Abstract
Zoonotic diseases are estimated to constitute 75% of all emerging infectious diseases, of which more than 70% come from wild species. The potential threat of zoonotic spillover from the consumption of wildmeat has been the subject of policy and media attention, especially in the context of the COVID-19 pandemic; however, little is known about the actual conditions that contribute to the risk of spillover and associated disease transmission. In this Review, we compile existing evidence from available literature on the conditions of spillover associated with wildmeat consumption, including the types of wild animal and disease, modes of transmission, and the conditions in which spillover is thought to have occurred. We suggest that stronger understanding of the context of spillover from wildmeat is needed to enable more targeted and effective policy responses that reduce the risk of future pandemics of zoonotic origin. Such interventions could also lead to the avoidance of unintended adverse consequences for human communities that rely on wild produce, including wildmeat, as sources of dietary protein, fat, and micronutrients.
Introduction
In December, 2019, a cluster of unusual pneumonia cases was detected in Wuhan, China, and was traced to the Huanan seafood market. The market was quickly shut down; however, COVID-19 spread across almost every country in the world in the ensuing months. Although the seafood market has not been substantiated as the origin of COVID-19,1, 2 the pandemic has prompted demands for immediate and comprehensive restrictions on the consumption and trading of wild animals. Bans on the trade and consumption of wild animals have since been enacted in several Asian countries and regions, notably in China and Vietnam. However, such responses are blunt policy instruments, and are not grounded in locally situated understandings of how and why zoonotic spillover might have occurred.
Zoonotic diseases are diseases that normally exist in (non-human) vertebrates but can jump to infect humans during spillover events. Zoonotic spillover has been documented as originating from (and involving as intermediate species) various domesticated and non-domesticated wild animals. Transmission from animal to human can be airborne, vector-borne, through food, or through close or direct contact of humans with animals or their blood, faeces, or saliva.3 Around 75% of emerging infectious diseases are estimated to be of zoonotic origin (or to have transmission pathways involving animals),4 with animals in the wildlife trade hosting approximately three-quarters of all known zoonotic viruses and associated diseases, not all of which are harmful to humans.5
Spillover requires infectious pathogens to overcome a succession of several disease-related, human, animal, and environmental barriers (figure 1 ) that lead to the coexistence of pathogen, animal reservoir, and a susceptible human population. With intensification and new anthropogenic pressures bringing animals and humans into ever-closer contact, the dynamics of spillover are changing. The rate at which diseases are emerging is accelerating, with more than five new diseases emerging every year7 as land-use changes, land-management practices, climate change, and other drivers shift the distribution of pathogens, wild animal hosts, and human populations. By understanding the conditions that have historically given rise to spillover, we might be better placed to intervene to mitigate both the risk of spillover occurring and the consequences in communities that might be more vulnerable to its effects.
Figure 1.
Barriers to the spillover process
Barriers occur in reservoir hosts (green), the environment and vectors (blue), and spillover hosts (orange). Adapted from Sokolow and colleagues.6
Because SARS-CoV-2, the causative agent of COVID-19, is thought to have originated in bats and the Wuhan wet market was posited as an early source of infection, questions continue to be raised around the safe consumption of wild animals, with widespread calls for total bans on their consumption and trade.8, 9 The biodiversity conservation community has responded to the pandemic by adding a public health narrative to its long-standing concerns around the effects of unsustainable consumption of wildmeat, excessive wildlife harvesting, and both legal and illegal wildlife trades.8, 10 Immediately after the emergence of COVID-19 in humans, more than 200 wildlife non-governmental organisations signed an open letter to WHO, calling on the organisation to encourage a highly precautionary approach to the wildlife trade among its member states, including enacting permanent bans on all live wildlife markets and prohibiting the use of wild animal products in traditional medicine. Temporary and permanent bans on wildlife trade and consumption have previously been enacted in response to outbreaks of zoonotic disease. These measures include restrictions at local scales (eg, specific live bird markets were banned in response to H5N1 in Northern Vietnam in 2013)11 and at national or regional scales (eg, the west African bushmeat ban in response to the outbreak of Ebola virus disease in 2013–16).12 Other bans have been imposed on particular species that are deemed to pose a high risk of particular diseases—for example, a ban on pangolin and bat consumption in Ghana in response to COVID-19.13
Key messages.
-
•
The potential threat of zoonotic spillover from the consumption of wildmeat has been the subject of much recent policy and media attention; however, little is known about the actual conditions that contribute to the risk of spillover through wildmeat consumption
-
•
Blanket bans on wildmeat consumption are blunt policy instruments that could have unintended consequences, such as pushing harvesting and consumption practices underground and heightening food insecurity in some rural and indigenous peoples’ communities
-
•
This Review highlights the diverse conditions that contribute to spillover risk relating to wildmeat consumption, as well as substantial knowledge gaps, and suggests the need for more nuanced approaches to protect an increasingly globalised and interconnected world from the potential risks of zoonotic spillover while not stigmatising the consumption practices and livelihoods of communities that rely on wild resources
-
•
As the dynamics of disease emergence are changing, there is need for increased global (pre-spillover) surveillance measures, including identification and monitoring of potential spillover hotspots, and ongoing, dynamic risk assessment of high-risk settings in which human and animal populations are in close proximity
-
•
Because knowledge about zoonotic disease is often poor and is variable across sites, interventions that work with local communities to improve understanding of high-risk and low-risk species, practices, and situations could help to reduce spillover risk from the consumption of both domesticated meat and wildmeat
-
•
Targeted interventions to reduce spillover risk from both domesticated meat and wildmeat consumption practices might be needed within particular regions or at particular times of the year; however, these measures must be developed collaboratively with local communities to mitigate disbenefits and improve legitimacy
By reducing contact rates between humans and high-risk animal species, preventing wild animal hunting should decrease the transmission of zoonotic diseases to humans. However, there are concerns that the risks of spillover from wildmeat consumption might be overemphasised in these immediate responses from policy makers and conservation organisations.14 Calls for total bans on the trade and consumption of wild animals and wildmeat often fail to appreciate the parallel risks from commercialised food systems and trade of domesticated livestock in poorly regulated conditions, and so provide cover for inaction on policies that could potentially result in more meaningful reductions in the risk of future pandemics. Estimates suggest that only 3% of emerging infectious diseases, between 1940 and 2004, were attributable to bushmeat consumption, compared with 17% from conventional agriculture and food industry change, and 31% from land-use change, including deforestation, habitat fragmentation, agricultural development, irrigation, and urbanisation.3, 15, 16
Calls for blanket bans on wild animal consumption and trade also overlook the detrimental effects of such bans on some human populations.17 Many rural communities and indigenous peoples rely on wild produce—particularly meat, fish, and insects—as important sources of dietary protein, fat, and micronutrients.18, 19, 20 Such communities are often already marginalised and have poorer health and nutritional outcomes than do non-indigenous, urban-dwelling communities.21 For people who live in urban areas, although alternative (non-wild) meat sources might be available, research has shown the cultural importance of wildmeat, indicating freshness and luxuriousness of produce.22 Disallowing the consumption and trade of wildmeat could threaten the continued food and nutritional security of communities that depend on it, and undermine the cultural importance of these consumption practices.
