Abstract
Background
Increasing evidence shows that genetic variants of genes in the diabetes mellitus (DM) metabolic pathway, such as the vitamin D receptor (VDR) gene rs739837 polymorphism, increase the risk of DM susceptibility. However, the findings have been inconsistent. The present study was performed to evaluate the association of VDR gene rs739837 and type 2 diabetes (T2DM) or gestational diabetes mellitus (GDM) risk.
Methods
A comprehensive meta-analysis and a subgroup analysis were conducted to assess the association between VDR rs739837 and T2DM or GDM among five genetic models (dominant, recessive, homozygote heterozygote, and allele models) using a fixed or random model.
Results
The meta-analysis included 9 studies. In the overall analysis, the results showed that VDR rs739837 was associated with an increased risk of T2DM or GDM in the allele model (T vs. G: OR = 1.088; 95% CI: 1.018–1.163; P = 0.012) and dominant model (TT + GT vs. GG: OR = 1.095; 95% CI: 1.001–1.197; P = 0.047). In the subgroup analysis, VDR rs739837 was also associated with an increased risk of T2DM in the allele model (T vs. G: OR = 1.159; 95% CI: 1.055–1.273; P = 0.002) and dominant model (TT + GT vs. GG: OR = 1.198; 95% CI: 1.048–1.370; P = 0.008). However, VDR rs739837 was not associated with GDM.
Conclusions
Significant associations were found between the VDR rs739837 polymorphism and T2DM susceptibility, but not with GDM.
Supplementary Information
The online version contains supplementary material available at 10.1186/s40001-022-00688-x.
Keywords: Type 2 diabetes mellitus, Gestational diabetes mellitus, Vitamin D receptor, rs739837, Susceptibility, Meta-analysis
Introduction
Diabetes mellitus (DM) is a common chronic disorder that includes type 1 DM (T1DM), type 2 DM (T2DM), gestational DM (GDM) and other types of DM with T2DM accounting for the majority of the DM cases [1]. T2DM is a disease of multifactorial etiologies caused by insulin resistance and impaired insulin secretion [2]. Increasing evidence indicates that genetic factors contribute to T2DM susceptibility [3]. Moreover, genetic variations associated with insulin resistance and β-cell dysfunction have been suggested to play key roles in the development of GDM [4, 5]. Studies have shown that women with a history of GDM are at an increased risk of developing T2DM [6–8], and women with a family history of diabetes may have an increased risk of GDM [9]. Thus, GDM may share similar genetic susceptibilities and risk factors with T2DM [10–12].
Vitamin D (Vit D) has important immunoregulatory immune characteristics. Supplementation with vitamin D has been shown to prevent the development of T2DM [13]. Conversely, the depletion of vitamin D may be involved in the etiology of T2DM by influencing insulin secretion [14, 15]. Increasing evidence shows that genetic variants of genes in the DM metabolic pathway may increase the risk of DM susceptibility, such as the vitamin D receptor (VDR) gene, which is located on human chromosome 12q13.11 and is primarily expressed in the pancreas [16]. Alterations in VDR activation and expression may increase fatty acid synthase expression and lipogenesis [17, 18], resulting in a reduction in lipolysis and lipid deposition [19]. In addition, abnormal VDR expression contributes to calcium signal-mediated lipid accumulation through the p38 MAPK pathway [20], which results in high lipid deposition in the liver, disturbs insulin signaling and causes β-cell dysfunction [21]. Abnormal VDR expression may play an important role in the development of T2DM.
