Abstract
Children are extremely liable to indoor air pollutants as their physiology and a few metabolic pathways are different from those of adults. The present cross-sectional study aimed to assess exposure of children living with parents who use hookah tobacco smoke to polycyclic aromatic hydrocarbons (PAHs) using a biomonitoring approach. The study was conducted on 25 children (7–13 years of age) exposed to hookah smoke at home and 25 unexposed age-matched children. Urinary levels of five metabolites of PAHs were quantified via headspace gas chromatography-mass spectrometry (GC–MS). Urinary malondialdehyde (MDA) was measured, as well. Information regarding the sociodemographic and lifestyle conditions was collected through interviews using managed questionnaires. The urinary 1-OH-NaP and 9-OH-Phe concentrations were respectively 1.7- and 4.6-folds higher in the case samples compared to the control group (p < 0.05). In addition, urinary MDA levels were 1.4 times higher in the exposed children than in the unexposed group, but the difference was not statistically significant (p > 0.05). Increasing the consumption of grilled and meat food in the diet increased the participants’ urinary 2-OH-Flu and 1-OH-Pyr levels, respectively. Moreover, sleeping in the living room instead of the bedroom at night was a significant predictor of high 1-OH-NaP and 2-OH-NaP concentrations in the children’s urine. Overall, the findings confirmed that children living in their homes with hookah-smoking parents were significantly exposed to naphthalene and phenanthrene. Hence, implementing protective measures is critical to reduce the exposure of this group of children.
Supplementary Information
The online version contains supplementary material available at 10.1007/s11356-022-20589-0.
Keywords: Risk assessment, Polycyclic aromatic hydrocarbons, Oxidative stress, Hookah smoke, Children
Introduction
There are many concerns about exposure to indoor air pollution, as the concentrations of indoor air pollutants are often higher compared to outdoors (Moradi et al. 2019, Dehghani et al. 2022). Furthermore, people spend approximately 80–90% of their time indoors (Tabatabaei et al. 2021). Therefore, indoor air quality is important for human health (Bernstein et al. 2008, Miri et al. 2018). Environmental tobacco smoke (ETS) is a major contributor to indoor air pollution (Mueller et al. 2011). In recent years, the use of tobacco products via waterpipe (hookah) has globally risen among adults and teenagers, especially in East Mediterranean countries (Maziak et al. 2017, Leavens et al. 2018). The important factors contributing to the global spread of waterpipe tobacco smoke (WTS) are the increasing pleasant flavors of tobacco, the wrong opinion that waterpipe is healthier than cigarettes, acceptance by communities, and lower costs (El-Zaatari et al. 2015, Kumar et al. 2015, Leavens et al. 2018). Based on the previous studies, mainstream and side stream waterpipe smoke has been found to contain seriously high quantities of carcinogenic substances such as polycyclic aromatic hydrocarbons (PAHs) (Sepetdjian et al. 2010, Nguyen et al. 2013). Thus, houses are becoming the predominant location for the exposure of children and adults to PAH compounds through hookah smoke (Health and Services 2006).
PAHs are compounds that are mostly colorless, white, or pale yellow and are formed during both biological processes and incomplete combustion. Although humans can be exposed to PAH compounds in a variety of routes such as inhalation of polluted air and dermal absorption, food and tobacco smoking are considered the main pathways of exposure (Abdel-Shafy and Mansour 2016).
Benzo[a]pyrene (BaP), as a part of the high molecular weight of PAH compounds, tends to accumulate in the adipose tissue and only a small part of it is metabolized in the body. However, most of the fluorine or phenanthrene is metabolized in the body as a low molecular weight of PAHs (Dejmek et al. 2000, Abdel-Shafy and Mansour 2016, Urbancova et al. 2020). Metabolism of PAHs occurs in the body by cytochrome P450 enzymes. This process occurs in two phases. In the first phase, mainly OH-PAHs are formed. In the second phase, these compounds with glucuronic acid or sulfate produce compounds that are excreted in the urine due to their high solubility in water (Tombolini et al. 2018, Urbancova et al. 2020).
PAHs can be harmful to human health during metabolic process. In the first phase, for example, formed reactive species can increase cancer in humans by reacting with DNA and proteins in the human body. PAHs can also have numerous negative effects on children’s health by increasing respiratory problems, bronchial asthma, and risk of cancer (Freire et al. 2009, Urbancova et al. 2020). In addition, PAH compounds share a similar mechanism of carcinogenic action in both experimental animals and humans (Watson et al. 1988; Shamsedini et al, 2022a). BaP and another compound of high concern, i.e., chrysene (CHRY) have been listed as carcinogenic to humans (group 1) and possibly carcinogenic to humans (Group 2B), respectively by the International Agency for Research on Cancer (IARC) (Ke et al. 2016).
Malondialdehyde (MDA) has been extensively used as a biomarker for lipid peroxidation, because it is a product of lipid oxidative damage. Previous research has shown that MDA is associated with various diseases and exposure to PAHs suggesting a potential relationship between PAHs exposure and health effects (Lin et al. 2016).
Up to now, published papers have mainly focused on the analysis of PAHs in urine samples to assess PAHs exposure in a wide range of populations including highly exposed people such as smokers (Freire et al. 2009) and kitchen workers (Campo et al. 2010). Additionally, recent studies have been done on children exposed to these compounds in areas with polluted outdoor air (Shahsavani et al. 2017, Murawski et al. 2020, Salami et al. 2021). However, there is a paucity of studies assessing PAHs exposure among children exposed to hookah smoke at home. Therefore, the present study aimed to evaluate the potential of urinary metabolized PAHs to be used as the biomarkers of environmental exposure to PAHs and oxidative stress by measuring MDA concentrations in the children exposed to hookah smoke daily in southwestern Iran.
Materials and methods
Study characteristics and participant eligibility
This cross-sectional study was performed in Khesht, a city in Kazerun, Fars Province, Southwest of Iran (29°33′49″N 51°20′13″E), with over 11 thousand residents. One of the important reasons for choosing this area was the high rate of hookah smoking (Tabatabaei et al. 2021). Another reason was the similarity of houses structure in the area, which largely controls the confounders. On the other hand, this area is a town with a climate and structure similar to the village, which is far from the highways, traffic, gas stations, and industries that are the sources of these compounds (Keyte et al. 2016).
A total of 25 healthy children aging 7–13 years with hookah smoking parents were recruited as the exposed group. At least one parent of these children smoked hookah at home on a daily basis. In addition, 25 age-matched children with no-smoking parents were enrolled as the unexposed control group.
The inclusion criteria for the recruited subjects in this study included the following: (1) they lived in the Khesht city; (2) children ranged in age from 7 to 13 years; (3) in the case group, at least one parent used hookah daily at home and for the control group, no one at home used any kind of tobacco (hookah, cigarettes, etc.); (4) for 3 days before sampling did not take any medication such as acetaminophen, adult cold pills, and dietary supplements (vitamins, minerals, iron, etc.); and (5) participants in both groups did not have any disease (heart, had no kidneys, etc.). Among the participants in the case group, whose parents smoked cigarettes in addition to hookah were excluded from the study.
