Abstract
Triple therapy consisting of clarithromycin (CLR), lansoprazole (LPZ), and amoxicillin (AMZ) is effective as eradication therapy for patients with peptic ulcer disease and Helicobacter pylori infection. We evaluated the effects of LPZ and AMZ on the uptake of [14C]CLR into the gastric tissue of rats. After administration of [14C]CLR alone or in combination with LPZ and AMZ, the distributions of [14C]CLR in the main organs and gastrointestinal tissues were compared. LPZ and AMZ had no effect on the distribution of [14C]CLR in any tissue except gastric tissue. The concentration of radioactivity in gastric tissue was several times higher when [14C]CLR was administered orally together with LPZ than when it was administered alone. The gastric emptying of [14C]CLR became smaller in the case of the coadministration of LPZ. AMZ had no apparent influence on the disposition of [14C]CLR. After the intravenous administration of [14C]CLR, no effects of drug coadministration were evident. In vitro uptake of [14C]CLR into gastric tissue was enhanced in the case of a high-pH environment. The uptake was not influenced by the concurrent presence of LPZ and AMZ. These results suggest that the penetration of [14C]CLR possibly depends on elevated gastric pH, as gastric acid secretion was inhibited by LPZ, and this may be a primary factor in explaining why the concentration of [14C]CLR at the target site, gastric tissue, was enhanced by the coadministration of LPZ.
The association between active gastritis and Helicobacter pylori was first reported by Warren (24) and Marshall (13), and H. pylori-associated gastritis may be the cause of gastric ulcer. Recently, clinical trials involving the use of antibiotics yielded evidence that antibiotics can eradicate H. pylori (5, 6, 8, 17).
The macrolide antibiotic clarithromycin (CLR) is, to date, one of the most active antimicrobial agents against H. pylori in vitro (4). It is relatively stable in the presence of gastric acid (15) and has a high affinity for tissue (10). The penicillin antibiotic amoxicillin (AMZ) also has strong in vitro activity against H. pylori (7) and is more stable than macrolides in gastric acid. Treatment with CLR or AMZ alone in vivo, however, is rarely effective. The eradication rate was 15 to 54% when CLR was administered alone (18) and 20 to 30% when AMZ was administered alone (7). The activities of both CLR and AMZ against H. pylori are affected by pH, as the activities are reduced under acidic conditions (1, 4) and the levels of distribution of the drugs to the gastric mucus and mucosa inhabited by H. pylori are insufficient. When a single antibiotic, CLR or AMZ, was combined with a proton pump inhibitor (lansoprazole [LPZ]) (19), the eradication rates were 57 to 77% (20, 23). H. pylori was eradicated from 84 to 95% of patients concomitantly prescribed LPZ, CLR, and AMZ (2, 11, 20).
To better comprehend the synergism, the effects of LPZ and AMZ on the distribution of 14C-labeled CLR in rats were investigated, with particular focus on the ability of CLR to penetrate gastric tissue.
MATERIALS AND METHODS
Chemicals.
[6-O-methyl-14C]CLR was obtained from Daiichi Pure Chemicals Co., Ltd. (Tokyo, Japan). The specific activity was 2.44 MBq/mg and the radiochemical purity was 96% or higher, as determined on the basis of high-pressure liquid chromatography (HPLC) analysis. Unlabeled CLR was synthesized at the Research Center, Taisho Pharmaceutical Co., Ltd. (Saitama, Japan). LPZ was obtained from Takeda Chemical Industries, Ltd. (Osaka, Japan). AMZ was purchased from Sigma Chemical Co. (St. Louis, Mo.). All other commercially available reagents and solvents were of either analytical or HPLC grade.
Animals.
Male Wistar rats (age, 7 weeks) were purchased from Nihon SLC Co., Ltd. (Shizuoka, Japan). The rats were fed a commercial food (MF; Oriental Yeast Co., Ltd., Tokyo, Japan) and water freely throughout the acclimatization period and the study period except for the day before the labeled compounds were administered. The rats weighed 193 to 221 g and were used for the study when they were 8 weeks of age.
Preparation of dosage form and administration of drug.