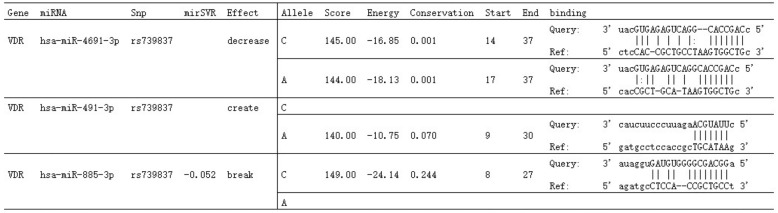
The rs739837 polymorphism is located in the 3′-untranslated region (UTR) of the VDR gene, and the rs739837 variation may affect VDR posttranscriptional regulation by binding with microRNA [22]. MiRNAs play an important role in the regulation of gene expression; thus, SNPs in the seed sites of miRNA targets may create or destroy miRNA-binding sites and further affect phenotypes and disease susceptibility [23]. We queried rs739837 polymorphism located in predicted miRNA target sites through the "MirSNP" database (http://bioinfo.bjmu.edu.cn/mirsnp/search/), which showed that rs739837 destroyed, created or disrupted putative miRNA target sites (Fig. 1). This polymorphism may affect the normal expression of VDR and further increase the risk of DM [24]. In recent years, several studies have shown associations of VDR rs739837 with T2DM [25–29] or GDM [30–32], but the results are controversial. Therefore, we conducted a meta-analysis to evaluate the association of VDR rs739837 with the risk of T2DM and GDM.
Fig. 1.
Effects of rs739837 on putative miRNA target sites
Materials and methods
The present meta-analysis was conducted according to Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) guidelines.
Literature search
The Google Scholar, PubMed and Chinese National Knowledge Infrastructure databases were systematically searched for relevant studies using the following terms:
(1) “VDR” or “vitamin D receptor” or “rs739837” or “polymorphism” or “type 2 diabetes mellitus” and “T2DM”; and (2) “VDR” or “vitamin D receptor” or “rs739837” or “polymorphism” and “gestational diabetes mellitus” and “GDM”.
The search was performed with no date or language restrictions. All the studies were evaluated by reading the title and abstract to exclude irrelevant studies. The full texts of eligible studies were then assessed by reading the full text to confirm inclusion in the study.
Inclusion and exclusion criteria
The inclusion criteria were as follows: (1) case–control/cohort studies; (2) studies that evaluated the association between VDR SNP rs739837 and T2DM/GDM; (3) adequate raw data or sufficient data to calculate odds ratios (ORs) with corresponding 95% confidence intervals (CIs); (4) a T2DM diagnosis based on the clinical criteria of the World Health Organization; and (5) a GDM diagnosis based on the clinical criteria of the World Health Organization.
The exclusion criteria were as follows: (1) not a case–control/cohort study; (2) not related to VDR SNP rs739837 and T2DM/GDM; (3) insufficient data; and (4) non-diabetic mellitus (NDM) subject data not in Hardy—Weinberg equilibrium (HWE).
Data extraction
Two authors independently extracted the following data from the included studies:
first author; origin; year of publication; type of DM; numbers of T2DM/GDM patients and NDM controls; distribution of alleles and genotypes; and ORs with 95% CIs of the allele distribution.
Statistical analysis
The following five genetic models were evaluated for rs739837: dominant model (TT + GT vs. GG), recessive model (TT vs. GG + GT), homozygote model (TT vs. GG), heterozygote model (GT vs. GG) and allele model (T vs. G). Genetic heterogeneity was estimated using the Q-test and I2 test. Lower heterogeneity was defined as I2 < 50% and P > 0.01 when using the fixed effects model (Mantel–Haenszel) to calculate ORs with corresponding 95% CIs. Otherwise, the random effects model (Mantel–Haenszel) was used [33, 34]. The significance of the ORs was evaluated using the Z test. Begg’s and Egger’s tests were used to determine publication bias. STATA v.14.0 software (Stata Corporation, TX, USA) was used to perform all statistical analyses.
Results
Study inclusion and characteristics
A total of 89 studies were searched using the inclusion and exclusion criteria. Figure 2 shows a flowchart of the study selection process. The following 9 eligible studies were included in the final analysis: 5 arteticles, which included 6 studies related to VDR SNP rs739837 and T2DM (one study only had allele mode data); and 3 articles, which included 3 studies related to VDR SNP rs739837 and GDM. The characteristics of each included study are shown in Table 1.
Fig. 2.
Flow diagram of study selection in the study
Table 1.