This study was approved by the Ethics Committee of Shiraz University of Medical Sciences (IR.SUMS.REC.1399.955). Written consent forms for the children’s participation in the project were obtained from all the parents.
Participant questionnaires and urine sample preparation
Sampling was done on mid-June 2020. In doing so, the researcher referred to Khesht city from Shiraz University of Medical Sciences with a letter of introduction and a code of ethics. The study conditions and the criteria for entering and leaving the study were explained to the parents of subjects by a trained researcher. One day was determined for urine sampling and on the sampling day; a checklist and informed consent form were completed by the parents. The children’s urine samples were taken once from 8 to 10 am and were collected in 100-ml polypropylene vials. In total, 50 urine samples were collected including 25 samples from the exposed children and 25 from the unexposed group. Immediately after collection, a portion of the samples was separated for measuring the urinary creatinine and malondialdehyde. Eventually, urine samples were stored in a cool box for tests related to PAH urinary metabolites in the laboratory of Amin monitoring scholars and to measure urinary creatinine and malondialdehyde in the laboratory of the Faculty of Nutrition, University of Science Shiraz Medical, was transferred. Information about lifestyle, living conditions, children’s indoor and outdoor activities, the type of food consumed during the week (seafood, grilled, fruits and vegetables, and meat), frequency of cooking at home, age of the building, use of hood in the kitchen, etc. were collected using face-to-face interviews and a comprehensive questionnaire.
The urinary creatinine levels were analyzed through Jaffè reaction (Butler 1975). Briefly, 3 ml of urine was mixed with 5 ml sodium acetate buffer (pH = 5) in the laboratory. Then, 5 µl β-glucuronidase/arylsulfatase enzyme was added. The fully blended mixture was incubated at 37 °C for 5 h (Liu et al. 2015). The rest of the samples were coded and stored at − 20 °C in the laboratory for further analysis.
Quantification of OH-PAHs in the urine samples
Solid-phase microextraction (SPME) was performed by immersing an SPME fiber (coated with an 85 µm polyacrylate layer) into the liquid sample (kept at 35 °C) for 45 min while the sample was magnetically stirred. The SPME fiber was then analyzed by being transferred to a hot gas chromatograph injection port and allowing the adsorbed chemicals to desorb at 270 °C for 3 min (Liu et al. 2015). Afterwards, the concentrations of five PAH metabolites including 1-hydroxynathalene (1-OH-NaP), 2-hydroxynathalene (2-OH-NaP), 2-hydroxyfluorene (2-OH-Flu), 1-hydroxypyrene (1-OHPyr), and 9-hydroxyphenanthrene (9-OH-Phe) were determined using gas chromatography (GC, Agilent 7890 N, Agilent Co.) coupled to a mass spectrometer (MS, Agilent 5975C, Agilent Co.). A 30 m × 0.25 µm × 0.5 µm polydimethylsiloxane capillary column (HP-5 MS (5%phenl)-95%) was used for the separation process. The MSD device software was ChemStation version E.02.01.1177 and the carrier gas was helium with the purity level of 99.99%. The initial GC oven temperature was 80 °C, which was held for 2 min, increased to 285 °C (at a rate of 8 °C/min), and held for 2 min. The splitless GC injector was kept at 290 °C. The source and quadruple temperatures were kept at 230 and 150 °C, respectively. The MS was operated in the electron impact ionization mode, and the ionization energy was 70 eV.
Determination of the oxidative stress marker (MDA) in the urine samples
To prepare the urine samples, the samples were centrifuged at 4 °C for 10 min. In summary, standards and reagents were prepared according to the kit instructions. All the samples were shaken to homogenize. After that, 100 µl standards/samples and 100 μl of R4 were added to all the tubes, which were swirled to mix and were incubated for 5 min. In the next step, 200 µl of the ready chromogenic solution was added to all the tubes. The tubes were then placed on vigorously boiling water for 1 h. After 1 h, the tubes were immediately placed on an ice bath for 10 min to stop the reactions. The tubes were then centrifuged at 10,000 × g at 4 °C for 10 min. Afterwards, 200 µl of the supernatant was transferred with a pipette to the associated well in the microplate. Eventually, fluorescence was read at Ex/Em 530/550 nm. Besides, MDA was corrected with creatinine.
Quality control of urine samples and method performance
To determine the efficiency of recovery, 5 μl of the internal standard was added to each sample. After extracting all the samples, the recovery rate was obtained, which ranged from 73 to 103%. Urine samples of the control group who had no tobacco exposure, did not consume foods with high levels of PAHs, and lived far away from emission sources were collected as the blank samples and were used to calculate the limit of detection (LOD) and limit of quantification (LOQ). These measures were computed as the concentration equal to three and ten times the noise of the quantifier ion. The LOD and LOQ values for all the target compounds are listed in Table S1.
Risk assessment
In this study, the risk of exposure to PAHs in children was assessed using the levels of these chemicals in the urine. The risk assessment in the probabilistic approach was also estimated by Monte Carlo simulation.
Using Eq. 1, the estimated daily intake (EDI) of naphthalene, fluorine, phenanthrene, and pyrene was calculated by combining the levels of the urinary metabolites (OH-NaPs, 2-OH-Flu, 9-OH-Phe, and 1-OHPyr) in the children (Shahsavani et al. 2022 and Shamsedini et al. 2022a, b). The parameters used to calculate the non-carcinogenic risk are given in Table S2.
| 1 |
The noncancerous risk of exposure to PAHs was assessed using the risk factor hazard quotient (HQ), which was calculated using Eq. 2.
| 2 |
To assess the risk of multiple compounds attacking a target organ in the body, another index called the risk index (HI) was used by summing all the HQ values. The HI equation has been shown below.
HI > 1 indicates a potential for a negative health impact on the study population due to exposure to PAHs, but HI < 1 means that exposure to PAHs in these conditions does not pose a significant risk to human health.
Uncertainty is mainly due to the lack of awareness of changes in model parameters. To consider the uncertainties in risk assessment, the Crystal Ball software (11.1.2.3) was used to perform a potential non-carcinogenic risk assessment based on the Monte Carlo simulation.
Statistical analysis
Statistical analysis was performed using R software (version 4.0.2). If the concentration of urinary OH-PAHs was less than the LOQ, the detection frequencies of less than 50% and more than 50% were calculated as 1.4 and 1.2, respectively. Spearman’s correlation was utilized to determine the correlation between two variables. In addition, Mann–Whitney U test was used to compare the differences between the two independent groups. Moreover, multivariate linear regression analysis was employed to investigate the association between urinary OH-PAHs and the independent variables affecting the urinary concentrations. The statistical significance level was set at p < 0.05.
