Abstract
PAM50 intrinsic subtyping and risk of recurrence (ROR) score are approved for risk profiling in postmenopausal women. We aimed to examine their long-term prognostic value in terms of breast cancer-free interval (BCFi) and overall survival (OS) (n = 437) in premenopausal women randomised to 2 years of tamoxifen versus no systemic treatment irrespective of hormone-receptor status. Intrinsic subtyping added independent prognostic information in patients with oestrogen receptor-positive/human epidermal growth factor 2-negative tumours for BCFi and OS after maximum follow-up (overall P-value 0.02 and 0.006, respectively) and those with high versus low ROR had worse prognosis (maximum follow-up: hazard ratio (HR)BCFi: 1.70, P = 0.04). The prognostic information by ROR was similar regarding OS and in multivariable analysis. These results support that PAM50 subtyping and ROR score provide long-term prognostic information in premenopausal women. Moreover, tamoxifen reduced the incidence of breast cancer events only in patients with Luminal APAM50 tumours (0–10 years: HRBCFi(Luminal A): 0.41, HRBCFi(Luminal B): 1.19, Pinteraction = 0.02).
Trial registration: This trial is registered in the ISRCTN database, trial ID: ISRCTN12474687.
Subject terms: Breast cancer, Tumour biomarkers, Predictive markers, Prognostic markers
Introduction
The classification of breast cancer tumours by gene expression analysis into intrinsic subtypes (Luminal A, Luminal B, human epidermal growth factor receptor 2-enriched [HER2-E], and Basal-like), is well-established1–3. Genomic testing is recommended as a complement to conventional risk assessment in postmenopausal patients with equivocal risk of recurrence4,5. PAM50 intrinsic subtyping and risk of recurrence (ROR) score, initially developed by Parker et al., are included in the Prosigna© Breast Cancer Prognostic Gene Signature Assay3,6. This is approved and validated for postmenopausal women with oestrogen receptor-positive/HER2-negative (ER+/HER2−) tumours allocated to 5 years of endocrine therapy, and ROR score provides prognostic information7–11.
The prognostic value of PAM50 subtypes and ROR score in premenopausal women remains unclear. Previous studies have indicated that PAM50 and ROR score are applicable also for premenopausal women12–15. Moreover, some of these studies also demonstrated a possible predictive effect of PAM50 subtypes for tamoxifen and chemotherapy benefit12,13. Surrogate classification of breast cancer tumours into Luminal ASurrogate Classification, (SC) and Luminal BSC, using immunohistochemistry (IHC)/in situ hybridisation (ISH), was used in clinical settings before multigene assays were broadly implemented for prognostication and guiding decisions about adjuvant treatments. However, surrogate classifications have shown poor concordance to the corresponding intrinsic subtypes16–19, resulting in suboptimal risk estimation for patients with ER+/HER2− tumours.
In the SBII:2pre trial, premenopausal women were randomised between 2 years of adjuvant tamoxifen or no adjuvant systemic therapy (control), irrespective of hormone-receptor status and thus one third of the patients had ER-negative tumours. Availability of long-term follow-up data (>30 years) and preserved archival tumour tissues from the participants allowed us to assess the long-term effects across gene expression profiles and subtypes in this cohort.
The primary aim of this study was to investigate the prognostic value of PAM50 intrinsic subtypes and ROR score in premenopausal patients with ER+/HER2− tumours. The secondary aims were to compare luminal PAM50 and St. Gallen 2013 surrogate subtypes and to evaluate if luminal PAM50 subtypes can be used to predict tamoxifen benefit in premenopausal patients.
Results
Sample availability and cohort characteristics
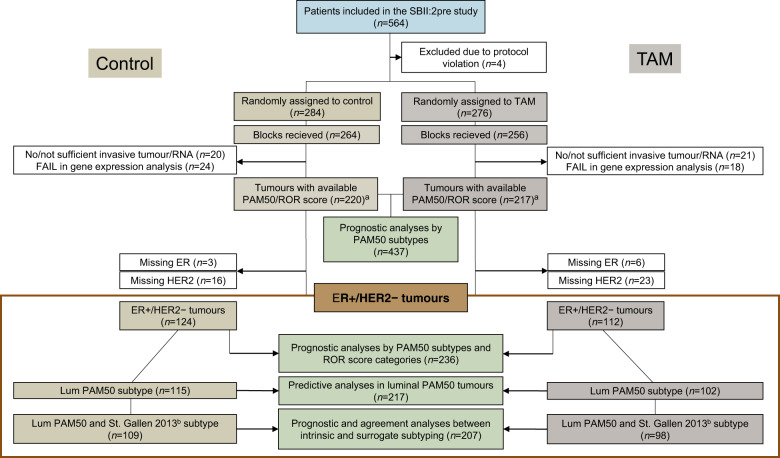
Gene expression analyses were successfully performed for 220 and 217 tumours (and the corresponding number of patients) in the control and tamoxifen arms, respectively (Fig. 1). The median follow-up time for those with no breast cancer events regarding breast cancer-free interval (BCFi) and overall survival (OS) were 28 and 33 years, respectively. Patient and tumour characteristics for the entire cohort and for the ER+/HER2− cohort are presented in Table 1.
Fig. 1. Flow chart of the included patients.
aAvailable ROR score categories in n = 219 and n = 216 patients in the control (no systemic treatment) and tamoxifen arm, respectively, due to n = 1 missing nodal status for one patient in each treatment arm. bDefined accordingly: LumASC, low Ki67 (<20%) and high PR (≥20%); LumBSC, high Ki67 (≥20%) and/or low PR (<20%). Cases with missing re-evaluated PR data were substituted (n = 2 in the control arm) with previously available IHC data for PR. ER oestrogen receptor, HER2 human epidermal growth factor receptor 2, IHC immunohistochemistry, Lum Luminal, PR progesterone receptor, ROR risk of recurrence, SC surrogate classification, TAM tamoxifen.
Table 1.
Patient and tumour characteristics for the whole study cohort (n = 560) by study arm and for the ER-positive/HER2-negative subgroup.
