DK, Schaapherder AF, Claas FHJ, van Miert PPMC, Roelen DL, van Kooten C, et al. Autologous bone marrow‐derived mesenchymal stromal cells for the treatment of allograft rejection after renal transplantation: results of a phase I study. Stem Cells Transl Med. 2013;2:107–111. doi: 10.5966/sctm.2012-0114 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Dreyer GJ, Groeneweg KE, Heidt S, Roelen DL, Pel M, Roelofs H, Huurman VAL, Bajema IM, Moes DJAR, Fibbe WE, et al. Human leukocyte antigen selected allogeneic mesenchymal stromal cell therapy in renal transplantation: the Neptune study, a phase I single‐center study. Am J Transplant. 2020;20:2905–2915. doi: 10.1111/ajt.15910 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Erpicum P, Weekers L, Detry O, Bonvoisin C, Delbouille M‐H, Grégoire C, Baudoux E, Briquet A, Lechanteur C, Maggipinto G, et al. Infusion of third‐party mesenchymal stromal cells after kidney transplantation: a phase I‐II, open‐label, clinical study. Kidney Int. 2019;95:693–707. doi: 10.1016/j.kint.2018.08.046 [DOI] [PubMed] [Google Scholar]
- 10. Lalu MM, Mazzarello S, Zlepnig J, Dong YYR, Montroy J, McIntyre L, Devereaux PJ, Stewart DJ, David Mazer C, Barron CC, et al. Safety and efficacy of adult stem cell therapy for acute myocardial infarction and ischemic heart failure (SafeCell Heart): a systematic review and meta‐analysis. Stem Cells Transl Med. 2018;7:857–866. doi: 10.1002/sctm.18-0120 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Zeier M, Van Der Giet M. Calcineurin inhibitor sparing regimens using m‐target of rapamycin inhibitors: an opportunity to improve cardiovascular risk following kidney transplantation? Transpl Int. 2011;24:30–42. doi: 10.1111/j.1432-2277.2010.01140.x [DOI] [PubMed] [Google Scholar]
- 12. McMullen JR, Sherwood MC, Tarnavski O, Zhang L, Dorfman AL, Shioi T, Izumo S. Inhibition of mTOR signaling with rapamycin regresses established cardiac hypertrophy induced by pressure overload. Circulation. 2004;109:3050–3055. doi: 10.1161/01.CIR.0000130641.08705.45 [DOI] [PubMed] [Google Scholar]
- 13. Shioi T, McMullen JR, Tarnavski O, Converso K, Sherwood MC, Manning WJ, Izumo S. Rapamycin attenuates load‐induce cardiac hypertrophy in mice. Circulation. 2003;107:1664–1670. doi: 10.1161/01.CIR.0000057979.36322.88 [DOI] [PubMed] [Google Scholar]
- 14. Gao XM, Wong G, Wang B, Kiriazis H, Moore XL, Su YD, Dart A, Du XJ. Inhibition of mTOR reduces chronic pressure‐overload cardiac hypertrophy and fibrosis. J Hypertens. 2006;24:1663–1670. doi: 10.1097/01.hjh.0000239304.01496.83 [DOI] [PubMed] [Google Scholar]
- 15. Topilsky Y, Hasin T, Raichlin E, Boilson BA, Schirger JA, Pereira NL, Edwards BS, Clavell AL, Rodeheffer RJ, Frantz RP, et al. Sirolimus as primary immunosuppression attenuates allograft vasculopathy with improved late survival and decreased cardiac events after cardiac transplantation. Circulation. 2012;125:708–720. doi: 10.1161/CIRCULATIONAHA.111.040360 [DOI] [PubMed] [Google Scholar]
- 16. Raichlin E, Chandrasekaran K, Kremers WK, Frantz RP, Clavell AL, Pereira NL, Rodeheffer RJ, Daly RC, McGregor CGA, Edwards BS, et al. Sirolimus as primary immunosuppressant reduces left ventricular mass and improves diastolic function of the cardiac allograft. Transplantation. 2008;86:1395–1400. doi: 10.1097/TP.0b013e318189049a [DOI] [PubMed] [Google Scholar]