Results
Characteristics of the study participants
The selected characteristics of the studied participants based on the questionnaires are summarized in Table 1. The results revealed no significant difference between the exposed and unexposed groups regarding these parameters (p > 0.05). Out of the 50 children, 58% were boys and 42% were girls. The mean age of the participants was 10.3 ± 1.8 years, ranging from 7 to 13 years. Anomalous Body Mass Index (BMI, greater than 30 kg⋅m2) was found in one child (2%). In contrast, 46% of the children had a BMI lower than 17 kg⋅m2. Since the study area was far from the main roads, industries, factories, bus terminals, and restaurants, the study population was far from the sources of PAHs emissions. The common mode of transportation was traveling by private cars. Besides, none of the participants used busses and subways to travel. Most of the exposed children (72%) had at least one hookah smoker parent, and 28% of them had two smoker parents. In addition, regular tobacco was the most common type of tobacco used in the exposed group.
Table 1.
The selected characteristics of the studied population
| Characteristics | Exposed group | Non-exposed group |
|---|---|---|
| No | 25 | 25 |
| Geo-mean ± SD | Geo-mean ± SD | |
| Age (years) | 10.79 ± 1.2 | 9.49 ± 1.2 |
| Height (cm) | 142.4 ± 1.2 | 134.4 ± 1.06 |
| Weight (kg) | 35.52 ± 1.28 | 30.94 ± 1.26 |
| BMI (kg/m2) | 17.51 ± 1.24 | 17.14 ± 1.16 |
| Gender (%) | ||
|
Girl Boy |
36 64 |
48 52 |
| Eating fast food (%) | ||
|
Once per week 2–3 times per week More than 3 times |
76 24 - |
64 24 12 |
| Eating grilled food (%) | ||
|
Once per week 2–3 times per week More than 3 times |
68 12 20 |
76 24 - |
| Eating meat (%) | ||
|
Once per week 2–3 times per week More than 3 times |
28 40 32 |
24 16 60 |
| Eating fruits and vegetables (%) | ||
|
Once per day 2–3 times per day More than 3 times a day |
40 60 - |
44 16 40 |
| Eating seafood (%) | ||
|
Once per week 2–3 times per week More than 3 times |
12 72 16 |
14 66 20 |
| Frequency of cooking at home (%) | ||
|
Once per day 2–3 times per day More than 3 times per day |
- 88 12 |
- 84 16 |
| Age of the house (%) | ||
|
< 5 5–10 10–15 > 15 |
20 56 8 16 |
32 56 8 4 |
| Baby’s sleeping place (%) | ||
|
Bedroom Living room |
76 24 |
76 24 |
| Home floor (%) | ||
|
1st 1 < |
80 20 |
76 24 |
| Using kitchen hoods (%) | ||
|
Yes No |
64 36 |
68 32 |
| Number of hookah smokers in the house (%) | ||
|
0 1 2 3 |
- 72 28 - |
100 - - - |
| Number of times of hookah smoking (%) | ||
|
None Once per day 2–3 times per day More than 3 times a day |
_ 12 72 16 |
100 - - - |
| Type of tobacco (%) | ||
|
None Fruity Regular |
- 20 80 |
100 - - |
OH-PAHs urinary concentrations profile in the studied groups
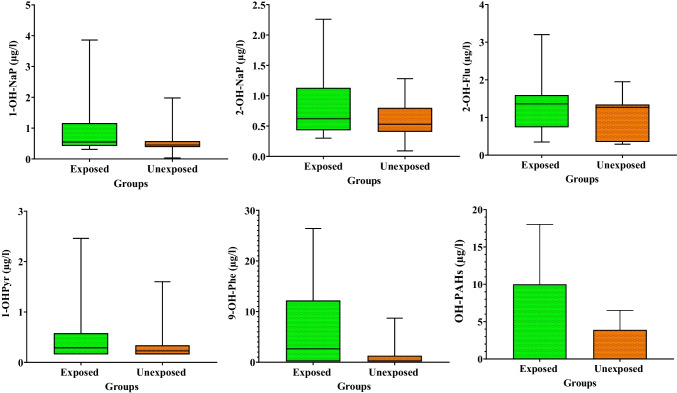
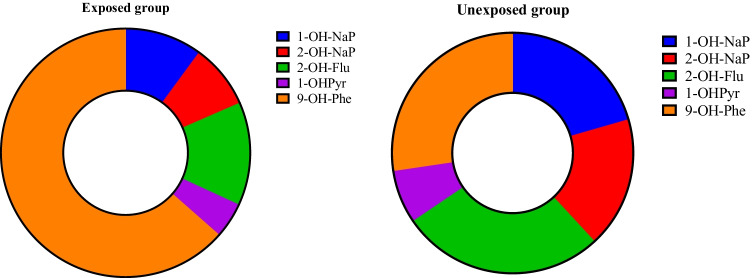
Descriptive statistics of the OH-PAHs urinary concentrations in the target population are summarized in Table 2 and Fig. 1. The results revealed a significant difference between the two groups regarding the measured concentrations of 1-OH-NaP, 9-OH-Phe, and ∑OH-PAHs (p < 0.05). Accordingly, the urinary levels of ∑OH-PAHs were 2.53 times higher in the exposed children than in the unexposed children. The compositions of OH-PAHs in the urine samples are shown in Fig. 2. As the figure depicts, the highest and lowest levels were related to 9-hydroxyphenanthrene and 1-hydroxypyrene, respectively in both exposed and unexposed groups.
Table 2.
Statistical analysis of the urinary OH-PAHs in the studied groups (µg/l)
| Exposure type | Exposed group | Unexposed group | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Statistical analysis | Mean ± SD (min–max) |
Geo-mean | SE | Median | Mean ± SD (min–max) |
Geo-mean | SE | Median | Comparison |
| 1-OH-NaP |
1.01 ± 0.96 (0.31–3.86) |
0.73 | 0.19 | 0.55 |
0.57 ± 0.38 (0.25–1.98) |
0.49 | 0.07 | 0.45 | P = 0.04 |
| 2-OH-NaP | 0.83 ± 0.55 (0.3–2.26) | 0.69 | 0.11 | 0.62 |
0.61 ± 0.27 (0.15–1.28) |
0.54 | 0.05 | 0.53 | P = 0.22 |
| 2-OH-Flu |
1.34 ± 0.76 (0.35–3.2) |
1.2 | 0.15 | 1.36 |
1.03 ± 0.51 (0.29–1.95) |
0.86 | 0.1 | 1.27 | P = 0.13 |
| 1-OH-Pyr |
0.46 ± 0.5 (0.16–2.46) |
0.33 | 0.1 | 0.29 |
0.34 ± 0.37 (0.16–1.6) |
0.26 | 0.07 | 0.23 | P = 0.23 |
| 9-OH-Phe |
6.34 ± 8.14 (0.17–26.41) |
1.67 | 1.62 | 2.65 |
1.36 ± 2.35 (0.17–8.7) |
0.47 | 0.47 | 0.27 | P = 0.02 |
| OH-PAHs |
9.94 ± 8.1 (2.18–29.62) |
7.53 | 1.6 | 6.1 |
3.92 ± 2.6 (1.2–10.69) |
3.23 | 0.52 | 3.1 | P = 0.00 |
Fig. 1.