Rats were given 5 mg of [14C]CLR, 10 mg of LPZ, and 10 mg of AMZ per kg of body weight. These drugs for oral administration were suspended in 5% gum arabic adjusted to pH 7.0 with 1 N KOH. For intravenous administration, [14C]CLR was given in a solution of saline, with equimolar amounts of HCl used to dissolve the drug. An intravenous dose of AMZ was dissolved in 2.33% KH2PO4–1.44% NaHCO3 isotonic buffer (pH 7.4). The rats were distributed randomly into six groups, with each group comprising three rats, and were given the drugs as indicated in Table 1.
TABLE 1.
Dosage information for six groups of ratsa
| Group | Treatment on day 1 to day 3 | Treatment on day 4 |
|---|---|---|
| 1 | Vehicle p.o., once daily | [14C]CLR p.o., 1 h after vehicle p.o. |
| 2 | LPZ p.o., once daily | [14C]CLR p.o., 1 h after LPZ p.o. |
| 3 | LPZ p.o., once daily | [14C]CLR p.o. + AMZ p.o. concomitantly, 1 h after LPZ p.o. |
| 4 | Vehicle p.o., once daily | [14C]CLR i.v., 1 h after vehicle p.o. |
| 5 | LPZ p.o., once daily | [14C]CLR i.v., 1 h after LPZ p.o. |
| 6 | LPZ p.o., once daily | [14C]CLR i.v. + AMZ i.v. concomitantly, 1 h after LPZ p.o. |
Rats were treated under nonfasting conditions from day 1 to day 3 and were fasted for 16 h before treatment on day 4. p.o., administration by gastric intubation; i.v., administration by bolus injection into an inferior vein. The doses were 5 mg/kg for [14C]CLR, 10 mg/kg for LPZ, and 10 mg/kg for AMZ. The vehicle was 5% gum arabic adjusted to pH 7.0 with 1 N KOH.
Effects of LPZ and AMZ on distribution of [14C]CLR.
At 15, 30, 60, and 240 min after oral administration of [14C]CLR (groups 1 to 3) and at 15 and 60 min after intravenous administration of [14C]CLR (groups 4 to 6), the rats were anesthetized with either and whole blood was withdrawn from the inferior aorta and placed into heparinized containers. Plasma was obtained by centrifugation of the blood at 1,600 × g rpm for 10 min at 4°C. After the collection of blood, the liver, kidney, heart, lung, stomach, and intestine (from duodenum to ileum) were immediately excised and the gastrointestinal contents were collected from each animal. The stomach was separated into the forestomach and the glandular stomach. The duodenum was separated from the intestine. The radioactivity in each biological sample was dissolved with 0.5 to 1 ml of Soluene-350 (Packard Instrument Co., Inc., Meriden, Conn.) and was decolored by adding 0.4 to 0.5 ml of 30% hydrogen peroxide. Then, 10 ml of Insta-gel plus scintillator (Packard Instrument Co., Inc.) was added to the sample after neutralization with 0.5 to 1 ml of 1 N HCl. The radioactivity was measured with a liquid scintillation counter (LS6000TA; Beckman Instruments Inc., Fullerton, Calif.).
Preparation of drug solution for in vitro studies.
[14C]CLR was dissolved in an isotonic buffer of pH 2.3 (12 N HCl, 6.1 ml; citric acid, 7.7 g, NaOH, 2.9 g; NaCl, 3.6 g [all per liter]), pH 4.0 (12 N HCl, 3.8 ml; citric acid, 12.6 g; NaOH, 4.8 g; NaCl, 2.1 g [all per liter]), pH 5.5 (NaCl, 5.0 g; Na2HPO4 · 12H2O, 1.2 g; KH2PO4, 8.6 g [all per liter]), or pH 7.4 (NaCl, 4.0 g; Na2HPO4 · 12H2O, 19.1 g; KH2PO4, 1.8 g [all per liter]) at a final concentration of 100 μg/ml. When the drug interaction was investigated, LPZ or LPZ and AMZ at final concentrations of 200 μg/ml were each added to the solution of [14C]CLR.
In vitro uptake of [14C]CLR into gastric tissue.
Rats were fasted for 16 h and were then killed by withdrawal of whole blood, following anesthetization with ether. The stomach was washed with about 10 ml of saline, the cardia and pylorus were tied, and then the entire stomach was immediately excised. A solution of 1 ml of [14C]CLR in an isotonic buffer of pH 2.3, 4.0, 5.5, or 7.4 was injected through a needle into the excised stomach sac, followed by incubation at 37°C for 1 h in 5 ml of isotonic buffer of pH 7.4. After incubation, the radioactivities of the buffers in the sac, incubation medium, and gastric tissue (forestomach and glandular stomach, respectively) were determined in the same manner as described above for the in vivo study.
