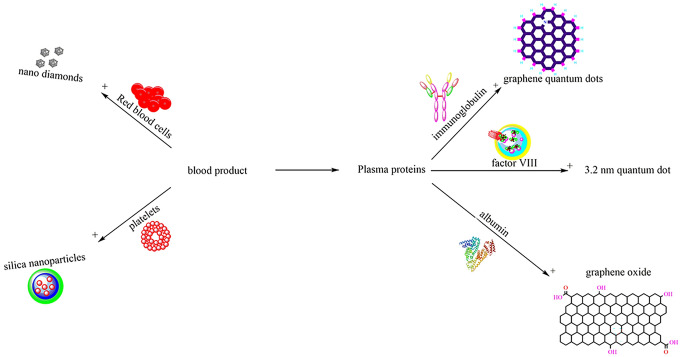
Abstract
Despite the importance of the proper quality of blood products for safe transfusion, conventional methods for preparation and their preservation, they lack significant stability. Non-metal nanoparticles with particular features may overcome these challenges. This review study for the first time provided a comprehensive vision of the interaction of non-metal nanoparticles with each blood product (red blood cells, platelets and plasma proteins). The findings of this review on the most effective nanoparticle for improving the stability of RBCs indicate that graphene quantum dots and nanodiamonds show compatibility with RBCs. For increasing the stability of platelet products, silica nanoparticles exhibited a suppressive impact on platelet aggregation. Pristine graphene also shows compatibility with platelets. For better stability of plasma products, graphene oxide was indicated to preserve free human serum albumin from thermal shocks at low ionic strength. For increased stability of Factor VIII, mesoporous silica nanoparticles with large pores exhibit the superb quality of recovered proteins. Furthermore, 3.2 nm quantum dots exhibited anticoagulant effects. As the best promising nanoparticles for immunoglobulin stability, graphene quantum dots showed compatibility with γ-globulins. Overall, this review recommends further research on the mentioned nanoparticles as the most potential candidates for enhancing the stability and storage of blood components.
Graphical abstract
Keywords: Blood products, Non-metal nanoparticles, Nanodiamonds, Stability, Mesoporous silica nanoparticles
Introduction
Nowadays, the transfusion of blood and its components play a life-saving role in different clinical indications such as bleeding, anemia, surgery, trauma, and etc. (Greening et al. 2010; Sen Gupta 2017). Blood products are derived from whole blood which is rapidly combined with anticoagulants that lead to chemical modifications. Blood derivatives are produced through various purification steps, resulting in damages related to preparation which also combines with storage lesions. To ensure proper storage of blood components and safe transfusion, various strategies have been conducted, including using anticoagulants, centrifugation, filtration, and keeping blood products in additive solutions. Blood products are also treated through the use of pathogen inactivation systems or by utilizing novel storage strategies that enhance their quality (Abonnenc et al. 2018). However, conventional approaches have adverse effects on these products in different aspects. For instance, their biofunction and stability are altered in methods for improving the shelf life using stabilizers and additives (Belousov 2014; Sen Gupta 2017). Blood cells and plasma proteins are also damaged or lost in the process of eradicating contaminations (Klein 2005). Furthermore, current plasma protein purification processes are not efficient and cost-effective (Evtushenko et al. 2005; Mehrizi and Hosseini 2021).
Nanotechnology as an advancing science has provided new opportunities for studying not only the mechanisms of cell damage but also the development of efficient and safe methods for storing cells outside the body which offers a potential solution to the challenges of blood products storage and separation (Belousov et al. 2019; Dashti Rahmat Abadi et al. 2014; Mehrizi 2021a; Shahabi et al. 2014; Zadeh Mehrizi 2021a, b, c; Zadeh Mehrizi and Amini Kafiabad 2021a, b; Zadeh Mehrizi et al. 2021; Zadeh Mehrizi and Eshghi 2021; Zadeh Mehrizi and Mousavi Hosseini 2021). Carbon nanoparticles have received much attention due to their particular optical, thermal, mechanical, electrical, and chemical features. They are very promising materials in gene and drug delivery systems, cancer treatment, bio-sensing, and stem cell therapy. Additionally, for enhanced efficacy, they can easily be functionalized with chemical groups and antibacterial and anti-inflammatory compounds (Fedel 2020).
Despite the potential application of nanotechnology in blood banking and the importance of the stability of blood products, few studies have been focused on the effect of non-metal nanoparticles on each blood component. Therefore, the current paper for the first time reviews the recent studies from 2011 to 2021, to evaluate the interaction of different types of non-metal nanoparticles such as silica, graphene, fullerenes, carbon nanotubes with RBCs, platelets, and plasma proteins including albumin, coagulation factor VIII and immunoglobulin. This study discusses the challenges of each blood product preparation or storage. It also provides an overview of conformational changes, oxidative stress conditions, toxicity concentrations, and the purification process of blood products in case of using non-metal nanomaterials.
Red blood cells (RBCs)
Among the blood components, RBCs are the most commonly transfused one for hemorrhage treatment and oxygen delivery improvement in patients with anemia (Hess 2010a; Klein et al. 2007). RBC concentrates are typically stored in 2–6 °C for up to 42 days but in certain conditions, they can also be preserved using cryopreservation methods (Greening et al. 2010). RBC storage lesion is primarily caused by metabolic reactions and oxidative injury in the storage bags caused by the acidic environment and presence of oxygen, heme, and iron together (Hess 2010b). During storage, ATP and 2,3-diphosphoglycerate decrease, and RBC deformation and hemolysis increase. Thus, the units are collected either as whole blood into bags containing anticoagulant citrate and nutrient phosphate and dextrose to maintain ATP, and 2,3-DPG levels or by apheresis into acid citrate dextrose. Furthermore, due to the lactic acid accumulation in the blood, the pH of RBCs decreases which results in increased phosphatase 3 activity, which induces 2,3-DPG degradation and reduced oxygen delivery to tissues (Hess 2010a; Zubair 2010). The interaction of nanomaterials with RBCs can induce toxicity through membrane alteration, inflammation and ROS production and is associated with their various properties (Pan et al. 2018; Reviakine et al. 2017).
Silica nanoparticles
As an extremely significant class of nanomaterials, silicon-based nanoparticles possess numerous unique properties, including superior physical, electronic, chemical, and optical properties, high surface-to-volume ratios, improved surface reactivity, and easy surface modification. In addition to these properties, due to their ease of fabrication, cost effectiveness, hydrophilicity, favorable biocompatibility, and controlled size, they are promising candidates for a broad range of biomedical applications such as gene carriers, drug delivery, biosensors and bio-imaging (He et al. 2010a, b; He and Su 2014; Jeelani et al. 2020; Murugadoss et al. 2017).
The adverse effect of nanoparticles on RBCs is initially associated with damage to the plasma membrane. There is a key role for the lipid composition of the membrane outer leaflet in nanoparticle-induced membrane damage in both vesicles and erythrocytes. The influence of membrane asymmetry loss and alterations of the lipid composition of the outer leaflets of RBCs on nanoparticle membrane interactions was investigated. In comparison with healthy cells, eryptotic cells had significantly less damage caused by unmodified silica nanoparticles. Neither amino- nor carboxyl-modified particles caused significant hemolysis. Unmodified silica nanoparticles also showed a significant reduction in membrane binding when membrane asymmetry was lost. These nanoparticles also caused noticeable cell deformation. Additionally, unmodified particles disrupted vesicles mimicking the erythrocyte outer leaflet lipid composition. The level of damage and nanoparticle binding to the membrane was diminished in vesicles mimicking the composition of scrambled membranes. Lipid adsorption was discovered as a mechanism for vesicle damage (Bigdelou et al. 2020).
In terms of the evaluation of size impact of nanoparticle on hemocompatibility, silica nanoparticles (d = ∼ 200 nm) induced faster hemolysis than nanowires (d = ∼ 200 nm, l = 1 m or 10 m). The RBC deformability showed no significant difference while the “aggregation index” for RBC aggregation was reduced in short silica nanowires in comparison with other silica nanomaterials. Silica nanoparticles caused larger rheological changes than nanowires (Kim et al. 2016a, b).
The concentration of the nanomaterials also determines the toxicity to RBCs as it was indicated that the 12.5 g/mL or lower concentration of silica nanomaterials did not lead to toxicity for RBCs (Kim et al. 2016a, b). Amorphous silica nanoparticles (SiNPs) lead to dose-dependent hemolysis of mouse erythrocytes in vitro and are taken up by the erythrocytes. SiNPs induced oxidative activity and apoptosis, and increased cytosolic Ca2+, which may explain the hemolytic activity induced by these nanoparticles. SiNPs possibly cause in vivo anemia and circulatory disorders (Nemmar et al. 2014).
The activity of silica nanoparticles can be enhanced through ion doping them with trace elements found in the human body like Ca, Mg, and Cu (Hoppe et al. 2011). Mg and Cu added to silica-based nanoparticles improved hemocompatibility by protecting erythrocytes from oxidative stress in comparison with pure silica nanoparticles (Tsamesidis et al. 2020).
Nanoparticles are immediately coated with a layer of proteins in contact with biological fluids like blood plasma which is called protein corona (Lundqvist et al. 2017). Compared with nanoparticles with a preformed corona on their surfaces, bare silica nanoparticles exhibit higher adhesion to the cell membrane and a higher internalization efficiency (Lesniak et al. 2012). It has been demonstrated that as a result of plasma corona formation, the hemolytic activity of silica nanoparticles of various sizes will be hindered (Shi et al. 2012).
Researchers indicated that in addition to pore size and morphology, mesoporous silica nanoparticles (MSNs) activity in RBCs will be affected by the formation of the protein corona which significantly reduces hemolysis activity. Ma et al. proved the hemocompatibility of MSNs for blood-compatible nanomaterials' production (Ma et al. 2014). MSN-SiOH, a spherical mesoporous silica nanoparticle with silanol groups on the outside surface, induced spherical protein corona formation during hemolysis. In phosphate buffer saline solution, this has been proven cytotoxicity to RBCs when dispersed as a colloid. The hemolytic effect caused by MSN-SiOH in a dose-dependent manner can be reduced by human blood proteins, such as albumin, plasma, hemoglobin, and RBC lysate. As a result, the toxicity and bioreactivity of silica nanoparticles must be interpreted as a function of the protein corona formation (Martinez et al. 2015).
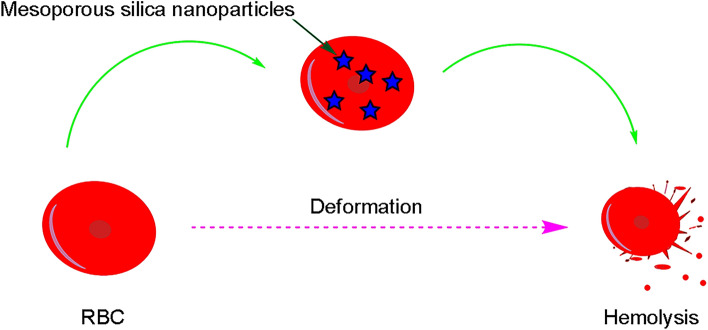
Several factors affect the hemolytic effect of MSNs, including their shape, size, surface charge, surface modification, and surface roughness. Bare MSNs induce the hemolysis of RBCs (Mukhopadhyay et al. 2019), as shown in Fig. 1.
Fig. 1.
The interaction of bare mesoporous silica nanoparticles with red blood cells. Bare MSNs lead to the hemolysis of RBCs
Recent research has shown that surface modification can effectively reduce the hemolytic activity of bare MSNs (He et al. 2010a, b; Yildirim et al. 2013). For instance, the cRGD functionalized Zein has an anti-hemolysis effect and prevents the damage of Au on SiO2 surface to RBCs through altering the protein conformation during the interaction with Au on SiO2 surface (Huang et al. 2021). A synthesized asymmetric MSNs with an adjustable head–tail structure exhibited significantly lower hemolytic activity than other conventional symmetric nanoparticles. The superb hemocompatibility of these asymmetrical head–tail MSNs was because of the low level of reactive oxygen species, flat contact with the RBC membrane, and reduced distortion of RBC (Abbaraju et al. 2017). It seems that the modified silica nanoparticle in comparison with unmodified type indicates more hemocompatibility caused by its morphology and reduced ROS.
Graphene
Graphene is a two-dimensional material that is composed of hexagonally arranged sp2-hybridized carbon atoms with a large surface area on either side of the planar axis. Graphene has remarkable features of electrical and thermal conductivity, and light absorption. Due to graphene's particular surface area, structural and mechanical properties, graphene and its derivatives including mono- and few-layer graphene, graphene oxide (GO), reduced graphene oxide (rGO), graphene nanosheets (GNS), etc. have been widely used for biomedical purposes, such as antibacterial agents, drug delivery, biosensors, and tissue engineering. However, several factors influence the toxicity of the graphene family such as size, functionalization, surface area, charge, and corona formation. Furthermore, each type of graphene has remarkably different physical and chemical properties, which leads to different toxicological effects. To make graphene materials safe for use in biomedical applications, safety evaluations are imperative (Ealia and Saravanakumar 2017; Ou et al. 2016; Seabra et al. 2014).
It was demonstrated that there was no significant toxicity difference between pristine and functionalized graphene (p–G and f–G) in human primary blood components and both types of graphene exhibited high compatibility with RBCs and did not disrupt the RBC plasma membrane (Sasidharan et al. 2012). In contrast, in another study, GN, GO, and rGO caused different levels of structural damage to the membrane of chicken embryo RBC and dose-dependent hemolysis according to methods of production and surface modification. Hydrophobic and reduced graphene nanoparticles (GN and rGO) demonstrated more toxic effects than the hydrophilic, oxidized form. Moreover, GN generated by physical exfoliation had higher hemolytic activity than rGO produced chemically (Jaworski et al. 2017).
In another investigation of the influence of different forms and concentrations of graphene quantum dots (GQDs) on human RBCs, the findings were indications to insignificant hemolysis and rheological changes of the RBCs at concentrations under 500 mg/mL. The carboxylated GQDs led to more substantial hemolytic activity and caused abrupt alterations in RBC deformability and aggregation than the non-functionalized or hydrophilic GQDs at concentrations > 750 mg/mL (Kim et al. 2016a, b).
