Highlights
-
•
Radiation-induced angiosarcomas (ASs) of the gynecologic tract are rare neoplasms.
-
•
We report a case of radiation-induced AS of the vagina and vulva.
-
•
Few cases of radiation-induced AS of the vagina and vulva have been reported.
-
•
This entity is to be considered when a tumor arises in a previously irradiated area.
Keywords: Angiosarcoma, Radiation, Vagina, Vulva
Abstract
Angiosarcomas are uncommon malignant mesenchymal neoplasms of endothelial origin. They may be primary or secondary to radiation exposure, chronic lymphedema or to other associated risk factors. They can occur anywhere in the body, with the most common location being the skin of the head and neck. Radiation-induced angiosarcomas of the gynecologic tract are very rare with only few cases reported in the literature. We report a case of a 54-year-old lady who developed angiosarcoma of the vagina and vulva 9 years following radiotherapy for cervical cancer. She was treated with chemoradiotherapy and died nine months following the diagnosis of angiosarcoma. We also performed a literature review of the radiation-induced angiosarcomas arising in the vagina and vulva. Angiosarcomas should always be considered in the differential diagnosis when dealing with a tumor located in a previously irradiated area, as they may clinically mimic recurrence of the original tumor the patient had.
1. Introduction
Angiosarcomas (ASs) are rare aggressive mesenchymal neoplasms of vascular origin. They can occur almost anywhere in the body: skin/subcutaneous tissue (most common location), superficial and deep soft tissue, and internal organs (Abraham et al., 2007). They carry a poor prognosis, with a 5-year overall survival of around 30% (Kruse et al., 2014). Almost half of the patients die within the first year following the diagnosis (Meis-Kindblom and Kindblom, 1998). In general, sarcomas of the gynecologic tract are uncommon; hence the rarity of ASs in this region. Post-radiation ASs of the gynecologic tract are exceedingly rare, with only few cases reported in the literature (Betancourt et al., 2019). Herein, we present a case of AS of the vagina and vulva following radiotherapy for cervical adenocarcinoma, mimicking clinically tumor recurrence. To our knowledge, 9 cases of radiation-induced AS of the vagina (6 cases) and vulva (3 cases) were previously reported in the literature (Morgan et al., 1989, Tohya et al., 1991, Chan and SenGupta, 1991, Morimura et al., 2001, Takeuchi et al., 2005, Guirguis et al., 2007, Brady et al., 2011, Yost et al., 2017, Meyer et al., 2021).
2. Case report
A 54-year-old female patient presented for pelvic pain and recurrent urinary tract infections of several month duration. Her past medical history was notable for cervical adenocarcinoma (FIGO stage IIB), diagnosed 10 years prior to presentation and treated with concurrent chemotherapy, pelvic radiotherapy and brachytherapy. Her treatment ended 9 years prior to presentation. At the time of presentation, the physical exam and pelvic ultrasound were unremarkable. PET scan and pelvic MRI did not show any recurrent disease. Urine culture, cystoscopy with random urinary bladder biopsies were performed and no lesions were identified. The patient was diagnosed with chronic cystitis and was treated with antibiotics accordingly. One month later, the patient presented again for vaginal pain and discharge with no improvement of her previous symptoms. On physical exam, the patient was found to have a vaginal mass and green vaginal secretions. Vaginal cultures were performed and antibiotic treatment was initiated. CT scan of the abdomen and pelvis showed dilatation of the right collecting duct system and ureter, raising the possibility of an obstruction, that may be due to progression of known cervical cancer or post-therapy fibrosis. It also showed new nodules in the right lower quadrant highly suspicious for peritoneal deposits. The overall clinical impression was a recurrence of cervical tumor in the vagina causing compression of the urethra with right-sided hydronephrosis, which in turn lead to pyelonephritis. Biopsy of the vaginal mass was then performed. On histologic examination, the biopsied material consisted predominantly of necroinflammatory tissue with few endocervical glands and scattered groups of highly atypical, pleomorphic, mitotically active tumor cells. The latter displayed focal luminal/vascular architecture (Fig. 1). By immunohistochemical staining, these cells were diffusely and strongly positive for CD31, CD34 and C-MYC (Fig. 2). They were negative for CKAE1/3, CK8/18, CK7, and CD45. Ki-67 proliferation index was high in the tumor cells, reaching 70%. In the setting of the patient’s remote history of radiotherapy, the overall histological and immunohistochemical findings were most consistent with high grade AS. CT scan of the brain and chest were negative for metastatic disease. The tumor was not amenable for surgical resection. Systemic chemotherapy (with weekly Paclitaxel, 80 mg/m2) was initiated. Five months later, the patient developed a new vulvar mass which was biopsied. The biopsy showed a malignant tumor that had a morphology similar to the previously biopsied vaginal mass. Vascular markers (CD31 and CD34) and C-MYC were diffusely and strongly positive within the tumor cells, confirming the diagnosis of high-grade AS. The patient developed a vesicovaginal fistula. Palliative radiotherapy was initiated. Two months later, the patient died from urosepsis.