| Initial study cohort (n = 560) | ER+/HER2− cohort (n = 280) | |||
|---|---|---|---|---|
| Characteristics | Control group n (%) | TAM-treated group n (%) | Control group n (%) | TAM-treated group n (%) |
| Follow-up BCFi/OS (years)a | ||||
| Median | 28/33 | 28/33 | 28/33 | 28/33 |
| Range (10th–90th percentiles) | (25–31)/(30–35) | (25–30)/(31–35) | (26–31)/(30–35) | (25–30)/(30–35) |
| Age (years) | ||||
| Median | 45 | 45 | 46 | 46 |
| Range | 27–58 | 26–57 | 27–54 | 33–57 |
| <40 | 59 (21) | 51 (19) | 24 (16) | 17 (13) |
| ≥40 | 225 (79) | 225 (82) | 124 (84) | 115 (87) |
| Tumour size (mm) | ||||
| Median | 22 | 25 | 22 | 23 |
| Range | 2–50 | 5–75 | 2–50 | 8–50 |
| ≤20 | 121 (43) | 86 (31) | 70 (47) | 48 (37) |
| >20 | 163 (57) | 189 (69) | 78 (53) | 83 (63) |
| Missing | 0 | 1 | 0 | 1 |
| Nodal status | ||||
| Median number of positive nodes | 1 | 1 | 1.5 | 1 |
| Range | 0–22 | 0–21 | 0–15 | 0–17 |
| Node-negative | 75 (27) | 83 (30) | 35 (24) | 36 (27) |
| Node-positive | 208 (74) | 192 (70) | 113 (76) | 96 (73) |
| Missing | 1 | 1 | 0 | 0 |
| NHG | ||||
| 1 | 32 (12) | 27 (11) | 25 (17) | 22 (17) |
| 2 | 115 (44) | 105 (42) | 88 (60) | 68 (53) |
| 3 | 116 (44) | 117 (47) | 33 (23) | 39 (30) |
| Missing | 21 | 27 | 2 | 3 |
| ER | ||||
| Positive | 191 (70) | 171 (65) | 148 (100) | 132 (100) |
| Negative | 84 (31) | 91 (35) | 0 | 0 |
| Missing | 9 | 14 | 0 | 0 |
| PR | ||||
| Positive | 185 (67) | 163 (61) | 139 (94) | 118 (90) |
| Negative | 92 (33) | 103 (39) | 9 (6) | 13 (10) |
| Missing | 7 | 10 | 0 | 1 |
| HER2 | ||||
| Negative | 203 (84) | 197 (87) | 148 (100) | 132 (100) |
| Positive | 38 (16) | 30 (13) | 0 | 0 |
| Missing | 43 | 49 | 0 | 0 |
| LVI | ||||
| Absent | 138 (56) | 124 (52) | 75 (55) | 64 (53) |
| Present | 109 (44) | 113 (48) | 62 (45) | 56 (47) |
| Missing | 37 | 39 | 11 | 12 |
| Ki67 (%) | ||||
| <14 | 18 (8) | 25 (11) | 13 (10) | 18 (16) |
| 14–19 | 25 (11) | 27 (12) | 22 (18) | 15 (13) |
| ≥20 | 184 (81) | 167 (76) | 91 (72) | 82 (71) |
| Missing | 57 | 57 | 22 | 17 |
| TILs (%) | ||||
| <10 | 129 (52) | 123 (52) | 90 (66) | 86 (72) |
| 10–49 | 86 (35) | 75 (32) | 36 (26) | 29 (24) |
| 50–74 | 27 (11) | 31 (13) | 11 (8) | 5 (4) |
| ≥75 | 7 (3) | 8 (3) | 0 | 0 |
| Missing | 35 | 39 | 11 | 12 |
| Histopathological type | ||||
| Ductal/NST | 209 (84) | 200 (83) | 123 (84) | 111 (84) |
| Lobular | 22 (9) | 21 (9) | 18 (12) | 14 (11) |
| Medullary | 14 (6) | 11 (5) | 2 (1) | 1 (1) |
| Other | 5 (2) | 10 (4) | 3 (2) | 6 (5) |
| Missing | 34 | 34 | 2 | 0 |
| Subtype (IHC/ISH) | ||||
| Luminal/HER2 | 148 (64) | 132 (61) | 148 (100) | 132 (100) |
| HER2+ | 38 (16) | 30 (14) | 0 | 0 |
| TNBC | 46 (20) | 54 (25) | 0 | 0 |
| Missing | 52 | 60 | 0 | 0 |
| PAM50 intrinsic subtype | ||||
| LumA | 101 (46) | 90 (42) | 82 (66) | 66 (59) |
| LumB | 41 (19) | 42 (19) | 33 (27) | 36 (32) |
| HER2-E | 39 (18) | 35 (16) | 8 (7) | 4 (4) |
| Basal-like | 39 (18) | 50 (23) | 1 (1) | 6 (5) |
| Missing | 64 | 59 | 24 | 20 |
| ROR scoreb | ||||
| Median | 56 | 56 | 45 | 50 |
| Range | 0–94 | 1–94 | 4–94 | 12–94 |
| Low | 22 (10) | 23 (11) | 16 (13) | 15 (13) |
| Intermediate | 48 (22) | 55 (26) | 35 (28) | 29 (26) |
| High | 149 (68) | 138 (64) | 73 (59) | 68 (61) |
| Missing | 65 | 60 | 24 | 20 |
| N0 (node-negative)c | ||||
| Low (0–40) | 19 (33) | 20 (32) | 15 (52) | 14 (45) |
| Intermediate (41–60) | 11 (19) | 21 (34) | 6 (21) | 7 (23) |
| High (61–100) | 27 (47) | 21 (34) | 8 (28) | 10 (32) |
| Missing | 18 | 21 | 6 | 5 |
| N1 (1–3 positive nodes)c | ||||
| Low (0–15) | 3 (3) | 3 (3) | 1 (2) | 1 (2) |
| Intermediate (16–40) | 37 (35) | 34 (32) | 29 (45) | 22 (38) |
| High (41–100) | 65 (62) | 71 (66) | 35 (54) | 35 (60) |
| Missing | 34 | 28 | 12 | 11 |
| N2 (≥4 positive nodes)c | ||||
| High (0–100) | 57 (100) | 46 (100) | 30 (100) | 23 (100) |
| Missing | 12 | 10 | 6 | 4 |
BCFi breast cancer-free interval, ER oestrogen receptor, HER2-E human epidermal growth factor receptor 2-enriched, IHC immunohistochemistry, ISH in situ hybridisation, Lum Luminal, LVI lymphovascular invasion, NHG Nottingham histological grade, NST no special type, OS overall survival, PR progesterone receptor, ROR risk of recurrence, TAM tamoxifen, TILs tumour infiltrating lymphocytes, TNBC triple-negative breast cancer.
aPatients without events.
bThe ROR score categories were defined by the following cut-offs based on N-status; N0; low: 0–40, intermediate: 41–60, high: 61–100, N1; low: 0–15, intermediate: 15–40, high: 41–100, N2; high: 0–100.
cROR score stratified by nodal status.
The proportions of Luminal A, Luminal B, HER2-E, and Basal-like intrinsic subtypes by PAM50 (n = 437) were 44%, 19%, 17%, and 20%, respectively (Supplementary Fig. 1). The median ROR score was 56 and the proportions among patients with available nodal status classified into the low, intermediate, and high ROR categories (n = 435) were 10%, 24%, and 66%, respectively.
Prognostic value of PAM50 subtypes
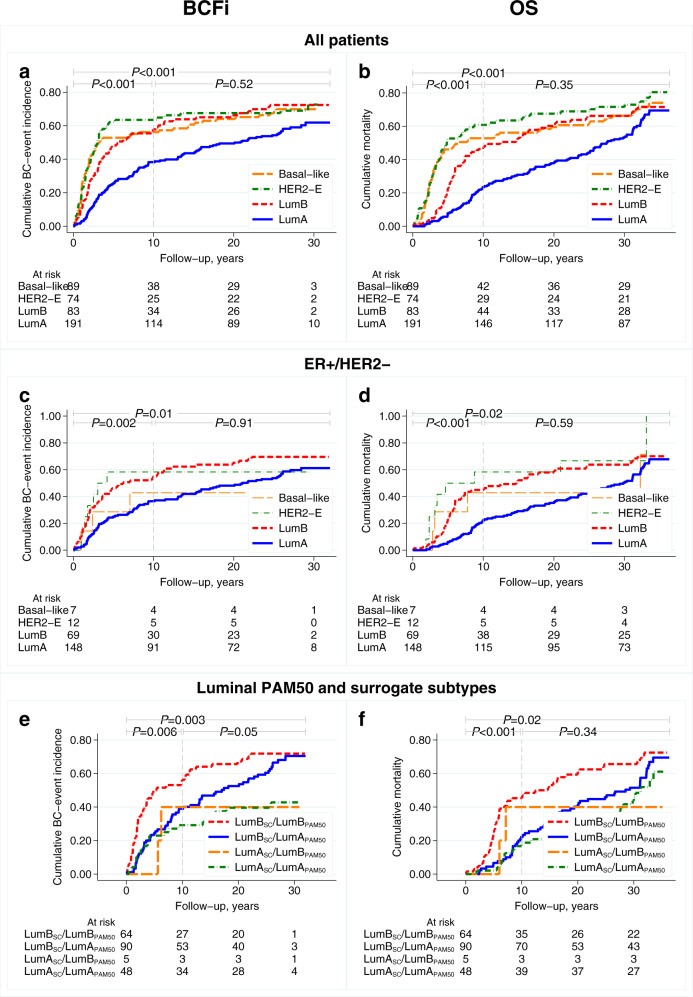
Cumulative incidence curves for BCFi and OS by PAM50 subtypes are presented in Fig. 2a, b for all patients and in Fig. 2c, d for patients with ER+/HER2− tumours. After the maximum follow-up period, patients with ER+/HER2− and Luminal BPAM50 tumours had a higher cumulative incidence of breast cancer events than patients whose tumours were categorised as Luminal APAM50 (hazard ratio [HR]BCFi: 1.56, 95% confidence interval [CI] 1.09–2.22, P = 0.01). The results were similar for the period 0–10 years (HRBCFi: 1.93, 95% CI: 1.27–2.93, P = 0.002; Table 2). The results also indicated increased overall mortality for patients whose tumours were Luminal BPAM50 as compared to Luminal APAM50 (maximum follow-up: HROS: 1.49, 95% CI: 1.05–2.12, P = 0.03; 0–10 years: HROS: 2.55, 95% CI: 1.56–4.17, P < 0.001). The results were similar after adjusting for other clinicopathological variables (Table 2) and for all included patients irrespective of hormone-receptor status (Supplementary Table 1). Additionally, cumulative incidence curves for recurrence-free interval (RFi) with essentially the same results are depicted in Supplementary Fig. 2a–d.