The unmodified GO nanosheets cause severe damage to RBCs and displayed diverse hemolytic properties under several incubation conditions (Wang et al. 2016). The hemolysis and toxicity of GO appear to be associated with the hydrophilic edges and hydrophobic bases of its chemical structure, which lead to strong interactions between GO and the cell plasma membrane (Kim et al. 2010). GO interacts with neutral and negatively charged lipid membranes and the binding is diminished beyond a certain concentration of negatively charged lipids and favored in high-salt buffers. It was also demonstrated that neutral membrane vesicles tend to break down and extend over the GO after this binding. In contrast, vesicles with negatively charged membranes tend to remain bound without disrupting the GO. GO also interacted with RBCs, and led to hemolysis. It seems that hemolysis is suppressed when GO is previously coated with lipid membranes, especially with pure phosphatidylcholine vesicles (Monasterio et al. 2017).
Functionalized graphene is more compatible with human RBCs by decreasing hemolysis. For enhanced hemocompatibility and particularly the hemolysis level of GO nanosheets, the conjugation of bovine serum albumin (BSA) with GO was suggested. The GO-g-BSA surface reduced the adhesion of RBC in a flowed condition, maintained the morphology of RBC, and decreased hemolysis effectively. Inert BSA resisted GO from interacting with RBC lipid bilayers, and the negative charge on BSA repelled negative charged RBCs from approaching (Cai et al. 2015). It was also proposed that functionalization of graphene nanoplatelets with dextran (GNP-Dex) shows no adverse effects on the hematological components and hemolysis of blood cells (Chowdhury et al. 2013). In consensus, the toxicity of functionalized graphene oxide with D-mannose to human RBCs and the amount of the proteins in the hard corona was diminished significantly (de Sousa et al. 2018). It was also proposed that the reduction process of graphene oxide by primary amines is an effective approach for achieving in vivo biocompatibility for biomedicine applications. Researchers also synthesized a novel nanomaterial based on GO and mesoporous amino silica nanoparticles (H2N-MSNs) that significantly demonstrated diminished erythrocyte lysis and interaction with proteins in human plasma (Fonseca et al. 2018).
Nanodiamonds
Nanodiamonds (NDs) are a newly discovered class of carbon-based nanoparticles whose core is basically composed of carbon sp3 structures with sp2 and defect carbons on the surface. These nanoparticles possess attractive chemical, physical and optical properties. They have gained wide popularity due to their low toxicity, adjustable size ranges, high hardness, photostability, and large surface area which can be functionalized easily. These properties lead to various applications of nanodiamonds in photodevices, biosensors, bio-imaging, energy storage, and drug delivery. Moreover, the characteristics of nanodiamonds can be enhanced through doping and surface modifications for the purpose of biomedical usages (Fedel 2020; Ho et al. 2015; Perevedentseva et al. 2013; Qin et al. 2021; Wąsowicz et al. 2017; Xing and Dai 2009). An in vitro study indicated the hemocompatibility of unmodified and oxygen and hydrogen-terminated surface-modified NDs with no hemolysis observed (Wąsowicz et al. 2017).
Nanodiamonds influence RBCs' deformability and aggregation in a concentration-dependent way. The size of nanodiamonds also affects their interaction with RBCs. In an in vitro study, 100-nm cNDs were found to be localized around RBCs, whereas 5-nm cNDs penetrated into the RBCs. Nanodiamonds did not cause hemolysis and did not alter cell viability or oxygenation/deoxygenation (Lin et al. 2012). In another study it was demonstrated that in contact with 100 nm carboxylated nanodiamonds, some RBCs form echinocytes which can be related to the intercellular contacts induced by the surface charge of the NDs and plasma membrane alteration. Avsievich et al. indicated that nanodiamonds lead to high aggregation strength of RBCs and therefore the formation of large cell aggregates (Avsievich et al. 2019). NDs (dimensions of 35–500 nm) produced using high-pressure, high-temperature (HPHT) methods and purified through air oxidation and strongly oxidizing acids, were not cytotoxic, nor did they have significant hemolytic and thrombogenic effects. Based on this study, SP3-carbon-based nanoparticles could be used to deliver drugs for the treatment of blood-related diseases and for other biomedicine applications (Li et al. 2013). More research has to be conducted for the application of these nanoparticles for blood stability purposes.
Fullerene
Fullerenes are a class of carbon allotropes composed of sp-bonded carbon atoms which are arranged in the form of hollow spheres, ellipses, or tubes. The different types of fullerenes include alkali doped fullerenes, endohedral fullerenes, endohedral metallofullerenes, exohedral fullerenes, and heterofullerenes. The most common fullerene is C60, which is symmetrical, spherical, and formed by joining carbon atoms together by sp2-hybridization and is popular for its mechanical strength, electrochemical characteristics, thermal and photoconductivity. The fullerene exhibits a variety of photo, physical, and electrochemical properties, including ionization potential, semiconducting band gap, binding energy, and enthalpy. In the biomedical field, these nanoparticles are used for anti-retroviral and antibacterial drugs, tumor therapy, free radical scavenging, MRI contrast agents, and drug carriers (Bakry et al. 2007; Bhatia 2016; Castro et al. 2017; Dellinger et al. 2013; Ealia and Saravanakumar 2017; Gogotsi 2015).
The hydrophobic structure of bare fullerenes results in cytotoxicity and must be altered to improve its solubility in polar solvents and to acquire specific functions (Castro et al. 2017). Functionalization of the surface of carbon nanomaterials is effective in their interplays with biological systems and proper surface coating reduces the side toxicity. C60 fullerenes that were functionalized with higher molecular weight polyethylene glycols demonstrated more biocompatibility and all the evaluated nanomaterials were found to diminish the cellular metabolic activity while two only interfered with the plasma membrane integrity, and none caused hemolysis and were safe for RBCs (Canapè et al. 2015).
Carbon nanotube
Carbon nanotubes (CNTs) are cylindrically shaped rolled-up graphene sheets that depend on the layer counts classified into single-walled carbon nanotubes (SWCNTs), with single graphene sheets, and multi-walled carbon nanotubes (MWCNTs), with two or more graphene layers. These materials have significant strength, flexibility, particular structure and size, and distinct chemical, thermal and electrical conductivity. By functionalizing these materials, their biocompatibility, solubility, penetration into cell cytoplasm and nuclei, and efficacy as genetic material and peptide carriers are improved (Bhatia 2016; Ealia and Saravanakumar 2017; Fedel 2020; Zhang et al. 2010). In addition, TPU-g-PEG/CNT (thermoplastic polyurethane-g-polyethylene glycol/carbon nanotube) nanofibers exhibited antibacterial function and superior hemocompatibility, including a reduction in blood cell adhesion and lower hemolysis rates (Shi et al. 2016).
Single-walled carbon nanotubes
Increased concentration of SWNTs or incubation time resulted in diminished RBC aggregation. Higher hemolytic activity of the bundled SWNTs compared to individual SWNTs was observed. In addition, bundled SWNTs significantly changed the shape and fusion of RBCs. Altogether, bundled SWNTs were more toxic than individual ones and these data may be used for evaluation of the risk of nanomaterial toxicity in the blood (Heo et al. 2017).
Multi-walled carbon nanotubes
A higher N atomic percentage led N+-bombarded MWCNTs led to enhanced hemocompatibility. MWCNTs containing nitrogen improved cell proliferation and growth and hemolysis was not observed on the surfaces (Zhao et al. 2014).
Quantum dots
Carbon quantum dots (CQDs) are biocompatible fluorescent nanomaterials based on carbon. CQDs which are smaller than 10 nm, have an organic coating on top of an inorganic semiconductor core to enhance optical properties. A cap covers quantum dots, which makes them more soluble in aqueous buffers. These materials are popular for high stability, low toxicity, rapid and low-cost production, and surface functionalization. These carbon nanostructures are used for various biological aims such as immunoassays, biosensors, gene, protein, and drug delivery, photosensitizers, bio-imaging, and antibacterial agents (Abd Rani et al. 2020; Bhatia 2016; Devi et al. 2019; El-Shabasy et al. 2021; Molaei 2019).
Cryopreservation of RBCs by freezing and adding high concentrations of cryoprotectants, (e.g., glycerol and trehalose) can be utilized to enhance the shelf life limitation of conventional RBCs storage in refrigerators and decreasing aging storage lesions. Nonetheless, intracellular ice crystals formation during the freezing process and osmotic changes can result in RBC membrane damages (Carneiro Dias 2019). Conventional cryoprotectants based on solvents do not prevent ice recrystallization (Deller et al. 2015). Antifreeze proteins have the ability to prevent ice growth and freezing damages but they are expensive and often unstable. In this order, researchers synthesized oxidized quasi-carbon nitride quantum dots (OQCNs) with thermal-hysteresis activity, ice-crystal shaping, and anti-recrystallization effects. It was demonstrated in the cryopreservation of sheep RBCs that the use of OQCNs increased the amount of cell recovery by over two times compared to using a commercial cryoprotectant (hydroxyethyl starch) without added organic solvents. This study suggested the utility of graphitic carbon nitride derivatives for more effective cryopreservation (Bai et al. 2017).
Colloidal apatite nanoparticles
Apatite is a group of inorganic compounds found primarily in vertebrate tissues. Apatite nanoparticles have been developed for nanomedicine applications. Colloid stabilized apatite nanoparticles demonstrated excellent biocompatibility, low cytotoxicity, low inflammatory potential, and high hemocompatibility. Moreover, NPs interact with phospholipid membranes strongly, allowing for protein and drug delivery (t et al. 2017; Okada and Matsumoto 2015).
The delivery of cryoprotectants into RBC can be eased by using apatite nanoparticles which induce local alteration of the RBC membrane. The interaction of these nanoparticles with RBC is affected by the size and charge of the apatite particles. In this case, researchers developed a glycerol-free cryopreservation method, using colloidal apatite NPs as bioactive promoters of trehalose-mediated RBC cryopreservation. In this study, 2-aminoethylphosphate was used for enhanced stability and hexametaphosphate anions for the dispersibility and purification stability of colloidal apatite NPs. Apatite NP significantly increased RBC cryosurvival, reaching 91%, which is comparable to protocols using glycerol. Moreover, the concentration of NPs and the incubation conditions affected the bioactivity of NP strongly. Apatite NPs do not cross the bilayer, but rather modulate its physical properties that trehalose and a fluorescent model molecule permeated through the membrane as a result of these changes. This study suggested a novel alternative method to using toxic glycerol for cells cryopreservation (Stefanic et al. 2017).
Hydroxyapatite particles
Hydroxyapatite (Ca10(PO4)6(OH)2, HAP) exhibits superior biocompatibility, biodegradability, nontoxicity, bioactivity, non-inflammatory and non-immunogenicity activity. Therefore, they can be used for bio-imaging and the delivery of genes, anticancer drugs, radioisotopes, antibiotics, and proteins (Han et al. 2012; Loo et al. 2010). It seems that the size and charge of HAP particles influence the aggregation of the RBCs. HAP nanoparticles despite HAP microparticles induced some aggregation of the RBCs in the unstructured agglutinates. The adhesion of HAP nanoparticles to the surface membrane of the RBCs was in the case of their higher adsorption capacity compared to HAP microparticles, causing their surface membranes to become sunken without damaging the lipid bilayer. The aggregation of the RBCs induced by the HAP nanoparticles was suppressed due to highly negatively charged HAP nanoparticles after heparin modification. RBC aggregation may be caused by the electrostatic interaction between the positively charged binding sites on the surface of the HAP nanoparticle and the negatively charged groups on the surface of the RBC (Han et al. 2012).
The results and main effects of non-metal NPs on RBCs and hemocompatibility studies are summarized in Table 1.
Table 1.