Fig. 1.
Angiosarcoma A- Pleomorphic spindled and epithelioid cells, haphazardly arranged, with focal luminal architecture (arrows) (hematoxylin-eosin, original magnification x20), B- Mitotic activity is elevated (arrows) (hematoxylin-eosin, original magnification x40).
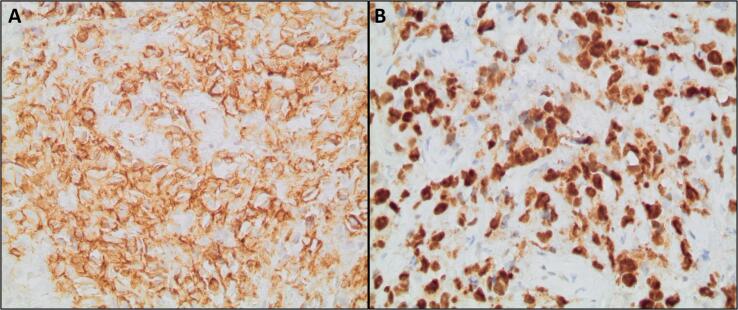
Fig. 2.
Immunohistochemistry A- CD31 showing diffuse membranous positivity in tumor cells (original magnification x40), B- C-MYC showing diffuse and strong nuclear positivity in tumor cells (hematoxylin-eosin, original magnification x40).
3. Discussion
ASs are uncommon malignant mesenchymal neoplasms of endothelial differentiation, accounting for 1–2% of soft tissue sarcomas in adults (Cao et al., 2019). More than half of the cases arise from the skin of the head and neck region, but ASs can occur in any body location (Abraham et al., 2007). The pathogenesis of this entity has not been completely understood, and in most cases, the etiology is unknown; however, several risk factors have been studied and proved to play an important role in the pathogenesis of AS. Chronic sun damage has been suggested as a risk factor for primary cutaneous ASs, given the fact that most of these cases arise in sun-damaged skin of the head and neck in elderly patients (Shustef et al., 2017). However, this relationship is still controversial. Definite predisposing factors for secondary cutaneous AS are radiation exposure (most commonly for breast cancer) and long-standing lymphedema. The typical scenario for the latter condition is Stewart Treves syndrome, which occurs in the setting of radical mastectomy with axillary lymph node dissection. However, AS secondary to chronic lymphedema or radiation exposure can also arise in other locations (Verdura et al., 2019). Risk factors for non-cutaneous AS, such as hepatic AS, include environmental carcinogens such as vinyl chloride monomer, colloidal thorium dioxide (Thorotrast, previously used as a radiocontrast material), androgen, phenylhydrazine, and chronic arsenic ingestion (Chaudhary et al., 2015). A small subset of ASs arises in association with pre-existing benign vascular lesions (hemangioma, vascular malformation, atypical vascular lesion), implanted foreign material, and at sites of prior trauma/surgery (Webster et al., 2011). Around 3% of ASs are associated with genetic syndromes, such as neurofibromatosis (von-Recklinghausen), Maffucci syndrome, Klippel-Trenaunay syndrome, bilateral retinoblastoma, xeroderma pigmentosa, and Ollier disease (Blatt et al., 2019).