The urinary levels of OH-PAHs in the study groups
Fig. 2.
Composition profiles of OH-PAHs in the study groups’ urine samples
MDA urinary concentrations profile in the study participants
The results of the MDA urinary levels in the study groups are presented in Table 3 and Fig. S2. The results revealed significant correlations between the MDA level and some parameters including weight, height, and BMI. Additionally, the MDA urinary level was 1.42 times higher in the exposed samples than in the unexposed samples, but the difference was not statistically significant (p > 0.05).
Table 3.
The urinary levels of MDA in the study groups
| Exposed group | Unexposed group | ||||||||
| Statistical analysis |
Mean ± SD (min–max) |
Geo-mean | SE | Median |
Mean ± SD (min–max) |
Geo-mean | SE | Median | Comparison |
| MDA (µg/g creatinine) |
6.82 ± 4.24 (1.3–17.75) |
5.61 | 0.84 | 6.52 |
5.05 ± 3.1 (0.43–10.17) |
3.94 | 0.6 | 4.06 | P = 0.16 |
| Correlation coefficient | |||||||||
| Weight | Height | BMI | |||||||
| MDA (μg/g creatinine) | 0.291 | 0.282 | 0.279 | ||||||
| P-value | 0.04 | 0.04 | 0.04 | ||||||
Correlations and predictors of exposure to PAHs
Spearman’s rank correlation coefficient was used to determine the relationship between the urinary concentrations of PAHs and the participants’ age. Based on the results, the concentrations of 2-OH-NaP and 1-OH-Pyr were significantly and positively associated with the children’s age (Fig. S3). The strongest correlation was observed for 1-OH-Pyr (CC = 0.423, p = 0.002). As shown in Fig. S4, the results of Mann–Whitney U test indicated that the mean concentrations of 1-OH-NaP and 2-OH-NaP were significantly higher in boys than in girls.
Linear regression
Regression models were designed to estimate the association between the urinary concentrations of OH-PAHs and independent variables including sleeping place, BMI, frequent use of meat, grilled food, fruits, and vegetables, creatinine and MDA concentrations, age of the house, number of cooking times during the day, and use of kitchen hood. The results of multiple regression analysis (β coefficient, p-value) for the association between the urinary concentrations of the target compounds and potentially confounding variables are presented in Table 4. The results showed a significant relationship between the children’s sleeping place and the levels of 1-hydroxy naphthalene and 2-hydroxy naphthalene in the participants’ urine samples (p < 0.05) (Fig. S5). Grilled food was also effective in the urinary concentration of 2-hydroxy fluorene. Accordingly, the concentration of this metabolite was higher among the participants who ate more grilled food during the week compared to those who ate grilled food to a lesser extent. The concentration of 1-OHPyr was also higher in the children who ate more meat compared to those who ate less meat (Fig. S6). The results of the linear regression on the assessment of the association between MDA level and OH-PAHs concentrations, BMI, gender, and some types of food are presented in Table 5. Based on the findings, MDA was associated with higher concentrations of 2-hydroxy naphthalene (β = 3.01, p = 0.01).
Table 4.
Multivariate linear regression analysis of the urinary OH-PAHs (µg/l) with the factors affecting OH-PAHs in the study participants (β coefficient (p-value))
| Factors | 1-OH-NaP | 2-OH-NaP | 2-OH-Flu | 1-OHP-yr | 9-OH-Phe |
|---|---|---|---|---|---|
| Grilled food | 0.02 (0.6) | 0.042 (0.54) | 0.23 (0.00)* | 0.015 (0.13) | 0.31 (0.53) |
| Eating meat | 0.47 (0.9) | 0.034 (0.81) | 0.53 (0.15) | 2.4 (0.03)* | 0.11 (0.59) |
| Sleeping place | 0.73 (0.02)* | 0.62 (0.04)* | 1.3 (0.78) | 0.07 (0.53) | 0.18 (0.73) |
Table 5.
Multivariate linear regression analysis of urinary MDA (µg/g creatinine) with the factors affecting the urinary MDA in the study participants
| Factors | β coefficient | P-value |
|---|---|---|
| Grilled food | − 0.03 | 0.95 |
| Fast food | 0.27 | 0.71 |
| BMI | 0.16 | 0.03* |
| Gender | 1.6 | 0.12 |
| 1-OH-NaP | 1.1 | 0.16 |
| 2-OH-NaP | 3.01 | 0.01* |
| 2-OH-Flu | 0.67 | 0.32 |
| 1-OH-Pyr | − 0.77 | 0.44 |
| 9-OH-Phe | 0.04 | 0.61 |
Risk assessment
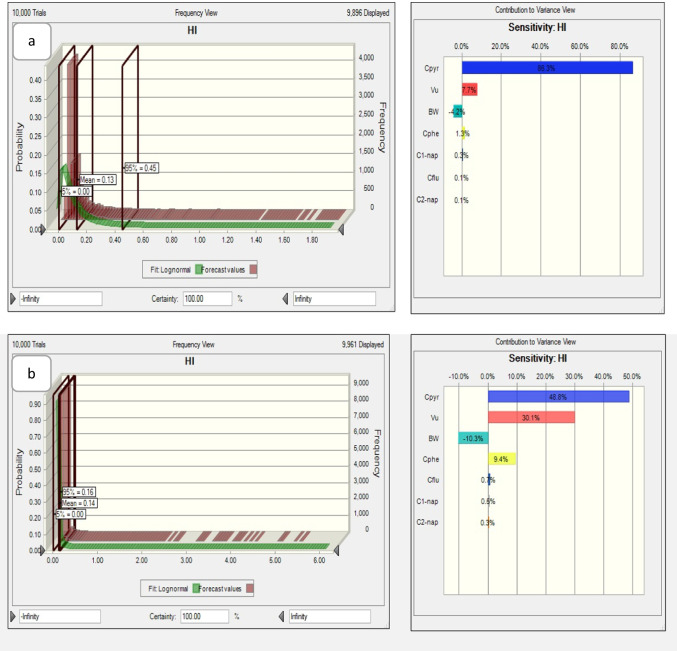
The Hazard Index (HI) was calculated to determine the potential health risks of OH-PAHs. As shown in Table 6, the HI was 3.4 times higher in the case group than in the control group. Moreover, HQ was estimated by Monte Carlo simulation with 10,000 replications using the Oracle Crystal ball software to determine the HQ variance (Fig. 3). The HQs of PAHs for the 95th percentile were 0.45 and 0.16 in the case and control groups, respectively, which indicated a low non-carcinogenic risk for these groups.
Table 6.