Furthermore, as has been evident in places where wildlife harvesting has been restricted, such bans could push wildlife consumption and trade underground, making the regulation of safe handling practices more difficult. For example, a 2013–16 bushmeat ban in response to the outbreak of Ebola virus disease seemed to encourage undercover hunting and trade in parts of west Africa.11 Although opposition to wildlife trade bans comes from diverse actors and sectors, including many with their own vested interests (eg, wildlife traders and wildlife farm owners),23 ensuring that public discourse and policy action is appropriately informed by the best available knowledge of the diverse conditions that lead to spillover remains important, to avoid adverse consequences of well intentioned but poorly informed policies.
Despite the widespread media and policy attention given to practices of wildmeat consumption, and a strong understanding of pathogenic prevalence within wild animals, research exploring the conditions for zoonotic spillover associated with wildmeat consumption has not been consolidated. In this Review, we seek to address this deficit in knowledge by identifying available evidence for reported spillover events that have been linked strongly to wildmeat consumption or consumption-related exposures between 1940 and 2021.
We extracted relevant data on the types of wild animal and disease, modes of transmission, and environments in which spillover is thought to have occurred. These links are often difficult to establish, and diagnostic analyses in animals, humans, or both can be scarce or absent, which limits the quality of the evidence. For the larger outbreaks identified, we qualitatively characterised the conditions for the initial take-off of human-to-human transmission. Although spillovers can be frequent, take-off within human populations is comparatively rare. Understanding the factors that have historically contributed to the take-off of zoonotic disease within human populations can support evidence-based policy to mitigate the occurrence of serious disease outbreaks in a world that is increasingly interconnected and globalised.
By examining available evidence of the conditions for spillover from wildmeat consumption, we discuss how such understanding could inform discussions about interventions that could offer more targeted approaches to mitigate spillover risks.
Zoonotic spillover linked to wildmeat consumption
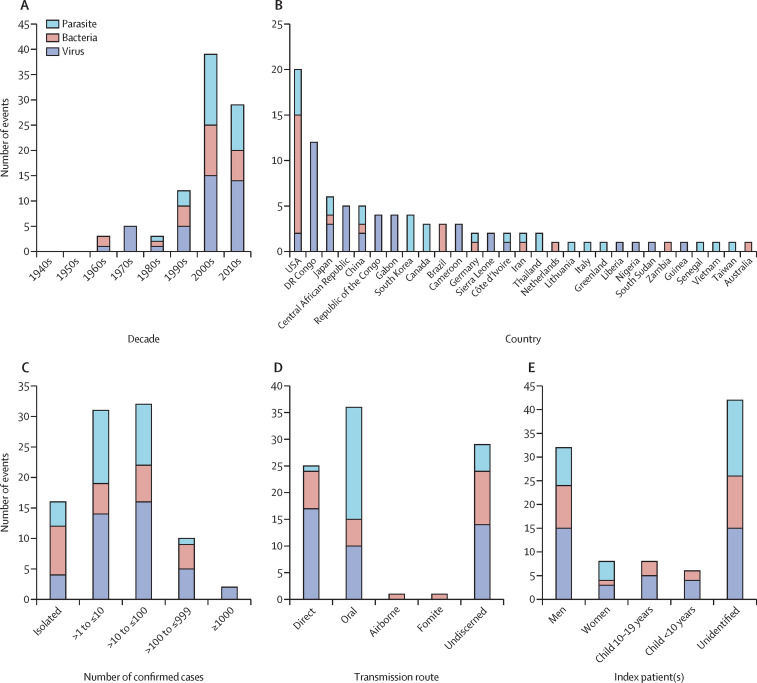
Zoonotic spillover linked to wildmeat consumption was assessed for 91 suspected spillover events of 25 zoonotic diseases (appendix pp 2–23). We identified 41 spillover events from ten viral pathogens, 23 events from ten bacterial pathogens, and 27 events from five different parasites. Owing to limitations in disease reporting—for example, difficulties in timely and accurate diagnoses, or in attributing disease to instances of wildmeat consumption—these events are very unlikely to represent the totality of spillover associated with wildmeat consumption. Furthermore, some of the studies retrieved could represent instances in which there were multiple disease introductions into humans rather than one independent event. Excluding HIV-1/SIVcpz, the earliest spillover event identified occurred in 1967, and the most recent was in 2018. We note that there have been more events reported since 2000 (n=68) than in the 60 years before 2000 (n=23; figure 2A ). This finding could reflect accelerating rates of spillover owing to increased interactions between humans and wild animals, or could simply result from temporal biases in reporting towards the present day due to enhanced diagnostic methods, surveillance, or renewed policy and research focus following the 2002–03 severe acute respiratory syndrome (SARS) pandemic and subsequent large-scale zoonotic outbreaks.
Figure 2.
Summary of spillover events
Events are reviewed by decade of reporting (A), country of reporting (B), size of resulting outbreak (C), transmission pathway (D), and suspected index case (E).
Spillover events associated with wildmeat consumption have been reported across all continents (excluding Antarctica), with the largest numbers in Africa (n=38), North America (n=23), and Asia (n=21), and lower frequencies in Europe (n=6), South America (n=3), and Oceania (n=1; figure 2B). Geographical differences in pathogen type can be identified: viral spillover was most frequently reported in Africa (n=35), bacterial spillover was most frequent in North America (n=13), and parasitic spillover was most frequent in Asia (n=13). Spillover locations mostly reflect known sites of zoonotic disease endemicity (within animal reservoirs and human populations).
Most spillover events identified did not lead to large-scale outbreaks (figure 2C). Ten events led to outbreaks of 100 or more confirmed cases, including two events of 1000 or more cases. 47 events resulted in ten or fewer confirmed cases, of which 16 events were isolated incidents.
