Abstract
Aims
Albuminuria is strongly associated with risk of renal dysfunction, cardiovascular disease and mortality. However, clinical guidelines diverge, and evidence is sparse on what risk factor levels regarding blood pressure, blood lipids and BMI are needed to prevent albuminuria in adolescents and young adults with type 1 diabetes.
Methods
A total of 9347 children and adults with type 1 diabetes [mean age 15.3 years and mean diabetes duration 1.4 years at start of follow-up] from The Swedish National Diabetes Registry were followed from first registration until end of 2017. Levels for risk factors for a risk increase in nephropathy were evaluated, and the gradient of risk per 1 SD (standard deviation) was estimated to compare the impact of each risk factor.
Results
During the follow-up period, 8610 (92.1%) remained normoalbuminuric, 737 (7.9%) individuals developed micro- or macroalbuminuria at any time period of whom 132 (17.9% of 737) individuals developed macroalbuminuria. Blood pressure ≥ 140/80 mmHg was associated with increased risk of albuminuria (p ≤ 0.0001), as were triglycerides ≥ 1.0 mmol/L (p = 0.039), total cholesterol ≥ 5.0 mmol/L (p = 0.0003), HDL < 1.0 mmol/L (p = 0.013), LDL 3.5– < 4.0 mmol/L (p = 0.020), and BMI ≥ 30 kg/m2 (p = 0.033). HbA1c was the strongest risk factor for any albuminuria estimated by the measure gradient of risk per 1 SD, followed by diastolic blood pressure, triglycerides, systolic blood pressure, cholesterol and LDL. In patients with HbA1c > 65 mmol/mol (> 8.1%), blood pressure > 140/70 mmHg was associated with increased risk of albuminuria.
Conclusions
Preventing renal complications in adolescents and young adults with type 1 diabetes need avoidance at relatively high levels of blood pressure, blood lipids and BMI, whereas very tight control is not associated with further risk reduction. For patients with long-term poor glycaemic control, stricter blood pressure control is advocated.
Supplementary Information
The online version contains supplementary material available at 10.1007/s00592-022-01863-6.
Keywords: Type 1 diabetes, Albuminuria, Lipids, Blood pressure, BMI
Introduction
Diabetic nephropathy (DN), also known as diabetic kidney disease, occurs in 15–40% of all persons with type 1 diabetes. DN is characterised by pathological urinary albumin excretion, glomerular lesions, and loss of glomerular filtration rate [1]. Microalbuminuria has been established as an early marker of progressive kidney disease, and macroalbuminuria an essential risk factor for cardiovascular disease, including stroke, atrial fibrillation, heart failure and mortality [2–4].
The Diabetes Control and Complication Trial (DCCT) and the follow-up Epidemiology of Diabetes Interventions and Complications (EDIC) demonstrated benefits of reducing HbA1c and decreasing the risk of DN with intensive therapy compared with conventional therapy [5, 6]. Blood pressure, dyslipidaemia, diabetes duration and albumin excretion rate have also been shown to be associated with progression of DN [7–16]. Recommendations regarding blood pressure diverge in guidelines from European Society of Cardiology (ESC), American College of Cardiology (ACC)/American Heart Association (AHA) and American Diabetes Association (ADA) [17–19]. Additionally, for blood lipid managements, evidence remains incomplete regarding adults with type 1 diabetes < 40 years old, as earlier studies have mainly been performed in older patients with long diabetes duration [9–14]. To fully understand the impact of a risk factor for development of diabetes complications, it is essential to have information from diagnosis of diabetes and onwards in large, unselected population-based patient cohorts. Correct estimations of risk factors are essential for prognosis, but also for resource allocation and avoiding adverse events from unnecessary medications.
Recently, we found in a large patient cohort, following patients from diagnosis and onwards, that risks of nephropathy gradually increased with HbA1c higher than 52 mmol/mol (7.0%) and no further risk reductions were seen at HbA1c < 48 mmol/mol (6.5%) [20]. In this population-based cohort study using paediatric and adult registries in Sweden, we aimed to evaluate what levels of blood lipids, blood pressure and BMI in persons with type 1 diabetes are related to risk of nephropathy. We also aimed to rank HbA1c, blood lipids, blood pressure, body mass index (BMI), and smoking regarding their risk contribution to development of DN.
Methods
We conducted a registry-based observational cohort study. Ethical approval was obtained from the Regional Ethical Review Board in Gothenburg, Sweden.
Data sources
Data were obtained from the paediatric registry, SWEDIABKIDS, and the adult registry, The Swedish National Diabetes Registry (NDR), which started in the years 2000 and 1996, respectively. The registries recently merged and include information on risk factors and complications. For inclusion in the registries, patients and/or guardians provide their informed consent. More than 95% of Swedish children and adolescents with type 1 diabetes are registered in the SWEDIABKIDS, the registries have included data on almost all children and adolescents with diabetes in Sweden [21]. In this study, information from NDR from 1998 onwards and for SWEDIABKIDS from 2000 onwards were included.
Study population
Current evaluations were performed from a recently used study cohort [20]. A total of 9347 persons with type 1 diabetes were included and followed up during period 1 January 1998 to 31 December 2017. Type 1 diabetes was defined as treatment with insulin and diagnosis < 30 years of age, and this definition has been validated in 97% of cases [22]. To be included, children should have a clinical diagnosis of type 1 diabetes in the register. HLA and diabetes-related autoantibodies are determined in all children with newly diagnosed diabetes. In brief, criteria for inclusion in the study were children and adults who had their type 1 diabetes- diagnosis for < 5 years when first recorded in the registries. In addition, a minimum of 4 visits with non-missing values during a minimum of 8-year follow-up from diagnosis were required.
Study procedures
Variables assessed were HbA1c (glycated haemoglobin), BMI (body mass index), low-density lipoprotein (LDL), high-density lipoprotein (HDL), cholesterol, triglycerides, systolic blood pressure (SBP), diastolic blood pressure (DBP) and smoking.
As earlier described [20], participants were included in the registers at different time points, whereby subcohorts were categorized by follow-up time from diabetes diagnosis to evaluate risk factors for a certain diabetes duration: 8–9, 10–11, 12–13, 14–15, and 16–20 years. This approach was used to standardize the diabetes duration which is essential for the development of complications. Further, conventional approaches of evaluating time to event by time-updated models could not be used since hazard ratios were not constant over time (assumption of proportional hazards was not fulfilled). We followed participants from the first observation until the end of each subcohort if having a registration regarding albuminuria (normo-, micro- or macroalbuminuria). The primary analysis was a pooled analysis using all the five different subcohorts described above. Two separate analyses were performed for evaluating risk factors for any albuminuria (micro- or macroalbuminuria) and macroalbuminuria, respectively.
Microalbuminuria was defined as two positive tests from three samples taken within 1 year, with an albumin/creatinine ratio of 3–30 mg/mmol (30–300 mg/g) or U-albumin of 20–200 μg/min (20–300 mg/L). Macroalbuminuria was defined as albumin/creatinine ratio > 30 mg/mmol. If the first estimation of elevated albumin/creatinine ratio was registered as macroalbuminuria, this event was included in both the microalbuminuria and macroalbuminuria analyses.
