Abstract
There is a potential risk for severe acute respiratory syndrome coronavirus 2 (SARS‐CoV‐2) spread through human contact with seafood and the inanimate materials contaminated by the virus. In this study, we examined the stability of the virus in artificial seawater (ASW) and on the surface of selected materials. SARS‐CoV‐2 (3.75 log10 TCID50) in ASW at 22℃ maintained infectious about 3 days and at 4℃ the virus survived more than 7 days. It should be noticed that viable virus at high titer (5.50 log10 TCID50) may survive more than 20 days in ASW at 4℃ and for 7 days at 22℃. SARS‐CoV‐2 on stainless steel and plastic bag maintained infectious for 3 days, and on nonwoven fabric for 1 day at 22℃. In addition, the virus remained infectious for 9 days on stainless steel and non‐woven fabric, and on plastic bag for 12 days at 4℃. It is important to highlight the role of inanimate material surfaces as a source of infection and the necessity for surface decontamination and disinfection.
Keywords: biostatistics and bioinformatics, epidemiology, SARS coronavirus, survival analysis, virus classification
According to the World Health Organization Commentaries, severe acute respiratory syndrome coronavirus 2 (SARS‐CoV‐2) is mainly transmitted through contact (<1.0 m) as well as droplet, airborne, fomite, fecal‐oral, bloodborne, mother‐to‐child, and animal‐to‐human transmission. Direct and close contact transmission occur by respiratory secretions or droplets (>5–10 μm in diameter). 1 , 2 Indirect contact transmission may be possible by contacting with a contaminated object or surface (fomite transmission). The virus may transmit via virus‐contaminated surfaces and hands. 3 SARS‐CoV‐2 virus has the ability to survive on different surfaces for extended periods, ranging from days up to months depending on environment temperature. 1 In our previous study, we have found that SARS‐CoV‐2 (1.2 × 103 PFU) was able to survive for 3 days in liquid medium or on dry filter paper at 22℃. 4 In addition, SARS‐CoV‐2 (1.2 × 103 PFU) can persist in acidic condition (pH 2.2) for 60 min. 4 SARS‐CoV‐2 have been isolated from a seafood package and a chopping board in the seafood market in China, underscoring risk assessment of SARS‐CoV‐2 transmission in environment. 5 , 6 , 7 Furthermore, WHO pointed out that crowded beaches or swimming pools did pose a risk of spreading SARS‐CoV‐2 through close contact with infected people or contaminated surfaces. 8 Sala‐Comorera et al. found that SARS‐CoV‐2 (4 log10 TCID50/ml) declined 1 log titer in seawater for 1.1 days at 20℃ and for 2.2 days at 4℃. 9 It suggested the risk of SARS‐CoV‐2 in seawater for the disease spreading. However, how long SARS‐CoV‐2 can survive in seawater remains to be investigated. In the present study, we examined the stability of SARS‐CoV‐2 in seawater (artificial seawater, ASW) and on the surface of selected materials (stainless steel, plastic bag, non‐woven fabric, etc), which would provide evidence for COVID‐19 epidemic control.
SARS‐CoV‐2 early cases have been linked to wholesale seafood markets in Wuhan, 10 and SARS‐CoV‐2 nucleic acid have been detected in the imported frozen seafood in Qingdao. 11 It suggests that the virus may survive in seawater and frozen seafood. We, therefore, evaluated the stability of the virus in artificial seawater (The chemical composition of ASW in Table S1 by Dr J Floor Anthoni). 12 The titer of plague‐purified SARS‐CoV‐2 strain nCoV‐SH01 (Genbank MT121215) 13 was determined using 50% cell culture infectious dose assay (TCID50). The viruses in ASW or medium as control were added into each well of 48‐well plates (Corning costar) and incubated in a wet box (~80% humility) at 22℃ or 4℃ for 1, 3, 5, 7, or 14 days. The viral solutions were then transferred to Vero‐E6 cell monolayer (80% confluent) supplemented with maintenance medium containing 5 µg/ml trypsin and incubated in a humidified chamber with 5% CO2 at 37℃. The cytopathic effects (CPE) were checked daily under a microscope for 5 days and viral titers were determined with TCID50. When viruses were kept in medium or ASW at 22℃, the viral titer dropped from the initial 3.75 log10 TCID50 to 1.50 log10 TCID50 (ASW, reduced to 0.56% viability) and 1.75 log10 TCID50 (medium, reduced to 1.00% viability) on Day 3 and no viable virus was detected on day 5 (Table 1), which was consistent to the findings of our previous study that SARS‐CoV‐2 was able to survive for 3 days in liquid medium at 22℃. 4 Then we increased the initial viral titer to 5.50 log10 TCID50, the viral titer decreased to 1.13 log10 TCID50 (ASW, reduced to 0.004% viability) or 1.75 log10 TCID50 (medium, reduced to 0.02% viability) by Day 7 and no viable virus could be detected on Day 14 (Table 1).