Transmission from wild animals to humans
We summarised the identified transmission pathways (from wild animal host to human index patient) within the spillover events documented (figure 2D). Although transmission routes could not be discerned for a third of events, direct contact or oral transmission were considered to be the most likely transmission routes for most unknowns. Disentangling whether transmission occurred through direct transmission (contact with animal bodily fluids) during carcass handling, or through oral transmission during consumption was difficult, especially for cases in which other supporting evidence is scarce or absent. For cases in which identifying the transmission route was possible, oral transmission (through the ingestion of wildmeat) was most commonly associated with zoonotic spillover (36 events). Cooking methods were frequently alluded to in case reports, with the consumption of raw or undercooked meat often mentioned. For cases in which this information was provided, statistical analyses show strong evidence of the importance of cooking method—for example, an investigation of trichinellosis spillover in Thailand found no association between human infection and eating wild boar cooked after a recent hunt, but consumption of the same meat raw significantly increased (17-fold) the odds of infection.24
Direct contact with animal bodily fluids before consumption was reported in more than a quarter of spillover events identified. Qualitative description of spillover implicated practices associated with hunting (eg, skinning, butchering, and field dressing) as probable sources of zoonotic transmission. Statistical analyses identified that, in some cases, involvement in hunting or wildmeat preparation was associated with infection, and not ingestion itself. One study25 reported no association between eating hippopotamus and having anthrax (adjusted odds ratio [aOR] 2·0; 95% CI 0·5–1·1), but skinning the animal (12·0; 4·3–36·5) and cutting the animal (8·1; 2·2–29·2) were significantly associated with infection. Only two studies identified airborne26 or fomite27 transmission as a viable transmission route; these studies were deemed to provide poor evidence that these were the transmission route in the cases described.
Characterising wild animal reservoirs and human hosts
Demographic information on suspected index cases was available for 42 (fewer than half) of the 91 events. Of those known, men were most frequently reported to be the index case (n=32; figure 2E). The occupation of index cases was often noted, and included hunters (professional) and wildlife traders—occupations predominantly held by men. Age-related patterns were discernible for some diseases; for example, children younger than 18 years were reported as the index cases for seven (41%) of 17 monkeypox spillovers and, of these, four occurred in children younger than 10 years. For hepatitis E, index cases were all adults older than 40 years. Recreational activities that exposed index patients to wild animals were also documented, including recreational hunting27 and tourism.28
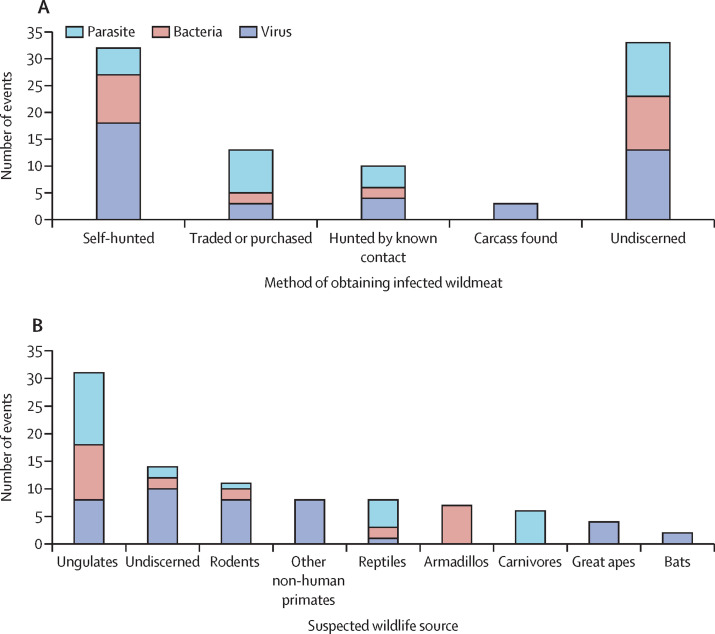
The source of wildmeat suspected as the animal reservoir was described in 58 (64%) of 91 events (figure 3A ). In 32 (53%) of these events, the wildmeat consumed had been hunted by the index case or cases; in a further ten events (12%), it was hunted by a known contact of the index case, such as a family member or neighbour. 12 events (27%) involved consumption of contaminated, purchased wildmeat.
Figure 3.
Wildmeat links to spillover effects
Events are listed by method of obtaining wildmeat (A) and suspected wildlife source of disease (B).
We identified various wild animal hosts of zoonotic disease (figure 3B). For cases in which the wildlife source was indicated, the consumption (or related activities) of non-human primates was most frequently associated with viral spillover (n=12), including Ebola virus, monkeypox virus, HLTV/STLV-1, and simian foamy virus. Rodents were suspected as wild animal hosts for plague,29 leptospirosis in the USA,30 and Lassa fever in Guinea.31 Wild boar was suspected as the source of brucellosis,32, 33 streptococcosis,34 hepatitis E,35, 36 trichinellosis,28, 37, 38, 39, 40, 41, 42, 43, 44 and sparganosis in Japan.45 Most events had animal pools that were consistent with existing understanding of disease endemicity; however, for some events, suspected wild animal hosts were inconsistent with known animal pools at the time,46 reinforcing the need to strengthen environmental surveillance of possible animal reservoirs.
Initial take-off in human populations
Six spillover events led to outbreaks of more than 100 suspected, probable, or confirmed cases of disease with human-to-human transmission. These events involved four well known viruses: HIV-1/SIVcpz,47, 48 Ebola virus,49, 50, 51 SARS,52 and monkeypox.53 We extracted details on the conditions that enabled initial rapid take-off in human-to-human transmission.
Introductions of HIV-1/SIVcpz into human populations are thought to have occurred many times before the 20th century; however, it was not until the early 1900s that conditions were favourable for the take-off of the disease as an epidemic. A combination of factors has been proposed as to why conditions might have been favourable at this time,47 including forced labour during construction of the Congo–Océan railway, urbanisation, improved road and rail networks supporting constant population movement, sex work, and other sociobehavioural changes that are still not fully understood.54
In a 2013 monkeypox outbreak in the Democratic Republic of the Congo, high rates of intrahousehold transmission were reported (median secondary attack rate 50%), with 104 possible cases across 16 households.55 Transmission occurs through respiratory droplets or contact with pox lesions. How interhousehold spread occurred is not certain, although a later study56 reported that school-aged males were most likely to be household index cases. Differences in intrahousehold transmission were noted between households with a previous history of smallpox vaccination and those without.
In the 2003 outbreak of SARS in China, the reason for the initial rapid surge in infections and extensive transmission chains in Guangdong province was unclear, because other regions with cases did not see similar trajectories. The analysis by Xu and colleagues52 of the characteristics of early-onset and late-onset cases supported nosocomial transmission as a reason for the surge. Approximately a third of early cases were seen in health-care workers (32% with onset in January, 2003), a proportion that declined as the epidemic progressed (27% in February and 18% in March). The surge in cases in January also coincided with the admission to hospital of a highly infectious index patient, who transmitted SARS to health-care workers at three different hospitals in Guangzhou city and several family members—a trigger that was absent in other regions.