According to instructions to the clinics, the registered blood pressure (SBP and DBP) was the mean value of two supine readings with a cuff of appropriate size and after at least 5 min of rest. HbA1c values were measured in mmol/mol and were also converted to levels in % according to the National Glycohaemoglobin Standardization Program for dual reporting [23]. Laboratory methods at participating care units for analysing HbA1c level and albuminuria are regularly checked with central reference samples of HbA1c and albuminuria to ensure high accuracy [24].
SBP, DBP, LDL, HDL, triglycerides, total cholesterol, BMI and HbA1c were evaluated as mean of longitudinally collected values, as continuous variables in relation to any albuminuria and to macroalbuminuria. Nonlinear effects were studied by analysing categories of the continuous variables; SBP categories (< 110 mmHg, 110– < 120 mmHg, 120– < 130 mmHg, 130– < 140 mmHg, ≥ 140 mmHg), DBP categories (< 60 mmHg, 60– < 70 mmHg, 70– < 80 mmHg, 80– < 85 mmHg, ≥ 85 mmHg), BMI categories (< 18.5 kg/m2, 18.5– < 25 kg/m2, 25– < 30 kg/m2, 30– < 35 kg/m2, ≥ 35 kg/m2), LDL categories (< 2.0 mmol/l, 2.0– < 2.5 mmol/L, 2.5– < 3.0 mmol/L, 3.0– < 3.5 mmol/L, 3.5– < 4.0 mmol/L, ≥ 4.0 mmol/L), HDL categories (< 1.0 mmol/L, 1.0– < 1.5 mmol/L, 1.5– < 2.0 mmol/L, ≥ 2.0 mmol/L), triglycerides categories (< 0.5 mmol/L, 0.5– < 1.0 mmol/L, 1.0– < 1.5 mmol/L, 1.5–2.0 mmol/L, ≥ 2.0 mmol/L), cholesterol categories (< 4.0 mmol/L, 4.0– < 5.0 mmol/L, 5.0– < 6.0 mmol/L, ≥ 6.0 mmol/L) and smoking (no versus yes). Odds ratios were described for the following changes in the risk factors: SBP (10 mmHg), DBP (5 mmHg), blood lipid levels (1 mmol/L), BMI (5 kg/m2) and HbA1c (10 mmol/mol, 1%). To rank the relative contribution of the risk factors, the metric gradient of risk per one SD was used [25] determining how much the risk of any and macroalbuminuria changes per one SD change in the risk factor. Additionally, sensitivity analyses on all persons > 13 years of age at diabetes diagnosis were performed for the above-mentioned analyses. The impact of risk factor categories on any albuminuria and macroalbuminuria were further analysed after subgrouping for a mean level of HbA1c above or below 65 mmol/mol (8.1%) during follow-up.
Statistical analysis
Numbers and percentages (with 95% confidence intervals) of complications were expressed for each category of SBP, DBP, BMI, LDL, HDL, triglycerides and cholesterol. Generalised estimating equations modelling was used to estimate the relation between the risk factors and endpoints for diabetic nephropathy, adjusting for within patient correlation for repeated data over the 5 follow-up cohorts. This method allows more than one observation per patient. Unstructured covariance matrix was used. The use of binomial distribution with logit link function resulted in odds ratios (95% confidence intervals) as risk estimates. Analyses were performed for continuous variables and by categories of HbA1c, SBP, DBP, BMI, LDL, HDL, triglycerides and cholesterol, and for smoking. Risk factors were analysed adjusted for age and sex and additionally for mean HbA1c. Mean HbA1c was analysed adjusted for age and sex and additionally for SBP, BMI, triglycerides, cholesterol and smoking.
Pearson correlation was calculated when analysing association between two continuous variables.
No data were imputed. Tests were two-tailed and conducted at 0.05 significance level. All statistical programming was performed using SAS Software version 9.4 (SAS Institute, Cary, NC).
Results
Patient characteristics
A total of 9347 persons with type 1 diabetes were included. Patient characteristics are shown in Table 1 for the total cohort and for patients without albuminuria during follow-up and with micro- and/or macroalbuminuria at any time period during follow-up, respectively.
Table 1.
Patient characteristics
| Variable | All patients (n=9347) | All patients with no albuminuria (n=8610) | All patients with micro- or macroalbuminuria in any time period (n=737) | All patients with no macroalbuminuria (n=9215) | All patients with macroalbuminuria in any time period (n=132) |
|---|---|---|---|---|---|
| Sex | |||||
| Male | 5244 (56.1%) | 4883 (56.7%) | 361 (49.0%) | 5184 (56.3%) | 60 (45.5%) |
| Female | 4103 (43.9%) | 3727 (43.3%) | 376 (51.0%) | 4031 (43.7%) | 72 (54.5%) |
| Age at first visit | 15.3 (7.9) n=9347 | 15.2 (7.9) n=8610 | 16.0 (7.9) n=737 | 15.2 (7.9) n=9215 | 17.2 (8.4) n=132 |
| Diabetes onset year | 2003 (4) n=9347 | 2003 (4) n=8610 | 2002 (4) n=737 | 2003 (4) n=9215 | 2002 (4) n=132 |
| HbA1c mean (%) for longest follow-up | 7.94 (1.04) n=9347 | 7.89 (1.01) n=8610 | 8.49 (1.27) n=737 | 7.93 (1.03) n=9215 | 8.57 (1.40) n=132 |
| HbA1c mean (mmol/mol) for longest follow-up | 63.2 (11.4) n=9347 | 62.7 (11.0) n=8610 | 69.3 (13.9) n=737 | 63.1 (11.3) n=9215 | 70.1 (15.3) n=132 |
| HbA1c mean category for longest follow-up | |||||