Characteristics of each study included in this meta-analysis
| Author | Year | Origin | Type | Cases/controls n | ORs with 95% CI (T vs G) | Allele distribution | Genotype distribution | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Cases, n | Controls, n | Cases, n | Controls, n | |||||||||||||
| G | T | G | T | GG | GT | TT | GG | GT | TT | HWE(P) | ||||||
| Zhang et al. | 2021 | Chinese (Henan) | T2DM | 324/1687 | 1.238 (1.028–1.492) | 459 | 189 | 2532 | 842 | 163 | 133 | 28 | 957 | 618 | 112 | 0.367 |
| Yu et al. | 2017 | Chinese (Han) | T2DM | 397/775 | 1.257(1.037–1.523) | 565 | 229 | 1172 | 378 | 202 | 161 | 34 | 448 | 276 | 51 | 0.339 |
| Lin et al. | 2016 | Chinese (Neimenggu) | T2DM | 319/387 | 0.987 (0.779–1.251) | 469 | 169 | 567 | 207 | 171 | 127 | 21 | 209 | 149 | 29 | 0.699 |
| Vimaleswaran et al. | 2014 | British | T2DM | – | 1.162 (0.937–1.441) | – | – | – | – | – | – | – | – | – | – | > 0.57 |
| Xu et al. | 2014 | Chinese (Ningxia Hui population) | T2DM | 154/115 | 1.089 (0.752–1.577) | 210 | 98 | 161 | 69 | 69 | 72 | 13 | 54 | 53 | 8 | > 0.05 |
| Xu et al. | 2014 | Chinese (Ningxia Han population) | T2DM | 201/148 | 1.066 (0.778–1.461) | 259 | 143 | 195 | 101 | 93 | 73 | 35 | 70 | 55 | 23 | > 0.05 |
| Chen et al. | 2021 | Chinese (Guangdong) | GDM | 555/646 | 0.953 (0.801–1.134) | 776 | 334 | 890 | 402 | 281 | 214 | 60 | 313 | 264 | 69 | 0.236 |
| Liu et al. | 2021 | Chinese (Hubei) | GDM | 816/851 | 1.058 (0.910–1.229) | 1152 | 480 | 1221 | 481 | 414 | 324 | 78 | 447 | 327 | 77 | > 0.05 |
| Wang et al. | 2015 | Chinese (Beijing) | GDM | 657/772 | 1.047 (0.889–1.232) | 935 | 379 | 1113 | 431 | 334 | 267 | 56 | 401 | 311 | 60 | 0.874 |
n number, T2DM type 2 diabetes mellitus, GDM gestational diabetes mellitus, OR odds ratio, CI confidence interval, HWE Hardy–Weinberg equilibrium, (-) not applicable
Heterogeneity analysis
Overall heterogeneity analysis
Low heterogeneity among studies was detected in the allele model (T vs. G: I2 = 0.0%; P = 0.472) [25–32], homozygote model (TT vs. GG: I2 = 0.0%; P = 0.787), heterozygote model (GT vs. GG: I2 = 0.0%; P = 0.996) and dominant model (TT + GT vs. GG: I2 = 1.5%; P = 0.418). High heterogeneity was detected in the recessive model (TT vs. GG + GT: I2 = 82.9%; P < 0.001) [25, 26, 28–32] (Fig. 3).
Fig. 3.
The overall meta-analysis for the association between VDR rs739837 and T2DM or GDM susceptibility. A Allele model: T vs. G (fixed effects model). B Homozygote model: TT vs. GG (fixed effects model). C Heterozygote model: GT vs. GG (fixed effects model). D Recessive model: TT vs. GG + GT (random effects model). E Dominant model, TT + GT vs. GG (fixed effects model). OR odds ratio, CI confidence interval, I2: measurement to quantify the degree of heterogeneity in meta-analyses
Subgroup heterogeneity analysis
In the T2DM subgroup, low heterogeneity among studies was detected in the allele model (T vs. G: I2 = 0.0%; P = 0.652) [25–29], homozygote model (TT vs. GG: I2 = 0.0%; P = 0.675), heterozygote model (GT vs. GG: I2 = 0.0%; P = 0.936) and dominant model (TT + GT vs. GG: I2 = 0.0%; P = 0.595). High heterogeneity was detected in the recessive model (TT vs. GG + GT: I2 = 86.6%; P < 0.001) [25, 26, 28, 29] (Fig. 4).