Immediate outbreaks of Ebola virus disease have been associated with nosocomial transmission; this was the reason for the initial rapid spread of the 1976 outbreak in Zaire (now Democratic Republic of the Congo). Case histories quickly suggested that shared use of contaminated needles and syringes (used to administer malaria treatments) at the Yambuku Mission Hospital (Mongala Province, Zaire; now Democratic Republic of the Congo) was a major source of early transmission of Ebola virus.49 Other early risk factors for household transmission included the direct care of, sleeping in the same room as, delivering children of, and attending the funerals of infected individuals, and preparing cadavers. Close contact with bodily fluids during the preparation of bodies was thought to contribute to early transmission in the 2007 outbreak of Ebola virus disease in the Democratic Republic of the Congo.51 The extreme inaccessibility of the affected area is thought to have delayed the detection of the outbreak, in which 267 people were infected and 187 (70%) people died.
Quality of evidence
The evidence that wildmeat consumption was the source of spillover was of variable quality. The evidence for almost half of spillover events (50%; n=45) examined was deemed to be of poor quality; for a fifth of events the evidence was deemed to be strong (20%; n=18). Broken down by pathogen type, the strongest evidence (by percentage of events) was seen for the spillover of parasitic pathogens, for which a third of events were deemed to be of high quality. For these events, samples of infected wildmeat were retrievable and the presence of the pathogen was confirmed. For five outbreak investigations involving parasitic spillover, formal statistical analyses reinforced the source of infection in (often) larger groups of infected people. Spillover of bacterial pathogens was often examined retrospectively in seroprevalence studies (ten of 23 studies; 43%), and index patients and exact wildmeat sources were rarely identifiable. Some studies with higher-quality evidence showed endemicity of bacterial infection within local wild animal populations, such as the presence of Leptospira sp in local squirrel populations in Iowa, USA.30 Viral spillover was poorly characterised in 32 of 37 qualitative case reports, which described plausible animal reservoirs and transmission routes with reference to the suspected index case. Some studies of viral spillover that were deemed to provide stronger-quality evidence suggested endemicity of infection within local reservoirs, such as the detection of monkeypox virus within local Congolese squirrel (Funisciurus sp and Heliosciurus sp) and rodent (Cricetomys emini) species.56 More confidence in the animal origin of infection was possible for studies in which exposure to the suspected wildmeat source was shared among multiple index patients (12 of 37 studies)—for example, studies in which the index cases were all involved in the same hunt, or consumed the same wildmeat.57
Discussion
This Review highlights some of the diverse pathways that link the consumption of wildmeat to zoonotic spillover, and the current scarcity of strong detailed evidence on spillover events associated with wildmeat consumption. For around a third of events, the index case, source of wildmeat, or transmission pathway could not be identified from the literature we reviewed. Therefore, the current narrative that implicates wildlife consumers as the source of zoonotic diseases is not substantiated by sufficiently robust evidence. More detailed understanding and characterisation of spillover events is needed to inform more context-sensitive and culturally sensitive and effective interventions aimed at prevention.
Political economists have noted the racist undertones inherent in disease attribution, arguing that apportioning blame at the “bad” consumption practices of low-income and middle-income countries shifts responsibility and attention away from the risks of zoonoses present in food production systems in high-income countries.58 Our Review shows that wildmeat consumption and the associated risk of spillover is a global occurrence, and is not just confined to wildlife markets and hunting practices in low-income and middle-income countries. Although spatial biases in surveillance and diagnostic capability make the assessment of where spillover events are really concentrated difficult, the highest number of spillover events was reported in the USA (n=20), linked with the recreational hunting of wild animals. However, these outbreaks were self-contained, and minimal human-to-human transmission was reported. The larger outbreaks we identified were mostly concentrated in resource-poor regions. In low-income settings, places where diseases are endemic, and places where symptoms are not diagnosed early, spillover is likely to be underreported. For regions where geographical centres of spillover might be inaccessible or where zoonotic disease might be easily misdiagnosed or undiagnosed, outbreaks might only be reported when they reach a threshold size or are distinctive (in clinical presentation or severity) from other endemic illnesses. In such settings, the cumulative effect of regular small outbreaks or endemic disease on local livelihoods and health—often in addition to other environmental shocks—might require the introduction of intensified surveillance in these areas, and improved understanding of routine wild animal consumptive practices and associated exposure risks that might be faced by local inhabitants.
We found that sources of wildmeat for consumption were variable. Some age-related and gender-related differences were also noted between index cases: men were most frequently reported to be the index case, and children were observed as frequent index cases for monkeypox. Several studies noted gender differences in wildmeat consumptive practices: in both high-income and low-and-middle-income countries, men and children were often procurers of wild animals, whereas women often prepared food and therefore might also be exposed to uncooked carcasses. We noted several risk factors associated with cooking and preparation methods, including skinning and butchering. Better understanding of gender-related and age-related differences in exposure risk during hunting and food preparation could inform tailored educational and preventative programmes that support the safer handling and consumption of wildmeat.
Our findings support previous serological studies on zoonoses, which have indicated that although spillover from wild animals is comparatively frequent, events that result in widespread human-to-human transmission are less common. We observed that few identified spillover events led to large outbreaks with extensive human-to-human transmission. Larger outbreaks were concentrated in remote, low-income settings that might already be vulnerable to environmental and socioeconomic shocks. Early human-to-human transmission was often a result of nosocomial transmission, close familial or community contacts, and cultural (eg, burial) practices. Simple outbreak containment measures—including isolation of patients, use of personal protective equipment by health-care workers, not sharing health-care equipment, and change in the cultural customs at funerals—emerge as important ways to break the chain of human-to-human transmission.49, 50, 51
Limitations
There are several limitations to this Review. First, spatial biases in surveillance and diagnostic capability make the assessment of the extent to which spillover really is concentrated in the regions identified difficult. Investigations into the spillover source at the start of an outbreak are often not a priority and so might be slow. Retrospective identification of the spillover source, especially when delayed, can be challenging. Second, although we attempted to characterise index cases, challenges in surveillance make it often possible that suspected index cases are not the real index cases, but merely the first patient to be identified or those with a plausible exposure history to wild animals known to be reservoirs for disease. Furthermore, the possibility of continued or extensive environmental sources of disease cannot be excluded, especially for larger or extended outbreak events, leading to multiple introductions of disease into a susceptible human population, and therefore multiple index cases. Finally, this Review attempted to identify available evidence for reported spillover events that have been linked strongly to wildmeat consumption or consumption-related exposures; however, this link is often difficult to establish, and the quality of evidence to support this link is often not strong. By including only those events for which evidence is available to support this direct link, our analysis is likely to understate the number of events associated with wildlife consumption and therefore should not be used to estimate the incidence of events. Further biases within our search could have been introduced as a result of the English language restriction.
Towards targeted policy responses and intervention
Following the COVID-19 pandemic, attitudes towards wildmeat consumption and trade practices will probably continue to evolve, shaped by pressures for regulation and legislative change and discourses of fear and contagion. This Review suggests the need for more nuanced approaches to protect an increasingly globalised and interconnected world from the potential risks of zoonotic spillover while not stigmatising the consumption practices and livelihoods of communities that rely upon wild resources.