| <48 mmol/mol | 667 (7.1%) | 642 (7.5%) | 25 (3.4%) | 662 (7.2%) | 5 (3.8%) |
| 48-52 mmol/mol | 932 (10.0%) | 889 (10.3%) | 43 (5.8%) | 924 (10.0%) | 8 (6.1%) |
| 53-57 mmol/mol | 1511 (16.2%) | 1419 (16.5%) | 92 (12.5%) | 1498 (16.3%) | 13 (9.8%) |
| 58-70 mmol/mol | 3963 (42.4%) | 3701 (43.0%) | 262 (35.5%) | 3914 (42.5%) | 49 (37.1%) |
| >70 mmol/mol | 2274 (24.3%) | 1959 (22.8%) | 315 (42.7%) | 2217 (24.1%) | 57 (43.2%) |
| SBP mean (mmHg) for longest follow-up | 116.7 (8.8) n=9330 | 116.5 (8.6) n=8594 | 118.2 (10.0) n=736 | 116.6 (8.7) n=9198 | 119.1 (12.0) n=132 |
| SBP mean category for longest follow-up | |||||
| <110 mmHg | 2048 (22.0%) | 1911 (22.2%) | 137 (18.6%) | 2021 (22.0%) | 27 (20.5%) |
| 110-<120 mmHg | 4129 (44.3%) | 3812 (44.4%) | 317 (43.1%) | 4074 (44.3%) | 55 (41.7%) |
| 120-<130 mmHg | 2490 (26.7%) | 2286 (26.6%) | 204 (27.7%) | 2461 (26.8%) | 29 (22.0%) |
| 130-<140 mmHg | 567 (6.1%) | 512 (6.0%) | 55 (7.5%) | 556 (6.0%) | 11 (8.3%) |
| >=140 mmHg | 96 (1.0%) | 73 (0.8%) | 23 (3.1%) | 86 (0.9%) | 10 (7.6%) |
| DBP mean (mmHg) for longest follow-up | 70.1 (5.9) n=9330 | 69.9 (5.8) n=8594 | 71.7 (6.8) n=736 | 70.0 (5.8) n=9198 | 72.4 (8.2) n=132 |
| DBP mean category for longest follow-up | |||||
| <60 mmHg | 317 (3.4%) | 298 (3.5%) | 19 (2.6%) | 314 (3.4%) | 3 (2.3%) |
| 60-<70 mmHg | 4425 (47.4%) | 4133 (48.1%) | 292 (39.7%) | 4368 (47.5%) | 57 (43.2%) |
| 70-<80 mmHg | 4086 (43.8%) | 3741 (43.5%) | 345 (46.9%) | 4036 (43.9%) | 50 (37.9%) |
| 80-<85 mmHg | 397 (4.3%) | 347 (4.0%) | 50 (6.8%) | 388 (4.2%) | 9 (6.8%) |
| >=85 mmHg | 105 (1.1%) | 75 (0.9%) | 30 (4.1%) | 92 (1.0%) | 13 (9.8%) |
| BMI mean (kg/m2) for longest follow-up | 22.9 (4.0) n=9316 | 22.9 (4.0) n=8583 | 23.6 (4.9) n=733 | 22.9 (4.0) n=9185 | 23.9 (5.0) n=131 |
| BMI mean category for longest follow-up | |||||
| <18.5 kg/m^2 | 1038 (11.1%) | 961 (11.2%) | 77 (10.5%) | 1025 (11.2%) | 13 (9.9%) |
| 18.5-<25 kg/m^2 | 5880 (63.1%) | 5450 (63.5%) | 430 (58.7%) | 5804 (63.2%) | 76 (58.0%) |
| 25-<30 kg/m^2 | 1917 (20.6%) | 1762 (20.5%) | 155 (21.1%) | 1889 (20.6%) | 28 (21.4%) |
| 30-<35 kg/m^2 | 383 (4.1%) | 336 (3.9%) | 47 (6.4%) | 373 (4.1%) | 10 (7.6%) |
| >=35 kg/m^2 | 98 (1.1%) | 74 (0.9%) | 24 (3.3%) | 94 (1.0%) | 4 (3.1%) |
| HDL mean (mmol/L) for longest follow-up | 1.52 (0.38) n=8534 | 1.52 (0.38) n=7844 | 1.47 (0.38) n=690 | 1.52 (0.38) n=8413 | 1.48 (0.42) n=121 |
| HDL mean category for longest follow-up | |||||
| <1.0 mmol/L | 383 (4.5%) | 332 (4.2%) | 51 (7.4%) | 372 (4.4%) | 11 (9.1%) |
| 1.0-<1.5 mmol/L | 4026 (47.2%) | 3681 (46.9%) | 345 (50.0%) | 3971 (47.2%) | 55 (45.5%) |
| 1.5-<2.0 mmol/L | 3233 (37.9%) | 2998 (38.2%) | 235 (34.1%) | 3191 (37.9%) | 42 (34.7%) |
| >=2.0 mmol/L | 892 (10.5%) | 833 (10.6%) | 59 (8.6%) | 879 (10.4%) | 13 (10.7%) |
| LDL mean (mmol/L) for longest follow-up | 2.53 (0.69) n=8539 | 2.51 (0.68) n=7846 | 2.65 (0.73) n=693 | 2.52 (0.68) n=8422 | 2.72 (0.79) n=117 |
| LDL mean category for longest follow-up | |||||
| <2.0 mmol/L | 1832 (21.5%) | 1709 (21.8%) | 123 (17.7%) | 1812 (21.5%) | 20 (17.1%) |
| 2.0-<2.5 mmol/L | 2617 (30.6%) | 2427 (30.9%) | 190 (27.4%) | 2589 (30.7%) | 28 (23.9%) |
| 2.5-<3.0 mmol/L | 2191 (25.7%) | 2013 (25.7%) | 178 (25.7%) | 2159 (25.6%) | 32 (27.4%) |
| 3.0-<3.5 mmol/L | 1175 (13.8%) | 1061 (13.5%) | 114 (16.5%) | 1155 (13.7%) | 20 (17.1%) |
| 3.5-<4.0 mmol/L | 501 (5.9%) | 440 (5.6%) | 61 (8.8%) | 490 (5.8%) | 11 (9.4%) |
| >=4.0 mmol/L | 223 (2.6%) | 196 (2.5%) | 27 (3.9%) | 217 (2.6%) | 6 (5.1%) |
| Cholesterol mean (mmol/L) for longest follow-up | 4.49 (0.78) n=8707 | 4.48 (0.77) n=8010 | 4.68 (0.85) n=697 | 4.49 (0.78) n=8587 | 4.89 (0.99) n=120 |
| Cholesterol mean category for longest follow-up | |||||
| <4.0 mmol/L | 2250 (25.8%) | 2107 (26.3%) | 143 (20.5%) | 2231 (26.0%) | 19 (15.8%) |
| 4.0-<5.0 mmol/L | 4394 (50.5%) | 4074 (50.9%) | 320 (45.9%) | 4344 (50.6%) | 50 (41.7%) |
| 5.0-<6.0 mmol/L | 1721 (19.8%) | 1533 (19.1%) | 188 (27.0%) | 1684 (19.6%) | 37 (30.8%) |
| >=6.0 mmol/L | 342 (3.9%) | 296 (3.7%) | 46 (6.6%) | 328 (3.8%) | 14 (11.7%) |
| Triglycerides mean (mmol/L) for longest follow-up | 1.07 (0.68) n=8300 | 1.04 (0.59) n=7630 | 1.38 (1.24) n=670 | 1.06 (0.64) n=8189 | 1.75 (1.94) n=111 |
| Triglycerides mean category for longest follow-up | |||||
| <0.5 mmol/L | 335 (4.0%) | 317 (4.2%) | 18 (2.7%) | 333 (4.1%) | 2 (1.8%) |
| 0.5-<1.0 mmol/L | 4550 (54.8%) | 4267 (55.9%) | 283 (42.2%) | 4513 (55.1%) | 37 (33.3%) |
| 1.0-<1.5 mmol/L | 2175 (26.2%) | 1980 (26.0%) | 195 (29.1%) | 2148 (26.2%) | 27 (24.3%) |
| 1.5-2.0 mmol/L | 687 (8.3%) | 613 (8.0%) | 74 (11.0%) | 670 (8.2%) | 17 (15.3%) |
| >=2.0 mmol/L | 553 (6.7%) | 453 (5.9%) | 100 (14.9%) | 525 (6.4%) | 28 (25.2%) |
| Smoking at any time before for longest follow-up | |||||
| No | 6518 (77.8%) | 6021 (78.4%) | 497 (71.3%) | 6436 (78.0%) | 82 (65.1%) |
| Yes | 1862 (22.2%) | 1662 (21.6%) | 200 (28.7%) | 1818 (22.0%) | 44 (34.9%) |
For categorical variables n (%) is presented.