The clinical presentation varies depending on the site of involvement. Cutaneous AS usually presents as solitary or multiple purple/red nodules, that may be rapidly growing. These nodules commonly bleed or ulcerate. Secondary cases are clinically similar to spontaneous ones, but tend to appear several years following the inciting agent (Shon and Billings, 2017).
Microscopically, ASs are infiltrative neoplasms that display a wide spectrum of appearances, ranging from well-differentiated to poorly-differentiated forms. Well-differentiated tumors consist of complex anastomosing vascular channels while poorly differentiated tumors consist of solid sheets of atypical cells without clear evidence of vasoformation, thus, making the diagnosis of AS challenging. A single tumor may show a combination of different patterns, from vasoformative areas to more solid areas (Marušić and Billings, 2017). The tumor cells may have a spindled or epithelioid appearance and atypia ranges from mild to marked. The cells may exhibit a characteristic hobnailing appearance. Immunohistochemically, ASs are positive for vascular markers: CD31, CD34, ERG, Factor VIII, FLI1, UEA1, and VEGF. Epithelioid ASs may express cytokeratin, CD30, and EMA. D2-40 (also known as podoplanin, a lymphatic marker) is variably expressed, and its positivity suggests lymphatic differentiation (Wang et al., 2017).
The relationship between radiation exposure and AS, among other types of sarcomas, has been thoroughly studied and demonstrated. Criteria have been established for the classification of post-radiotherapy sarcomas. These include: sarcoma development within the field of prior radiotherapy, previous exposure to a high amount of radiation, period of at least 3–5 years between radiotherapy and sarcoma development, different histology between primary malignancy and secondary sarcoma (Kiyohara et al., 2002). These criteria are met by the present case.
Genes involved in angiogenesis have been found to play an important role in the pathogenesis of post-radiation ASs. MYC gene amplification is the most notorious genetic aberration in secondary ASs. It has even been thought to be the exclusive genetic abnormality seen in most secondary ASs, and was found to be absent in other radiation-induced sarcomas as well as in benign vascular lesions (Guo et al., 2011). MYC gene amplification has been proved to be an early and necessary event in the development of radiation-induced ASs. In fact, it promotes inappropriate progression from G1 phase to S phase, leading to uncontrolled cellular proliferation. This aberration is translated by consistent MYC expression by immunohistochemistry (Habeeb and Rubin, 2019). Positivity for C-MYC immunohistochemical stain further confirms the diagnosis of post-radiation AS as in the present case.
In general, sarcomas of the gynecologic tract are uncommon; hence the rarity of ASs (both primary and secondary) in this location. Vagina and vulva are two of the least affected sites. In fact, among the 52 patients with primary AS of the gynecologic tract reviewed by Kruse et al. in 2014, only two had AS of the vagina, and two had AS of the vulva (Kruse et al., 2014). Radiation-induced ASs of the gynecologic tract are exceedingly rare, and only few cases were reported in the literature. We found 9 case reports of radiation-induced AS of the vagina (6 cases) and vulva (3 cases), summarized in Table 1 (Morgan et al., 1989, Tohya et al., 1991, Chan and SenGupta, 1991, Morimura et al., 2001, Takeuchi et al., 2005, Guirguis et al., 2007, Brady et al., 2011, Yost et al., 2017, Meyer et al., 2021). The primary malignancies were: adenocarcinoma of endometrium (3 cases), squamous cell carcinoma of cervix (1 case), adenocarcinoma of cervix (1 case), carcinoma of cervix (not specified, 1 case), ovarian dysgerminoma (1 case), squamous cell carcinoma of vulva (1 case), and squamous cell carcinoma of anal canal (1 case). The corresponding patients were above the age of 50. The interval between radiation exposure and AS development ranged from 4 to 21 years. The histopathological features were variable, but all of them showed at least focal vascular formation. Two cases displayed a pure epithelioid morphology, leading to the diagnosis of epithelioid AS. By immunohistochemistry, vascular markers were positive in these 9 cases. CD34 was negative in one of them, a case of epithelioid AS of the vulva, which reflects the lower degree of this marker’s sensitivity, when compared to other vascular markers, notably CD31 and ERG (Sullivan et al., 2015). Moreover, CD34 was shown to be variably positive particularly in epithelioid AS, with a positivity ranging from 40 to 100% (Hart and Mandavilli, 2011). The other case of epithelioid AS was positive for cytokeratin (Cam5.2), a common feature of epithelioid malignancies. Ki-67 stain, when performed, revealed a high proliferation index. C-MYC stain was only done in two cases, and was positive in them.