Fig. 4.
Subgroup meta-analysis for the association between VDR rs739837 and T2DM or GDM susceptibility. A Allele model: T vs. G (fixed effects model). B Homozygote model: TT vs. GG (fixed effects model). C Heterozygote model: GT vs. GG (fixed effects model). D Recessive model (T2DM): TT vs. GG + GT (random effects model). E Recessive model (GDM): TT vs. GG + GT (fixed effects model). F Dominant model, TT + GT vs. GG (fixed effects model). OR odds ratio, CI confidence interval, I2: measurement to quantify the degree of heterogeneity in meta-analyses
In the GDM subgroup, low heterogeneity among studies was detected in the allele model (T vs. G: I2 = 0.0%; P = 0.635), homozygote model (TT vs. GG: I2 = 0.0%; P = 0.850), heterozygote model (GT vs. GG: I2 = 0.0%; P = 0.971), recessive model (TT vs. GG + GT: I2 = 0.0%; P = 0.829) and dominant model (TT + GT vs. GG: I2 = 0.0%; P = 0.553) [30–32] (Fig. 4).
Overall meta-analysis results
In the overall analysis, a fixed effects model was used to analyze the allele, homozygote, heterozygote and dominant models. VDR rs739837 was shown to be significantly associated with increased DM (T2DM and GDM) risk in the allele model (T vs. G: OR = 1.088; 95% CI: 1.018–1.163; P = 0.012) and dominant model (TT + GT vs. GG: OR = 1.095; 95% CI: 1.001–1.197; P = 0.047). No significant associations were found under the homozygote model (TT vs. GG: OR = 1.144; 95% CI: 0.973–1.346; P = 0.103) and heterozygote model (GT vs. GG: OR = 1.073; 95% CI: 0.909–1.266; P = 0.406). A random effects model indicated no significant difference for the recessive model (TT vs. GG + GT: OR = 0.764; 95% CI: 0.517–1.129; P = 0.177) (Fig. 3).
Subgroup meta-analysis results
We performed subgroup analysis according to the type of DM to evaluate the association between VDR rs739837 and T2DM or GDM susceptibility.
In the T2DM subgroup, the results showed that rs739837 was significantly related to an increased risk of T2DM in the allele model (T vs. G: OR = 1.159; 95% CI: 1.055–1.273; P = 0.002) and dominant model (TT + GT vs. GG: OR = 1.198; 95% CI: 1.048–1.370; P = 0.008) using a fixed effects model. No significant associations were found under the homozygote model (TT vs. GG: OR = 1.273; 95% CI: 0.992–1.633; P = 0.058) or heterozygote model (GT vs. GG: OR = 1.094; 95% CI: 0.849–1.410; P = 0.486) using a fixed effects model. A random effects model also showed that no significant difference was found for the recessive model (TT vs. GG + GT: OR = 0.269; 95% CI: 0.684–0.349; P = 0.269) (Fig. 4).
In the GDM subgroup, no significant associations were found under the allele model (T vs. G: OR = 1.023; 95% CI: 0.932–1.123; P = 0.631), homozygote model (TT vs. GG: OR = 1.060; 95% CI: 0.857–1.313; P = 0.590), heterozygote model (GT vs. GG: OR = 1.057; 95% CI: 0.850–1.315; P = 0.618), recessive model (TT vs. GG + GT: OR = 0.931; 95% CI: 0.758–1.143; P = 0.493) or dominant model (TT + GT vs. GG: OR = 1.018; 95% CI: 0.903–1.148; P = 0.765) using a fixed effects model (Fig. 4).
Publication bias
According to Begg’s and Egger’s tests, no significant publication bias was found in any of the genetic models (all P > 0.05, Additional file 1: Tables S1–S3), and the funnel plots are shown in Figs. 5, 6.