Current global disease-control efforts are almost exclusively reactive, responding to the threats posed by disease after a spillover event has occurred.59 Literature in the past decade, but especially in the context of the COVID-19 pandemic, has examined potential policy responses to proactively mitigate the risk and burdens of future disease spillover events due to wildlife trade and wildmeat consumption, with relevance to the findings of this Review. These responses can be broken down into improved wildlife monitoring, health surveillance and risk assessment in harvesting, consumption, and trading environments; awareness campaigns; and targeted interventions and regulation related to wildmeat hunting and consumption. Different policy options will be feasible, appropriate, practical, and affordable in different settings.60
Surveillance and risk assessment in relevant environments
As the risks of zoonotic spillover directly from wildlife are increasing, there is arguably a need for increased global pre-spillover surveillance measures, including identification and monitoring of potential spillover hotspots, and ongoing, dynamic risk assessment of high-risk settings and human and animal populations. Existing efforts to identify key hotspots for spillover and subsequent take-off in transmission have indicated an increased risk of spillover in forested tropical regions that have rapid land-use changes, sites where wildlife biodiversity (mammal species richness) and human population densities are high.61, 62 Elsewhere, there has been early exploration into instruments to assess risk from wildlife consumption and trade in given settings, which can be used to inform local policy. For example, Wikramanayake and colleagues63 developed a risk-assessment tool to evaluate disease risk presented by the food processing and handling of traded species in specific markets, therefore enabling the identification of high-risk trading conditions. Enhanced surveillance of human population groups who could be more readily exposed through the consumption of at-risk animal species, and of the animal populations to which they are exposed, could help to predict spillover risk and enable early intervention.59 Further investigation of practical tools that can be easily adapted for use in other contexts in which wildmeat consumption takes place is needed. Given that accumulated evidence also points to the dynamic nature of spillover risk over time (including seasonally as animals migrate) and in response to environmental change (such as deforestation), such tools should monitor change in spillover risk and therefore enable adaptive policy responses. This research speaks to the broader agenda of the One Health approach,64, 65 which recognises and seeks to better understand the complex entanglement of pathogenic, human, animal, and environmental factors that influence the emergence, spread, and heterogeneous effects of zoonotic disease, and drive effective research and policy responses.
Improving local awareness of spillover risk
Interventions that work with local communities to improve understanding of high-risk and low-risk species, practices, and situations could help to reduce spillover risk from wildmeat consumption. The adoption of precautions by wildmeat users assumes existing knowledge of associated risks and the capacity to adopt protective behaviours. Accumulated evidence suggests that the perception of and knowledge about zoonotic diseases is generally low, is variable between and within locations, and is often negatively associated with participation in wildmeat consumption and related activities.66 In some contexts, attitudes towards disease risk associated with wildlife hunting have been dismissive of the risk factors, and appeal to the perceived masculine performance associated with hunting.67 Such attitudes could be counterproductive to current risk-reduction efforts.
This Review and previous work have also indicated the diverse social determinants of exposure to wildmeat, including gender, education, and age.68, 69 Knowing who is most exposed to high-risk species, and targeting awareness-building activities (eg, of high-risk species, practices, conditions, and seasonal risks) towards those most exposed, could result in positive behaviour change, such as encouraging individuals to consider additional safety measures when hunting or butchering wildmeat. Furthermore, increasing knowledge of alternative, lower-risk wildlife products could reduce the demand for high-risk species while satiating taste and the need for wild food sources.60 Understanding the socioeconomic context of human–wildlife interactions, including existing perceptions of disease risk and who is most exposed, is therefore important for awareness-building activities. Working with leading, trusted actors within communities is important to support effective engagement and the credible communication of risks.
Targeted intervention and regulation related to wildmeat hunting and consumption
Preventing or reducing the hunting, consumption, and trading of wild species should decrease the transmission of zoonotic diseases to livestock and people.61 Interventions could include banning, penalising, or regulating the sale, possession, and consumption of particular high-risk species, such as bats.70 For species that are deemed lower-risk, interventions to ensure good hygiene practices (eg, licensing and standards) will probably be more effective than total bans on consumption.11 Restrictions on harvesting, consumption, or trade activities within particular regions or at particular times of the year (when high-risk species are migrating or breeding, and therefore animal densities are likely to be high) might also be warranted.
However, the benefits of such interventions must be weighed against the risks of unintended consequences of such interventions, including on local community livelihoods. Consultation with community leaders and stakeholders before intervention is imperative to ensure that policies are attentive to local disease exposures (and practices that might lead to these) while being sensitive to local needs. For example, Leroy and colleagues51 noted that migratory bats in the Democratic Republic of the Congo provide an important seasonal food source for men, postmenopausal women, and children. The 2007 outbreak of Ebola virus disease in this country coincided with a large-scale migration of bats; as such, hunters had to adopt non-traditional killing methods and used shotguns to shoot hordes of bats, resulting in direct contact with blood-covered animals. In such examples, given that contact between humans and wild animals is inevitable (ie, bats will continue migrating to areas of human habitation), an effective policy response would need to better understand the nature of these exposures to recommend alternative behaviours that would limit the risk of disease spillover. Engaging with community stakeholders can mitigate the disbenefits of interventions, improve acceptance and therefore increase the success of interventions.71
Although there are unarguably risks of disease spillover associated with wildlife consumption, policies that aim to reduce these risks represent only one strand of solutions that seek to mitigate the risk of future pandemics. Recognition of land-use changes as a substantial driver of disease outbreaks is growing, including agricultural expansion, resource extraction, deforestation, urbanisation, and infrastructural development. Media and policy focus on wildmeat consumption should not distract from important research and context-sensitive intervention into the risks of spillover presented by ongoing landscape alteration and destruction, and if or how these processes could interfere with risks of spillover from consumption.5, 72, 73
Global political discourse continues to identify wildmeat consumption and trade as high-risk activities for zoonotic disease spillover, despite the incomplete understanding and the scarcity of strong evidence to support these claims. By compiling existing evidence, this Review has highlighted important gaps in knowledge about the context of spillovers, and indicated that more careful understanding of spillover conditions is necessary to support more sensitive and targeted policy and public health responses.14, 23 Developing stronger understanding of the nature of consumptive exposures to wildmeat with spillover potential needs to be a future research priority for all contexts, especially in low-income settings that could be vulnerable to larger outbreaks that exacerbate existing vulnerabilities. The ongoing (and perhaps accelerating) threat of zoonotic disease calls not for blunt, knee-jerk policy reactions aimed at disease prevention, which prohibit all forms of wildmeat consumption practices, but for context-sensitive and effective interventions grounded in understanding of the vulnerabilities of diverse local communities, and their continued need for access to these sources of nutrition and food security.