Table 1.
Table summarizing reported cases of radiation-induced AS of the vagina and vulva.
| Case report | Primary malignancy | Site of origin of AS | Age at diagnosis of AS | Interval between radiation exposure and AS development (years) | Epithelioid AS variant | Immunohisto-chemical profile | Therapy | Outcome | |
|---|---|---|---|---|---|---|---|---|---|
|
Positive stains |
Negative stains |
||||||||
| (Morgan et al., 1989) (1) | Adenocarcinoma of endometrium | Vagina and bladder | 72 | 9 | No | Factor VIII | _ | Chemo-therapy | Died 7 months after diagnosis |
| (Tohya et al., 1991) (2) | Squamous cell carcinoma of cervix | Vagina | 73 | 20 | No | Factor VIII | _ | Surgery, chemo-therapy, radiation | Died 30 months after diagnosis |
|
Chan and SenGupta, 1991 (3) |
Carcinoma of cervix | Vaginal vault | 80 | 21 | No | _ | Surgery | Died postoperatively | |
| (Morimura et al., 2001) (4) | Adenocarcinoma of cervix | Vagina | 61 | 14 | No | Factor VIII, CD34 | CK, SMA | Chemo-therapy, interleukin-2 | Disease free for 14 months, then recurrence |
| (Takeuchi et al., 2005) (5) | Dysgerminoma of right ovary | Vagina | 53 | 9 | No | _ | Brachy-therapy, interleukin-2 | Died 16 months after diagnosis | |
| (Guirguis et al., 2007) (6) |
Squamous cell carcinoma of vulva | Vulva (mons pubis) | 78 | 4 | Yes | Factor VIII, CK, CD31, CD34 | _ | Surgery, chemo-therapy | Still being treated at time of writing |
| (Brady et al., 2011) (7) |
Adenocarcinoma of endometrium | Vagina | 73 | 13 | No | CD31, Vim, CD34, EMA | CK, D2-40 | Chemo-therapy | Still being treated at time of writing |
| Ki-67 proliferation index increased | |||||||||
| (Yost et al., 2017) (8) | Adenocarcinoma of endometrium | Vulva | 90 | 5 | Yes | Vim, ERG, CD31 | CD34, CK, p63, MART1, S100, ER, PAX8, CD45 | None | _ |
| (Meyer et al., 2021) (9) | Squamous cell carcinoma of anal canal | Vulva | 65 | 15 | _ | CD31, C-MYC |
CK | Surgery | _ |
| Ki-67 proliferation index increased | |||||||||
| Khaled et al. (present case) | Adenocarcinoma of cervix | Vagina and vulva | 54 | 9 | No | CD31, CD34 and C-MYC | CK, CD45 | Chemo-therapy and radiation | Died 9 months after diagnosis |
| Ki-67 proliferation index increased | |||||||||
AS: Angiosarcoma; CK: Cytokeratin, Vim: Vimentin.
The main differential diagnoses of AS in the setting of prior radiation exposure are atypical vascular lesions (AVLs) and other sarcomas. AVLs are benign entities that usually develop on the skin in previously irradiated areas. The typical scenario is the development of one or more small purple papules/nodules on the skin overlying a patient’s breast or chest 2–3 years following radiotherapy for breast cancer. Histologically, these are well-circumscribed lesions, confined to the dermis with no extension into the subcutis. They consist of anastomosing irregular vascular channels, lined by a single layer of endothelial cells that display no or minimal atypia. No mitoses are usually seen. AVLs can be of lymphatic or vascular type (Patton et al., 2008). The endothelial cells are positive for vascular markers and are negative for C-MYC. In addition to AVLs and AS, any type of sarcoma may occur secondary to radiation exposure. The histopathological findings combined with the immunohistochemical profile should be helpful in making the distinction.