Hazard Index in the case and control groups
| HI | ||||
|---|---|---|---|---|
| Min | Mean | Max | SD | |
| Case | 0.004 | 0.073 | 0.276 | 0.09 |
| Control | 0.003 | 0.021 | 0.119 | 0.03 |
Fig. 3.
Monte Carlo simulation histogram for a the case group and b the control group
Discussion
Up to now, numerous studies have assessed PAHs exposure in the general population and occupational settings. In the present study, a biomonitoring method was used to assess PAHs exposure among children with hookah-smoking parents. Khesht was selected as the study area, because it has been located at a distance of 5000 m from the main roads, gas stations, and industries that are the main ambient sources of PAH compounds emissions (Nadali et al. 2021). The results indicated significantly higher ∑OH-PAHs urinary levels in the samples collected from the exposed individuals compared to the unexposed group. Tobacco smoking has been regarded as the main source of PAHs exposure (Slezakova et al. 2014).
In the present study, the effect of hookah smoking at home on naphthalene and phenanthrene exposure was investigated by comparing the exposed and non-exposed groups concerning the statically significant differences in the levels of urinary 1-hydroxy naphthalene and 9-hydroxy phenanthrene. In the exposed group, 80% of the parents used regular tobacco. A recent study conducted on the indoor air of cafés serving waterpipe reported the high concentrations of naphthalene and phenanthrene emitted from hookah smoke with regular tobacco (Rostami et al. 2019). This might account for the higher levels of urinary 1-hydroxy naphthalene and 9-hydroxy phenanthrene among the metabolites of PAHs in the urine samples of the case group compared to the control group in the current research. Furthermore, the median levels of urinary ∑OH-PAHs were 1.96 times higher in the samples collected from the children who had hookah-smoking parents in comparison to the unexposed group. These findings were comparable with those reported previously in other studies. For instance, some studies demonstrated that the mean level of 9-hydroxy phenanthrene was higher in the urine samples of smokers compared to non-smokers (Hoepfner et al. 1987, Martin et al. 1989). Another study conducted on the evaluation of PAHs exposure in smoking pregnant women’s urine samples confirmed the higher risk of exposure to PAHs in this population (Florek et al. 2005). Overall, tobacco smoking was identified as a major source of PAHs exposure (Thai et al. 2020).
The present study findings indicated that the highest and lowest were related to 9-hydroxyphenanthrene and 1-hydroxypyrene, respectively, in both exposed and unexposed groups. These results were consistent with the findings of a prior research on the children aged 7–13 years in Chongqing, China (Liu et al. 2017), but not with those reported in Canada’s general population (Ratelle et al. 2020), Canadian pregnant women (Nethery et al. 2012), and Spanish children (Fernández et al. 2021). In these studies, 2-hydroxynaphthalene and 9-hydroxyphenanthrene had the highest and lowest concentrations, respectively. Breathing is one of the most common ways for PAHs entrance into the body. People may also be exposed by eating contaminated food and drinking contaminated water. Additionally, the most important sources of naphthalene emissions include the use of deodorants and pollutants emitted from nearby garages and vehicle traffic (Batterman et al. 2012). However, the current study area was located at a distance of more than 5000 m from the mentioned sources. Thus, the higher exposure of the study participants to phenanthrene was assumed to be associated with indoor air pollution via incomplete fossil fuel, wood combustion, and oil-burning, as the sources of phenanthrene emission (Gad 2014; Lee and McCain 2015). In a previous research, seafood was considered one of the sources of phenanthrene (Gad 2014). In the current study also, the results obtained from the questionnaires (Table 1) revealed the high consumption of seafood compared to other kinds of food in Khesht.
According to Table 2, although the geometric mean of urinary 1-OH-NaP was higher than that of 2-OH-NaP in the exposed group, it was lower than 2-OH-NaP in the unexposed group. There was also a significant difference between the case and control groups regarding the concentration of 1-OH-NaP. The findings in the unexposed group were comparable with those reported previously in other studies (Li et al. 2008, Guo et al. 2013, Scinicariello and Buser 2014, Liu et al. 2017). Thus, tobacco smoking was a source of exposure to naphthalene. Several studies have also demonstrated that naphthalene concentrations in residential areas could be partly related to tobacco smoke (Jia and Batterman 2010, Batterman et al. 2012).
The results of regression analysis showed that the 1-OH-NaP and 2-OH-NaP concentrations were associated with the sleeping place through the night. Accordingly, the urinary levels of 1-OH-NaP and 2-OH-NaP were higher in the children who slept in the living room than in those who slept in the bedroom (Fig. S5). One possible reason is that the living room is generally more polluted than the bedrooms. Free air diffusion from cooking in the kitchen to the living room may be a reason for the higher concentrations and exposure. A previous study also reported a strong association between cooking fumes and indoor sources of PAHs (Yao et al. 2015).
The urinary concentrations of 2-OH-Flu in the unexposed children in the present investigation were comparable to those observed among children in China (Liu et al. 2017) (Table 7). In this study, the children who ate more grilled food were more likely to have higher urinary concentrations of 2-OHFlu compared to those who ate less grilled food (p < 0.05). In general, the Iranian diet is rich in deep-fried and grilled food, particularly grilled meat known as kebab. Kebab is one of the foremost consumed and favorite foods in Iran. Due to the incomplete combustion of charcoal, broiling meat results in high levels of PAHs within the grill (Shahsavani et al. 2017). This might account for the higher levels of urinary 2-OH-Flu in the current investigation.
Table 7.
The measured urinary concentrations of OH-PAHs in other papers
| 1-OH-NaP | 2-OH-NaP | 2-OH-Flu | 1-OH-Pyr | 9-OH-Phe | Ref | |
|---|---|---|---|---|---|---|
| Exposed | 0.73 | 0.69 | 1.2 | 0.33 | 1.67 | Present study |
| Unexposed | 0.49 | 0.54 | 0.86 | 0.26 | 0.47 | Present study |
| Germany (7–13 years) | 0.78 | 0.23 | 0.38 | 0.09 | 0.05 | (Murawski et al. 2020) |
| USA (6–11 years) | 1.43 | 1.69 | 0.24 | 0.06 | 0.02 | (Li et al. 2008) |
| India (30 to 51 years) | 3.39 | 8.71 | 1.1 | 0.69 | 0.28 | (Guo et al. 2013) |
| China (7–13 years) | - | 0.25 | 0.83 | 0.18 | 2.54 | (Liu et al. 2017) |
| USA (6–19 years) | 1.6 | 2.53 | 0.24 | 0.09 | - | (Scinicariello and Buser 2014) |
In the present research, the urinary concentrations of 1-hydroxypyrene were 3.7 and 2.6 times higher in the children who ate meat more than three times a week in their diet compared to those who ate meat once and 2–3 times a week, respectively (Fig. S6). In a study conducted on the effects of lifestyle on the urinary concentration of 1-hydroxypyrene, the participants who ate more meat excreted 1.5–2 times more 1-hydroxypyrene in urine compared to those who ate more vegetables (Kawamoto et al. 2007).