In order to clarify the effects of LPZ and AMZ on the penetration of [14C]CLR, rats which were given drugs as indicated in Table 1 were used. One hour after the final administration of LPZ, their stomachs were washed with saline and excised. Then, isotonic buffer plus [14C]CLR and LPZ or isotonic buffer plus [14C]CLR, LPZ, and AMZ was injected into the sacs and the experiments were continued as described above.
Statistics.
The results are expressed as the means and standard deviations. The significance of differences was evaluated by variance analysis with the SAS/STAT package. A significance level of 0.01 was used for all tests.
RESULTS
Concentrations of radioactivity in main organs.
The concentrations of radioactivity in the main organs 60 min after administration of [14C]CLR are presented in Table 2. When [14C]CLR was administered orally, the highest concentration of radioactivity was found in the liver, followed in descending order by the concentrations in the lung, kidney, and heart. On the other hand, after intravenous administration, the concentration in the lung was the highest, being approximately twice as high as that in the liver. LPZ and AMZ were not observed to have any effect on the concentration of [14C]CLR in any tissue.
TABLE 2.
Concentrations of radioactivity in tissues 60 min after oral or intravenous administration of [14C]CLR to rats (groups 1 to 6)
| Route of administration and groupa | Concntration (μg eq/ml or μg eq/g)b
|
|||||
|---|---|---|---|---|---|---|
| Blood | Plasma | Liver | Kidney | Heart | Lung | |
| p.o. | ||||||
| Group 1 | 0.23 ± 0.24 | 0.21 ± 0.15 | 13.21 ± 8.52 | 3.14 ± 2.46 | 1.31 ± 1.17 | 7.48 ± 6.81 |
| Group 2 | 0.20 ± 0.09 | 0.20 ± 0.12 | 12.91 ± 12.78 | 2.87 ± 1.77 | 1.15 ± 0.57 | 6.68 ± 3.93 |
| Group 3 | 0.22 ± 0.12 | 0.19 ± 0.12 | 12.34 ± 5.88 | 3.10 ± 1.92 | 1.35 ± 0.81 | 8.71 ± 5.64 |
| i.v. | ||||||
| Group 4 | 0.48 ± 0.06 | 0.45 ± 0.12 | 15.54 ± 8.46 | 8.67 ± 1.32 | 3.00 ± 0.72 | 34.91 ± 4.53 |
| Group 5 | 0.53 ± 0.06 | 0.46 ± 0.06 | 17.28 ± 2.70 | 8.45 ± 1.77 | 3.08 ± 0.21 | 42.13 ± 3.96 |
| Group 6 | 0.55 ± 0.06 | 0.44 ± 0.03 | 14.67 ± 1.47 | 8.59 ± 1.71 | 3.26 ± 0.81 | 36.65 ± 1.50 |
p.o., oral route; i.v., intravenous route.
Each value represents the mean ± standard deviation of three animals.
Concentration of radioactivity in gastrointestinal tissues.
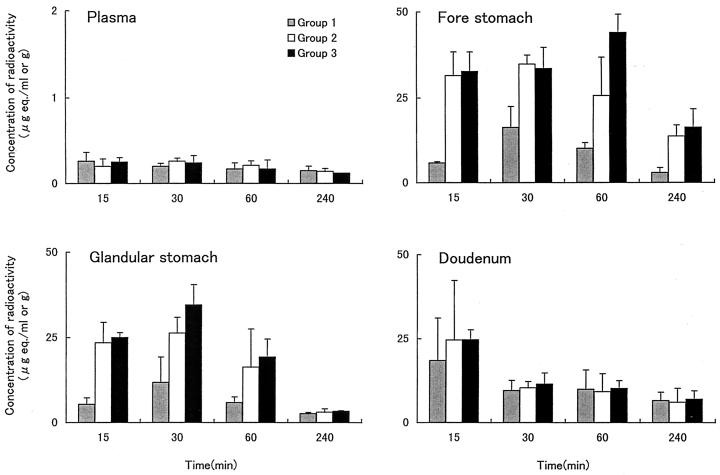
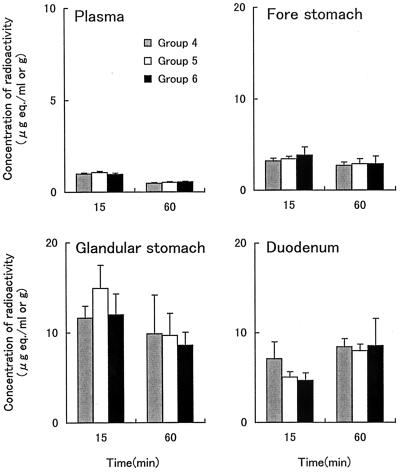