Biocompatibility studies of nanoparticles with RBC
| Row | Nanoparticle type | Surface modification | Nanoparticle size (nm) | Effects | Study type | References |
|---|---|---|---|---|---|---|
| Silica | ||||||
| 1 | Silica | Unmodified, amine and carboxyl groups |
Unmodified: 39.2 ± 10.3 amine-modified: 38.0 ± 9.1 carboxyl-altered: 38.0 ± 12.6 |
Unmodified silica nanoparticles: less damage, reduction in membrane binding in membrane asymmetry loss, cell deformation, disruption of vesicles mimicking the erythrocyte outer leaflet lipid composition. no significant hemolysis by amino- and carboxyl-modified particles | In vitro | Bigdelou et al. (2020) |
| 2 | Silica | – | Nanoparticles: d = 200 nanowires: d = 200 nm, l = 1 m or 10 m |
Fasterhemolysis by nanoparticles than nanowires reduced RBC aggregation in the short silica nanowires compared with others |
In vitro | Kimet al. (2016a, b) |
| 3 | Amorphous silica | – | 50 nm | Dose-dependent hemolysis, oxidative activity, apoptosis and increased cytosolic Ca2+ in RBCs | In vitro | Nemmar et al. (2014) |
| 4 | Ion-doped silica | Calcium, magnesium and copper ions |
100% SiO2: 2.05 ± 0.39 60–40% SiO2–CaO: 2.05 ± 0.33 60–30– 10% SiO2–CaO–MgO,: 1.52 ± 0.27 60–30-7.5–2.5% SiO2–CaO–MgO–CuO: 5.7 ± 1.06 |
Improved hemocompatibility by Mg and Cu added to silica in comparison with pure silica nanoparticles | In vitro | Tsamesidis et al. (2020) |
| 5 | Amorphous silica nanoparticles | – | 7–14 (hydrophilic vs. hydrophobic), 5–15, 20 and 50 | Hindered hemolytic activity as a result of corona formation | In vitro | Shi et al. (2012) |
| 6 | Meso/macroporous silica particles | Pore diameters of 40 and 170 | ||||
| 7 | Sphere- and rod-shaped Mesoporous Silica | – |
Large pore size sphere-shaped: 68 ± 4 Small pore size sphere-shaped: 67 ± 3 Rod-shaped: (107 ± 8) × (343 ± 16) |
Hemocompatibility, pore size and morphology of the MSNs influence the conformation of absorbed HSA and HSF | In vitro | Ma et al. (2014) |
| 8 | Spherical mesoporous silica (MSN-SiOH) | Silanol groups | 115 ± 5 | Spherical protein corona formation during hemolysis, reduced hemolytic effect in a dose-dependent manner by human blood proteins | In vitro | Martinez et al. (2015) |
| 9 | Au@silica | Au, cRGD-Zein |
0%methanol: 150 thickness 16.2%methanol: 215.0 ± 35.0 |
Anti-hemolysis effect of cRGD-Zein | In vitro | Huang et al. (2021) |
| 10 | Asymmetric mesoporous silica | Adjustable head–tail structure | 200 | Superb hemocompatibility, low reactive oxygen species, flat contact to the RBC membrane, and reduced distortion of RBC | In vitro | Abbaraju et al. (2017) |
| Graphene | ||||||
| 11 | GN | – |
GN: 400 nm–1.5 μm |
Dose-dependent hemolysis according to methods of production and surface modification | In vivo | Jaworski et al. (2017) |
| GO |
GO: 100 nm–2.3 μm |
|||||
| rGO |
rGO: 100 nm–1.5 μm, aggregates > 5 μm |
|||||
| 12 | Graphene |
Carboxyl, epoxy, and hydroxyl groups |
Average thickness: ≈ 0.4 | Hemocompatibility of both p-G and f-G | In vitro | Sasidharan et al. (2012) |
| 13 | GQDs | Hydroxyl and carboxyl groups |
Non-functionalized GQDs: 122 hydroxylated/carboxylated GQDs: 190 |
Concentration dependent hemolytic activity and RBC deformability and aggregation | In vitro | Kimet al. (2016a, b) |
| 14 | GO | – | 156 ± 213 | Severe damage to RBCs, diverse hemolytic properties under several incubation conditions | In vitro | Wang et al. (2016) |
| 15 | GO | Lipid bilayers | – | Hemolysis of bare GO, suppressed hemolysis by coated GO with lipid membranes | In vitro | Monasterio et al. (2017) |
| 16 | GO nanosheets | BSA | – | Hemocompatibility | In vitro | Cai et al. (2015) |
| 17 | GNP-Dex | Dextran | 81.3 nm | Hemocompatibility | In vitro and In vivo | Chowdhury et al. (2013) |
| 18 | GO | d-mannose | – | Hemocompatibility | In vitro | de Sousa et al. (2018) |
| 19 | GO-MSN nanocomposite | MSN | 51 ± 8 | Decrease of hemolysis and minimization of the interaction with human plasma proteins | In vitro | Fonseca et al. (2018) |
| Nanodiamonds | ||||||
| 20 | Nanodiamonds | Oxygen and hydrogen-terminated surface-modified NDs |
H2 ND: 5–6 O2 ND: 10–15 Unmodified ND: 10 |
Hemocompatibility | In vitro | Wąsowicz et al. (2017) |
| 21 | Nanodiamonds | – | 5 and 100 | Localization 100-nm cNDs around RBCs, penetration of 5-nm cNDs into the RBCs, no hemolysis and no alteration in cell viability or oxygenation/deoxygenation, RBC deformability and aggregation in a concentration-dependent way | In vitro | Lin et al. (2012) |
| 22 | Nanodiamonds | Carboxyl group | 100 | Echinocyte formation, plasma membrane alteration, higher aggregation strength of RBCs, formation of larger cell aggregates | In vitro | Avsievich et al. (2019) |
| 23 | HPHT-NDs | 35–500 | Not cytotoxic, no significant hemolytic and thrombogenic effects | In vitro | Li et al. (2013) | |
| Fullerene | ||||||
| 24 | Fullerene | PEG, PEI, carboxyl and hydroxyl group | – | No hemolysis | In vitro | Canapè et al. (2015) |
| Carbon nanotube | ||||||
| 25 | TPU-g-PEG/CNT | TPU grafting PEG | – | Hemocompatibility, reduction in blood cell adhesion and lower hemolysis rates | In vitro | Shi et al. (2016) |
| 26 | SWNTs | - |
Diameter of 0.8–1.2 length of 100–1000 |
Diminished RBC aggregation by increased concentration of SWNTs or incubation time, more toxic effect of bundled SWNTs than individual SWNTs | In vitro | Heo et al. (2017) |
| 27 | N+-bombarded MWCNTs | N+ | 40 to 60 | Enhanced hemocompatibility by higher N atomic percentage | In vitro | Zhao et al. (2014) |
| Others | ||||||
| 28 | OQCNs | Oxidized | 10 | Increased recovery times of sheep RBCs cryopreservation compared to using a commercial cryoprotectant | In vitro | Bai et al. (2017) |
| 29 | Colloidal apatite nanoparticles | AEP/HMP | Monomodal distribution centered on d50 ~ 40, with d10 ~ 22 and d90 ~ 80 | Increased RBC cryosurvival | In vitro | Stefanic et al. (2017) |
| 30 | HAP particles | Heparin |
HAP nanoparticles: 18–30 × 45–120 heparin-modified HAP nanoparticles: 10–20 × 40–100 HAP microparticles: 0.15–0.3 μm |
Aggregation of the RBCs by HAP nanoparticles despite HAP microparticles, suppression of RBCs aggregation by highly negatively charged HAP nanoparticles after heparin modification | In vitro | Han et al. (2012) |
Platelets
Platelets play a significant role in blood coagulation and are given to treat hemorrhage or to prevent bleeding in thrombocytopenic patients (Sharma et al. 2011). Platelets can be stored at 20–24 °C in flat bags for up to 5 days with gentle agitation to prevent aggregation and hypoxia. Some procedures for platelet collection may induce partial or complete activation of them. Additionally, during the process of storage at room temperature, platelets continue to metabolize and undergo a range of physiochemical changes that have a detrimental effect on their structure and function and also decrease their shelf life (Greening et al. 2010; Hess 2010a). The bacterial contamination is another challenge of platelet storage (Mathai 2009). The best platelet storage can be achieved by gentle methods of platelet collection, storage in buffered Ringer's acetate that feeds the platelet mitochondria, and storage in polyvinyl chloride bags to maximize oxygen diffusion and stabilize the platelet membranes (Hess 2010a; Mehrizi 2021b; Mehrizi and Eshghi 2021; Wąsowicz et al. 2017; Zadeh Mehrizi and Amini Kafiabad 2021a, b).
Silica
SiNPs hindered ADP-induced platelet-rich plasma aggregation but simultaneously partly activated platelets. Small SiNPs (10 nm) could activate the living cells, while larger ones (up to 40 nm) absorbed fibrinogen and hampered the aggregation of platelets. These findings highlights the importance of the concentration and size of SiNPs in their application in nanotechnology (Gryshchuk and Galagan 2016).
Graphene
For the purpose of investigating the effect of f-G on platelets, both f-G and p-G exhibited compatibility with platelets, and plasma coagulation pathways and did not disrupt the platelets function (Sasidharan et al. 2012). Furthermore, GNP-Dex formulations did not lead to activation of platelets (Chowdhury et al. 2013). Another study on the anticoagulant impact of albumin-functionalized graphene oxide (albumin-GO) was conducted and showed that GO has a high loading capacity for albumin and fibrinogen while not disrupting the secondary structure and conformational stability of albumin. Furthermore, albumin-functionalized GO was shown to have enhanced anticoagulant activity and could potentially be used as an anticoagulant coating material for devices in contact with blood under dynamic flow conditions (Loh and Lim 2015). Researchers have also developed a double-layer-coated vascular stent with anti-proliferation and antithrombosis properties with GO loaded with docetaxel (DTX) internal layer coating. The coating suppresses the adhesion and activation of platelets and also the proliferation and migration of smooth muscle cells, demonstrating good biocompatibility and anti-proliferation ability without the shedding problem (Ge et al. 2019). Du et al. suggested using polyethylenimine (PEI) molecules as linkers, to immobilize l-cysteine onto graphene nanosheets, developing a hemocompatible nitric oxide-generating l-cysteine-grafted graphene film that suppressed the activation, adhesion, and aggregation of platelets (Du et al. 2016).
Fullerene
Anticoagulants are used to inhibit the activation of platelets and coagulation (Prakash and Thakur 2011). The application of hydrated fullerenes for thrombolytic enzymes and the development of novel anticoagulants was suggested. The data from an in vitro study demonstrated the increased cleavage of the blood plasma clot in the presence of hydrated C60 fullerene (C60HyFn). Biological activity of very small C60HyFn doses was mediated by specific and ordered aqueous structures that the C60 molecule organizes around it and not the biological properties of the C60 fullerene molecule itself. In addition, it was also shown that the activation of fibrinolysis may happen because of the stabilization/protection of active conformation of protease molecules in the presence of specific aqueous structures ordered by C60HyFn (Andrievsky et al. 2010).
Nanotubes
The hemocompatibility and cytotoxicity of halloysite nanotubes (HNTs) was studied in vitro for biomedical usage. The results showed the non-hemolysis effect of HNTs. In addition, HNTs shortened the plasma recalcification time in a dose-dependent manner and exhibited procoagulant activity with citrated plasma. Micrographs taken with SEM demonstrated that HNTs could activate platelets in vitro. Overall, HNTs indicated good cell compatibility over a low concentration range (Liu et al. 2015).
Single-walled carbon nanotubes
SWCNTs activated platelets and exhibited prothrombotic effects in microcirculation in vivo. SWCNTs remarkably enhanced the expression of platelet P-selectin, the number of platelet-granulocyte complexes, and platelet aggregability in vitro, and diminished the occlusion time in mesenteric arteries and cremasteric arterioles (Bihari et al. 2010).
Multi-walled carbon nanotubes
MWCNTs infiltrate the plasma membrane of platelets and interact with the dense tubular system leading to depletion of their intracellular stores of Ca2+. This process is followed by the clustering of the stromal interaction molecule 1 colocalized with Orai1, which demonstrates the activation of store-operated Ca2+ entry. Researchers indicated the molecular mechanism of CNT-induced platelet activation which is essential to assess the biocompatibility of carbon nanomaterials with blood (De Paoli Lacerda et al. 2011). MWCNTs bombarded with N+ that had a higher nitrogen content, demonstrated better cytocompatibility and less platelet adhesion (Zhao et al. 2014).
The properties of the protein that forms the corona have also a significant impact on the outcome of the interaction of carboxylated-MWCNTs (CNTCOOH) with platelet. Platelet aggregation and the release of platelet membrane microparticles (PMPs) were activated by bare carboxylated-MWCNTs (CNTCOOH). The platelet aggregating activity of CNTCOOH was reduced by HSA corona, while fibrinogen attenuated the effect of CNTCOOH on platelet by agglomeration of CNTCOOH. In contrast, the IgG and the histone H1 corona enhanced the release of PMP, respectively, by inducing platelet fragmentation, and aggregation of platelet (De Paoli et al. 2014).
Carbon dot
Carbon dots (CDOTs) with diameters less than 10 nm, have appealing photo-physical properties, an easy synthetic process, and facile surface modification. They possess superb biocompatibility and low toxicity due to their biological features such as photoluminescence, organic carbon sources, and high stability in water media. These nanoparticles can be used for bio-imaging, drug and gene delivery, bio-sensing, and photothermal and photodynamic therapy applications (Fedel 2020; Gayen et al. 2019; Ghosal and Ghosh 2019; Zuo et al. 2015).
Lee et al. synthesized a new type of CDOTNP from fresh garlic cloves and investigated its potential role as a new anti-platelet agent. This NP exhibited a suppressive impact on collagen-stimulated human platelet aggregation by inhibiting PKC activation and Akt, JNK1/2, and p38 MAPK phosphorylation but it did not cause cytotoxic effects. The in vivo study on mice models indicated that this CDOT could diminish mortality in mice with ADP-induced acute pulmonary thromboembolism. Overall, this CDOT is effective against platelet activation in vitro via reduction of the phospholipase C/PKC cascade, which results in the inactivation of MAPK. This CDOT can be used for therapeutic purposes for arterial thromboembolic disorders (Lee et al. 2020).
Table 2 summarizes the hemocompatibility studies of non-metal NPs on platelets.
Table 2.
Biocompatibility studies of nanoparticles with platelets
| Row | Nanoparticle | Surface modification | Nanoparticle size | Effects | Study type | References |
|---|---|---|---|---|---|---|
| Silica | ||||||
| 1 | Silica | – | 10–40 | Concentration and size-dependent ADP-induced platelet-rich plasma aggregation suppression and partly activation of platelets | In vitro | Gryshchuk and Galagan (2016) |
| Graphene | ||||||
| 2 | Graphene | Carboxyl, epoxy, and hydroxyl groups | Average thickness: ≈ 0.4 | Compatibility with platelets | In vitro | Sasidharan et al. (2012) |
| 3 | GNP-Dex | Dextran | 81.3 | No activation of platelet | In vitro and in vivo | Chowdhury et al. (2013) |
| 4 | Albumin-GO | Albumin | – | Enhanced anticoagulant activity | In vitro | Loh and Lim (2015) |
| 5 | GO | Docetaxel | – | Antiproliferation and antithrombosis | In vitro and in vivo | Ge et al. (2019) |
| 6 | Graphene nanosheets | PEI, l-cysteine | 4–5 | Suppression of the activation, adhesion, and aggregation of platelets | In vitro | Du et al. (2016) |
| Fullerene( | ||||||
| 7 | C60 Fullerene | Hydrated |
Single C60HyFn: 1.6–1.8 13 hydrated C60: 3.4 |
Clot lysis effect | In vitro | Andrievsky et al. (2010) |
| Nanotubes | ||||||
| 8 | HNTs | - |
Length: 0.2–1 µm Diameter: 20–50 Inner diameter: 10–20 nm |
Nonhemolysis effect and procoagulant activity in a dose-dependent manner | In vitro | Liu et al. (2015) |
| 9 | SWNTs | – | – | Activation of platelets and prothrombotic effects in vivo, enhanced expression of platelet P-selectin, the number of platelets–granulocyte complexes, and platelet aggregability in vitro | In vitro and in vivo | Bihari et al. (2010) |
| 10 | MWCNTs | – | – | Platelet activation by depletion of platelet intracellular stores of Ca2+ | In vitro | De Paoli Lacerda et al. (2011) |
| 11 | MWCNTs bombarded with N+ | N+ | – | Better cytocompatibility and less platelet adhesion by higher nitrogen content | In vitro | Zhao et al. (2014) |
| 12 | MWCNTs | Carboxyl group | Outer diameters: 60–100 nm lengths: 1–2 µ m | Significant impact of the properties of the protein corona on the outcome of the interaction with PLT, Activation of PLT aggregation and the release of PMPs by Bare CNTCOOH, reduced PLT aggregating activity by HSA corona, attenuation of the effect of CNTCOOH on PLT by agglomeration of CNTCOOH by FBG, enhanced release of PMP by IgG and the H1 corona, respectively, by inducing PLT fragmentation, and aggregation of PLT | In vitro | De Paoli et al. (2014) |
| Carbon dot | ||||||
| 13 | Carbon dot | – | 3 | Suppression of platelet aggregation via reduction of the phospholipase C/PKC cascade and the inactivation of MAPK | In vitro and in vivo | Lee et al. (2020) |
Plasma proteins
A variety of life-saving proteins are made from human plasma including albumin, coagulation factors (e.g., factor VIII and IX, prothrombin, fibrinogen, etc.), immunoglobulins, protease inhibitors, and anticoagulants. These products are administrated to patients who lack blood components or with active bleeding (Burnouf 2018). Plasma is often preserved frozen, at − 18 ℃ for one year or at − 65 ℃ for seven years. After thawing, it can be kept at 1–6 ℃ for 5 days (Hess 2010a). However, storage of plasma for more than 7 days at – 20 ℃, will lead to the alteration of metabolites concentration and protein aggregation through damaging reactions and enzymatic activities (Pinto et al. 2014).