Table 1.
Survival of SARS‐CoV‐2 in artificial seawater (ASW) or medium.
| Virus | Temperature (℃) | Treated | Titer (log10 TCID50) of virus kept for days* | ||||
|---|---|---|---|---|---|---|---|
| Day 1 | Day 3 | Day 5 | Day 7 | Day 14 | |||
|
Low titer (3.75 log10 TCID50) |
22 | Medium | 2.00 | 1.75 | UD | UD | UD |
| ASW | 2.25 | 1.50 | UD | UD | UD | ||
| 4 | Medium | 2.75 | 2.25 | 2.00 | 1.50 | UD | |
| ASW | 2.75 | 2.50 | 1.75 | 1.38 | UD | ||
|
High titer (5.50 log10 TCID50) |
22 | Medium | 3.25 | 3.00 | 2.00 | 1.75 | UD |
| ASW | 3.50 | 3.25 | 2.00 | 1.13 | UD | ||
| 4 | Medium | 3.50 | 3.25 | 3.25 | 3.25 | 2.75 | |
| ASW | 3.25 | 3.25 | 3.00 | 3.00 | 3.00 | ||
Abbreviations: SARS‐CoV‐2, severe acute respiratory syndrome coronavirus 2; UD, under detective level.
The nCoV‐SH01 at TCID50 5.50 log10 or 3.75 log10 were treated with medium or artificial seawater for days at 22℃ or 4℃, and the titers were detected by TCID50 assay at Day 1, 3, 5, 7, or 14.
We further investigated virus survival at 4℃ by inoculated with low titer (3.75 log10 TCID50) or high titer (5.50 log10 TCID50) virus. By keeping in ASW (4℃) till Day 14, the viral titer decreased from 5.50 to 3.00 log10 TCID50, and in medium (4℃) dropped to 2.75 log10 TCID50 (Table 1, Figure 1A). When the virus with 3.75 log10 TCID50 kept in ASW (4℃), by Day 7 the viral titer dropped to 1.38 log10 TCID50 (ASW) and 1.50 log10 TCID50 (medium), and no CPE was observed on Day 14 (Table 1, Figure 1B).
Figure 1.
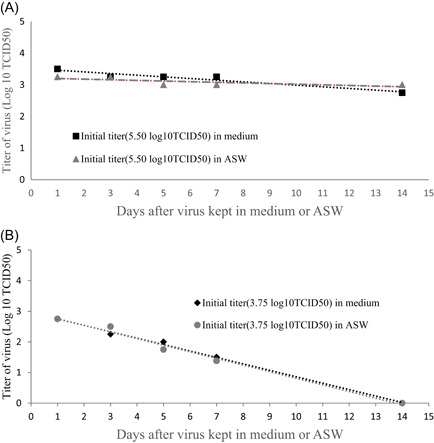
(A) Survival of SARS‐CoV‐2 (high titer) in artificial seawater (ASW) or medium at 4℃. The nCoV‐SH01 at 5.50 log10 TCID50 were treated with artificial seawater or medium for days at 4℃, and the titers were detected by TCID50 assay at Day 1, 3, 5, 7, or 14. (B) Survival of SARS‐CoV‐2 (low titer) in artificial seawater (ASW) or medium at 4℃. The nCoV‐SH01 at 3.75 log10 TCID50 were treated with artificial seawater or medium for days at 4℃, and the titers were detected by TCID50 assay at Day 1, 3, 5, 7, or 14. SARS‐CoV‐2, severe acute respiratory syndrome coronavirus 2
Salinity has previously been demonstrated to have a negative effect on stability of an enveloped RNA virus such as influenza virus. 14 Sala‐Comorera et al. found that infectious SARS‐CoV‐2 titers (4.00 log10 TCID50) remained stable for 24 h in river water at both 4 or 20℃ and in seawater at 4℃. 9 By 20 days at either 4 or 20℃ the virus RNA in both river water and seawater were detected with no decline. 9 In the present study, we found that SARS‐CoV‐2 (3.75 log10 TCID50) in ASW and liquid medium at 22℃ maintained infectious for about 3 days, and at 4℃ the virus survived more than 7 days. When we put the viral survival times in high titer group and low titer group together, it suggested that the virus at a high titer (5.50 log10 TCID50) may survive more than 20 days in ASW at 4℃. At either 4 or 20℃, there was no significant difference of the SARS‐CoV‐2 stability between in ASW and in liquid medium. It suggested that the viability of the virus was not significantly affected by seawater and has similar risk of infection. Therefore, it is necessary to pay attention to protect those in contact with seafood and the environment contaminated by SARS‐COV‐2.