Fig. 2. Cumulative incidence curves for BCFi and OS by PAM50 subtypes.
(a, b) All included patients, (c, d) patients with ER-positive/HER2-negative tumours, and (e, f) patients with available intrinsic PAM50 and surrogate subtyping by St. Gallen 2013. Overall P-values from log rank test, Gehan’s version for BCFi, for maximum follow-up and for different time intervals. BCFi breast cancer-free interval, HER2-E human epidermal growth factor receptor 2-enriched, Lum Luminal, OS overall survival, SC surrogate classification.
Table 2.
Cox regression uni- and multivariable models for BCFi and OS by PAM50 subtypes and St. Gallen 2013 surrogate subtypes for different time intervals in patients with ER-positive/HER2-negative tumours.
| Univariable | Multivariablea | |||
|---|---|---|---|---|
| BCFi | OS | BCFi | OS | |
| HR (95% CI); P-value | ||||
|
PAM50 subtype (ER+/HER2− cohort) |
0–10 years | |||
| (n = 236, n = 102 events) overall P-value = 0.01d | (n = 236, n = 74 events) overall P-value < 0.001d | (n = 233, n = 102 events) overall P-value = 0.04d | (n = 233, n = 74 events) overall P-value = 0.01d | |
| LumA (Ref.) | 1.00 | 1.00 | 1.00 | 1.00 |
| LumB | 1.93 (1.27–2.93); 0.002 | 2.55 (1.56–4.17); <0.001 | 2.02 (1.26–3.26); 0.004 | 2.42 (1.37–4.28); 0.002 |
| HER2-E | 2.22 (1.01–4.91); 0.05 | 4.34 (1.92–9.85); <0.001 | 1.74 (0.69–4.40); 0.24 | 3.35 (1.25–8.98); 0.02 |
| Basal-like | 1.25 (0.39–4.04); 0.70 | 2.38 (0.72–7.85); 0.16 | 1.17 (0.34–4.03); 0.80 | 1.67 (0.46–6.00); 0.43 |
| >10 yearsb | ||||
| (n = 130, n = 43 events) | (n = 162, n = 77 events) overall P-value = 0.74d | (n = 127, n = 42 events) | (n = 159, n = 77 events) overall P-value = 0.31d | |
| LumA (Ref.) | 1.00 | 1.00 | 1.00 | 1.00 |
| LumB | 0.92 (0.45–1.87); 0.81 | 0.86 (0.50–1.48); 0.58 | 1.91 (0.79–4.59); 0.15 | 1.66 (0.87–3.16); 0.12 |
| HER2-E | – | 0.76 (0.18–3.12); 0.70 | – | 3.50 (0.63–19.45); 0.15 |
| Basal-like | – | 0.38 (0.05–2.76); 0.34 | – | 2.36 (0.25–22.11); 0.45 |
| Maximum follow-up timec | ||||
| (n = 236, n = 145 events) overall P-value = 0.08d | (n = 236, n = 151 events) overall P-value = 0.05d | (n = 233, n = 144 events) overall P-value = 0.02d | (n = 233, n = 151 events) overall P-value = 0.006d | |
| LumA (Ref.) | 1.00 | 1.00 | 1.00 | 1.00 |
| LumB | 1.56 (1.09–2.22); 0.01 | 1.49 (1.05–2.12); 0.03 | 1.89 (1.25–2.86); 0.003 | 1.93 (1.28–2.92); 0.002 |
| HER2-E | 1.35 (0.62–2.93); 0.45 | 2.11 (1.06–4.20); 0.04 | 1.50 (0.62–3.67); 0.37 | 3.04 (1.33–6.95); 0.008 |
| Basal-like | 0.73 (0.23–2.30); 0.59 | 1.05 (0.38–2.87); 0.93 | 0.82 (0.24–2.74); 0.75 | 1.45 (0.49–4.28); 0.50 |
| St. Gallen 2013/PAM50 subtype | 0–10 years | |||
| (n = 207, n = 87 events) overall P-value = 0.007d | (n = 207, n = 60 events) overall P-value = 0.001d | (n = 205, n = 87 events) overall P-value = 0.04d | (n = 205, n = 60 events) overall P-value = 0.03d | |
| LumBSC/LumBPAM50 (Ref.) | 1.00 | 1.00 | 1.00 | 1.00 |
| LumBSC/LumAPAM50 | 0.52 (0.33–0.83); 0.006 | 0.37 (0.21–0.66); 0.001 | 0.50 (0.29–0.84); 0.009 | 0.38 (0.20–0.74); 0.004 |
| LumASC/LumBPAM50 | 0.49 (0.12–2.05); 0.33 | 0.76 (0.18–3.20); 0.71 | 0.77 (0.18–3.32); 0.72 | 1.12 (0.25–5.07); 0.88 |
| LumASC/LumAPAM50 | 0.39 (0.21–0.73); 0.003 | 0.32 (0.15–0.68); 0.003 | 0.45 (0.23–0.91); 0.03 | 0.44 (0.19–1.01); 0.05 |
| >10 yearsb | ||||
| (n = 117, n = 42 events) overall P-value = 0.06e | (n = 147, n = 70 events) overall P-value = 0.56e | (n = 115, n = 41 events) overall P-value = 0.06e | (n = 145, n = 70 events) overall P-value = 0.56e | |
| LumBSC/LumBPAM50 (Ref.) | 1.00 | 1.00 | 1.00 | 1.00 |
| LumBSC/LumAPAM50 | 1.30 (0.62–2.69); 0.49 | 1.11 (0.62–1.98); 0.72 | 0.64 (0.26–1.60); 0.34 | 0.59 (0.30–1.15); 0.12 |
| LumASC/LumBPAM50 | – | – | – | – |
| LumASC/LumAPAM50 | 0.45 (0.16–1.23); 0.12 | 0.81 (0.41–1.59); 0.54 | 0.26 (0.08–0.80); 0.02 | 0.38 (0.18–0.84);0.02 |
| Maximum follow-up timec | ||||
| (n = 207, n = 129 events) overall P-value = 0.004d | (n = 207, n = 130 events) overall P-value = 0.02d | (n = 205, n = 128 events) overall P-value = 0.01d | (n = 205, n = 130 events) overall P-value = 0.008d | |
| LumBSC/LumBPAM50 (Ref.) | 1.00 | 1.00 | 1.00 | 1.00 |
| LumBSC/LumAPAM50 | 0.70 (0.47–1.02); 0.06 | 0.65 (0.44–0.96); 0.03 | 0.58 (0.37–0.90); 0.02 | 0.50 (0.32–0.79); 0.003 |
| LumASC/LumBPAM50 | 0.34 (0.08–1.41); 0.14 | 0.37 (0.09–1.52); 0.17 | 0.52 (0.12–2.24); 0.38 | 0.48 (0.11–2.05): 0.32 |
| LumASC/LumAPAM50 | 0.39 (0.23–0.67); 0.001 | 0.51 (0.32–0.83); 0.007 | 0.39 (0.21–0.70); 0.002 | 0.43 (0.25–0.74); 0.003 |
BCFi breast cancer-free interval, CI confidence interval, ER oestrogen receptor, HER2-E human epidermal growth factor receptor 2-enriched, HR hazard ratio, Lum Luminal, NHG Nottingham histological grade, OS overall survival, SC surrogate classification.
aAll analyses are stratified by study region and adjusted for age (continuous), tumour size (>20 vs ≤20 mm), NHG (1 vs 2 vs 3), nodal status (N0 vs N1 vs N2) and treatment arm.
bFrom year 10 to maximum follow-up time.
c32 and 36 years regarding BCFi and OS, respectively.
dOverall P-value, three degree of freedom Wald test.
eOverall P-value, two degree of freedom Wald test.