- 17. Kushwaha SS, Raichlin E, Sheinin Y, Kremers WK, Chandrasekaran K, Brunn GJ, Platt JL. Sirolimus affects cardiomyocytes to reduce left ventricular mass in heart transplant recipients. Eur Heart J. 2008;29:2742–2750. doi: 10.1093/eurheartj/ehn407 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Reinders MEJ, Groeneweg KE, Hendriks SH, Bank JR, Dreyer GJ, Vries APJ, Pel M, Roelofs H, Huurman VAL, Meij P, et al. Autologous bone marrow derived mesenchymal stromal cell therapy with early tacrolimus withdrawal: the randomized prospective, single‐center, open‐label TRITON study. Am J Transplant. 2021;21:3055–3065. doi: 10.1111/ajt.16528 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Levey AS, Stevens LA, Schmid CH, Zhang YL, Castro AF, Feldman HI, Kusek JW, Eggers P, Van Lente F, Greene T, et al. A new equation to estimate glomerular filtration rate. Ann Intern Med. 2009;150:604–612. doi: 10.7326/0003-4819-150-9-200905050-00006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Devereux RB, Alonso DR, Lutas EM, Gottlieb GJ, Campo E, Sachs I, Reichek N. Echocardiographic assessment of left ventricular hypertrophy: comparison to necropsy findings. Am J Cardiol. 1986;57:450. doi: 10.1016/0002-9149(86)90771-X [DOI] [PubMed] [Google Scholar]
- 21. Lang RM, Bierig M, Devereux RB, Flachskampf FA, Foster E, Pellikka PA, Picard MH, Roman MJ, Seward J, Shanewise JS, et al. Recommendations for chamber quantification: a report from the American Society of Echocardiography’s Guidelines and Standards Committee and the Chamber Quantification Writing Group, developed in conjunction with the European Association of Echocardiography, a branch of the European Society of Cardiology. J Am Soc Echocardiogr. 2005;18:1440. doi: 10.1016/j.echo.2005.10.005 [DOI] [PubMed] [Google Scholar]
- 22. Baumgartner H, Hung J, Bermejo J, Chambers JB, Edvardsen T, Goldstein S, Lancellotti P, LeFevre M, Miller F Jr, Otto CM. Recommendations on the echocardiographic assessment of aortic valve stenosis: a focused update from the European Association of Cardiovascular Imaging and the American Society of Echocardiography. J Am Soc Echocardiogr. 2017;30:372–392. doi: 10.1016/j.echo.2017.02.009 [DOI] [PubMed] [Google Scholar]
- 23. Nagueh SF, Smiseth OA, Appleton CP, Byrd BF III, Dokainish H, Edvardsen T, Flachskampf FA, Gillebert TC, Klein AL, Lancellotti P, et al. Recommendations for the evaluation of left ventricular diastolic function by echocardiography: an update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. Eur Heart J Cardiovasc Imaging. 2016;17:1321–1360. doi: 10.1093/ehjci/jew082 [DOI] [PubMed] [Google Scholar]
- 24. Ommen SR, Nishimura RA, Appleton CP, Miller FA, Oh JK, Redfield MM, Tajik AJ. Clinical utility of Doppler echocardiography and tissue Doppler imaging in the estimation of left ventricular filling pressures: a comparative simultaneous Doppler‐catheterization study. Circulation. 2000;102:1788. doi: 10.1161/01.CIR.102.15.1788 [DOI] [PubMed] [Google Scholar]
- 25. Rudski LG, Lai WW, Afilalo J, Hua L, Handschumacher MD, Chandrasekaran K, Solomon SD, Louie EK, Schiller NB. Guidelines for the echocardiographic assessment of the right heart in adults: a report from the American Society of Echocardiography endorsed by the European Association of Echocardiography, a registered branch of the European Society of Cardiology, and the Canadian Society of Echocardiography. J Am Soc Echocardiogr. 2010;23:685–713. doi: 10.1016/j.echo.2010.05.010 [DOI] [PubMed] [Google Scholar]