Search strategy and selection criteria
First, we identified all unique zoonotic pathogens (n=183) from a publicly available database of 335 emerging infectious disease events. From this database, only four emerging infectious diseases were identified for which the driver of emergence was ascribed to bushmeat. The full texts of these sources were retrieved. Given that these events represented only the first time that such zoonoses were reported in humans (eg, Ebola virus in 1976), we searched for subsequent spillover events for these diseases up to 2021, using keyword searches. To identify spillover events for endemic and epidemic zoonoses associated with wild animal consumption, we searched PubMed and Google Scholar using keywords and their synonyms relating to the wildlife exposure (eg, “wild meat”, “bushmeat”, and “hunt”), the zoonotic disease outcome (eg, “zoonotic” and “infect*”), and the association between the two. Studies were included in our analysis if they described unique events of zoonotic spillover, provided evidence to support the association of wildmeat consumption practices with zoonotic spillover, hypothesised that exposure to wild animals or meat preceded illness onset within an expected incubation period and was biologically plausible, featured zoonotic disease outcomes that were clinically diagnosed in human participants, were published between 1940 and 2021, and could be retrieved in full in English. Studies were excluded if they were published before 1940, if domesticated species (including livestock) were identified as the driver of zoonotic disease emergence (as reservoir host species or intermediate species involved in wildlife-to-human spillover), or if zoonotic disease was not diagnosed according to recognised clinical criteria. We excluded vector-borne diseases from this Review, because an indirect transmission route to humans can be considered to be a less immediate source of zoonotic transmission for hunters and consumers of wild animals than handling and eating, and policy recommendations are likely to differ. To identify events that might have been missed by the literature search, we examined the reference lists of retrieved studies. The quality of the available evidence was assessed as poor, moderate, or strong through use of a framework based on Bradford Hill criteria, to indicate whether evidence was weakly or more strongly supportive of wildmeat consumption as the cause of spillover. Detailed information on the methods used can be found in the appendix (pp 2–4).
Declaration of interests
We declare no competing interests.
Acknowledgments
Acknowledgments
CM is funded by the Economic and Social Research Council for doctoral research (award ES/J500033/1).
Contributors
CM and BV conceptualised the study and designed the methods. CM searched the literature. CM and BV interpreted the data. CM wrote the original draft of the manuscript, which was reviewed and edited by CM and BV. BV acquired funding and supervised the project.
Supplementary Material
References
- 1.Huang C, Wang Y, Li X, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395:497–506. doi: 10.1016/S0140-6736(20)30183-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Zhou P, Shi Z-L. SARS-CoV-2 spillover events. Science. 2021;371:120–122. doi: 10.1126/science.abf6097. [DOI] [PubMed] [Google Scholar]
- 3.Loh EH, Zambrana-Torrelio C, Olival KJ, et al. Targeting transmission pathways for emerging zoonotic disease surveillance and control. Vector Borne Zoonotic Dis. 2015;15:432–437. doi: 10.1089/vbz.2013.1563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Taylor LH, Latham SM, Woolhouse MEJ. Risk factors for human disease emergence. Philos Trans R Soc Lond B Biol Sci. 2001;356:983–989. doi: 10.1098/rstb.2001.0888. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Shivaprakash KN, Sen S, Paul S, Kiesecker JM, Bawa KS. Mammals, wildlife trade, and the next global pandemic. Curr Biol. 2021;31:3671–3677. doi: 10.1016/j.cub.2021.06.006. e3. [DOI] [PubMed] [Google Scholar]
- 6.Sokolow SH, Nova N, Pepin KM, et al. Ecological interventions to prevent and manage zoonotic pathogen spillover. Philos Trans R Soc Lond B Biol Sci. 2019;374 doi: 10.1098/rstb.2018.0342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.International Livestock Research Institute Mapping of poverty and likely zoonoses hotspots. Zoonoses Project 4. Report to Department for International Development, UK. Jan 1, 2012. https://assets.publishing.service.gov.uk/media/57a08a63ed915d622c0006fd/ZooMapDFIDreport18June2012FINALsm.pdf
- 8.Yang N, Liu P, Li W, Zhang L. Permanently ban wildlife consumption. Science. 2020;367 doi: 10.1126/science.abb1938. [DOI] [PubMed] [Google Scholar]
- 9.Borzée A, McNeely J, Magellan K, et al. COVID-19 highlights the need for more effective wildlife trade legislation. Trends Ecol Evol. 2020;35:1052–1055. doi: 10.1016/j.tree.2020.10.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Xiao L, Lu Z, Li X, Zhao X, Li BV. Why do we need a wildlife consumption ban in China? Curr Biol. 2021;31:R168–R172. doi: 10.1016/j.cub.2020.12.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Fournié G, Guitian J, Desvaux S, et al. Interventions for avian influenza A (H5N1) risk management in live bird market networks. Proc Natl Acad Sci USA. 2013;110:9177–9182. doi: 10.1073/pnas.1220815110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Bonwitt J, Dawson M, Kandeh M, et al. Unintended consequences of the ‘bushmeat ban' in west Africa during the 2013–2016 Ebola virus disease epidemic. Soc Sci Med. 2018;200:166–173. doi: 10.1016/j.socscimed.2017.12.028. [DOI] [PubMed] [Google Scholar]
- 13.Booth H, Arias M, Brittain S, et al. “Saving lives, protecting livelihoods, and safeguarding nature”: risk-based wildlife trade policy for sustainable development outcomes post-COVID-19. Front Ecol Evol. 2020;9 doi: 10.3389/fevo.2021.639216. [DOI] [Google Scholar]
- 14.Eskew EA, Carlson CJ. Overselling wildlife trade bans will not bolster conservation or pandemic preparedness. Lancet Planet Health. 2020;4:e215–e216. doi: 10.1016/S2542-5196(20)30123-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Jones KE, Patel NG, Levy MA, et al. Global trends in emerging infectious diseases. Nature. 2008;451:990–993. doi: 10.1038/nature06536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Gottdenker NL, Streicker DG, Faust CL, Carroll CR. Anthropogenic land use change and infectious diseases: a review of the evidence. EcoHealth. 2014;11:619–632. doi: 10.1007/s10393-014-0941-z. [DOI] [PubMed] [Google Scholar]