The concentrations of radioactivity in gastrointestinal tissues after the administration of [14C]CLR are shown in Fig. 1 and 2. For both the oral and intravenous routes of administration, the concentrations of radioactivity in all gastrointestinal tissues were much higher than those in plasma. After oral administration, the concentrations in the forestomach and glandular stomach were altered significantly (P < 0.01) by coadministration of LPZ (Fig. 1). During the first 60 min, the concentrations in the forestomach and the glandular stomach were two to six times higher when [14C]CLR was given together with LPZ (groups 2 and 3) than when it was given alone (group 1). LPZ had no apparent effect on the concentration of radioactivity in the duodenum. There were no significant differences in the concentrations of radioactivity in any of the gastrointestinal tissues between the dual-treatment group (group 2) and the triple-treatment group (group 3). On the other hand, in the case of intravenous administration, the radioactivity in the glandular stomach was three to four times higher than that in the forestomach (Fig. 2). The coadministration of drugs had no effect on the concentrations of radioactivity in gastric tissues, which is different from the case for oral administration.
FIG. 1.
Concentrations of radioactivity in gastrointestinal tissues after oral administration of [14C]CLR to rats (groups 1 to 3). Each value represents the mean±standard deviation for three animals. Significant differences (P < 0.01) in the concentrations of radioactivity in the forestomachs and glandular stomachs were observed in groups 2 and 3 compared with those in group 1.
FIG. 2.
Concentrations of radioactivity in gastrointestinal tissues after intravenous administration of [14C]CLR to rats (groups 4 to 6). Each value represents the mean ± standard deviation for three animals.
Recovery of radioactivity in gastrointestinal contents.
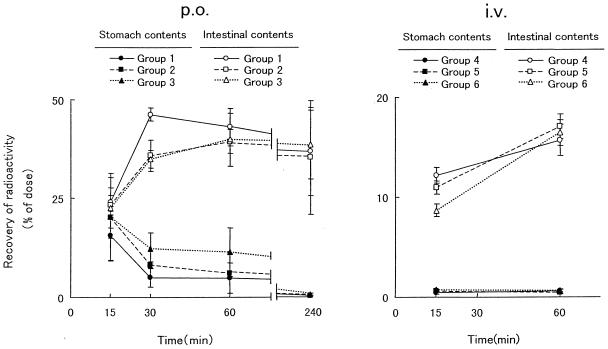
The recoveries of radioactivity in the stomach contents and the intestinal contents after administration of [14C]CLR are shown in Fig. 3. After oral administration, there was a pronounced tendency (P < 0.0487) for the recovery of radioactivity in the stomach contents observed after [14C]CLR was administered together with LPZ (Groups 2 and 3) to be higher than that observed after [14C]CLR was administered alone (group 1). During the first 60 min after administration, the levels of recovery of radioactivity in the intestinal contents of groups 2 and 3 were lower than that for group 1. On the other hand, after the intravenous administration of [14C]CLR, no effect of LPZ on the recovery of radioactivity in the gastrointestinal contents was detected. The recovery of radioactivity in stomach contents (about 0.6%) and intestinal contents (10 to 20%) was lower after intravenous administration than that after oral administration.
FIG. 3.
Recovery of radioactivity in gastrointestinal contents after oral (p.o.) or intravenous (i.v.) administration of [14C]CLR to rats (groups 1 to 6). Each value represents the mean ± standard deviation for three animals.
In vitro uptake of radioactivity into gastric tissue.