For continuous variables Mean (SD)/n = is presented.
The proportion of women in the cohort was 43.9%, the mean HbA1c for the longest follow-up was 63.2 ± 11.4 mmol/mol (7.9 ± 1.0%), mean blood pressure 116 ± 9/70 ± 6 mmHg, mean BMI 22.9 ± 4.0 kg/m2, mean LDL 2.5 ± 0.7 mmol/L, mean HDL 1.5 ± 0.4 mmol/L, mean cholesterol 4.5 ± 0.8 mmol/L, and mean triglycerides 1.1 ± 0.7 mmol/L. The mean age at first registered visit was 15.3 ± 7.9 years, and the mean duration of diabetes at first registration was 1.4 ± 1.7 years. Median follow-up was 12.0 years (range 8.0–20.0). In Supplemental Table 1, patient characteristics for the different subcohorts are shown.
Relative contribution of risk factors for DN
Among 9347 children and adults with type 1 diabetes from the Swedish National Diabetes Registry and observed over a median of 12.0 years, 737 (7.9%) developed any albuminuria; 132 out of these 737 (17.9%) developed macroalbuminuria. Levels of risk factors for a significant risk increase in albuminuria were evaluated and ranking of risk factors was estimated (Table 2). For both any albuminuria and macroalbuminuria, HbA1c was the strongest risk factor. For any albuminuria, the remaining risk factors were ranked as follows: DBP, triglycerides, SBP, total cholesterol and LDL. No impact for: HDL, BMI and smoking. For macroalbuminuria, the remaining risk factors were ranked as follows: total cholesterol, SBP, DBP, and triglycerides. No impact existed for: BMI, LDL, HDL and smoking.
Table 2.
Generalized estimating equation (GEE) models for the impact of risk factors on nephropathy endpoints
| Adjusted for age and sex | Multivariable adjusted* | |||||||
|---|---|---|---|---|---|---|---|---|
| Variable | n (%) (95% CI) events** | n (%) (95% CI) persons with events*** | OR (95% CI) per specified unit increase | OR (95% CI) per 1 SD increase | p-value | OR (95% CI) per specified unit increase | OR (95% CI) per 1 SD increase | p-value |
| Microalbuminuria/Macroalbuminuria vs None | ||||||||
| Mean HbA1c (mmol/mol) (by 10 unit increase) | 1027 (6.1%) (5.7%–6.5%) | 737 (7.9%) (7.3%–8.4%) | 1.55 (1.46–1.66) | 1.65 (1.53–1.78) | <.0001 | 1.49 (1.39–1.61) | 1.58 (1.45–1.72) | <.0001 |
| Mean HbA1c (%) (by 1 unit increase) | 1027 (6.1%) (5.7%–6.5%) | 737 (7.9%) (7.3%–8.4%) | 1.62 (1.51–1.73) | 1.65 (1.53–1.77) | <.0001 | 1.55 (1.43–1.68) | 1.58 (1.45–1.71) | <.0001 |
| Mean SBP (mmHg) (by 10 unit increase) | 1026 (6.1%) (5.7%–6.5%) | 736 (7.9%) (7.3%–8.5%) | 1.28 (1.16–1.42) | 1.24 (1.13–1.35) | <.0001 | 1.25 (1.12–1.38) | 1.21 (1.10–1.32) | <.0001 |
| Mean DBP (mmHg) (by 5 unit increase) | 1026 (6.1%) (5.7%–6.5%) | 736 (7.9%) (7.3%–8.5%) | 1.30 (1.21–1.41) | 1.35 (1.24–1.48) | <.0001 | 1.23 (1.14–1.34) | 1.27 (1.16–1.39) | <.0001 |
| Mean BMI (kg/m2) (by 5 unit increase) | 1022 (6.1%) (5.7%–6.5%) | 733 (7.9%) (7.3%–8.4%) | 1.19 (1.06–1.34) | 1.15 (1.05–1.25) | 0.0031 | 1.10 (0.98–1.23) | 1.07 (0.98–1.17) | 0.12 |
| Mean LDL (mmol/L) (by 1 unit increase) | 957 (6.2%) (5.8%–6.5%) | 689 (8.1%) (7.5%–8.7%) | 1.32 (1.18–1.47) | 1.21 (1.12–1.31) | <.0001 | 1.17 (1.05–1.30) | 1.11 (1.03–1.20) | 0.0056 |
| Mean HDL (mmol/L) (by 1 unit increase) | 959 (6.2%) (5.8%–6.6%) | 688 (8.1%) (7.5%–8.7%) | 0.68 (0.51–0.89) | 0.86 (0.77–0.95) | 0.0046 | 0.82 (0.63–1.05) | 0.92 (0.84–1.02) | 0.12 |
| Mean triglycerides (mmol/L) (by 1 unit increase) | 930 (6.2%) (5.8%–6.6%) | 668 (8.0%) (7.5%–8.7%) | 1.58 (1.44–1.73) | 1.35 (1.27–1.44) | <.0001 | 1.37 (1.25–1.50) | 1.23 (1.16–1.31) | <.0001 |
| Mean cholesterol (mmol/L) (by 1 unit increase) | 968 (6.1%) (5.8%–6.5%) | 695 (8.0%) (7.4%–8.6%) | 1.36 (1.24–1.51) | 1.28 (1.19–1.39) | <.0001 | 1.20 (1.09–1.32) | 1.16 (1.07–1.25) | 0.0002 |
| Smoking at any time before (yes vs no) | 975 (6.2%) (5.9%–6.6%) | 695 (8.3%) (7.7%–8.9%) | 1.42 (1.19–1.69) | 1.15 (1.07–1.24) | <.0001 | 1.05 (0.88–1.26) | 1.02 (0.95–1.10) | 0.58 |
| Macroalbuminuria vs None/Microalbuminuria | ||||||||
| Mean HbA1c (mmol/mol) (by 10 unit increase) | 163 (1.0%) (0.8%–1.1%) | 132 (1.4%) (1.2%–1.7%) | 1.59 (1.35–1.86) | 1.69 (1.41–2.03) | <.0001 | 1.46 (1.22–1.74) | 1.54 (1.26–1.88) | <.0001 |