Agreement and prognostic effect of luminal PAM50 and St. Gallen 2013 surrogate subtypes
In the agreement analyses (ER + /HER2 − cohort, n = 207), 67% and 33% were assessed as Luminal APAM50 and Luminal BPAM50, respectively. The corresponding figures for St. Gallen 2013 surrogate subtypes were 26% and 74%, respectively. In total, 58% (90/154) of patients classified as Luminal BSC were classified as Luminal APAM50 (Table 3).
Table 3.
Distribution of luminal subtypes according to PAM50 and St. Gallen 2013 surrogate subtyping (n = 207) and corresponding agreement analyses (percentage and kappa [κ] statistic).
| PAM50 subtype | St. Gallen 2013 surrogate subtyping n (%) | |
|---|---|---|
| LumASC (n = 53) | LumBSC (n = 154) | |
| LumAPAM50 (n = 138) | 48 (91) | 90 (58) |
| LumBPAM50 (n = 69) | 5 (9) | 64 (42) |
| Agreement (%) | 54 | |
| Kappa (κ) (95% CI) | 0.21 (0.12–0.30) | |
CI confidence interval, Lum Luminal, SC surrogate classification.
The difference in outcomes between the four different combinations of the St. Gallen 2013 surrogate and PAM50 subtypes (Luminal BSC/Luminal BPAM50 [n = 64], Luminal BSC/Luminal APAM50 [n = 90], Luminal ASC/Luminal BPAM50 [n = 5], and Luminal ASC/Luminal APAM50 [n = 48]) is illustrated in Fig. 2e, f and Supplementary Fig. 2e, f. After 10 years of follow-up, patients with tumours classified as Luminal BSC/Luminal APAM50, had better prognosis than those with tumours classified as Luminal BSC/Luminal BPAM50 (HRBCFi: 0.52, 95% CI: 0.33–0.83, P = 0.006; HROS: 0.37, 95% CI: 0.21–0.66, P = 0.001; Table 2). A proportion (91/207) of the cohort was at risk for breast cancer events at 20 years of follow-up, hence the prognostic effects became weaker with long-term (>30 years) follow-up and the results were similar in multivariable analyses (Table 2).
Predictive value of luminal PAM50 subtyping for tamoxifen benefit
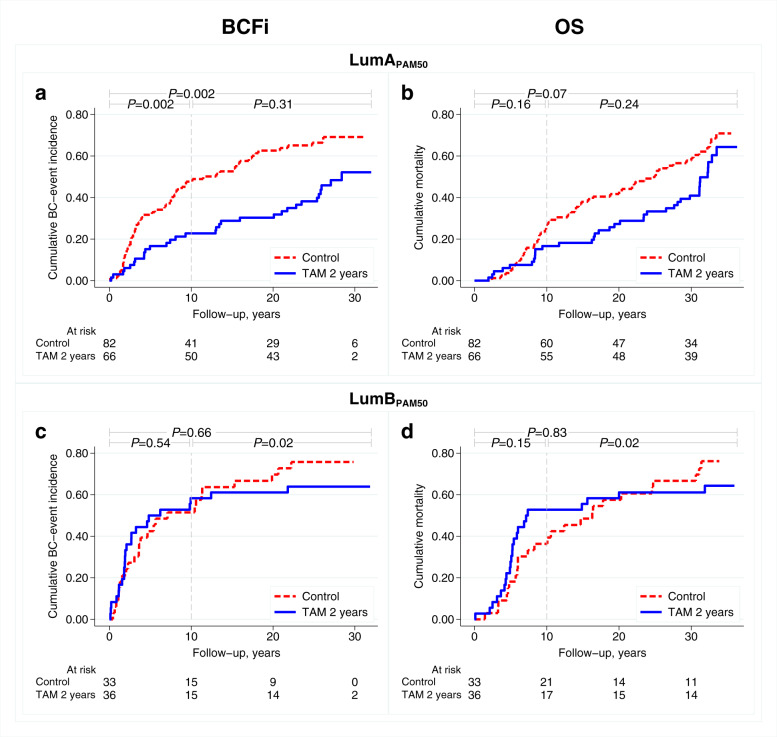
After 10 years of follow-up, a beneficial effect of adjuvant tamoxifen was observed in patients with ER+/HER2− and Luminal APAM50 tumours (HRBCFi: 0.41, 95% CI: 0.23–0.74, P = 0.003; Fig. 3a); however, not for patients with Luminal BPAM50 tumours (HRBCFi: 1.19, 95% CI: 0.63–2.27, P = 0.59; Fig. 3c). Hence, the effect of tamoxifen was threefold better in patients with Luminal APAM50 tumours as compared with those with Luminal BPAM50 tumours (interaction: HRBCFi: 0.34, 95% CI: 0.14–0.83, P = 0.02). Similar results were observed for OS (Table 4 and Fig. 3b, d) and in the multivariable analyses (Supplementary Table 2), but was not as evident after maximum follow-up. Corresponding cumulative incidence curves for RFi are presented in Supplementary Fig. 3. When selecting all patients with luminal PAM50 tumours, regardless of ER/HER2 status (n = 274), the findings after 10 years of follow-up were essentially the same (interaction: HRBCFi: 0.45, 95% CI: 0.21–0.96, P = 0.04)).
Fig. 3. Cumulative incidence curves for BCFi and OS according to treatment arm.
(a, b) Patients with LumA and (c, d) LumB tumours by PAM50. P-values from log rank test, Gehan’s version for BCFi, for maximum follow-up and for different time intervals. BCFi breast cancer-free interval, Lum Luminal, OS overall survival, TAM tamoxifen.
Table 4.
Cox regression models for BCFi and OS by luminal PAM50 subtypes, tamoxifen treatment, and PAM50 subtype by treatment interaction in patients with ER-positive/HER2-negative tumours.
| BCFi | OS | |
|---|---|---|
| HR (95% CI); P-value | ||
| 0–10 years | ||
| (n = 217, n = 92 events) | (n = 217, n = 64 events) | |
| TAM vs control in LumAPAM50 | 0.41 (0.23–0.74); 0.003 | 0.61 (0.30–1.26); 0.18 |
| TAM vs control in LumBPAM50 | 1.19 (0.63–2.27); 0.59 | 1.76 (0.85–3.63); 0.13 |
| Interaction luminal PAM50 subtype x TAM | 0.34 (0.14–0.83); 0.02 | 0.35 (0.13–0.97); 0.04 |
| >10 yearsa | ||
| (n = 121, n = 43 events) | (n = 153, n = 74 events) | |
| TAM vs control in LumAPAM50 | 0.69 (0.35–1.37); 0.29 | 0.74 (0.44–1.25); 0.26 |
| TAM vs control in LumBPAM50 | 0.17 (0.04–0.80); 0.03 | 0.25 (0.08–0.77); 0.02 |
| Interaction luminal PAM50 subtype x TAM | 4.05 (0.74–22.1); 0.11 | 2.95 (0.85–10.2); 0.09 |
| Maximum follow-up timeb | ||
| (n = 217, n = 135 events) | (n = 217, n = 138 events) | |
| TAM vs control in LumAPAM50 | 0.52 (0.34–0.81); 0.004 | 0.71 (0.46–1.08); 0.11 |
| TAM vs control in LumBPAM50 | 0.80 (0.45–1.41); 0.44 | 0.87 (0.49–1.54); 0.63 |
| Interaction luminal PAM50 subtype x TAM | 0.65 (0.32–1.34); 0.24 | 0.82 (0.40–1.65); 0.57 |
All analyses are stratified by study region.