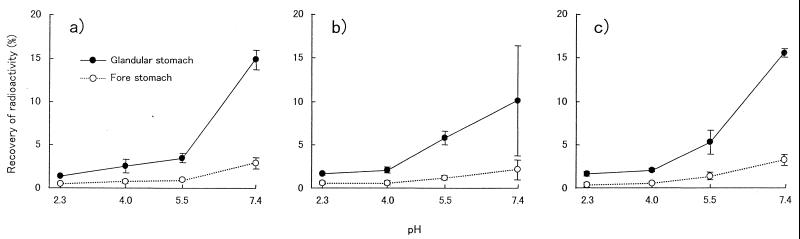
[14C]CLR dissolved in isotonic buffer at various pHs was injected into stomach sacs, and then the stomach sacs were incubated at 37°C. The uptake of radioactivity into glandular stomach and forestomach tissues depended significantly (P < 0.01) on the pH environment. The highest level of uptake was observed at pH 7.4; the recoveries were 14.86% in glandular stomach tissue and 2.93% in forestomach tissue. The uptake decreased in the descending order of pH 5.5, 4.0, and 2.3 (Fig. 4a). The coadministration of LPZ and AMZ on the uptake of [14C]CLR into the gastric tissue was not observed to have any effect (Fig. 4b and c). In all cases, there was little recovery of radioactivity in the incubation medium.
FIG. 4.
In vitro uptake of radioactivity in rat gastric tissue after injection of [14C]CLR (100 μg/ml) into rat stomach sacs. Each value represents the mean ± standard deviation of three experiments. A significant difference (P < 0.01) in the recovery of radioactivity in the glandular stomach and the forestomach was observed among the pH environments. (a) [14C]CLR (100 μg/ml) alone; (b) [14C]CLR (100 μg/ml) plus LPZ (200 μg/ml) after administration of LPZ (10 mg/kg for 4 days, orally); (c) [14C]CLR (100 μg/ml) plus LPZ (200 μg/ml) and AMZ (200 μg/ml) after administration of LPZ (10 mg/kg for 4 days orally).
DISCUSSION
H. pylori naturally infects humans; over its evolution the organism has become well suited to the environment of the gastric mucosa and mucus (12). The present study shows the synergistic effects of LPZ and AMZ on the uptake of [14C]CLR into rat gastric tissue, with both gastric mucosa and mucus being target sites.
After both the oral and the intravenous administration of [14C]CLR, the concentrations of radioactivity in the liver, lung, kidney, heart, and gastrointestinal tissues, which were much higher than those in blood and plasma, were the result of the high degree of affinity of [14C]CLR for tissue. When [14C]CLR was administered intravenously, radioactivity was detected in the stomach contents, suggesting that [14C]CLR may be secreted from gastric tissue. This may explain why [14C]CLR was distributed selectively into the glandular stomach containing foveolae gastricae and fundic glands which had secreting cells, whereas the concentration of radioactivity in the forestomach, which had no secreting cells, was low after intravenous administration. In the case of oral administration, the concentrations of radioactivity in both the forestomach and the glandular stomach were similar, but these levels were higher than those after intravenous administration. Thus, we propose two routes of [14C]CLR uptake into the gastric tissue after oral administration: one is penetration from gastric lumen, and the other is secretion through the blood circulation. The synergistic effect of LPZ on the uptake of radioactivity into the gastric tissue would be due to the penetration of [14C]CLR and would have no influence on the secretion of [14C]CLR. CLR is metabolized to the active metabolite 14-hydroxy CLR in humans (21), but the metabolic reaction occurs at a low level in rats (22). Furthermore, CLR is more stable in a high-pH environment (treatment with LPZ). Hence, in the present study, the radioactivity that penetrated into the gastric tissue and that was secreted through the blood circulation may mainly be unchanged CLR in rats.
The level of gastric emptying of the radioactivity into the intestinal contents was lower when [14C]CLR was coadministered with LPZ than when it was administered alone. Proton pump inhibitors such as omeprazole (16) and LPZ (9) had no effect on the gastric emptying of a liquid meal. Therefore, it was interesting that the gastric emptying of [14C]CLR after oral administration was influenced by the coadministration of LPZ. A part of the [14C]CLR dosed was trapped in the gastric mucus, and the trapping ratio might be increased by the coadministration of LPZ.
To better comprehend the mechanism of enhanced penetration of [14C]CLR by the coadministration of LPZ, the effects of both pH and the presence of additional drugs were demonstrated in in vitro experiments with excised stomach sacs. The uptake of radioactivity into gastric tissue depended on the pH. The level of penetration of CLR was greater at neutral pH and less at acidic pH. As CLR is a base with a pKa of 8.76 (15), the percentage of the nonionized form is larger at basic pH and the solubility of CLR in lipid may be enhanced. In contrast, no direct drug interaction would exist regarding the penetration of [14C]CLR. It was reported that the pH of the gastric surface after oral dosing to rats with LPZ (10 mg/kg) was elevated to about 6 and was significantly higher than that for the vehicle (pH 2 to 3) (14). In addition, when omeprazole was administered to healthy subjects, the viscosity of the gastric mucus decreased when the intragastric pH was increased (3). Therefore, in vivo the increase in the level of [14C]CLR penetration would be due to the effect of LPZ on the gastric pH.