In the current research, the concentrations of 2-OH-NaP and 1-OH-Pyr in the children’s urine samples significantly increased from 7 to 13 years of age. Another study also reported an increase in the 2-OH-NaP concentration from 5 to 9–10 years of age (Jung et al. 2015), which represented an increase in inhalation and consumption levels over time (Jung et al. 2015). Nonetheless, a biomonitoring study in the Australian population did not show any relationships between the OH-PAHs concentrations and age (Thai et al. 2020). In contrast, a Canadian study revealed a significant positive relationship between the OH-PAH concentration and age in all the cycles studied (Khoury et al. 2018).
The present study findings indicated significant differences between boys and girls regarding the levels of 1-OH-NaP and of 2-OH-NaP. Up to now, limited studies have been conducted in this area. On the other hand, this study was performed on a small sample size. Therefore, further studies are recommended to be performed on OH-PAHs to identify the potential differences between the two genders.
In the current research, the concentration of MDA (µg/g creatinine) was higher in the children exposed to ETS than in those who were not exposed to ETS, but the difference was not statistically significant (Fig. S2). These results were in agreement with those of another study conducted on Korean adults and children (Yoon et al. 2012). In one other research carried out in Tehran, the MDA concentrations in the exposed and non-exposed samples were respectively 1.87- and 2.58-folds lower compared to the children aged 7–13 years working as street flower sellers (Rafiee et al. 2022). Moreover, the levels of MDA measured in the exposed and non-exposed groups were 17–80- and 23–108-folds lower than those measured in a group of children and their mothers living in Korea and China (Lee et al. 2004).
The present study findings revealed a positive relationship between the urinary 2-OH-NaP level and the urinary level of MDA as the biomarker of oxidative stress, which was in line with the findings of a prior study on children and their mothers in Korea and China (Lee et al. 2004). There was also a significant positive correlation between the urinary levels of MDA and children’s BMI (CC = 0.27, p = 0.04). Similarly, another study found that MDA was significantly associated with BMI (r = 0.29, p = 0.01) (Meguid et al. 2014).
The HQ results obtained in the current investigation were consistent with those of the research conducted by Fernandez among the children exposed to OH-PAHs in Spain (Fernández et al. 2021). Statistical analysis was performed to determine the most effective variable in increasing the health risk in the exposed population. Accordingly, 1-hydroxypyrene was the most influential risk factor for non-carcinogenic health due to PAH compounds. Therefore, the results of definitive and probabilistic risk assessment showed that exposure to PAHs had no significant effects on the participants’ health. Yet, further epidemiological studies are recommended to monitor the exposure of residents to PAHs and to determine the nutritional and environmental factors in the PAHs exposure to identify other important factors.
The present study had some limitations. This was the first study assessing the exposure of children with hookah-smoking parents to PAH compounds. Since the findings showed significant levels of ∑OH-PAHs in the urine samples of the exposed children, mitigation techniques have to be carried out to limit children’s exposure to PAH compounds through inhalation, thereby eliminating the harm arising through the oxidative stress related to PAHs exposure.
Also, due to the high cost of measuring PAH urinary metabolites and financial constraints, as well as the prevalence of coronavirus during sampling and the reluctance of some parents to cooperate, it was not possible to take more samples.
Conclusion
The present study aimed to characterize PAHs exposure and MDA levels (a biomarker of oxidative stress) among the children who had hookah-smoking parents in Khesht, Iran. The findings revealed significant differences between the exposed and unexposed groups regarding the urinary concentrations of 1-OH-NaP and 9-OH-Phe. Although the urinary levels of MDA were higher in the exposed samples than in the unexposed samples, this difference was not statistically significant. Because of their unique body structure and metabolic pathways, children are particularly sensitive to environmental pollutants and their exposure to chemical compounds such as PAHs can has destructive consequences on their health.
The current study results demonstrated that sleeping in the living room instead of the bedroom could be a significant predictor of the high concentrations of 1-OH-NaP and of 2-OH-NaP in the children’s urine samples. In addition, eating more grilled food and meat could increase the concentrations of 2-OH-Flu and 1-OHPyr in the children’s urine samples. Future studies are suggested to investigate the effects of lifestyle and diet on the urinary levels of OH-PAHs among children.
Overall, children should be protected from PAHs by stopping hookah use by family members at home. Future studies are recommended to determine whether interventional approaches such as dietary changes and banning tobacco burning at home can reduce children’s exposure to PAHs and the related oxidative stress. The concentrations of MDA and PAH metabolites can help define mitigation strategies and public health policies to reduce children’s exposure to PAHs.
Supplementary Information
Below is the link to the electronic supplementary material.
Acknowledgements
The authors would like to thank the Vice-chancellor for Research and Technology of Shiraz University of Medical Sciences for financially supporting the research (proposal No. 21524). They would also like to appreciate Ms. A. Keivanshekouh at the Research Consultation Center (RCC) of Shiraz University of Medical Sciences for her invaluable assistance in editing the manuscript.
Author contribution
Zeynab Tabatabaei: methodology, investigation, writing—original draft. Mohammad Ali Baghapour: writing—review and editing, supervision. Mohammad Hoseini and Amin Mohammadpour: software, formal analysis, data curation, review and editing. Narges Shamsedini: writing—review and editing.
Funding
This work was supported by Shiraz University of Medical Sciences. Mohammad Hosseini has received research support from Shiraz University of Medical Sciences.”
Data availability
The datasets obtained in this study are available from the corresponding author on reasonable request.
Declarations
Ethics approval
The research was approved by the Ethics Committee of Shiraz University of Medical Sciences, Shiraz, Iran (ethics code: IR.SUMS.REC.1399.955).