| Mean HbA1c (%) (by 1 unit increase) | 163 (1.0%) (0.8%–1.1%) | 132 (1.4%) (1.2%–1.7%) | 1.66 (1.39–1.98) | 1.69 (1.41–2.03) | <.0001 | 1.51 (1.25–1.83) | 1.54 (1.26–1.88) | <.0001 |
| Mean SBP (mmHg) (by 10 unit increase) | 163 (1.0%) (0.8%–1.1%) | 132 (1.4%) (1.2%–1.7%) | 1.47 (1.13–1.90) | 1.39 (1.11–1.73) | 0.0037 | 1.40 (1.09–1.80) | 1.33 (1.08–1.65) | 0.0083 |
| Mean DBP (mmHg) (by 5 unit increase) | 163 (1.0%) (0.8%–1.1%) | 132 (1.4%) (1.2%–1.7%) | 1.36 (1.12–1.65) | 1.42 (1.14–1.76) | 0.0017 | 1.28 (1.05–1.56) | 1.32 (1.06–1.66) | 0.014 |
| Mean BMI (kg/m2) (by 5 unit increase) | 162 (1.0%) (0.8%–1.1%) | 131 (1.4%) (1.2%–1.7%) | 1.15 (0.90–1.47) | 1.11 (0.92–1.35) | 0.27 | 1.04 (0.80–1.35) | 1.03 (0.84–1.26) | 0.78 |
| Mean LDL (mmol/L) (by 1 unit increase) | 144 (0.9%) (0.8%–1.1%) | 116 (1.4%) (1.1%–1.6%) | 1.38 (1.03–1.84) | 1.25 (1.02–1.53) | 0.031 | 1.27 (0.98–1.66) | 1.18 (0.98–1.42) | 0.076 |
| Mean HDL (mmol/L) (by 1 unit increase) | 148 (1.0%) (0.8%–1.1%) | 120 (1.4%) (1.2%–1.7%) | 0.65 (0.32–1.31) | 0.84 (0.64–1.11) | 0.23 | 0.89 (0.45–1.74) | 0.96 (0.73–1.24) | 0.73 |
| Mean triglycerides (mmol/L) (by 1 unit increase) | 137 (0.9%) (0.8%–1.1%) | 110 (1.3%) (1.1%–1.6%) | 1.66 (1.44–1.91) | 1.40 (1.27–1.53) | <.0001 | 1.50 (1.31–1.72) | 1.31 (1.20–1.43) | <.0001 |
| Mean cholesterol (mmol/L) (by 1 unit increase) | 147 (0.9%) (0.8%–1.1%) | 119 (1.4%) (1.1%–1.6%) | 1.64 (1.28–2.09) | 1.48 (1.22–1.81) | <.0001 | 1.49 (1.21–1.84) | 1.38 (1.16–1.63) | 0.0002 |
| Smoking at any time before (yes vs no) | 156 (1.0%) (0.8%–1.2%) | 125 (1.5%) (1.2%–1.8%) | 1.81 (1.23–2.67) | 1.28 (1.09–1.50) | 0.0027 | 1.30 (0.87–1.95) | 1.11 (0.94–1.31) | 0.21 |
*HbA1c mean adjusted for age, sex, SBP, BMI, triglycerides, cholesterol and smoking. All other variables adjusted for age, sex and HbA1c mean.
**The number of events pooled over all periods.
***The number of individuals with at least one event pooled over all periods.
Thresholds for risk of DN
The variables were analysed as categorical variables for the impact on any albuminuria endpoints evaluated for different thresholds adjusted for age, sex and mean HbA1c, as a study of potential nonlinear effects.
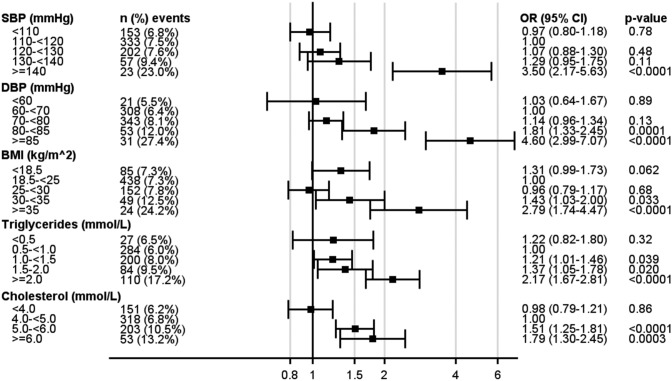
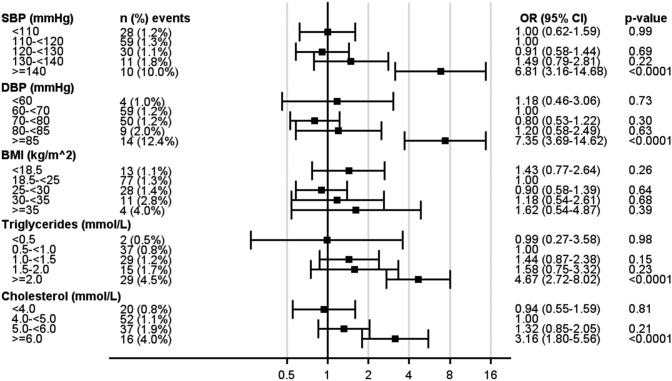
As seen in Fig. 1, the risk of any albuminuria increased at SBP ≥ 140 mmHg compared with the reference category (110–120 mmHg) and at DBP ≥ 80 mmHg compared with the reference category (60– < 70 mmHg). For macroalbuminuria (Fig. 2), blood pressure ≥ 140/85 mmHg increased the risk. For blood lipids, the risk of any albuminuria increased for LDL 3.5– < 4.0 mmol/L, HDL < 1.0 mmol/L, triglycerides 1.0– < 1.5 mmol/L, and cholesterol 5.0– < 6.0 mmol/L compared to each reference category. For BMI, the risk of any albuminuria increased at ≥ 30 kg/m2. For macroalbuminuria, mean triglycerides and cholesterol remained significant risk factors for the highest category compared to the respective reference category. Associations between BMI categories and macroalbuminuria, showed numerically higher OR, although not being significant.
Fig. 1.
Generalized Estimating Equation (GEE) models for the impact of various variable categories on any albuminuria
Fig. 2.
Generalized Estimating Equation (GEE) models for the impact of various variable categories on macroalbuminuria
In the supplementary appendix, the categorized levels of SBP, DBP, LDL, HDL, triglycerides, cholesterol and BMI analysed for the impact on albuminuria endpoints are shown and, additionally, adjusted for age and sex (Supplemental Table 2).
Impact of risk factors on subgrouping of HbA1c
The impact of various risk factors on albuminuria was also evaluated based on mean HbA1c levels during follow-up (≤ 65 mmol/mol and > 65 mmol/mol [8.1%]) adjusted for age, sex and mean HbA1c, as shown in Supplemental Table 3. For persons with mean HbA1c > 65 mmol/mol during follow-up, a blood pressure > 140/70 mmHg was associated with increased risk of albuminuria, whereas for persons with HbA1c ≤ 65 mmol/mol, blood pressure > 140/80 mmHg was associated with increased risk.