- 26. Jankowski J, Floege J, Fliser D, Böhm M, Marx N. Cardiovascular disease in chronic kidney disease: pathophysiological insights and therapeutic options. Circulation. 2021;143:1157–1172. doi: 10.1161/CIRCULATIONAHA.120.050686 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Mark AL. Cyclosporine, sympathetic activity, and hypertension. N Engl J Med. 1990;323:748–750. doi: 10.1056/NEJM199009133231109 [DOI] [PubMed] [Google Scholar]
- 28. Opelz G, Döhler B; Collaborative Transplant Study . Improved long‐term outcomes after renal transplantation associated with blood pressure control. Am J Transplant. 2005;5:2725–2731. doi: 10.1111/j.1600-6143.2005.01093.x [DOI] [PubMed] [Google Scholar]
- 29. Opelz G, Wujciak T, Ritz E. Association of chronic kidney graft failure with recipient blood pressure. Collaborative Transplant Study. Kidney Int. 1998;53:217–222. doi: 10.1046/j.1523-1755.1998.00744.x [DOI] [PubMed] [Google Scholar]
- 30. Rigatto C, Foley R, Jeffery J, Negrijn C, Tribula C, Parfrey P. Electrocardiographic left ventricular hypertrophy in renal transplant recipients: prognostic value and impact of blood pressure and anemia. J Am Soc Nephrol. 2003;14:462–468. doi: 10.1097/01.ASN.0000043141.67989.39 [DOI] [PubMed] [Google Scholar]
- 31. Dounousi E, Mitsis M, Naka KK, Pappas C, Lakkas L, Harisis C, Pappas K, Koutlas V, Tzalavra I, Spanos G, et al. Differences in cardiac structure assessed by echocardiography between renal transplant recipients and chronic kidney disease patients. Transplant Proc. 2014;46:3194–3198. doi: 10.1016/j.transproceed.2014.10.034 [DOI] [PubMed] [Google Scholar]
- 32. Slubowska K, Lichodziejewska B, Pruszczyk P, Szmidt J, Durlik M. Left ventricular hypertrophy in renal transplant recipients in the first year after transplantation. Transplant Proc. 2014;46:2719–2723. doi: 10.1016/j.transproceed.2014.08.019 [DOI] [PubMed] [Google Scholar]
- 33. Dudziak M, Debska‐Slizień A, Rutkowski B. Cardiovascular effects of successful renal transplantation: a 30‐month study on left ventricular morphology, systolic and diastolic functions. Transplant Proc. 2005;37:1039–1043. doi: 10.1016/j.transproceed.2004.12.201 [DOI] [PubMed] [Google Scholar]
- 34. Ferreira SR, Moisés VA, Tavares A, Pacheco‐Silva A. Cardiovascular effects of successful renal transplantation: a 1‐year sequential study of left ventricular morphology and function, and 24‐hour blood pressure profile. Transplantation. 2002;74:1580–1587. doi: 10.1097/00007890-200212150-00016 [DOI] [PubMed] [Google Scholar]
- 35. Karpe KM, Talaulikar GS, Walters GD. Calcineurin inhibitor withdrawal or tapering for kidney transplant recipients. Cochrane Database Syst Rev. 2017;7:CD006750. doi: 10.1002/14651858.CD006750.pub2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Khater NA, Selim SA, Abd El‐Baset SA, Abd El Hameed SH. Therapeutic effect of mesenchymal stem cells on experimentally induced hypertensive cardiomyopathy in adult albino rats. Ultrastruct Pathol. 2017;41:36–50. doi: 10.1080/01913123.2016.1260080 [DOI] [PubMed] [Google Scholar]
- 37. Eirin A, Zhu XY, Ebrahimi B, Krier JD, Riester SM, van Wijnen AJ, Lerman A, Lerman LO. Intrarenal delivery of mesenchymal stem cells and endothelial progenitor cells attenuates hypertensive cardiomyopathy in experimental renovascular hypertension. Cell Transplant. 2015;24:2041–2053. doi: 10.3727/096368914X685582 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Dai W, Hale SL, Kloner RA. Role of a paracrine action of mesenchymal stem cells in the improvement of left ventricular function after coronary artery occlusion in rats. Regen Med. 2007;2:63–68. doi: 10.2217/17460751.2.1.63 [DOI] [PubMed] [Google Scholar]