Serum albumin
Albumin, the most frequent protein in plasma, plays a critical role in the maintenance of blood osmotic pressure and pH, and the translocation and disposition of various proteins, metabolites, and fatty acids. The function of albumin is determined by the affinity of different ligands to it (He and Carter 1992). Despite the wide application of nanomaterials, their interaction study with specific proteins or biological systems is crucial for their effective and safe utilization (Žūkienė and Snitka 2015). It was indicated that there is a strong interaction of carbon nanoparticles with BSA and HSA. As a result of this interaction, partial unfolding and significant changes in protein conformation was observed which may alter the stability and transport properties of BSA and HSA in various biological processes and indicates the possible risk of carbon nanoparticles for bio-application (Mandal et al. 2013).
Over the past few years, mesoporous silica nanoparticles with pore sizes between 2 and 50 nm, have gained a lot of attention. Their outstanding features of easy independent functionalization of the surface, cost-effective synthesize, large surface area, stable aqueous dispersion, and favorable biocompatibility and biodegradability make them a potential nanoparticle for biomedical purposes. Particle size, pore size, and morphology of MSNs can be modulated as required (Jafari et al. 2019; Narayan et al. 2018). The pore size and morphology of the MSNs influence the conformation of absorbed HSA. Adsorption capacity will be affected by the conformational changes of the adsorbed proteins. Nevertheless, the initial adsorption rate is determined only by the properties of MSNs and proteins (Ma et al. 2014).
Graphene
Researchers found a significant binding interaction between BSA and GO through weak bonds, which resulted in alterations in the conformation of BSA (Kuchlyan et al. 2015; Nan et al. 2019). Multi-non-covalent bonds between BSA and GO surface leads to the change of the conformation and some activities of BSA which results in the diminished thermal stability of BSA on the GO surface. Additionally, the Esterase-like activity and the Non-enzymatic glycosylation of BSA increase on the GO surface while the ability of BSA in drug binding decreases on the GO surface (Zhang et al. 2018). Figure 2 summarizes the results of the binding of graphene oxide to BSA.
Fig. 2.
The results of graphene oxide binding to bovine serum albumin
In another study, the results of calorimetry indicated that there is a strong interaction between GO and free HSA that preserves free HSA from thermal shocks at low ionic strength. The reduced ζ-potential of HSA/GO complex, in comparison with free GO, supports the results. However, the ionic strength plays a key role in the binding of HSA and GO that this interaction is hindered in blood plasma. Docking analysis demonstrated that there is a significantly higher affinity between HSA and GO in its partly unfolded structure at low-ionic-strength compared to native protein conformation at physiological conditions. The results indicate that GO does not cause a toxic effect under physiological conditions in blood plasma (Taneva et al. 2021).
Moreover, the hemocompatibility of p-GO and f-GO (GO-COOH and GO-PEI) with HSA was tested in vitro. The findings demonstrated the considerable toxicity of p-GO and GO-PEI which led to malfunctioning of HSA while the GO-COOH had minor effects on HSA's conformation and its ability of bilirubin binding was unaffected (Ding et al. 2014). The strong hydrogen bonds between GQDs and HSA was observed due to compact structure of HSA but the conformation of albumin was not altered significantly (Ba et al. 2020). In contrast, another study proved the binding of GQDs to site I of HSA and their complex formation and GQDs led to conformational changes in HSA that indicated the possibility of toxicity of GQDs (Huang et al. 2015).
Researchers suggested the usage of nanocomposites instead of individual nanoparticles for enhanced efficacy of them in biomedical applications. The effect of GO-based nanocomposites on BSA have been studied (Baral et al. 2020; Naskar et al. 2017). Naskar et al. demonstrated the interaction of synthesized ZnO graphene nanocomposite with BSA and suggested the application of these nanocomposites for biomedicine (Naskar et al. 2017). The secondary structure of BSA was altered insignificantly by nanocomposites of graphene oxide with reduced manganese dioxide content (MnO2 on RGO). The percentage of GO in NCs influenced the molecular interplays between NC and BSA. BSA esterase-like activity was slightly affected after adsorption on NCs. Aggregates of various sizes were formed from the protein-NC complexes consisting of nanorod and sheet-like shapes (Baral et al. 2020).
Fullerene
The biological activity of pristine C60 is associated with its concentration, conformation, water solubility, and stability. Due to the poor aqueous solubility of C60, the biological functions are significantly decreased (Liu et al. 2004). Wu et al. proposed a direct way to solubilize pristine C60 by using BSA directly as a solubilizer. However, solubilized C60 remained cytotoxic (Wu et al. 2011).
C60 nanoparticles are able to quench the fluorescence of serum albumins in non-specific adsorption. The amino residue microenvironment was minimally altered (only for BSA), whereas slight damage was caused to the protein secondary structure of HSA/BSA (Liu et al. 2012). Another research demonstrated that the binding of nC60 and HSA led to conformational changes of the protein resulting in a compacter structure. The interaction of HSA with nC60 aggregates affected the function of drug-binding sites of HSA (Song et al. 2011). A docking analysis and binding site alignment study indicated that while the HIV protease and serum albumin-binding sites were similar, the fullerene-specific antibody-binding site was different from those observed in others (Benyamini et al. 2006).
The interaction of C60 and a group of proteins that possibly bind to it such as antibody Fab fragment and albumin was investigated. The results provided useful information for a wide variety of bioapplications including anti-inflammatory drugs, antitumor, and antiprotozoal therapies, novel antibiotics, and etc. (Calvaresi and Zerbetto 2010).
Single-walled carbon nanotubes
BSA and SWCNTs formed hydrophobic forces and covalent bonds in solid nanocomposites, dependent on the nanotube diameter utilized. This interaction led to the alteration of the conformation of albumin. Furthermore, there was a decrease in intensity in the high-frequency region of the absorption bands. Additionally, the covalent attachments to the graphene surface led to distortion in SWCNTs (Gerasimenko et al. 2020).
Quantum dot
Some research has been conducted in the investigation of the interaction of QDs with BSA and HSA (Dzagli et al. 2010; Lai et al. 2012; Xiao et al. 2018; Yang et al. 2013). Glutathione-capped CdTe quantum dots (GSH-CdTe QDs) was indicated to form a ground-state complex with trivial changes in the conformation of HSA. This study provided insight into QD bioactivities in vivo and how they can be used to design biocompatible and stable QDs (Yang et al. 2013). Ag2Te QDs and CdTe QDs were shown to statically quench the fluorescence of HSA by electrostatic interactions, but Ag2Te QDs had weaker quenching and binding ability than CdTe QDs. Micro-environmental and structural variations of HSA were induced by their binding interactions with both QDs but Ag2Te QDs led to a less conformational change in HSA, demonstrating the lower toxicity risk (Xiao et al. 2018). As a result of the interaction of CdSe/ZnS core–shell QDs with BSA, QDs on BSA bioconjugates had been formed. The temperature dependence of the QDs on BSA emission was more complex than the monotonically declining fluorescence intensity of non-conjugated QDs and was highly dependent on the protein's conformation (Dzagli et al. 2010).
In the evaluation of the charge effect, a study indicated that negative coated CdTe QDs and HSA formed a protein corona and showed an adsorption behavior while the adsorption of HSA onto the surface of positive coated CdTe QDs led to the aggregation of QDs which can act as the nuclei adsorbing larger amounts of proteins (Lai et al. 2012). Xiao et al. studied the interplays between different charge-capped CdSe/ZnS QDs and BSA. The formation of QDs–BSA complex was observed and the biological activity of BSA was altered as a result of the structural changes of BSA by QDs (Xiao et al. 2012).
Carbon dot
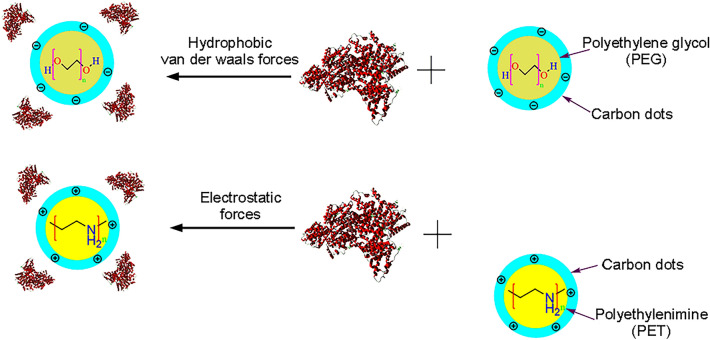
In the investigation of the photochemical interaction of CDs, Maity et al. studied the interaction of the synthesized amine-coated Ru (III) doped carbon nanodots (Ru:CNDEDAs) with BSA and HSA. The results of the circular dichroism study indicated the perturbation of albumin structure by Ru:CNDEDAs. In contrast, the absorption study implied the formation of a ground-state complex between Ru:CNDEDAs and BSA/HSA. The transfer of energy from BSA/HSA to Ru:CNDEDAs was also demonstrated and the efficiency of this energy transfer was highly dependent on the stability of the protein nanoparticle complex (Maity et al. 2019). In another study, dynamic quenching of protein fluorescence was observed and C-dots had no significant effect on HSA or γ-globulin structure. C-dots affected the transport function of proteins by varying the affinity of drugs with HSA and γ-globulins (Xu et al. 2016). It was also reported that the quenching mechanism of nitrogen and three kinds of sulfur co-doped carbon dots (N,S-CDs) was different but All N,S-CDs led to conformational changes in HSA (Liang et al. 2020). The role of surface charge on the interaction of negative PEG C-dots and positive PEI C-dots with HSA was also studied by researchers. The fluorescence of HSA was quenched by both C-dots. The binding affinity of PEG and PEI C-dots to HSA was approximately similar. PEG C-dots interacted with the site I of HSA through electrostatic forces, whereas PEI C-dots interacted with the site II through hydrophobic and van der Waals forces. Moreover, the structure of HSA was insignificantly affected by PEG C-dots. In contrast, only the high content of PEI C-dots caused notable distortion of HSA conformation (Li et al. 2018). Figure 3 indicates the interaction of charged carbon nanodots with HSA.
Fig. 3.
The interaction of charged carbon nanodots with human serum albumin. PEG C-dots interact with the site I of HSA via electrostatic forces whereas PEI C-dots interacted with the site II via hydrophobic and van der Waals forces
Table 3 summarizes the studies on interactions of non-metal NPs and serum albumin.
Table 3.
Biocompatibility studies of nanoparticles with serum albumin
| Row | Nanoparticle | Surface modification | Nanoparticle size | Results | Study type | References |
|---|---|---|---|---|---|---|
| 1 | Carbon nanoparticles | – | 4.0 | Strong interaction of carbon nanoparticles with BSA and HSA | In vitro | Mandal et al. (2013) |
| Silica | ||||||
| 2 | Sphere- and rod-shaped Mesoporous Silica | – |
Large pore size sphere–shaped: 68 ± 4 Small pore size sphere–shaped: 67 ± 3 Rod–shaped: (107 ± 8) × (343 ± 16) |
Conformational change of HSA dependent to the pore size and morphology of the MSNs | In vitro | Ma et al. (2014) |
| Graphene | ||||||
| 3 | GO | – | – | Interaction between BSA and GO and alterations in BSA structure | In vitro | Nan et al. (2019) |
| 4 | GO | – |
Height: 1.2 Average thickness: 4 |
Significant interaction of BSA on the GO surface and conformational changes of BSA after the addition of GO | In vitro | Kuchlyan et al. (2015) |
| 5 | GO | – | – | The conformational change, diminished thermal stability, increased Esterase-like activity and the Non-enzymatic glycosylation, decreased drug binding of BSA | In vitro and in silico | Zhang et al. (2018) |
| 6 | GO | – | 1.0 ± 0.3 μm | Ionic strength-dependent interaction between GO and free HSA, hindered interaction in blood plasma | In vitro | Taneva et al. (2021) |
| 7 | GO | PEI, COOH | – |
Minimal conformational change and HSA’s binding capacity to bilirubin by the binding of GO-COOH, strong toxicity by binding of p-GO and GO-PEI |
In vitro | Ding et al. (2014) |
| 8 | GQDs | – | 2.14 ± 0.05 | Strong hydrogen bonds with HSA, insignificant conformational change | In vitro | Ba et al. (2020) |
| 9 | GQDs | – | 1.2 | Conformational changes and possibility of toxicity of GQDs | In vitro | Huang et al. (2015) |
| 10 | ZnO–rGO | ZnO | 20 | Interaction of ZnO–rGO with BSA | In vitro | Naskar et al. (2017) |
| 11 | MnO2@RGO | MnO2 | – | Insignificantly Changes in secondary structure of BSA, the NC GO percentage dependent interplays of NC and BSA, slight BSA esterase-like activity changes, aggregate forming from the protein-NC complexes | In vitro | Baral et al. (2020) |
| Fullerene | ||||||
| 12 | C60 | – | 20–60 | Using BSA as a solubilizer, sustained cytotoxic effect of Solubilized C60 | In vitro | Wu et al. (2011) |
| 13 | C60 | – | 120–260 | Slight damage to the secondary structure of HSA/BSA | In vitro | Liu et al. (2012) |
| 14 | nC60 | – | – | Conformational changes of the HSA resulting in compacter structure, Alteration of drug binding sites | In vitro | Song et al. (2011) |
| 15 | C60-fullerene and carboxy fullerenes | Carboxyl group | – | Similar binding sites of HIV protease and serum albumin | In silico | Benyamini et al. (2006) |
| 16 | C60 Fullerene | – | – | Antibody Fab fragment and serum albumin as target Candidates | In silico | Calvaresi and Zerbetto (2010) |
| SWCNTs | ||||||
| 17 | SWCNTs | – |
SWCNT I: d = 1.4 ± 1.8 nm, l = 0.3 ± 0.8 mm SWCNT II: 3.2 ± 4 nm, l = 5 mm |
Nanotube diameter dependent hydrophobic forces and covalent bonds of BSA and SWCNTs in solid nanocomposites, alteration of the conformation of albumin, decrease in intensity in the high-frequency region of the absorption bands, distortion in SWCNTs by the covalent attachments to the graphene surface | In vitro | Gerasimenko et al. (2020) |
| Quantum dot | ||||||
| 18 | GSH–CdTe QDs | GSH–CdTe | 1.7, 2.5, 2.8, 3.0 | Ground-state complex with HSA, no significant changes in the conformation of HSA | In vitro | Yang et al. (2013) |
| 19 | Ag2Te QDs and CdTe QDs | Ag2Te and CdTe | 2.9 | Weaker quenching and binding ability of Ag2Te QDs than CdTe QDs, less conformational change in HSA by Ag2Te QDs demonstrating the lower toxicity risk | In vitro | Xiao et al. (2018) |
| 20 | CdSe/ZnS core–shell quantum dots | CdSe/ZnS | 3.3 | Formation of QDs@BSA bioconjugates | In vitro | Dzagli et al. (2010) |
| 21 | CdTe quantum dots | CdTe | 4 | Formation of protein corona by negative QDs and HSA, adsorption of HSA onto the surface of positive QDs and the aggregation of QDs | In vitro | Lai et al. (2012) |
| 22 | CdSe/ZnS quantum dots | CdSe/ZnS |
Negative QDs: 2.8 Positive QDs:2.6 |
Formation of QDs–BSA complex, structural AND biological activity changes of BSA | In vitro | Xiao et al. (2012) |
| Carbon dots | ||||||
| 23 | Ru:CNDEDAs | Amine–coated Ru (III) | – | Perturbation of albumin in circular dichroism study, the formation of a ground-state complex with BSA/HSA in the absorption study, the higher stability of Ru:CNDEDA-BSA than Ru:CNDEDA-HSA | In vitro and in silico | Maity et al. (2019) |
| 24 | C-dots | – | 2.4 ± 0.6 | No significant effect on HSA or γ -globulin structure, impact on the transport function of proteins by varying the affinity of drugs with HSA and γ –globulins | In vitro | Xu et al. (2016) |
| 25 | N,S-CDs | Nitrogen and sulfur | 2.0, 7.6, 13.9 | Conformational changes in HSA |
In vitro and in silico |
Liang et al. (2020) |
| 26 | negative PEG Cdots and positive PEI Cdots | PEG, PEI | 2.5 ± 0.9 | Similar binding affinity of PEG and PEI Cdots to HSA, interaction of PEG Cdots with site I, interaction of PEI Cdots with site II, minimal effect of PEG Cdots on HSA structure, notable distortion of HSA conformation by the high content of PEI Cdots | In vitro | Li et al. (2018) |
Factor VIII
Coagulation factor VIII (FVIII) is a co-factor for the proteolytic activation of factor X by factor IXa (Bhopale and Nanda 2003). This product is used for hemophilia patients. The final quality of this product is a crucial factor and is influenced by the protein purification procedure (Gouw et al. 2013). The in vivo and in vitro stability of FVIII is low and needs frequent drug administration and also lyophilization for long-term storage which leads to challenging self-administration of it for hemophilia patients (Wang et al. 2003). NPs can also be used for the purpose of improved stability of coagulation factors, anticoagulant or procoagulant effects, or as drug carriers. Therefore, the interactions of NPs with the blood coagulation system have to be investigated (Ilinskaya and Dobrovolskaia, 2013; Mehrizi and Hosseini, 2021).