Since SARS‐CoV‐2 nucleic acids have been detected on the surface of agricultural products, package of imported goods or cold chain products, and farmer's market environment, 5 , 7 , 15 , 16 there is a potential risk for virus spread through human contact with these inanimate materials. We, therefore, determined survival of SARS‐CoV‐2 on the surface of selected materials including stainless steel, plastic bag, nonwoven fabric, rubber glove, cardboard, and wood board. 4.0 log10 TCID50 of nCoV‐SH01 in 10 µl was inoculated on a square piece (1 cm × 1 cm) of the material in 12‐well plates (Corning costar) and placed at 4℃ or 22℃ for 1, 3, 5, 7, 9, 12, or 14 days after air dry in a biosafety cabinet. After the incubation, viruses were eluted with DMEM and viral titers were determined by TCID50 assay. Before the testing, we first measured virus elution efficiencies from the different materials, and it showed that 100% (104.00/104.00) from stainless steel, 83.18% (103.92/104.00) from plastic bag, 6.76% (102.83/104.00) from nonwoven fabric and 0.18% (101.25/104.00) from rubber glove respectively. Since the elution efficiencies of cardboard and wood board used were too low (<0.1%), the two materials were excluded in the following study (Table S2). At 22℃, the titers of the viruses eluted from stainless steel and plastic bag were 1.33 log10 TCID50 (initial 4.00 log10 TCID50) and 2.00 log10 TCID50 (initial 3.92 log10 TCID50) on Day 3, respectively and no CPE was observed on Day 5. The titer of the viruses eluted from non‐woven fabric decreased from the initial 2.83–1.08 log10 TCID50 on Day 1 and no CPE was observed on Day 3. No viable virus was detected in the eluent from the rubber glove by Day 1, although after 2‐h incubation virus could be detected (1.25 log10 TCID50) (Table 2, Figure S1A).
Table 2.
Stability of SARS‐CoV‐2 on the surface of materials at 4℃ or room temperature (RT, 22℃).
| Materials | Assay | Survived virus on the surface of the materials for days* | |||||||
|---|---|---|---|---|---|---|---|---|---|
| 2 h** | Day 1 | Day 3 | Day 5 | Day 7 | Day 9 | Day 12 | Day 14 | ||
| Stainless steel at 4℃ | CPE*** | / | ++++ | ++++ | ++++ | ++++ | +++ | ‐ | ‐ |
|
logTCID50 **** percentage (%) |
/ | 3.50 ± 0.25 (31.62) | 3.08 ± 0.29 (12.02) | 2.83 ± 0.14 (6.76) | 2.58 ± 0.14 (3.80) | 1.50 ± 0.37 (0.32) | UD | UD | |
| Stainless steel at RT | CPE | ++++ | ++++ | ++++ | ‐ | ‐ | / | / | ‐ |
|
logTCID50 percentage (%) |
4.00 ± 0.38 | 2.25 ± 0.25 (1.78) | 1.33 ± 0.14 (0.21) | UD | UD | / | / | UD | |
| Plastic bag at 4℃ | CPE | / | ++++ | ++++ | ++++ | ++++ | ++++ | +++ | ‐ |
|
logTCID50 percentage (%) |