Fig. 5.
Funnel plot of the odds ratios in the overall meta-analysis. A Allele model: T vs. G. B Homozygote model: TT vs. GG. C Heterozygote model: GT vs. GG. D Recessive model, TT vs. GG + GT. E Dominant model: TT + GT vs. GG
Fig. 6.
Funnel plot of the odds ratios in the subgroup meta-analysis. A Allele model: T vs. G. B Homozygote model: TT vs. GG. C Heterozygote model: GT vs. GG. D Recessive model, TT vs. GG + GT. E Dominant model: TT + GT vs. GG
Discussion
In this systematic review and meta-analysis study, we performed a systematic and objective assessment of the associations between the VDR rs739837 polymorphism and DM. The findings of the meta-analysis of 9 case–control studies in the overall type of DM determined a significant association of the T allele and TT + GT genotype with DM risk. Moreover, the subgroup analysis also revealed that rs739837 was significantly related to an increased risk of T2DM in the T allele and TT + GT genotype, but no significant associations were found under any models in the GDM subgroup.
The VDR gene has been confirmed to be significantly involved in the regulation of the endocrine system, suggesting that it is a potential candidate gene for metabolic disorders. Rs739837 is located in the 3′-untranslated region (UTR) of the VDR gene, which regulates gene expression. A series of investigations have reported that the rs739837 SNP is associated with diabetes risk. Zhang et al. demonstrated a significant association between the T allele and TT + GT genotype of rs739837 and T2DM risk [25]. Yu et al. found a significant relationship between the rs739837 polymorphism and T2DM in the T allele, recessive model (GG/GT + TT) and additive model (GG/TT) [26]. A previous study has identified that the rs739837 genotype distributions show significant differences across T2DM cases and controls [28]. Interestingly, Jia et al. found that the T and C allele frequencies of rs739837 are 70.2% and 29.8% in cases, respectively, and 73 and 27%, in controls, respectively. The control group results reported by Jia et al. were inconsistent with other reports; they reported that rs739837 is significantly associated with an increased risk of T2DM in the additive model (TT vs. TC vs. CC) and dominant (TT vs. TC/CC) model [35]. Due to the differences in allele frequencies from other reports, the study by Jia et al. was not included in the meta-analysis. Vimaleswaran and Lin showed that rs739837 is not associated with T2DM risk [27, 29]. Moreover, four studies demonstrated no relationship between the genotypic model of rs739837 and GD [30–32, 36]. A previous meta-analysis has suggested that women with a history of GDM are almost 10 times more likely to develop T2DM than those with a normoglycemic pregnancy [7]. The magnitude of this risk is consistent with evidence that T2DM and GDM share common pathogenic mechanisms and risk factors.
Interestingly, in the overall analysis, low heterogeneity among studies was detected in the allele, dominant, homozygote and heterozygote genetic models, and high heterogeneity among studies was detected under the recessive model as well as in the subgroup analysis. Although the GDM studies only contained women, there was no significant heterogeneity in the meta-analysis with T2DM research involving men and women, except for the recessive model. The subtype analysis revealed that the T2DM group had higher heterogeneity, while the GDM group had lower heterogeneity. Therefore, it was possible to combine T2DM and GDM for meta-analysis to explore potential common susceptibility factors. However, due to the small GDM sample size, there was no powerful conclusion of the results.
There were several limitations in the present meta-analysis. First, there were limited studies that estimated VDR rs739837 and T2DM or GDM risk. In particular, few articles have researched the association between VDR rs739837 and GDM. Only three articles contained data from five genetic models, and one article did not have available data for meta-analysis, which may affect the overall estimation. Moreover, the present study included only Chinese studies. Thus, studies using larger sample sizes of other ethnic groups worldwide need to be performed. Finally, the present study only evaluated the association between rs739837 genotypes and T2DM or GDM risk without adjusting the effects of other risk factors, such as interacting gene–gene and gene–environment factors [37]. Therefore, further study is required to evaluate the susceptibility factors of T2DM or GDM.