- 17.Roe D, Lee TM. Possible negative consequences of a wildlife trade ban. Nat Sustain. 2021;4:5–6. [Google Scholar]
- 18.FAO and Alliance of Bioversity International and CIAT Indigenous peoples' food systems: insights on sustainability and resilience from the front line of climate change. 2021. [DOI]
- 19.Rowland D, Ickowitz A, Powell B, Nasi R, Sunderland T. Forest foods and healthy diets: quantifying the contributions. Environ Conserv. 2017;44:102–114. [Google Scholar]
- 20.Nasi R, Taber A, Van Vliet N. Empty forests, empty stomachs? Bushmeat and livelihoods in the Congo and Amazon basins. Int For Rev. 2011;13:355–368. [Google Scholar]
- 21.Anderson I, Robson B, Connolly M, et al. Indigenous and tribal peoples' health (The Lancet–Lowitja Institute Global Collaboration): a population study. Lancet. 2016;388:131–157. doi: 10.1016/S0140-6736(16)00345-7. [DOI] [PubMed] [Google Scholar]
- 22.Zhong S, Crang M, Zeng G. Constructing freshness: the vitality of wet markets in urban China. Agric Human Values. 2020;37:175–185. [Google Scholar]
- 23.Wikramanayake E, Pfeiffer D, Magouras I, et al. Evaluating wildlife markets for pandemic disease risk. Lancet Planet Health. 2021;5:e400–e401. doi: 10.1016/S2542-5196(21)00143-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Kusolsuk T, Kamonrattanakun S, Wesanonthawech A, et al. The second outbreak of trichinellosis caused by Trichinella papuae in Thailand. Trans R Soc Trop Med Hyg. 2010;104:433–437. doi: 10.1016/j.trstmh.2009.12.005. [DOI] [PubMed] [Google Scholar]
- 25.Lehman MW, Craig AS, Malama C, et al. Role of food insecurity in outbreak of anthrax infections among humans and hippopotamuses living in a game reserve area, rural Zambia. Emerg Infect Dis. 2017;23:1471–1477. doi: 10.3201/eid2309.161597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Eales KM, Norton RE, Ketheesan N. Brucellosis in northern Australia. Am J Trop Med Hyg. 2010;83:876–878. doi: 10.4269/ajtmh.2010.10-0237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Rosenkranz M, Elsner HA, Stürenburg HJ, Weiller C, Röther J, Sobottka I. Streptococcus suis meningitis and septicemia contracted from a wild boar in Germany. J Neurol. 2003;250:869–870. doi: 10.1007/s00415-003-1103-3. [DOI] [PubMed] [Google Scholar]
- 28.Dupouy-Camet J, Yera H, Dahane N, Bouthry E, Kapel CMO. A cluster of three cases of trichinellosis linked to bear meat consumption in the Arctic. J Travel Med. 2016;23 doi: 10.1093/jtm/taw037. [DOI] [PubMed] [Google Scholar]
- 29.Li M, Song Y, Li B, et al. Asymptomatic Yersinia pestis infection, China. Emerg Infect Dis. 2005;11:1494–1496. doi: 10.3201/eid1109.041147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Diesch SL, Crawford RP, McCulloch WF, Top FH. Human leptospirosis acquired from squirrels. N Engl J Med. 1967;276:838–842. doi: 10.1056/NEJM196704132761504. [DOI] [PubMed] [Google Scholar]
- 31.Ter Meulen J, Lukashevich I, Sidibe K, et al. Hunting of peridomestic rodents and consumption of their meat as possible risk factors for rodent-to-human transmission of Lassa virus in the Republic of Guinea. Am J Trop Med Hyg. 1996;55:661–666. doi: 10.4269/ajtmh.1996.55.661. [DOI] [PubMed] [Google Scholar]
- 32.Carrington M, Choe U, Ubillos S, et al. Fatal case of brucellosis misdiagnosed in early stages of Brucella suis infection in a 46-year-old patient with Marfan syndrome. J Clin Microbiol. 2012;50:2173–2175. doi: 10.1128/JCM.00573-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Centers for Disease Control and Prevention Brucella suis infection associated with feral swine hunting—three states, 2007–2008. MMWR Morb Mortal Wkly Rep. 2009;58:618–621. [PubMed] [Google Scholar]
- 34.Halaby T, Hoitsma E, Hupperts R, Spanjaard L, Luirink M, Jacobs J. Streptococcus suis meningitis, a poacher's risk. Eur J Clin Microbiol Infect Dis. 2000;19:943–945. doi: 10.1007/pl00011230. [DOI] [PubMed] [Google Scholar]
- 35.Li TC, Chijiwa K, Sera N, et al. Hepatitis E virus transmission from wild boar meat. Emerg Infect Dis. 2005;11:1958–1960. doi: 10.3201/eid1112.051041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Masuda J, Yano K, Tamada Y, et al. Acute hepatitis E of a man who consumed wild boar meat prior to the onset of illness in Nagasaki, Japan. Hepatol Res. 2005;31:178–183. doi: 10.1016/j.hepres.2005.01.008. [DOI] [PubMed] [Google Scholar]
- 37.Greene YG, Padovani T, Rudroff JA, Hall R, Austin C, Vernon M. Trichinellosis caused by consumption of wild boar meat—Illinois, 2013. MMWR Morb Mortal Wkly Rep. 2014;63:451. [PMC free article] [PubMed] [Google Scholar]
- 38.Holzbauer SM, Agger WA, Hall RL, et al. Outbreak of Trichinella spiralis infections associated with a wild boar hunted at a game farm in Iowa. Clin Infect Dis. 2014;59:1750–1756. doi: 10.1093/cid/ciu713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Khumjui C, Choomkasien P, Dekumyoy P, et al. Outbreak of trichinellosis caused by Trichinella papuae, Thailand, 2006. Emerg Infect Dis. 2008;14:1913–1915. doi: 10.3201/eid1412.080800. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Cui J, Wang ZQ, Xu BL. The epidemiology of human trichinellosis in China during 2004–2009. Acta Trop. 2011;118:1–5. doi: 10.1016/j.actatropica.2011.02.005. [DOI] [PubMed] [Google Scholar]
- 41.Sohn WM, Kim HM, Chung DI, Yee ST. The first human case of Trichinella spiralis infection in Korea. Korean J Parasitol. 2000;38:111–115. doi: 10.3347/kjp.2000.38.2.111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Van De N, Thi Nga V, Dorny P, et al. Trichinellosis in Vietnam. Am J Trop Med Hyg. 2015;92:1265–1270. doi: 10.4269/ajtmh.14-0570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Kia EB, Meamar AR, Zahabiun F, Mirhendi H. The first occurrence of Trichinella murrelli in wild boar in Iran and a review of Iranian trichinellosis. J Helminthol. 2009;83:399–402. doi: 10.1017/S0022149X09990319. [DOI] [PubMed] [Google Scholar]
- 44.Fichi G, Stefanelli S, Pagani A, et al. Trichinellosis outbreak caused by meat from a wild boar hunted in an Italian region considered to be at negligible risk for Trichinella. Zoonoses Public Health. 2015;62:285–291. doi: 10.1111/zph.12148. [DOI] [PubMed] [Google Scholar]