Sensitivity analysis
Similar patterns were observed in a sensitivity analysis when patients > 13 years of age at diabetes diagnosis were included regarding their impact on development of any albuminuria. The patient characteristics are shown in the Supplemental Table 4. In brief, 4606 patients were included in the analyses. Numerically, they had a lower mean HbA1c, higher mean blood pressure, comparable mean blood lipids and higher mean BMI at baseline compared to the full cohort population. The same target levels for blood pressure (≥ 140/80 mmHg) and BMI (≥ 30 kg/m2) were associated with increased risk of any albuminuria (Supplement Table 5).
As observed in Supplemental Table 6, the impact of continuous risk factors expressed by per specified unit and 1 SD increase on nephropathy endpoints are shown. For any albuminuria, HbA1c followed by DBP, SBP, triglycerides, BMI and HDL remained the significant risk factors. For macroalbuminuria, blood pressure and triglycerides had a significant impact, but not other studied variables.
Conclusions
In this population-based cohort study, we used both pediatric and adult registries to evaluate the impact of various risk factors on the development of albuminuria in persons with type 1 diabetes. HbA1c was the strongest risk factor for any albuminuria, followed by blood pressure, blood lipids and BMI. No association was seen for smoking. For macroalbuminuria, systolic and diastolic blood pressure, triglycerides and cholesterol remained significant non-glycaemic risk factors for the development of albuminuria. In persons with poor glycaemic control (mean HbA1c > 65 mmol/mol [8.1%]), blood pressure > 140/70 mmHg was associated with increased risk of albuminuria.
In 1993, DCCT showed beneficial effects of reducing HbA1c and decreasing the risk of nephropathy with intensive therapy compared with conventional therapy [5]. In the follow-up EDIC-study, the beneficial effects on albumin excretion and the reduced incidence of hypertension after the DCCT suggested that previous intensive treatment had extended benefit in delaying progression of diabetic nephropathy [6]. The importance of glycaemic control on nephropathy has been shown in several other studies [7–12]. Since then, multiple studies have evaluated other risk factors for progression of albuminuria. Hypertension, dyslipidaemia, and diabetes duration are well-known risk factors, whereas the impact of gender, smoking and BMI have shown diverging results [7–12]. Recently, a study, using the DCCT/EDIC cohort, showed that the risk of macroalbuminuria was associated with sex, blood pressure and lipids (LDL, HDL and triglycerides) [16]. Our results expand upon the results from prior studies by providing data from persons with type 1 diabetes in a population-based contemporary cohort with short diabetes duration at baseline (mean 1.4 years) followed over 8–20 years, as well as what levels of these risk factors increases the risk of albuminuria.
In our study, SBP and DBP were evaluated both as continuous and categorical variables. Both analyses showed that blood pressure has an important impact on the risk of albuminuria. As a continuous variable, an increase in 10 mmHg for mean SBP increased the risk of any albuminuria by 25% and for mean DBP by 35% with an increase in each 5 mmHg. When evaluating SBP and DBP as a categorical variable, levels above 140/80 increased risk of albuminuria. For DBP ≥ 85 mmHg, the OR was increased to 4.6. According to guidelines from ADA and ESC, hypertension is defined as ≥ 140/90 mmHg, whereas ACC/AHA define hypertension as > 130/90 mmHg. Additionally, the guidelines differ in recommendations regarding risk assessment and target goals in adults. Further, the recommendations are mainly based on clinical trials on an older population with or without type 2 diabetes [17–19]. Hence, few high-quality data exist for blood pressure targets in type 1 diabetes from diagnosis and onwards.
Another important risk factor for albuminuria was blood lipids. Our results show that the risk of albuminuria increased at following levels: triglycerides ≥ 1.0 mmol/L, total cholesterol ≥ 5.0 mmol/L, LDL 3.5– < 4.0 mmol/L and HDL < 1.0 mmol/L compared to each group selected reference. Of the blood lipids, triglycerides had the strongest impact on risk of albuminuria after HbA1c and diastolic blood pressure. In a cross-sectional analysis, it was shown that glycaemic control was an important mediator of lipid abnormalities in youth with type 1 diabetes [26]. Additionally, in the FinnDiane study, it was shown that triglyceride and cholesterol levels were associated with incident albuminuria [27]. The mechanisms responsible for the dyslipidaemia in type 1 diabetes remain unclear, but the subcutaneous route of insulin administration, that is responsible for peripheral hyperinsulinemia, may play a role [28]. Another possible explanation is weight gain, and central obesity from a sedentary lifestyle. The individuals with type 1 diabetes and an associated metabolic syndrome have an increased cardiovascular risk compared to other type 1 diabetes patients related to the development of atherogenic lipid profile, in which hypertriglyceridemia is an essential component [29].
In clinical practice, caregivers are more hesitant to treat younger individuals with medications for increased blood pressure and blood lipids. There is a risk of side effects with medications, evidence is less robust due to lack of randomized trials in this patient group and younger individuals have lower adherence to treatment [30]. It is, therefore, important to know at what levels risk factors besides HbA1c really increases the risk of albuminuria. Systolic blood pressure of 130 compared to 140 mmHg has been debated for long periods [31]. This study supports the higher blood pressure level, but at repeated determinations above these values, younger individuals should also get antihypertensive drugs. Additionally, since risk factors can be more difficult to judge among young children, we analysed in a sensitivity analysis patient with diabetes diagnosis after 13 years of age, the same age span as in the DCCT study, with confirming results. In several diseases, strict risk factor targets have been found to be beneficial in prevention to more severe disease, e.g. lower blood lipid targets after myocardial infarction. In the current study, patients with high mean HbA1c (> 8.1%) during follow-up seem to benefit of an even lower diastolic blood pressure of < 70 mmHg. Hence, stricter blood pressure control is indicated in persons with type 1 diabetes with poor glycaemic control.
In the case of statins, these are more commonly used at slightly older ages when the risk of cardiovascular disease increases. With clearly increased LDL and cholesterol levels in younger individuals, one should first exclude familial hypercholesterolemia and then consider medication if dietary advice is not enough [32]. Regarding BMI, previous studies have not found that obesity is a strong risk factor for myocardial infarction and mortality [33]. However, the risk of heart failure, which is significantly more common in type 1 diabetes than in the general population, clearly increases above BMI 30 [34, 35]. In addition, in a study from National Diabetes Audit data, which included individuals with type 1 and type 2 diabetes in UK and Wales, association between obesity and kidney disease was shown [36]. The fact that we see that the risk of albuminuria increases above BMI 30 indicates that an important focus in diabetes care among adolescents and young adults should be to avoid obesity. Furthermore, a weight reduction can contribute to reduced blood pressure in obese patients [37].