Conclusions
To our knowledge, this study is the first to assess the role of VDR rs739837 and T2DM or GDM risk. Significant associations were found between the VDR rs739837 polymorphism and T2DM susceptibility but not association with GDM.
Supplementary Information
Additional file 1: Table S1 Begg’s and Egger’s tests in overall analysis. Table S2 Begg’s and Egger’s tests in T2DM subgroup analysis. Table S3 Begg’s and Egger’s tests in GDM subgroup analysis.
Acknowledgements
Not applicable.
Author contributions
QZ, DZ and YW contributed equally to this work. QZ, DZ and YW wrote the main manuscript text and prepared all table and figures. QZ and DZ managed the literature searches and analyses. The study was supervised by Y-OY, ZL and RG. All authors reviewed the manuscript. All authors read and approved the final manuscript.
Funding
Support for this work includes funding from the National Natural Science Foundation of China (81873649); Doctoral scientific research Initiate funding project of Shunde Women and Children’s Hospital of Guangdong Medical University (Maternity & Child Healthcare Hospital of Shunde Foshan) (2020BSQD007); Guangdong Medical University Research Foundation (GDMUM2020008 and GDMUM2020012); Medical Research Project of Foshan Health Bureau (20210188 and 20210289).
Availability of data and materials
All data used or generated during the study are available from the corresponding author on reasonable request: Runmin Guo, E-mail: 1314ivu@126.com.
Declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Qiaoli Zeng, Dehua Zou and Yue Wei contributed equally to this work
Contributor Information
Yingguang Ouyang, Email: 178512944@qq.com.
Zhaohang Lao, Email: xilinold@sohu.com.
Runmin Guo, Email: 1314ivu@126.com.
References
- 1.Kautzky Willer A, Harreiter J, Pacini G. Sex and gender differences in risk pathophysiology and complications of type diabetes mellitus. Endocr Rev. 2016;37(3):278–316. doi: 10.1210/er.2015-1137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Banerjee M, Vats P. Reactive metabolites and antioxidant gene polymorphisms in type 2 diabetes mellitus. Indian J Hum Genet. 2014;20(1):10–19. doi: 10.4103/0971-6866.132747. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Tian Y, Xu J, Huang T, et al. A novel polymorphism (rs35612982) in CDKAL1 is a risk factor of type 2 diabetes: a case-control study. Kidney Blood Press Res. 2019;44(6):1313–1326. doi: 10.1159/000503175. [DOI] [PubMed] [Google Scholar]
- 4.Kwak SH, Kim SH, Cho YM, et al. A genome-wide association study of gestational diabetes mellitus in Korean women. Diabetes. 2012;61(2):531–541. doi: 10.2337/db11-1034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Wu L, Cui WH, Tam, , et al. Genetic variants associated with gestational diabetes mellitus: a meta-analysis and subgroup analysis. Sci Rep. 2016;6:30539. doi: 10.1038/srep30539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Bellamy L, Casas JP, Hingorani AD, et al. Type 2 diabetes mellitus after gestational diabetes: a systematic review and meta-analysis. Lancet. 2009;373(9677):1773–1779. doi: 10.1016/S0140-6736(09)60731-5. [DOI] [PubMed] [Google Scholar]
- 7.Vounzoulaki E, Khunti K, Abner SC, et al. Progression to type 2 diabetes in women with a known history of gestational diabetes systematic review and meta-analysis. BMJ (Clin Res Ed) 2020;369:m1361. doi: 10.1136/bmj.m1361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.You H, Hu J, Liu Y, et al. Risk of type 2 diabetes mellitus after gestational diabetes mellitus: a systematic review & meta-analysis. Indian J Med Res. 2021;154(1):62–77. doi: 10.4103/ijmr.IJMR_852_18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Williams MA, Qiu C, Dempsey JC, et al. Familial aggregation of type 2 diabetes and chronic hypertension in women with gestational diabetes mellitus. J Reprod Med. 2003;48(12):955–962. [PubMed] [Google Scholar]