BCFi, breast cancer-free interval; CI, confidence interval; ER, oestrogen receptor; HER2, human epidermal growth factor receptor 2; HR, hazard ratio; Lum, Luminal; OS, overall survival; TAM, tamoxifen.
aFrom year 10 to maximum follow-up time.
b32 and 36 years regarding BCFi and OS, respectively.
Prognostic value of ROR score (ER+/HER2− subgroup)
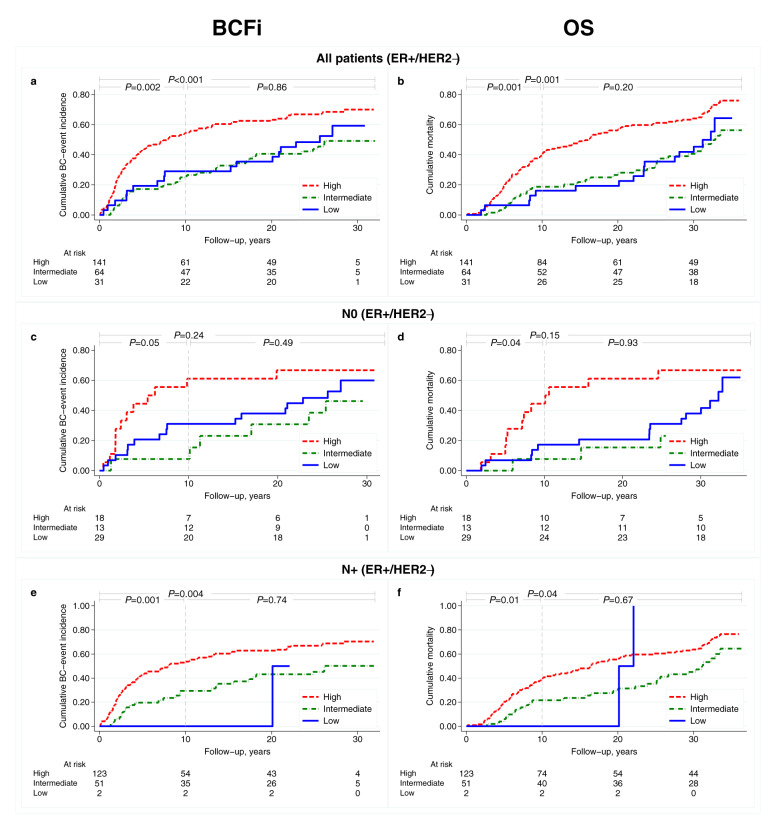
Among all patients (ER+/HER2− subgroup, n = 236), the distributions of low, intermediate, and high ROR score categories were: 13%, 27%, and 60%, respectively. The outcomes are illustrated in Fig. 4 and in Supplementary Fig. 4. For all patients with ER+/HER2− tumours, high vs low ROR score was associated with worse outcomes after 10 years of follow-up (HRBCFi: 2.36, 95% CI: 1.18–4.72, P = 0.02; Table 5). This effect was less pronounced after maximum long-term follow-up (HRBCFi: 1.70, 95% CI: 1.01–2.85, P = 0.04). The corresponding results for OS and the multivariable analyses are presented in Table 5 and in Supplementary Table 3, respectively.
Fig. 4. Cumulative incidence curves for BCFi and OS by ROR score categories for patients with ER-positive/HER2-negative tumors.
(a, b) all patients (c, d) node-negative, and (e, f) node-positive patients. Overall P-values from log rank test, Gehan’s version for BCFi, for maximum follow-up and for different time intervals. BCFi breast cancer-free interval, ER oestrogen receptor, HER2 human epidermal growth factor receptor 2, OS overall survival, ROR risk of recurrence.
Table 5.
Cox regression univariable models for BCFi and OS by ROR score categories for different time intervals for patients with ER-positive/HER2-negative tumours in all patients, node-negative, and node-positive (1–3 positive nodes) subgroups of patients.
| All patients (ER+/HER2−) | Node-negative (ER+/HER2−) | Node-positive (ER+/HER2−)a | ||||
|---|---|---|---|---|---|---|
| BCFi | OS | BCFi | OS | BCFi | OS | |
| HR (95% CI); P-value | ||||||
| 0–10 years | ||||||
| ROR score | (n = 236, n = 102 events) overall P-value < 0.001d | (n = 236, n = 74 events) overall P-value = 0.002d | (n = 60, n = 21 events) overall P-value = 0.01d | (n = 60, n = 14 events) overall P-value = 0.04d | (n = 121, n = 49 events) | (n = 121, n = 36 events) |
| Low (Ref.) | 1.00 | 1.00 | 1.00 | 1.00 | – | – |
| Intermediate | 0.83 (0.37–1.88); 0.66 | 1.19 (0.42–3.37); 0.75 | 0.20 (0.03–1.59); 0.13 | 0.40 (0.05–3.43); 0.40 | 1.00 | 1.00 |
| High | 2.36 (1.18–4.72); 0.02 | 2.99 (1.19–7.46); 0.02 | 2.53 (1.04–6.12); 0.04 | 3.17 (1.03–9.75); 0.04 | 1.99 (1.08–3.66); 0.03 | 1.84 (0.91–3.74); 0.09 |
| >10 yearsb | ||||||
| ROR score | (n = 130, n = 43 events) overall P-value = 0.93d | (n = 162, n = 77 events) overall P-value = 0.19d | (n = 39, n = 13 events) overall P-value = 0.56d | (n = 46, n = 16 events) overall P-value = 0.40d | (n = 69, n = 21 events) | (n = 84, n = 38 events) |
| Low (Ref.) | 1.00 | 1.00 | 1.00 | 1.00 | – | – |
| Intermediate | 0.85 (0.36–2.01); 0.71 | 0.76 (0.37–1.56); 0.45 | 1.22 (0.38–3.92); 0.74 | 0.38 (0.08–1.75); 0.21 | 1.00 | 1.00 |
| High | 0.88 (0.39–2.01); 0.77 | 1.24 (0.65–2.35); 0.51 | 0.37 (0.05–3.03); 0.36 | 1.18 (0.36–3.79); 0.79 | 1.19 (0.50–2.80); 0.70 | 1.02 (0.54–1.93); 0.96 |
| Maximum follow-upc | ||||||
| ROR score | (n = 236, n = 145 events) overall P-value = 0.001d | (n = 236, n = 151 events) overall P-value = 0.001d | (n = 60, n = 34 events) overall P-value = 0.15d | (n = 60, n = 30 events) overall P-value = 0.03d | (n = 121, n = 70 events) | (n = 121, n = 74 events) |
| Low (Ref.) | 1.00 | 1.00 | 1.00 | 1.00 | – | – |
| Intermediate | 0.84 (0.47–1.53); 0.58 | 0.89 (0.49–1.61); 0.70 | 0.66 (0.26–1.69); 0.39 | 0.39 (0.11–1.35); 0.14 | 1.00 | 1.00 |
| High | 1.70 (1.01–2.85); 0.04 | 1.77 (1.06–2.97); 0.03 | 1.69 (0.79–3.58); 0.17 | 1.96 (0.91–4.22); 0.09 | 1.68 (1.03–2.75); 0.04 | 1.34 (0.84–2.14); 0.22 |
All analyses are stratified by study region. The ROR score categories are defined by the following cut-offs based on N-status; N0; low: 0–40, intermediate: 41–60, high: 61–100, N1; low: 0–15, intermediate: 16–40, high: 41–100, N2: high: 0–100.