Consent to participate
Informed consent was obtained from all individual participants included in the study.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- Abdel-Shafy HI, Mansour MS. A review on polycyclic aromatic hydrocarbons: source, environmental impact, effect on human health and remediation. Egypt J Pet. 2016;25(1):107–123. doi: 10.1016/j.ejpe.2015.03.011. [DOI] [Google Scholar]
- Batterman S, et al. Sources, concentrations, and risks of naphthalene in indoor and outdoor air. Indoor Air. 2012;22(4):266–278. doi: 10.1111/j.1600-0668.2011.00760.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bernstein JA, et al. The health effects of nonindustrial indoor air pollution. J Allergy Clin Immunol. 2008;121(3):585–591. doi: 10.1016/j.jaci.2007.10.045. [DOI] [PubMed] [Google Scholar]
- Butler AR. The Jaffé reaction. Identification of the coloured species. Clin Chim Acta. 1975;59(2):227–232. doi: 10.1016/0009-8981(75)90033-9. [DOI] [PubMed] [Google Scholar]
- Campo L, et al. Urinary profiles to assess polycyclic aromatic hydrocarbons exposure in coke-oven workers. Toxicol Lett. 2010;192(1):72–78. doi: 10.1016/j.toxlet.2008.12.018. [DOI] [PubMed] [Google Scholar]
- Dehghani S, Fararouei M, Rafiee A, Hoepner L, Oskoei V, Hoseini M (2022) Prenatal exposure to polycyclic aromatic hydrocarbons and effects on neonatal anthropometric indices and thyroid-stimulating hormone in a Middle Eastern population. Chemosphere 286(Pt 1):131605 [DOI] [PubMed]
- Dejmek J, et al. The impact of polycyclic aromatic hydrocarbons and fine particles on pregnancy outcome. Environ Health Perspect. 2000;108(12):1159–1164. doi: 10.1289/ehp.001081159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- El-Zaatari ZM, et al. Health effects associated with waterpipe smoking. Tob Control. 2015;24(Suppl 1):i31. doi: 10.1136/tobaccocontrol-2014-051908. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fernández SF, et al. Children’s exposure to polycyclic aromatic hydrocarbons in the Valencian Region (Spain): urinary levels, predictors of exposure and risk assessment. Environ Int. 2021;153:106535. doi: 10.1016/j.envint.2021.106535. [DOI] [PubMed] [Google Scholar]
- Florek E, et al. Evaluation exposition to polycyclic aromatic hydrocarbons of pregnant women who smoking tobacco. Przegl Lek. 2005;62(10):1013–1018. [PubMed] [Google Scholar]
- Freire C, et al. Urinary 1-hydroxypyrene and PAH exposure in 4-year-old Spanish children. Sci Total Environ. 2009;407(5):1562–1569. doi: 10.1016/j.scitotenv.2008.10.068. [DOI] [PubMed] [Google Scholar]
- Gad SE (2014) Phenanthrene. Encyclopedia of Toxicology (Third Edition). P. Wexler. Oxford, Academic Press: 865-867
- Guo Y, et al. Concentrations and profiles of urinary polycyclic aromatic hydrocarbon metabolites (OH-PAHs) in several asian countries. Environ Sci Technol. 2013;47(6):2932–2938. doi: 10.1021/es3052262. [DOI] [PubMed] [Google Scholar]
- Health, U. D. o. and H. Services (2006) The health consequences of involuntary exposure to tobacco smoke: a report of the Surgeon General, Atlanta, GA: US Department of Health and Human Services, Centers for Disease …. [PubMed]
- Hoepfner I, et al. Hydroxy-phenanthrenes in the urine of non-smokers and smokers. Toxicol Lett. 1987;35(1):67–71. doi: 10.1016/0378-4274(87)90087-7. [DOI] [PubMed] [Google Scholar]
- Jia C, Batterman S. A critical review of naphthalene sources and exposures relevant to indoor and outdoor air. Int J Environ Res Public Health. 2010;7(7):2903–2939. doi: 10.3390/ijerph7072903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jung KH, et al. Repeatedly high polycyclic aromatic hydrocarbon exposure and cockroach sensitization among inner-city children. Environ Res. 2015;140:649–656. doi: 10.1016/j.envres.2015.05.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kawamoto T, et al. Effects of lifestyle on urinary 1-hydroxypyrene concentration. J Occup Health. 2007;49(3):183–189. doi: 10.1539/joh.49.183. [DOI] [PubMed] [Google Scholar]
- Ke Y, et al. Comparative study of oxidative stress biomarkers in urine of cooks exposed to three types of cooking-related particles. Toxicol Lett. 2016;255:36–42. doi: 10.1016/j.toxlet.2016.05.017. [DOI] [PubMed] [Google Scholar]
- Keyte IJ, et al. On-road traffic emissions of polycyclic aromatic hydrocarbons and their oxy- and nitro- derivative compounds measured in road tunnel environments. Sci Total Environ. 2016;566–567:1131–1142. doi: 10.1016/j.scitotenv.2016.05.152. [DOI] [PubMed] [Google Scholar]
- Khoury C, et al. Human biomonitoring reference values for some non-persistent chemicals in blood and urine derived from the Canadian Health Measures Survey 2009–2013. Int J Hyg Environ Health. 2018;221(4):684–696. doi: 10.1016/j.ijheh.2018.03.003. [DOI] [PubMed] [Google Scholar]
- Kumar SR, et al. A review of air quality, biological indicators and health effects of second-hand waterpipe smoke exposure. Tob Control. 2015;24(Suppl 1):i54. doi: 10.1136/tobaccocontrol-2014-052038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leavens EL, et al. Comparison of a preferred versus non-preferred waterpipe tobacco flavour: subjective experience, smoking behaviour and toxicant exposure. Tob Control. 2018;27(3):319. doi: 10.1136/tobaccocontrol-2016-053344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee KH, et al. Urinary PAHS metabolites and oxidative stress biomarkers as environmental air pollution in Chinese and Korean. Epidemiology. 2004;15(4):S74–S75. doi: 10.1097/00001648-200407000-00183. [DOI] [Google Scholar]
- Lee SH and McCain WC (2015) Chapter 31 - wildlife toxicity assessment for phenanthrene. Wildlife Toxicity Assessments for Chemicals of Military Concern. M. A. Williams, G. Reddy, M. J. Quinn and M. S. Johnson, Elsevier: 591–598
- Li Z, et al. Concentration and profile of 22 urinary polycyclic aromatic hydrocarbon metabolites in the US population. Environ Res. 2008;107(3):320–331. doi: 10.1016/j.envres.2008.01.013. [DOI] [PubMed] [Google Scholar]
- Lin Y, et al. Urinary metabolites of polycyclic aromatic hydrocarbons and the association with lipid peroxidation: a biomarker-based study between Los Angeles and Beijing. Environ Sci Technol. 2016;50(7):3738–3745. doi: 10.1021/acs.est.5b04629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu L, et al. Quantification of selected monohydroxy metabolites of polycyclic aromatic hydrocarbons in human urine. Sci China Chem. 2015;58(10):1579–1584. doi: 10.1007/s11426-015-5357-2. [DOI] [Google Scholar]
- Liu S, et al. Levels and risk factors for urinary metabolites of polycyclic aromatic hydrocarbons in children living in Chongqing, China. Sci Total Environ. 2017;598:553–561. doi: 10.1016/j.scitotenv.2017.04.103. [DOI] [PubMed] [Google Scholar]