CLR shows excellent in vitro activity against H. pylori, and the MIC is lower at basic pH (1, 4). In the present study, not only the biological activity of CLR against H. pylori but also the penetration of CLR were enhanced in a high-pH environment (treatment with LPZ). The penetration would be the primary factor that affects CLR uptake into gastric tissue soon after administration. On the other hand, the concentration related to secretion is lower than that related to penetration, but it may exceed the MIC in the high-pH environment (treatment with LPZ) for a long time. In humans, CLR is metabolized to its active metabolite, 14-hydroxy CLR. Therefore, the penetration of CLR and the secretion of both CLR and 14-hydroxy CLR may play important roles in the eradication of H. pylori, with a time lag. These synergistic effects would lead to a highly effective means of eradication of H. pylori in clinical therapy.
REFERENCES
- 1.Cederbrant G, Kahlmeter G, Schalén C, Kamme C. Additive effect of clarithromycin combined with 14-hydroxy clarithromycin, erythromycin, amoxycillin, metronidazole or omeprazole against Helicobacter pylori. J Antimicrob Chemother. 1994;34:1025–1029. doi: 10.1093/jac/34.6.1025. [DOI] [PubMed] [Google Scholar]
- 2.Fennerty M B, Kovacs T O G, Krause R, Haber M, Weissfeld A, Siepman N, Rose P. A comparison of 10 and 14 days of lansoprazole triple therapy for eradication of Helicobacter pylori. Arch Intern Med. 1998;158:1651–1656. doi: 10.1001/archinte.158.15.1651. [DOI] [PubMed] [Google Scholar]
- 3.Goddard A F, Spiller R C. The effect of omeprazole on gastric juice viscosity, pH and bacterial counts. Aliment Pharmacol Ther. 1996;10:105–109. doi: 10.1111/j.1365-2036.1996.tb00183.x. [DOI] [PubMed] [Google Scholar]
- 4.Hardy D J, Hanson C W, Hensey D M, Beyer J M, Fernandes P B. Susceptibility of Campylobacter pylori to macrolides and fluoroquinolones. J Antimicrob Chemother. 1988;22:631–636. doi: 10.1093/jac/22.5.631. [DOI] [PubMed] [Google Scholar]
- 5.Heatley R V. Review article: the treatment of Helicobacter pylori infection. Aliment Pharmacol Ther. 1992;6:291–303. doi: 10.1111/j.1365-2036.1992.tb00051.x. [DOI] [PubMed] [Google Scholar]
- 6.Hirschl A M, Hentschel E, Schütze K, Nemec H, Pötzi R, Gangl A, Weiss W, Pletschette M, Stanek G, Rotter M L. The efficacy of antimicrobial treatment in Campylobacter pylori-associated gastritis and duodenal ulcer. Scand J Gastroenterol. 1988;23:76–81. doi: 10.3109/00365528809091718. [DOI] [PubMed] [Google Scholar]
- 7.Hirschl A M, Rotter M L. Amoxicillin for the treatment of Helicobacter pylori infection. J Gastroenterol. 1996;31:44–47. [PubMed] [Google Scholar]
- 8.Isenberg J I, Mcquaid K R, Laine L, Rubin W. Acid-peptic disorders. In: Yamada T, Alpers D H, Owyang C, Powell D W, Silverstein F E, editors. Textbook of gastroenterology. Vol. 1. Philadelphia, Pa: J. B. Lippincott Company; 1991. pp. 1241–1339. [Google Scholar]
- 9.Kito G, Yoshimura H, Sato H, Inatomi N, Inada I. General pharmacological studies on AG-1749 Jpn. Pharmacol Ther. 1990;18:3907–3925. [Google Scholar]