BCFi breast cancer-free interval, CI confidence interval, ER oestrogen receptor, HER2 human epidermal growth factor receptor 2, HR hazard ratio, OS overall survival, ROR risk of recurrence.
aOnly N1 (1–3 positive nodes) are included in the node-positive definition. Since only n = 2 patients are defined as ROR low in the N1 category, these are omitted from the analyses.
bFrom year 10 to maximum follow-up time.
c32 and 36 years regarding BCFi and OS, respectively.
dTwo-degree of freedom Wald test.
Stratified by nodal status, the distributions of ROR score categories were: node-negative (N0) (n = 60): 48%, 22% and 30%, N+ (1–3 positive nodes, n = 123): 2%, 42% and 57%, respectively. High vs low ROR score yielded 2.5- and 1.7- fold increased incidence of breast cancer events in N0 patients after 10 years of follow-up and maximum follow-up, respectively (Table 5). Due to small sample size (n = 2), the low ROR category was omitted in the analysis of N+ (1–3 positive nodes) patients and the results indicated that intermediate ROR score was associated with lower incidence of breast cancer events than high ROR score (Table 5).
Discussion
This study demonstrates that PAM50 subtypes and ROR score could provide long-term prognostic information in premenopausal patients, and indicates a possible tamoxifen-predictive effect by luminal intrinsic subtyping after 10 years of follow-up. The Prosigna© Breast Cancer Prognostic Gene Signature Assay, which can use formalin-fixed paraffin-embedded tissues in a decentralised mode20, is validated in postmenopausal women7,8. Our results demonstrated that premenopausal patients with Luminal BPAM50 as compared with Luminal APAM50 tumours, had a > 1.4-fold higher incidence of breast cancer events and death after long-term follow-up.
Our results confirmed the suboptimal agreement between luminal intrinsic and surrogate subtyping16–19, and we demonstrated that over 50% of Luminal BSC tumours were re-classified as Luminal APAM50. These results are in agreement with the report by Viale et al. using BluePrint/MammaPrint17. Importantly, the re-classification in our study was translated into improved prognosis and this emphasises the possibility of overestimating the risk for breast cancer events in patients with surrogate Luminal B tumours, which could affect treatment decisions.
Two years of adjuvant tamoxifen was previously reported to be associated with a long-term survival benefit and reduction of breast cancer events for ER-positive patients by the SBII:2pre trial21,22. In this study, during the first 10 years of follow-up, a tamoxifen-treatment effect was seen in patients with Luminal APAM50 tumours, but not in those with Luminal BPAM50 tumours. However, the evidence of treatment interaction was weak in the subsequent follow-up time periods. This indicated that the luminal PAM50 subtype could be a predictive marker for tamoxifen benefit in addition to ER status. Our finding was observed regardless of ER and HER2 status by IHC/ISH, suggesting the PAM50 subtyping could potentially be clinically used upfront to stratify premenopausal patients to tamoxifen therapy. The luminal PAM50 subtypes have previously been reported to be associated with benefit from 5 years adjuvant tamoxifen treatment in premenopausal women (n = 398); however, no separate treatment effects in patients with Luminal A and B were reported12. Yu et al. presented a long-term beneficial effect of 5-year tamoxifen treatment in postmenopausal women with Luminal APAM50 tumours, and the effect attenuated over time in patients with Luminal BPAM50 tumours23.
This study demonstrated better long-term prognosis for patients with low vs high ROR score. However, the trial was underpowered to define the prognostic value of ROR score by nodal status. In general, our cohort had more aggressive tumour characteristics and the outcome for node-negative patients with low ROR score was worse compared with postmenopausal women in the validation studies7,8,20. It remains unclear if premenopausal patients with low ROR score are potential candidates for abstaining adjuvant chemotherapy. Data from the TAILORx trial indicated a beneficial effect of adjuvant chemotherapy in patients ≤50 years and a recurrence score of 16–2524 and the results from the RxPONDER demonstrated that node-positive premenopausal women with a recurrence score ≤25 did benefit from additional chemotherapy25. These results are emphasising that further studies of multigene assays including premenopausal patients are warranted.
A strength of this study is the long-term follow-up data and the fact that it is based on a trial including only premenopausal women randomised to tamoxifen monotherapy vs control (systemically untreated patients). Moreover, reassessments of the progesterone receptor and Ki67 were performed for the surrogate subtypes and a well-established gene expression method was used. A limitation of this study is that the quality of old preserved tissues may result in uncertainty, especially regarding Ki67 assessment26. However, the required RNA quantity is minimal, and quality check of the RNA assured that gene expression output data were reliable. Further limitations include that the duration of endocrine treatment was shorter than current recommendation and, the power of this study was low due to the limited number of included patients. Even though we demonstrated that PAM50 subtyping and ROR score could separate premenopausal into groups with different risks of recurrence and death, it remains unclear if these can be used for de-escalation of adjuvant chemotherapy27.
In conclusion, PAM50 subtypes and ROR score provided independent prognostic information after long-term follow-up. After 10 years of follow-up, the re-classification of Luminal BSC tumours into Luminal APAM50 was associated with a lower incidence of breast cancer events. Moreover, the tamoxifen effect was associated with the Luminal A intrinsic subtype, independent of ER status.
Methods
Study population
The patients in this study were included in the SBII:2pre study, which randomised 564 premenopausal women between 2 years of adjuvant tamoxifen or no systemic treatment. Inclusion and exclusion criteria have been described previously and demonstrated long-term beneficial effect of tamoxifen treatment21,22,28,29. Patients were classified as premenopausal until one year after menstrual periods had stopped according to the study protocol (Supplementary Reference 1). The cohort included in this study is illustrated in Fig. 1.
Compliance with ethical requirements
Oral informed consent was obtained from all participants included in the SBII:2pre trial, and approval was given by the ethical committees in Lund and Linköping, Sweden. The oral consent was verified by a signature of the investigator in the registration form which was sent to the coordinating centre. The follow-up study was approved by the ethical committee of Lund (Dnr LU 2015/350) for extended follow-up as well as for genomic analysis (Dnr LU 2017/97). Biobank approval was cleared for all involved pathology departments.
Study endpoints and follow-up data
The primary endpoints were BCFi including any of the following first events: local, regional, or distant recurrence; contralateral breast cancer (invasive or ductal cancer in situ); or breast cancer-related death (data cut-off Nov 30 2016)22, according to the DATECAN recommendation30. The secondary endpoint was OS and follow-up data were retrieved from the Swedish Causes of Death Register (data cut-off 10 December 2020). In sensitivity analysis, we additionally reported on RFi excluding contralateral breast cancer events30. Results for maximum follow-up and the two time intervals 0–10 years and >10 years were reported.
Tumour characteristics and assessments of progesterone receptor (PR) and Ki67 status
Archival formalin-fixed paraffin-embedded (FFPE) tissues (n = 520) from breast tumours of the study participants were collected. Reassessments of PR (n = 464) and Ki67 (n = 463) status using whole tissue sections were performed according to Swedish national guidelines by a breast pathologist (UK)31. Data on ER, Nottingham histological grade (NHG), and HER2 was available as described previously21,28,29,32,33. These assessments were performed retrospectively and independently by two national reference breast pathologists28,33.
Gene expression analyses
1–5 sections (10 µm thick) from FFPE tissue with invasive breast carcinoma, were used to extract RNA (AllPrep DNA/RNA FFPE kit (Qiagen Cat:80234, Hilden, Germany)). Gene expression analysis was performed according to the manufacturer’s instructions using the NanoString Breast Cancer 360TM assay on an nCounter® SPRINT Profiler instrument (NanoString Technologies)34.