/ | 3.25 ± 0.43 (21.38) | 3.58 ± 0.14 (45.71) | 2.83 ± 0.38 (8.13) | 2.25 ± 0.43 (2.14) | 2.51 ± 0.29 (3.89) | 1.75 ± 0.57 (0.68) | UD | |
| Plastic bag at RT | CPE | ++++ | ++++ | ++++ | ‐ | ‐ | / | / | ‐ |
|
logTCID50 percentage (%) |
3.92 ± 0.38 | 2.50 ± 0.50 (3.80) | 2.00 ± 0.50 (1.20) | UD | UD | / | / | UD | |
| Nonwoven fabric at 4℃ | CPE | / | ++++ | ++++ | ++++ | ++ | ++ | ‐ | ‐ |
|
logTCID50 percentage (%) |
/ | 3.08 ± 0.58 (177.82) | 2.42 ± 0.58 (38.90) | 2.08 ± 0.58 (17.78) | 0.58 ± 1.01 (0.56) | 0.52 ± 0.47 (0.50) | UD | UD | |
| Nonwoven fabric at RT | CPE | ++++ | ++++ | ‐ | ‐ | ‐ | / | / | ‐ |
|
logTCID50 percentage (%) |
2.83 ± 1.13 | 1.08 ± 0.95 (1.77) | UD | UD | UD | / | / | UD | |
| Rubber gloves at 4℃ | CPE | / | ++ | ‐ | / | / | / | / | / |
|
logTCID50 percentage (%) |
/ | 0.33 ± 0.47 (12.02) | UD | / | / | / | / | / | |
| Rubber gloves at RT | CPE | ++++ | ‐ | ‐ | / | / | / | / | / |
|
logTCID50 percentage (%) |
1.25 ± 2.16 | UD | UD | / | / | / | / | / | |
Abbreviations: CPE, cytopathic effects; SARS‐CoV‐2, severe acute respiratory syndrome coronavirus 2.
The nCoV‐SH01 (4.00 log10 TCID50) in 10 µl DMEM was added on the surface of each material and placed on the biosafety cabinet to dry naturally for 30 min at 22℃. The eluting virus was detected by CPE and TCID50 at Day 1, 3, 5, 7, 9, 12, or 14. The experiments were carried out in triplicate wells for each dilution.
The virus was naturally dried at room temperature for 2 h. Then the eluted virus activity and titer were measured to calculate the elution efficiency (stainless steel: 100%, plastic bag: 83.18%, nonwoven fabric: 6.76%, rubber gloves: 0.18%).
CPE of Vero E6 cells was checked under a microscope at 48 h post infected. Degree of CPE, “++++”, >75% of cells; “+++”, 50%–75%; “++”, 25%–50%; “+”, 0–25%; “±”, not clear‐cut; “‐”, no CPE. The cytopathic effects were observed under a microscope daily for 5 days.
The titer of virus were determined for log TCID50 presented as mean ± SD, and percentage (%): viral survival rate in percentages = 10(viral titer at days)/10(viral titer at 2 h). UD: under detective level./: not done.
At 4℃, the eluted viruses from stainless steel and non‐woven fabric dropped to 1.50 log10 TCID50 and 0.52 log10 TCID50 by Day 9, respectively, and no CPE was observed on Day 12, while that from plastic bag dropped to 1.75 log10 TCID50 by Day 12 and no CPE was observed on Day 14. Loss of viral infectivity was confirmed by passing the culture supernatant blindly for three generations. Thus, the virus can survive much longer on stainless steel, nonwoven fabric, and plastic bag (>9 days) at 4℃ (Table 2, Figure S1B).