Regardless of grade and etiology (primary vs secondary), ASs are highly malignant and aggressive tumors, with an overall 5-year survival of around 30% (Kruse et al., 2014). Among the 9 reported cases of radiation-induced AS of the vagina and vulva, 4 patients died, and death occurred weeks to 30 months following the diagnosis of AS (Table 1). The dismal prognosis of ASs is attributed to the high rates of recurrence and metastasis. The most common sites of metastasis are the bones, lymph nodes, and lungs, and around 20–50% of patients have metastases at the time of presentation (Young et al., 2010). Several factors have been studied and associated with a poor prognosis; they include: large tumor size, advanced patient age (>70 years), epithelioid component, high mitotic rate, necrosis, greater tumor depth, and positive margins on resection (Deyrup et al., 2008). However, none of these associations has been validated. Moreover, some studies have demonstrated that the original site and etiology of AS affect the prognosis. For instance, ASs originating from retroperitoneal sites, visceral organs, as well as post-radiation cases may be associated with a more adverse outcome, as shown in some series (Young et al., 2010).
Due to the rarity of ASs, there is no consensus regarding the optimal therapeutic strategies. The current treatment options include surgery, chemotherapy, and radiotherapy. Implementation of immunotherapy has been studied and has shown promising results (Morimura et al., 2001, Takeuchi et al., 2005). Until now, radical surgical resection has been the standard of care for AS. In view of the rapidly progressive and infiltrative nature of this entity, it is not uncommon for margins to be involved, thus, wide margins are recommended. Because of the high risk of local recurrence and metastasis, adjuvant chemoradiotherapy is usually indicated (Young et al., 2010). In our patient’s case, the tumor was not amenable for surgical resection. Therefore, she was only treated with chemoradiotherapy and died nine months following the diagnosis of AS.
Despite the increased use of radiotherapy in treating gynecologic neoplasms, the incidence of radiation-induced ASs in this body region remains low. AS should always be considered in the differential diagnosis when dealing with a tumor located in a previously irradiated area, as they may clinically mimic recurrence of the original tumor the patient had. More studies are needed in order to better elucidate the pathogenesis behind these tumors and to help reach more targeted treatment options.
Informed consent statement
Consent to publish could not be obtained from the patient due to patient’s death. Patient’s relatives could not be reached.
CRediT authorship contribution statement
Chirine S. Khaled: Conceptualization, Methodology, Visualization, Writing – original draft. Sara A.J. Sinno: Conceptualization, Methodology, Supervision, Writing – review & editing.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Contributor Information
Chirine S. Khaled, Email: chirinekhaled@hotmail.com.
Sara A.J. Sinno, Email: ss262@aub.edu.lb.
References
- Abraham J.A., Hornicek F.J., Kaufman A.M., Harmon D.C., Springfield D.S., Raskin K.A., Mankin H.J., Kirsch D.G., Rosenberg A.E., Nielsen G.P., Desphpande V., Suit H.D., DeLaney T.F., Yoon S.S. Treatment and outcome of 82 patients with angiosarcoma. Ann. Surg. Oncol. 2007;14(6):1953–1967. doi: 10.1245/s10434-006-9335-y. [DOI] [PubMed] [Google Scholar]