Silica
SiNPs have procoagulant effects and reduce the time of coagulation in activated partial thromboplastin time (APTT) and prothrombin time (PT) tests. SiNPs also increase the factor X activation by RVV in plasma by absorption of intrinsic pathway factors on their surface. SiNPs are introduced as hemostatic agents for therapeutic purposes (Gryshchuk and Galagan 2016).
The effect of the surface morphology of silica nanoparticles on the loading and release of three small, medium, and large size model proteins, respectively, including Myoglobin, BSA, and FVIII was investigated using porous silica nanoparticles with different pore sizes. The results indicated that the release of the small pore silica particles follows a controlled diffusion mechanism, whereas the kinetic analysis can distinguish two components associated with the surface adsorbed protein and the portion of protein allocated to the pores. Particles characterized by the largest pore sizes (approximately 25 nm) exhibit a non-Fickien release profile and mono-exponential dependence, indicating a mono-modal distribution of proteins on the silica matrix. In addition, the conformation or activity of the released proteins was evaluated by in vitro methods and the data showed the superb quality of the recovered proteins. Mesoporous structures of the silica nanoparticles relative to the size of protein confirmed the results (Zampini et al. 2019).
Fullerene
Liu et al. suggested the development of a fullerene-based anticoagulant by impeding the activity of coagulation factor X (FXa). Researchers analyzed the interplays between FXa and fullerene derivatives including fullerenol molecules (C60(OH)24)/carboxy fullerene molecules (C60(C(COOH)2)2) with different hydrophilic–hydrophobic properties using molecular docking. The results demonstrated that C60 (C(COOH)2)2 binds more stably to the active site of FXa than C60 (OH)24 with lower binding energy during competitive absorptions. The uptake of C60(OH)24 is due to hydrophilic interactions, while the uptake of C60 (C(COOH)2) is due to hydrophobic interactions (Liu et al. 2019).
Carbon nanotube
Researchers designed a platform for biocompatible nitrogen-doped carbon nanotube for extraction of plasma from human blood by nanomaterial synthesis with microfabrication. They developed a microfluidic device with a double helix channel with cross-flow filtration. This canal was made of aligned CNTs with an average distance of 80 nm and a porosity value of 93%. While blood extraction through this canal, the larger molecules such as blood cells were trapped and smaller ones like proteins and clotting factors passed through it. It also recovered albumin with an efficiency of 80% (Yeh et al. 2018).
Quantum dots
The anticoagulant effect of 3.2 nm QDs compared to 3.6 nm counterparts in plasma was demonstrated. QDs by affecting the intrinsic pathway increased the coagulation time in APTT and PT tests. The activity of intrinsic pathway factors including Factors VIII, IX, XI, and XII was normal. The Factor VIII activity was reduced to 51% but led to normal hemostasis. The anticoagulant role of QDs was significantly affected by the concentration of calcium ions, but not by cadmium ions leached from the QDs. This role was not dependent on platelets and phospholipids. The results of this study indicated the potential role of the physicochemical properties of the QDs in the modulating of hemostasis and the coagulation cascade, but the underlying mechanism is unclear (Maguire et al. 2018).
Table 4 summarizes the biocompatibility studies of non-metal NPs with plasma factor VIII.
Table 4.
Biocompatibility studies of nanoparticles with plasma factor VIII
| Row | Nanoparticle type | Surface modification | Nanoparticle size | Effects | Study type | References |
|---|---|---|---|---|---|---|
| 1 | Silica | – | 10–40 | Procoagulant effects, increased the factor X activation | In vitro | Gryshchuk and Galagan (2016) |
| 2 | Porous silica | – | 74 ± 11, 83 ± 9 and 94 ± 9 | Controlled diffusion mechanism of the release of the small pore silica particles, a non-Fickien release profile and mono-exponential dependence by the nanaoparticles with largest pore sizes (approximately 25 nm),superb quality of the recovered proteins | in vitro | Zampini et al. (2019) |
| 3 | Fullerenol and carboxy fullerene | Carboxyl and hydroxyl groups | – | Anticoagulant by impeding the activity of FXa, more stably binding of carboxy fullerene to the active site of FXa than fullerenol | In silico | Liu et al. (2019) |
| 4 | Carbon nanotube | Nitrogen | – | Albumin recovery with an efficiency of 80%, biocompability of CNTs | In vitro | Yeh et al. (2018) |
| 5 | Quantum dots | – | 3.2, 3.6 | The anticoagulant effect of 3.2 nm QDs calcium ions concentration dependent, increased coagulation time, reduced Factor VIII activity | In vitro | Maguire et al. (2018) |
Immunoglobulins
Immunoglobulin G (IgG) is present in blood plasma as the second abundant protein after albumin (Strengers 2017). Normal polyvalent IgG contains multiple antibodies and is used in inflammatory and autoimmune diseases and immune deficiencies. Hyperimmune IgG is derived from typically vaccinated donors and exhibits higher titer in neutralizing antibodies against particular antigens such as the D antigen, cytomegalovirus, or hepatitis B or A (Burnouf 2018). The high quality and purity of IgG are crucial for the therapeutic application of this product. Nonetheless, current methods of IgG purification contain multiple steps and are not cost-effective. Thus alternative low-cost and high-efficient approaches are in demand (Azevedo et al. 2007; Ferreira et al. 2016).
Silica
Recently, researchers modified silica nanoparticles with antibodies for different purposes (Moore et al. 2015; Ngamcherdtrakul et al. 2018). Ngamcherdtrakul et al. developed a small-interfering RNA nanoparticle carrier consisting of 50-nm mesoporous silica nanoparticles modified with a copolymer of polyethylenimine and polyethyleneglycol, and antibody. It was demonstrated that the lyophilized material can be stored stably for 2 months at 4 ℃ and at least 6 months at − 20 ℃. The findings suggested this process could be utilized for other similar nanoparticle systems (Ngamcherdtrakul et al. 2018). Moore et al. also synthetized antibody-coated silica NPs (Ab-NPs) using multivalent polyamidoamine dendrimers and dextran as crosslinkers which maintained colloidal stability during NP-linker binding and the subsequent conjugation reaction between linker-coated NPs and proteins to fabricate monodisperse Ab-NPs. It was discovered that freeze-drying Ab-NPs from a 1% BSA solution enabled the colloid in solid form to be reconstituted into a stable state by adding solvent and shaking the sample vial using hand (Moore et al. 2015).
Graphene
The interplays between GQDs and γ-globulins are through electrostatic forces. The presence of binding sites on the surface of γ-globulins results in a high binding constant of these proteins with GQDs. GQDs had a negligible effect on the secondary structures of γ-globulins, suggesting possible biological usage of GQDs at the molecular level (Ba et al. 2020).
There are few studies on the interaction of IgG with non-metal NPs and more investigations are required for enhanced purification of IgG.
Conclusion
Blood products are used in various life-threatening clinical indications such as hemorrhage, anemia, and other blood deficiencies and disorders. This paper discussed the challenges of the shelf-time, stability, and purification of each blood product and provided comprehensive data on the interaction and hemocompatibility of different non-metal nanoparticles with these components and their side effects, to design alternative methods of blood products improvement in the future using nanoparticles. The compatibility of p-G with RBCs was observed by a study but researchers suggested functionalization of it for better compatibility. GQDs also indicated insignificant adverse effects on RBCs in a concentration-dependent manner. Nanodiamonds also showed the hemocompatibility with RBCs in a concentration and size-dependent way that 100-nm cNDs localized around RBCs, whereas 5-nm cNDs penetrated them. But they did not cause hemolysis and did not alter cell viability or oxygenation/deoxygenation. However, some studies demonstrated the aggregation of RBCs by nanodiamonds.
SiNPs hampered ADP-induced platelet-rich plasma aggregation but activated some platelets in a concentration and size-dependent way. The p–G also exhibited compatibility with platelets, and plasma coagulation pathways and did not disrupt the platelets' function. A novel synthesized CDOT NP from fresh garlic cloves exhibited a suppressive impact on collagen-stimulated human platelet aggregation by inhibiting PKC activation and Akt, JNK1/2, and p38 MAPK phosphorylation. It also inactivated the platelets via reduction of the phospholipase C/PKC cascade and inactivation of MAPK in vitro.
There is a strong interaction between GO and free HSA that preserves free HSA from thermal shocks at low ionic strength. Thus, under physiological conditions, GO does not cause toxicity in blood plasma. But other researchers proposed the usage of functionalized GO and GO-based nanocomposites instead of individual nanoparticles for enhanced compatibility with albumin.
In the investigation of loading and release of FVIII, large pore size MSNs resulted in superb quality of the recovered protein due to mesoporous structures of the silica nanoparticles relative to the size of the protein. The anticoagulant effect of 3.2 nm QDs through affecting the intrinsic pathway was also demonstrated. This effect is dependent on calcium ions concentration.
There are electrostatic forces between GQDs and γ-globulins. The presence of binding sites on the surface of γ-globulins results in a high binding constant of these proteins with GQDs. GQDs had a negligible effect on the secondary structures of γ-globulins.
In conclusion, this review study suggests GQDs and nanodiamonds as the most promising nanoparticles for improving the stability of RBCs. For platelets, SiNPs exhibited a suppressive impact on platelet aggregation. Pristine graphene also shows compatibility with platelets and is a potential candidate for increasing the stability of platelets. For better stability of plasma products, GO was indicated to preserve free HSA from thermal shocks at low ionic strength. For increased stability of FVIII, MSNs with large pores exhibited the superb quality of recovered proteins. Additionally, 3.2 nm QDs exhibited anticoagulant effects. As the preferred nanoparticles for immunoglobulin stability, GQDs showed compatibility with γ-globulins.
Altogether, despite some promising reports, most carbon-based nanoparticles does not show good hemocompatibility. However, it seems that nanotechnology may have great potential to improve the blood banking system by controlling physicochemical properties of non-metal nanomaterials and introducing novel alternatives to conventional methods of blood storage. Nevertheless, current information on using nanoparticles for improved blood storage suffers from serious studies which need to be conducted in this field. This review provided a useful vision for the recognition of promising hemocompatible non-metal nanomaterials for enhanced blood banking through novel nanoparticle-based approaches.
Funding
The authors declare that no funding was received for this study.
Declarations
Conflict of interest
The authors declare that there is no conflict of interest to declare.
Ethical approval
This study does not involve any human or animal testing.
Informed consent
Not applicable.