Chin et al. have reported that SARS‐CoV‐2 (6.8 log10 TCID50) remained viable after 14 days at 4℃ in the transport medium with a reduction of a 0.6 log10 TCID50 in viral titer by 7 days at 22℃, and 1 day at 37℃. They also found that the virus is extremely stable (with no significant decrease in titer) under a wide range of pH values (pH 3–10) for 60 min at room temperature. 17 Doremalen et al. compared SARS‐CoV‐2 and SARS‐CoV‐1, and found that both viruses were more stable on surfaces of plastic and stainless steel (3 days) than on copper and cardboard (1 day). 18 Many factors may affect the viral survival on the surface of various materials, including virus inactivation by materials and virus adsorption by porous materials. Hirose et al. found that SARS‐COV‐2 could survive 59.8 h on the surface of plain paper, and paper surface treatments that enable the rapid evaporation of liquid might reduce the stability of virus. 19 Additionally, Hirose et al. found that the survival times of SARS‐CoV‐2 VOCs (Variants of Concern) on the plastic surface were much longer than on the human skin, and the Omicron variant (5.0 × 104 TCID50) survived for 193.5 h on the plastic surface and only 21.1 h on the human skin. 20 , 21 The importance of surface‐mediated transmission was demonstrated by Rawlinson et al. who used a DNA oligonucleotide surrogate for contaminated bodily fluid to determine how SARS‐CoV‐2 would spread within a clinical surface environment. It showed that within 10 h, the surrogate moved from the isolation room and transferred to 41% of all surfaces sampled. 22 We found that at 22℃ SARS‐CoV‐2 (4.0 log10 TCID50) on stainless steel and plastic bag maintained infectious for 3 days, and on non‐woven fabric for 1 day. In addition, at 4℃ the virus was more stable with remaining infectious for 9 days on stainless steel and non‐woven fabric, and on plastic bag for 12 days. It suggests that virus‐contaminated items and packaging surfaces pose a risk of infection by close contacts.
Taken together, the data suggest that contaminated viruses may survive on the surfaces of the materials in cold‐chain shipping (e.g., stainless steel, plastic, and nonwoven fabric) for a long time. It is important to highlight the role of inanimate material surfaces as a source of infection and the necessity for surface decontamination and disinfection. In addition, more stringent personal protection, as well as hand hygiene, should be implemented on the personnel engaged in the transportation and handling of cold‐chain shipped food.
AUTHOR CONTRIBUTIONS
Zhi‐ping Sun, Si‐yu Yang, and Xia Cai performed the viral experiment in BSL‐3 lab, analyzed the data, and participated in writing the paper. Wen‐dong Han, Gao‐wei Hu, Yun Qian, Yu‐yan Wang, and Rong Zhang participated in experiments in BSL‐3 lab. Di Qu and You‐hua Xie designed the experiments, planned the approach, and wrote and edited the paper.
CONFLICTS OF INTEREST
The authors declare no conflicts of interest.
Supporting information
Supplementary information.
Supplementary information.
Supplementary information.
ACKNOWLEDGMENTS
This study was supported by NSTMP (2018ZX10734401, 2018ZX10301208, and 2019ZX09721001), Development Programs for COVID‐19 of Shanghai Science and Technology Commission (Grant No. 20431900401), and Project of Novel Coronavirus of Fudan University.
Sun Z‐P, Yang S‐Y, Cai X, et al. Survival of SARS‐CoV‐2 in artificial seawater and on the surface of inanimate materials. J Med Virol. 2022;94:3982‐3987. 10.1002/jmv.27807
Zhi‐Ping Sun, Si‐Yu Yang, and Xia Cai are co‐authors.
Contributor Information
You‐Hua Xie, Email: yhxie@fudan.edu.cn.
Di Qu, Email: dqu@shmu.edu.cn.
DATA AVAILABILITY STATEMENT
The data that support the findings of this study are available from the corresponding author upon reasonable request.
REFERENCES
- 1. Organization. WH . Transmission of SARS‐CoV‐2: implications for infection prevention precautions. (https://www.who.int/news-room/commentaries/detail/transmission-of-sars-cov-2-implications-for-infection-prevention-precautions).
- 2. Marques M, Domingo JL. Contamination of inert surfaces by SARS‐CoV‐2: persistence, stability and infectivity. A review. Environ Res. 2021;193:110559. 10.1016/j.envres.2020.110559 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Bueckert M, Gupta R, Gupta A, Garg M, Mazumder A. Infectivity of SARS‐CoV‐2 and other coronaviruses on dry surfaces: potential for indirect transmission. Materials. 2020;13:22. 10.3390/ma13225211 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Sun Z, Cai X, Gu C, et al. Survival of SARS‐COV‐2 under liquid medium, dry filter paper and acidic conditions. Cell Discov. 2020;6:57. 10.1038/s41421-020-00191-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Liu P, Yang M, Zhao X, et al. Cold‐chain transportation in the frozen food industry may have caused a recurrence of COVID‐19 cases in destination: Successful isolation of SARS‐CoV‐2 virus from the imported frozen cod package surface. Biosaf Health. 2020;2(4):199‐201. 10.1016/j.bsheal.2020.11.003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Daily C . Experts see similarity in Beijing, Dalian outbreaks. 2020. https://www.chinadaily.com.cn/a/202007/30/WS5f228da3a31083481725d32c.html
- 7. Times G . Beijing supermarkets stop selling salmon after wholesalers test positive for coronavirus. 2020a. https://www.globaltimes.cn/content/1191462.shtml
- 8. Organization . WH. Coronavirus disease (COVID‐19): Environmental Surveillance. (https://www.who.int/news-room/questions-and-answers/item/environmental-surveillance).