- Betancourt R., Randall C., Kuan-Celarier A., West L., Soper J., Bookhout C. Post-radiation angiosarcoma of the uterine cervix. Gynecologic Oncology Rep. 2019;29:13–15. doi: 10.1016/j.gore.2019.05.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blatt J., Finger M., Price V., Crary S.E., Pandya A., Adams D.M. Cancer Risk in Klippel-Trenaunay Syndrome. Lymphatic Res. Biol. 2019;17(6):630–636. doi: 10.1089/lrb.2018.0049. [DOI] [PubMed] [Google Scholar]
- Brady A., Al-Kalbani M., Al Rawahi T., Nagar H., McCluggage W.G. Postirradiation primary vaginal angiosarcoma with widespread intra-abdominal metastasis. Int. J. Gynecol. Pathol.: Official J. Int. Soc. Gynecol. Pathol. 2011;30(5):514–517. doi: 10.1097/PGP.0b013e318214bd05. [DOI] [PubMed] [Google Scholar]
- Cao J., Wang J., He C., Fang M. Angiosarcoma: a review of diagnosis and current treatment. Am. J. Cancer Res. 2019;9(11):2303–2313. [PMC free article] [PubMed] [Google Scholar]
- Chan W.W., SenGupta S.K. Postirradiation angiosarcoma of the vaginal vault. Arch. Pathol. Lab. Med. 1991;115(5):527–528. [PubMed] [Google Scholar]
- Chaudhary P., Bhadana U., Singh R.A.K., Ahuja A. Primary hepatic angiosarcoma. Eur. J. Surg. Oncol.: J. Eur. Soc. Surg. Oncol. Br. Assoc. Surg. Oncol. 2015;41(9):1137–1143. doi: 10.1016/j.ejso.2015.04.022. [DOI] [PubMed] [Google Scholar]
- Deyrup A.T., McKenney J.K., Tighiouart M., Folpe A.L., Weiss S.W. Sporadic cutaneous angiosarcomas: a proposal for risk stratification based on 69 cases. Am. J. Surg. Pathol. 2008;32(1):72–77. doi: 10.1097/PAS.0b013e3180f633a3. [DOI] [PubMed] [Google Scholar]
- Guirguis A., Kanbour-Shakir A., Kelley J. Epithelioid angiosarcoma of the mons after chemoradiation for vulvar cancer. Int. J. Gynecological Pathology : Official J. Int. Soc. Gynecol. Pathol. 2007;26(3):265–268. doi: 10.1097/01.pgp.0000250145.67462.c9. [DOI] [PubMed] [Google Scholar]
- Guo T., Zhang L., Chang N.-E., Singer S., Maki R.G., Antonescu C.R. Consistent MYC and FLT4 gene amplification in radiation-induced angiosarcoma but not in other radiation-associated atypical vascular lesions. Genes Chromosom. Cancer. 2011;50(1):25–33. doi: 10.1002/gcc.20827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Habeeb O., Rubin B.P. The Molecular Diagnostics of Vascular Neoplasms. Surgical Pathology Clinics. 2019;12(1):35–49. doi: 10.1016/j.path.2018.10.002. [DOI] [PubMed] [Google Scholar]
- Hart J., Mandavilli S. Epithelioid angiosarcoma: a brief diagnostic review and differential diagnosis. Arch. Pathol. Lab. Med. 2011;135(2):268–272. doi: 10.5858/135.2.268. [DOI] [PubMed] [Google Scholar]
- Kiyohara T., Kumakiri M., Kobayashi H., Itoh K., Lao L.-M., Ohkawara A., Nakamura H. Spindle cell angiosarcoma following irradiation therapy for cervical carcinoma. J. Cutan. Pathol. 2002;29(2):96–100. doi: 10.1034/j.1600-0560.2002.290206.x. [DOI] [PubMed] [Google Scholar]
- Kruse A.-J., Sep S., Slangen B.F.M., Vandevijver N.M., Van Gorp T., Kruitwagen R.F., Van de Vijver K.K. Angiosarcomas of primary gynecologic origin: a clinicopathologic review and quantitative analysis of survival. Int. J. Gynecol. Cancer : Official J. Int. Gynecol. Cancer Soc. 2014;24(1):4–12. doi: 10.1097/IGC.0000000000000020. [DOI] [PubMed] [Google Scholar]
- Marušić Z., Billings S.D. Histopathology of Spindle Cell Vascular Tumors. Surgical Pathology Clinics. 2017;10(2):345–366. doi: 10.1016/j.path.2017.01.006. [DOI] [PubMed] [Google Scholar]
- Meis-Kindblom J.M., Kindblom L.-G. Angiosarcoma of soft tissue: a study of 80 cases. Am. J. Surg. Pathol. 1998;22(6):683–697. doi: 10.1097/00000478-199806000-00005. [DOI] [PubMed] [Google Scholar]