Data deposition information
No datasets have been used.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- Abbaraju PL, Meka AK, Song H, Yang Y, Jambhrunkar M, Zhang J, Xu C, Yu M, Yu C. Asymmetric silica nanoparticles with tunable head–tail structures enhance hemocompatibility and maturation of immune cells. J Am Chem Soc. 2017;139(18):6321–6328. doi: 10.1021/jacs.6b12622. [DOI] [PubMed] [Google Scholar]
- Abd Rani U, Ng LY, Ng CY, Mahmoudi E. A review of carbon quantum dots and their applications in wastewater treatment. Adv Colloid Interface Sci. 2020;278:102124. doi: 10.1016/j.cis.2020.102124. [DOI] [PubMed] [Google Scholar]
- Abonnenc M, Tissot J-D, Prudent M. General overview of blood products in vitro quality: Processing and storage lesions. Transfus Clin Biol. 2018;25(4):269–275. doi: 10.1016/j.tracli.2018.08.162. [DOI] [PubMed] [Google Scholar]
- Andrievsky G, Shakhnin D, Tronza A, Zhernosekov D, Tykhomyrov A. The acceleration of blood plasma clot lysis in the presence of hydrated C60 fullerene nanostructures in super-small concentration. Fuller Nanotub Carbon Nanostruct. 2010;18(3):303–311. doi: 10.1080/15363831003785257. [DOI] [Google Scholar]
- Avsievich T, Popov A, Bykov A, Meglinski I. Mutual interaction of red blood cells influenced by nanoparticles. Sci Rep. 2019;9(1):1–6. doi: 10.1038/s41598-019-44659-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Azevedo AM, Rosa PA, Ferreira IF, Aires-Barros MR. Optimisation of aqueous two-phase extraction of human antibodies. J Biotechnol. 2007;132(2):209–217. doi: 10.1016/j.jbiotec.2007.04.002. [DOI] [PubMed] [Google Scholar]
- Ba X-X, Gao T, Yang M, Jiang P, Jiang F-L, Liu Y. Thermodynamics of the interaction between graphene quantum dots with human serum albumin and γ-globulins. J Solut Chem. 2020;49(1):100–116. doi: 10.1007/s10953-019-00941-8. [DOI] [Google Scholar]
- Bai G, Song Z, Geng H, Gao D, Liu K, Wu S, Rao W, Guo L, Wang J. Oxidized quasi-carbon nitride quantum dots inhibit ice growth. Adv Mater. 2017;29(28):1606843. doi: 10.1002/adma.201606843. [DOI] [PubMed] [Google Scholar]
- Bakry R, Vallant RM, Najam-ul-Haq M, Rainer M, Szabo Z, Huck CW, Bonn GK. Medicinal applications of fullerenes. Int J Nanomed. 2007;2(4):639–649. [PMC free article] [PubMed] [Google Scholar]
- Baral A, Satish L, Das DP, Sahoo H, Ghosh MK. Molecular interactions of MnO2@ RGO (manganese dioxide-reduced graphene oxide) nanocomposites with bovine serum albumin. J Biomol Struct Dyn. 2020;38(7):2038–2046. doi: 10.1080/07391102.2019.1640131. [DOI] [PubMed] [Google Scholar]
- Belousov A. Nanotechnology and discovery of a new factor which influences on permeability of erythrocytes and eryptosis. J Mater Sci Eng. 2014;4(11):367–372. doi: 10.17265/2161-6213/2014.11.008. [DOI] [Google Scholar]
- Belousov A, Malygon E, Yavorskiy V, Belousova E. Stabilization of molecular structure membranes of preserved rbcs by means nanotechnology. Ann Med Surg Case Rep AMSCR. 2019 doi: 10.23880/mjccs-16000204. [DOI] [Google Scholar]
- Benyamini H, Shulman-Peleg A, Wolfson HJ, Belgorodsky B, Fadeev L, Gozin M. Interaction of C60-fullerene and carboxyfullerene with proteins: docking and binding site alignment. Bioconjug Chem. 2006;17(2):378–386. doi: 10.1021/bc050299g. [DOI] [PubMed] [Google Scholar]
- Bhatia S (2016) Nanoparticles types, classification, characterization, fabrication methods and drug delivery applications. In: Natural Polymer Drug Delivery Systems. Springer, pp 33–93
- Bhopale G, Nanda R. Blood coagulation factor VIII: an overview. J Biosci. 2003;28(6):783–789. doi: 10.1007/BF02708439. [DOI] [PubMed] [Google Scholar]
- Bigdelou P, Vahedi A, Kiosidou E, Farnoud AM. Loss of membrane asymmetry alters the interactions of erythrocytes with engineered silica nanoparticles. Biointerphases. 2020;15(4):041001. doi: 10.1116/6.0000246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bihari P, Holzer M, Praetner M, Fent J, Lerchenberger M, Reichel CA, Rehberg M, Lakatos S, Krombach F. Single-walled carbon nanotubes activate platelets and accelerate thrombus formation in the microcirculation. Toxicology. 2010;269(2–3):148–154. doi: 10.1016/j.tox.2009.08.011. [DOI] [PubMed] [Google Scholar]
- Burnouf T. An overview of plasma fractionation. Ann Blood. 2018;3:33. doi: 10.21037/aob.2018.05.03. [DOI] [Google Scholar]
- Cai B, Hu K, Li C, Jin J, Hu Y. Bovine serum albumin bioconjugated graphene oxide: Red blood cell adhesion and hemolysis studied by QCM-D. Appl Surf Sci. 2015;356:844–851. doi: 10.1016/j.apsusc.2015.08.178. [DOI] [Google Scholar]
- Calvaresi M, Zerbetto F. Baiting proteins with C60. ACS Nano. 2010;4(4):2283–2299. doi: 10.1021/nn901809b. [DOI] [PubMed] [Google Scholar]
- Canapè C, Foillard S, Bonafè R, Maiocchi A, Doris E. Comparative assessment of the in vitro toxicity of some functionalized carbon nanotubes and fullerenes. RSC Adv. 2015;5(84):68446–68453. doi: 10.1039/C5RA11489F. [DOI] [Google Scholar]
- Carneiro Dias AE (2019) Study of RBC shape transitions induced by nanoparticles Universitat Rovira i Virgili]. http://hdl.handle.net/10803/668080. Accessed date: RDL 1/1996
- Castro E, Garcia AH, Zavala G, Echegoyen L. Fullerenes in biology and medicine. J Mater Chem B. 2017;5(32):6523–6535. doi: 10.1039/C7TB00855D. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chowdhury SM, Kanakia S, Toussaint JD, Frame MD, Dewar AM, Shroyer KR, Moore W, Sitharaman B. In vitro hematological and in vivo vasoactivity assessment of dextran functionalized graphene. Sci Rep. 2013;3(1):1–10. doi: 10.1038/srep02584. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dashti Rahmat Abadi F, Ebrahimi Shahmabadi H, Abedi A, Alavi SE, Movahedi F, Koohi Moftakhari Esfahani M, Zadeh Mehrizi T, Akbarzadeh A. Polybutylcyanoacrylate nanoparticles and drugs of the platinum family: last status. Indian J Clin Biochem. 2014;29(3):333–338. doi: 10.1007/s12291-013-0364-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Paoli SH, Diduch LL, Tegegn TZ, Orecna M, Strader MB, Karnaukhova E, Bonevich JE, Holada K, Simak J. The effect of protein corona composition on the interaction of carbon nanotubes with human blood platelets. Biomaterials. 2014;35(24):6182–6194. doi: 10.1016/j.biomaterials.2014.04.067. [DOI] [PubMed] [Google Scholar]
- De Paoli Lacerda SH, Semberova J, Holada K, Simakova O, Hudson SD, Simak J. Carbon nanotubes activate store-operated calcium entry in human blood platelets. ACS Nano. 2011;5(7):5808–5813. doi: 10.1021/nn2015369. [DOI] [PubMed] [Google Scholar]
- de Sousa M, Martins CH, Franqui LS, Fonseca LC, Delite FS, Lanzoni EM, Martinez DST, Alves OL. Covalent functionalization of graphene oxide with d-mannose: Evaluating the hemolytic effect and protein corona formation. J Mater Chem B. 2018;6(18):2803–2812. doi: 10.1039/c7tb02997g. [DOI] [PubMed] [Google Scholar]
- Deller RC, Vatish M, Mitchell DA, Gibson MI. Glycerol-free cryopreservation of red blood cells enabled by ice-recrystallization-inhibiting polymers. ACS Biomater Sci Eng. 2015;1(9):789–794. doi: 10.1021/acsbiomaterials.5b00162. [DOI] [PubMed] [Google Scholar]
- Dellinger A, Zhou Z, Connor J, Madhankumar A, Pamujula S, Sayes CM, Kepley CL. Application of fullerenes in nanomedicine: an update. Nanomedicine. 2013;8(7):1191–1208. doi: 10.2217/nnm.13.99. [DOI] [PubMed] [Google Scholar]
- Devi P, Saini S, Kim K-H. The advanced role of carbon quantum dots in nanomedical applications. Biosen Bioelectron. 2019;141:111158. doi: 10.1016/j.bios.2019.02.059. [DOI] [PubMed] [Google Scholar]
- Ding Z, Zhang Z, Ma H, Chen Y. In vitro hemocompatibility and toxic mechanism of graphene oxide on human peripheral blood T lymphocytes and serum albumin. ACS Appl Mater Interfaces. 2014;6(22):19797–19807. doi: 10.1021/am505084s. [DOI] [PubMed] [Google Scholar]
- Drouet C, Fleury JB, Stefanic M, Choimet M, Al-Kattan A, Kim HM, Oh JM, Tourrette A, Santran V, Pipy B. Colloidal apatite nanoparticles: insights on their interaction with cells and artificial lipid membranes. Key Eng Mater. 2017;720:95–101. doi: 10.4028/www.scientific.net/KEM.720.95. [DOI] [Google Scholar]
- Du Z, Dou R, Zu M, Liu X, Yin W, Zhao Y, Chen J, Yan L, Gu Z. Nitric oxide-generating L-cysteine-grafted graphene film as a blood-contacting biomaterial. Biomater Sci. 2016;4(6):938–942. doi: 10.1039/c6bm00074f. [DOI] [PubMed] [Google Scholar]
- Dzagli MM, Canpean V, Iosin M, Mohou MA, Astilean S. Study of the interaction between CdSe/ZnS core-shell quantum dots and bovine serum albumin by spectroscopic techniques. J Photochem Photobiol a: Chem. 2010;215(1):118–122. doi: 10.1016/j.jphotochem.2010.08.008. [DOI] [Google Scholar]
- Ealia SAM, Saravanakumar M. A review on the classification, characterisation, synthesis of nanoparticles and their application. IOP Conf Ser Mater Sci Eng. 2017;263:032019. doi: 10.1088/1757-899X/263/3/032019. [DOI] [Google Scholar]
- El-Shabasy RM, Elsadek MF, Ahmed BM, Farahat MF, Mosleh KM, Taher MM. Recent developments in carbon quantum dots: properties, fabrication techniques, and bio-applications. Processes. 2021;9(2):388. doi: 10.3390/pr9020388. [DOI] [Google Scholar]
- Evtushenko M, Wang K, Stokes HW, Nair H. Blood protein purification and simultaneous removal of nonenveloped viruses using tangential-flow preparative electrophoresis. Electrophoresis. 2005;26(1):28–34. doi: 10.1002/elps.200406150. [DOI] [PubMed] [Google Scholar]
- Fedel M. Hemocompatibility of carbon nanostructures. C. 2020;6(1):12. doi: 10.3390/c6010012. [DOI] [Google Scholar]
- Ferreira AM, Faustino VF, Mondal D, Coutinho JA, Freire MG. Improving the extraction and purification of immunoglobulin G by the use of ionic liquids as adjuvants in aqueous biphasic systems. J Biotechnol. 2016;236:166–175. doi: 10.1016/j.jbiotec.2016.08.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fonseca LC, de Araújo MM, de Moraes ACM, da Silva DS, Ferreira AG, Franqui LS, Martinez DST, Alves OL. Nanocomposites based on graphene oxide and mesoporous silica nanoparticles: Preparation, characterization and nanobiointeractions with red blood cells and human plasma proteins. Appl Surf Sci. 2018;437:110–121. doi: 10.1016/j.apsusc.2017.12.082. [DOI] [Google Scholar]
- Gayen B, Palchoudhury S, Chowdhury J. Carbon dots: a mystic star in the world of nanoscience. J Nanomater. 2019 doi: 10.1155/2019/3451307. [DOI] [Google Scholar]
- Ge S, Xi Y, Du R, Ren Y, Xu Z, Tan Y, Wang Y, Yin T, Wang G. Inhibition of in-stent restenosis after graphene oxide double-layer drug coating with good biocompatibility. Regen Biomater. 2019;6(5):299–309. doi: 10.1093/rb/rbz010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gerasimenko AY, Ten GN, Ryabkin DI, Shcherbakova NE, Morozova EA, Ichkitidze LP. The study of the interaction mechanism between bovine serum albumin and single-walled carbon nanotubes depending on their diameter and concentration in solid nanocomposites by vibrational spectroscopy. Spectrochim Acta A Mol Biomol Spectrosc. 2020;227:117682. doi: 10.1016/j.saa.2019.117682. [DOI] [PubMed] [Google Scholar]
- Ghosal K, Ghosh A. Carbon dots: The next generation platform for biomedical applications. Mater Sci Eng: C. 2019;96:887–903. doi: 10.1016/j.msec.2018.11.060. [DOI] [PubMed] [Google Scholar]
- Gogotsi Y. Not just graphene: The wonderful world of carbon and related nanomaterials. MRS Bull. 2015;40(12):1110–1121. doi: 10.1557/mrs.2015.272. [DOI] [Google Scholar]
- Gouw SC, Van Der Bom JG, Ljung R, Escuriola C, Cid AR, Claeyssens-Donadel S, Van Geet C, Kenet G, Mäkipernaa A, Molinari AC. Factor VIII products and inhibitor development in severe hemophilia A. N Engl J Med. 2013;368(3):231–239. doi: 10.1056/NEJMoa1208024. [DOI] [PubMed] [Google Scholar]
- Greening DW, Glenister KM, Sparrow RL, Simpson RJ. International blood collection and storage: clinical use of blood products. J Proteom. 2010;73(3):386–395. doi: 10.1016/j.jprot.2009.07.011. [DOI] [PubMed] [Google Scholar]
- Gryshchuk V, Galagan N. Silica nanoparticles effects on blood coagulation proteins and platelets. Biochem Res Int. 2016 doi: 10.1155/2016/2959414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Han Y, Wang X, Dai H, Li S. Nanosize and surface charge effects of hydroxyapatite nanoparticles on red blood cell suspensions. ACS Appl Mater Interfaces. 2012;4(9):4616–4622. doi: 10.1021/am300992x. [DOI] [PubMed] [Google Scholar]