- 10.Mao H, Li Q, Gao S. Meta-analysis of the relationship between common type 2 diabetes risk gene variants with gestational diabetes mellitus. PLoS ONE. 2012;7(9):e45882. doi: 10.1371/journal.pone.0045882. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Rhee SY, Kim JY, Woo JT, et al. Familial clustering of type 2 diabetes in Korean women with gestational diabetes mellitus. Korean J Intern Med. 2010;25(3):269–272. doi: 10.3904/kjim.2010.25.3.269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Pu J, Zhao B, Wang EJ, et al. Racial/Ethnic differences in gestational diabetes prevalence and contribution of common risk factors. Paediatr Perinat Epidemiol. 2015;29(5):436–443. doi: 10.1111/ppe.12209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.de Boer IH, Tinker LF, Connelly S, et al. Calcium plus vitamin D supplementation and the risk of incident diabetes in the women's health initiative. Diabetes Care. 2008;31(4):701–707. doi: 10.2337/dc07-1829. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Müller K, Bendtzen K. Inhibition of human T lymphocyte proliferation and cytokine production by 1,25-dihydroxyvitamin D3. differential effects on CD45RA+ and CD45R0+ cells. Autoimmunity. 1992;14(1):37–43. doi: 10.3109/08916939309077355. [DOI] [PubMed] [Google Scholar]
- 15.Palomer X, González-Clemente JM, Blanco-Vaca F, et al. Role of vitamin D in the pathogenesis of type 2 diabetes mellitus obesity metabolism. Diabetes Obes Metab. 2008;10(3):185–197. doi: 10.1111/j.1463-1326.2007.00710.x. [DOI] [PubMed] [Google Scholar]
- 16.Johnson JA, Grande JP, Roche PC, et al. Immunohistochemical localization of the 1,25(OH)2D3 receptor and calbindin D28k in human and rat pancreas. Am J Physiol. 1994;267(3 Pt 1):E356–E360. doi: 10.1152/ajpendo.1994.267.3.E356. [DOI] [PubMed] [Google Scholar]
- 17.McCann LM, Beto J. Roles of calcium-sensing receptor and vitamin d receptor in the pathophysiology of secondary hyperparathyroidism. J Ren Nutr. 2010;20(3):141–150. doi: 10.1053/j.jrn.2010.01.004. [DOI] [PubMed] [Google Scholar]
- 18.Zemel MB. Regulation of adiposity and obesity risk by dietary calcium: mechanisms and implications. J Am Coll Nutr. 2002;21(2):146S–151S. doi: 10.1080/07315724.2002.10719212. [DOI] [PubMed] [Google Scholar]
- 19.Duncan RE, Ahmadian M, Jaworski K, et al. Regulation of lipolysis in adipocytes. Annu Rev Nutr. 2007;27:79–101. doi: 10.1146/annurev.nutr.27.061406.093734. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Sun C, Qi R, Wang L, et al. p38 MAPK regulates calcium signal-mediated lipid accumulation through changing VDR expression in primary preadipocytes of mice. Mol Biol Rep. 2012;39(3):3179–3184. doi: 10.1007/s11033-011-1084-8. [DOI] [PubMed] [Google Scholar]
- 21.Sonnenberg J, Luine VN, Krey LC, et al. 1,25-Dihydroxyvitamin D3 treatment results in increased choline acetyltransferase activity in specific brain nuclei. Endocrinology. 1986;118(4):1433–1439. doi: 10.1210/endo-118-4-1433. [DOI] [PubMed] [Google Scholar]
- 22.Bartel DP. MicroRNAs: genomics, biogenesis, mechanism, and function. Cell. 2004;116(2):281–297. doi: 10.1016/S0092-8674(04)00045-5. [DOI] [PubMed] [Google Scholar]
- 23.Ryan BM, Robles AI, Harris CC. Genetic variation in microRNA networks: the implications for cancer research. Nat Rev Cancer. 2010;10(6):389–402. doi: 10.1038/nrc2867. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Saccone D, Asani F, Bornman L. Regulation of the vitamin D receptor gene by environment, genetics and epigenetics. Gene. 2015;561(2):171–180. doi: 10.1016/j.gene.2015.02.024. [DOI] [PubMed] [Google Scholar]