- 39. Healy ME, Bergin R, Mahon BP, English K. Mesenchymal stromal cells protect against caspase 3‐mediated apoptosis of CD19+ peripheral B cells through contact‐dependent upregulation of VEGF. Stem Cells Dev. 2015;24:2391–2402. doi: 10.1089/scd.2015.0089 [DOI] [PubMed] [Google Scholar]
- 40. Oliveira‐Sales EB, Maquigussa E, Semedo P, Pereira LG, Ferreira VM, Câmara NO, Bergamaschi CT, Campos RR, Boim MA. Mesenchymal stem cells (MSC) prevented the progression of renovascular hypertension, improved renal function and architecture. PLoS One. 2013;8:e78464. doi: 10.1371/journal.pone.0078464 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Wang C, Dobrzynski E, Chao J, Chao L. Adrenomedullin gene delivery attenuates renal damage and cardiac hypertrophy in Goldblatt hypertensive rats. Am J Physiol Renal Physiol. 2001;280:F964–F971. doi: 10.1152/ajprenal.2001.280.6.F964 [DOI] [PubMed] [Google Scholar]
- 42. Hatzistergos KE, Quevedo H, Oskouei BN, Hu Q, Feigenbaum GS, Margitich IS, Mazhari R, Boyle AJ, Zambrano JP, Rodriguez JE, et al. Bone marrow mesenchymal stem cells stimulate cardiac stem cell proliferation and differentiation. Circ Res. 2010;107:913–922. doi: 10.1161/CIRCRESAHA.110.222703 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Lim HW, De Windt LJ, Steinberg L, Taigen T, Witt SA, Kimball TR, Molkentin JD. Calcineurin expression, activation, and function in cardiac pressure‐overload hypertrophy. Circulation. 2000;101:2431. doi: 10.1161/01.CIR.101.20.2431 [DOI] [PubMed] [Google Scholar]
- 44. Sakata Y, Masuyama T, Yamamoto K, Nishikawa N, Yamamoto H, Kondo H, Ono K, Otsu K, Kuzuya T, Miwa T, et al. Calcineurin inhibitor attenuates left ventricular hypertrophy, leading to prevention of heart failure in hypertensive rats. Circulation. 2000;102:2269. doi: 10.1161/01.CIR.102.18.2269 [DOI] [PubMed] [Google Scholar]
- 45. Gelpi RJ, Gao S, Zhai P, Yan L, Hong C, Danridge LMA, Ge H, Maejima Y, Donato M, Yokota M, et al. Genetic inhibition of calcineurin induces diastolic dysfunction in mice with chronic pressure overload. Am J Physiol Heart Circ Physiol. 2009;297:H1814. doi: 10.1152/ajpheart.00449.2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Ding B, Price RL, Borg TK, Weinberg EO, Halloran PF, Lorell BH. Pressure overload induces severe hypertrophy in mice treated with cyclosporine, an inhibitor of calcineurin. Circ Res. 1999;84:729. doi: 10.1161/01.RES.84.6.729 [DOI] [PubMed] [Google Scholar]
- 47. Anthony C, Imran M, Pouliopoulos J, Emmanuel S, Iliff JW, Moffat KJ, Ross J, Graham RM, Kotlyar E, Muthiah K, et al. Everolimus for the prevention of calcineurin inhibitor‐induced left ventricular hypertrophy after heart transplantation (RADTAC Study). JACC Heart Fail. 2021;9:301–313. doi: 10.1016/j.jchf.2021.01.007 [DOI] [PubMed] [Google Scholar]
- 48. Mourer JS, Ewe SH, Mallat MJ, Ng AC, Rabelink TJ, Bax JJ, Delgado V, de Fijter JW. Late calcineurin inhibitor withdrawal prevents progressive left ventricular diastolic dysfunction in renal transplant recipients. Transplantation. 2012;94:721–728. doi: 10.1097/TP.0b013e3182603297 [DOI] [PubMed] [Google Scholar]
- 49. Paoletti E, Cassottana P, Amidone M, Gherzi M, Rolla D, Cannella G. ACE inhibitors and persistent left ventricular hypertrophy after renal transplantation: a randomized clinical trial. Am J Kidney Dis. 2007;50:133–142. doi: 10.1053/j.ajkd.2007.04.013 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Table S1
Figures S1–S2