- He XM, Carter DC. Atomic structure and chemistry of human serum albumin. Nature. 1992;358(6383):209–215. doi: 10.1038/358209a0. [DOI] [PubMed] [Google Scholar]
- He Y, Su Y (2014) Silicon nanostructures. In: Silicon Nano-biotechnology. Springer Berlin Heidelberg, pp 19–38. 10.1007/978-3-642-54668-6_2
- He Q, Zhang J, Shi J, Zhu Z, Zhang L, Bu W, Guo L, Chen Y. The effect of PEGylation of mesoporous silica nanoparticles on nonspecific binding of serum proteins and cellular responses. Biomaterials. 2010;31(6):1085–1092. doi: 10.1016/j.biomaterials.2009.10.046. [DOI] [PubMed] [Google Scholar]
- He Y, Fan C, Lee S-T. Silicon nanostructures for bioapplications. Nano Today. 2010;5(4):282–295. doi: 10.1016/j.nantod.2010.06.008. [DOI] [Google Scholar]
- Heo Y, Li C-A, Kim D, Shin S. Rheological alteration of erythrocytes exposed to carbon nanotubes. Clin Hemorheol Microcirc. 2017;65(1):49–56. doi: 10.3233/CH-15081. [DOI] [PubMed] [Google Scholar]
- Hess JR. Conventional blood banking and blood component storage regulation: opportunities for improvement. Blood Transfus. 2010;8(Suppl 3):s9–15. doi: 10.2450/2010.003S. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hess JR. Red cell storage. J Proteom. 2010;73(3):368–373. doi: 10.1016/j.jprot.2009.11.005. [DOI] [PubMed] [Google Scholar]
- Ho D, Wang C-HK, Chow EK-H. Nanodiamonds: the intersection of nanotechnology, drug development, and personalized medicine. Sci Adv. 2015;1(7):e1500439. doi: 10.1126/sciadv.1500439. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hoppe A, Güldal NS, Boccaccini AR. A review of the biological response to ionic dissolution products from bioactive glasses and glass-ceramics. Biomaterials. 2011;32(11):2757–2774. doi: 10.1016/j.biomaterials.2011.01.004. [DOI] [PubMed] [Google Scholar]
- Huang S, Qiu H, Lu S, Zhu F, Xiao Q. Study on the molecular interaction of graphene quantum dots with human serum albumin: combined spectroscopic and electrochemical approaches. J Hazard Mater. 2015;285:18–26. doi: 10.1016/j.jhazmat.2014.11.019. [DOI] [PubMed] [Google Scholar]
- Huang W, Deng Y, Ye L, Xie Q, Jiang Y. Enhancing hemocompatibility and the performance of Au@ silica nanoparticles by coating with cRGD functionalized zein. Mater Sci Eng C. 2021;125:112064. doi: 10.1016/j.msec.2021.112064. [DOI] [PubMed] [Google Scholar]
- Ilinskaya AN, Dobrovolskaia MA. Nanoparticles and the blood coagulation system. Part I: benefits of nanotechnology. Nanomedicine. 2013;8(5):773–784. doi: 10.2217/nnm.13.48. [DOI] [PubMed] [Google Scholar]
- Jafari S, Derakhshankhah H, Alaei L, Fattahi A, Varnamkhasti BS, Saboury AA. Mesoporous silica nanoparticles for therapeutic/diagnostic applications. Biomed Pharmacother. 2019;109:1100–1111. doi: 10.1016/j.biopha.2018.10.167. [DOI] [PubMed] [Google Scholar]
- Jaworski S, Hinzmann M, Sawosz E, Grodzik M, Kutwin M, Wierzbicki M, Strojny B, Vadalasetty KP, Lipińska L, Chwalibog A. Interaction of different forms of graphene with chicken embryo red blood cells. Environ Sci Pollut Res. 2017;24(27):21671–21679. doi: 10.1007/s11356-017-9788-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jeelani PG, Mulay P, Venkat R, Ramalingam C. Multifaceted application of silica nanoparticles. A review. SILICON. 2020;12(6):1337–1354. doi: 10.1007/s12633-019-00229-y. [DOI] [Google Scholar]
- Kim J, Cote LJ, Kim F, Yuan W, Shull KR, Huang J. Graphene oxide sheets at interfaces. J Am Chem Soc. 2010;132(23):8180–8186. doi: 10.1021/ja102777p. [DOI] [PubMed] [Google Scholar]
- Kim J, Heo Y-J, Shin S. Haemocompatibility evaluation of silica nanomaterials using hemorheological measurements. Clin Hemorheol Microcirc. 2016;62(2):99–107. doi: 10.3233/CH-151953. [DOI] [PubMed] [Google Scholar]
- Kim J, Nafiujjaman M, Nurunnabi M, Lee Y-K, Park H-K. Hemorheological characteristics of red blood cells exposed to surface functionalized graphene quantum dots. Food Chem Toxicol. 2016;97:346–353. doi: 10.1016/j.fct.2016.09.034. [DOI] [PubMed] [Google Scholar]
- Klein HG. Pathogen inactivation technology: cleansing the blood supply. J Intern Med. 2005;257(3):224–237. doi: 10.1111/j.1365-2796.2005.01451.x. [DOI] [PubMed] [Google Scholar]
- Klein HG, Spahn DR, Carson JL. Red blood cell transfusion in clinical practice. Lancet. 2007;370(9585):415–426. doi: 10.1016/S0140-6736(07)61197-0. [DOI] [PubMed] [Google Scholar]
- Kuchlyan J, Kundu N, Banik D, Roy A, Sarkar N. Spectroscopy and fluorescence lifetime imaging microscopy to probe the interaction of bovine serum albumin with graphene oxide. Langmuir. 2015;31(51):13793–13801. doi: 10.1021/acs.langmuir.5b03648. [DOI] [PubMed] [Google Scholar]
- Lai L, Lin C, Xu Z-Q, Han X-L, Tian F-F, Mei P, Li D-W, Ge Y-S, Jiang F-L, Zhang Y-Z. Spectroscopic studies on the interactions between CdTe quantum dots coated with different ligands and human serum albumin. Spectrochim Acta A Mol Biomol Spectrosc. 2012;97:366–376. doi: 10.1016/j.saa.2012.06.025. [DOI] [PubMed] [Google Scholar]
- Lee T-Y, Jayakumar T, Thanasekaran P, Lin K-C, Chen H-M, Veerakumar P, Sheu J-R. Carbon dot nanoparticles exert inhibitory effects on human platelets and reduce mortality in mice with acute pulmonary thromboembolism. Nanomterials. 2020;10(7):1254. doi: 10.3390/nano10071254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lesniak A, Fenaroli F, Monopoli MP, Åberg C, Dawson KA, Salvati A. Effects of the presence or absence of a protein corona on silica nanoparticle uptake and impact on cells. ACS Nano. 2012;6(7):5845–5857. doi: 10.1021/nn300223w. [DOI] [PubMed] [Google Scholar]
- Li H-C, Hsieh F-J, Chen C-P, Chang M-Y, Hsieh PC, Chen C-C, Hung S-U, Wu C-C, Chang H-C. The hemocompatibility of oxidized diamond nanocrystals for biomedical applications. Sci Rep. 2013;3(1):1–8. doi: 10.1038/srep03044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li M-Y, Xiao C-Q, Xu Z-Q, Yin M-M, Yang Q-Q, Yin Y-L, Liu Y. Role of surface charge on the interaction between carbon nanodots and human serum albumin. Spectrochim Acta A Mol Biomol Spectrosc. 2018;204:484–494. doi: 10.1016/j.saa.2018.06.082. [DOI] [PubMed] [Google Scholar]
- Liang C-Y, Pan J, Bai A-M, Hu Y-J. Insights into the interaction of human serum albumin and carbon dots: Hydrothermal synthesis and biophysical study. Int J Biol Macromol. 2020;149:1118–1129. doi: 10.1016/j.ijbiomac.2020.01.238. [DOI] [PubMed] [Google Scholar]
- Lin Y-C, Tsai L-W, Perevedentseva EV, Mona J, Cheng C-L, Chang H-H, Lin C-H, Sun D-S, Lugovtsov AE, Priezzhev AV. The influence of nanodiamond on the oxygenation states and micro rheological properties of human red blood cells in vitro. J Biomed Opt. 2012;17(10):101512. doi: 10.1117/1.JBO.17.10.101512. [DOI] [PubMed] [Google Scholar]
- Liu Y, Wang H, Liang P, Zhang HY. Water-soluble supramolecular fullerene assembly mediated by metallobridged β-cyclodextrins. Angew Chem Int Ed. 2004;43(20):2690–2694. doi: 10.1002/anie.200352973. [DOI] [PubMed] [Google Scholar]
- Liu S, Sui Y, Guo K, Yin Z, Gao X. Spectroscopic study on the interaction of pristine C 60 and serum albumins in solution. Nanoscale Res Lett. 2012;7(1):1–7. doi: 10.1186/1556-276X-7-433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu H-Y, Du L, Zhao Y-T, Tian W-Q. In vitro hemocompatibility and cytotoxicity evaluation of halloysite nanotubes for biomedical application. J Nanomater. 2015 doi: 10.1155/2015/685323. [DOI] [Google Scholar]
- Liu J, Liu R, Li L, Wang X, Gao X, Xing G, Jiang H, Zhao L. Interaction principle between coagulation factor X and fullerene derivatives with different hydrophilic-hydrophobic properties for anticoagulation. J Nanosci Nanotechnol. 2019;19(8):4603–4610. doi: 10.1166/jnn.2019.16365. [DOI] [PubMed] [Google Scholar]
- Loh KP, Lim CT. Molecular hemocompatibility of graphene oxide and its implication for antithrombotic applications. Small. 2015;11(38):5105–5117. doi: 10.1002/smll.201500841. [DOI] [PubMed] [Google Scholar]
- Loo S, Moore T, Banik B, Alexis F. Biomedical applications of hydroxyapatite nanoparticles. Curr Pharm Biotechnol. 2010;11(4):333–342. doi: 10.2174/138920110791233343. [DOI] [PubMed] [Google Scholar]
- Lundqvist M, Augustsson C, Lilja M, Lundkvist K, Dahlbäck B, Linse S, Cedervall T. The nanoparticle protein corona formed in human blood or human blood fractions. PLoS ONE. 2017;12(4):e0175871. doi: 10.1371/journal.pone.0175871. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ma Z, Bai J, Wang Y, Jiang X. Impact of shape and pore size of mesoporous silica nanoparticles on serum protein adsorption and RBCs hemolysis. ACS Appl Mater Interfaces. 2014;6(4):2431–2438. doi: 10.1021/am404860q. [DOI] [PubMed] [Google Scholar]
- Maguire CM, Lavin M, Doyle M, Byrne M, Prina-Mello A, O'Donnell JS, Volkov Y. The anticoagulant properties of cadmium telluride quantum dots. J Interdiscip Nanomed. 2018;3(1):16–28. doi: 10.1002/jin2.35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maity A, Pal U, Chakraborty B, Sengupta C, Sau A, Chakraborty S, Basu S. Preferential photochemical interaction of Ru (III) doped carbon nano dots with bovine serum albumin over human serum albumin. Int J Biol Macromol. 2019;137:483–494. doi: 10.1016/j.ijbiomac.2019.06.126. [DOI] [PubMed] [Google Scholar]
- Mandal S, Hossain M, Devi PS, Kumar GS, Chaudhuri K. Interaction of carbon nanoparticles to serum albumin: elucidation of the extent of perturbation of serum albumin conformations and thermodynamical parameters. J Hazard Mater. 2013;248:238–245. doi: 10.1016/j.jhazmat.2013.01.009. [DOI] [PubMed] [Google Scholar]
- Martinez DST, Paula AJ, Fonseca LC, Luna LAV, Silveira CP, Durán N, Alves OL. Monitoring the hemolytic effect of mesoporous silica nanoparticles after human blood protein corona formation. Eur J Inorg Chem. 2015;27:4595–4602. doi: 10.1002/ejic.201500573. [DOI] [Google Scholar]
- Mathai J. Problem of bacterial contamination in platelet concentrates. Transfus Apher Sci. 2009;41(2):139–144. doi: 10.1016/j.transci.2009.07.012. [DOI] [PubMed] [Google Scholar]
- Mehrizi TZ. Hemocompatibility and hemolytic effects of functionalized nanoparticles on red blood cells: a recent review study. NANO. 2021;16(08):2130007. doi: 10.1142/S1793292021300073. [DOI] [Google Scholar]
- Mehrizi TZ. An overview of the latest applications of platelet-derived microparticles and nanoparticles in medical technology 2010–2020. Curr Mol Med. 2021 doi: 10.2174/1566524021666210928152015. [DOI] [PubMed] [Google Scholar]
- Mehrizi TZ, Eshghi P. Investigation of the effect of nanoparticles on platelet storage duration 2010–2020. Int Nano Lett. 2021 doi: 10.1007/s40089-021-00340-2. [DOI] [Google Scholar]
- Mehrizi TZ, Hosseini KM. An overview on investigation of nanomaterials' effect on plasma components: Immunoglobulins and coagulant factor VIII, 2010–2020 review. Nanoscale Adv. 2021 doi: 10.1039/D1NA00119A. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Molaei MJ. A review on nanostructured carbon quantum dots and their applications in biotechnology, sensors, and chemiluminescence. Talanta. 2019;196:456–478. doi: 10.1016/j.talanta.2018.12.042. [DOI] [PubMed] [Google Scholar]
- Monasterio BG, Alonso B, Sot JS, García-Arribas AB, Gil-Cartón D, Valle M, Zurutuza A, Goñi FM. Coating graphene oxide with lipid bilayers greatly decreases its hemolytic properties. Langmuir. 2017;33(33):8181–8191. doi: 10.1021/acs.langmuir.7b01552. [DOI] [PubMed] [Google Scholar]
- Moore C, Monton H, O'Kennedy R, Williams D, Nogues C, Crean C, Gubala V. Controlling colloidal stability of silica nanoparticles during bioconjugation reactions with proteins and improving their longer-term stability, handling and storage. J Mater Chem B. 2015;3(10):2043–2055. doi: 10.1039/c4tb01915f. [DOI] [PubMed] [Google Scholar]
- Mukhopadhyay S, Veroniaina H, Chimombe T, Han L, Zhenghong W, Xiaole Q. Synthesis and compatibility evaluation of versatile mesoporous silica nanoparticles with red blood cells: an overview. RSC Adv. 2019;9(61):35566–35578. doi: 10.1039/C9RA06127D. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Murugadoss S, Lison D, Godderis L, Van Den Brule S, Mast J, Brassinne F, Sebaihi N, Hoet PH. Toxicology of silica nanoparticles: an update. Arch Toxicol. 2017;91(9):2967–3010. doi: 10.1007/s00204-017-1993-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nan Z, Hao C, Ye X, Feng Y, Sun R. Interaction of graphene oxide with bovine serum albumin: A fluorescence quenching study. Spectrochim Acta A Mol Biomol Spectrosc. 2019;210:348–354. doi: 10.1016/j.saa.2018.11.028. [DOI] [PubMed] [Google Scholar]