- 9. Sala‐Comorera L, Reynolds LJ, Martin NA, O'Sullivan JJ, Meijer WG, Fletcher NF. Decay of infectious SARS‐CoV‐2 and surrogates in aquatic environments. Water Res. 2021;201:117090. 10.1016/j.watres.2021.117090 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Zhu N, Zhang D, Wang W, et al. A novel coronavirus from patients with pneumonia in China, 2019. N Engl J Med. 2020;382(8):727‐733. 10.1056/NEJMoa2001017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Jia J, Yuan Q, Hui JW, et al. Investigation of contamination of SARS‐CoV‐2 in imported frozen seafood from a foreign cargo ship and risk factors for infection in stevedores in Qingdao. Zhonghua Liu Xing Bing Xue Za Zhi. 2021;42(8):1360‐1364. 10.3760/cma.j.cn112338-20210209-00107 [DOI] [PubMed] [Google Scholar]
- 12. Anthoni JF. The chemical composition of seawater. http://www.seafriends.org.nz/oceano/seawater.htm
- 13. Zhang Rong YZ. Isolation of a 2019 novel coronavirus strain from a coronavirus disease 19 patient in Shanghai. J Microbes Infect. 2020;15:15‐20. [Google Scholar]
- 14. Dublineau A, Batejat C, Pinon A, Burguiere AM, Leclercq I, Manuguerra JC. Persistence of the 2009 pandemic influenza A (H1N1) virus in water and on non‐porous surface. PLoS One. 2011;6(11):e28043. 10.1371/journal.pone.0028043 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Times G . COVID‐19 outbreaks in Wuhan, Beijing and Dalian share certain similarities: China's top epidemiologist. 2020b. https://www.globaltimes.cn/content/1196130.shtml
- 16. Han J, Zhang X, He S, Jia P. Can the coronavirus disease be transmitted from food? A review of evidence, risks, policies and knowledge gaps. Environ Chem Lett. 2020;19:1‐12. 10.1007/s10311-020-01101-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Chin AWH, Chu JTS, Perera MRA, et al. Stability of SARS‐CoV‐2 in different environmental conditions. Lancet Microbe. 2020;1(1):e10. 10.1016/S2666-5247(20)30003-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. van Doremalen N, Bushmaker T, Morris DH, et al. Aerosol and surface stability of SARS‐CoV‐2 as compared with SARS‐CoV‐1. N Engl J Med. 2020;382(16):1564‐1567. 10.1056/NEJMc2004973 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Hirose R, Miyazaki H, Bandou R, et al. Stability of SARS‐CoV‐2 and influenza virus varies across different paper types. J Infect Chemother. 2022;28(2):252‐256. 10.1016/j.jiac.2021.11.006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Hirose R, Ikegaya H, Naito Y, et al. Survival of severe acute respiratory syndrome coronavirus 2 (SARS‐CoV‐2) and influenza virus on human skin: importance of hand hygiene in coronavirus disease 2019 (COVID‐19). Clin Infect Dis. 2021;73(11):e4329‐e4335. 10.1093/cid/ciaa1517 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Ryohei Hirose YI, Ikegaya H, Miyazaki H, et al. Differences in environmental stability among SARS‐CoV‐2 variants of concern: omicron has higher stability. bioRxiv . 2022. 2022.01.18.476607 10.1101/2022.01.18.476607 [DOI] [PMC free article] [PubMed]
- 22. Rawlinson S, Ciric L, Cloutman‐Green E. COVID‐19 pandemic ‐ let's not forget surfaces. J Hosp Infect. 2020;105(4):790‐791. 10.1016/j.jhin.2020.05.022 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary information.
Supplementary information.
Supplementary information.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.