- Meyer M., Smith E.S., Leitao Jr. M.M. Angiosarcoma of the vulva following radiation for colorectal cancer. Gynecol. Oncol. Rep. 2021;36:100711. doi: 10.1016/j.gore.2021.100711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morgan M.A., Moutos D.M., Pippitt C.H., Suda R.R., Smith J.J., Thurnau G.R. Vaginal and bladder angiosarcoma after therapeutic irradiation. South. Med. J. 1989;82(11):1434–1436. doi: 10.1097/00007611-198911000-00025. [DOI] [PubMed] [Google Scholar]
- Morimura Y., Hashimoto T., Soeda S., Fujimori K., Yamada H., Yanagida K., Sato A. Angiosarcoma of vagina successfully treated with interleukin-2 therapy and chemotherapy: a case report. J. Obstetrics Gynaecology Res. 2001;27(4):231–235. doi: 10.1111/j.1447-0756.2001.tb01257.x. [DOI] [PubMed] [Google Scholar]
- Patton K.T., Deyrup A.T., Weiss S.W. Atypical vascular lesions after surgery and radiation of the breast: a clinicopathologic study of 32 cases analyzing histologic heterogeneity and association with angiosarcoma. Am. J. Surg. Pathol. 2008;32(6):943–950. doi: 10.1097/pas.0b013e31815bf8fe. [DOI] [PubMed] [Google Scholar]
- Shon W., Billings S.D. Cutaneous Malignant Vascular Neoplasms. Clin. Lab. Med. 2017;37(3):633–646. doi: 10.1016/j.cll.2017.06.004. [DOI] [PubMed] [Google Scholar]
- Shustef, E., Kazlouskaya, V., Prieto, V.G., Ivan, D., Aung, P.P., 2017. Cutaneous angiosarcoma: a current update. J. Clin. Pathol. 70(11):917-25. [DOI] [PubMed]
- Sullivan H.C., Edgar M.A., Cohen C., Kovach C.K., HooKim K., Reid M.D. The utility of ERG, CD31 and CD34 in the cytological diagnosis of angiosarcoma: an analysis of 25 cases. J. Clin. Pathol. 2015;68(1):44–50. doi: 10.1136/jclinpath-2014-202629. [DOI] [PubMed] [Google Scholar]
- Takeuchi K., Deguchi M., Hamana S., Motoyama S., Kitazawa S., Maruo T. A case of postirradiation vaginal angiosarcoma treated with recombinant interleukin-2 therapy. Int. J. Gynecological Cancer : Official J. Int. Gynecol. Cancer Soc. 2005;15(6):1163–1165. doi: 10.1111/j.1525-1438.2005.00268.x. [DOI] [PubMed] [Google Scholar]
- Tohya T., Katabuchi H., Fukuma K., Fujisaki S., Okamura H. Angiosarcoma of the vagina. A light and electronmicroscopy study. Acta Obstet. Gynecol. Scand. 1991;70(2):169–172. doi: 10.3109/00016349109006203. [DOI] [PubMed] [Google Scholar]
- Verdura V., Di Pace B., Concilio M., Guastafierro A., Fiorillo G., Alfano L., Nicoletti G.F., Savastano C., Cascone A.M., Rubino C. A new case of radiation-induced breast angiosarcoma. Int. J. Surg. Case Rep. 2019;60:152–155. doi: 10.1016/j.ijscr.2019.06.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang L., Lao I.W., Yu L., Wang J. Clinicopathological features and prognostic factors in angiosarcoma: A retrospective analysis of 200 patients from a single Chinese medical institute. Oncol. Lett. 2017;14(5):5370–5378. doi: 10.3892/ol.2017.6892. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Webster P., Wujanto L., Fisher C., Walker M., Ramakrishnan R., Naresh K., Thomas J.M., Papalois V., Crane J., Taube D., Duncan N. Malignancies confined to disused arteriovenous fistulae in renal transplant patients: an important differential diagnosis. Am. J. Nephrol. 2011;34(1):42–48. doi: 10.1159/000328908. [DOI] [PubMed] [Google Scholar]
- Yost S., Bradish J., Grossheim L., Hoekstra A. Epithelioid angiosarcoma of the vulva: A case report. Gynecol. Oncology Rep. 2017;21:91–93. doi: 10.1016/j.gore.2017.07.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Young R.J., Brown N.J., Reed M.W., Hughes D., Woll P.J. Angiosarcoma. Lancet Oncol. 2010;11(10):983–991. doi: 10.1016/S1470-2045(10)70023-1. [DOI] [PubMed] [Google Scholar]