Housekeeping gene geomean quality control (QC) categorised samples as PASS/BORDERLINE (≥202) or FAIL (<202) with 91% (437/479) of the samples passing QC. PAM50 genes were normalised to the PAM50 housekeeper gene geomean. The correlation between the observed scaled expression for the PAM50 genes and a centroid for each of the four subtypes was then determined. The subtype with the greatest correlation value defined the intrinsic subtype. In the ROR score, a weighted sum of the proliferation score, the four subtype correlations and tumour size were used to calculate a score between 0 and 100. The categorisation of ROR score was determined based on nodal status according to the following definitions: N0 low: 0–40, intermediate: 41–60, high: 61–100, N1 (1–3 positive nodes); low: 0–15, intermediate: 16–40, high: 41–100, N2 (≥4 positive nodes); high: 0–10020.
Surrogate subtyping
The ER+/HER2− breast cancer tumours were classified as Luminal ASC or Luminal BSC according to the St. Gallen 2013 guidelines: Luminal ASC; low Ki67 (<20%) and high PR (≥20%), Luminal BSC; high Ki67 (≥20%) and/or low PR (<20%)35.
Statistical analyses
Cumulative incidence curves were used to illustrate outcomes for patient subgroups. The estimates for BCFi take the competing event death without a preceding breast cancer event into account. In analyses of RFi, also contralateral breast cancer as first event was treated as a competing risk. Evidence against equality of two or more cumulative incidence curves was evaluated using the log rank test. We used a trend version of the test for ordered groups and a modified version, derived by Geskus36, for comparison of cause-specific cumulative incidence curves (BCFi and RFi). Cox regression models, stratified for region, were used to estimate HRs with 95% CIs Cause-specific Cox regression was used for the endpoint BCFi, censoring the follow-up at time of death for patients who died without a registered breast cancer event. Similarly, the follow-up time was censored at the time of death without a preceding breast cancer event or at the diagnosis of contralateral breast cancer as first breast cancer related event in analyses of RFi. Proportional hazards assumptions were in general not met in analyses of long-term follow-up. The corresponding HRs should therefore be interpreted cautiously as average effects over time. Our way of handling this problem was to also calculate the relative effects with the follow-up restricted to 10 years. The evidence against proportional hazards, as measured by Schoenfeld’s test, was in general much lower for the two intervals 0–10 years and 10+ years compared to the evidence in analyses of maximum follow-up.
Log rank tests of prognostic or predictive effects are presented in figures whereas tests based on Cox regression models are presented in tables. For Cox models, we present both effects relative to a chosen reference category, with 95% CIs and P-values, and an overall Wald test of each factor. Multivariable analyses of PAM50 subtype and ROR categories were adjusted for established prognostic factors, but since nodal status and tumour stage are included in the definition of ROR, these factors were excluded from the set of adjustment variables in analyses of the independent prognostic effect of ROR categories.
For prognostic differences between luminal PAM50 and surrogate subtyping, a variable including the four combinations of Luminal APAM50, Luminal BPAM50, Luminal ASC and Luminal BSC tumours, was created. Percentage agreement and kappa (κ) statistics were used in agreement analyses. To evaluate the differential effect of tamoxifen benefit in luminal PAM50 tumours, a Cox model was fitted including an interaction variable between luminal PAM50 subgroup and treatment arm. The prognostic effect of ROR score was studied in ER+/HER2− patients and also stratified by nodal status.
The results are presented in accordance with the Reporting Recommendations for Tumour Marker Prognostic Studies (REMARK) where applicable37,38. All statistical tests were two-sided, and a P-value <0.05 was considered statistically significant. No adjustment for multiple testing was performed. All calculations were performed using IBM SPSS, version 25.0 (IBM Corp., Armonk, NY, USA) and the cumulative incidence curves were drawn using STATA, version 17.0 (StataCorp LLC, College Station, TX, USA).
Reporting summary
Further information on research design is available in the Nature Research Reporting Summary linked to this article.
Supplementary information
Acknowledgements
The authors thank all patients who participated in the original SBII:2pre trial and collaborators at the participating hospitals including the pathology departments and the Department of Oncology, Region Jönköping County, for making this study possible. This study was funded by grants from the Swedish Cancer Society, Anna and Edwin Bergers Foundation, The Berta Kamprad Foundation, The Swedish Cancer and Allergy Fund, Governmental Funding of Clinical Research within the Swedish National Health Service, Percy Falk Foundation, and Futurum – the Academy for Health and Care, Region Jönköping County.
Author contributions
Conception and design: B.N., C.L., L.R., P.-O.B. Data acquisition: C.F., C.L., M.E., L.R., P.-O.B., UK. Formal analysis: C.L., L.R., P.-O.B. Data interpretation: B.N., C.L., L.R., M.F., O.S., P.-O.B. Funding acquisition: C.L., L.R. Project administration: C.F., L.R., P.-O.B. Software: C.L, P.-O.B. Supervision: L.R., C.F., P.-O.B. Validation: C.L., P.-O.B. Writing—original draft: C.L., L.R. Writing—review & editing: all authors. The final version was approved by all authors.
Funding
Open access funding provided by Lund University.
Data availability
The datasets used and/or analysed during the current study could be available from the corresponding author upon reasonable request if this is in line with current laws.
Code availability
No code was generated in the present study. Assignment of PAM50 and ROR score, were provided by the manufacturer’s code (NanoString Breast Cancer 360TM assay on an nCounter® SPRINT Profiler instrument, NanoString Technologies)34.
Competing interests
M.E. received consultancy/advisory role for Pfizer and Novartis. S.C. is a shareholder and employed by Nanostring. U.K. has ownership in proHISTO. All other authors declare no competing interests.
Footnotes
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
The online version contains supplementary material available at 10.1038/s41523-022-00423-z.
References
- 1.Sorlie T, et al. Gene expression patterns of breast carcinomas distinguish tumor subclasses with clinical implications. Proc. Natl Acad. Sci. USA. 2001;98:10869–10874. doi: 10.1073/pnas.191367098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Nielsen TO, et al. A comparison of PAM50 intrinsic subtyping with immunohistochemistry and clinical prognostic factors in tamoxifen-treated estrogen receptor-positive breast cancer. Clin. Cancer Res. 2010;16:5222–5232. doi: 10.1158/1078-0432.CCR-10-1282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Parker JS, et al. Supervised risk predictor of breast cancer based on intrinsic subtypes. J. Clin. Oncol. 2009;27:1160–1167. doi: 10.1200/JCO.2008.18.1370. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Harris LN, et al. Use of biomarkers to guide decisions on adjuvant systemic therapy for women with early-stage invasive breast cancer: American Society of Clinical Oncology Clinical Practice Guideline. J. Clin. Oncol. 2016;34:1134–1150. doi: 10.1200/JCO.2015.65.2289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.National Institute for Health and Care Excellence, Early and locally advanced breast cancer: diagnosis and management (NG101), https://www.nice.org.uk/guidance/ng101 (2018). [PubMed]
- 6.Wallden B, et al. Development and verification of the PAM50-based Prosigna breast cancer gene signature assay. BMC Med Genomics. 2015;8:54. doi: 10.1186/s12920-015-0129-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Gnant M, et al. Predicting distant recurrence in receptor-positive breast cancer patients with limited clinicopathological risk: using the PAM50 Risk of Recurrence score in 1478 postmenopausal patients of the ABCSG-8 trial treated with adjuvant endocrine therapy alone. Ann. Oncol. 2014;25:339–345. doi: 10.1093/annonc/mdt494. [DOI] [PubMed] [Google Scholar]