- 45.Tanaka S, Maruyama H, Ishiwata K, Nawa Y. A case report of pleural sparganosis. Parasitol Int. 1997;46:73–75. [Google Scholar]
- 46.Tu ZC, Zeitlin G, Gagner JP, Keo T, Hanna BA, Blaser MJ. Campylobacter fetus of reptile origin as a human pathogen. J Clin Microbiol. 2004;42:4405–4407. doi: 10.1128/JCM.42.9.4405-4407.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Hahn BH, Shaw GM, De Cock KM, Sharp PM. AIDS as a zoonosis: scientific and public health implications. Science. 2000;287:607–614. doi: 10.1126/science.287.5453.607. [DOI] [PubMed] [Google Scholar]
- 48.Peeters M, Jung M, Ayouba A. The origin and molecular epidemiology of HIV. Expert Rev Anti Infect Ther. 2013;11:885–896. doi: 10.1586/14787210.2013.825443. [DOI] [PubMed] [Google Scholar]
- 49.WHO Ebola haemorrhagic fever in Zaire, 1976. Bull World Health Organ. 1978;56:271–293. [PMC free article] [PubMed] [Google Scholar]
- 50.Formenty P, Libama F, Epelboin A, et al. L'épidémie de fièvre hémorragique à virus Ebola en République du Congo, 2003: une nouvelle stratégie? Med Trop. 2003;63:291–295. [PubMed] [Google Scholar]
- 51.Leroy EM, Epelboin A, Mondonge V, et al. Human Ebola outbreak resulting from direct exposure to fruit bats in Luebo, Democratic Republic of Congo, 2007. Vector Borne Zoonotic Dis. 2009;9:723–728. doi: 10.1089/vbz.2008.0167. [DOI] [PubMed] [Google Scholar]
- 52.Xu RH, He JF, Evans MR, et al. Epidemiologic clues to SARS origin in China. Emerg Infect Dis. 2004;10:1030–1037. doi: 10.3201/eid1006.030852. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Nolen LD, Osadebe L, Katomba J, et al. Introduction of monkeypox into a community and household: risk factors and zoonotic reservoirs in the Democratic Republic of the Congo. Am J Trop Med Hyg. 2015;93:410–415. doi: 10.4269/ajtmh.15-0168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Pepin J. Cambridge University Press; Cambridge: 2011. The origins of AIDS. [Google Scholar]
- 55.Nolen LD, Osadebe L, Katomba J, et al. Extended human-to-human transmission during a monkeypox outbreak in the Democratic Republic of the Congo. Emerg Infect Dis. 2016;22:1014–1021. doi: 10.3201/eid2206.150579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Hutin YJF, Williams RJ, Malfait P, et al. Outbreak of human monkeypox, Democratic Republic of Congo, 1996 to 1997. Emerg Infect Dis. 2001;7:434–438. doi: 10.3201/eid0703.010311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Georges AJ, Leroy EM, Renaut AA, et al. Ebola hemorrhagic fever outbreaks in Gabon, 1994–1997: epidemiologic and health control issues. J Infect Dis. 1999;179:S65–S75. doi: 10.1086/514290. [DOI] [PubMed] [Google Scholar]
- 58.Wallace R. NYU Press; New York, NY: 2016. Big farms make big flu. [Google Scholar]
- 59.Kurpiers LA, Schulte-Herbrüggen B, Ejotre I, Reeder DAM. In: Problematic wildlife. Angelici FM, editor. Springer; Cham: 2016. Bushmeat and emerging infectious diseases: lessons from Africa; pp. 507–551. [Google Scholar]
- 60.Petrovan SO, Aldridge DC, Bartlett H, et al. Post COVID-19: a solution scan of options for preventing future zoonotic epidemics. Biol Rev Camb Philos Soc. 2021;96:2694–2715. doi: 10.1111/brv.12774. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Johnson CK, Hitchens PL, Pandit PS, et al. Global shifts in mammalian population trends reveal key predictors of virus spillover risk. Proc Biol Sci. 2020;287 doi: 10.1098/rspb.2019.2736. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Allen T, Murray KA, Zambrana-Torrelio C, et al. Global hotspots and correlates of emerging zoonotic diseases. Nat Commun. 2017;8 doi: 10.1038/s41467-017-00923-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Wikramanayake E, Pfeiffer DU, Magouras I, et al. A tool for rapid assessment of wildlife markets in the Asia-Pacific Region for risk of future zoonotic disease outbreaks. One Health. 2021;13 doi: 10.1016/j.onehlt.2021.100279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Ferri M, Lloyd-Evans M. The contribution of veterinary public health to the management of the COVID-19 pandemic from a One Health perspective. One Health. 2021;12 doi: 10.1016/j.onehlt.2021.100230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Decaro N, Martella V, Saif LJ, Buonavoglia C. COVID-19 from veterinary medicine and one health perspectives: what animal coronaviruses have taught us. Res Vet Sci. 2020;131:21–23. doi: 10.1016/j.rvsc.2020.04.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Kamins AO, Rowcliffe JM, Ntiamoa-Baidu Y, Cunningham AA, Wood JL, Restif O. Characteristics and risk perceptions of Ghanaians potentially exposed to bat-borne zoonoses through bushmeat. EcoHealth. 2015;12:104–120. doi: 10.1007/s10393-014-0977-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Massey PD, Polkinghorne BG, Durrheim DN, Lower T, Speare R. Blood, guts and knife cuts: reducing the risk of swine brucellosis in feral pig hunters in north-west New South Wales, Australia. Rural Remote Health. 2011;11 [PubMed] [Google Scholar]
- 68.Lawson E, Ayivor JS, Ohemeng F, Ntiamoa-Baidu Y. Social determinants of a potential spillover of bat-borne viruses to humans in Ghana. Int J Biol. 2016;8:66. [Google Scholar]
- 69.Pernet O, Schneider BS, Beaty SM, et al. Evidence for henipavirus spillover into human populations in Africa. Nat Commun. 2014;5 doi: 10.1038/ncomms6342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Guth S, Mollentze N, Renault K, et al. Bats host the most virulent—but not the most dangerous—zoonotic viruses. bioRxiv. 2021 doi: 10.1101/2021.07.25.453574. published online July 26. (preprint). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Wilkinson A, Parker M, Martineau F, Leach M. Engaging ‘communities': anthropological insights from the west African Ebola epidemic. Philos Trans R Soc Lond B Biol Sci. 2017;372 doi: 10.1098/rstb.2016.0305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Rohr JR, Barrett CB, Civitello DJ, et al. Emerging human infectious diseases and the links to global food production. Nat Sustain. 2019;2:445–456. doi: 10.1038/s41893-019-0293-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Bloomfield LSP, McIntosh TL, Lambin EF. Habitat fragmentation, livelihood behaviors, and contact between people and nonhuman primates in Africa. Landsc Ecol. 2020;35:985–1000. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.