A strength of the current study is that patients were followed from close after type 1 diabetes diagnosis (mean diabetes duration 1.4 years at inclusion) over 8–20 years. It is essential to have the complete historical risk factor profile from patients before development of diabetes complications. However, this study was limited by the absence of other factors contributing to albuminuria which were not documented in the registry (e.g., other renal diseases). Since the current study was not randomized, residual confounding cannot be excluded. However, since randomized trials are lacking in the current age group, this first large population-based study following non-glycaemic risk factors from diagnosis of type 1 diabetes is an important contribution to the evidence for risk factor targets in adolescents and young adults with type 1 diabetes.
In this analysis of nationwide registers, it was shown that beside the importance of HbA1c, prevention of renal complications in adolescents and young adults with type 1 diabetes was associated with certain target levels for non-glycaemic risk factors: blood pressure ≥ 140/80 mmHg, LDL 3.5– < 4.0 mmol/L, BMI ≥ 30 kg/m2, HDL < 1.0 mmol/L and total cholesterol ≥ 5.0 mmol/L. Risk was evident already at triglycerides levels ≥ 1.0 mmol/L, which had the strongest impact on risk after glycaemic control and diastolic blood pressure. The current findings are essential for patients and care-givers where treatment for blood pressure, lipids and obesity have potential adverse effects and require major health care resources. Somewhat more tight blood pressure control is indicated in patients with poor glycaemic control being at high risk of nephropathy.
Supplementary Information
Below is the link to the electronic supplementary material.
Author contributions
SSA wrote the first draft of the manuscript. AP performed statistical calculations. All authors were involved in the study design, interpretation of data, and review of the manuscript.
Funding
Open access funding provided by University of Gothenburg. This study was supported by the Swedish government (Agreement for Medical Education and Research) and Novo Nordisk Foundation. The funders had no role in the study design, data collection, data analysis, data interpretation, or writing of the report.
Data availability
Data are not publicly available. Data may be available on request to the Swedish National Diabetes Registry.
Code availability
N/A.
Declarations
Conflict of interest
TN has received honoraria on expert group participation from AstraZeneca, Merck Shark & Dohme, Novo Nordisk, Eli Lilly and Company, Boerhinger Ingelheim, Abbot and Amgen. ML has received grants from Eli Lilly and Novo Nordisk, and consulting fees from AstraZeneca, Boehringer Ingelheim, Dexcom, Eli Lilly, MSD and Novo Nordisk. SSA, AP, AS, HW, BR, JL: no conflicts of interests.
Consent to participate
Each patient provides informed consent for inclusion in the NDR.
Consent for publication
Not required.
Ethics approval
This study was approved by the regional ethical and review board of the University of Gothenburg, Gothenburg, Sweden (Dnr 977–17).
Footnotes
Johnny Ludvigsson, Marcus Lind shared last author position.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Lim A. Diabetic nephropathy—Complications and treatment. Int J Nephrol Renov Dis. 2014;7:361–381. doi: 10.2147/IJNRD.S40172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.de Boer IH, Gao X, Cleary PA, et al. Albuminuria changes and cardiovascular and renal outcomes in type 1 diabetes: the DCCT/EDIC Study. Clin J Am Soc Nephrol CJASN. 2016;11(11):1969–1977. doi: 10.2215/CJN.02870316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Hallström S, Pivodic A, Rosengren A, Ólafsdóttir AF, Svensson AM, Lind M. Risk factors for atrial fibrillation in people with type 1 diabetes: an observational cohort study of 36,258 patients from the swedish national diabetes registry. Diabetes Care. 2019;42(8):1530–1538. doi: 10.2337/dc18-2457. [DOI] [PubMed] [Google Scholar]
- 4.Vestberg D, Rosengren A, Olsson M, et al. Decreased eGFR as a risk factor for heart failure in 13 781 individuals with type 1 diabetes. J Diabetes Sci Technol. 2015;10(1):131–136. doi: 10.1177/1932296815596174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Nathan DM, Genuth S, Lachin J, Diabetes Control and Complications Trial Research Group et al. The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. New Engl J Med. 1993;329(14):977–986. doi: 10.1056/NEJM199309303291401. [DOI] [PubMed] [Google Scholar]
- 6.DCCT/EDIC research group Effect of intensive diabetes treatment on albuminuria in type 1 diabetes: long-term follow-up of the diabetes control and complications trial and epidemiology of diabetes interventions and complications study. Lancet Diabetes Endocrinol. 2014;2(10):793–800. doi: 10.1016/S2213-8587(14)70155-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Coonrod BA, Ellis D, Becker DJ, et al. Predictors of microalbuminuria in individuals with IDDM. Pittsburgh epidemiology of diabetes complications study. Diabetes Care. 1993;16(10):1376–1383. doi: 10.2337/diacare.16.10.1376. [DOI] [PubMed] [Google Scholar]
- 8.Bojestig M, Arnqvist HJ, Hermansson G, Karlberg BE, Ludvigsson J. Declining incidence of nephropathy in insulin-dependent diabetes mellitus. N Engl J Med. 1994;330(1):15–18. doi: 10.1056/NEJM199401063300103. [DOI] [PubMed] [Google Scholar]
- 9.The Microalbuminuria Collaborative Study Group Predictors of the development of microalbuminuria in patients with Type 1 diabetes mellitus: a seven-year prospective study. Diabetic Med J British Diabetic Assoc. 1999;16(11):918–925. doi: 10.1046/j.1464-5491.1999.00182.x. [DOI] [PubMed] [Google Scholar]
- 10.Rossing P, Hougaard P, Parving HH. Risk factors for development of incipient and overt diabetic nephropathy in type 1 diabetic patients: a 10-year prospective observational study. Diabetes Care. 2002;25(5):859–864. doi: 10.2337/diacare.25.5.859. [DOI] [PubMed] [Google Scholar]
- 11.Giorgino F, Laviola L, Cavallo Perin P, Solnica B, Fuller J, Chaturvedi N. Factors associated with progression to macroalbuminuria in microalbuminuric Type 1 diabetic patients: the EURODIAB prospective complications study. Diabetologia. 2004;47(6):1020–1028. doi: 10.1007/s00125-004-1413-8. [DOI] [PubMed] [Google Scholar]
- 12.Nordwall M, Arnqvist HJ, Bojestig M, Ludvigsson J. Good glycemic control remains crucial in prevention of late diabetic complications–the Linköping diabetes complications study. Pediatr Diabetes. 2009;10(3):168–176. doi: 10.1111/j.1399-5448.2008.00472.x. [DOI] [PubMed] [Google Scholar]