- 25.Zhang D, Cheng C, Wang Y, et al. The influence of VDR polymorphisms on the type 2 diabetes susceptibility in Chinese: an interaction with hypertriglyceridemia. Mol Genet Genomics. 2021;296(4):837–844. doi: 10.1007/s00438-021-01784-z. [DOI] [PubMed] [Google Scholar]
- 26.Yu C, WangWang L, et al. Study and evaluation the impact of vitamin D receptor variants on the risk of type 2 diabetes mellitus in Han Chinese. J Diabetes. 2017;9(3):275–284. doi: 10.1111/1753-0407.12413. [DOI] [PubMed] [Google Scholar]
- 27.Vimaleswaran KS, Power C, Hyppönen E. Interaction between vitamin D receptor gene polymorphisms and 25-hydroxyvitamin D concentrations on metabolic and cardiovascular disease outcomes. Diabetes Metab. 2014;40(5):386–389. doi: 10.1016/j.diabet.2014.01.003. [DOI] [PubMed] [Google Scholar]
- 28.Xu JR, Yang Y, Liu XM, et al. Association of VDR polymorphisms with type 2 diabetes mellitus in Chinese Han and Hui populations. Genet Mol Res. 2014;13(4):9588–9598. doi: 10.4238/2014.November.14.2. [DOI] [PubMed] [Google Scholar]
- 29.XH Lin Study the relationship of clinical index and gene polymorphisms with the diabetic retinopathy Doctoral Dissertation of Chongqing Medical University (in Chinese)
- 30.Liu J, Dai Q, Li W, et al. Association of vitamin D receptor gene polymorphisms with gestational diabetes mellitus-a case control study in Wuhan China. BMC Pregnancy Childbirth. 2021;21(1):142. doi: 10.1186/s12884-021-03621-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Wang Y, Wang O, Li W, et al. Variants in vitamin D binding protein gene are associated with gestational diabetes mellitus. Medicine. 2015;94(40):e1693. doi: 10.1097/MD.0000000000001693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Chen H, XIAO M, SUN J, et al. Association of polymorphisms in vitamin D receptor gene with gestational diabetes mellitus. J Practical Med. 2021;37(22):2866–2870. [Google Scholar]
- 33.Zeng Q, Zou D, Zeng Q, et al. Association between insulin-like growth factor-1 rs35767 polymorphism and type 2 diabetes mellitus susceptibility: a meta-analysis. Front Genet. 2021;12:774489. doi: 10.3389/fgene.2021.774489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Han Q, Geng W, Zhang D, et al. ADIPOQ rs2241766 gene polymorphism and predisposition to diabetic kidney disease. J Diabetes Res. 2020;2020:5158497. doi: 10.1155/2020/5158497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Jia J, Ding H, Yang K, et al. Vitamin D receptor genetic polymorphism is significantly associated with risk of type 2 diabetes mellitus in Chinese Han population. Arch Med Res. 2015;46(7):572–579. doi: 10.1016/j.arcmed.2015.09.006. [DOI] [PubMed] [Google Scholar]
- 36.Shi A, Wen J, Liu G, et al. Genetic variants in vitamin D signaling pathways and risk of gestational diabetes mellitus. Oncotarget. 2016;7(42):67788–67795. doi: 10.18632/oncotarget.11984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Wu Y, Ding Y, Tanaka Y, et al. Risk factors contributing to type 2 diabetes and recent advances in the treatment and prevention. Int J Med Sci. 2014;11(11):1185–1200. doi: 10.7150/ijms.10001. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Additional file 1: Table S1 Begg’s and Egger’s tests in overall analysis. Table S2 Begg’s and Egger’s tests in T2DM subgroup analysis. Table S3 Begg’s and Egger’s tests in GDM subgroup analysis.
Data Availability Statement
All data used or generated during the study are available from the corresponding author on reasonable request: Runmin Guo, E-mail: 1314ivu@126.com.