- Narayan R, Nayak UY, Raichur AM, Garg S. Mesoporous silica nanoparticles: A comprehensive review on synthesis and recent advances. Pharmaceutics. 2018;10(3):118. doi: 10.3390/pharmaceutics10030118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Naskar A, Khan H, Bera S, Jana S. Soft chemical synthesis, characterization and interaction of ZnO graphene nanocomposite with bovine serum albumin protein. J Mol Liq. 2017;237:113–119. doi: 10.1016/j.molliq.2017.04.074. [DOI] [Google Scholar]
- Nemmar A, Beegam S, Yuvaraju P, Yasin J, Shahin A, Ali BH. Interaction of amorphous silica nanoparticles with erythrocytes in vitro: role of oxidative stress. Cell Physiol Biochem. 2014;34(2):255–265. doi: 10.1159/000362996. [DOI] [PubMed] [Google Scholar]
- Ngamcherdtrakul W, Sangvanich T, Reda M, Gu S, Bejan D, Yantasee W. Lyophilization and stability of antibody-conjugated mesoporous silica nanoparticle with cationic polymer and PEG for siRNA delivery. Int J Nanomed. 2018;13:4015. doi: 10.2147/IJN.S164393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Okada M, Matsumoto T. Synthesis and modification of apatite nanoparticles for use in dental and medical applications. Jpn Dent Sci Rev. 2015;51(4):85–95. doi: 10.1016/j.jdsr.2015.03.004. [DOI] [Google Scholar]
- Ou L, Song B, Liang H, Liu J, Feng X, Deng B, Sun T, Shao L. Toxicity of graphene-family nanoparticles: a general review of the origins and mechanisms. Part Fibre Toxicol. 2016;13(1):1–24. doi: 10.1186/s12989-016-0168-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pan DC, Myerson JW, Brenner JS, Patel PN, Anselmo AC, Mitragotri S, Muzykantov V. Nanoparticle properties modulate their attachment and effect on carrier red blood cells. Sci Rep. 2018;8(1):1–12. doi: 10.1038/s41598-018-19897-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Perevedentseva E, Lin Y-C, Jani M, Cheng C-L. Biomedical applications of nanodiamonds in imaging and therapy. Nanomedicine. 2013;8(12):2041–2060. doi: 10.2217/nnm.13.183. [DOI] [PubMed] [Google Scholar]
- Pinto J, Domingues MRM, Galhano E, Pita C, do Céu AlmeidaCarreiraGil MIMAM. Human plasma stability during handling and storage: impact on NMR metabolomics. Analyst. 2014;139(5):1168–1177. doi: 10.1039/c3an02188b. [DOI] [PubMed] [Google Scholar]
- Prakash S, Thakur A. Platelet concentrates: past, present and future. J Maxillofac Oral Surg. 2011;10(1):45–49. doi: 10.1007/s12663-011-0182-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Qin J-X, Yang X-G, Lv C-F, Li Y-Z, Liu K-K, Zang J-H, Yang X, Dong L, Shan C-X. Nanodiamonds: synthesis, properties, and applications in nanomedicine. Mater Des. 2021;210:110091. doi: 10.1016/j.matdes.2021.110091. [DOI] [Google Scholar]
- Reviakine I, Jung F, Braune S, Brash JL, Latour R, Gorbet M, van Oeveren W. Stirred, shaken, or stagnant: What goes on at the blood–biomaterial interface. Blood Rev. 2017;31(1):11–21. doi: 10.1016/j.blre.2016.07.003. [DOI] [PubMed] [Google Scholar]
- Sasidharan A, Panchakarla LS, Sadanandan AR, Ashokan A, Chandran P, Girish CM, Menon D, Nair SV, Rao C, Koyakutty M. Hemocompatibility and macrophage response of pristine and functionalized graphene. Small. 2012;8(8):1251–1263. doi: 10.1002/smll.201102393. [DOI] [PubMed] [Google Scholar]
- Seabra AB, Paula AJ, de Lima R, Alves OL, Durán N. Nanotoxicity of graphene and graphene oxide. Chem Res Toxicol. 2014;27(2):159–168. doi: 10.1021/tx400385x. [DOI] [PubMed] [Google Scholar]
- Sen Gupta A. Bio-inspired nanomedicine strategies for artificial blood components. Wiley Interdiscip Rev Nanomed Nanobiotechnol. 2017;9(6):e1464. doi: 10.1002/wnan.1464. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shahabi J, Ebrahimi Shahmabadi H, Alavi SE, Movahedi F, Koohi Moftakhari Esfahani M, Zadeh Mehrizi T, Akbarzadeh A. Effect of gold nanoparticles on properties of nanoliposomal hydroxyurea: an in vitro study. Indian J Clin Biochem. 2014;29(3):315–320. doi: 10.1007/s12291-013-0355-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sharma S, Sharma P, Tyler LN. Transfusion of blood and blood products: indications and complications. Am Fam Phys. 2011;83(6):719–724. [PubMed] [Google Scholar]
- Shi J, Hedberg Y, Lundin M, Wallinder IO, Karlsson H, Möller L. Hemolytic properties of synthetic nano-and porous silica particles: the effect of surface properties and the protection by the plasma corona. Acta Biomater. 2012;8(9):3478–3490. doi: 10.1016/j.actbio.2012.04.024. [DOI] [PubMed] [Google Scholar]
- Shi H, Liu H, Luan S, Shi D, Yan S, Liu C, Li RK, Yin J. Effect of polyethylene glycol on the antibacterial properties of polyurethane/carbon nanotube electrospun nanofibers. RSC Adv. 2016;6(23):19238–19244. doi: 10.1039/C6RA00363J. [DOI] [Google Scholar]
- Song M, Liu S, Yin J, Wang H. Interaction of human serum album and C60 aggregates in solution. Int J Mol Sci. 2011;12(8):4964–4974. doi: 10.3390/ijms12084964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stefanic M, Ward K, Tawfik H, Seemann R, Baulin V, Guo Y, Fleury J-B, Drouet C. Apatite nanoparticles strongly improve red blood cell cryopreservation by mediating trehalose delivery via enhanced membrane permeation. Biomaterials. 2017;140:138–149. doi: 10.1016/j.biomaterials.2017.06.018. [DOI] [PubMed] [Google Scholar]
- Strengers PF. Evidence-based clinical indications of plasma products and future prospects. Ann Blood. 2017 doi: 10.21037/aob.2017.12.03. [DOI] [Google Scholar]
- Taneva SG, Krumova S, Bogár F, Kincses A, Stoichev S, Todinova S, Danailova A, Horváth J, Násztor Z, Kelemen L. Insights into graphene oxide interaction with human serum albumin in isolated state and in blood plasma. Int J Biol Macromol. 2021;175:19–29. doi: 10.1016/j.ijbiomac.2021.01.151. [DOI] [PubMed] [Google Scholar]
- Tsamesidis I, Pouroutzidou GK, Lymperaki E, Kazeli K, Lioutas CB, Christodoulou E, Perio P, Reybier K, Pantaleo A, Kontonasaki E. Effect of ion doping in silica-based nanoparticles on the hemolytic and oxidative activity in contact with human erythrocytes. Chem-Biol Interact. 2020;318:108974. doi: 10.1016/j.cbi.2020.108974. [DOI] [PubMed] [Google Scholar]
- Wang W, Wang YJ, Kelner DN. Coagulation factor VIII: structure and stability. Int J Pharm. 2003;259(1–2):1–15. doi: 10.1016/s0378-5173(03)00227-8. [DOI] [PubMed] [Google Scholar]
- Wang Y, Zhang B, Zhai G. The effect of incubation conditions on the hemolytic properties of unmodified graphene oxide with various concentrations. RSC Adv. 2016;6(72):68322–68334. doi: 10.1039/C6RA13607A. [DOI] [Google Scholar]
- Wąsowicz M, Ficek M, Wróbel MS, Chakraborty R, Fixler D, Wierzba P, Jędrzejewska-Szczerska M. Haemocompatibility of Modified Nanodiamonds. Materials. 2017;10(4):352. doi: 10.3390/ma10040352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu H, Lin L, Wang P, Jiang S, Dai Z, Zou X. Solubilization of pristine fullerene by the unfolding mechanism of bovine serum albumin for cytotoxic application. ChemComm. 2011;47(38):10659–10661. doi: 10.1039/c1cc13953c. [DOI] [PubMed] [Google Scholar]
- Xiao Q, Huang S, Ma J, Su W, Li P, Cui J, Liu Y. Systematically investigation of interactions between BSA and different charge-capped CdSe/ZnS quantum dots. J Photochem Photobiol A Chem. 2012;249:53–60. doi: 10.1016/j.jphotochem.2012.08.019. [DOI] [Google Scholar]
- Xiao Q, Liang Y, Liu Y, Lu S, Huang S. Comparison of molecular interactions of Ag2Te and CdTe quantum dots with human serum albumin by spectroscopic approaches. Luminescence. 2018;33(1):181–189. doi: 10.1002/bio.3391. [DOI] [PubMed] [Google Scholar]
- Xing Y, Dai L. Nanodiamonds for nanomedicine. Nanomedicine. 2009 doi: 10.2217/17435889.4.2.207. [DOI] [PubMed] [Google Scholar]
- Xu Z-Q, Yang Q-Q, Lan J-Y, Zhang J-Q, Peng W, Jin J-C, Jiang F-L, Liu Y. Interactions between carbon nanodots with human serum albumin and γ-globulins: the effects on the transportation function. J Hazard Mater. 2016;301:242–249. doi: 10.1016/j.jhazmat.2015.08.062. [DOI] [PubMed] [Google Scholar]
- Yang B, Liu R, Hao X, Wu Y, Du J. Effect of CdTe quantum dots size on the conformational changes of human serum albumin: results of spectroscopy and isothermal titration calorimetry. Biol Trace Elem Res. 2013;155(1):150–158. doi: 10.1007/s12011-013-9771-z. [DOI] [PubMed] [Google Scholar]
- Yeh Y-T, Lin Z, Zheng S-Y, Terrones M. A carbon nanotube integrated microfluidic device for blood plasma extraction. Sci Rep. 2018;8(1):1–8. doi: 10.1038/s41598-018-31810-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yildirim A, Ozgur E, Bayindir M. Impact of mesoporous silica nanoparticle surface functionality on hemolytic activity, thrombogenicity and non-specific protein adsorption. J Mater Chem B. 2013;1(14):1909–1920. doi: 10.1039/c3tb20139b. [DOI] [PubMed] [Google Scholar]
- Zadeh Mehrizi T. Hemocompatibility and hemolytic effects of functionalized nanoparticles on red blood cells: a recent review study. NANO. 2021;16(08):27. doi: 10.1142/S1793292021300073. [DOI] [Google Scholar]
- Zadeh Mehrizi T. Impact of metallic, quantum dots and carbon-based nanoparticles on quality and storage of albumin products for clinical use. NANO. 2021;16(14):2130013. doi: 10.1142/S1793292021300139. [DOI] [Google Scholar]
- Zadeh Mehrizi T. An overview of the latest applications of platelet-derived microparticles and nanoparticles in medical technology 2010–2020. Curr Mol Med. 2021 doi: 10.2174/1566524021666210928152015. [DOI] [PubMed] [Google Scholar]
- Zadeh Mehrizi T, Amini Kafiabad S. Evaluation of the effects of nanoparticles on the therapeutic function of platelet: a review. J Pharm Pharmacol. 2021;74(2):179–190. doi: 10.1093/jpp/rgab089. [DOI] [PubMed] [Google Scholar]
- Zadeh Mehrizi T, Amini Kafiabad S. A review study of the influences of dendrimer nanoparticles on stored platelet in order to treat patients (2001–2020) Curr Nanosci. 2021;17:1–15. doi: 10.2174/157341371701210304152534. [DOI] [Google Scholar]
- Zadeh Mehrizi T, Eshghi P. Investigation of the effect of nanoparticles on platelet storage duration 2010–2020. Int Nano Lett. 2021;12:15–45. doi: 10.1007/s40089-021-00340-2. [DOI] [Google Scholar]
- Zadeh Mehrizi T, Mousavi Hosseini K. An overview on investigation of nanomaterials' effect on plasma components: immunoglobulins and coagulant factor VIII, 2010–2020 review. Nanoscale Adv. 2021;3:3730–3745. doi: 10.1039/D1NA00119A. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zadeh Mehrizi T, Amini Kafiabad S, Eshghi P. Effects and treatment applications of polymeric nanoparticles on improving platelets’ storage time: a review of the literature from 2010 to 2020. Blood Res. 2021;56(4):215–228. doi: 10.5045/br.2021.2021094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zampini G, Matino D, Quaglia G, Tarpani L, Gargaro M, Cecchetti F, Iorio A, Fallarino F, Latterini L. Experimental evidences on the role of silica nanoparticles surface morphology on the loading, release and activity of three proteins. Micropor Mesopor Mat. 2019;287:220–227. doi: 10.1016/j.micromeso.2019.06.005. [DOI] [Google Scholar]
- Zhang Y, Bai Y, Yan B. Functionalized carbon nanotubes for potential medicinal applications. Drug Discov Today. 2010;15(11–12):428–435. doi: 10.1016/j.drudis.2010.04.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang H, Zhu Z, Wang Y, Fei Z, Cao J. Changing the activities and structures of bovine serum albumin bound to graphene oxide. Appl Surf Sci. 2018;427:1019–1029. doi: 10.1016/j.apsusc.2017.08.130. [DOI] [Google Scholar]
- Zhao M, Cao Y, Liu X, Deng J, Li D, Gu H. Effect of nitrogen atomic percentage on N+-bombarded MWCNTs in cytocompatibility and hemocompatibility. Nanoscale Res Lett. 2014;9(1):1–9. doi: 10.1186/1556-276X-9-142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zubair AC. Clinical impact of blood storage lesions. Am J Hematol. 2010;85(2):117–122. doi: 10.1002/ajh.21599. [DOI] [PubMed] [Google Scholar]
- Žūkienė R, Snitka V. Zinc oxide nanoparticle and bovine serum albumin interaction and nanoparticles influence on cytotoxicity in vitro. Colloids Surf B Biointerfaces. 2015;135:316–323. doi: 10.1016/j.colsurfb.2015.07.054. [DOI] [PubMed] [Google Scholar]
- Zuo J, Jiang T, Zhao X, Xiong X, Xiao S, Zhu Z. Preparation and application of fluorescent carbon dots. J Nanomater. 2015 doi: 10.1155/2015/787862. [DOI] [Google Scholar]