- 8.Dowsett M, et al. Comparison of PAM50 risk of recurrence score with oncotype DX and IHC4 for predicting risk of distant recurrence after endocrine therapy. J. Clin. Oncol. 2013;31:2783–2790. doi: 10.1200/JCO.2012.46.1558. [DOI] [PubMed] [Google Scholar]
- 9.Sestak I, et al. Comparison of the performance of 6 prognostic signatures for estrogen receptor–positive breast cancer: a secondary analysis of a randomized clinical trial. JAMA Oncol. 2018;4:545–553. doi: 10.1001/jamaoncol.2017.5524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Laenkholm AV, et al. PAM50 risk of recurrence score predicts 10-year distant recurrence in a comprehensive danish cohort of postmenopausal women allocated to 5 years of endocrine therapy for hormone receptor-positive early breast cancer. J. Clin. Oncol. 2018;36:735–740. doi: 10.1200/JCO.2017.74.6586. [DOI] [PubMed] [Google Scholar]
- 11.Sestak I, et al. Prediction of late distant recurrence after 5 years of endocrine treatment: a combined analysis of patients from the austrian breast and colorectal cancer study group 8 and Arimidex, Tamoxifen alone or in combination randomized trials using the PAM50 risk of recurrence score. J. Clin. Oncol. 2014;33:916–922. doi: 10.1200/JCO.2014.55.6894. [DOI] [PubMed] [Google Scholar]
- 12.Chia SK, et al. A 50-gene intrinsic subtype classifier for prognosis and prediction of benefit from adjuvant tamoxifen. Clin. Cancer Res. 2012;18:4465–4472. doi: 10.1158/1078-0432.CCR-12-0286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Jensen M-B, et al. The Prosigna gene expression assay and responsiveness to adjuvant cyclophosphamide-based chemotherapy in premenopausal high-risk patients with breast cancer. Breast Cancer Res. 2018;20:79. doi: 10.1186/s13058-018-1012-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Pu M, et al. Research-based PAM50 signature and long-term breast cancer survival. Breast Cancer Res. Treat. 2020;179:197–206. doi: 10.1007/s10549-019-05446-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Ohnstad HO, et al. Prognostic value of PAM50 and risk of recurrence score in patients with early-stage breast cancer with long-term follow-up. Breast Cancer Res. 2017;19:120. doi: 10.1186/s13058-017-0911-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Lundgren C, et al. Agreement between molecular subtyping and surrogate subtype classification: a contemporary population-based study of ER-positive/HER2-negative primary breast cancer. Breast Cancer Res. Treat. 2019;178:459–467. doi: 10.1007/s10549-019-05378-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Viale G, et al. Immunohistochemical versus molecular (BluePrint and MammaPrint) subtyping of breast carcinoma. Outcome results from the EORTC 10041/BIG 3-04 MINDACT trial. Breast Cancer Res Treat. 2017;167:123–131. doi: 10.1007/s10549-017-4509-9. [DOI] [PubMed] [Google Scholar]
- 18.Holm J, et al. Concordance of immunohistochemistry-based and gene expression-based subtyping in breast. Cancer JNCI Cancer Spectr. 2021;5:pkaa087. doi: 10.1093/jncics/pkaa087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Prat A, et al. Clinical implications of the intrinsic molecular subtypes of breast cancer. Breast. 2015;24:S26–S35. doi: 10.1016/j.breast.2015.07.008. [DOI] [PubMed] [Google Scholar]
- 20.Veracyte, Prosigna® Breast Cancer Prognostic Gene Signature Assay, https://www.prosigna.com (2022).
- 21.Ekholm M, et al. Two years of adjuvant tamoxifen provides a survival benefit compared with no systemic treatment in premenopausal patients with primary breast cancer: long-term follow-up (>25 years) of the phase III SBII:2pre Trial. J. Clin. Oncol. 2016;34:2232–2238. doi: 10.1200/JCO.2015.65.6272. [DOI] [PubMed] [Google Scholar]
- 22.Ekholm M, et al. Effects of adjuvant tamoxifen over three decades on breast cancer-free and distant recurrence-free interval among premenopausal women with oestrogen receptor-positive breast cancer randomised in the Swedish SBII:2pre trial. Eur. J. Cancer. 2019;110:53–61. doi: 10.1016/j.ejca.2018.12.034. [DOI] [PubMed] [Google Scholar]
- 23.Yu NY, et al. Assessment of long-term distant recurrence-free survival associated with Tamoxifen therapy in postmenopausal patients with luminal A or luminal B breast cancer. JAMA Oncol. 2019;5:1304–1309. doi: 10.1001/jamaoncol.2019.1856. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Sparano JA, et al. Adjuvant chemotherapy guided by a 21-gene expression assay in breast cancer. N. Engl. J. Med. 2018;379:111–121. doi: 10.1056/NEJMoa1804710. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kalinsky K, et al. 21-gene assay to inform chemotherapy benefit in node-positive breast cancer. N. Engl. J. Med. 2021;385:2336–2347. doi: 10.1056/NEJMoa2108873. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Nielsen TO, et al. Assessment of Ki67 in breast cancer: updated recommendations from the international Ki67 in Breast Cancer Working Group. J. Natl Cancer Inst. 2021;113:808–819. doi: 10.1093/jnci/djaa201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Stein RC, et al: OPTIMA: a prospective randomized trial to validate the clinical utility and cost-effectiveness of gene expression test-directed chemotherapy decisions in high clinical risk early breast cancer. Cancer Res.79, (suppl; abstr OT1-05-02) (2019).
- 28.Rydén L, et al. Two years of adjuvant tamoxifen in premenopausal patients with breast cancer: a randomised, controlled trial with long-term follow-up. Eur. J. Cancer. 2005;41:256–264. doi: 10.1016/j.ejca.2004.06.030. [DOI] [PubMed] [Google Scholar]
- 29.Lundgren C, et al. Tumour-infiltrating lymphocytes as a prognostic and tamoxifen predictive marker in premenopausal breast cancer: data from a randomised trial with long-term follow-up. Breast Cancer Res. 2020;22:140. doi: 10.1186/s13058-020-01364-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Gourgou-Bourgade, S. et al. Guidelines for time-to-event end point definitions in breast cancer trials: results of the DATECAN initiative (Definition for the Assessment of Time-to-event Endpoints in CANcer trials). Ann. Oncol.26, 873–879 (2015). [DOI] [PubMed]
- 31.Svensk förening för patologi, Bröstpatologi, Bröstcancer, https://www.svfp.se/brostpatologi (2018).
- 32.Jirstrom K, et al. Pathology parameters and adjuvant tamoxifen response in a randomised premenopausal breast cancer trial. J. Clin. Pathol. 2005;58:1135–1142. doi: 10.1136/jcp.2005.027185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Rydén L, et al. Tumor-specific expression of vascular endothelial growth factor receptor 2 but not vascular endothelial growth factor or human epidermal growth factor receptor 2 is associated with impaired response to adjuvant tamoxifen in premenopausal breast cancer. J. Clin. Oncol. 2005;23:4695–4704. doi: 10.1200/JCO.2005.08.126. [DOI] [PubMed] [Google Scholar]
- 34.NanoString Technologies, nCounter® Breast Cancer 360™ Panel, https://www.nanostring.com/products/gene-expression-panels/gene-expression-panels-overview/360-series-panel-collection/ncounter-breast-cancer-360-panel (2022).
- 35.Goldhirsch A, et al. Personalizing the treatment of women with early breast cancer: highlights of the St Gallen International Expert Consensus on the Primary Therapy of Early Breast Cancer 2013. Ann. Oncol. 2013;24:2206–2223. doi: 10.1093/annonc/mdt303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Geskus RB. Cause-specific cumulative incidence estimation and the fine and gray model under both left truncation and right censoring. Biometrics. 2011;67:39–49. doi: 10.1111/j.1541-0420.2010.01420.x. [DOI] [PubMed] [Google Scholar]
- 37.Altman DG, McShane LM, Sauerbrei W, Taube SE. Reporting recommendations for tumor marker prognostic studies (REMARK): explanation and elaboration. PLoS Med. 2012;9:e1001216. doi: 10.1371/journal.pmed.1001216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.McShane LM, et al. Reporting recommendations for tumor MARKer prognostic studies (REMARK) Breast Cancer Res. Treat. 2006;100:229–235. doi: 10.1007/s10549-006-9242-8. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The datasets used and/or analysed during the current study could be available from the corresponding author upon reasonable request if this is in line with current laws.
No code was generated in the present study. Assignment of PAM50 and ROR score, were provided by the manufacturer’s code (NanoString Breast Cancer 360TM assay on an nCounter® SPRINT Profiler instrument, NanoString Technologies)34.