- 13.Thomas MC, Rosengård-Bärlund M, Mills V. Serum lipids and the progression of nephropathy in type 1 diabetes. Diabetes Care. 2006;29(2):317–322. doi: 10.2337/diacare.29.02.06.dc05-0809. [DOI] [PubMed] [Google Scholar]
- 14.Skrivarhaug T, Bangstad HJ, Stene LC, Sandvik L, Hanssen KF, Joner G. Low risk of overt nephropathy after 24 yr of childhood-onset type 1 diabetes mellitus (T1DM) in Norway. Pediatr Diabetes. 2006;7(5):239–246. doi: 10.1111/j.1399-5448.2006.00204.x. [DOI] [PubMed] [Google Scholar]
- 15.Raile K, Galler A, Hofer S, et al. Diabetic nephropathy in 27,805 children, adolescents, and adults with type 1 diabetes: effect of diabetes duration, A1C, hypertension, dyslipidemia, diabetes onset, and sex. Diabetes Care. 2007;30(10):2523–2528. doi: 10.2337/dc07-0282. [DOI] [PubMed] [Google Scholar]
- 16.Perkins BA, Bebu I, de Boer IH, et al. Risk factors for kidney disease in type 1 diabetes. Diabetes Care. 2019;42(5):883–890. doi: 10.2337/dc18-2062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Unger T, Borghi C, Charchar F, et al. 2020 International society of hypertension global hypertension practice guidelines. Hypertension. 2020;75(6):1334–1357. doi: 10.1161/HYPERTENSIONAHA.120.15026. [DOI] [PubMed] [Google Scholar]
- 18.Williams B, Mancia G, Spiering W, et al. 2018 ESC/ESH Guidelines for the management of arterial hypertension. Eur Heart J. 2018;39(33):3021–3104. doi: 10.1093/eurheartj/ehy339. [DOI] [PubMed] [Google Scholar]
- 19.American Diabetes Association 10 Cardiovascular disease and risk management: standards of medical care in diabetes-2021. Diabetes Care. 2021;44(Suppl 1):S125–S150. doi: 10.2337/dc21-S010. [DOI] [PubMed] [Google Scholar]
- 20.Lind M, Pivodic A, Svensson AM, Ólafsdóttir AF, Wedel H, Ludvigsson J. HbA1c level as a risk factor for retinopathy and nephropathy in children and adults with type 1 diabetes: Swedish population based cohort study. BMJ (Clin Res ed) 2019;366:l4894. doi: 10.1136/bmj.l4894. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Samuelsson U, Åkesson K, Peterson A, Hanas R, Hanberger L. Continued improvement of metabolic control in Swedish pediatric diabetes care. Pediatr Diabetes. 2018;19(1):150–157. doi: 10.1111/pedi.12467. [DOI] [PubMed] [Google Scholar]
- 22.Eeg-Olofsson K, Cederholm J, Nilsson PM, et al. Glycemic control and cardiovascular disease in 7454 patients with type 1 diabetes: an observational study from the Swedish National Diabetes Register (NDR) Diabetes Care. 2010;33(7):1640–1646. doi: 10.2337/dc10-0398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Jeppsson JO, Kobold U, Barr J, et al. Approved IFCC reference method for the measurement of HbA1c in human blood. Clin Chem Laborat Med. 2002;40(1):78–89. doi: 10.1515/CCLM.2002.016. [DOI] [PubMed] [Google Scholar]
- 24.Lindblad B, Nordin G. External quality assessment of HbA1c and its effect on comparison between Swedish pediatric diabetes clinics. Experiences from the Swedish pediatric diabetes quality register (Swediabkids) and Equalis. Clin Chem Laborat Med. 2013;51(10):2045–2052. doi: 10.1515/cclm-2013-0226. [DOI] [PubMed] [Google Scholar]
- 25.Hopper JL. Odds per adjusted standard deviation: comparing strengths of associations for risk factors measured on different scales and across diseases and populations. Am J Epidemiol. 2015;182(10):863–867. doi: 10.1093/aje/kwv193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Guy J, Ogden L, Wadwa RP, et al. Lipid and lipoprotein profiles in youth with and without type 1 diabetes: the SEARCH for Diabetes in Youth case-control study. Diabetes Care. 2009;32(3):416–420. doi: 10.2337/dc08-1775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Mäkinen VP, Soininen P, Kangas AJ, et al. Triglyceride-cholesterol imbalance across lipoprotein subclasses predicts diabetic kidney disease and mortality in type 1 diabetes: the FinnDiane Study. J Inter Med. 2013;273(4):383–395. doi: 10.1111/joim.12026. [DOI] [PubMed] [Google Scholar]
- 28.Vergès B. Dyslipidemia in type 1 diabetes: AMaskedDanger. Trends Endocrinol Metab. 2020;31(6):422–434. doi: 10.1016/j.tem.2020.01.015. [DOI] [PubMed] [Google Scholar]
- 29.Hartz JC, de Ferranti S, Gidding S. Hypertriglyceridemia in diabetes mellitus: implications for pediatric care. J Endocrine Soc. 2018;2(6):497–512. doi: 10.1210/js.2018-00079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Rosina R, Crisp J, Steinbeck K. Treatment adherence of youth and young adults with and without a chronic illness. Nurs Health Sci. 2003;5(2):139–147. doi: 10.1046/j.1442-2018.2003.00149.x. [DOI] [PubMed] [Google Scholar]
- 31.Grassi G, Mancia G, Nilsson PM. Specific blood pressure targets for patients with diabetic nephropathy? Diabetes Care. 2016;39(Suppl 2):S228–S233. doi: 10.2337/dcS15-3020. [DOI] [PubMed] [Google Scholar]
- 32.Wiegman A, Gidding SS, Watts GF, et al. Familial hypercholesterolaemia in children and adolescents: gaining decades of life by optimizing detection and treatment. Eur Heart J. 2015;36(36):2425–2437. doi: 10.1093/eurheartj/ehv157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Vestberg D, Rosengren A, Eeg-Olofsson K, et al. Body mass index as a risk factor for coronary events and mortality in patients with type 1 diabetes. Open Heart. 2018;5(1):e000727. doi: 10.1136/openhrt-2017-000727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Rosengren A, Vestberg D, Svensson AM, et al. Long-term excess risk of heart failure in people with type 1 diabetes: a prospective case-control study. Lancet Diabetes Endocrinol. 2015;3(11):876–885. doi: 10.1016/S2213-8587(15)00292-2. [DOI] [PubMed] [Google Scholar]
- 35.Vestberg D, Rosengren A, Olsson M, Gudbjörnsdottir S, Svensson AM, Lind M. Relationship between overweight and obesity with hospitalization for heart failure in 20,985 patients with type 1 diabetes: a population-based study from the Swedish National Diabetes Registry. Diabetes Care. 2013;36(9):2857–2861. doi: 10.2337/dc12-2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Hill CJ, Cardwell CR, Maxwell AP, et al. Obesity and kidney disease in type 1 and 2 diabetes: an analysis of the National Diabetes Audit. QJM Monthly J Assoc Phys. 2013;106(10):933–942. doi: 10.1093/qjmed/hct123. [DOI] [PubMed] [Google Scholar]
- 37.Harsha DW, Bray GA. Weight loss and blood pressure control (Pro) Hypertension. 2008;51(6):1420–1425. doi: 10.1161/HYPERTENSIONAHA.107.094011. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Data are not publicly available. Data may be available on request to the Swedish National Diabetes Registry.
N/A.