- Martin F, et al. Urinary excretion of hydroxy-phenanthrenes after intake of polycyclic aromatic hydrocarbons. Environ Int. 1989;15(1–6):41–47. doi: 10.1016/0160-4120(89)90008-1. [DOI] [Google Scholar]
- Maziak W, et al. Consensus statement on assessment of waterpipe smoking in epidemiological studies. Tob Control. 2017;26(3):338. doi: 10.1136/tobaccocontrol-2016-052958. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meguid N et al (2014) Efficacy of selected treadmill training programme on oxidative stress in adolescents with Down syndrome. East Mediterr Health J 9(19):131-137 [PubMed]
- Miri M, et al. Environmental determinants of polycyclic aromatic hydrocarbons exposure at home, at kindergartens and during a commute. Environ Int. 2018;118:266–273. doi: 10.1016/j.envint.2018.06.006. [DOI] [PubMed] [Google Scholar]
- Moradi M, et al. Exposure to BTEX in beauty salons: biomonitoring, urinary excretion, clinical symptoms, and health risk assessments. Environ Monit Assess. 2019;191(5):286. doi: 10.1007/s10661-019-7455-7. [DOI] [PubMed] [Google Scholar]
- Mueller D, et al. Tobacco smoke particles and indoor air quality (ToPIQ)-the protocol of a new study. J Occup Med Toxicol. 2011;6(1):35. doi: 10.1186/1745-6673-6-35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Murawski A, et al. Polycyclic aromatic hydrocarbons (PAH) in urine of children and adolescents in Germany–human biomonitoring results of the German Environmental Survey 2014–2017 (GerES V) Int J Hyg Environ Health. 2020;226:113491. doi: 10.1016/j.ijheh.2020.113491. [DOI] [PubMed] [Google Scholar]
- Nadali A, et al. Phase distribution and risk assessment of PAHs in ambient air of Hamadan, Iran. Ecotoxicol Environ Saf. 2021;209:111807. doi: 10.1016/j.ecoenv.2020.111807. [DOI] [PubMed] [Google Scholar]
- Nethery E, et al. Urinary polycyclic aromatic hydrocarbons as a biomarker of exposure to PAHs in air: a pilot study among pregnant women. J Eposure Sci Environ Epidemiol. 2012;22(1):70–81. doi: 10.1038/jes.2011.32. [DOI] [PubMed] [Google Scholar]
- Nguyen T, et al. Charcoal burning as a source of polyaromatic hydrocarbons in waterpipe smoking. J Environ Sci Health B. 2013;48(12):1097–1102. doi: 10.1080/03601234.2013.824300. [DOI] [PubMed] [Google Scholar]
- Rafiee A, et al. Exploring urinary biomarkers to assess oxidative DNA damage resulting from BTEX exposure in street children. Environ Res. 2022;203:111725. doi: 10.1016/j.envres.2021.111725. [DOI] [PubMed] [Google Scholar]
- Ratelle M, et al. Polycyclic aromatic hydrocarbons (PAHs) levels in urine samples collected in a subarctic region of the Northwest Territories, Canada. Environ Res. 2020;182:109112. doi: 10.1016/j.envres.2020.109112. [DOI] [PubMed] [Google Scholar]
- Rostami R, et al. Exposure and risk assessment of PAHs in indoor air of waterpipe cafés in Ardebil, Iran. Build Environ. 2019;155:47–57. doi: 10.1016/j.buildenv.2019.03.031. [DOI] [Google Scholar]
- Salami F et al (2021) Urinary levels of PAH metabolites in pregnant women and their correlation with sociodemographic factors and PM 2.5 exposure in an urban and a suburban area. Air Qual Atmos Health 14:653–665
- Scinicariello F, Buser MC. Urinary polycyclic aromatic hydrocarbons and childhood obesity: NHANES (2001–2006) Environ Health Perspect. 2014;122(3):299–303. doi: 10.1289/ehp.1307234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sepetdjian E, et al. Carcinogenic PAH in waterpipe charcoal products. Food Chem Toxicol. 2010;48(11):3242–3245. doi: 10.1016/j.fct.2010.08.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shahsavani S, et al. Biological monitoring of urinary 1-hydroxypyrene by PAHs exposure among primary school students in Shiraz, Iran. Int Arch Occup Environ Health. 2017;90(2):179–187. doi: 10.1007/s00420-016-1184-9. [DOI] [PubMed] [Google Scholar]
- Shahsavani, S., et al.(2022). Exposure to polycyclic aromatic hydrocarbon-induced oxidative stress in Shiraz, Iran: urinary levels, health risk assessment and mediation effect of MDA on the risk of metabolic syndromes. Int Arch Occup Environ Health. 10.1007/s00420-021-01822-8 [DOI] [PubMed]
- Shamsedini N, et al. Health risk assessment of polycyclic aromatic hydrocarbons in individuals living near restaurants: a cross-sectional study in Shiraz. Iran: Scientific report; 2022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shamsedini N, et al. Exposure to polycyclic aromatic hydrocarbon-induced oxidative stress in individuals living near restaurants: a cross-sectional study in Shiraz, Iran. Environ Monit Assess. 2022;194(4):285. doi: 10.1007/s10661-022-09868-y. [DOI] [PubMed] [Google Scholar]
- Slezakova K, et al. Levels and risks of particulate-bound PAHs in indoor air influenced by tobacco smoke: a field measurement. Environ Sci Pollut Res. 2014;21(6):4492–4501. doi: 10.1007/s11356-013-2391-5. [DOI] [PubMed] [Google Scholar]
- Tabatabaei Z, et al. Assessing BTEX concentrations emitted by hookah smoke in indoor air of residential buildings: health risk assessment for children. J Environ Health Sci Eng. 2021;19(2):1653–1665. doi: 10.1007/s40201-021-00721-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thai PK, et al. Analysis of urinary metabolites of polycyclic aromatic hydrocarbons and cotinine in pooled urine samples to determine the exposure to PAHs in an Australian population. Environ Res. 2020;182:109048. doi: 10.1016/j.envres.2019.109048. [DOI] [PubMed] [Google Scholar]
- Tombolini F, et al. Levels of urinary metabolites of four PAHs and cotinine determined in 1016 volunteers living in Central Italy. Environ Sci Pollut Res. 2018;25(29):28772–28779. doi: 10.1007/s11356-018-1650-x. [DOI] [PubMed] [Google Scholar]
- Urbancova K, et al. Comparison of polycyclic aromatic hydrocarbon metabolite concentrations in urine of mothers and their newborns. Sci Total Environ. 2020;723:138116. doi: 10.1016/j.scitotenv.2020.138116. [DOI] [PubMed] [Google Scholar]
- Watson AY et al (1988) Potential carcinogenic effects of polynuclear aromatic hydrocarbons and nitroaromatics in mobile source emissions. Air Pollution, the Automobile, and Public Health, National Academies Press (US) [PubMed]
- Yao Z, et al. Characteristics of PAHs from deep-frying and frying cooking fumes. Environ Sci Pollut Res. 2015;22(20):16110–16120. doi: 10.1007/s11356-015-4837-4. [DOI] [PubMed] [Google Scholar]
- Yoon H-S, et al. Polycyclic aromatic hydrocarbon (1-OHPG and 2-naphthol) and oxidative stress (malondialdehyde) biomarkers in urine among Korean adults and children. Int J Hyg Environ Health. 2012;215(4):458–464. doi: 10.1016/j.ijheh.2012.02.007. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The datasets obtained in this study are available from the corresponding author on reasonable request.