- 10.Kohno Y, Yoshida H, Suwa T, Suga T. Comparative pharmacokinetics of clarithromycin (TE-031), a new macrolide antibiotic, and erythromycin in rats. Antimicrob Agents Chemother. 1989;33:751–756. doi: 10.1128/aac.33.5.751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Lamouliatte H, Cayla R, Zerbib F, Forestier S, de Mascarel A, Joubert-Collin M, Mégraud F. Dual therapy using a double dose of lansoprazole with amoxicillin versus triple therapy using a double dose of lansoprazole, amoxicillin, and clarithromycin to eradicate Helicobacter pylori infection: results of a prospective randomized open study. Am J Gastroenterol. 1998;93:1531–1534. doi: 10.1111/j.1572-0241.1998.00280.x. [DOI] [PubMed] [Google Scholar]
- 12.Lee A, Hazell S L. Campylobacter pylori in health and disease: an ecological perspective. Microb Ecol Health Dis. 1988;1:1–16. [Google Scholar]
- 13.Marshall B. Unidentified curved bacilli on gastric epithelium in active chronic gastritis. Lancet. 1983;ii:1273–1275. [PubMed] [Google Scholar]
- 14.Nagaya H, Inatomi N, Satoh H. Differences in the antisecretory actions of the proton pump inhibitor AG-1749 (lansoprazole) and the histamine H2-receptor antagonist famotidine in rats and dogs. Jpn J Pharmacol. 1991;55:425–436. doi: 10.1254/jjp.55.425. [DOI] [PubMed] [Google Scholar]
- 15.Nakagawa Y, Itai S, Yoshida T, Nagai T. Physicochemical properties and stability in the acid solution of a new macrolide antibiotic, clarithromycin, in comparison with erythromycin. Chem Pharm Bull. 1992;40:725–728. doi: 10.1248/cpb.40.725. [DOI] [PubMed] [Google Scholar]
- 16.Pendleton R G, Cook P G, Shepherd-Rose A, Mangel A W. Effects of H2-receptor antagonists upon physiological acid secretory states in animals. J Pharmacol Exp Ther. 1985;233:64–69. [PubMed] [Google Scholar]
- 17.Peterson W L. Helicobacter pylori and peptic ulcer disease. N Engl J Med. 1991;324:1043–1048. doi: 10.1056/NEJM199104113241507. [DOI] [PubMed] [Google Scholar]
- 18.Peterson W L, Graham D Y, Marshall B, Blaser M J, Genta R M, Klein P D, Stratton C W, Drnec J, Prokocimer P, Siepman N. Clarithromycin as monotherapy for eradication of Helicobacter pylori: a randomized, double-blind trial. Am J Gastroenterol. 1993;88:1860–1864. [PubMed] [Google Scholar]
- 19.Satoh H, Inatomi N, Nagaya H, Inada I, Nohara A, Nakamura N, Maki Y. Antisecretory and antiulcer activities of a novel proton pump inhibitor AG-1749 in dogs and rats. J Pharmacol Exp Ther. 1989;248:806–815. [PubMed] [Google Scholar]
- 20.Schwartz H, Krause R, Sahba B, Haber M, Weissfeld A, Rose P, Siepman N, Freston J. Triple versus dual therapy for eradicating Helicobacter pylori and preventing ulcer recurrence: a randomized, double-blind, multicenter study of lansoprazole, clarithromycin, and/or amoxicillin in different dosing regimens. Am J Gastroenterol. 1998;93:584–590. doi: 10.1111/j.1572-0241.1998.169_b.x. [DOI] [PubMed] [Google Scholar]
- 21.Suwa T, Ohtake T, Urano H, Kodama T, Nakamura M, Iwatate C, Watanabe T. Metabolic fate of TE-031 (A-56268) (IX) absorption and excretion in humans (HPLC method) Chemotherapy (Tokyo) 1988;36:933–940. [Google Scholar]
- 22.Suwa T, Yoshida H, Yoshitomi S, Kamei K. Metabolic fate of TE-031 (A-56268) (IV) metabolism of 14C-TE-031 in rats and dogs. Chemotherapy (Tokyo) 1988;36:227–237. [Google Scholar]
- 23.Takimoto T, Kimura K, Taniguchi Y, Satoh K, Saifuku K, Kihira K, Yoshida Y, Ido K. Dual therapy with lansoprazole and clarithromycin for eradication of Helicobacter pylori. Eur J Gastroenterol Hepatol. 1995;7:S63–S66. [PubMed] [Google Scholar]
- 24.Warren J R. Unidentified curved bacilli on gastric epithelium in active chronic gastritis. Lancet. 1983;ii:1273. [PubMed] [Google Scholar]